Time Dependent Lyotropic Chromonic Textures in Microfluidic Confinements
Abstract
1. Introduction
2. Materials and Methods
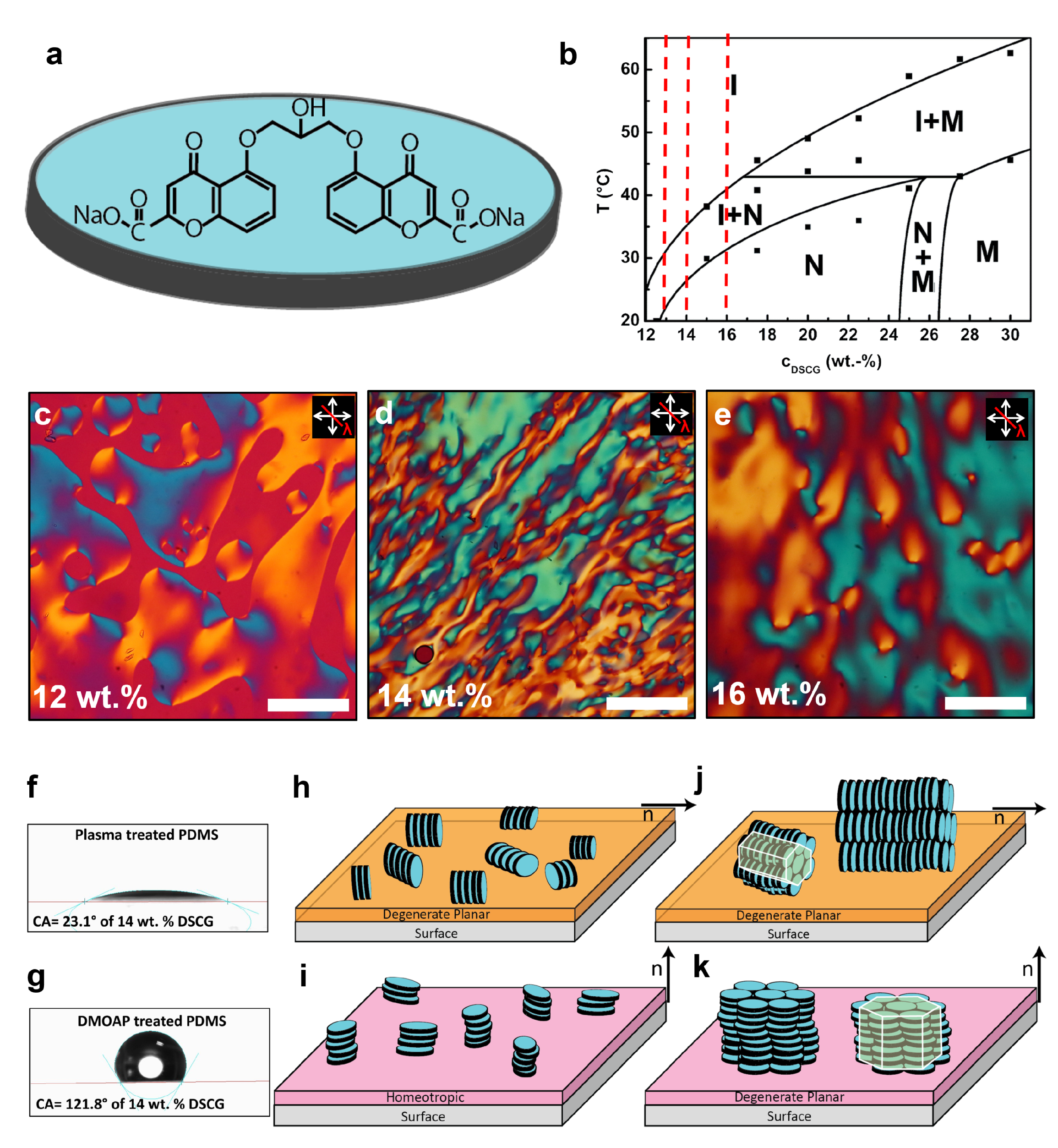
2.1. Materials and Solution Preparation
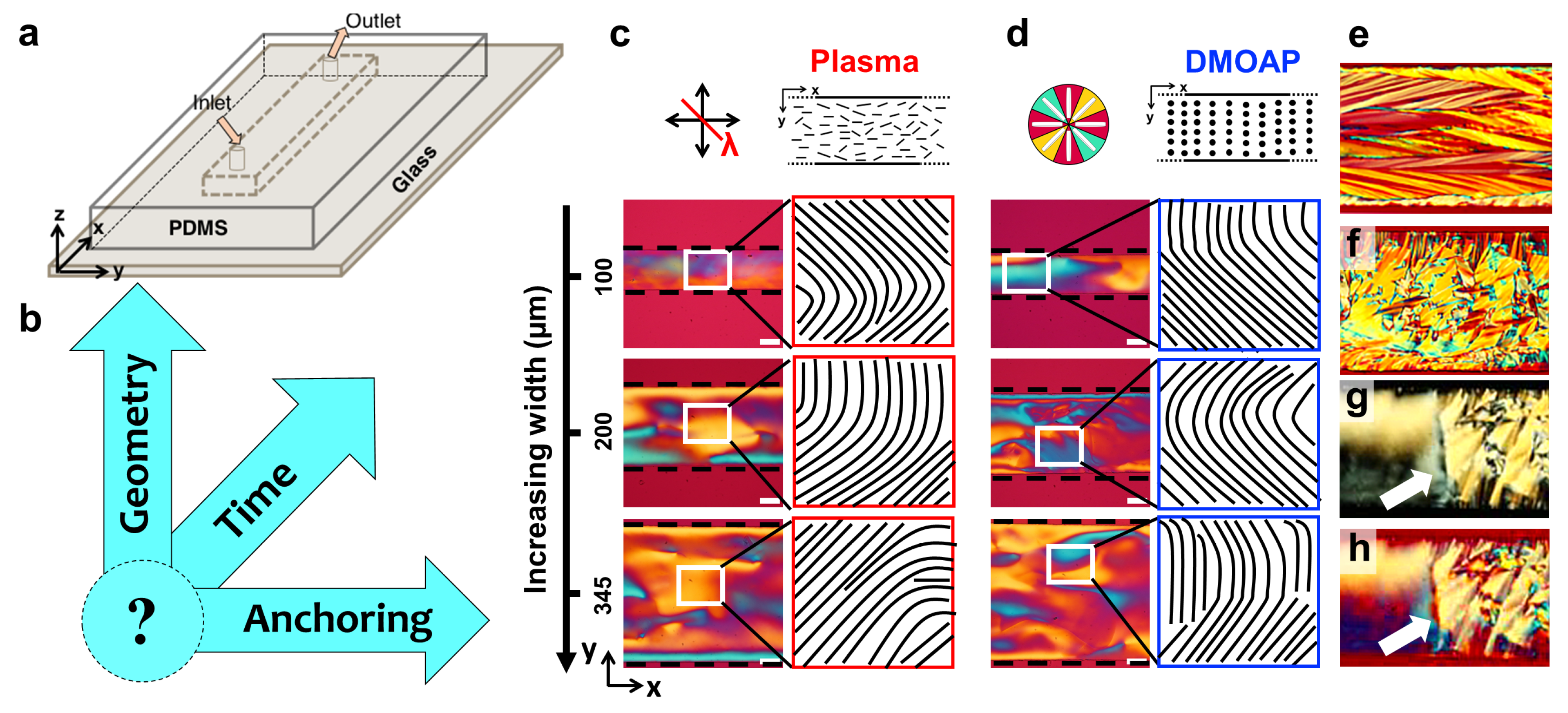
2.2. Microfluidic Confinement
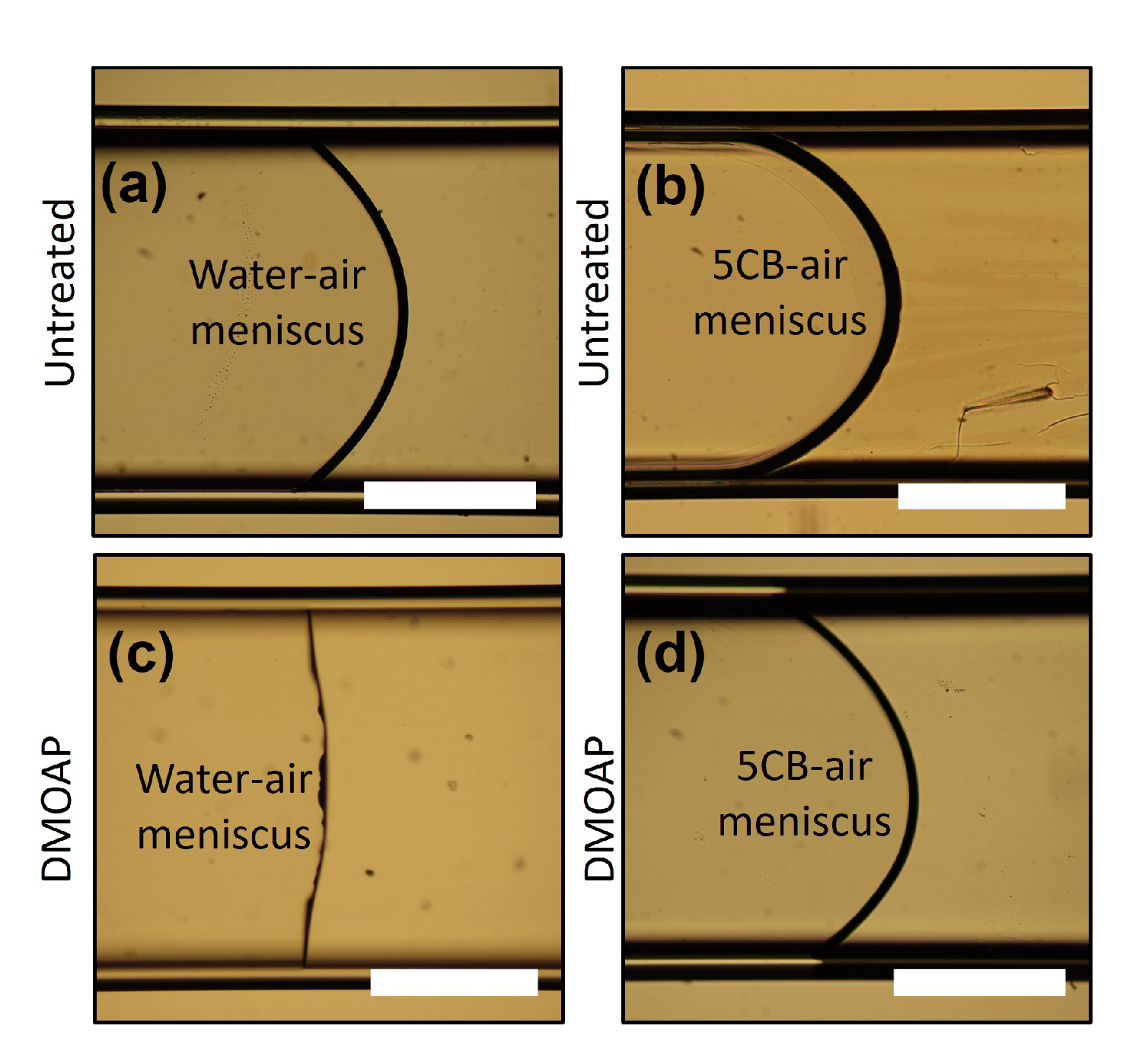
2.3. Surface Anchoring and Boundary Conditions
2.4. Filling the Microfluidic Devices
2.5. Optical Microscopy and Image Acquisition
3. Results and Discussion
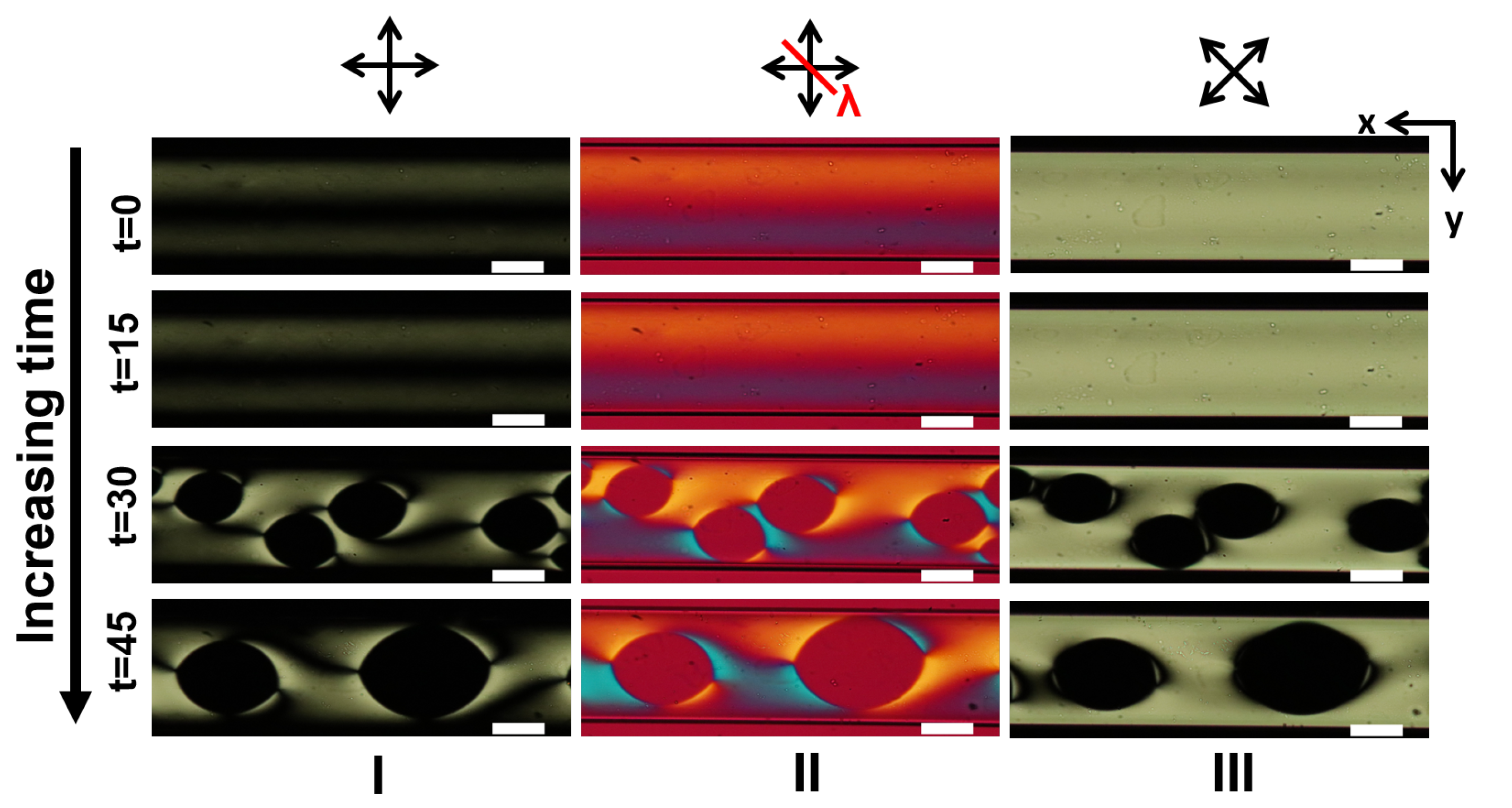
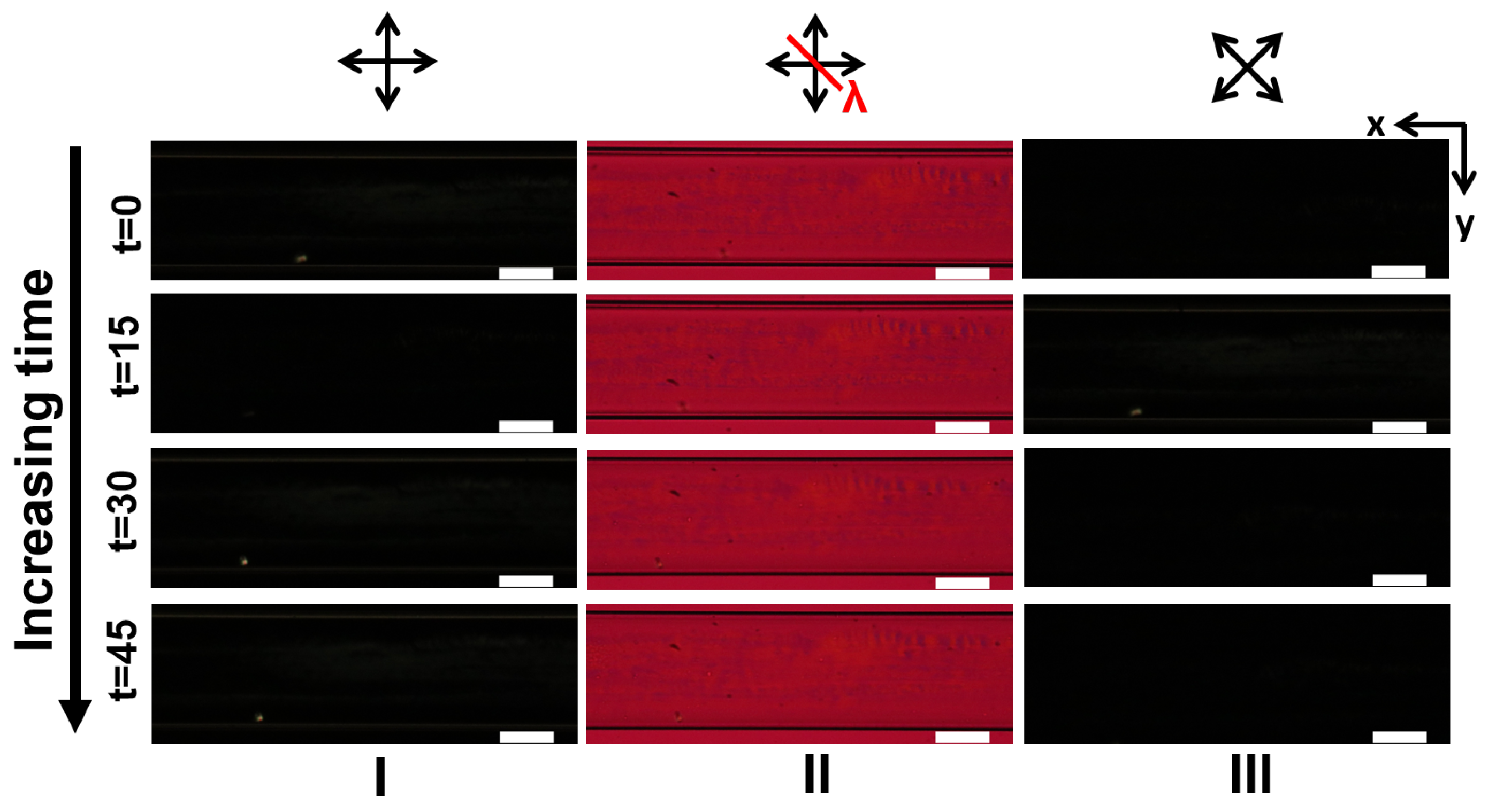
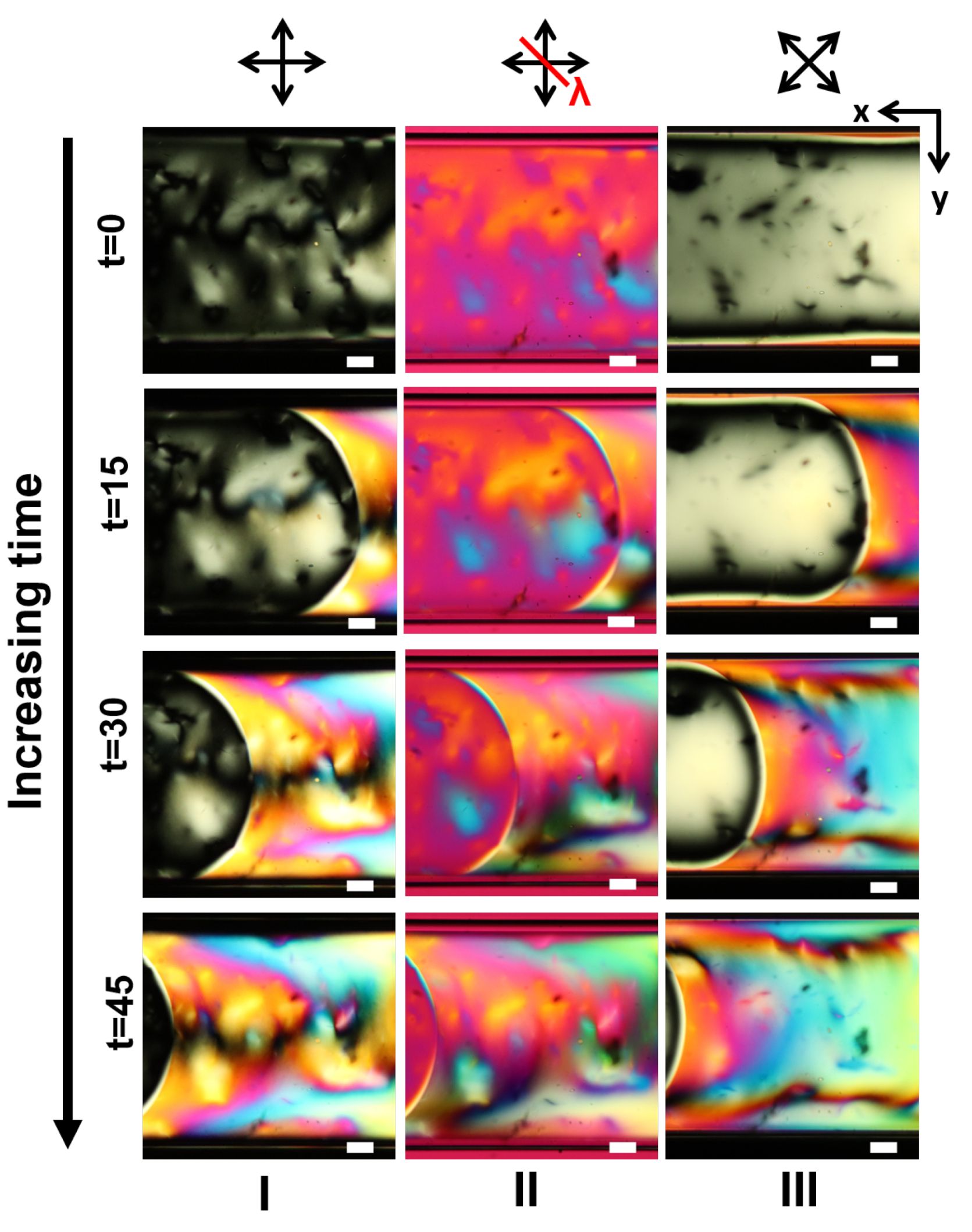
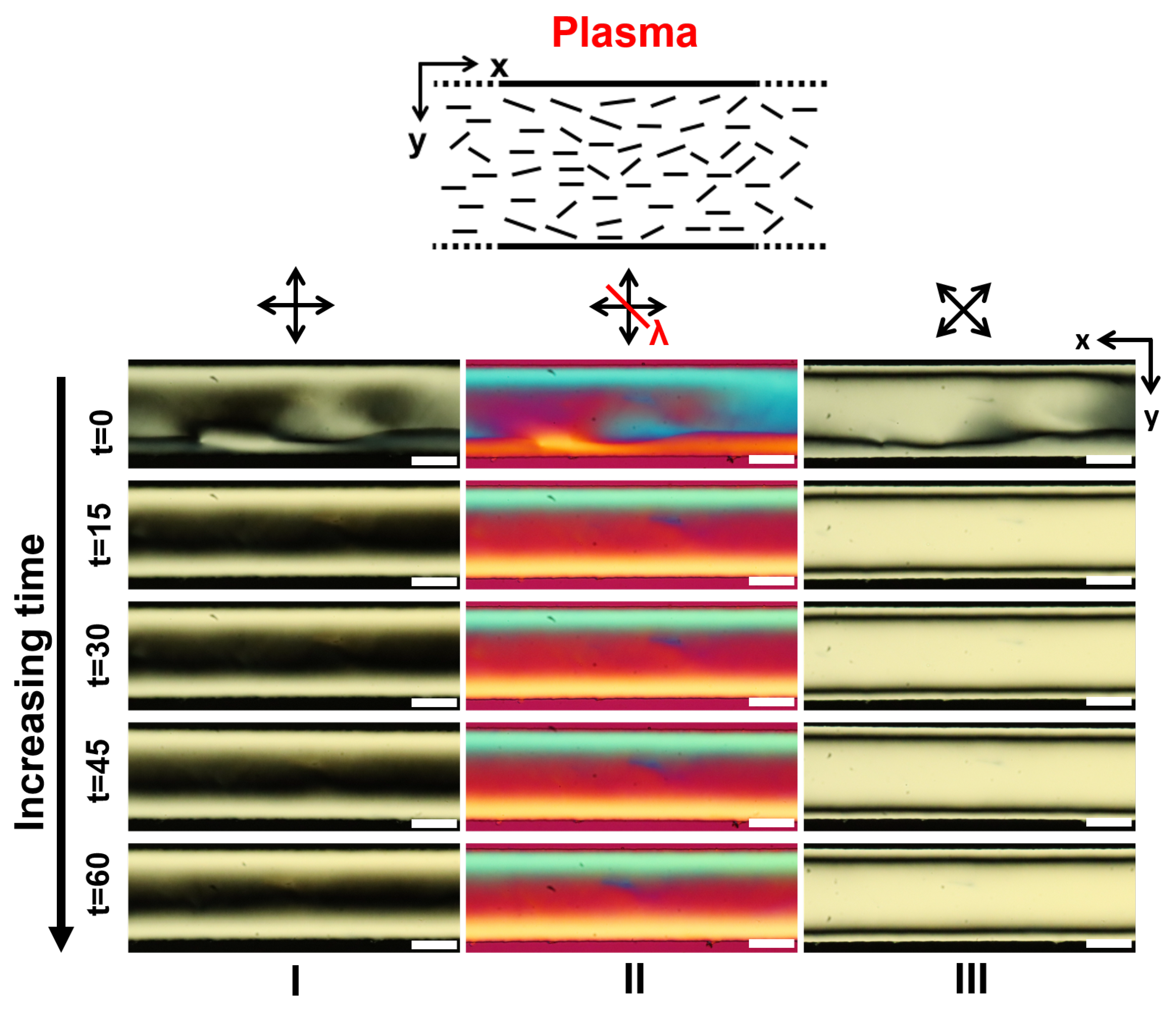
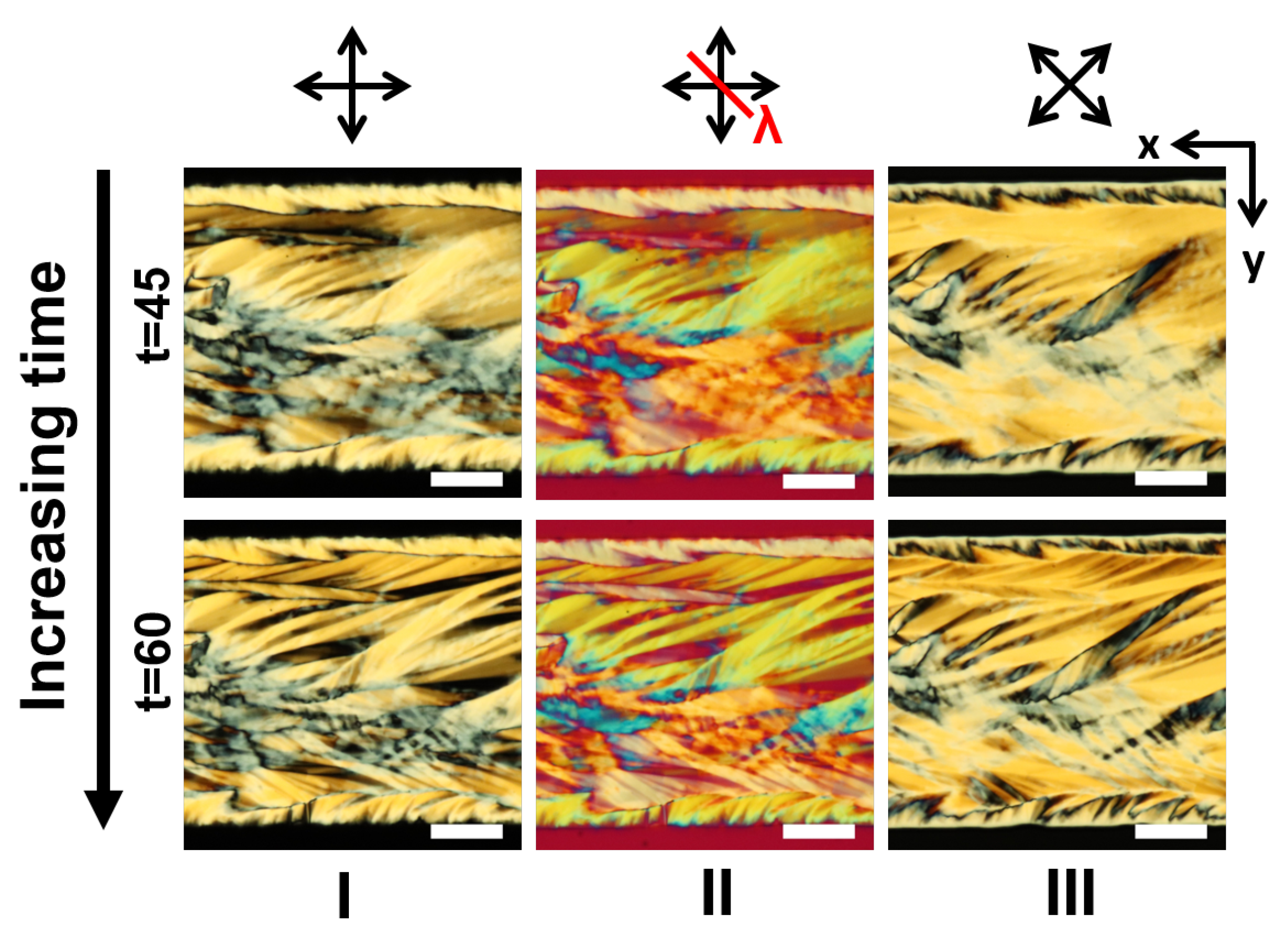
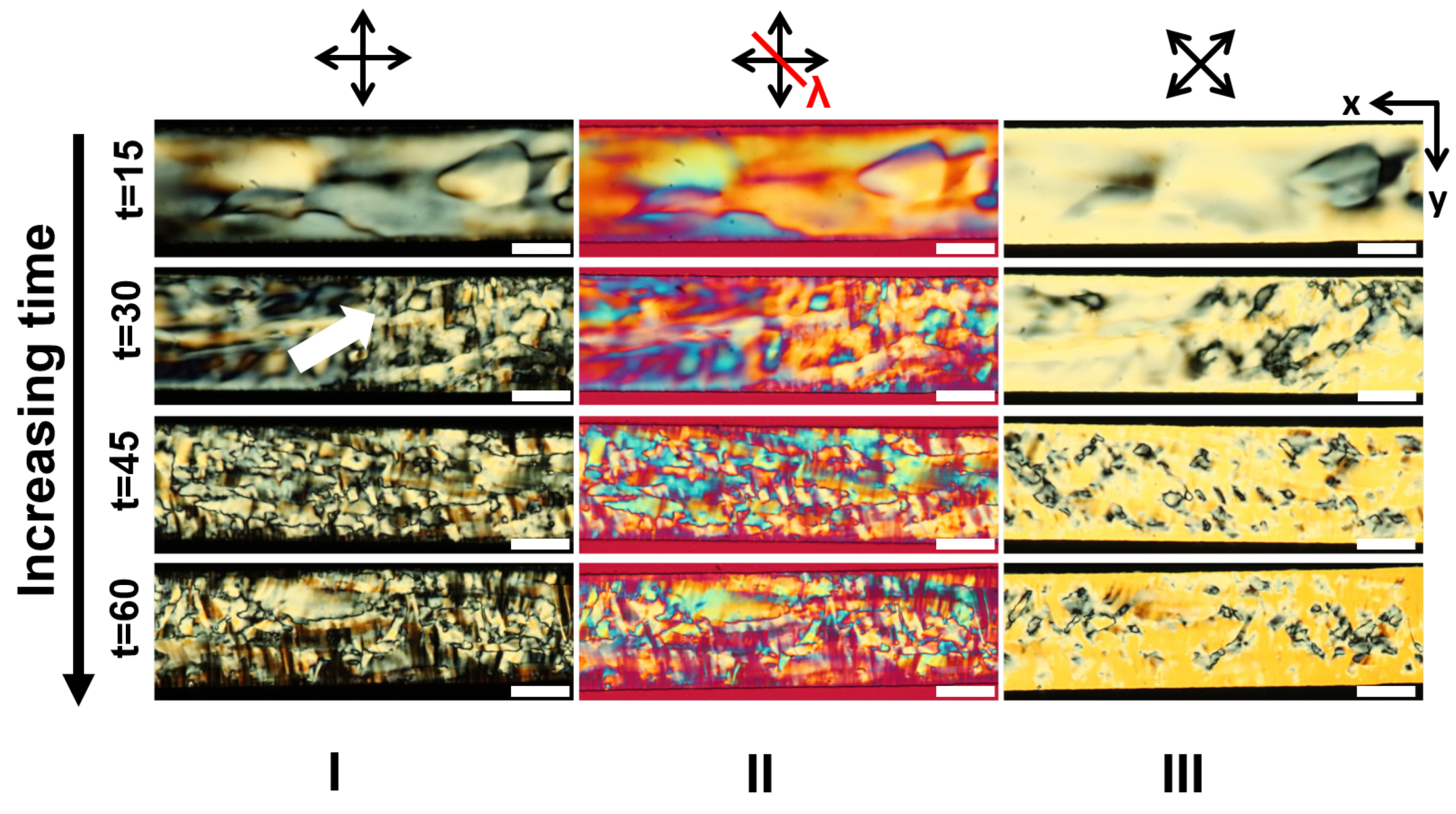
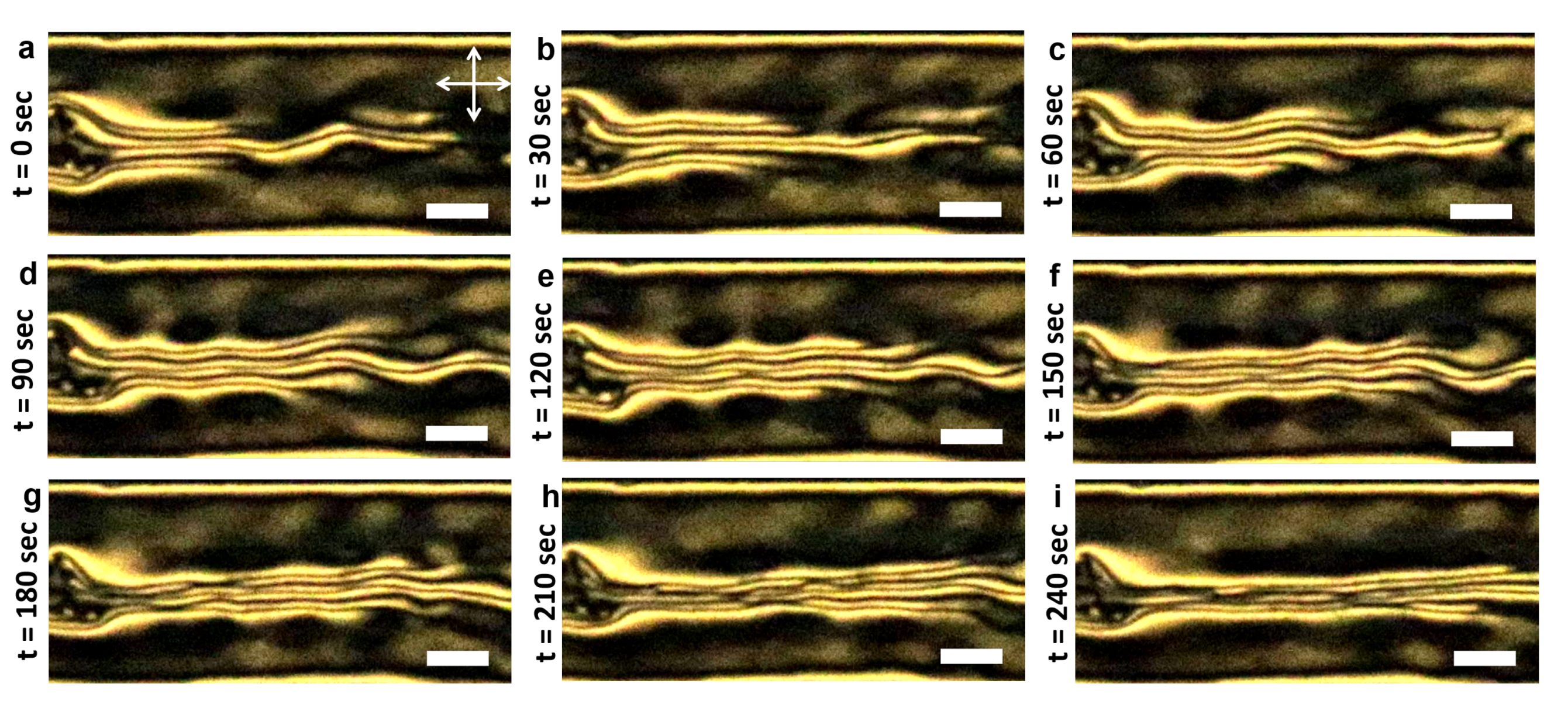
3.1. LCLC Microfluidic Textures Due to Degenerate Planar Anchoring
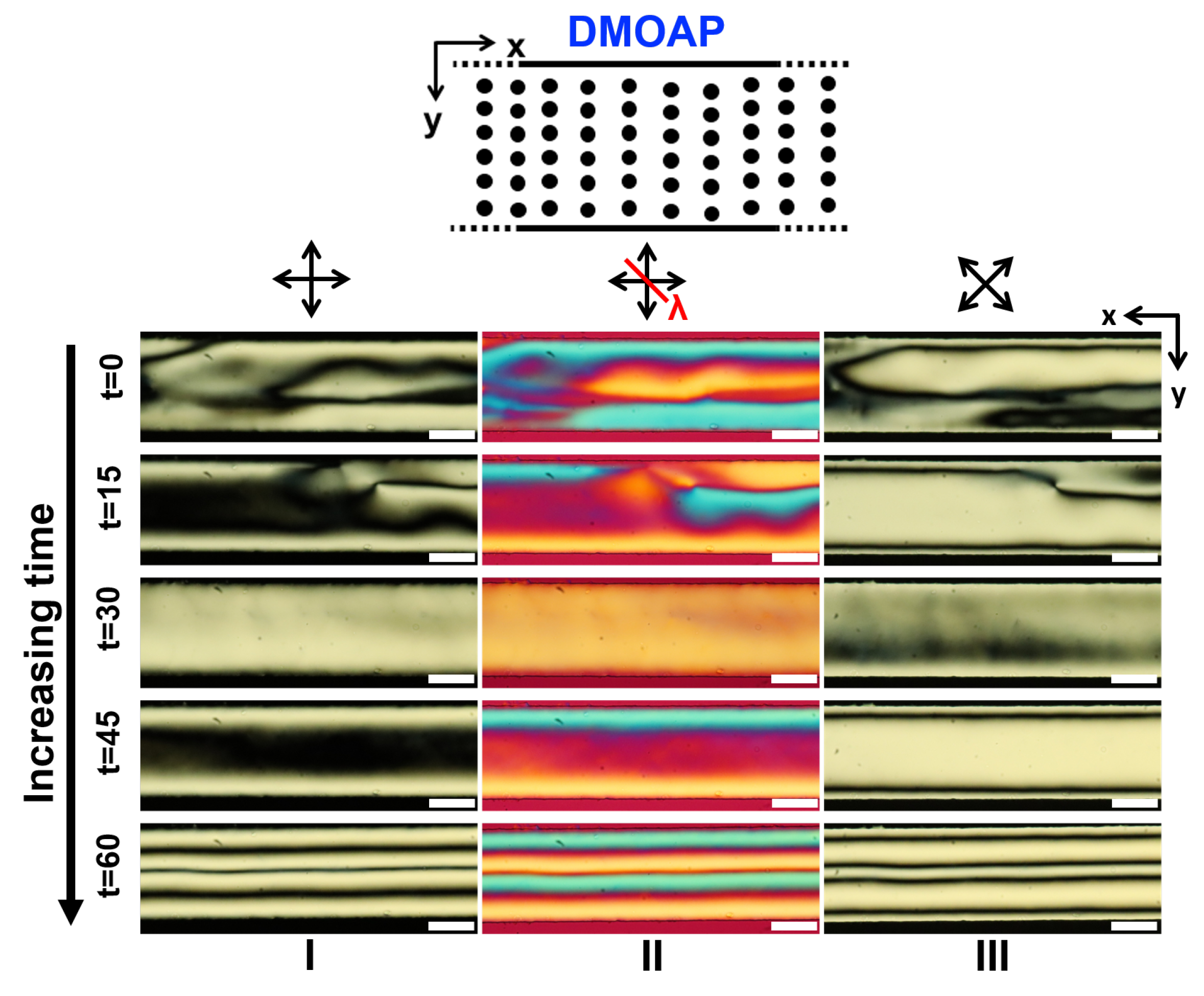
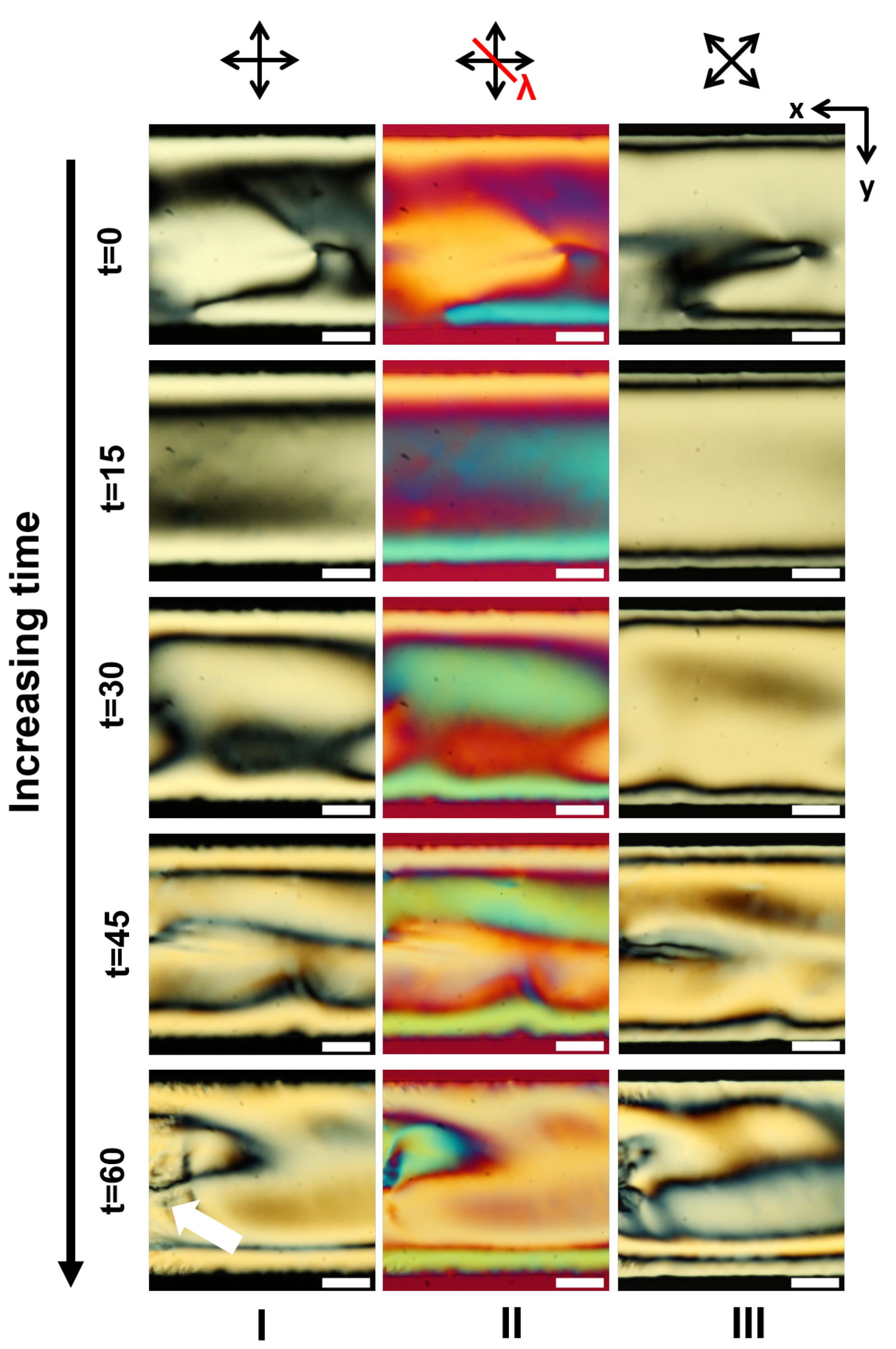
3.2. LCLC Texture in Homeotropic Microchannels
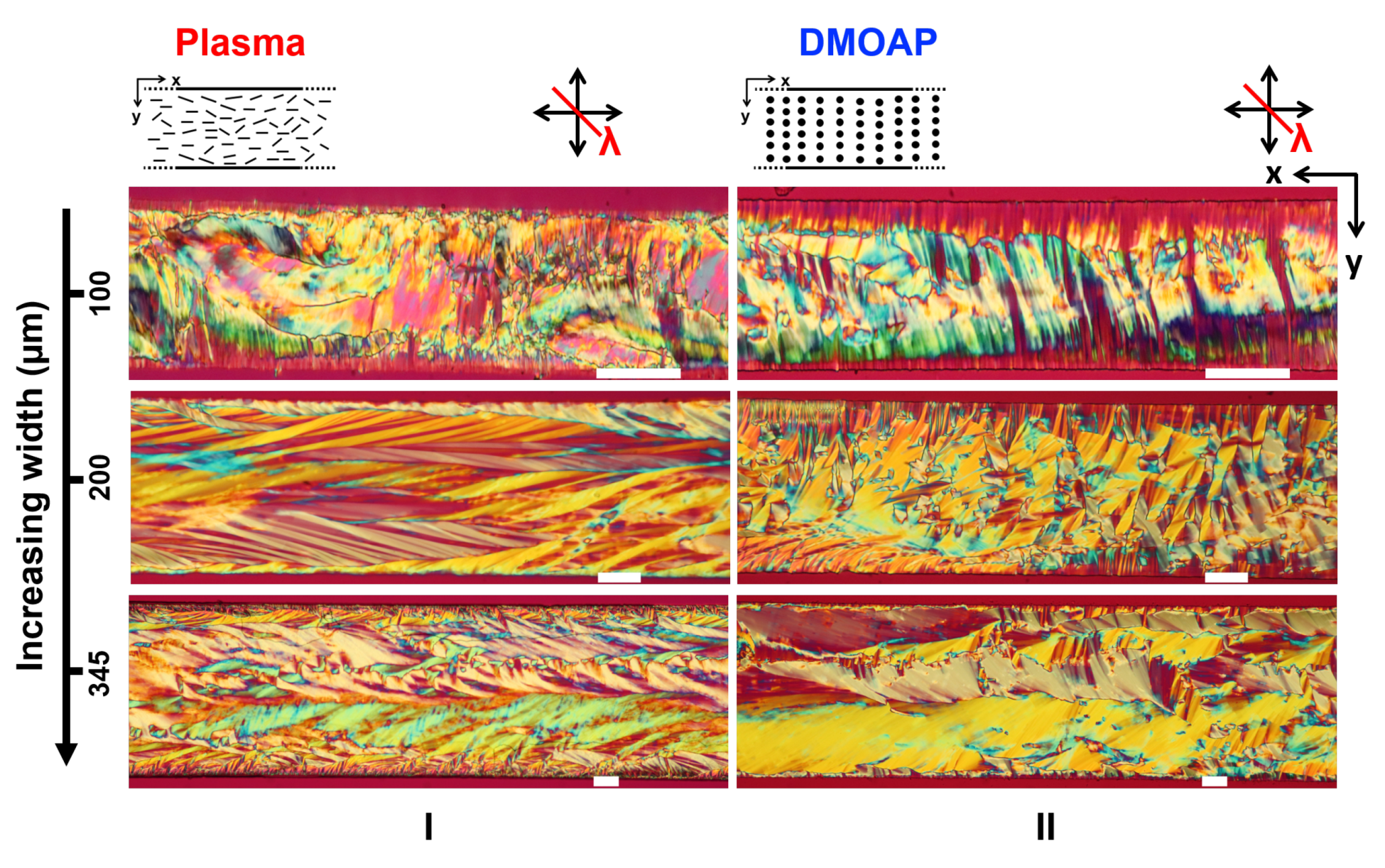
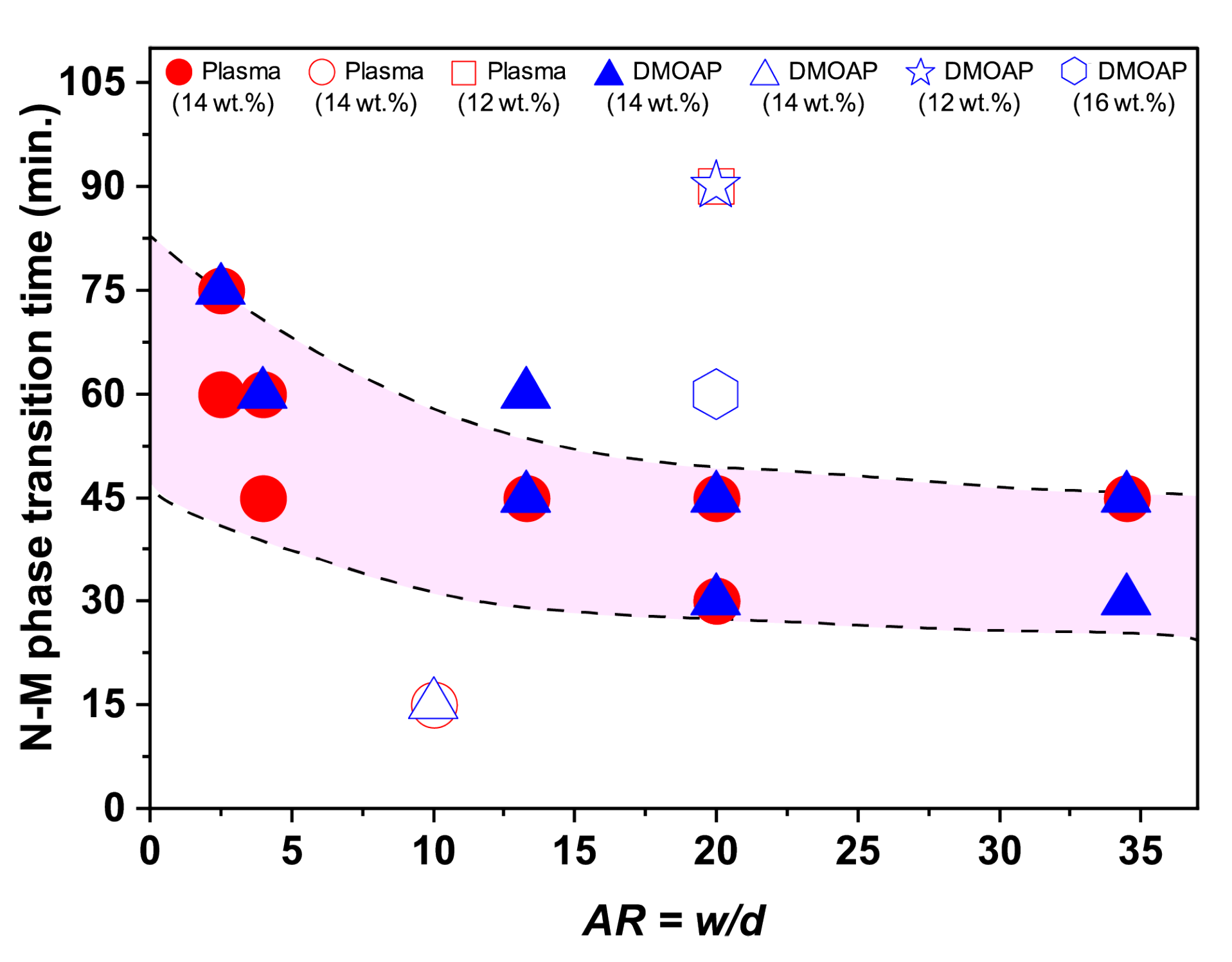
3.3. Role of the Channel Dimensions on the Evolution of the LCLC Textures
3.3.1. Role of the Channel Width
3.3.2. Role of the Channel Depth
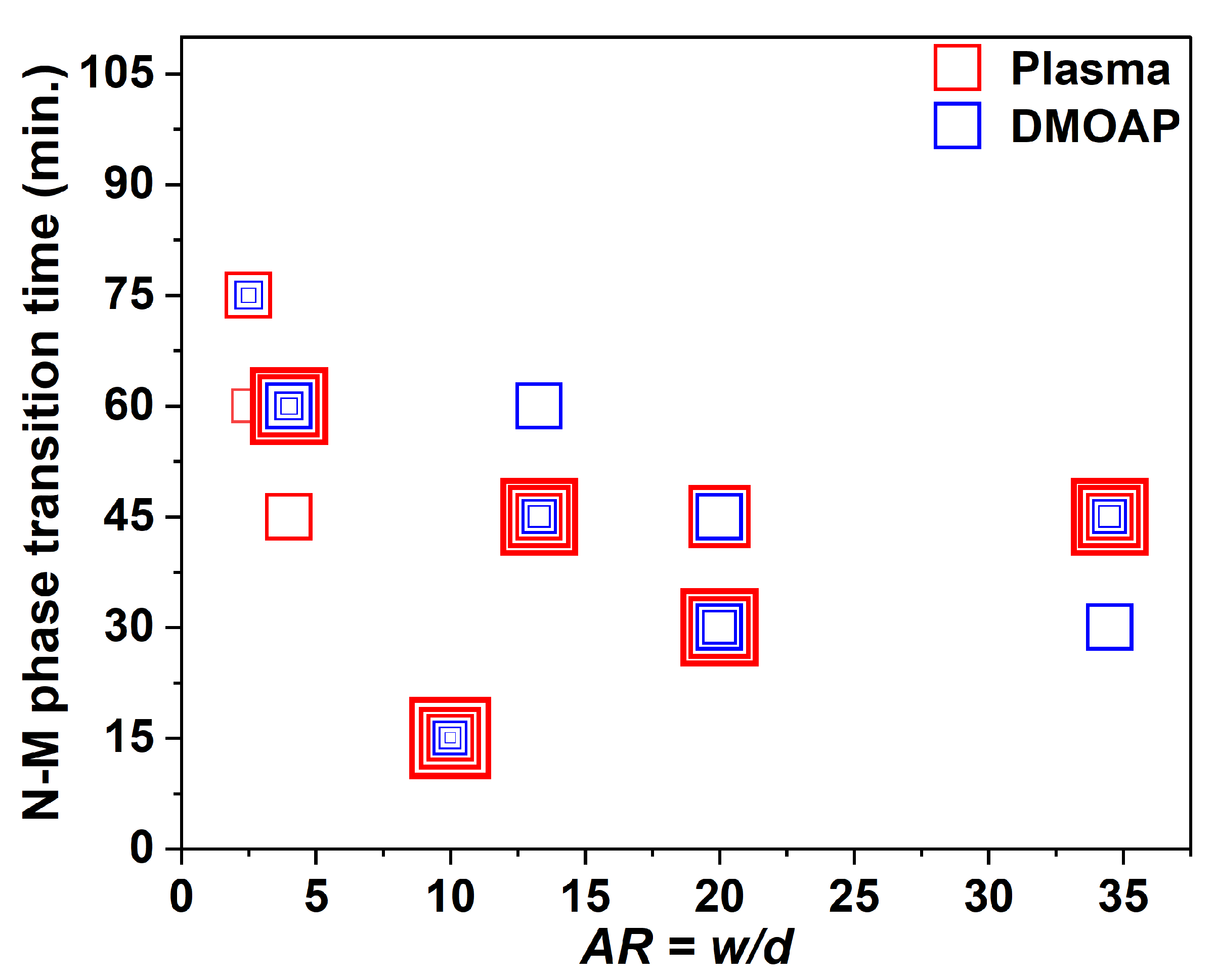
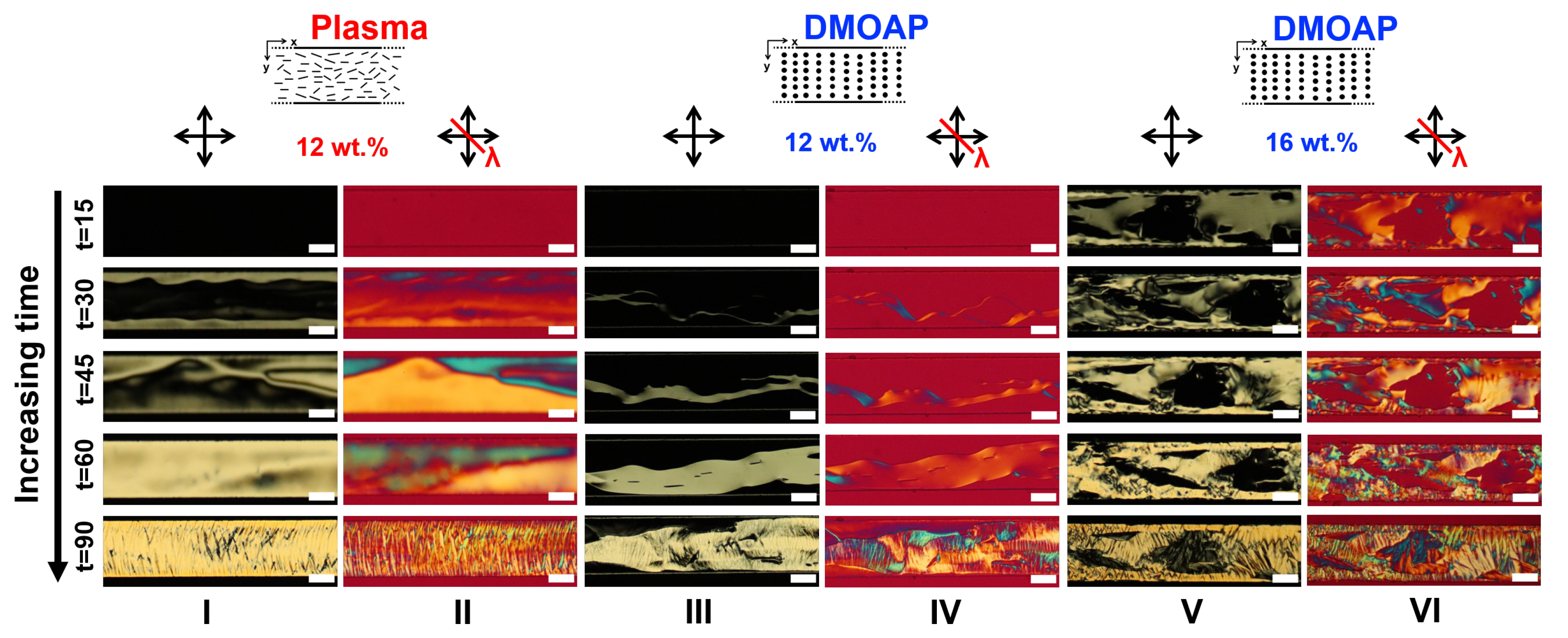
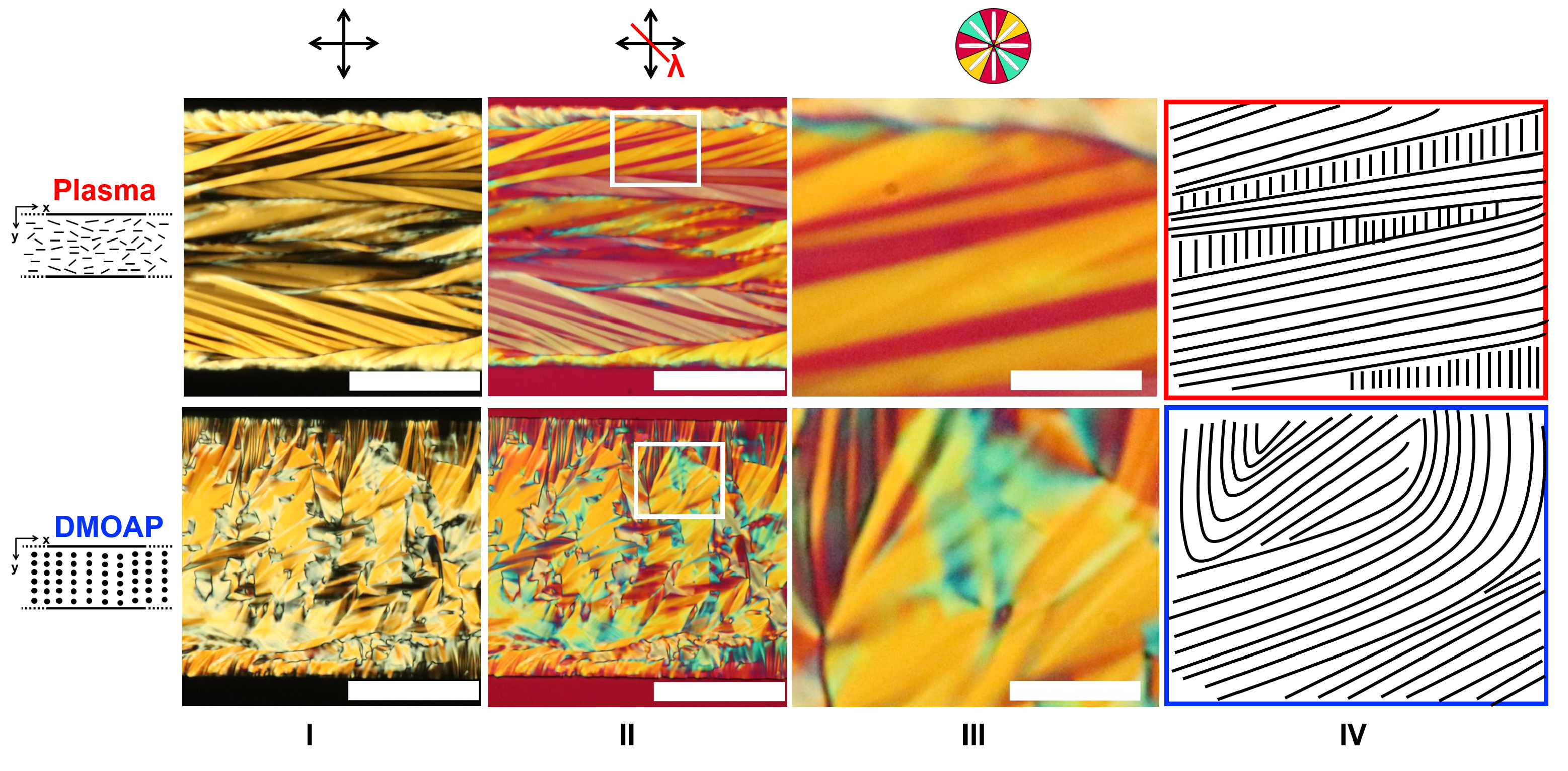
3.4. Comparing the Effects of Surface Treatment on the Stable LCLC Microfluidic Textures
3.5. Discussions
4. Conclusions
- (i)
- microchannel dimensions (confinement effect),
- (ii)
- surface anchoring (anchoring effects, specifically in relation to the PDMS surface), and
- (iii)
- experimental timescale (spontaneous surface-mediated effects).
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
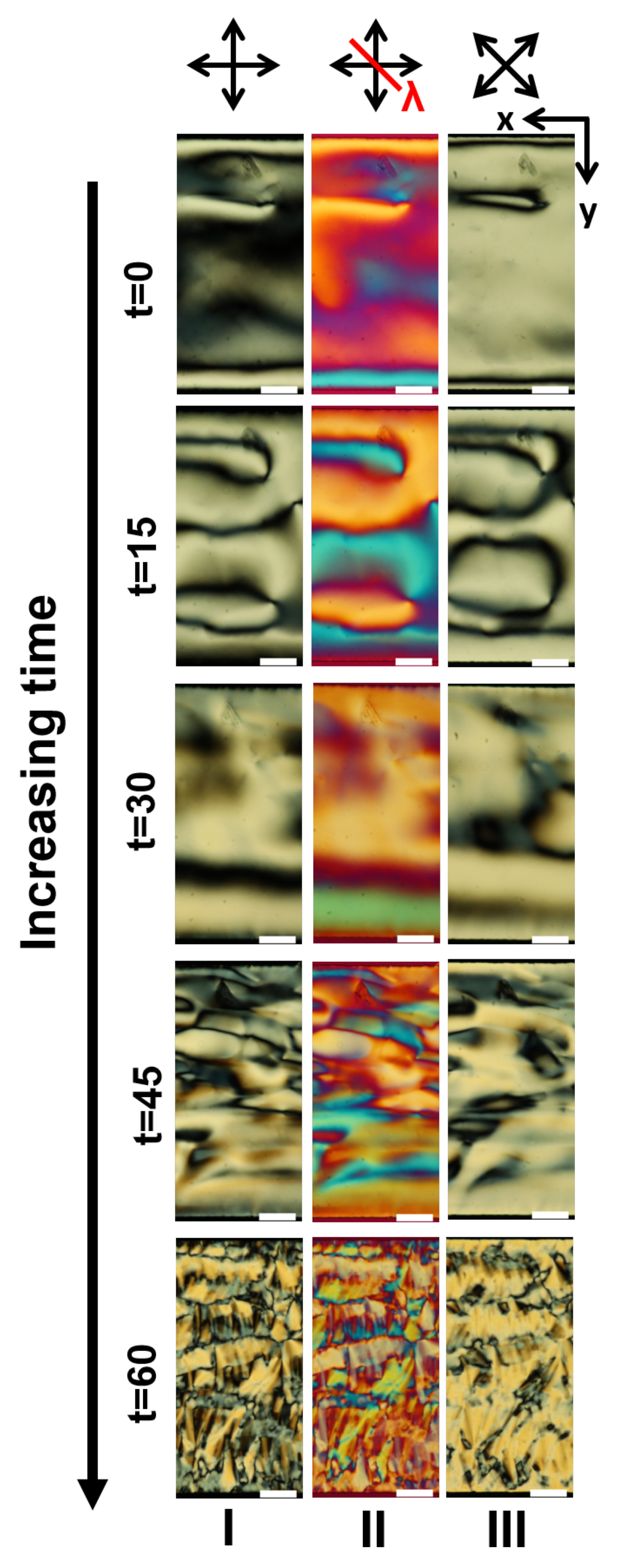
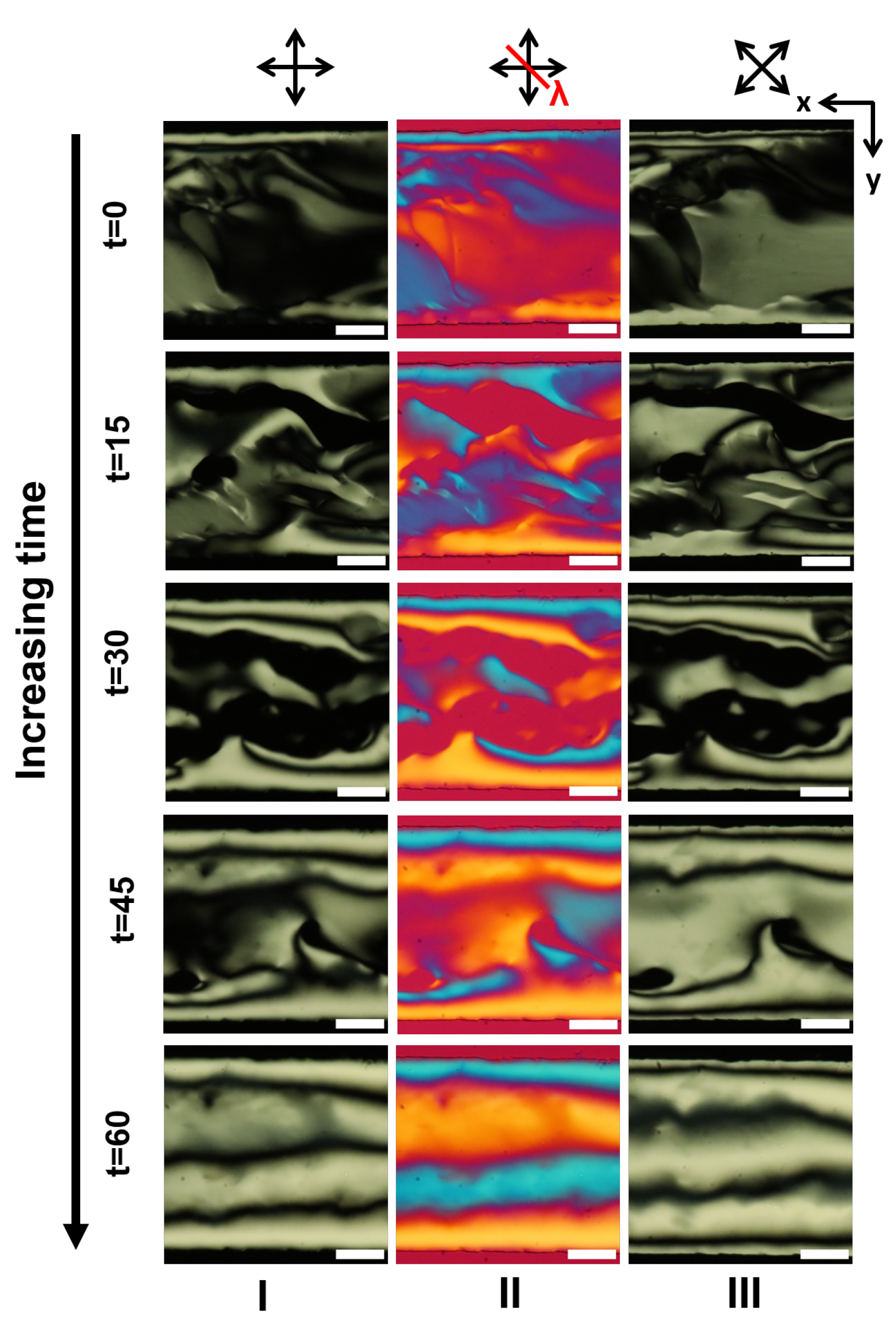
Appendix A. Supplementary Figures


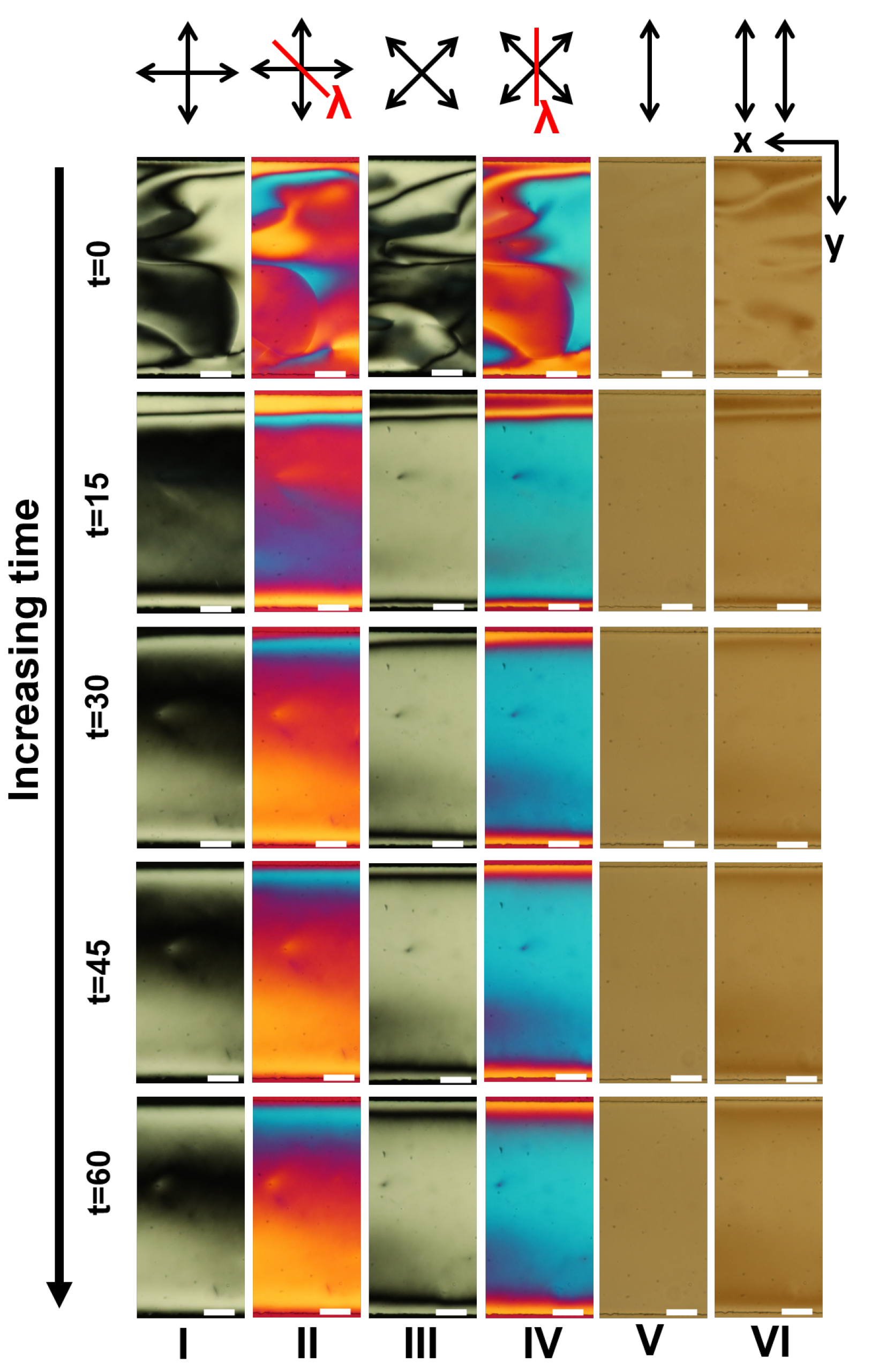
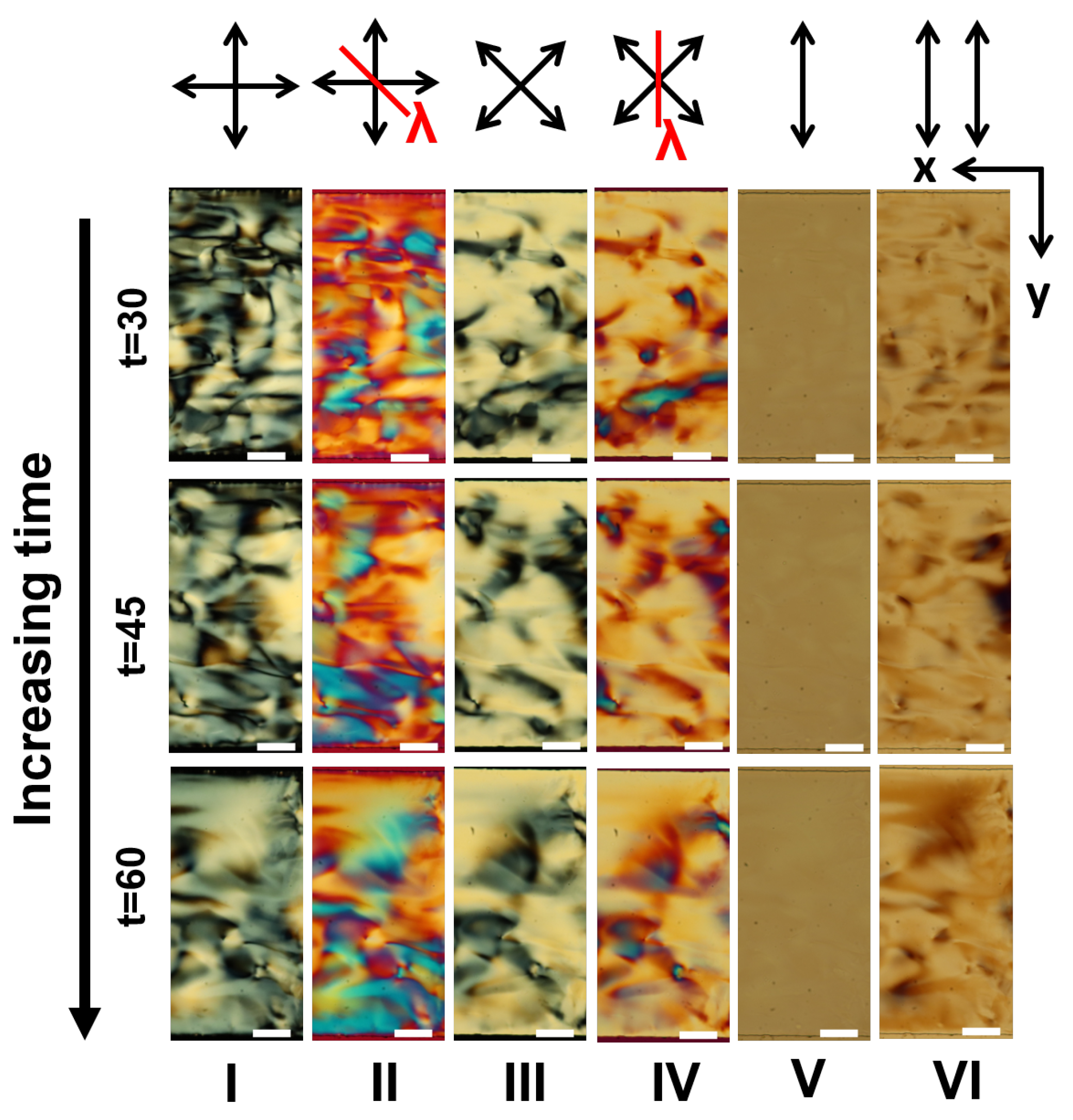
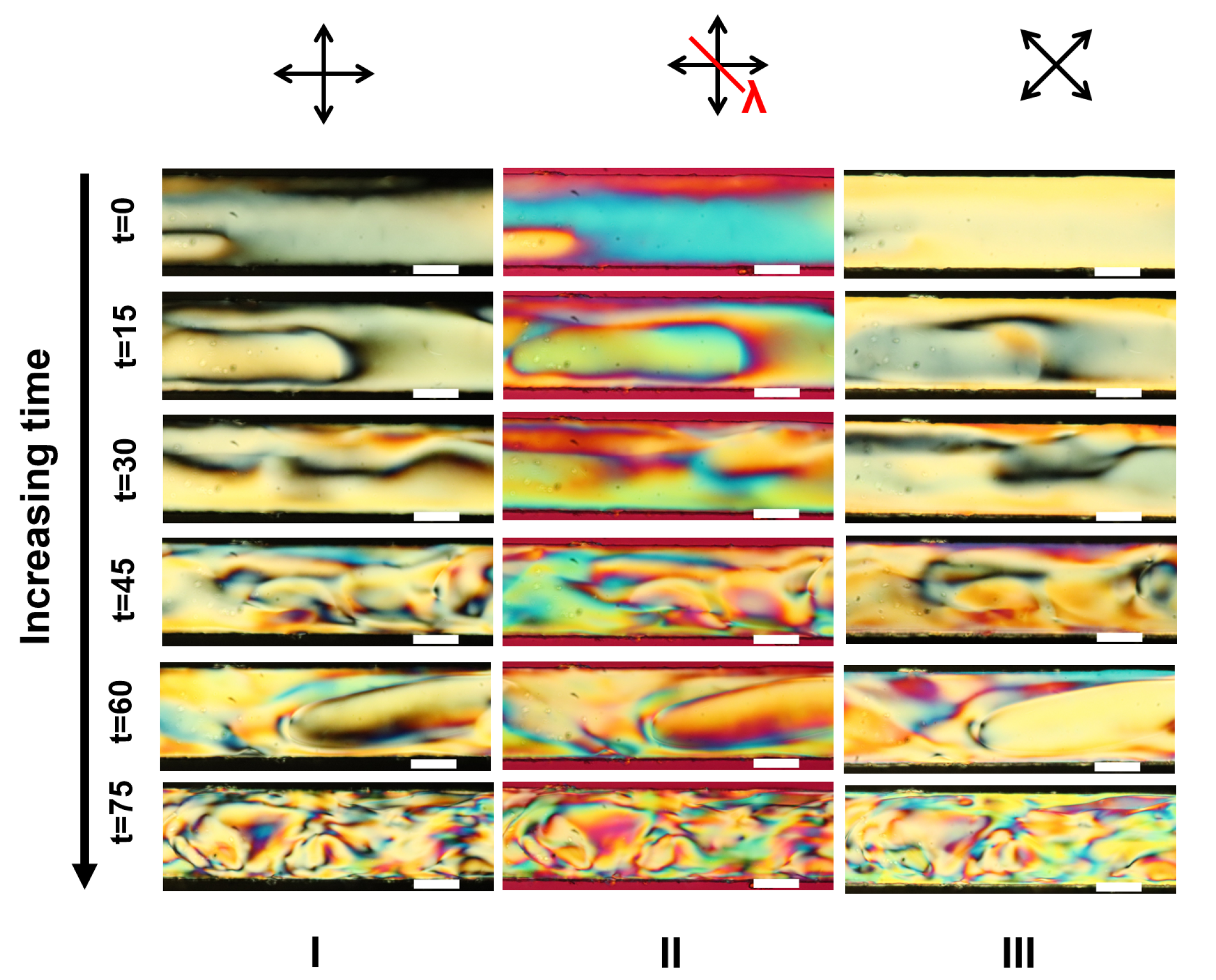
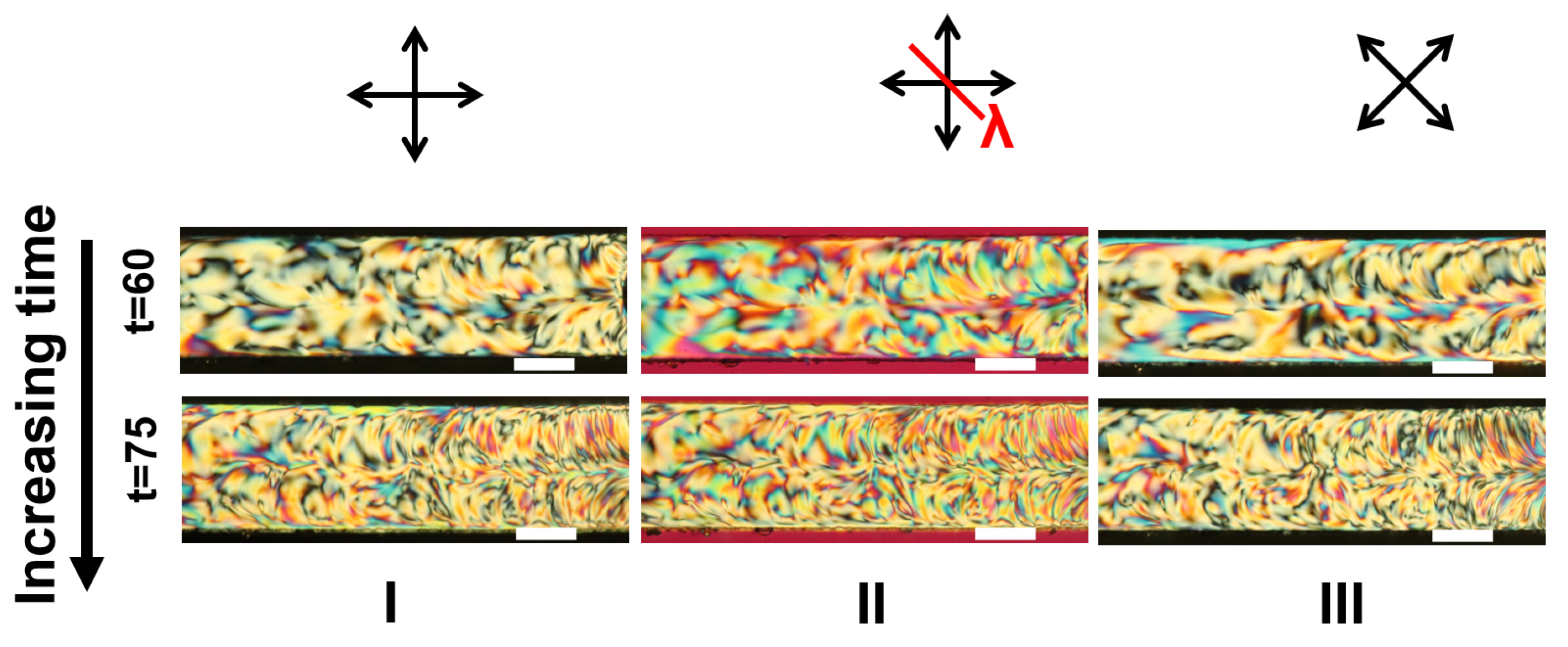

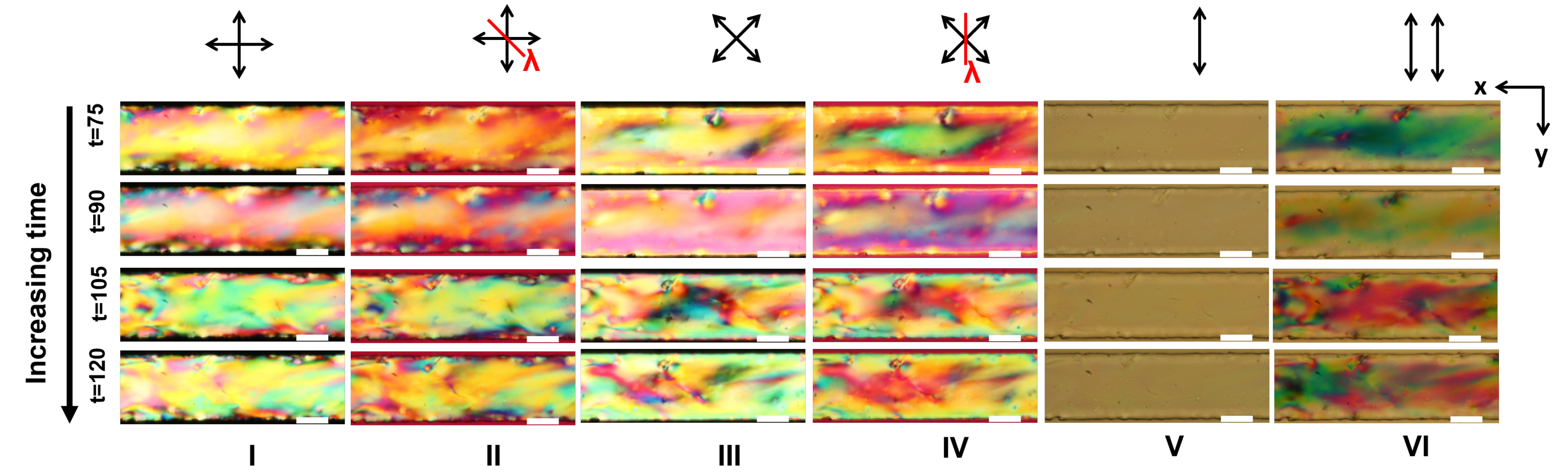
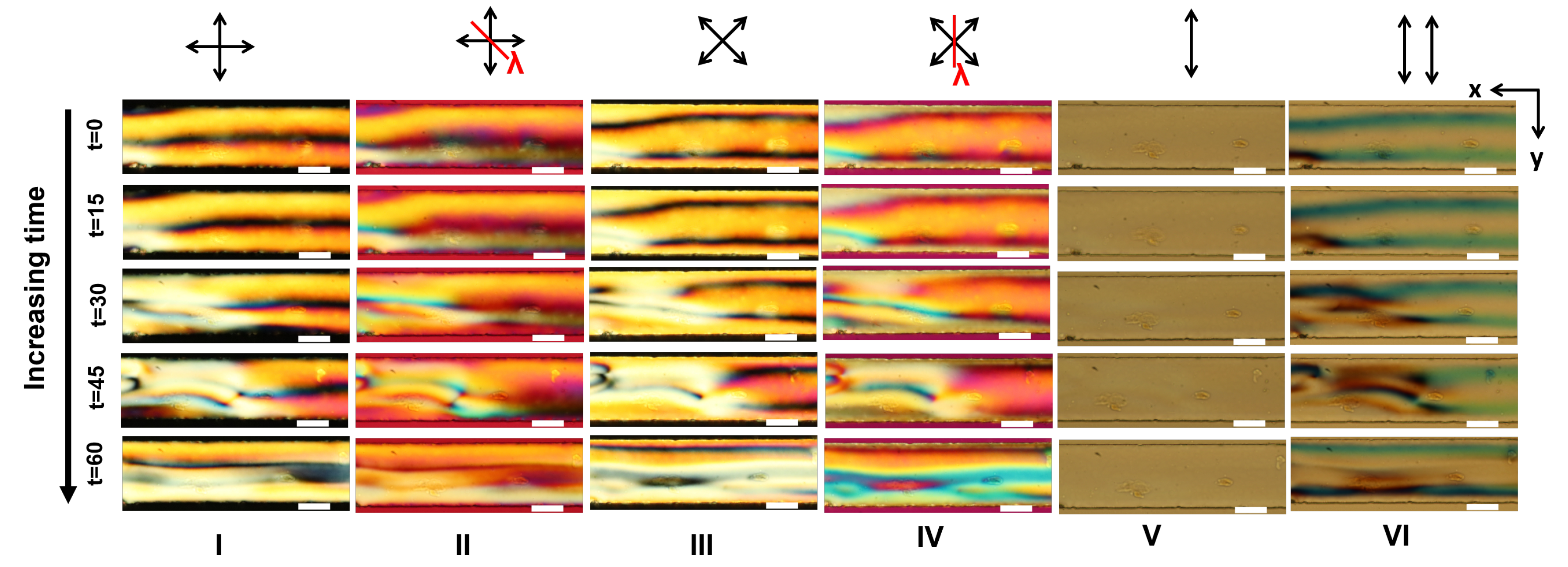
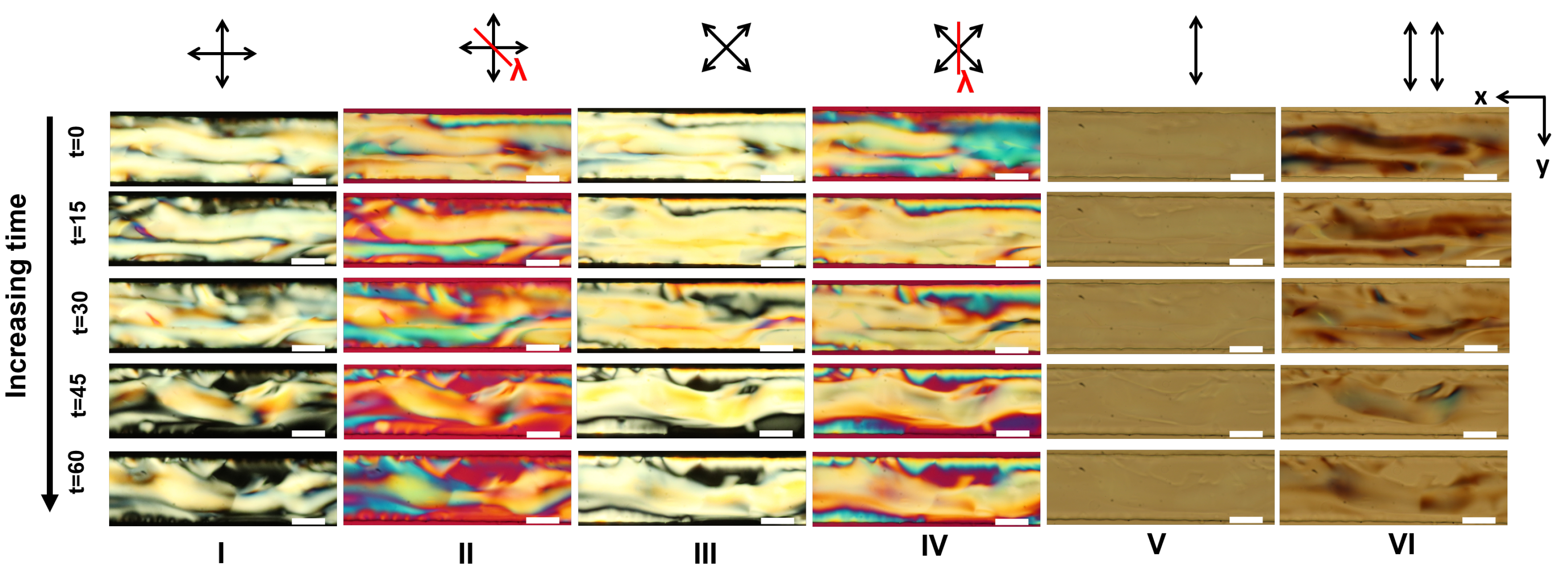
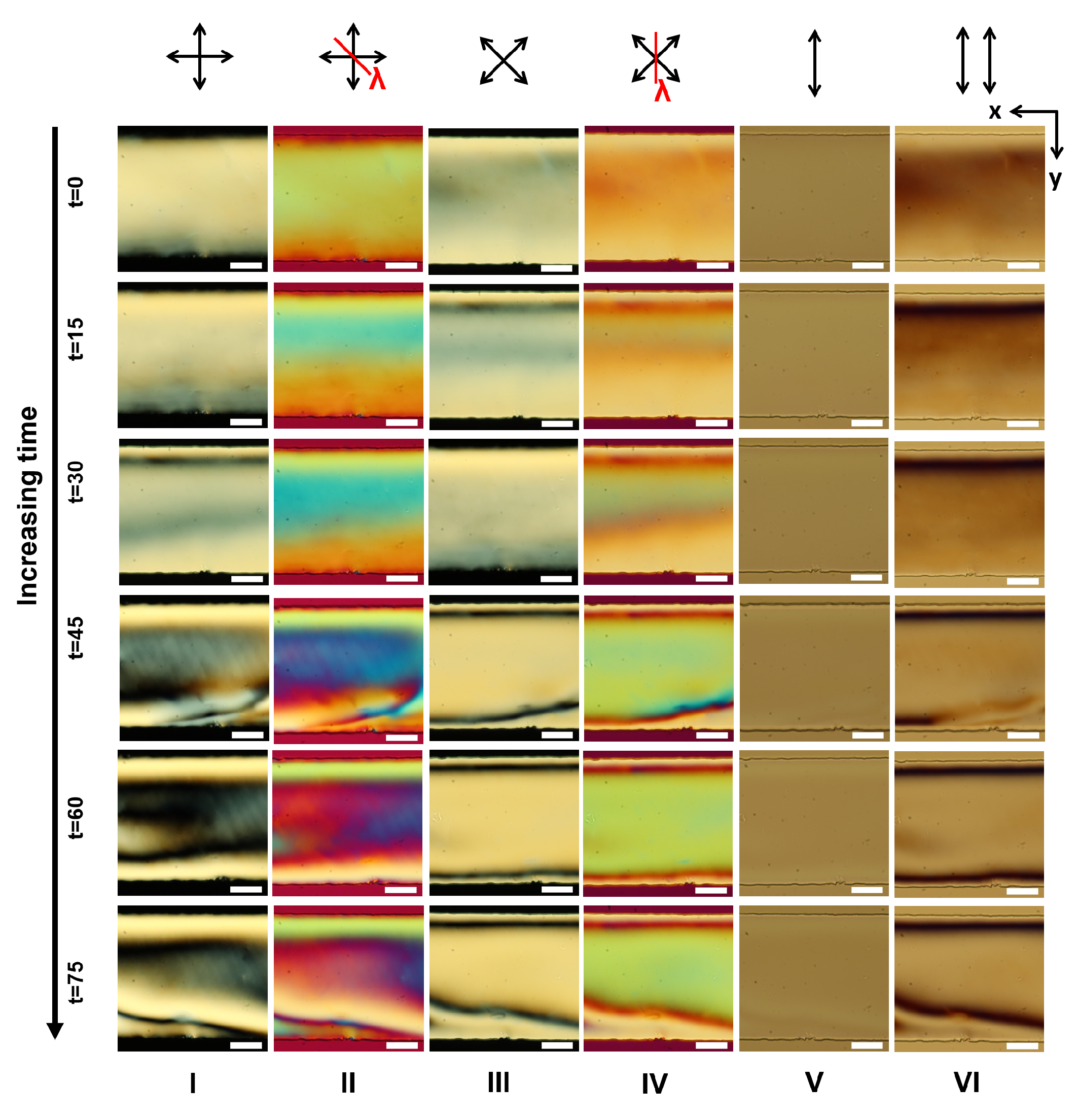



References
- Lydon, J. Chromonic review. J. Mater. Chem. 2010, 20, 10071–10099. [Google Scholar] [CrossRef]
