Simultaneous Measurements of Structure and Water Permeability in an Isolated Human Skin Stratum Corneum Sheet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of SC Samples
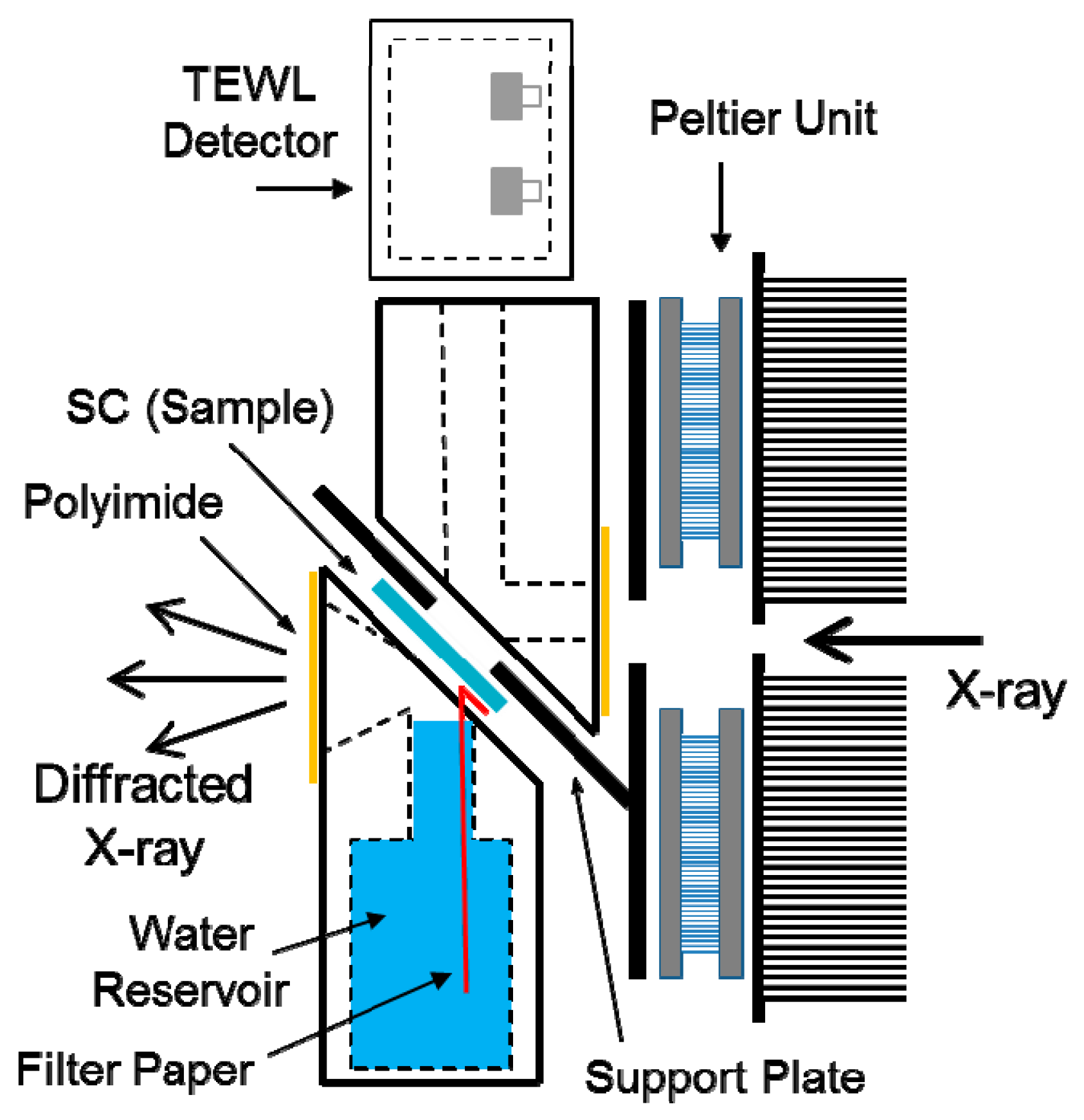
2.2. Sample Holder for Simultaneous Measurements of SC Structure and Water Permeability
2.3. Synchrotron X-ray Diffraction
2.4. Simultaneous Measurements of Structure and Water Permeability in SC
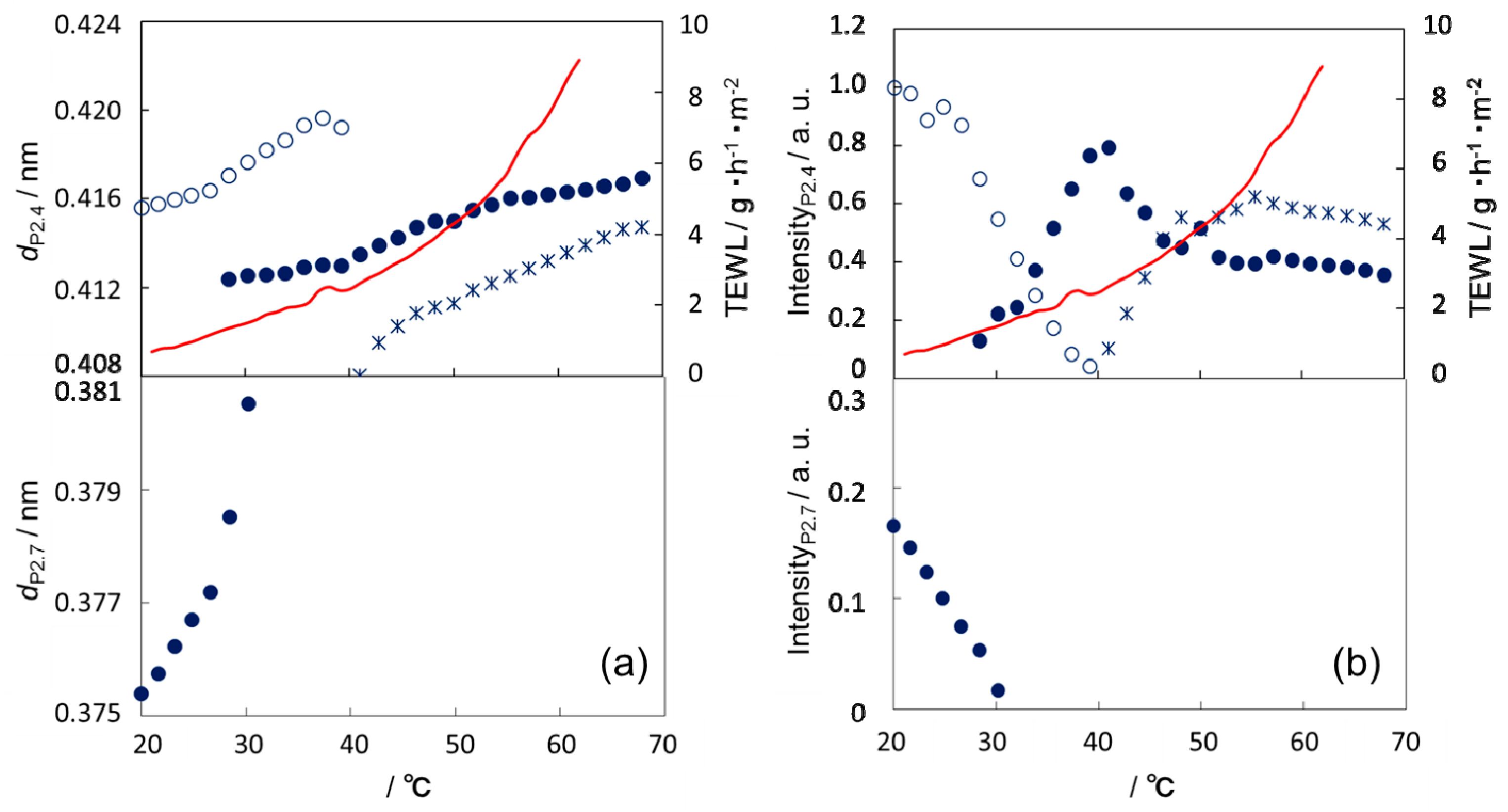
3. Results
3.1. Development of a Method for Simultaneous Analysis of the SC Structure and Function
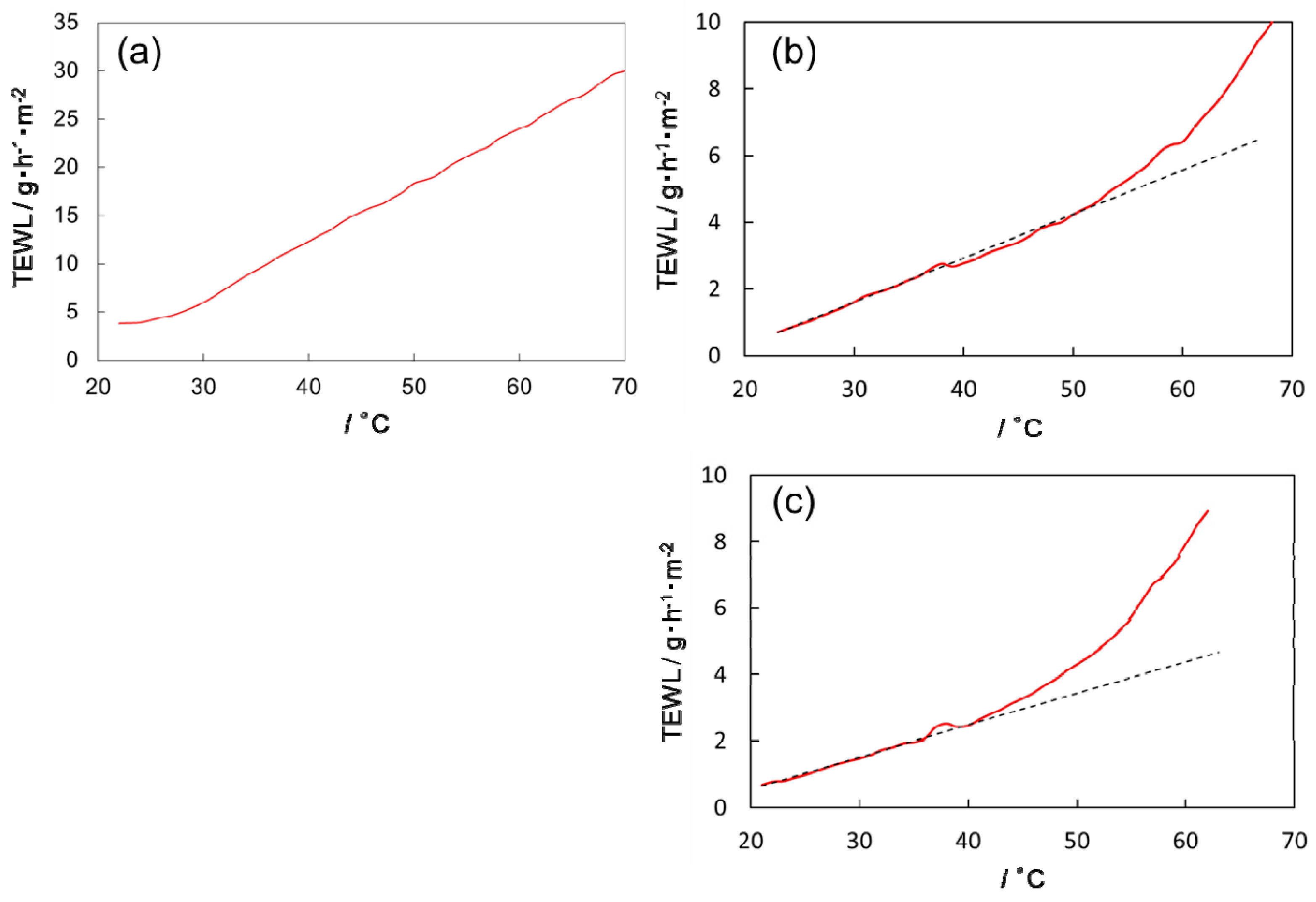
3.2. Temperature Dependence of Water Permeability
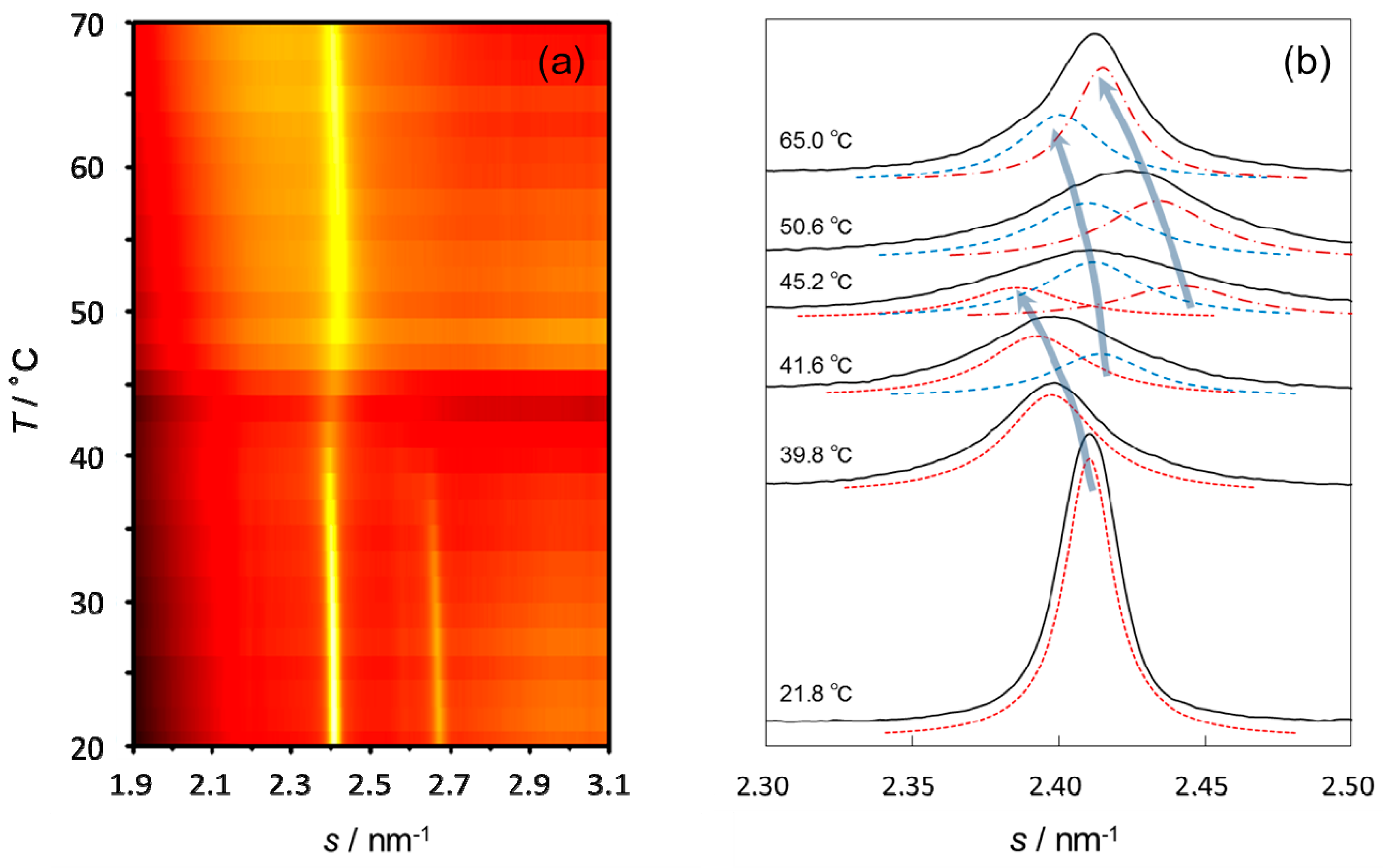
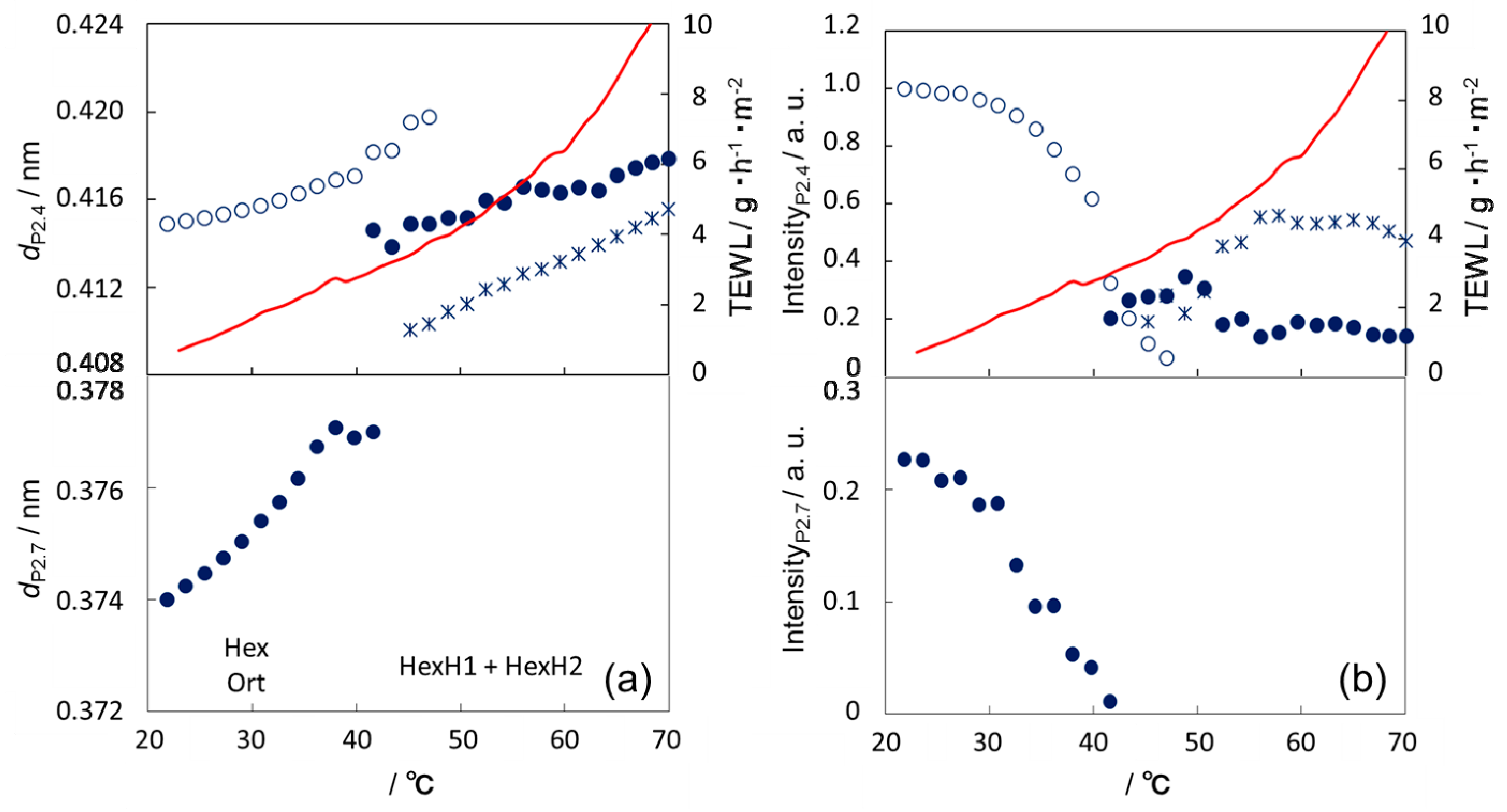
3.3. Thermal Behavior of the Lipid Organization in SC
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wertz, P.W. The nature of the epidermal barrier: Biochemical aspect. Adv. Drug Deliv. Rev. 1996, 18, 283–294. [Google Scholar] [CrossRef]
- Elias, P.M. Epidermal lipids, barrier function, and desquamation. J. Investig. Dermatol. 1983, 80, 44–49. [Google Scholar] [CrossRef]
- Landmann, L. The epidermal permeability barrier. Comparison between in vivo and in vitro lipid structures. Eur. J. Cell Biol. 1984, 33, 258–264. [Google Scholar]
