Fabrication of Mesoporous V2O5@g-C3N4 Nanocomposite as Photocatalyst for Dye Degradation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nanostructures’ Description Methods
2.2. Batch Experiments
3. Results
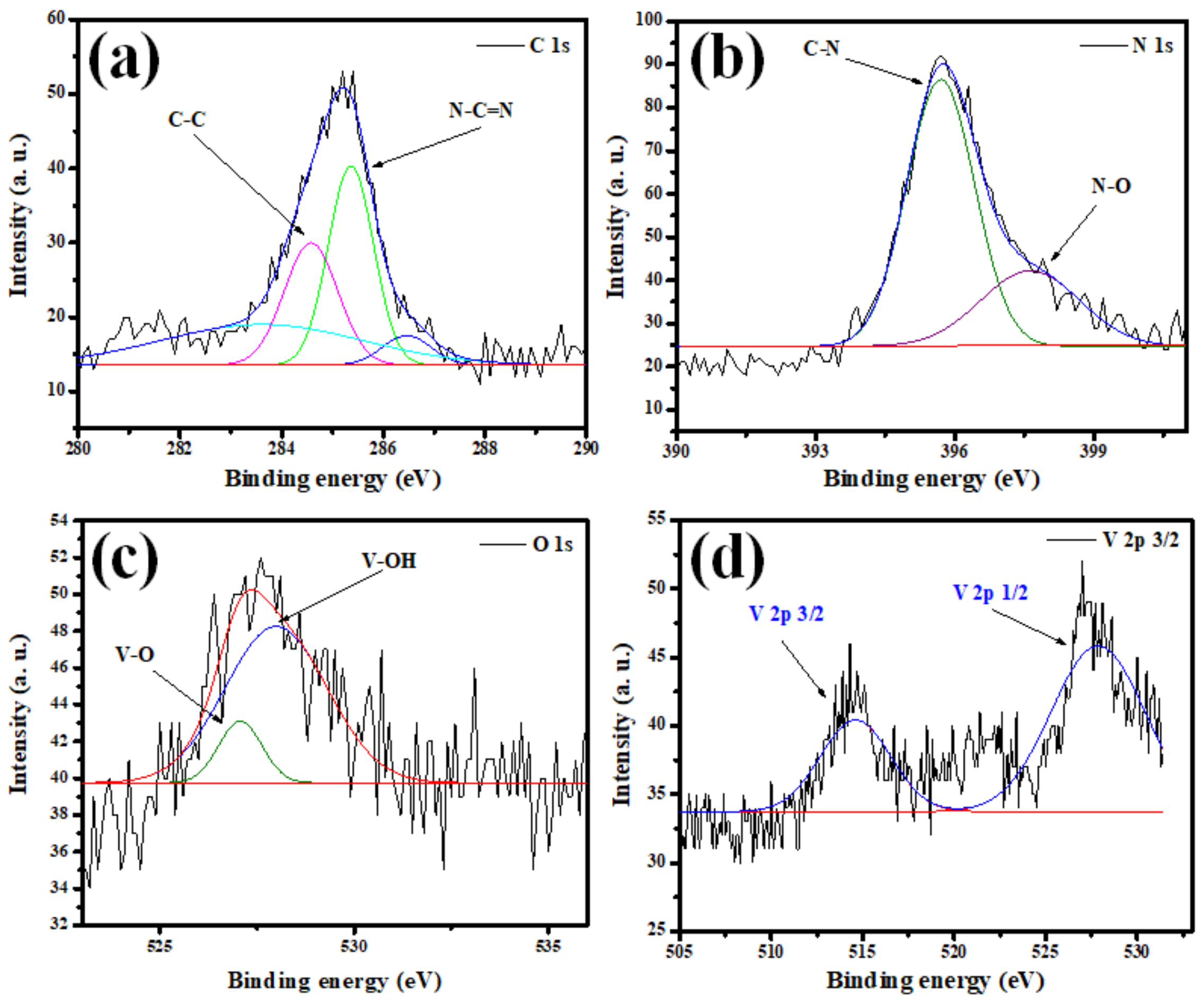
3.1. Nanostructures’ Characterizations
3.2. Photocatalysis Performance Part
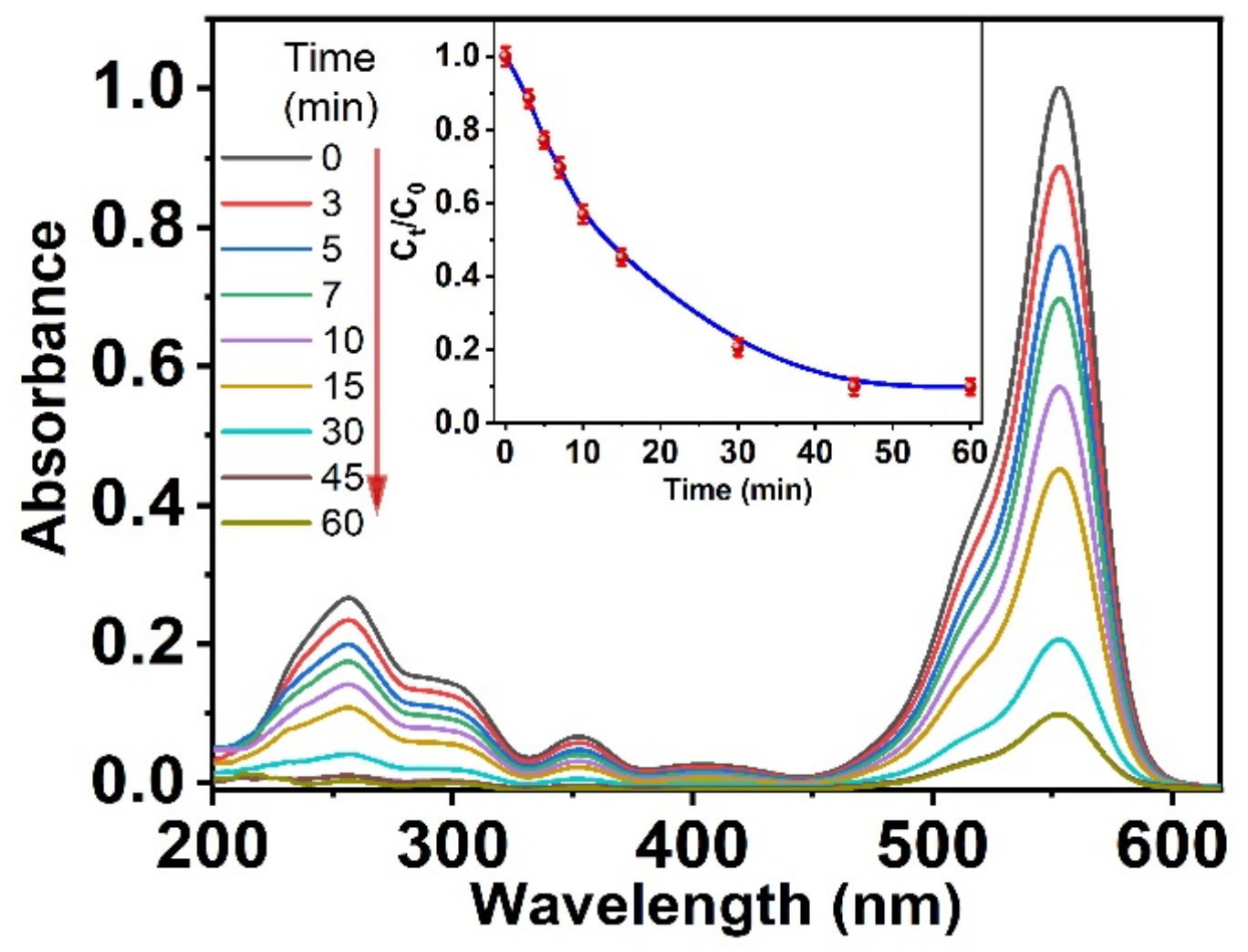
3.2.1. Absorption Spectra Experiments
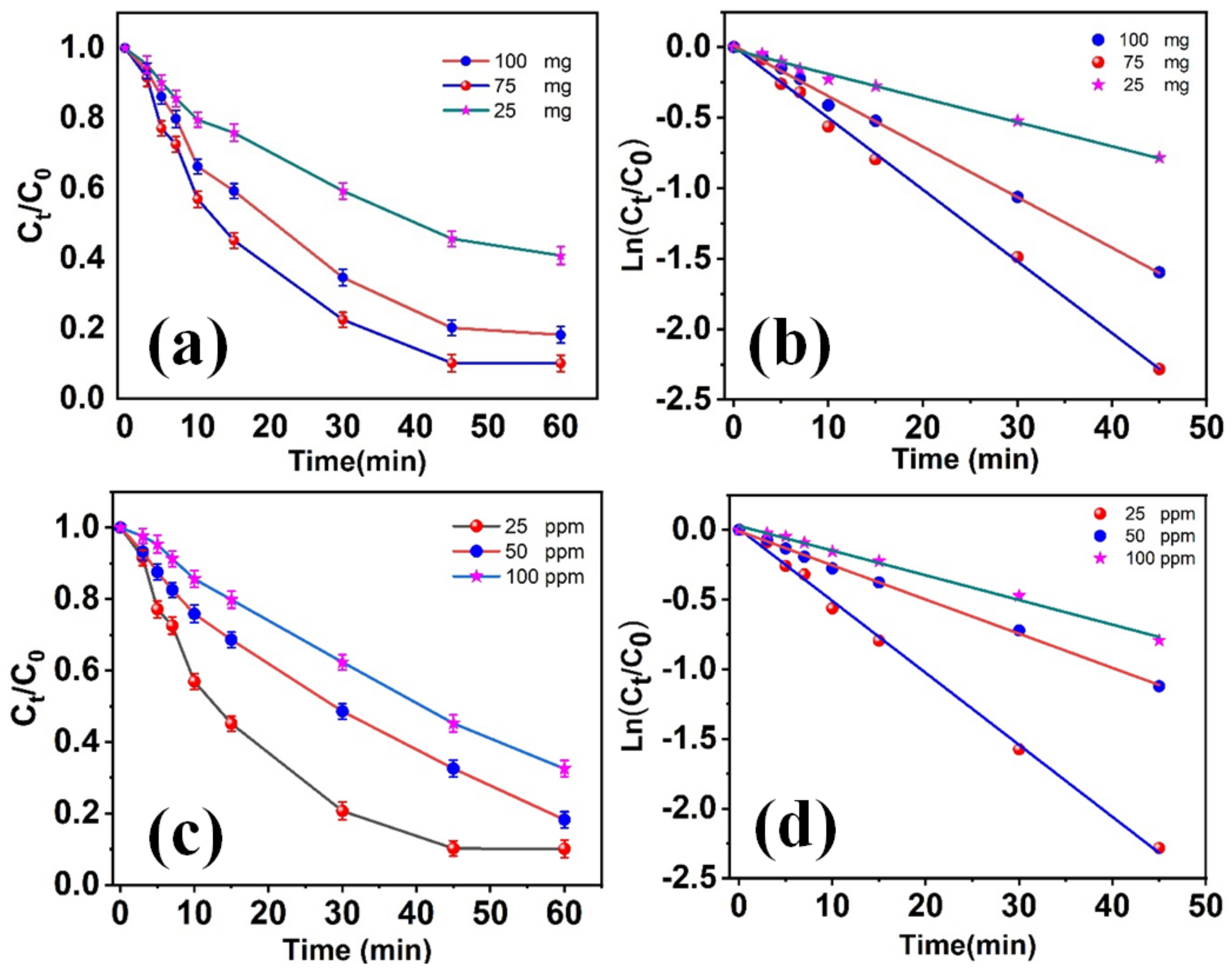
3.2.2. Effect of the Nanomaterials’ Dosage and RB Content on Photodegradation
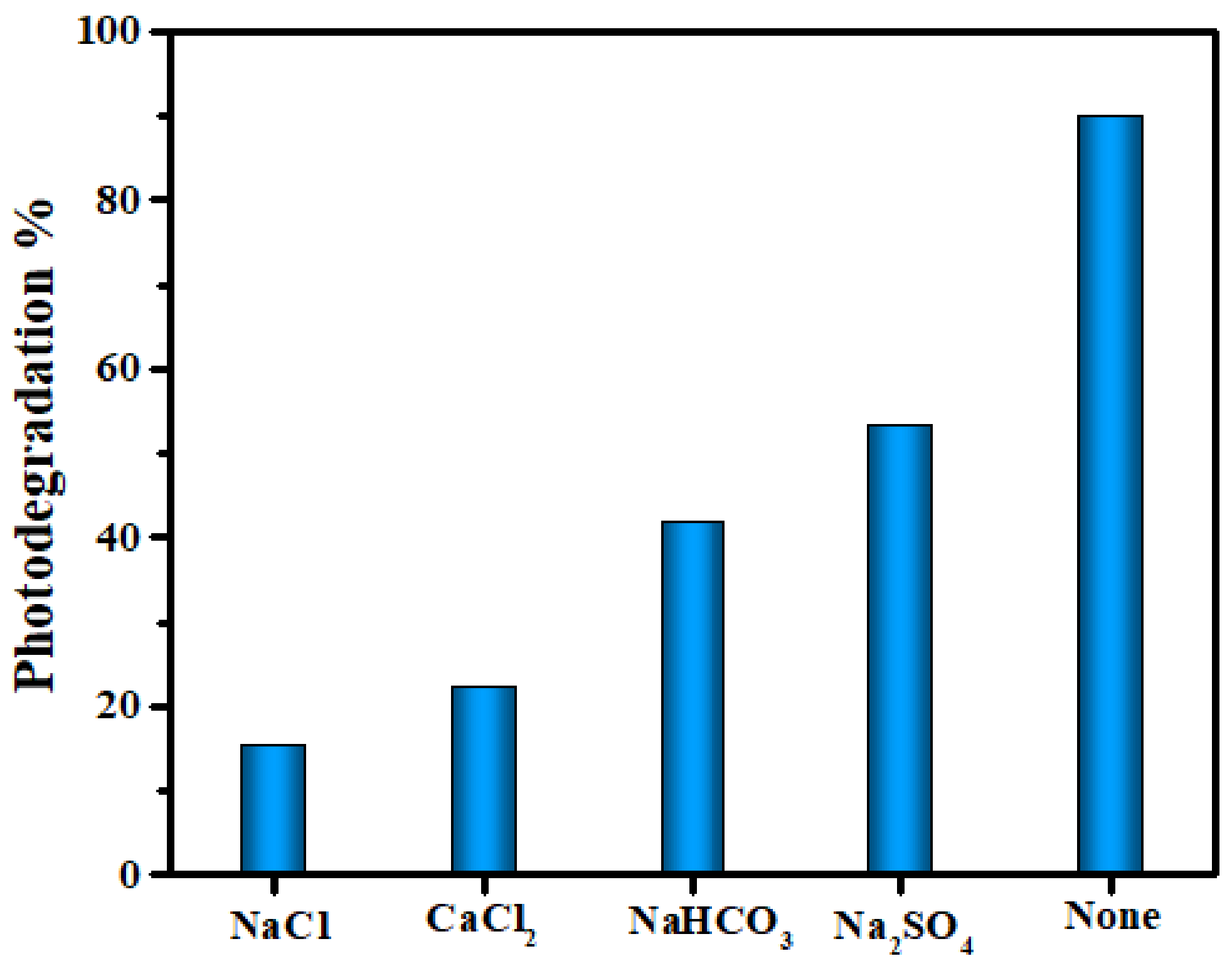
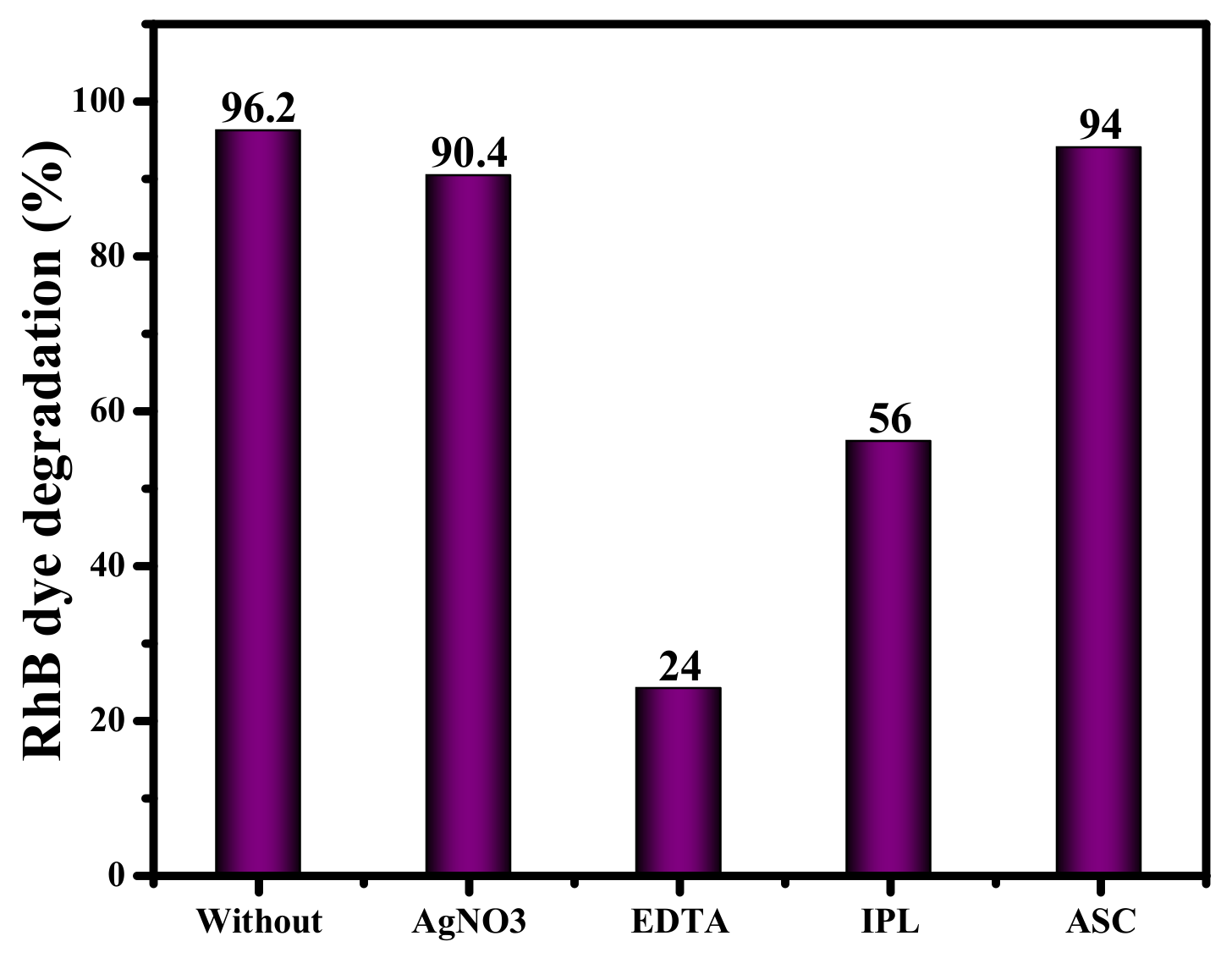
3.2.3. Effect of Inorganic Ions
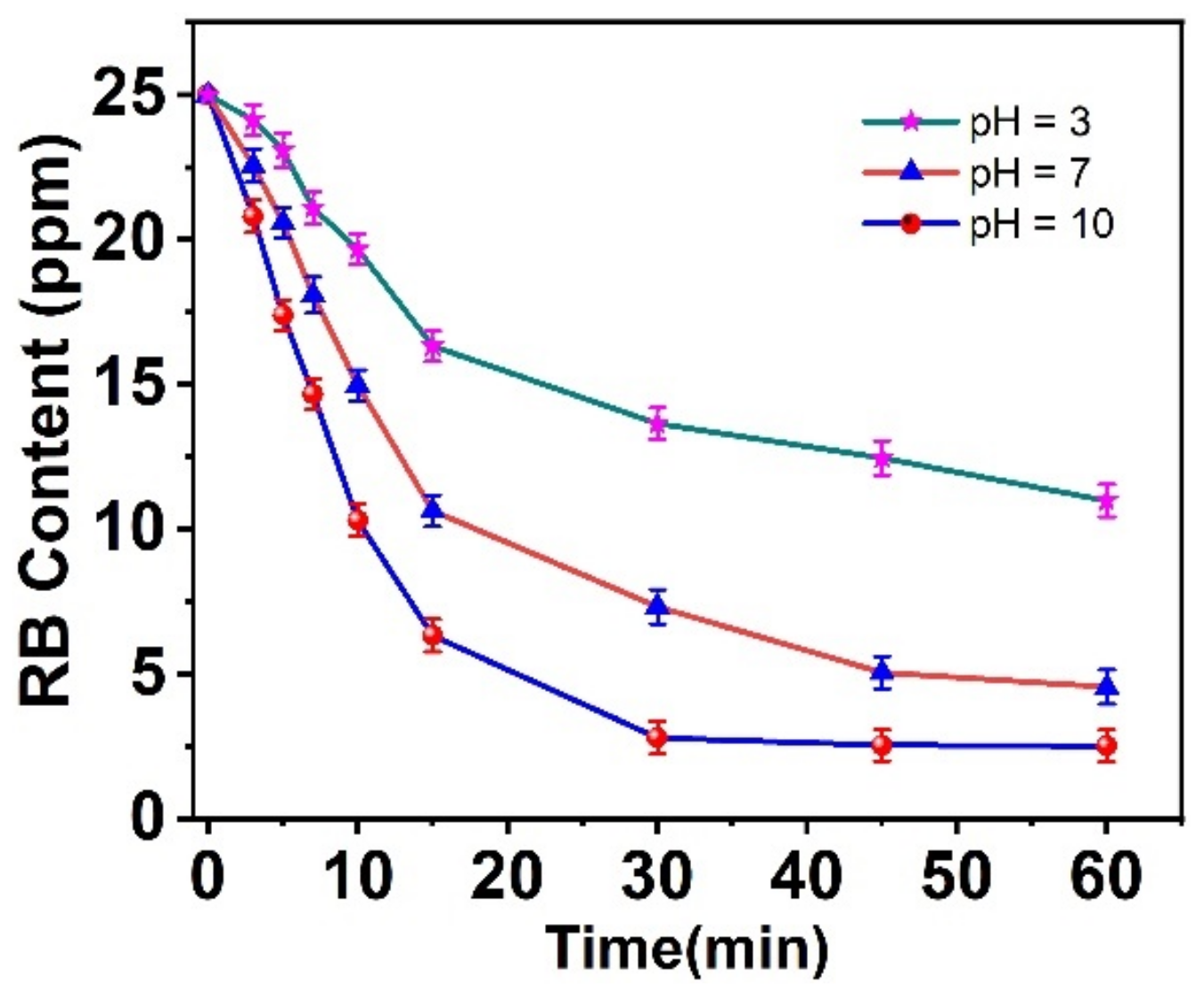
3.2.4. pH Influence on RB Dye Degradation
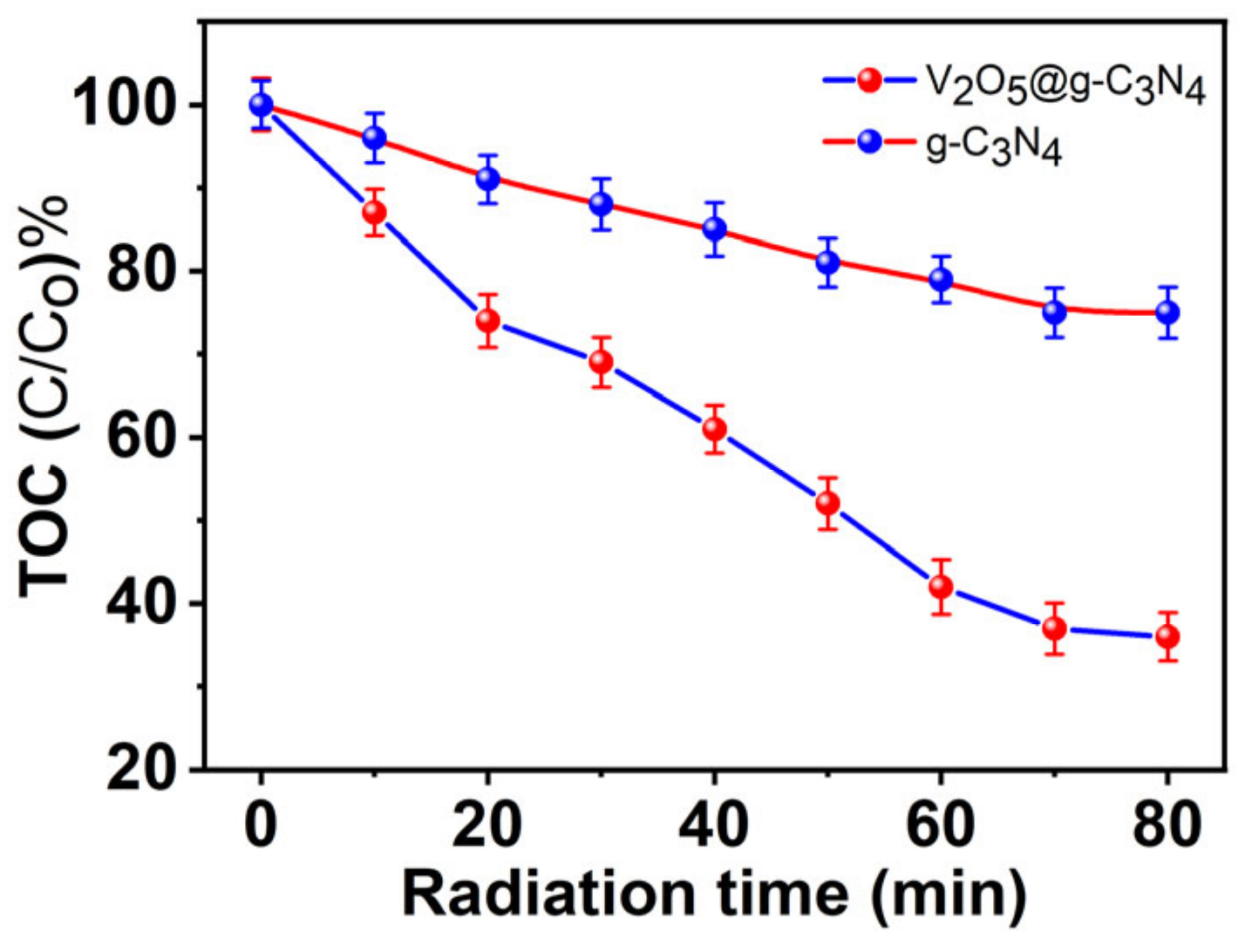
3.2.5. TOC Measurements
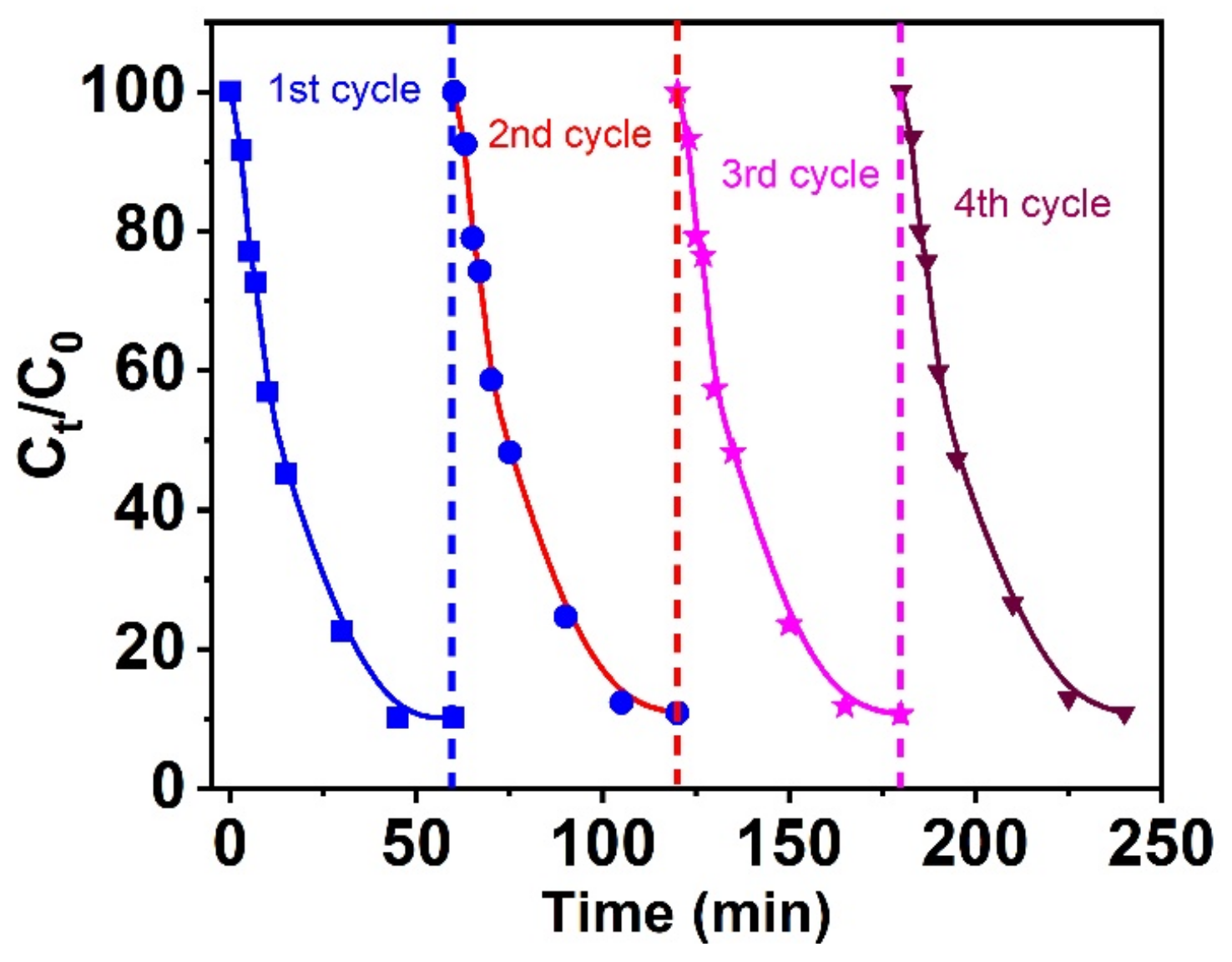
3.2.6. Nano-Catalysts Reused Study
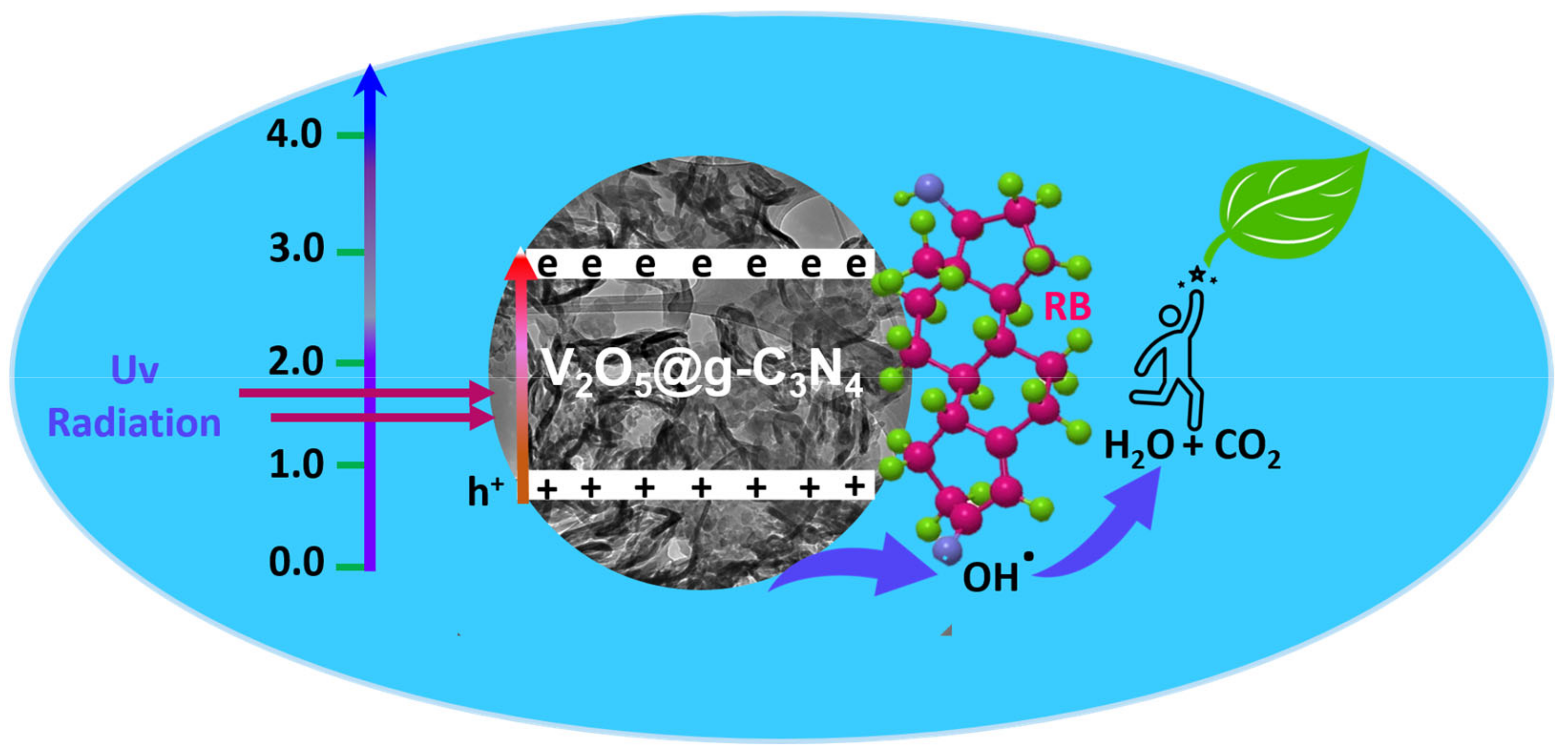
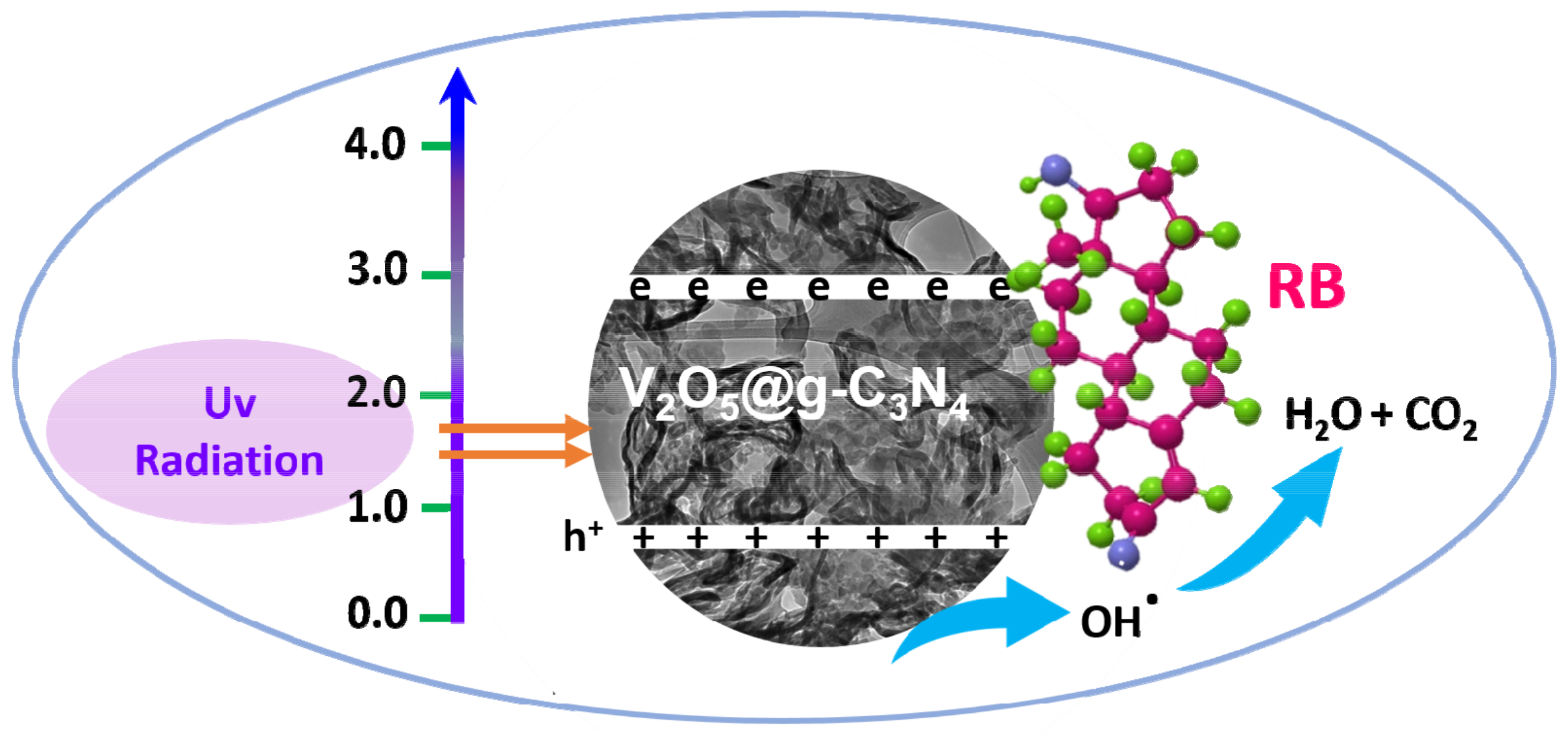
3.2.7. Photocatalytic Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Toghan, A.; Modwi, A. Boosting unprecedented indigo carmine dye photodegradation via mesoporous MgO@ g-C3N4 nanocomposite. J. Photochem. Photobiol. A Chem. 2021, 419, 113467. [Google Scholar] [CrossRef]
- Hu, H.; Xin, J.H.; Hu, H.; Wang, X.; Miao, D.; Liu, Y. Synthesis and stabilization of metal nanocatalysts for reduction reactions—A review. J. Mater. Chem. A 2015, 3, 11157–11182. [Google Scholar] [CrossRef]
- Ghosh, B.K.; Ghosh, N.N. Applications of metal nanoparticles as catalysts in cleaning dyes containing industrial effluents: A review. J. Nanosci. Nanotechnol. 2018, 18, 3735–3758. [Google Scholar] [CrossRef]
- Gogoi, D.; Makkar, P.; Ghosh, N.N. Solar light-irradiated photocatalytic degradation of model dyes and industrial dyes by a magnetic CoFe2O4–gC3N4 S-scheme heterojunction photocatalyst. ACS Omega 2021, 6, 4831–4841. [Google Scholar] [CrossRef] [PubMed]
- Manu, B.; Chaudhari, S. Anaerobic decolorisation of simulated textile wastewater containing azo dyes. Bioresour. Technol. 2002, 82, 225–231. [Google Scholar] [CrossRef]
