A Novel Glucose Biosensor Based on Hierarchically Porous Block Copolymer Film
Abstract
1. Introduction
2. Materials and Methods
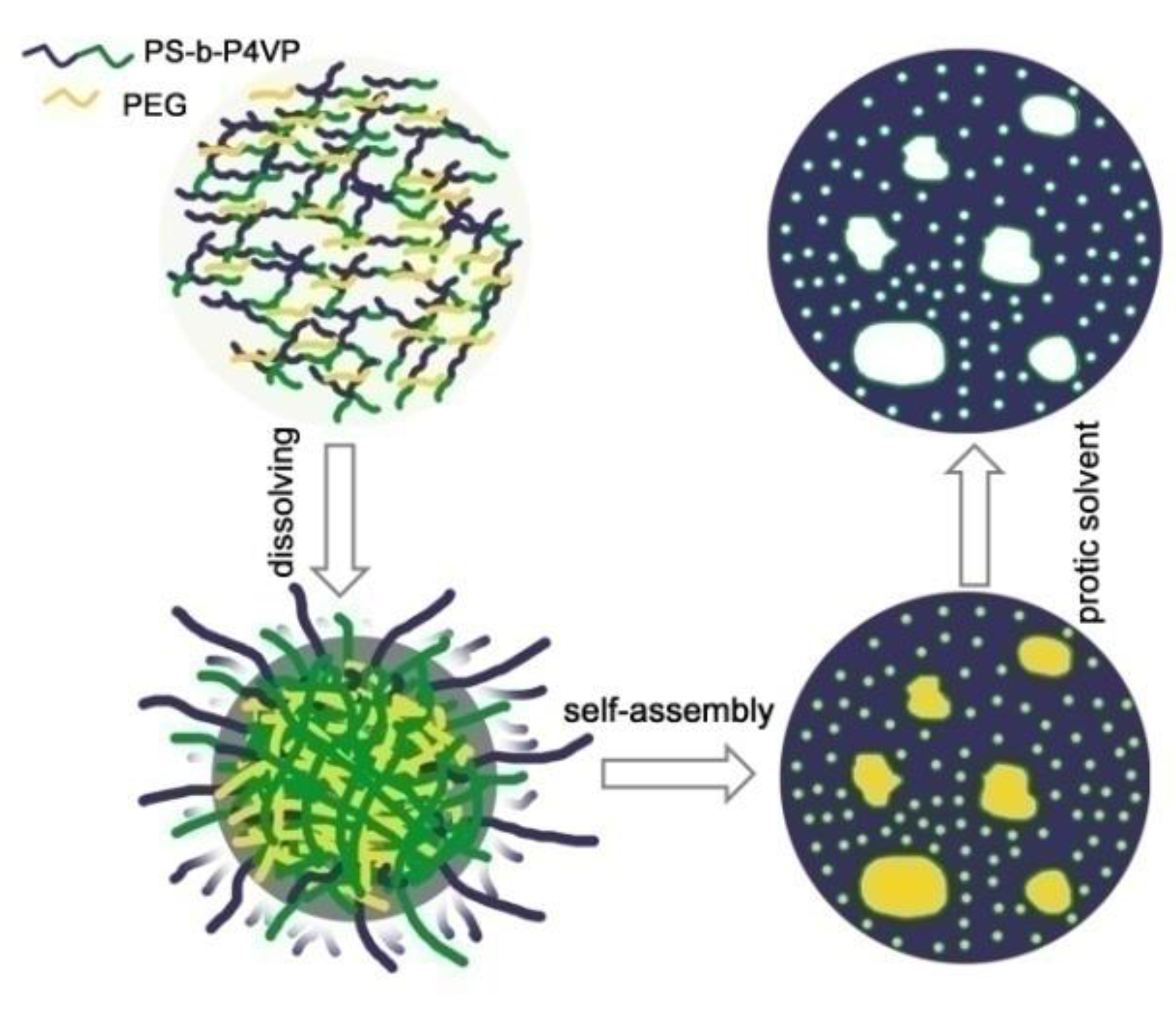
2.1. Preparation of Hierarchically Porous PS-b-P4VP Film
2.2. Construction of Glucose Biosensor
3. Results and Discussions
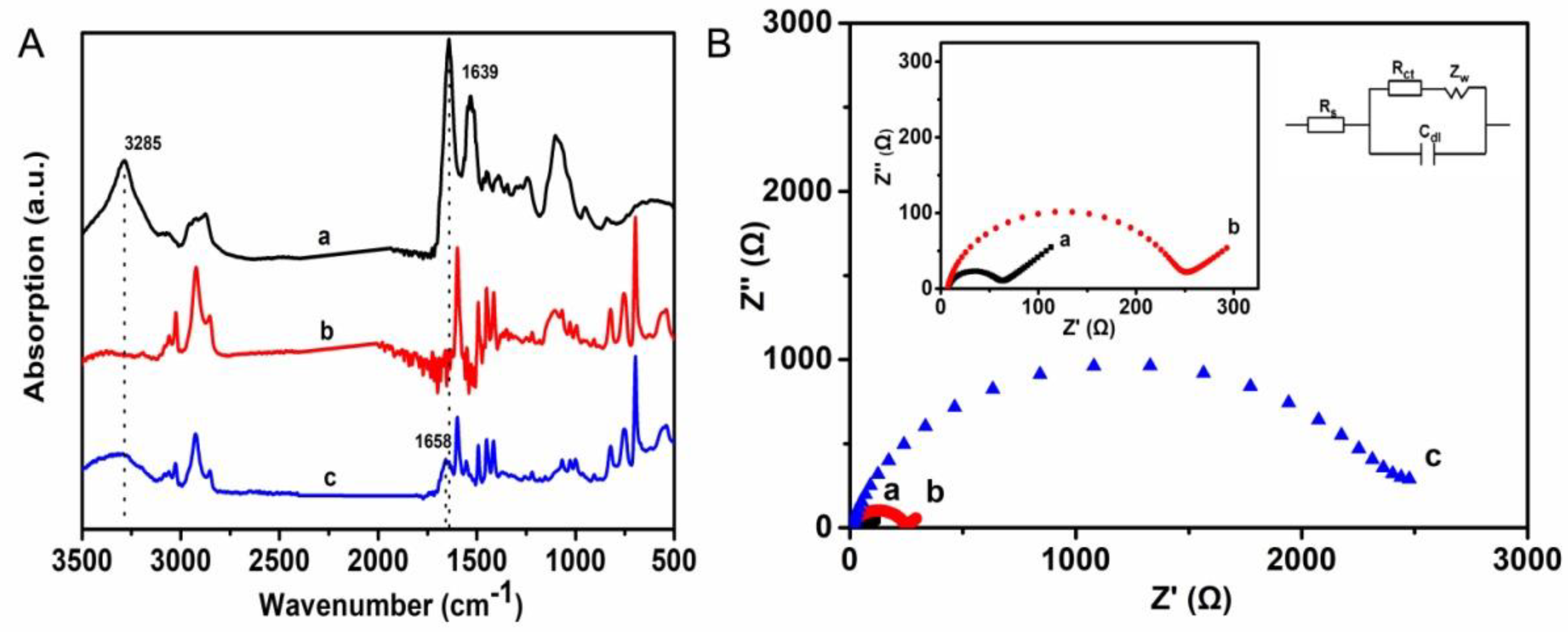
3.1. Characterizations of the Hierarchically Porous Film and Glucose Biosensor
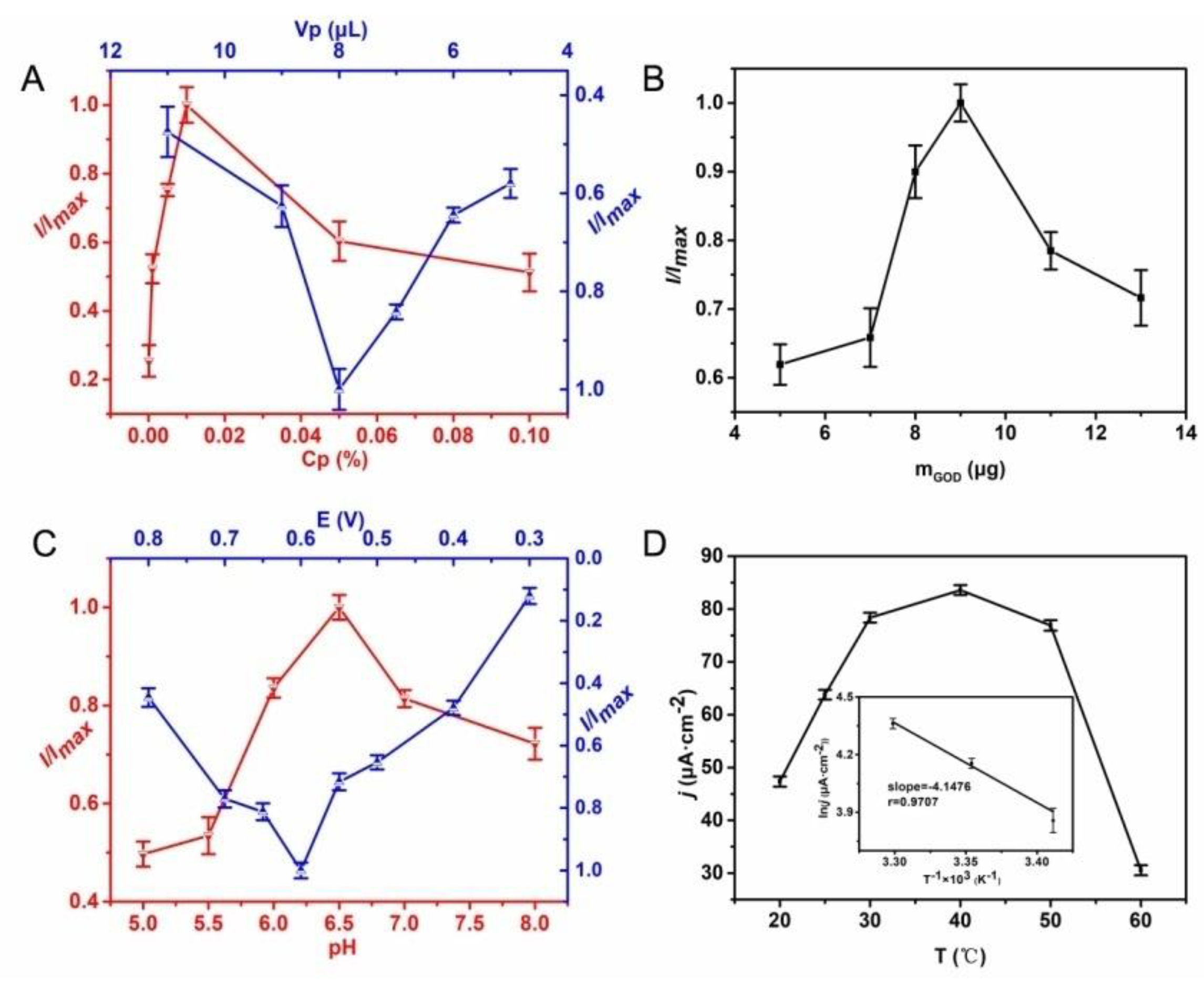
3.2. Optimization of the Glucose Biosensor
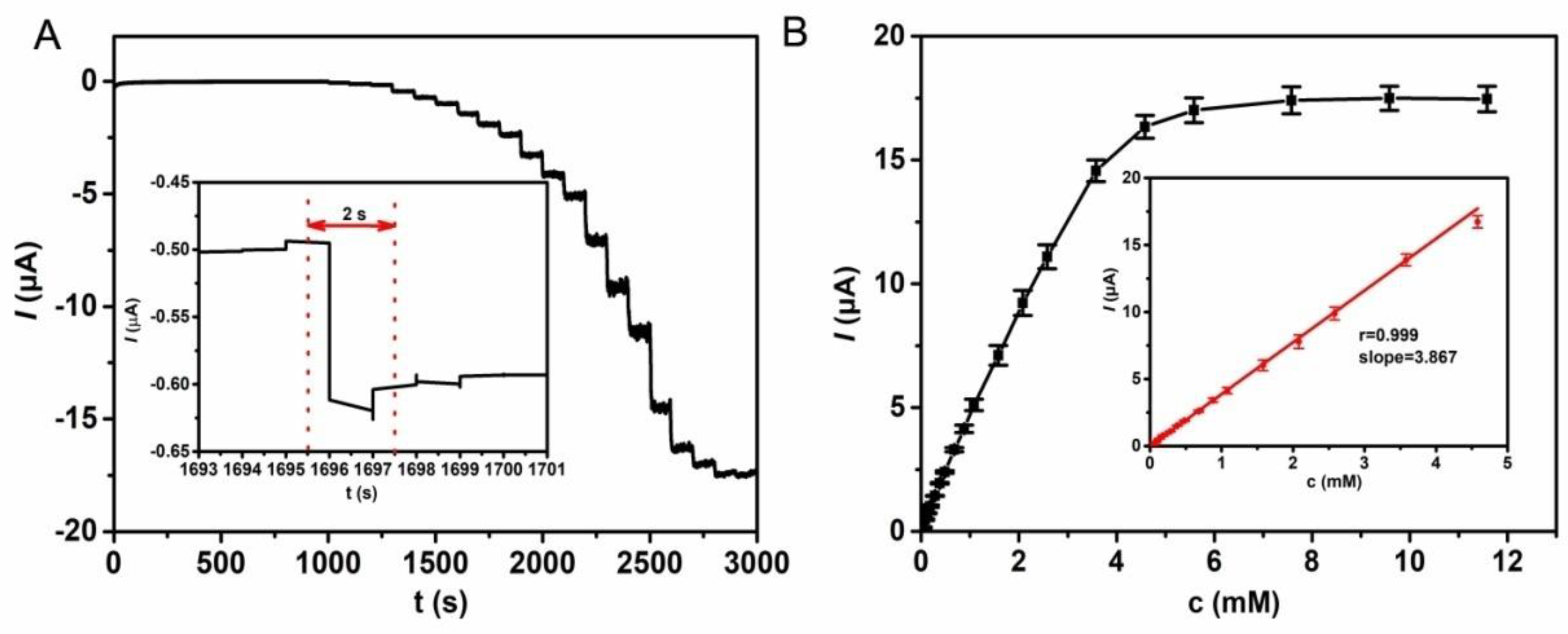
3.3. Performance of the Proposed Glucose Biosensor
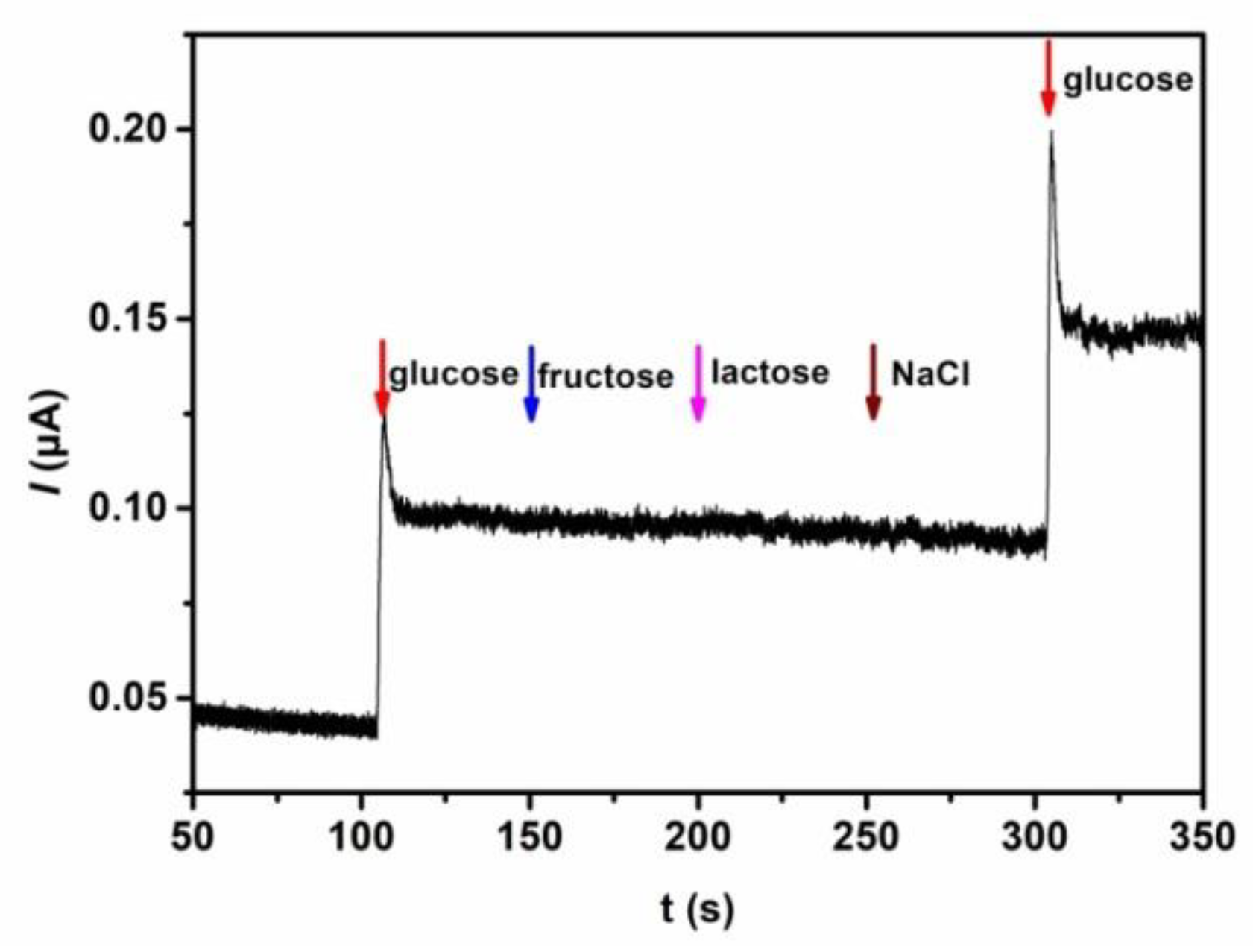
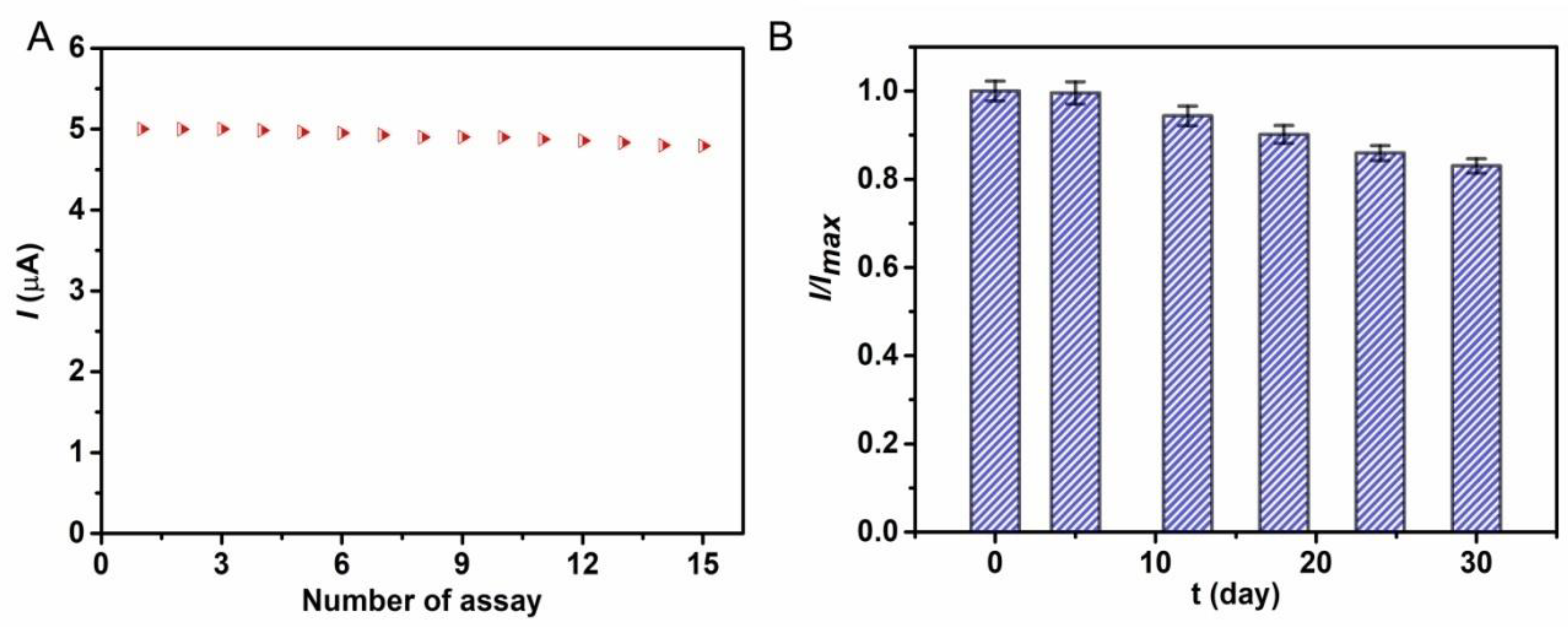
3.4. Interference Study and Stability of the Biosensor
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chen, K.J.; Lee, C.F.; Rick, J.; Wang, S.H.; Liu, C.C.; Hwang, B.J. Fabrication and application of amperometric glucose biosensor based on a novel PtPd bimetallic nanoparticle decorated multi-walled carbon nanotube catalyst. Biosens. Bioelectron. 2012, 33, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Cornell, B.A.; Braachmaksvytis, V.L.B.; King, L.G.; Osman, P.D.J.; Raguse, B.; Wieczorek, L.; Pace, R.J. A biosensor that uses ion-channel switches. Nature 1997, 387, 580. [Google Scholar] [CrossRef] [PubMed]
- Cosnier, S.; Holzinger, M. Electrosynthesized polymers for biosensing. Chem. Soc. Rev. 2011, 40, 2146–2156. [Google Scholar] [CrossRef] [PubMed]
