Nanotechnology-Based Strategy to Upgrade the Performances of Plastic Flexible Film Waste
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Production of Copolyamide-Based Nanocomposite Masterbatch
2.3. Production of Neat and Nanocomposite Fil-s/Copolyamide Blends
2.4. Characterization Techniques
3. Results and Discussion
3.1. Neat Fil-s/CoPA Blends
3.2. Nanocomposite Fil-s/CoPA/PM15 Blends
3.3. Mechanical Performances of Neat and Nanocomposite CoPA/Fil-s Blends
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alam, O.; Billah, M.; Yajie, D. Characteristics of plastic bags and their potential environmental hazards. Resour. Conserv. Recyl. 2018, 132, 121–129. [Google Scholar] [CrossRef]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Al-Salem, S.M.; Antelava, A.; Constantinou, A.; Manos, G.; Dutta, A. A review on thermal and catalytic pyrolysis of plastic solid waste (PSW). J. Environ. Manag. 2017, 197, 177–198. [Google Scholar] [CrossRef]
- Lahtela, V.; Hyvärinen, M.; Kärki, T. Composition of Plastic Fractions in Waste Streams: Toward More Efficient Recycling and Utilization. Polymers 2019, 11, 69. [Google Scholar] [CrossRef]
- Botta, L.; Scaffaro, R.; Sutera, F.; Mistretta, M.C. Reprocessing of PLA/Graphene Nanoplatelets Nanocomposites. Polymers 2018, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Shojaeiarani, J.; Bajwa, D.S.; Rehovsky, C.; Bajwa, S.G.; Vahidi, G. Deterioration in the Physico-Mechanical and Thermal Properties of Biopolymers Due to Reprocessing. Polymers 2019, 11, 58. [Google Scholar] [CrossRef]
- Plastics—The Facts 2018. An Analysis of European Plastics, Plastics Europe. 2018. Available online: https://www.plasticseurope.org/it/resources/publications/619-plastics-facts-2018 (accessed on 2 May 2019).
