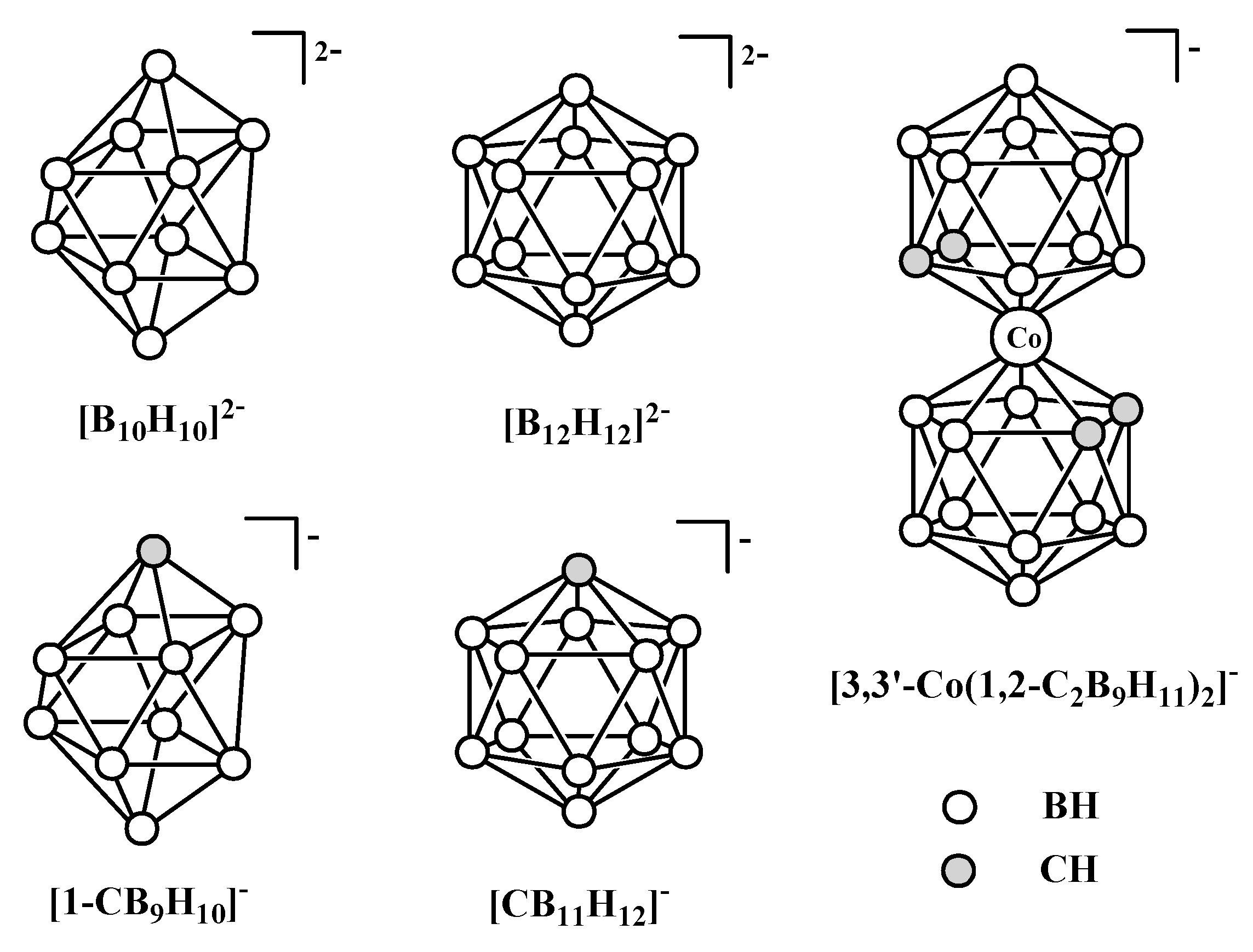
2. Copper and Silver Complexes with the closo-Decaborate anion [B10H10]2−
The
closo-decaborate anion [B
10H
10]
2− was the first representative of the
closo-borane family, which was reported by Hawthorne and Pitochelli in 1959 [
15]. The
closo-decaborate anion has structure of bicapped square antiprism with two different types of boron atoms (five-coordinated apical (B
a) and six-coordinated equatorial (B
e)) that makes some aspects of its chemistry unique and having no analogues in the chemistry of icosahedral boron hydrides. One of these peculiarities is the possibility to form various coordination isomers. Despite rather rapid development of its chemistry [
16,
17], the coordination chemistry of the
closo-decaborate anion was poorly understood until the last fifteen years, when a number of studies which cast light on this subject were published.
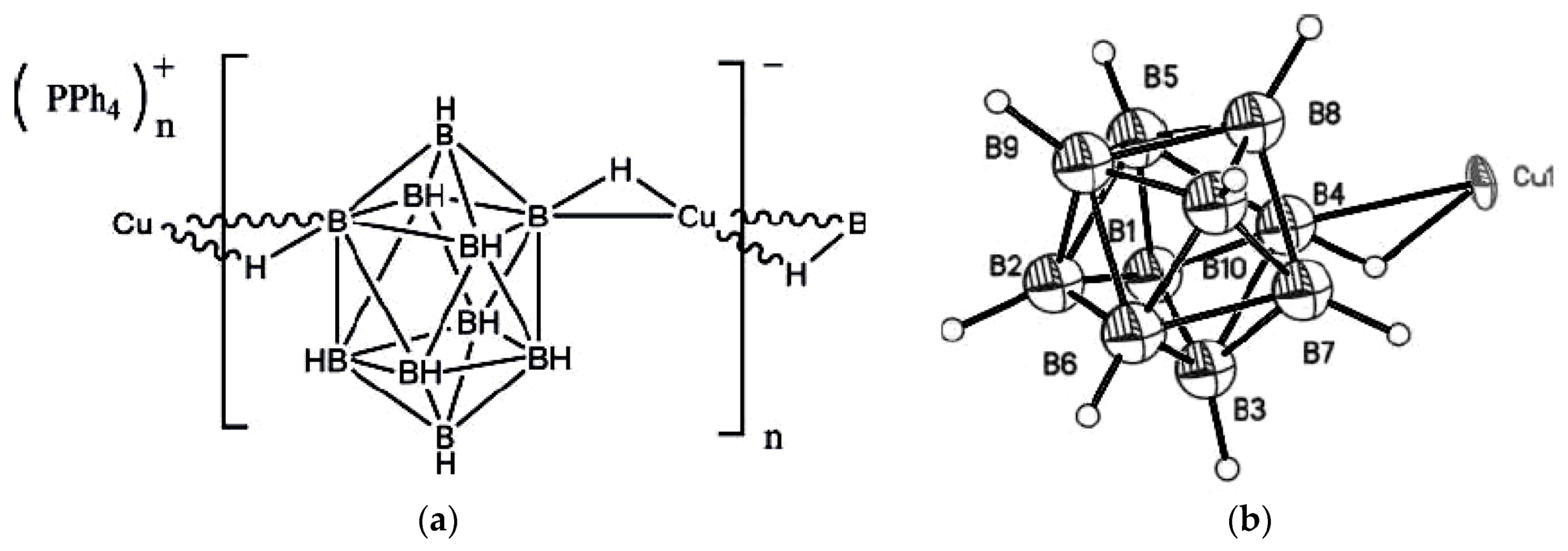
Copper(I)
closo-decaborate {Cu
2[B
10H
10]}
n was the first crystallographically characterized salt with the
closo-decaborate anion [
18]. The complex was prepared by reaction of K
2[B
10H
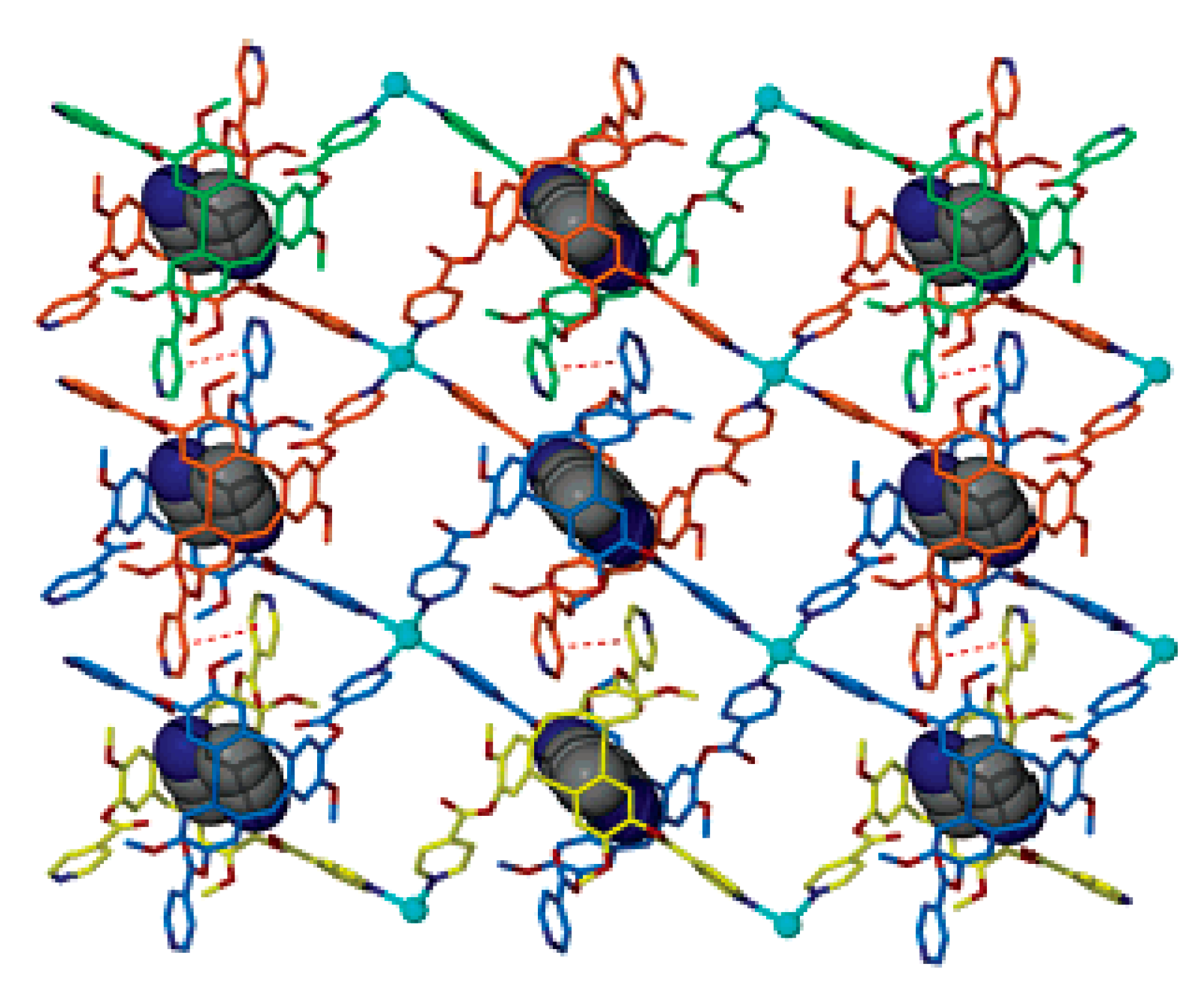
10] with copper(II) acetate in water. According to single crystal X-ray diffraction data, copper atoms are arranged near the centers of opposite polyhedral faces providing each boron cage to be surrounded by four copper atoms with the Cu…B distances varying from 2.06 to 2.33 Å (
Figure 1) [
18]. Based on the X-ray data, the formation of three-centered two-electron Cu-H-B bonds between copper and the boron cage was suggested. This suggestion was supported by the IR spectrum wherein two absorption bands in the B-H stretching region are observed: one of them (with maximum near 2450 cm
−1) was assigned to non-coordinated BH groups and the other (with maximum at 2300 cm
−1) was assigned to the BH groups involved in the Cu-H-B interactions [
19].
The similar silver(I) salt Ag
2[B
10H
10] can be prepared by metathetical reactions in aqueous solution [
20,
21]. Its structure was not determined, however the existence of Ag-H-B interactions in the solid state was supported by the IR spectroscopy data [
21].
In addition to the three-dimensional polymeric binary complexes {M
2[B
10H
10]}, the
closo-decaborate anion is able to form one-dimensional chain polymers of the Cat{M[B
10H
10]} type both with Cu
+ and Ag
+ cations. The complexes are formed at equimolar amounts of Cat
2[B
10H
10] and copper(II) and silver(I) salts, whereas in the case of two-fold excess of metal salts or in the presence of small cations (e.g., NH
4+) formation of binary complexes {M
2[B
10H
10]} takes place. It is worth noting that copper(I) complexes Cat{Cu[B
10H
10]} are formed from copper(II) salts due to reductive properties of the decahydro-
closo-decaborate anion. As a result, copper(I) complexes Cat{Cu[B
10H
10]} (where Cat
+ = K
+, Cs
+, R
2NH
2+, R
3NH
+, R
4N
+ (R = Me, Et, Bu), Ph
4P
+, NaphCH
2PPh
3+, Ph
4As
+) [
22,
23,
24,
25] and silver(I) complexes Cat{Ag[B
10H
10]} (where Cat = Cs
+, R
2NH
2+, R
3NH
+, R
4N
+ (R = Me, Et, Pr, Bu)) [
21] were prepared. IR spectra of all these compounds demonstrate characteristic bonds of M-H-B stretching at 2150–2400 cm
−1 due to interactions of Cu
+ and Ag
+ cations with the
closo-decaborate anion.
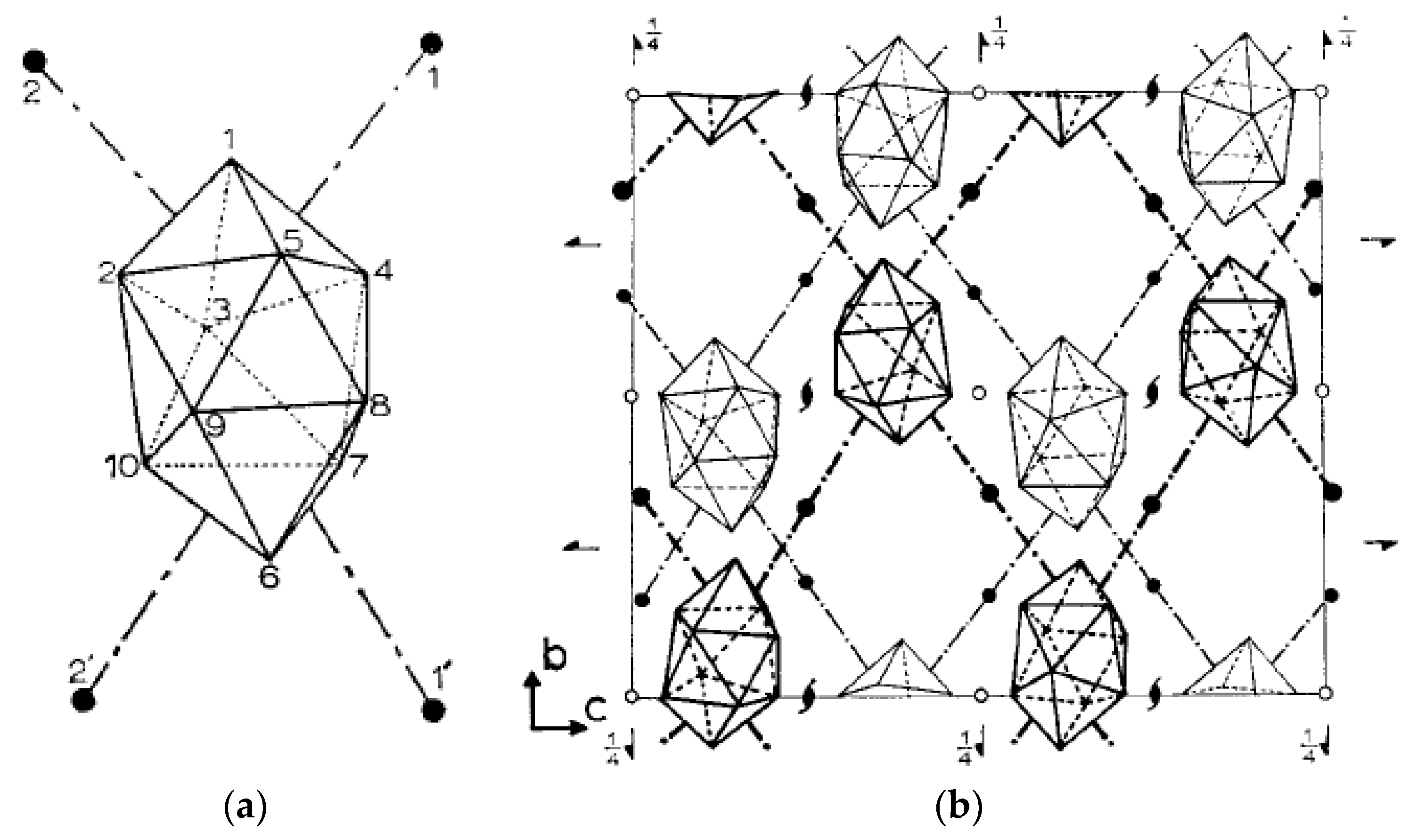
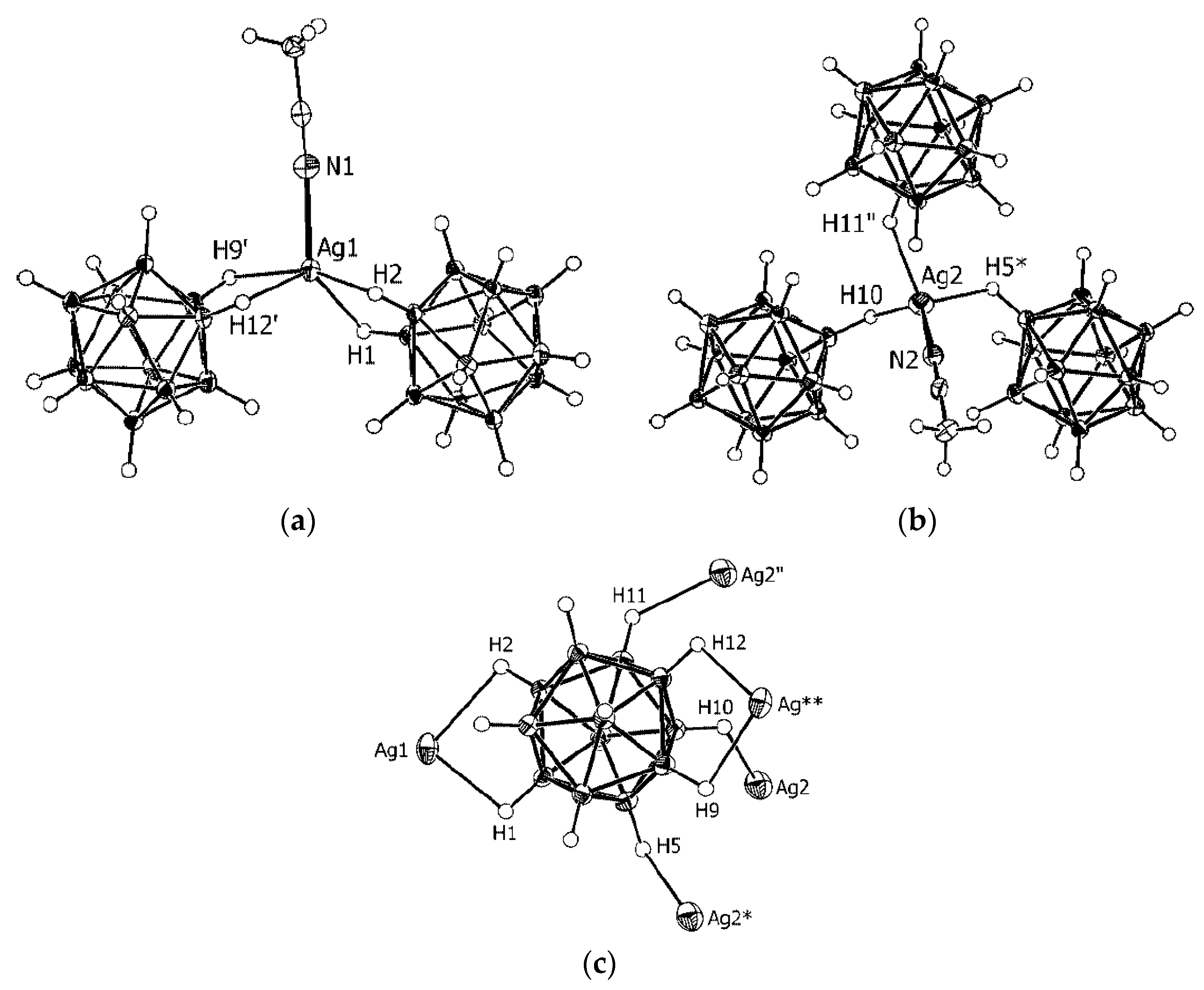
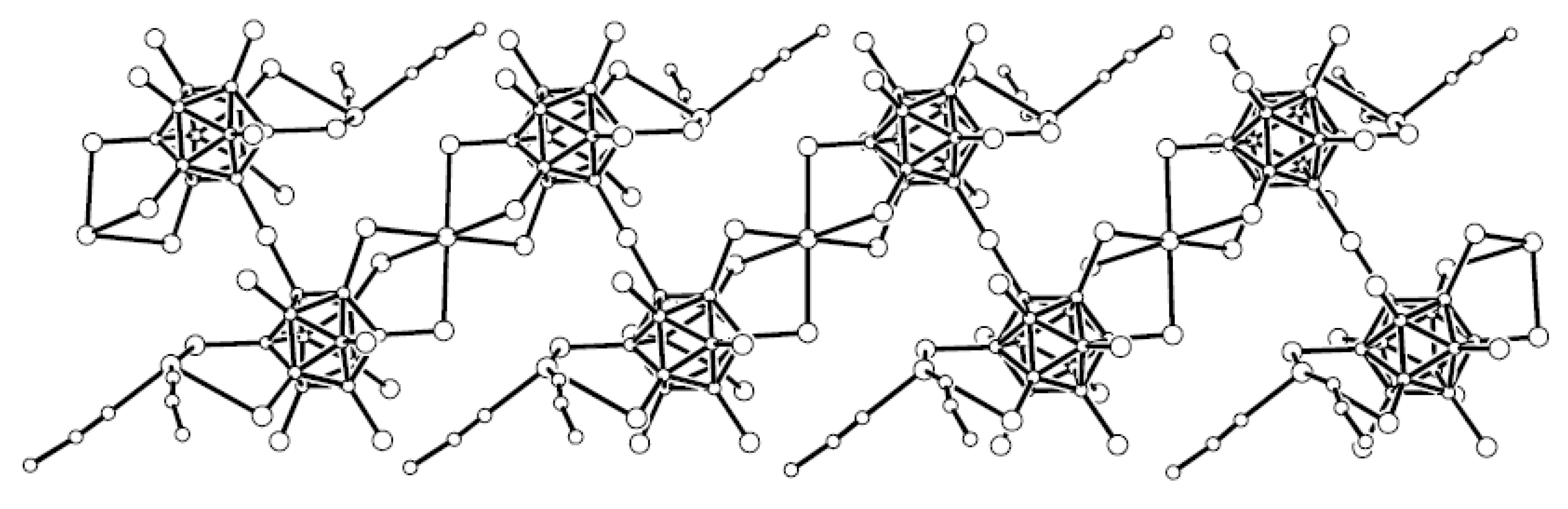
In the copper(I) complex {Cs(Cu[B
10H
10])}
n the Cu
+ cation is in distorted tetrahedral arrangement formed by two bridging
closo-decaborate anions which are coordinated through B
a-B
e and B
e-B
e’ edges. The Cu…B
a distances (2.159(6) Å) are significantly shorter than the Cu…B
e distances (2.228–2.287(6) Å). The binding of Cu
+ by
closo-decaborate edges of different types results in formation of zigzag chains running along the
c axis (
Figure 2a) [
25,
26]. In the silver(I) complex {Cs(Ag[B
10H
10])}
n the
closo-decaborate anion is coordinated to copper cations through two B
a-B
a edges with a shared vertex (Ag…B distances of 2.645(4) and 2.593(6) Å) to form zigzag chains. The chains are connected into layers owing to additional contacts with BH groups of the neighboring [B
10H
10]
2− anions (Ag…B distances of 3.183(6) and 3.337(6) Å) (
Figure 2b) [
21].
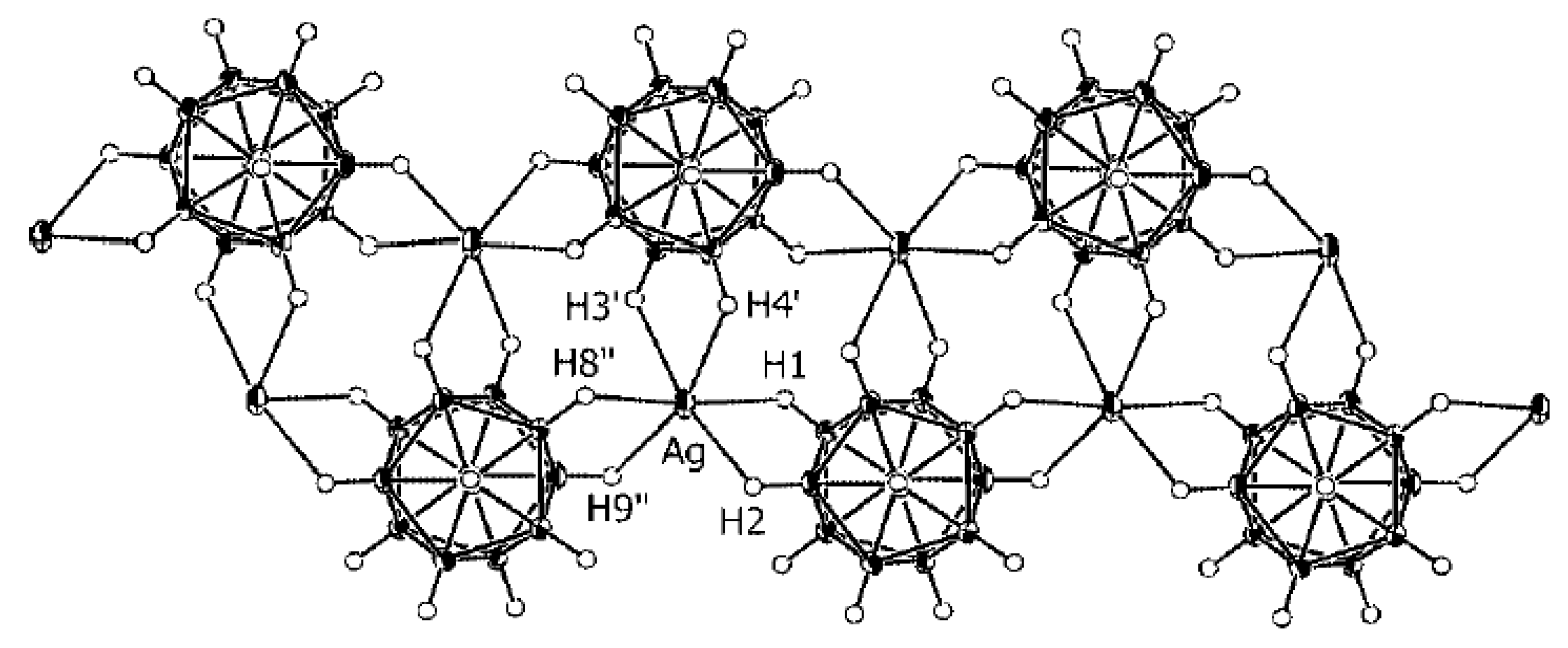
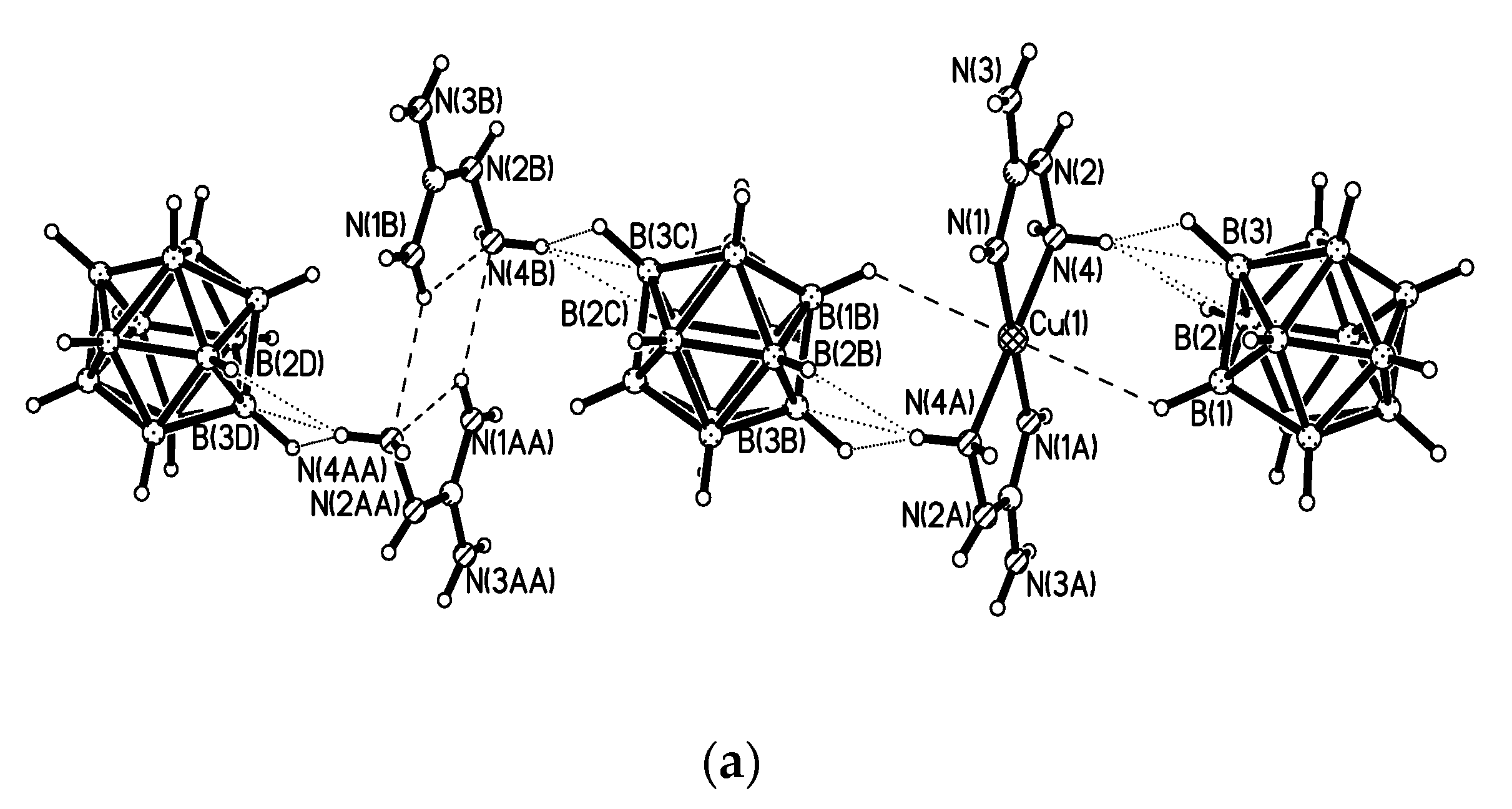
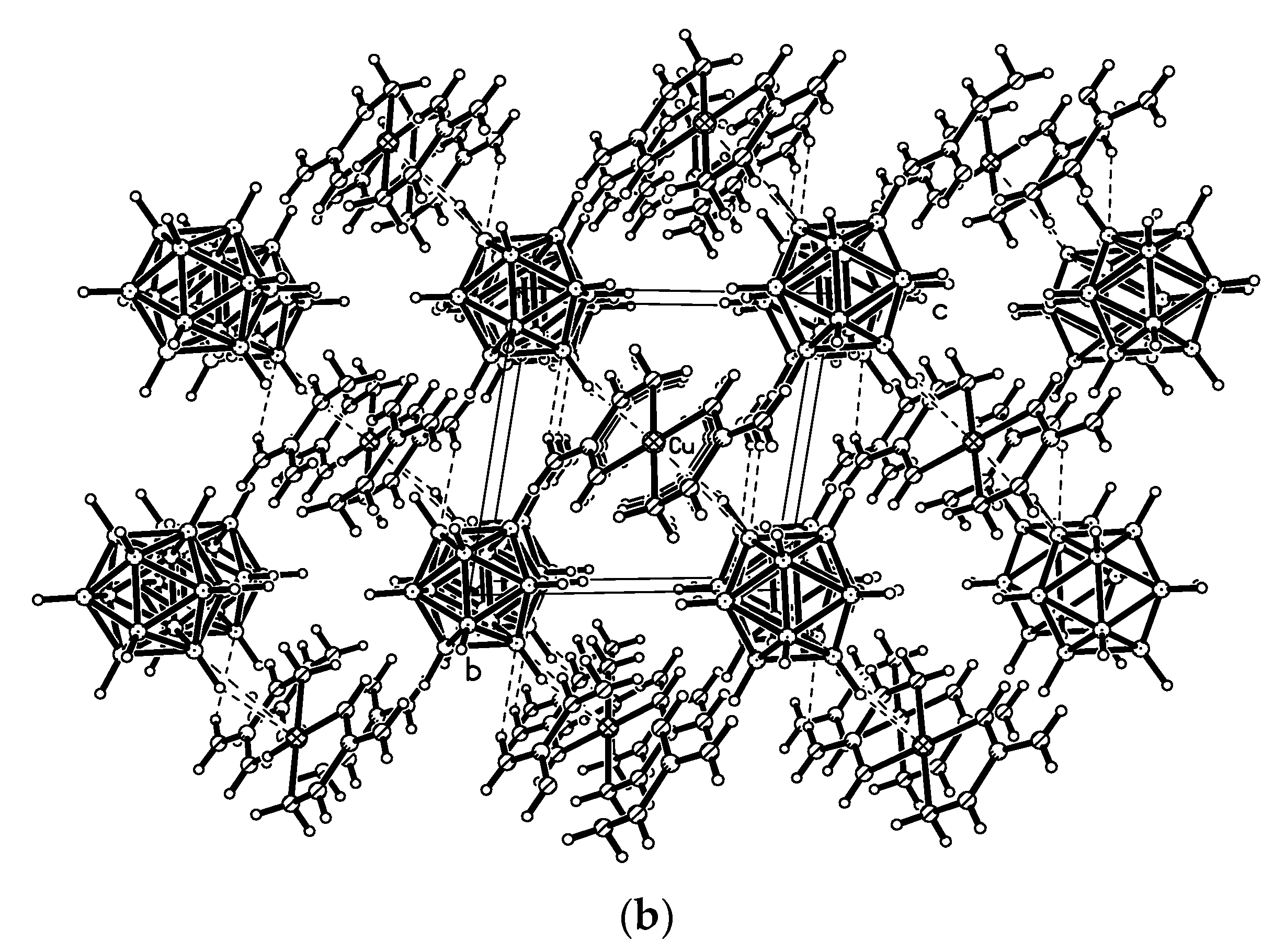
In the structure of {Me
2NH
2(Cu[B
10H
10])}
n [
25,
26], the copper cation has a distorted tetrahedral environment formed by two bridging
closo-decaborate anions which are coordinated through B
a-B
e edges resulting in the formation of straight {(Cu[B
10H
10])
−}
n chains that run along the
b axis. In both independent chains, the polyhedra that coordinate the Cu atoms by the B(1)-B(2) and B(7)-B(10) edges alternate with those coordinating Cu atoms by the B(1)-B(2) and B(8)-B(10) edges. The Cu…B
a and Cu…B
e bond lengths substantially overlap (2.145–2.285(9) and 2.130–2.268(9) Å, respectively). The symmetrically related chains are packed into layers parallel to the
bc plane. The layers alternate along the
a axis. The [Me
2NH
2]
+ cations are located between the anionic layers and form the NH…HB dihydrogen bonds with the polyhedral anions (
Figure 3) [
26].
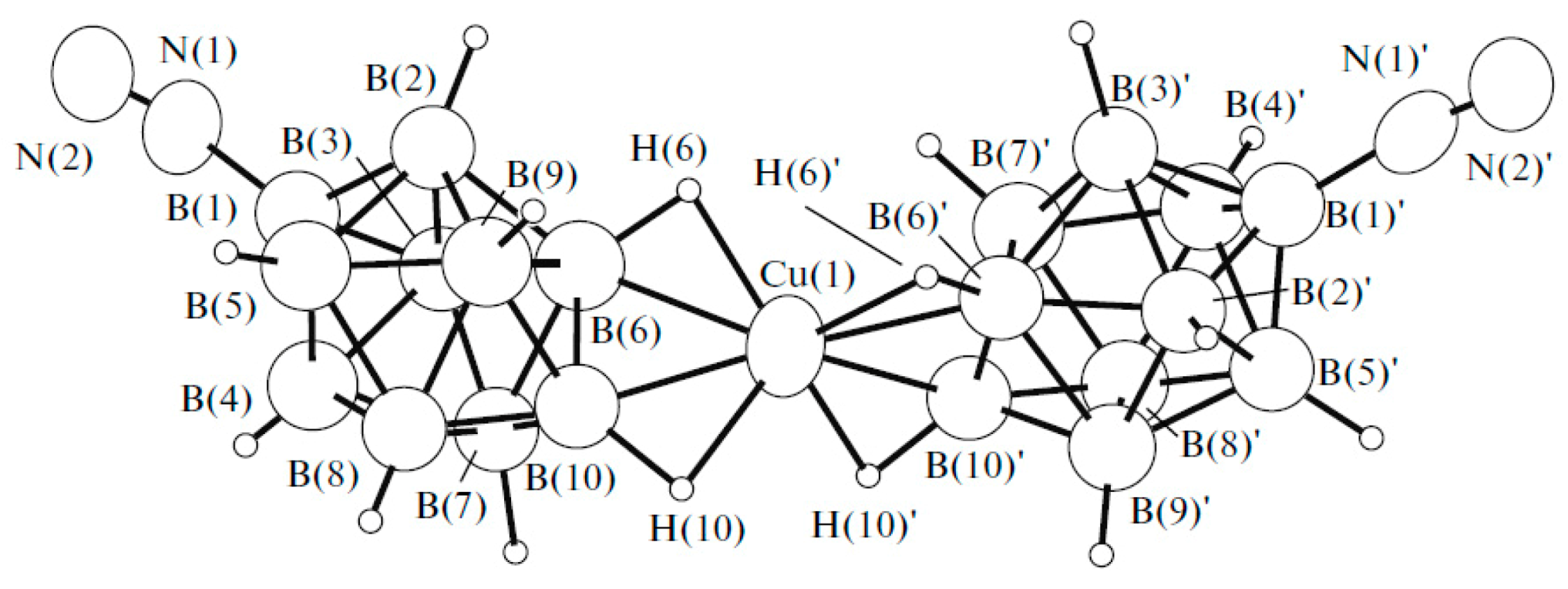
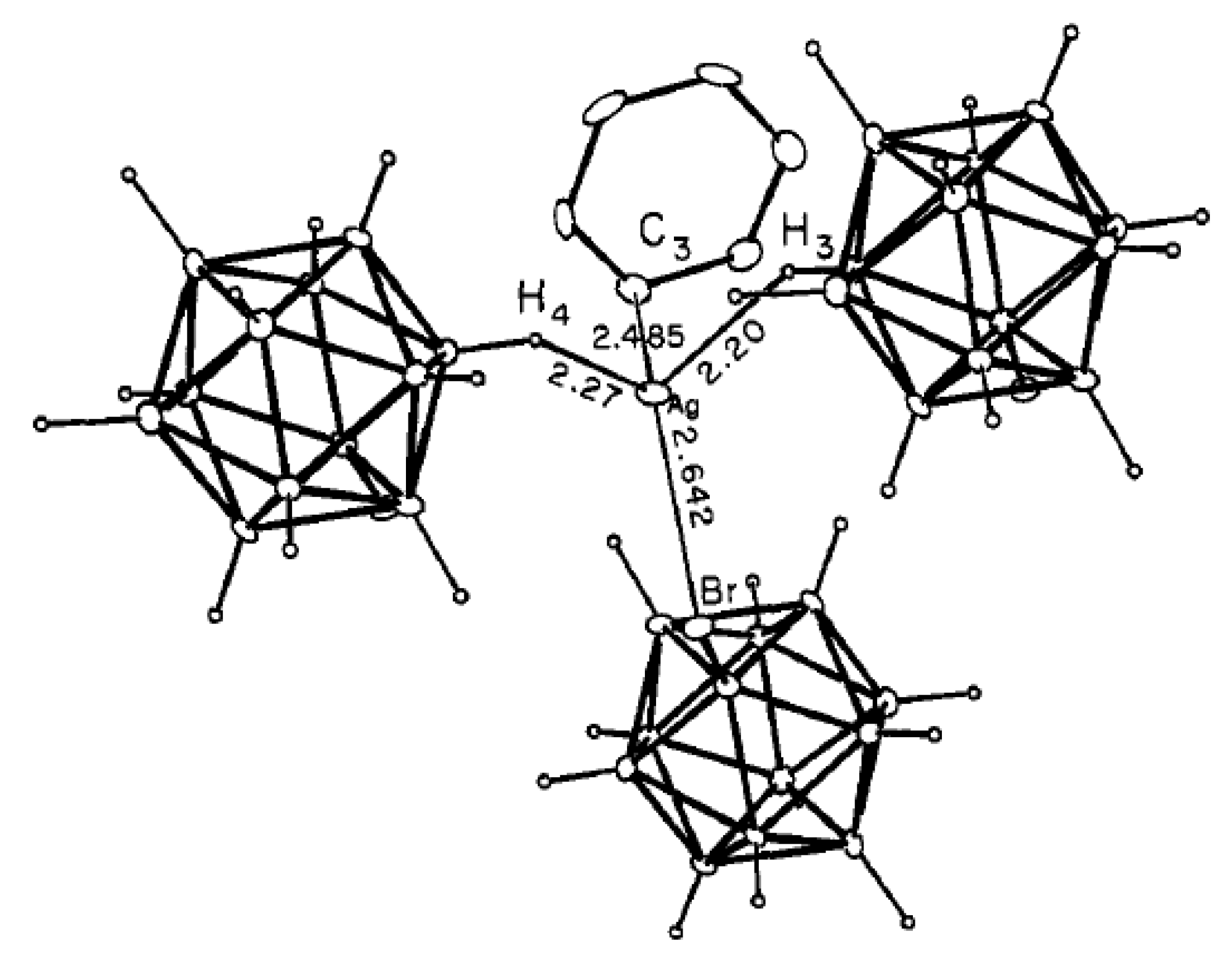
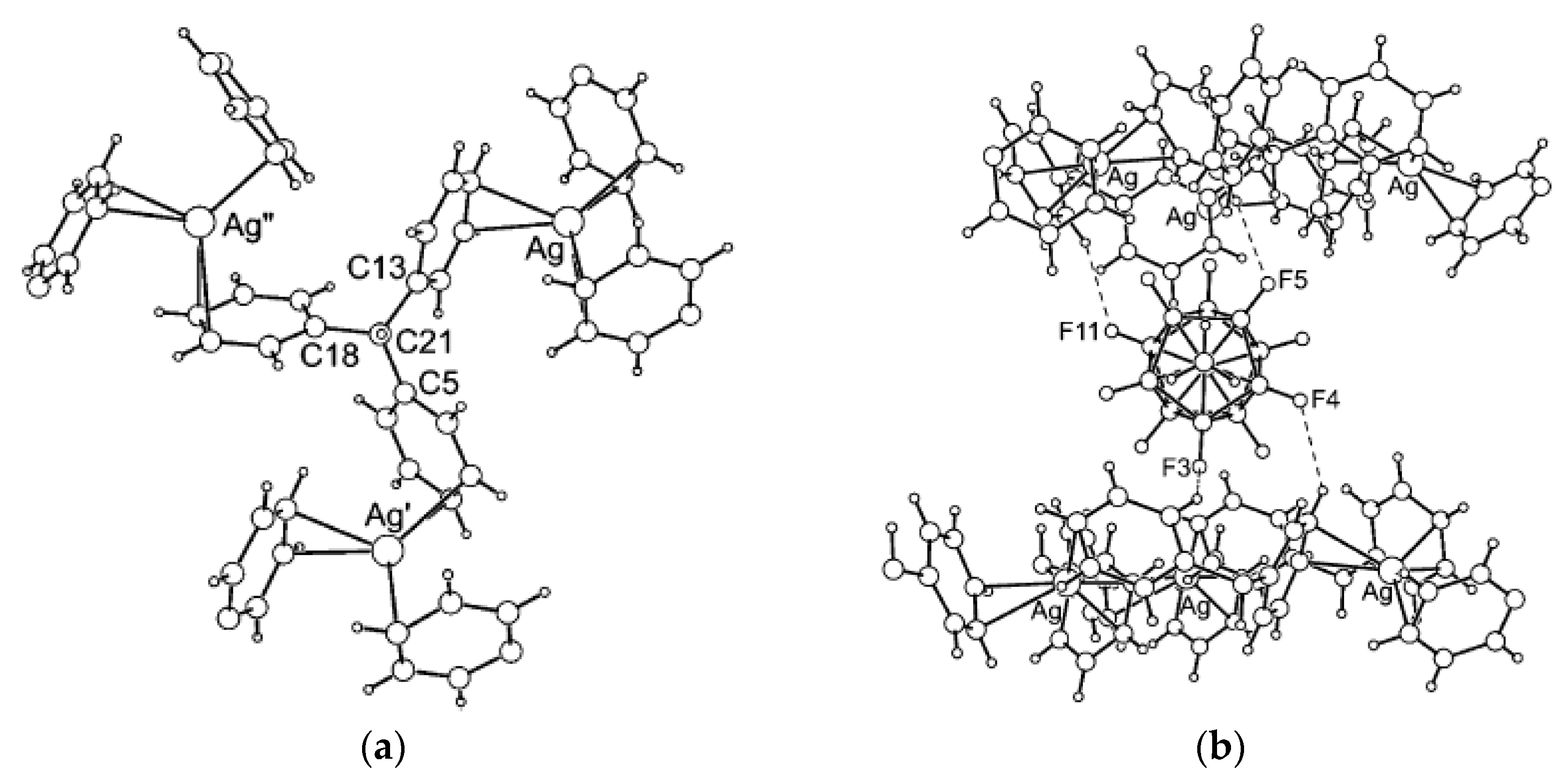
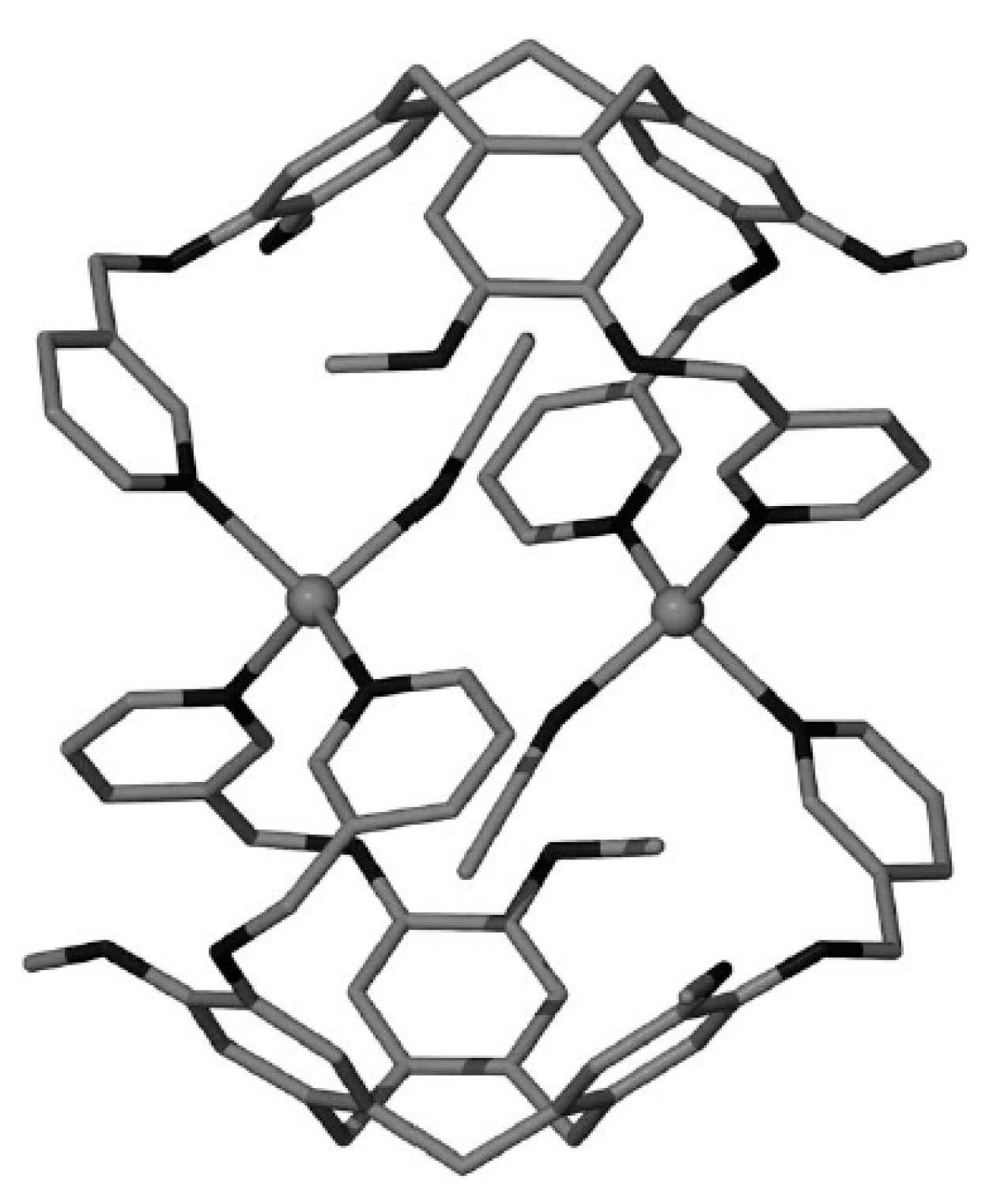
Comparison of structures of {(Et
3NH)(M[B
10H
10])}
n (M = Cu [
22] or Ag [
21]) revealed that different polymeric chains are formed in these two complexes. In the copper(I) complex, the polymeric chain is formed by coordination of the boron cage to a copper atom via B
a-B
e and B
e-B
e’ edges (Cu…B
a 2.172 Å, Cu…B
e 2.213(4)–2.234(4) Å) (
Figure 4а). The Et
3NH
+ cations form the NH···HB dihydrogen bonds with the polyhedral anions (NH…HB 2.20(6)–2.92(6) Å). In the silver(I) complex the metal atom is coordinated by two
closo-decaborate anions via B
a-B
e edges and one
closo-decaborate anion via B
e-B
e’ edge (Ag…B 2.561(5)–2.907(5) Å) (
Figure 4b). Similar to the copper(I) complex, the Et
3NH
+ cations in {(Et
3NH)(Ag[B
10H
10])}
n form the NH…HB dihydrogen bonds with
closo-decaborate anions.
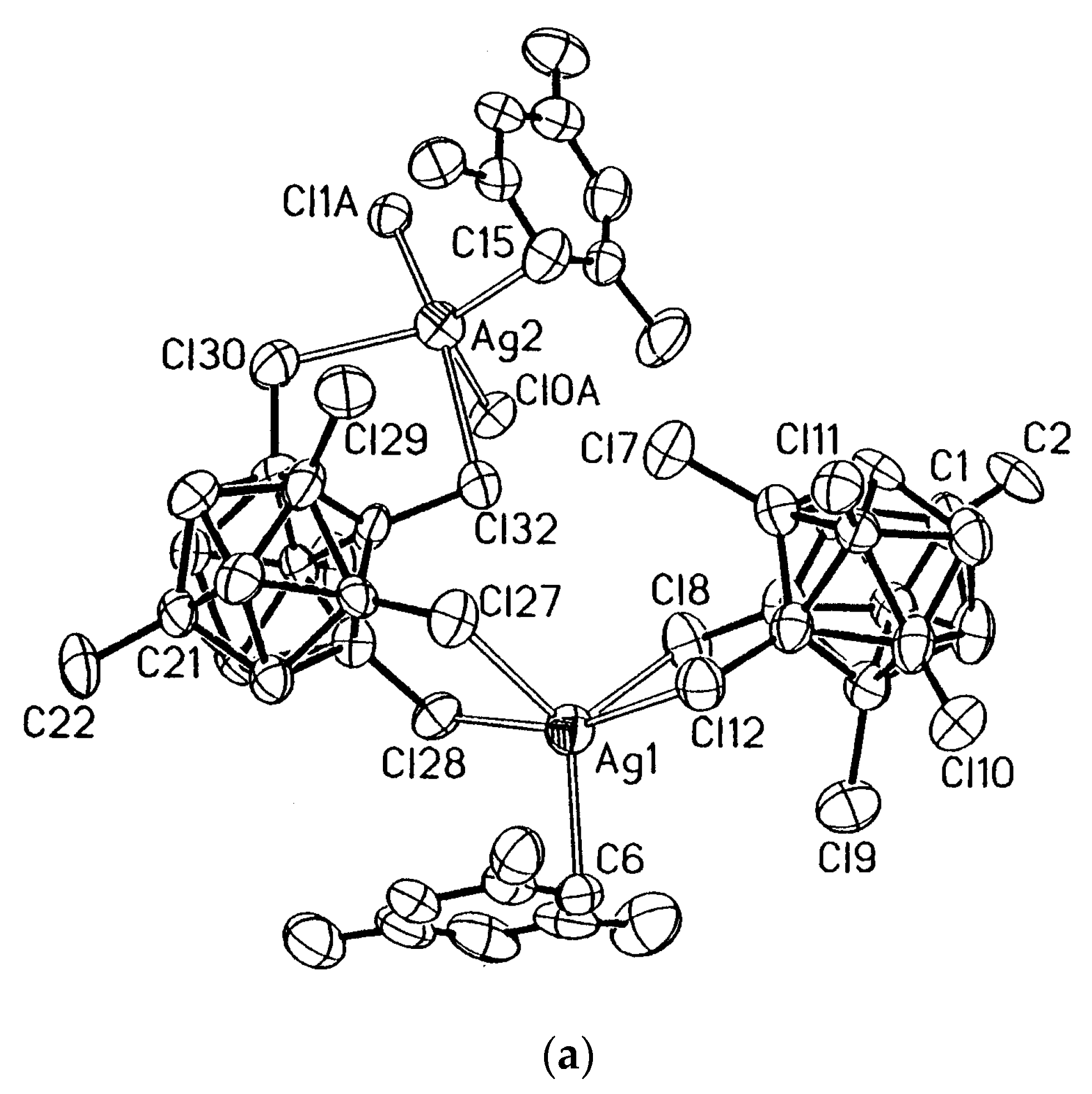
In the structure of {(Ph
4P)(Cu[B
10H
10])}
n the copper cation is coordinated by two
closo-decaborate anions through single B
e-H groups (Cu…B 2.160 and 2.187 Å) [
27] forming infinite polymeric chain (
Figure 5).