- Lydon, J. Chromonic liquid crystalline phases. Liq. Cryst. 2011, 38, 1663–1681. [Google Scholar] [CrossRef]
- Zhou, S. Recent progresses in lyotropic chromonic liquid crystal research: Elasticity, viscosity, defect structures, and living liquid crystals. Liq. Cryst. Today 2018, 27, 91–108. [Google Scholar] [CrossRef]
- Lubensky, T.C. Confined chromonics and viral membranes. Mol. Cryst. Liq. Cryst. 2017, 646, 235–241. [Google Scholar] [CrossRef][Green Version]
- Hartshorne, N.H.; Woodard, G.D. Mesomorphism in the system disodium cromoglycate-water. Mol. Cryst. Liq. Cryst. 1973, 23, 343–368. [Google Scholar] [CrossRef]
- Cox, J.S.; Woodard, G.D.; McCrone, W.C. Solid-state chemistry of cromolyn sodium (disodium cromoglycate). J. Pharm. Sci. 1971, 60, 1458–1465. [Google Scholar] [CrossRef]
- Agra-Kooijman, D.M.; Singh, G.; Lorenz, A.; Collings, P.J.; Kitzerow, H.S.; Kumar, S. Columnar molecular aggregation in the aqueous solutions of disodium cromoglycate. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2014, 89, 062504. [Google Scholar] [CrossRef]
- Simon, K.A.; Sejwal, P.; Falcone, E.R.; Burton, E.A.; Yang, S.; Prashar, D.; Bandyopadhyay, D.; Narasimhan, S.K.; Varghese, N.; Gobalasingham, N.S.; et al. Noncovalent polymerization and assembly in water promoted by thermodynamic incompatibility. J. Phys. Chem. B 2010, 114, 10357–10367. [Google Scholar] [CrossRef]
- Shiyanovskii, S.V.; Lavrentovich, O.D.; Schneider, T.; Ishikawa, T.; Smalyukh, I.I.; Woolverton, C.J.; Niehaus, G.D.; Doane, K.J. Lyotropic chromonic liquid crystals for biological sensing applications. Mol. Cryst. Liq. Cryst. 2005, 434, 259/[587]–270/[598]. [Google Scholar] [CrossRef]
- Guo, F.; Mukhopadhyay, A.; Sheldon, B.W.; Hurt, R.H. Vertically Aligned Graphene Layer Arrays from Chromonic Liquid Crystal Precursors. Adv. Mater. 2011, 23, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Mushenheim, P.C.; Trivedi, R.R.; Tuson, H.H.; Weibel, D.B.; Abbott, N.L. Dynamic self-assembly of motile bacteria in liquid crystals. Soft Matter 2014, 10, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Sokolov, A.; Lavrentovich, O.D.; Aranson, I.S. Living liquid crystals. Proc. Natl. Acad. Sci. USA 2014, 111, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Collings, P.J.; van der Asdonk, P.; Martinez, A.; Tortora, L.; Kouwer, P.H. Anchoring strength measurements of a lyotropic chromonic liquid crystal on rubbed polyimide surfaces. Liq. Cryst. 2017, 44, 1165–1172. [Google Scholar] [CrossRef]
