1. Introduction
Boron (Z = 5) is one of the lightest elements of the periodic table, coming just before carbon (Z = 6). It is also certainly the least well-known element with 2p valence shell. In comparison to the very popular carbon, it has not attracted as much attention (despite more than two centuries of use), but it has fascinated and exasperated generations of scientists. We, the contributors and editors of the present special issue, are proud to belong to the current generation, and we are equally enthralled but no longer exasperated.
Discovered in 1808, boron was isolated by Joseph-Louis Gay-Lussac (French chemist, 1778-1850) and Louis-Jacques Thénard (French chemist, 1777–1857), and independently by Sir Humphry Davy (English chemist, 1778–1829). The years that followed can be summarized by citing Grimes [
1]. Boron “
appeared normal—even boring—for a century following its isolation […]. It behaved exactly as expected, forming trivalent compounds […], and everyone […] believed that its simplest hydride had to be BH3 […]. It took the great German chemist Alfred Stock [(1876-1946)] to uncover the truth about the boron hydrides […]. Not until another half-century had passed did another towering scientist, William Nunn Lipscomb [(American chemist, 1919–2011)], finally crack the mystery of the three-center-bonded polyboranes”. Crystallinity of these polyboranes and the development of powerful characterization tools like X-ray diffraction within the 20th century, among other factors, have allowed the recent great advances in boron science.
Hosmane, in the preface of the book he edited, asks the question “
what is boron?” and right after gives the answer “
the question itself may not seem very significant to many people” [
2]. Boron is indeed little known. It may be confused with borax, which is the ore from which boron is extracted. This may appear to be unfair in comparison to the reputation of carbon, but obviously boron has shown to be versatile after two centuries of research and development. Let us again cite Hosmane [
2]. “
How many of us know that a regular intake of boron can lessen the chance of prostate cancer? How many of us know that boron plays a direct and critical role in combating cancer through a treatment called boron neutron capture therapy?” Medicine is clearly a highly important and crucial area of application. In fact, boron—especially boron-based compounds—have been widely used in many areas of application, including health (e.g., fungicides, detergents, soaps, antiseptics, and cosmetics), nutrition (e.g., additives, fertilizers), electronics, energy (e.g., nuclear energy, hydrogen storage, fuel cell), organic and inorganic chemistry, catalysis, metallurgy, enamels, and ceramics.
Accordingly, the present special issue focuses on fundamentals and applications of crystalline boron-based (nano-)materials, and aims at illustrating the recent great advances in boron science through several examples.
2. The Journal Crystals, an Already-Identified Forum for Boron-Based (Nano-)Materials
Boron scientists have not waited for the present special issue to report their cutting-edge research in this journal, Crystals. All of them are briefly cited hereafter, as they also illustrate the versatility of boron and the importance of crystalline boron-based (nano-)materials in several areas of application.
2.1. Attractive Physical Properties
Borophosphates are intermediate compounds made from the system M
xO
y−B
2O
3−P
2O
5−(H
2O), where M is a transition metal like Fe. They are generally used as glasses, but borophosphates with open frameworks have been shown to be of interest as magnetic materials. This is the case, for example, for the new KFe[BP
2O
8(OH)] reported by Wang and Mudring [
3]. It shows a three-dimensional framework crystallizing in the monoclinic structure with a space group
P2
1/
c; it is formed by alternating {BO
3(OH)} and {PO
4} tetrahedrons.