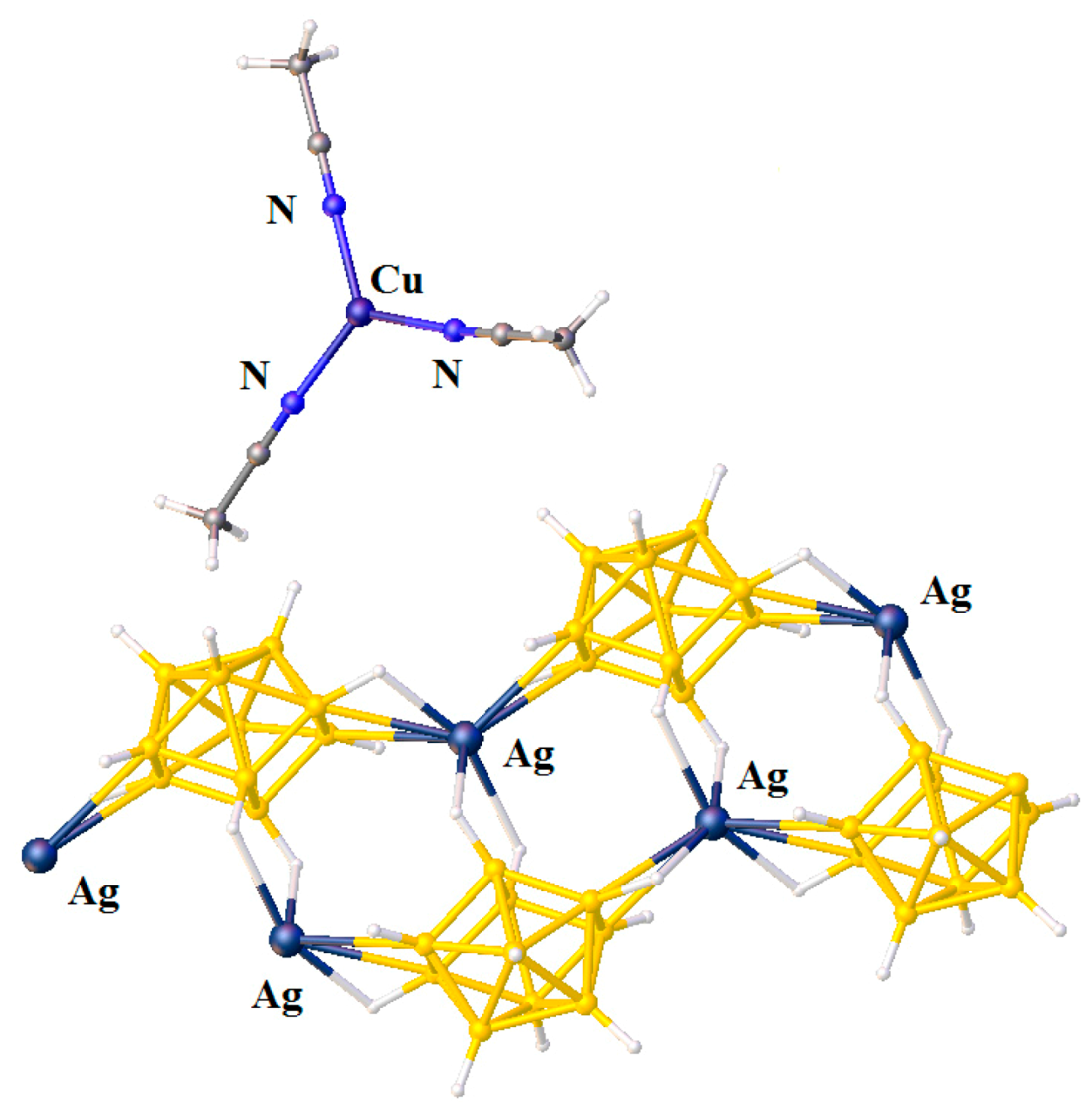
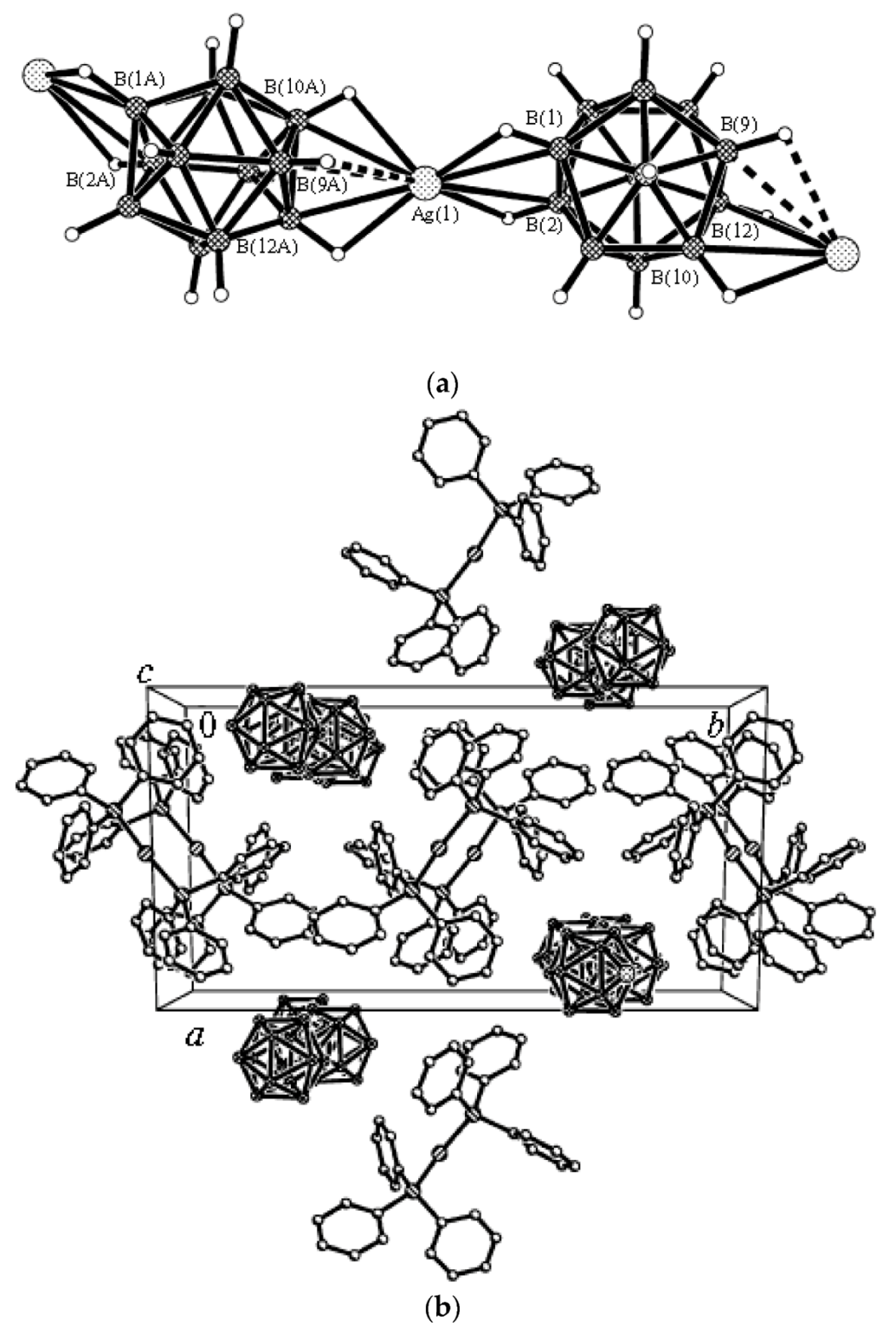
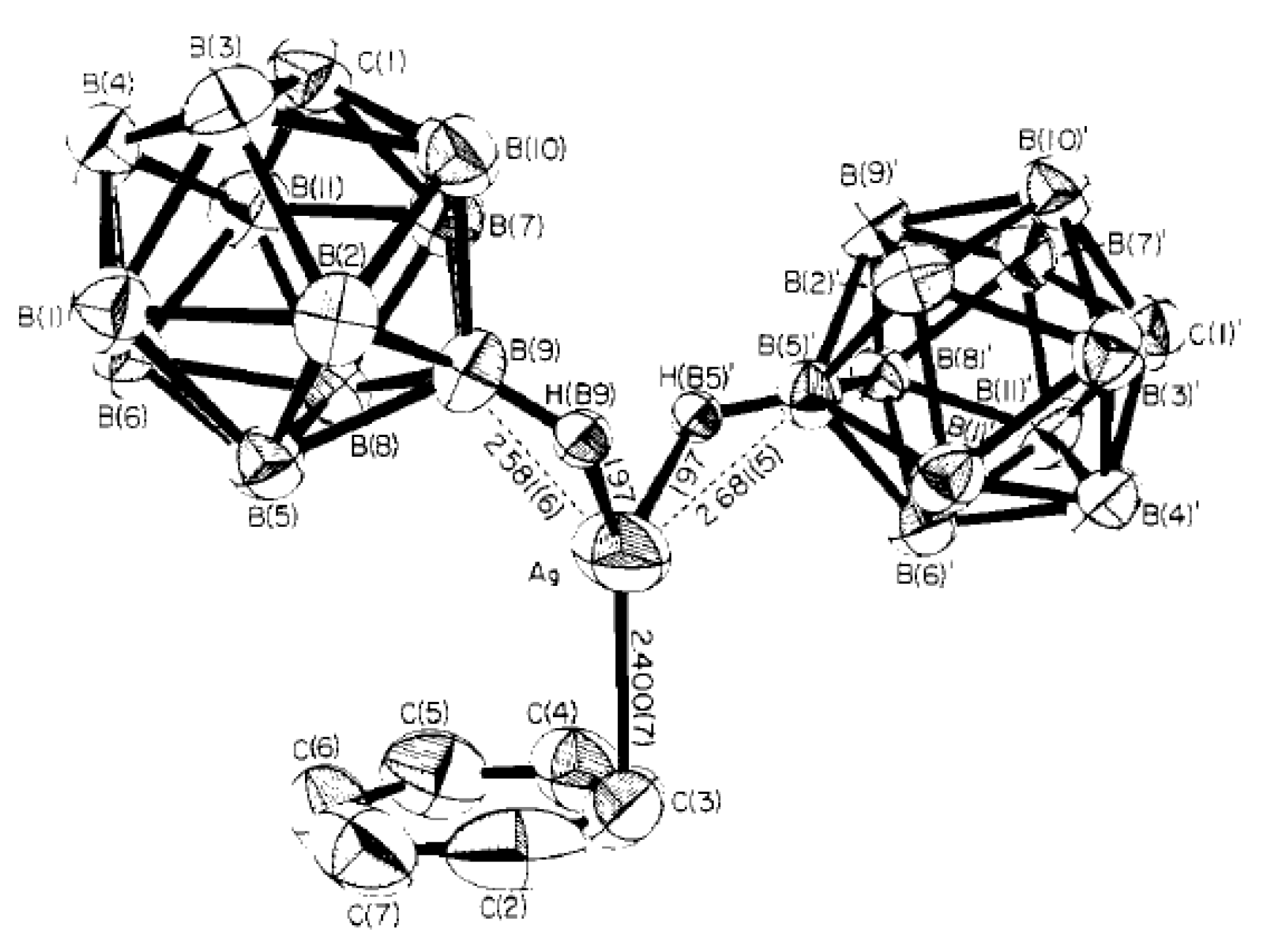
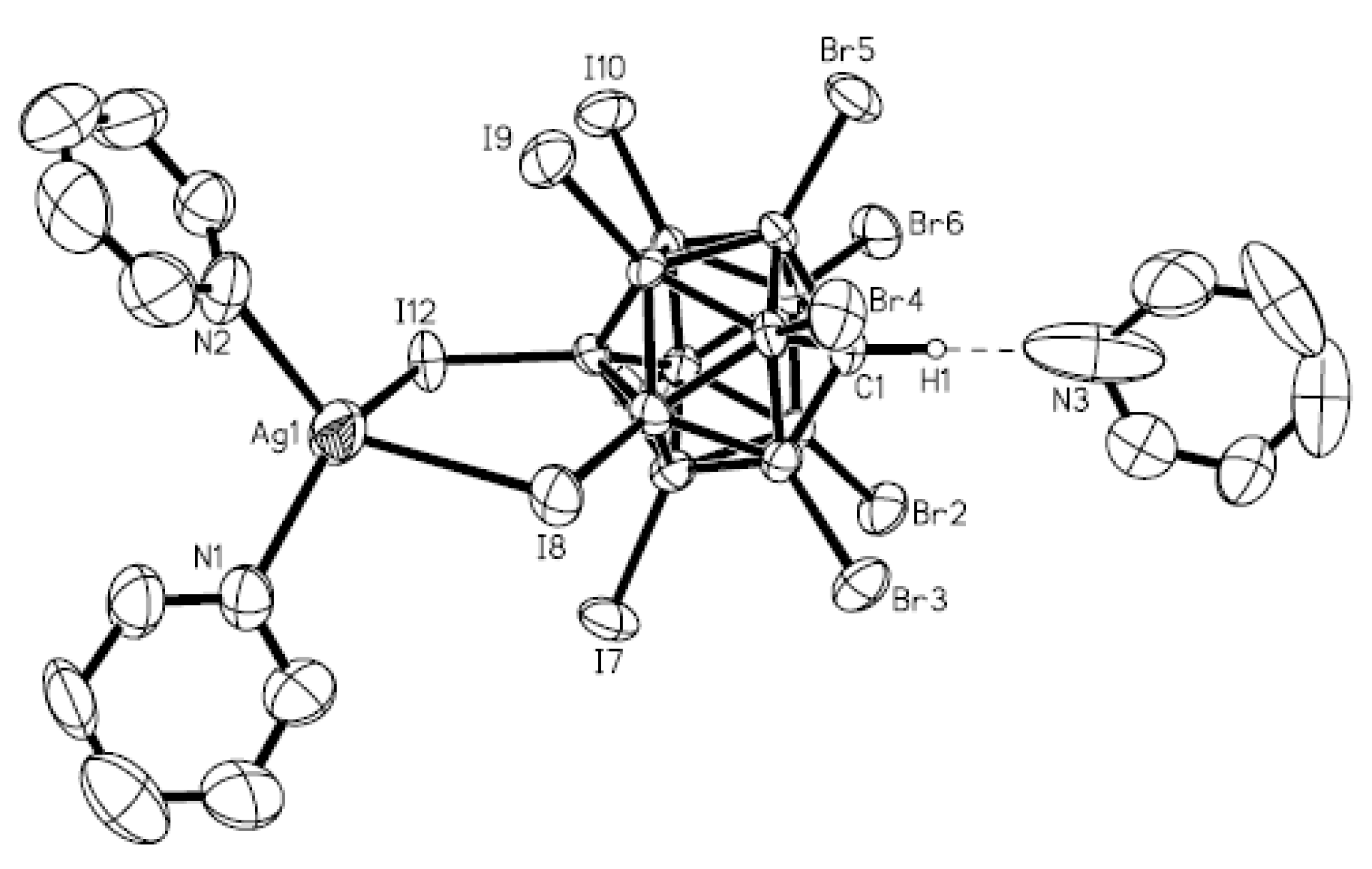
The unusual complex {[Cu(MeCN)
3](Ag[B
10H
10])}
n was prepared by the reaction of Cu
2[B
10H
10] and Ag
2[B
10H
10] in acetonitrile in the presence of trifluoroacetic acid. The silver cation is coordinated by two
closo-decaborate anions through B
a-B
e edges (Ag…B
a and Ag…B
e bond lengths are 2.656(8) and 2.547(7) Å, respectively) and one
closo-decaborate anion through B
e-B
e’ edge (Ag…B
e 2.758(9) Å). The trigonal-planar environment of copper cation is formed by acetonitrile nitrogen atoms. Two elongated contacts with the BH groups supplements the Cu
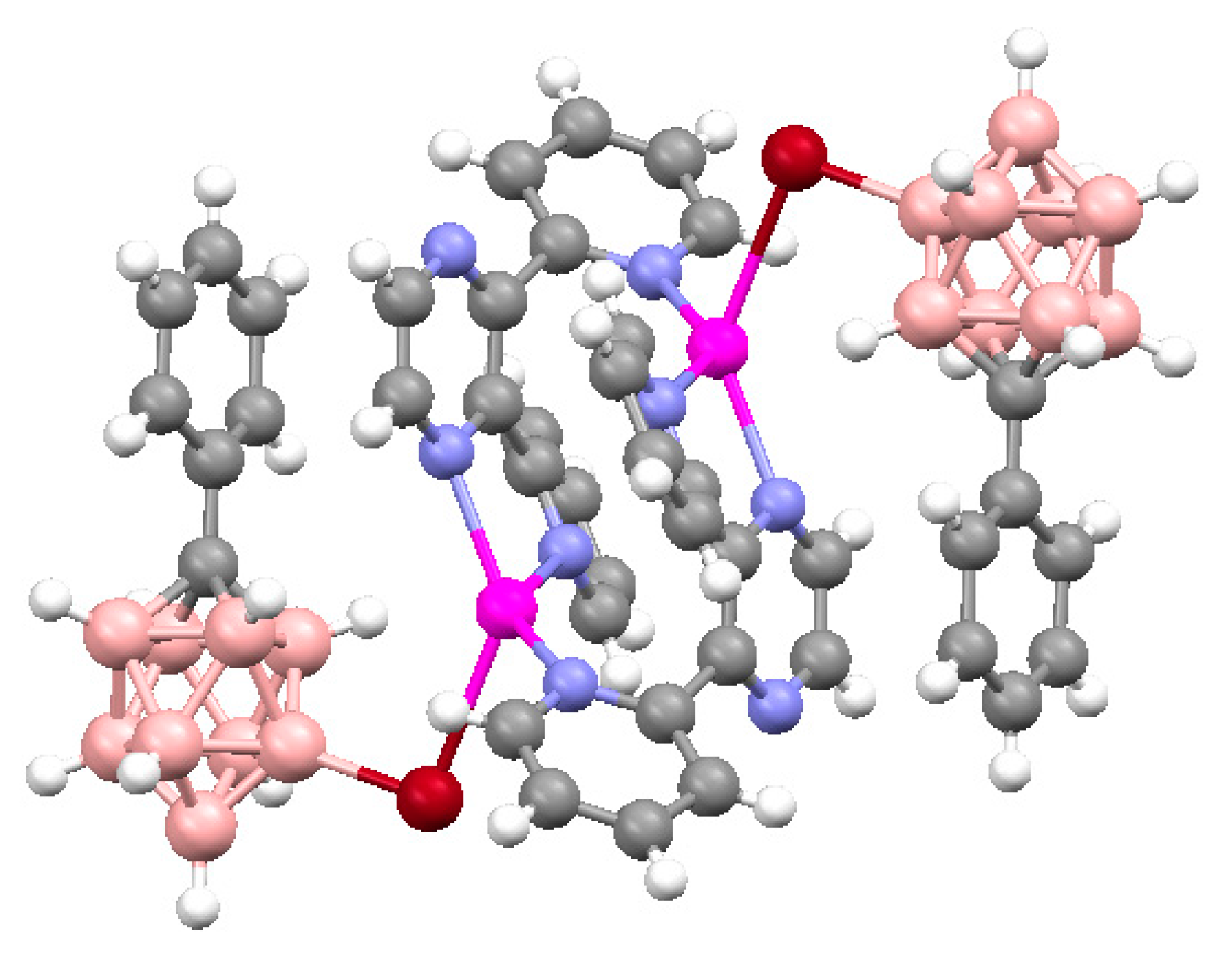
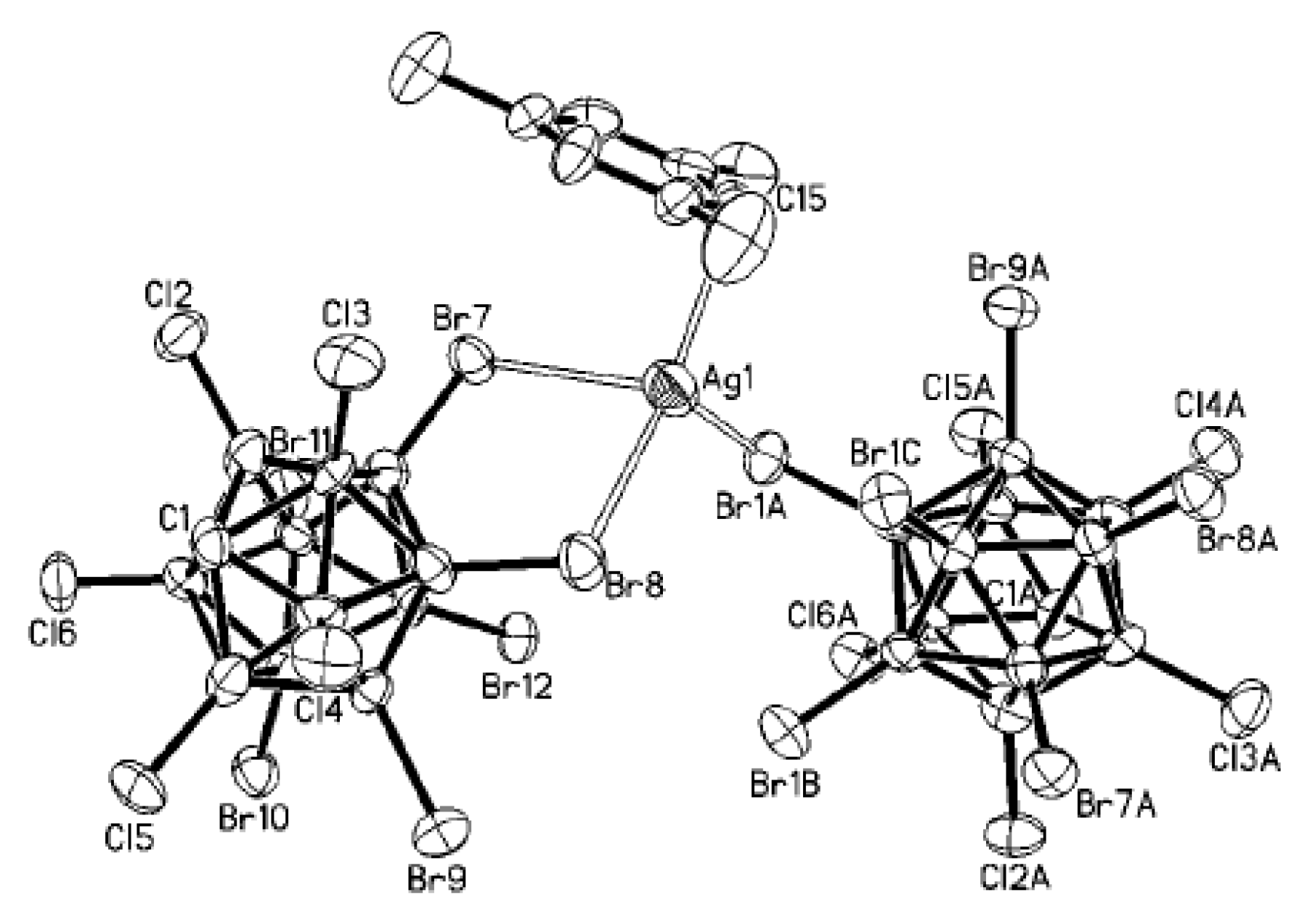
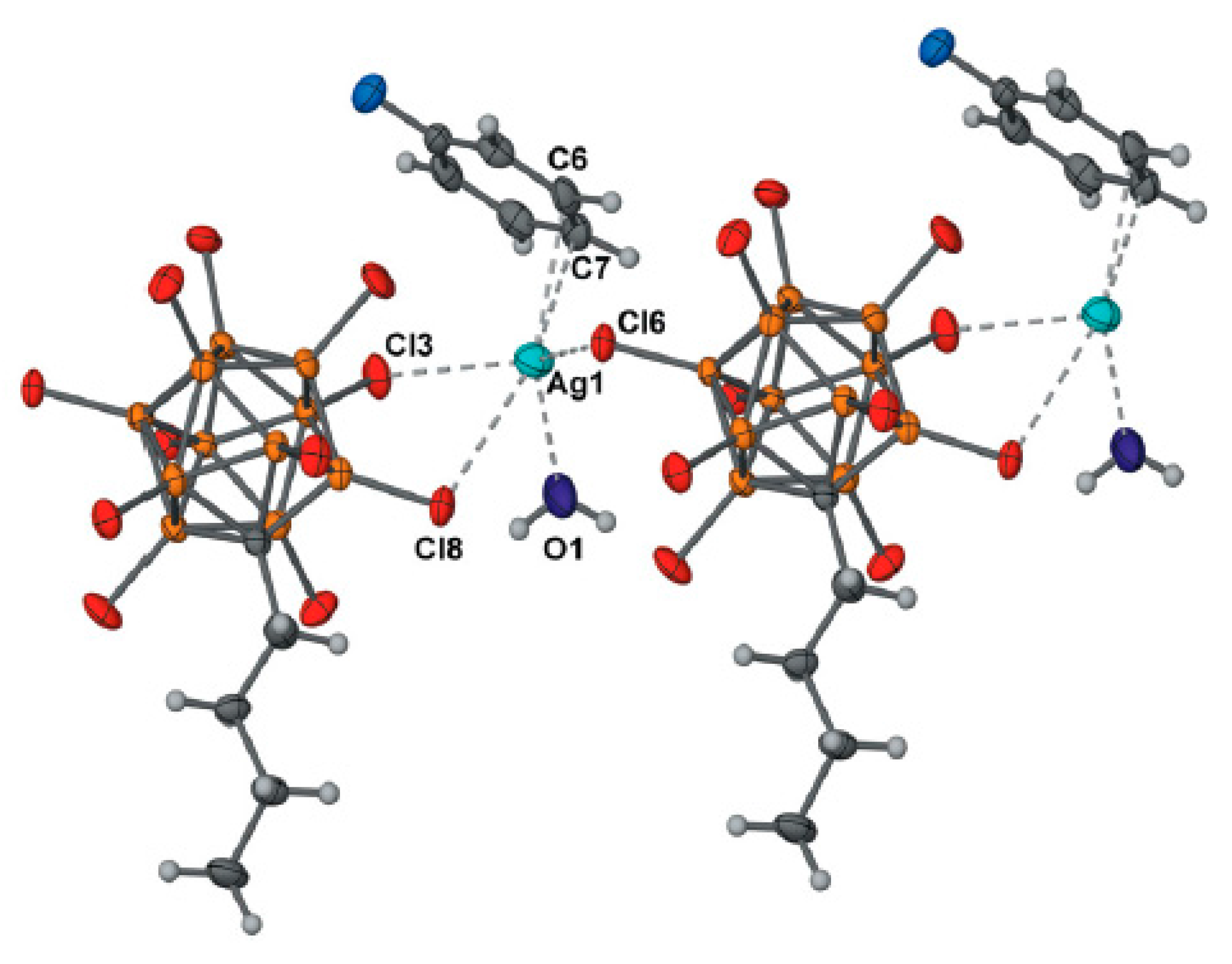
+ coordination to trigonal bipyramidal (Cu…B(H), 3.319(7) Å; Cu…H(B), 2.40(7) Å) (
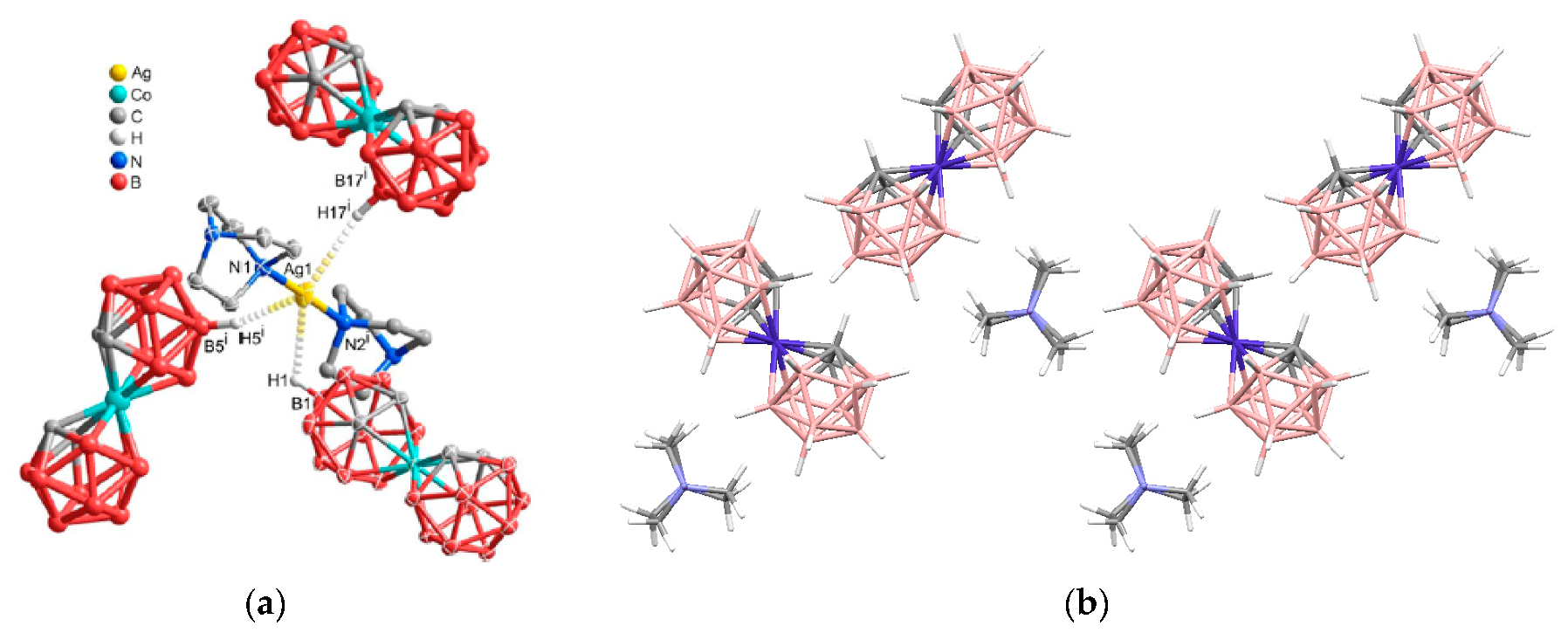
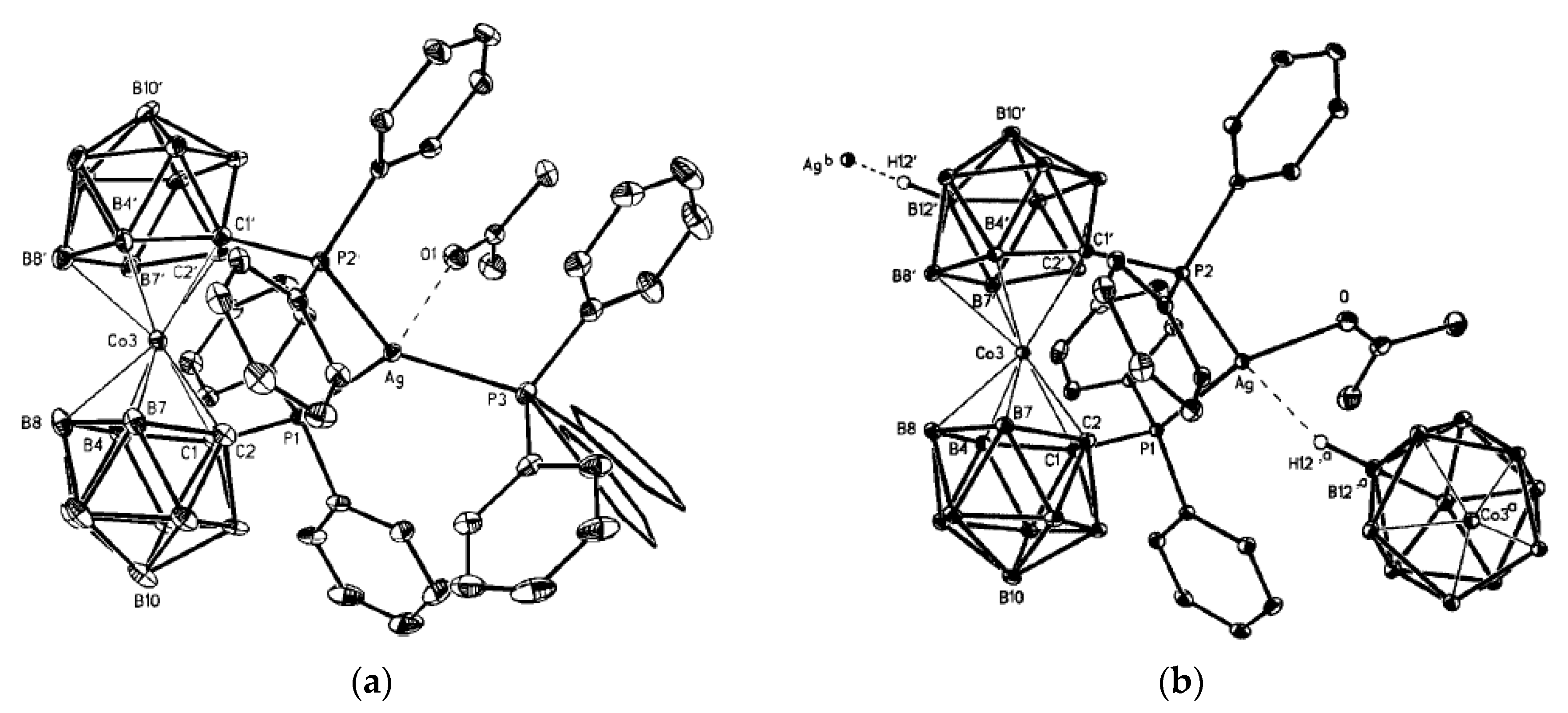
Figure 6) [
28].
As it was demonstrated above, all the {Cat(M[B
10H
10])}
n complexes in the solid state form polymeric chains with cations located between the chains. The discrete complex (Et
3NH)([Cu[1-B
10H
9N
2]
2) was prepared by the reaction of the charge-compensated diazonium derivative of the
closo-decaborate anion (Et
3NH)[1-B
10H
9N
2] with CuCl. The copper cation in this complex is coordinated by two [1-B
10H
9N
2]
− anions through B(6)-B(10) edges that are the most distant from the substituent. The Cu…B(6) and Cu…B(10) distances are 2.184(9) and 2.168(8) Å, the Cu…H(6) and Cu…H(10) distances are 1.96 and 1.99 Å (
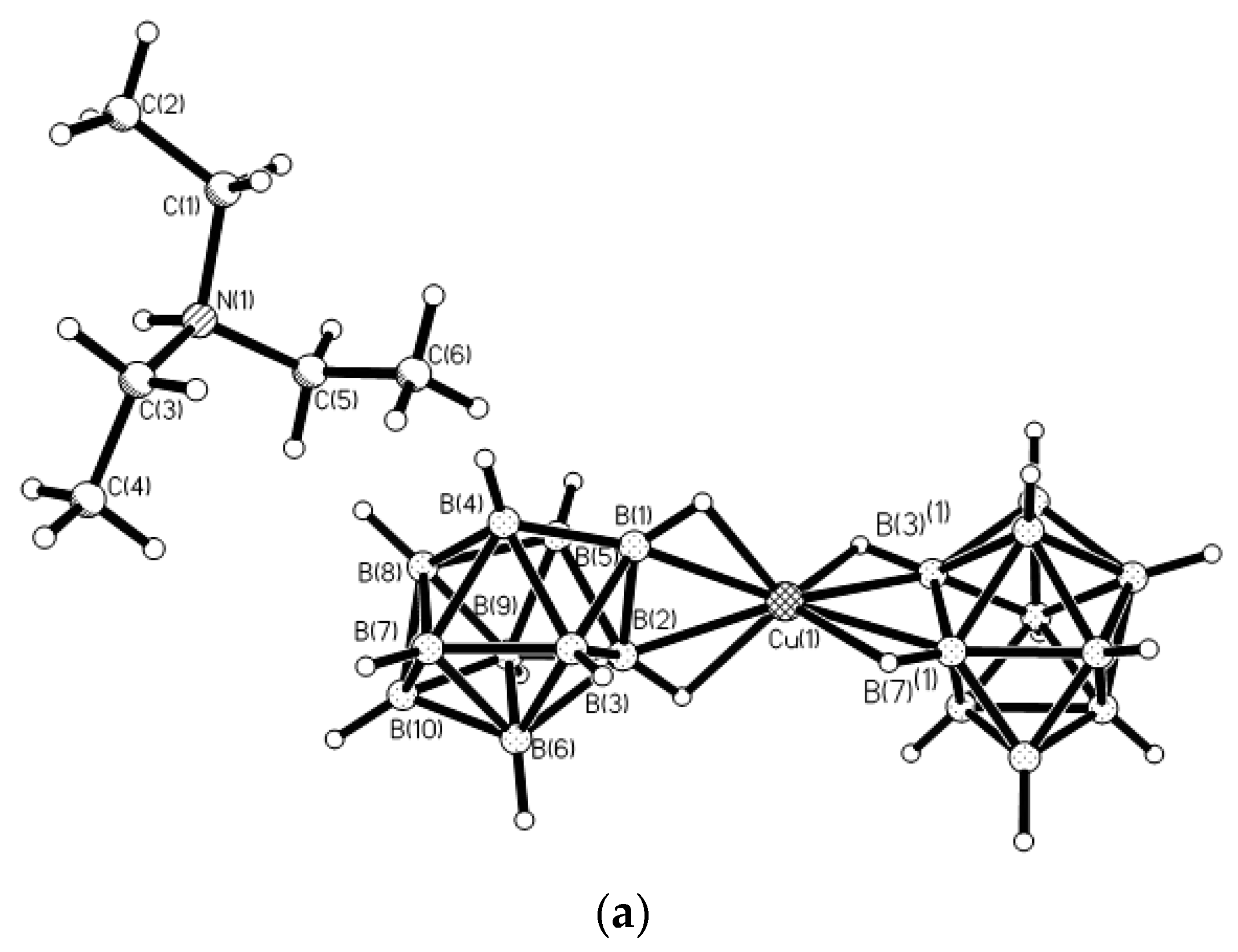
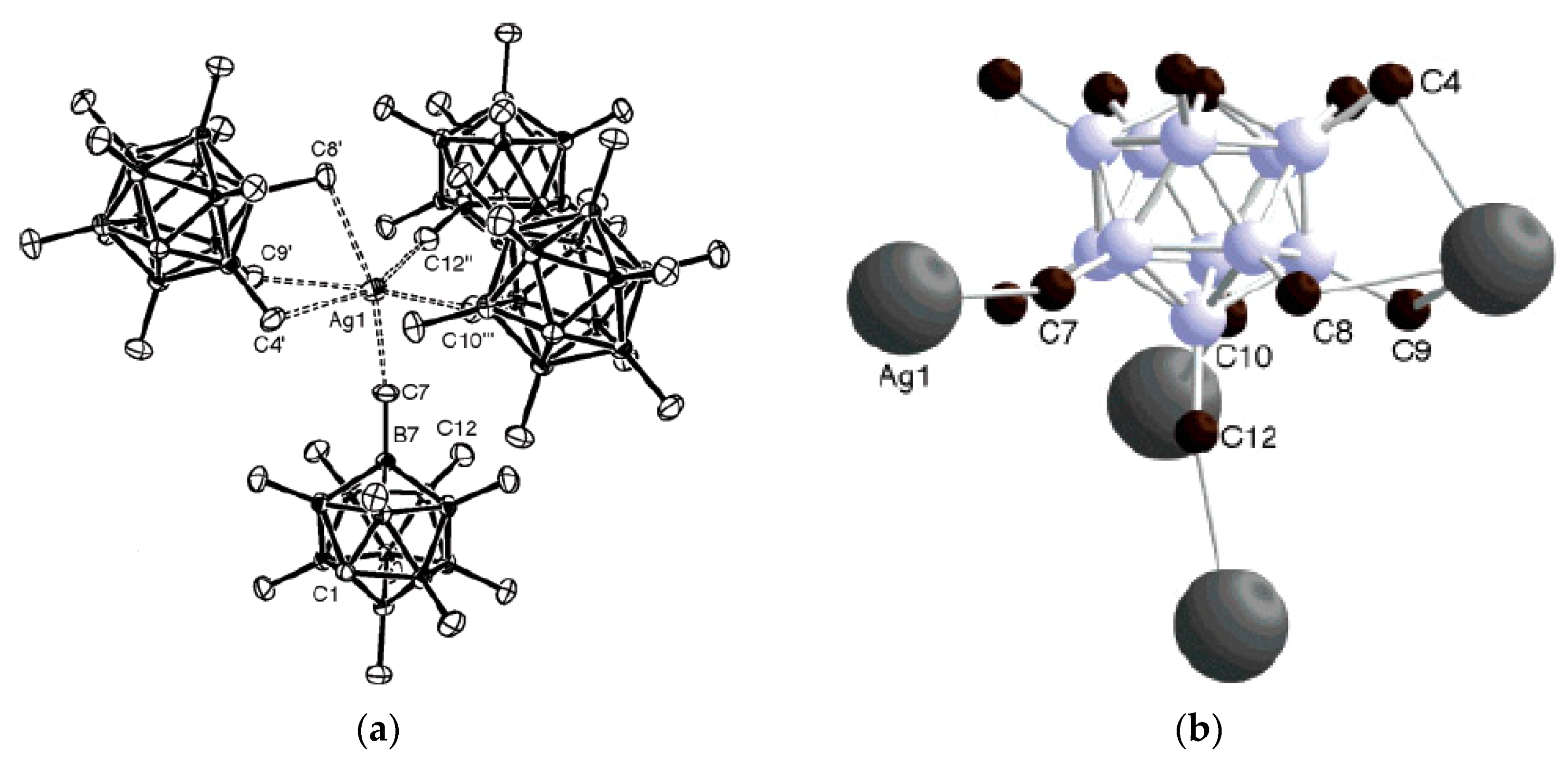
Figure 7) [
29].
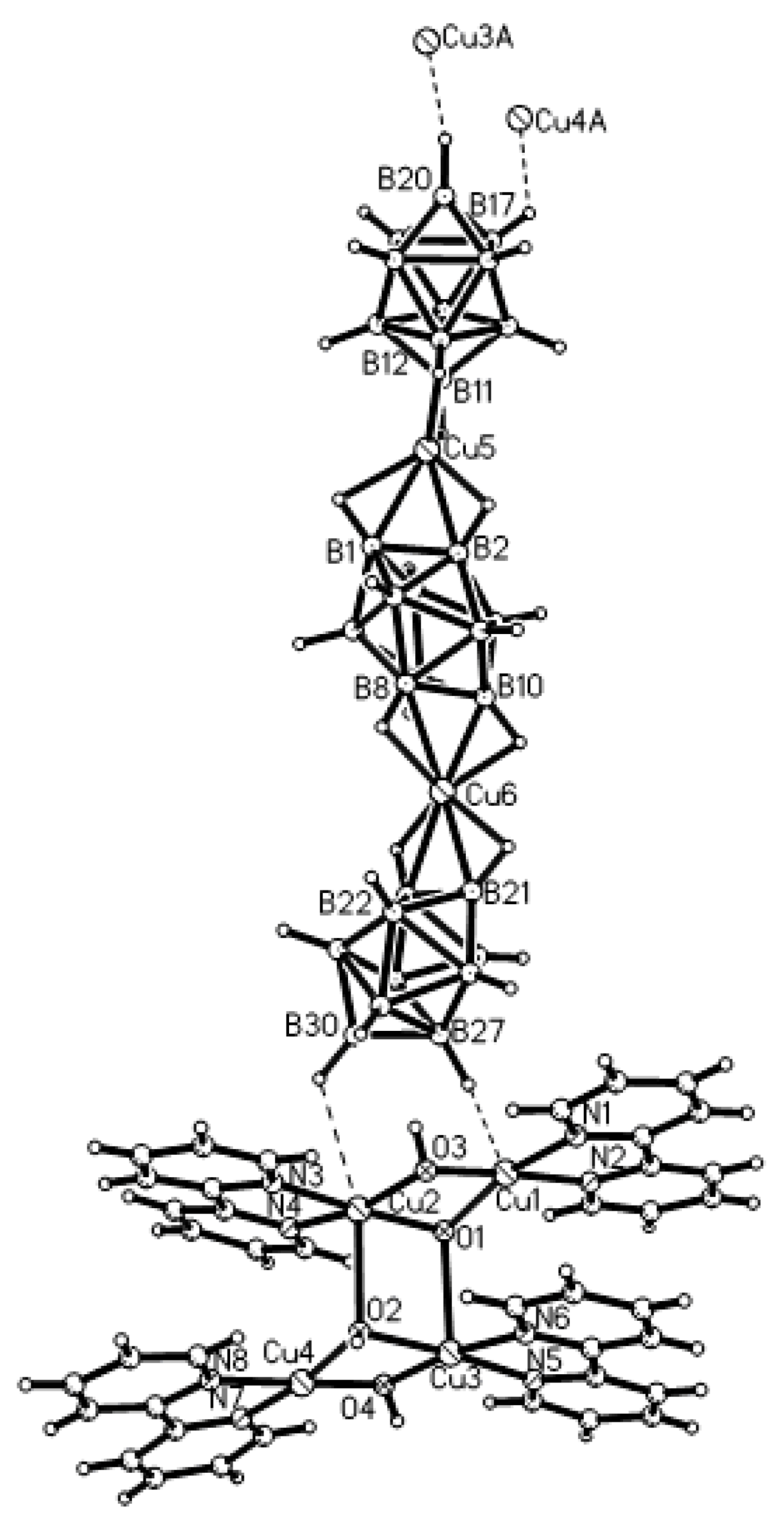
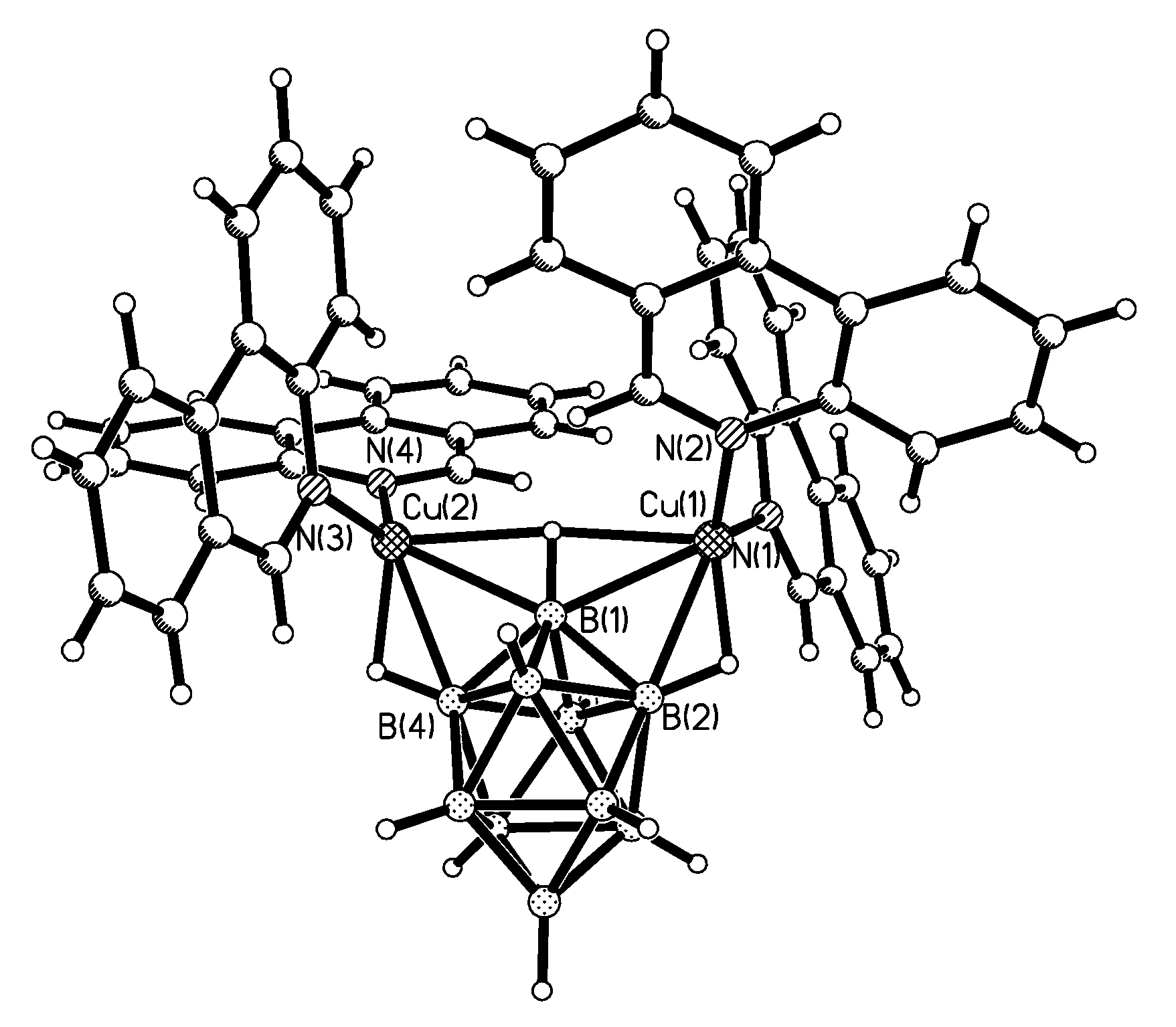
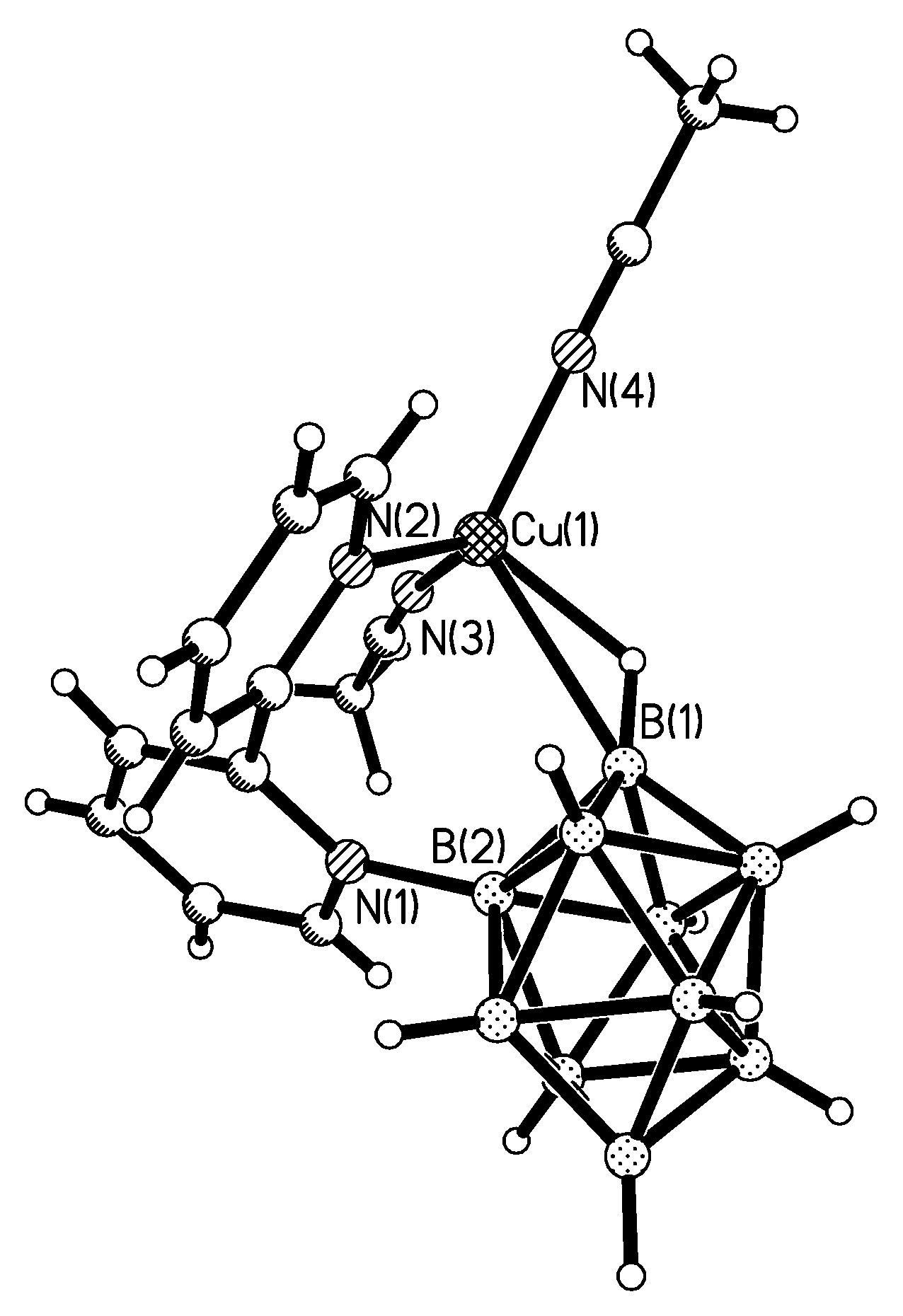
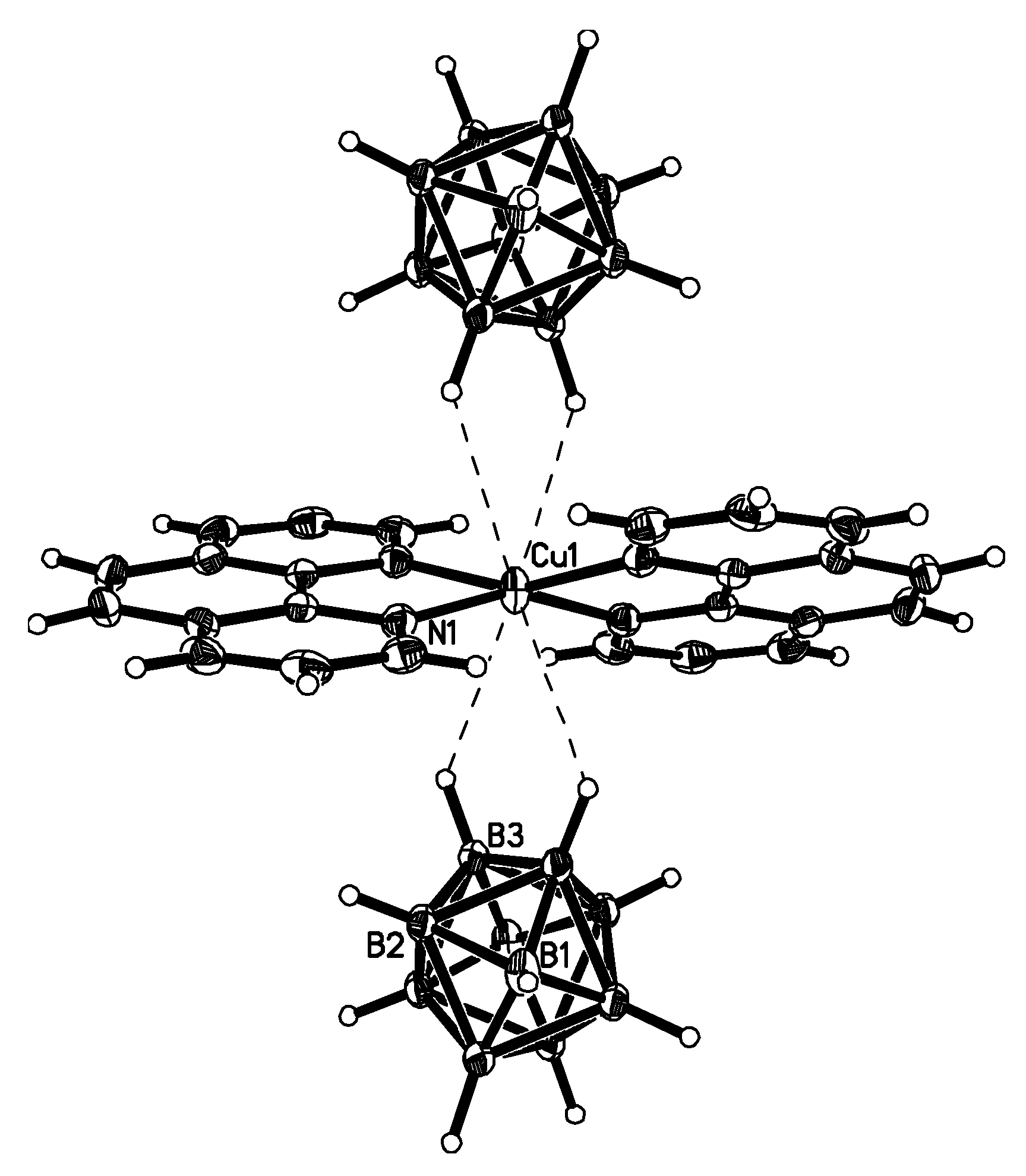
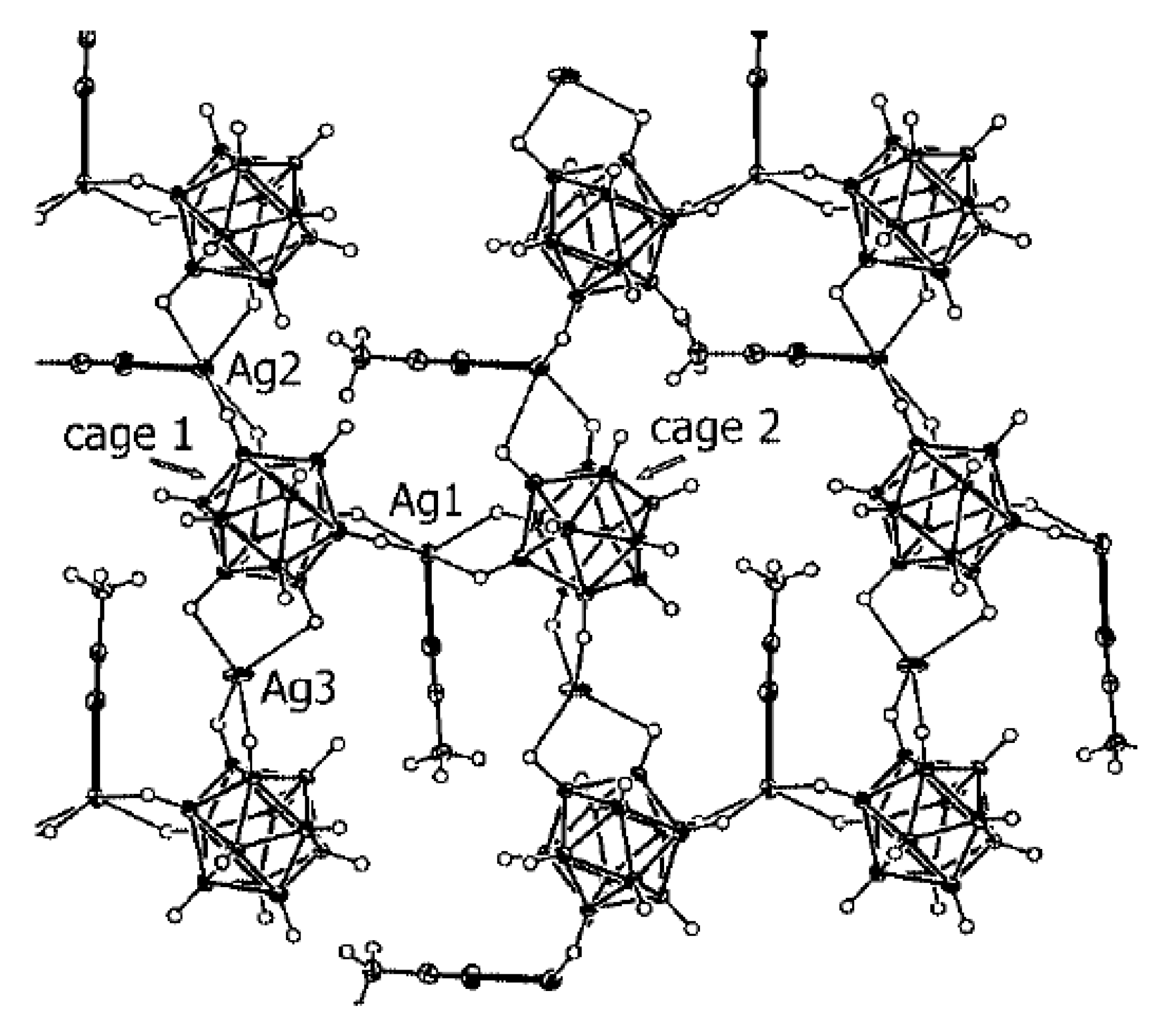
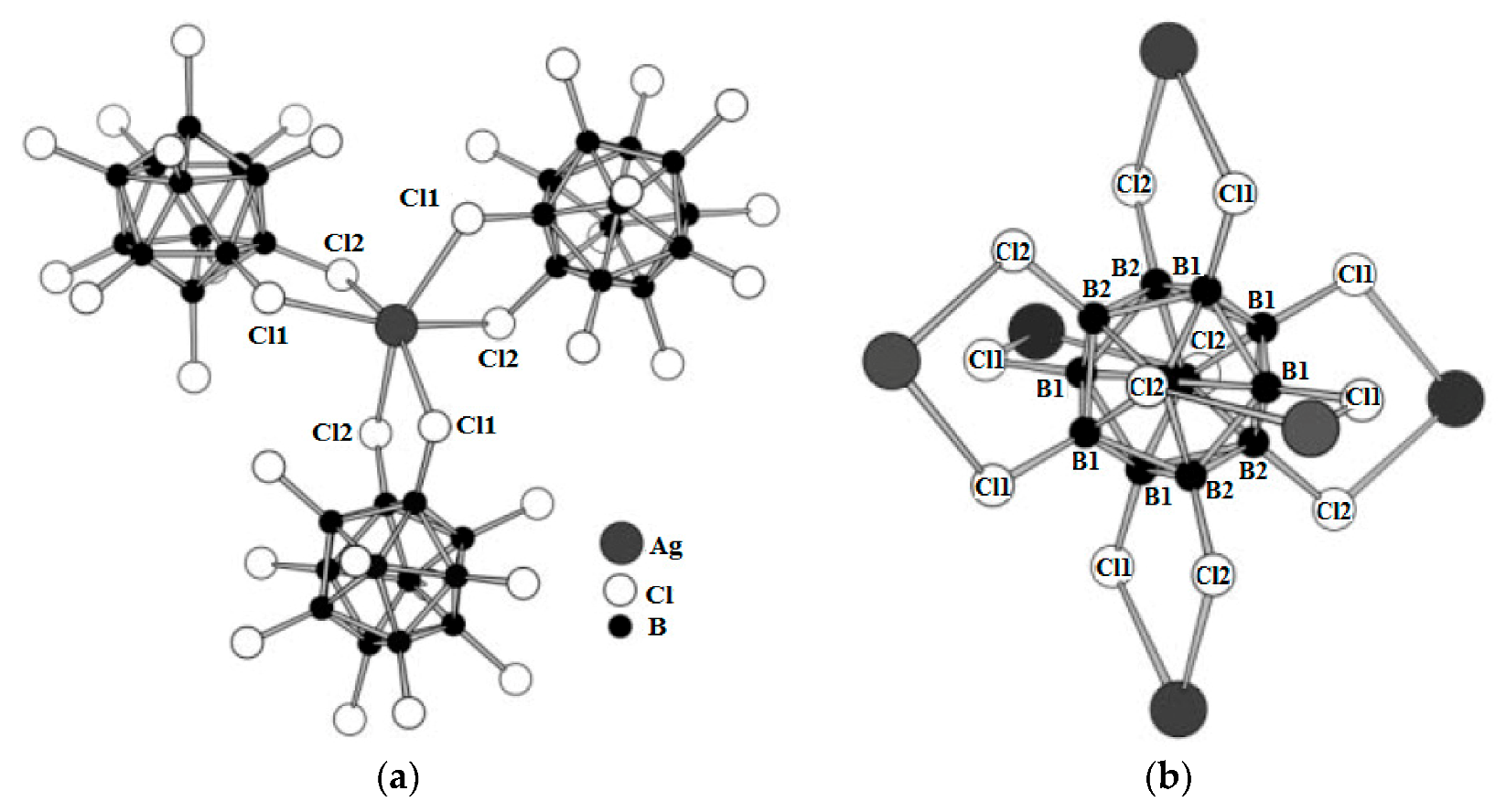
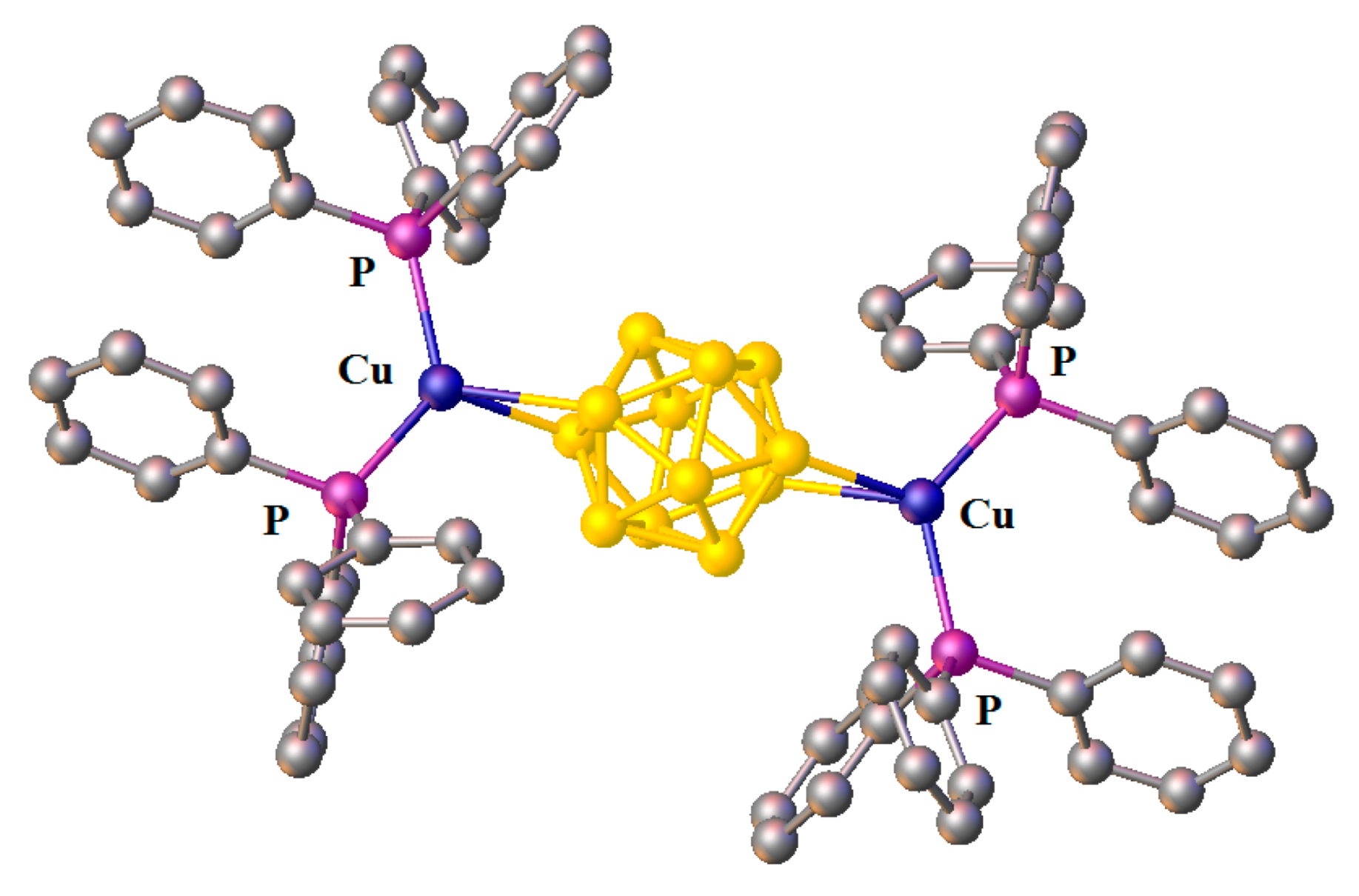
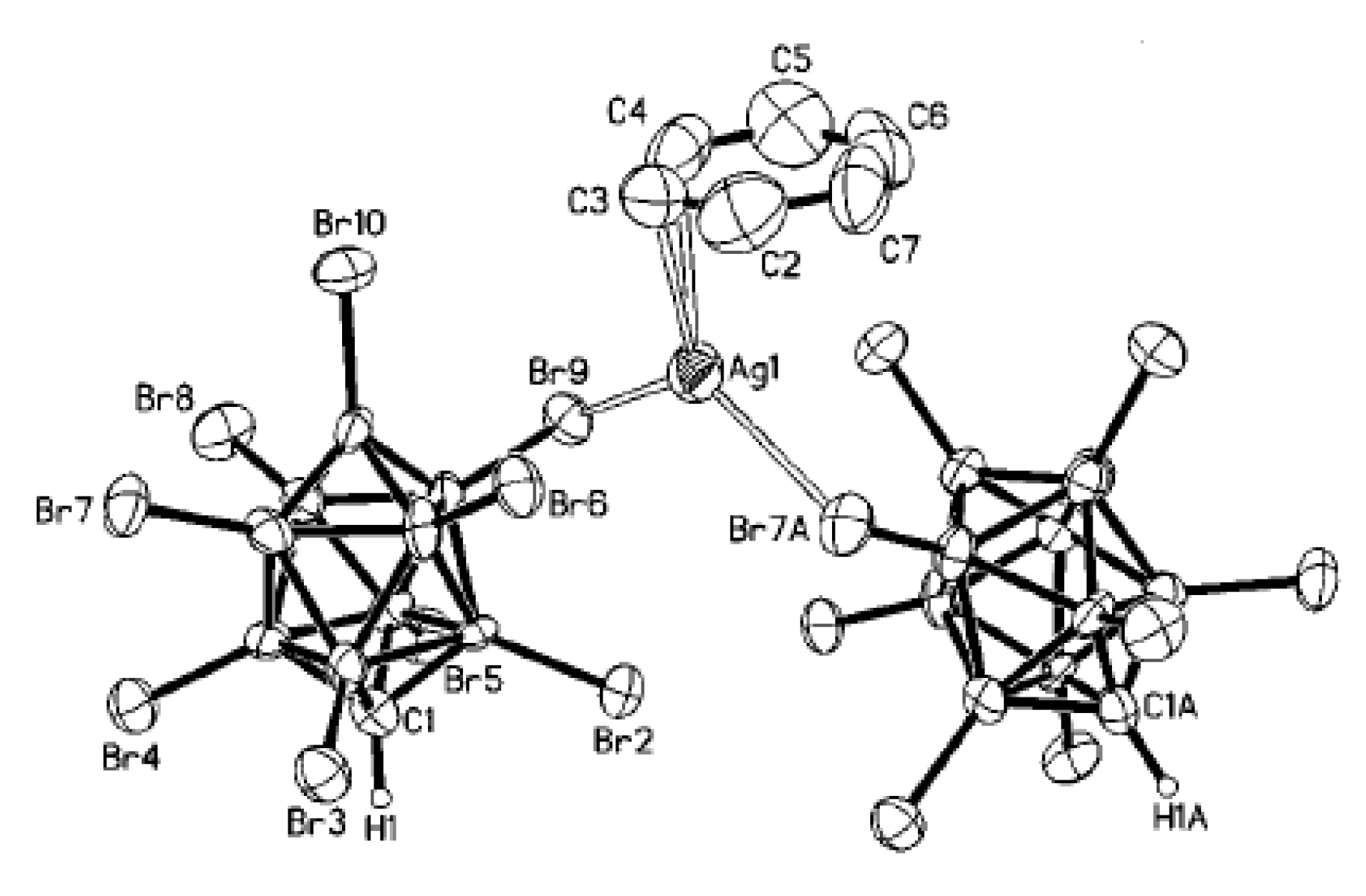
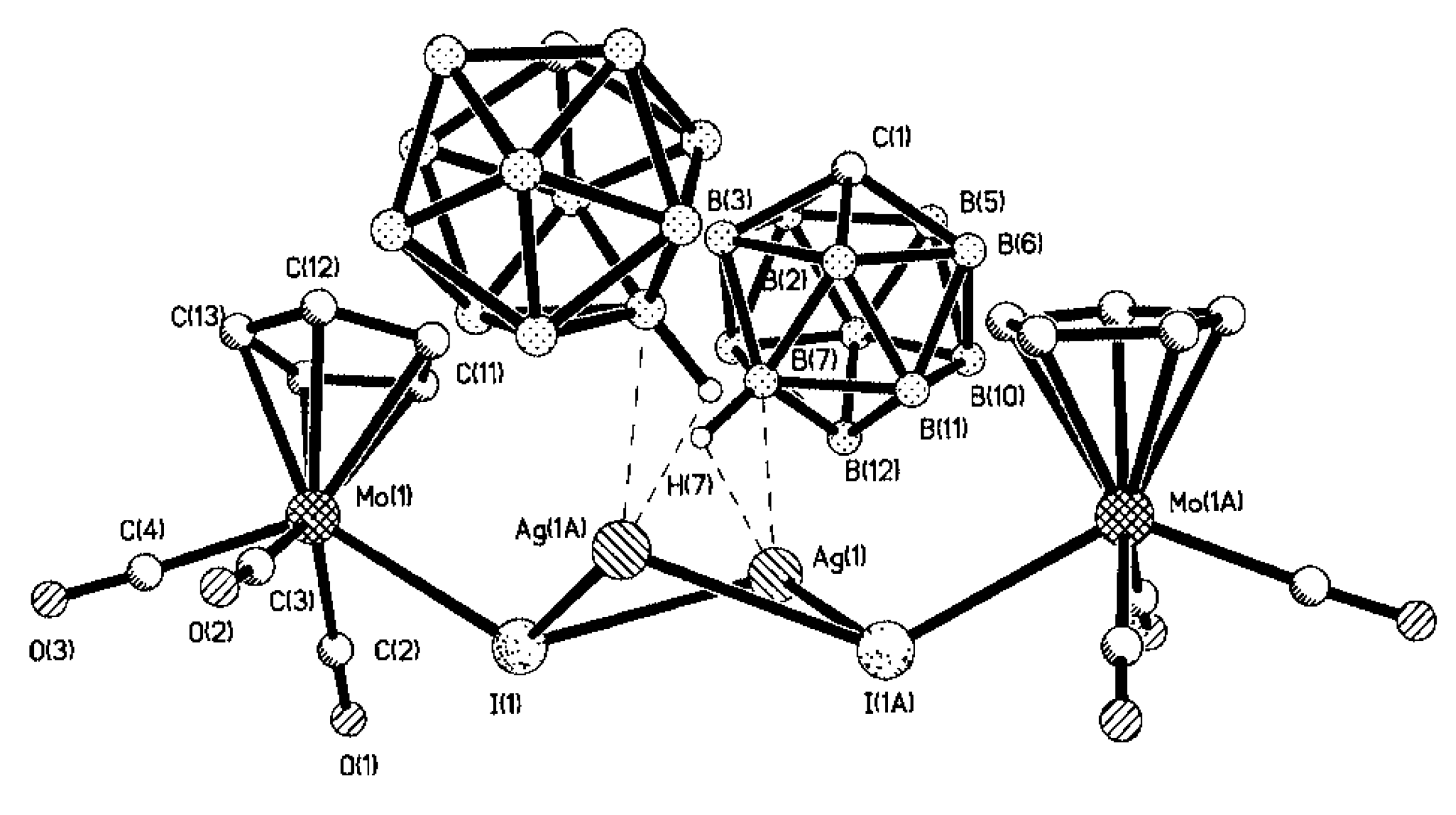
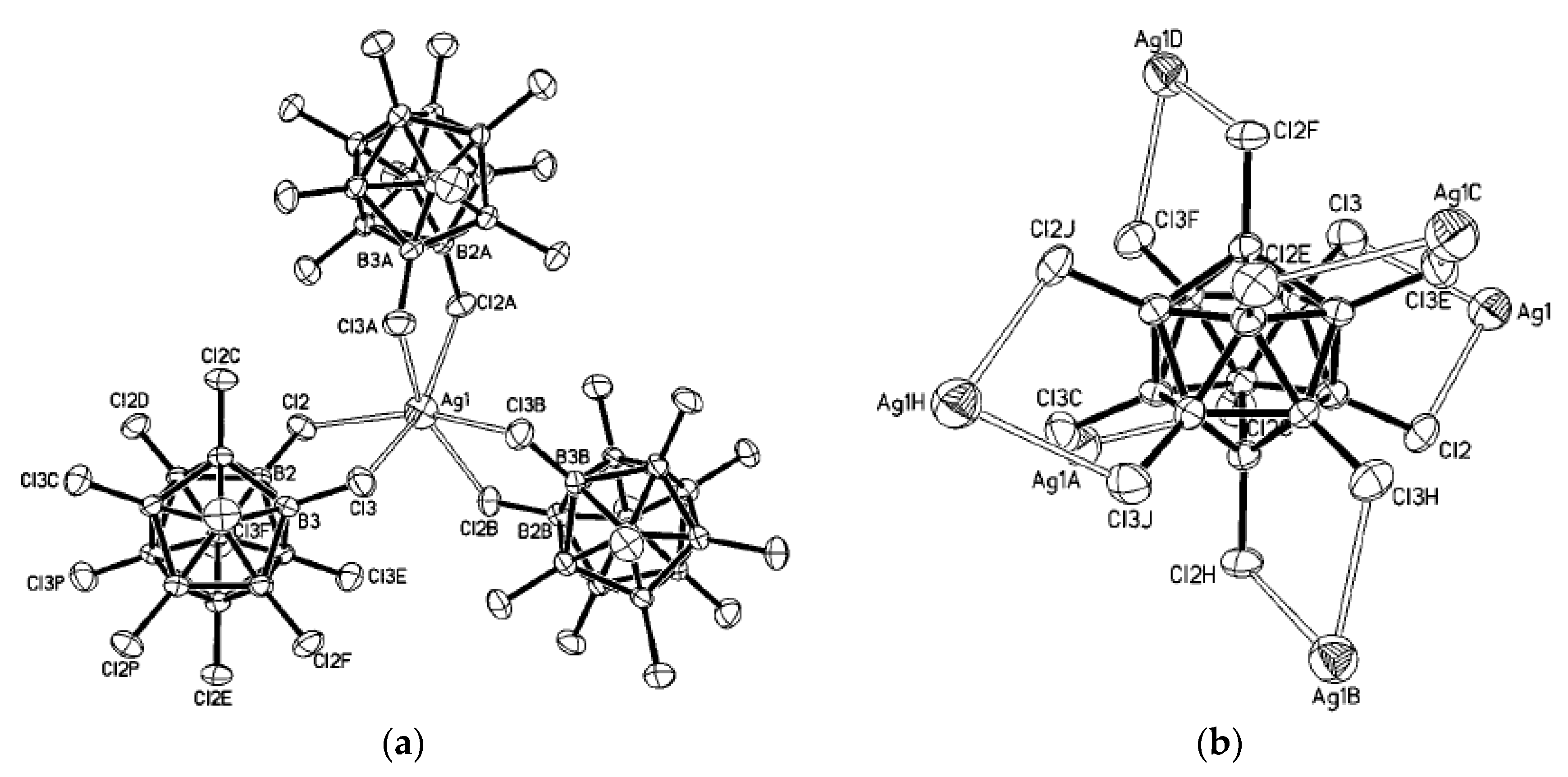
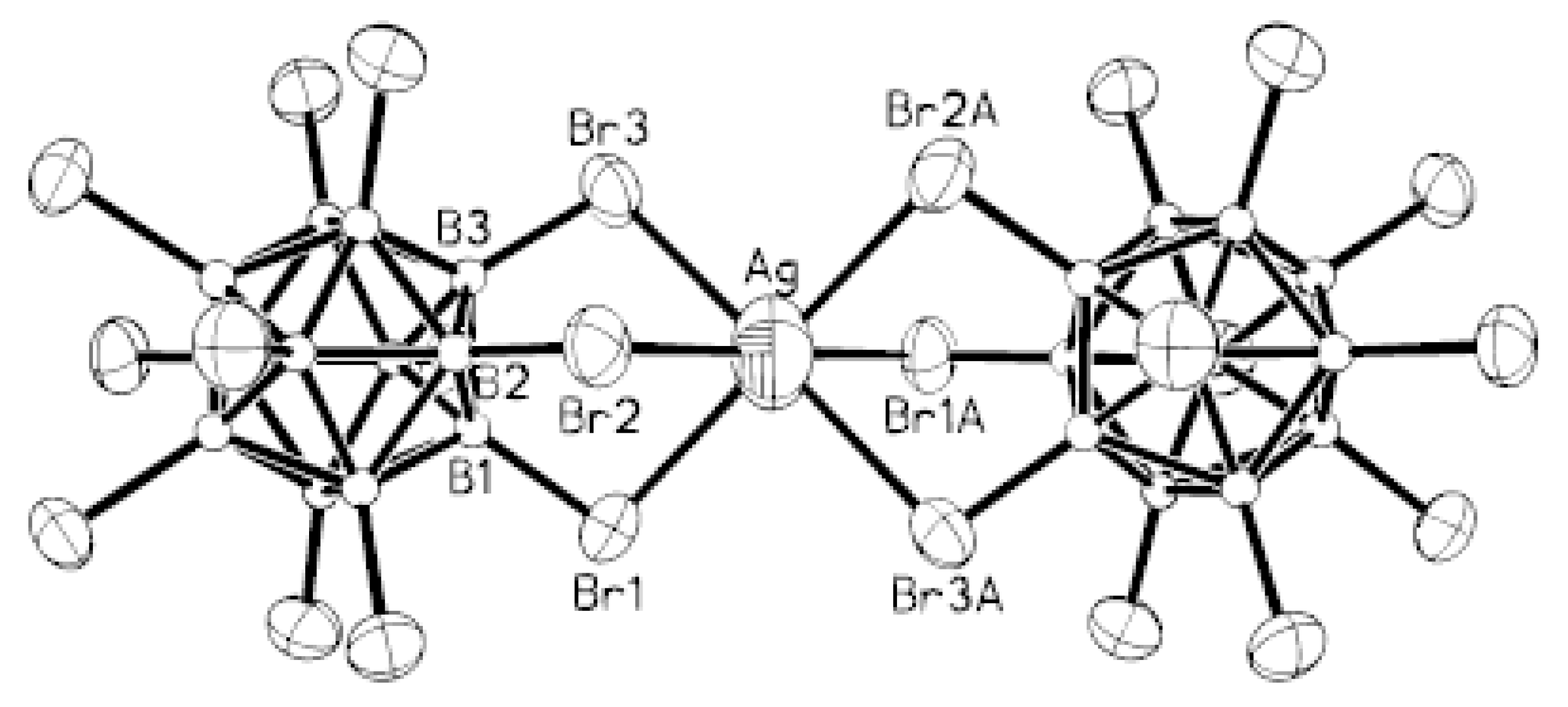
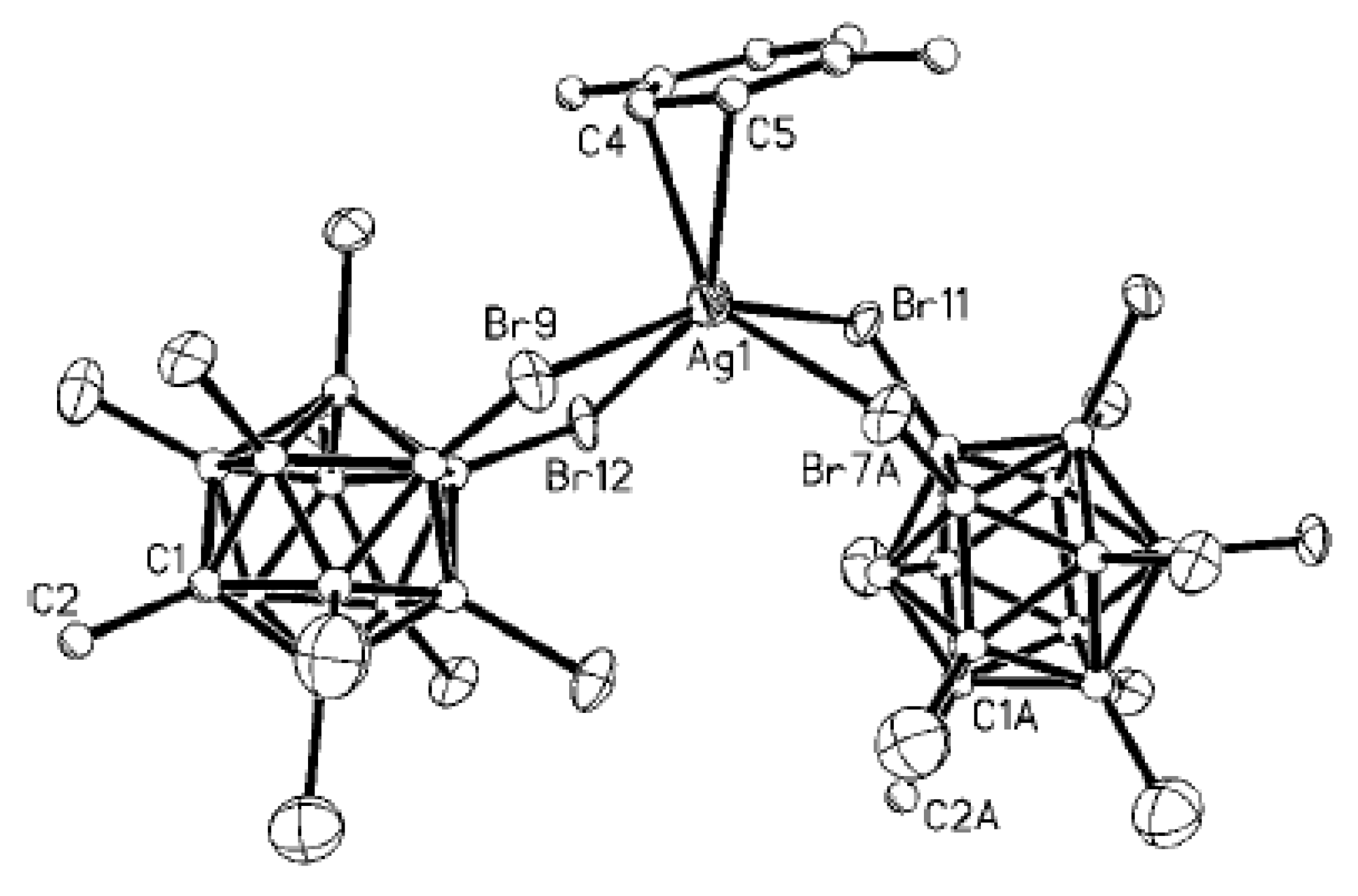
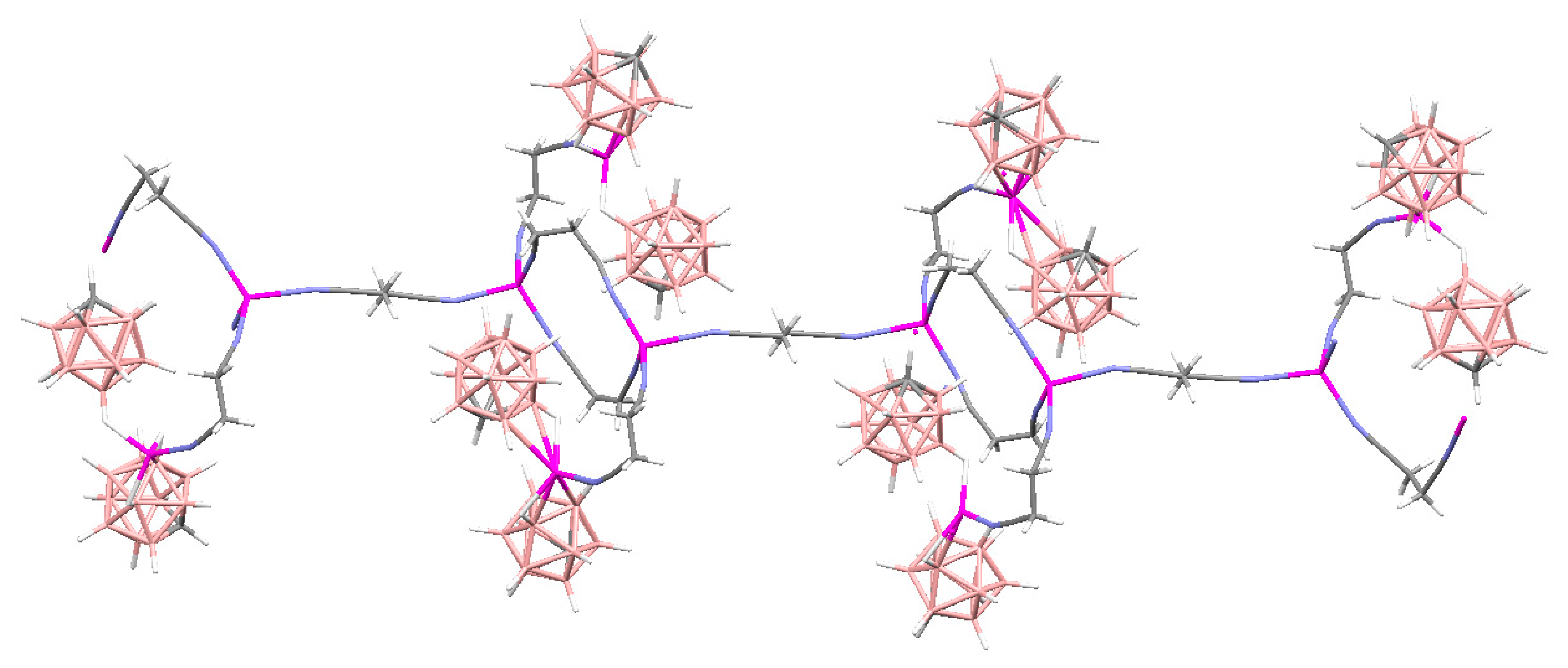
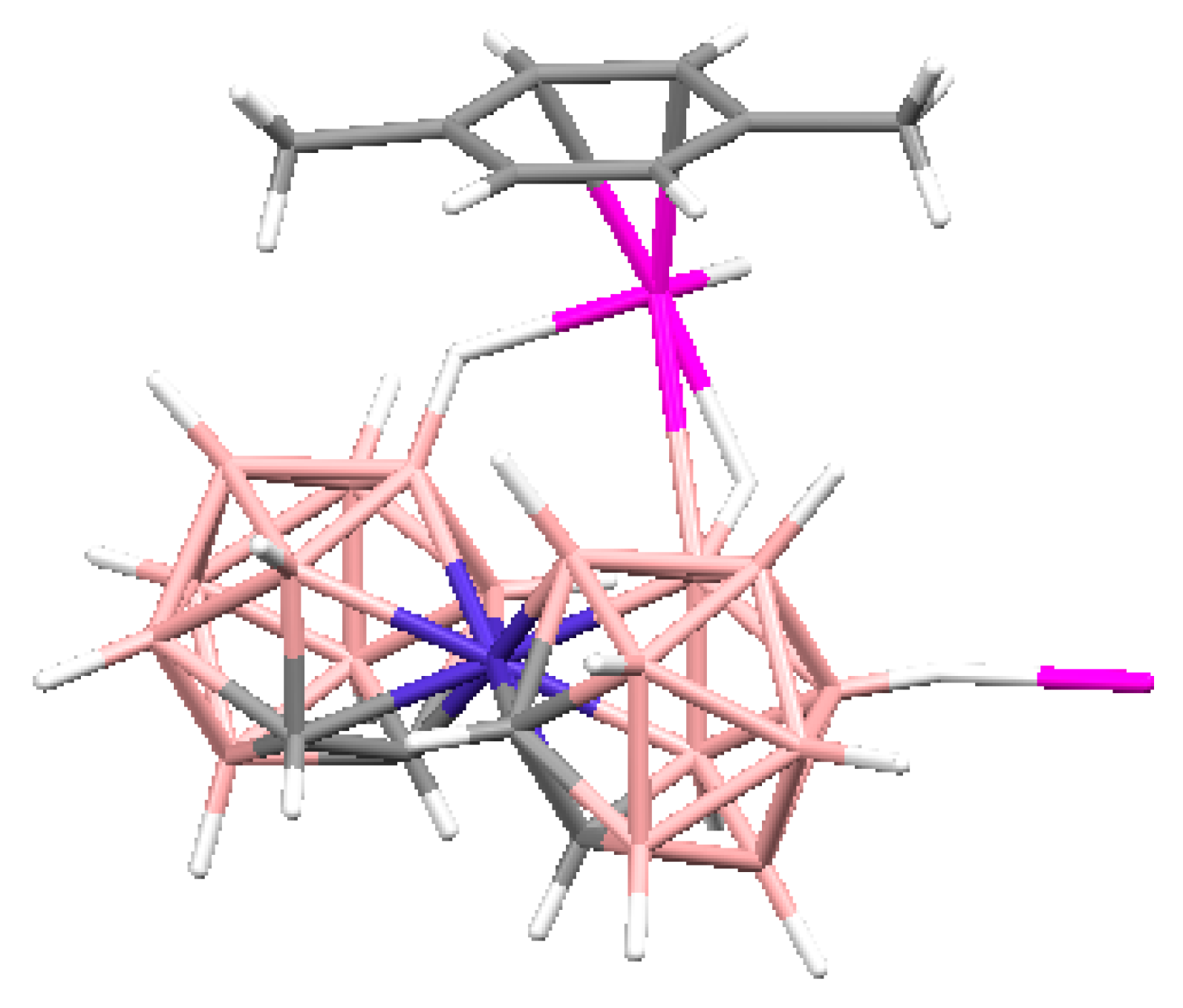
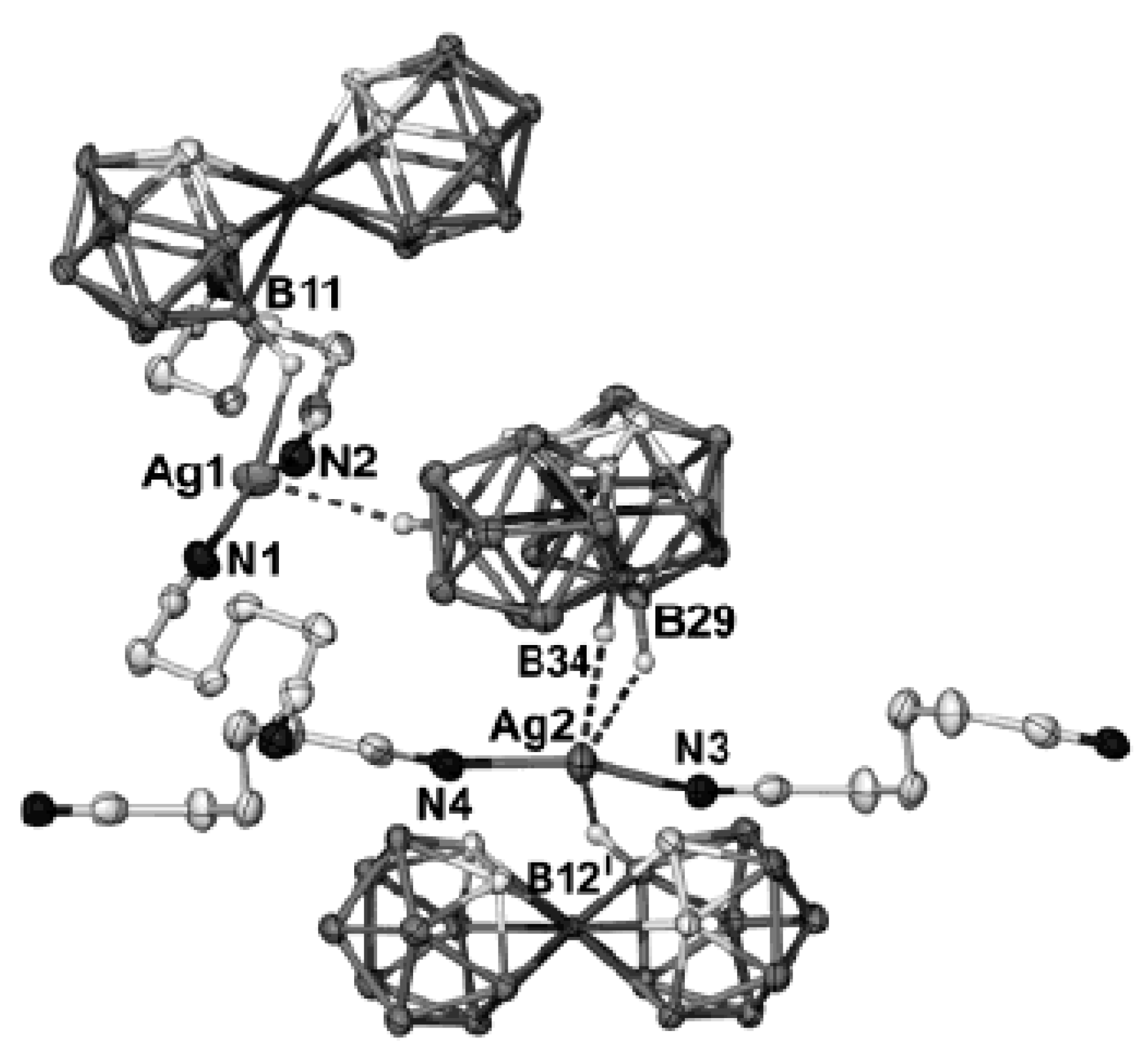
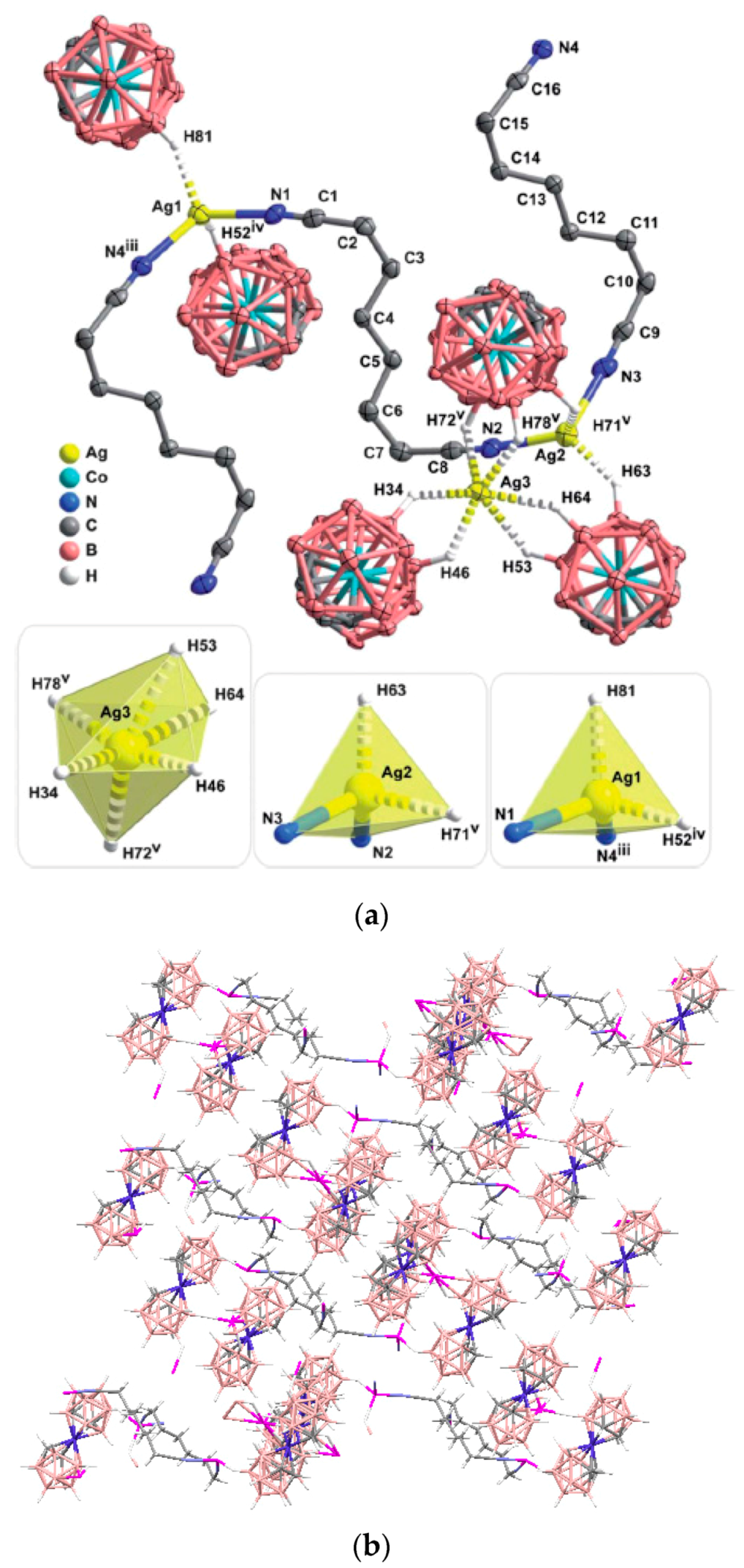
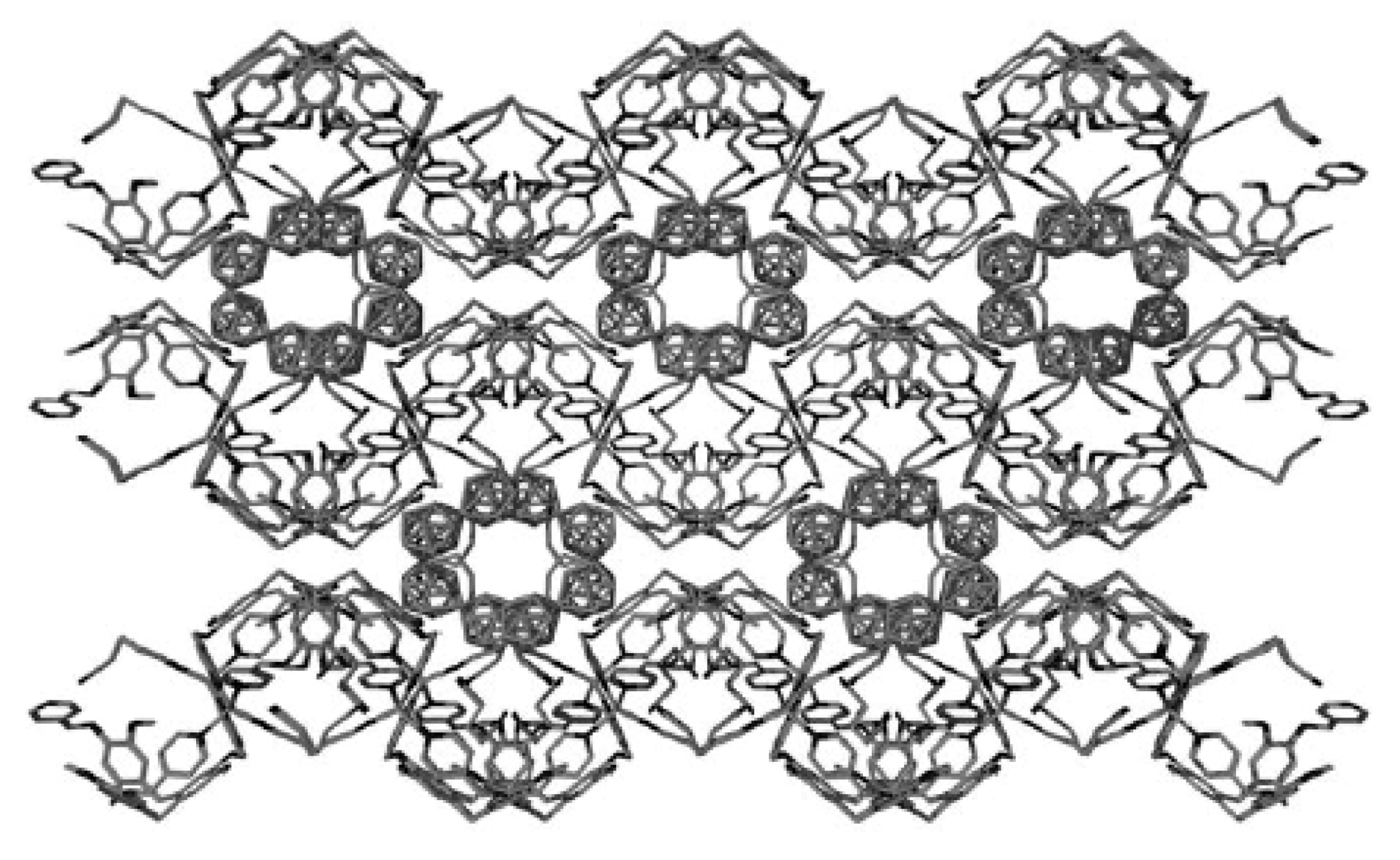
Complex [Cu
II4(OH)
4(2,2′-Bipy)
4][Cu
I2[B
10H
10]
3]·4(MeCN) containing discrete [Cu
I2[B
10H
10]
3]
4− anions and [Cu
II4(OH)
4(Bipy)
4]
4+ cations was prepared by the reaction of Cu
2[B
10H
10] with 2,2′-bipyridine in refluxing acetonitrile in air [
30,
31]. In the anionic part three
closo-decaborate anions are connected by two Cu
+ cations into a linear {Cu
I2[B
10H
10]
3}
4− oligomer (Cu…B and Cu…H(B) distances are 2.139–2.205(8) and 1.69–2.01 Å, respectively). The [Cu
4(OH)
4(Bipy)
4]
4+ cation has a double-decker structure. Each of the Cu
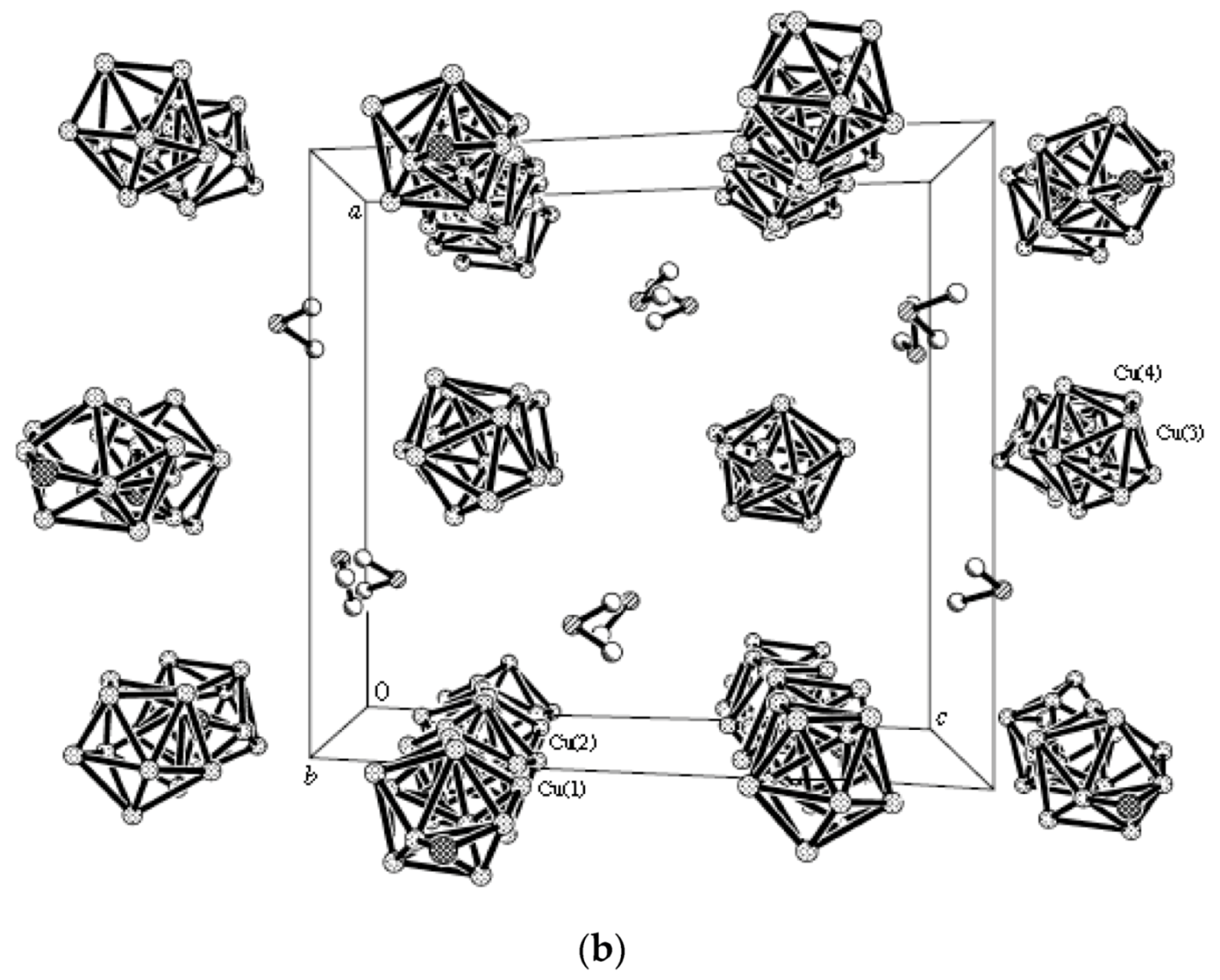
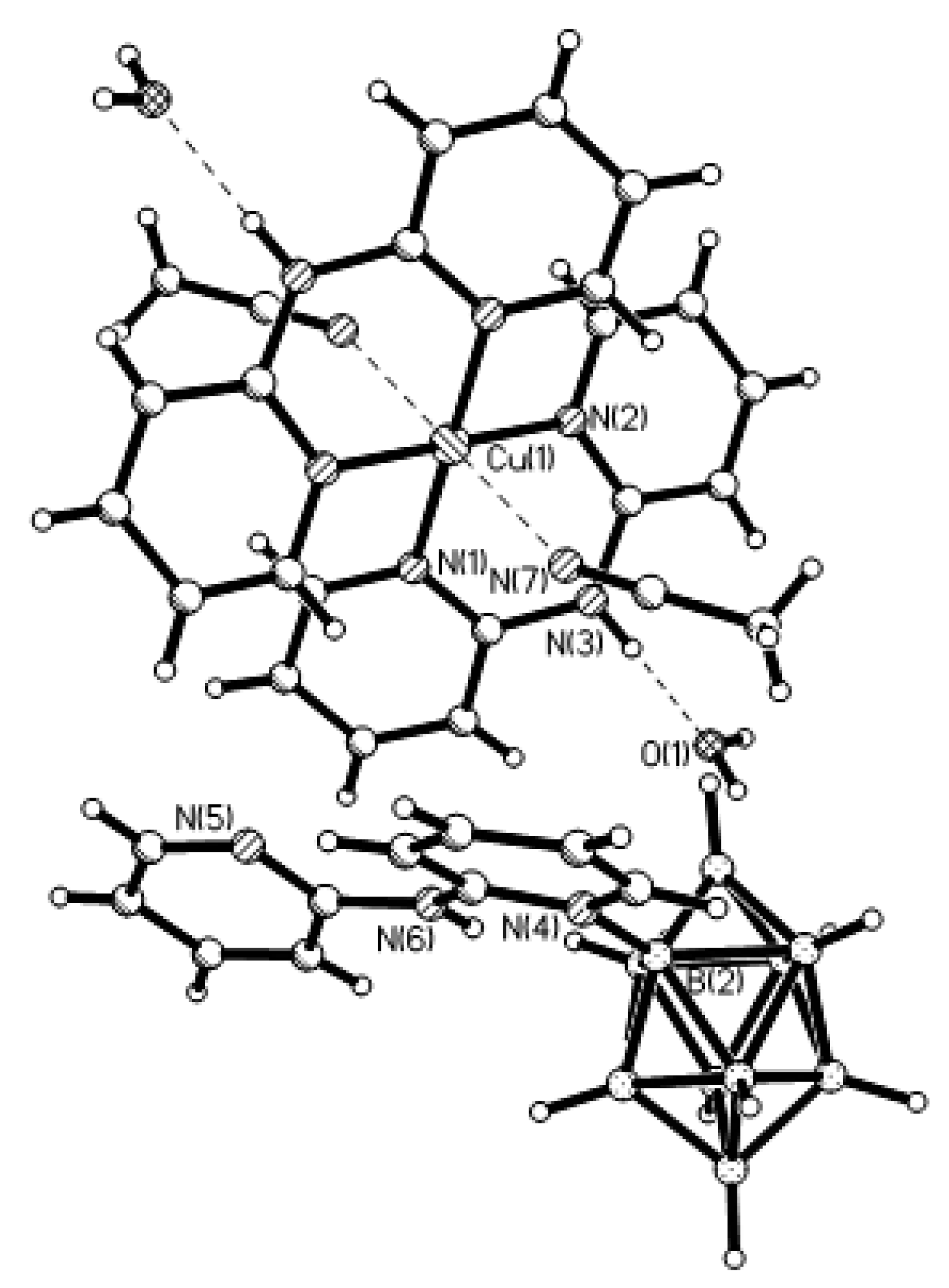
