New BEDT-TTF Radical Cation Salt with Mixed Anions: α'-[BEDT-TTF]2[CuBr2]0.4[CuCl2]0.6
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation
2.2. Molecular Structure of Cation and Anion Moieties of 1
| Formula | C20H16Br0.8Cl1.2CuS16 |
| Formula weight | 938.85 |
| Crystal size (mm) | 0.16 × 0.16 × 0.02 |
| Crystal system | monoclinic |
| Space group | P2/c |
| a (Å) | 7.8124(14) |
| b (Å) | 6.6564(11) |
| c (Å) | 30.444(6) |
| β (º) | 96.062(2) |
| V (Å3) | 1574.3(5) |
| Z | 2 |
| Dcalc (g cm−3) | 1.981 |
| F(000) | 938 |
| μ(Mo- Kα) (mm−1) | 2.878 |
| T (K) | 93 |
| Measured 2 θ range (°) | 6.16–54.94 |
| No. of reflections collected | 12296 |
| Independent reflections | 3598 |
| No. of reflections with I > 2σ(I) | 3031 |
| R (I > 2σ(I)) a | 0.0349 |
| Rw (all data) b | 0.0747 |
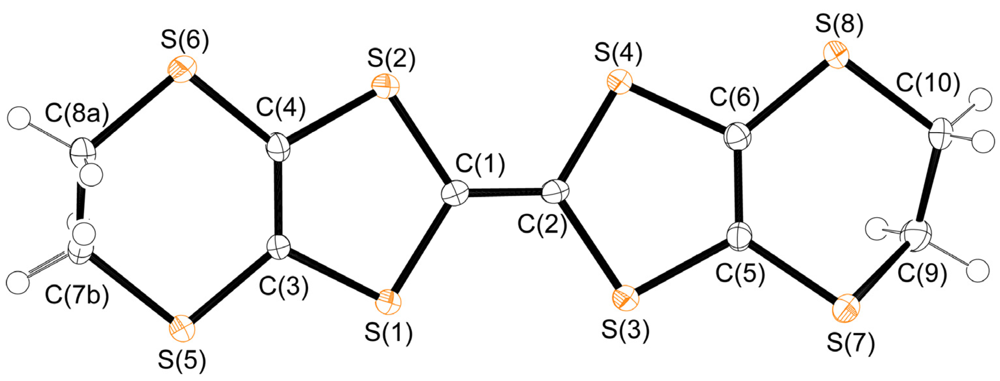
| Bond | Bond length | Bond | Bond length |
|---|---|---|---|
| C(1)–C(2) | 1.336(4) | C(3)–C(4) | 1.351(3) |
| C(5)–C(6) | 1.353(3) | S(1)–C(1) | 1.732(3) |
| S(2)–C(1) | 1.737(2) | S(3)–C(2) | 1.740(2) |
| S(4)–C(2) | 1.743(3) | S(1)–C(3) | 1.755(2) |
| S(2)–C(4) | 1.753(3) | S(3)–C(5) | 1.748(3) |
| S(4)–C(6) | 1.757(3) | S(5)–C(3) | 1.748(3) |
| S(6)–C(4) | 1.742(3) | S(7)–C(5) | 1.742(3) |
| S(8)–C(6) | 1.743(3) | ||
| Bond | Bond angle | Bond | Bond Angle |
| S(1)–C(1)–C(2) | 122.48(19) | S(2)–C(1)–C(2) | 122.66(19) |
| S(3)–C(2)–C(1) | 123.02(19) | S(4)–C(2)–C(1) | 122.19(19) |
| S(1)–C(1)–S(2) | 114.84(14) | S(3)–C(2)–S(4) | 114.74(14) |
| S(1)–C(3)–C(4) | 116.50(19) | S(2)–C(4)–C(3) | 116.75(19) |
| S(3)–C(2)–C(1) | 123.02(19) | S(4)–C(2)–C(1) | 122.19(19) |
| C(1)–S(1)–C(3) | 95.38(12) | C(1)–S(2)–C(4) | 95.18(12) |
| C(2)–S(3)–C(5) | 94.97(12) | C(2)–S(4)–C(6) | 95.31(12) |
| C(3)–C(4)–S(6) | 128.6(2) | C(4)–C(3)–S(5) | 127.97(19) |
| C(5)–C(6)–S(8) | 128.8(2) | C(6)–C(5)–S(7) | 125.0(2) |
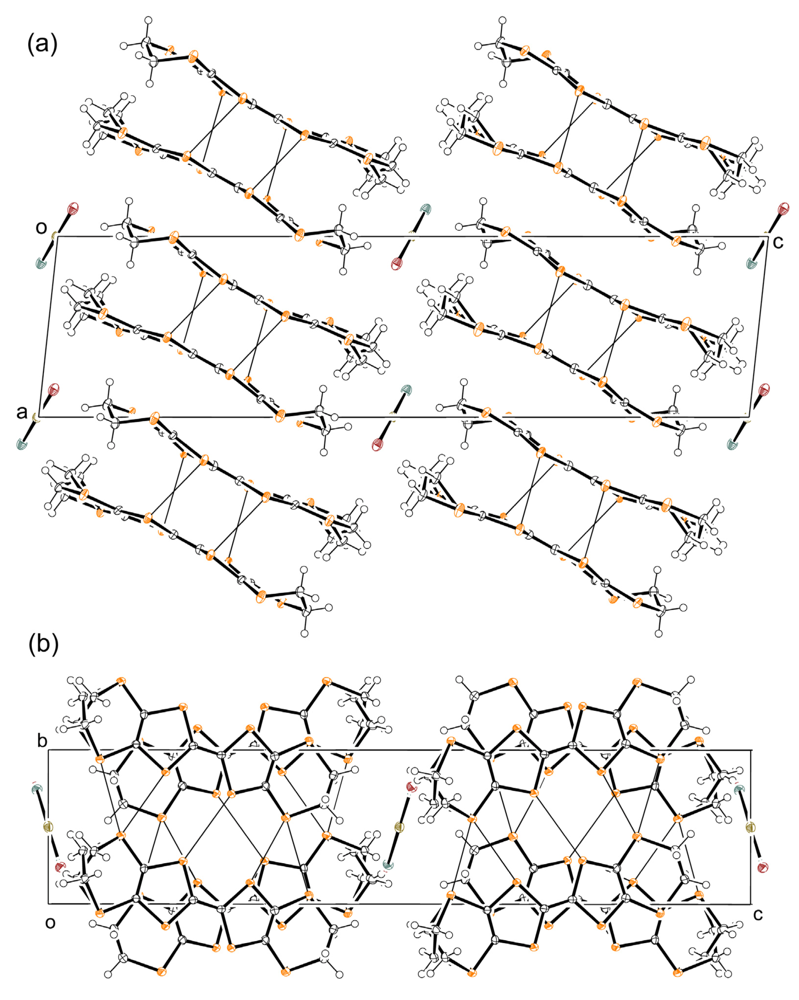
2.3. Crystal Structure of 1
2.4. Calculated Band Structure of 1
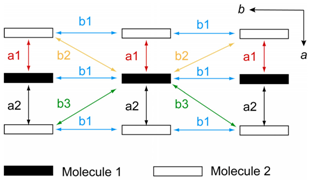
| Interaction | S | Interaction | S |
| a1 | −13.1 | b1 | 1.6 |
| a2 | −8.1 | b2 | −0.33 |
| b3 | −0.73 |
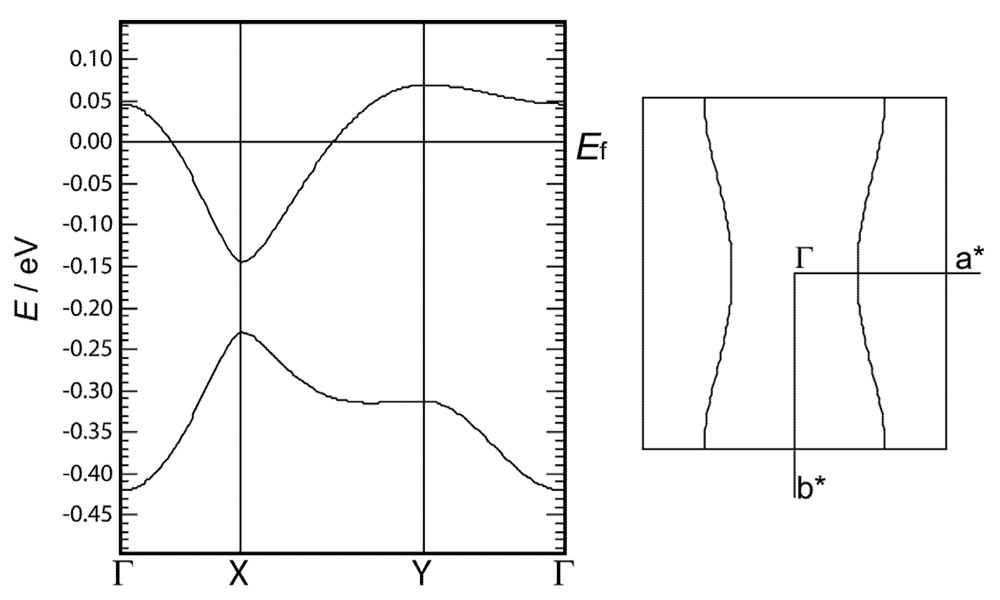
2.5. Electrical Resistivity of 1
3. Experimental Section