- White, S.H.; Mirejovsky, D.; King, G.I. Structure of lamellar lipid domains and corneocyte envelopes of murine stratum corneum. An X-ray diffraction study. Biochemictry 1988, 27, 3725–3732. [Google Scholar] [CrossRef]
- Bouwstra, J.A.; Gooris, G.S.; van der Spek, J.A.; Bras, W. Structural investigations of human stratum corneum by small-angle X-ray scattering. J. Investig. Dermatol. 1991, 97, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Nakazawa, H.; Ohta, N.; Hatta, I. A possible regulation mechanism of water content in human stratum corneum via intercellular lipid matrix. Chem. Phys. Lipids 2012, 165, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Ohta, N.; Ban, S.; Tanaka, H.; Nakata, S.; Hatta, I. Swelling of intercellular lipid lamellar structure with short repeat distance in hairless mouse stratum corneum as studied by X-ray diffraction. Chem. Phys. Lipids 2003, 123, 1–8. [Google Scholar] [CrossRef]
- Charalambopoulou, G.C.; Steriotis, T.A.; Hauss, T.; Stubos, A.K.; Kanellopoulos, N.K. Structure alterations of fully hydrated human stratum corneum. Phys. B Condens. Matter 2004, 350, e603–e606. [Google Scholar] [CrossRef]