2+ ions is bound to a bidentate 2,2′-bipyridine molecule and two or three bridging OH groups, which form a square (the Cu(1) and Cu(4) atoms) or a tetragonal pyramid (the Cu(2) and Cu(3) atoms). The Cu-N bond lengths are within 1.973–2.014(5) Å. The Cu-O bond lengths in the squares formed by the Cu(1) and Cu(4) atoms and in the bases of the pyramids formed by the Cu(2) and Cu(3) atoms are within 1.909–1.956 and 1.921–1.986 Å, respectively. The apical O atoms are at Cu-O distances of 2.342(4) and 2.384(4) Å from the Cu(2) and Cu(3) atoms, respectively. The BH groups of the terminal polyhedral anions of the oligomer complete the coordination of the Cu(1) and Cu(4) atoms to a square pyramid (4 + 1) (Cu…B(H), 3.089(7) and 3.175(7) Å; Cu…H(B), 2.17 and 2.33 Å) and that of the Cu(2) and Cu(3) atoms, to a square bipyramid (4 + 1 + 1) (Cu…B(H), 3.485(8) and 3.364(7) Å; Cu…H(B), 2.91 and 2.54 Å). Weak Cu…B(H) interactions connect the [Cu
4(OH)
4(Bipy)
4]
4+ complex cations and [Cu
2[B
10H
10]
3]
4− complex anions into chains. The terminal [В
10H
10]
2− anions of the oligomeric anion link the Cu
2+ and Cu
+ atoms forming Cu
2+···В
10H
10···Cu
+ bridges (
Figure 8).
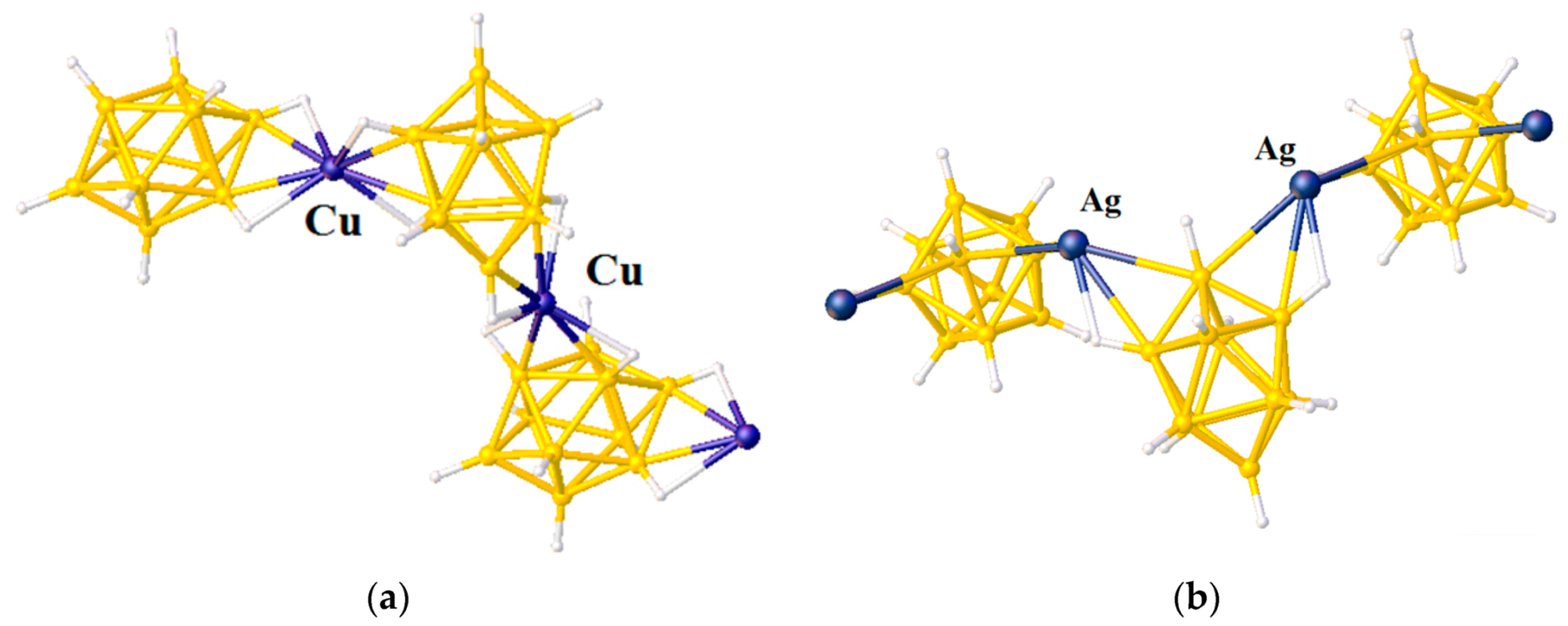
Solvent coordinated complexes of transition metals are widely regarded as useful starting materials in synthetic chemistry since their weakly coordinating solvent ligands, e.g., acetonitrile and tetrahydrofuran, can be easily replaced by more strongly coordinating ligands. In this way, they can be also applied as building blocks for inorganic and organometallic macromolecules.
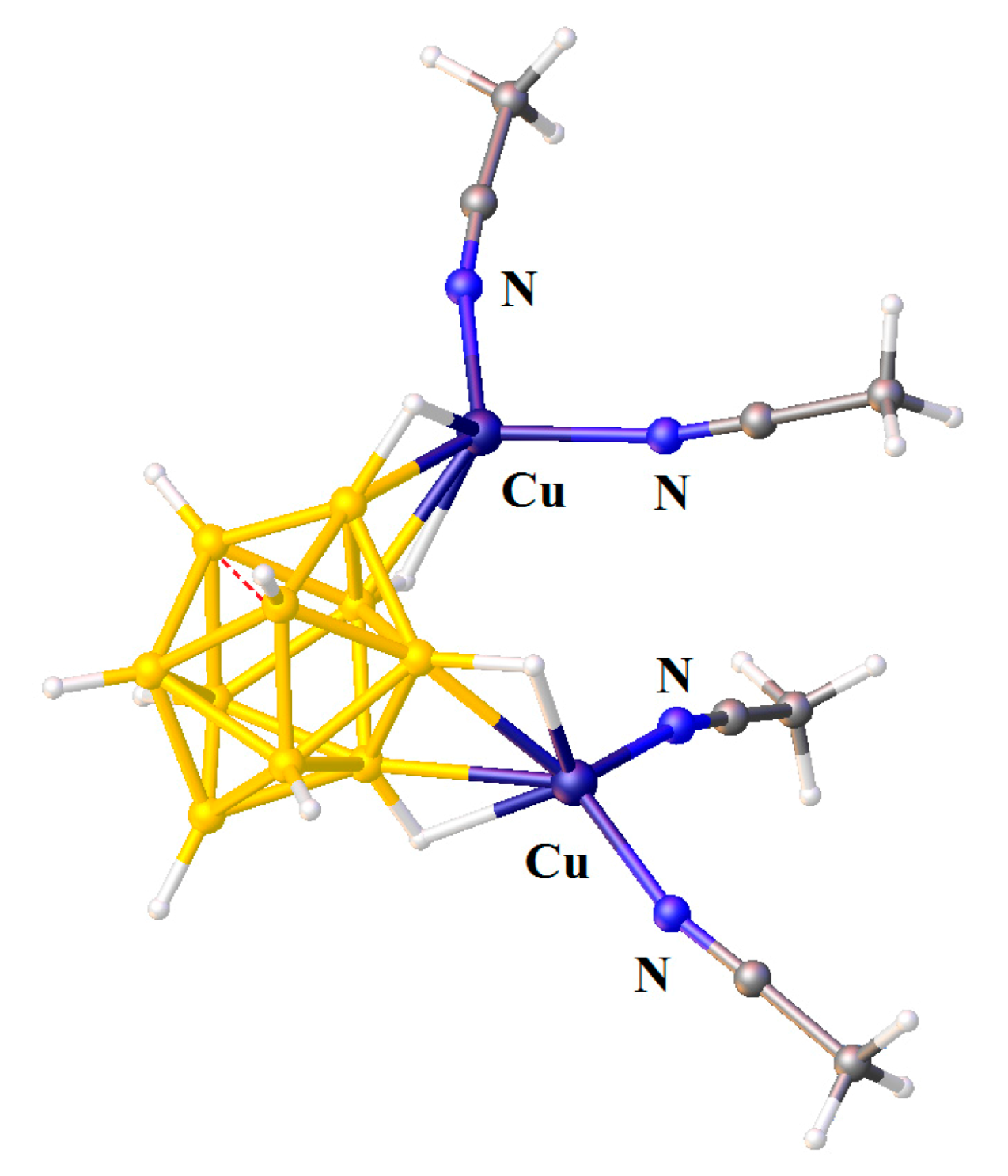
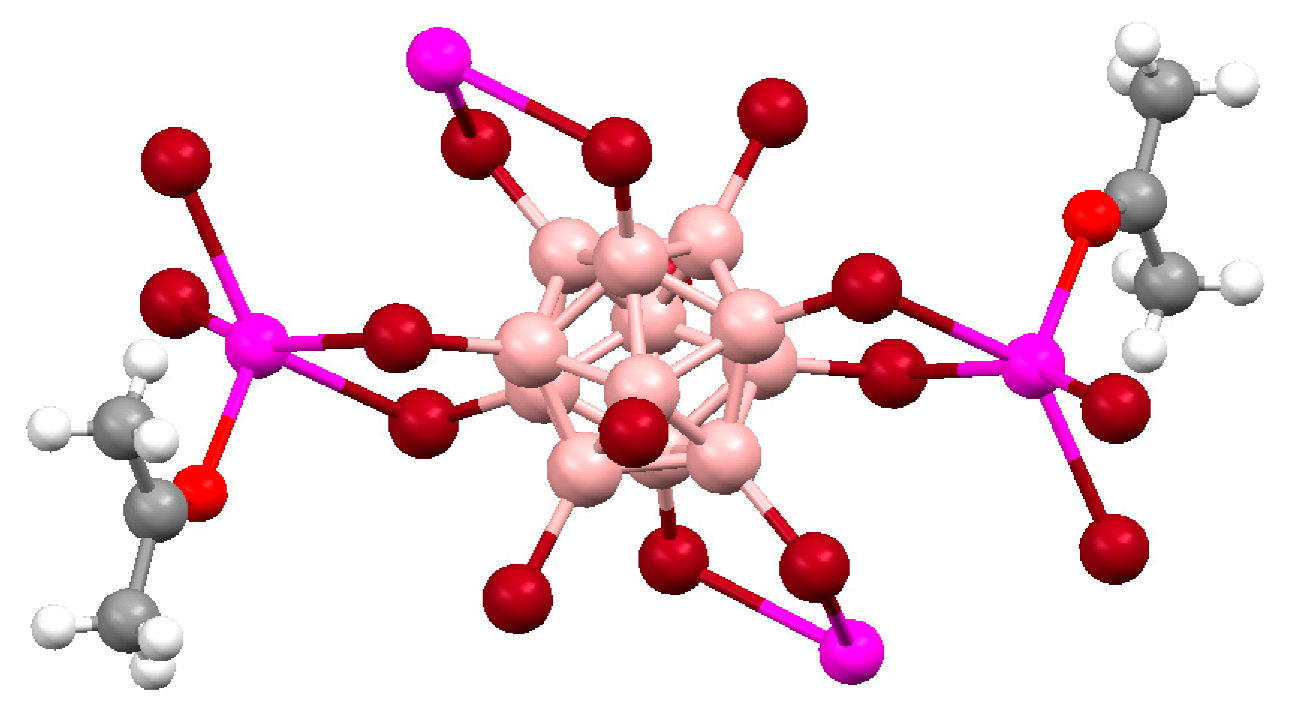
Recrystallization of {Cu
2[B
10H
10]}
n from acetonitrile solution in the presence of trifluoroacetic acid gave complex {((MeCN)
2Cu)
2[B
10H
10]} with two [(MeCN)
2Cu]
+ fragments coordinated to B(1)-B(2) and B(3)-B(6) edges [
28] (
Figure 9).
Complex {[(MeCN)
3Ag](Ag[2-B
10H
9F])}
2 was prepared by the treatment of (Ph
4P)
2[2-B
10H
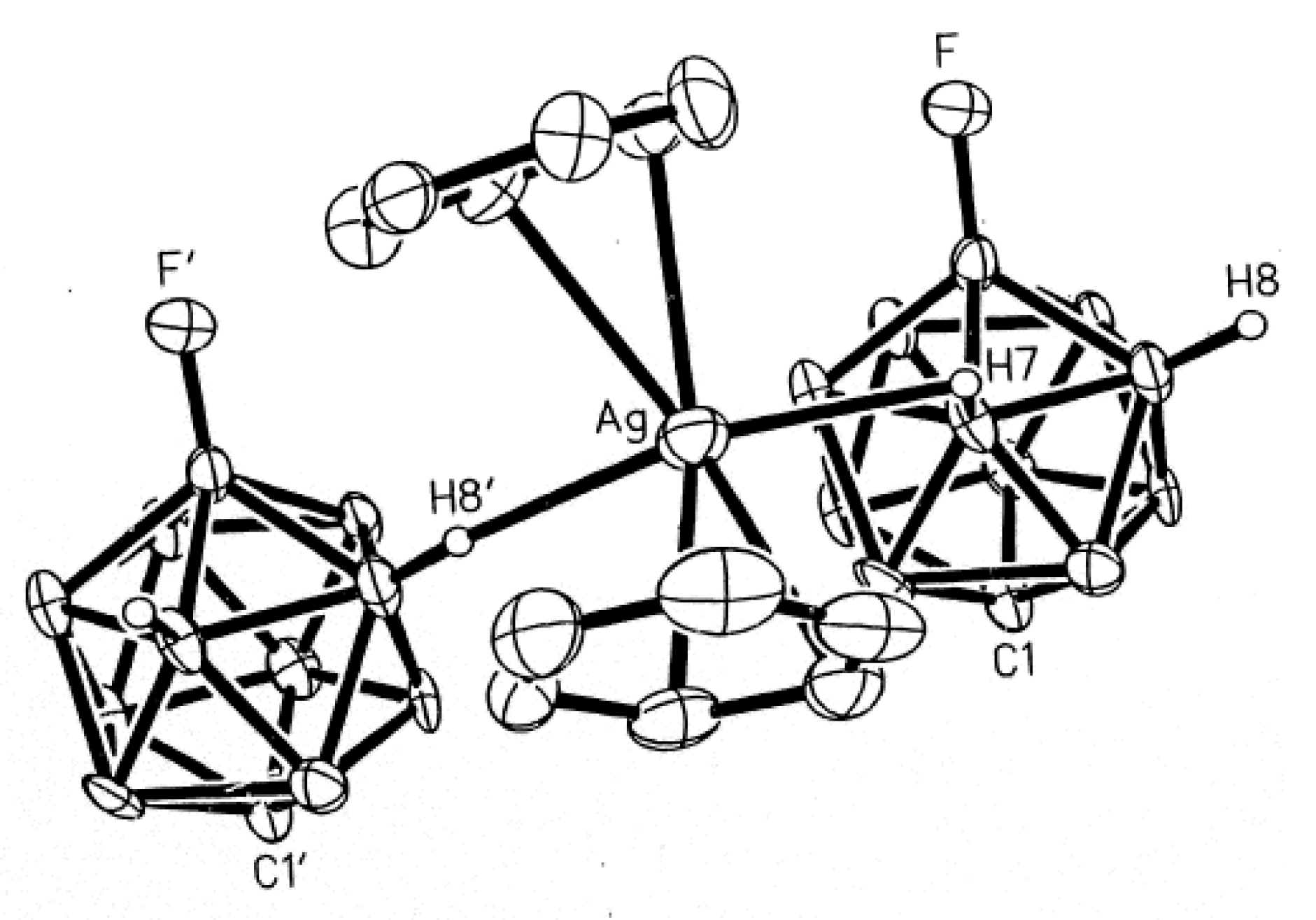
9F] with silver trifluoroacetate in 9:1 dichloromethane-toluene solution followed by crystallization from acetonitrile. The structure of the {[(MeCN)
3Ag](Ag[2-B
10H
9F])}
2 complex consists of dimeric {Ag[2-B
10H
9F]}
22− units weakly interacting with two nearly planar [(MeCN)
3Ag]
+ cations (Ag…B and Ag…H(B) distances are 2.520–2.644(5) and 2.02–2.36(5) Å, respectively) [
32]. There is no interaction between the fluorine substituent in boron cage and either silver cation (
Figure 10) [
14].
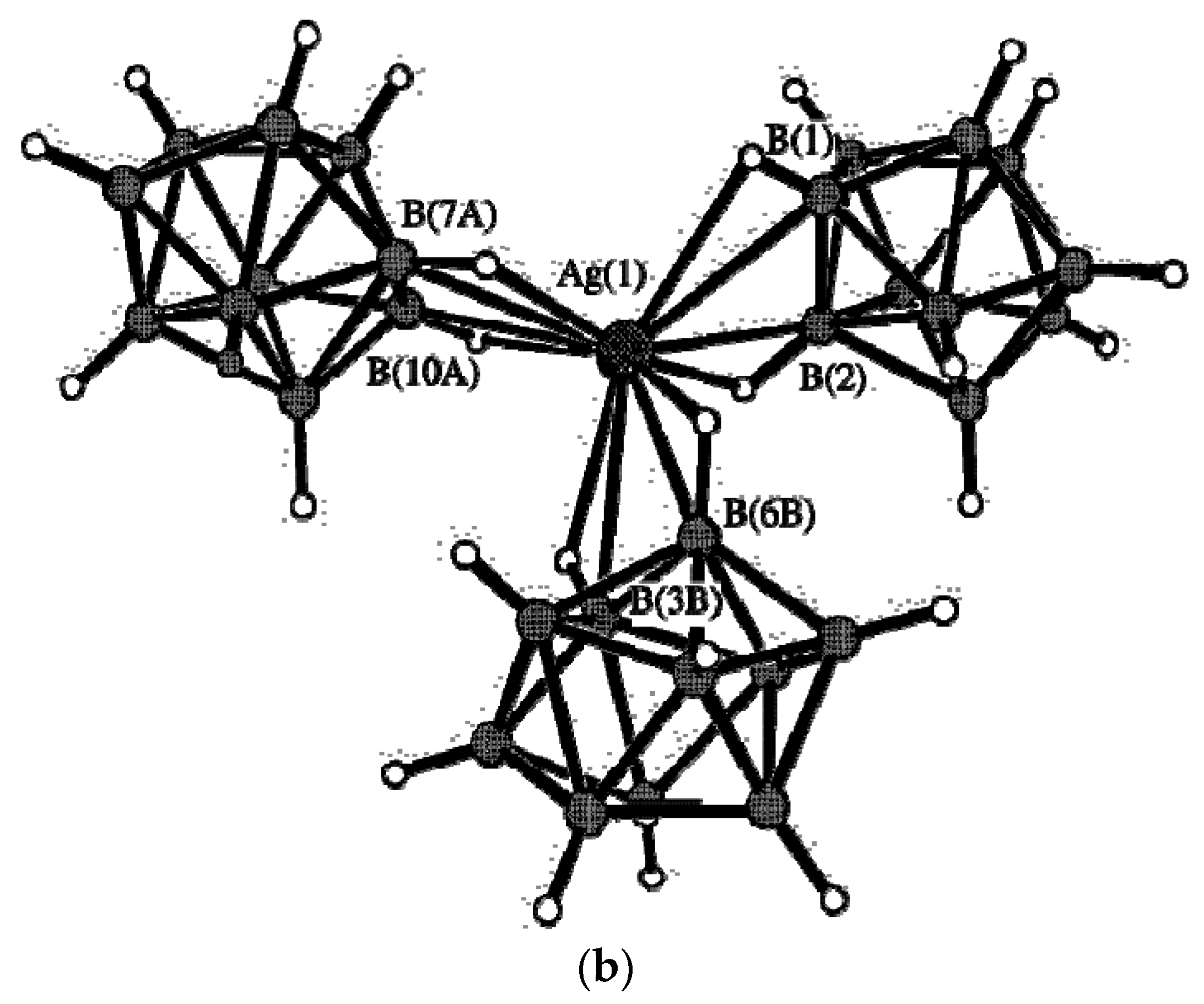
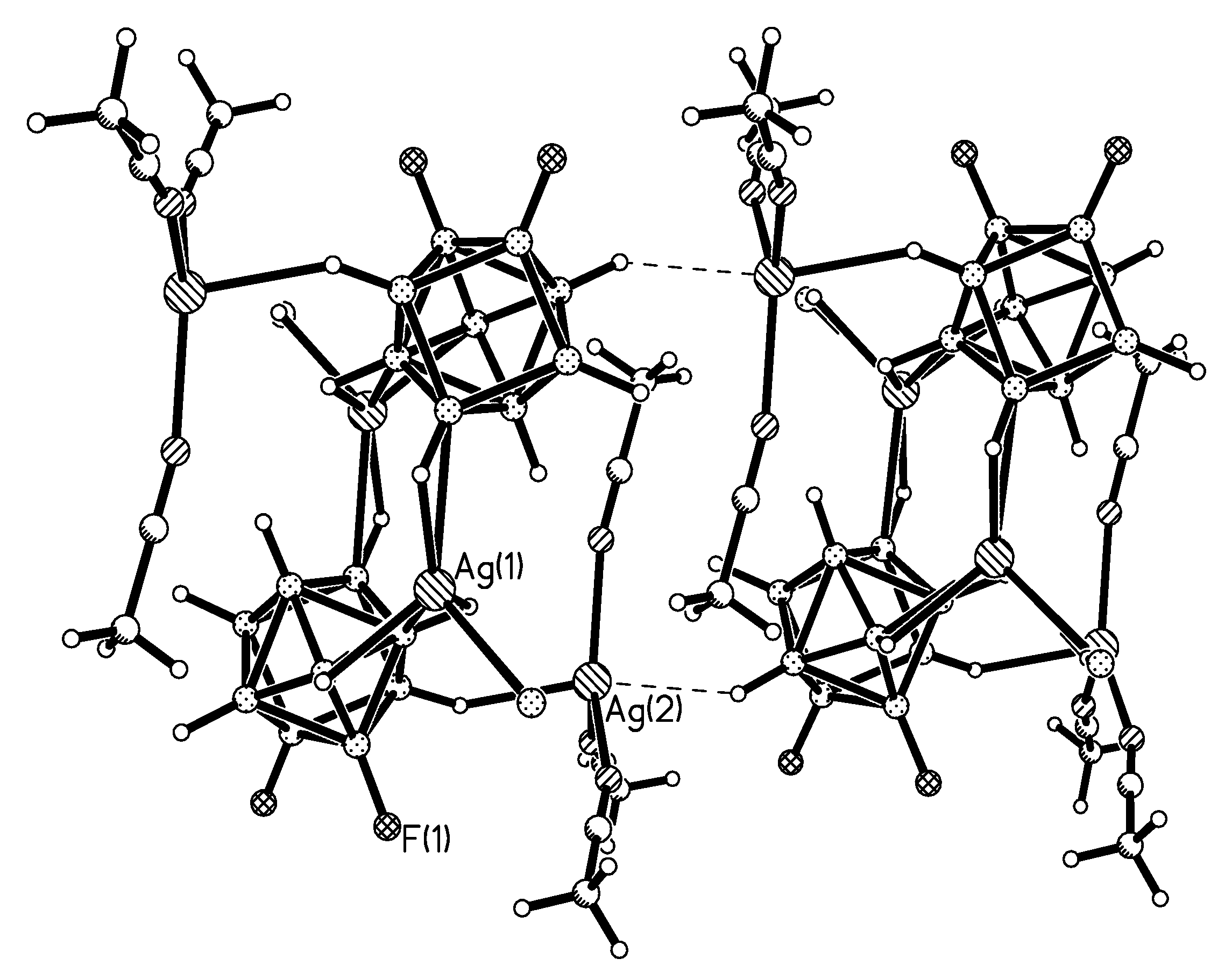
Complex {Ag
2(DMF)[B
10H
10]}
n was prepared by crystallization of Ag
2[B
10H
10] from N,N-dimethylformamide [
33]. The compound contains four types of silver atoms. The environment of the Ag(1) and Ag(4) atoms are formed by the O atoms of two DMF molecules and two BH groups of two
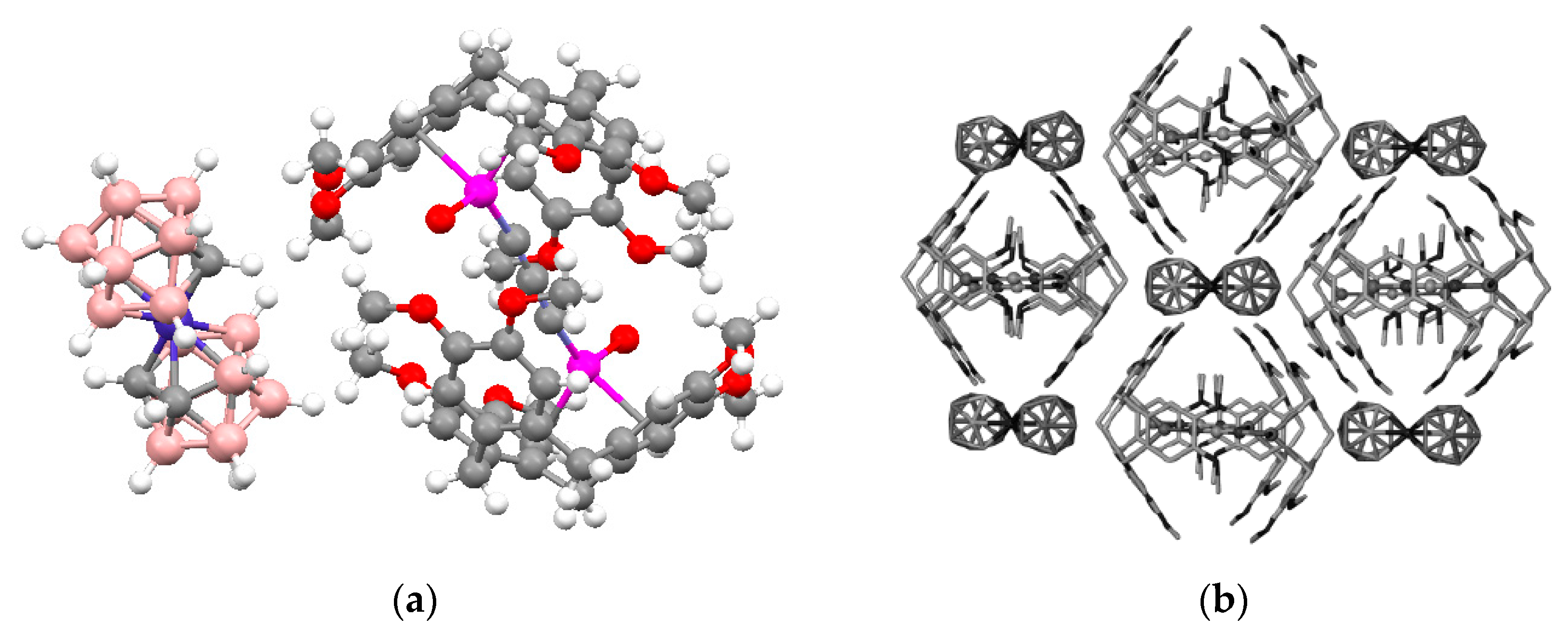
closo-decaborate anions (Ag…B distances 2.555(10) and 2.646(12) Å). Two other BH groups form additional elongated bonds (Ag…B distances 2.891(11) and 2.929(10) Å). Although the environment of Ag(1) and Ag(4) have the identical composition, they differ in the structure (
Figure 11a,b). The Ag(1) atom forms short Ag…B contacts (2.613(11) and 2.696(11) Å) with one
closo-decaborate anion and long Ag…B contacts (2.974(10) and 3.086(12) Å) with other one anion. The Ag(1) atom is coordinated by the B
a-B
e and B
e-B
e’ edges of the
closo-decaborate anion, whereas the Ag(4) atom is coordinated only by B
a-B
e edges. The environment of the Ag(2) and Ag(3) atoms includes only
closo-decaborate anions (
Figure 11c,d).
