Inhibition of Poly(ethylenediaminetetraacetic acid-diethanolamine) on Deposition of Calcium Sulfate Crystal in Simulated Industrial Water
Abstract
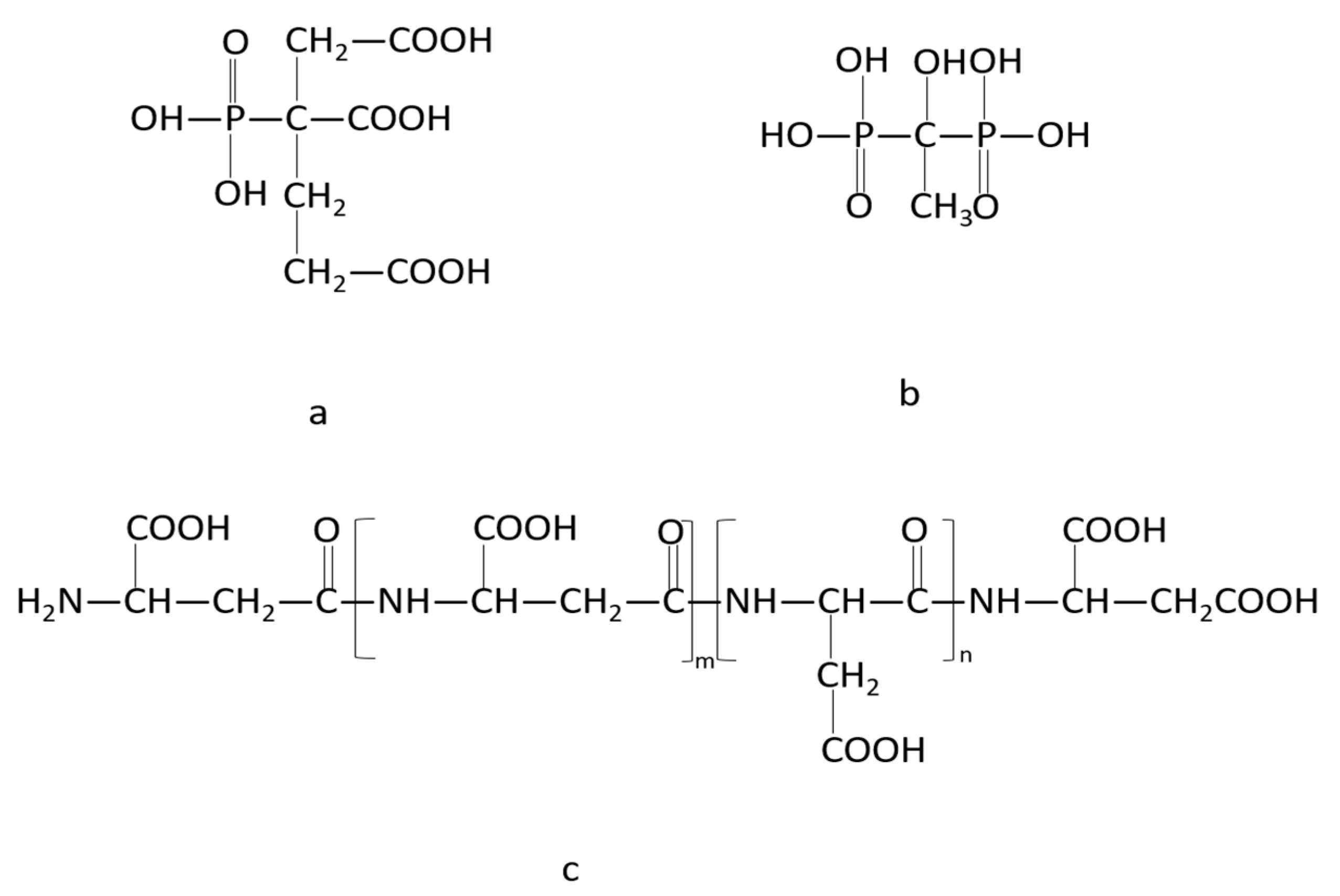