- Patel, R.; Suresh, S. Decolourization of azo dyes using magnesium–palladium system. J. Hazard. Mater. 2006, 137, 1729–1741. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Tang, J.; Shi, Y.; Wang, H.; Jiang, X. Improved in situ synthesis of heterostructured 2D/2D BiOCl/g-C3N4 with enhanced dye photodegradation under visible-light illumination. ACS Omega 2019, 4, 22187–22196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.-J.; Qi, P.; Li, J.; Zheng, X.-C.; Liu, P.; Guan, X.-X.; Zheng, G.-P. Three-dimensional Fe2O3–TiO2–graphene aerogel nanocomposites with enhanced adsorption and visible light-driven photocatalytic performance in the removal of RhB dyes. J. Ind. Eng. Chem. 2018, 61, 407–415. [Google Scholar] [CrossRef]
- Rather, M.A.; Bhat, S.A.; Pandit, S.A.; Bhat, F.A.; Rather, G.M.; Bhat, M.A. As catalytic as silver nanoparticles anchored to reduced graphene oxide: Fascinating activity of imidazolium based surface active ionic liquid for chemical degradation of rhodamine B. Catal. Lett. 2019, 149, 2195–2203. [Google Scholar] [CrossRef]
- Sang, Y.; Cao, X.; Dai, G.; Wang, L.; Peng, Y.; Geng, B. Facile one-pot synthesis of novel hierarchical Bi2O3/Bi2S3 nanoflower photocatalyst with intrinsic pn junction for efficient photocatalytic removals of RhB and Cr (VI). J. Hazard. Mater. 2020, 381, 120942. [Google Scholar] [CrossRef]
- Khan, M.S.; Khalid, M.; Shahid, M. A Co (II) coordination polymer derived from pentaerythritol as an efficient photocatalyst for the degradation of organic dyes. Polyhedron 2021, 196, 114984. [Google Scholar] [CrossRef]
- Refat, M.S.; Saad, H.A.; Gobouri, A.A.; Alsawat, M.; Adam, A.M.A.; Shakya, S.; Gaber, A.; Alsuhaibani, A.M.; El-Megharbel, S.M. Synthesis and spectroscopic characterizations of nanostructured charge transfer complexes associated between moxifloxacin drug donor and metal chloride acceptors as a catalytic agent in a recycling of wastewater. J. Mol. Liq. 2022, 349, 118121. [Google Scholar] [CrossRef]
- Alminderej, F.M.; Younis, A.M.; Albadri, A.E.; El-Sayed, W.A.; El-Ghoul, Y.; Ali, R.; Mohamed, A.M.A.; Saleh, S.M. The superior adsorption capacity of phenol from aqueous solution using Modified Date Palm Nanomaterials: A performance and kinetic study. Arab. J. Chem. 2022, 15, 104120. [Google Scholar] [CrossRef]