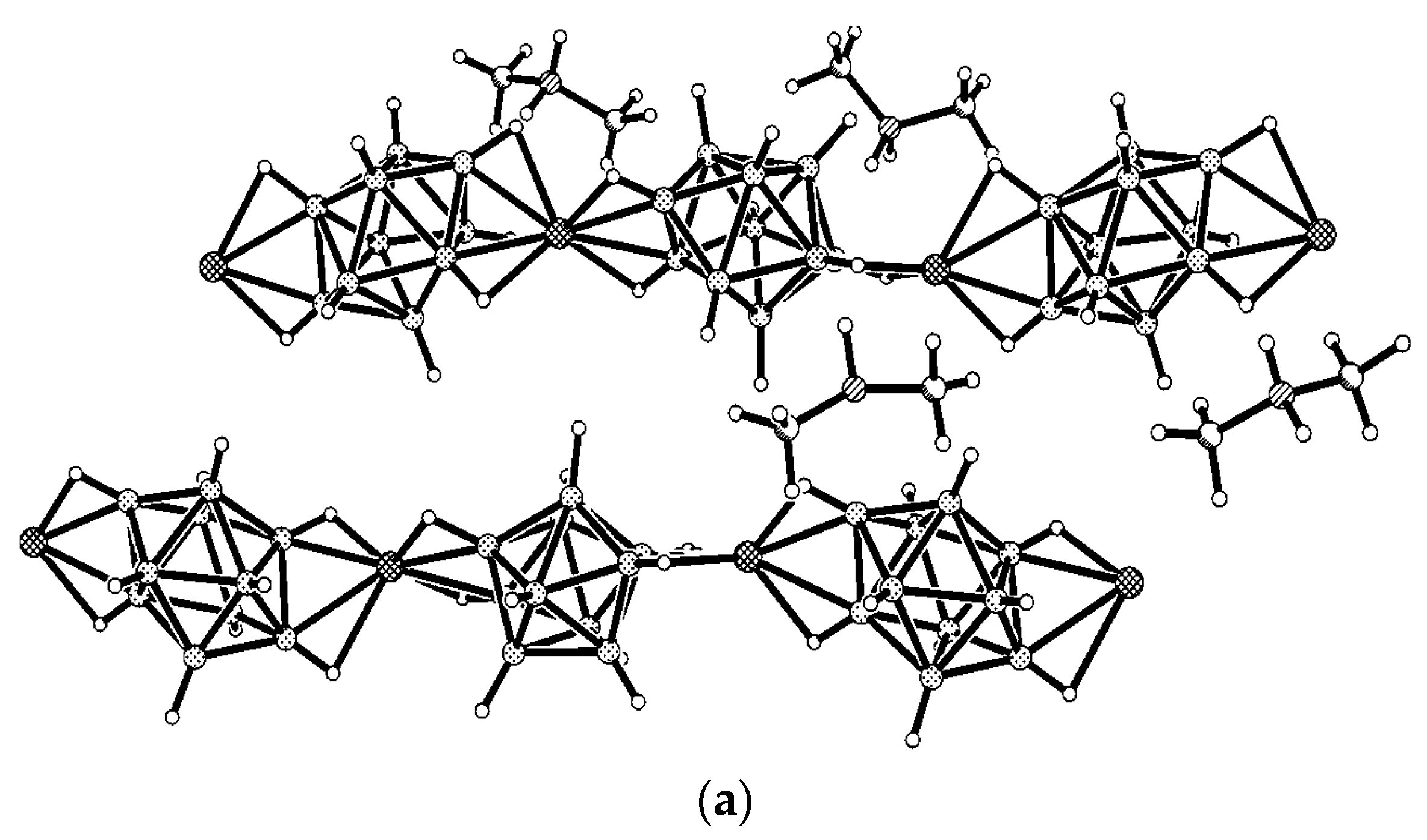
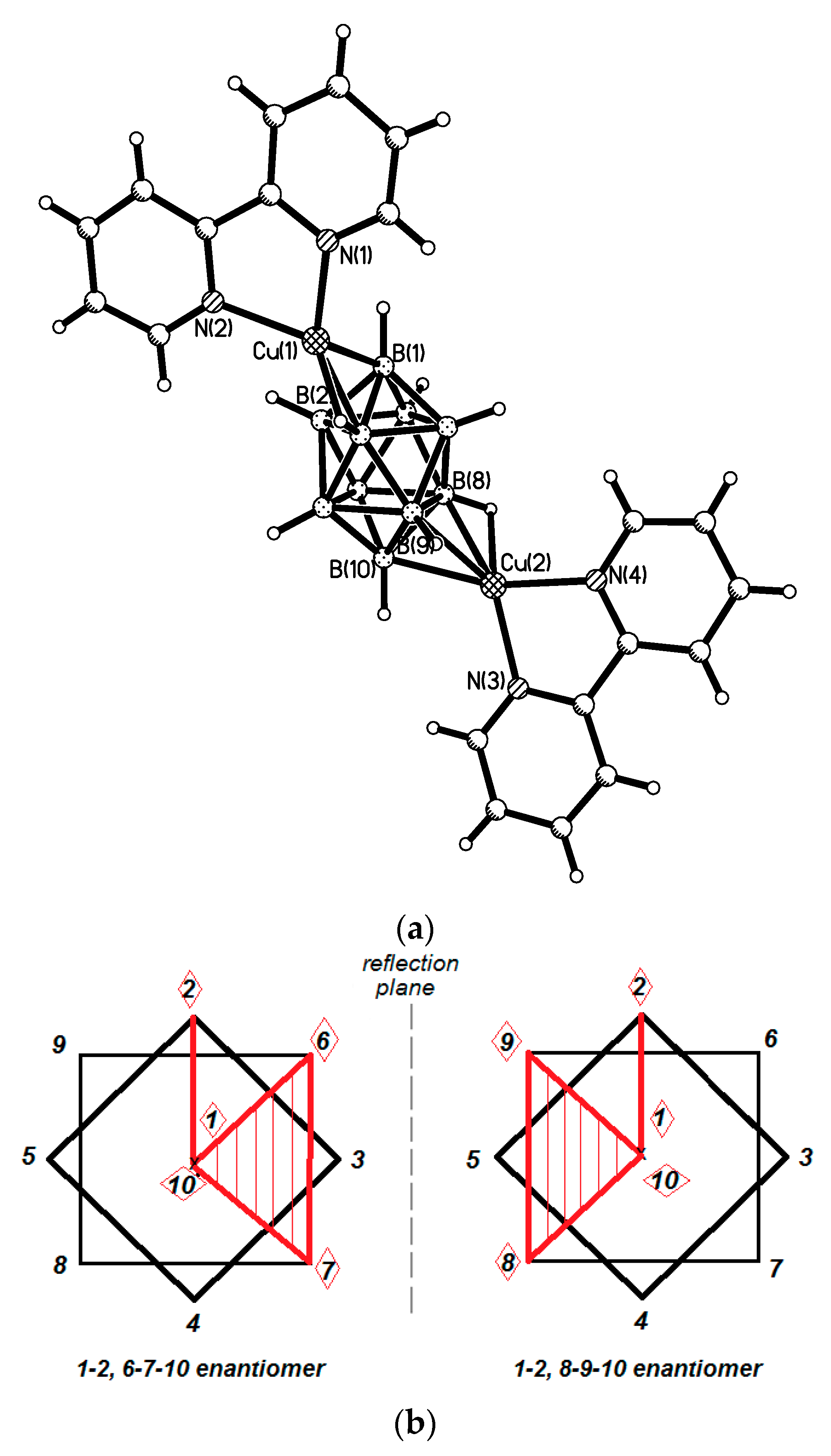
Formation of polymeric complexes with the closo-decaborate anion can be avoided using ligands that can saturate effectively the coordination sphere of the metal atom. Taking in the account that tetrahedral environment is the most preferred both for copper(I) and for silver(I) complexes, it can be expected that the closo-decaborate anion should form discrete complexes of {(L2M)2[B10H10]} type (where M = Cu, Ag). Indeed, a large number of such complexes were synthesized and structurally characterized. The {L2M}+ fragments have two free coordination places and are coordinated to the [B10H10]2− anion through two BH groups of one edge in chelate-like fashion. Due to geometrical features of the closo-decaborate anion there are a total of 25 possible isomers for the {(L2M)2[B10H10]} complexes, 17 of which are chiral. Naturally, not all of these coordination modes can be realized due to the reaction ability of the boron clusters, steric hindrances of ligands, and other factors. Nevertheless, a number of these positional isomers were already obtained and structurally characterized.
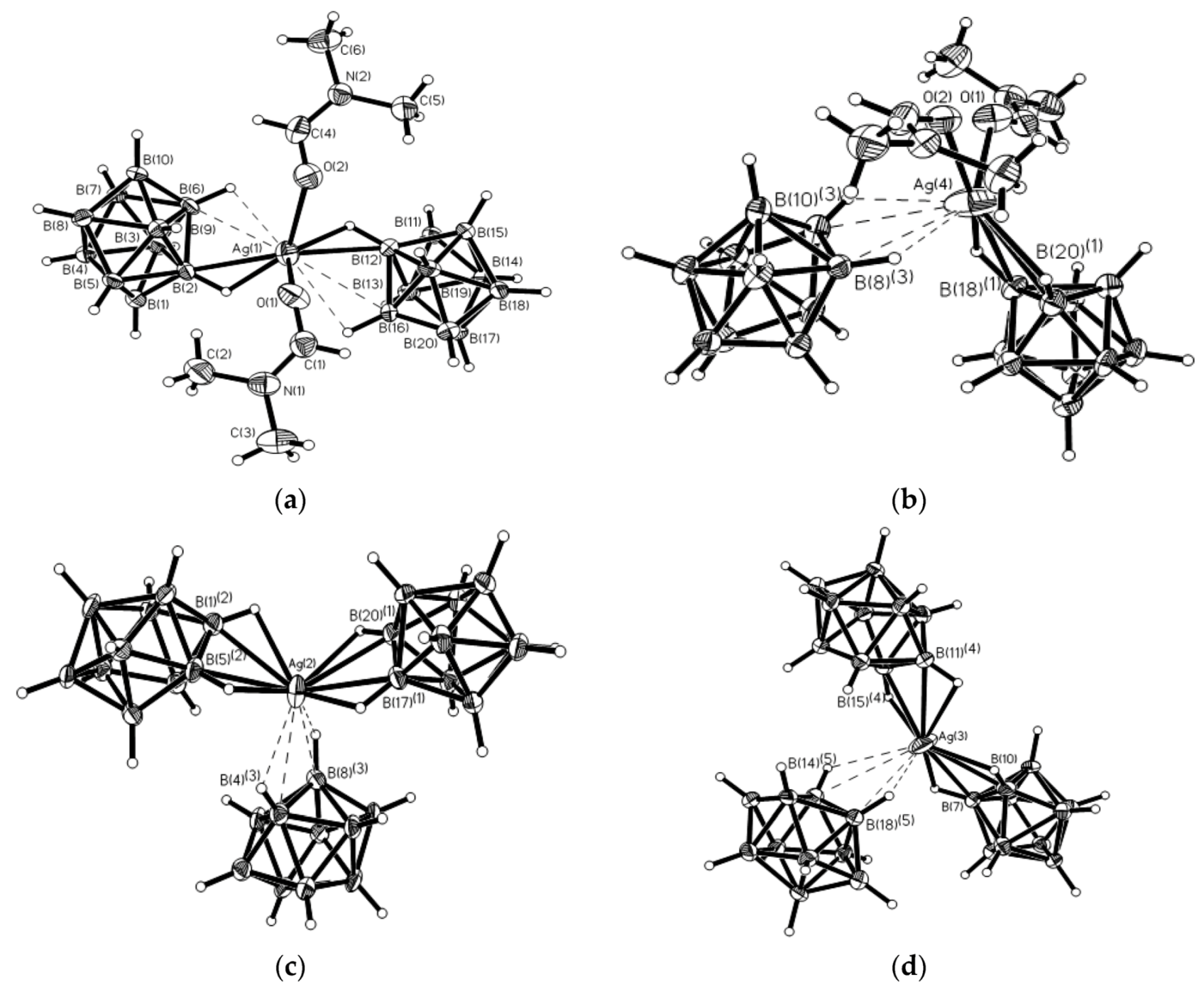
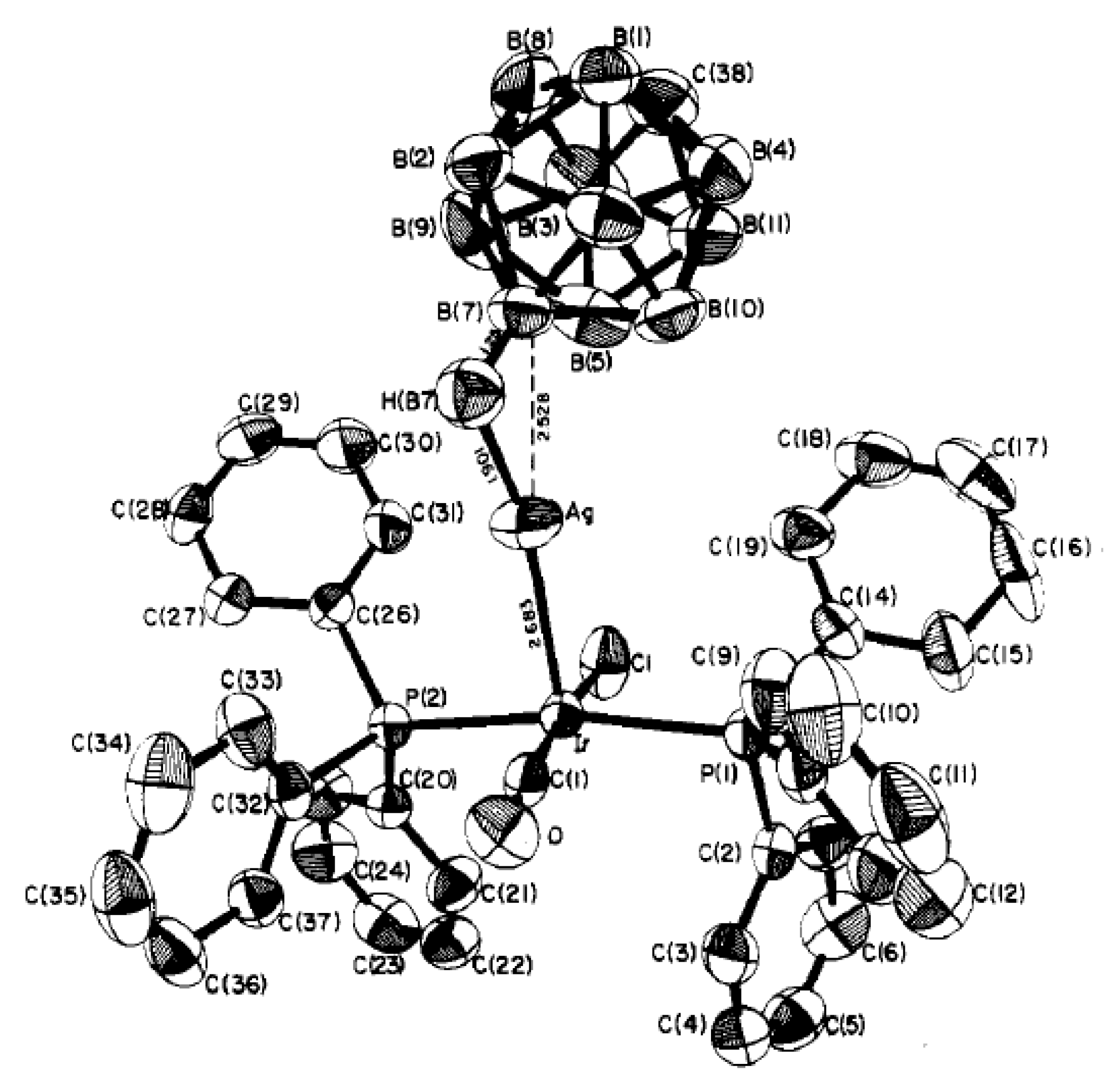
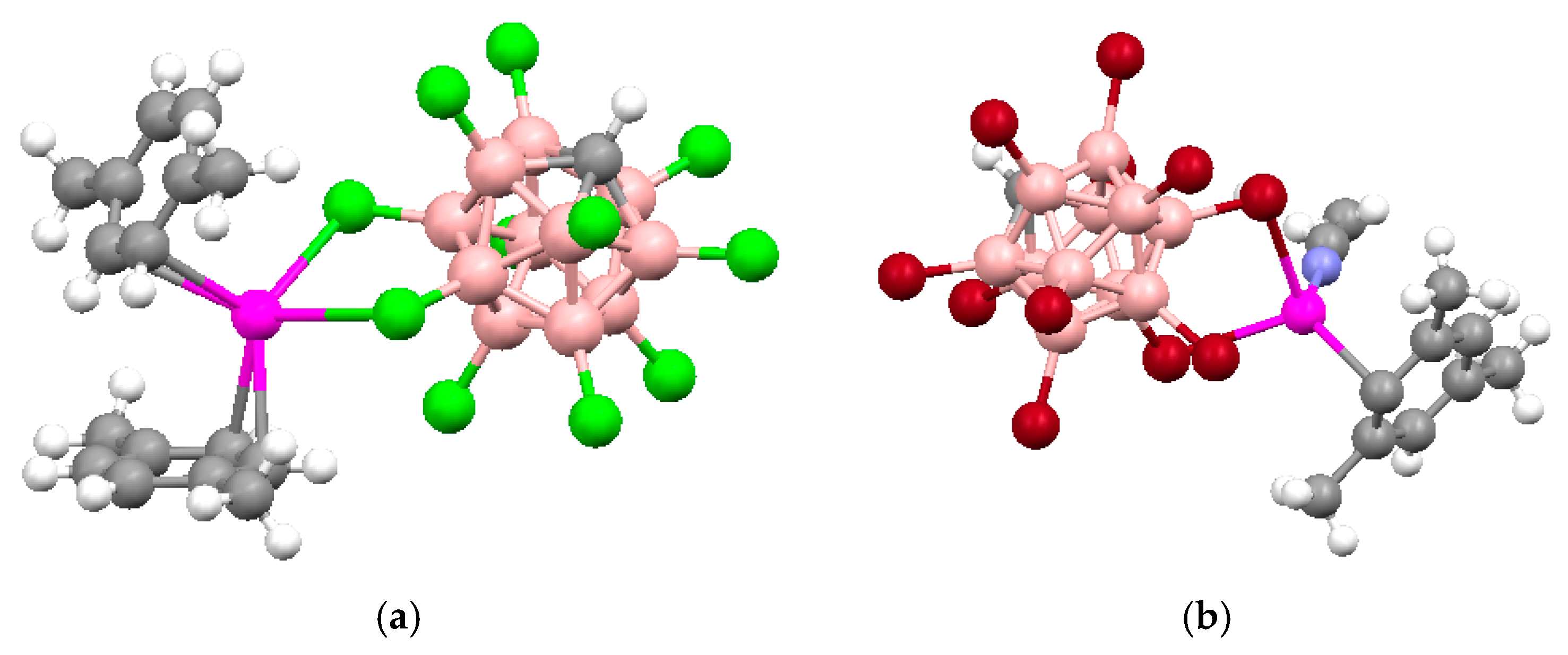
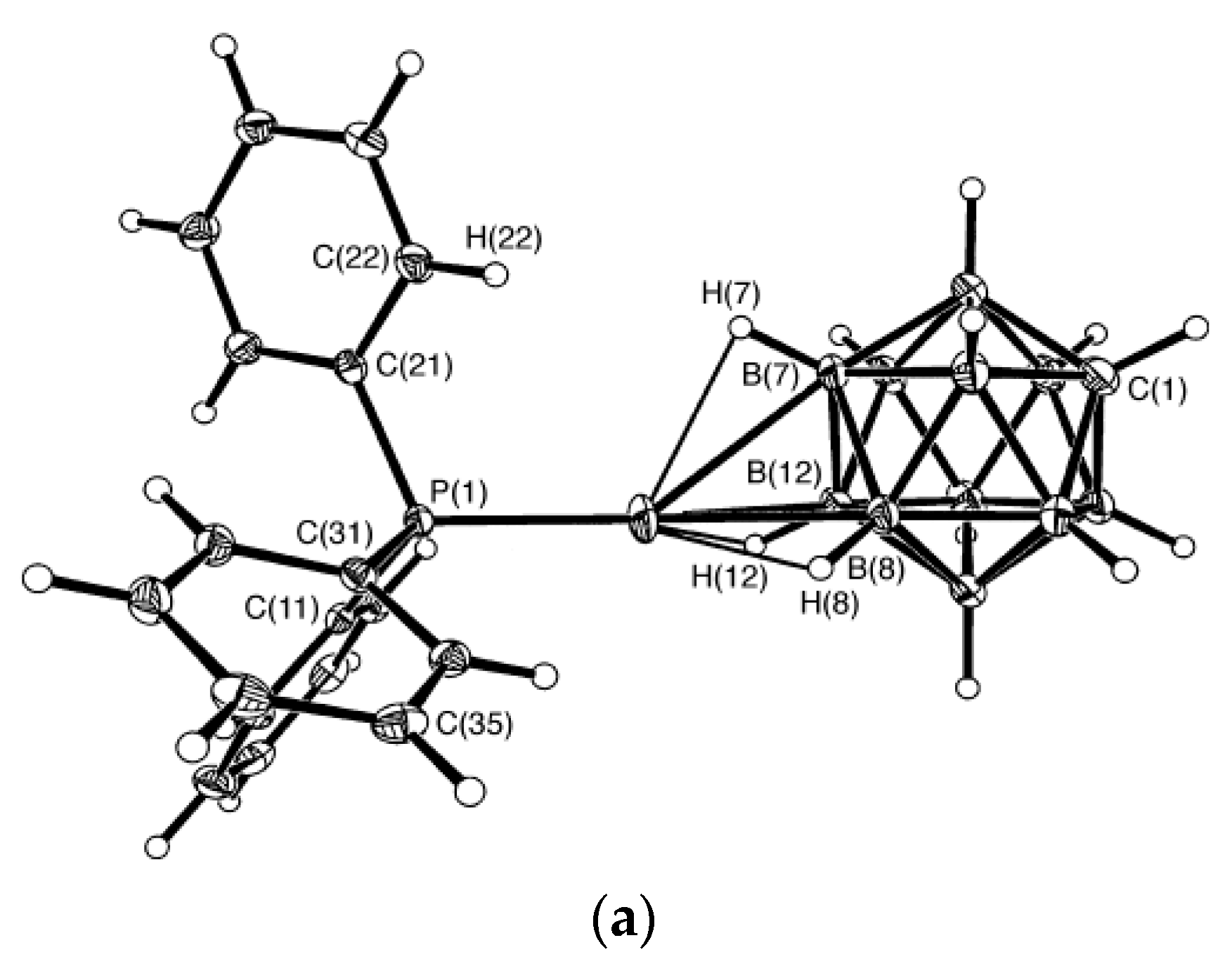
Synthesis of the first complex of this type {((Ph
3P)
2Cu)
2[B
10H
10]} by the reaction of (Et
3NH)
2[B
10H
10] with [(Ph
3P)
3CuCl] in chloroform was described by Gill and Lippard in 1975 [
34]. The copper atoms are in a quasi-tetrahedral environment formed by two phosphorus atoms of the Ph
3P ligands and two BH groups of the boron polyhedron. The
closo-decaborate anion coordinates metal atoms by B
a-B
e edges at different apical vertices of the boron polyhedron. Two enantiomers of {((Ph
3P)
2Cu)
2[B
10H
10]} were found in the crystal. In one enantiomer, coordination proceeds via B(1)-B(2) and B(6)-B(10) edges, whereas the other enantiomer—via B(1)-B(2) and B(9)-B(10) edges. The same compound was isolated by interaction of Cat
2[B
10H
10] (Cat
+ = Bu
4N
+, Ph
4P
+) with [(Ph
3P)
3CuCl] in acetonitrile [
35]. Structure of the 1–2, 9–10 enantiomer of {((Ph
3P)
2Cu)
2[B
10H
10]} is presented in
Figure 12a. The Cu…B bonds vary in the range from 2.260(9) to 2.317(9) Å, the Cu…HB distances range from 1.78 to 2.05 Å, the Cu-Н-В angles are 109–122°. The existence of the Cu-H-B interactions in the solid state is supported by the presence of the Cu-H-B stretching band in the IR spectrum of {((Ph
3P)
2Cu)
2[B
10H
10]} at 2150–2400 cm
−1 [
36]. The molecular conductance measurements indicate that the {((Ph
3P)
2Cu)
2[B
10H
10]} complex does not dissociate in dichloromethane solution [
34].
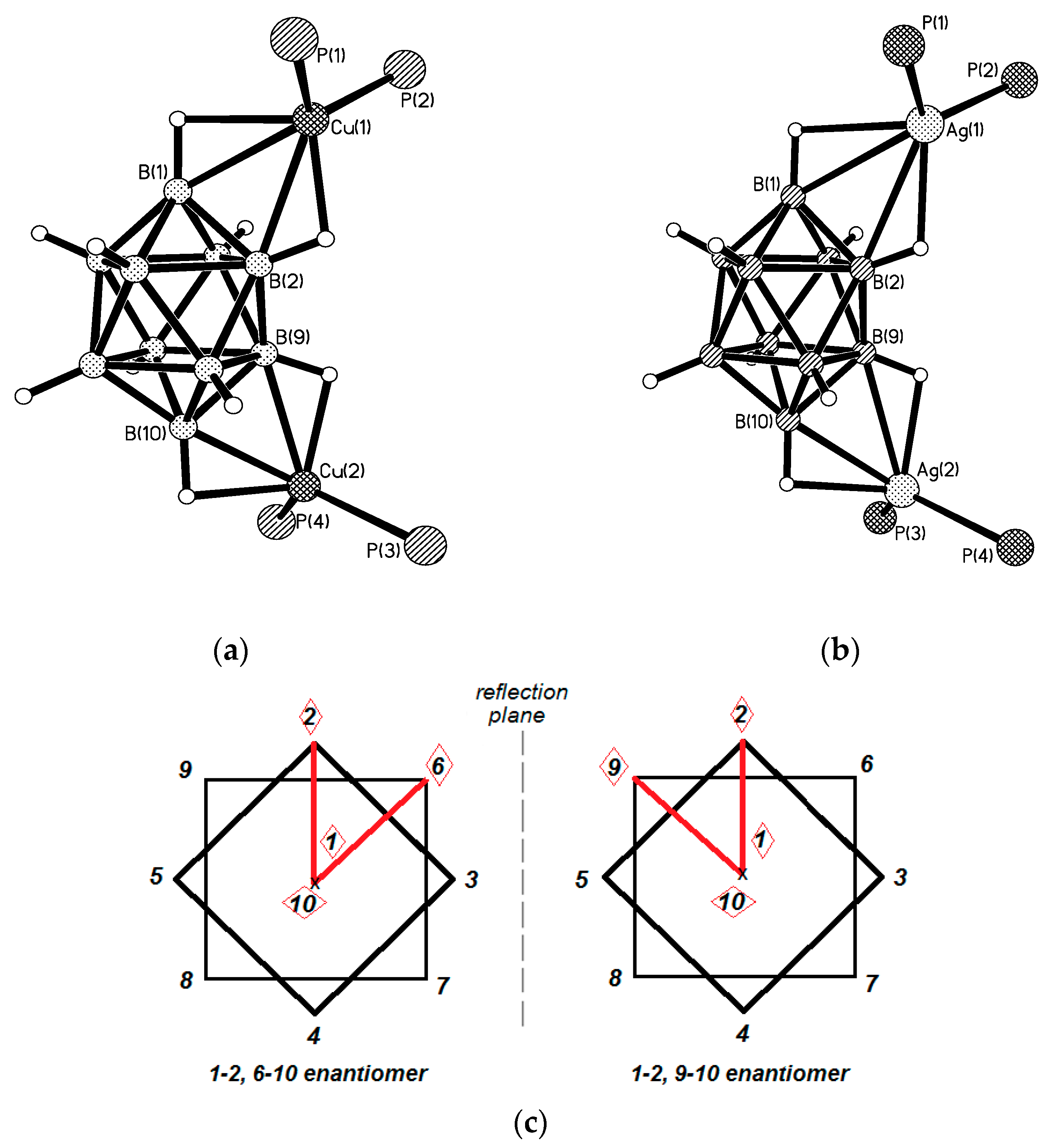
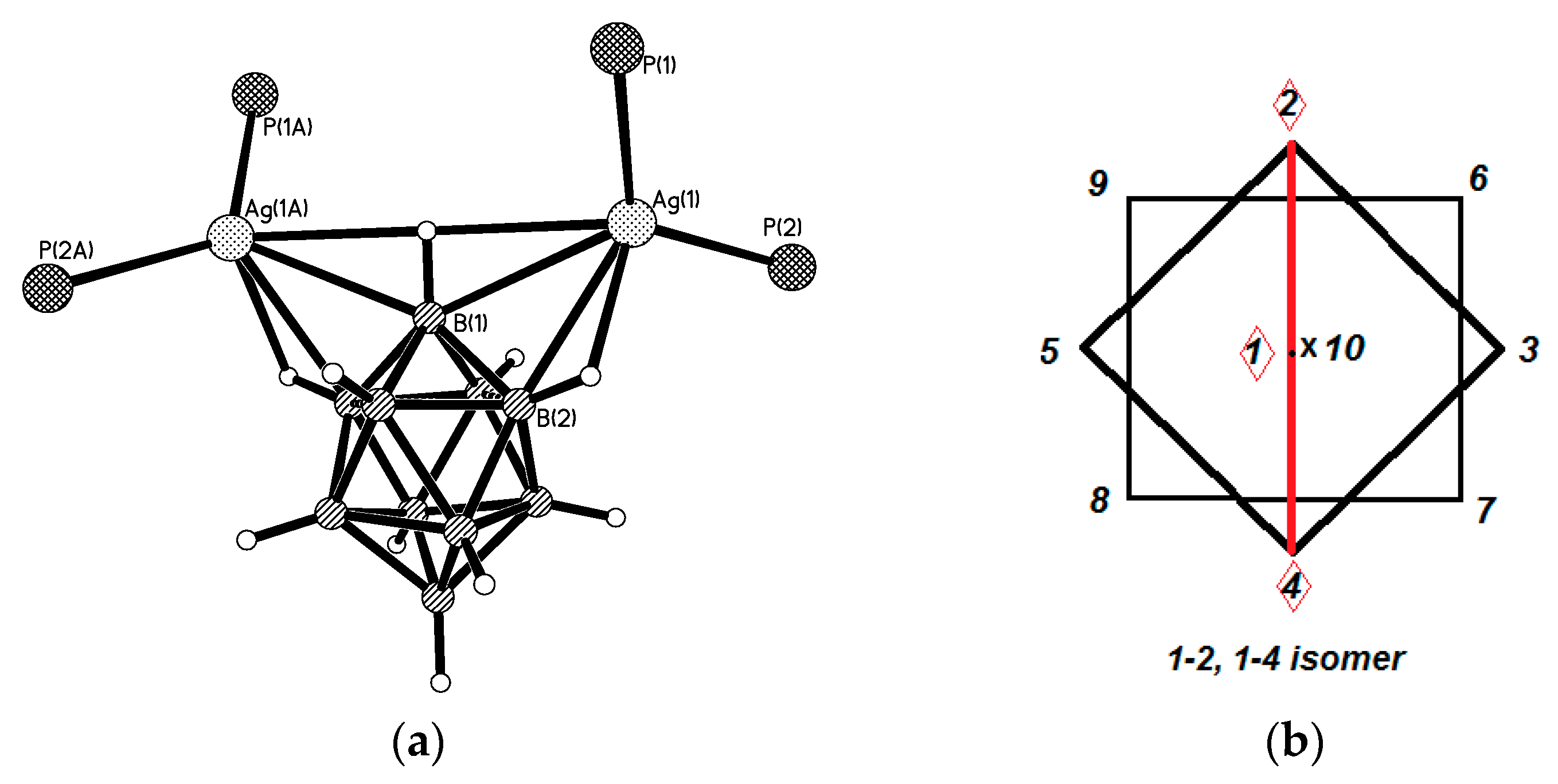
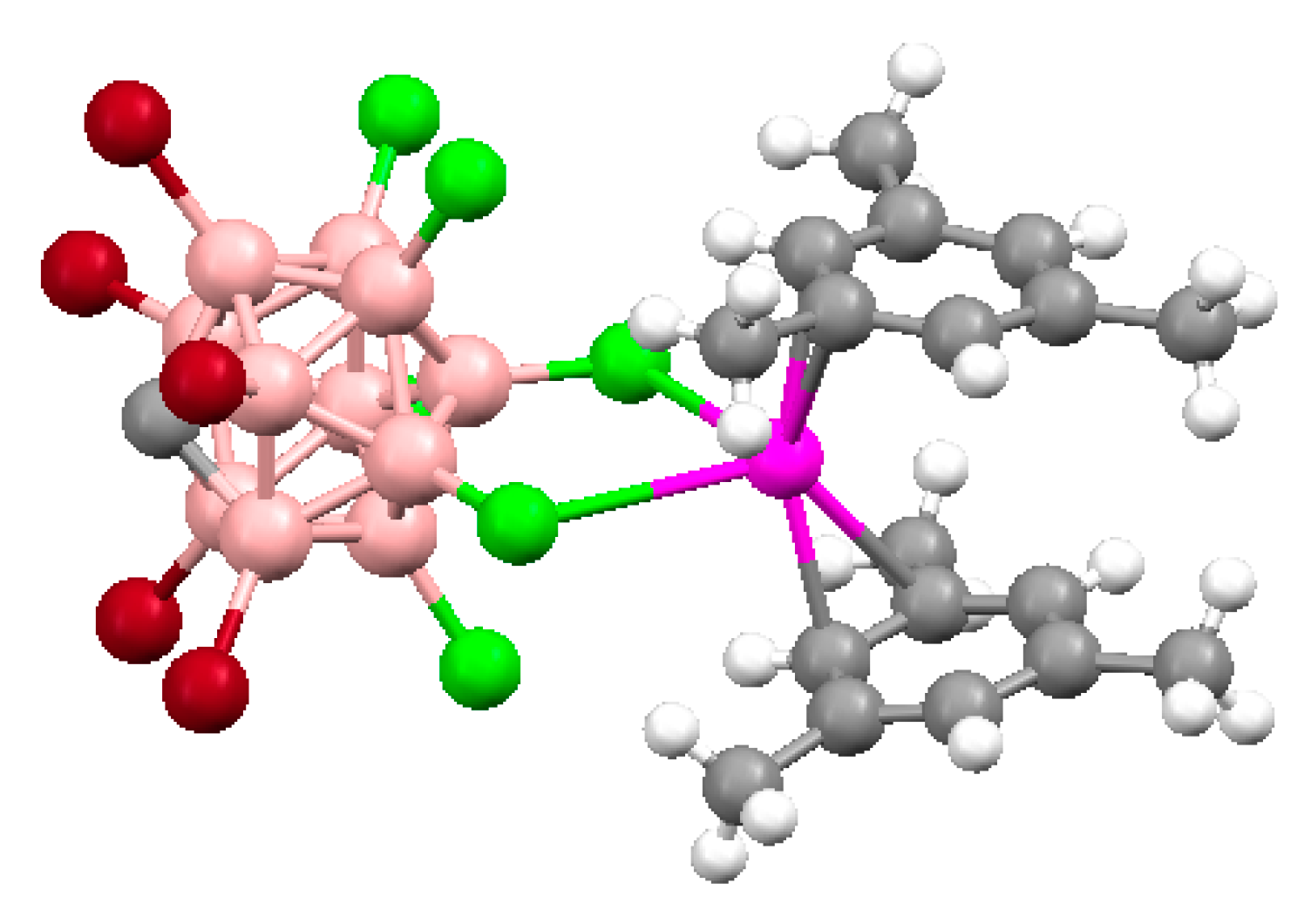
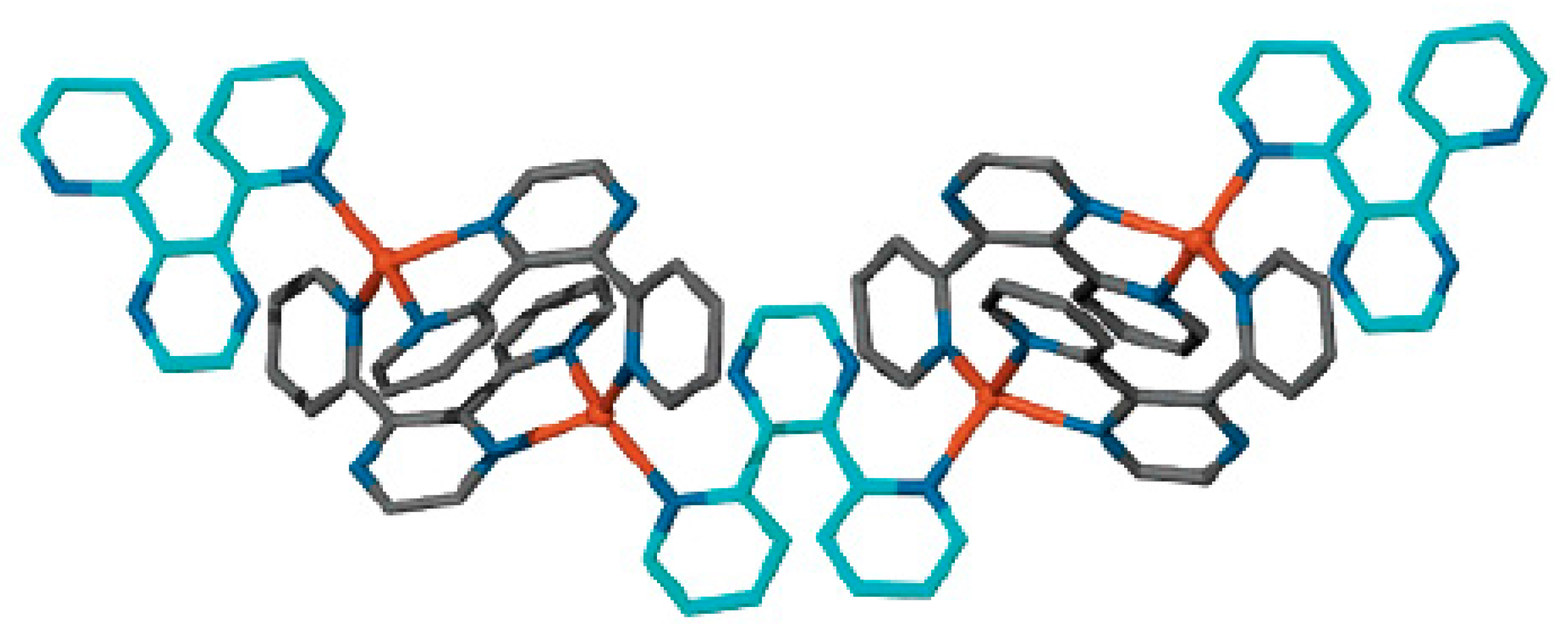
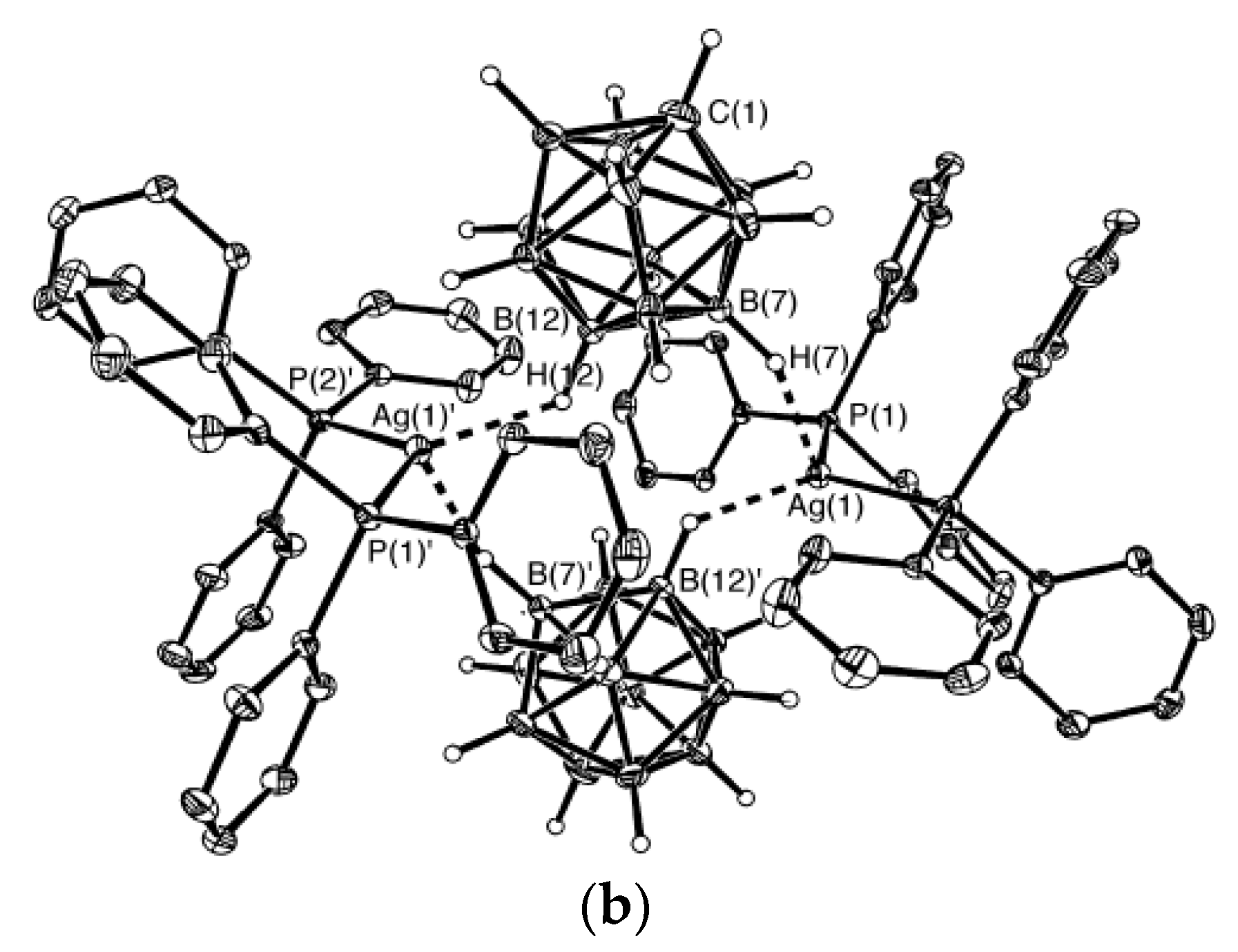
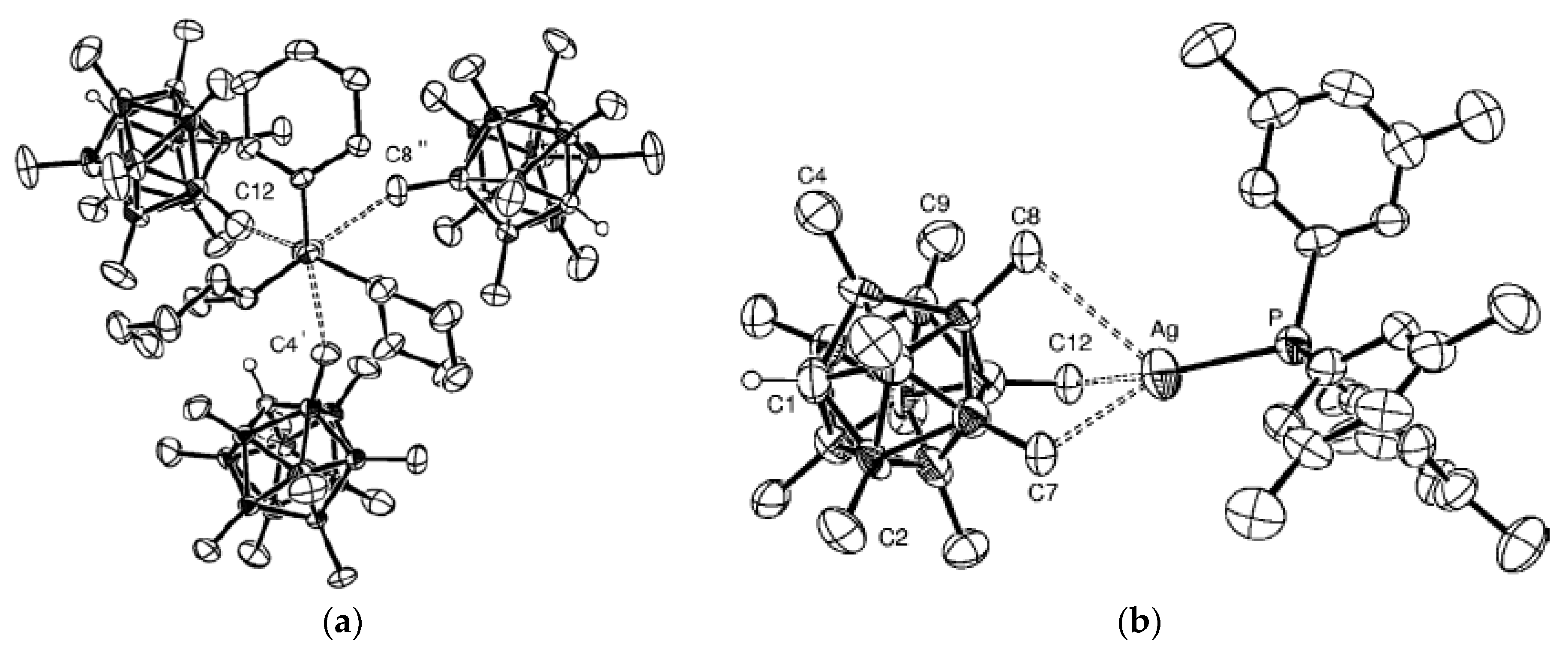
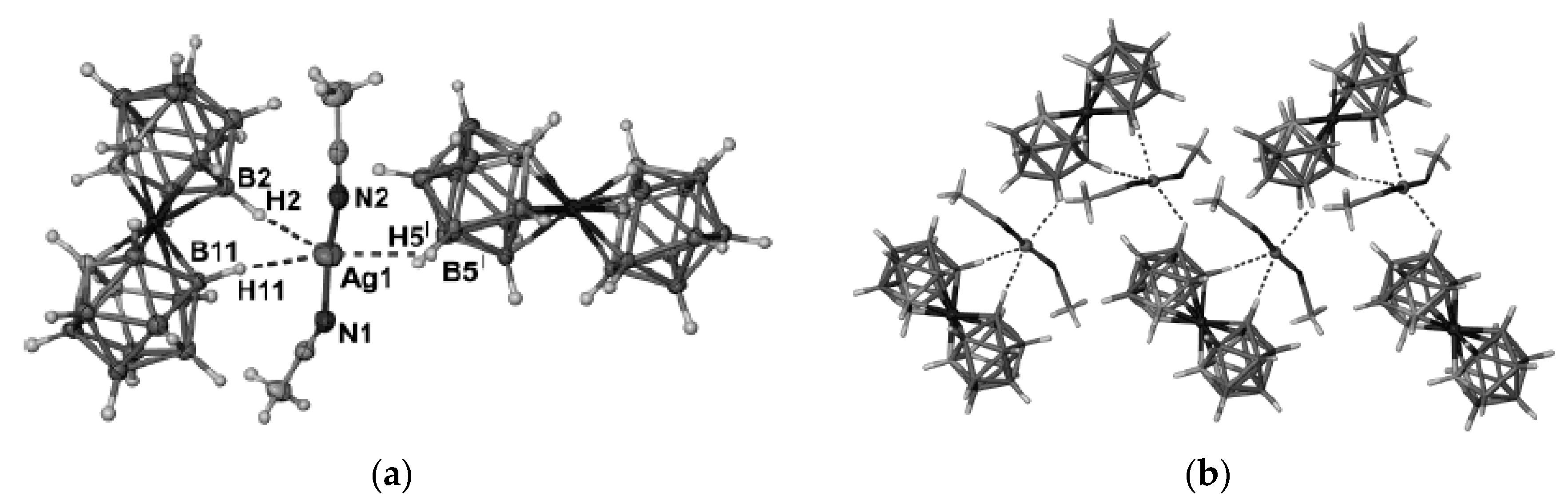
Similar silver(I) complex 1–2, 6(9)–10 {((Ph
3P)
2Ag)
2[B
10H
10]} was prepared by the reaction of (Et
3NH)
2[B
10H
10] with [(PPh
3)
3Ag]NO
3 in acetonitrile (
Figure 12b). The geometrical parameters of MHB bond are: Ag…B and Ag…HB bonds are 2.561(4)–2.617(4) and 1.97–2.29 Å, respectively; the Ag-Н-В angles are 109–122° [
35]. Schematic representation of both 1–2, 6–10 and 1–2, 9–10 enantiomers which are present in {((Ph
3P)
2M)
2[B
10H
10]} are shown in
Figure 12c.
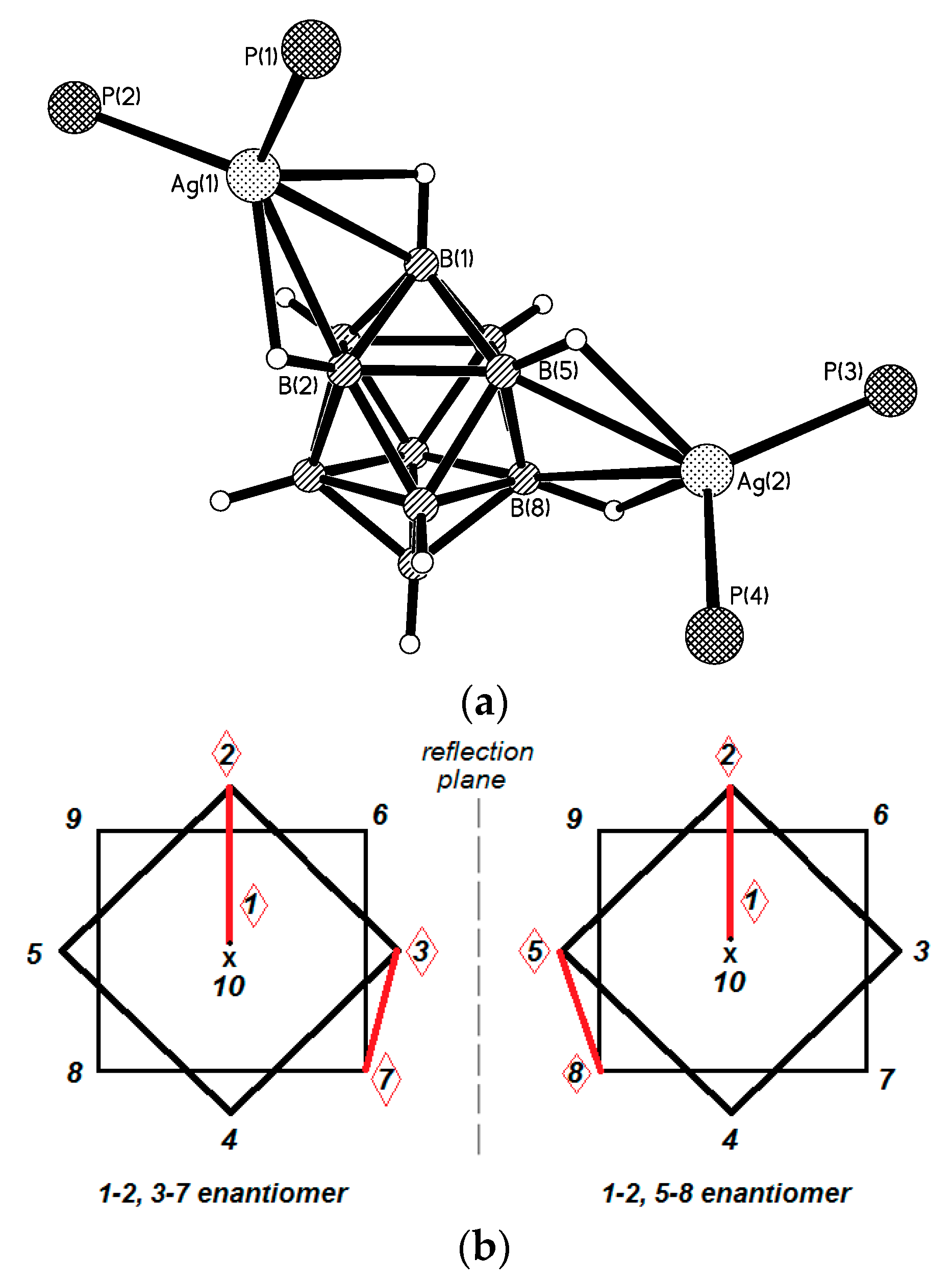
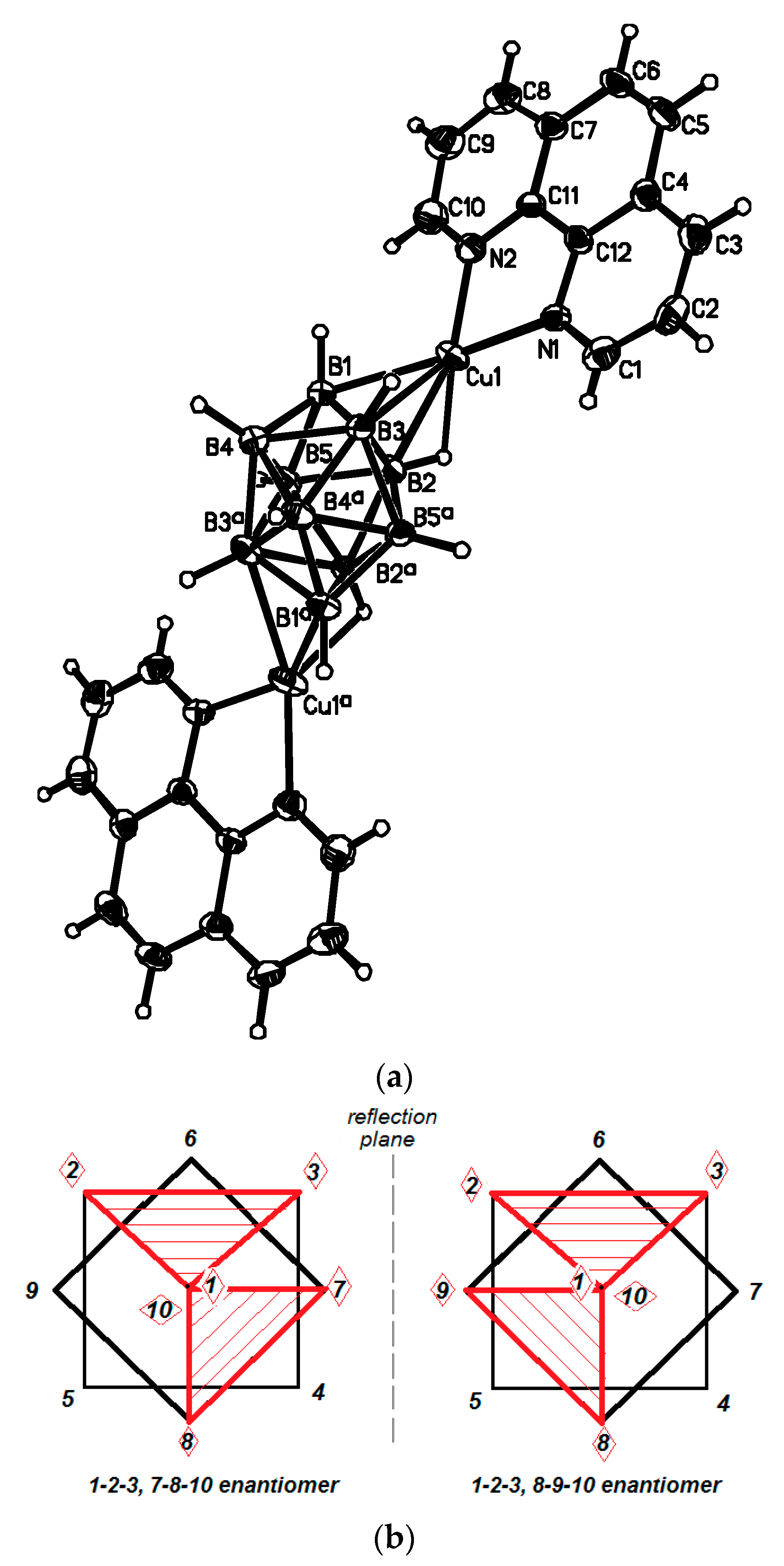
Another positional isomer of {((Ph
3P)
2Ag)
2[B
10H
10]} was prepared by the reaction of (Et
3NH)
2[B
10H
10] with [(Ph
3P)
3Ag]NO
3 in the acetonitrile in the presence of trifluoroacetic acid. In the obtained {((Ph
3P)
2Ag)
2[B
10H
10]} complex (
Figure 13), the [В
10Н
10]
2− anion is coordinated to silver atoms by B
a-B
e and B
e-B
e’ edges (1–2, 3–7 for one enantiomer and 1–2, 5–8 for another enantiomer) [
37]. The Ag(1)…B bonds with the B
a-B
e edge (2.577(3) and 2.519(3) Å) are shorter than the Ag(2)…B bonds with the B
e-B
e’ edge (2.653(3) and 2.799(3) Å). The Ag…H distances range from 2.06 to 2.34 Å, the Ag-H-B angles are 90.0–105.3°.
When the protonated form of the
closo-decaborate anion was used in complexation reactions, complexes with coordination of the both metal atoms by B
a-B
e edges near the same apical vertex were obtained. The reaction of (Ph
4P)[B
10H
11] with [(Ph
3P)
3Ag]NO
3 in acetonitrile results in 1–2, 1–4 {((Ph
3P)
2Ag)
2[B
10H
10]} [
35]. The Ag…B and Ag…H distances are 2.642(3)–2.783(4) and 2.12–2.55 Å, respectively, the Ag-H-В angles vary from 118 to 120°. It should be noted that the boron cage in the complex is distorted: the В(2)–В(3) bond is elongated and equal to 1.947(10), the В(4)–B(5) bond is shortened to 1.543(9) Å (
Figure 14).
The reactions of Ag
2[B
10H
10] with complex [(Ph
3P)
3Ag]NO
3 or free triphenylphosphine in refluxing acetonitrile unexpectedly afforded polymeric complex {((Ph
3P)Ag)
2[B
10Н
10]}. In the crystal each
closo-decaborate anion is coordinated by four {(Ph
3P)Ag}
+ fragments to form polymeric chain (
Figure 15) [
38].