1. Introduction
2. Materials and Methods
2.1. Materials
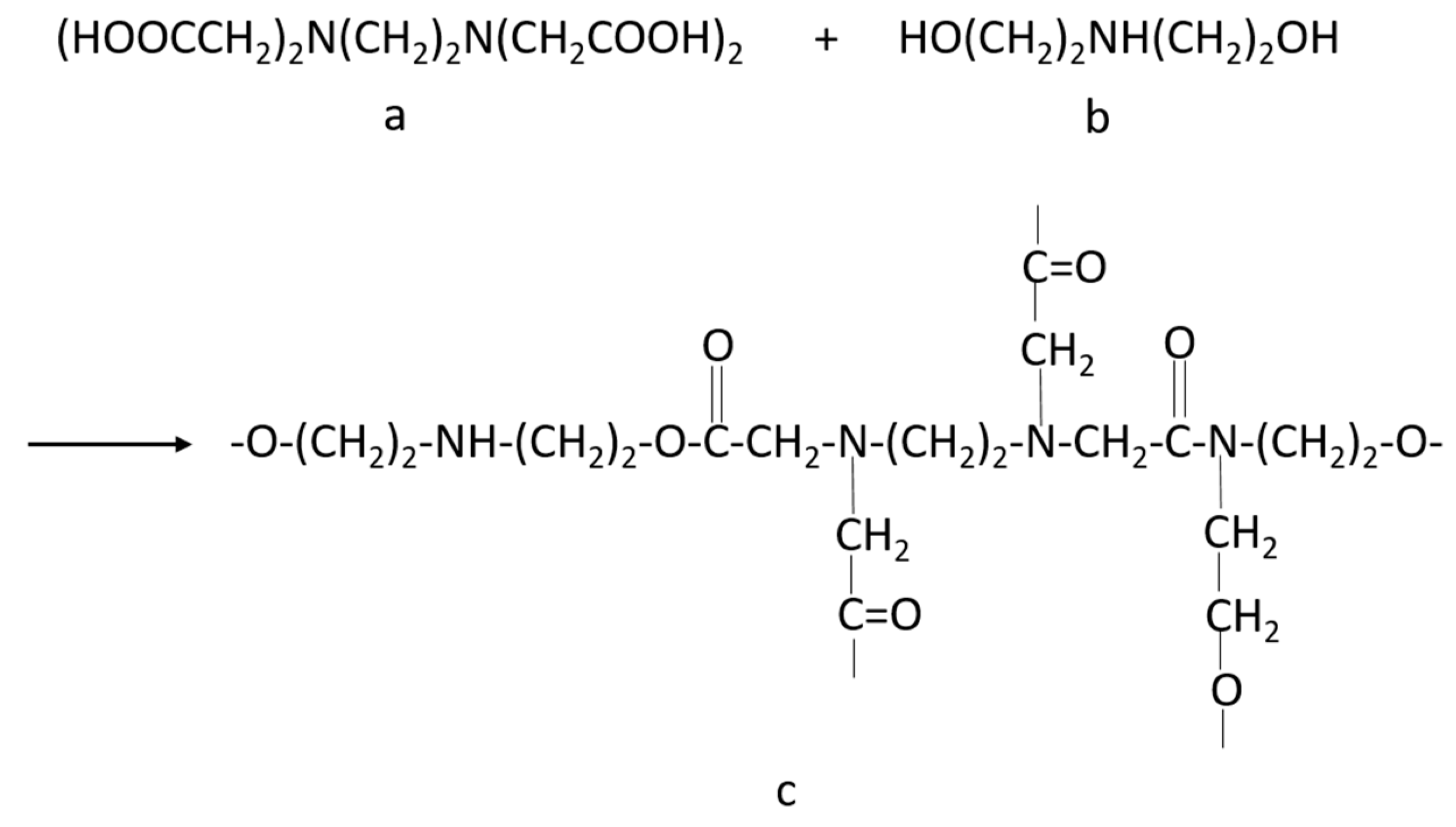
2.2. Synthesis of Polymer
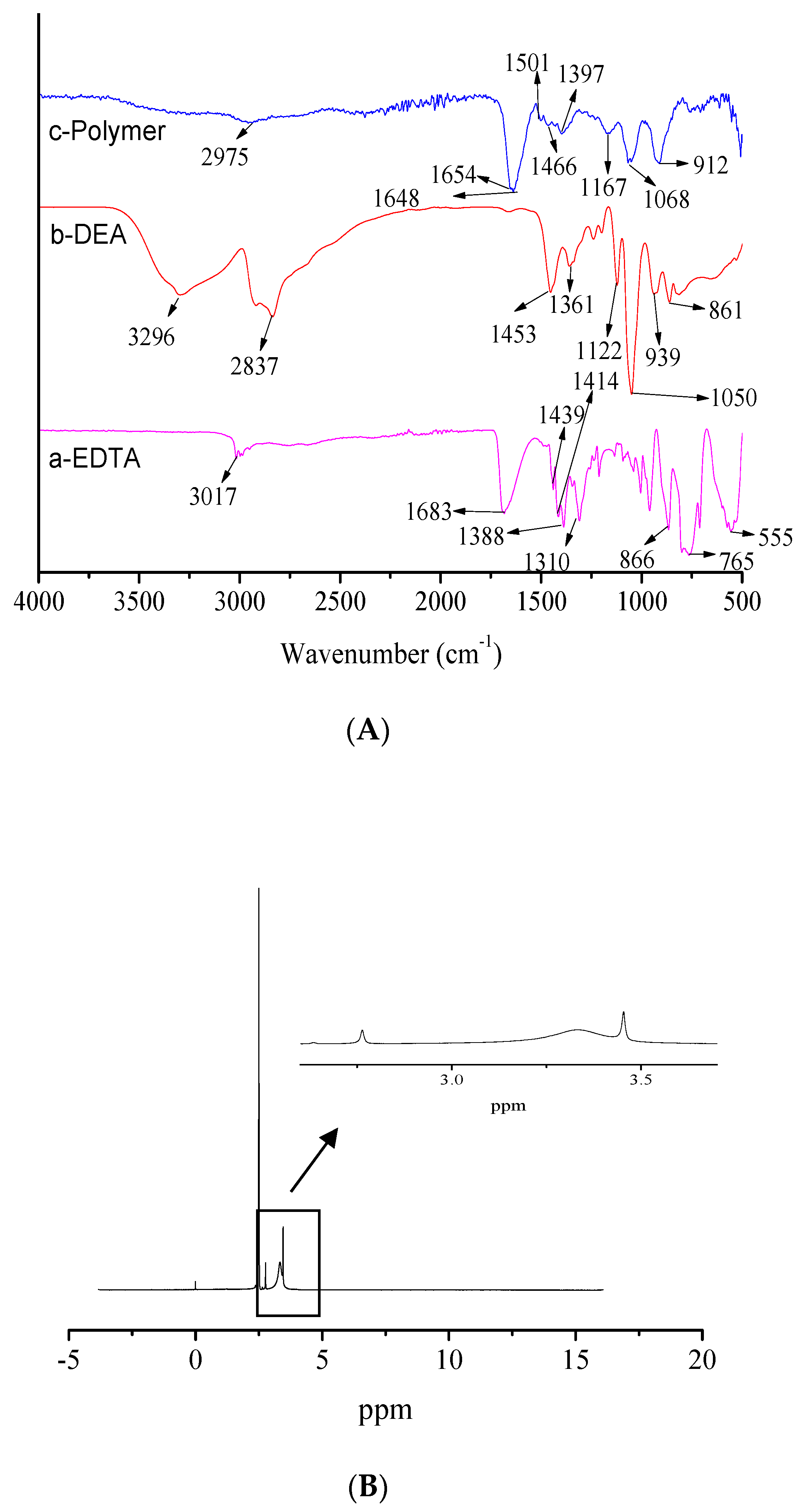