3.1. Materials
3.2. Preparation of α'-[BEST-TTF]2(CuBr2)0.4(CuCl2)0.6
3.3. Crystal Structure Determination
3.4. Overlap Integral Calculation
3.5. Electrical Resistivity
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Shibaeva, R.P.; Yagubskii, E.B. Molecular conductors and superconductors based on trihalides of BEDT-TTF and some of its analogues. Chem. Rev. 2004, 104, 5347–5378. [Google Scholar] [CrossRef]
- Taniguchi, H.; Miyashita, M.; Uchiyama, K.; Satoh, K.; Môri, N.; Okamoto, H.; Miyagawa, K.; Kanoda, K.; Hedo, M.; Uwatoko, Y. Superconductivity at 14.2 K in Layered Organics under Extreme Pressure. J. Phys. Soc. Jpn. 2003, 72, 468–471. [Google Scholar] [CrossRef]
- Kini, A.M.; Geiser, U.; Wang, H.H.; Carlson, K.D.; Williams, J.M.; Kwok, W.K.; Vandervoort, K.G.; Thompson, J.E.; Stupka, D.L.; Jung, D.; et al. A new ambient-pressure organic superconductor, κ-(ET)2Cu[N(CN)2]Br, with the highest transition temperature yet observed (inductive onset Tc = 11.6 K, resistive onset = 12.5 K). Inorg. Chem. 1990, 29, 2555–2557. [Google Scholar]
- Williams, J.M.; Kini, A.M.; Wang, H.H.; Carlson, K.D.; Geiser, U.; Montgomery, L.K.; Pyrka, G.J.; Watkins, D.M.; Kommers, J.M.; Boryschuk, S.J.; et al. From semiconductor-semiconductor transition (42 K) to the highest-Tc organic superconductor, κ-(ET)2Cu[N(CN)2]Cl (Tc = 12.5 K). 1990, 29, 3272–3274. [Google Scholar]
- Posselt, H.; Müller, H.; Andres, K.; Sushko, Y.V.; Saito, G. Reentrant meissner effect in the organic superconductor κ-ET2Cu[N(CN)2]Cl1−xBrx under pressure. Synth. Met. 1995, 70, 917–918. [Google Scholar] [CrossRef]
- Ishiguro, T.; Ito, H.; Yamauchi, Y.; Ohmichi, E.; Kubota, M.; Yamochi, H.; Saito, G.; Kartsovnik, M.V.; Tanatar, M.A.; Sushko, U.V.; et al. Electronic phase diagrams and fermi surfaces of κ-(ET)2X, the high Tc organic superconductors. Synth. Met. 1997, 85, 1471–1478. [Google Scholar] [CrossRef]
- Mori, H.; Okano, T.; Tanaka, S.; Tamura, M.; Nishio, Y.; Kajita, K.; Mori, T. Discontinuous Change of Metal-Insulator Transition in the Alloyed θ-system: θ-(BEDT-TTF)2(Rb1−xCsx)Zn(SCN)4. J. Phys. Soc. Jpn. 2000, 69, 1751–1756. [Google Scholar]