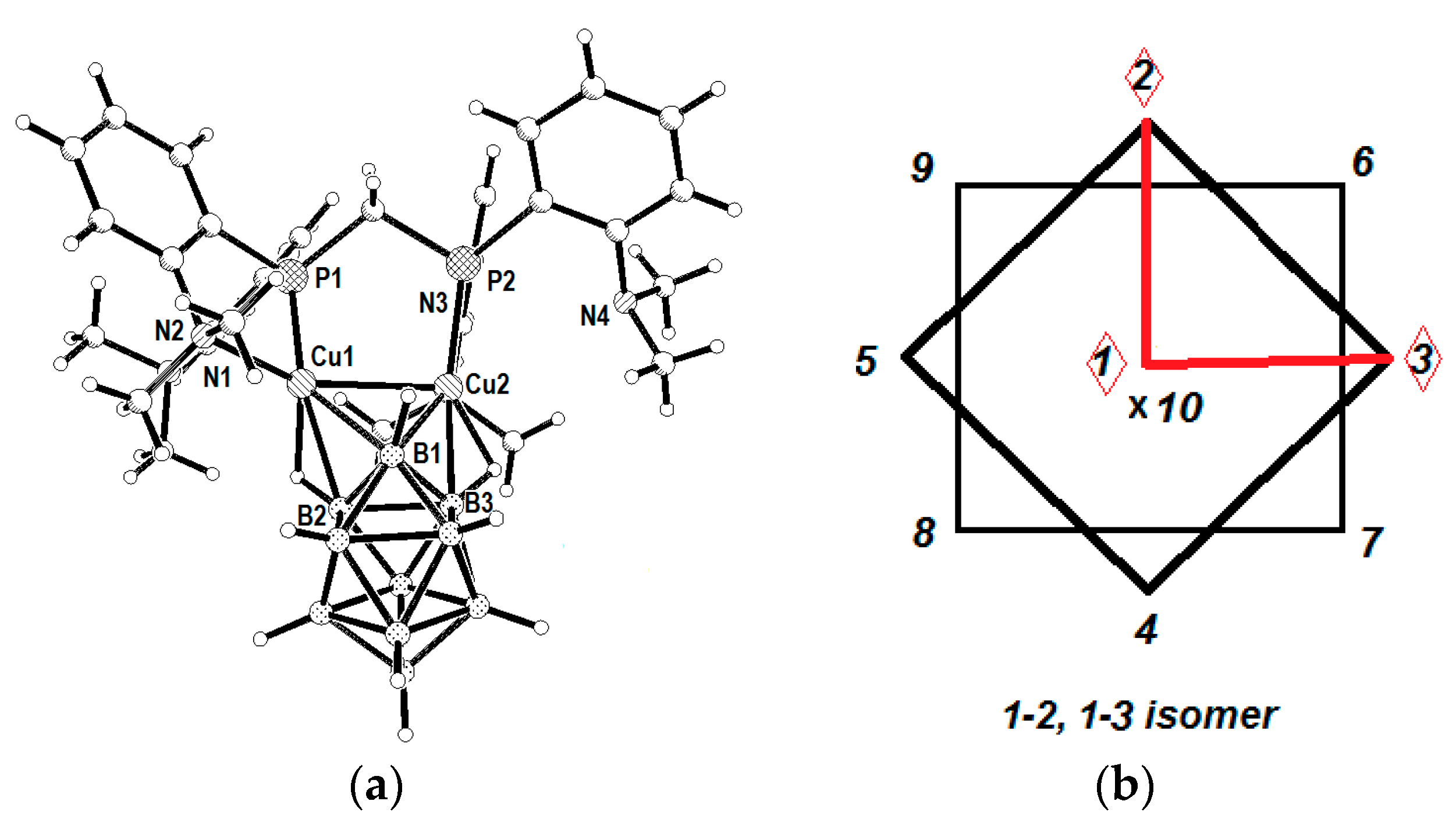
The unusual 1–2, 1–3 coordination isomer {(
dmapm)Cu
2[B
10H
10]} (where
dmapm is 1,1-bis{di-(o-
N,N-dimetylanilinyl)phosphino}methane) was prepared by the reaction of distanna-
closo-decaborate (Et
4N)
2[Sn
2B
10H
10] with dinuclear complex [Cu
2(MeCN)
2(
dmapm)][BF
4]
2 [
39]. The
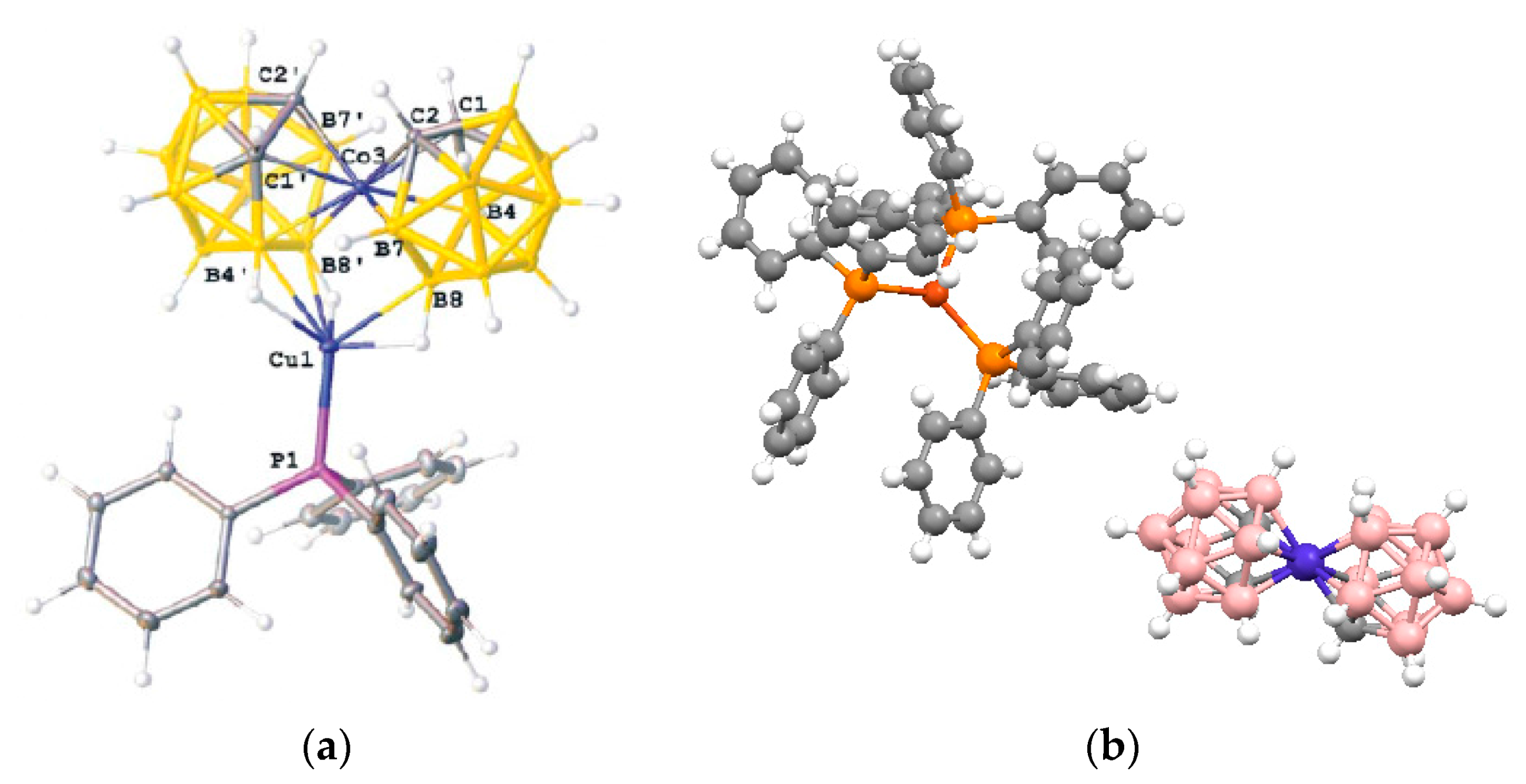
closo-decaborate anion coordinates two copper(I) atoms via 1–2 and 1–3 apical edges forming a relative short Cu…Cu bond (2.6631(5) Å). The Cu(1)…B(2) and Cu(2)…B(3) distances are 2.214(3) and 2.201(3) Å, respectively, and Cu(1)…B(1) and Cu(2)…B(1) distances are 2.227(3) and 2.541(3) Å, respectively. The close positions of metal atoms (near the 1–2 and 1–3 edges) can be caused by structure of the starting copper(I) complex and general tendency of the
closo-borate anion to form complexes first with less coordinated apical vertex (
Figure 16).
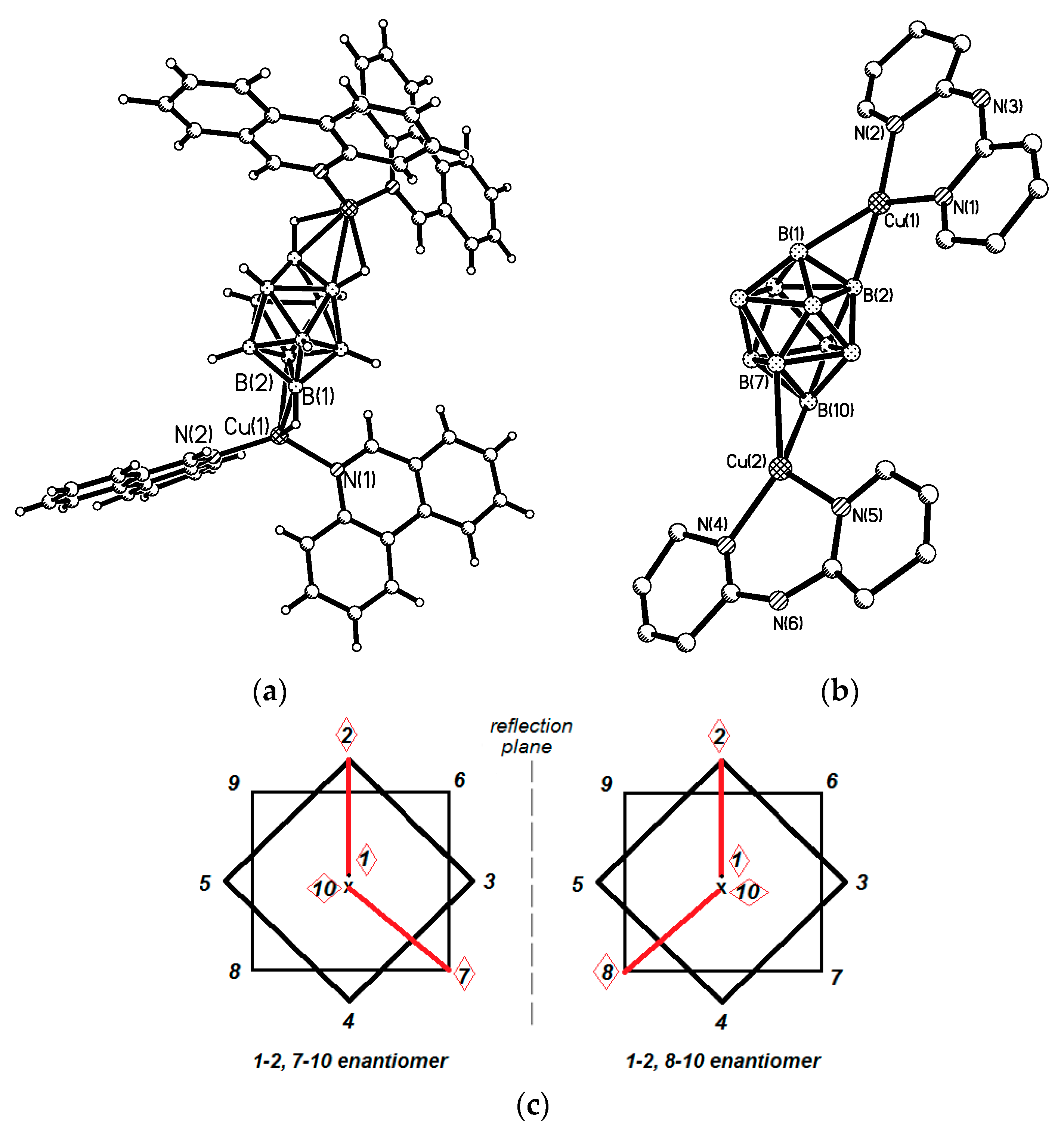
Reaction of Cu
2[B
10H
10] with phenanthridine (5NPhen) in acetonitrile resulted in formation of a mixture of 1–2, 1–4 and 1–2, 7(8)-10 complexes {((5NPhen)
2Cu)
2[B
10H
10]} [
40]. In the 1–2, 1–4 isomer both copper atoms are bonded with apical edges near the same boron vertex, the Cu-B bonds with the shared apical boron atom are elongated (2.316(3) and 2.313(3) Å) as compared to the Cu-B bonds between copper atoms and equatorial boron atoms (2.215(4) and 2.245(4) Å) (
Figure 17). In the 1–2, 7(8)–10 isomer the Cu-B bonds with apical boron atoms of both edges are shorter that those with equatorial edges (Cu(1)…B 2.219(4) and 2.244(4) Å; Cu(2)…B 2.253(4) and 2.280(4) Å) (
Figure 18a). The similar reaction of Cu
2[B
10H
10] with di(pyrid-2-yl)amine (BPa) in acetonitrile at 0 °C under N
2 atmosphere gives the 1–2, 7(8)–10 complex {((BPa)Cu)
2[B
10H
10]} with the same coordination mode of the
closo-decaborate anion [
41] (
Figure 18b). Positional isomers found in both complexes are schematically presented in
Figure 18c.
The listed above positional isomers belong to complexes with the
closo-decaborate anion that coordinates metal atoms by two edges. However, a few examples of the facial coordination of the
closo-decaborate anion were revealed. The reactions of Cu
2[B
10H
10] with chelate ligands 2,2′-bipyridine (2,2′-Bipy) and 1,10-phenanthroline (Phen) in acetonitrile result in formation of copper(I) complexes {((L)Cu)
2[B
10H
10]} with facial and mixed edge-facial coordination mode of metal atoms by the
closo-decaborate anion [
42]. In the complex {((2,2′-Bipy)Cu)
2[B
10H
10]} the boron cluster coordinates metal atoms by apical B(1)-B(2) edge and the opposite B(6)-B(7)-B(10) face (or the corresponding B(8)-B(9)-B(10) face in other enantiomer). The Cu…B bonds fall in the range from 2.156(7) to 2.256(7) Å (
Figure 19).
The {((Phen)Cu)
2[B
10H
10]} complex is centrosymmetrical with crystallographically equivalent copper atoms [
43]. The
closo-decaborate anion coordinates both copper atoms asymmetrically by two opposite apical faces B(1)-B(2)-B(3) and B(7)-B(8)-B(10) (or B(8)-B(9)-B(10)). The Cu…B bonds with each boron face are 2.269(5), 2.119(5), and 2.497(6) Å (
Figure 20).
The metal coordination mode in the
closo-decaborate complexes depends on steric effects of ligands and their basicity, whereas the formation of specific positional isomers is strongly governed by the reaction conditions. A more detailed discussion of this subject was presented in the recent review [
14].
It should be noted that complexation reactions proceeding under redox conditions could be accompanied with substitution reactions in the
closo-decaborate anion. For example, the reaction of Cu
2[B
10H
10] and 2,2′-Bipy in acetonitrile gives not only the above mentioned complex {((2,2′-Bipy))Cu
2[B
10H
10]} but also a complex with substituted
closo-decaborate anion {(MeCN)
2Cu[2-B
10H
9(N-2,2′-Bipy)]} [
44]. In this complex the substituted anion [2-B
10H
9(N-2,2′-Bipy)]
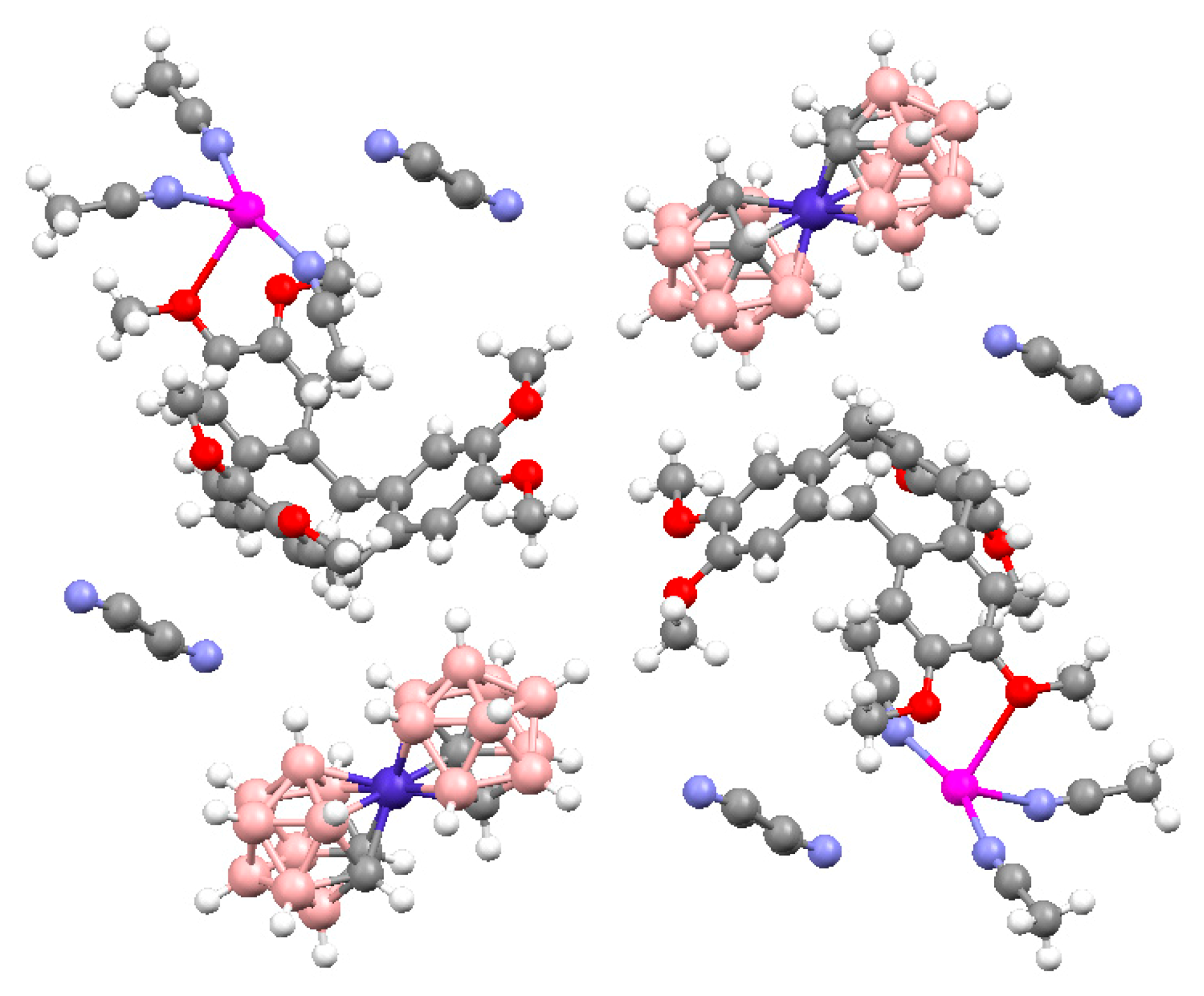
− acts as bidentate ligand. The tetrahedral environment of the Cu atom is formed by two N atoms of acetonitrile molecules (Cu-N 1.995(3) and 1.976(3) Å), the N atom of the pendant pyridine (Cu-N 2.028(2) Å), and the B(1)-Н(1) apical group of the cluster anion (Cu...B 2.601(3) Å, Cu...Н 1.85(3)Å, Cu-Н-В angle 123(2)°) (
Figure 21).
On the other hand, under definite conditions the reactions can result in the complete dissociation of the starting complex and formation of complexes without Cu…HB interactions. For example, the reaction of Cu
2[B
10H
10] and 1,10-phenanthroline in refluxing acetonitrile gives complexes [Cu
I(Phen)
2]
2[B
10H
10]·MeCN and [Cu
I(Phen)
2]
2[Cu
II(Phen)
3][B
10H
10]
2 [
43]. The complex [Cu
I(Phen)
2]
2[B
10H
10]·MeCN is built of [Cu(Phen)
2]
+ cations, [B
10H
10]
2− anions, and solvate MeCN molecules. Both independent Cu atoms have a severely distorted tetrahedral coordination. The Cu-N bond lengths fall in the range 1.997(2)–2.082(2) Å. The endocyclic N-Cu-N bond angles are approximately equal (82.06(10)–2.31(9)°), whereas the exocyclic angles around two Cu atoms vary in the ranges 105.12(10)–156.58(10)° and 107.88(9)–146.71(10)°. The complex [Cu
I(Phen)
2]
2[Cu
II(Phen)
3][B
10H
10]
2 consist of cationic Cu
I and Cu
II complexes [Cu
II(Phen)
3]
2+ and [Cu
I(Phen)
2]
+, respectively, and [B
10H
10]
2− anions. The coordination polyhedra of the Cu
II and Cu
I atoms are a distorted octahedron and a tetrahedron, respectively. The Cu-N bond lengths are 2.026(6)–2.330(6) Å for Cu
II and 2.022(7)–2.056(7) Å for Cu
I complex cations (
Figure 22) [
43].
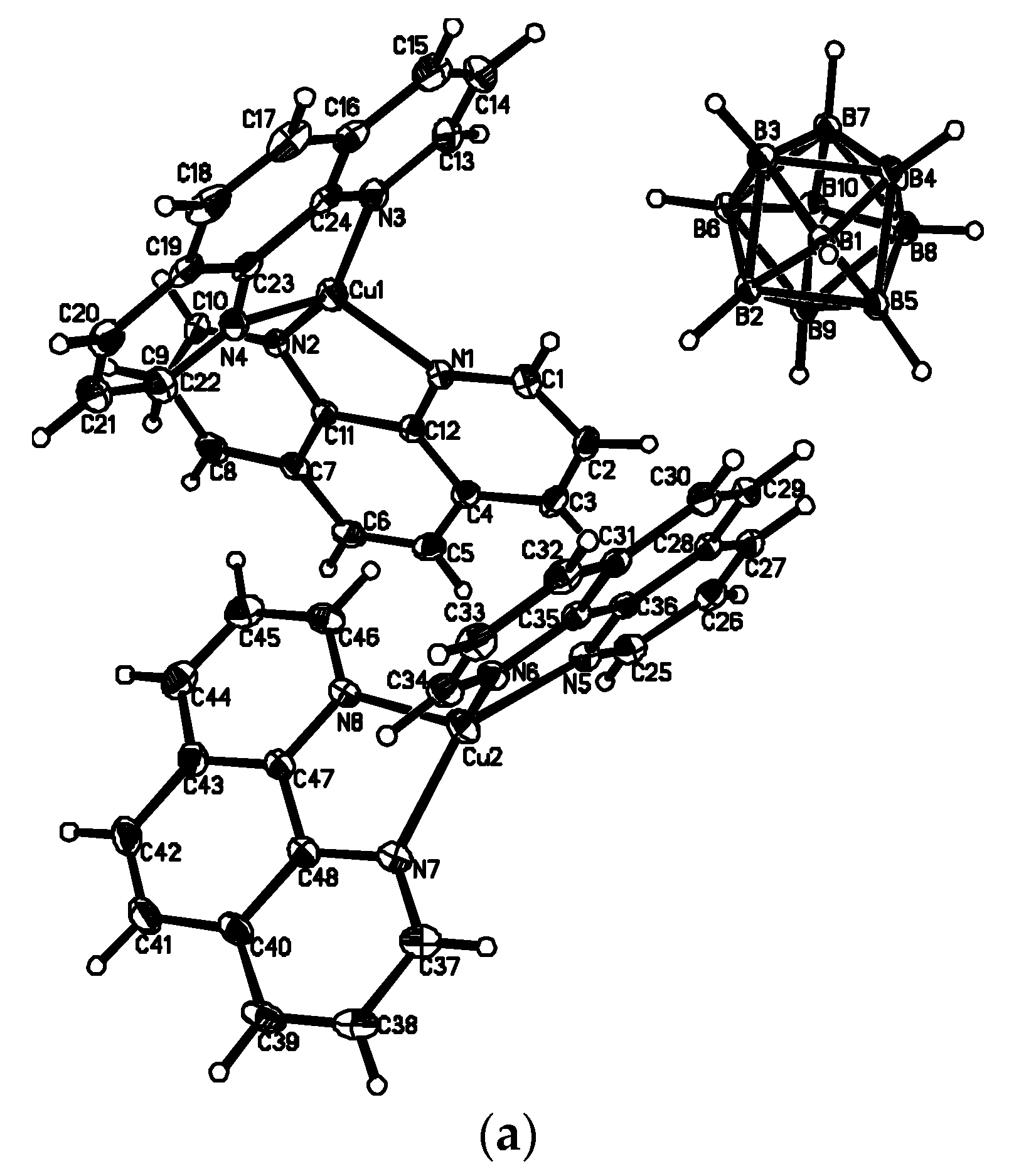
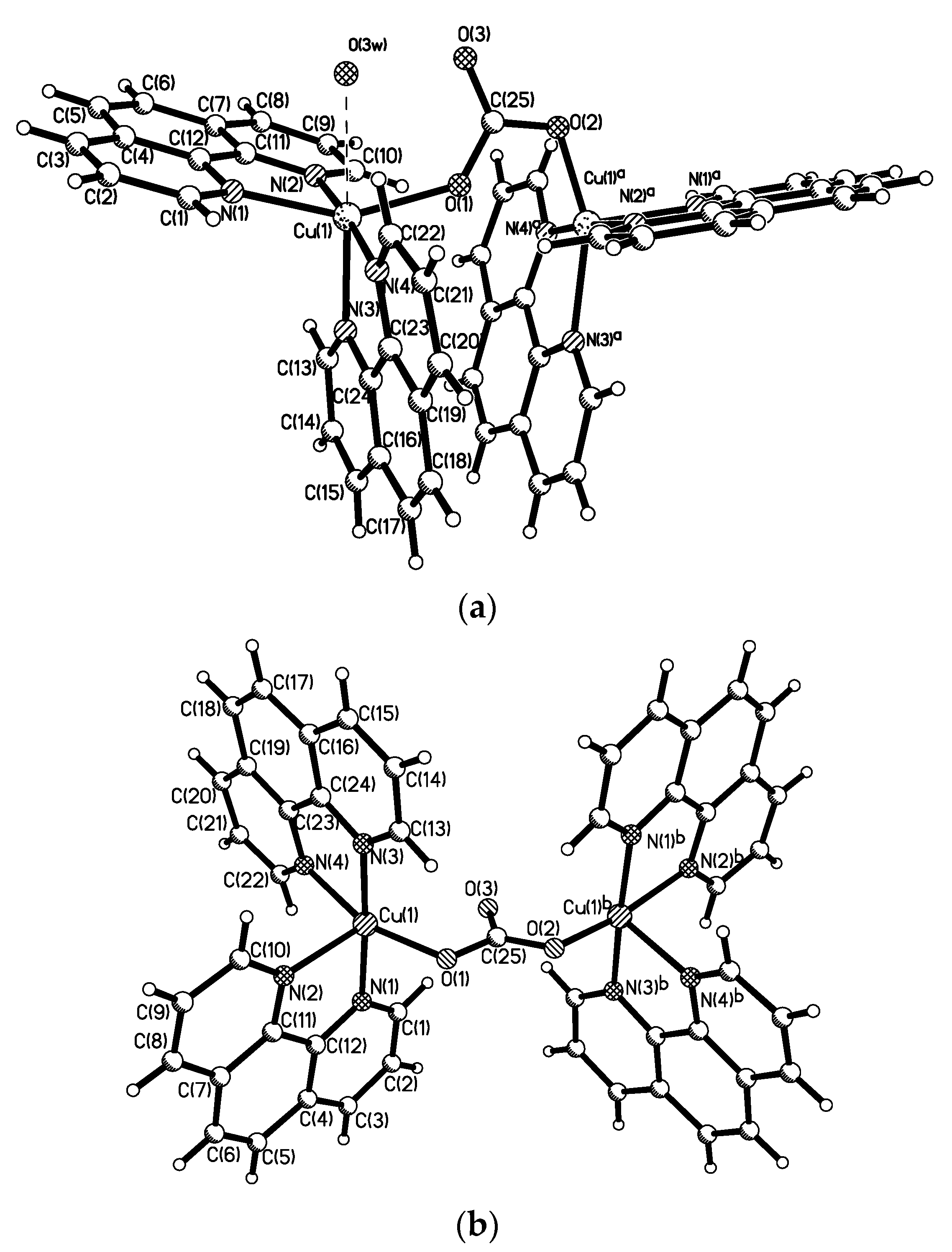
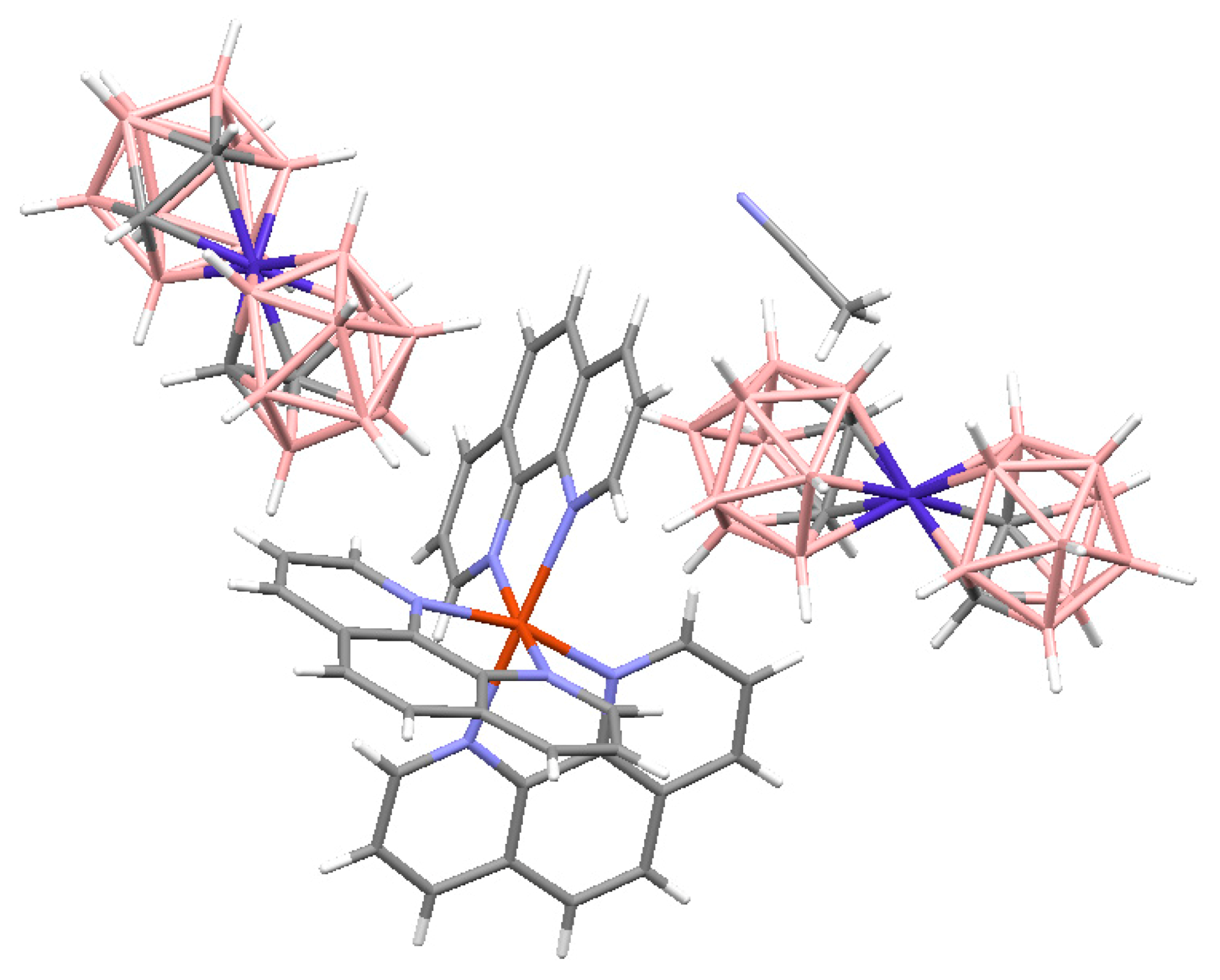
The last complex contains both Cu
I and Cu
II cationic complexes. Another example of heterovalent copper(I,II) complex is [Cu
II4(OH)
4(Bipy)
4][Cu
I2[B
10H
10]
3]·4(MeCN) (See
Figure 8). It should be noted that although copper(II) is easily reduced to copper(I) with
closo-decaborate anion in solutions, upon crystallization of complexes containing azaheterocyclic ligands in air a reverse process, namely the oxidation of copper(I) to copper(II), often occurs.
The reaction of CuSO
4·5H
2O with an excess of 1,10-phenanthroline in DMF results in qualitative precipitation of copper(II) complex [Cu(Phen)
2][B
10H
10]. The crystal of [Cu(Phen)
2][B
10H
10] is built of the [Cu(Phen)
2]
2+ cations and the [B
10H
10]
2− anions. The copper cation is in distorted plane square coordination (Cu-N bonds are 1.9962(12) Å, N-Cu-N angles are 83.04(7), 101.29(7) and 157.73(7)°). The
closo-decaborate anions approach the complex cation on either side (Cu…B and Cu…H contacts are 3.545(17) and 2.85(19) Å, respectively). The [Cu(Phen)
2]
2+ cations and the [B
10H
10]
2− anions alternate in the
a direction (
Figure 23) [
43].
Reaction of Cu
2[B
10H
10] with 1,10-phenanthroline in acetonitrile followed by addition of DMF or DMSO afforded
anti,anti- and
anti,syn-isomers of complex [Cu
2(Phen)
4(μ-CO
3)][B
10H
10] with bridging CO
3 group [
45]. These complexes were structurally characterized as solvates [Cu
2(Phen)
4(μ-CO
3)][B
10H
10]·2.5(DMSO)·2(H
2O) and [Cu
2(Phen)
4(μ-CO
3)][B
10H
10]·4(DMF) (
Figure 24). In both structures the CO
32− anion acts as a bridge between two {Cu(Phen)
2}
2+ fragments. The basic difference between both isomers is the spatial orientation of the {Cu(Phen)
2}
2+ fragments:
anti,syn in the DMSO solvate and
anti,anti in the DMF solvate. As a result, the Cu…Cu distance in the dimer with the
anti,syn configuration (4.4409(14) Å) is considerably shorter that in the dimers with the
anti,anti configuration (5.2886(7) Å) (
Figure 24) [
14].
The
anti,anti-complex [Cu
2(Phen)
4(μ-CO
3)][B
10H
10]·3.5(DMF)·1.25(H
2O) was also prepared by directed reaction of (Et
3NH)
2[B
10H
10] with previously prepared copper(II) complex [Cu
2(Phen)
4(μ-CO
3)]Cl
2 with the
anti,anti configuration of the CO
3 group in DMF [
43].
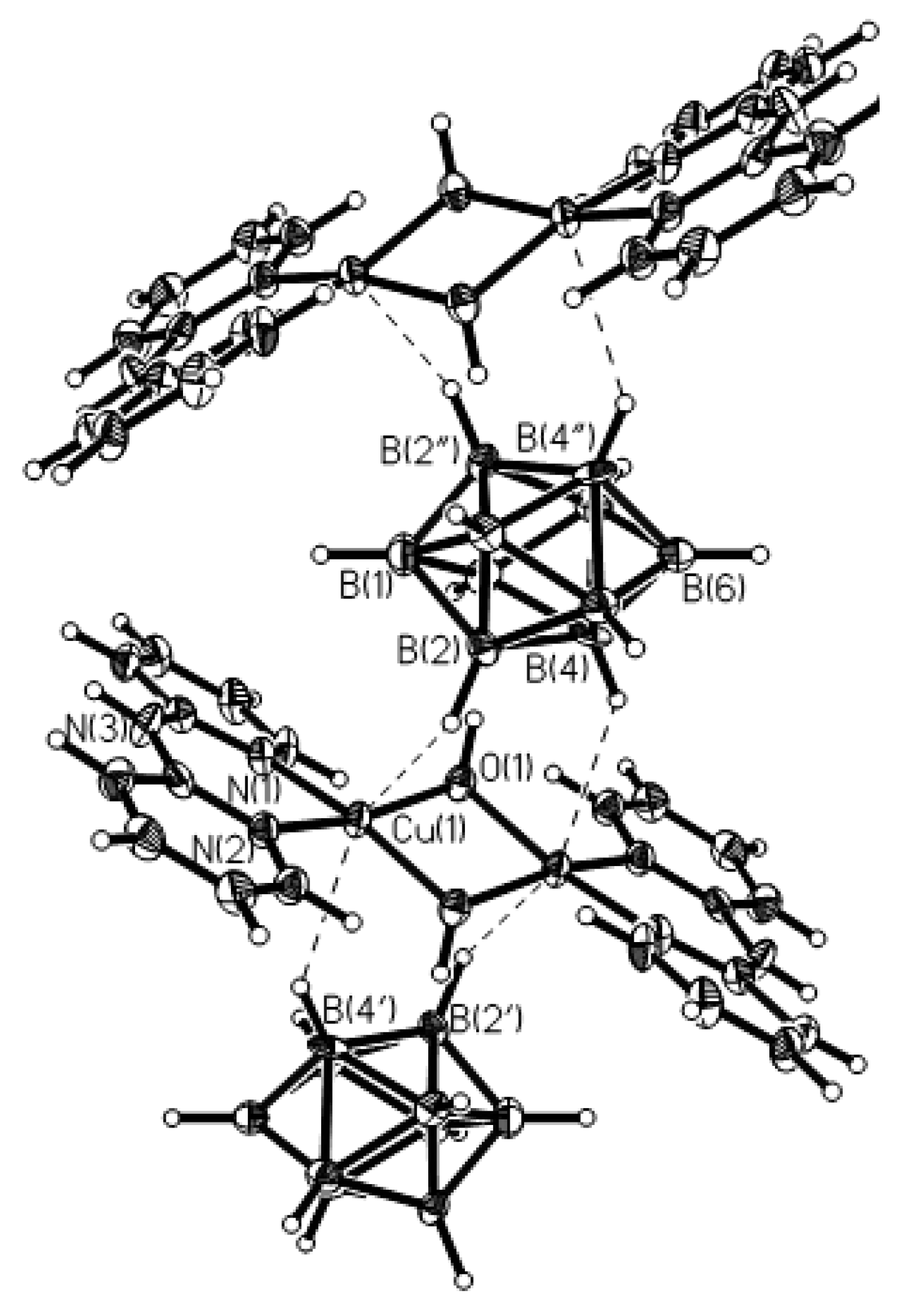
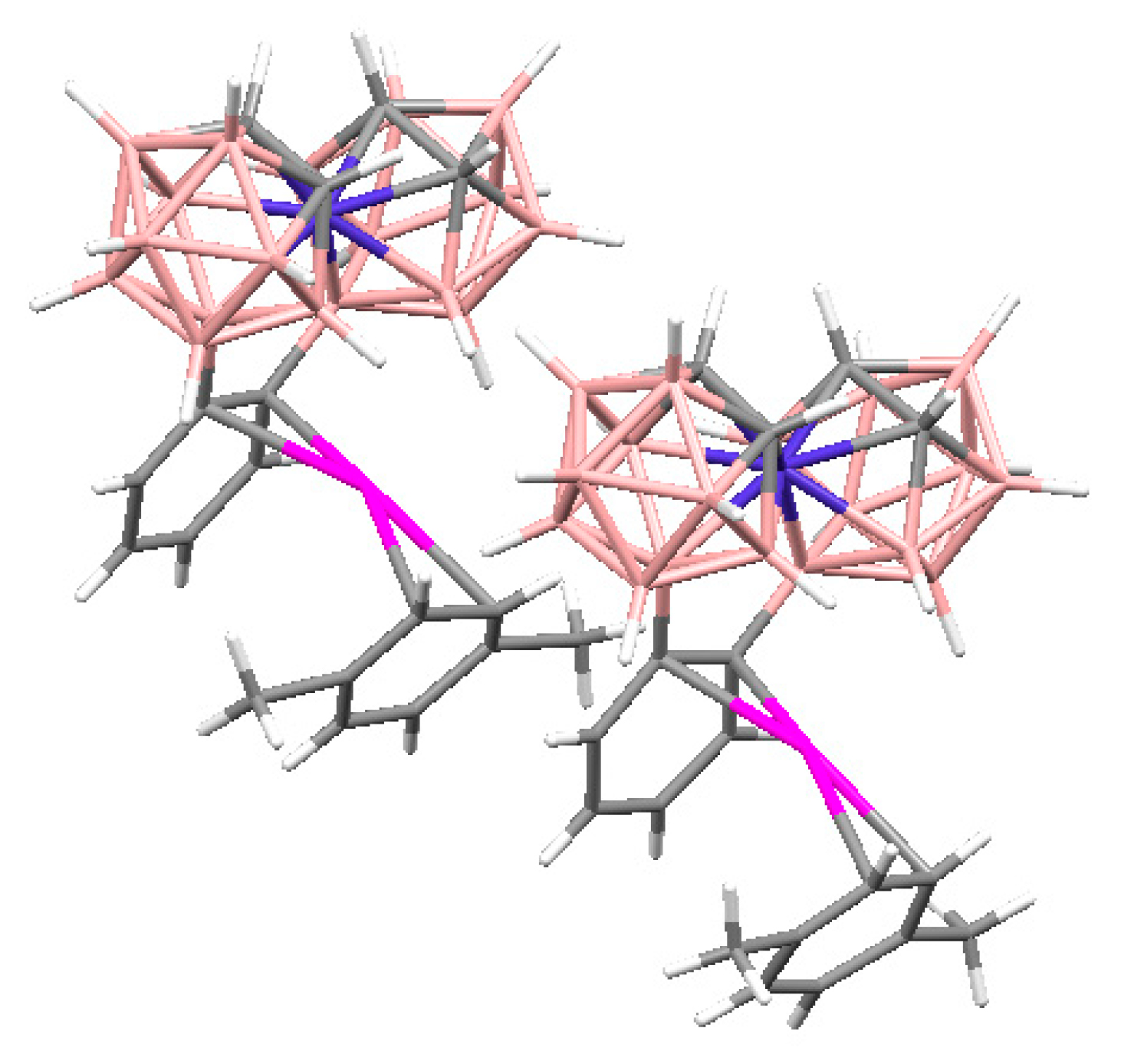
The reaction of Cu
2[B
10H
10] with 2,2′-bipyridine in an acetonitrile/DMSO mixture produces tetranuclear hydroxo bridged copper(II) cluster {[Cu
4(2,2′-Bipy)
4(OH)
4][B
10H
10]
2·2(DMSO)} [
46]. The crystals are composed of tetranuclear [Cu
4(2,2′-Bipy)
4(OH)
4]
4+ cations, [B
10H
10]
2− anions and DMSO molecules. The cation has a centrosymmetric double-decker structure of the Z type (open cubane-like complex). The asymmetric unit of the complex consists of two {(2,2′-Bipy)Cu}
2+ fragments connected to each other by OH
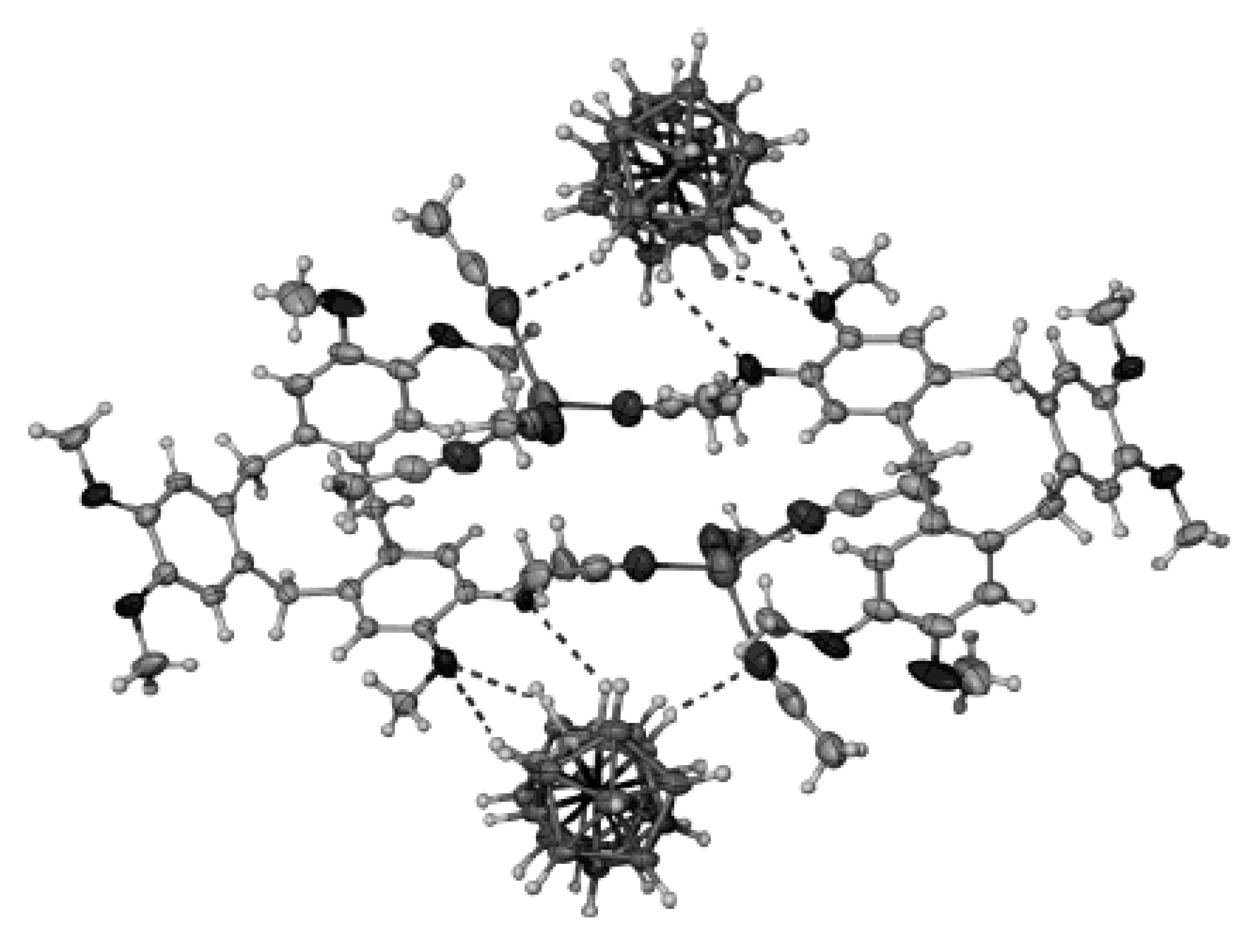
− groups. The Cu(1) and Cu(2) atoms have a flattened environment of two nitrogen atoms and two oxygen atoms. The coordination of the Cu(1) and Cu(2) atoms is completed to 4 + 1 by the H(2B) atom of the polyhedral anion and the O(2′) atom of the second part of the complex, respectively. The axial Cu(2)-O(2′) bond (2.332(1) Å) is elongated as compared with the equatorial Cu-O bonds (1.922-1.946(1) Å). The Cu(1)-H(2B) bond length is 2.22(2) Å. The Cu(1) and Cu(2) atoms form long axial contacts (Сu(1)…N(4′) 3.123 Å; Cu(2)…H(1B) 2.86(2) Å), each of which completes the polyhedron to an asymmetrically elongated tetragonal bipyramid. The O(2)-H(2) hydroxyl group forms a hydrogen bond with the DMSO molecule (O(2)…O(3) 2.676(2) Å), and the O(1)-H(1) group is involved in the formation of dihydrogen bond with the B(3)-H(3) group of the anion (H(1)…H(3B) 2.16(3) Å). The DMSO molecule is additionally bonded to the complex cation by the С(1)-Н…О(3) hydrogen bond. In the complex cation, there are π-π stacking interactions between overlapping 2,2′-Bipy molecules (
Figure 25). The same tetranuclear hydroxo-bridged cations were found in complex {[Cu
II4(OH)
4(Bipy)
4][Cu
I2[B
10H
10]
3]·4(MeCN)} (See
Figure 8).
Reaction of Cu
2[B
10H
10] with di(pyrid-2-yl)amine in acetonitrile at ambient temperature results in hydroxo-bridged copper(II) complex {[Cu
2(Bpa)
2(OH)
2][B
10H
10]} [
41]. The crystals are composed of the dimeric cations [Cu
2(Bpa)
2(OH)
2]
2+ and the
closo-decaborate anions. A distorted square planar environment of copper atoms is formed by two nitrogen atoms of di(pyrid-2-yl)amine and oxygen atoms of two bridging ОН
− groups with the Cu(1)…Cu(1’) distance of 2.9180(14) Å. The [B
10H
10]
2− anion is located on the two-fold rotation axis that passes through the apical vertices B(1) and B(6). The H(2) and H(4) atoms of the anion form Cu…HB contacts of 2.73 and 2.63 Å with copper atoms of the complex cation thus completing copper atom coordination to 4 + 2 and connecting the cations and the anions into polymeric chains (
Figure 26) [
41].
The similar reaction at −20 °C gave a mixture of {[(Cu
II2(Bpa)
2(OH)
2]
2[Cu
I2[B
10H
10]
3]·n(MeCN)}, {[Cu
II(Bpa)(CO
3)]
2·H
2O} and {[Cu
II(Bpa)
2(MeCN)
2][2-B
10H
9(N-Bpa)]
2·2(H
2O)} complexes. The crystal of {[Cu
II(Bpa)
2(MeCN)
2][2-B
10H
9(N-Bpa)]
2·2(H
2O)} consists of the [Cu
II(Bpa)
2(MeCN)
2]
2+ cations and the [2-B
10H
9(N-Bpa)]
− anions, where molecule of di(pyrid-2-yl)amine is attached through nitrogen atom of the pyridine ring to the B(2) position of the boron cluster anion (
Figure 27) [
41].
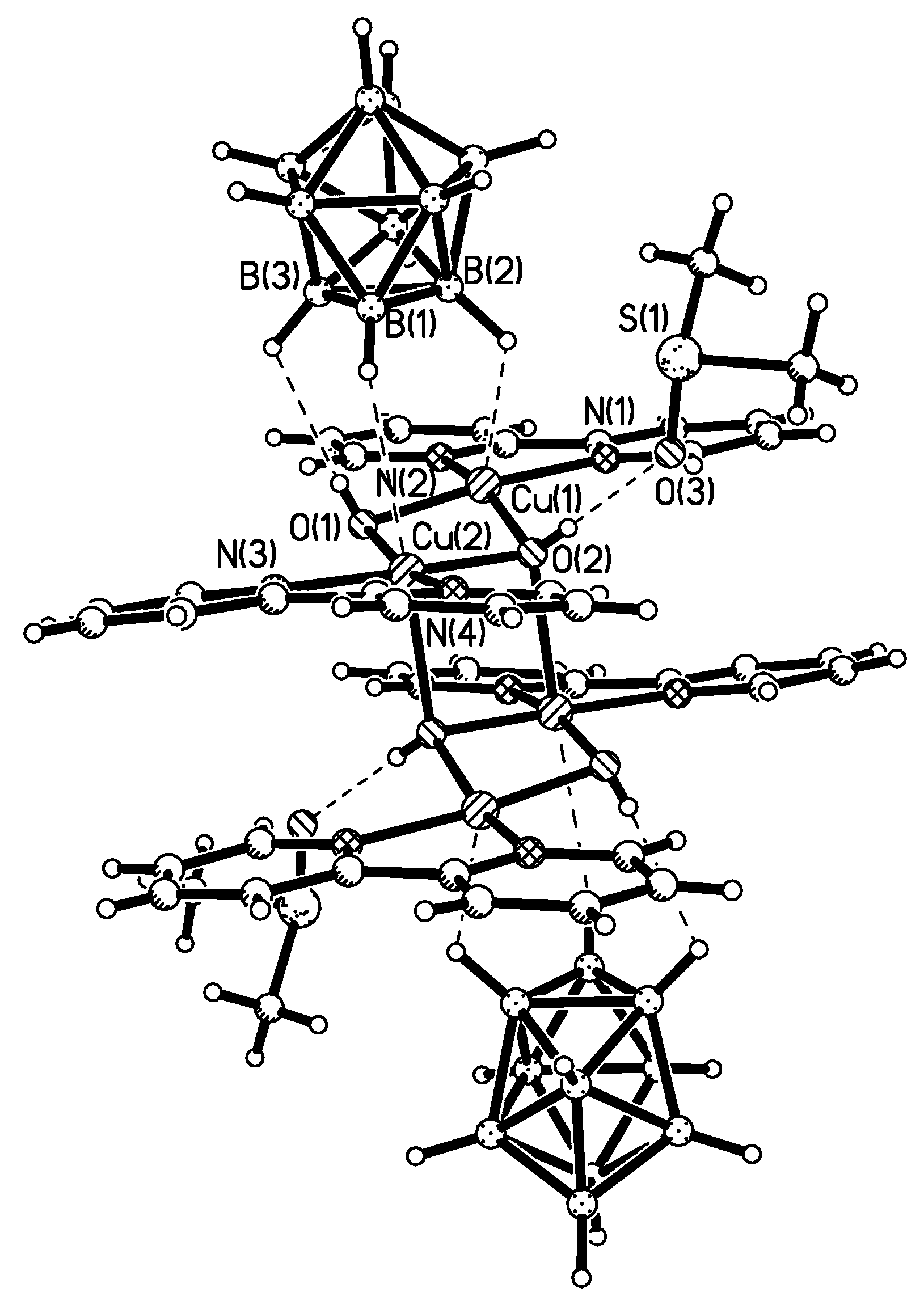
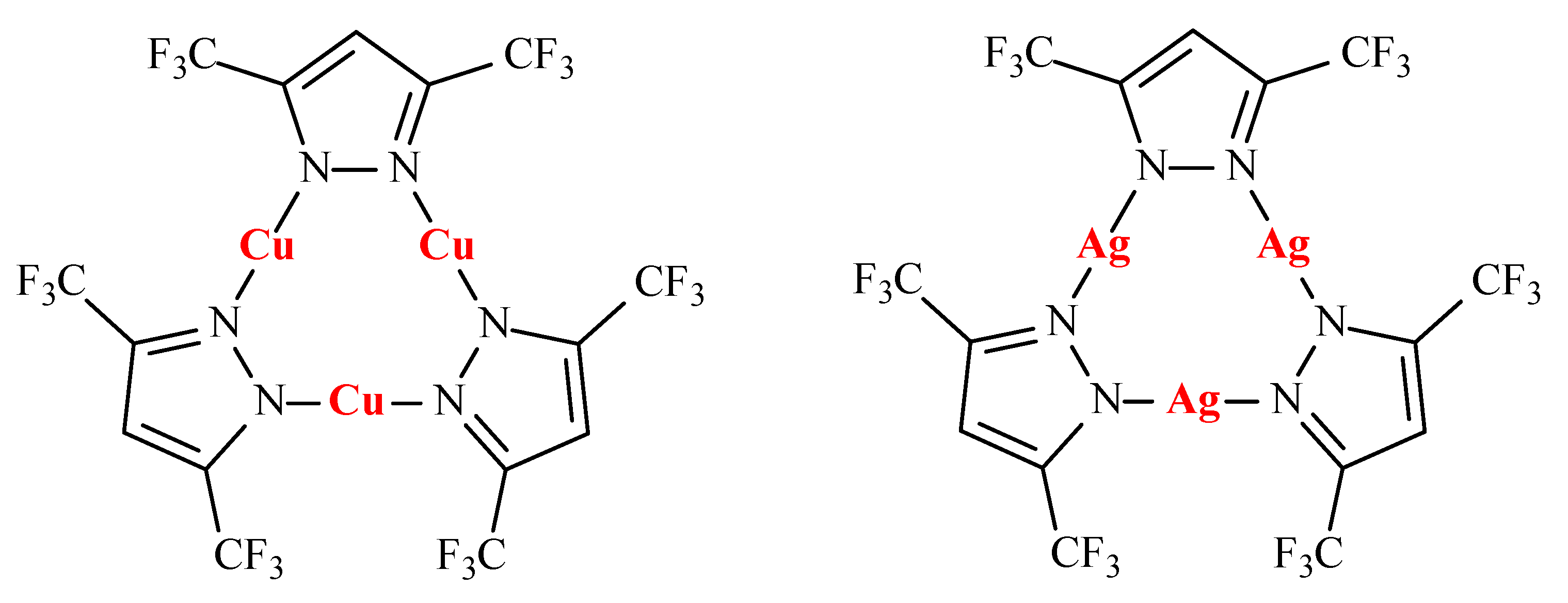
The complexation between the
closo-decaborate anion and cyclic copper(I) and silver(I) 3,5-bis(trifluoromethyl)pyrazolates {[3,5-(CF
3)
2Pz]M}
3 (M = Cu, Ag) (
Scheme 2) in CH
2Cl
2 was studied by IR spectroscopy. Formation of two types of complexes, 1:1 and 1:2, was revealed and their stability constants were determined [
47].
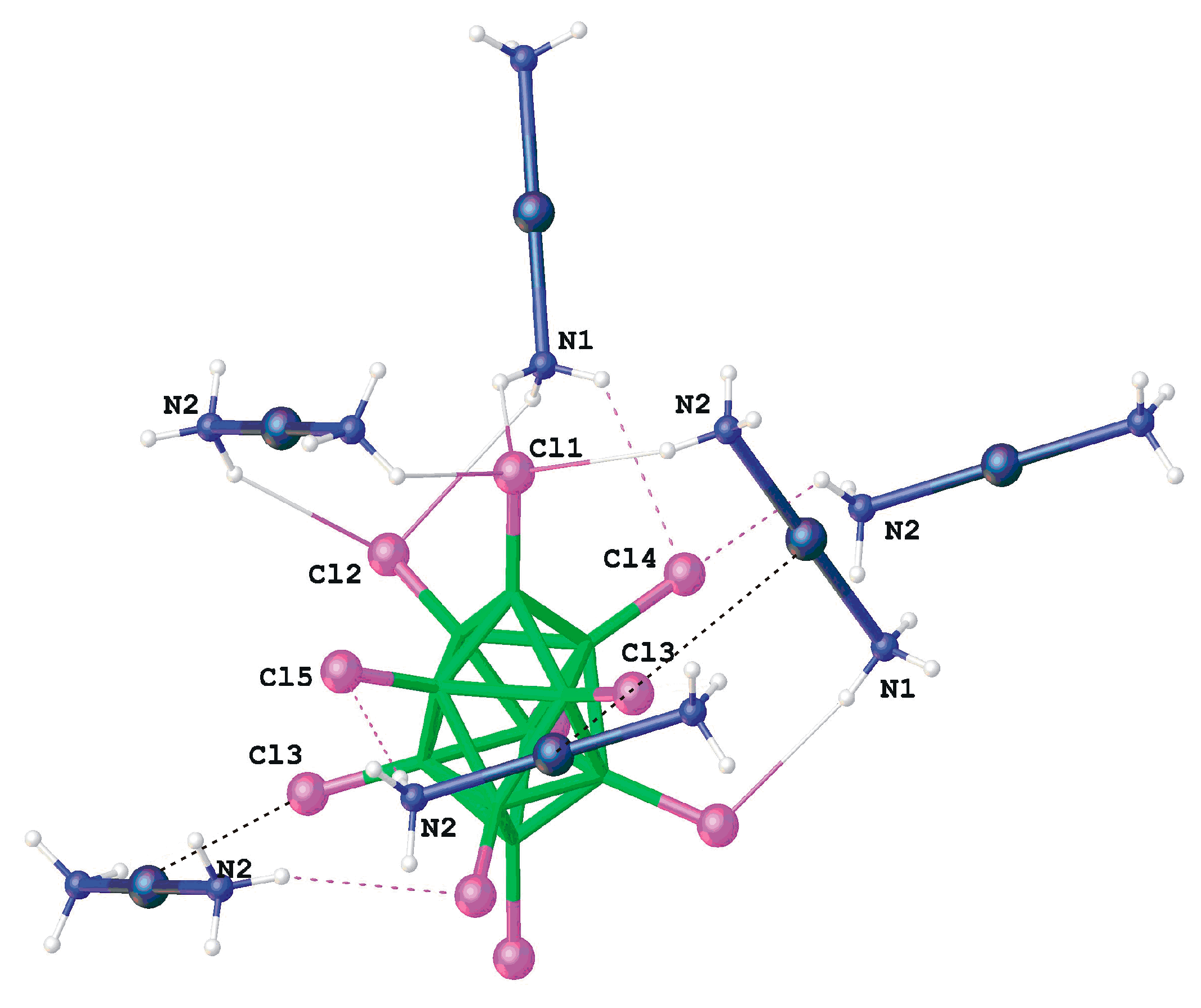
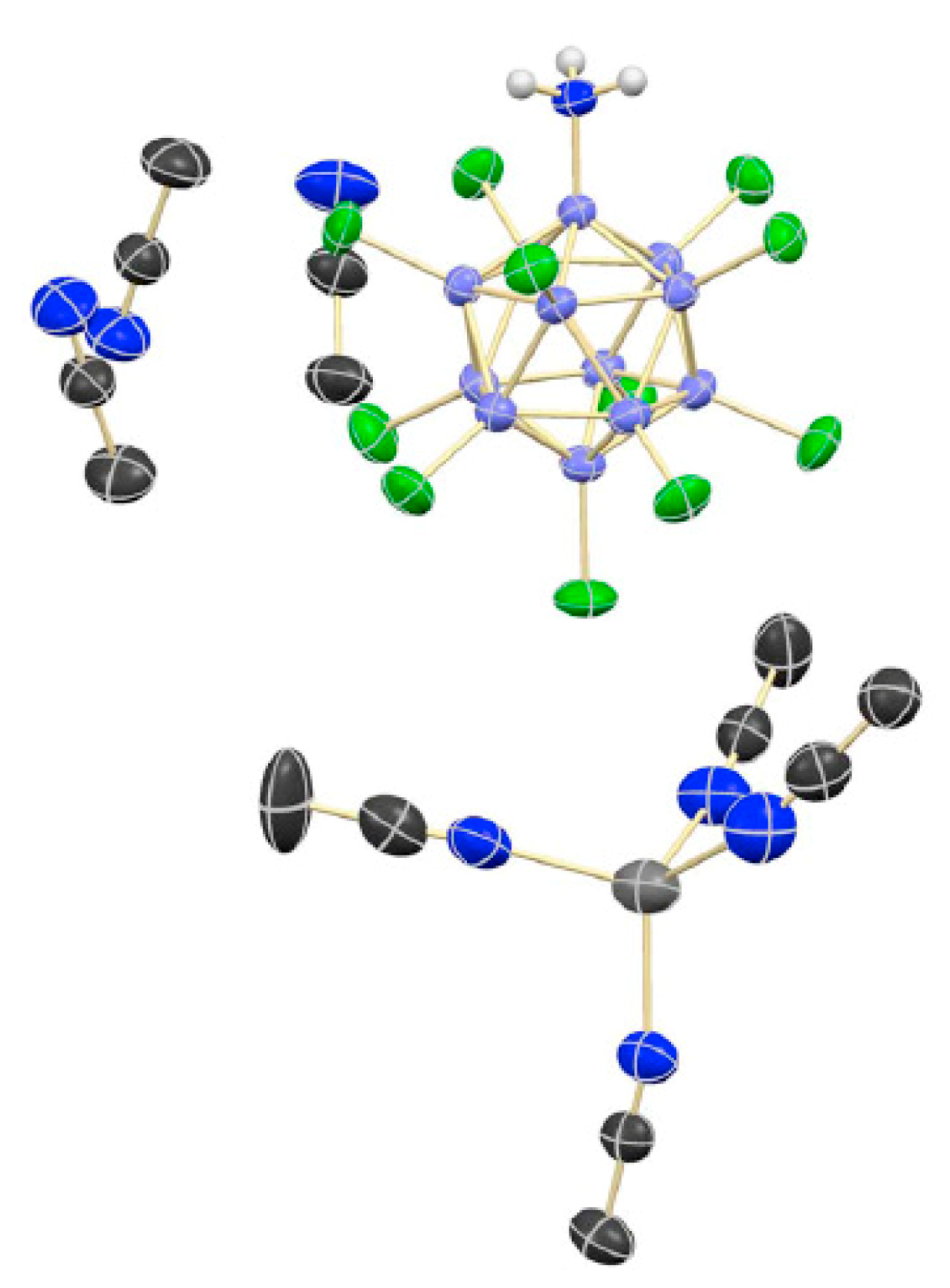
Silver ammonia complex with decachloro-
closo-decaborate anion [Ag(NH
3)
2]
2[B
10Cl
10] was prepared by reaction of (Et
3NH)
2[B
10Cl
10] with AgNO
3 in aqueous ammonia. According to X-ray diffraction, the crystal is built of the [Ag(NH
3)
2]
+ cations and [B
10Cl
10]
2− anions. The Ag-N distances are equal to 2.128(3) and 2.140(3) Å, the NAgN angle is almost linear (178.9°). There are rather short N–H...Cl (<2.95 Å) and Ag...Cl (<3.5 Å) contacts between the cations and anions (
Figure 28) [
48].
