Reversible Single-Crystal-to-Single-Crystal Phase Transition of Chiral Salicylidenephenylethylamine
Abstract
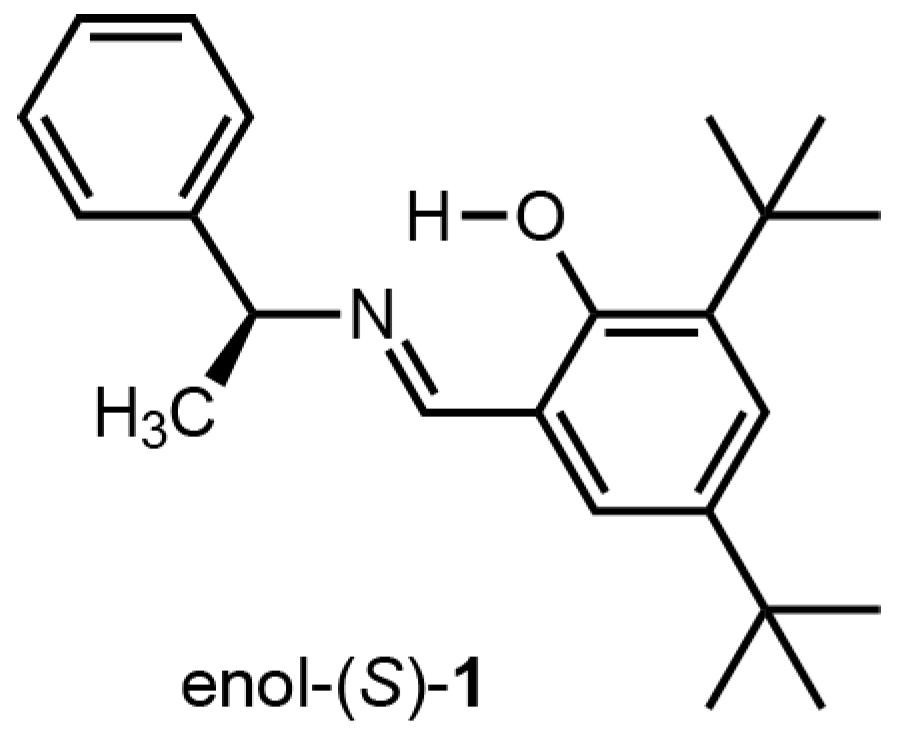
:1. Introduction
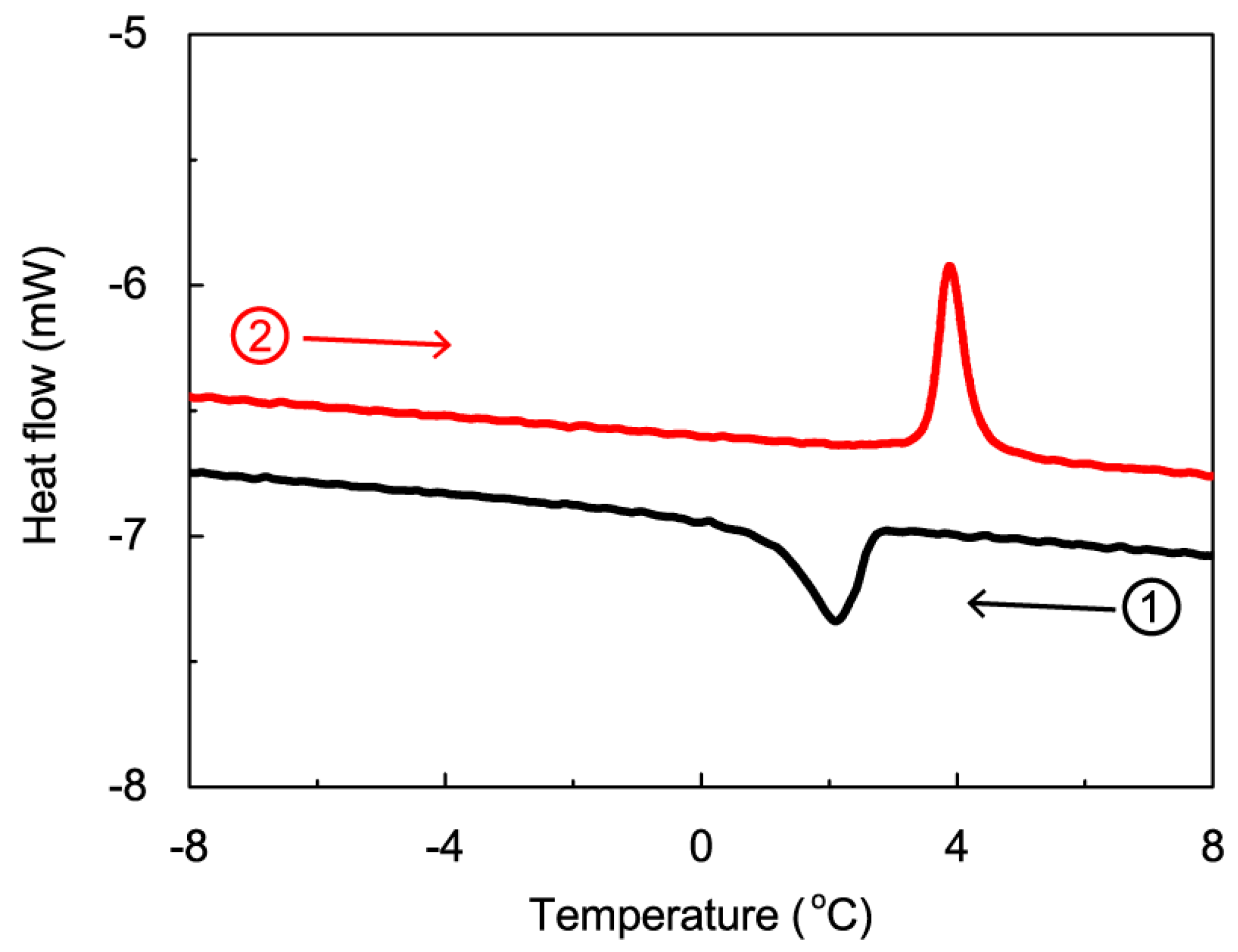
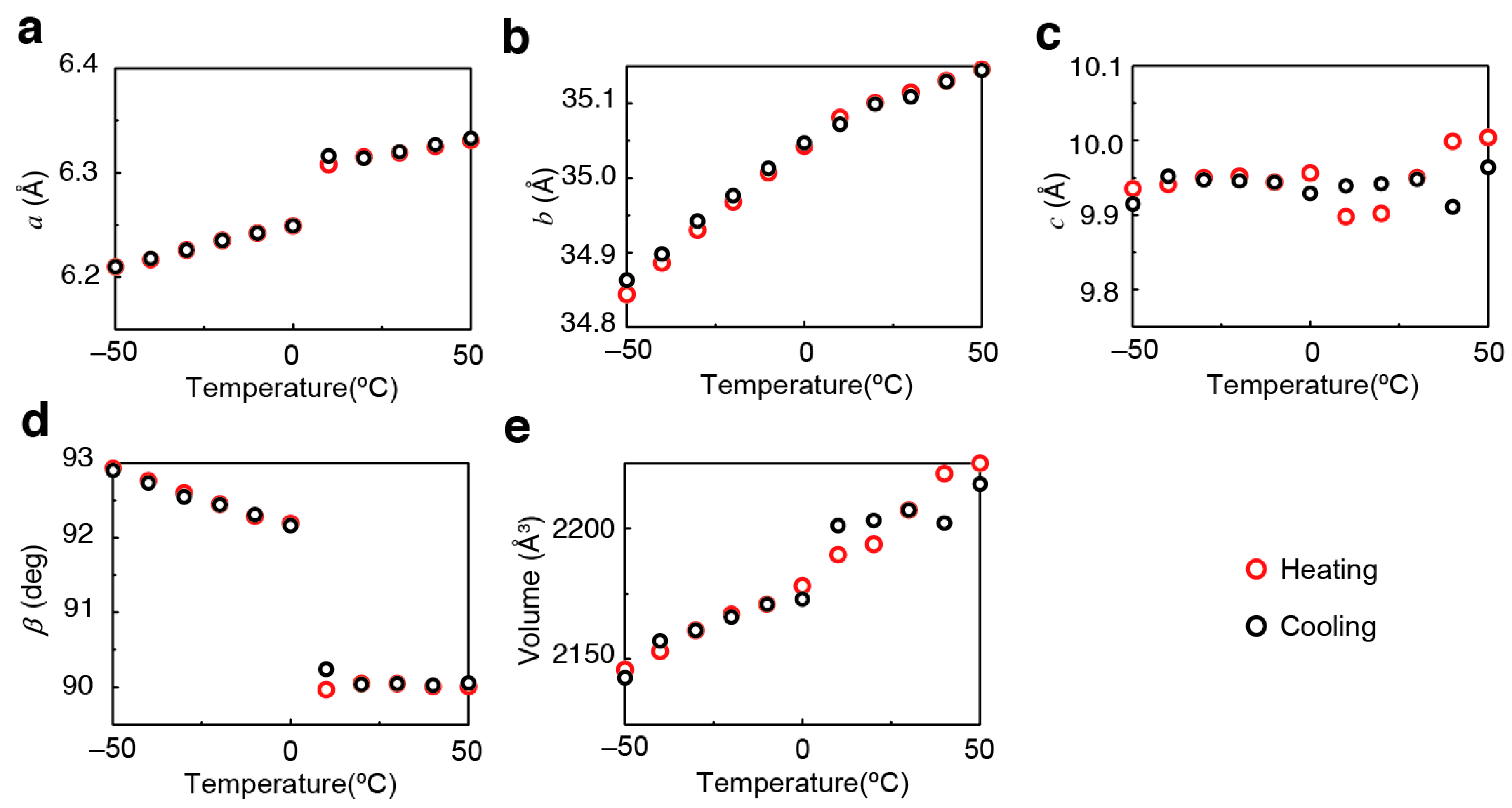
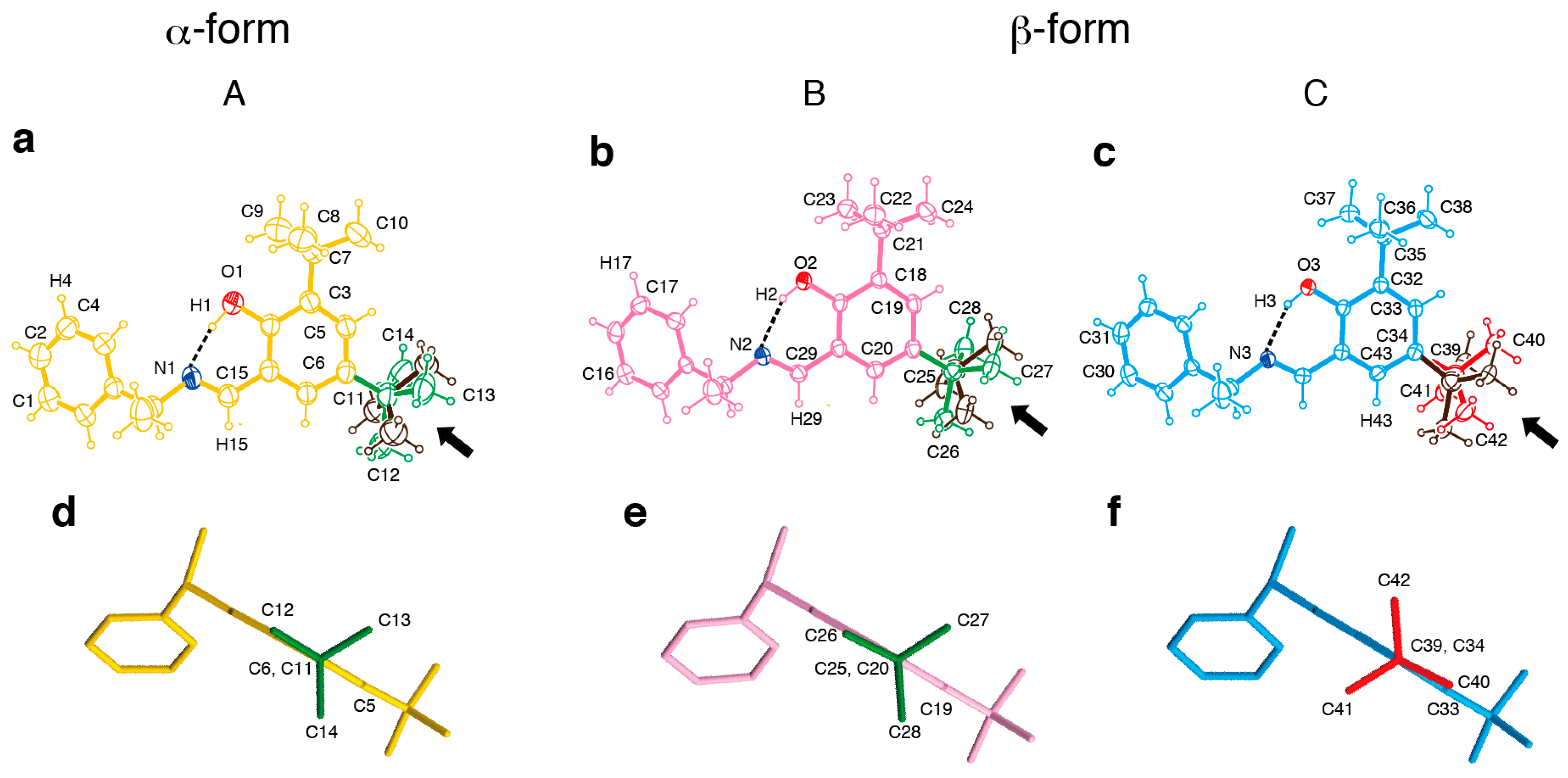
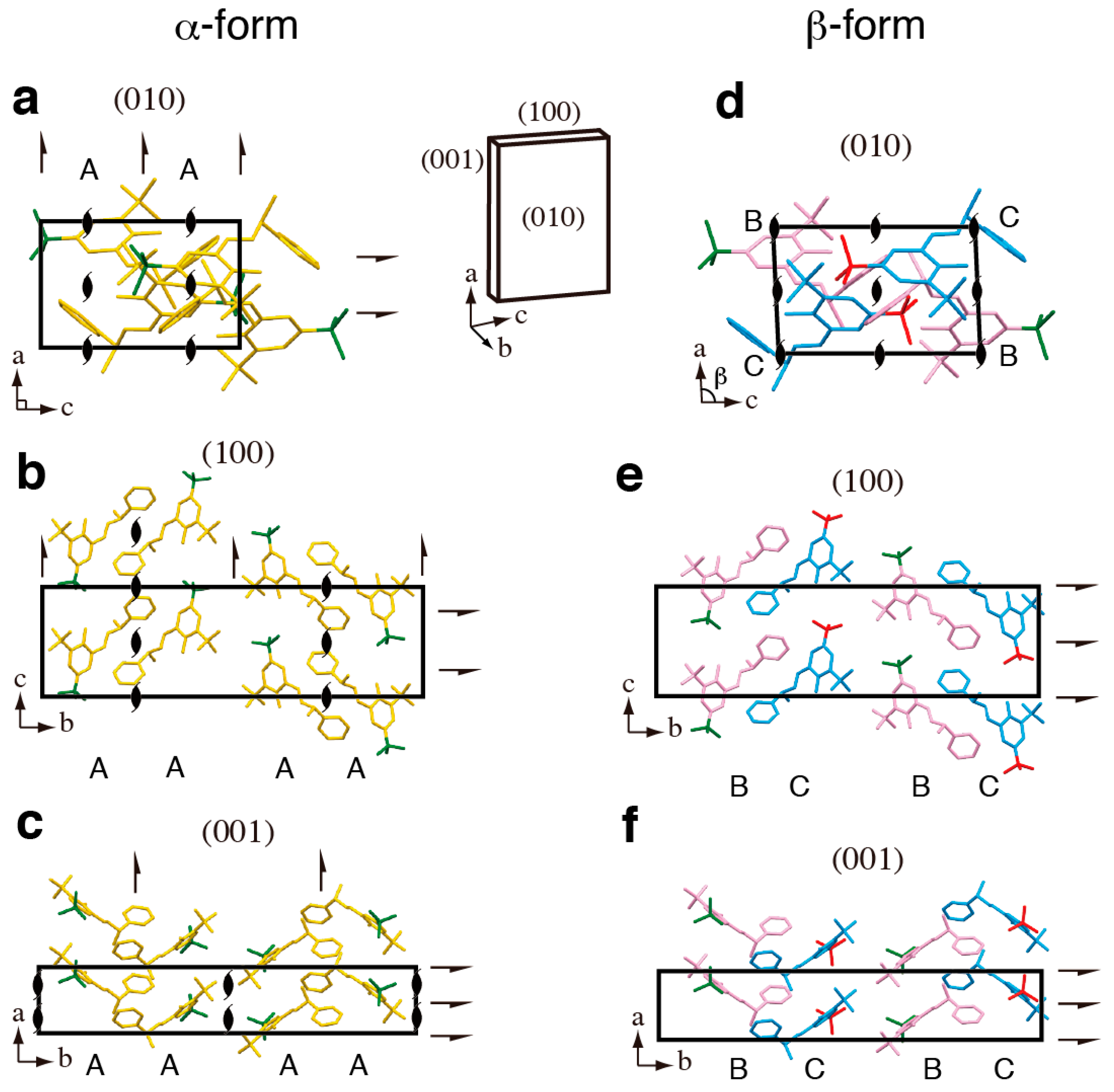
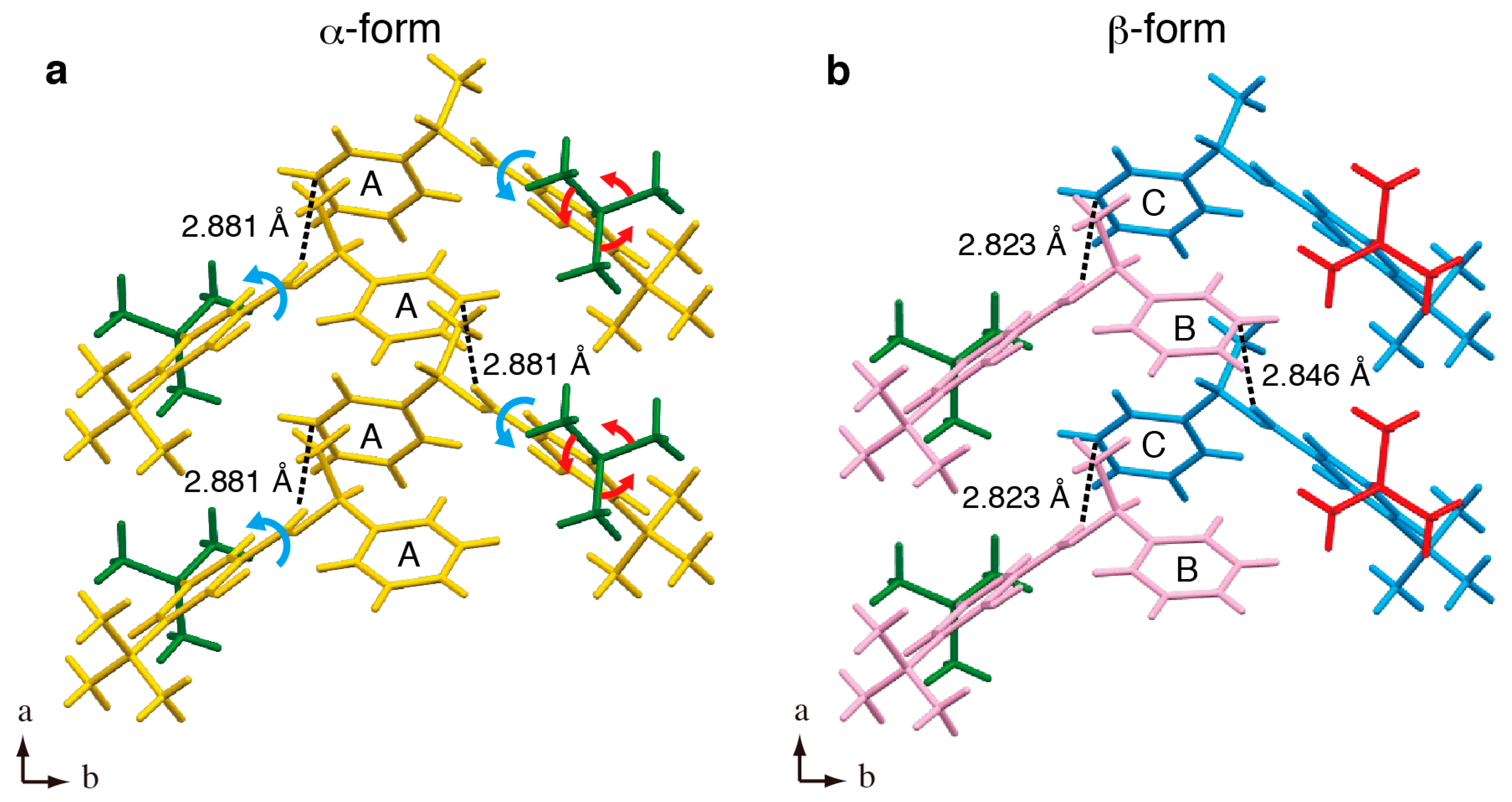
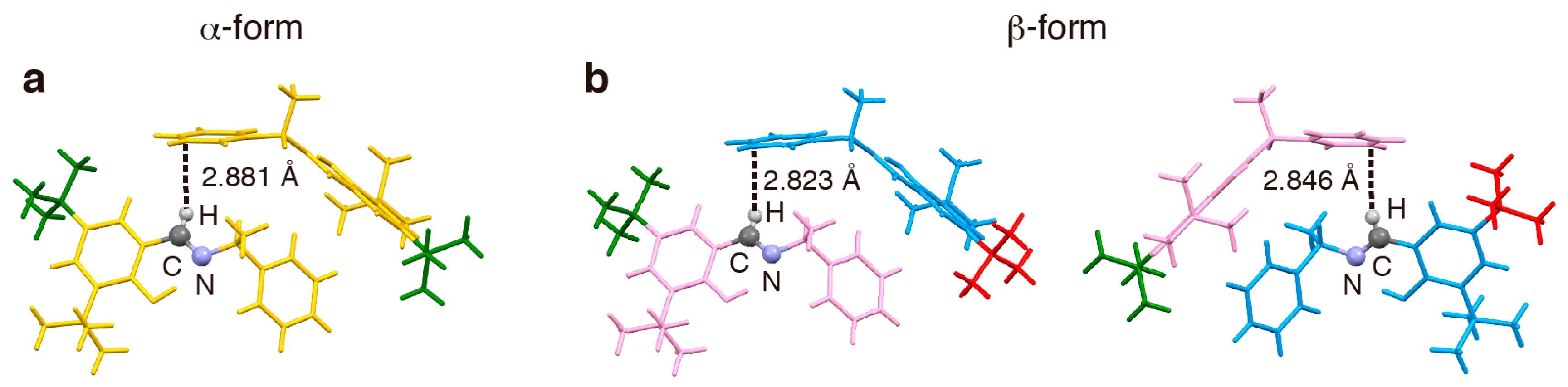
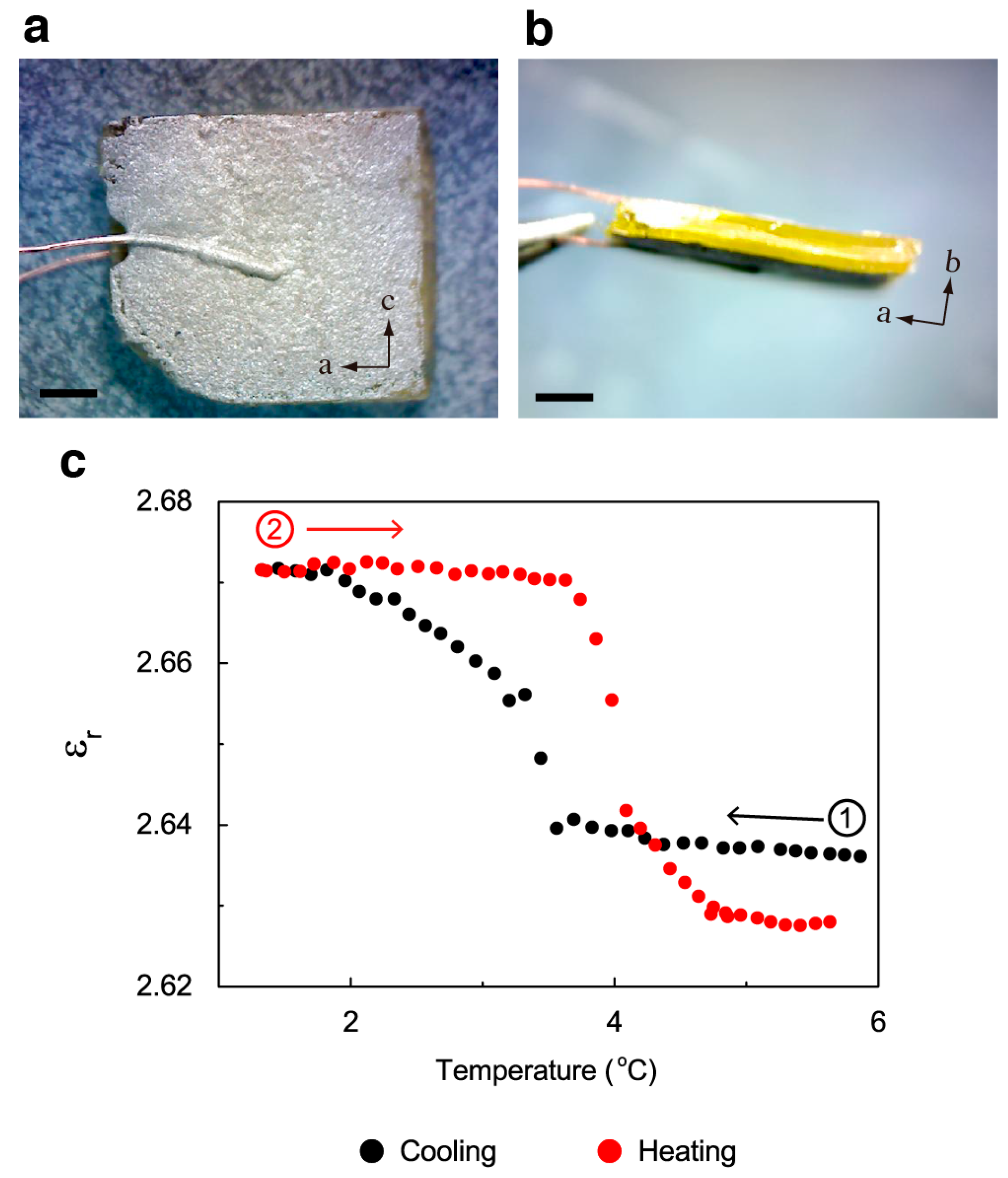
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Differential Scanning Calorimetry
3.3. Crystallography
3.4. Dielectric Measurements
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Horiuchi, S.; Tokura, Y. Organic ferroelectrics. Nat. Mater. 2008, 7, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.S.; Yamamoto, K.; Cai, H.L.; Takahashi, K.; Sato, O. Above Room Temperature Organic Ferroelectrics: Diprotonated 1,4-Diazabicyclo[2.2.2]octane Shifts between Two 2-Chlorobenzoates. J. Am. Chem. Soc. 2016, 138, 12005–12008. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.Z.; Gu, Z.F.; Xiong, J.B.; Gao, J.X.; Liu, Y.; Wang, B.; Tan, Y.H.; Xu, Q. Unusual Sequential Reversible Phase Transitions Containing Switchable Dielectric Behaviors in Cyclopentyl Ammonium 18-Crown-6 Perchlorate. Chem. Mater. 2016, 28, 4476–4482. [Google Scholar] [CrossRef]
- Kaftory, M.; Botoshansky, M.; Kapon, M.; Shteiman, V. Irreversible single-crystal to polycrystal and reversible single-crystal to single-crystal phase transformations in cyanurates. Acta Crystallogr. Sect. B Struct. Sci. 2001, 57, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.A.; Levendis, D.C.; Schoening, F.R.L. A new polymorph of ortho-ethoxy-trans-cinnamic acid: Single-to-single-crystal phase transformation and mechanism. Acta Crystallogr. Sect. B Struct. Sci. 2004, 60, 300–314. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Ito, Y. Low-temperature-induced reversible single-crystal-to-single-crystal phase transition of 3,4-dichloro-2′,4′,6′-triethylbenzophenone. CrystEngComm 2010, 12, 1628–1634. [Google Scholar] [CrossRef]
- Schmidt, A.; Kababya, S.; Appel, M.; Khatib, S.; Botoshansky, M.; Eichen, Y. Measuring the Temperature Width of a First-Order Single Crystal to Single Crystal Phase Transition Using Solid-State NMR: Application to the Polymorphism of 2-(2,4-Dinitrobenzyl)-3-methylpyridine. J. Am. Chem. Soc. 1999, 121, 11291–11299. [Google Scholar] [CrossRef]
- Das, D.; Engel, E.; Barbour, L.J. Reversible single-crystal to single-crystal polymorphic phase transformation of an organic crystal. Chem. Commun. 2010, 46, 1676–1678. [Google Scholar] [CrossRef] [PubMed]
- Girard, J.; Fromm, K. Single crystal to single crystal polymorphic phase transition of a silver nitrate 24-crown-8 complex and its pseudo-polymorphism. CrystEngComm 2012, 14, 6487–6491. [Google Scholar] [CrossRef]