- Lee, H.; Labes, M.M. Lyotropic cholesteric and nematic phaes of disodium cromoglycate in magnetic fields. Mol. Cryst. Liq. Cryst. 1982, 84, 137–157. [Google Scholar] [CrossRef]
- Tone, C.M.; De Santo, M.P.; Buonomenna, M.G.; Golemme, G.; Ciuchi, F. Dynamical homeotropic and planar alignments of chromonic liquid crystals. Soft Matter 2012, 8, 8478–8482. [Google Scholar] [CrossRef]
- Simon, K.A.; Burton, E.A.; Cheng, F.; Varghese, N.; Falcone, E.R.; Wu, L.; Luk, Y.Y. Controlling thread assemblies of pharmaceutical compounds in liquid crystal phase by using functionalized nanotopography. Chem. Mater. 2010, 22, 2434–2441. [Google Scholar] [CrossRef]
- Jeong, J.; Han, G.; Johnson, A.T.C.; Collings, P.J.; Lubensky, T.C.; Yodh, A.G. Homeotropic Alignment of Lyotropic Chromonic Liquid Crystals Using Noncovalent Interactions. Langmuir 2014, 30, 2914–2920. [Google Scholar] [CrossRef]
- Kim, J.Y.; Nayani, K.; Jeong, H.S.; Jeon, H.J.; Yoo, H.W.; Lee, E.H.; Park, J.O.; Srinivasarao, M.; Jung, H.T. Macroscopic alignment of chromonic liquid crystals using patterned substrates. Phys. Chem. Chem. Phys. 2016, 18, 10362–10366. [Google Scholar] [CrossRef]
- Peng, C.; Guo, Y.; Turiv, T.; Jiang, M.; Wei, Q.H.; Lavrentovich, O.D. Patterning of Lyotropic Chromonic Liquid Crystals by Photoalignment with Photonic Metamasks. Adv. Mater. 2017, 29, 1606112. [Google Scholar] [CrossRef]
- Dhakal, N.P.; Jiang, J.; Guo, Y.; Peng, C. Self-Assembly of Aqueous Soft Matter Patterned by Liquid-Crystal Polymer Networks for Controlling the Dynamics of Bacteria. ACS Appl. Mater. Interfaces 2020, 12, 13680–13685. [Google Scholar] [CrossRef]
- Peng, C.; Turiv, T.; Guo, Y.; Wei, Q.H.; Lavrentovich, O.D. Command of active matter by topological defects and patterns. Science 2016, 354, 882–885. [Google Scholar] [CrossRef] [PubMed]
- Turiv, T.; Koizumi, R.; Thijssen, K.; Genkin, M.M.; Yu, H.; Peng, C.; Wei, Q.H.; Yeomans, J.M.; Aranson, I.S.; Doostmohammadi, A.; et al. Polar jets of swimming bacteria condensed by a patterned liquid crystal. Nat. Phys. 2020, 16, 481–487. [Google Scholar] [CrossRef]