An interesting case presents a silver complex with the dimeric icosaborate anion
trans-[B
20H
18]
2- which can be considered as product of mild oxidation of the
closo-decaborate anion [B
10H
10]
2− [
49]. Complex {((Ph
3P)
3Ag)
2[
trans-B
20H
18]} was prepared by the reaction of (Et
3NH)
2[
trans-B
20H
18] with [(Ph
3P)
3Ag]NO
3 in N,N-dimethylformamide. In the obtained complex each silver atom is coordinated by the apical BH group of the dimeric boron anion and three molecules of triphenylphosphine. The Ag…B distance is 3.382(3) Å and the AgHB angle is 161.8º that reflects much weaker cation-anion bonding in comparison with the parent
closo-decaborate anion (
Figure 29) [
50].
The UV radiation of a crystal of {((Ph
3P)
3Ag)
2[
trans-B
20H
18]} results in its single-crystal-to-single-crystal isomerization to {((Ph
3P)
3Ag)
2[
iso-B
20H
18]} without the crystal degradation with only minor (0.75%) contraction of the unit cell. There are two linkage isomers co-crystallize in the final crystal of {((Ph
3P)
3Ag)
2[
iso-B
20H
18]}: in one of them, the BHAg angle is practically linear (172°), while in other isomer with facial coordination its value (116°) is typical for polydentate coordination of silver atom. The Ag…B distances increase to 3.517(5) and 3.394(8) Å, respectively (
Figure 29). The isomerization is reversible, and heating {((Ph
3P)
3Ag)
2[
iso-B
20H
18]} at 150ºC produces {((Ph
3P)
3Ag)
2[
trans-B
20H
18]} the
iso-isomer was converted completely to the described
trans-isomer [
50].
3. Copper and Silver Complexes with the closo-dodecaborate anion [B12H12]2−
The
closo-dodecaborate anion [B
12H
12]
2− is the most studied representative of the
closo-borane family [
51]. The [B
12H
12]
2− anion has close to sphere icosahedral structure that causes decrease of its coordination ability. Nevertheless, a large number of its copper(I) and silver(I) complexes has been described.
The binary copper(I) and silver(I) salts of
closo-dodecaborate anion M
2[B
12H
12] (M = Cu, Ag) were prepared [
20,
52,
53]. Their X-ray structures were not determined, however the existence of M-H-B interactions in the solid state was supported by the IR spectroscopy data.
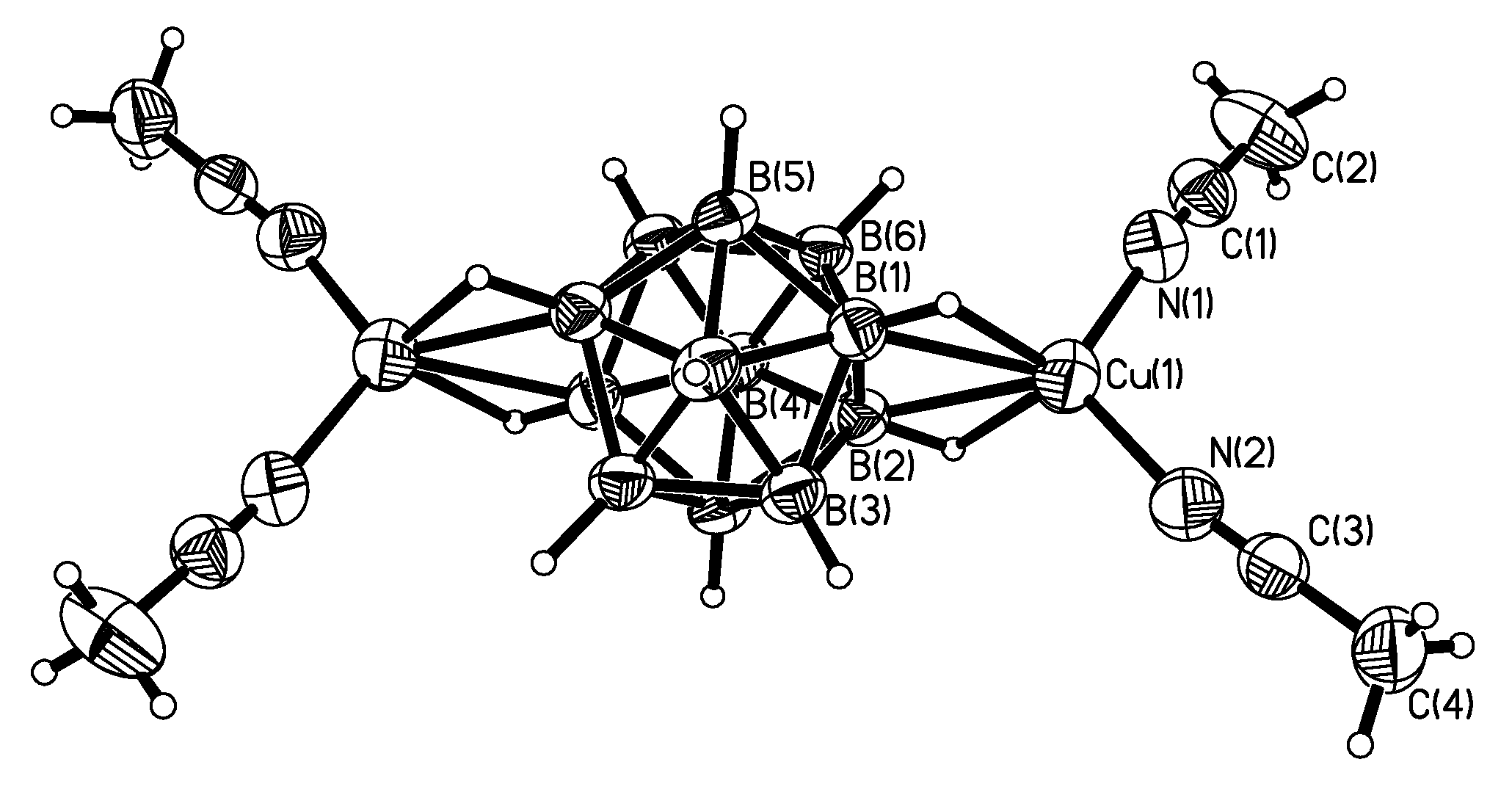
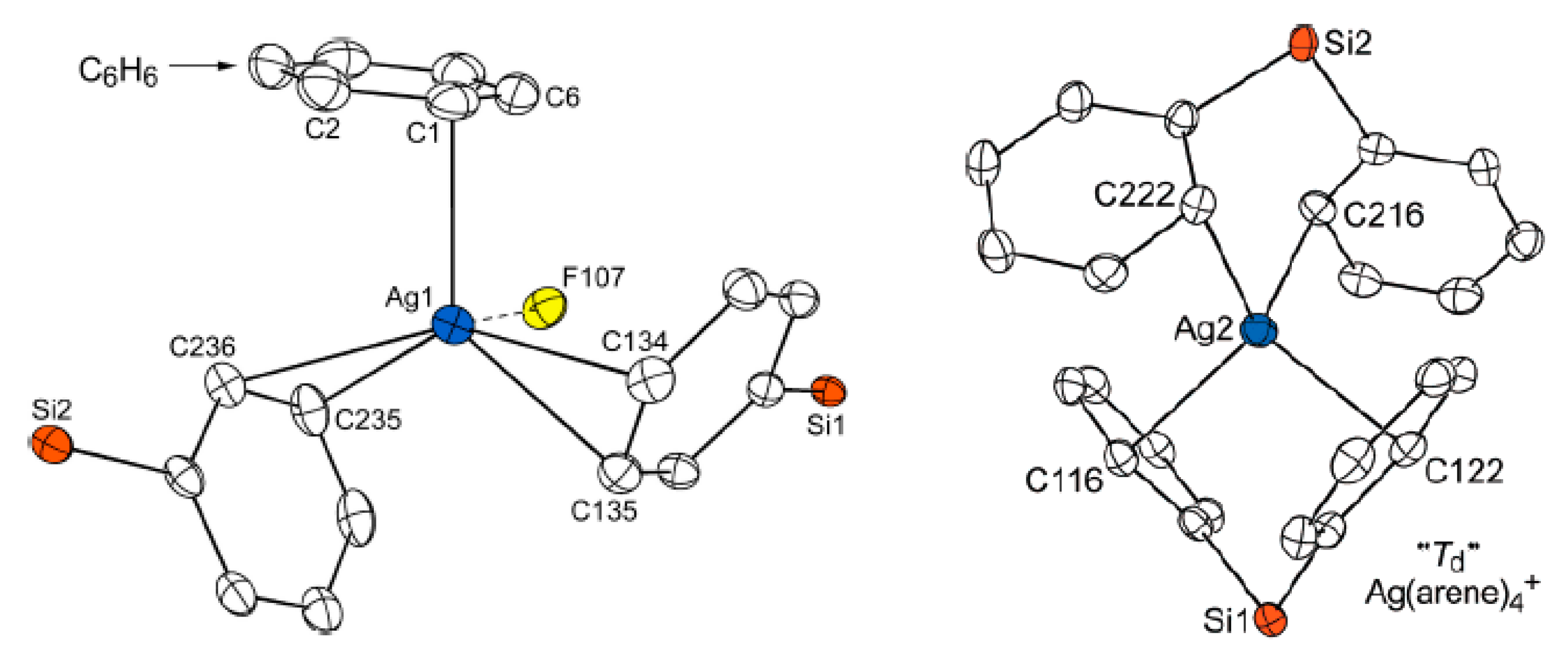
Complex {((MeCN)
2Cu)
2[B
12H
12]} was prepared by the reaction of the acid (H
3O)
2[B
12H
12] with Cu
2O followed by recrystallization from acetonitrile. In the structure of {((MeCN)
2Cu)
2[B
12H
12]} two {(MeCN)
2Cu}
+ fragments are coordinated to opposite edges of the
closo-dodecaborate anion. The Сu…B bonds are 2.288(3) and 2.351(4) Å, Сu…Н(B) bonds are 1.86 and 1.95 Å (
Figure 30) [
54].
Complex {((MeCN)Ag)
2[B
12H
12]}
n which was prepared by crystallization of Ag
2[B
12H
12] from acetonitrile has a 3D-polymeric framework structure with two crystallographically different Ag cations. The Ag(1) cation is coordinated via Ag…(BH)
2 bridges by two
closo-dodecaborate anions and one acetonitrile molecule forming slightly distorted square pyramid environments. The Ag(2) cation is coordinated via single Ag…BH bridges by three
closo-dodecaborate anions and one acetonitrile molecule forming tetrahedral arrangement. The Ag…B and Ag…H bonds are 2.582(3)–2.873(3) and 2.02(3)–226(4) Å, respectively (
Figure 31) [
55].
Similar to complexes of the
closo-decaborate anion, a series of complexes {Cat(Cu[B
12H
12])} (Cat
+ = Cs, R
3NH
+ and R
4N
+ (R = Me, Et, Pr, Bu), Ph
4P
+, Ph
4As
+) was prepared by reaction of Cat
2[B
12H
12] with CuSO
4 or CuCl
2 in aqueous solution in the presence of reducing agent (SO
2 or Na
2SO
3) [
23,
54]. In the IR spectra of the Cat{Cu[B
12H
12]} complexes along with the B-H stretching band at 2450–2540 cm
−1 a new band appears at 2220–2165 cm
−1, that can be assigned to Cu-H-B stretching by analogy with similar complexes of the
closo-decaborate anion.
The similar silver(I) complexes {Cat(Ag[B
12H
12])} (Cat
+ = Cs, R
3NH
+ and R
4N
+ (R = Me, Et, Pr, Bu), Et
3NBz
+, Ph
4P
+, Ph
4As
+, Ph
3PCH
2Naph
+) were prepared by the reaction of Cat
2[B
12H
12] with AgNO
3, in aqueous solution [
53,
55]. Structures of complexes {Cat(Ag[B
12H
12])}
n (Cat
+ = Et
3NBz
+ and Ph
3PCH
2Naph
+) were determined by single crystal X-ray diffraction.
The structure of complex {(Ph
3PCH
2Naph)(Ag[B
12H
12])}
n is built of the {Ag[B
12H
12]
−}
n chains and the [Ph
3PCH
2Naph]
+ cations. Each silver cation is coordinated by two
closo-dodecaborate anions, to which silver is bound by Ag…(BH)
2 bonds (Ag…HB distances are 2.474(6)–2.580(6) Å). There are two types of
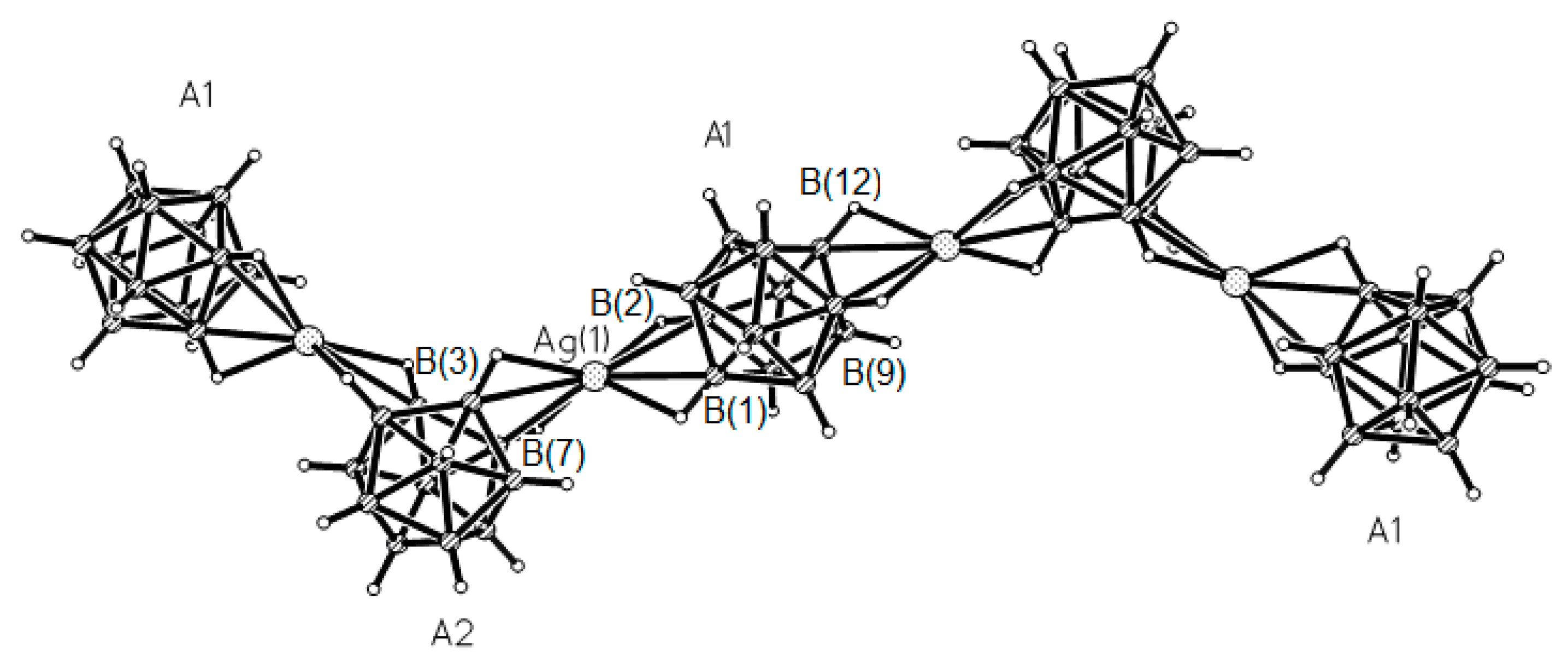
closo-dodecaborate anions: the first one (A1) coordinate silver via B(1)–B(2) and B(9)-B(12) edges, whereas the second one (A2) coordinates silver via B(1)-B(2) and B(7)-B(8) edges. The centrosymmetric A1 anion provides linearity of the chain fragments of the chain, whereas the A2 anion provides a bend of the silver anionic chain (
Figure 32) [
53].
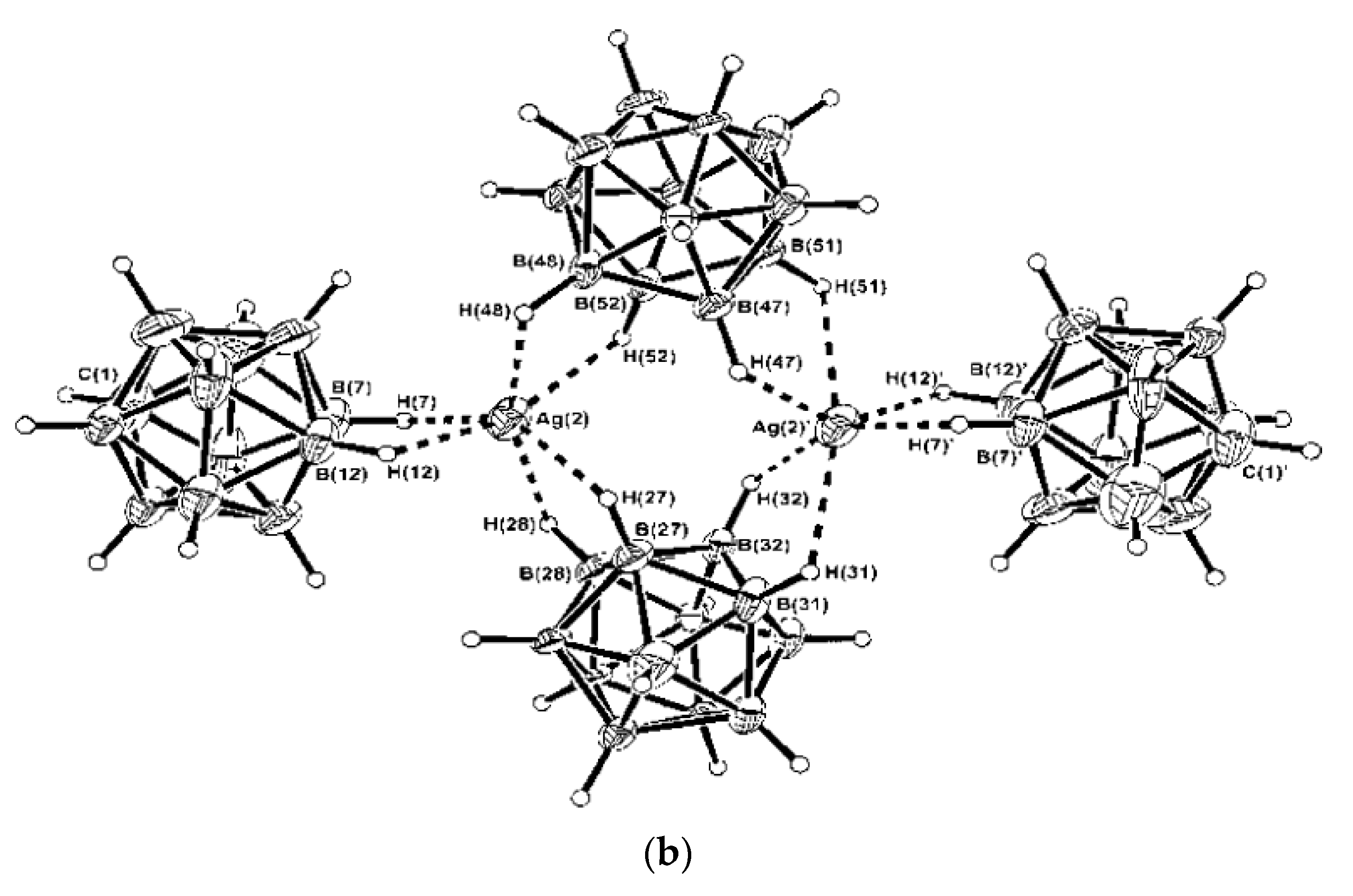
The structure of complex {(Et
3NBz)(Ag[B
12H
12])}
n is characterized by double anionic chain structure of {Ag[B
12H
12]}
−. Each silver cation is coordinated by three icosahedral cages, to which silver is bound by Ag…(BH)
2 bonds. Each
closo-dodecaborate anion cage is coordinated by three silver cations. The Ag…B and Ag-H bonds fall in the ranges 263.8(2)–292.0(2) and 2.18(2)–240(2) Å, respectively (
Figure 33) [
55].
Complex {(Ph
3PCH
2Naph){(MeCN)Ag}[B
12H
12])}
n was obtained by crystallization of {(Ph
3PCH
2Naph)(Ag[B
12H
12])}
n from acetonitrile. The crystals are built of the silver anionic chains and the [Ph
3PCH
2Naph]
+ cations. The silver chain contains one centrosymmetric Ag…[B
12H
12]…Ag fragment similar to that found in the acetonitrile-free complex (see
Figure 30, the A1 anion) and one additional fragment, in which silver atom is coordinated by two
closo-dodecaborate anions and an acetonitrile molecule. Thus, the environment of silver Ag(1) and Ag(2) is formed by two edges of two symmetrically unequivalent icosahedra (A3 and A4) (
Figure 34) [
53].
Complex {(Et
3NBz)((MeCN)Ag)
2Ag[B
12H
12])}
n was prepared by addition of aqueous AgNO
3 to a solution of {(Et
3NBz)(Ag[B
12H
12])}
n in acetonitrile. The asymmetric unit contains three silver atoms, two
closo-dodecaborate anions, two acetonitrile molecules and the organic cation. Two silver atoms Ag(1) and Ag(2) are coordinated by two icosahedral cages via Ag…(HB)
2 bridges and acetonitrile molecule, giving a five-fold arrangement. The Ag(3) atom is coordinated by two
closo-dodecaborate anions giving a distorted tetrahedron. The Ag…B bonds fall in the range of 2.426(3)–2.987(3) Å (
Figure 35) [
55].
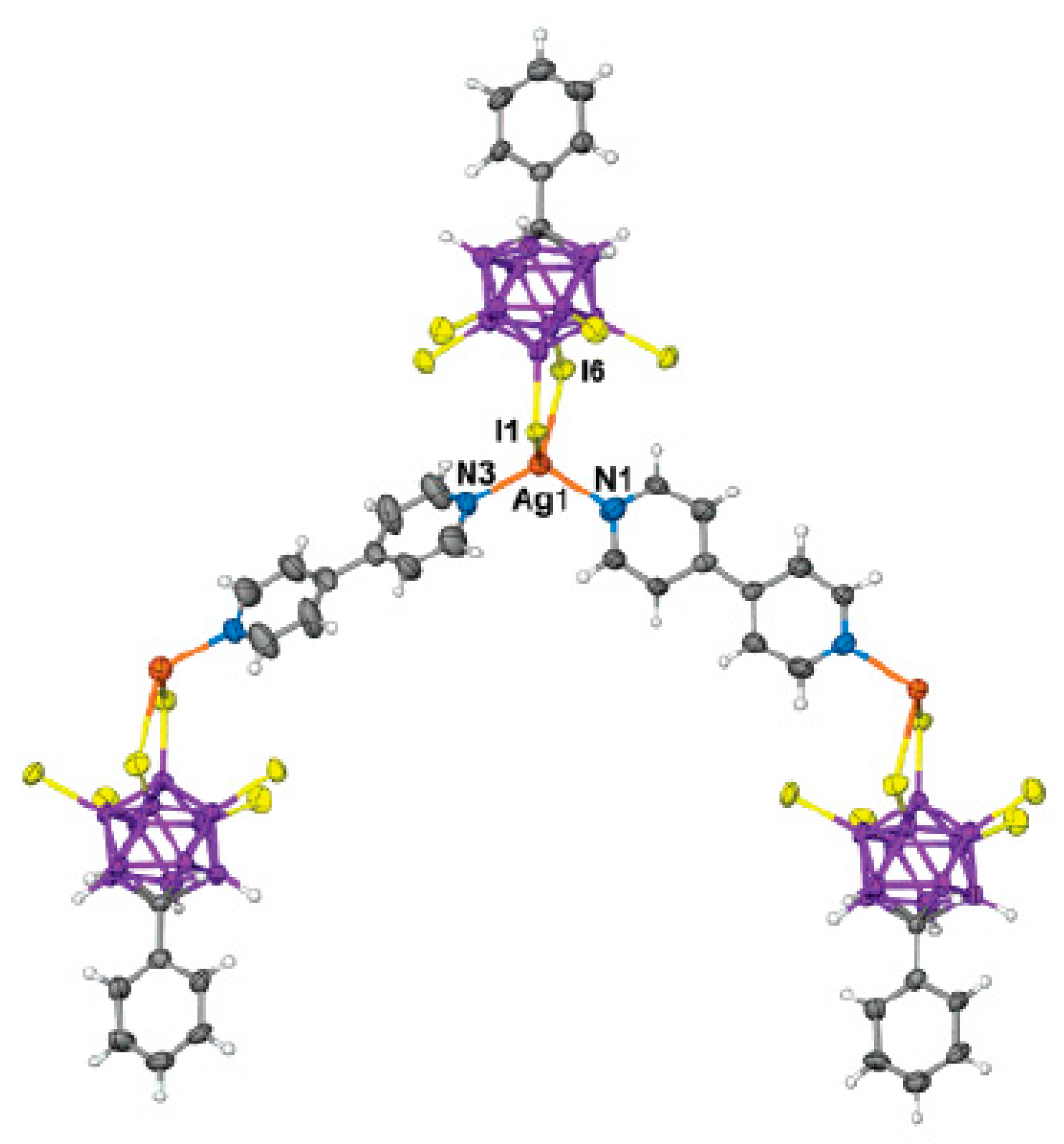
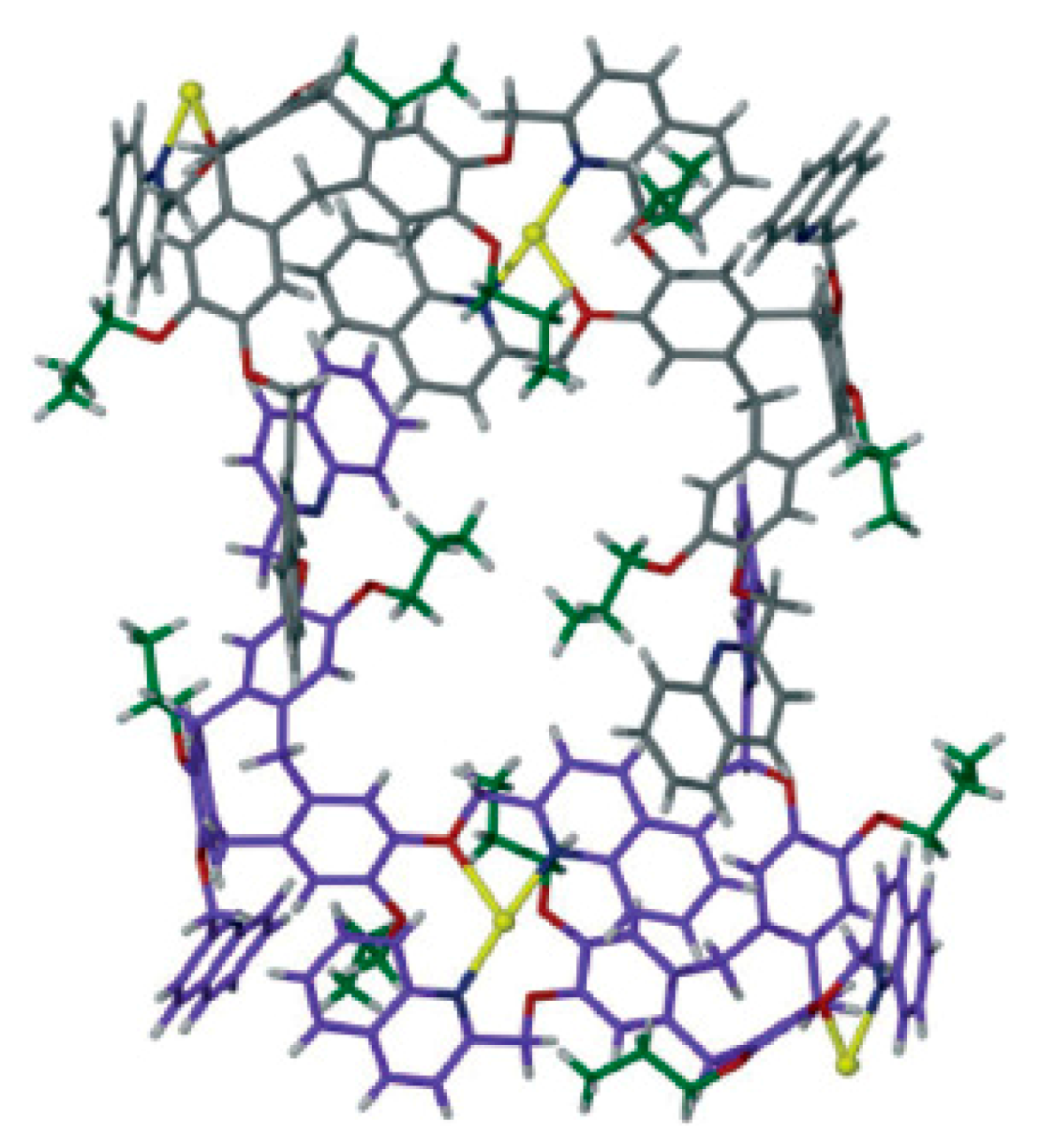
Complex with bis(triphenylphosphine)gold(I) cation {[(Ph
3P)
2Au](Ag[B
12H
12])}
n was prepared by reaction of Ag
2[B
12H
12] with [(Ph
3P)AuCl] in acetonitrile/benzene. The structure of the complex consists of polymeric anionic chains {Ag[B
12H
12]}
+ which are extended along axis с and [(Ph
3P)
2Au]
+ complex cations. The silver atom is coordinated by the B(1)-B(2) edge of one anion and the B(9)-B(10)-B(12) face of the other anion. The Ag…B bond lengths vary from 2.498(11) to 2.738(11) Å. In the [(Ph
3P)
2Au]
+ cation the gold atom has a nearly linear coordination (Au-P bond lengths are 2.338(2) and 2.341(2) Å, P-Au-P angle is 170.91(7)°) (
Figure 36) [
56].
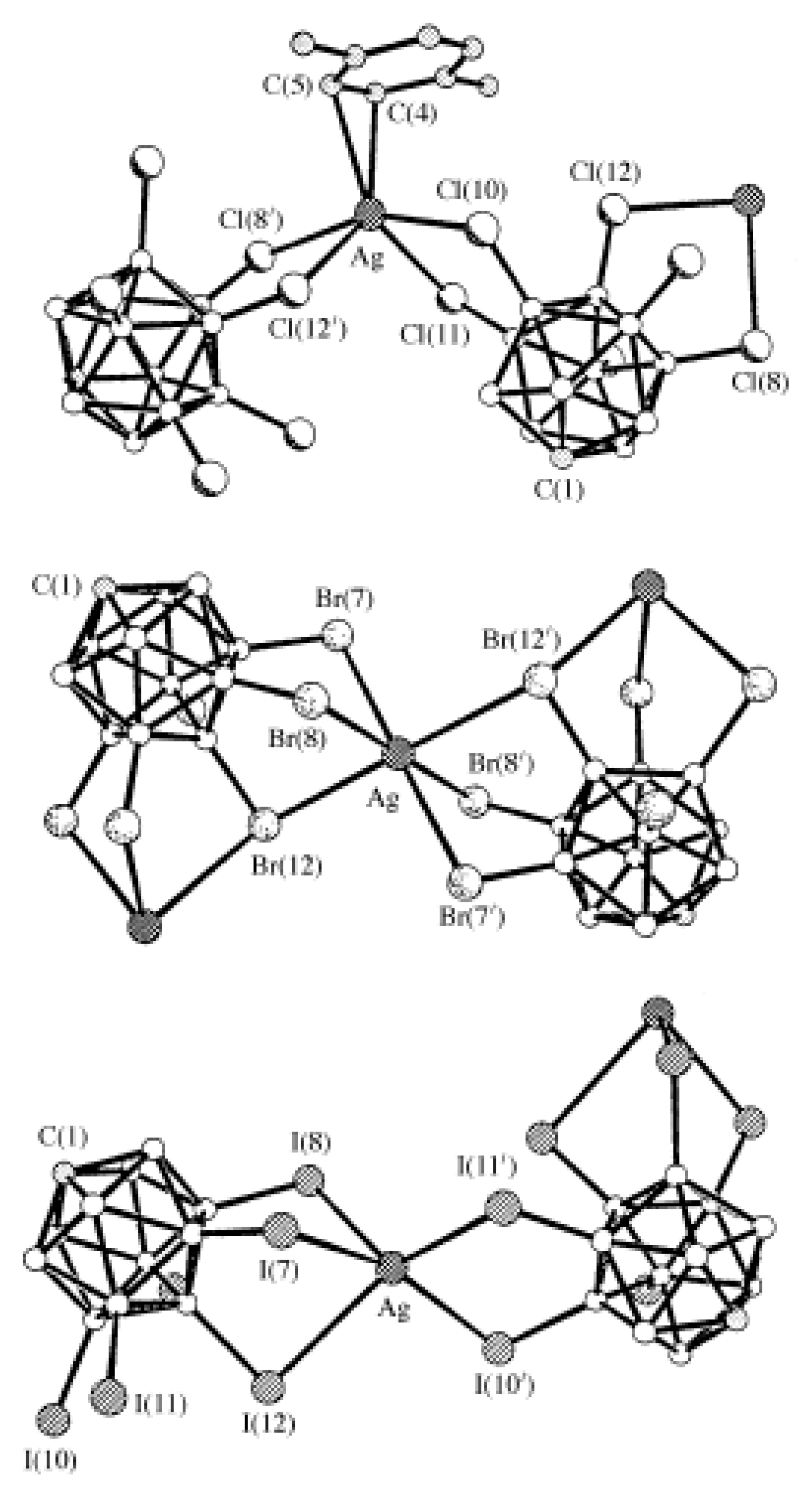
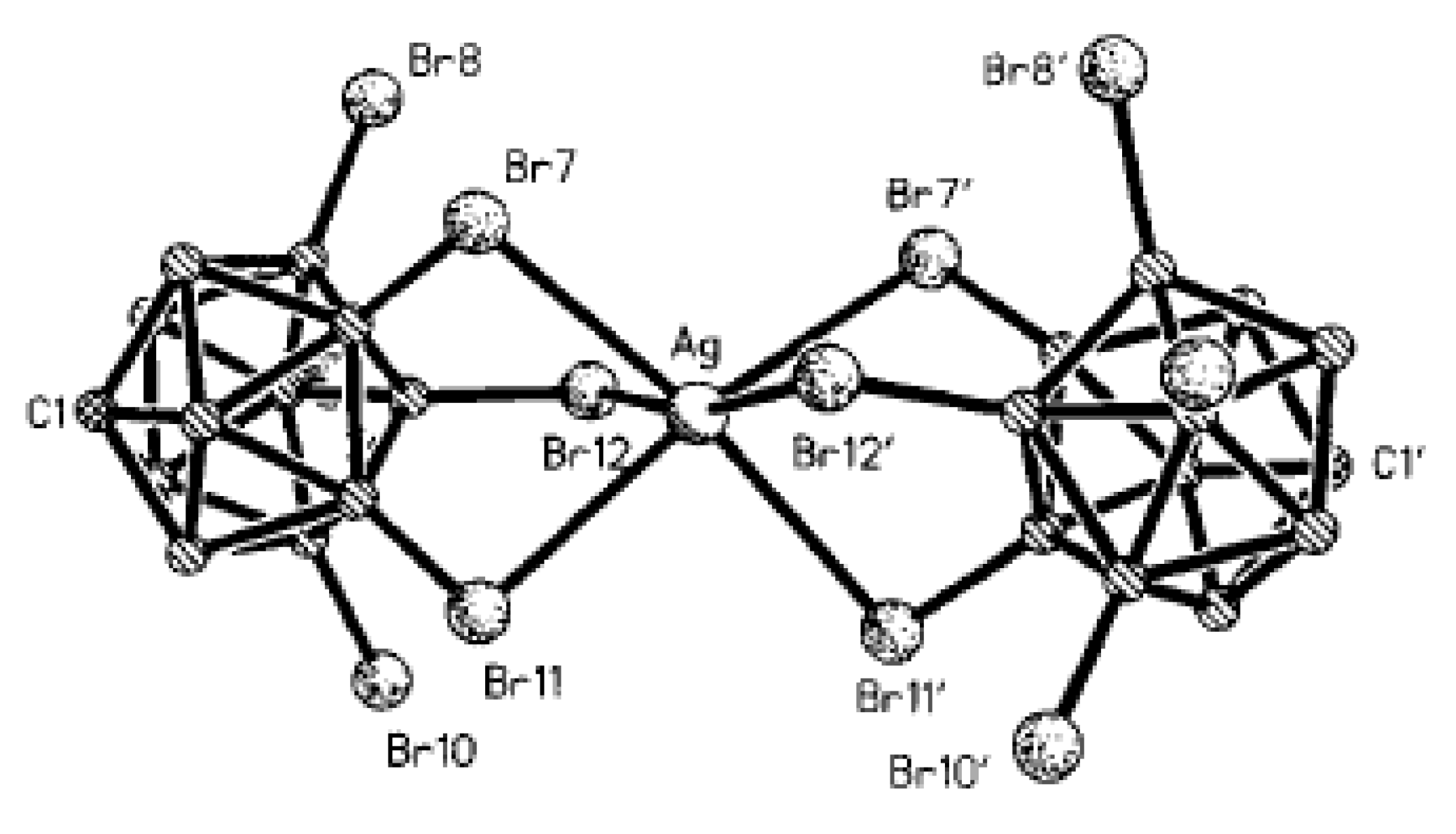
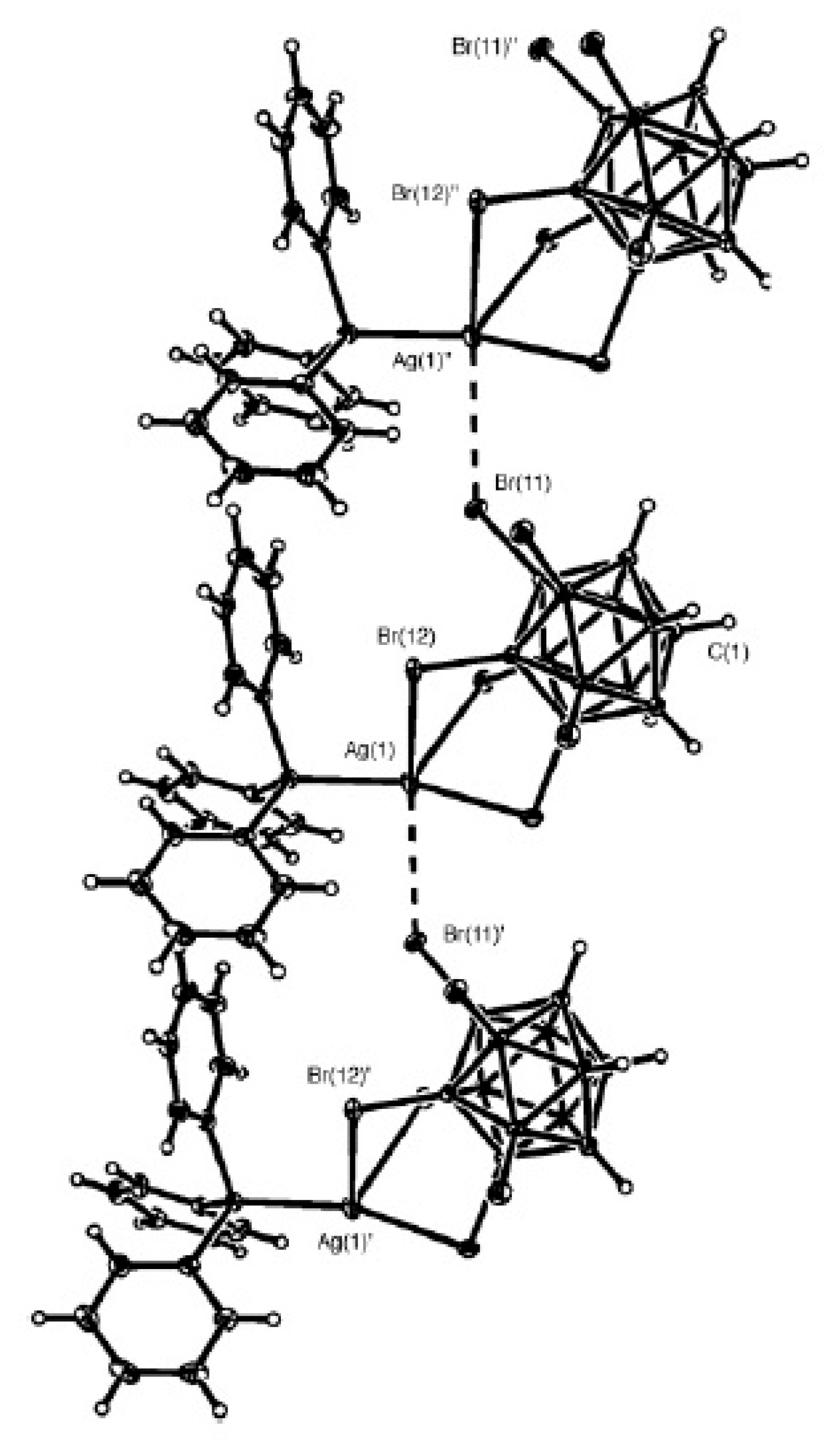
Silver dodecachloro-
closo-dodecaborate {Ag
2[B
12Cl
12]}
n was obtained by the metathesis reaction of Cs
2[B
12Cl
12] with an aqueous solution of AgNO
3 [
57] or by reaction of an aqueous solution of the free acid (H
3O)
2[B
12Cl
12] with Ag
2O [
57,
58]. In the crystal each Ag
+ cation is coordinated by six chlorine atoms from the edges of three [B
12Cl
12]
2− anions providing a distorted octahedral environment (Ag…Cl distances 2.83–2.85 Å) (
Figure 37) [
57].
Complex {((Me
2CO)Ag)
2[B
12Br
12]}
n was prepared by the metathesis reaction of (Et
3NBz)
2[B
12Br
12] with AgOTf in acetone [
55]. In the structure, the Ag
+ cations are coordinated in a square-pyramidal way by four bromine atoms from two [B
12Br
12]
2− anions at the basis and by acetone molecule at the apex. Each dodecabromo-
closo-dodecaborate anion is coordinated by four {(Me
2CO)Ag}
+ cations. The Ag…Br bonds fall in the range of 2.731–2.906 Å, the Ag-O bond is 2.344 Å (
Figure 38).
Lowering the anion charge from 2- to 1- by an introduction of a charge-compensating group decreases its coordination ability. Complex [Ag(MeCN)
4][B
12Cl
11NH
3]·3(MeCN) prepared by crystallization from acetonitrile consists of the discrete [Ag(MeCN)
4]
+ cations, the [B
12Cl
11NH
3]
− anions and acetonitrile solvate molecules (
Figure 39) [
59].
Several complexes of
closo-dodecaborate anion with triphenylphosphine and di(2-pyridyl)amine ligands were prepared. In the structure of {((Ph
3P)
2Cu)
2[B
12H
12]} two {(Ph
3P)
2Cu}
+ fragments coordinated to opposite edges of the
closo-dodecaborate anion. The Сu…B bonds are 2.392(18) and 2.387(17) Å, Сu…Н(B) bonds are 1.82(13) and 1.88(15) Å (
Figure 40) [
60].
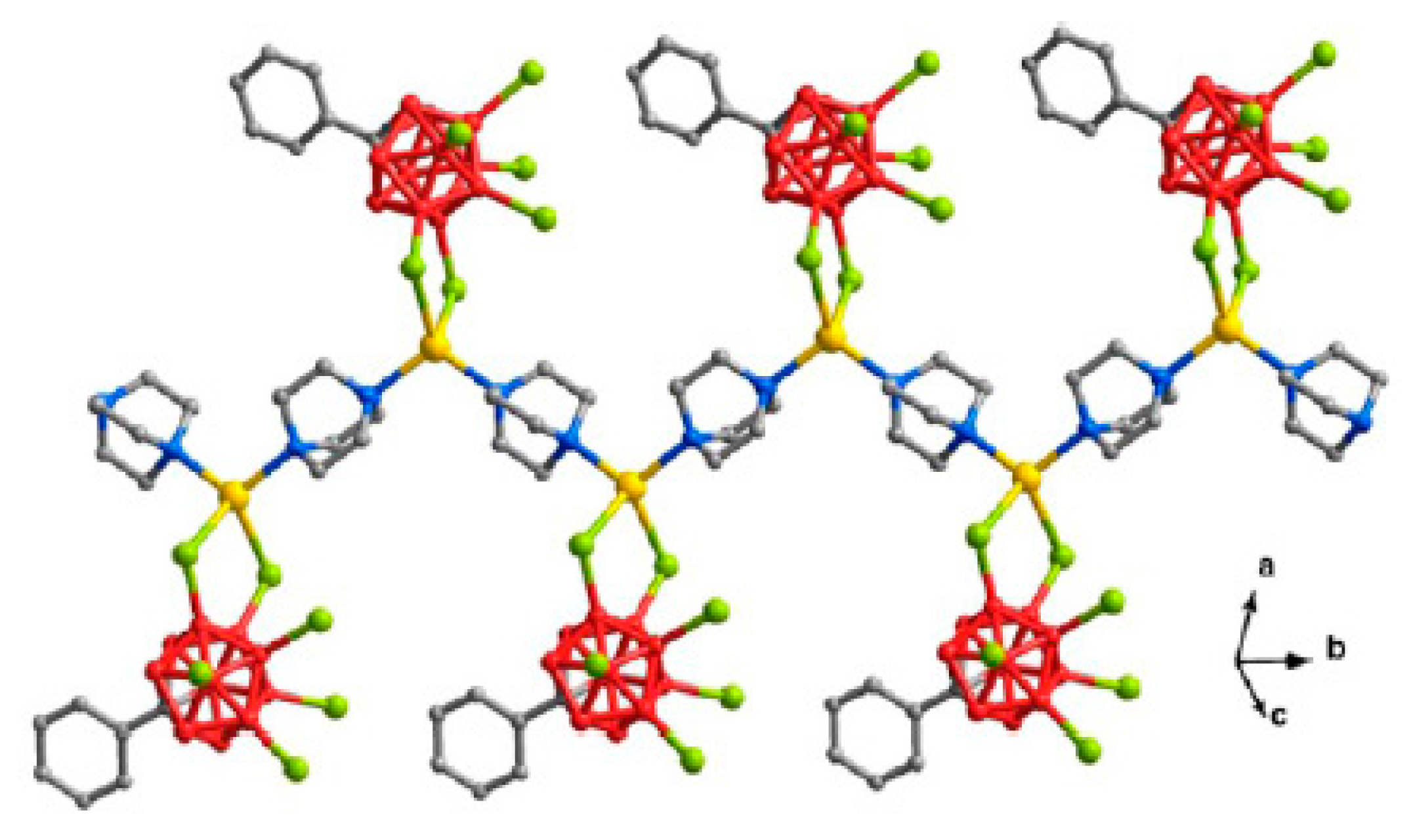
The polymeric complex {((BPa)Ag)
2[B
12Н
12]}
n was prepared by the reaction of Cs(Ag[B
12Н
12]) or Ag
2[B
12H
12] with di(2-pyridyl)amine in acetonitrile. The complex is built of centrosymmetric binuclear {(BPa)Ag[B
12H
12]Ag(BPa)} units connected into chains running along the
a axis. The silver cation is coordinated by the nitrogen atoms of the chelating di(2-pyridyl)amine ligand (Ag-N bonds are 2.288(3) and 2.320(3) Å), the B(1)H group of the
closo-dodecaborate anion (Ag…B and Ag…H bonds are 2.544(4) and 1.87 Å, respectively, Ag-H-B angle is 114º), and the H(4) atom of the neighboring anion. The interaction between the neighboring complexes is apparently weak: Ag…H distance is 2.14 Å, the Ag-H-B angle is open to 143º, and Ag…B distance of 3.108(4) Å is too long to be considered a bond. In the crystal di(2-pyridyl)amine molecules pack into stacks with the interplanar spacing of ~3.50 Å (
Figure 41) [
61].
The reaction of Cs(Ag[B
12Н
12]) or Ag
2[B
12H
12] with di(2-pyridyl)amine in DMF produces discrete complex {((Bpa)Ag)
2[B
12Н
12]·2(DMF)}. The {((Bpa)Ag)
2[B
12Н
12]} fragment is centrosymmetric. The silver atoms has a trigonal coordination formed by two nitrogen atoms of the chelating di(2-pyridyl)amine ligand (Ag-N, 2.2745(15) Å) and the BH group of the
closo-dodecaborate anion (Ag…B and Ag…H bonds are 2.413(3) and 1.92(4) Å, respectively, Ag-H-B angle is 99(2)º). In addition, silver atom has long contacts with two other BH groups (Ag…B, 2.7954(18) Å; Ag…H, 2.60(2) Å). In the crystal two DMF molecules are attached to amino groups of di(2-pyridyl)amine ligands by N-H…O hydrogen bonds (
Figure 42) [
61].
The mixed-ligand complex {((BPa)(Ph
3P)Ag)
2[B
12Н
12]} was prepared by reaction of Cs(Ag[B
12H
12]) with triphenylphosphine and di(2-pyridyl)amine in a DMF/acetonitrile/benzene mixture. Crystals of this compound are built of centrosymmetric binuclear complexes [(BPa)(PPh
3)Ag(B
12H
12)Ag(Bpa)(PPh
3)]. The pyridine nitrogen atoms of di(2-pyridyl)amine and the phosphorus atom of triphenylphosphine form strong bonds with the silver cation (Ag-N, 2.356(2) and 2.369(3) Å; Ag-P, 2.3963(7) Å). The trigonal coordination of silver is complemented by two weak bonds with the BH groups of the
closo-dodecaborate anion (Ag(1)…B(1), 2.831(3); Ag(1)…H(1), 2.32(3) Å; Ag(1)…B(2), 2.886(3); Ag(1)…H(2), 2.43(3) Å) (
Figure 43). Although the Ag…B distances in this complex are much longer than in {((BPa)Ag)
2[B
12Н
12]·2(DMF)} and {((BPa)Ag)
2[B
12Н
12]}
n (2.413-2.544 Å), the Ag-H-B stretching band at 2389 cm
−1 in the IR spectrum unambiguously indicates the existence of Ag-H-B interactions between the silver atom and the
closo-dodecaborate anion [
61].
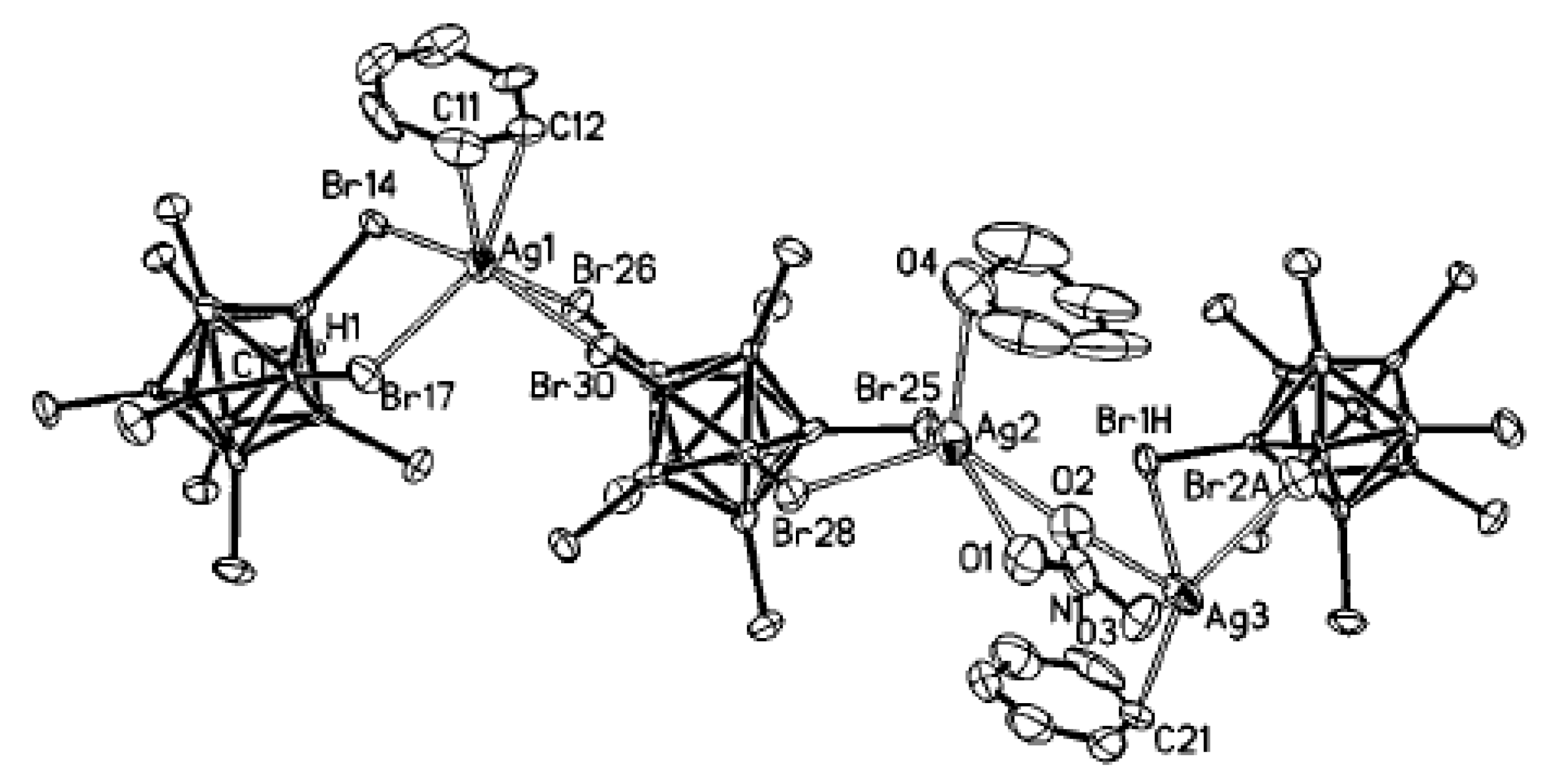
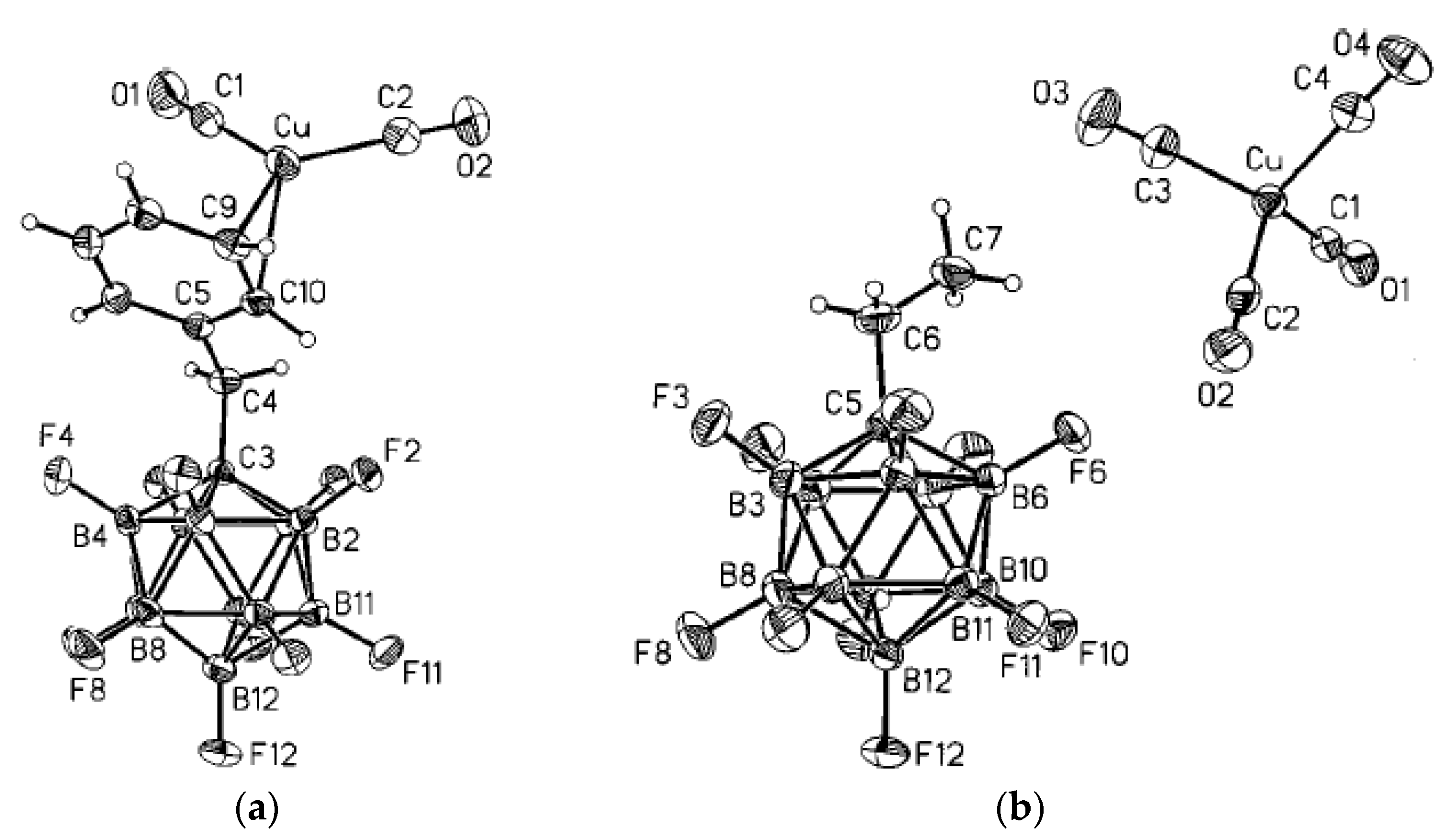
Complex {(HAgu)
2[Cu(Agu)
2][B
12H
12]
2} was prepared by reaction of CuSO
4 with (НAgu)
2[B
12H
12] in aqueous solution. The structure is built of [Cu(Agu)
2]
2+ and [(HAgu)
2]
2+ cations and the [B
12H
12]
2− anions. The square planar environment of copper atom is formed by two flat aminoguanidine molecules, which close five-membered chelate cycles (Cu-N, 1.9702(13) and 2.0261(10) Å; N-Cu-N angle, 81.53(4)°). In addition, copper atom forms two weak bonds with hydrogen atoms of two
closo-dodecaborate anions (Cu…H, 2.805(14) Å; B-H-Cu angle, 132.5(10)°). In the cells where copper positions are vacant, pairs of HAgu
+ cations are connected by the intermolecular N(1)-H(13)…N(4) hydrogen bonds into centrosymmetric dimers. The N(4)H atom is also involved in intermolecular dihydrogen bond with the B(3)H atom (
Figure 44) [
62].
The complexation between the
closo-dodecaborate anion and cyclic copper(I) and silver(I) 3,5-bis(trifluoromethyl)pyrazolates {[3,5-(CF
3)
2Pz]M}
3 (M = Cu, Ag) in CH
2Cl
2 was studied by IR spectroscopy. Formation of two types of complexes, 1:1 and 1:2, was revealed and their stability constants were determined. It was demonstrated that
closo-dodecaborate anion forms less stable complexes than the
closo-decaborate anion [
47].
4. Silver Complexes with the 1-carba-closo-decaborate anion [1-CB9H10]−
Synthesis of the 1-carba-
closo-decaborate anion [1-CB
9H
10]
− anion was reported for the first time almost fifty years ago in the late 1960s [
63,
64], its coordination chemistry has been studied much less than chemistry of other weakly coordinated anions due to the absence of practical methods of its synthesis until 2000-s [
65]. The situation was changed when convenient approach to synthesis of the parent anion and its
C-aryl derivatives via reaction of decaborane with aldehydes in alkali media was elaborated [
66,
67,
68,
69,
70,
71,
72].
