Synthesis and Characterization of Clay Polymer Nanocomposites of P(4VP-co-AAm) and Their Application for the Removal of Atrazine
Abstract
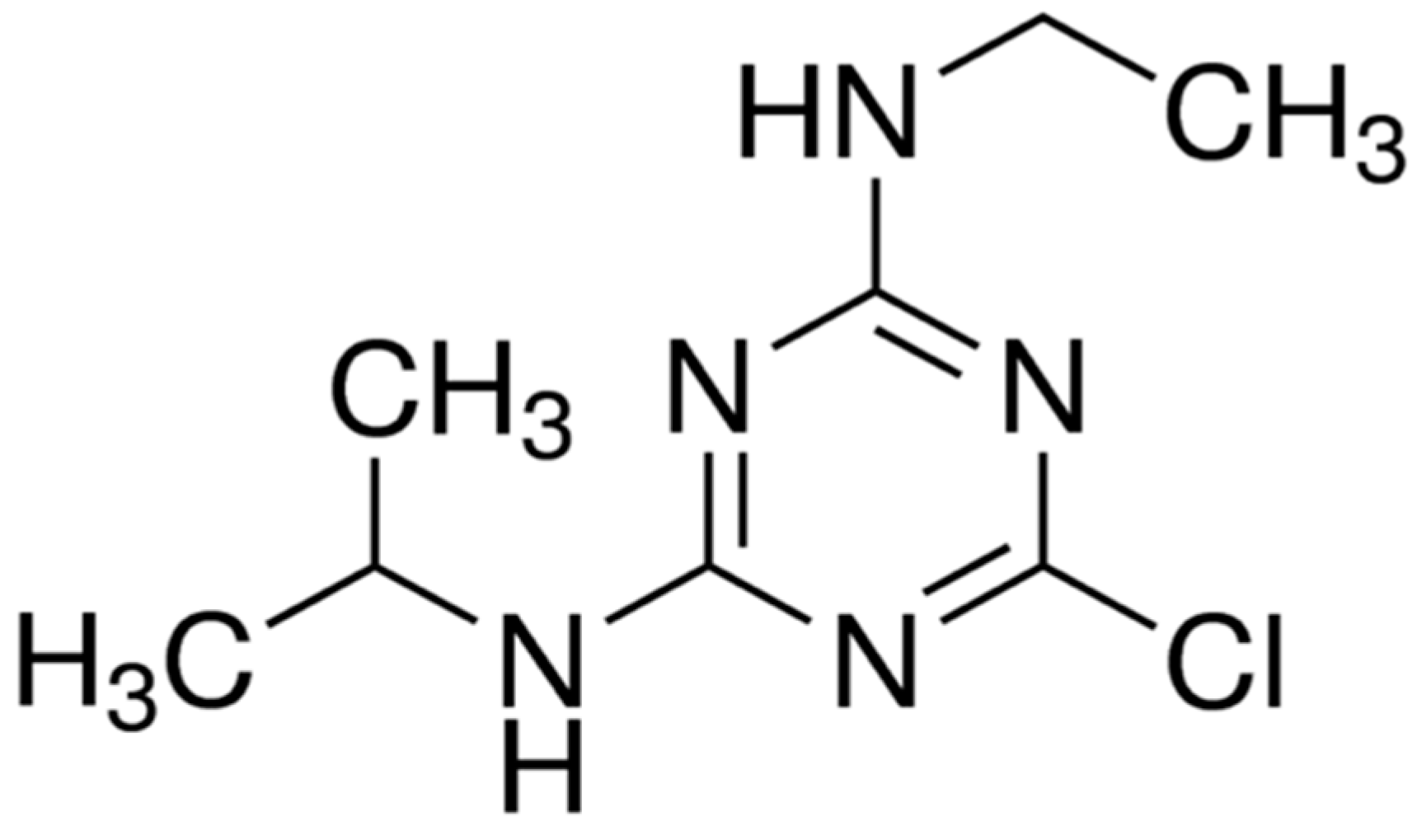
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Raw Clay Mineral Organo-Modification and Cationic Exchange Capacity (CEC)
2.3. Modification of the Homoionized Clay Mineral with HDTMA-Br and PTMA-Cl
2.4. Synthesis of the Organo-Modified Clay Nanocomposites (CC)
2.5. Swelling Behavior
2.6. Characterization
2.6.1. Raw Clay Mineral, Organo-Modified Clay Materials, and CC
2.6.2. ATZ Quantification and Adsorption Kinetics Evaluation
3. Results
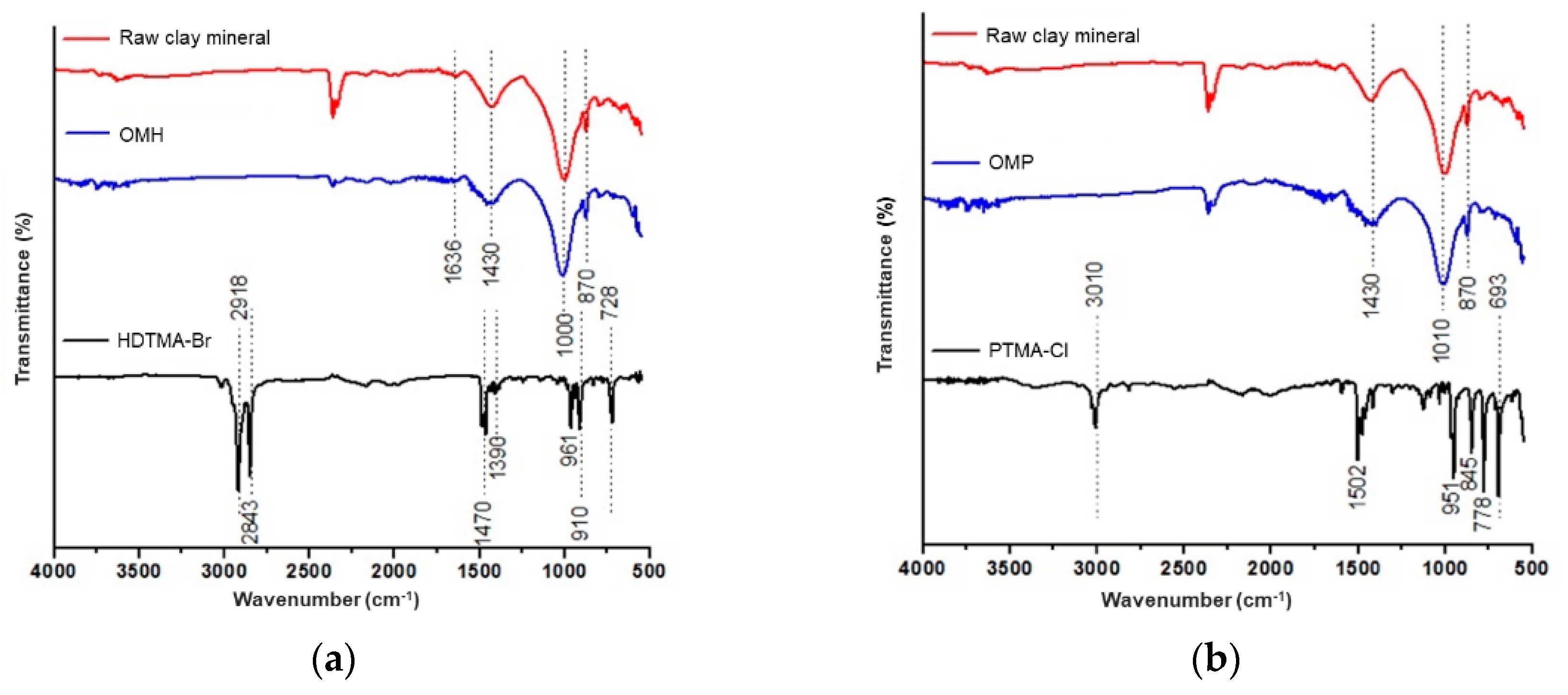
3.1. Organo-Modification of the Raw Clay Mineral
3.1.1. CEC
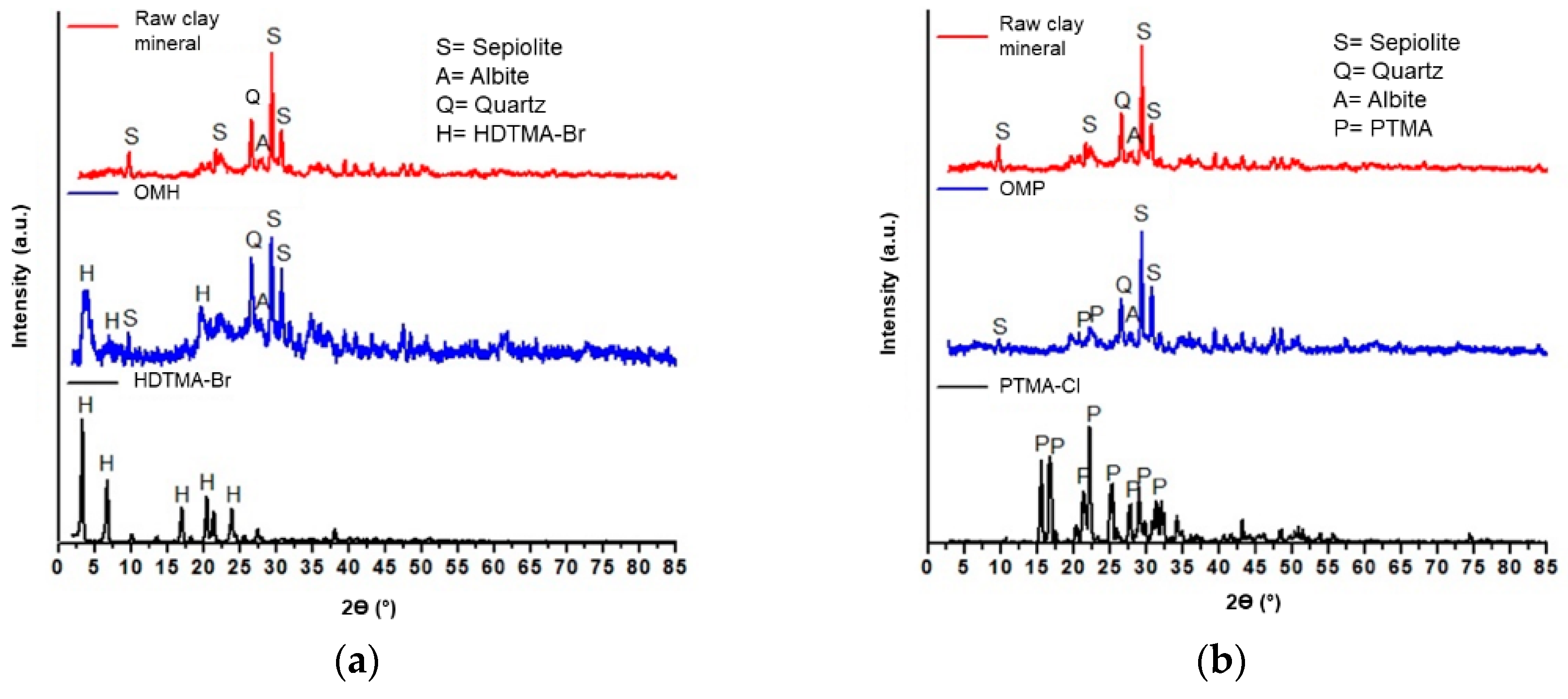
3.1.2. XRD Analysis
3.2. Organo-Modified Clay Nanocomposite (CC) Characterization
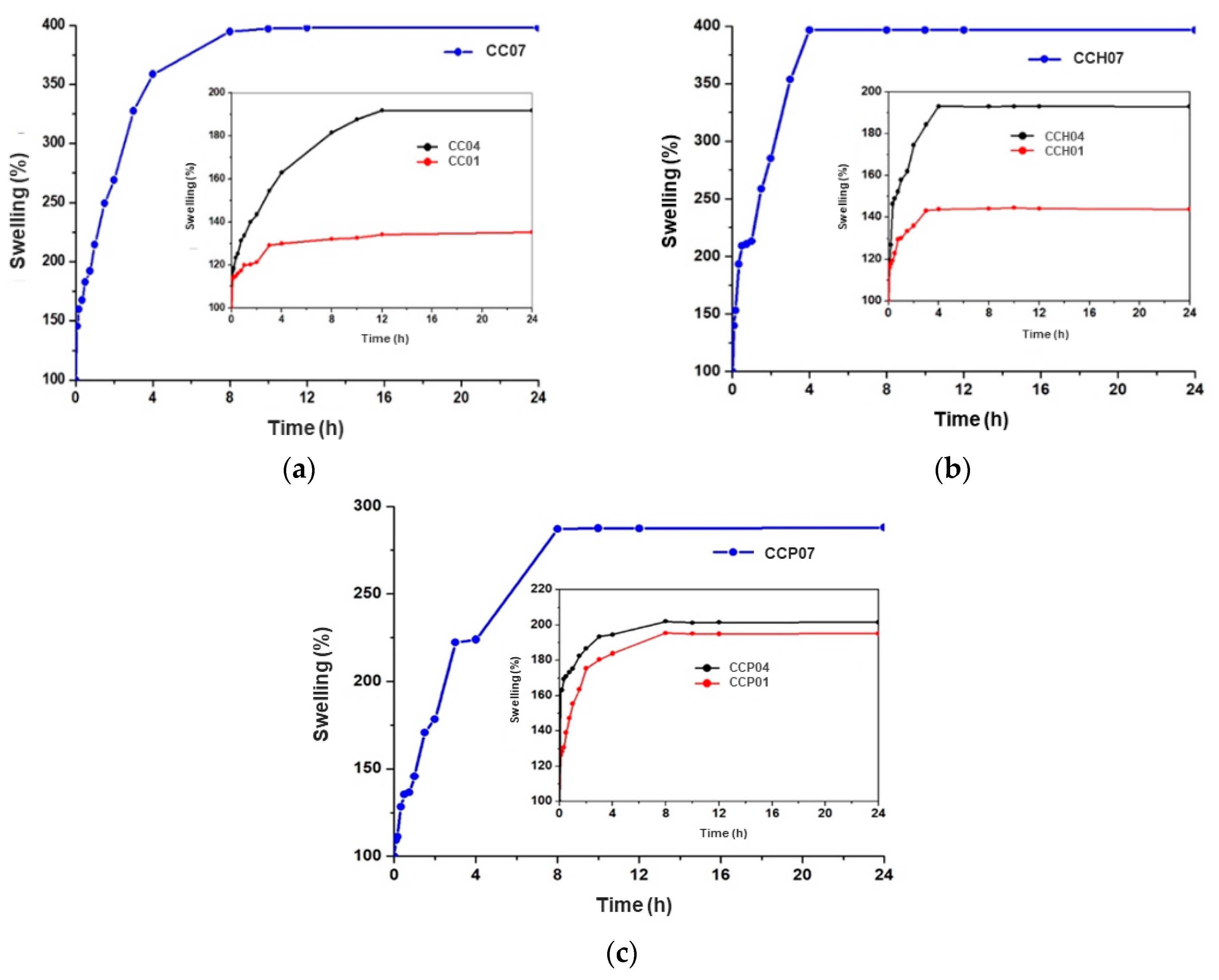
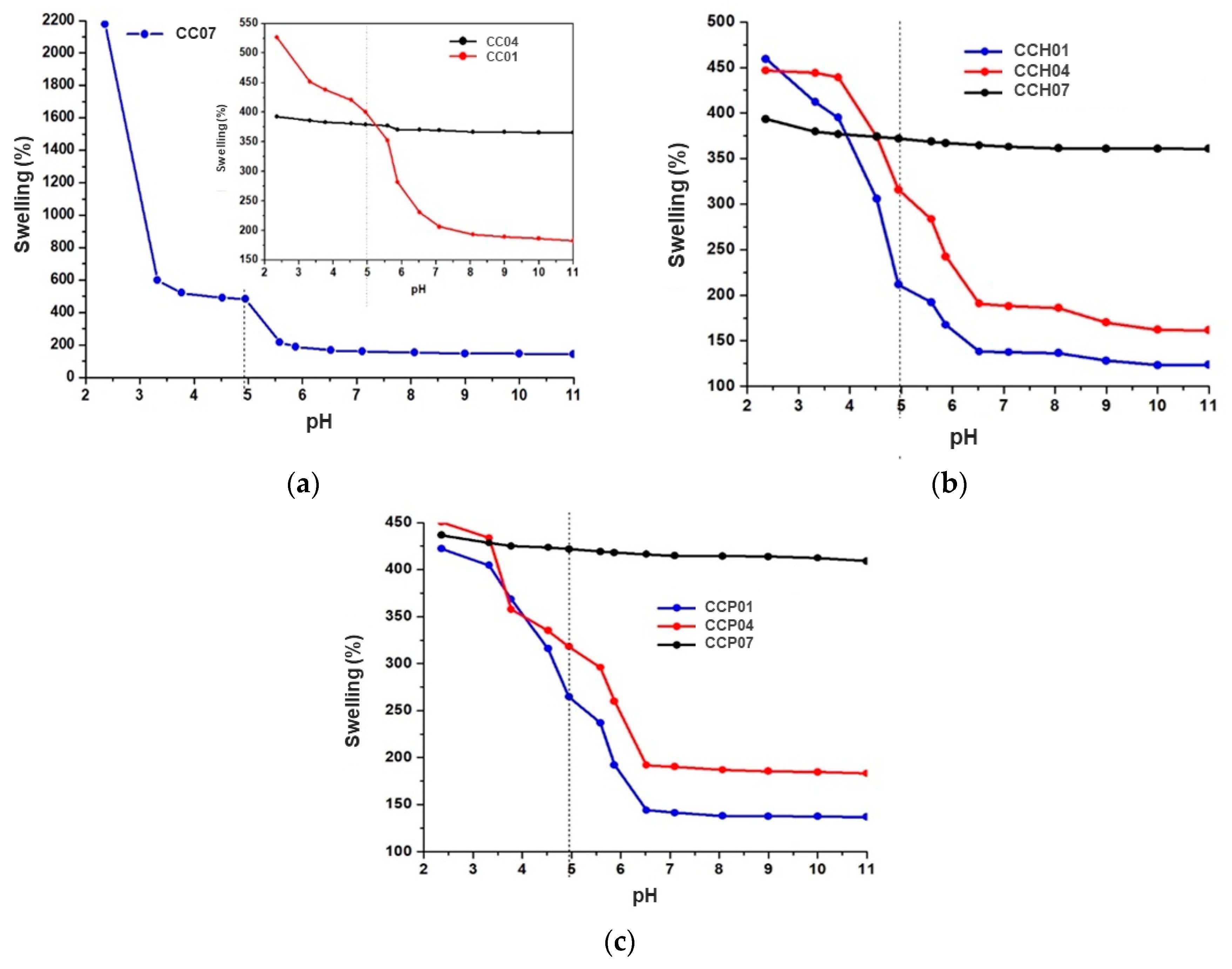
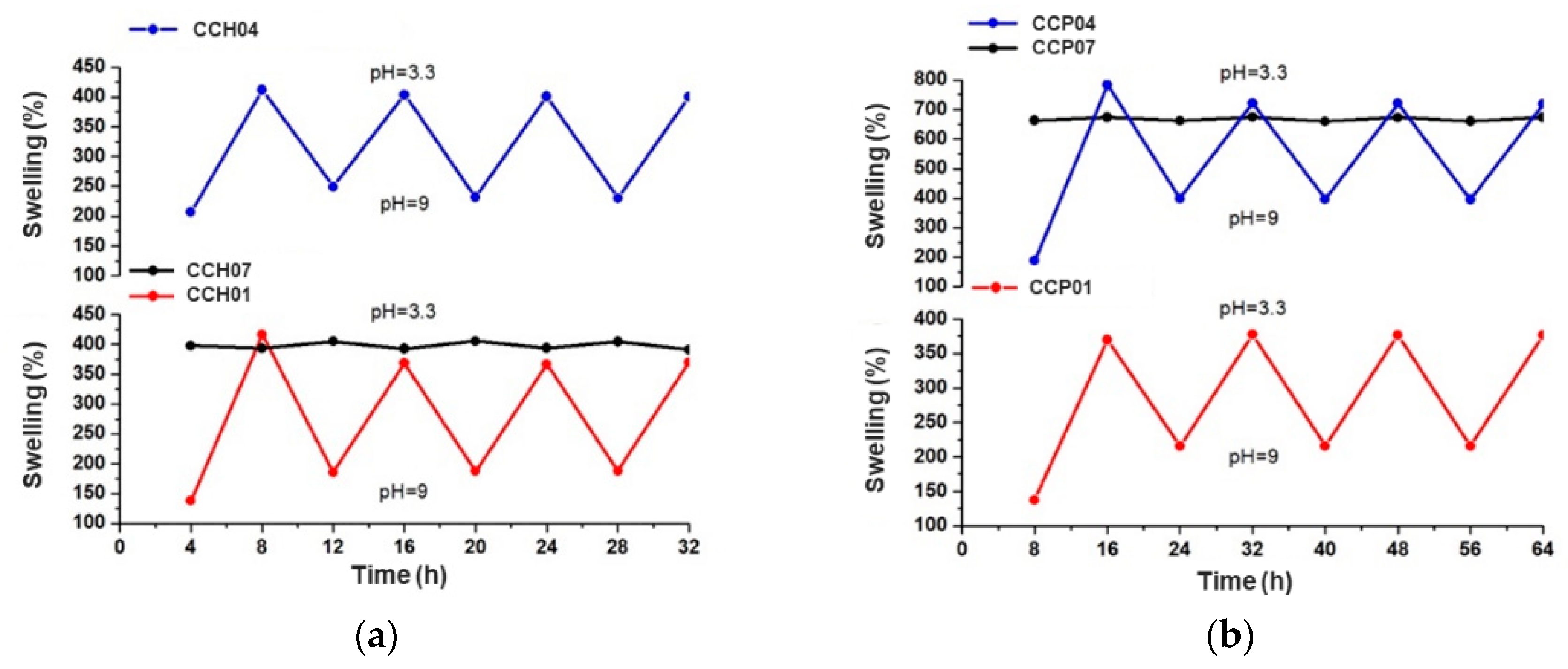
3.2.1. Swelling Behavior
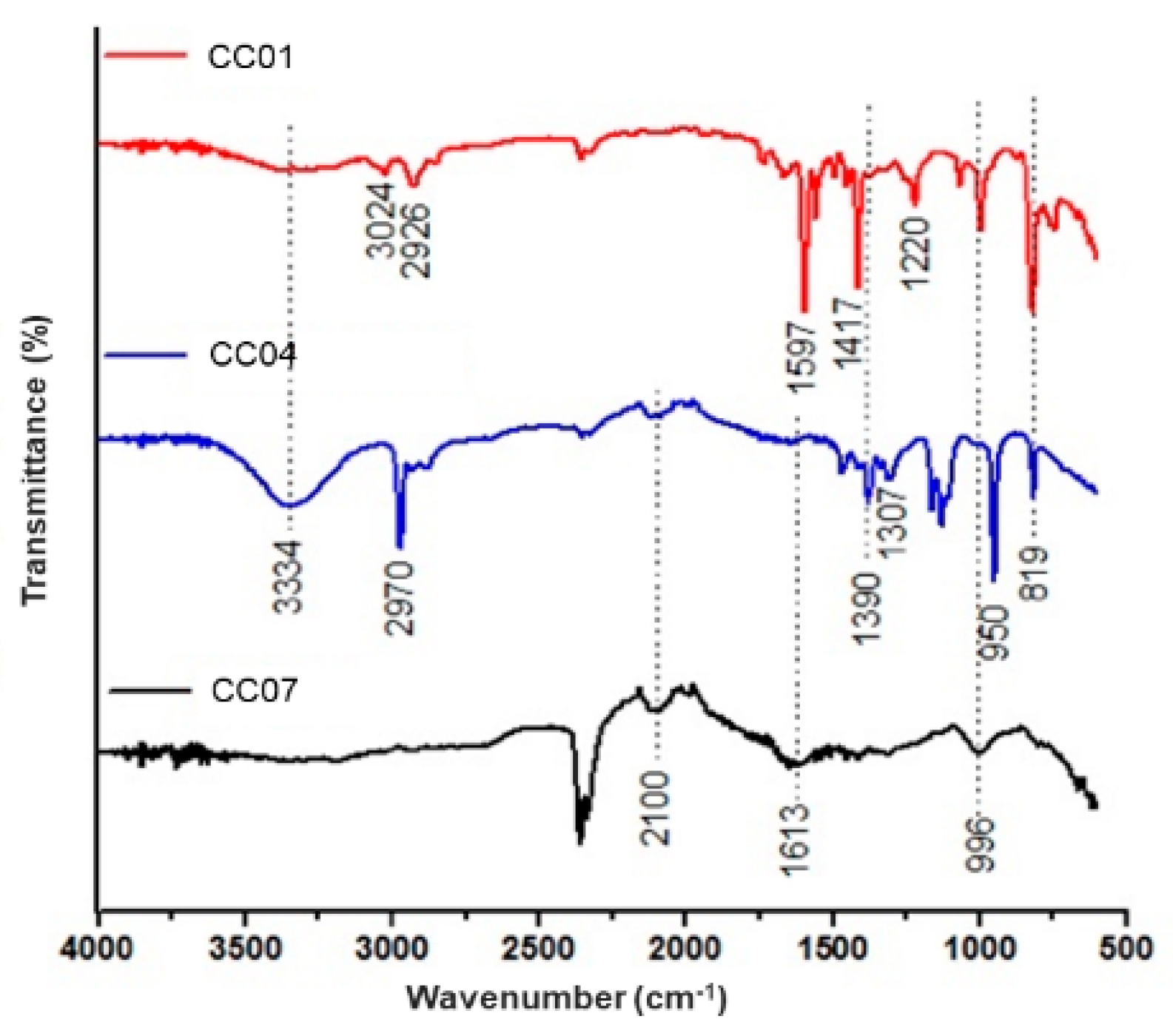
3.2.2. FTIR
3.2.3. SEM
3.2.4. Thermal Analysis
3.3. Adsorption Experiments
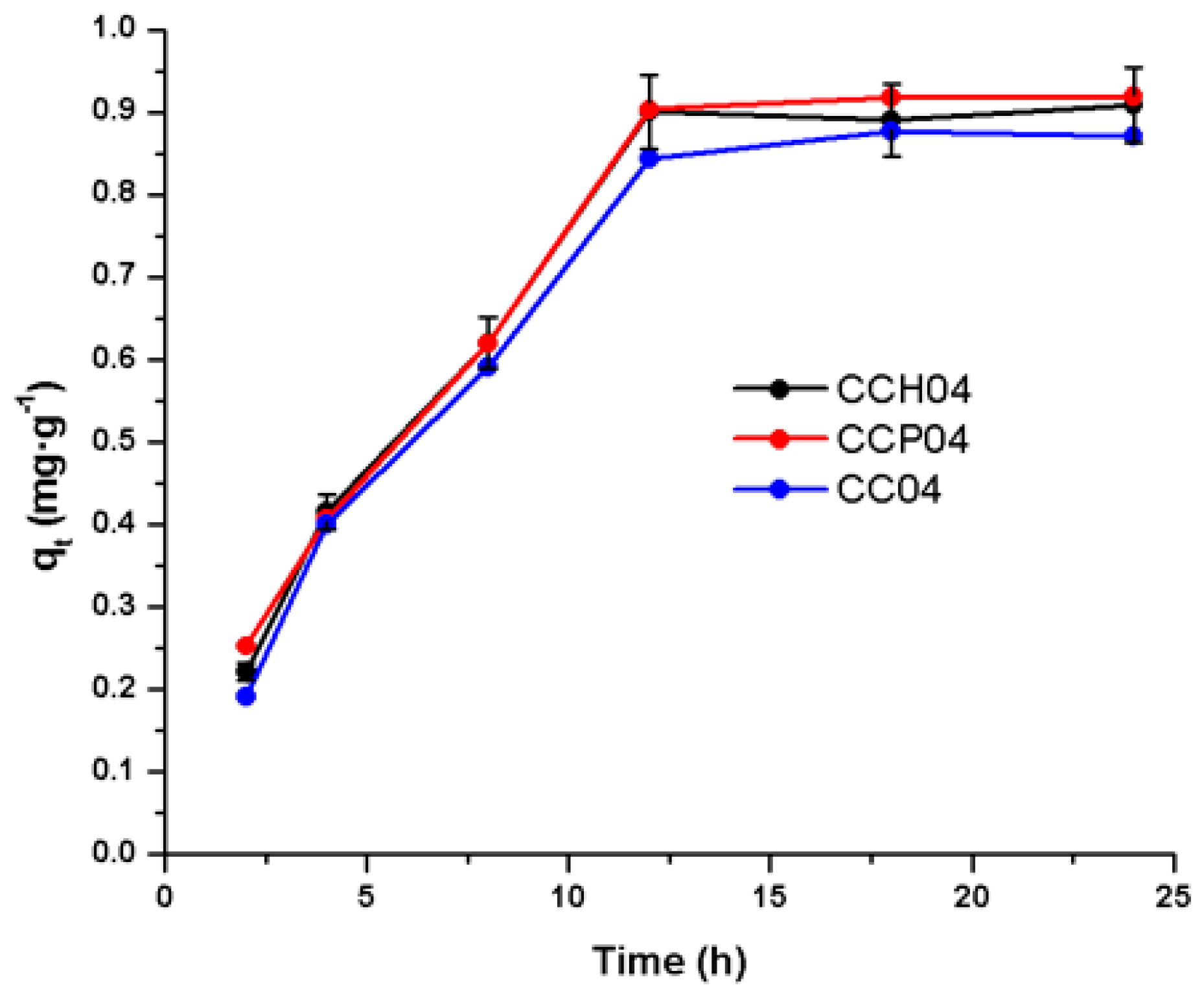
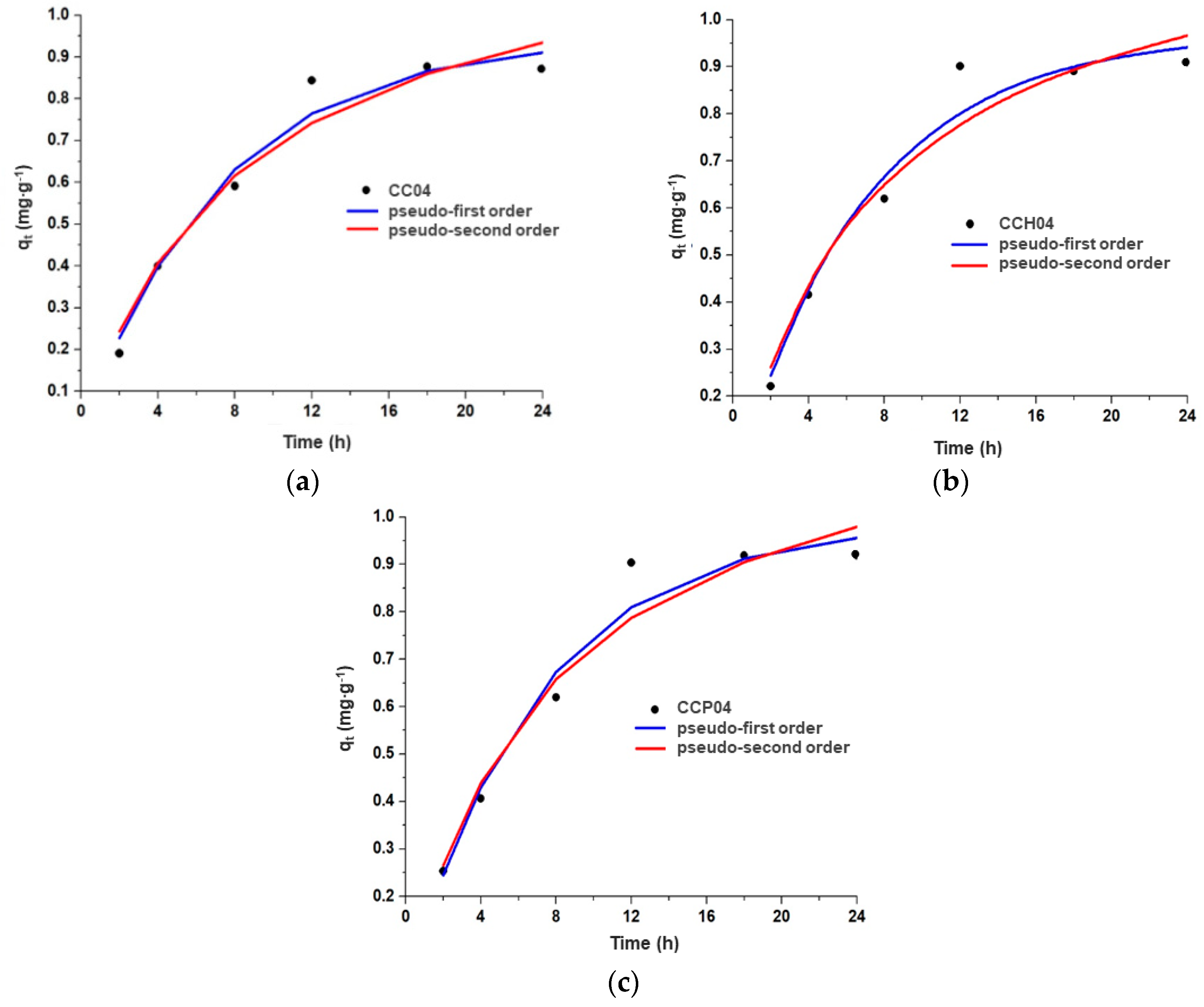
3.3.1. Adsorption Kinetics of the Synthesized Nanocomposites with the CC04 Copolymer
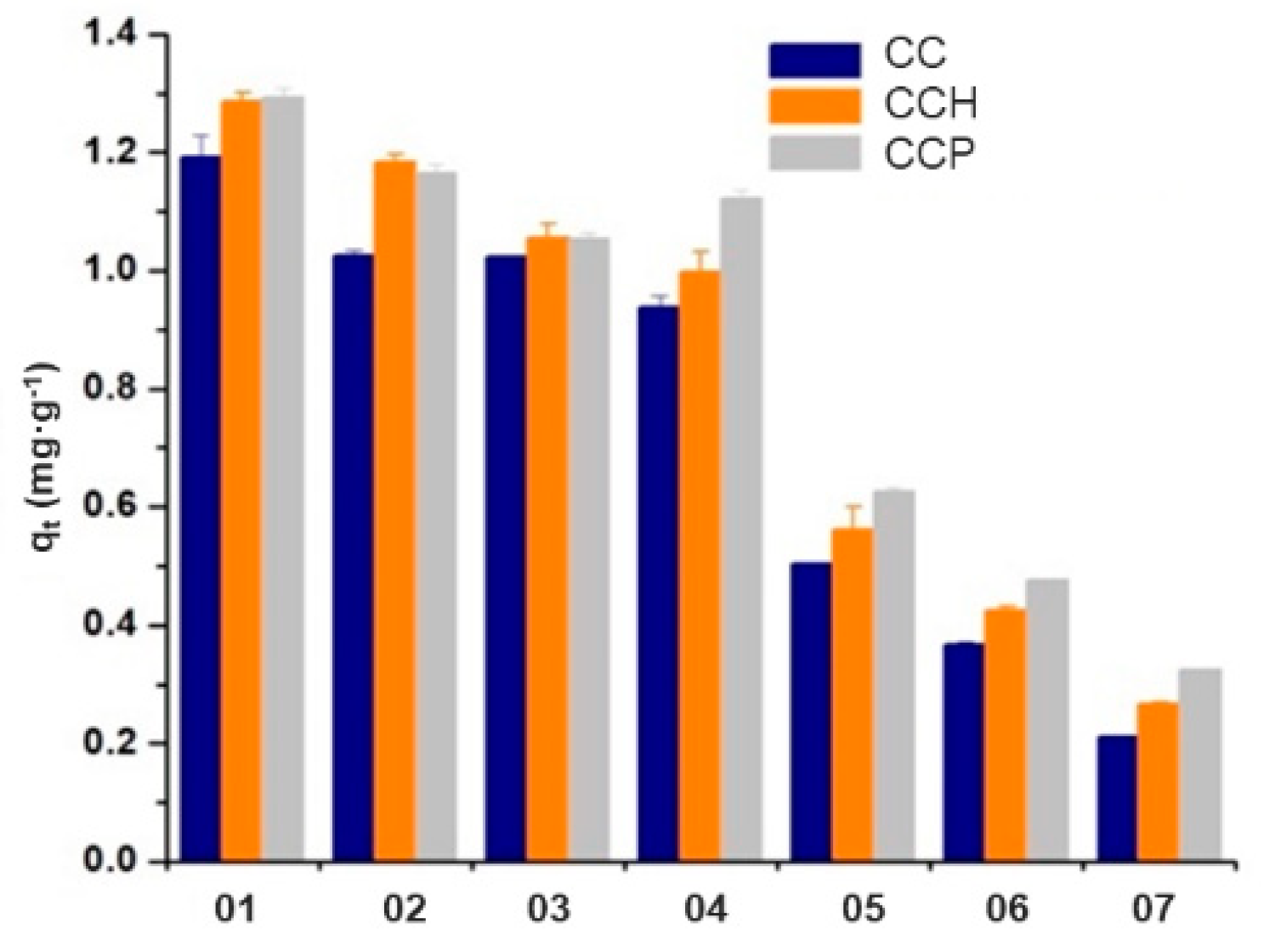
3.3.2. Effect of the 4VP and AAm Molar Fractions in the Obtained Nanocomposites
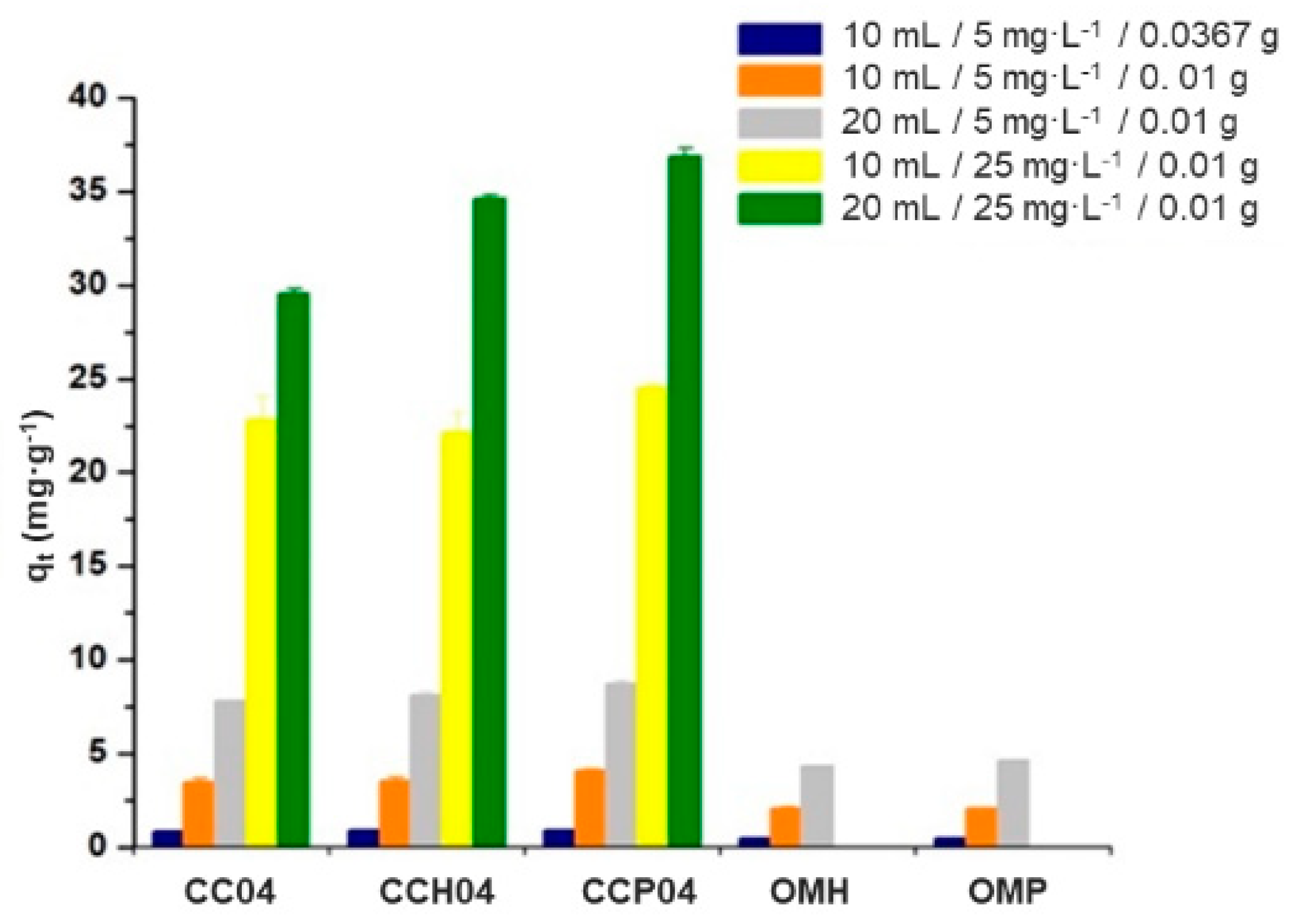
3.3.3. Effect of the Mass–Volume Ratio on the Capacity to Remove ATZ with the Modified and Synthesized Materials
