Tablet of Ximenia Americana L. Developed from Mucoadhesive Polymers for Future Use in Oral Treatment of Fungal Infections
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Plant Material
2.3. Obtaining the X. Americana Extracts
2.4. Evaluation of Antifungal Activity
2.5. Binary Mixtures
2.6. Thermal Analysis
2.7. FTIR
2.8. Formulation Development
2.9. Mucoadhesion
3. Results and Discussion
3.1. Evaluation of Antifungal Activity
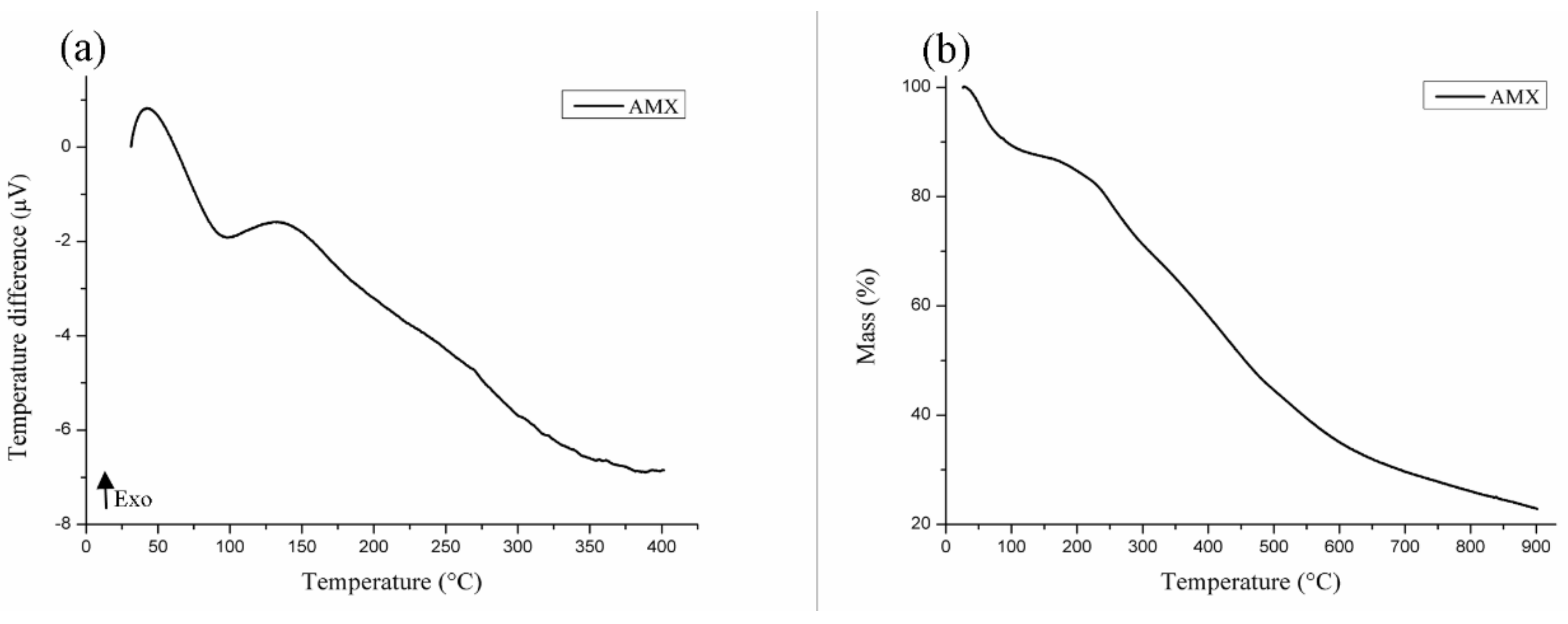
3.2. Thermal Characterization of AMX
3.3. Compatibility Study
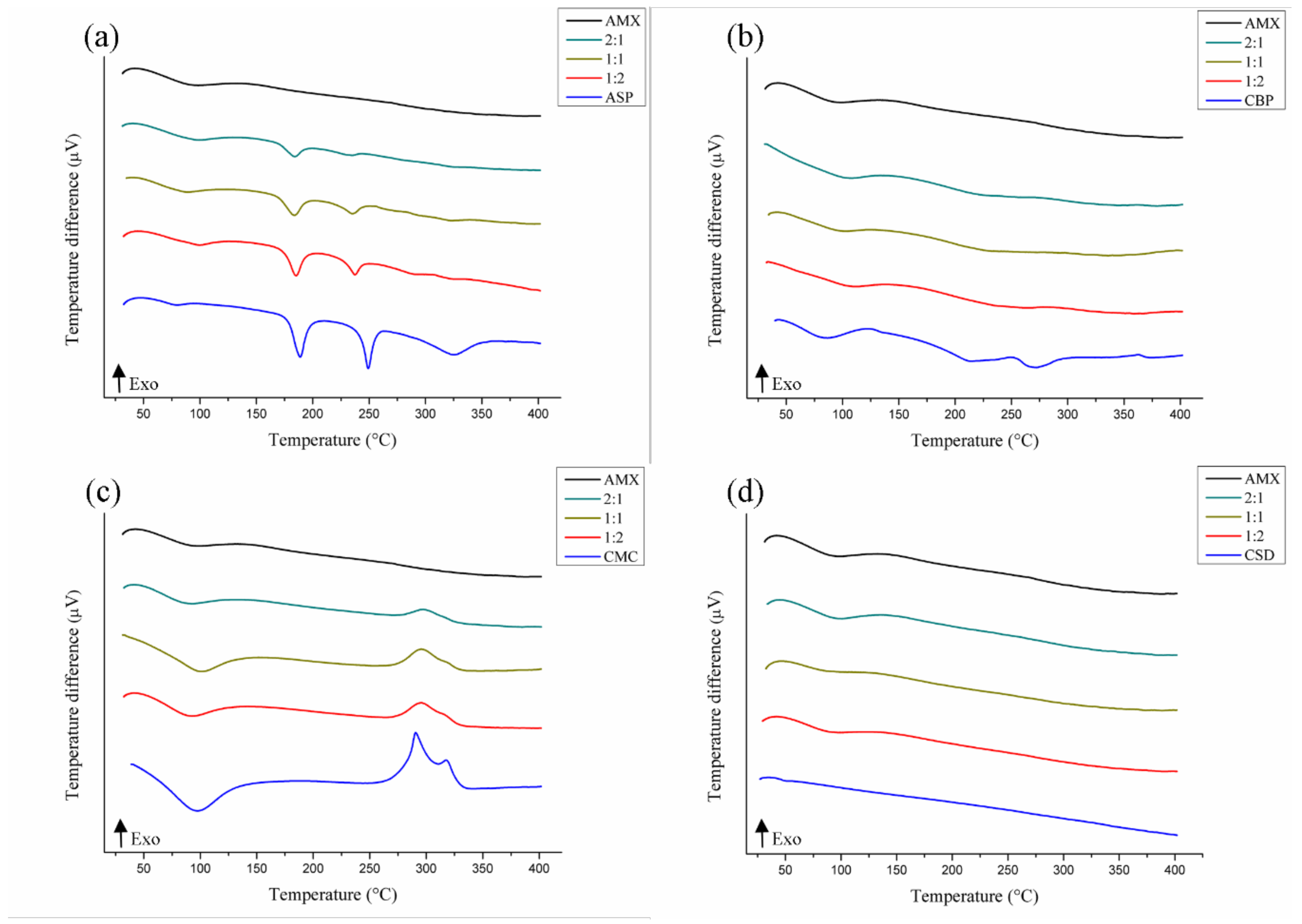
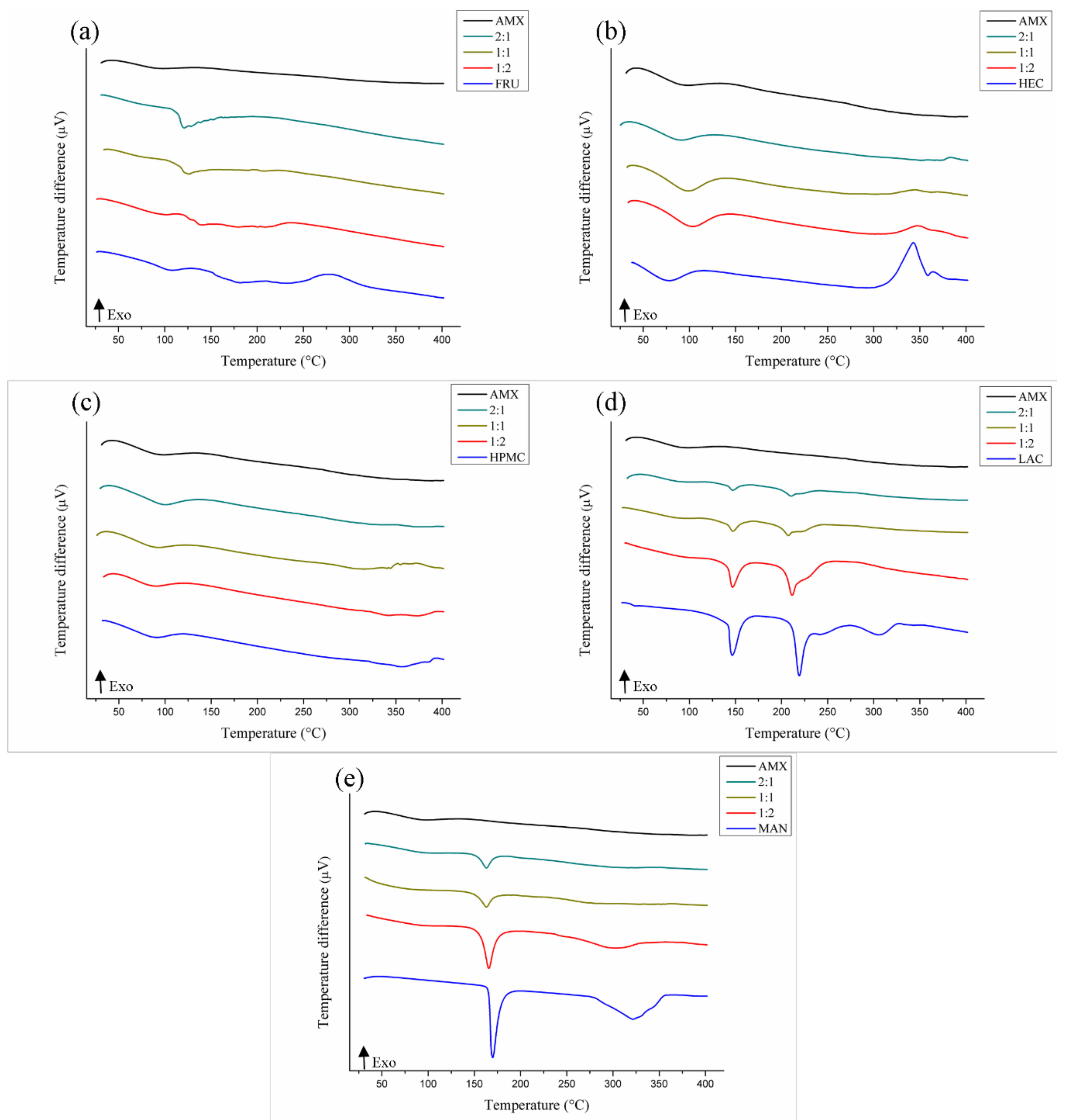
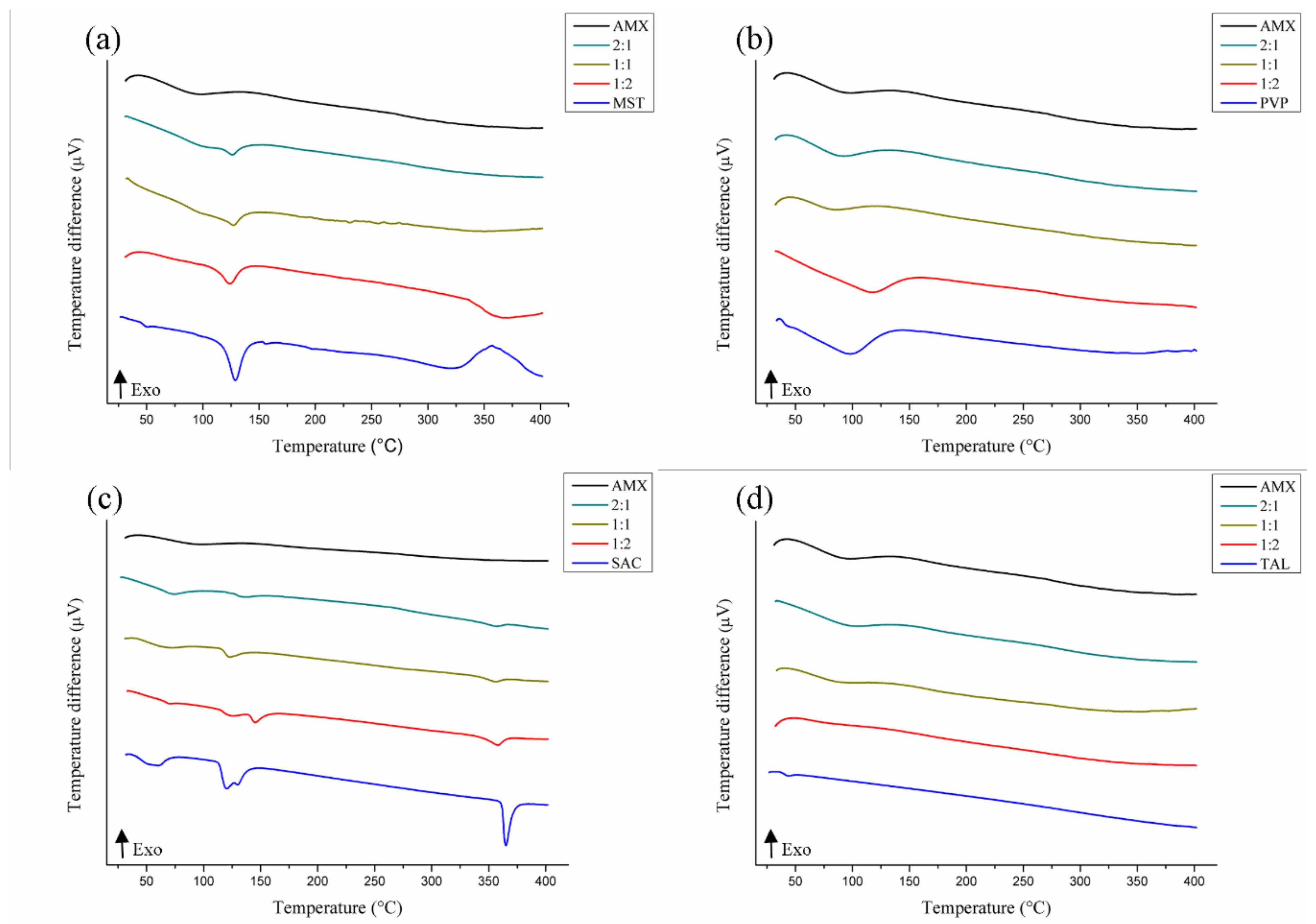
3.3.1. DTA
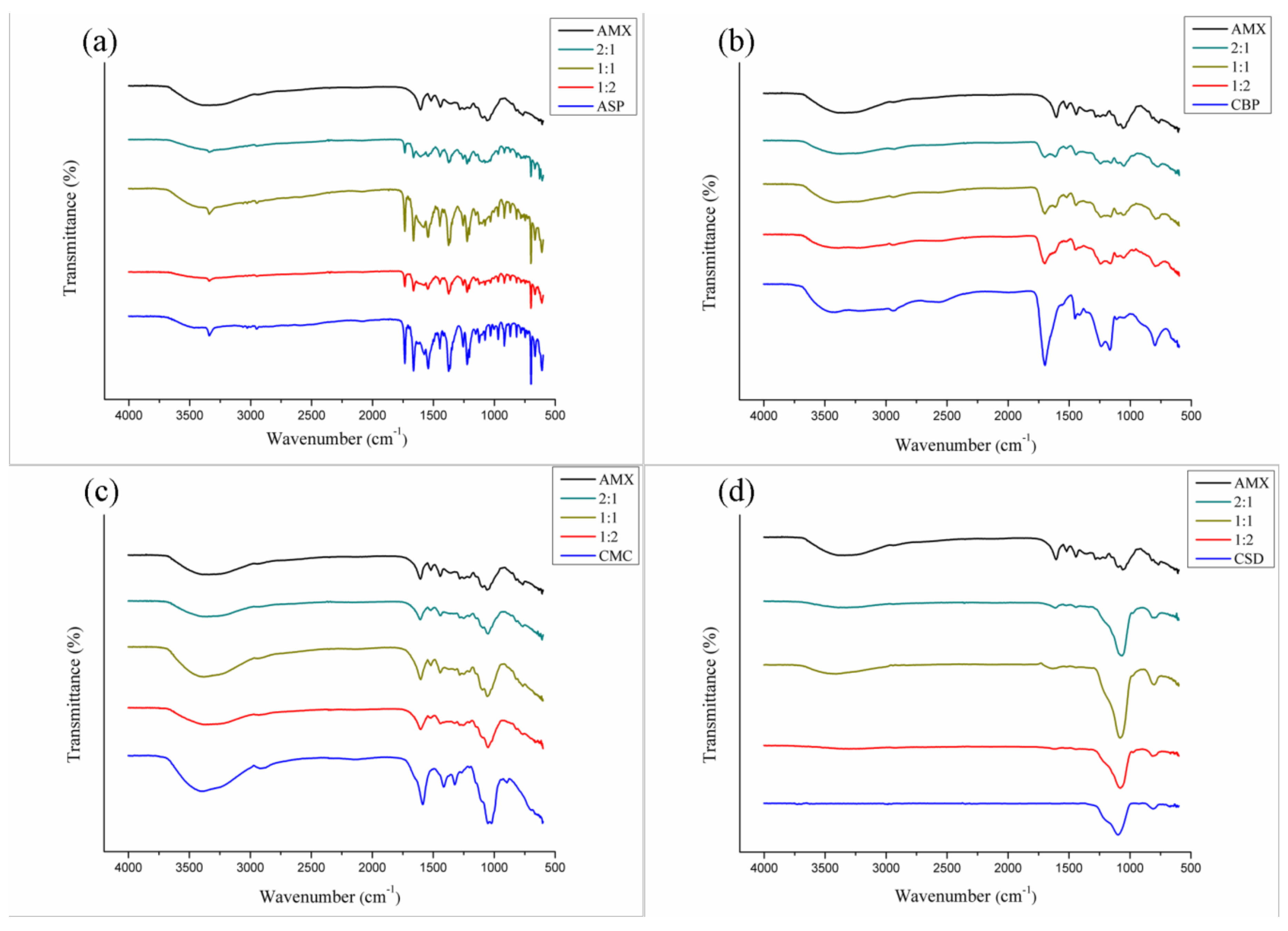
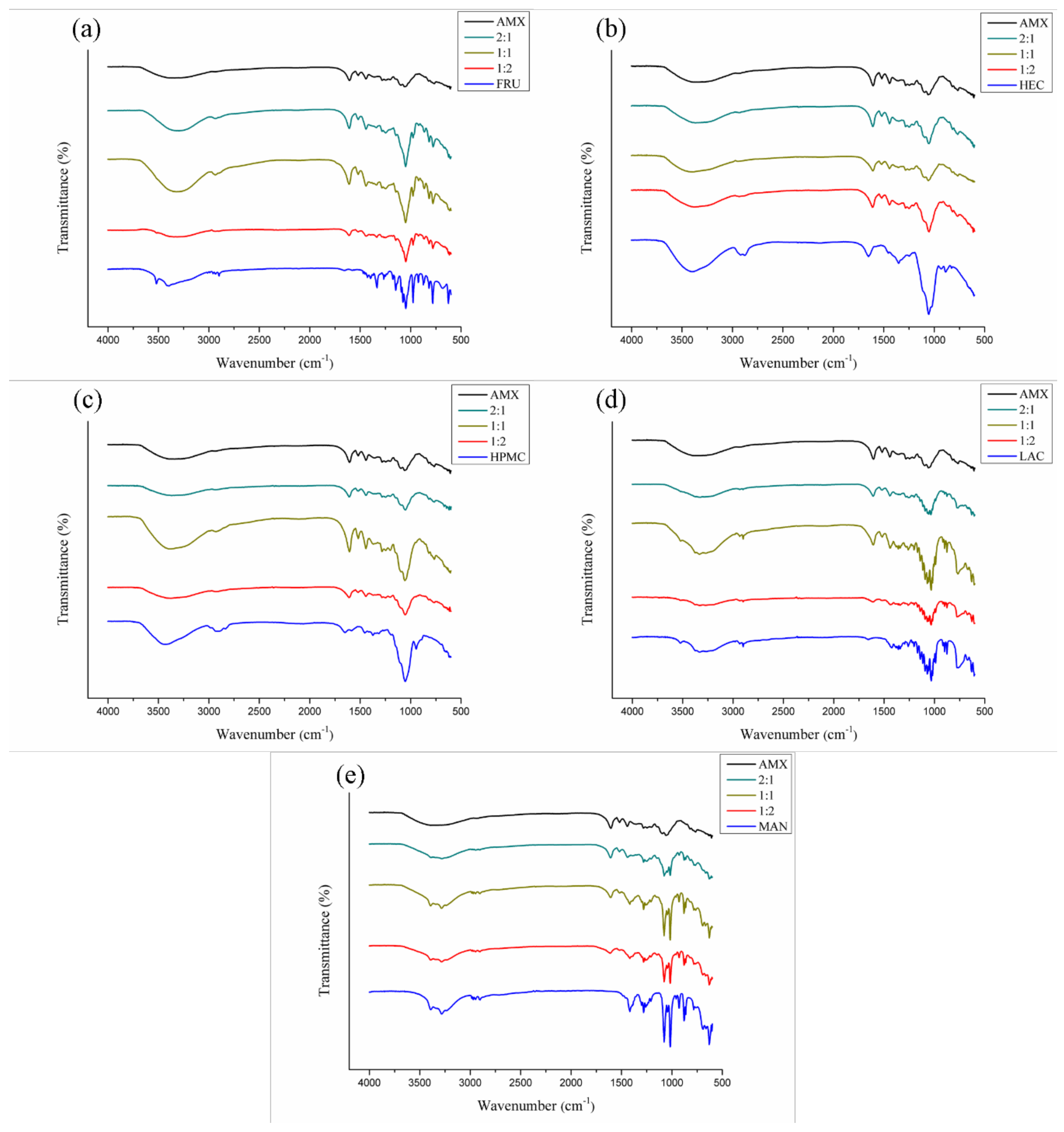
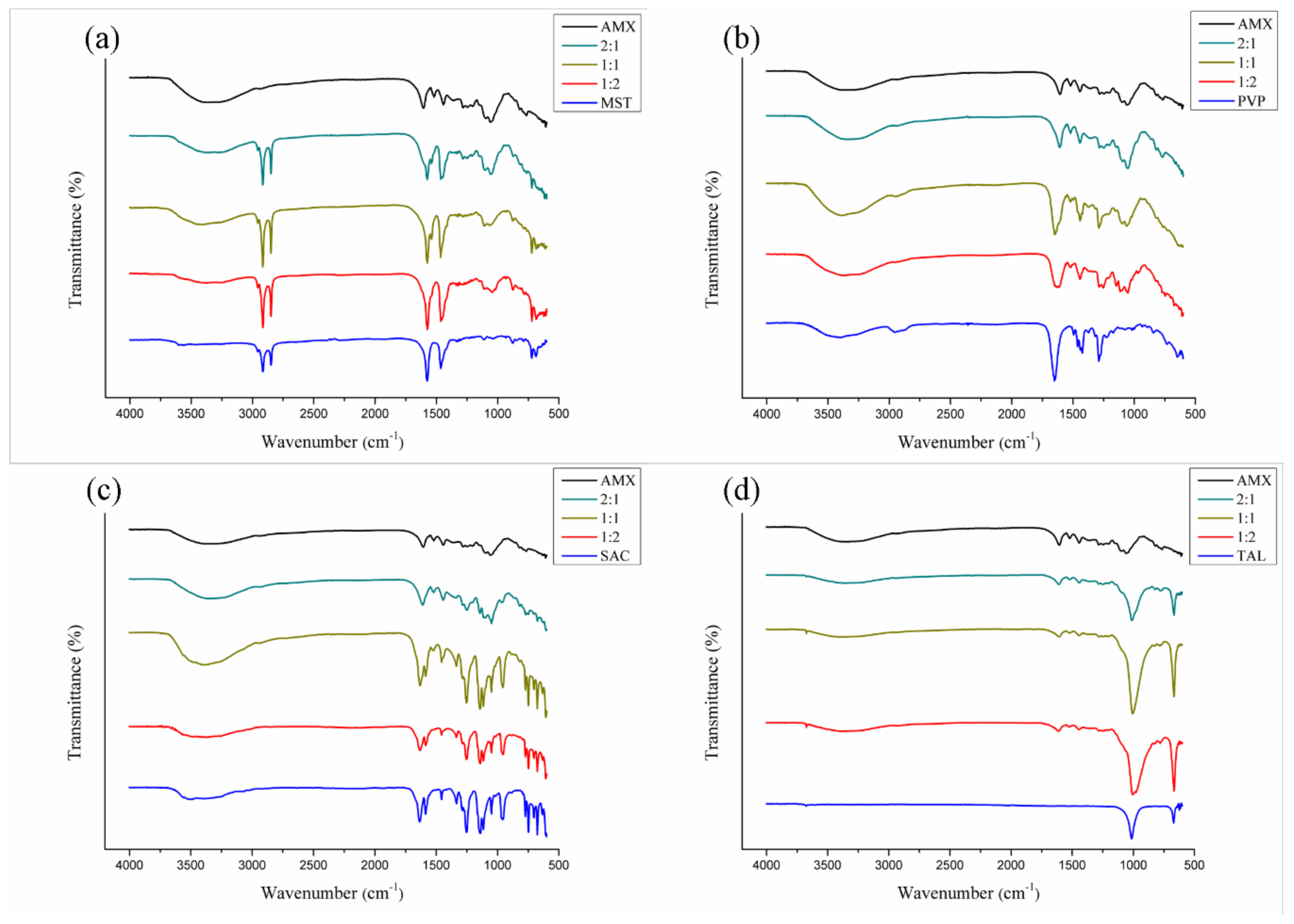
3.3.2. FTIR
3.4. Mucoadhesive Tablet Development
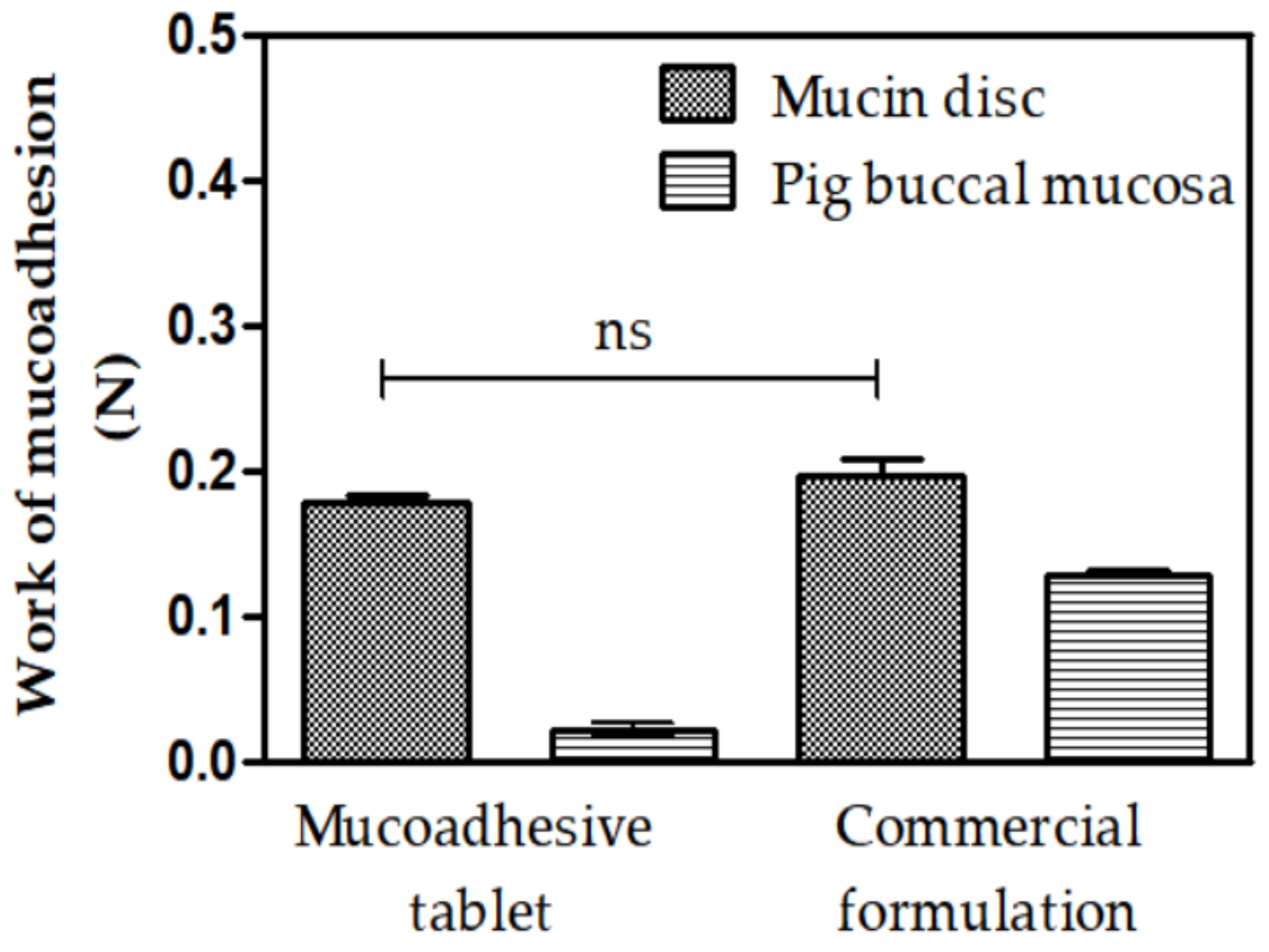
3.5. Mucoadhesion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Excipient/Abbreviation | Functional Category |
|---|---|
| Aspartame (ASP) | Sweetening agent |
| Sodium saccharin (SAC) | Sweetening agent |
| Fructose (FRU) | Sweetening agent |
| Carbopol (CBP) | Mucoadhesive agent |
| Sodium carboxymethylcellulose (CMC) | Mucoadhesive agent |
| Hydroxyethylcellulose (HEC) | Mucoadhesive agent |
| Hydroxypropylmethylcellulose (HPMC) | Mucoadhesive agent |
| Colloidal silicon dioxide (CSD) | Disintegrant |
| Polyvinylpyrrolidone K-30 (PVP)* | Disintegrant |
| Magnesium stearate (MST) | Lubricant |
| Talc (TAL) | Lubricant |
| Mannitol (MAN) | Sweetening agent/diluent |
| Lactose monohydrate (LAC) | Diluent |
| Sample | Event | Start–End (°C) | Peak (°C) | ΔH (J·g−1) |
|---|---|---|---|---|
| AMX | First | 66.41–126.28 | 98.67 | −283.69 |
| ASP | First | 59.72–91.16 | 80.23 | −35.43 |
| Second | 178.70–196.78 | 188.87 | −229.97 | |
| Third | 243.79–255.77 | 249.17 | −183.82 | |
| Fourth | 303.23–347.95 | 325.47 | −281.75 | |
| AMX + ASP (1:1) | First | 55.18–121.32 | 89.03 | −148.52 |
| Second | 170.02–193.65 | 183.90 | −123.74 | |
| Third | 222.86–244.21 | 235.55 | −64.54 | |
| Fourth | 309.01–337.42 | 322.89 | −14.97 | |
| AMX + ASP (1:2) | First | 53.71–118.83 | 98.93 | −138.94 |
| Second | 175.18–193.76 | 184.97 | −164.40 | |
| Third | 226.99–244.48 | 237.58 | −87.29 | |
| Fourth | 283.28–304.42 | 283.53 | −6.79 | |
| Fifth | 308.47–332.22 | 329.86 | −13.74 | |
| AMX + ASP (2:1) | First | 50.39–130.82 | 98.62 | −193.24 |
| Second | 169.74–193.95 | 184.25 | −85.90 | |
| Third | 217.74–241.31 | 234.78 | −25.09 | |
| CBP | First | 56.25–113.69 | 86.63 | −185.54 |
| Second | 181.79–246.38 | 213.91 | −182.44 | |
| Third | 255.62–293.73 | 272.20 | −95.02 | |
| Fourth | 351.44–368.78 | 362.72 | 14.49 | |
| AMX + CBP (1:1) | First | 83.02–122.89 | 104.39 | −70.91 |
| Second | 303.89–383.38 | 336.16 | −100.08 | |
| AMX + CBP (1:2) | First | 56.25–113.69 | 86.63 | −185.54 |
| Second | 181.79–246.38 | 213.91 | −182.44 | |
| Third | 255.62–293.73 | 272.20 | −95.02 | |
| Fourth | 351.44–368.78 | 362.72 | 14.49 | |
| AMX + CBP (2:1) | First | 58.93–134.65 | 106.95 | −195.85 |
| CMC | First | 71.60–126.66 | 97.97 | −397.78 |
| Second | 286.00–301.70 | 290.81 | 133.76 | |
| Third | 312.77–319.61 | 317.74 | 7.90 | |