- Kato, R.; Kobayashi, H.; Kobayashi, A.; Nishio, Y.; Kajita, K.; Sasaki, W. Partially” superconducting behavior of θ-(BEDT-TTF)2(I3)1−x(A)x (A = AuI2, I2Br) system. Chem. Lett. 1986, 6, 957–960. [Google Scholar]
- Mori, T. Structural genealogy of BEDT-TTF-based organic conductors III. Twisted molecules: δ and α' phases. Bull. Chem. Soc. Jpn. 1999, 72, 2011–2027. [Google Scholar]
- Beno, M.A.; Firestone, M.A.; Leung, P.C.W.; Sowa, L.M.; Wang, H.H.; Williams, J.M.; Whangbo, M.-H. Crystal and band electronic structures of a new class of 2:1 organic conducting salts α'-(BEDT-TTF)2X, X− = Ag(CN)2−, Au(CN)2− and AuBr2−. Solid State Commun. 1986, 57, 735–739. [Google Scholar] [CrossRef]
- Kurmoo, M.; Talham, D.R.; Day, P.; Howard, J.A.K.; Stringer, A.M.; Obertelli, D.S.; Friend, R.H. BEDT-TTF)2CuCl2, a new conducting charge transfer salt. Synth. Met. 1988, 22, 415–418. [Google Scholar] [CrossRef]
- Guionneau, P.; Kepert, C.J.; Bravic, G.; Chasseau, D.; Truter, M.R.; Kurmoo, M.; Day, P. Determining the charge distribution in BEDT-TTF salts. Synth. Met. 1997, 86, 1973–1974. [Google Scholar] [CrossRef]
- Asplund, M.; Jagner, S.; Nilsson, M. Crystal structures of tetrabutylammonium dichlorocuprate(I) and tetrabutylammonium dibromocuprate(I), [N(C4H9)4][CuCl2] and [N(C4H9)4][CuBr2]. Acta Chem. Scand. 1983, A37, 57–62. [Google Scholar]
- Enberg, A. An X-ray refinement of the crystal structure of copper(II) chloride dihydrate. Acta Chem. Scand. 1970, 24, 3510–3520. [Google Scholar] [CrossRef]
- Helmholz, L. The crystal structure of anhydrous cupric bromide. J. Am. Chem. Soc. 1947, 69, 886–889. [Google Scholar] [CrossRef]
- Obertelli, S.D.; Friend, R.H.; Talham, D.R.; Kurmoo, M.; Day, P. The magnetic susceptibility and EPR of the organic conductors α'-(BEDT-TTF)2X, X = AuBr2, CuCl2 and Ag(CN)2. J. Phys. Condens. Matter 1989, 1, 5671–5680. [Google Scholar] [CrossRef]
- Horiuchi, S.; Yamochi, H.; Saito, G.; Sakaguchi, K.; Kusunoki, M. Nature and origin of stable metallic state in organic charge-transfer complexes of bis(ethylenedioxy)tetrathiafulvalene. J. Am. Chem. Soc. 1996, 118, 8604–8622. [Google Scholar]