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rinsky, J.L.; Hopenhayn, C.; Golla, V.; Browning, S.; Bush, H.M. Atrazine exposure in public drinking water and preterm birth. Public Health Rep. 2012, 127, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Golla, V.; Nelms, J.; Taylor, R.; Mishra, S. Pesticide concentrations in drinking water from farm homes: Variation between community water supply and well-water. J. Environ. Sci. Eng. 2011, 5, 955–961. [Google Scholar]
- Nödler, K.; Licha, T.; Voutsa, D. Twenty years later—Atrazine concentrations in selected coastal waters of the Mediterranean and the Baltic Sea. Mar. Pollut. Bull. 2013, 70, 112–118. [Google Scholar] [CrossRef]
- Graymore, M.; Stagnitti, F.; Allinson, G. Impacts of atrazine in aquatic ecosystems. Environ. Int. 2001, 26, 483–495. [Google Scholar] [CrossRef]
- Gardi, I.; Nir, S.; Mishael, Y. Filtration of triazine herbicides by polymer-clay sorbents: Coupling an experimental mechanistic approach with empirical modeling. Water Res. 2015, 70, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Fu, L.; Jin, C.; Meng, Z.; Yang, N. Study on the Isolation of Two Atrazine-Degrading Bacteria and the Development of a Microbial Agent. Microorganisms 2019, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Katoon, H.; Nai, J.P.N. Augmentation of Atrazine biodegradation by two Bacilli immobilized on α-Fe2O3 magnetic nanoparticles. Sci. Rep. UK 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Monard, C.; Vandekoornhuyse, P.; Le Bot, B.; Binet, F. Relationship between bacterial diversity and function under biotic control: The soil pesticide degraders as a case study. ISME J. 2011, 5, 1048–1056. [Google Scholar] [CrossRef]
- Meakins, N.C.; Bubb, J.M.; Lester, J.N. The mobility, partitioning and degradation of atrazine and simazine in the salt marsh environment. Mar. Pollut. Bull. 1995, 30, 812–819. [Google Scholar] [CrossRef]