- Saleh, S.; Younis, A.; Ali, R.; Elkady, E. Phenol removal from aqueous solution using amino modified silica nanoparticles. Korean J. Chem. Eng. 2019, 36, 529–539. [Google Scholar] [CrossRef]
- Khezami, L.; Ben Aissa, M.A.; Modwi, A.; Guesmi, A.; Algethami, F.K.; Bououdina, M. Efficient removal of organic dyes by Cr-doped ZnO nanoparticles. Biomass Convers. Biorefinery 2022, 2022, 1–14. [Google Scholar] [CrossRef]
- Modwi, A.; Khezami, L.; Ghoniem, M.G.; Nguyen-Tri, P.; Baaloudj, O.; Guesmi, A.; AlGethami, F.K.; Amer, M.S.; Assadi, A.A. Superior removal of dyes by mesoporous MgO/g-C3N4 fabricated through ultrasound method: Adsorption mechanism and process modeling. Environ. Res. 2022, 205, 112543. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V. Application of low-cost adsorbents for dye removal—A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef]
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Divyapriya, G.; Nambi, I.M.; Senthilnathan, J. Nanocatalysts in Fenton based advanced oxidation process for water and wastewater treatment. J. Bionanosci. 2016, 10, 356–368. [Google Scholar] [CrossRef]
- Khan, M.S.; Shahid, M. Improving Water Quality Using Metal−Organic Frameworks. In Metal-Organic Frameworks for Environmental Remediation; American Chemical Society: New York, NY, USA, 2021; pp. 171–191. [Google Scholar]
- Khan, M.S.; Khalid, M.; Ahmad, M.S.; Kamal, S.; Shahid, M. Effect of structural variation on enzymatic activity in tetranuclear (Cu4) clusters with defective cubane core. J. Biomol. Struct. Dyn. 2021, 1–14. [Google Scholar] [CrossRef]
- Younis, A.M.; Elkady, E.M.; Saleh, S.M. Novel eco-friendly amino-modified nanoparticles for phenol removal from aqueous solution. Environ. Sci. Pollut. Res. 2020, 27, 30694–30705. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, M.; Kakkar, R. Facile synthesis of mesoporous magnesium oxide–graphene oxide composite for efficient and highly selective adsorption of hazardous anionic dyes. Res. Chem. Intermed. 2020, 46, 2497–2521. [Google Scholar] [CrossRef]