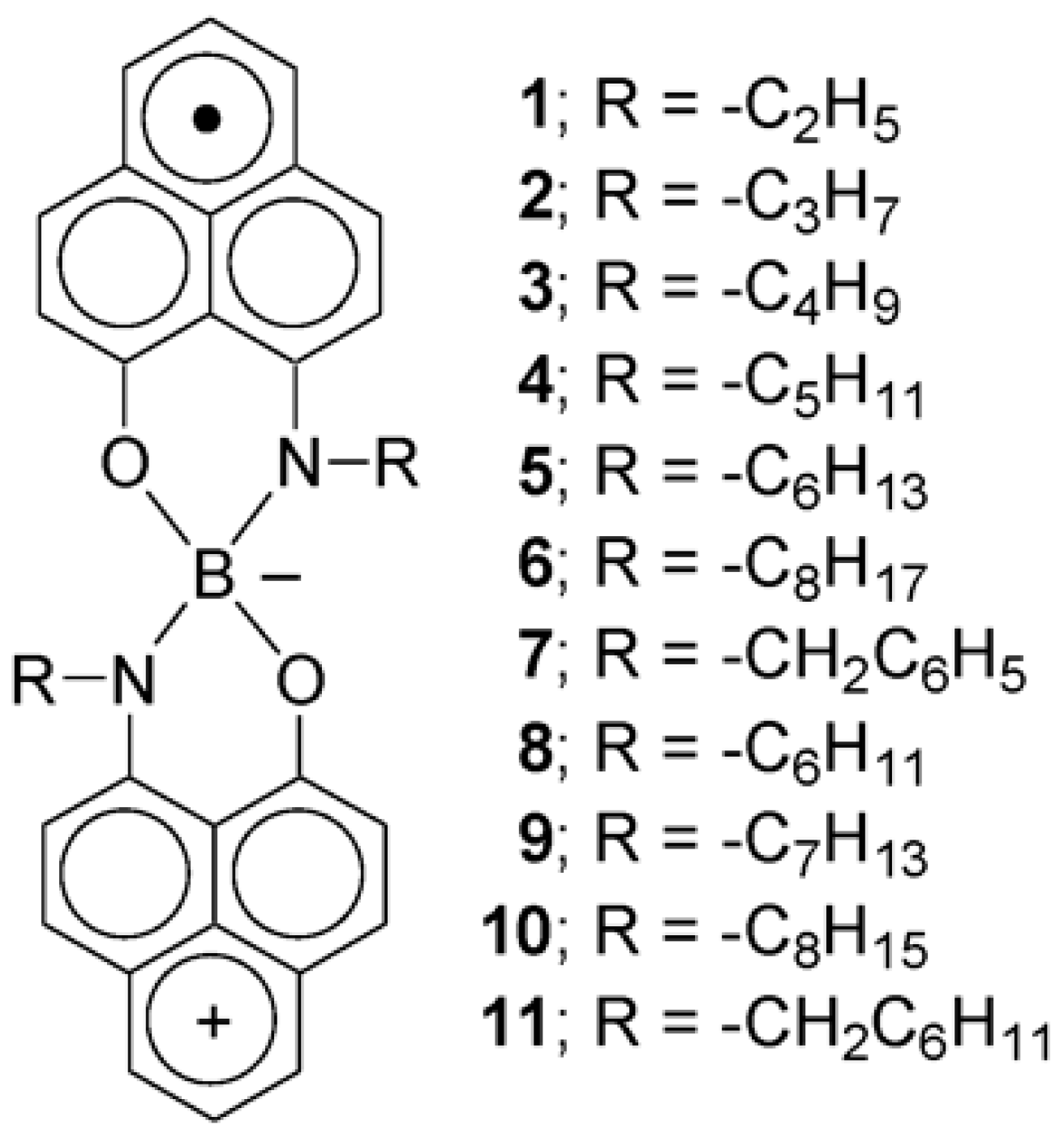
Another class of compounds that show particular magnetic properties is constituted of substituted spirobiphenalenyl boron radicals (
Figure 1). They are paramagnetic. Another of their physical properties interested Pag et al. [
4]. These radicals are indeed attractive molecular conductors for optoelectronics, magnetooptics, and spintronics.
Borates are mainly used as heat-resistant glass and insulator fiberglass, as detergent and soap, and as agricultural micronutrients. Other applications are also prospected as borates have interesting optical properties. Gallium borate GaBO
3 has notably been well studied with respect to luminescence properties. This was reported by Wang et al. [
5], who especially worked on the single crystal structure (trigonal calcite type, space group
R-3c) and the band gap (3.62 eV) of GaBO
3.
