Natural vs. Synthetic Zeolites
Abstract
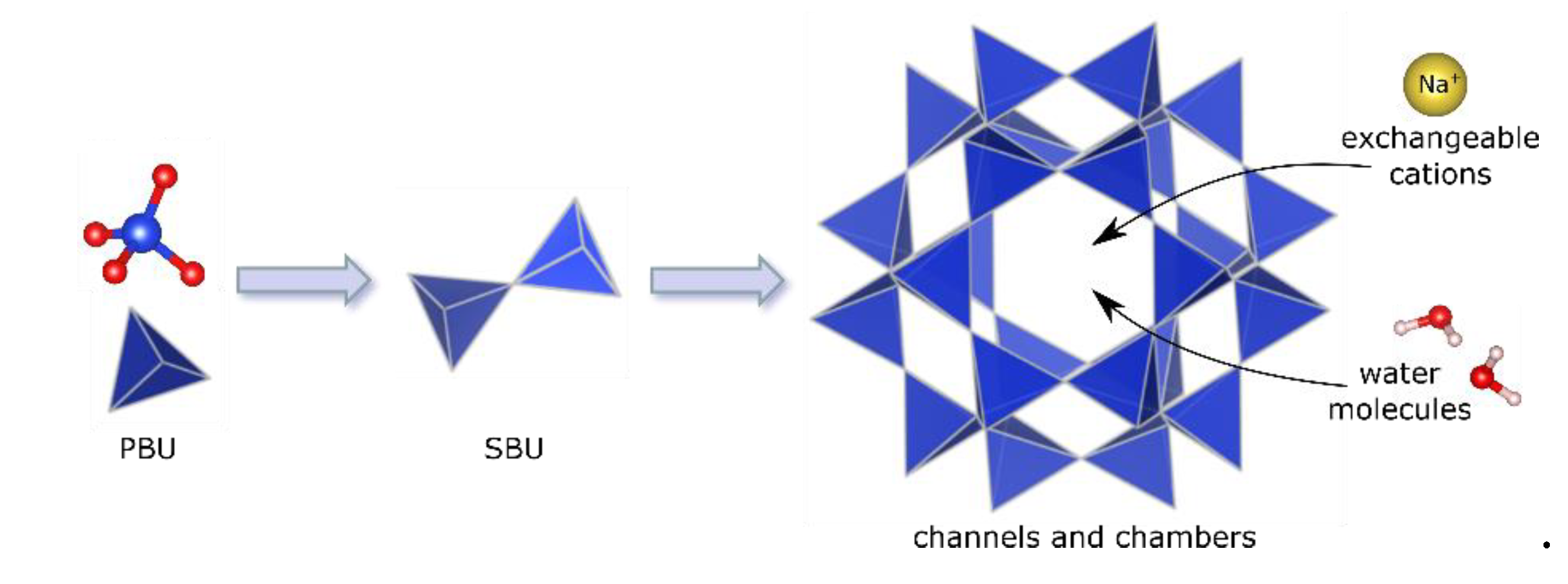
1. What Are Zeolites?
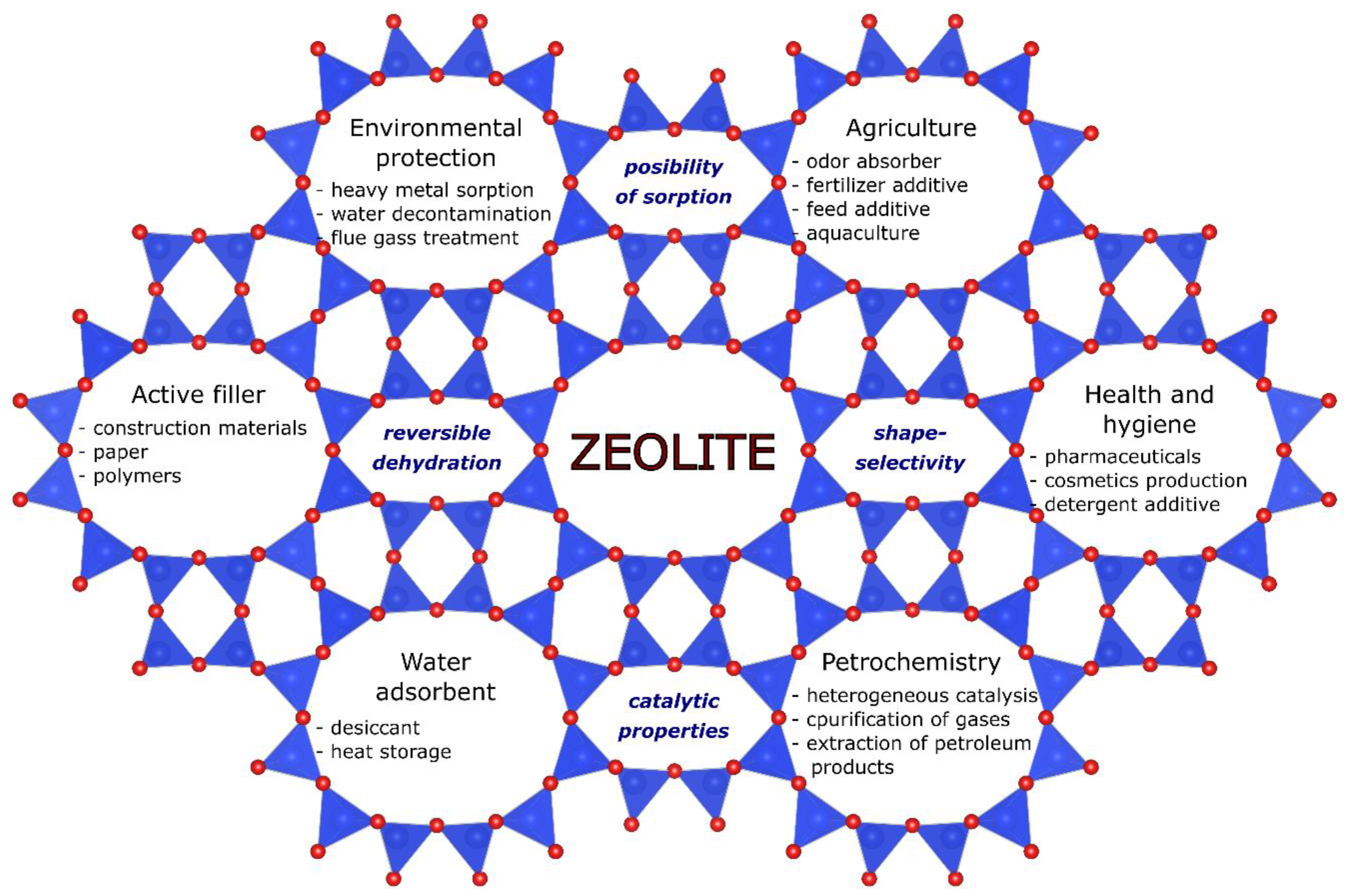
- Low density and large volume of free spaces;
- The existence of channels and chambers of strictly defined dimensions (shape-selectivity);
- High degree of hydration and the presence of so-called "zeolite water";
- High degree of crystallinity;
- Possibility of sorption of molecules and ions;
- Ion exchange capacity;
- Catalytic properties.
2. Natural Zeolites
Application Examples
3. Synthetic Zeolites
3.1. Substrates and Products
3.2. Synthesis Methods
3.3. Zolitization Products
3.4. Benefits of Synthetic Zeolite
4. Summary
- Accurate analysis of used raw material composition;
- Selection of appropriate parameters of zeolite synthesis;
- Comprehensive structural characterization of zeolites;
- Potential modification of zeolites to improve their properties;
- Zeolite granulation;
- Assessment of ion exchange and sorption potential;
- Behavior and mechanisms in zeolite-catalyzed reactions.