| AMX + CMC (1:1) | First | 83.19–128.16 | 100.75 | −177.71 |
| Second | 277.55–318.74 | 295.42 | 198.44 | |
| AMX + CMC (1:2) | First | 75.51–119.36 | 92.61 | −152.56 |
| Second | 275.67–316.37 | 295.63 | 179.56 | |
| AMX + CMC (2:1) | First | 52.53–128.94 | 91.95 | −239.41 |
| Second | 278.07–320.79 | 297.34 | 114.52 | |
| CSD | First | 27.62–48.97 | 35.59 | 21.10 |
| AMX + CSD (1:1) | First | 80.02–107.25 | 105.60 | −9.19 |
| AMX + CSD (1:2) | First | 55.12–124.84 | 97.85 | −129.91 |
| AMX + CSD (2:1) | First | 54.21–134.64 | 100.47 | −181.98 |
| FRU | First | 87.79–121.41 | 107.92 | −47.51 |
| Second | 190.63–210.33 | 210.13 | 28.19 | |
| Third | 254.48–307.24 | 279.24 | 309.91 | |
| AMX + FRU (1:1) | First | 118.69–134.69 | 126.09 | −76.91 |
| Second | 191.94–203.81 | 195.05 | 6.15 | |
| Third | 220.61–235.04 | 222.28 | 13.5 | |
| AMX + FRU (1:2) | First | 90.91–108.74 | 101.92 | −12.26 |
| Second | 141.26–164.2 | 149.76 | 30.95 | |
| Third | 218.17–248.33 | 237.24 | 136.48 | |
| AMX + FRU (2:1) | First | 115.32–138.45 | 121.37 | −294.63 |
| HEC | First | 59.73–101.83 | 77.73 | −113.76 |
| Second | 325.83–355.15 | 343.28 | 237.51 | |
| Third | 359.21–368.49 | 364.60 | 10.15 | |
| AMX + HEC (1:1) | First | 56.25–128.83 | 98.58 | −313.54 |
| Second | 320.82–353.96 | 344.65 | 26.04 | |
| AMX + HEC (1:2) | First | 59.67–134.47 | 104.51 | −345.83 |
| Second | 322.98–369.57 | 346.98 | 181.95 | |
| AMX + HEC (2:1) | First | 45.45–119.58 | 91.22 | −281.17 |
| Second | 376.04–396.15 | 383.22 | 17.62 | |
| HPMC | First | 61.68–112.83 | 91.18 | −138.66 |
| Second | 337.66–373.63 | 356.09 | −89.52 | |
| AMX + HPMC (1:1) | First | 64.97–117.03 | 93.44 | −11.91 |
| Second | 285.24–334.35 | 316.21 | −46.32 | |
| AMX + HPMC (1:2) | First | 61.99–112.82 | 89.43 | −103.01 |
| Second | 336.40–349.92 | 341.99 | −14.46 | |
| Third | 382.45–428.34 | 374.75 | −12.19 | |
| AMX + HPMC (2:1) | First | 92.12–125.29 | 101.68 | −86.69 |
| Second | 338.29–368.38 | 344.15 | 9.27 | |
| LAC | First | 142.89–156.77 | 146.52 | −183.12 |
| Second | 214.47–226.32 | 219.34 | −226.34 | |
| Third | 237.71–246.58 | 241.72 | −3.62 | |
| Fourth | 284.67–323.80 | 304.85 | −137.36 | |
| AMX + LAC (1:1) | First | 84.56–110.61 | 99.90 | −2.19 |
| Second | 141.61–156.27 | 147.46 | −25.28 | |
| Third | 199.51–211.00 | 207.32 | −45.78 | |
| AMX + LAC (1:2) | First | 142.76–157.07 | 146.90 | −216.02 |
| Second | 204.29–216.80 | 211.31 | −463.07 | |
| AMX + LAC (2:1) | First | 82.80–126.49 | 97.99 | −15.28 |
| Second | 139.37–155.15 | 147.19 | −59.60 | |
| Third | 202.50–218.73 | 210.83 | −72.55 | |
| MAN | First | 165.99–177.62 | 169.89 | −450.53 |
| Second | 280.49–353.91 | 321.83 | −812.74 | |
| AMX + MAN (1:1) | First | 157.97–171.08 | 163.10 | −50.80 |
| AMX + MAN (1:2) | First | 157.32–175.31 | 165.63 | −503.47 |
| Second | 237.23–329.28 | 304.15 | −585.71 | |
| AMX + MAN (2:1) | First | 85.87–125.00 | 112.56 | −22.99 |
| Second | 154.30–171.26 | 162.94 | −130.24 | |
| Third | 283.03–340.83 | 325.73 | −47.41 | |
| MST | First | 45.31–54.35 | 50.91 | −7.99 |
| Second | 119.80–137.59 | 129.00 | −255.25 | |
| Third | 154.00–159.72 | 156.07 | −4.08 | |
| Fourth | 276.41–343.01 | 319.75 | −379.49 | |