The silver salt of the parent 1-carba-
closo-decaborate anion, Ag[1-CB
9H
10], can be prepared by metathesis of the cesium salt with AgNO
3 in aqueous solution followed by crystallization from toluene [
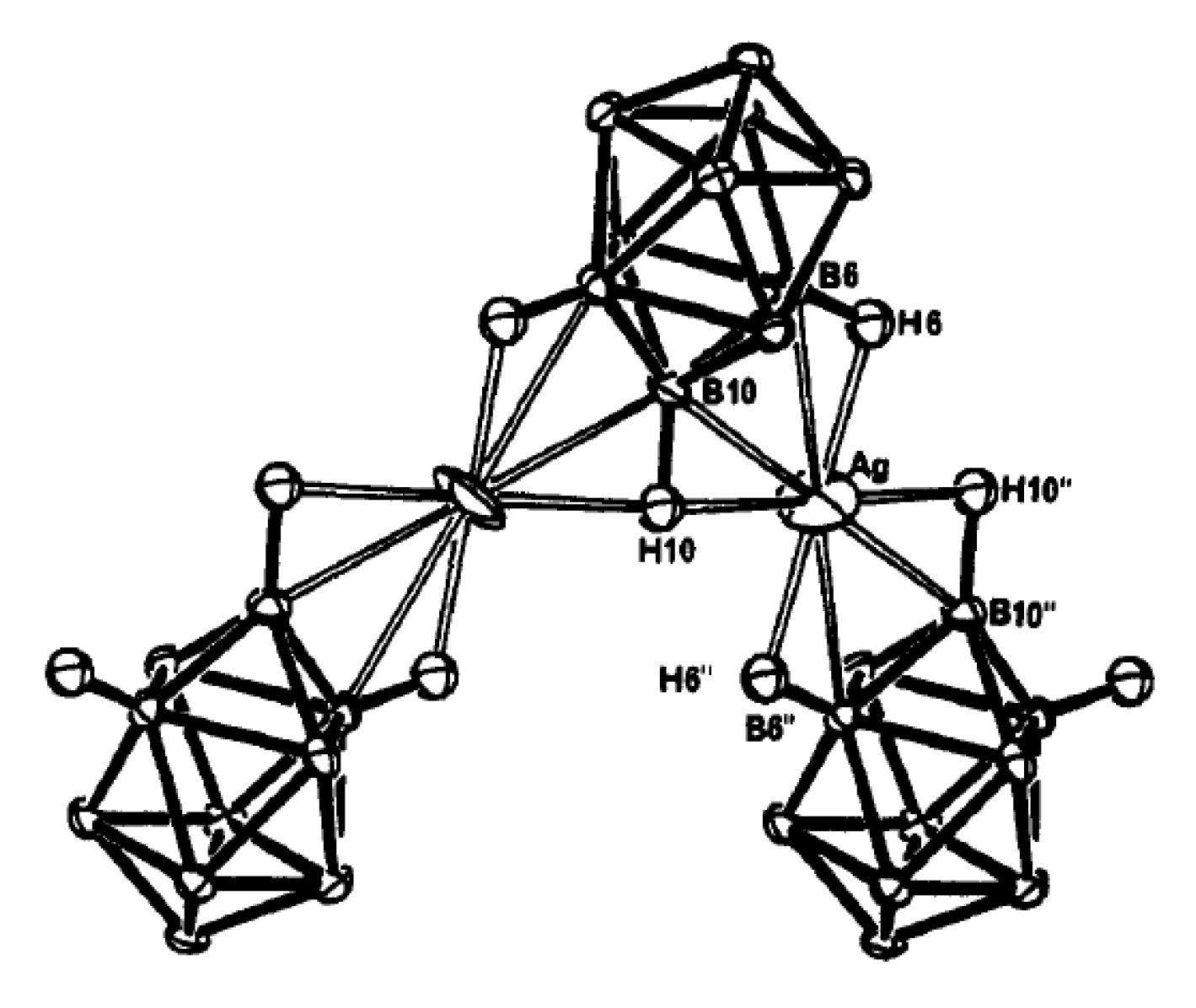
73]. In the crystal the coordination geometry of silver approximates linear two-coordination except that the [1-CB
9H
10]
− anions provide η
2-like coordination via the antipodal to carbon B(6)-B(10) and B(8)-B(10) edges to form infinite polymeric chain. The silver atom is closer to B(6) (2.496(3) Å) than to B(10) (2.578(2) Å); however, this probably does not indicate site nucleophilicity on the carborane cage, but rather reflects the fact that the B(10)-H bond is shared by two silver atoms (
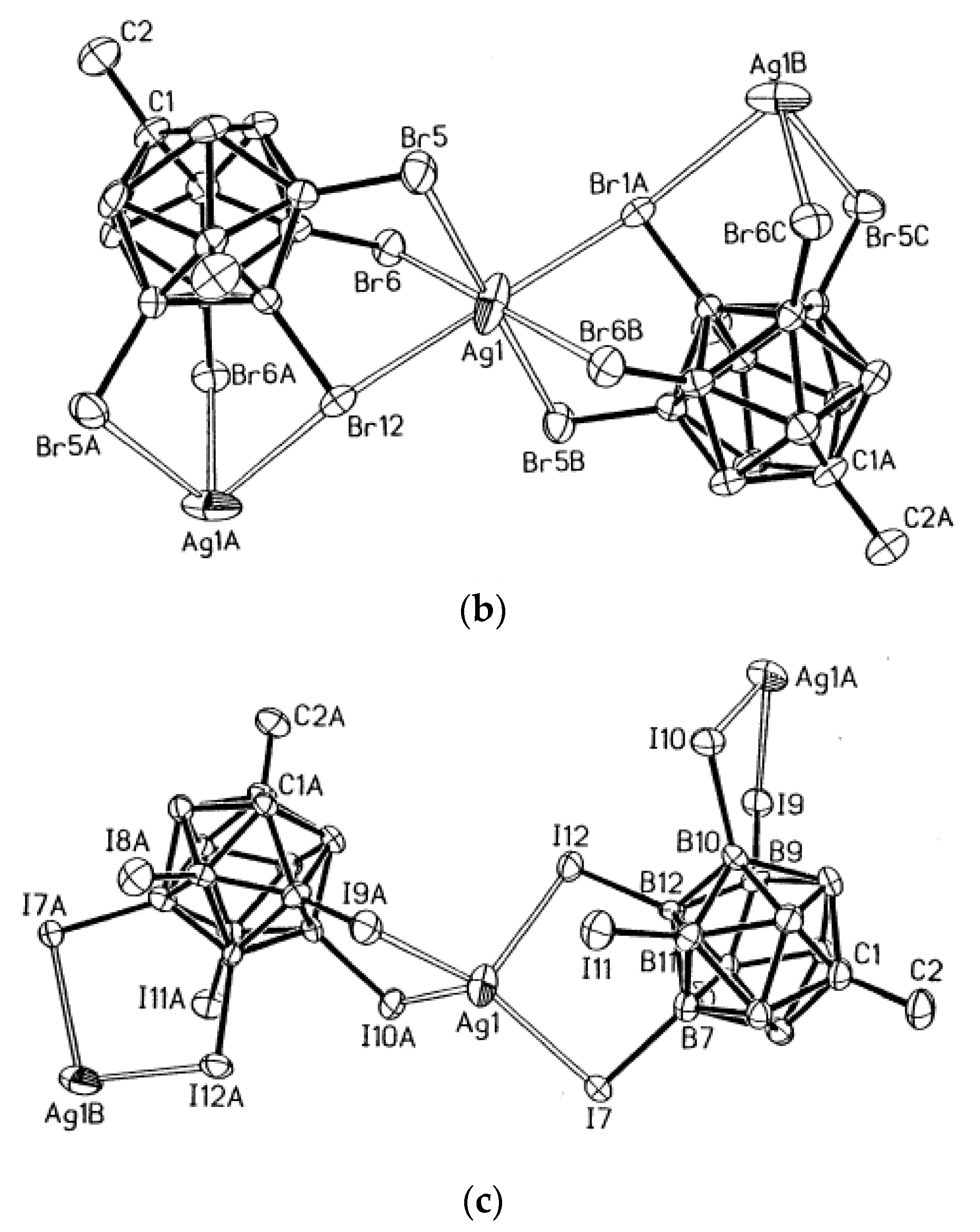
Figure 45) [
73].
The silver salt of 6,7,8,9,10-pentabromo-1-carba-
closo-decaborate anion, Ag[1-CB
9H
5-6,7,8,9,10-Br
5], was prepared by the metathesis of Cs[1-CB
9H
5-6,7,8,9,10-Br
5] with AgNO
3 from aqueous solution [
73]. The toluene solvate {Ag(η
2-toluene)[1-CB
9H
5-6,7,8,9,10-Br
5]}
n was obtained by crystallization from toluene/
n-hexane. The coordination sphere of silver is formed by η
2-toluene, one bidentate carborane anion, and one monodentate carborane anion. The anions act as bridging ligands to a zigzag linear chain of silver ions. The η
2 coordination of toluene has Ag-C distances of about 2.62 Ǻ. The coordination of the anion is notable on two counts. Firstly, the bridging disposition is quite unsymmetrical, making it “belong” more to one silver atom than another. This is true of both denticity and bond length. The bidentate binding is characterized by Ag-Br bond lengths of ~2.78 Å, whereas the monodentate interaction is weaker at 2.842(6) Å. Secondly, in the bidentate interaction, the Ag-Br(10) and Ag-Br(6) bond lengths are practically indistinguishable (2.783(6) and 2.775(6) Å, respectively) (
Figure 46) [
73].
The silver salts of C-substituted perchloro and perbromo derivatives of 1-carba-
closo-decaborate anion, Ag[1-Bn-1-CB
9Cl
9] and Ag[1-H
2N-1-CB
9Br
9], were prepared by treatment of the corresponding sodium salts with AgNO
3 in aqueous solution [
74]. The silver salt of the perbromo derivative of 1-carba-
closo-decaborate anion, Ag[1-HCB
9Br
9], was prepared by the metathesis of Cs[1-HCB
9Br
9] with AgNO
3 from aqueous solution and crystallized from benzene/THF solution to give {Ag(η
2-benzene)[1-HCB
9Br
9]}
n [
74]. The [1-HCB
9Br
9]
− anions act as bridging ligands in a one-dimensional coordination polymer chain, an increasingly familiar motif in silver carborane structures. The silver atom is in a trigonal arrangement of one η
2-benzene and two monodentate bridging carborane anions. It is noteworthy that the [1-HCB
9Br
9]
− anion coordinates to the silver atom through the Br(6) atom rather than Br(10) antipodal to the carbon atom. The average Ag-Br distance of 2.778 Å is close to those found in {Ag(η
2-toluene)[1-CB
9H
5-6,7,8,9,10-Br
5]}
n. The asymmetric η
2 benzene coordination has Ag-C distances of 2.629(8) and 2.649(7) Å (
Figure 47) [
74].
At the same time crystals of {(η
2-benzene)(η
1-benzene)Ag
3[1-HCB
9Br
9]
2[NO
3](THF)}
n were obtained. The X-ray analysis reveals that this complex is a one-dimensional coordination polymer with both carborane and nitrate anions acting as bridging ligands. Each Ag atom in asymmetric unit has a unique coordination environment although they are all five-coordinated. Ag(1) is in a propeller-like arrangement having one η
2-benzene and two bidentate bridging [1-HCB
9Br
9]
− anions. Ag(2) is bound to one bidentate [1-HCB
9Br
9]
− anion, one bidentate NO
3− anion, and the oxygen atom of the coordinated THF molecule in a distorted square-pyramidal geometry. Ag(3) is in a highly distorted square-pyramidal arrangement having one η
1-benzene, one bidentate [1-HCB
9Br
9]
− anion, and one bidentate NO
3− anion. It is noteworthy that a bromine atom from the upper tetragonal belt can also coordinate to an Ag
+ ion (
Figure 48) [
74].
Synthesis of silver salts of some other derivatives of the 1-carba-
closo-decaborate anion (Ag[1-Ph-1-CB
9H
9] [
75], Ag[1-PhCH
2-1-CB
9Cl
9] [
74], Ag[1-NH
2-1-CB
9Br
9] [
74], Ag[1-Ph-1-CB
9H
8-6-I] [
76], Ag[1-Ph-1-CB
9H
4-6,7,8,9,10-I
5] [
75]) was described as well.
While the relatively labile chloride ligand in complexes MCl(CO)(PPh
3)
2 (M = Ir, Rh) can be readily abstracted with silver perchlorate or triflate, the reactions with Ag[1-CB
9H
5-6,7,8,9,10-Br
5] result in M→Ag donor-acceptor metal-metal bonded adducts (PPh
3)
2(CO)ClM·Ag[1-CB
9H
5-6,7,8,9,10-Br
5] that were isolated in the solid state [
73]. The iridium complex was characterized by single crystal X-ray diffraction to adopt the expected square pyramid silver-iridium Lewis acid-base structure. The [1-CB
9H
5-6,7,8,9,10-Br
5]
− anion is coordinated to silver atom through three bromine atoms realizing the η
3-coordination mode. The Ag-Br distances range from 2.721(8) to 2.973(6) Å (
Figure 49) [
73].
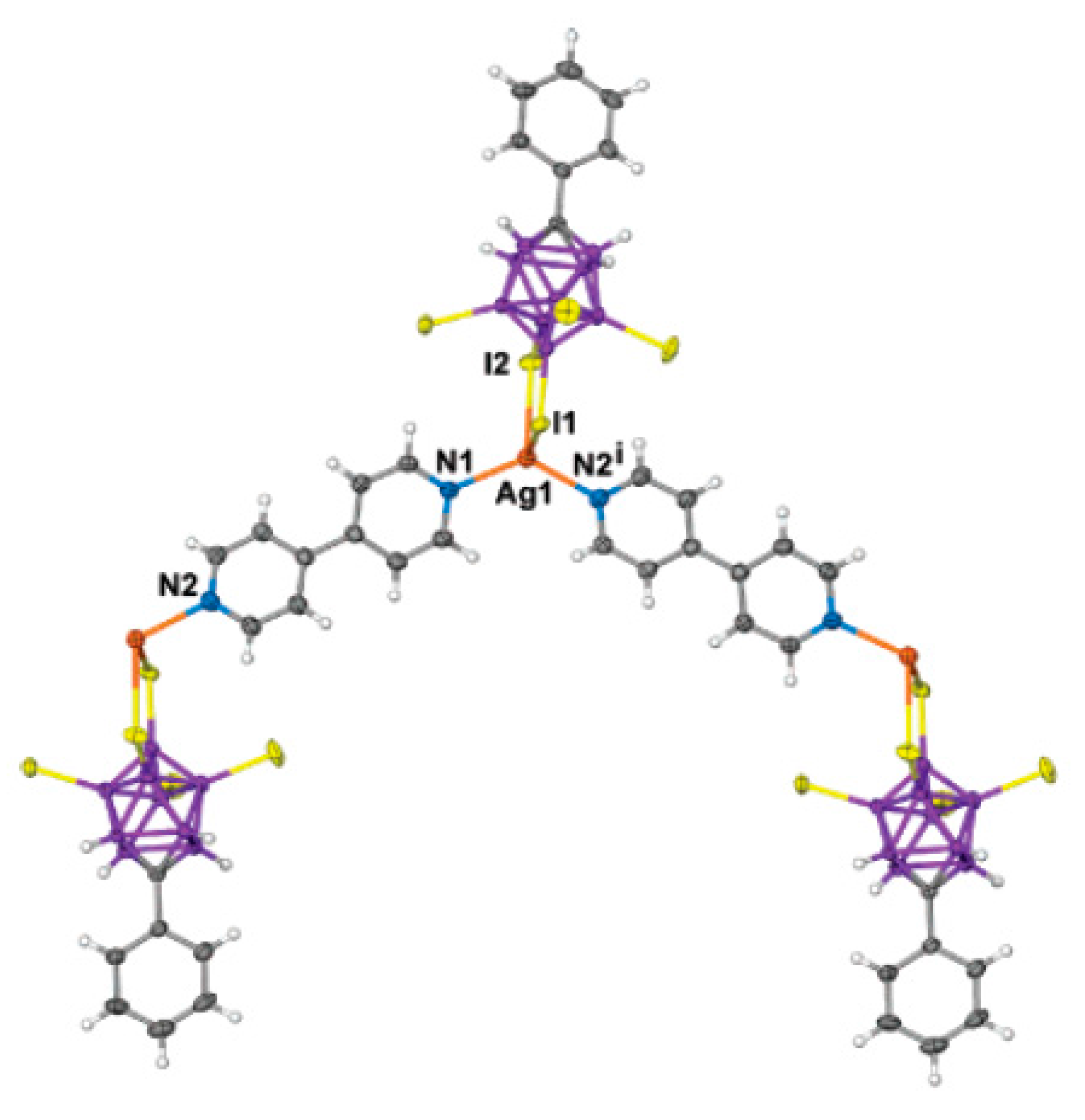
Complex {[Ag(4,4′-Bipy)][1-Ph-1-CB
9H
9]·MeCN}
n was prepared by reaction of Ag[1-Ph-1-CB
9H
9] with 4,4′-bipyridine in acetonitrile [
75]. The Ag(I) has two coordinate bonds to two 4,4′-Bipy ligands in a near-linear fashion, with an angle N(1)-Ag-(1)-N(2) of 177.42(15)° and with Ag-N distances of Ag(1)-N(1) 2.132(4) and Ag(1)-N(2) 2.144(5) Å. Each 4,4′-bipyridine ligand bridges two silver atoms to give an infinite linear coordination chain. The chains group together in pairs, with the Ag(I) center of one chain positioned directly above the center of the C(4)-C(4′) bond of the 4,4′-bipyridine of the second chain at a closest Ag…C distance of 3.585 Å. There are face-to-face π-stacking interactions between the arene rings of each chain at a centroid separation of 3.793 Å. The carborane anions do not form any close interactions with other molecular components. Both [1-Ph-1-CB
9H
9]
− anion and MeCN solvent molecules are arranged in a fashion that creates unidirectional channels of rhomboid cross section that run along the
y-direction and contain the [Ag(4,4′-Bipy)]
+ coordination chains (
Figure 50).
Similar complex with iodinated carborane anion {Ag(4,4′-Bipy)[1-Ph-1-CB
9H
4-6,7,8,9,10-I
5]· MeCN}
n was prepared by the reaction of Ag[1-Ph-1-CB
9H
4-6,7,8,9,10-I
5] with 4,4′-bipyridine in acetonitrile [
75]. In contrast to the previous complex, the silver atom here has a distorted tetrahedral geometry with two coordinate bonds to two 4,4′-Bipy ligands with an angle N(1)-Ag(1)-N(2) of 127.42(11)° and two symmetrically equivalent Ag-N bonds of 2.271(4) Å. The coordination sphere is completed by two iodine atoms of the chelating [1-Ph-1-CB
9H
4-6,7,8,9,10-I
5]
− anion, which coordinates to the metal center at Ag-I distances of Ag(1)-I(1) 2.9851(5) and Ag(1)-I(2) 2.8642(10)Å. The [Ag(4,4′-Bipy)]
+ coordination chain is puckered to create a zigzag chain. At each silver center along the chain the coordinated [1-Ph-1-CB
9H
4-6,7,8,9,10-I
5]
− anions point in alternating directions (
Figure 51) [
75].
Complex {Ag(bppz)[1-Ph-1-CB
9H
9]·0.5MeCN}
n was prepared by reaction of Ag[1-Ph-1-CB
9H
9] with 2,3-bis(2-pyridyl)pyrazine in acetonitrile [
75]. The silver atom has a distorted six-coordinate geometry, arising from two bidentate chelating interactions with two symmetry-related bppz ligands with Ag-N distances in the range of 2.325(2) and 2.593(2) Å and from two Ag…H-B interactions at distances of 2.291 Å (Ag(1)…H(103)) and 2.471 Å (Ag(1)…H(101)), both involving the same [1-Ph-1-CB
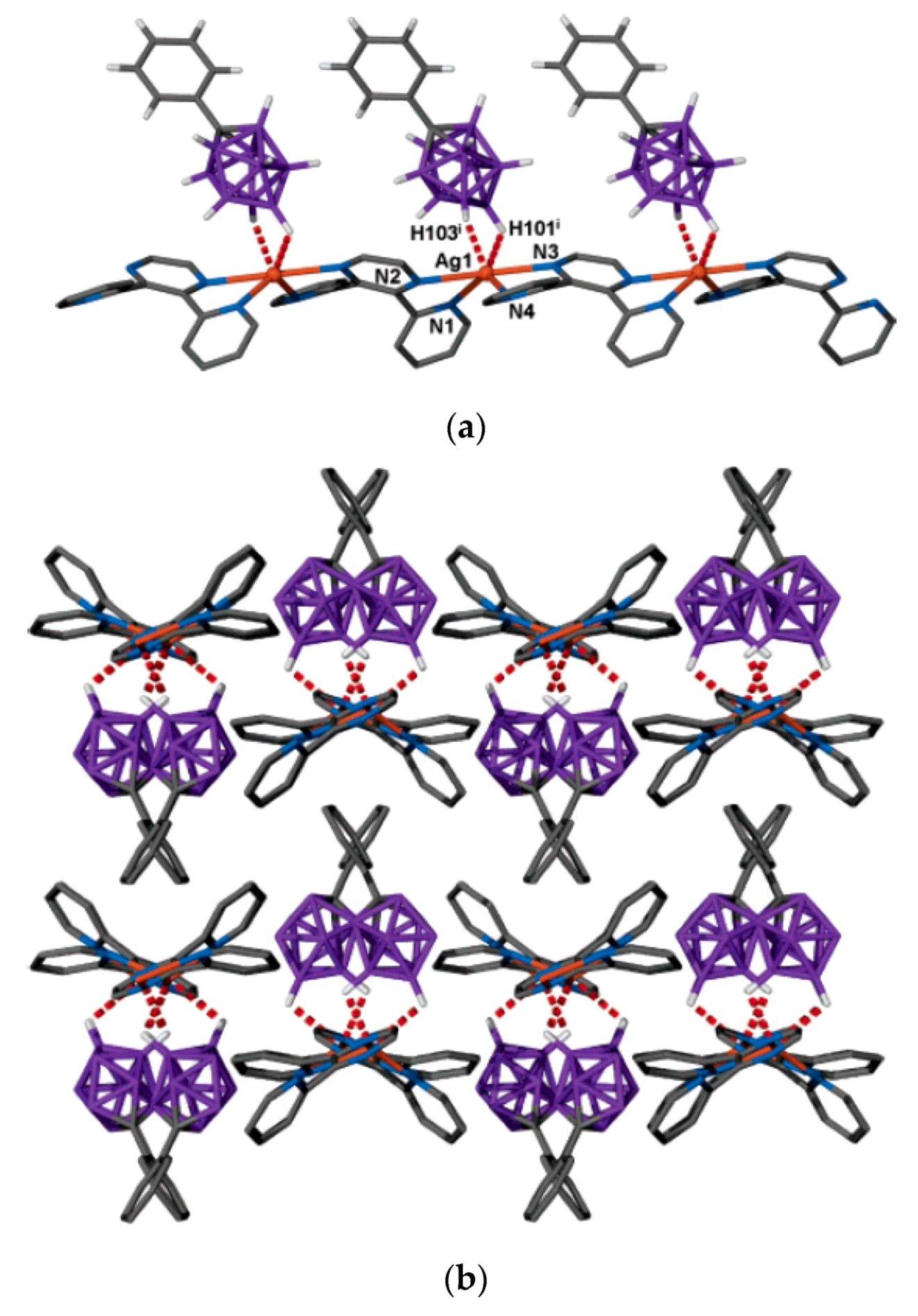
9H
9]
− anion. The 2,3-bis(2-pyridyl)pyrazine ligand acts as a bis-chelating bridging ligand between the Ag(I) centers and thereby propagates a coordination chain that runs parallel with the crystallographic
c-axis (
Figure 52a) [
75]. The coordinated anions are all on one side of the chain but show two alternating orientations along the chain. The chains stack with an identical orientation in the
x-direction, such that the phenyl groups of the anions of one chain slot into the grooves created by the pyridine rings of the bppz ligands of an adjacent chain. The orientation of the chains alternates along the
y-direction (
Figure 52b) [
75].
In contrast to the other complexes with the 1-carba-
closo-decaborate anion reported here, the reaction of Ag[1-Ph-1-CB
9H
8-6-Br] with 2,3-bis(2-pyridyl)pyrazine in acetonitrile produces discrete dimeric complex {Ag(bppz)[1-Ph-1-CB
9H
8-6-Br]}
2 [
75] rather than polymeric one. The silver atom has an irregular tetrahedral geometry: Ag(1) is coordinated with two bppz ligands, to form a chelating interaction to one ligand [Ag-N distances of Ag(1)-N(1) 2.332(10) and Ag(1)-N(3) 2.372(9) Å], with the third coordination bond occurring to the pyridyl nitrogen atom of a second bppz ligand (Ag(1)-N(2) 2.268(9) Å). The coordination environment of silver is completed with the bromine atom of the [1-Ph-1-CB
9H
8-6-Br]
− (Ag(1)-Br(1) 2.9382(11) Å) (
Figure 53).
Complex {Ag(DABCO)[1-Ph-1-CB
9H
9]}
n was prepared by reaction of Ag[1-Ph-1-CB
9H
9] with 1,4-diazabicyclo[2.2.2]octane in acetonitrile [
77]. The silver atom has a distorted tetrahedral geometry with two coordinate bonds to two crystallographic equivalent DABCO ligands with an angle N(1)-Ag(1)-N(2) of 123.22(1)° and Ag-N bonds of 2.2750(18) and 2.2713(18) Å for Ag(1)-N(1) and Ag(1)-N(2), respectively. Furthermore, it interacts with two BH groups of the [1-Ph-1-CB
9H
9]
− anion, the Ag…H-B interaction with equatorial BH group being much stronger (1.8623(4) Å) than the Ag…H-B interaction with apical BH group (2.3529(6) Å) which can be considered as an agnostic interaction. The 1D coordination network is puckered to create the [Ag(DABCO)]
+ zigzag chain. At each silver center along the chain the coordinated [1-Ph-1-CB
9H
9]
− anions point in alternating directions (
Figure 54) [
77]. These coordination-chain structural features (zigzag chain with the adjacent coordinative anions pointing in opposite directions) are considerably similar to that reported for the complex {Ag(4,4′-Bipy)[1-Ph-1-CB
9H
4-6,7,8,9,10-I
5]·MeCN}
n (see above).
Similar complex with monoiodinated carborane {Ag(DABCO)[1-Ph-1-CB
9H
8-6-I]}
n was prepared by reaction of Ag[1-Ph-1-CB
9H
8-6-I] with 1,4-diazabicyclo[2.2.2]octane in acetonitrile as well [
77]. The silver atom establishes coordination bonds with two nitrogen atoms from two crystallographically equivalent DABCO ligands (Ag(1)-N(1) and Ag(1)-N(2) bonds of 2.263(2) and 2.260(2) Å, respectively), and the iodine atom of the [1-Ph-1-CB
9H
8-6-I]
− anion (Ag(1)-I(1) distance of 2.9155(4) Å). The Ag(1)-N and Ag(1)-I coordinative interactions originate the formation of 1D coordination chains with the carborane anions incorporated in the chains. As was observed in the previous case, the coordination chains run along the
a axis with some zigzag arrangement. However, in contrast with the previous chain, all the carbaborane anions point in the same direction (
Figure 55) [
77].
Cyclotriveratrylene (CTV) and its analogues (
Scheme 3) represent a class of shape-persistent C
3-symmetric molecular hosts derived from the tribenzo[a,d,g]cyclononatriene scaffold. CTV is able to form crystalline clathrate complexes with various small organic molecules, in which the solvent guest may be contained in the molecular cavity of cyclotriveratrylene. CTV and its analogues have also demonstrated an affinity for globular, electron-poor guests, such as fullerenes and carboranes, which form “ball-in-socket” superstructures. Therefore, study of interactions of cyclotriveratrylene and its analogues with weakly-coordinating carboranes and metallacarboranes is very exciting [
78].
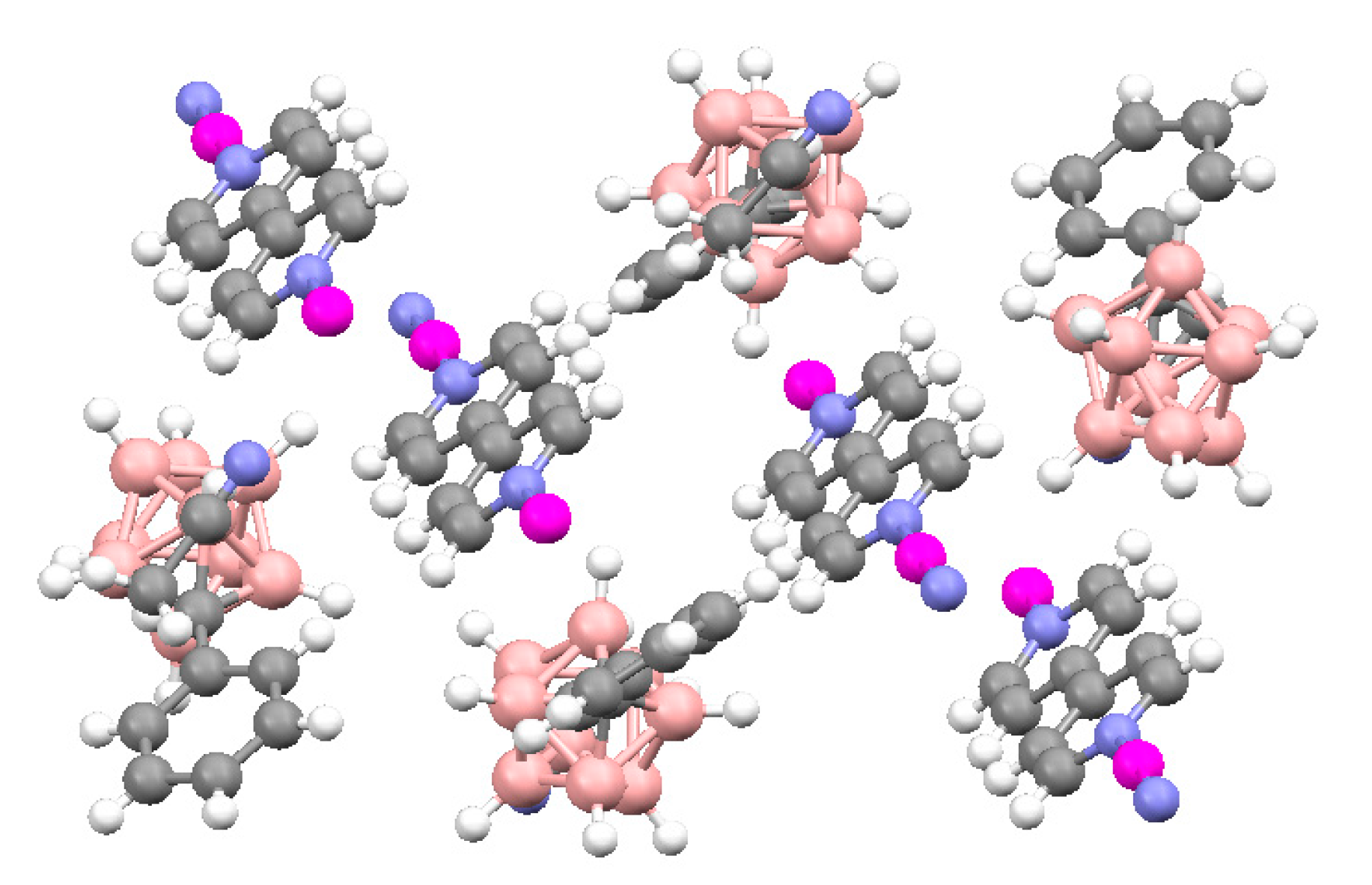
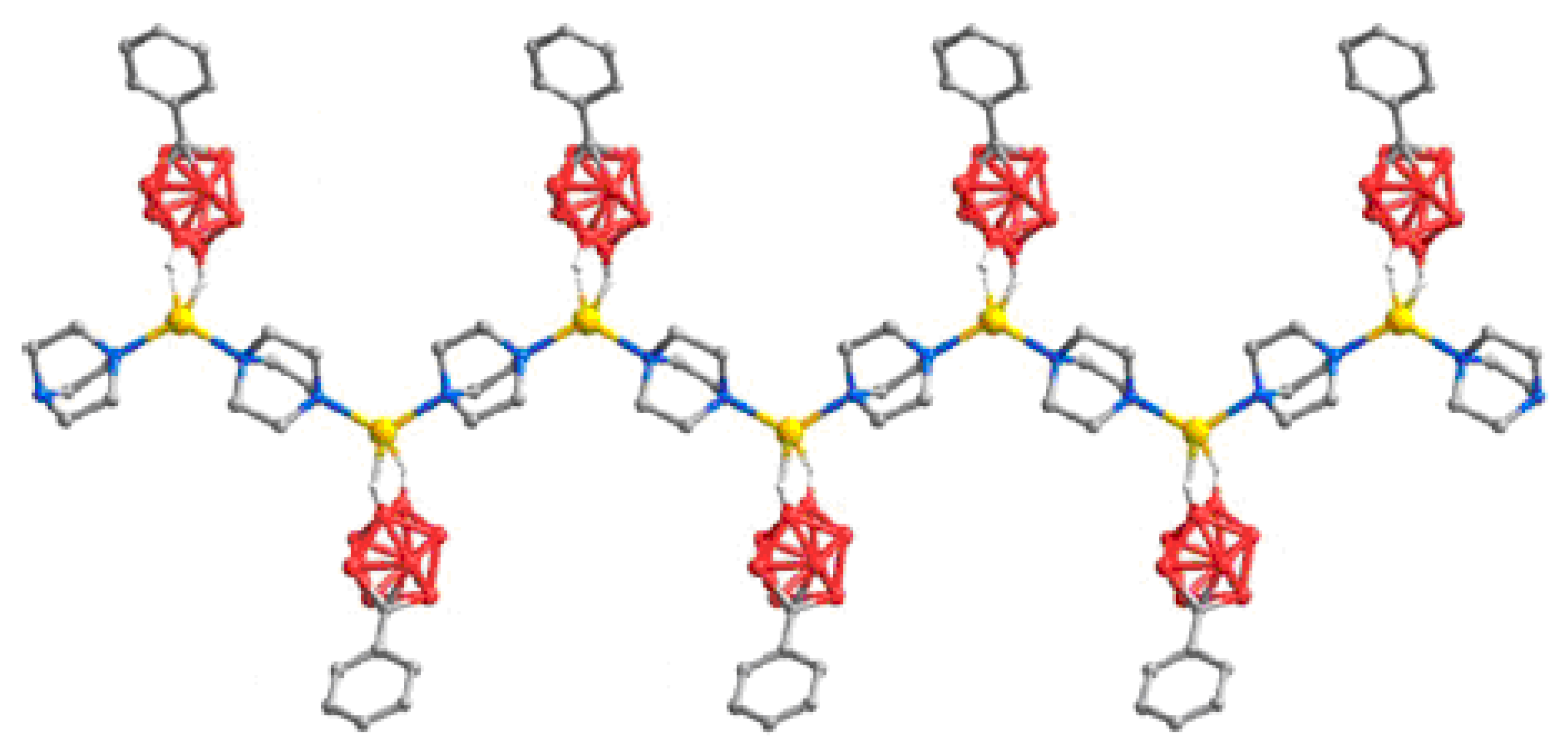
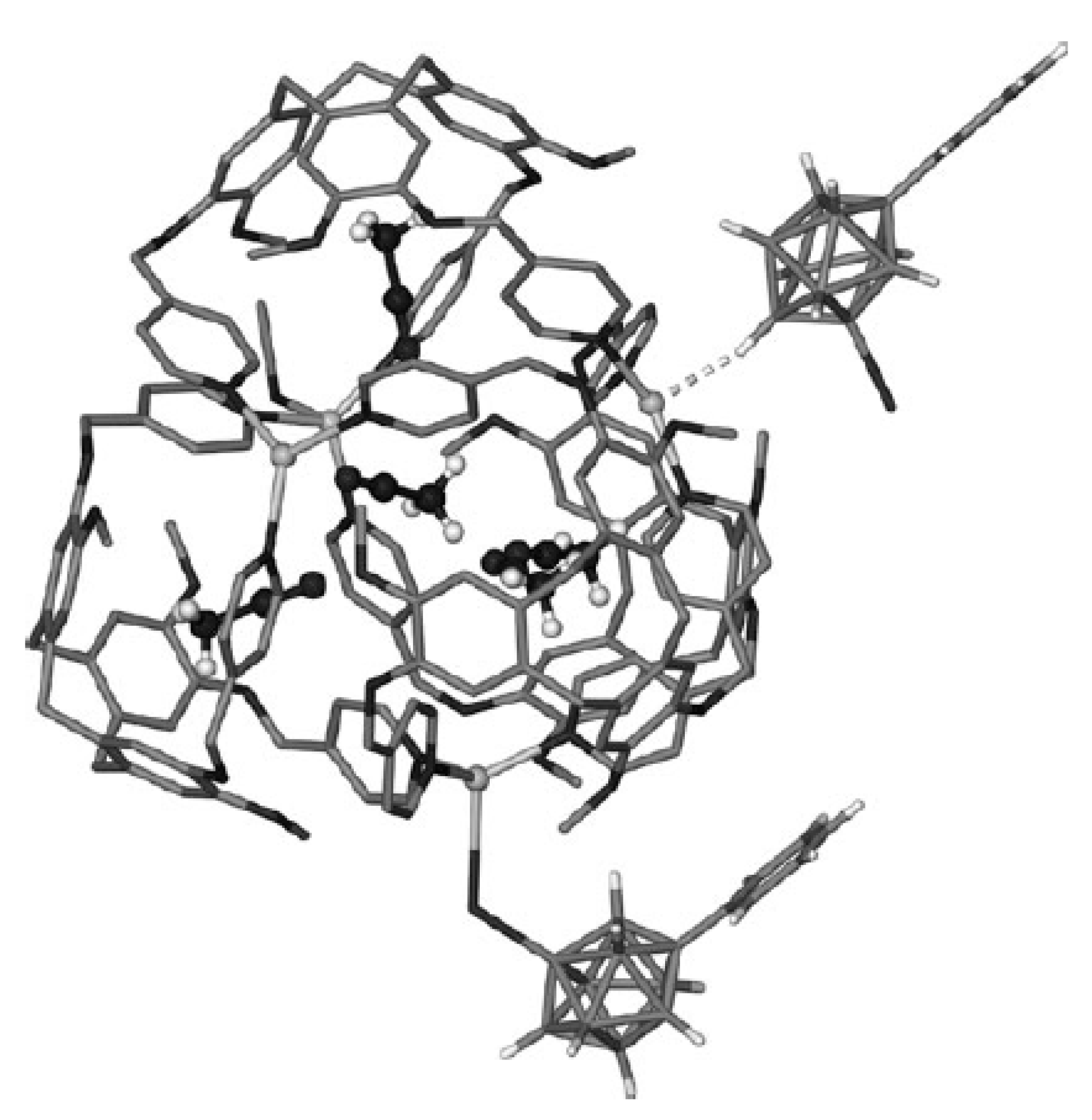
Star-burst tetrahedron complex {Ag
4(tris(isonic)CTG)
4(MeCN)[1-Ph-1-CB
9H
8-6-I]
2} [1-Ph-1-CB
9H
8-6-I]
2·12(MeCN) was obtained by the reaction of Ag[1-Ph-1-CB
9H
8-6-I] with tris(isonicotinoyl)cyclotriguaiacylene (tris(isonic)CTG) in acetonitrile [
76]. The two carborane anions coordinate to two external vertices of the tetrahedron, giving a {Ag
4L
4(MeCN)
2[1-Ph-1-CB
9H
8-6-I]
2}
2+ prism. Each of the four silver centers in the tetrahedron have a slightly different coordination environment. All are coordinated by three pyridyl arms from three different ligands. Two Ag(I) centers are irregular tetrahedral with terminal ligands external to the prism: either the MeCN ligand, or the [1-Ph-1-CB
9H
8-6-I]
− anion that coordinates through its iodine atom at Ag-I distance of 2.9305(9) Ǻ. The coordinated [1-Ph-1-CB
9H
8-6-I]
− anion is oriented such that its phenyl ring accepts an edge-to-face π-stacking interaction with a pyridyl ring of one of the ligands with a CH…π
centroid distance of 2.60 Ǻ. One of the other Ag(I) centers forms a weak Ag…H-B
eq interaction (2.332 Ǻ) to another [1-Ph-1-CB
9H
8-6-I]
− anion, again external to the prism. The final Ag(I) has an irregular trigonal-planar geometry but with an additional weak coordination to a guest MeCN molecule located inside the prism (
Figure 56) [
76].
5. Silver Complexes with the carba-closo-dodecaborate anion [CB11H12]−
The 1-carba-
closo-dodecaborate anion [CB
11H
12]
− was reported for the first time together with the [1-CB
9H
10]
− anion [
63,
64], however its chemistry received much more extensive development due to its better availability, especially after 2000, when a convenient method of its synthesis avoiding usage of highly toxic and expensive decaborane was proposed [
79]. A powerful impetus to the development of chemistry of the [CB
11H
12]
− anion was given by the possibility of its use as weakly coordinating anion, that resulted in the synthesis of huge amounts of its derivatives [
80,
81].
The silver salt of the parent carba-
closo-dodecaborate anion, Ag[CB
11H
12], was prepared by metathesis of the cesium salt with AgNO
3 in aqueous solution crystallized from benzene/hexane solution to give {Ag(η
1-benzene)[CB
11H
12]·C
6H
6}
n [
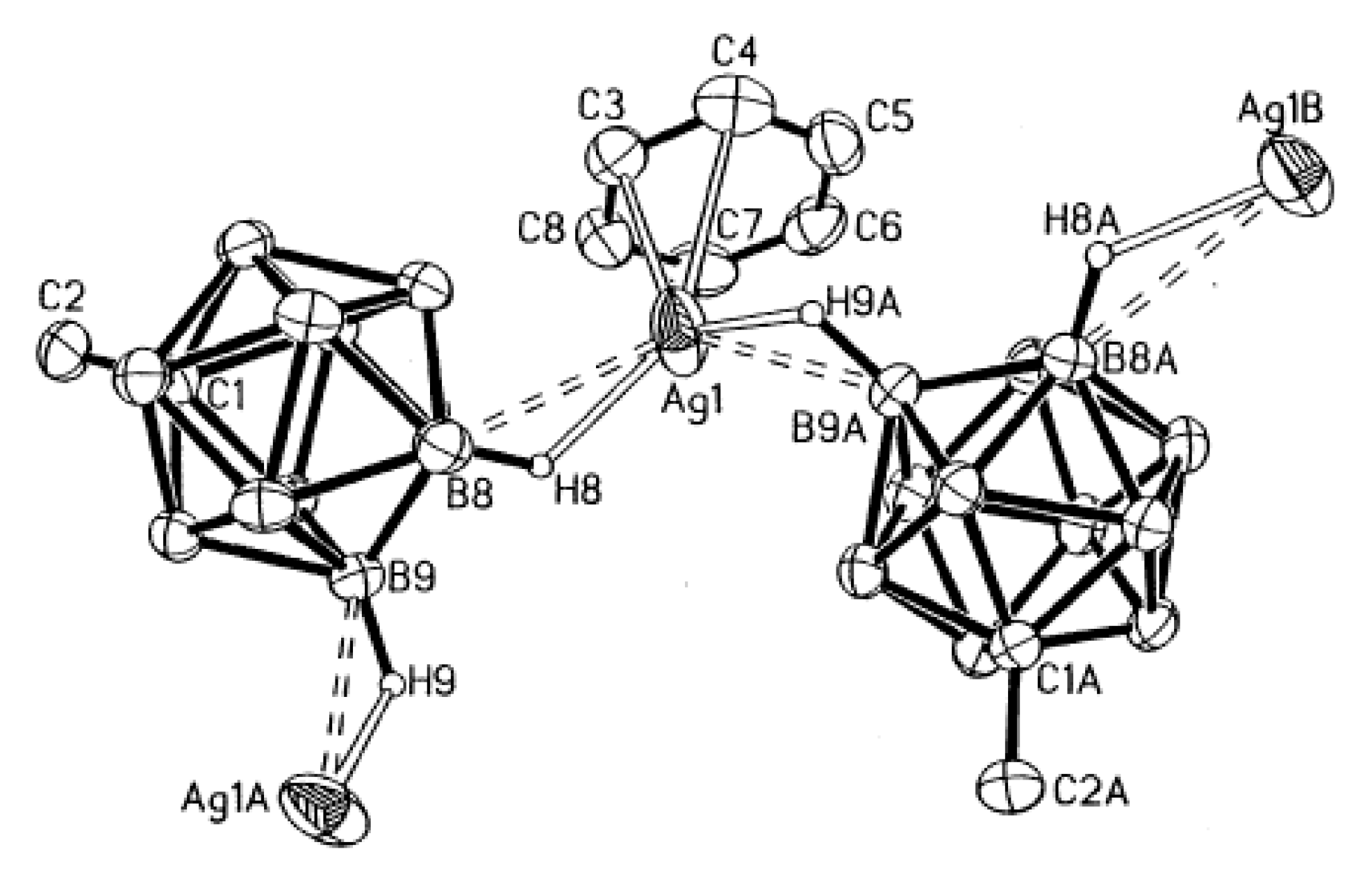
82]. The silver atom has a formal coordination number of three, bonding to two carborane anions and one benzene molecule. The interatomic distances Ag…H(B5′) and Ag…H(B9) are 1.968 and 1.974 Å, respectively (Ag…B(5′) and Ag…B(9) distances are 2.581(6) and 2.682(5) Å). The benzene molecule is coordinated to the silver atom in a η
1-fashion with short Ag-C distance of 2.400(7) Å (
Figure 57). The carborane anion acts as a bridging ligand to give an alternating cation-anion chain which is maintained in one dimension throughout the lattice. Noteworthy that, according to IR spectral data, the Ag…HB interactions preserve in the benzene solution (2380 cm
−1) [
82]. As it was revealed by
11B NMR spectroscopy, the same is true for the solution of Ag[CB
11H
12] in dichloromethane [
83].
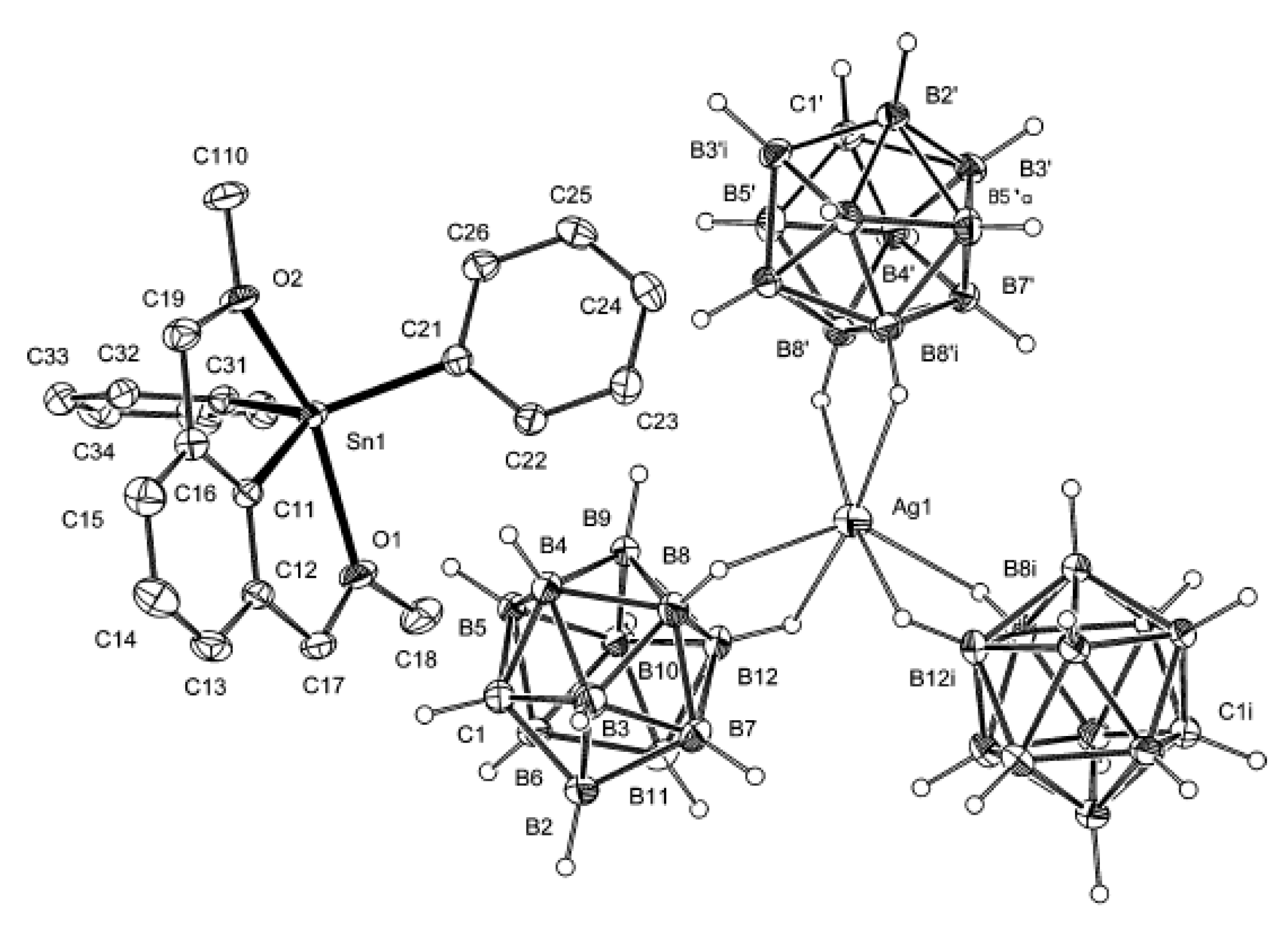
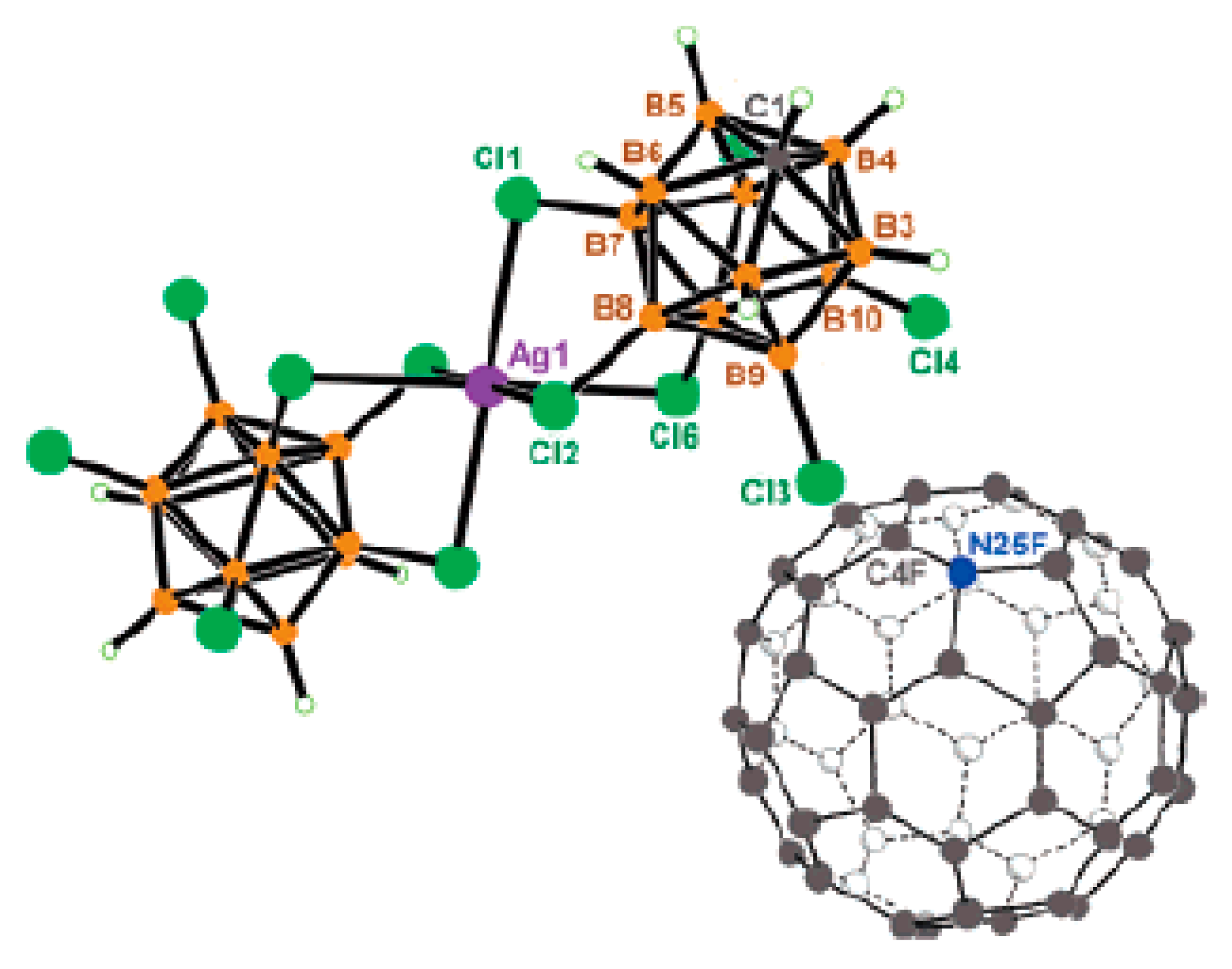
As it was mentioned above, one of the most common ways to obtain compounds with stabilized reactive cations are metathesis reactions of weak or sometimes even covalently bonded halide complexes with silver salts of weakly coordinating anions. In particular, triaryltin(IV) cation containing
O,C,O-coordinating pincer ligand [1-{2,6-(MeO)
2C
6H
3}SnPh
2]
+[CB
11H
12]
− was prepared by the treatment of the corresponding triaryltin chloride with the equimolar amount of Ag[CB
11H
12] in tetrahydrofuran. However, a change in the stoichiometry of the starting compounds (2:3) and solvent (CH
2Cl
2) resulted in the complex with new complex anion [1-{2,6-(MeO)
2C
6H
3}SnPh
2][Ag{CB
11H
12}
3]. The complex anion consists of the central Ag
+ ion, which is surrounded by three [CB
11H
12]
− anionic carborane ligands. Each of them is bonded to the Ag
+ center via bridging Ag…H(B12) (opposite to the cage carbon) and Ag…H(B8) (adjacent to B12) bonds, which results in a 6-fold coordination of Ag
+ by the B-H bonds. The coordination polyhedron around the silver atom approximates a distorted octahedral shape. The Ag…H distances are 2.2388–2.2583 Å (
Figure 58). The IR spectrum of the complex contains two bands at 2551 and 2347 cm
−1, which are diagnostic for ν(BH) and ν(AgHB) stretching, respectively [
84].
An even more complex anion was found in the complex [Ag(IMes)
2]
2[Ag
2{CB
11H
12}
4] formed by the treatment of 1,3-dimesitylimidazol-2-ylidene (IMes) with Ag[CB
11H
12] in toluene. The complex anion contains two silver atoms bridged by two [CB
11H
12]
− anions in addition to one terminally bound carborane on each metal. The coordination geometry around each silver atom is approximately trigonal prismatic, with each metal center involved in four shorter and two longer interactions with the carborane BH groups. The shorter Ag-H-B interactions vary in distance ranging from 1.96 to 2.23 Å (Ag…H) and 2.586(7) to 2.79(2) Å (Ag…B). The longer interactions are in the range 2.84(2)-2.94(2) Å (Ag…B) (
Figure 59) [
83].
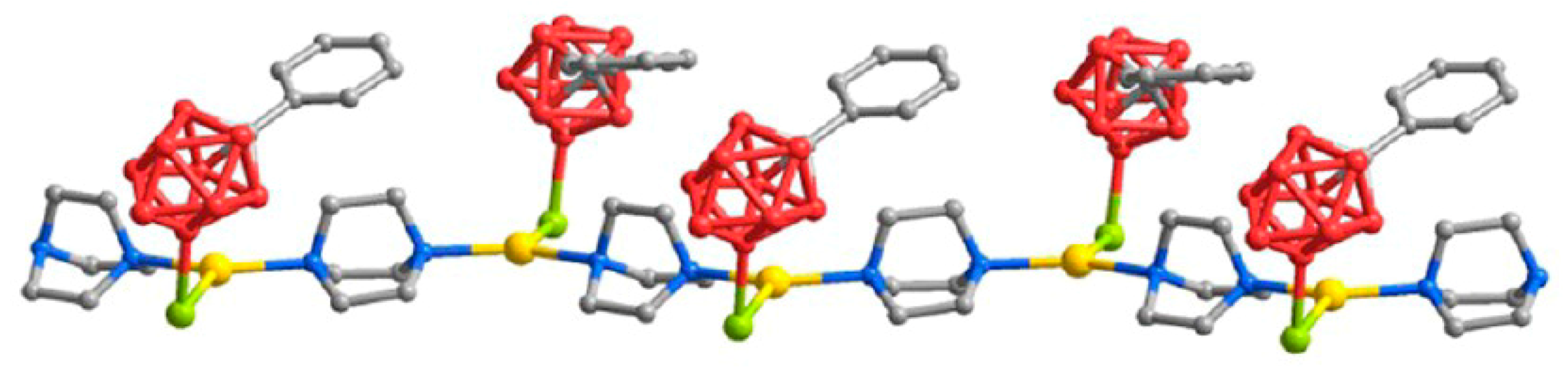
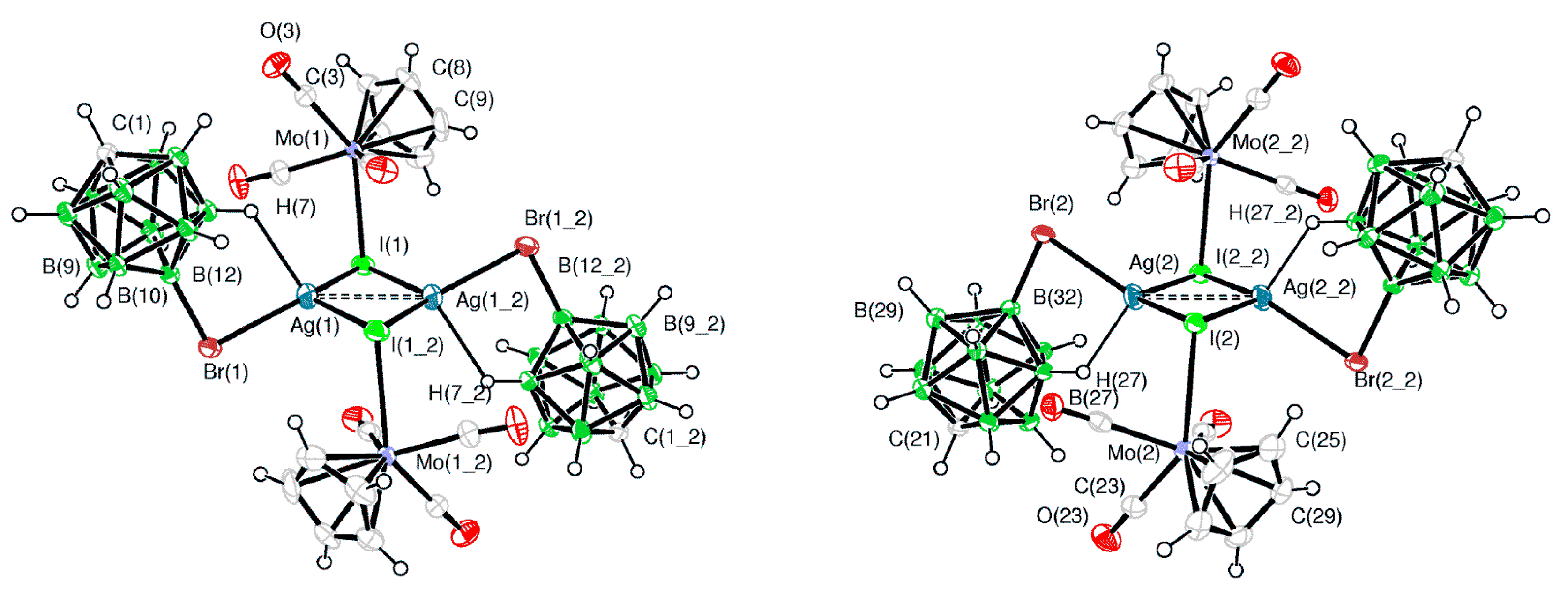
Similar to Ag[1-CB
9H
5-6,7,8,9,10-Br
5], the reaction of Ag[CB
11H
12] with iridium chloride complex IrCl(CO)(PPh
3)
2 in toluene results in the square pyramidal Ir→Ag donor-acceptor metal-metal bonded complex (PPh
3)
2(CO)ClIr·Ag[CB
11H
12] [
85,
86]. The [CB
11H
12]
− anion is coordinated to silver atom through the antipodal to the carbon atom BH group (the Ag…H(B) and Ag…B distances are 1.901 and 2.528 Å, respectively) (
Figure 60) [
85]. The metathesis proceeds only in donor solvents, such as acetone, giving cationic species with coordinated solvent [
86].
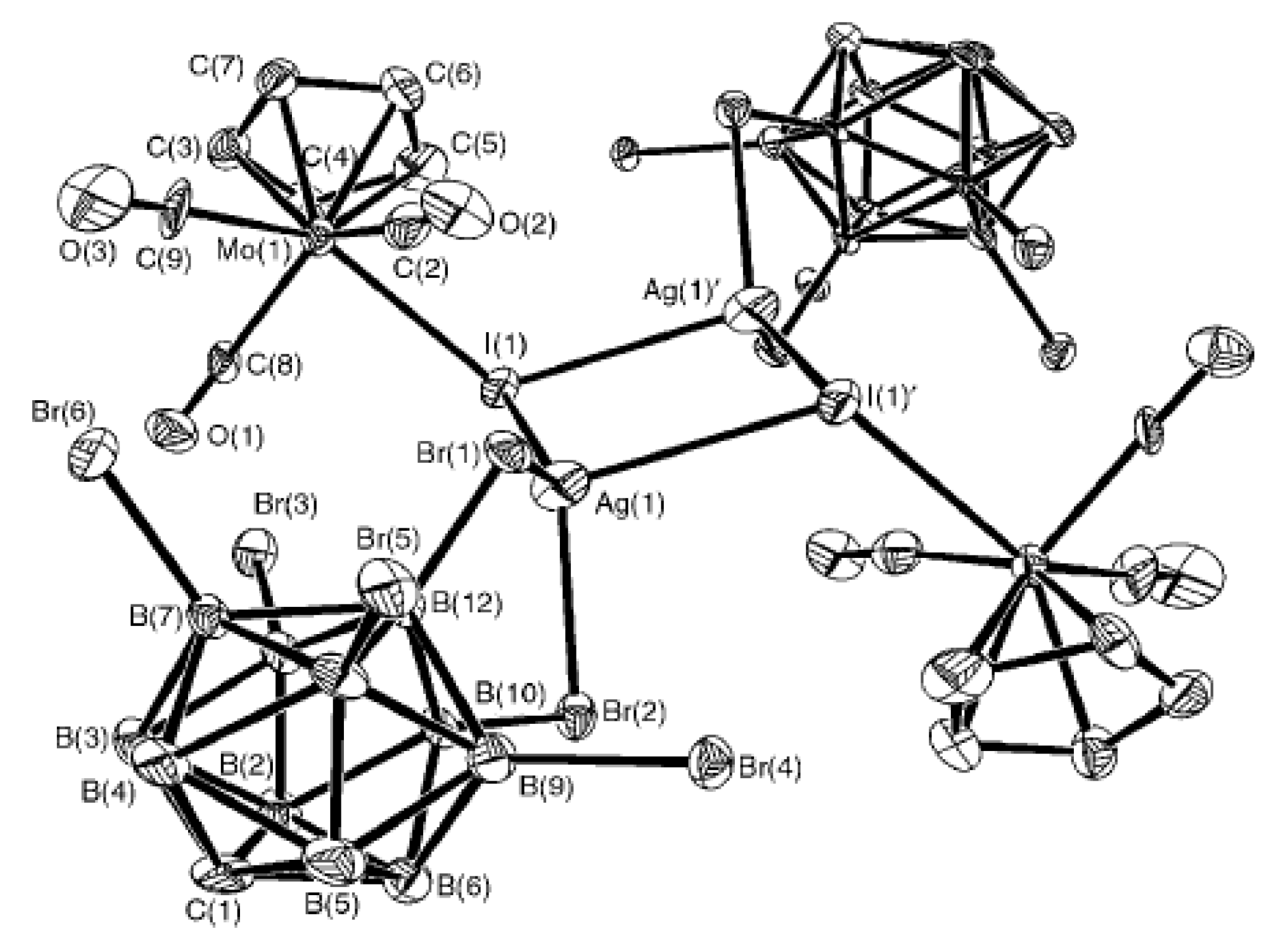
The long-lived (up to 1 week at room temperature) intermediate complexes (Cp)Fe(CO)
2X·Ag[CB
11H
12] with {(Cp)Fe(CO)
2[CB
11H
12]} as the ultimate product [
86] were obtained by reacting Ag[CB
11H
12] with iron halide complexes (Cp)Fe(CO)
2X (X = Cl, Br, I) in toluene [
86]. The similar reaction with (Cp)Mo(CO)
3I in dichloromethane also proceeds through formation of stable dimeric {[(Cp)Mo(CO)
3I]Ag[CB
11H
12]}
2 intermediate which was characterized by single crystal X-ray diffraction (
Figure 61) [
87,
88].
The silver salt of
C-alkyl substituted carba-
closo-dodecaborate anion Ag[1-R-1-CB
11H
11] (R = Me [
89], Et [
90]) were prepared as well. The crystal structure of {Ag(η
2-benzene)[1-Me-1-CB
11H
11]}
n resembles structure of the corresponding salt of the parent anion with the silver atom bonding to two carborane anions and one benzene molecule. The interatomic Ag…B(8) and Ag…B(9A) distances are 2.613(1) and 2.613(1) Å, respectively. The benzene molecule is coordinated to the silver atom in asymmetric η
2-fashion with Ag-C distances of 2.460(1) and 2.698(1) Å (
Figure 62) [
89].