Funding
Conflicts of Interest
References
- Meier, W.M. Zeolites and Zeolite-Like Materials. In Proceedings of the Seventh International Zeolite Conference, Tokyo, Japan, 17–22 August 1986; Elsevier: Amsterdam, The Netherlands, 1986; pp. 13–22. [Google Scholar]
- Reháková, M.; Čuvanová, S.; Dzivák, M.; Rimár, J.; Gaval’ova, Z. Agricultural and agrochemical uses of natural zeolite of the clinoptilolite type. Curr. Opin. Solid State Mater. Sci. 2004, 8, 397–404. [Google Scholar] [CrossRef]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry, and Use; Wiley: Hoboken, NJ, USA, 1973. [Google Scholar]
- Ciciszwili, G.W.; Andronikaszwili, T.G.; Kirow, G.N.; Filizowa, Ł.D. Zeolity Naturalne; Wydawnictwo Naukowo-Techniczne: Warszawa, Poland, 1990. [Google Scholar]
- Gottardi, G.; Galli, E. Natural Zeolites, Mineral and Rocks 18; Springer: Berlin/Heidelberg, Germany, 1985. [Google Scholar]
- Dyer, A. Ion-exchange properties of zeolites. In Zeolites and Ordered Mesoporous Materials: Progress and Prospects, Studies in Surface Science and Catalysis 157; Čejka, J., Bekkum, H., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 181–204. [Google Scholar]
- Sprynskyy, M.; Buszewski, B.; Terzyk, A.P.; Namieśnik, J. Study of the selection mechanism of heavy metal (Pb2+, Cu2+, Ni2+, and Cd2+) adsorption on clinoptilolite. J. Colloid Interface Sci. 2006, 304, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Gorimbo, J.; Taenzana, B.; Muleja, A.A.; Kuvarega, A.T.; Jewell, L.L. Adsorption of cadmium, nickel and lead ions: Equilibrium, kinetic and selectivity studies on modified clinoptilolites from the USA and RSA. Environ. Sci. Pollut. Res. Int. 2018, 25, 30962–30978. [Google Scholar] [CrossRef]
- Vassileva, P.; Voikova, D. Investigation on natural and pretreated Bulgarian clinoptilolite for ammonium ions removal from aqueous solutions. J. Hazard. Mater. 2009, 170, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Ciosek, A.L.; Luk, G.K. An innovative dual-column system for heavy metallic ion sorption by natural zeolite. Appl. Sci. 2017, 7, 795. [Google Scholar] [CrossRef]
- Belova, T.P. Adsorption of heavy metal ions (Cu2+, Ni2+, Co2+, and Fe2+) from aqueous solutions by natural zeolite. Heliyon 2019, 5, e02320. [Google Scholar] [CrossRef] [PubMed]
- Belousov, P.; Semenkova, A.; Egorova, T.; Romanchuk, A.; Zakusin, S.; Dorzhieva, O.; Tyupina, E.; Izosimova, Y.; Tolpeshta, I.; Chernov, M.; et al. Cesium sorption and desorption on glauconite, gentonite, zeolite, and diatomite. Minerals 2019, 9, 625. [Google Scholar] [CrossRef]
- Ivanova, E.; Karsheva, M.; Koumanova, B. Adsorption of ammonium ions onto natural zeolite. J. Chem. Technol. Metall. 2010, 45, 295–302. [Google Scholar]
- Sangeetha, C.; Baskar, P. Zeolite and its potential uses in agriculture: A critical review. Agric. Rev. 2016, 37, 101–108. [Google Scholar] [CrossRef]
- Junaid, A.S.M.; Yin, H.; Koenig, A.; Swenson, P.; Chowdhury, J.; Burland, G.; McCaffrey, W.C.; Kuznicki, S.M. Natural zeolite catalyzed cracking-assisted light hydrocarbon extraction of bitumen from Athabasca oil sands. Appl. Catal. A 2009, 354, 44–49. [Google Scholar] [CrossRef]
- Copcia, V.E.; Luchian, C.; Dunca, S.; Bilba, N.; Hristodor, C.M. Antibacterial activity of silver-modified natural clinoptilolite. J. Mater. Sci. 2011, 46, 7121–7128. [Google Scholar] [CrossRef]
- Baerlocher, C.; McCusker, L.B. Database of Zeolite Structures. Available online: http://www.iza-structure.org/databases/ (accessed on 15 July 2020).