- Negash, N.; Alemu, H.; Tessema, M. Determination of Phenol and Chlorophenols at Single-Wall Carbon Nanotubes/Poly(3,4-ethylenedioxythiophene) Modified Glassy Carbon Electrode Using Flow Injection Amperometry. Anal. Chem. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Song, Y.; Luo, Y.; Zhu, C.; Li, H.; Du, D.; Lin, Y. Recent advances in electrochemical biosensors based on graphene two-dimensional nanomaterials. Biosens. Bioelectron. 2016, 76, 195. [Google Scholar] [CrossRef] [PubMed]
- Valiūnienė, A.; Rekertaitė, A.I.; Ramanavičienė, A.; Mikoliūnaitė, L.; Ramanavičius, A. Fast Fourier transformation electrochemical impedance spectroscopy for the investigation of inactivation of glucose biosensor based on graphite electrode modified by Prussian blue, polypyrrole and glucose oxidase. Colloid Surf. A Physicochem. Eng. Asp. 2017, 5, 165–171. [Google Scholar] [CrossRef]
- Wang, Y.H.; Huang, K.J.; Wu, X. Recent advances in transition-metal dichalcogenides based electrochemical biosensors: A review. Biosens. Bioelectron. 2017, 97, 305. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Dong, S.; Huang, G.; Xie, Q.; Huang, T. MOF-derived Fe2O3 nanoparticle embedded in porous carbon as electrode materials for two enzyme-based biosensors. Sens. Actuators B 2018, 1, 189–197. [Google Scholar] [CrossRef]
- Márquez, A.; Jiménez-Jorquera, C.; Domínguez, C.; Muñoz-Berbel, X. Electrodepositable alginate membranes for enzymatic sensors: An amperometric glucose biosensor for whole blood analysis. Biosens. Bioelectron. 2017, 97, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Ren, Y.; Zhang, Y.; Li, J.; Li, H.; Hu, X.H. Nanoflake-like SnS2 matrix for glucose biosensing based on direct electrochemistry of glucose oxidase. Biosens. Bioelectron. 2011, 26, 4337–4341. [Google Scholar] [CrossRef] [PubMed]