- Mori, T.; Kobayashi, A.; Sasaki, Y.; Kobayashi, H.; Saito, G.; Inokuchi, H. The intermolecular interaction of tetrathiafulvalene and bis(ethylenedithio)tetrathiafulvalene in organic metals. calculation of orbital overlaps and models of energy-band structures. Bull. Chem. Soc. Jpn. 1985, 125, 125–134. [Google Scholar]
- Kobayashi, H.; Kato, R.; Mori, T.; Kobayashi, A.; Sasaki, Y.; Saito, G.; Inokuchi, H. Organic conductors based on multi-sulfur π-donor and/or π-acceptor molecules-BEDT-TTF, BMDT-TTF, BPDT-TTF, and M(dmit)2−. Mol. Cryst. Liq. Cryst. 1985, 125, 125–134. [Google Scholar] [CrossRef]
- Miller, J.S. Extended Linear Chain Compounds; Plenum Press: New York, NY, USA, 1983; Volume 1–3. [Google Scholar]
- Altomare, A.; Burla, M.C.; Gamalli, M.; Cascarano, M.; Giacovazzo, G.L.; Guagliardi, A.; Moliterni, A. SIR97: A new tool for crystal structure determination and refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A. 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Summerville, R.H.; Hoffmann, R. Tetrahedral and other M2L6 transition metal dimers. J. Am. Chem. Soc. 1976, 98, 7240–7254. [Google Scholar] [CrossRef]
- Joergensen, K.A.; Hoffmann, R. Binding of alkenes to the ligands in OsO2X2 (X = O and NR) and CpCo(NO)2. A frontier orbital study of the formation of intermediates in the transition-metal-catalyzed synthesis of diols, amino alcohols, and diamines. J. Am. Chem. Soc. 1986, 108, 1867–1876. [Google Scholar]
- Schilling, B.E.R.; Hoffmann, R.; Faller, J.W. Effect of ligand asymmetry on the structure and reactivity of CpMLL'(allyl) (Cp = cyclopentadienyl, M = metal, L = ligand) and -(ethylene) complexes. J. Am. Chem. Soc. 1979, 101, 592–598. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kubo, K.; Yamashita, M. New BEDT-TTF Radical Cation Salt with Mixed Anions: α'-[BEDT-TTF]2[CuBr2]0.4[CuCl2]0.6. Crystals 2012, 2, 284-293. https://doi.org/10.3390/cryst2020284
Kubo K, Yamashita M. New BEDT-TTF Radical Cation Salt with Mixed Anions: α'-[BEDT-TTF]2[CuBr2]0.4[CuCl2]0.6. Crystals. 2012; 2(2):284-293. https://doi.org/10.3390/cryst2020284
Chicago/Turabian StyleKubo, Kazuya, and Masahiro Yamashita. 2012. "New BEDT-TTF Radical Cation Salt with Mixed Anions: α'-[BEDT-TTF]2[CuBr2]0.4[CuCl2]0.6" Crystals 2, no. 2: 284-293. https://doi.org/10.3390/cryst2020284