- World Economic Forum. The New Plastics Economy: Rethinking the Future of Plastics. Ellen MacArthur Foundation and McKinsey & Company, 2016. Available online: http://www.ellenmacarthurfoundation.org/publications (accessed on 2 May 2019).
- Directive 2004/12/EC of the European Parliament and of the Council of 11 February 2004 amending Directive 94/62/EC on packaging and packaging waste. Off. J. Eur. Union 2004, L47, 26–31.
- Directive (EU) 2018/852 of the European Parliament and of the Council of 30 May 2018 amending Directive 94/62/EC on packaging and packaging waste. Off. J. Eur. Union 2018, L150, 141–154.
- Horodytska, O.; Valdés, F.J.; Fullana, A. Plastic flexible films waste management—A state of art review. Waste Manag. 2018, 77, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, E.; Claro, M.; Scarfato, P.; Di Maio, L.; Incarnato, L. Upgrading of recycled plastics obtained from flexible packaging waste by adding nanosilicates. AIP Conf. Proc. 2015, 1695, 020053. [Google Scholar] [CrossRef]
- Garofalo, E.; Di Maio, L.; Scarfato, P.; Di Gregorio, F.; Incarnato, L. Reactive compatibilization and melt compounding with nanosilicates of post-consumer flexible plastic packagings. Polym. Degrad. Stabil. 2018, 152, 52–63. [Google Scholar] [CrossRef]
- Coppola, B.; Courard, L.; Michel, F.; Incarnato, L.; Di Maio, L. Investigation on the use of foamed plastic waste as natural aggregates replacement in lightweight mortar. Compos. Part B Eng. 2016, 99, 75–83. [Google Scholar] [CrossRef]
- Garofalo, E.; Scarfato, P.; Di Maio, L.; Incarnato, L. Tuning of co-extrusion processing conditions and film layout to optimize the performances of PA/PE multilayer nanocomposite films for food packaging. Polym. Compos. 2018, 39, 3157–3167. [Google Scholar] [CrossRef]
- Di Maio, L.; Scarfato, P.; Garofalo, E.; Galdi, M.R.; D’Arienzo, L.; Incarnato, L. Processing-structure-properties relationships in PLA nanocomposite films. AIP Conf. Proc. 2014, 1593, 308–311. [Google Scholar] [CrossRef]