2.2. Attractive Chemical Properties
Boron hydrides (e.g., alkali borohydrides) are well-known as effective reducing agents. Sodium borohydride (NaBH
4) is certainly the most widely used in both organic and inorganic chemistry. Potassium borohydride (KBH
4) is an alternative. It was, for example, successfully used to reduce the oxide layer forming on the surface of the intermetallic material MlNi
3.6Co
0.85Al
0.3Mn
0.3 used as a fuel cell anode. This work was reported by Chen et al. [
6].
Alkali borohydrides are also of interest in the field of energy, especially as solid-state chemical hydrogen storage materials. For this application, lithium borohydride (LiBH
4) is more suitable than the sodium and potassium counterparts, owing to the lightness of lithium (M
w = 6.94 g/mol) and the highest gravimetric hydrogen density (18.5 wt % H). Destabilization of LiBH
4 has been widely investigated with the objective to decrease the dehydrogenation temperature, and Zavorotynska et al.’s contribution concerns the solid solution LiBH
4–LiCl [
7]. Liquid-state chemical hydrogen storage is also a possible application for alkali borohydrides, but NaBH
4 is better than the lithium and potassium counterparts in that case [
8]. Other candidates for chemical hydrogen storage are boranes, the simplest candidate being ammonia borane (NH
3BH
3). Sagan et al. [
9], for their part, considered derivatives with the following molecular structures: LiN(CH
3)
2BH
3 and KN(CH
3)
2BH
3.
The BH
3 entity is not stable enough to exist in an isolated form. It exists as a dimer, which is the diborane molecule B
2H
6. Otherwise, it can be stabilized by complexation with a Lewis base like ammonia NH
3 or tetrahydrofuran (CH
2)
4O, leading to the formation of a Lewis adduct. Accordingly, BH
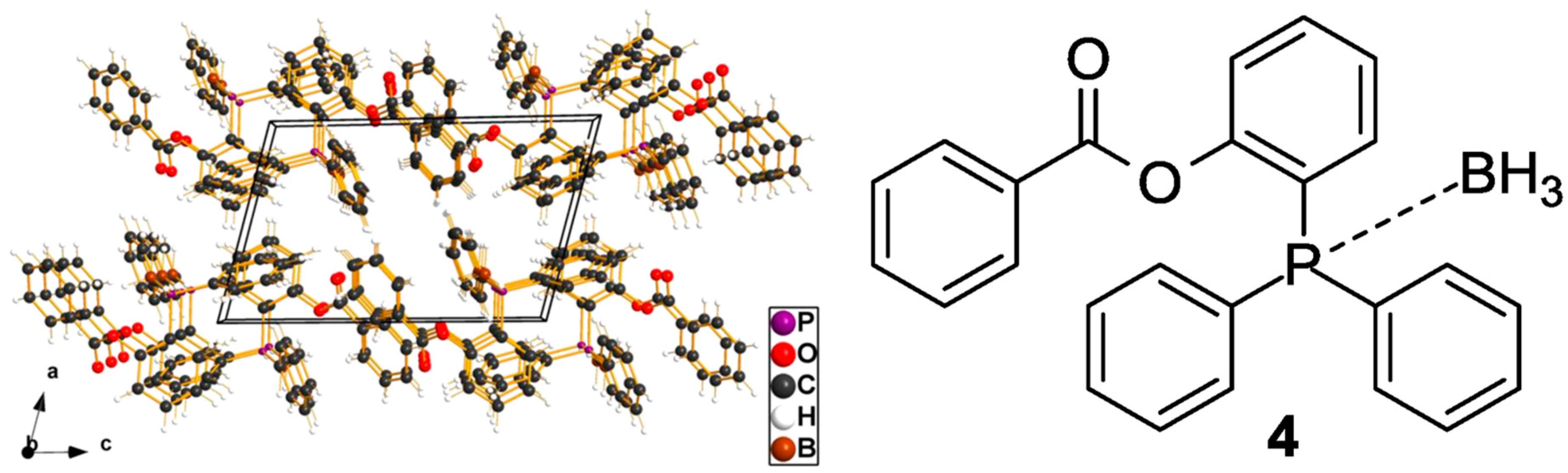
3 is of key importance in molecular (organic) chemistry, as it can be used as protecting group of a potential Lewis base. For example, one may cite the work reported by Mamat and Köckerling, who used BH
3 to protect a phosphane (
Figure 2) [
10]; another work belongs to Bourque et al. [
11]. Both NaBH
4 and a complex of BH
3 were used for the synthesis of a maltolato compound; the former was used as reducing agent, and the latter as Lewis acid.