| AMX + MST (1:1) | First | 123.76–138.22 | 127.43 | −71.44 |
| AMX + MST (1:2) | First | 112.93–134.13 | 123.68 | −126.77 |
| Second | 341.03–401.02 | 366.74 | −204.32 | |
| AMX + MST (2:1) | First | 120.27–135.90 | 126.36 | −51.45 |
| PVP K-30 | First | 53.58–125.06 | 97.61 | −515.18 |
| AMX + PVP (1:1) | First | 54.43–116.18 | 87.01 | −147.70 |
| AMX + PVP (1:2) | First | 89.84–141.02 | 116.75 | −202.44 |
| AMX + PVP (2:1) | First | 51.22–126.61 | 92.02 | −273.58 |
| SAC | First | 42.16–67.71 | 60.76 | −139.27 |
| Second | 114.9–136.39 | 120.25 | −281.00 | |
| Third | 126.89–131.67 | 129.85 | −5.52 | |
| Fourth | 361.66–370.92 | 365.15 | −210.17 | |
| AMX + SAC (1:1) | First | 42.16–67.71 | 60.76 | −139.27 |
| Second | 114.9–136.39 | 120.25 | −281.00 | |
| Third | 361.66–370.92 | 365.15 | −26.91 | |
| AMX + SAC (1:2) | First | 64.6–74.86 | 70.58 | −10.73 |
| Second | 115.34–135.1 | 126.04 | −40.79 | |
| Third | 140.88–152.57 | 145.45 | −55.66 | |
| Fourth | 349.31–364.54 | 357.99 | −65.39 | |
| AMX + SAC (2:1) | First | 66.61–83.47 | 73.97 | −29.27 |
| Second | 128.34–147.75 | 137.68 | −26.45 | |
| Third | 348.6–365.37 | 356.49 | −18.02 | |
| TAL | First | 38.13–51.22 | 44.70 | −11.76 |
| AMX + TAL (1:1) | First | 74.88–111.55 | 99.76 | −27.33 |
| AMX + TAL (1:2) | - | - | - | - |
| AMX + TAL (2:1) | First | 65.31–129.85 | 106.16 | −113.68 |
References
- Kassem, A.A.; Mohsen, A.M.; Ahmed, R.S.; Essam, T.M. Self-nanoemulsifying drug delivery system (SNEDDS) with enhanced solubilization of nystatin for treatment of oral candidiasis: Design, optimization, in vitro and in vivo evaluation. J. Mol. Liq. 2016, 218, 219–232. [Google Scholar] [CrossRef]
- Muadcheingka, T.; Tantivitayakul, P. Distribution of Candida albicans and non-albicans Candida species in oral candidiasis patients: Correlation between cell surface hydrophobicity and biofilm forming activities. Arch. Oral Biol. 2015, 60, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Pilmis, B.; Jullien, V.; Sobel, J.; Lecuit, M.; Lortholary, O.; Charlier, C. Antifungal drugs during pregnancy: An updated review. J. Antimicrob. Chemother. 2015, 70, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Lins, R.; Vasconcelos, F.H.P.; Leite, R.B.; Coelho-Soares, R.S.; Barbosa, D.N. Avaliação clínica de bochechos com extratos de aroeira (Schinus terebinthifolius) e camomila (Matricaria recutita L.) sobre a placa bacteriana e a gengivite. Rev. Bras. Plant. Med. 2013, 15, 112–120. [Google Scholar] [CrossRef]
- Chanda, W.; Joseph, T.P.; Wang, W.; Padhiar, A.A.; Zhong, M. The potential management of oral candidiasis using anti-biofilm therapies. Med. Hypotheses 2017, 106, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Pantoja, P.S.; Assreuy, A.M.S.; Silva, R.O.; Damasceno, S.R.B.; Mendonça, V.A.; Mendes, T.S.; Morais, J.A.V.; Almeida, S.L.; Teixeira, A.E.E.A.; Souza, M.H.L.P.; et al. The polysaccharide-rich tea of Ximenia americana barks prevents indomethacin-induced gastrointestinal damage via neutrophil inhibition. J. Ethnopharmacol. 2018, 224, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Sobeh, M.; Mahmoud, M.F.; Abdelfattah, M.A.O.; El-Beshbishy, H.A.; El-Shazly, A.M.; Wink, M. Hepatoprotective and hypoglycemic effects of a tannin rich extract from Ximenia americana var. caffra root. Phytomedicine 2017, 33, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.L.B.; Freitas, W.E.S.; de Morais, P.L.D.; Sarmento, J.D.A.; Alves, R.E. Bioactive compounds and antioxidant potential fruit of Ximenia americana L. Food chem. 2016, 192, 1078–1082. [Google Scholar]