- Pal, R.; Reddy, M.B.M.; Dinesh, B.; Balaram, P.; Row, T.N.G. Temperature-Induced Reversible First-Order Single Crystal to Single Crystal Phase Transition in Boc-γ4(R)Val-Val-OH: Interplay of Enthalpy and Entropy. J. Phys. Chem. A 2014, 118, 9568–9574. [Google Scholar] [CrossRef] [PubMed]
- Kawato, T.; Koyama, H.; Kanatomi, H.; Shigemizu, H. Difference in the Rate of Photo-induced Unimolecular Motion of Chiral Salicylideneamines in the Chiral Crystal Environments. Chem. Lett. 1997, 26, 401–402. [Google Scholar] [CrossRef]
- Koshima, H.; Matsuo, R.; Matsudomi, M.; Uemura, Y.; Shiro, M. Light-Driven Bending Crystals of Salicylidenephenylethylamines in Enantiomeric and Racemate Forms. Cryst. Growth Des. 2013, 13, 4330–4337. [Google Scholar] [CrossRef]
- Takanabe, A.; Tanaka, M.; Johmoto, K.; Uekusa, H.; Mori, T.; Koshima, H.; Asahi, T. Optical Activity and Optical Anisotropy in Photomechanical Crystals of Chiral Salicylidenephenylethylamines. J. Am. Chem. Soc. 2016, 45, 15066–15077. [Google Scholar] [CrossRef] [PubMed]