- Youssef, L.; Younes, G.; Al-Oweini, R. Photocatalytic degradation of atrazine by heteropolyoxotungstates. J. Taibah Univ. Sci. 2019, 13, 274–279. [Google Scholar] [CrossRef]
- Díaz-Nava, M.C.; Solache-Ríos, M.; Contreras-Olivares, N. Enhanced decolorization of dyes by an iron modified clay and thermodynamic parameters. Water Sci. Technol. 2016, 73, 2007–2016. [Google Scholar]
- Hernández-Hernández, K.A.; Illescas, J.; Díaz-Nava, M.C.; Muro-Urista, C.; Martínez-Gallegos, S.; Ortega-Aguilar, R.E. Polymer-Clay Nanocomposites and Composites: Structures, Characteristics, and their Applications in the Removal of Organic Compounds of Environmental Interest. Med. Chem. 2016, 6, 201–210. [Google Scholar] [CrossRef]
- Hernández-Hernández, K.A.; Solache-Ríos, M.; Díaz-Nava, M.C. Removal of brilliant blue FCF from aqueous solutions using an unmodified and iron modified bentonite and the thermodynamic parameters of the process. Water Air Soil Poll. 2013, 24, 1–11. [Google Scholar] [CrossRef]
- Díaz-Nava, M.C.; Olguín, M.T.; Solache-Ríos, M. Adsorption of phenol onto surfactants modified bentonite. J. Incl. Phenom. Macrocycl. 2012, 74, 67–75. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, T.; He, T.; Chen, L. Removal of crystal violet by clay/PNIPAm nanocomposite hydrogels with various clay contents. Appl. Clay Sci. 2014, 90, 1–5. [Google Scholar] [CrossRef]
- Zadaka, D.; Nir, S.; Radian, A.; Mishael, A.G. Atrazine removal from water by polycation–clay composites: Effect of dissolved organic matter and comparison to activated carbon. Water Res. 2009, 43, 677–683. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layerd silicate nanocomposites preparation, properties and uses of a new class of materials. Mater. Sci. Eng. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Saavedra-Labastida, E.; Díaz-Nava, M.C.; Illescas, J.; Muro, C. Comparison of the removal of an anionic dye from aqueous solutions by adsorption with organically modified clays and their composites. Water Air Soil Pollut. 2019, 230, 1–26. [Google Scholar] [CrossRef]
- Ortega-Aguirre, S.; Díaz-Nava, M.C.; Solache-Ríos, M.J.; Illescas, J. Alginate-iron modified zeolite beads biocomposite for removal of azo dye from water medium. MRS Adv. 2018, 3, 3769–3773. [Google Scholar] [CrossRef]
- Remigio-Reyes, G.D.; Illescas, J.; Díaz-Nava, M.C. Removal of bromophenol blue dye from aqueous solutions through the synthesis and characterization of a polymer-clay composite. MRS Adv. 2018, 3, 3751–3755. [Google Scholar] [CrossRef]
- Hernández-Hernández, K.A.; Illescas, J.; Díaz-Nava, M.C.; Martínez-Gallegos, S.; Muro-Urista, C.; Ortega-Aguilar, R.E.; Rodríguez-Alba, E.; Rivera, E. Preparation of nanocomposites for the removal of phenolic compounds from aqueous solutions. Appl. Clay Sci. 2018, 157, 212–217. [Google Scholar] [CrossRef]
- Yanovska, E.S.; Vretik, L.O.; Nikolaeva, O.A.; Polonska, Y.; Sternik, D.; Kichkiruk, O.Y. Synthesis and Adsorption Properties of 4-Vinylpyridine and Styrene Copolymer in Situ Immobilized on Silica Surface. Nanoscale Res. Lett. 2017, 12, 1–6. [Google Scholar] [CrossRef]
- Rassu, M.; Alzari, V.; Nuvoli, D.; Nuvoli, L.; Sanna, D.; Sanna, V.; Malucelli, G.; Mariani, A. Semi-interpenetrating polymer networks of methyl cellulose and polyacrylamide prepared by frontal polymerization. J. Polym. Sci. Pol. Chem. 2017, 55, 1268–1274. [Google Scholar] [CrossRef]