- Shumyantseva, V.V.; Sigolaeva, L.V.; Agafonova, L.E.; Bulko, T.V.; Pergushov, D.V.; Schacher, F.H.; Archakov, A.I. Facilitated biosensing via direct electron transfer of myoglobin integrated into diblock copolymer/multi-walled carbon nanotube nanocomposites. J. Mater. Chem. B 2015, 3, 5467–5477. [Google Scholar] [CrossRef]
- Li, Z.; Sheng, L.; Meng, A.; Xie, C.; Zhao, K. A glassy carbon electrode modified with a composite consisting of reduced graphene oxide, zinc oxide and silver nanoparticles in a chitosan matrix for studying the direct electron transfer of glucose oxidase and for enzymatic sensing of glucose. Microchim. Acta 2016, 183, 1625–1632. [Google Scholar] [CrossRef]
- Li, H.; Hu, X.; Zhu, H.; Zang, Y.; Xue, H. Amperometric Phenol Biosensor Based on a New Immobilization Matrix: Polypyrrole Nanotubes Derived from Methyl Orange as Dopant. Int. J. Electrochem. Sci. 2017, 6714–6728. [Google Scholar] [CrossRef]
- Liu, X.; Yang, W.; Chen, L.; Jia, J. Three-Dimensional Copper Foam Supported CuO Nanowire Arrays: An Efficient Non-enzymatic Glucose Sensor. Electrochim. Acta 2017, 235, 519–526. [Google Scholar] [CrossRef]