- Yu, Y.; Hu, X.; Li, M.; Fang, J.; Leng, C.; Zhu, X.; Xu, W.; Qin, J.; Yao, L.; Liu, Z.; et al. Constructing mesoporous Zr-doped SiO2 onto efficient Z-scheme TiO2/g-C3N4 heterojunction for antibiotic degradation via adsorption-photocatalysis and mechanism insight. Environ. Res. 2022, 214, 114189. [Google Scholar] [CrossRef]
- Du, J.; Ma, S.; Zhang, N.; Liu, W.; Lv, M.; Ni, T.; An, Z.; Li, K.; Bai, Y. Efficient photocatalytic organic degradation and disinfection performance for Ag/AgFeO2/g-C3N4 nanocomposites under visible-light: Insights into the photocatalysis mechanism. Colloids Surf. A Physicochem. Eng. Asp. 2022, 654, 130094. [Google Scholar] [CrossRef]
- Huang, X.; Xu, X.; Yang, R.; Fu, X. Synergetic adsorption and photocatalysis performance of g-C3N4/Ce-doped MgAl-LDH in degradation of organic dye under LED visible light. Colloids Surf. A Physicochem. Eng. Asp. 2022, 643, 128738. [Google Scholar] [CrossRef]
- Zhou, G.; Meng, L.; Ning, X.; Yin, W.; Hou, J.; Xu, Q.; Yi, J.; Wang, S.; Wang, X. Switching charge transfer of g-C3N4/BiVO4 heterojunction from type II to Z-scheme via interfacial vacancy engineering for improved photocatalysis. Int. J. Hydrog. Energy 2022, 47, 8749–8760. [Google Scholar] [CrossRef]
- Yang, Z.; Xing, Z.; Feng, Q.; Jiang, H.; Zhang, J.; Xiao, Y.; Li, Z.; Chen, P.; Zhou, W. Sandwich-like mesoporous graphite-like carbon nitride (Meso-g-C3N4)/WP/Meso-g-C3N4 laminated heterojunctions solar-driven photocatalysts. J. Colloid Interface Sci. 2020, 568, 255–263. [Google Scholar] [CrossRef]
- Vellaichamy, B.; Paulmony, T. Visible light active metal-free photocatalysis: N-doped graphene covalently grafted with g-C3N4 for highly robust degradation of methyl orange. Solid State Sci. 2019, 94, 99–105. [Google Scholar]
- Zhang, C.; Fu, Z.; Hong, F.; Pang, G.; Dong, T.; Zhang, Y.; Liu, G.; Dong, X.; Wang, J. Non-metal group doped g-C3N4 combining with BiF3: Yb3+, Er3+ upconversion nanoparticles for photocatalysis in UV–Vis–NIR region. Colloids Surf. A Physicochem. Eng. Asp. 2021, 627, 127180. [Google Scholar] [CrossRef]
- Shen, J.-H.; Chiang, T.H.; Tsai, C.K.; Jiang, Z.W.; Horng, J.J. Mechanistic insights into hydroxyl radical formation of Cu-doped ZnO/g-C3N4 composite photocatalysis for enhanced degradation of ciprofloxacin under visible light: Efficiency, kinetics, products identification and toxicity evaluation. J. Environ. Chem. Eng. 2022, 10, 107352. [Google Scholar] [CrossRef]