- Prasetyoko, H.D.; Santoso, M.; Qoniah, I.; Leaw, W.L.; Firda, P.B.D. A review on synthesis of kaolin-based zeolite and the effect of impurities. J. Chin. Chem. Soc-Taip. 2020, 67, 911–936. [Google Scholar]
- Hadi NurPereira, P.M.; Ferreira, B.F.; Oliveira, N.P.; Nassar, E.J.; Ciuffi, K.J.; Vicente, M.A.; Trujillano, R.; Rives, V.; Gil, A.; Korili, S.; et al. Synthesis of zeolite A from metakaolin and its application in the adsorption of cationic dyes. Appl. Sci. 2018, 8, 608. [Google Scholar]
- Biel, O.; Rożek, P.; Florek, P.; Mozgawa, W.; Król, M. Alkaline activation of kaolin group minerals. Crystals 2020, 10, 268. [Google Scholar] [CrossRef]
- Gualtieri, A.F. Synthesis of sodium zeolite from natural halloysite. Phys. Chem. Miner. 2001, 28, 719–728. [Google Scholar] [CrossRef]
- Dyer, A.; Tangkawanit, S.; Rangsriwatananon, K. Exchange diffusion of Cu2+, Ni2+, Pb2+ and Zn2+ into analcime synthesized from perlite. Microporous Mesoporous Mater. 2004, 75, 273–279. [Google Scholar] [CrossRef]
- Christidis, G.E.; Phapaliars, I.; Kontopoulos, A. Zeolitization of perlite fines: Mineralogical characteristics of the end product and mobilization of chemical elements. Appl. Clay Sci. 1999, 15, 305–324. [Google Scholar] [CrossRef]
- Rujiwatra, A. A selective preparation of phillipsite and sodalite from perlite. Mater. Lett. 2004, 58, 2012–2015. [Google Scholar] [CrossRef]
- Burriesci, N.; Crisafulli, M.L.; Saija, L.M. Hydrothermal synthesis of zeolites from rhyolitic pumice of different geological origins. Mater. Lett. 1983, 2, 74–78. [Google Scholar] [CrossRef]
- Garcia, G.; Cardenasa, E.; Cabrera, S.; Hedlund, J.; Mouzon, J. Synthesis of zeolite Y from diatomite as silica source. Microporous Mesoporous Mater. 2016, 219, 29–37. [Google Scholar] [CrossRef]
- Querol, X.; Moreno, N.; Umaña, J.C.; Alastuey, A.; Hernández, E.; López-Soler, A.; Plana, F. Synthesis of zeolites from coal fly ash: An overview. Int. J. Coal Geol. 2002, 50, 413–423. [Google Scholar] [CrossRef]
- Kunecki, P.; Panek, R.; Wdowin, M.; Bień, T.; Franus, W. Influence of the fly ash fraction after grinding process on the hydrothermal synthesis efficiency of Na-A, Na-P1, Na-X and sodalite zeolite types. Int. J. Coal Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Ojha, K.; Pradhan, N.C.; Samanta, A.N. Zeolite from fly ash: Synthesis and characterization. Bull. Mater. Sci. 2004, 27, 555–564. [Google Scholar] [CrossRef]
- Prasetyoko, D.; Ramli, Z.; Endud, S.; Hamdan, H.; Sulikowski, B. Conversion of rice husk ash to zeolite beta. Waste Manag. 2006, 26, 1173–1179. [Google Scholar] [CrossRef] [PubMed]
- Król, M.; Mozgawa, W.; Morawska, J.; Pichór, W. Spectroscopic investigation of hydrothermally synthesized zeolites from expanded perlite. Microporous Mesoporous Mater. 2014, 196, 216–222. [Google Scholar] [CrossRef]
- Barrer, R.M.; White, E.A.D. The hydrothermal chemistry of silicates. Part II. Synthetic crystalline sodium aluminosilicate. J. Chem. Soc. (Resumed) 1952, 286, 1561–1571. [Google Scholar] [CrossRef]
- Park, M.; Choi, C.L.; Lim, W.T.; Kim, M.C.; Choi, J.; Heo, N.H. Molten-salt method for the synthesis of zeolitic materials: I. Zeolite formation in alkaline molten-salt system. Microporous Mesoporous Mater. 2000, 37, 81–89. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Xu, D.; Han, L.; Niu, D.; Tian, B.; Zhang, J.; Zhang, L.; Wu, W. Removal of ammonium from aqueous solutions using zeolite synthesized from fly ash by a fusion method. Desalination 2011, 271, 111–121. [Google Scholar] [CrossRef]