- Lin, L.; Yan, J.; Li, J. Small-molecule triggered cascade enzymatic catalysis in hour-glass shaped nanochannel reactor for glucose monitoring. Anal. Chem. 2014, 86, 10546–10551. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Wiesner, U. Tailoring Pore Size of Graded Mesoporous Block Copolymer Membranes: Moving from Ultrafiltration toward Nanofiltration. Macromolecules 2015, 48, 6153–6159. [Google Scholar] [CrossRef]
- Ji, S.; Nagpal, U.; Liao, W.; Liu, C.C.; Pablo, J.J.D.; Nealey, P.F. Three-dimensional Directed Assembly of Block Copolymers together with Two-dimensional Square and Rectangular Nanolithography. Adv. Mater. 2011, 23, 3692–3697. [Google Scholar] [CrossRef] [PubMed]
- Orilall, M.C. Block copolymer based composition and morphology control in nanostructured hybrid materials for energy conversion and storage: solar cells, batteries, and fuel cells. Chem. Soc. Rev. 2011, 40, 520–535. [Google Scholar] [CrossRef] [PubMed]
- Jones, B.H.; Lodge, T.P. Hierarchically structured materials from block polymer confinement within bicontinuous microemulsion-derived nanoporous polyethylene. ACS Nano. 2011, 5, 8914–8927. [Google Scholar] [CrossRef] [PubMed]
- Schöttner, S.; Schaffrath, H.J.; Gallei, M. Poly(2-hydroxyethyl methacrylate)-Based Amphiphilic Block Copolymers for High Water Flux Membranes and Ceramic Templates. Macromolecules 2016, 49, 7286–7295. [Google Scholar] [CrossRef]
- Xu, J.; Sheng, Q.; Shen, Y.; Zheng, J. Enhanced direct electron transfer of glucose oxidase based on gold nanoprism and its application in biosensing. Colloids Surf. A Physicochem. Eng. Asp. 2017, 529, 113–118. [Google Scholar] [CrossRef]