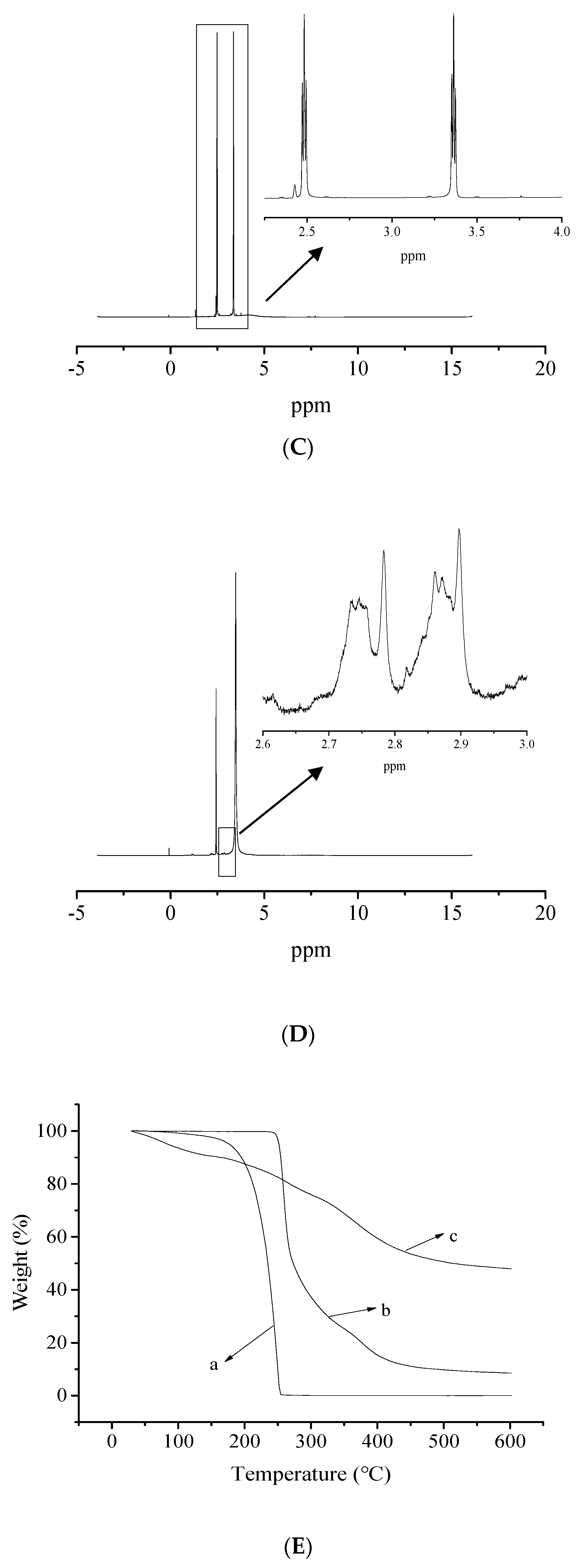
2.3. Characterization of the Prepared Polymers
2.4. Evaluation of Polymer Inhibition Performance of Precipitation