- Centore, R.; Jazbinsek, M.; Tuzi, A.; Roviello, A.; Capobianco, A.; Peluso, A. A series of compounds forming polar crystals and showing single-crystal-to-single-crystal transitions between polar phases. CrystEngComm 2012, 14, 2645–2653. [Google Scholar] [CrossRef]
- Smith, H.E.; Cook, S.L.; Warren, M.E., Jr. Optically Active Amines. II. The Optical Rotatory Dispersion Curves of the N-Benzylidene and Substituted N-Benzylidene Derivatives of Some Open-Chain Primary Amines. J. Org. Chem. 1964, 29, 2265–2272. [Google Scholar] [CrossRef]
- Johmoto, K.; Ishida, T.; Sekine, A.; Uekusa, H.; Ohashi, Y. Relation between photochromic properties and molecular structures in salicylideneaniline crystals. Acta Crystallogr. Sect. B Struct. Sci. 2012, 68, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.; Dos Santos, M.L.; Chaves, M.R.; Amaral, M.H.; Tolédano, J.C.; Périgaud, A.; Savary, H. Pyroelectric effect in benzil. Ferroelectrics 1988, 79, 253–256. [Google Scholar] [CrossRef]
- Horiuchi, S.; Tokunaga, Y.; Giovannetti, G.; Picozzi, S.; Itoh, H.; Shimano, R.; Kumai, R.; Tokura, Y. Above-room-temperature ferroelectricity in a single-component molecular crystal. Nature 2010, 463, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Rigaku. ABSCOR; Rigaku Corporation: Tokyo, Japan, 1995. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Adv. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Rigaku. NUMABS; Rigaku Corporation: Tokyo, Japan, 1999. [Google Scholar]
- Sheldrick, G.M. A Short History of SHELX. Acta Crystallogr. Sect. A Found. Adv. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
| α-Form | β-Form | |
|---|---|---|
| Temperature (°C) | 10 | −50 |
| Crystal system | Orthorhombic | Monoclinic |
| Space group | P212121 | P21 |
| a (Å) | 6.3058 (4) | 6.20653 (11) |
| b (Å) | 35.071 (2) | 34.8358 (7) |
| c (Å) | 9.8851 (6) | 9.92593 (18) |
| β (deg) | 90 | 92.7570 (10) |
| Volume (Å3) | 2186.1 (2) | 2143.59 (7) |
| Z | 4 | 4 |
| ρcalc (g·cm−3) | 1.025 | 1.046 |
| R1 [I > 2σ(I)] | 0.0681 | 0.0475 |
| Goodness of fit | 1.052 | 1.030 |
| α-Form | β-Form | ||