3. The Journal Crystals, a Key Forum for the Present Special Issue
The present special issue is constituted of nine review and research papers of high quality, reporting different boron-based (nano-)materials for different possible areas of application. They are briefly discussed hereafter.
3.1. Boranes and Borohydrides for Solid-State Hydrogen Storage
Owarzany et al. [
12] present an overview on metal amidoboranes M(NH
2BH
3) with M = Li, Na, K, Rb, Cs, Mg, Ca, Sr, Y, Al, and Zn. Amidoboranes constitute a constantly growing family of NH
3BH
3 derivatives, especially investigated as solid-state chemical hydrogen storage materials. The authors propose the most exhaustive survey of the amidoborane reported so far. Their crystal structures are discussed first and in detail. Additional pieces of information are given based on nuclear magnetic resonance, infrared, and Raman spectra. The authors also scan the synthesis paths, while giving details about reactivity and selected properties, such as (among others) ionic conductivity, storage capacity, and interatomic distance. The conclusion of the article points out the issues that hinder the development and implementation of these chemical hydrogen storage materials.
We [
13] have also contributed to the special issue with an article dealing with the in situ Synchrotron X-ray diffraction of boranes. Pristine ammonia borane (NH
3BH
3), hydrazine borane (N
2H
4BH
3), and two hydrazinidoborane derivatives (MN
2H
3BH
3; M = Li, Na), hydrazine
bisborane (BH
3N
2H
4BH
3) and sodium triborane (NaB
3H
8) are analyzed. All are currently considered as potential chemical hydrogen storage materials. The boranes were investigated over a wide range of temperatures (80–300 K). Differences in crystal structures, the existence of phase transition, evolutions of unit cell parameters and volumes, and variation of coefficients of thermal expansion can be observed. The main results are presented and discussed.
Like Zavorotynska et al. [
7] (cf.
Section 2.2), Javadian et al. [
14] investigated LiBH
4. However, their approach is much different. They consider the destabilization of LiBH
4 via the combination of two approaches; that is, chemical doping with another hydride (sodium alanate, NaAlH
4) and nanoconfinement into a mesoporous carbon aerogel with high specific surface area (689 m
2/g). The reciprocal is also true, because one may consider the destabilization of NaAlH
4 by the presence of LiBH
4. In comparison to the pure and bulk parent materials, improved dehydrogenation and rehydrogenation properties are reported, making the authors positively conclude on the viability of the proposed approach.
3.2. Polyhedral Boranes and Carboranes
There are two review article contributions addressing boron-rich clusters, such as polyhedral boranes and carboranes.
The first is proposed by Planas et al. [
15]. The review summarizes the synthetic routes, coordination chemistry, and properties of a series of carboranyl ligands containing N, O donors (e.g.,
closo-carbonylmethylalcohols with nitrogenated aromatic rings) as well as of metal (e.g., cobalt, iron, platinum, titanium) complexes of these N,O-type carborane ligands. The wealth of carborane chemistry is particularly remarked. It is of even more importance that N,O-type carborane ligand-based complexes show a variety of properties, such as those used in magnetic, chiroptical, nonlinear optical, catalytic, and biomedical applications. It is worth citing the last sentence of the authors’ conclusion, as it is true for all of the articles of the present special issue: “
New developments are appearing constantly and are limited only by our imagination”.