- Shan, D.; He, Y.; Wang, S.; Xue, H.; Zheng, H. A porous poly(acrylonitrile-co-acrylic acid) film-based glucose biosensor constructed by electrochemical entrapment. Anal. Biochem. 2006, 356, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Sun, H.; Xu, F.; Yuwen, L.; Fan, C.; Wang, L. Direct electrochemistry of glucose oxidase and a biosensor for glucose based on a glass carbon electrode modified with MoS2 nanosheets decorated with gold nanoparticles. Microchim. Acta 2014, 181, 1497–1503. [Google Scholar] [CrossRef]
- Ruan, J.L.; Chen, C.; Shen, J.H.; Zhao, X.L.; Qian, S.H.; Zhu, Z.G. A Gelated Colloidal Crystal Attached Lens for Noninvasive Continuous Monitoring of Tear Glucose. Polymers 2017, 9, 125. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, L.; Zhang, Y.; Xiao, S.; Bi, F.; Zhao, J.; Gai, G.; Ding, J. Glucose Oxidase-Based Glucose-Sensitive Drug Delivery for Diabetes Treatment. Polymers 2017, 9, 255. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Pan, Y.; Min, L.; Zhang, Y.; Hu, X.; Yang, Z. Platinum nanoparticle-assembled nanoflake-like tin disulfide for enzyme-based amperometric sensing of glucose. Microchim. Acta 2017, 7, 1–7. [Google Scholar] [CrossRef]
- Liu, A.; Lang, Q.; Liang, B.; Shi, J. Sensitive detection of maltose and glucose based on dual enzyme-displayed bacteria electrochemical biosensor. Biosens. Bioelectron. 2017, 87, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Ye, H.; Zhang, Z.; Zhao, F.; Zeng, B. Metal–Organic Framework Derived Hollow Polyhedron CuCo2O4 Functionalized Porous Graphene for Sensitive Glucose Sensing. Sens. Actuators B 2016, 242, 728–735. [Google Scholar] [CrossRef]