|---|---|---|---|
| Molecule A | Molecule B | Molecule C | |
| Intramolecular hydrogen bond | |||
| N---O distance (Å) | 2.591 | 2.596 | 2.591 |
| N---H−O angle (deg) | 160.4 | 148.67 | 148.94 |
| Dihedral angle between phenyl and salicyl plane (deg) | 41.06 | 46.82 | 43.97 |
| Torsion angle (deg) | |||
| 5-t-Bu group | 61.5 | 63.29 | 126.86 |
| (C5−C6−C11−C13) | (C19−C20−C25−C27) | (C33−C34−C39−C42) | |
| −59.21 | −55.24 | 6.27 | |
| (C5−C6−C11−C14) | (C19−C20−C25−C28) | (C33−C34−C39−C40) | |
| 178.68 | −175.97 | −115.53 | |
| (C5−C6−C11−C12) | (C19−C20−C25−C26) | (C33−C34−C39−C41) | |
| 3-t-Bu group | 123.57 | 121.41 | 125.7 |
| (C5−C3−C7−C9) | (C19−C18−C21−C23) | (C33−C32−C35−C37) | |
| 3.95 | 1.53 | 6.05 | |
| (C5−C3−C7−C10) | (C19−C18−C21−C24) | (C33−C32−C35−C38) | |
| −115.50 | −117.63 | −112.10 | |
| (C5−C3−C7−C8) | (C19−C18−C21−C22) | (C33−C32−C35−C36) | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takanabe, A.; Katsufuji, T.; Johmoto, K.; Uekusa, H.; Shiro, M.; Koshima, H.; Asahi, T. Reversible