The second review by Avdeeva et al. [
16] is substantial, with 85 pages about silver and copper complexes with
closo-polyhedral borane (10 or 12 boron atoms), carborane (1 carbon atom with 9 or 11 boron atoms) and metallacarborane (e.g., [3,3’-Co(1,2-C
2B
9H
11)
2]
−) anions (
Figure 3). Each of the complexes is reported in terms of synthesis and crystal structures. They demonstrate a wide variety of structural types, relating to both the metal coordination environment and coordination modes of boron hydride anions, which is discussed in detail by the authors. This review paper is of fundamental importance, with a major part dedicated to crystal structures and coordination chemistry.
3.3. Boronate Ligands and Derivatives
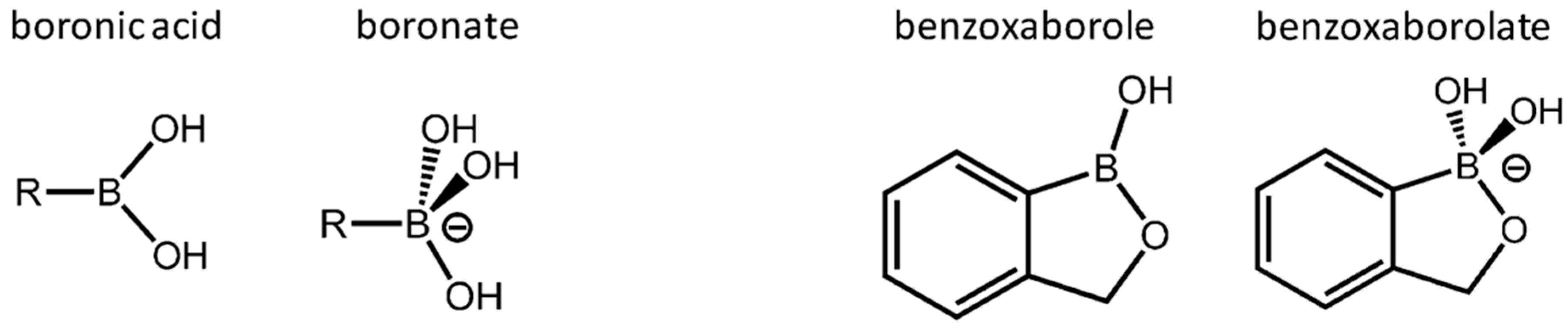
Sene et al.’s review [
17] is about some of the key results on crystalline structures with boronate and benzoxaborolate ligands (anions) that can be used as building blocks (
Figure 4). The authors emphasize the fact that such ligands “
had up until recently hardly been looked into as possible building blocks for materials applications”. With respect to these mentioned “
materials applications”, the authors have in mind coordination networks as well as metal organic networks (MOFs). The article is structured around crystal structures involving simple boronates and benzoxaborolates, spectroscopic signatures in materials, and finally emerging applications like functional coordination polymers/networks with temperature-dependent luminescent properties or promising magnetic properties and benzoxaborole drugs (nanomedicine).
3.4. Boron Nitride
Boron nitride is a non-oxide ceramic material of high interest. A first illustration of this is provided by Matarin and Rimola [
18]. By using computational methods (B3LYP-D2* periodic simulations), they investigated the adsorption of molecules like water (H
2O), ammonia (NH
3), formic acid (HCOOH), benzene (C
6H
6), and methane (CH
4) onto boron nitride nanotubes showing defects derived from monovacancies of boron and nitrogen atoms. Distinction is made between the aforementioned polar (H
2O, NH
3, HCOOH) and non-polar (C
6H
6, CH
4) molecules. It is shown that nitrogen-rich boron nitride nanotubes are more reactive towards the adsorption of polar molecules (dictated by dative interactions), whereas the adsorption of non-polar molecules (physisorption governed by, e.g., π-stacking for C
6H
6) does not depend on the type of the nanotube. It is therefore concluded that adsorption onto boron nitride nanotubes may be modulated by the presence of surface defects.