- Scognamillo, S.; Alzari, V.; Nuvoli, D.; Mariani, A. Thermoresponsive super water absorbent hydrogels prepared by frontal polymerization. J. Polym. Sci. Pol. Chem. 2010, 48, 2486–2490. [Google Scholar] [CrossRef]
- Martino, L.; Guigo, N.; Van Berkel, J.G.; Sbirrazzuoli, N. Influence of organically modified montmorillonite and sepiolite clays on the physical properties of bio-based poly(ethylene 2,5-furandicarboxylate). Compos. Part B-Eng. 2017, 110, 96–105. [Google Scholar] [CrossRef]
- Chen, H.; Lu, H.; Zhou, Y.; Zheng, M.; Ke, C.; Zeng, D. Study on thermal properties of polyurethane nanocomposites based on organo-sepiolite. Polym. Degrad. Stabil. 2012, 97, 242–247. [Google Scholar] [CrossRef]
- Lowe, D.J.; Chapman, A.V.; Cook, S.; Busfield, J.J.C. Natural Rubber Nanocomposites by In Situ Modification of Clay. Macromol. Mater. Eng. 2011, 296, 693–702. [Google Scholar] [CrossRef]
- Mariani, A.; Nuvoli, D.; Alzari, V.; Pini, M. Phosphonium-Based ionic liquids as a new class of radical initiators and their use in gas-free frontal polymerization. Macromolecules 2008, 41, 5191–5196. [Google Scholar] [CrossRef]
- ANSI/API 13B-1. Recommended Practice for Field Testing-Water-Based Drilling Fluids, 5th ed.; API Publishing Services: Washington, DC, USA, 2017. [Google Scholar]
- ASTM C837-09. Standard Test Method for Methylene Blue Index of Clay; ASTM International: West Conshohocken, PA, USA, 2014. [Google Scholar]
- Zhuang, G.; Gao, J.; Chen, H.; Zhang, Z. A new one-step method for physical purification and organic modification of sepiolite. Appl. Clay Sci. 2018, 153, 1–8. [Google Scholar] [CrossRef]
- Moreira, M.A.; Ciuffi, K.J.; Rives, V.; Vicente, M.A.; Trujillano, R.; Gil, A.; Korili, S.A.; de Faria, E.H. Effect of chemical modification of palygorskite and sepiolite by 3-aminopropyltriethoxisilane on adsorption of cationic and anionic dyes. Appl. Clay Sci. 2017, 135, 394–404. [Google Scholar] [CrossRef]