- Garofalo, E.; Scarfato, P.; Di Maio, L.; Incarnato, L. Effect of nanocomposite composition on shear and elongational rheological behaviour of PLA/MMT hybrids. AIP Conf. Proc. 2014, 1599, 422–425. [Google Scholar] [CrossRef]
- Di Maio, L.; Garofalo, E.; Scarfato, P.; Incarnato, L. Effect of polymer/organoclay composition on morphology and rheological properties of polylactide nanocomposites. Polym. Comp. 2015, 36, 1135–1144. [Google Scholar] [CrossRef]
- Raji, M.; Mekhzoum, M.M.; Rodrigue, D.; Qaiss, A.K.; Bouhfid, R. Effect of silane functionalization on properties of polypropylene/clay nanocomposites. Compos. Part B Eng. 2018, 146, 106–115. [Google Scholar] [CrossRef]
- Garofalo, E.; Fariello, M.L.; Di Maio, L.; Incarnato, L. Effect of biaxial drawing on morphology and properties of copolyamide nanocomposites produced by film blowing. Eur. Polym. J. 2013, 49, 80–89. [Google Scholar] [CrossRef]
- Scarfato, P.; Incarnato, L.; Di Maio, L.; Dittrich, B.; Schartel, B. Influence of a novel organo-silylated clay on the morphology, thermal and burning behavior of low density polyethylene composites. Compos. Part B Eng. 2016, 98, 444–452. [Google Scholar] [CrossRef]
- Shah, D.; Maiti, P.; Gunn, E.; Schmidt, D.F.; Jiang, D.D.; Batt, C.A.; Giannelis, E.P. Dramatic enhancements in toughness of polyvinylidiene fluoride nanocomposites via nanoclay directed crystal structure and morphology. Adv. Mater. 2004, 16, 1173–1177. [Google Scholar] [CrossRef]
- Rashmi, B.J.; Prashantha, K.; Lacrampe, M.-F.; Krawczak, P. Toughening of poly(lactic acid) without sacrificing stiffness and strength by melt-blending with polyamide 11 and selective localization of halloysite nanotubes. Express Polym. Lett. 2015, 9, 721–735. [Google Scholar] [CrossRef]