- Yu, X.; Yin, W.; Wang, T.; Zhang, Y. Decorating g-C3N4 nanosheets with Ti3C2 MXene nanoparticles for efficient oxygen reduction reaction. Langmuir 2019, 35, 2909–2916. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, N.A.; Ullah, H.; Safaei, J.; Ismail, A.F.; Mohamad Noh, M.F.; Soh, M.F.; Ibrahim, M.A.; Ludin, N.A.; Teridi, M.A.M. Efficient photoelectrochemical performance of γ irradiated g-C3N4 and its g-C3N4@ BiVO4 heterojunction for solar water splitting. J. Phys. Chem. C 2019, 123, 9013–9026. [Google Scholar] [CrossRef] [Green Version]
- Rawool, S.A.; Samanta, A.; Ajithkumar, T.G.; Kar, Y.; Polshettiwar, V. Photocatalytic hydrogen generation and CO2 conversion using g-C3N4 decorated dendritic fibrous nanosilica: Role of interfaces between silica and g-C3N4. ACS Appl. Energy Mater. 2020, 3, 8150–8158. [Google Scholar] [CrossRef]
- Cao, S.; Yu, J. g-C3N4-based photocatalysts for hydrogen generation. J. Phys. Chem. Lett. 2014, 5, 2101–2107. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Wang, L.; Zong, R.; Zhu, Y. Photocatalytic activity enhanced via g-C3N4 nanoplates to nanorods. J. Phys. Chem. C 2013, 117, 9952–9961. [Google Scholar] [CrossRef]
- Zou, H.; Xiao, G.; Chen, K.; Peng, X. Noble metal-free V 2 O 5/gC 3 N 4 composites for selective oxidation of olefins using hydrogen peroxide as an oxidant. Dalton Trans. 2018, 47, 13565–13572. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, D.; Uma Shankar, V.; Gopalakrishnan, R. Supercapacitor behavior and characterization of RGO anchored V2O5 nanorods. J. Mater. Sci. Mater. Electron. 2019, 30, 16142–16155. [Google Scholar] [CrossRef]
- Khezami, L.; Aissa, M.A.B.; Modwi, A.; Ismail, M.; Guesmi, A.; Algethami, F.K.; Ticha, M.B.; Assadi, A.A.; Nguyen-Tri, P. Harmonizing the photocatalytic activity of g-C3N4 nanosheets by ZrO2 stuffing: From fabrication to experimental study for the wastewater treatment. Biochem. Eng. J. 2022, 182, 108411. [Google Scholar] [CrossRef]