In the crystal structure of {Ag[1-Et-1-CB
11H
11]}
n the silver atom is coordinated to three monocarborane anions. The coordination occurs through BH groups, with the first anion coordinating via BH group in position 7 (Ag(1)…H(B7) is 2.078 Å), the second anion coordinating via BH group in position 10 (Ag(1)…H(B10) is 1.943 Å), and with the third anion coordinating via two adjacent BH groups in positions 8 and 9 (Ag(1)…H(B8) is 2.352 Å and Ag(1)…H(B9) is 2.301 Å). These results in slightly distorted tetrahedral geometry around the Ag
+ cation. The extended solid state structure is characterized by layers that pack parallel to the
bc plane of the unit cell (
Figure 63) [
90].
The silver salt of 12-fluoro-1-carba-
closo-dodecaborate anion, Ag[1-CB
11H
11-12-F], was prepared by cation exchange from the corresponding cesium salt. The crystals of the silver salt of 12-fluoro-1-carba-
closo-dodecaborate anion {Ag(η
2-benzene)
2[1-CB
11H
11-12-F]}
n were obtained by crystallization from benzene. The silver atom has a formal coordination number of four, bonding to two carborane anions and two benzene molecules. Both [1-CB
11H
11-12-F]
− anions are coordinated to the silver atom via BH groups (Ag…H(B) distances of 2.18 and 2.28 Å), the benzene molecules are coordinated to the silver atom in a η
2-fashion (
Figure 64) [
91]. Significantly, there is no interaction between the silver ion and the fluorine atom.
The silver salt of 12-bromo-1-carba-
closo-dodecaborate anion, Ag[1-CB
11H
11-12-Br], was prepared using the standard metathesis approach [
92]. The crystals {Ag(η
1-benzene) [1-CB
11H
11-12-Br]}
n were obtained by crystallization from benzene. The silver atom has a formal coordination number of four, bonding to tree carborane anions and one benzene molecule. Two [1-CB
11H
11-12-Br]
− anions are coordinated to the silver atom via BH groups (Ag…H(B) distances of 2.20 and 2.27 Å) and the third anion is coordinated by the bromine atom (Ag…Br(B) distance of 2.642 Å), the benzene molecule is coordinated to the silver atom in a η
1-fashion with Ag-C distance of 2.485 Å (
Figure 65) [
92].
The reaction with (Cp)Mo(CO)
3I with Ag[CB
11H
11-12-Br] in dichloromethane produces dimeric complex {[(Cp)Mo(CO)
3I]Ag[CB
11H
11-12-Br]}
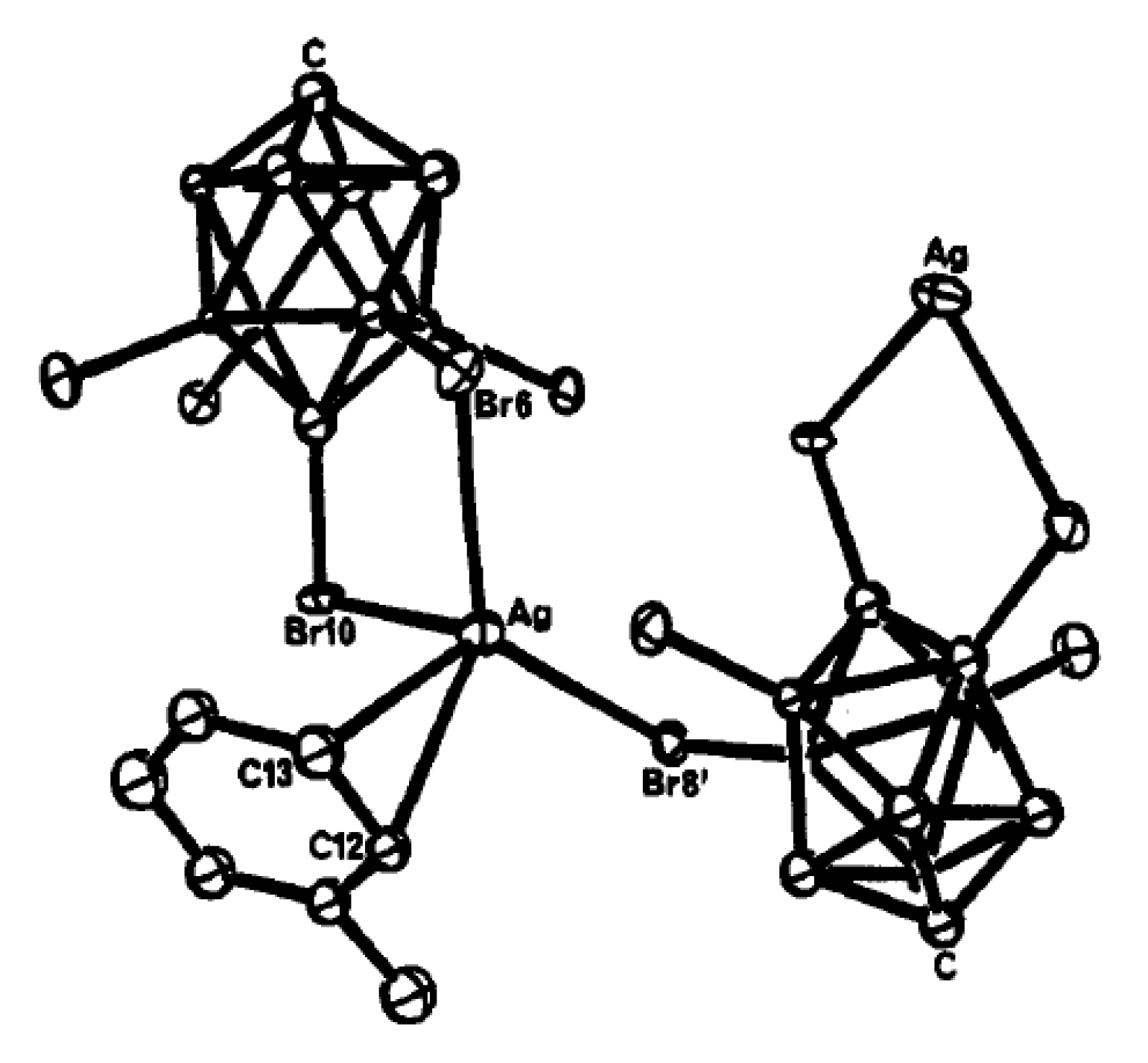
2 which was characterized by single crystal X-ray diffraction. Its structure contains a centrosymmetric planar {Ag
2I
2} core appended with two {CpMo(CO)
3I} fragments and two bidentate carborane anions. Each carborane anion is coordinated to a silver atom through one Ag-Br (2.6456(8) Å) and one Ag-HB (Ag…B 3.159(3) Å, Ag…H 2.43(2) Å) bonds. The two metal fragments are orientated
trans with respect to one another and the central {Ag
2I
2} core (
Figure 66) [
93].
The silver salts of 6,7,8,9,10,11,12-hexahalogeno-1-carba-
closo-dodecaborate anions, Ag[1-CB
11H
6-6,7,8,9,10,11,12-X
6] (X = Cl [
94], Br [
86], I [
94]), were prepared from the treatment of the corresponding cesium salts with AgNO
3 in aqueous solution. The crystals of {Ag(η
2-
para-xylene)[1-CB
11H
6-6,7,8,9,10,11,12-Cl
6]}
n were obtained by crystallization from 1,4-xylene. The silver atom is in the five-coordinated propeller arrangement built of one η
2-
p-xylene and two bidentate bridging carborane anions. The Ag…Cl distances range from 2.640(1) to 2.926(1) Å. The
p-xylene is coordinated in asymmetric η
2 fashion with Ag-C bonds of 2.506(3) and 2.481(4) Å (
Figure 67) [
94].
The crystals of {Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6]}
n were obtained by crystallization from toluene/hexane in the presence of
i-Pr
2SiHCl. The silver atom is in distorted octahedral arrangement with two tridentate bridging carborane anions. The bromine atom attached to B(12) is shared by two silver ions. The average Ag…Br distance is 2.862(2) Å (
Figure 67) [
95]. In the crystals of {Ag[1-CB
11H
6-6,7,8,9,10,11,12-I
6]·0.5(benzene)}
n the silver atom is complexed by iodine atoms to one anion in bidentate fashion and to another in tridentate fashion adopting a square-pyramidal arrangement (
Figure 67) [
95].
The discrete {Ag[1-CB
11H
6-6,7,8,9,10,11,12-Cl
6]
2}
− anions with six-coordinated silver atom is in adistorted octahedral arrangement (
Figure 68) were found in the X-ray structure of the azafullerene carbocation salt [C
59N]{Ag[1-CB
11H
6-6,7,8,9,10,11,12-Cl
6]
2}·3(
o-dichlorobenzene) which was prepared by reaction of (C
59N)
2 dimer with [HBPC·]{Ag[1-CB
11H
6-6,7,8,9,10,11,12-Cl
6]
2} (HBPC = hexabromo(phenyl)carbazole) in
o-dichlorobenzene [
96].
The similar discrete {Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6]
2}
− anions (
Figure 69) were found in the X-ray structure of the [Fe(TPP)]{Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6]
2}·4(
p-xylene) complex which was prepared by the treatment of [Fe(TPP)Br] with Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6] in
p-xylene at 100 °C, subsequent removal of the resulting AgBr precipitate, and vapor diffusion of
n-hexane [
97].
Similar to Ag[CB
11H
12], the reaction of Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6] with iridium chloride complex IrCl(CO)(PPh
3)
2 in fluorobenzene results in the donor-acceptor metal-metal bonded complex (PPh
3)
2(CO)ClIr·Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6] identified by IR spectroscopy [
86].
The similar reaction with (Cp)Mo(CO)
3I in dichloromethane also proceeds through formation of stable dimeric {[(Cp)Mo(CO)
3I]Ag[1-CB
11H
6-6,7,8,9,10,11,12-Br
6]}
2 intermediate which was characterized by single crystal X-ray diffraction. Its structure bears close similarities with that found in {[(Cp)Mo(CO)
3I]Ag[1-CB
11H
12]}
2, with a centrosymmetric planar {Ag
2I
2} core appended with two {CpMo(CO)
3I} fragments and two bidentate carborane anions. The two metal fragments are orientated
trans with respect to one another and the central {Ag
2I
2} core (
Figure 70) [
88].
The silver salts of
C-methyl hexahalogen carba-
closo-dodecaborate anions Ag[1-Me-1-CB
11H
5-6,7,8,9,10,11,12-X
6] (X = Cl, Br, I) were prepared using standard metathesis methodology. The crystal structure of {Ag(η
1-mesitylene)[1-Me-1-CB
11H
5-6,7,8,9,10,11,12-Cl
6]}
n resembles structure of {Ag(η
2-
p-xylene)[1-CB
11H
6-6,7,8,9,10,11,12-Cl
6]}
n with five-coordinated silver atom is in a distorted square pyramidal arrangement of one η
1-mesitylene and two bidentate bridging [1-Me-CB
11H
5-6,7,8,9,10,11,12-Cl
6]
− anions. The Ag…Cl distances range from 2.683(2) to 2.874(2) Å (
Figure 71) [
89]. The solid state structure of {Ag[1-Me-1-CB
11H
5-6,7,8,9,10,11,12-Br
6]}
n is almost identical to that of {Ag(1-CB
11H
6-6,7,8,9,10,11,12-Br
6)}
n with the average Ag…Br distance of 2.858(1) Å (
Figure 71) [
89]. In the solid state structure of {Ag[1-Me-1-CB
11H
5-6,7,8,9,10,11,12-I
6]}
n the silver atom is complexed by four iodine atoms from two bidentate bridging [1-Me-1-CB
11H
5-6,7,8,9,10,11,12-I
6]
− anions in a distorted tetrahedral arrangement with the Ag…I distances varying from 2.798(2) to 2.872(2) Å (
Figure 71) [
89].
The silver salts of the perchloro and perbromo derivatives of 1-carba-
closo-dodecaborate anion, Ag[1-HCB
11Cl
11] and Ag[1-HCB
11Br
11], were prepared by treatment of the corresponding sodium salt with AgNO
3 in aqueous solution [
98]. Single crystals of {(η
2-
p-xylene)
2Ag[1-HCB
11Cl
11]}·1/2(
p-xylene) were grown from
p-xylene. The complex has a monomeric structure with four-coordinated silver atom in a propeller arrangement of two η
2-
p-xylene molecules and a single bidentate [1-HCB
11Cl
11]
− anion (
Figure 72a) [
99]. Single crystals of {(η
1-mesitylene)(MeCN)Ag[1-HCB
11Br
11]} were grown from a mesitylene-acetonitrile solution. The silver atom has a distorted tetrahedral arrangement consisting of one η
1-bound mesitylene molecule, one acetonitrile molecules and two bromine atoms of the bidentate [1-HCB
11Br
11]
− anion (Ag…Br bonds are 2.772(1) and 2.776(1) Å) (
Figure 72b) [
99].
Silver salts of mixed halocarborane anions Ag[1-HCB
11-2,3,4,5,6-Br
5-7,8,9,10,11,12-Cl
6], Ag[1-HCB
11-2,3,4,5,6-I
5-7,8,9,10,11,12-Cl
6], Ag[1-HCB
11-2,3,4,5,6-Cl
5-7,8,9,10,11,12-Br
6], Ag[1-HCB
11-2,3,4,5,6-I
5-7,8,9,10,11,12-Br
6] and Ag[1-HCB
11-2,3,4,5,6-Br
5-7,8,9,10,11,12-I
6] were prepared by treatment of the corresponding sodium salts with AgNO
3 in aqueous solution. Single crystals of {(η
2-mesitylene)
2Ag[1-HCB
11-2,3,4,5,6-Br
5-7,8,9,10,11,12-Cl
6]}·mesitylene and ({(η
1-mesitylene)Ag[1-HCB
11-2,3,4,5,6-Cl
5-7,8,9,10,11,12-Br
6]}·mesitylene)
n were grown from mesitylene and mesitylene/acetone solutions, respectively. Similar to structure of {(η
2-
p-xylene)
2Ag[1-HCB
11Cl
11]}·1/2(
p-xylene), {(η
2-mesitylene)
2Ag[1-HCB
11-2,3,4,5,6-Br
5-7,8,9,10,11,12-Cl
6]}·mesitylene has monomeric structure with four-coordinated silver atom in a propeller arrangement of two η
2-mesitylene molecules and a single bidentate [1-HCB
11Br
5Cl
6]
− anion which is coordinated through its Cl(8) and Cl(12) atoms (the Ag…Cl(8) and Ag…Cl(12) distances are 2.986(2) and 2.889(2) Å, respectively) (
Figure 73) [
99]. It is noteworthy that no interaction between Ag
+ and any one of the bromine atoms from the upper belt of the cage is observed, although Ag
+ usually prefers to bind to a bromine atom rather than a chlorine atom according to the hard-soft acid-base principle.
In contrast, a polymeric structure is observed in ({(η
1-mesitylene)Ag[1-HCB
11-2,3,4,5,6-Cl
5-7,8,9,10,11,12-Br
6]}·mesitylene)
n. The four-coordinated silver atom is in a distorted tetrahedral arrangement of one η
1-mesitylene molecule, two bromine atoms from a bridging [1-HCB
11Cl
5Br
6]
− anion and one bromine atom from another bridging [1-HCB
11Cl
5Br
6]
− anion (the Ag…Br distances of 2.750(2)–2.873(1) Å) (
Figure 74) [
99]. There are no interactions between Ag
+ and any one of the five chlorine atoms from the upper belt of the carborane cage, which is understandable because Br
− is a softer base than Cl
− and the lower belt atoms bear more negative charge than the upper belt ones. These results may suggest that the charge distribution on the icosahedral cage plays a predominant role in the coordination chemistry of carborane anions.
The silver salts of
C-methyl perhalogen derivatives of the carba-
closo-dodecaborate anion Ag[1-Me-1-CB
11X
11] (X = Cl, Br) were prepared by treatment of the sodium salts with AgNO
3 in aqueous solution [
98]. Single crystals of {Ag[1-Me-1-CB
11Cl
11]}
n were grown from benzene solution. As expected, the complex has polymeric structure. Each silver atom coordinates to six chlorine atoms from the three [1-Me-CB
11Cl
11]
− anions in a distorted-octahedral arrangement at an average Ag…Cl distance of 2.882(1) Å, and each anion is surrounded by six silver atoms in a regular octahedral arrangement to form a three-dimensional coordination polymer (
Figure 75) [
99].
Single crystals of {Ag[1-Me-1-CB
11Br
11]}
n were grown from a chlorobenzene solution. Like many other silver(I) salts of icosahedral carborane anions, it is a one-dimensional coordination polymer with the carborane anions acting as bridging ligands. The silver atom has six-coordinate geometry which is very similar to that found in {Ag[1-HCB
11H
5-7,8,9,10,11,12-Br
6]}
n with the metal atom bonding to three bromine atoms from each of two bridging [1-Me-CB
11Br
11]
− anions (the average Ag…Br distance of 2.848(5) Å) (
Figure 76) [
98]. The methyl group is completely mixed with eleven bromine atoms of the cage anion. Therefore, it is not possible to distinguish the BBr vertex antipodal to carbon from other vertexes. Unlike in the solid state structure of {Ag[1-HCB
11H
5-7,8,9,10,11,12-Br
6]}
n, where the most basic bromine atom attached to B(12) antipodal to carbon is shared by two silver ions, there is no bromine atom shared by the silver ion in {Ag[1-Me-1-CB
11Br
11]}
n, perhaps reflecting the steric effect and/or the fact that there is no significantly electron-rich bromine atom on the cage due to donor effect of the methyl group
Fractional crystallization of Ag[1-Me-1-CB
11H
11-nBr
n] (
n = 8–10) from a hot
p-xylene solution containing a very small amount of acetonitrile gave X-ray-quality crystals of {(
p-xylene)Ag[1-Me-CB
11HBr
10]}
n. The complex has the five-coordinated silver atom in a propeller arrangement of one η
2-
p-xylene and two bidentate bridging [1-Me-CB
11HBr
10]
− anions, which is similar to the solid-state structure of {(η
2-
p-xylene)Ag(1-HCB
11H
6-7,8,9,10,11,12-Cl
6]}
n. The Ag…Br distances range from 2.818(2) to 2.972(2) Å. The
p-xylene is coordinated in asymmetric η
2 fashion with Ag-C bonds of 2.514(5) and 2.485(5) Å (
Figure 77) [
98].
The silver salt of the
C-butyl perchloro-carba-
closo-dodecaborate anion Ag[1-Bu-1-CB
11Cl
11] was prepared by treatment of the corresponding sodium salt with AgNO
3 in aqueous solution. Single crystals of {Ag(η
2-C
6H
5F)(H
2O)[1-Bu-1-CB
11Cl
11]}
n were grown from fluorobenzene solution. The silver atom is coordinated by η
2-fluorobenzene (the Ag-C bonds of 2.553(3) and 2.587(3) Å), water (the Ag-O bond of 2.317(2) Å), one bidentate (the Ag…Cl distances are 2.847(1) and 2.878(1) Å) and one monodentate (the Ag…Cl distance is 3.127(1) Å) bridging [1-Bu-1-CB
11Cl
11]
− anions (
Figure 78) [
100].
The silver salt of the
C-methyl perfluoro-carba-
closo-dodecaborate anion Ag(C
6H
6)[1-Me-1-CB
11F
11] was prepared by metathesis reaction of the corresponding cesium salt and AgNO
3 in refluxing benzene [
101]. Single crystals of {Ag(CHPh
3)[1-Me-1-CB
11F
11]}
n were obtained from the reaction of Ag(C
6H
6)[1-Me-1-CB
11F
11] with Ph
3CCl in dichloromethane. The structure consists of layers of {Ag(CHPh
3)
+}
n polycations with [1-Me-1-CB
11F
11]
− anions filling half of the trigonal prismatic holes defined by six Ag
+ ions from successive {Ag(CHPh
3)
+}
n layers. The {Ag(CHPh
3)
+}
n triangular-net layers contain the first example of a rigorously trigonal planar Ag(arene)
3+ complex. In the {Ag(CHPh
3)
+}
n layer three phenyl groups from three different CHPh
3 ligands in asymmetric η
2-fashion coordinate to the Ag
+ ion being in trigonal planar arrangement. There is no bonding between the Ag
+ cations and the [1-Me-1-CB
11F
11]
− anions (
Figure 79) [
101].
The silver salt {Ag[1-Me-1-CB
11F
10-12-SiPh
3]·0.5(benzene)}
n was obtained by metathesis of the corresponding cesium salt and AgBF
4 followed by crystallization from benzene solution. The structure contains two types of silver cations in different arrangement. The coordination sphere of one of Ag
+ cations is trigonal planar and consists of two η
2-coordinated phenyl rings of two different [1-Me-1-CB
11F
10-12-SiPh
3]
− anions and one η
1-coordinated benzene molecule. The other Ag
+ cation is coordinated in η
1-fashion by four phenyl groups of two different [1-Me-1-CB
11F
10-12-SiPh
3]
− anions forming the tetrahedral coordination sphere (
Figure 80) [
100]. Again, there are no Ag…F interactions between the Ag
+ cations and the [1-Me-1-CB
11F
10-12-SiPh
3]
− anions.
Copper(I) complexes (CO)
2Cu[1-Bn-1-CB
11F
11] and [Cu(CO)
4][1-Et-1-CB
11F
11] were prepared by treating the corresponding silver salts with CuCl in dichloromethane under an CO atmosphere. In the first complex the copper(I) dicarbonyl moiety is coordinated in asymmetrical η
2-fashion by the phenyl ring of the [1-Bn-1-CB
11F
11]
− anion (
Figure 81a), whereas in the second complex there is no interactions between the [Cu(CO)4]
+ cation and the [1-Et-1-CB
11F
11]
− anion (
Figure 81b) [
103]. Again, there are no Ag…F interactions between the Cu
+ cations and the fluorinated carborane anions that can be explained taking in the account the hard-soft acid-base principle.
The silver salt of the permethylated carba-
closo-dodecaborate anion Ag[HCB
11Me
11] was prepared by metathesis reaction of the corresponding cesium salt and AgNO
3 in water-Et
2O. Single crystals of {Ag[HCB
11Me
11]}
n were obtained by crystallization from dichloromethane/hexanes. One tridentate and three monodentate carborane anions affords a distorted octahedral coordination environment around the silver cation. Each carborane anion is closely associated with four silver cations. This results in a three-dimensional coordination polymer (
Figure 82). The Ag…C distances span the range 2.652(2)–3.089(2) Å [
104].
In contrast to the silver salts of the parent carba-
closo-dodecaborate anion and its halogenderivatives, the reaction of Ag[HCB
11Me
11] with (Cp)Mo(CO)
3I in dichloromethane results in complex {([(Cp)Mo(CO)
3I]
3Ag)
2}[HCB
11HMe
11]
2, where each silver atom in the central {Ag
2I
2} core is surrounded by three {(Cp)Mo(CO)
3I} units, one of which bridges the two silver centres (
Figure 83) [
93].
Single crystals of {(MeCN)
2Ag[CB
11H
12]}
n were grown by crystallization of Ag[CB
11H
12] from acetonitrile/trifluoroethanol solution. The silver atom is linearly coordinated by two crystallographically equivalent acetonitrile ligands at the Ag-N distance 2.172(5) Å and N-Ag-N angle of 157.6(2)°. There are four additional interactions to BH groups of three surrounding [CB
11H
12]
− anions at Ag…H-B distances 2.421 and 2.405 Å forming a distorted octahedral coordination environment (
Figure 84a). Each [CB
11H
12]
− anion forms Ag…H-B interactions with three silver centers, and every silver atom interacts with three [CB
11H
12]
− anions forming a 2D network. These networks pack in the crystal lattice through interdigitation of the terminal acetonitrile ligands (
Figure 84b) [
105].
Crystals of {Ag(NCCH
2CN)
2[CB
11H
12]}
2 were grown by crystallization of Ag[CB
11H
12] from a malonitrile/ethanol solution. The complex is a discrete dimeric complex. There are two types of the malonitrile ligand. One type of the ligand bridges between two Ag
+ cations to form a cyclic dimer. The other coordinates in a terminal fashion with a pendant nitrile group that is not involved in coordination with adjacent dimers. The silver atom has a distorted tetrahedral geometry with coordination by three malonitrile nitrogen atoms and one Ag…H-B interaction (2.038 Å) (
Figure 85) [
105].
Single crystals of {Ag
4(NC(CH
2)
2CN)
5[CB
11H
12]
4}
n were grown by crystallization of Ag[CB
11H
12] from succinonitrile/ethanol solution. The silver cations fall into two groups, either coordinated by four succinonitrile ligands (Ag2, Ag3, Ag5) or by one succinonitrile ligand and two [CB
11H
12]
− anions (Ag1, Ag4, Ag6). The first type of silver cations propagates a coordination chain. All have distorted tetrahedral geometry with Ag-N distances ranging from 2.194 to 2.350(9) Å and N-Ag-N angles ranging from 97.8 to 121.3(3)°. Silver cations of the other type are not involved in propagating the chain structure but are effectively terminal branches from the chain. Each is connected to the chain via a single bridging succinonitrile, and the other coordination sites are occupied by Ag-H-B interactions. For Ag1, the coordination environment is distorted tetrahedral with three Ag…HB bonds of 1.971, 2.062, and 2.139 Å. The coordination environment around Ag4 is very similar to that of Ag1, while for Ag6 the environment shows two short Ag…HB interactions and two long Ag…HB interactions at around 2.5 Å (
Figure 86) [
105].
Complex {Ag(pyz)[CB
11H
12]}
n was prepared by the reaction of Ag[CB
11H
12] with pyrazine in acetonitrile [
106]. The silver cation is coordinated to two crystallographically equivalent pyrazine ligands at Ag-N distance of 2.203(3) Å in an almost linear fashion (N-Ag-N angle 174.76(14)°). There are also four Ag-H-B interactions between the silver cation and four [CB
11H
12]
− anions with Ag…HB distances in the range 2.287–2.460 Å. Each pyrazine molecule bridges between two silver cations to form an infinite planar chain, which runs along the b-axis. Each carborane anion interacts with four silver cations. On the whole, the coordination network has 3D pillared checkerboard arrangement (
Figure 87).
Complex {[Ag(4,4′-Bipy)][1-Ph-1-CB
11H
5-7,8,9,10,11,12-I
6]·MeCN}
n was prepared by reaction of Ag[1-Ph-1-CB
11H
5-7,8,9,10,11,12-I
6] with 4,4′-bipyridine in acetonitrile [
75]. Structure of the complex is similar to those found in complex {Ag(4,4′-Bipy)[1-Ph-1-CB
9H
4-6,7,8,9,10-I
5]·MeCN}
n. Each silver atom is coordinated through two Ag-N bonds of two crystallographically distinct 4,4′-Bipy ligands at distances of Ag(1)-N(1) 2.274(6) and Ag(1)-N(3) 2.242(7) Å and by a chelating interaction with the [1-Ph-1-CB
11H
5I
6]
− anion resulting from two Ag-I coordination bonds. Each 4,4′-Bipy ligand bridges between two silver cations giving a zigzag coordination chain where the coordinated [1-Ph-1-CB
11H
5I
6]
− anion points in two alternating directions above or below the chain (
Figure 88) [
75].
Complexes {[Ag
2(bppz)
3][CB
11H
12]
2·MeCN}
n [
106] and {[Ag
2(bppz)
3][1-(4-BrC
6H
4)-1-CB
11H
11]
2·1.5(MeCN)}
n [
75] were prepared by the corresponding silver salts with 2,3-bis(2-pyridyl)pyrazine in acetonitrile. Both complexes contain virtually identical {[Ag
2(bppz)
3]
2+}
n coordination chains. Each silver cation has a distorted tetrahedral geometry and is coordinated by N-atoms belonging at three different bppz molecules with one chelating interaction per metal atom. Two types of ligands with different binding motifs can be distinguished. One bridges between two silver cations with a chelating interaction to one metal and a single bond through a pyridine nitrogen atom to the other. There are two such ligands, forming a double bridge between two adjacent silver cations. The other type of bppz ligand forms a single bridge between two silver cations and coordinates only through its pyridine nitrogen atoms (
Figure 89).
Complex {Ag(DABCO)[1-Ph-1-CB
11H
5-7,8,9,10,11,12-I
6]}
n was prepared by reaction of Ag[1-Ph-1-CB
11H
5-7,8,9,10,11,12-I
6] with 1,4-diazabicyclo[2.2.2]octane in acetonitrile [
77]. The silver atom has a slightly distorted tetrahedral geometry with two coordinate bonds to two crystallographic equivalent DABCO ligands with an angle N-Ag-N of 111.40(14)° and Ag-N bonds of 2.346(4) and 2.321(4) Å. Furthermore, it interacts with two iodine atoms of the [1-Ph-1-CB
11H
5I
6]
− anion with an angle I-Ag-I of 95.41(3)º and Ag…I distances of 2.8276(9) and 2.817(1) Å. The {Ag(DABCO)[1-Ph-1-CB
11H
5-7,8,9,10,11,12-I
6]}
n coordination chains are comparable to those observed in {Ag(DABCO)[1-Ph-1-CB
9H
9]}
n (
Figure 90) [
77].
Complex {(Py)
2Ag[1-HCB
11Br
5I
6]·Py} was obtained by recrystallization of Ag[1-HCB
11Br
5I
6] from pyridine/acetonitrile. The four-coordinated silver atom is in a distorted-tetrahedral arrangement of two iodine atoms from the [1-HCB
11Br
5I
6]
− anion (Ag-I bonds 2.894(1) and 2.835(1) Å) and two pyridine molecules (Ag-N bonds 2.258(11) and 2.267(4) Å). It is interesting to note that the third pyridine molecule points toward the C-H cage proton with the N…C distance of 3.061(15) Å and C-H-N angle of 172(1)°, indicating there is a hydrogen bonding between pyridine molecule and the acidic C-H cage proton (
Figure 91) [
107].
Capsule-like complex {[Ag
2(tris(pyamino)CTG)
2(MeCN)
2][CB
11H
12]
2} was obtained by co-crystallization of Ag[CB
11H
12] with tris(3-pyridylmethylamino)cyclotriguaiacylene cavitand (tris(pyamino)CTG) from acetone. It structure is very close to those found in the complex with cobalt bis(dicarbollide) anion which will be discussed below [
108].
Triphenylphosphine complex {(Ph
3P)Ag[CB
11H
12]} was prepared by the reaction of Ag[CB
11H
12] with equimolar amount of PPh
3 in dichloromethane, whereas the excess of phosphine results in the formation of dimeric complex {(Ph
3P)
2Ag[CB
11H
12]}
2 [
109,
110]. In the complex {(Ph
3P)Ag[CB
11H
12]} the {AgPPh
3}
+ fragment interacts with the carborane anion through the three BH units of triangular face that is the most distant from the carbon atom with Ag…B distances of 2.504(1)-2.619(2) Å (
Figure 92a) [
109,
110]. In the complex {(Ph
3P)
2Ag[CB
11H
12]}
2 each carborane unit bridges two silver centers. Each boron cage interacts with the two silver cations through one shorter (Ag(1)-H(12′) 2.17(2) Å, Ag(1)-B(12′) 2.892(2) Å) and one longer (Ag(1)-H(7) 2.51(2) Å, Ag(1)-B(7) 3.494(2) Å) Ag-H-B interactions (
Figure 92b) [
110]. Notably, according to
11B NMR spectral data, the Ag-H-B interactions are preserved in dichloromethane solution of {(Ph
3P)Ag[CB
11H
12]} but not in {(Ph
3P)
2Ag[CB
11H
12]}
2, which reflects the significantly weaker interactions in the dimeric complex [
110].
In a similar way, complex {(Ph
3P)Ag[HCB
11H
5-7,8,9,10,11,12-Br
6]} was prepared by the reaction of Ag[HCB
11H
5-7,8,9,10,11,12-Br
6] with the equimolar amount of triphenylphosphine in dichloromethane, whereas the excess of phosphine give complex {(Ph
3P)
2Ag[HCB
11H
5-7,8,9,10,11,12-Br
6]} [
109,
110]. The silver center in {(Ph
3P)Ag[HCB
11H
5Br
6]} is coordinated with the carborane anion through two shorter (2.6963(5) Å and 2.8710(5) Å) Ag…Br interactions and one longer one (Ag(1)-B(12) 3.0953(5) Å). There is also a long interaction (3.4901(5) Å) between Ag(1) and Br(11’) on a symmetry related cage in the lattice; this bromine atom approaches Ag(1) approximately
trans to Br(12) (Br(12)-Ag(1)-Br(11’) 157.15(1)°). Although this distance is long, the coordination motif around the silver suggests that it is significant in the solid state. This interaction completes the coordination sphere at the silver center (
Figure 93) [
110].
In a similar way, a series of complexes {(R
3P)Ag[HCB
11Me
11]} (R = C
6H
5, C
6H
11, C
6H
3-3,5-Me
2) was prepared by reactions of Ag[HCB
11Me
11] with the corresponding phosphines in dichloromethane [
104,
111]. In the structure of {(Ph
3P)Ag[HCB
11Me
11]} there is a close contact between the silver cation and carborane anion through Ag…MeB interaction (Ag(1)-C(7) 2.544(2) Å). There are two more, albeit substantially longer, contacts from adjacent carborane anions in the lattice Ag(1)…C(9’) 3.154(2) Å and Ag(1)….C(12”) 3.336(2) Å) (
Figure 94) [
104,
111]. Similarly in the structure of {(Cy
3P)Ag[HCB
11Me
11]} there is a single close Ag…MeB contact, this time from the antipodal B(12)Me vertex (Ag-C(12) 2.608(2) Å). In the lattice there are two other relatively close Ag-C distances from neighboring carborane methyl groups (Ag-C(4′) 2.888(1) Å and Ag-C(8’’) 2.898(1) Å), resulting in an approximate C
3v motif around the central silver atom (
Figure 95a) [
104]. In the structure of {((3,5-Me
2C
6H
3)
3P)Ag[HCB
11Me
11]} the steric bulk of the 3,5-methyl groups has switched off any significant extra interactions from carborane anions adjacent in the lattice, there being only three close intermolecular Ag…MeB contacts from a
single carborane anion. These come from three methyl groups all on the lower surface of the cage. Two of these contacts are relatively short (Ag…C(7) 2.785(10) Å and Ag-C(12) 2.554(10) Å), while the other is longer (Ag-C(8) 3.166(10) Å), however, all are well within the sum of the van der Waals radius of the methyl group and the ionic radius of Ag
+ (3.29 Å) (
Figure 95b) [
104].
The reaction of {(Ph
3P)Ag[HCB
11Me
11]} with triphenylphosphine results in [(Ph
3P)
2Ag][HCB
11Me
11] which also can be obtained by the reaction of Ag[HCB
11Me
11] with an excess of Ph
3P (
Figure 96) [
104]. The attempt to recrystallize {(Ph
3P)Ag[HCB
11Me
11]} from diethyl ether resulted in [(Et
2O)
2Ag(PPh
3)][HCB
11Me
11] [
104].
It is known that the carba-
closo-dodecaborate anion has slightly acidic CH group but its acidity increases significantly on halogenation of the cage. This open a way to synthesis of carborane derivatives with covalent bonds C-M (M = Ag, Cu). The deprotonation of the cage C-H proton of the [HCB
11I
5Br
6]
− anion with KOH followed by treatment with AgNO
3 and recrystallization from acetonitrile gave complex {((MeCN)
4Ag
3)(Ag[HCB
11I
5Br
6]
2)·2(MeCN)}
n. The C-Ag(1)-C fragment is linear and Ag(1)-C distance is 2.142(7) Å. The interesting structural feature is the strong bonding interactions between the second silver and the upper belt iodine atoms. This is a brand new coordination motif that has not been observed before in the silver salts of any carborane monoanions. Each six-coordinated silver Ag(3) links two cages together, leading to the formation of a zig-zag coordination polymeric chain. The Ag…IB and Ag…BrB distances vary from 2.953(2) to 3.008(1) Å and from 2.954(1) to 3.030(2) Å, respectively (
Figure 97) [
107].
Reaction of (Bu
4N)[HCB
11F
11] with [Cu(mesityl)]
n and Bu
4NCl in dichloromethane resulted in formation of (Bu
4N)
2[1-ClCu-1-CB
11F
11]. The C-Cu-Cl fragment in the [1-ClCu-1-CB
11F
11]
− anion is near linear (176.0(2)°) and the Cu-C and Cu-Cl bonds are 1.917(5) and 2.136(1) Å, respectively (
Figure 98) [
112].
6. Silver and Copper Complexes with Cobalt bis(dicarbollide) anion [3,3′-Co(1,2-C2B9H11)2]−
Cobalt bis(dicarbollide) [3,3′-Co(1,2-C
2B
9H
11)
2]
− was prepared for the first time fifty years ago [
113,
114] and it still remains the subject of the increasing interest [
115] due to good perspectives of its application in various fields starting from medicine [
116,
117,
118] and finishing with nuclear waste treatment [
119], and weakly coordinating anions is one of them.
The silver salt of cobalt bis(dicarbollide) Ag[3,3′-Co(1,2-C
2B
9H
11)
2] was prepared by metathesis of the cesium salt or acid form (H
3O)[3,3′-Co(1,2-C
2B
9H
11)
2] with AgNO
3 in aqueous solution [
73,
120]. Crystals of {Ag(η
2-
p-xylene)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n were grown by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from
p-xylene [
116]. The silver atom is bound to one
p-xylene molecule in an η
2-fashion (Ag-C bonds are 2.562(7) and 2.411(7) Å) and three B-H bonds from the three dicarbollide cages through H(8), H(8’) and H(9A) atoms (the Ag…B distances are very long—2.705(6), 2.926(6) and 2.938(6) Å, however all Ag…H distances fail in a range 2.01–2.08 Å) in a distorted tetrahedral geometry. The location of the three coordinating BH groups involves that portion of the bis(dicarbollide) anion which is believed to be the most electron-rich. The bis(dicarbollide) anion has
cisoid conformation, that can be stabilized by B(8)H…Ag…HB(8’) interactions. The tridentate [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion and Ag
+ cations serve as alternating bridging ligands to give a zigzag cation-anion chain (
Figure 99) [
121].
The silver salt of 8,8’,9,9’,12,12′-hexabromo cobalt bis(dicarbollide) Ag[8,8’,9,9’,12,12′-Br
6- 3,3′-Co(1,2-C
2B
9H
8)
2] was prepared by metathesis of the corresponding cesium salt with AgNO
3 in aqueous solution [
73]. Complex [(MeCN)
4Ag][8,8’,9,9’,12,12′-Br
6-3,3′-Co(1,2-C
2B
9H
8)
2] were prepared by crystallization of Ag[8,8’,9,9’,12,12′-Br
6-3,3′-Co(1,2-C
2B
9H
8)
2] from an acetonitrile/
p-xylene solution. The crystal composes of discrete [(MeCN)Ag]
+ cations and metallacarborane anions (
Figure 100) [
121].
An unusual complex {Ag(η
2-
p-xylene)[8,8’-μ-C
6H
4-3,3′-Co(1,2-C
2B
9H
10)
2]} was prepared by metathesis of the corresponding cesium salt with AgNO
3 in aqueous solution followed by crystallization from
p-xylene [
121]. It is interesting to note that no interaction between the Ag
+ and any B-H bonds from the cage is observed. The Ag atom is η
2-bound to each of two aromatic rings in an approximately linear geometry. The average distances from the Ag atom to two aromatic rings are about the same (2.45(2) and 2.42(2) Å), perhaps indicating that the π-donor ability of the
p-xylene molecule is no different than that of the bridging aromatic ring in the [8,8’-μ-C
6H
4-3,3′-Co(1,2-C
2B
9H
10)
2]
− anion. The two aromatic rings are not parallel to each other and they are separated by about 4.1 Å. There are two crystallographically independent molecules in the unit cell, which are associated to each other through a π-π interaction between the bridging aromatic ring and the
p-xylene of the second complex (
Figure 101) [
121].
Complex {((MeCN)
2Ag)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n was obtained by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from an acetonitrile/2,2,2-trifluoroethanol solution. The silver cation is coordinated by two acetonitrile molecules arranged in a nearly linear fashion with a N-AgN angle of 167.26(16)°. The coordination sphere of silver is completed by weak interactions to B-H groups of the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion. Two BH groups from one metallacarborane anion, and one from a symmetry related another anion interact with the Ag
+ at Ag…B distances ranging from 2.35 to 2.56 Å. The rather long distances indicate that these interactions are weak. The geometry around the Ag
+ is distorted trigonal bipyramidal, with the three coordinating H atoms forming the equatorial plane. The bis(dicarbollide) anions have
cisoid conformation. Each [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion coordinates to two Ag
+ cations through three BH groups, and a stepped cation-anion coordinate chain is formed (
Figure 102) [
122].
Single crystals of {((NC(CH
2)
3CN)Ag)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n were grown by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from ethanol in the presence of glutaronitrile (1:1). The silver cation has two glutaronitrile ligands at Ag-N distances 2.160(3) and 2.171(4) Å and two close agostic Ag…H interactions at 2.036 and 2.118 Å. The corresponding B…Ag distances are not close however at 2.90 and 3.05 Å. The glutaronitrile ligands deviate from linearity with an N-Ag-N angle of 153.32(13)°. The Ag…BH interactions come from a single η
2-coordinated cobalt bis(dicarbollide) anion and run roughly perpendicular to the N-Ag-N axis, to form a silver coordination environment that can be regarded as a distorted T-shape. The glutaronitrile ligand bridges between two symmetry-equivalent silver cations, and a single-stranded coordination chain is propagated (
Figure 103) [
105].
Single crystals of {[Ag(NC(CH
2)
3CN)
2][3,3′-Co(1,2-C
2B
9H
11)
2]}
n were grown by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from ethanol in the presence of glutaronitrile (1:2) [
105]. The silver cations are coordinated by glutaronitrile ligands with distorted tetrahedral geometries. Each glutaronitrile ligand bridges between two silver cations to give a {Ag(NC(CH
2)
3CN)
2}
+ coordination chain with a criss-crossed ladder topology. The ladder is of unusual topology for a 4-connected polymer, and the Ag…Ag separations within the ladder are 6.95 Å along the sides and 4.52 Å along the zigzagging struts. The {Ag(NC(CH
2)
3CN)
2}
+ coordination chains do not show any close interactions with the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anions, and the crystal packing shows isolated coordination chains and double columns of cobalt bis(dicarbollide) anions (
Figure 104) [
105].
Single crystals of {((NC(CH
2)
4CN)Ag)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n were grown by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from ethanol in the presence of adiponitrile (1:1) [
105]. Likewise complex {((NC(CH
2)
3CN)Ag)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n, each silver cation is coordinated by two adiponitrile ligands with a distorted linear geometry. The Ag-N distances range from 2.151(4) to 2.174(3) Å, and N-Ag-N angles are 148.60(16) and 157.33(14)°. There is one close Ag…H interaction to Ag(1) at a distance 2.160 Å and a further long interaction at Ag…H distance 2.49 Å. The geometry around Ag(1) is best described as distorted trigonal pyramidal. There are three long Ag…H interactions to Ag(2) at distances ranging from 2.31 to 2.57 Å, with overall distorted trigonal bipyramidal geometry (
Figure 105) [
105].
Complex {((NC(CH
2)
5CN)Ag)[3,3′-Co(1,2-C
2B
9H
11)
2](Ag[3,3′-Co(1,2-C
2B
9H
11)
2])}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from acetone/trifluoroethanol in the presence of pimelonitrile [
123]. The two crystallographic independent silver cations Ag(1) and Ag(2a) exhibit a very distorted tetrahedral coordination. Ag(1) connects two N-atoms belonging to two crystallographic equivalent pimelonitrile ligands (Ag(1)-N(1) and Ag(1)-N(2i) distances of 2.2278(1) and 2.2358(1) Å, respectively, N(1)-Ag(1)-N(2i) angle of 124.530(2)°) and further interacts with two BH groups of an adjacent cobalt bis(dicarbollide) anion at Ag…H distances of 2.012(1) Å (Ag(1)…H(18)-B(8)) and 2.073(1) Å (Ag(1)…H(28)-B(17)) to complete the distorted tetrahedral coordination environment. The short Ag(1)…H(18) and Ag(1)…H(28) distances indicate that these interactions exhibit strong character. Ag(2) interacts with four BH groups belonging to two neighbouring crystallographic equivalent [3,3′-Co(1,2-C
2B
9H
11)
2]
− anions: Ag(2)…H(35a”) and Ag(2)…H(43”) at short Ag…H distances of 2.097(1) and 2.032(1) Å, respectively, and two weaker interactions for Ag(2)…H(45a) (2.247(1) Å) and Ag(2)…H(54a) (2.192(1) Å) (
Figure 106) [
123].
Complex {Ag
3(NC(CH
2)
6CN)
2[3,3′-Co(1,2-C
2B
9H
11)
2]
3}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from ethanol in the presence of suberonitrile [
123]. The structural unit is composed of three Ag
+ cations, two bridging ligands, and three cobalt bis(dicarbollide) anions. Two crystallographic independent atoms, Ag(1) and Ag(2), reveal identical coordination environment, whereas the Ag(3) is distinct. Both Ag(1) and Ag(2) are four coordinated centers, bounded by two N-atoms of two crystallographic independent suberonitrile ligands and also two BH groups belonging to two distinct [3,3′-Co(1,2-C
2B
9H
11)
2]
− anions, and mutually exhibiting distorted tetrahedral geometry. In Ag(1) center the Ag-N bond lengths are 2.228(4) Å (Ag(1)-N(1)) and 2.208(4) Å (Ag(1)-N(4
iii) and Ag…H distances are found to be 1.981(7) Å (Ag(1)…H(52
iv)) and 1.9728 Å (Ag(1)…H(81)), suggesting the existence of two strong interactions. The Ag-N distances in the Ag(2) center are analogous to those observed for Ag(1) at 2.245(4) Å (Ag(2)-N(3)) and 2.198(4) Å (Ag(2)-N(2)). However, while the two Ag…H-B interactions involving Ag(1) are similar, those of the Ag(2) center are significantly distinct: Ag(2)…H(63) is a stronger interaction and Ag(2)…H(71
v) has a considerable weaker nature, as evidently indicated by Ag…H distances of 1.877(1) and 2.314(1) Å, respectively. The Ag(3) coordination environment consists exclusively of six Ag-H-B interactions with the BH groups belonging to three crystallographic independent cobalt bis(dicarbollide) anions, leading to a center with highly distorted octahedral geometry (
Figure 107) [
123].
Complex {(Ag(pyz)(MeCN)
2)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] with pyrazine in acetonitrile [
106]. The silver cation is in a distorted tetrahedral geometry, coordinating to two acetonitrile molecules and to two pyrazine ligands. (Ag-N bonds range from 2.266(2) to 2.471(3) Å). Each pyrazine ligand is bridging, and a coordination chain is formed which runs along the
c-axis. The chains occur in pairs with a second chain running parallel but offset with the first, so that silver cations of one chain lies over the center of the pyrazine ring of the other one with an Ag…centroid distance of 3.732 Å. The coordinated acetonitrile molecules are directed away from the chain and lie parallel to acetonitrile molecules from two adjacent chains. The orientation of the acetonitrile molecules of adjacent coordination chains creates a “box-like” arrangement and the cobalt bis(dicarbollide) anions form rows and lie in the channels created by this packing arrangement. Each [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion is in close proximity to neighboring anions with a closest contact of 2.271 Å between the BH…HC groups, indicating the presence of weak dihydrohen interactions between the metallacarborane anions (
Figure 108) [
106].
Two polymorphs of complex {(Ag(4,4′-Bipy)(MeCN))[3,3′-Co(1,2-C
2B
9H
11)
2]}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] with 4,4′-bipyridine in acetonitrile [
106]. The silver coordination environment is essentially the same for both polymorphic forms, with silver cation coordinated by an acetonitrile and by two 4,4′-bipyridine molecules in a T-shape arrangement. In both polymorphs, the silver atom is in close proximity to a neighboring cobalt bis(dicarbollide) anion with Ag…H distances of 2.733 and 2.696 Å, too long to be considered effective interactions. The bridging 4,4′-bipyridine ligands form coordination chains that occur in infinite π-stacked columns. Each chain shows the coordinated acetonitrile molecules alternating in opposite directions. A box-like channels are generated that contains metallacarborane anions (
Figure 109) [
106].
The crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from dimethylsulfoxide in the presence of 4,4′-bipyridine gave complex {(Ag(4,4′-Bipy)(DMSO)
2)[3,3′-Co(1,2-C
2B
9H
11)
2]·DMSO}
n [
106]. The silver cation is coordinated by two 4,4′-bipyridine molecules (Ag-N bond length 2.187(8) and 2.161(8) Å) in an almost linear arrangement (N-Ag-N angle 165.4(3)°). The four-coordinated silver sphere is completed by two DMSO molecules. The {Ag(4,4′-Bipy)(DMSO)
2}
+ cationic fragments form coordination chains. The chains are arranged in pairs with the chain offset so that the silver cations are opposite the center of one of the arene rings of a ligand from the neighboring chain, at Ag…centroid distance of 3.874 Å. There are π…π staking interactions between the chais at a centroid separation of 3.576 Å. The extended structure reveals a herring-bone arrangement of the cobalt bis(dicarbollide) anions in the
bc-plane (
Figure 110).
Complex {[Ag
2(bppz)
3][3,3′-Co(1,2-C
2B
9H
11)
2]
2·3.5(MeCN)}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from acetonitrile in the presence of 2,3-bis(2-pyridyl)pyrazine [
106]. The crystals contain the {[Ag
2(bppz)
3]
2+}
n coordination chain which is similar to those found in the related complexes with the carba-
closo-dodecaborate anion (See
Figure 89). In the extended structure, the chains are separated by snaking chains of cobalt bis(dicarbollide) anions. The anionic chains have anions in one of two orientations and show dihydrogen bonding with closest BH…HC distances of 1.947 and 2.312 Å. Both the chains of polymeric {[Ag
2(bppz)
3]
2+}
n cations and the cobaltacarborane anions run in a parallel fashion along the c-axis forming a square grid or checkerboard pattern (
Figure 111).
Complex {Ag(DABCO)[3,3′-Co(1,2-C
2B
9H
11)
2]}
n was prepared by crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] from acetonitrile in the presence of 1,4-diazabicyclo[2.2.2]octane [
77]. The silver atom coordinates to two nitrogen atoms belonging at two crystallographically equivalent DABCO ligands (Ag-N distances 2.1752(17) and 2.180(17) Å) and further interacts with three BH groups belonging at three equivalent cobalt bis(dicarbollide) anions (Ag…H distances 2.8889(2), 2.4465(2), and 2.8958(1) Å). With consideration of the Ag-N coordination bonds and all the Ag-H-B interactions, the coordination geometry around silver atom can be visualized as intermediate between a deformed trigonal bipyramid and a distorted square pyramid. The bridging nature of the DABCO ligand between the Ag centers lead to the formation of cationic linear polymer chains {Ag(DABCO)}
+, which run along the
a axis with the Ag…Ag intrachain distance of 6.9544(5) Å. These cationic coordination chains are intercalated by the metallacarborane anions, ultimately generating a zigzag arrangement of both the coordination chains and carborane anions (
Figure 112) [
77].
The supramolecular complexes [Ag(MeCN)(H
2O)(CTV)
2][3,3′-Co(1,2-C
2B
9H
11)
2] and [Ag(MeCN)
3(CTV)][3,3′-Co(1,2-C
2B
9H
11)
2]·0.5MeCN were prepared by co-crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] and cyclotriveratrylene (CVT) from a acetonitrile/2,2,2-trifluoroethanol solution depending on the solvent ratio [
122]. In the structure of [Ag(MeCN)(H
2O)(CTV)
2][3,3′-Co(1,2-C
2B
9H
11)
2] the silver cation is located within the CTV molecular cavity. The coordination sphere of silver atom is four-coordinate with distorted tetrahedral geometry. There is one aquo ligand at Ag-O distance 2.369(15) Å, an acetonitrile molecule at Ag-N 2.038(13) Å and Ag-C bonds to the C6 position of one veratrole ring within the CTV (Ag-C distance 2.607(7) Å) and to the symmetry equivalent C3 position of an adjacent ring. The aquo ligand is exo to the CTV cavity, while the acetonitrile ligand is inside the CTV cavity and points along the plane of the arene ring not involved in coordination to silver. The bis(CTV) capsule actually comprises a {Ag(H
2O)(MeCN)(CTV)}
+ complex with a second molecule of CTV in an inverted orientation, associated with the first via host-guest interactions with the coordinated acetonitrile (
Figure 113a). The capsules stack in the
c direction, possibly through a weak hydrogen bond between the coordinated aquo ligand and dimethoxy group of neighboring capsules (O…O separation 3.43 Å). Capsules that are adjacent in the
bc plane associate through π-π interactions between arene rings of the CTV molecules where the centroid separation is 3.696 Å. The cobalt bis(dicarbollide) anions are located in hydrophobic pockets and show no significant interactions with neighboring molecules (
Figure 113b) [
122].
In the structure of [Ag(MeCN)
3(CTV)][3,3′-Co(1,2-C
2B
9H
11)
2]·0.5MeCN the silver cation has an unusual coordination sphere, comprising three acetonitrile ligands and two methoxy O atoms from the CTV. The Ag-N bond lengths are 2.184(2), 2.206(2), and 2.466(2) Å, and the MeCN ligands are arranged in a distorted T-shape. The coordination sphere of silver is completed by coordination to both O atoms of one of the veratrole moieties within the CTV at long Ag-O distances 2.685(2) and 2.834(2) Å to form a five-membered chelate ring. The coordination geometry around the silver atom may be described as highly distorted trigonal bipyramidal with MeCN ligands in the axial positions, and the dimethoxy unit and other MeCN comprising the equatorial plane. One of the coordinated acetonitrile molecules occupies the CTV molecular cavity to form a host-guest interaction. The guest MeCN is parallel to the coordinating veratrole ring of the CTV. In the crystal layers of the [Ag(MeCN)
3CTV]
+ cations separated by layers of the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anions. The [Ag(MeCN)
3CTV]
+ cations form pairs with the roughly linear MeCN-Ag-NCMe links nearly parallel to each other and with the CTV hosts in opposite orientations. Each [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion is at hydrogen bonding distances to one CTV arene ring at C-H…π distance 2.78 Å, but do not show other close contacts to other molecular components (
Figure 114) [
122].
The [Ag(MeCN)
4](CTV)[3,3′-Co(1,2-C
2B
9H
11)
2]·0.25MeCN were prepared by co-crystallization of Ag[3,3′-Co(1,2-C
2B
9H
11)
2] and cyclotriveratrylene from acetonitrile [
122]. The silver atom forms tetrahedral [Ag(MeCN)
4]
+ complex ion, which is a guest molecule with one coordinated MeCN ligand forming a host-guest interaction with the CTV. The guest MeCN extends into the CTV molecular cavity and approaches over one of the -CH
2- links of the CTV. The cobalt bis(dicarbollide) anion forms close interactions with both the [Ag(MeCN)
4]
+ cation and CTV molecule. There are three C-H…O hydrogen bonds between the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion and the methoxy groups of a single veratrole moiety of CTV (2.52, 2.59, 2.79 Å). The fourth C-H group of the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion is in close contact with a N atom of a coordinated MeCN at C-H…N distance 2.51 Å, leading to the dimeric {([Ag(MeCN)4]
+∩CTV)[3,3-Co(1,2-C
2B
9H
11)
2]}
2 assembly (
Figure 115) [
122].
Another supramolecular host-guest complex [Ag(tris(isonic)CTG)
2][3,3-Co(1,2-C
2B
9H
11)
2]·9(MeCN) was prepared by co-crystallization of Ag[3,3-Co(1,2-C
2B
9H
11)
2] with tris-(isonicotinoyl)cyclotriguaiacylene (tris(isonic)CTG) from acetonitrile. The silver atom has a tetrahedral geometry (Ag-N bond lengths 2.233(7)–2.448(7) Å) and is bound by four different ligand molecules. Each molecule of L acts as a bidentate bridge of two symmetry-related silver atoms to form a 1-D {Ag(tris(isonic)CTG)
2+}
n coordination polymer. The 1-D coordination polymers are interwoven through dimeric host-guest associations between the coordinated molecular host moieties of adjacent chains and π…π stacking interactions to give an extended 2-D network (
Figure 116). The cobalt bis(dicarbollide) anions are within channels created by packing the interwoven 2-D sheets [
124].
Complex [Ag
2(tris(3-pyamino)CTG)
2(MeCN)
2][3,3′-Co(1,2-C
2B
9H
11)
2]
2·2(MeCN) was prepared by co-crystallization of Ag[3,3-Co(1,2-C
2B
9H
11)
2] with tris(3-pyridylmethylamino)- cyclotriguaiacylene (tris(3-pyamino)CTG) from a acetonitrile/acetone solution [
108]. The structure is formed by the [Ag
2(tris(3-pyamino)CTG)
2(MeCN)
2]
+ cations, cobalt bis(dicarbollide) anions and MeCN solvent molecules. The cation [Ag
2(tris(3-pyamino)CTG)
2(MeCN)
2]
2+ is a dinuclear, dimeric species with a capsule-like structure. The centrosymmetric capsule forms through the head-to-head coordination of two ligands to two silver atoms. The silver cation is coordinated by a terminal acetonitrile ligand at an Ag-N distance of 2.321) Å, and three pyridyl groups at Ag-N distances of 2.327, 2.350), and 2.292 Å in a distorted tetrahedral geometry (
Figure 117) [
108].
Polymeric complex {[Ag(tris(4-pyamino)CTG){NC(CH
2)
3CN}]∩(NC(CH
2)
3CN) [3,3′-Co(1,2-C
2B
9H
11)
2]·n(NC(CH
2)
3CN)}
n was prepared by co-crystallization of Ag[3,3-Co(1,2-C
2B
9H
11)
2] with tris(4-pyridylmethylamino)cyclotriguaiacylene (tris(4-pyamino)CTG) from acetonitrile-2,2,2-trifluoroethanol in the presence of glutaronitrile [
108]. The silver cation has a tetrahedral geometry coordinated by three pyridyl moieties from three different molecules of tris(4-pyamino)CTG (Ag-N bond lengths of 2.343(6)–2.369(6) Å) and a monodentate capping glutaronitrile molecule (Ag-N bond length of 2.25(1) Å). The three cavitands coordinating to each silver cation are orientated such that two have the top face orientated in the same direction with the third directed in an opposing direction. The cavitand is likewise coordinated to three different symmetry-related silver cations and acts as a host for the second molecule of glutaronitrile. This results in 2D-network structure incorporating roughly rectangular channels. These channels are largely occupied by the globular metallacarbaborane anions and glutaronitrile molecules (
Figure 118) [
108].
Complex {[Ag(tris(isonic)CTG-Pr)[3,3′-Co(1,2-C
2B
9H
11)
2]]·2.5(DMF)·H
2O}
n was prepared by co-crystallization of Ag[3,3-Co(1,2-C
2B
9H
11)
2] with tris(isonicotinoyl)-tris(propoxy)- cyclotriguaiacylene (tris(isonic)CTG-Pr) from solution in N,N-dimethylformamide [
125]. In the crystal, the tris(isonic)CTG-Pr ligands coordinate to three symmetry-equivalent silver atoms, all of which are of distorted tetrahedral geometry, and the [3,3′-Co(1,2-C
2B
9H
11)
2]
− anion coordinates to the metal through a hydridic interaction. The Ag-N and Ag…H(B) bond lengths were measured at 2.264(4), 2.280(4) and 2.176 Å, respectively, alongside N-Ag-N bond angles of 127.48(14), 101.76(14) and 97.97(13)°. It results in a1D ladder-type motif, whereby the tris(isonic)CTG-Pr ligands are all inwardly orientated to afford a quasi-cylindrical arrangement of head-to-head ligands (
Figure 119) [
125].
Complex {[Ag
2(tris(2-quinamino)CTG-Pr)
2][3,3′-Co(1,2-C
2B
9H
11)
2]
2·1.5(MeNO
2)}
n was prepared by co-crystallization of Ag[3,3-Co(1,2-C
2B
9H
11)
2] with tris(2-quinolylmethyl)tris(propoxy)- cyclotriguaiacylene (tris(2-quinamino)CTG-Pr) from a solution in acetonitrile/nitromethane [
125]. In the crystal each silver cation is in approximately linear arrangement and is coordinated by two independent tris(2-quinaminoc)CTG-Pr ligands, with Ag-N bond lengths and N-Ag-N angles of 2.189(7)–2.197(7) Å and 174.3(3) and 176.2(3)°, respectively, that results in 1D coordination polymer. There is some bonding contribution from a proximal ethereal oxygen, with O…Ag distance of 2.513(7) Å. The host-guest interactions between quinolyl arm and the shallow hydrophobic cavity of the ligand core are stabilised by π…π interactions between the quinolyl moiety and the [a,d,g]cyclononatriene core, with centroid separation of 3.809 Å. Neighbouring 1D chains aggregate through π…π interactions, with aromatic centroid separations of 3.687 Å, and aggregation of propyl moieties which affords small pockets that are filled with disordered solvent and cobalt bis(dicarbollide) anions (
Figure 120) [
125].
Syntheses of two silver(I) complexes with cobalt bis(dicarbollide) based on the chelating phosphine ligand [1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2]
− were described [
126]. The complex {(Ph
3P)Ag[1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2]}·acetone was prepared by the reaction of (Me
4N)[1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2] with [(Ph
3P)AgCl] in ethanol, whereas the complex {(Me
2CO)Ag[1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2]} was prepared by the reaction of (Me
4N)[1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2] with AgClO
4 in ehanol/acetone. The silver cation in both complexes is tetrahedrally coordinated, with one long and three short contacts. In the first complex two of the short contacts are due to the two P atoms from the [1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2]
− ligand (Ag-P 2.5507(9) and 2.5470(10) Å), and the third is due to PPh
3 (Ag-P 2.4713(10) Å); the fourth bond is due to the O atom of acetone molecule (Ag…O 2.906(3) Å). In the second complex two short contacts are due to the P atoms of the [1,1’-(Ph
2P)
2-3,3′-Co(1,2-C
2B
9H
10)
2]
− ligand (Ag-P 2.4894(6) and 2.4933(6) Å), and the third is due to the O atom of acetone molecule (Ag-O 2.3895(17) Å), whereas the bond is due to Ag…H(12′) contact (2.17 Å), assembling the molecules to 1D-polymeric chain (
Figure 121) [
126].
Unlike the silver complexes, copper complexes with cobalt bis(dicarbollide) anion are represented by only a few examples. A mixture of complexes {(Ph
3P)Cu)[3,3′-Co(1,2-C
2B
9H
11)
2]} and [(Ph
3P)
3Cu][3,3′-Co(1,2-C
2B
9H
11)
2] was prepared by the reaction of Cs[3,3′-Co(1,2-C
2B
9H
11)
2] with CuCl
2 and triphenylphosphine or, alternatively, with complex [(Ph
3P)CuCl]
4 in dichloromethane, whereas the reaction of Cs[3,3′-Co(1,2-C
2B
9H
11)
2] with complex [(Ph
3P)
3CuCl] gives only the second complex [(Ph
3P)
3Cu][3,3′-Co(1,2-C
2B
9H
11)
2]. The last one can be obtained by the treatment of the first complex {(Ph
3P)Cu)[3,3′-Co(1,2-C
2B
9H
11)
2]} with excess of PPh
3. The copper(I) cation in the complex {(Ph
3P)Cu)[3,3′-Co(1,2-C
2B
9H
11)
2]} is in distorted tetrahedral arrangement formed by the P atom of triphenylphosphine ligand (Cu-P bond is 2.2264(2) Å) and three B(8)H, B(8’)H and B(4′)H groups of the cobalt bis(dicarbollide) anion (Cu…B and Cu…H distances are 2.2265(9)–2.3243(9) and 1.721(14)–2.014(14) Å, respectively) (
Figure 122) [
127,
128].
The copper(II) complex [Cu(Phen)
3][3,3′-Co(1,2-C
2B
9H
11)
2]
2 was prepared by the reaction of [Cu(Phen)
3](OAc)
2 with Cs[3,3′-Co(1,2-C
2B
9H
11)
2] in aqueous solution (
Figure 123) [
129,
130].