- Si, M.; Araki, T.; Ade, H.; Kilcoyne, A.L.D.; Fisher, R.; Sokolov, J.C.; Rafailovich, M.H. Compatibilizing bulk polymer blends by using organoclays. Macromolecules 2006, 39, 4793–4801. [Google Scholar] [CrossRef]
- Ginzburg, V.V. Influence of nanoparticles on miscibility of polymer blends. A simple theory. Macromolecules 2005, 38, 2362–2367. [Google Scholar] [CrossRef]
- Taguet, A.; Cassagnau, P.; Lopez-Cuesta, J.M. Structuration, selective dispersion and compatibilizing effect of (nano)fillers in polymer blends. Prog. Polym. Sci. 2014, 39, 1526–1563. [Google Scholar] [CrossRef]
- Scaffaro, R.; Botta, L. Nanofilled thermoplastic-thermoplastic polymer blends. In Nanostructured Polymer Blends; Thomas, S., Shanks, R., Chandrasekharakurup, S., Eds.; Elsevier: Oxford, UK, 2014; pp. 133–160. [Google Scholar]
- Salzano de Luna, M.; Filippone, G. Effects of nanoparticles on the morphology of immiscible polymer blends–Challenges and opportunities. Eur. Polym. J. 2016, 79, 198–218. [Google Scholar] [CrossRef]
- Chen, R.S.; Ahmad, S.; Gan, S. Characterization of recycled thermoplastics-based nanocomposites: Polymer-clay compatibility, blending procedure, processing condition, and clay content effects. Compos. Part B Eng. 2017, 131, 91–99. [Google Scholar] [CrossRef]
- Fang, C.; Nie, L.; Liu, S.; Yu, R.; Li, S. Characterization of polypropylene–polyethylene blends made of waste materials with compatibilizer and nano-filler. Compos. Part B Eng. 2013, 55, 498–505. [Google Scholar] [CrossRef]
- Palacios, J.K.; Sangroniz, A.; Eguiazabal, J.I.; Etxeberria, A.; Müller, A.J. Tailoring the properties of PP/PA6 nanostructured blends by the addition of nanosilica and compatibilizer agents. Eur. Polym. J. 2016, 85, 532–552. [Google Scholar] [CrossRef]
- Fenouillot, F.; Cassagnau, P.; Majesté, J.-C. Uneven distribution of nanoparticles in immiscible fluids: Morphology development in polymer blends. Polymer 2009, 5, 1333–1350. [Google Scholar] [CrossRef]