- Pilgram, G.S.K.; Van Pelt, A.M.; Spies, F.; Bouwstra, J.A.; Koerten, H.K. Cryo-electron diffraction as a tool to study local variations in the lipid organization of human stratum corneum. J. Microsc. 1998, 189, 71–78. [Google Scholar] [CrossRef]
- Nakazawa, H.; Imai, T.; Hatta, I.; Sakai, S.; Inoue, S.; Kato, S. Low-flux electron diffraction study for the intercellular lipid organization on a human corneocyte. Biochim. Biophys. Acta (BBA)-Biomembr. 2013, 1828, 1424–1431. [Google Scholar] [CrossRef]
- Boncheva, M.; Damien, F.; Normand, V. Molecular organization of the lipid matrix in intact Stratum Corneum using ATR-FTIR spectroscopy. Biochim. Biophys. Acta (BBA)-Biomembr. 2008, 1778, 1344–1355. [Google Scholar] [CrossRef]
- Imokawa, G.; Kuno, H.; Kawai, M. Stratum corneum lipids serve as a boundwater modulator. J. Investig. Dermatol. 1991, 96, 845–851. [Google Scholar] [CrossRef]
- Caspers, P.J.; Lucassen, W.G.; Carter, E.A.; Bruining, H.A.; Puppels, G.J. In vivo confocal Raman microspectroscopy of the skin: Non-invasive determination of molecular concentration profiles. J. Investig. Dermatol. 2001, 116, 434–442. [Google Scholar] [CrossRef]