- Lemić, J.; Tomašević-Čanović, M.; Djuričić, M.; Stanić, T. Surface modification of sepiolite with quaternary amines. J. Colloid Interface Sci. 2005, 292, 11–19. [Google Scholar] [CrossRef]
- Ortega, A.; Alarcón, D.; Muñoz-Muñoz, F.; Garzón-Fontecha, A.; Burillo, G. Radiation grafting of pH-sensitive acrylic acid and 4-vinylpyridine onto nylon-6 using one- and two-step methods. Radiat. Phys. Chem. 2015, 109, 6–12. [Google Scholar] [CrossRef]
- Clara-Rahola, J.; Moscoso, A.; Ruiz-Muelle, A.B.; Laurenti, M.; Formanek, P.; Lopez-Romero, J.M.; Fernández, I.; Diaz, J.F.; Jorge Rubio-Retama, J.; Fery, A.; et al. Au@p4VP core@shell pH-sensitive nanocomposites suitable for drug entrapment. J. Colloid Interface Sci. 2018, 514, 704–714. [Google Scholar] [CrossRef] [PubMed]
- Lazo, J.C.; Navarro, A.E.; Sun-Kou, M.R.; Llanos, B.P. Síntesis y caracterización de arcillas organofílicas y su aplicación como adsorbentes del fenol. Rev. Soc. Quím. Perú 2008, 74, 3–19. [Google Scholar]
- Kleinübing, S.J.; Gaia, F.; Bertagnolli, C.; Carlos da Silva, M.G. Extraction of alginate biopolymer present in marine alga sargassum filipendula and bioadsorption of metallic ions. Mater. Res. 2013, 16, 481–488. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]


| Sample Codes | 4VP (mol. %) | AAm (mol. %) | OMH/OMP (wt. %) | BIS (mol. %) | TBPPS (mol. %) |
|---|---|---|---|---|---|
| CC01 | 100 | 0 | - | 3 | 0.25 |
| CC02 | 85 | 15 | - | 3 | 0.25 |
| CC03 | 65 | 35 | - | 3 | 0.25 |
| CC04 | 50 | 50 | - | 3 | 0.25 |
| CC05 | 30 | 70 | - | 3 | 0.25 |
| CC06 | 10 | 90 | - | 3 | 0.25 |
| CC07 | 0 | 100 | - | 3 | 0.25 |
