Multi-Alkenylsilsesquioxanes as Comonomers and Active Species Modifiers of Metallocene Catalyst in Copolymerization with Ethylene
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
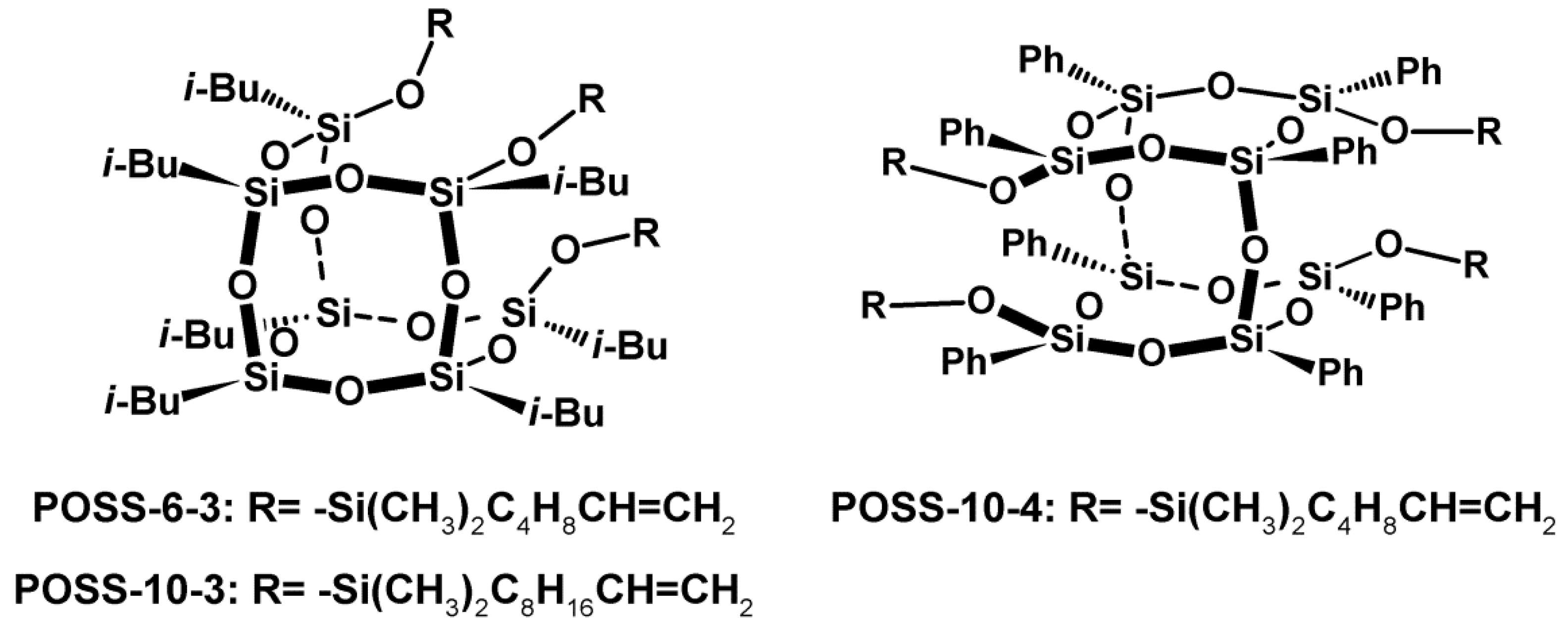
2.2. Experimental Section for the Synthesis of Trialkenyl- and Tetraalkenyl-Substituted Silsesquioxanes
2.3. Procedure for Ethylene Homopolymerization and Copolymerization of Ethylene with POSS
2.4. Analysis of (Co)polymer, Comonomer, and Catalytic System
3. Results
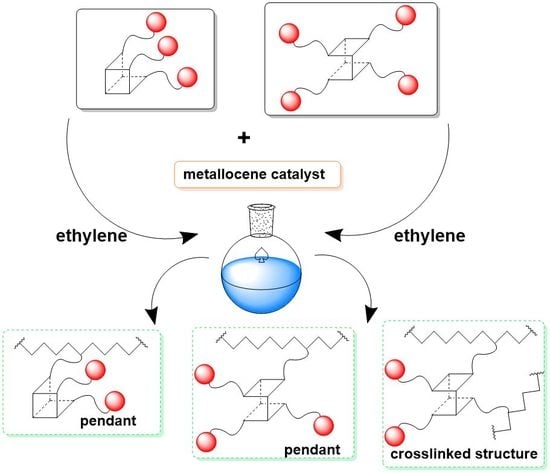
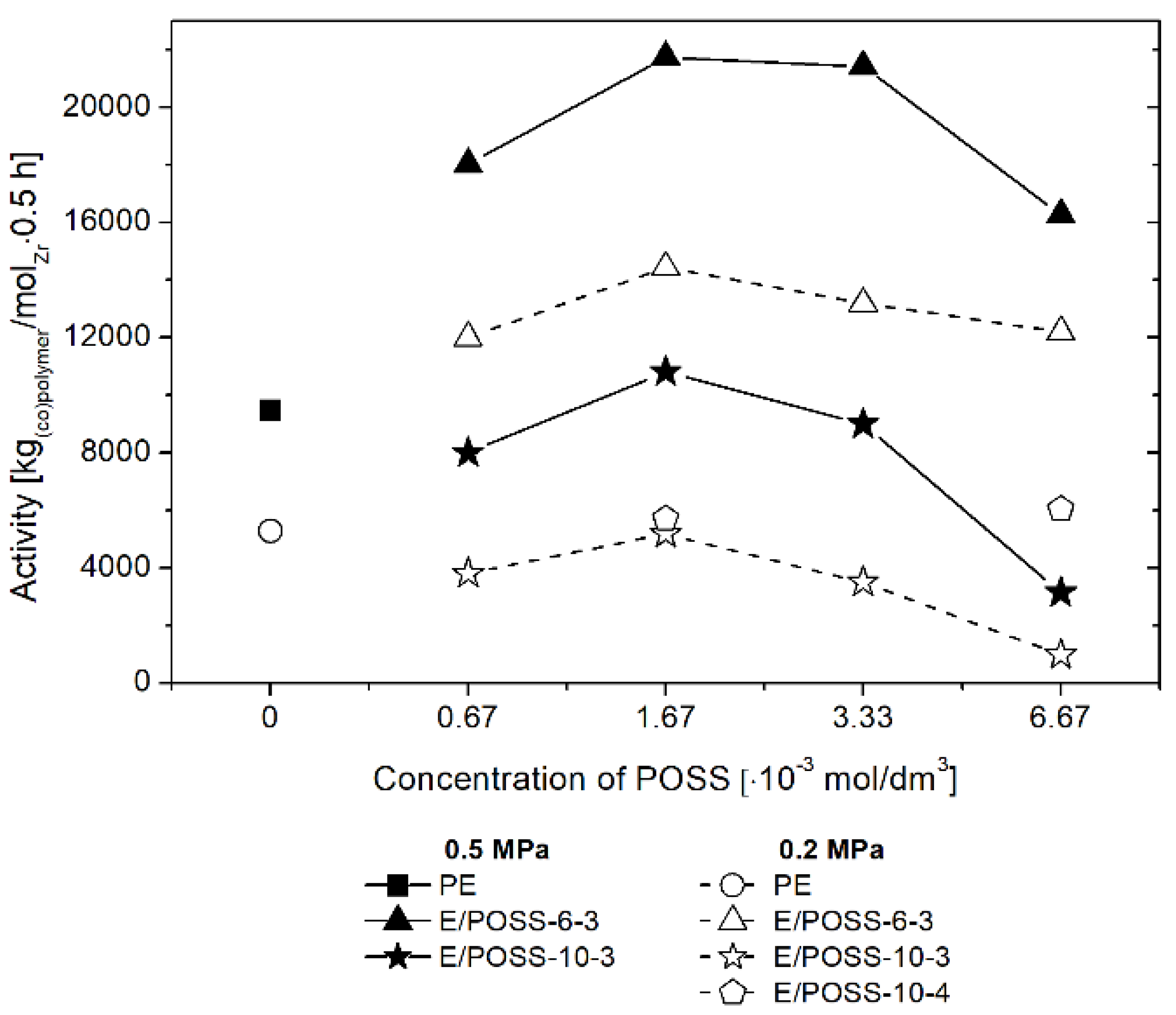
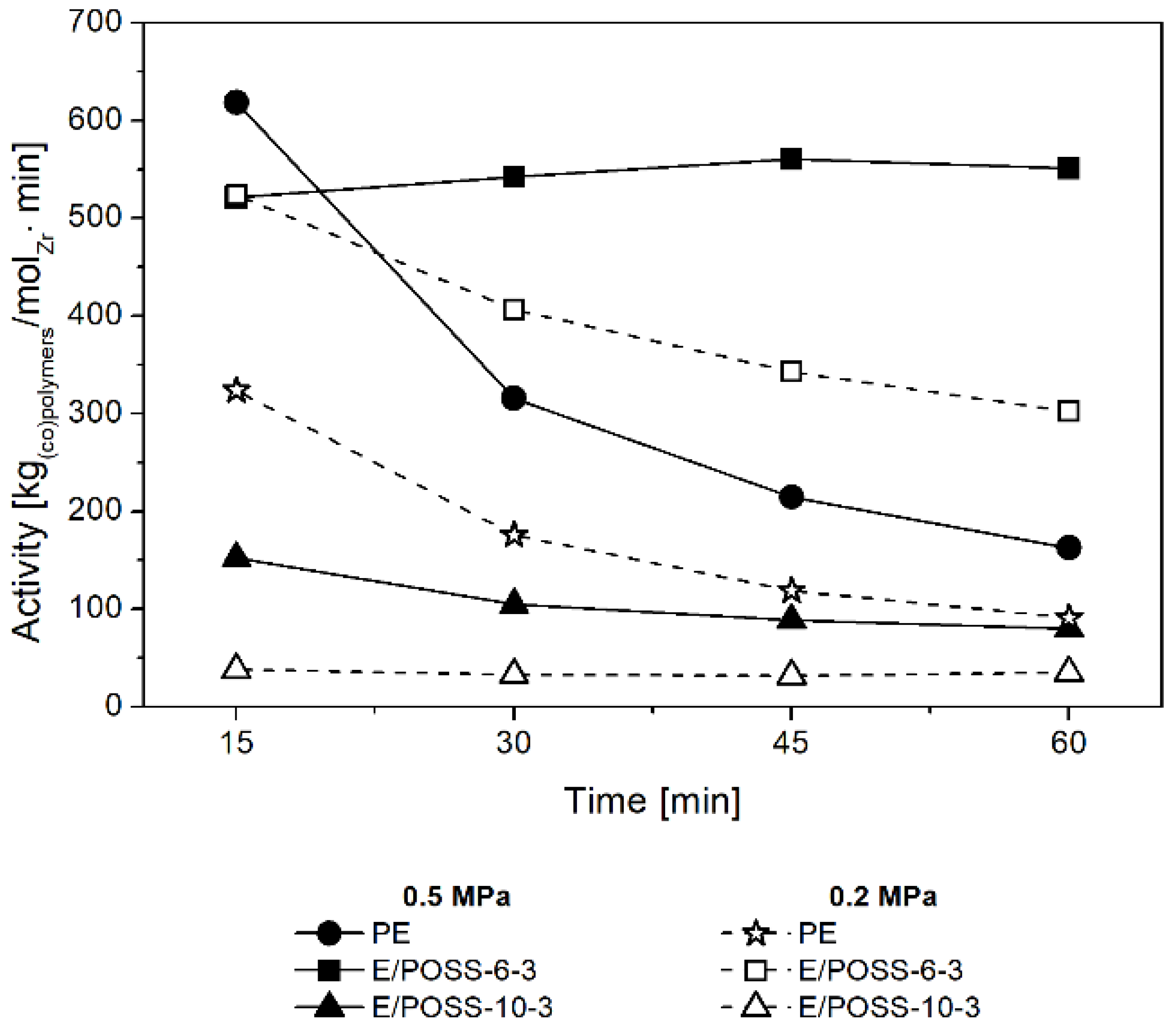
3.1. Performance of E/POSS Copolymerization
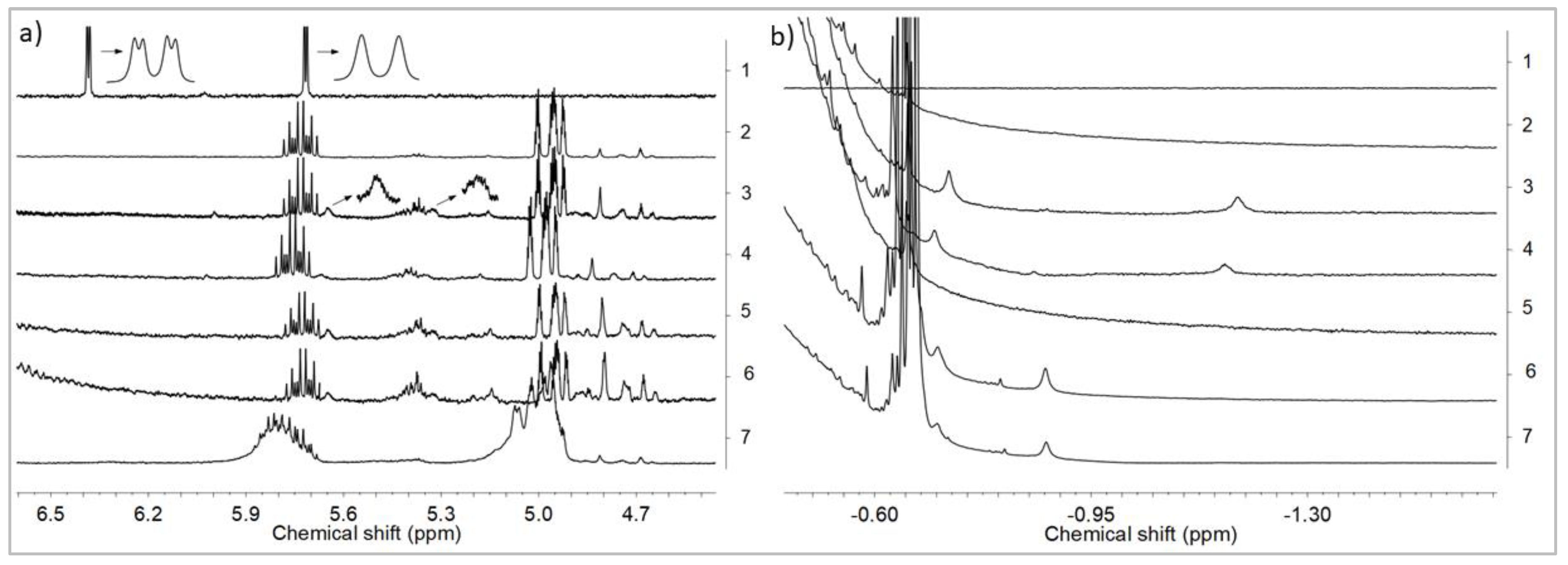
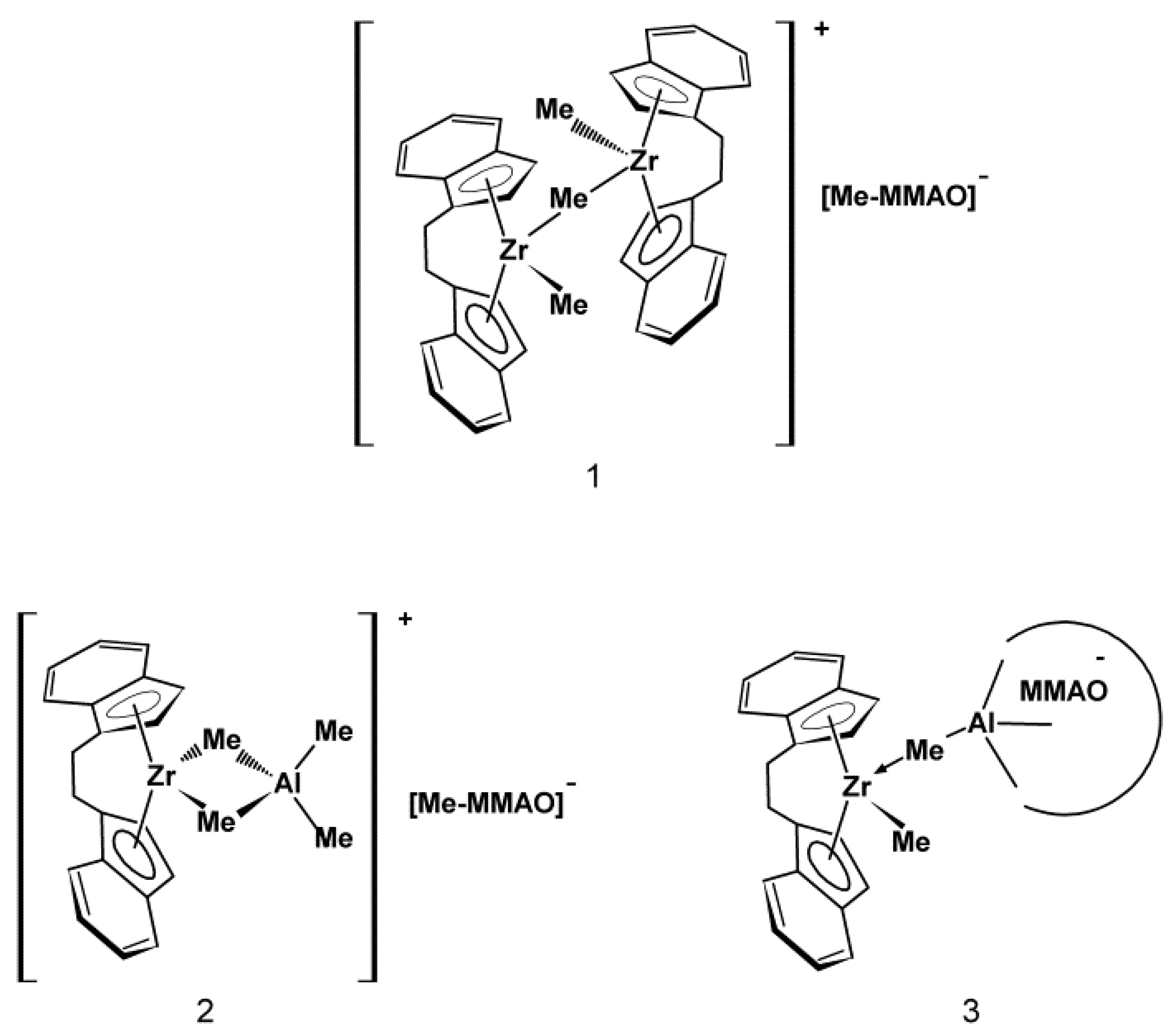
3.2. Influence of POSS on Ansa-Metallocene/MMAO Catalytic System
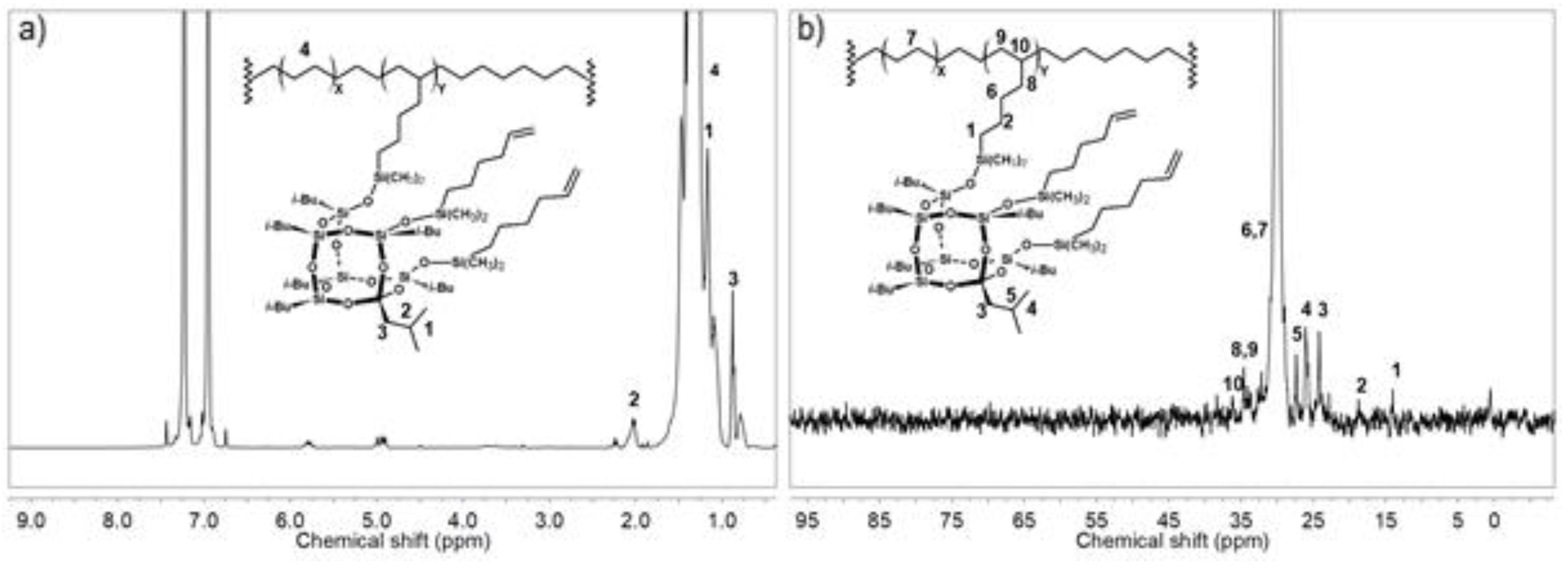
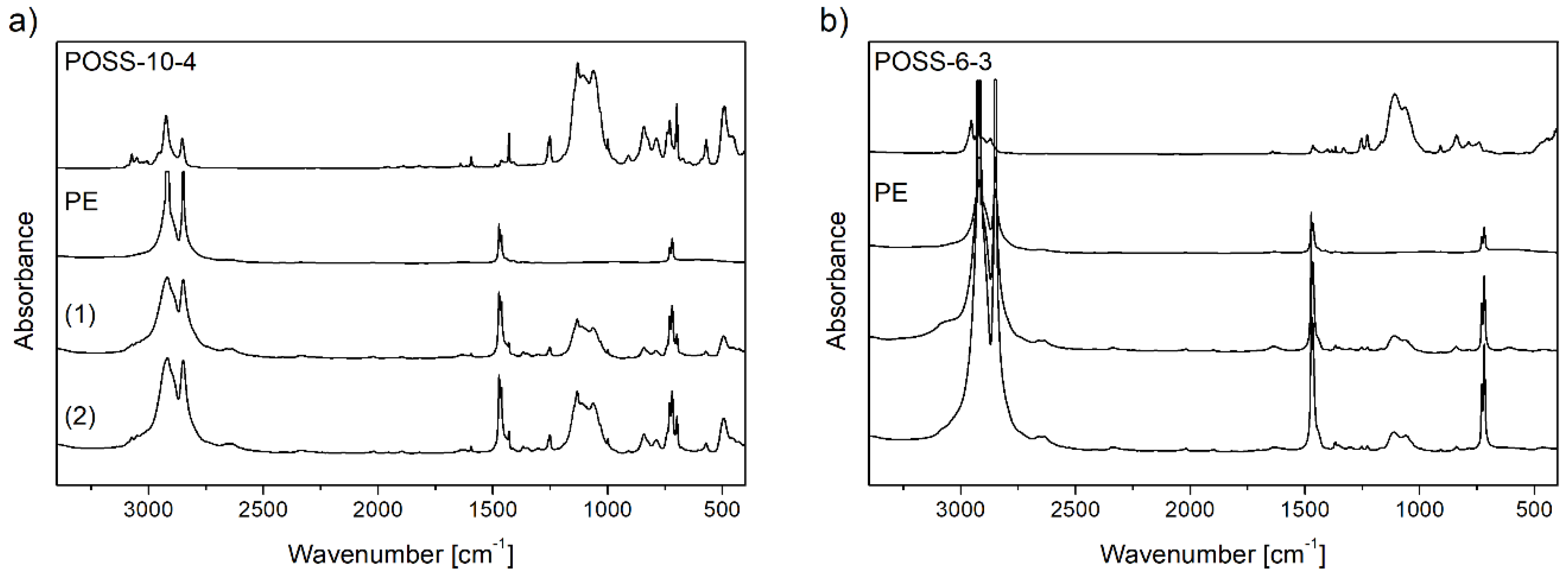
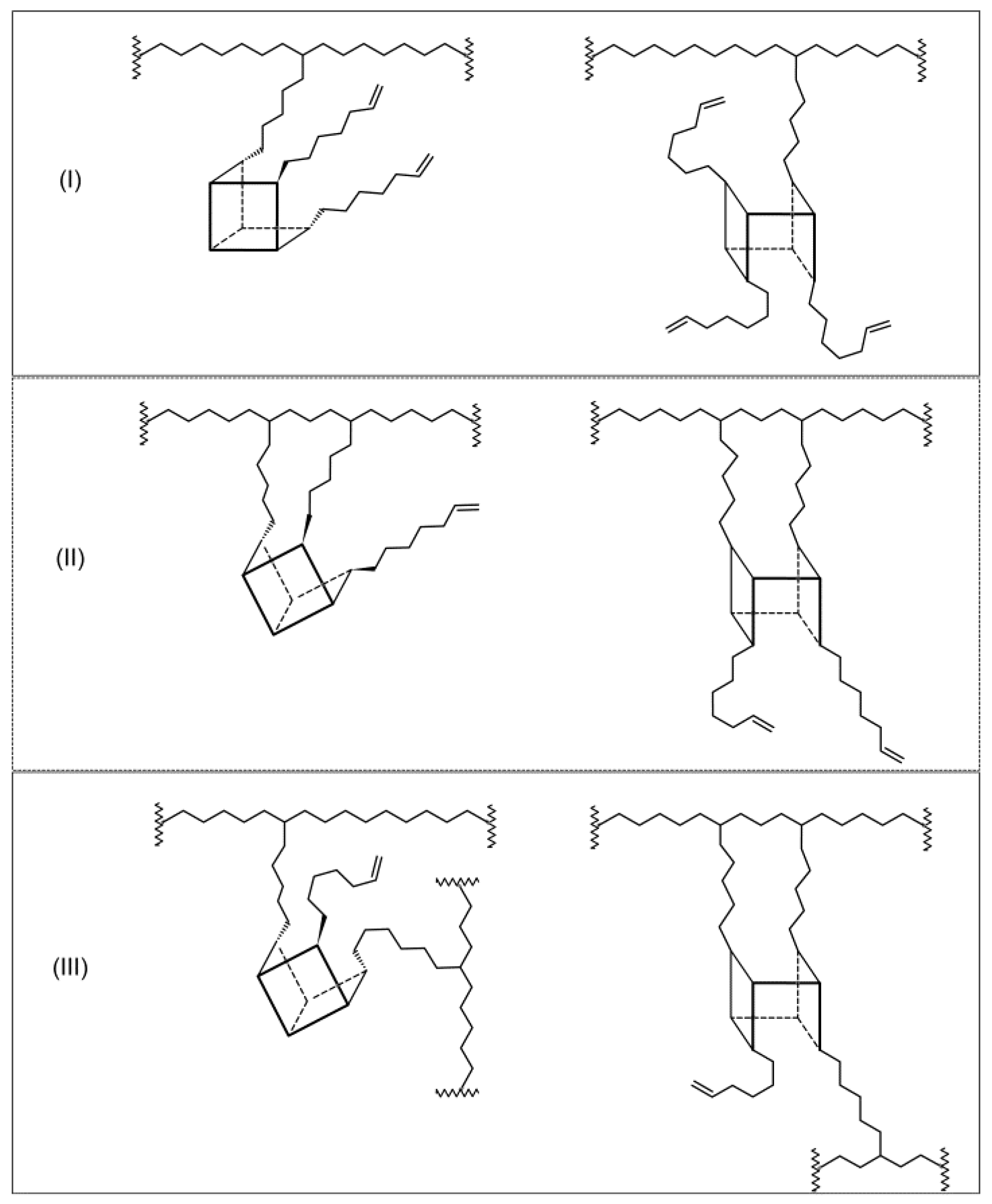
3.3. Structures of E/POSS Copolymers
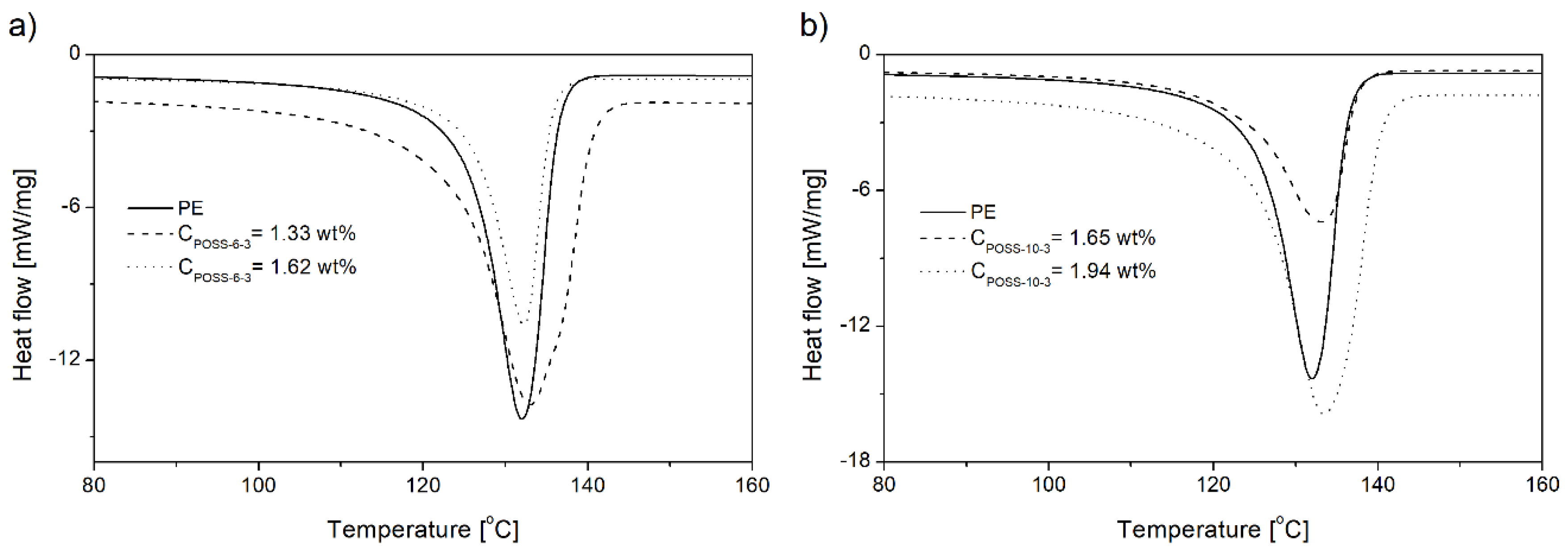
3.4. Crystallization and Melting Behavior of E/POSS Copolymers
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baney, R.H.; Itoh, M.; Sakakibara, A.; Suzuki, T. Silsesquioxanes. Chem. Rev. 1995, 95, 1409–1430. [Google Scholar] [CrossRef]
- Kickelbick, G. Introduction to Hybrid Materials. In Hybrid Materials; Wiley-VCH Verlag GmbH & Co., KGaA: Weinheim, Germany, 2007; pp. 1–48. [Google Scholar]
- Tsuchida, A.; Bolln, C.; Sernetz, F.G.; Frey, H.; Mülhaupt, R. Ethene and propene copolymers containing silsesquioxane side groups. Macromolecules 1997, 30, 2818–2824. [Google Scholar] [CrossRef]
- Zhang, H.-X.; Shin, Y.-J.; Yoon, K.-B.; Lee, D.-H. Preparation and properties of propylene/POSS copolymer with rac-Et(Ind)2ZrCl2 catalyst. Eur. Polym. J. 2009, 45, 40–46. [Google Scholar] [CrossRef]
- Zhang, H.-X.; Jung, M.-S.; Shin, Y.-J.; Yoon, K.-B.; Lee, D.-H. Preparation and properties of ethylene/POSS copolymer with rac-Et(Ind)2ZrCl2 catalyst. J. Appl. Polym. Sci. 2009, 111, 2697–2702. [Google Scholar] [CrossRef]