- Nazarenko, V.G.; Boiko, O.P.; Park, H.S.; Brodyn, O.M.; Omelchenko, M.M.; Tortora, L.; Nastishin, Y.A.; Lavrentovich, O.D. Surface alignment and anchoring transitions in nematic lyotropic chromonic liquid crystal. Phys. Rev. Lett. 2010, 105, 017801. [Google Scholar] [CrossRef]
- Tone, C.M.; De Santo, M.P.; Ciuchi, F. Alignment of chromonic liquid crystals: A difficult task. In Molecular Crystals and Liquid Crystals; Taylor & Francis Group: Abingdon, UK, 2013; Volume 576, pp. 2–7. [Google Scholar] [CrossRef]
- Jeong, J.; Kang, L.; Davidson, Z.S.; Collings, P.J.; Lubensky, T.C.; Yodh, A.G. Chiral structures from achiral liquid crystals in cylindrical capillaries. Proc. Natl. Acad. Sci. USA 2015, 112, E1837–E1884. [Google Scholar] [CrossRef]
- Guo, Y.; Shahsavan, H.; Davidson, Z.S.; Sitti, M. Precise Control of Lyotropic Chromonic Liquid Crystal Alignment through Surface Topography. ACS Appl. Mater. Interfaces 2019, 11, 36110–36117. [Google Scholar] [CrossRef]
- Zhou, S.; Nastishin, Y.A.; Omelchenko, M.M.; Tortora, L.; Nazarenko, V.G.; Boiko, O.P.; Ostapenko, T.; Hu, T.; Almasan, C.C.; Sprunt, S.N.; et al. Elasticity of lyotropic chromonic liquid crystals probed by director reorientation in a magnetic field. Phys. Rev. Lett. 2012, 109, 037801. [Google Scholar] [CrossRef]
- Nayani, K.; Chang, R.; Fu, J.; Ellis, P.W.; Fernandez-Nieves, A.; Park, J.O.; Srinivasarao, M. Spontaneous emergence of chirality in achiral lyotropic chromonic liquid crystals confined to cylinders. Nat. Commun. 2015, 6, 8067. [Google Scholar] [CrossRef]
- Tortora, L.; Lavrentovich, O.D. Chiral symmetry breaking by spatial confinement in tactoidal droplets of lyotropic chromonic liquid crystals. Proc. Natl. Acad. Sci. USA 2011, 108, 5163–5168. [Google Scholar] [CrossRef]
- Davidson, Z.S.; Kang, L.; Jeong, J.; Still, T.; Collings, P.J.; Lubensky, T.C.; Yodh, A.G. Chiral structures and defects of lyotropic chromonic liquid crystals induced by saddle-splay elasticity. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2015, 91, 050501. [Google Scholar] [CrossRef]
- Jeong, J.; Davidson, Z.S.; Collings, P.J.; Lubensky, T.C.; Yodh, A.G. Chiral symmetry breaking and surface faceting in chromonic liquid crystal droplets with giant elastic anisotropy. Proc. Natl. Acad. Sci. USA 2014, 111, 1742–1747. [Google Scholar] [CrossRef]