- Yang, M.H.; Jeong, J.M.; Lee, K.G.; Kim, D.H.; Lee, S.J.; Choi, B.G. Hierarchical porous microspheres of the Co3O4@graphene with enhanced electrocatalytic performance for electrochemical biosensors. Biosens. Bioelectron. 2017, 89, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zheng, J. Synthesis of single-crystal α-MnO2 nanotubes-loaded Ag@C core-shell matrix and their application for electrochemical sensing of nonenzymatic hydrogen peroxide. Talanta 2016, 159, 231–237. [Google Scholar]
- Jia, N.; Huang, B.; Chen, L.; Tan, L.; Yao, S. A simple non-enzymatic hydrogen peroxide sensor using gold nanoparticles-graphene-chitosan modified electrode. Sens. Actuators B 2014, 195, 165–170. [Google Scholar] [CrossRef]
- Li, J.; Tang, Y.; Yang, J.; Yang, Z.; Zhang, Y.; Hu, X. Cage-like PbS nanostructure for the construction of novel glucose electrochemical biosensor. Sens. Actuators B 2014, 190, 549–554. [Google Scholar] [CrossRef]
- Li, J.; Yang, Z.; Tang, Y.; Zhang, Y.; Hu, X. Carbon nanotubes-nanoflake-like SnS2 nanocomposite for direct electrochemistry of glucose oxidase and glucose sensing. Biosens. Bioelectron. 2013, 41, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Xu, B.; Hu, C.; Shao, H.B.; Qu, L. Direct electrochemistry and electrocatalysis of glucose oxidase on three-dimensional interpenetrating, porous graphene modified electrode. Electrochim. Acta 2013, 98, 48–53. [Google Scholar] [CrossRef]