- Groch, P.; Dziubek, K.; Czaja, K.; Dudziec, B.; Marciniec, B. Copolymers of ethylene with monoalkenyl- and monoalkenyl(siloxy)silsesquioxane (POSS) comonomers—Synthesis and characterization. Eur. Polym. J. 2017, 90, 368–382. [Google Scholar] [CrossRef]
- Liu, N.; Li, L.; Wang, L.; Zheng, S. Organic-inorganic polybenzoxazine copolymers with double decker silsesquioxanes in the main chains: Synthesis and thermally activated ring-opening polymerization behavior. Polymer 2017, 109, 254–265. [Google Scholar] [CrossRef]
- Hao, J.; Wei, Y.; Chen, B.; Mu, J. Polymerization of polyhedral oligomeric silsequioxane (POSS) with perfluoro-monomers and a kinetic study. RSC Adv. 2017, 7, 10700–10706. [Google Scholar] [CrossRef]
- Wu, S.; Hayakawa, T.; Kikuchi, R.; Grunzinger, S.J.; Kakimoto, M.-A.; Oikawa, H. Synthesis and characterization of semiaromatic polyimides containing POSS in main chain derived from double-decker-shaped silsesquioxane. Macromolecules 2007, 40, 5698–5705. [Google Scholar] [CrossRef]
- Wu, S.; Hayakawa, T.; Kakimoto, M.-A.; Oikawa, H. Synthesis and characterization of organosoluble aromatic polyimides containing POSS in main chain derived from double-decker-shaped silsesquioxane. Macromolecules 2008, 41, 3481–3487. [Google Scholar] [CrossRef]
- Liu, N.; Wei, K.; Wang, L.; Zheng, S. Organic-inorganic polyimides with double decker silsesquioxane in the main chains. Polym. Chem. 2016, 7, 1158–1167. [Google Scholar] [CrossRef]
- Miyasaka, M.; Fujiwara, Y.; Kudo, H.; Nishikubo, T. Synthesis and characterization of hyperbranched polymer consisting of silsesquioxane derivatives. Polym. J. 2010, 42, 799–803. [Google Scholar] [CrossRef]
- Wei, K.; Wang, L.; Li, L.; Zheng, S. Synthesis and characterization of bead-like poly(N-isopropylacrylamide) copolymers with double decker silsesquioxane in the main chains. Polym. Chem. 2015, 6, 256–269. [Google Scholar] [CrossRef]