- Nayani, K.; Fu, J.; Chang, R.; Park, J.O.; Srinivasarao, M. Using chiral tactoids as optical probes to study the aggregation behavior of chromonics. Proc. Natl. Acad. Sci. USA 2017, 114, 3826–3831. [Google Scholar] [CrossRef] [PubMed]
- DIetrich, C.F.; Rudquist, P.; Lorenz, K.; Giesselmann, F. Chiral structures from achiral micellar lyotropic liquid crystals under capillary confinement. Langmuir 2017, 33, 5852–5862. [Google Scholar] [CrossRef] [PubMed]
- Baza, H.; Turiv, T.; Li, B.X.; Li, R.; Yavitt, B.M.; Fukuto, M.; Lavrentovich, O.D. Shear-induced polydomain structures of nematic lyotropic chromonic liquid crystal disodium cromoglycate. Soft Matter 2020, 16, 8565–8576. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef]
- Sengupta, A.; Tkalec, U.; Bahr, C. Nematic textures in microfluidic environment. Soft Matter 2011, 7, 6542–6549. [Google Scholar] [CrossRef]
- Sengupta, A.; Pieper, C.; Enderlein, J.; Bahr, C.; Herminghaus, S. Flow of a nematogen past a cylindrical micro-pillar. Soft Matter 2013, 9, 1937–1946. [Google Scholar] [CrossRef]
- Sengupta, A. Topological microfluidics: Present and prospects. Liq. Cryst. Today 2015, 24, 70–80. [Google Scholar] [CrossRef]
- Emeršič, T.; Zhang, R.; Kos, Z.; Čopar, S.; Osterman, N.; de Pablo, J.J.; Tkalec, U. Sculpting stable structures in pure liquids. Sci. Adv. 2019, 5, eaav4283. [Google Scholar] [CrossRef]
- Sengupta, A.; Schulz, B.; Ouskova, E.; Bahr, C. Functionalization of microfluidic devices for investigation of liquid crystal flows. Microfluid. Nanofluidics 2012, 13, 941–955. [Google Scholar] [CrossRef]
- Sengupta, A.; Tkalec, U.; Ravnik, M.; Yeomans, J.M.; Bahr, C.; Herminghaus, S. Liquid crystal microfluidics for tunable flow shaping. Phys. Rev. Lett. 2013, 110, 048303. [Google Scholar] [CrossRef]
- Stieger, T.; Agha, H.; Schoen, M.; Mazza, M.G.; Sengupta, A. Hydrodynamic cavitation in Stokes flow of anisotropic fluids. Nat. Commun. 2017, 8, 15550. [Google Scholar] [CrossRef] [PubMed]