- Shafique, S.; Yang, S.; Iqbal, T.; Cheng, B.; Wang, Y.; Sarwar, H.; Woldu, Y.T.; Ji, P. Improving the performance of V2O5/rGO hybrid nanocomposites for photodetector applications. Sens. Actuators A Phys. 2021, 332, 113073. [Google Scholar] [CrossRef]
- Modwi, A.; Abbo, M.A.; Hassan, E.A.; Taha, K.K.; Khezami, L.; Houas, A. Influence of Annealing Temperature on the Properties of ZnO Synthesized Via 2.3. Dihydroxysuccinic Acid Using Flash Sol-Gel Method. J. Ovonic Res. 2016, 12, 59–66. [Google Scholar]
- Condon, J.B. Surface Area and Porosity Determinations by Physisorption: Measurements and Theory; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Rouquerol, J.; Rouquerol, F.; Llewellyn, P.; Maurin, G.; Sing, K.S. Adsorption by Powders and Porous Solids: Principles, Methodology and Applications; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Xiong, G.; Luo, L.; Li, C.; Yang, X. Synthesis of mesoporous ZnO (m-ZnO) and catalytic performance of the Pd/m-ZnO catalyst for methanol steam reforming. Energy Fuels 2009, 23, 1342–1346. [Google Scholar] [CrossRef]
- Dumeignil, F.; Sato, K.; Imamura, M.; Matsubayashi, N.; Payen, E.; Shimada, H. Modification of structural and acidic properties of sol–gel-prepared alumina powders by changing the hydrolysis ratio. Appl. Catal. A Gen. 2003, 241, 319–329. [Google Scholar] [CrossRef]
- Hao, R.; Wang, G.; Jiang, C.; Tang, H.; Xu, Q. In situ hydrothermal synthesis of g-C3N4/TiO2 heterojunction photocatalysts with high specific surface area for Rhodamine B degradation. Appl. Surf. Sci. 2017, 411, 400–410. [Google Scholar] [CrossRef]
- Li, Y.; Lv, K.; Ho, W.; Dong, F.; Wu, X.; Xia, Y. Hybridization of rutile TiO2 (rTiO2) with g-C3N4 quantum dots (CN QDs): An efficient visible-light-driven Z-scheme hybridized photocatalyst. Appl. Catal. B Environ. 2017, 202, 611–619. [Google Scholar] [CrossRef]
- Shawky, A.; Albukhari, S.M.; Amin, M.S.; Zaki, Z.I. Mesoporous V2O5/g-C3N4 nanocomposites for promoted mercury (II) ions reduction under visible light. J. Inorg. Organomet. Polym. Mater. 2021, 31, 4209–4221. [Google Scholar] [CrossRef]
- Fang, S.; Xia, Y.; Lv, K.; Li, Q.; Sun, J.; Li, M. Effect of carbon-dots modification on the structure and photocatalytic activity of g-C3N4. Appl. Catal. B Environ. 2016, 185, 225–232. [Google Scholar] [CrossRef]