- Hara, M.; Ma, T.; Verkman, A.S. Selectively reduced glycerol in skin of Aquaporin-3-deficient mice may account for impaired skin hydration, elasticity, and barrier recovery. J. Biol. Chem. 2002, 277, 46616–46621. [Google Scholar] [CrossRef] [PubMed]
- Rogiers, V. EEMCO guidance for the assessment of transepidermal water loss in cosmetic sciences. Skin Pharmacol. Physiol. 2001, 14, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Kompaore, F.; Dupont, C.; Marty, J.P. In vivo evaluation in man by two non-invasive methods of the stratum corneum barrier function after physical and chemical modification. Int. J. Cosmet. Sci. 1991, 13, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Tagami, H.; Kobayashi, H.; Kikuchi, K. A portable device using a closed chamber system for measuring transepidermal water loss: Comparison with the conventional method. Skin Res. Technol. 2002, 8, 7–12. [Google Scholar] [PubMed]
- Grubauer, G.; Elias, P.M.; Feingold, K.R. Transepidermal water loss; the signal for recovery of barrier structure and function. J. Lipid Res. 1989, 30, 323–333. [Google Scholar] [PubMed]
- Damien, F.; Boncheva, M. The extent of orthorhombic lipid phases in the stratum corneum determines the barrier efficiency of human skin in vivo. J. Investig. Dermatol. 2010, 130, 611–614. [Google Scholar] [CrossRef]