- Huang, J.; Jiang, P.; Wen, Y.; Deng, J.; He, J. Soy-castor oil based polyurethanes with octaphenylsilsesquioxanetetraol double-decker silsesquioxane in the main chains. RSC Adv. 2016, 6, 69521–69529. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, J.; Li, X.; Song, G.; Mu, J. Preparation, characterization, and properties of poly(aryl ether sulfone) systems with double-decker silsesquioxane in the main chains by reactive blending. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 780–788. [Google Scholar] [CrossRef]
- Żak, P.; Dudziec, B.; Dutkiewicz, M.; Ludwiczak, M.; Marciniec, B.; Nowicki, M. A new class of stereoregular vinylene-arylene copolymers with double-decker silsesquioxane in the main chain. J. Polym. Sci. Part A Polym. Chem. 2016, 54, 1044–1055. [Google Scholar] [CrossRef]
- Groch, P.; Dziubek, K.; Czaja, K.; Białek, M.; Mituła, K.; Dudziec, B.; Marciniec, B. Synthesis and structural characterization of ethylene copolymers containing double-decker silsesquioxane as pendant groups and cross-linkage sites by coordinative copolymerization. Eur. Polym. J. 2018, 100C, 187–199. [Google Scholar] [CrossRef]
- Liu, N.; Zheng, S. Organic–inorganic poly(N-vinylpyrrolidone) copolymers with double-decker silsesquioxane in the main chains: Synthesis, glass transition, and self-assembly behavior. J. Polym. Sci. Part A Polym. Chem. 2016, 54 18, 2949–2961. [Google Scholar] [CrossRef]
- Chen, D.; Yi, S.; Fang, P.; Zhong, Y.; Huang, C.; Wu, X. Synthesis and characterization of novel room temperature vulcanized (RTV) silicone rubbers using octa[(trimethoxysilyl)ethyl]-POSS as cross-linker. React. Funct. Polym. 2011, 71, 502–511. [Google Scholar] [CrossRef]
- Liu, H.; Zheng, S.; Nie, K. Morphology and Thermomechanical Properties of Organic−Inorganic Hybrid Composites Involving Epoxy Resin and an Incompletely Condensed Polyhedral Oligomeric Silsesquioxane. Macromolecules 2005, 38, 5088–5097. [Google Scholar] [CrossRef]
- Matějka, L.; Strachota, A.; Pleštil, J.; Whelan, P.; Steinhart, M.; Šlouf, M. Epoxy Networks Reinforced with Polyhedral Oligomeric Silsesquioxanes (POSS). Structure and Morphology. Macromolecules 2004, 37, 9449–9456. [Google Scholar] [CrossRef]
- Strachota, A.; Kroutilová, I.; Kovářová, J.; Matějka, L. Epoxy Networks Reinforced with Polyhedral Oligomeric Silsesquioxanes (POSS). Thermomechanical Properties. Macromolecules 2004, 37, 9457–9464. [Google Scholar] [CrossRef]