| CCH01 | 100 | 0 | 5 | 3 | 0.25 |
| CCH02 | 85 | 15 | 5 | 3 | 0.25 |
| CCH03 | 65 | 35 | 5 | 3 | 0.25 |
| CCH04 | 50 | 50 | 5 | 3 | 0.25 |
| CCH05 | 30 | 70 | 5 | 3 | 0.25 |
| CCH06 | 10 | 90 | 5 | 3 | 0.25 |
| CCH07 | 0 | 100 | 5 | 3 | 0.25 |
| CCP01 | 100 | 0 | 5 | 3 | 0.25 |
| CCP02 | 85 | 15 | 5 | 3 | 0.25 |
| CCP03 | 65 | 35 | 5 | 3 | 0.25 |
| CCP04 | 50 | 50 | 5 | 3 | 0.25 |
| CCP05 | 30 | 70 | 5 | 3 | 0.25 |
| CCP06 | 10 | 90 | 5 | 3 | 0.25 |
| CCP07 | 0 | 100 | 5 | 3 | 0.25 |
| Raw Clay Mineral (g) | Volume of Methylene Blue (mL) | CEC (meq/100 g) |
|---|---|---|
| 0.5006 | 15 | 29.96 |
| 0.5003 | 15 | 29.86 |
| 0.4996 | 14 | 28.02 |
| 0.1006 | 3 | 29.82 |
| 0.1004 | 3 | 29.88 |
| 0.1002 | 3 | 29.94 |
| Sample Code | T5 (°C) | Tg (°C) |
|---|---|---|
| Raw clay mineral | 500 | ND 1 |
| OMH | 480 | ND 1 |
| OMP | 520 | ND 1 |
| CC04 | 150 | 165 |
| CCH04 | 140 | 167 |
| CCP04 | 140 | 168 |
| Material | qe(exp) (mg∙g−1) | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||
|---|---|---|---|---|---|---|---|
| KL (h−1) | qe (mg∙g−1) | R2 | K2 (g∙mg−1·h−1) | qe (mg∙g−1) | R2 | ||
| CC04 | 0.845 | 0.137 | 0.945 | 0.986 | 0.095 | 1.258 | 0.978 |
| CCH04 | 0.900 | 144 | 0.972 | 0.983 | 0.100 | 1.278 | 0.974 |
| CCP04 | 0.903 | 143 | 0.988 | 0.984 | 0.098 | 1.298 | 0.976 |
| Material | Copolymer (CC) | HDTMA Nanocomposites (CCH) | PTMA Nanocomposites (CCP) | |||
|---|---|---|---|---|---|---|
| Removal (%) | qt Average (mg∙g−1) | Removal (%) | qt Average (mg∙g−1) | Removal (%) | qt Average (mg∙g−1) | |
| 01 | 92 | 1.192 ± 0.036 | 94 | 1.286 ± 0.015 | 95 | 1.293 ± 0.014 |
| 92 | 94 | 95 | ||||
| 02 | 81 | 1.026 ± 0.007 | 82 | 1.185 ± 0.011 | 83 | 1.165 ± 0.015 |
| 81 | 82 | 83 | ||||