- Ye, D.; Liang, G.; Li, H.; Luo, J.; Zhang, S.; Chen, H.; Kong, J. A novel nonenzymatic sensor based on CuO nanoneedle/graphene/carbon nanofiber modified electrode for probing glucose in saliva. Talanta 2013, 116, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhou, C.; Luo, J.; Long, Y.; Wang, C.; Yu, T.; Xiao, D. A polyaniline microtube platform for direct electron transfer of glucose oxidase and biosensing applications. J. Mater. Chem. B 2015, 3, 1116–1124. [Google Scholar] [CrossRef]
- Cao, H.; Yang, A.; Li, H.; Wang, L.; Li, S.; Kong, J.; Bao, X.; Yang, R. A non-enzymatic glucose sensing based on hollow cuprous oxide nanospheres in a Nafion matrix. Sens. Actuators B 2015, 214, 169–173. [Google Scholar] [CrossRef]
- Zhang, E.; Xie, Y.; Ci, S.; Jia, J.; Wen, Z. Porous Co3O4 hollow nanododecahedra for nonenzymatic glucose biosensor and biofuel cell. Biosens. Bioelectron. 2016, 81, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Mani, V.; Devadas, B.; Chen, S.M. Direct electrochemistry of glucose oxidase at electrochemically reduced graphene oxide-multiwalled carbon nanotubes hybrid material modified electrode for glucose biosensor. Biosens. Bioelectron. 2013, 41, 309–315. [Google Scholar] [CrossRef] [PubMed]


| Electrode Taterials | Sensitivity (µA·mM−1·cm−2) | Detection Limit (µM) | Linear Range (mM) | Response Time (s) | Reference |
|---|---|---|---|---|---|
| PtNPs@SnS2/nafion | 10.56 | 2.5 | 0.1–1 | 5 | [26] |
| GA-bacteria/GDH-bacteria/MWNTs | - | 40 | 0.1–2 | 5 | [27] |
| Cu-Co-ZIFs/PGO | 2426 | 0.15 | 0.0005–3.354 | 3 | [28] |
| Co3O4@G | 628 | 0.038 | 0.02–8 | 4 | [29] |