- Yusa, S.; Ohno, S.; Honda, T.; Imoto, H.; Nakao, Y.; Naka, K.; Nakamura, Y.; Fujii, S. Synthesis of silsesquioxane-based element-block amphiphiles and their self-assembly in water. RSC Adv. 2016, 6, 73006–73012. [Google Scholar] [CrossRef]
- Mituła, K.; Dutkiewicz, M.; Dudziec, B.; Marciniec, B.; Czaja, K. A library of monoalkenylsilsesquioxanes as potential comonomers for synthesis of hybrid materials. J. Therm. Anal. Calorim. 2017. in review. [Google Scholar]
- Mituła, K.; Duszczak, J.; Brzakalski, D.; Dudziec, B.; Kubicki, M.; Marciniec, B. Tetra-functional double-decker silsesquioxanes as anchors for reactive functional groups and potential synthons for hybrid materials. Chem. Commun. 2017, 53, 10370–10373. [Google Scholar] [CrossRef] [PubMed]
- Mituła, K.; Dudziec, B.; Marciniec, B. Synthesis of dialkenyl-substituted double-decker silsesquioxanes as precursors for linear copolymeric systems. J. Inorg. Organomet. Polym. Mater. 2017, 1–8. [Google Scholar] [CrossRef]
- Widman, G.; Riesen, R. Thermal Analysis: Terms, Methods, Application; Alfred Huthing Verlag: Heidelberg, Germany, 1987. [Google Scholar]
- Kaminsky, W.; Laban, A. Metallocene catalysis. Appl. Catal A Gen. 2001, 222, 47–61. [Google Scholar] [CrossRef]
- Lipponen, S.H.; Seppälä, J.V. Ethylenebis(indenyl)zirconium dichloride/methylaluminoxane-catalyzed copolymerization of ethylene and 1-alkene-n-trimethylsilanes. Organometallics 2011, 30, 528–533. [Google Scholar] [CrossRef]
- Hu, P.; Wang, J.-Q.; Wang, F.; Jin, G.-X. Preparation, Structure, and Ethylene (Co)Polymerization Behavior of Group IV Metal Complexes with an [OSSO]-Carborane Ligand. Chem. Eur. J. 2011, 17, 8576–8583. [Google Scholar] [CrossRef] [PubMed]
- Kaminsky, W.; Winkelbach, H. Influence of supported metallocene catalysts on polymer tacticity. Top. Catal. 1999, 7, 61–67. [Google Scholar] [CrossRef]
- Olabisi, O.; Atiqullah, M.; Kaminsky, W. Group 4 Metallocenes: Supported and Unsupported. J. Macromol. Sci. C 1997, 37, 519–554. [Google Scholar] [CrossRef]