- Chen, J.; Chen, J.-W.; Chen, H.-M.; Yang, J.-H.; Wang, Y. Effect of compatibilizer and clay on morphology and fracture resistance of immiscible high-density polyethylene/polyamide 6 blend. Compos. Part B Eng. 2013, 54, 422–430. [Google Scholar] [CrossRef]
- Scaffaro, R.; Botta, L.; Mistretta, M.C.; La Mantia, F.P. Processing–morphology–property relationships of polyamide 6/polyethylene blend–clay nanocomposites. Express Polym. Lett. 2013, 7, 873–884. [Google Scholar] [CrossRef]
- Garofalo, E.; D’Arienzo, L.; Scarfato, P.; Di Maio, L.; Incarnato, L. Copolyamide/Sepiolite Nanocomposites with Enhanced Stiffness and Toughness. AIP Conf. Proc. 2016, 1736, 020154. [Google Scholar] [CrossRef]
- Zare, Y. Recent progress on preparation and properties of nanocomposites from recycled polymers: A review. Waste Manag. 2013, 33, 598–604. [Google Scholar] [CrossRef]
- Vasanthan, N. Crystallinity determination of nylon 66 by density measurement and Fourier Transform Infrared (FTIR) spectroscopy. J. Chem. Educ. 2012, 89, 387–390. [Google Scholar] [CrossRef]
- Dayma, N.; Satapathy, B.K. Microstructural correlations to micromechanical properties of polyamide-6/low density polyethylene-grafted-maleic anhydride/nanoclay ternary nanocomposites. Mater. Des. 2012, 33, 510–522. [Google Scholar] [CrossRef]
- Labaume, I.; Médéric, P.; Huitric, J.; Aubry, T. Comparative study of interphase viscoelastic properties in polyethylene/polyamide blends compatibilized with clay nanoparticles or with a graft copolymer. J. Rheol. 2013, 57, 377–392. [Google Scholar] [CrossRef]
- Silva, J.M.; Machado, A.V.; Moldenaers, P.; Maia, J.M. The role of interfacial elasticity on the rheological behaviour of polymer blends. Korea Aust. Rheol. J. 2010, 22, 21–29. [Google Scholar]