| MnO2-Ag@C | 127.2 | 0.17 | 0.0005–5.7 | 2 | [30] |
| GNPs/GN-CS | - | 1.6 | 0.005–0.035 | 5 | [31] |
| Cage-like PbS/Nafion | 11.02 | 0.1 | 0.05–1.45 | 7 | [32] |
| MWCNTs-SnS2 | 21.65 | 4 | 0.02–1.95 | 7 | [33] |
| ERGO-chitosan hybrid | 6.82 | 1.7 | 0.02–3.2 | 6 | [34] |
| CuOnanoneedle/graphene/CNF | - | 0.1 | 1–5.3 | 2 | [35] |
| PANI microtube | 35.42 | 0.8 | 0.004–0.80 | 3 | [36] |
| Cu2O/Nafion | 2038.2 | 0.4 | 0.00125–0.0375 | 3 | [37] |
| Co3O4-HND | 708.4 | 0.58 | 0.002–6.06 | 2 | [38] |
| ERGO-MWCNT | - | 4.7 | 0.01–6.5 | 5 | [39] |
| PtPd-MWCNTs | 112 | 0.31 | 0.062–14.07 | 5 | [1] |
| PS-b-P4VP film | 54.74 | 0.05 | 0.01–4.5 | 2 | this work |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, T.; Gao, J.; Qin, X.; Zhang, X.; Xue, H. A Novel Glucose Biosensor Based on Hierarchically Porous Block Copolymer Film. Polymers 2018, 10, 723. https://doi.org/10.3390/polym10070723
Guo T, Gao J, Qin X, Zhang X, Xue H. A Novel Glucose Biosensor Based on Hierarchically Porous Block Copolymer Film. Polymers. 2018; 10(7):723. https://doi.org/10.3390/polym10070723
Chicago/Turabian StyleGuo, Teng, Jiefeng Gao, Xiang Qin, Xu Zhang, and Huaiguo Xue. 2018. "A Novel Glucose Biosensor Based on Hierarchically Porous Block Copolymer Film" Polymers 10, no. 7: 723. https://doi.org/10.3390/polym10070723
APA StyleGuo, T., Gao, J., Qin, X., Zhang, X., & Xue, H. (2018). A Novel Glucose Biosensor Based on Hierarchically Porous Block Copolymer Film. Polymers, 10(7), 723. https://doi.org/10.3390/polym10070723