- Pilgram, G.S.K.; Vissers, D.C.J.; van der Meulen, H.; Pavel, S.; Lavrijsen, S.P.M.; Bouwstra, J.A.; Koerten, H.K. Aberrant lipid organization in stratum corneum of patients with atopic dermatitis and lamellar ichthyosis. J. Investig. Dermatol. 2001, 117, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Hatta, I.; Nakazawa, H.; Obata, Y.; Ohta, N.; Inoue, K.; Yagi, N. Novel method to observe subtle structural modulation of stratum corneum on applying chemical agents. Chem. Phys. Lipids 2010, 163, 381–389. [Google Scholar] [CrossRef]
- Imai, T.; Nakazawa, H.; Kato, S. Thermal phase transition behavior of lipid layers on a single human corneocyte cell. Chem. Phys. Lipids 2013, 174, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Bouwstra, J.A.; Gooris, G.S.; Salomons-de Vries, M.A.; van der Spek, J.A.; Bras, W. Structure of human stratum corneum as a function of temperature and hydration: A wide-angle X-ray diffraction study. Int. J. Pharm. 1992, 84, 205–216. [Google Scholar] [CrossRef]
- Masunaga, H.; Ogawa, H.; Takano, T.; Sasaki, S.; Goto, S.; Tanaka, T.; Ohashi, H. Multipurpose soft-material SAXS/WAXS/GISAXS beamline at SPring-8. Polym. J. 2011, 43, 471–477. [Google Scholar] [CrossRef]