- Tritto, I.; Donetti, R.; Sacchi, M.C.; Locatelli, P.; Zannoni, G. Dimethylzirconocene−Methylaluminoxane Catalyst for Olefin Polymerization: NMR Study of Reaction Equilibria. Macromolecules 1997, 30, 1247–1252. [Google Scholar] [CrossRef]
- Lyakin, O.Y.; Bryliakov, K.P.; Semikolenova, N.V.; Lebedev, A.Y.; Voskoboynikov, A.Z.; Zakharov, V.A.; Talsi, E.P. 1H and 13C NMR Studies of Cationic Intermediates Formed upon Activation of “Oscillating” Catalyst (2-PhInd)2ZrCl2 with MAO, MMAO, and AlMe3/[CPh3]+[B(C6F5)4]. Organometallics 2007, 26, 1536–1540. [Google Scholar] [CrossRef]
- Bryliakov, K.P.; Talsi, E.P.; Bochmann, M. 1H and 13C NMR Spectroscopic Study of Titanium(IV) Species Formed by Activation of Cp2TiCl2 and [(Me4C5)SiMe2NtBu]TiCl2 with Methylaluminoxane (MAO). Organometallics 2004, 23, 149–152. [Google Scholar] [CrossRef]
- Talsi, E.P.; Bryliakov, K.P.; Semikolenova, N.V.; Zakharov, V.A.; Ystenes, M.; Rytterc, E. 1H NMR characterization of intermediates formed by the activation of zirconocenes with methylaluminoxane at high Al/Zr ratios. Mendeleev Commun. 2003, 13, 46–48. [Google Scholar] [CrossRef]
- Bryliakov, K.P.; Semikolenova, N.V.; Yudaev, D.V.; Ystenes, M.; Rytter, E.; Zakharov, V.A.; Talsi, E.P. 1H and 13C NMR Study of the Intermediates Formed by (Cp-R)2ZrCl2 Activation with MAO and AlMe3/[CPh3][B(C6F5)4]. Correlation of Spectroscopic and Ethene Polymerization Data. Macromol. Chem. Phys. 2003, 204, 1110–1117. [Google Scholar] [CrossRef]
- Bryliakov, K.P.; Semikolenova, N.V.; Yudaev, D.V.; Zakharov, V.A.; Brintzinger, H.H.; Ystenes, M.; Rytter, E.; Talsi, E.P. 1H-, 13C-NMR and ethylene polymerization studies of zirconocene/MAO catalysts: Effect of the ligand structure on the formation of active intermediates and polymerization kinetics. J. Organomet. Chem. 2003, 683, 92–102. [Google Scholar] [CrossRef]
- Babushkin, D.E.; Brintzinger, H.-H. Activation of Dimethyl Zirconocene by Methylaluminoxane (MAO) Size Estimate for Me-MAO- Anions by Pulsed Field-Gradient NMR. J. Am. Chem. Soc. 2002, 124, 12869–12873. [Google Scholar] [CrossRef] [PubMed]