- Manaure, A.C.; Müller, A.J. Nucleation and crystallization of blends of poly(propylene) and ethylene/a-olefin copolymers. Macromol. Chem. Phys. 2000, 201, 958–972. [Google Scholar] [CrossRef]
- Frensch, H.; Harnischfeger, P.; Jungnickel, B.J. Fractionated crystallization in incompatible polymer blends. In Multiphase Polymers: Blends and Ionomers; Utracki, L.A., Weiss, R.A., Eds.; ASC Symposium Series vol. 395; American Chemical Society, 1989; pp. 101–125. [Google Scholar]
- Ongen, A.; Ozcan, H.K.; Ozbas, E.E.; Balkaya, N. Adsorption of Astrazon Blue FGRL onto sepiolite from aqueous solutions. Desalin. Water Treat. 2012, 40, 129–136. [Google Scholar] [CrossRef]
- Sumita, M.; Sakata, K.; Asai, S.; Miyasaka, K.; Nakagawa, H. Dispersion of fillers and the electrical conductivity of polymer blends filled with carbon black. Polym. Bull. 1991, 25, 265–271. [Google Scholar] [CrossRef]
- Wu, S. Polymer Interface and Adhesion; Marcel Dekker: New York, NY, USA, 1982. [Google Scholar]
- Zonder, L.; Mccarthy, S.; Rios, F.; Ophir, A.; Kenig, S. Viscosity ratio and interfacial tension as carbon nanotubes distributing factors in melt-mixed blends of polyamide 12 and high-density polyethylene. Adv. Polym. Technol. 2014, 33. [Google Scholar] [CrossRef]
- Vazquez, Y.V.; Barbosa, S.E. Compatibilization of HIPS/ABS blends from WEEE by using Styrene-Butadiene Rubber (SBR). J. Environ. Manag. 2018, 217, 381–390. [Google Scholar] [CrossRef]
- Walker, I.; Collyer, A.A. Rubber toughening mechanisms in polymeric materials Cap 2. In Rubber Toughened Engineering Plastics; Collyer, A.A., Ed.; Springer Science+Business Media: Dordrecht, The Netherlands, 1994. [Google Scholar]
- Silberberg, J.; Man, C.D. The effect of rubber particle size on the mechanical properties of high-impact polystyrene. J. Appl. Polym. Sci. 1978, 22, 599–609. [Google Scholar] [CrossRef]