- Miura, K.; Kawamoto, M.; Inoue, K.; Yamamoto, M.; Kumasaka, T.; Sugiura, M.; Yamane, A.; Moriyama, H. Commissioning for wide-angle routine proteomix bemline BL40B2: Protein crystallography and small angle scattering. SPring-8 User Exp. Rep. 2000, 4, 168. [Google Scholar]
- De Paepe, K.; Houben, E.; Adam, R.; Wiesemann, F.; Rogiers, V. Validation of the VapoMeter, a closed unventilated chamber system to assess transepidermal water loss vs. the open chamber Tewameter. Skin Res. Technol. 2005, 11, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Iwai, I.; Han, H.; Hollander, L.; Svensson, S.; Öfverstedt, L.G.; Anwar, J.; Brewer, J.; Bloksgaard, M.; Laloeuf, A.; Nosek, D.; et al. The human skin barrier is organized as stacked bilayers of fully extended ceramides with cholesterol molecules associated with the ceramide sphingoid moiety. J. Investig. Dermatol. 2012, 132, 2215–2225. [Google Scholar] [CrossRef] [PubMed]
- Swartzendruber, D.C.; Manganoro, A.; Madison, K.C.; Kremer, M.; Wertz, P.W.; Squier, C.A. Organization of the intercellular spaces of porcine epidermal and palatal stratum corneum: A quantitative study employing ruthenium tetroxide. Cell Tissue Res. 1995, 279, 271–276. [Google Scholar] [CrossRef]