- Babushkin, D.E.; Semikolenova, N.V.; Zakharov, V.A.; Talsi, E.P. Mechanism of dimethylzirconocene activation with methylaluminoxane: NMR monitoring of intermediates at high Al/Zr ratios. Macromol. Chem. Phys. 2000, 201, 558–567. [Google Scholar] [CrossRef]
- Rocchigiani, L.; Busico, V.; Pastore, A.; Macchioni, A. Probing the interactions between all components of the catalytic pool for homogeneous olefin polymerisation by diffusion NMR spectroscopy. Dalton Trans. 2013, 42, 9104–9111. [Google Scholar] [CrossRef] [PubMed]
- Ciancaleoni, G.; Fraldi, N.; Budzelaar, P.H.M.; Busico, V.; Macchioni, A. Structure and Dynamics in Solution of Bis(phenoxy-amine)Zirconium Catalysts for Olefin Polymerization. Organometallics 2011, 30, 3096–3105. [Google Scholar] [CrossRef]
- Groch, P.; Dziubek, K.; Czaja, K.; Białek, M.; Adamczyk-Tomiak, K.; Rabiej, S.; Dudziec, B. Ethylene/POSS copolymerization behavior of postmetallocene catalysts and copolymer characteristics. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 3918–3934. [Google Scholar] [CrossRef]
- Bellamy, L.J. The Infrared Spectra of Complex Molecules; Chapman and Hall: London, UK, 1975; Chapter 20. [Google Scholar]
- Anderson, D.R. Analysis of Silicones; Wiley-Interscience: New York, NY, USA, 1974; Chapter 10. [Google Scholar]
- Launer, P.J.; Arkles, B. Reprinted from Silicon Compounds: Silanes & Silicones; Gelest, Inc.: Morrisville, PA, USA, 2013. [Google Scholar]
- Fineman, M.; Ross, S.D. Linear method for determining monomer reactivity ratios in copolymerization. J. Polym. Sci. 1950, 5, 259–262. [Google Scholar] [CrossRef]
- Bruaseth, I.; Rytter, E. Dual site ethene/1-hexene copolymerization with MAO activated (1,2,4-Me3Cp)2ZrCl2 and (Me5Cp)2ZrCl2 catalysts. Possible transfer of polymer chains between the sites. Macromolecules 2003, 36, 3026–3034. [Google Scholar] [CrossRef]
- Kokko, E.; Pietikäinen, P.; Koivunen, J.; Seppälä, J.V. Long-chain-branched polyethene by the copolymerization of ethene and nonconjugated α,ω-dienes. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 3805–3817. [Google Scholar] [CrossRef]