- Uchôa, V.T.; Sousa, C.M.M.; Carvalho, A.A.; Sant’Ana, A.E.G.; Chaves, M.H. Free radical scavenging ability of Ximenia americana L. stem bark and leaf extracts. J. Appl. Pharm. Sci. 2016, 6, 091–096. [Google Scholar]
- Soro, T.Y.; Zahoui, O.S.; Néné-bi, A.S.; Traoré, F. Antipyretic activity of the fractions of the aqueous extract of Ximenia americana (Linnaeus)(Olacaceae). Int. J. Pharmacol. Toxicol. 2015, 5, 104–108. [Google Scholar]
- Ribeiro, D.A.; Macêdo, D.G.; Oliveira, L.G.S.; Saraiva, M.E.; Oliveira, S.F.; Souza, M.M.A.; Menezes, I.R.A. Potencial terapêutico e uso de plantas medicinais em uma área de Caatinga no estado do Ceará, nordeste do Brasil. Rev. Bras. Plant. Med. 2014, 16, 912–930. [Google Scholar] [CrossRef]
- Siddaiah, M.; Veera, K.J.; Rao, P.M.; Reddy, K.Y.; Chetty, C.M. Screening of Ximenia americana L. for it’s anti-iflammatory activity. J. Res. Educ. Indian Med. 2012, 18, 51–54. [Google Scholar]
- Okhale, S.E.; Nnachor, A.C.; Bassey, U.E. Evaluation of HPLC-DAD and antiproliferative characteristics of the leaf infusion of Ximenia americana Linn. MicroMedicine 2017, 5, 45–52. [Google Scholar]
- Santana, C.P.; Medeiros, F.D.; Correia, L.P.; Diniz, P.H.G.; Véras, G.; Medeiros, A.C.D. Dissolution and uniformity of contente of tablets developed with extract of Ximenina americana L. PLoS ONE 2018, 13. [Google Scholar] [CrossRef]
- Kabran, G.R.M.; Mamyrbekova-Bekro, J.A.; Pirat, J.L.; Lecouvey, M.; Sainte-Cathérine, O.; Sommerer, N.; Verbaere, A.; Meudec, E.; Békro, Y.A. UPLC-MS quantification and anticancer potential of Ximenia americana hydro-acetonic crude extract leaves. Der Chem. Sin. 2017, 8, 70–74. [Google Scholar]
- Santana, C.P.; Fernandes, F.H.; Brandão, D.O.; Silva, P.C.D.; Correia, L.P.; Nóbrega, F.P.; medeiros, F.D.; Diniz, P.H.G.D.; Véras, G.; Medeiros, A.C.D. Compatibility study of dry extract of Ximenia americana L. and pharmaceutical excipients used in solid state. J. Therm. Anal. Calorim. 2018, 133, 603–617. [Google Scholar] [CrossRef]
- Sheetar, A.K.; Kotresha, K.; Kaliwal, B.B.; Vedamurthy, A.B. Evaluation of in vitro antioxidant and anti-inflammatory activities of Ximenia americana extracts. Asian Pac. J. Trop. Dis. 2015, 5, 918–923. [Google Scholar] [CrossRef]
- Sheetar, A.K.; Sateesh, M.K.; Kaliwal, B.B.; Vedamurthy, A.B. In vitro antidiabetic activities and GC-MS phytochemical analysis of Ximenia americana extracts. S. Afr. J. Bot. 2017, 111, 202–211. [Google Scholar]
- Sá, L.L.F.; Nogueira, N.C.; da Silva Filho, E.C.; Figueiras, A.; Veiga, F.; Nunes, L.C.C.; Soares-Sobrinho, J.L. Design of buccal mucoadhesive tablets: Understanding and development. J. Appl. Pharm. Sci. 2018, 8, 150–163. [Google Scholar]
- Fitaihi, R.A.; Aleanizy, F.S.; Elsamaligy, S.; Mahmoud, H.A.; Bayomi, M.A. Role of chitosan on controlling the characteristics and antifungal activity of bioadhesive fluconazole vaginal tablets. Saudi Pharm. J. 2018, 26, 151–161. [Google Scholar]