- Giomi, L.; Kos, Z.; Ravnik, M.; Sengupta, A. Cross-talk between topological defects in different fields revealed by nematic microfluidics. Proc. Natl. Acad. Sci. USA 2017, 114, E5771–E5777. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, A.; Bahr, C.; Herminghaus, S. Topological microfluidics for flexible micro-cargo concepts. Soft Matter 2013, 9, 7251–7260. [Google Scholar] [CrossRef]
- Na, Y.J.; Yoon, T.Y.; Park, S.; Lee, B.; Lee, S.D. Electrically Programmable Nematofluidics with a High Level of Selectivity in a Hierarchically Branched Architecture. ChemPhysChem 2010, 11, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Cuennet, J.G.; Vasdekis, A.E.; De Sio, L.; Psaltis, D. Optofluidic modulator based on peristaltic nematogen microflows. Nat. Photonics 2011, 5, 234–238. [Google Scholar] [CrossRef]
- Cuennet, J.G.; Vasdekis, A.E.; Psaltis, D. Optofluidic-tunable color filters and spectroscopy based on liquid-crystal microflows. Lab Chip 2013, 13, 2721–2726. [Google Scholar] [CrossRef]
- Wee, D.; Hwang, S.H.; Song, Y.S.; Youn, J.R. Tunable optofluidic birefringent lens. Soft Matter 2016, 12, 3868–3876. [Google Scholar] [CrossRef]
- Sengupta, A. Tuning Fluidic Resistance via Liquid Crystal Microfluidics. Int. J. Mol. Sci. 2013, 14, 22826–22844. [Google Scholar] [CrossRef]
- Wiese, O.; Marenduzzo, D.; Henrich, O. Microfluidic flow of cholesteric liquid crystals. Soft Matter 2016, 12, 9223–9237. [Google Scholar] [CrossRef]
- Guo, Y.; Afghah, S.; Xiang, J.; Lavrentovich, O.D.; Selinger, R.L.; Wei, Q.H. Cholesteric liquid crystals in rectangular microchannels: Skyrmions and stripes. Soft Matter 2016, 12, 6312–6320. [Google Scholar] [CrossRef]
- Sengupta, A.; Herminghaus, S.; Bahr, C. Liquid crystal microfluidics: Surface, elastic and viscous interactions at microscales. Liq. Cryst. Rev. 2014, 2, 73–110. [Google Scholar] [CrossRef]
- Čopar, S.; Kos, Z.; Emeršič, T.; Tkalec, U. Microfluidic control over topological states in channel-confined nematic flows. Nat. Commun. 2020, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, N.; Jünnemann-Held, G.; Collings, P.J.; Kitzerow, H.S. Self-organized assemblies of colloidal particles obtained from an aligned chromonic liquid crystal dispersion. Soft Matter 2015, 11, 1547–1553. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Ong, I.; Mazza, M.; Sengupta, A. Surface-mediated stability of lyotropic textures in microfluidic environments. 2020. in prepration. [Google Scholar]