- Song, S.; Wu, A.; Yu, Y.; Yang, P.; Fu, Z.; Fan, Z. Nonconjugated diene homopolymerization and copolymerization with ethylene catalyzed by α-diimine Ni(II) complex/Et2AlCl. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 1900–1909. [Google Scholar] [CrossRef]

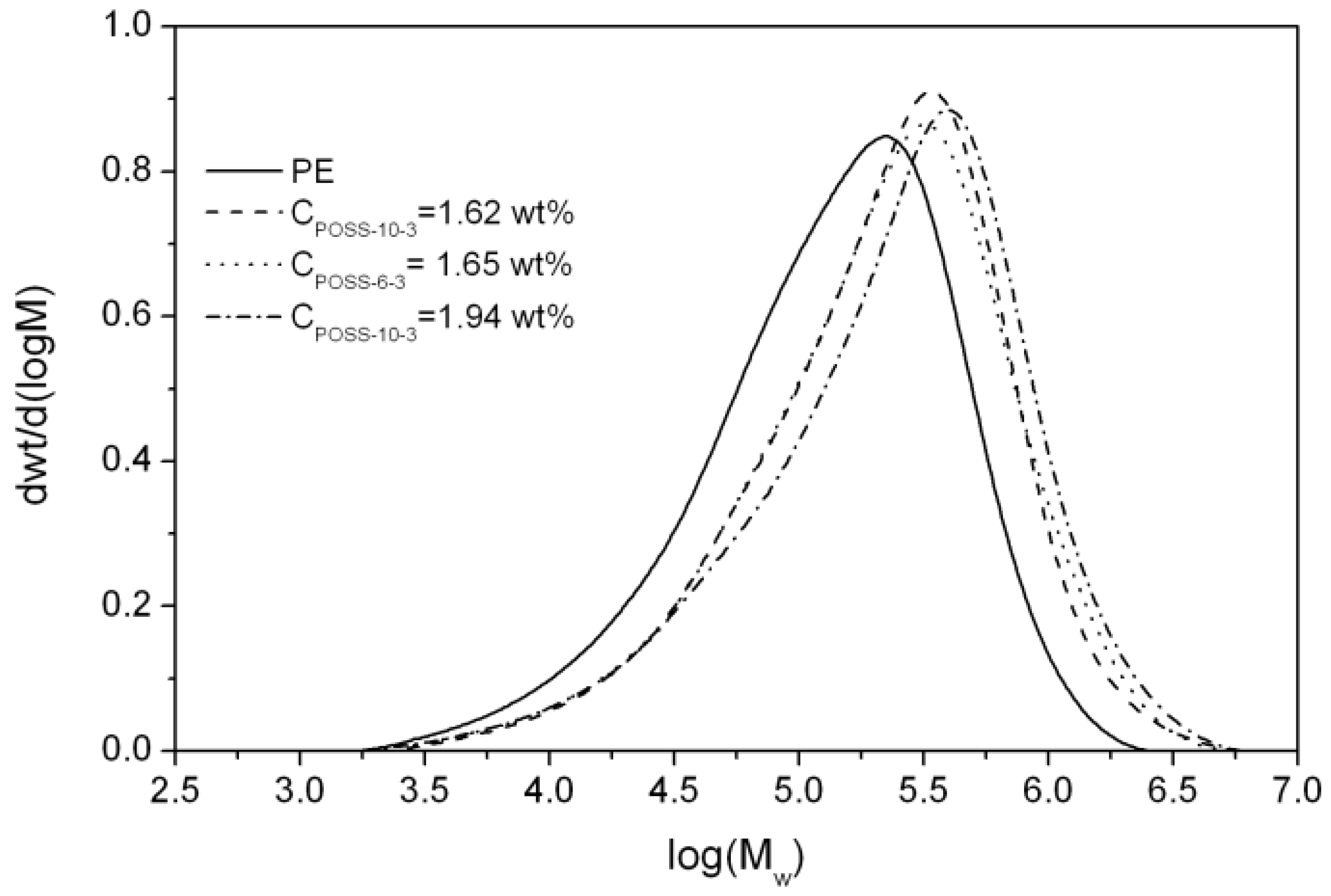
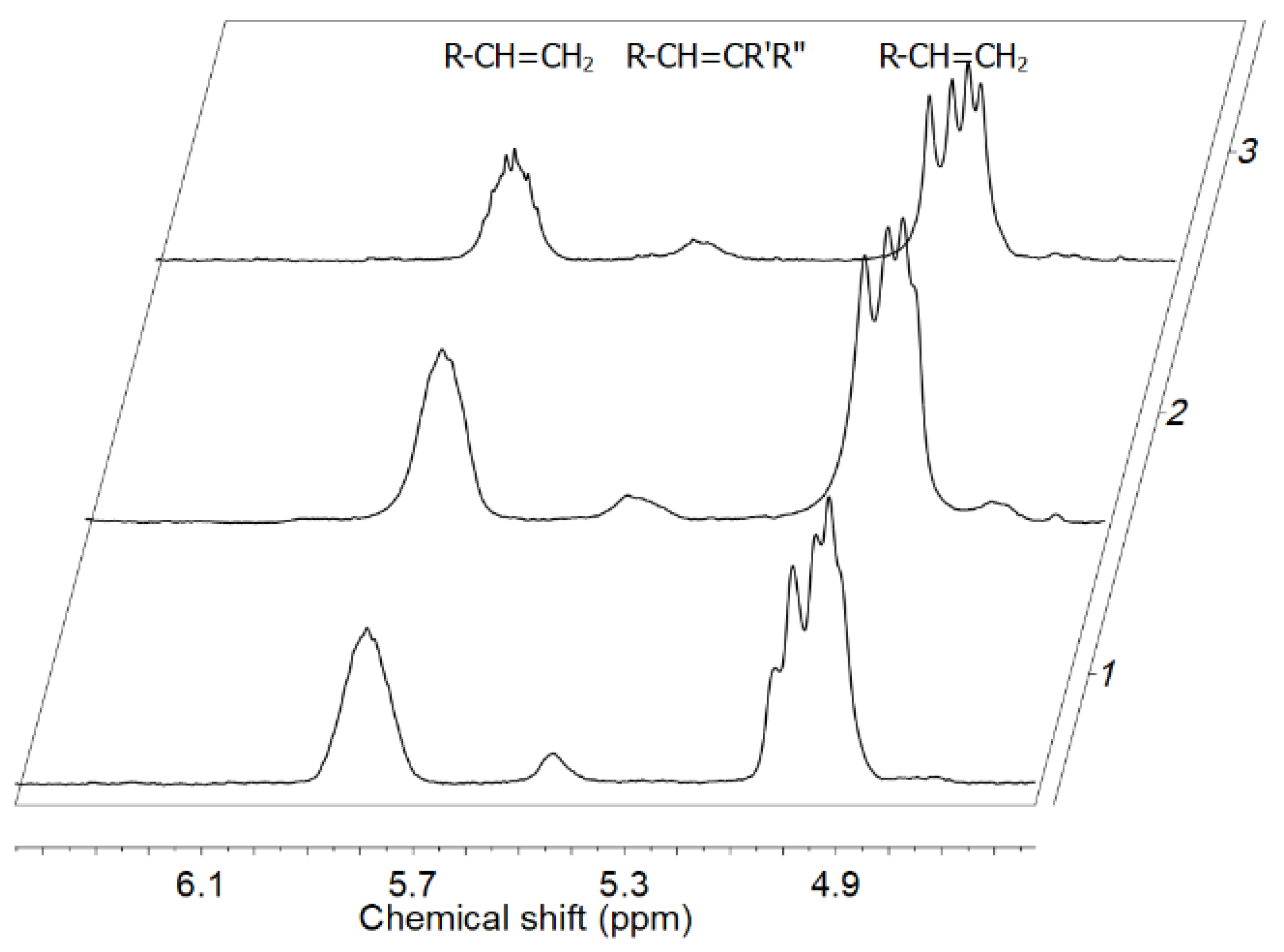
| Item | (Co)polymers | CPOSS e | ne e | MD e | Mw·103 (g/mol) g | Analysis of Unsaturated End Groups f | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| (mol %) | (wt %) | A908/A2020 | A800/A2020 | |||||||
| 1 | PE | 0 | 0 | 0 | 0 | 0 | 165 | 1.9 | 0.460 | 0 |
| 2 a | E/POSS-6-3 | 7.86 | 0.031 | 1.33 | 3226 | 99.969 | 326 | 3.9 | 0.649 | 0.145 |
| 3 b | 9.81 | 0.038 | 1.62 | 2632 | 99.962 | 364 | 4.0 | 0.857 | 0.110 | |
| 4 b,c | 12.13 | 0.047 | 2.00 | − | − | − | − | 0.984 | 0.328 | |
| 5 a | E/POSS-10-3 | 8.83 | 0.034 | 1.65 | 2941 | 99.966 | 377 | 4.4 | 0.843 | 0.175 |
| 6 b | 9.98 | 0.040 | 1.94 | 2500 | 99.960 | 434 | 4.9 | 1.053 | 0.187 | |
| 7 b,c | 8.25 | 0.031 | 1.51 | − | − | 526 | 4.8 | 0.769 | 0.541 | |
| 8 c,d | PE | 0 | 0 | 0 | 0 | 0 | 226 | 3.9 | 0.291 | 0 |
| 9 a,d | E/POSS-6-3 | 14.46 | 0.056 | 2.37 | 1786 | 99.944 | − | − | 0.945 | 0.161 |
| 10 b,d | 23.78 | 0.093 | 3.87 | 1075 | 99.907 | − | − | 1.664 | 0 | |
| 11 b,c,d | 16.43 | 0.064 | 2.70 | − | − | − | − | 0.928 | 0.463 | |
| 12 a,d | E/POSS-10-3 | 14.66 | 0.057 | 2.74 | 1754 | 99.943 | − | − | 1.062 | 0 |
| 13 b,d | 17.46 | 0.068 | 3.25 | 1471 | 99.932 | − | − | 1.481 | 0.429 | |
| 14 b,c,d | 11.07 | 0.043 | 2.08 | − | − | − | − | 1.337 | 0.309 | |
| 15 a,d | E/POSS-10-4 | 19.11 | f 0.075 | 4.74 | f 1333 | 99.925 | − | − | 0.565 | 2.663 |
| 16 b,c | 30.76 | f 0.121 | 7.43 | f 826 | 99.879 | − | − | 0.971 | 4.933 | |
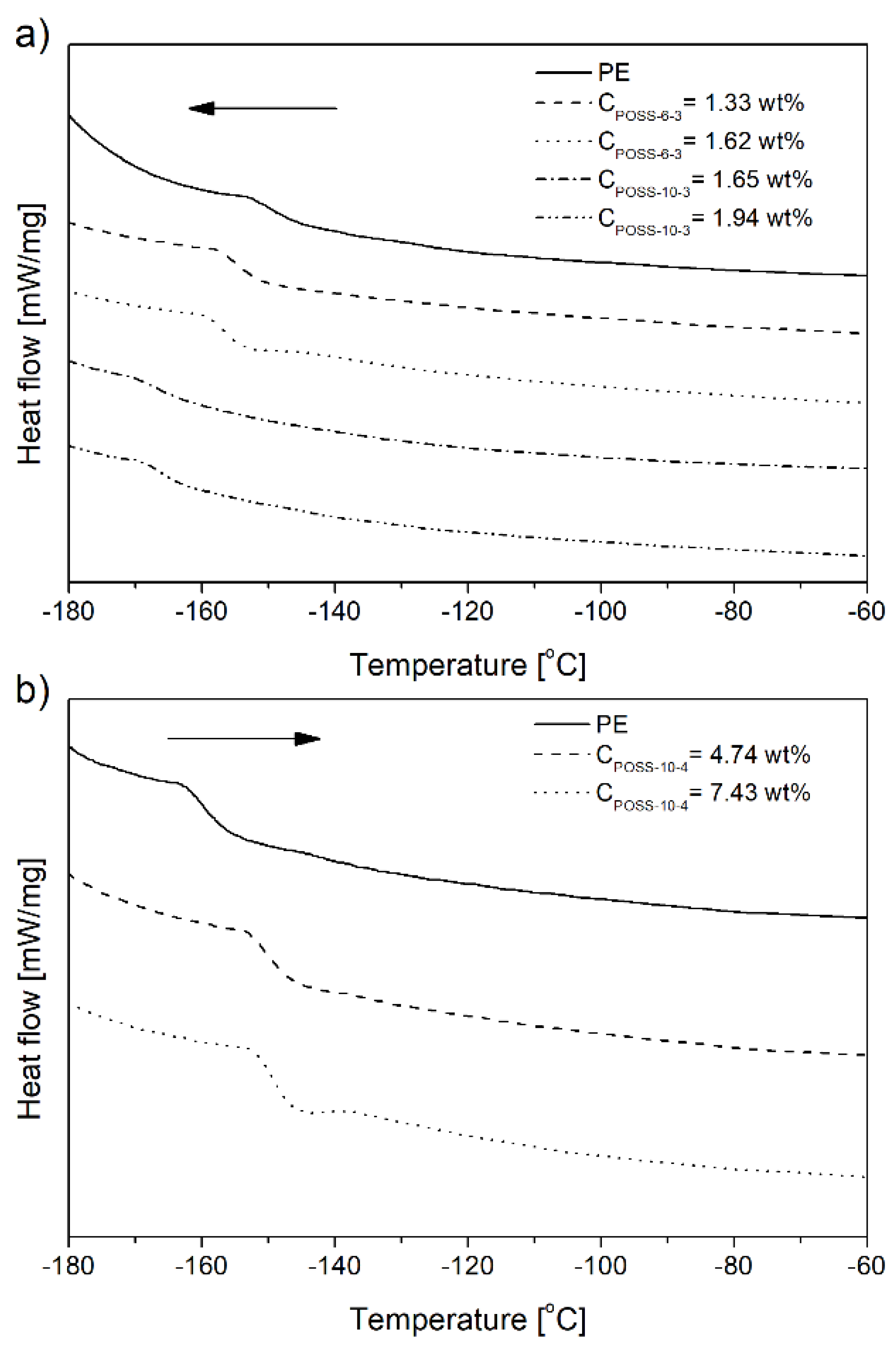
| Item | (Co)polymers | Tg [°C] | Xc c [%] | Melting Temperaturec | Crystallization Temperature c | ||||
|---|---|---|---|---|---|---|---|---|---|
| Tmo | Tmp | Tme | Tco | Tcp | Tce | ||||
| 1 | PE e | −149.7 | 63.2 | 124.8 | 131.0 | 136.4 | 119.7 | 116.5 | 111.9 |