- Reddy, P.D.; Balanjineyulu, R.; Swarnalatha, D.; Badarinath, A.V.; Gopinath, C. Design, development and in vitro characterization of felodipine mucoadhesive buccal tablets. J. Pharm. Res. 2015, 9, 170–176. [Google Scholar]
- Khutoryanskiy, V.V. Advances in mucoadhesion and mucoadhesive polymers. Macromol. Biosci. 2011, 11, 748–764. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, A.; Piras, A.M.; Uccello-Barretta, G.; Balzano, F.; Cesari, A.; Testai, L.; Citi, V.; Zambito, Y. Impact of mucoadhesive polymeric nanoparticulate systems on oral bioavailability of a macromolecular model drug. Eur. J. Pharm. Biopharm. 2018, 130, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Sahatsapan, N.; Rojanarata, T.; Ngawhirunpat, T.; Opanasopit, P.; Tonglairoum, P. 6-Maleimidohexanoic acid-grafted chitosan: A new generation mucoadhesive polymer. Carbohydr. Polym. 2018, 202, 258–264. [Google Scholar] [PubMed]
- Wasnik, M.N.; Godse, R.D.; Nair, H.A. Development and evaluation of buccoadhesive tablet for selegiline hydrochloride based on thiolated polycarbophil. Drug Dev. Ind. Pharm. 2014, 40, 632–638. [Google Scholar] [PubMed]
- Da Costa, R.S.; Negrão, C.A.B.; Camelo, S.R.P.; Ribeiro-Costa, R.M.; Barbosa, W.L.R.; da Costa, C.E.F.; Júnior, J.O.C.S. Investigation of thermal behavior of Heliotropium indicum L. lyophilized extract by TG and DSC. J. Therm. Anal. Calorim. 2013, 111, 1959–1964. [Google Scholar] [CrossRef]
- Liltorp, K.; Larsen, T.G.; Willumsen, B.; Holm, R. Solid state compatibility studies with tablet excipients using non thermal methods. J. Pharm. Biomed. Anal. 2011, 55, 424–428. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts: Approved Standard, 2nd ed.; Clinical and Laboratory Standards Institute (CLSI): Wayne, NJ, USA, 2002; Volume 22, 30p. [Google Scholar]
- Rowe, R.C.; Sheskey, P.J.; Quinn, M.C. Handbook of Pharmaceutical Excipients, 6th ed.; Pharmaceutical Press: Washington, DC, USA, 2009. [Google Scholar]
- Gupta, R.; Sharma, P.; Garg, A.; Soni, A.; Sahu, A.; Rai, S.; Rai, S.; Shukla, A. Formulation and evaluation of herbal effervescent granules incorporated with Calliandra haematocephala leaves extract. Indo Am. J. Pharm. Res. 2013, 3, 4366–4371. [Google Scholar]
- Lago, V.V.; Pereira, R.N.; Bertol, C.D. Propriedades micromeríticas e análise físico-química de matérias-primas de alopurinol. Rev. Ciênc. Farm. Básica Apl. 2012, 33, 385–393. [Google Scholar]
- Bernatoniete, J.; Petkeviciute, Z.; Kalveniene, Z.; Masteikova, R.; Draksiene, G.; Muselik, J.; Bernatoniene, R.; Lazauskas, R.; Savickas, A. The investigation of phenolic compounds and technological properties of Leonurus, Crataegus and Ginkgo extracts. J. Med. Plants Res. 2010, 4, 925–931. [Google Scholar]
- Oshiro Junior, J.A.; Carvalho, F.C.; Soares, C.P.; Chorilli, M.; Chiavacci, L.A. Development of Cutaneous Bioadhesive Ureasil-Polyether Hybrid Films. Int. J. Polym. Sci. 2015. [Google Scholar] [CrossRef]
- Oshiro Junior, J.A.; Nasser, N.J.; Chiari-Andréo, B.G.; Cuberes, T.; Chiavacci, L.A. Study of triamcinolone release and mucoadhesive properties of macroporous hybrid films for oral disease treatment. Biomed. Phys. Eng. Express 2018, 4, 035009. [Google Scholar]