- Sengupta, A.; Herminghaus, S.; Bahr, C. Nematic liquid crystals and nematic colloids in microfluidic environment. Mol. Cryst. Liq. Cryst. 2011, 547, 203–212. [Google Scholar] [CrossRef]
- Sengupta, A. Topological Microfluidics: Nematic Liquid Crystals and Nematic Colloids in Microfluidic Environment; Springer International Publishing: Cham, Switzerland, 2013; Volume 1, pp. 1–150. [Google Scholar] [CrossRef]
- Nastishin, Y.A.; Liu, H.; Schneider, T.; Nazarenko, V.; Vasyuta, R.; Shiyanovskii, S.V.; Lavrentovich, O.D. Optical characterization of the nematic lyotropic chromonic liquid crystals: Light absorption, birefringence, and scalar order parameter. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2005, 72, 041711. [Google Scholar] [CrossRef]
- Mirri, G.; Jampani, V.S.; Cordoyiannis, G.; Umek, P.; Kouwer, P.H.; Muševič, I. Stabilisation of 2D colloidal assemblies by polymerisation of liquid crystalline matrices for photonic applications. Soft Matter 2014, 10, 5797–5803. [Google Scholar] [CrossRef]
- Bodas, D.; Khan-Malek, C. Hydrophilization and hydrophobic recovery of PDMS by oxygen plasma and chemical treatment-An SEM investigation. Sens. Actuators B Chem. 2007, 123, 368–373. [Google Scholar] [CrossRef]
- Mata, A.; Fleischman, A.J.; Roy, S. Characterization of Polydimethylsiloxane (PDMS) Properties for Biomedical Micro/Nanosystems. Biomed. Microdevices 2005, 7, 281–293. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Datta, A.; Berg, J.M.; Gangopadhyay, S. Studies on surface wettability of poly(dimethyl) siloxane (PDMS) and glass under oxygen-plasma treatment and correlation with bond strength. J. Microelectromech. Syst. 2005, 14, 590–597. [Google Scholar] [CrossRef]
- Hillborg, H.; Ankner, J.F.; Gedde, U.W.; Smith, G.D.; Yasuda, H.K.; Wikström, K. Crosslinked polydimethylsiloxane exposed to oxygen plasma studied by neutron reflectometry and other surface specific techniques. Polymer 2000, 41, 6851–6863. [Google Scholar] [CrossRef]
- Ruben, B.; Elisa, M.; Leandro, L.; Victor, M.; Gloria, G.; Marina, S.; Mian, S.K.; Pandiyan, R.; Nadhira, L. Oxygen plasma treatments of polydimethylsiloxane surfaces: Effect of the atomic oxygen on capillary flow in the microchannels. Micro Nano Lett. 2017, 12, 754–757. [Google Scholar] [CrossRef]