| 2 a | E/POSS-6-3 e | −156.0 | 60.9 | 122.4 | 134.8 | 140.1 | 121.4 | 115.6 | 106.8 |
| 3 b | −157.1 | 59.1 | 125.4 | 132.3 | 135.6 | 120.1 | 118.2 | 114.1 | |
| 4 a | E/POSS-10-3 e | −166.5 | 58.5 | 122.5 | 134.3 | 138.3 | 120.8 | 115.1 | 108.2 |
| 5 b | −166.6 | 60.4 | 124.2 | 133.6 | 140.3 | 120.2 | 116.4 | 107.4 | |
| 6 | PE d | −155.0 | 61.2 | 123.2 | 128.6 | 134.6 | 119.8 | 116.5 | 112.8 |
| 7 a | E/POSS-6-3 d | −157.8 | 56.2 | 121.7 | 132.2 | 135.9 | 120.5 | 116.4 | 108.8 |
| 8 b | −158.0 | 56.7 | 121.8 | 131.0 | 135.9 | 120.1 | 116.3 | 108.9 | |
| 9 a | E/POSS-10-3 d | −157.3 | 54.7 | 119.3 | 129.8 | 133.6 | 119.8 | 115.1 | 108.2 |
| 10 b | −162.5 | 50.6 | 122.0 | 129.4 | 135.7 | 118.6 | 116.0 | 108.2 | |
| 11 | PE d | −159.3 | 62.2 | 123.2 | 129.6 | 134.6 | 119.8 | 117.5 | 112.8 |
| 12 a | E/POSS-10-4 d | −150.4 | 53.7 | 116.4 | 129.5 | 135.4 | 119.0 | 114.7 | 104.7 |
| 13 b | −149.5 | 50.2 | 117.2 | 127.9 | 132.5 | 118.4 | 114.7 | 106.0 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Groch, P.; Dziubek, K.; Czaja, K.; Mituła, K.; Dudziec, B. Multi-Alkenylsilsesquioxanes as Comonomers and Active Species Modifiers of Metallocene Catalyst in Copolymerization with Ethylene. Polymers 2018, 10, 223. https://doi.org/10.3390/polym10020223
Groch P, Dziubek K, Czaja K, Mituła K, Dudziec B. Multi-Alkenylsilsesquioxanes as Comonomers and Active Species Modifiers of Metallocene Catalyst in Copolymerization with Ethylene. Polymers. 2018; 10(2):223. https://doi.org/10.3390/polym10020223
Chicago/Turabian StyleGroch, Paweł, Katarzyna Dziubek, Krystyna Czaja, Katarzyna Mituła, and Beata Dudziec. 2018. "Multi-Alkenylsilsesquioxanes as Comonomers and Active Species Modifiers of Metallocene Catalyst in Copolymerization with Ethylene" Polymers 10, no. 2: 223. https://doi.org/10.3390/polym10020223
APA StyleGroch, P., Dziubek, K., Czaja, K., Mituła, K., & Dudziec, B. (2018). Multi-Alkenylsilsesquioxanes as Comonomers and Active Species Modifiers of Metallocene Catalyst in Copolymerization with Ethylene. Polymers, 10(2), 223. https://doi.org/10.3390/polym10020223