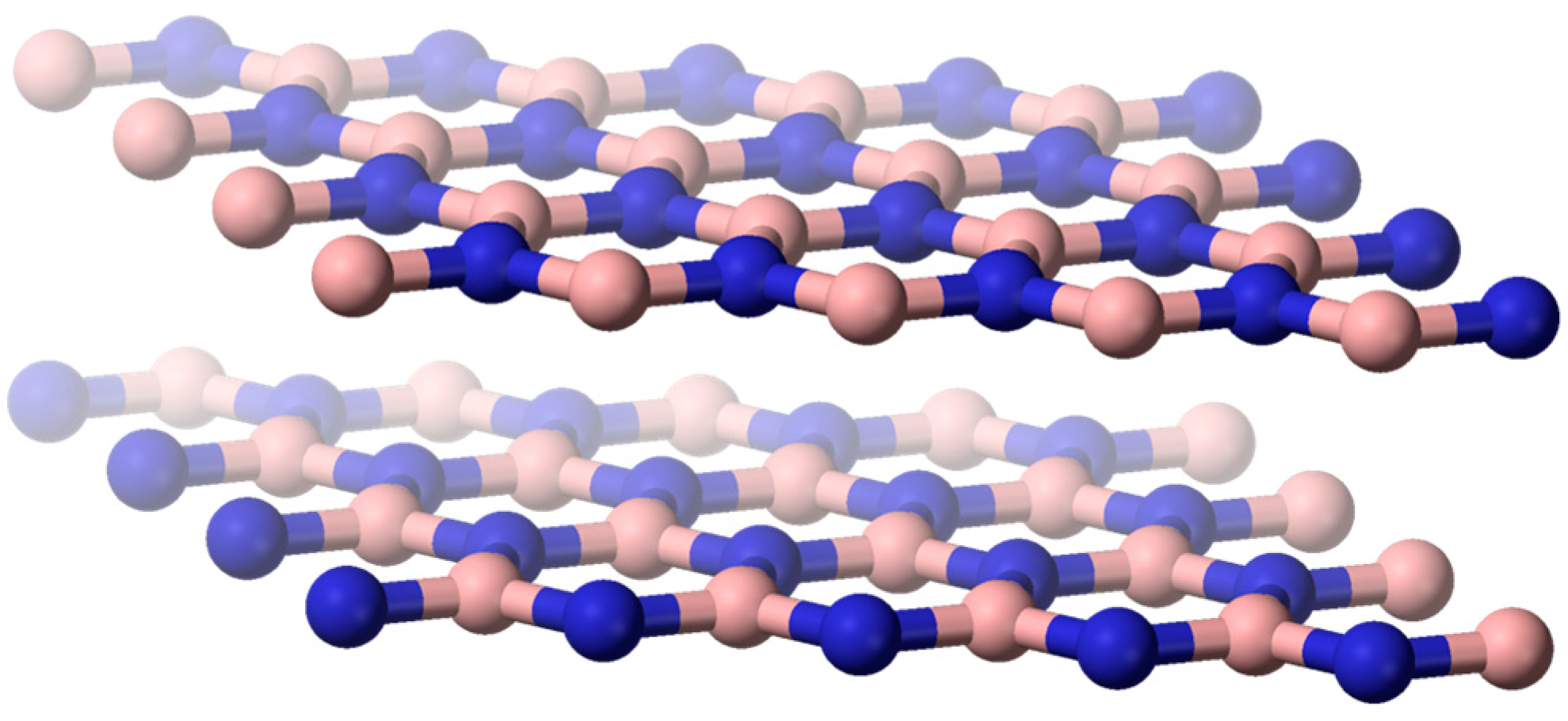
Hexagonal boron nitride (h-BN;
Figure 5) is principally used as lubricant in domains ranging from metallurgy to cosmetics. According to Yuan et al. [
19], h-BN has other properties that may open new perspectives for engineering applications, for example, as a solid lubricant in aeronautics or as a perfect substrate to graphene for electronic devices. Such promising developments require tailored h-BN shapes displaying a high level of crystallization, ensuring its properties for the long term. This article aims at giving an overview of the strategies (rapid thermal annealing, Li
3N as crystallization promoter, and spark plasma sintering) developed by the authors to prepare highly crystallized supported thick coatings and self-standing nanosheets. The advantages and limitations of the three strategies, as well as the main achievements, are reported and discussed. As a concluding remark, the authors optimistically expect the development of h-BN-based high performance devices in the future.