- Toepke, M.W.; Beebe, D.J. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip 2006, 6, 1484–1486. [Google Scholar] [CrossRef] [PubMed]
- Waters, L.J.; Finch, C.V.; Bhuiyan, A.K.H.; Hemming, K.; Mitchell, J.C. Effect of plasma surface treatment of poly(dimethylsiloxane) on the permeation of pharmaceutical compounds. J. Pharm. Anal. 2017, 7, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Yoshimi, A.; Hashizume, H.; Tamaki, S.; Tsuda, H.; Fukata, F.; Nishimura, K.i.; Yata, N. Importance of Hydrolysis of Amino Acid Moiety in Water-Soluble Prodrugs of Disodium Cromoglycate for Increased Oral Bioavailability. J. Pharmacobio Dyn. 1992, 15, 339–345. [Google Scholar] [CrossRef] [PubMed]

| Channel Dimension w × d (m) | Anchoring Condition | DSCG Concentration (%) | Nematic to M-Phase Transition Time (min) |
|---|---|---|---|
| 100 × 10 | Degenerate planar | 14 | 15 |
| 100 × 10 | Homeotropic | 14 | 15 |
| 200× 10 | Degenerate planar | 14 | 30–45 |
| 200 × 10 | Homeotropic | 14 | 30–45 |
| 345× 10 | Degenerate planar | 14 | 45 |
| 345 × 10 | Homeotropic | 14 | 30–45 |
| 100 × 25 | Degenerate planar | 14 | 45–60 |
| 100 × 25 | Homeotropic | 14 | 60 |
| 100 × 40 | Degenerate planar | 14 | 60–75 |
| 100 × 40 | Homeotropic | 14 | 75 |
| 200 × 15 | Degenerate planar | 14 | 45 |
| 200 × 15 | Homeotropic | 14 | 45–60 |
| 200 × 10 | Degenerate planar | 12 | 90 |
| 200 × 10 | Homeotropic | 12 | 90 |
| 200 × 10 | Homeotropic | 16 | 60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, A.; Ong, I.L.H.; Sengupta, A. Time Dependent Lyotropic Chromonic Textures in Microfluidic Confinements. Crystals 2021, 11, 35. https://doi.org/10.3390/cryst11010035
Sharma A, Ong ILH, Sengupta A. Time Dependent Lyotropic Chromonic Textures in Microfluidic Confinements. Crystals. 2021; 11(1):35. https://doi.org/10.3390/cryst11010035
Chicago/Turabian StyleSharma, Anshul, Irvine Lian Hao Ong, and Anupam Sengupta. 2021. "Time Dependent Lyotropic Chromonic Textures in Microfluidic Confinements" Crystals 11, no. 1: 35. https://doi.org/10.3390/cryst11010035
APA StyleSharma, A., Ong, I. L. H., & Sengupta, A. (2021). Time Dependent Lyotropic Chromonic Textures in Microfluidic Confinements. Crystals, 11(1), 35. https://doi.org/10.3390/cryst11010035