- Donald, A.M.; Kramer, E.J. Craze initiation and growth in high-impact polystyrene. J. Appl. Polym. Sci. 1982, 27, 3729–3741. [Google Scholar] [CrossRef]
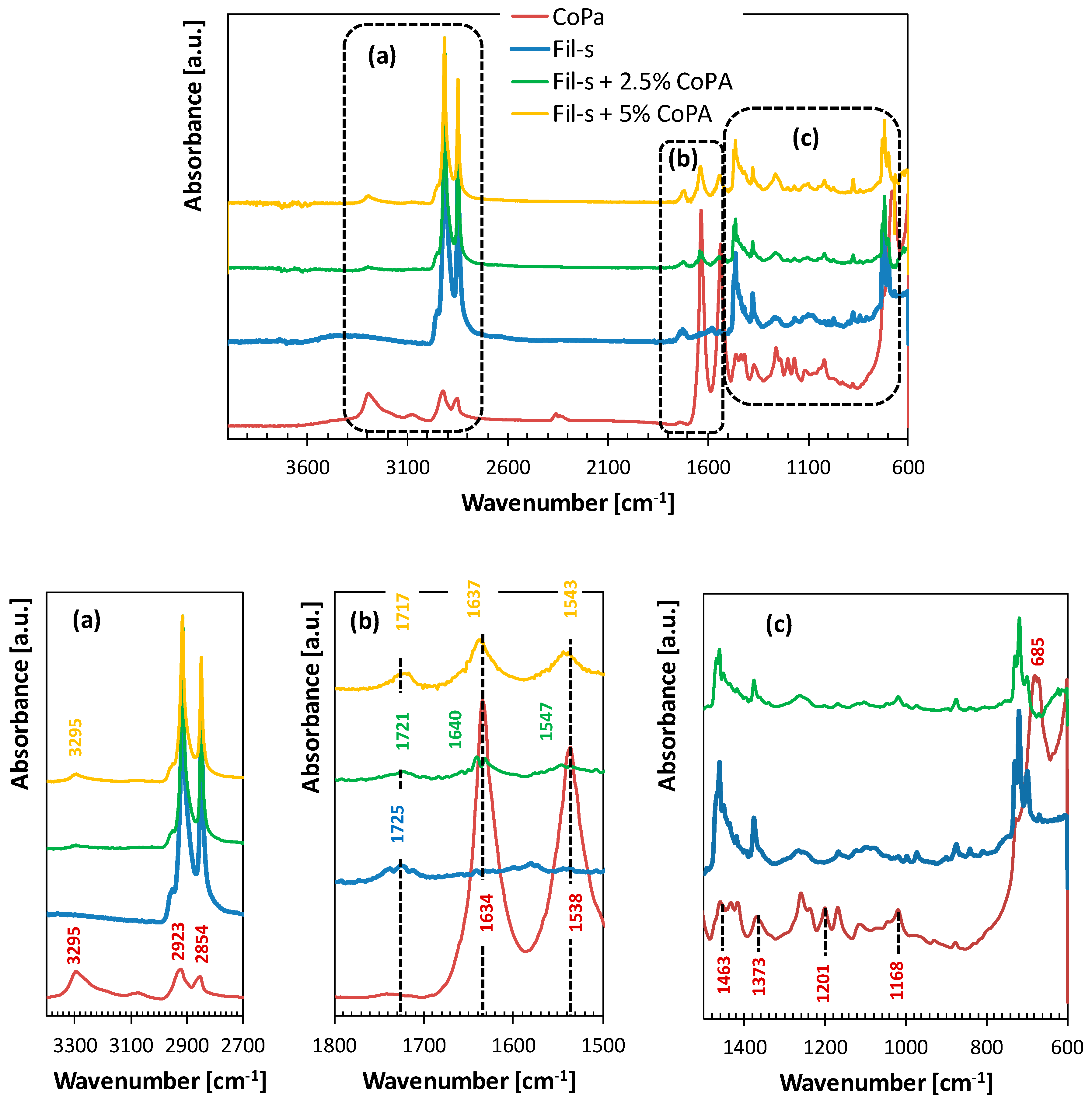
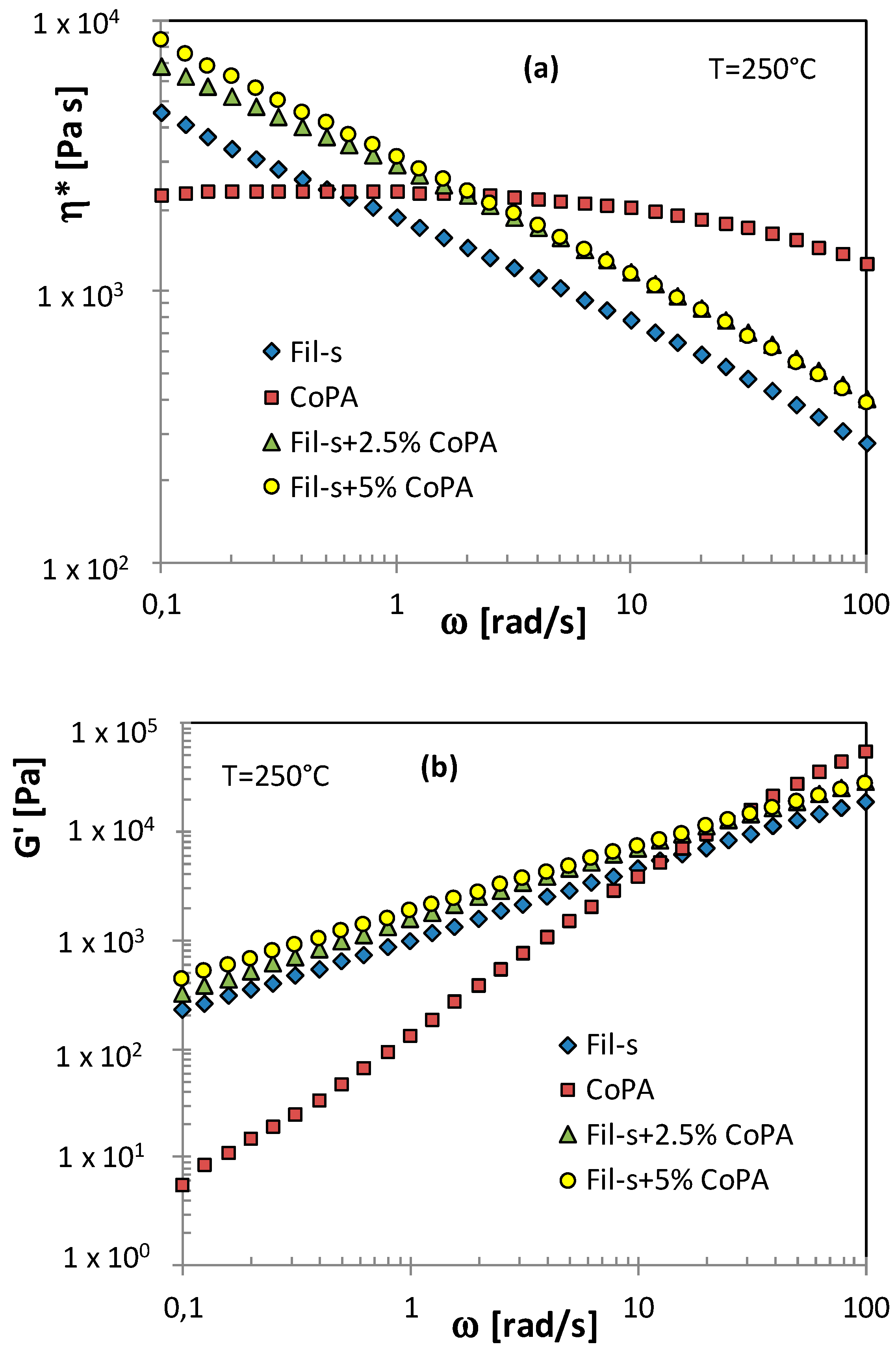
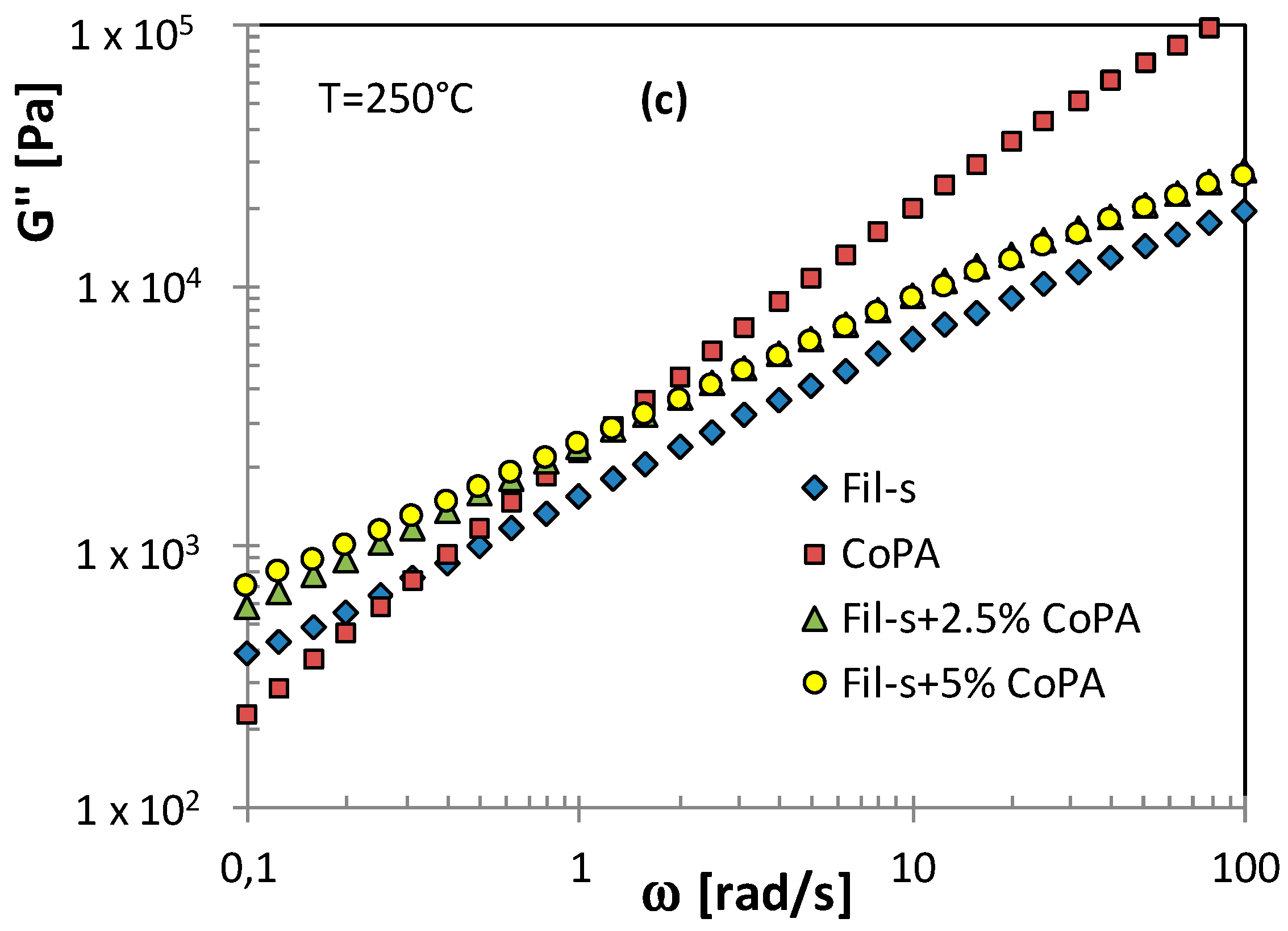


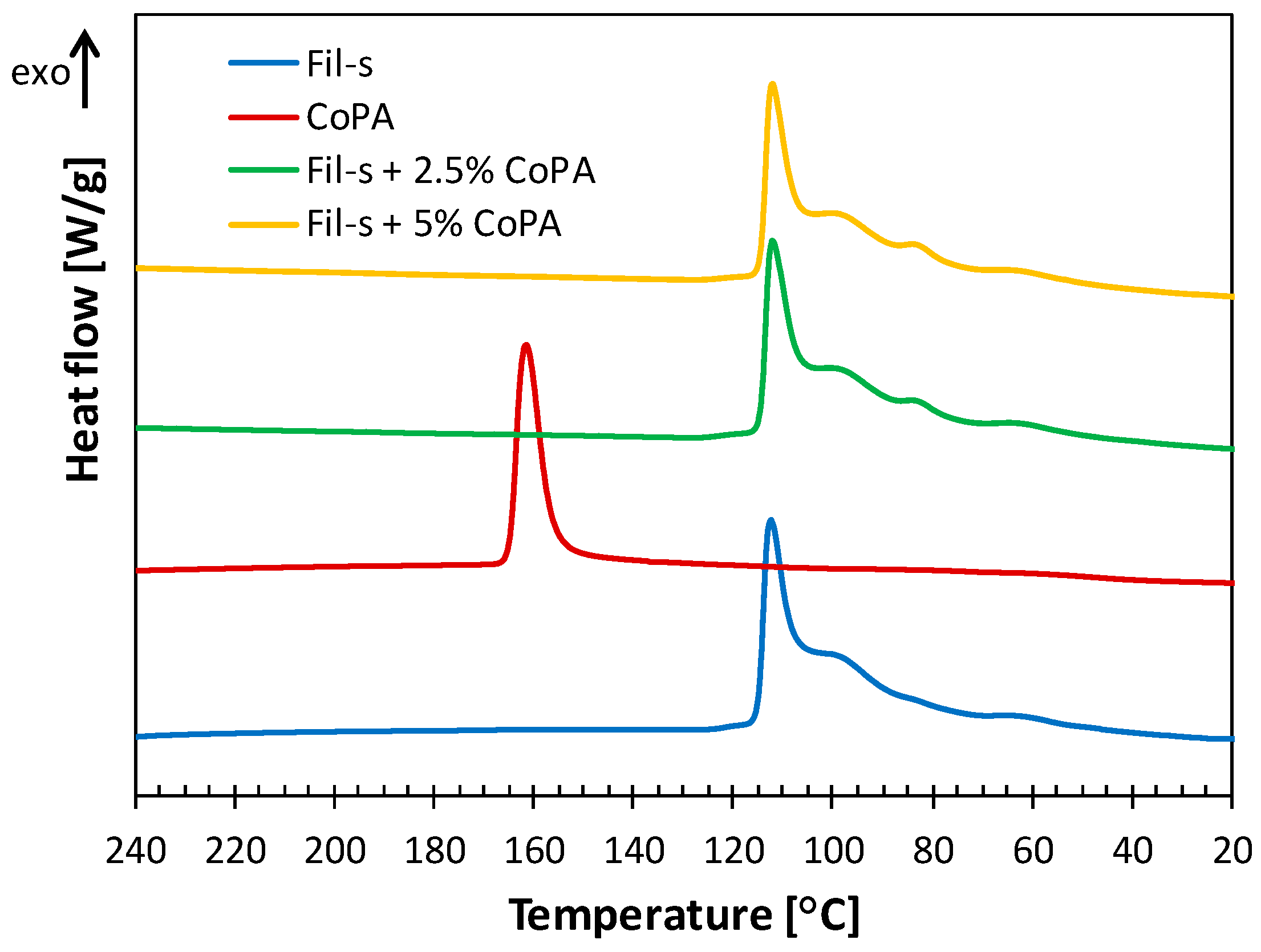
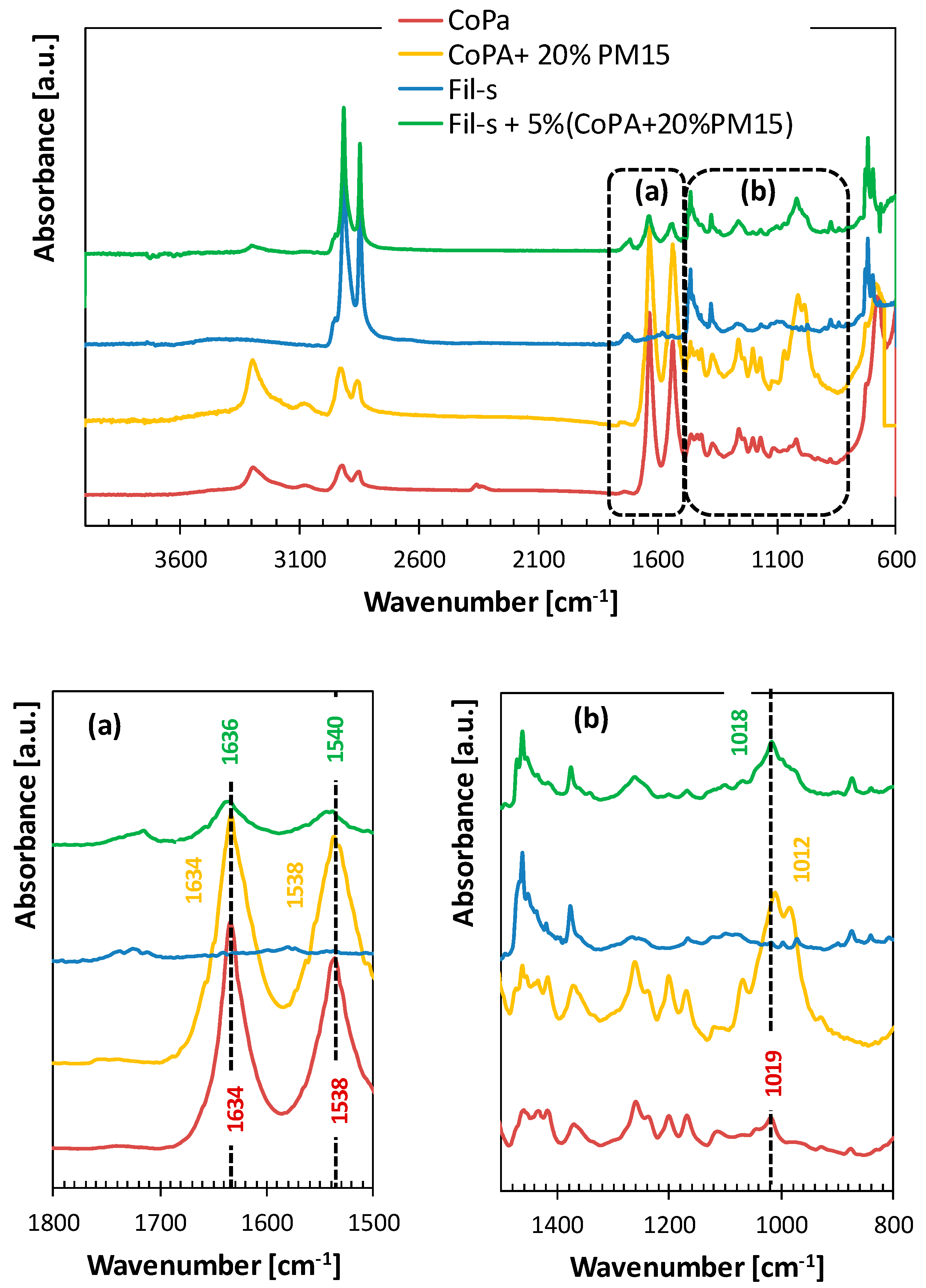
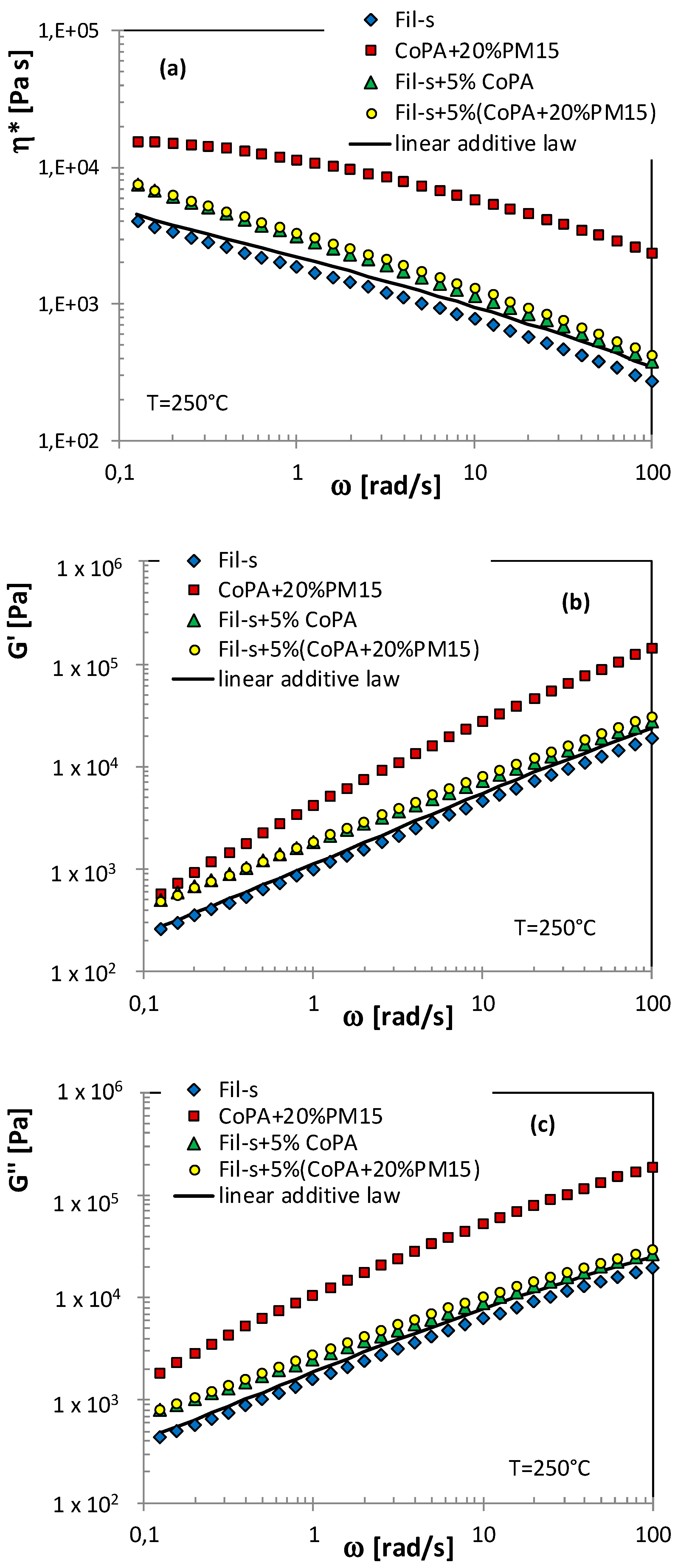
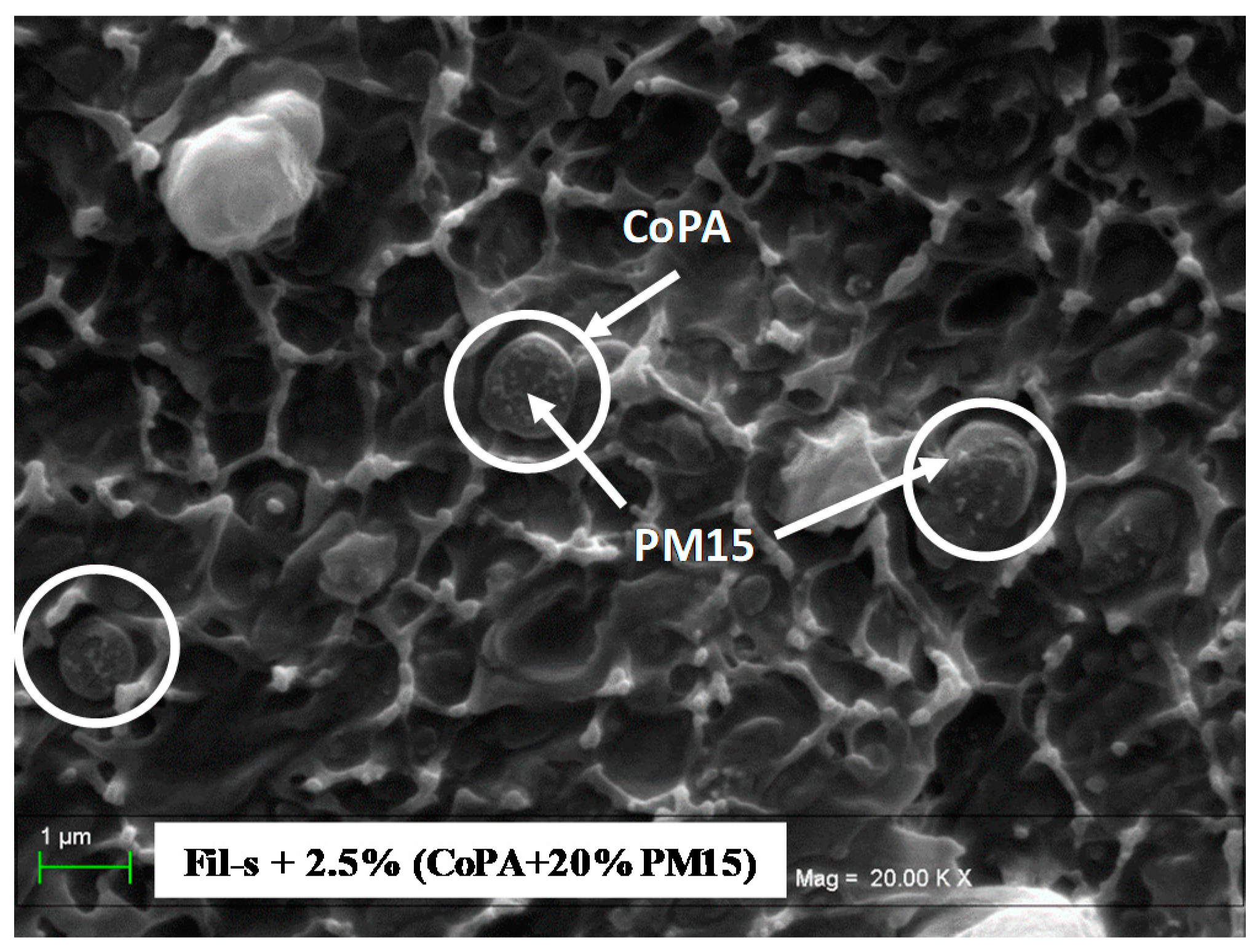

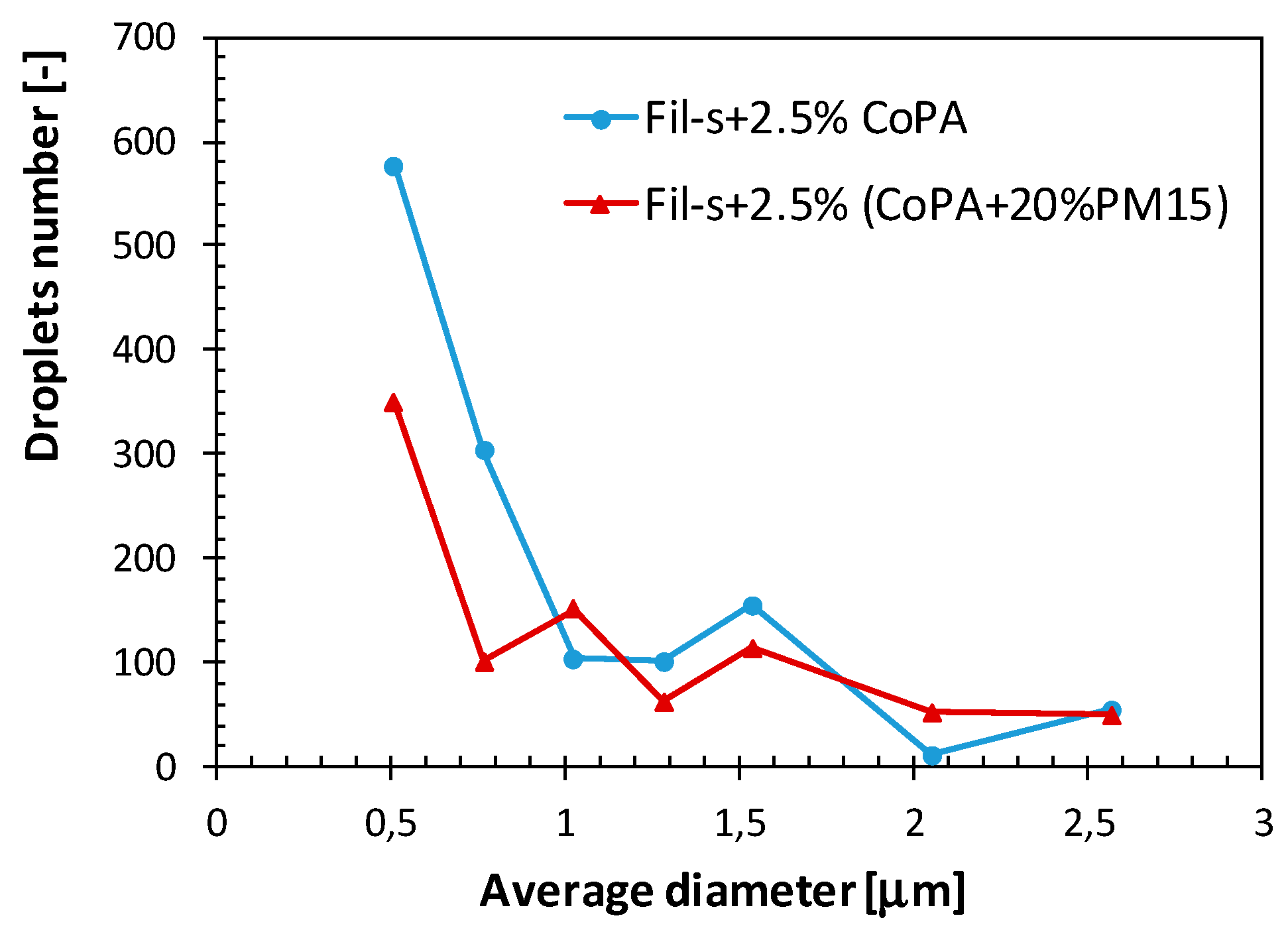
| SAMPLE | Actual CoPA Content [wt %] | Actual PM15 Content [wt %] |
|---|---|---|
| CoPA+20%PM15 | – | 18.03 ± 0.08 |
| Fil-s + 2.5% (CoPA+20% PM15) | 1.67 ± 0.21 | 0.30 ± 0.04 |
| Fil-s + 5% (CoPA+20% PM15) | 5.28 ± 0.23 | 0.95 ± 0.04 |
| Wavenumber (cm−1) | Assignment |
|---|---|
| 3500–3200 | –OH and –NH stretching |
| 2915 | –CH2 asymmetric stretching |
| 2847 | –CH2 symmetric stretching |
| 16001–565 | conjugated (C=C) stretching, –NH stretching |
| 1462 | –CH2 scissoring |
| 1376 | –CH3 scissoring |
| 740–690 | –CH2 rocking, (=C–H) bending, –NH stretching |
| Wavenumber [cm−1] | Assignment |
|---|---|
| 3295 | N–H stretch H-bonded |
| 2923 | Asymmetric CH2 stretch |
| 2854 | Symmetric CH2 stretch |
| 1634 | C=O stretch |
| 1538 | N–H bend |
| 1463 | CH2 scissors |
| 1373 | CH2 wagging |
| 1201 | CH2 twist-wagging |
| 1168 | CH2 wagging |
| 685 | N–H bend |
| Material | Surface Energy [mN/m] | ||
|---|---|---|---|
| Fil-s | 32.0 | 31.1 | 0.9 |
| CoPA | 39.5 | 32.7 | 6.7 |
| Sepiolite PM15 | 33.5 | 30.5 | 3.0 |
| Material | Interfacial Energy [mN/m] | |
|---|---|---|
| Harmonic Mean | Geometric Mean | |
| Fil-s/CoPA | 7.74 | 7.72 |
| Fil-s/PM15 | 3.91 | 3.90 |
| CoPA/PM15 | 1.59 | 0.87 |
| SAMPLE | Tc,PE [°C] | ΔHc,PE [J/g] | Tc,CoPA [°C] | ΔHc,CoPA[J/g] |
|---|---|---|---|---|
| Fil-s | 112 ± 0.7 | 94.2 ± 3.5 | – | – |