- Omer, M.E.F.A.; Elnima, E.I. Antimicrobial activity of Ximenia americana. Fitoterapia 2003, 74, 122–126. [Google Scholar]
- Maikai, V.A. Antimicrobial properties of stem bark extracts of Ximenia americana. J. Agric. Sci. 2009, 1, 30–34. [Google Scholar]
- Runyoro, D.K.B.; Ngassapa, O.D.; Matee, M.I.N.; Joseph, C.C.; Moshi, M.J. Medicinal plants used by Tanzanian traditional healers in the management of Candida infections. J. Ethnopharmacol. 2006, 106, 158–165. [Google Scholar] [CrossRef]
- Traoré, M.S.; Baldé, M.A.; Camara, A.; Baldé, E.S.; Diané, S.; Diallo, M.S.T.; Keita, A.; Cos, P.; Maes, L.; Pieters, L.; et al. The malaria co-infection challenge: An investigation into the antimicrobial activity of selected Guinean medicinal plants. J. Ethnopharmacol. 2015, 174, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Maroyi, A. Ximenia caffra Sond. (Ximeniaceae) in sub-Saharan Africa: A synthesis and review of its medicinal potential. J. Ethnopharmacol. 2016, 184, 81–100. [Google Scholar] [PubMed]
- Van Vuuren, S.; Holl, D. Antimicrobial natural product research: A review from a South African perspective for the years 2009–2016. J. Ethnopharmacol. 2017, 208, 236–252. [Google Scholar]
- Lagrouh, F.; Dakka, N.; Bakri, Y. The antifungal activity of Moroccan plants and the mechanism of action of secondary metabolites from plants. J. Mycol. Med. 2017, 27, 303–311. [Google Scholar]
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Umair, M.; Zafar, M.H.; Bahadar, K. Role of secondary metabolites in plant defense against pathogens. Microb. Pathog. 2018, 124, 198–202. [Google Scholar] [CrossRef]
- Chadha, R.; Bhandari, S. Drug-excipient compatibility screening: Role of thermoanalytical and spectroscopic techniques. J. Pharm. Biomed. Anal. 2014, 87, 82–97. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, H.A.; Santana, C.S.; Silva, P.C.D.; Simões, M.O.S.; Kaneko, T.M.; Medeiros, A.C.D. Development of a sunscreen by thermal compatibility study using Schinopsis brasiliensis Engler extract as preservative. J. Therm. Anal. Calorim. 2018, 131, 753–763. [Google Scholar] [CrossRef]
- Neil, M.J. (Ed.) The Merck Index, 15th ed.; Merck & Co., Inc.: Whitehouse Station, NJ, USA, 2013. [Google Scholar]
- Tita, B.; Fulias, A.; Bangur, G.; Marian, E.; Tita, D. Compatibility study between ketoprofen and pharmaceutical excipients used in solid dosage forms. J. Pharm. Biomed. Anal. 2011, 56, 221–227. [Google Scholar] [PubMed]
- Lawrence, J.F. Saccharin. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: New York, NY, USA, 2003; pp. 5033–5035. [Google Scholar]
- Ledeti, I.; Bolintineanu, S.; VLASE, G.; Circioban, D.; Ledeti, A.; Vlase, T.; Suta, L.; Caunii, A.; Murariu, M. Compatibility study between antiparkinsonian drug levodopa and excipients by FTIR spectroscopy, X-ray diffraction and thermal analysis. J. Therm. Anal. Calorim. 2017, 130, 433–441. [Google Scholar] [CrossRef]
- Chaves, L.L.; Rolim, L.A.; Gonçalves, M.L.C.M.; Vieira, A.C.C.; Alves, L.D.S.; Soares, M.F.R.; Soares-Sobrinho, J.L.; Lima, M.C.A.; Rolim-Neto, P.J. Study of stability and drug-excipient compatibility of diethylcarbamazine citrate. J. Therm. Anal. Calorim. 2013, 111, 2179–2186. [Google Scholar]