- Ma, W.; Li, D.; Wen, B.; Ma, X.; Jiang, D.; Chen, M. Construction of novel Sr0. 4H1. 2Nb2O6·H2O/g-C3N4 heterojunction with enhanced visible light photocatalytic activity for hydrogen evolution. J. Colloid Interface Sci. 2018, 526, 451–458. [Google Scholar] [CrossRef]
- Shanmugam, M.; Alsalme, A.; Alghamdi, A.; Jayavel, R. Enhanced photocatalytic performance of the graphene-V2O5 nanocomposite in the degradation of methylene blue dye under direct sunlight. ACS Appl. Mater. Interfaces 2015, 7, 14905–14911. [Google Scholar] [CrossRef]
- Yan, C.; Liu, L. Sn-doped V2O5 nanoparticles as catalyst for fast removal of ammonia in air via PEC and PEC-MFC. Chem. Eng. J. 2020, 392, 123738. [Google Scholar] [CrossRef]
- Katowah, D.F.; Saleh, S.M.; Mohammed, G.I.; Alkayal, N.S.; Ali, R.; Hussein, M.A. Ultra-efficient hybrid material-based cross-linked PANI@ Cs-GO-OXS/CuO for the photocatalytic degradation of Rhodamine-B. J. Phys. Chem. Solids 2021, 157, 110208. [Google Scholar] [CrossRef]
- Saleh, S.M. ZnO nanospheres based simple hydrothermal route for photocatalytic degradation of azo dye. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 211, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Katowah, D.F.; Saleh, S.M.; Alqarni, S.A.; Ali, R.; Mohammed, G.I.; Hussein, M.A. Network structure-based decorated CPA@ CuO hybrid nanocomposite for methyl orange environmental remediation. Sci. Rep. 2021, 11, 5056. [Google Scholar] [CrossRef]
- Zhang, L.; Cheng, H.; Zong, R.; Zhu, Y. Photocorrosion suppression of ZnO nanoparticles via hybridization with graphite-like carbon and enhanced photocatalytic activity. J. Phys. Chem. C 2009, 113, 2368–2374. [Google Scholar] [CrossRef]
- Ahmad, M.; Ahmed, E.; Hong, Z.L.; Ahmed, W.; Elhissi, A.; Khalid, N.R. Photocatalytic, sonocatalytic and sonophotocatalytic degradation of Rhodamine B using ZnO/CNTs composites photocatalysts. Ultrason. Sonochemistry 2014, 21, 761–773. [Google Scholar] [CrossRef]