| 03 | 76 | 1.024 ± 0.002 | 79 | 1.056 ± 0.023 | 80 | 1.054 ± 0.009 |
| 75 | 78 | 80 | ||||
| 04 | 71 | 0.938 ± 0.019 | 73 | 0.998 ± 0.033 | 78 | 1.122 ± 0.015 |
| 69 | 73 | 78 | ||||
| 05 | 37 | 0.505 ± 0.002 | 39 | 0.562 ± 0.038 | 44 | 0.625 ± 0.007 |
| 36 | 41 | 46 | ||||
| 06 | 28 | 0.366 ± 0.005 | 31 | 0.425 ± 0.007 | 35 | 0.476 ± 0.001 |
| 27 | 31 | 36 | ||||
| 07 | 15 | 0.210 ± 0001 | 19 | 0.266 ± 0.003 | 22 | 0.324 ± 0.002 |
| 16 | 21 | 24 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramírez-Gómez, J.A.; Illescas, J.; Díaz-Nava, M.d.C.; Muro-Urista, C.; Martínez-Gallegos, S.; Rivera, E. Synthesis and Characterization of Clay Polymer Nanocomposites of P(4VP-co-AAm) and Their Application for the Removal of Atrazine. Polymers 2019, 11, 721. https://doi.org/10.3390/polym11040721
Ramírez-Gómez JA, Illescas J, Díaz-Nava MdC, Muro-Urista C, Martínez-Gallegos S, Rivera E. Synthesis and Characterization of Clay Polymer Nanocomposites of P(4VP-co-AAm) and Their Application for the Removal of Atrazine. Polymers. 2019; 11(4):721. https://doi.org/10.3390/polym11040721
Chicago/Turabian StyleRamírez-Gómez, Jorge A., Javier Illescas, María del Carmen Díaz-Nava, Claudia Muro-Urista, Sonia Martínez-Gallegos, and Ernesto Rivera. 2019. "Synthesis and Characterization of Clay Polymer Nanocomposites of P(4VP-co-AAm) and Their Application for the Removal of Atrazine" Polymers 11, no. 4: 721. https://doi.org/10.3390/polym11040721
APA StyleRamírez-Gómez, J. A., Illescas, J., Díaz-Nava, M. d. C., Muro-Urista, C., Martínez-Gallegos, S., & Rivera, E. (2019). Synthesis and Characterization of Clay Polymer Nanocomposites of P(4VP-co-AAm) and Their Application for the Removal of Atrazine. Polymers, 11(4), 721. https://doi.org/10.3390/polym11040721