- Pinto, M.F.; Moura, E.A.; Souza, F.S.; Macêdo, R.O. Thermal compatibility studies of nitroimidazoles and excipients. J. Therm. Anal. Calorim. 2010, 102, 323–329. [Google Scholar]
- Bento da Silva, P.; Calixto, G.M.F.; Oshiro Júnior, J.A.; Bombardelli, R.L.Á.; Fonseca-Santos, B.; Rodero, C.F.; Chorilli, M. Structural features and the anti-inflammatory effect of green tea extract-loaded liquid crystalline systems intended for skin delivery. Polymers 2017, 9, 30. [Google Scholar] [CrossRef]
- Andrews, G.P.; Laverty, T.P.; Jones, D.S. Mucoadhesive polymeric platforms for controlled drug delivery. Eur. J. Pharm. Biopharm. 2009, 71, 505–518. [Google Scholar]
- Bruschi, M.L.; Jones, D.S.; Panzeri, H.; Gremião, M.P.; de Freitas, O.; Lara, E.H. Semisolid systems containing propolis for the treatment of periodontal disease: In vitro release kinetics, syringeability, rheological, textural, and mucoadhesive properties. J. Pharm. Sci. 2007, 96, 2074–2089. [Google Scholar] [CrossRef]
- Salmazi, R.; Calixto, G.; Bernegossi, J.; Ramos, M.A.S.; Bauab, T.M.; Chorilli, M. A curcumin-loaded liquid crystal precursor mucoadhesive system for the treatment of vaginal candidiasis. Int. J. Nanomed. 2015, 10, 4815. [Google Scholar]
- Perioli, L.; Ambrogi, V.; Giovagnoli, S.; Blasi, P.; Mancini, A.; Ricci, M.; Rossi, C. Influence of compression force on the behaviour of mucoadhesive buccal tablets. AAPS PharmSciTech 2008, 9, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Guilhotra, R.M.; Ikram, M.; Srivastava, S.; Gilhotra, N. A clinical perspective on mucoadhesive buccal drug delivery systems. J. Biomed. Res. 2014, 28, 81–97. [Google Scholar]
- Mansuri, S.; Kesharwani, P.; Jain, K.; Tekade, R.K.; Jain, N.K. Mucoadhesion: A promising approach in drug delivery system. React. Funct. Polym. 2016, 100, 151–172. [Google Scholar]
- Partenhauser, A.; Bernkop-Schnuerch, A. Mucoadhesive polymers in the treatment of dry X syndrome. Drug Discov. Today 2016, 21, 1051–1062. [Google Scholar] [PubMed]
- Chakraborti, C.K.; Sahoo, S.; Behera, P.K. Role of different biodegradable polymers on the permeability of ciprofloxacin. J. Adv. Pharm. Technol. Res. 2014, 5, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Bartkowiak, A.; Rojewska, M.; Hyla, K.; Zembrzuska, J.; Prochaska, K. Surface and swelling properties of mucoadhesive blends and their ability to release fluconazole in a mucin environment. Colloids Surf. B Biointerfaces 2018, 172, 586–593. [Google Scholar] [CrossRef]
- Novak, S.D.; Šporar, E.; Baumgartner, S.; Vrečer, F. Characterization of physicochemical properties of hydroxypropyl methylcellulose (HPMC) type 2208 and their influence on prolonged drug release from matrix tablets. J. Pharm. Biomed. Anal. 2012, 66, 136–143. [Google Scholar]
- Viridén, A.; Wittgren, B.; Larsson, A. Investigation of critical polymer properties for polymer release and swelling of HPMC matrix tablets. Eur. J. Pharm. Sci. 2009, 36, 297–309. [Google Scholar] [CrossRef] [PubMed]
- Pereirinha, P.R.P.; Figueiras, A.R.R. Desenvolvimento de sistemas mucoadesivo para a administração controlada de fármacos. Boletim Inform. Geum 2016, 7, 63–81. [Google Scholar]