- Takeda, H.; Hashimoto, S.; Yokoyama, H.; Iwamoto, Y.; Iwamoto, Y. Characterization of zeolite in zeolite-geopolymer hybrid bulk materials derived from kaolinitic clays. Materials 2013, 6, 1767–1778. [Google Scholar] [CrossRef]
- Król, M.; Minkiewicz, J.; Mozgawa, W. IR spectroscopy studies of zeolites in geopolymeric materials derived from kaolinite. J. Mol. Struct. 2016, 1126, 200–206. [Google Scholar] [CrossRef]
- Rożek, P.; Król, M.; Mozgawa, W. Geopolymer-zeolite composites: A review. J. Clean. Prod. 2019, 230, 557–579. [Google Scholar] [CrossRef]
- Querol, X.; Alastuey, A.; Lopez-Soler, A.; Plana, P.; Andres, J.M.; Juan, R.; Perrer, P.; Ruiz, C. A fast method for recycling fly ash: Microwave-assisted zeolite synthesis. Env. Sci. Technol. 1997, 31, 2527–2533. [Google Scholar] [CrossRef]
- Tanaka, H.; Eguchi, H.; Fujimoto, S.; Hino, R. Two-step process for synthesis of a single phase Na–A zeolite from coal fly ash by dialysis. Fuel 2006, 85, 1329–1334. [Google Scholar] [CrossRef]
- Cundy, C.S.; Cox, P.A. The hydrothermal synthesis of zeolites: Precursors, intermediates and reaction mechanism. Microporous Mesoporous Mater. 2005, 82, 1–78. [Google Scholar] [CrossRef]
- Gonthier, S.; Gora, L.; Güray, I.; Thompson, R.W. Further comments on the role of autocatalytic nucleation in hydrothermal zeolite syntheses. Zeolites 1993, 13, 414–418. [Google Scholar] [CrossRef]
- Lonin, A.Y.; Levenets, V.V.; Neklyudov, I.M.; Shchur, A.O. The usage of zeolites for dynamic sorption of cesium from wastewaters of nuclear power plants. J. Radioanal. Nucl. Chem. 2015, 303, 831–836. [Google Scholar] [CrossRef]
- Abdel Moamen, O.A.; Ismail, I.M.; Abdelmonem, N.; Abdel Rahmana, R.O. Factorial design analysis for optimizing the removal of cesium and strontium ions on synthetic nano-sized zeolite. J. Taiwan Inst. Chem. Eng. 2015, 55, 133–144. [Google Scholar] [CrossRef]
- He, K.; Chen, Y.; Tang, Z.; Hu, Y. Removal of heavy metal ions from aqueous solution by zeolite synthesized from fly ash. Environ. Sci. Pollut. Res. 2016, 3, 2778–2788. [Google Scholar] [CrossRef]
- Kozera-Sucharda, B.; Gworek, B.; Kondzielski, I. The simultaneous removal of zinc and cadmium from multicomponent aqueous solutions by their sorption onto selected natural and synthetic zeolites. Minerals 2020, 10, 343. [Google Scholar] [CrossRef]
- Bandura, L.; Franus, M.; Józefaciuk, G.; Franus, W. Synthetic zeolites from fly ash as effective mineral sorbents for land-based petroleum spills cleanup. Fuel 2015, 147, 100–107. [Google Scholar] [CrossRef]
- Nizami, A.S.; Ouda, O.K.M.; Rehan, M.; El-Maghraby, A.M.O.; Gardy, J.; Hassanpour, A.; Kumar, S.; Ismail, I.M.I. The potential of Saudi Arabian natural zeolites in energy recovery technologies. Energy 2016, 108, 162–171. [Google Scholar] [CrossRef]
- Ward, J.W. The nature of active sites on zeolites: I. The decationated Y zeolite. J. Catal. 1967, 9, 225–236. [Google Scholar] [CrossRef]
- Corma, A. State of the art and future challenges of zeolites as catalysts. J. Catal. 2003, 216, 298–312. [Google Scholar] [CrossRef]


© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Król, M. Natural vs. Synthetic Zeolites. Crystals 2020, 10, 622. https://doi.org/10.3390/cryst10070622
Król M. Natural vs. Synthetic Zeolites. Crystals. 2020; 10(7):622. https://doi.org/10.3390/cryst10070622
Chicago/Turabian StyleKról, Magdalena. 2020. "Natural vs. Synthetic Zeolites" Crystals 10, no. 7: 622. https://doi.org/10.3390/cryst10070622
APA StyleKról, M. (2020). Natural vs. Synthetic Zeolites. Crystals, 10(7), 622. https://doi.org/10.3390/cryst10070622