Single-Crystal-to-Single-Crystal Phase Transition of Chiral Salicylidenephenylethylamine. Crystals 2017, 7, 7. https://doi.org/10.3390/cryst7010007
Takanabe A, Katsufuji T, Johmoto K, Uekusa H, Shiro M, Koshima H, Asahi T. Reversible Single-Crystal-to-Single-Crystal Phase Transition of Chiral Salicylidenephenylethylamine. Crystals. 2017; 7(1):7. https://doi.org/10.3390/cryst7010007
Chicago/Turabian StyleTakanabe, Akifumi, Takuro Katsufuji, Kohei Johmoto, Hidehiro Uekusa, Motoo Shiro, Hideko Koshima, and Toru Asahi. 2017. "Reversible Single-Crystal-to-Single-Crystal Phase Transition of Chiral Salicylidenephenylethylamine" Crystals 7, no. 1: 7. https://doi.org/10.3390/cryst7010007
APA StyleTakanabe, A., Katsufuji, T., Johmoto, K., Uekusa, H., Shiro, M., Koshima, H., & Asahi, T. (2017). Reversible Single-Crystal-to-Single-Crystal Phase Transition of Chiral Salicylidenephenylethylamine. Crystals, 7(1), 7. https://doi.org/10.3390/cryst7010007