- Hatta, I.; Ohta, N.; Inoue, K.; Yagi, N. Coexistence of two domains in intercellular lipid matrix of stratum corneum. Biochim. Biophys. Acta (BBA)-Biomembr. 2006, 1758, 1830–1836. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakazawa, H.; Imai, T.; Suzuki, M.; Akakabe, N.; Hatta, I.; Kato, S. Simultaneous Measurements of Structure and Water Permeability in an Isolated Human Skin Stratum Corneum Sheet. Polymers 2019, 11, 829. https://doi.org/10.3390/polym11050829
Nakazawa H, Imai T, Suzuki M, Akakabe N, Hatta I, Kato S. Simultaneous Measurements of Structure and Water Permeability in an Isolated Human Skin Stratum Corneum Sheet. Polymers. 2019; 11(5):829. https://doi.org/10.3390/polym11050829
Chicago/Turabian StyleNakazawa, Hiromitsu, Tomohiro Imai, Mika Suzuki, Natsuki Akakabe, Ichiro Hatta, and Satoru Kato. 2019. "Simultaneous Measurements of Structure and Water Permeability in an Isolated Human Skin Stratum Corneum Sheet" Polymers 11, no. 5: 829. https://doi.org/10.3390/polym11050829
APA StyleNakazawa, H., Imai, T., Suzuki, M., Akakabe, N., Hatta, I., & Kato, S. (2019). Simultaneous Measurements of Structure and Water Permeability in an Isolated Human Skin Stratum Corneum Sheet. Polymers, 11(5), 829. https://doi.org/10.3390/polym11050829