| CoPA+20%PM15 | – | – | 158 ± 1.8 | 36.4 ± 2.3 |
| Fil-s+2.5%(CoPA+20%PM15) | 112 ± 1.2 | 100.9 ± 2.3 | – | – |
| Fil-s+5%(CoPA+20%PM15) | 112 ± 0.9 | 97.5 ± 3.1 | – | – |
| SAMPLE | E [MPa] | εy [%] | σy [MPa] | εb [%] | σb [MPa] |
|---|---|---|---|---|---|
| * CoPA | 540 ± 60 | 13.7 ± 2.1 | 39.9 ± 1.8 | 400 ± 30 | 38.8 ± 2.8 |
| * CoPA + 20%PM15 | 1900 ± 80 | 18.4 ± 2.1 | 84.6 ± 3.2 | 36 ± 8 | 69.9 ± 3.2 |
| Fil-s | 440 ± 40 | 7.3 ± 0.2 | 15.8 ± 0.5 | 25 ± 4 | 13.0 ± 0.8 |
| Fil-s + 2.5% CoPA | 411 ± 7 | 14.0 ±0.6 | 15.3 ± 0.3 | 150 ± 50 | 11.8 ± 0.2 |
| Fil-s + 2.5% (CoPA + 20%PM15) | 474 ± 8 | 15.2 ± 0.5 | 15.8 ± 0.4 | 260 ± 90 | 12.4 ± 0.3 |
| Fil-s + 5% CoPA | 425 ± 5 | 12.8 ± 0.7 | 15.7 ± 0.4 | 90 ± 20 | 10.9 ± 0.5 |
| Fil-s + 5% (CoPA + 20%PM15) | 518 ± 8 | 12.9 ± 0.6 | 15.0 ± 0.5 | 50 ± 10 | 11.1 ± 0.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garofalo, E.; Di Maio, L.; Scarfato, P.; Di Gregorio, F.; Incarnato, L. Nanotechnology-Based Strategy to Upgrade the Performances of Plastic Flexible Film Waste. Polymers 2019, 11, 830. https://doi.org/10.3390/polym11050830
Garofalo E, Di Maio L, Scarfato P, Di Gregorio F, Incarnato L. Nanotechnology-Based Strategy to Upgrade the Performances of Plastic Flexible Film Waste. Polymers. 2019; 11(5):830. https://doi.org/10.3390/polym11050830
Chicago/Turabian StyleGarofalo, Emilia, Luciano Di Maio, Paola Scarfato, Fabrizio Di Gregorio, and Loredana Incarnato. 2019. "Nanotechnology-Based Strategy to Upgrade the Performances of Plastic Flexible Film Waste" Polymers 11, no. 5: 830. https://doi.org/10.3390/polym11050830
APA StyleGarofalo, E., Di Maio, L., Scarfato, P., Di Gregorio, F., & Incarnato, L. (2019). Nanotechnology-Based Strategy to Upgrade the Performances of Plastic Flexible Film Waste. Polymers, 11(5), 830. https://doi.org/10.3390/polym11050830