It is worth mentioning that boron nitride is the featured molecule of a special issue of a sister journal; that is,
Molecules MDPI [
20]. The issue title is “boron nitride: synthesis and application”, and is co-edited by one of the present co-editors, Philippe MIELE. This special issue is dedicated to the most recent development on the synthesis and applications of boron nitride, including synthesis of high-quality crystal, nanopowders, nanosheets, nanostructured and porous ceramic materials, high-performance composites, and optoelectronic and electronic devices.
3.5. Boron-Tread Steels
The last paper of the series falls in a different field. Through a research paper, Gao et al. [
21] give an overview of the use of elemental boron as an effective alloying element employed to increase the hardenability of high strength low alloy steels. They particularly investigated the influence of boron on initial austenite grain size and hot deformation behavior of boron microalloyed steels. Among the various findings, the following one may be cited. Increasing the boron content of boron-tread steel increases the peak and critical strain of dynamic recrystallization, indicating that boron addition can delay the onset of dynamic recrystallization. We believe that the present work will give the reader a wider view of the application prospects of boron and related boron-based materials.
4. Conclusions
The present special issue gives an overview of the current works dedicated to crystalline boron-based (nano-)materials. Though the original review and research articles dedicate large sections to crystallinity and crystal structures, the authors also give details about various aspects, such as synthesis, spectroscopic signatures, physicochemical properties, and application prospects. With respect to this last aspect, advanced technologies, such as energy storage, gas sorption (energy and environment), biomedicine and nanomedicine, and optical and electronic devices are targeted. We cordially invite the reader curious about science, and especially boron science, to dip into each of the nine articles of the present special issue.
In conclusion, we would like to thank all of the authors who have contributed to this special issue. We again thank the main/correspondence authors who accepted our invitation several months ago. The present special issue is an important scientific contribution to the field of boron science, and we, the contributors, have all been actors of this successful publication. “
New developments are appearing constantly and are limited only by our imagination” [
15].
We wish all of the readers compelling reading.
Acknowledgments
We acknowledge all of the students (undergraduate, graduate, PhD) and post-doctorate fellows who worked or are working on boron science under our supervision.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Grimes, R.N. Foreword. In Boron Science—New Technologies and Applications; Hosmane, N.S., Ed.; CRC Press: Boca Raton, FL, USA, 2012; pp. xi–xii. [Google Scholar]
- Hosmane, N.S. Preface. In Boron Science—New Technologies and Applications; Hosmane, N.S., Ed.; CRC Press: Boca Raton, FL, USA, 2012; pp. xiii–xiv. [Google Scholar]
- Wang, G.; Mudring, A.V. A New Open-Framework Iron Borophosphate from Ionic Liquids: KFe[BP2O8(OH)]. Crystals 2011, 1, 22–27. [Google Scholar] [CrossRef]
- Pag, P.; Itkis, M.E.; Pal, S.K.; Bekyarova, E.; Donnadieu, B.; Haddon, R.C. Synthesis, Structure and Solid State Properties of Cyclohexanemethylamine Substituted Phenalenyl Based Molecular Conductor. Crystals 2012, 2, 446–465. [Google Scholar]