- Cevher, E.; Açma, A.; Sinani, G.; Aksu, B.; Zloh, M.; Mülazımoğlu, L. Bioadhesive tablets containing cyclodextrin complex of itraconazole for the treatment of vaginal candidiasis. Int. J. Biol. Macromol. 2014, 69, 124–136. [Google Scholar] [CrossRef]
- Tonglairoum, P.; Ngawhirunpat, T.; Rojanarata, T.; Panomsuk, S.; Kaomongkolgit, R.; Opanasopit, P. Fabrication of mucoadhesive chitosan coated polyvinylpyrrolidone/ cyclodextrin/clotrimazole sandwich patches for oral candidiasis. Carbohydr. Polym. 2015, 132, 173–179. [Google Scholar] [PubMed]
- Yan, Z.; Liu, X.; Liu, Y.; Han, Y.; Lin, M.; Wang, W.; Guan, X.; Zhu, S.; Zhang, H.; Wang, Q.; et al. The efficacy and safety of miconazole nitrate mucoadhesive tablets versus itraconazole capsules in the treatment of oral candidiasis: An open-label, randomized, multicenter trial. PLoS ONE 2016, 11, e0167880. [Google Scholar] [CrossRef] [PubMed]
- Llabot, J.M.; Manzo, R.H.; Allemandi, D.A. Novel mucoadhesive extended release tablets for treatment of oral candidosis: “in vivo” evaluation of the biopharmaceutical performance. J. Pharm. Sci. 2009, 98, 1871–1876. [Google Scholar] [PubMed]
| MIC (µg·mL−1) | ||||
|---|---|---|---|---|
| EtOH (%) | Maceration | Turbolysis | Ultrasound | |
| X. americana | 50 | 250.00 | 125.00 | 125.00 |
| 70 | 125.00 | 125.00 | 250.00 | |
| 90 | 125.00 | 250.00 | 250.00 | |
| Excipient | Concentration (%) | Class |
|---|---|---|
| HPMC | 48.97 | Adhesive |
| MAN | 10.00 | Diluent/sweetener |
| MST | 2.00 | Lubricant |
| PVP | 2.00 | Desintegrator |
| AMX | 37.03 | PAPI |
| Parameters | Formulation | Granulate |
|---|---|---|
| Gross density (g·mL−1) | 0.526 | 0.550 |
| Density of compaction (g·mL−1) | 0.769 | 0.625 |
| Hausner factor | 1.460 | 1.13 |
| Carr index (%) | 31.60 | 12.0 |
| Index of densification (mL) | 2.00 | 1.0 |
| Angle of repose (°) | 33.00 | 0.15 |
| Flow time (s) | 4.57 | 3.12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almeida, L.; Oshiro Júnior, J.A.; Silva, M.; Nóbrega, F.; Andrade, J.; Santos, W.; Ribeiro, A.; Conceição, M.; Veras, G.; Medeiros, A.C. Tablet of Ximenia Americana L. Developed from Mucoadhesive Polymers for Future Use in Oral Treatment of Fungal Infections. Polymers 2019, 11, 379. https://doi.org/10.3390/polym11020379
Almeida L, Oshiro Júnior JA, Silva M, Nóbrega F, Andrade J, Santos W, Ribeiro A, Conceição M, Veras G, Medeiros AC. Tablet of Ximenia Americana L. Developed from Mucoadhesive Polymers for Future Use in Oral Treatment of Fungal Infections. Polymers. 2019; 11(2):379. https://doi.org/10.3390/polym11020379
Chicago/Turabian StyleAlmeida, Lucas, João Augusto Oshiro Júnior, Milena Silva, Fernanda Nóbrega, Jéssica Andrade, Widson Santos, Angélica Ribeiro, Marta Conceição, Germano Veras, and Ana Cláudia Medeiros. 2019. "Tablet of Ximenia Americana L. Developed from Mucoadhesive Polymers for Future Use in Oral Treatment of Fungal Infections" Polymers 11, no. 2: 379. https://doi.org/10.3390/polym11020379
APA StyleAlmeida, L., Oshiro Júnior, J. A., Silva, M., Nóbrega, F., Andrade, J., Santos, W., Ribeiro, A., Conceição, M., Veras, G., & Medeiros, A. C. (2019). Tablet of Ximenia Americana L. Developed from Mucoadhesive Polymers for Future Use in Oral Treatment of Fungal Infections. Polymers, 11(2), 379. https://doi.org/10.3390/polym11020379