- Padovini, D.; Magdalena, A.; Capeli, R.; Longo, E.; Dalmaschio, C.; Chiquito, A.; Pontes, F. Synthesis and characterization of ZrO2@ SiO2 core-shell nanostructure as nanocatalyst: Application for environmental remediation of rhodamine B dye aqueous solution. Mater. Chem. Phys. 2019, 233, 1–8. [Google Scholar] [CrossRef]
- Rauf, M.; Meetani, M.; Hisaindee, S. An overview on the photocatalytic degradation of azo dyes in the presence of TiO2 doped with selective transition metals. Desalination 2011, 276, 13–27. [Google Scholar] [CrossRef]
- Lops, C.; Ancona, A.; Di Cesare, K.; Dumontel, B.; Garino, N.; Canavese, G.; Hérnandez, S.; Cauda, V. Sonophotocatalytic degradation mechanisms of Rhodamine B dye via radicals generation by micro-and nano-particles of ZnO. Appl. Catal. B Environ. 2019, 243, 629–640. [Google Scholar] [CrossRef] [PubMed]



| V2O5 Nanomaterials | Pure g-C3N4 | V2O5@g-C3N4 | |
|---|---|---|---|
| Energy gap (eV) | 2.84 | 2.27 | 2.41 |
| Crystallite size (nm) | 45.62 | 44.08 | 44.38 |
| Lattice parameters | a = 11.531Å, b = 4.377Å, and c = 3.564 Å | a = 6.237Å | a = 11.543Å, b = 4.382 Å, and c = 3.570 Å |
| d-spacing (Å) | 3.88 | 4.74 | 3.96 |
| BET surface area (m2/g) | 6.752 | 154 | 61.042 |
| Pore volume (cm3/g) | 0.057 | 0.912 | 0.353 |
| Pore radius (Å) | 17.10 | 27.8 | 18.46 |
| Sample | K (min−1) | t1/2 (min) | R2 | Degradation % | Energy Gap (ev) |
|---|---|---|---|---|---|
| g-C3-N4 | 0.0080 | 86.64 | 0.92 | 11.5 | 2.27 |
| V2O5 | 0.0026 | 266.6 | 0.95 | 31.6 | 2.84 |
| V2O5@g-C3N4 | 0.0517 | 13.41 | 0.99 | 93.4 | 2.41 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saleh, S.M.; Albadri, A.E.A.E.; Aissa, M.A.B.; Modwi, A. Fabrication of Mesoporous V2O5@g-C3N4 Nanocomposite as Photocatalyst for Dye Degradation. Crystals 2022, 12, 1766. https://doi.org/10.3390/cryst12121766
Saleh SM, Albadri AEAE, Aissa MAB, Modwi A. Fabrication of Mesoporous V2O5@g-C3N4 Nanocomposite as Photocatalyst for Dye Degradation. Crystals. 2022; 12(12):1766. https://doi.org/10.3390/cryst12121766
Chicago/Turabian StyleSaleh, Sayed M., Abuzar E. A. E. Albadri, Mohamed Ali Ben Aissa, and Abueliz Modwi. 2022. "Fabrication of Mesoporous V2O5@g-C3N4 Nanocomposite as Photocatalyst for Dye Degradation" Crystals 12, no. 12: 1766. https://doi.org/10.3390/cryst12121766