- Wang, S.; Ye, N.; Poeppelmeier, K.R. Flux Growth and Crystal Structure Refinement of Calcite Type Borate GaBO3. Crystals 2015, 5, 252–260. [Google Scholar] [CrossRef]
- Chen, Y.; Santos, D.M.F.; Sequeira, C.A.C.; Lobo, R.F.M. Studies of Modified Hydrogen Storage Intermetallic Compounds Used as Fuel Cell Anodes. Crystals 2012, 2, 22–33. [Google Scholar] [CrossRef]
- Zavorotynska, O.; Corno, M.; Pinatel, E.; Rude, L.H.; Ugliengo, P.; Jensen, T.R.; Baricco, M. Theoretical and Experimental Study of LiBH4-LiCl Solid Solution. Crystals 2012, 2, 144–148. [Google Scholar]
- Anderson, S.P.; Kalu, E.E. Electroless Nickel-Based Catalyst for Diffusion Limited Hydrogen Generation through Hydrolysis of Borohydride. Crystals 2013, 3, 405–418. [Google Scholar] [CrossRef]
- Sgan, F.; Filas, R.; Mitoraj, M.P. Non-Covalent Interactions in Hydrogen Storage Materials LiN(CH3)2BH3 and KN(CH3)2BH3. Crystals 2016, 6, 28. [Google Scholar] [CrossRef]
- Mamat, C.; Köckerling, M. Synthesis and Molecular Structure of 2-(Diphenylphosphano)phenyl Benzoate Borane Adduct. Crystals 2015, 5, 9–13. [Google Scholar] [CrossRef]
- Bourque, J.L.; Geier, S.J.; Vogels, C.M.; Decken, A.; Wescott, S.A. Synthesis and Reactivity of Novel Boranes based from Bulky Salicylaldimines: the Molecular Structure of a Moltato Compound. Crystals 2015, 5, 91–99. [Google Scholar] [CrossRef]
- Owarzany, R.; Leszczynski, P.J.; Fijalkowski, K.J.; Grochala, W. Mono- and Bimetallic Amidoboranes. Crystals 2016, 6, 88. [Google Scholar] [CrossRef]
- Yot, P.G.; Miele, P.; Demirci, U.B. In situ Synchrotron X-ray Thermodiffraction of Boranes. Crystals 2016, 6, 16. [Google Scholar] [CrossRef]
- Javadian, P.; Sheppard, D.A.; Buckley, C.E.; Jensen, T.R. Hydrogen Desorption Properties of Bulk and Nanoconfined LiBH4-NaAlH4. Crystals 2016, 6, 70. [Google Scholar] [CrossRef]
- Planas, J.G.; Teixidor, F.; Viñas, C. N,O-Type Carborane-Based Materials. Crystals 2016, 5, 50. [Google Scholar] [CrossRef]
- Avdeeva, V.V.; Malinina, E.A.; Sivaev, I.B.; Bregadze, V.I.; Kuznetsov, N.T. Silver and Copper Complexes with closo-Polyhedral Borane, Carborane and Metallacarborane Anions: Synthesis and X-ray Structure. Crystals 2016, 6, 60. [Google Scholar] [CrossRef]
- Sene, S.; Pizzoccaro, M.A.; Vezzani, J.; Reinholdt, M.; Gaveau, P.; Berthomieu, D.; Bégu, S.; Gervais, C.; Bonhomme, C.; Renaudin, G.; et al. Coordination Networks Based on Boronate and Benzoxaborolate Ligands. Crystals 2016, 6, 48. [Google Scholar] [CrossRef]
- Matarin, O.; Rimola, A. Influence of Defects in Boron Nitride Nanotubes in the Adsorption of Molecules. Insights from B3LYP-D2* Periodic Simulations. Crystals 2016, 6, 63. [Google Scholar] [CrossRef]
- Yuan, S.; Journet, C.; Linas, S.; Garnier, V.; Steyer, P.; Benayoun, S.; Brioude, A.; Toury, B. How to Increase the h-BN Crystallinity of Microfilms and Self-Standing Nanosheets: A Review of the Different Strategies Using the PDCs Route. Crystals 2016, 6, 55. [Google Scholar] [CrossRef]
- Special Issue “Boron Nitride: Synthesis and Application”. Available online: https://www.mdpi.com/journal/molecules/special_issues/Boron_Nitride (accessed on 25 August 2016).
- Gao, Y.L.; Xue, X.X.; Yang, H. Influence of Boron on Initial Austenite Grain Size and Hot Deformation Behavior of Boron Microalloyed Steels. Crystals 2015, 5, 592–607. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).