2.5. The Morphology of the Calcium Sulfate Crystal Precipitation
3. Results and Discussion
3.1. Structure of Prepared Polymers
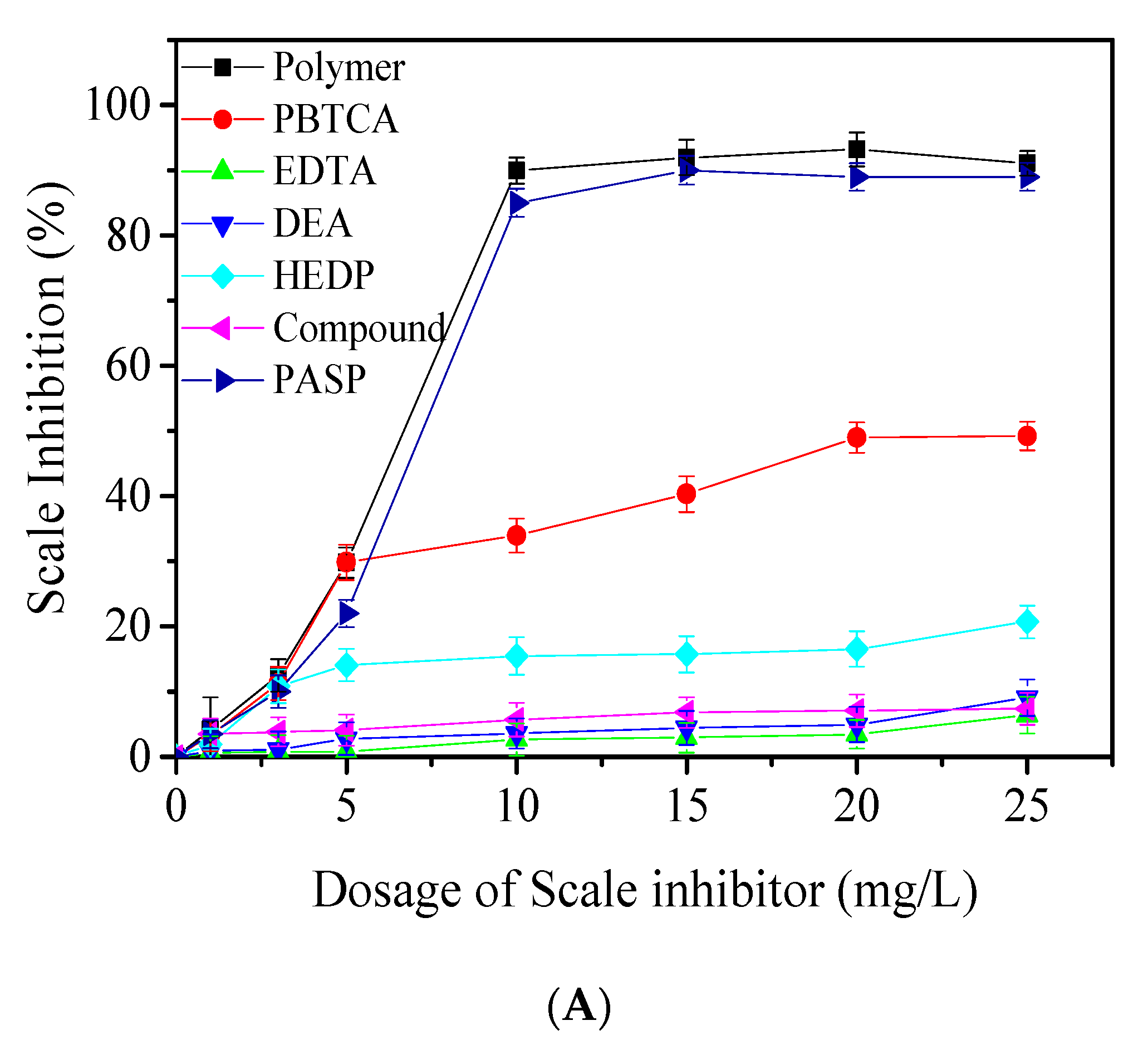
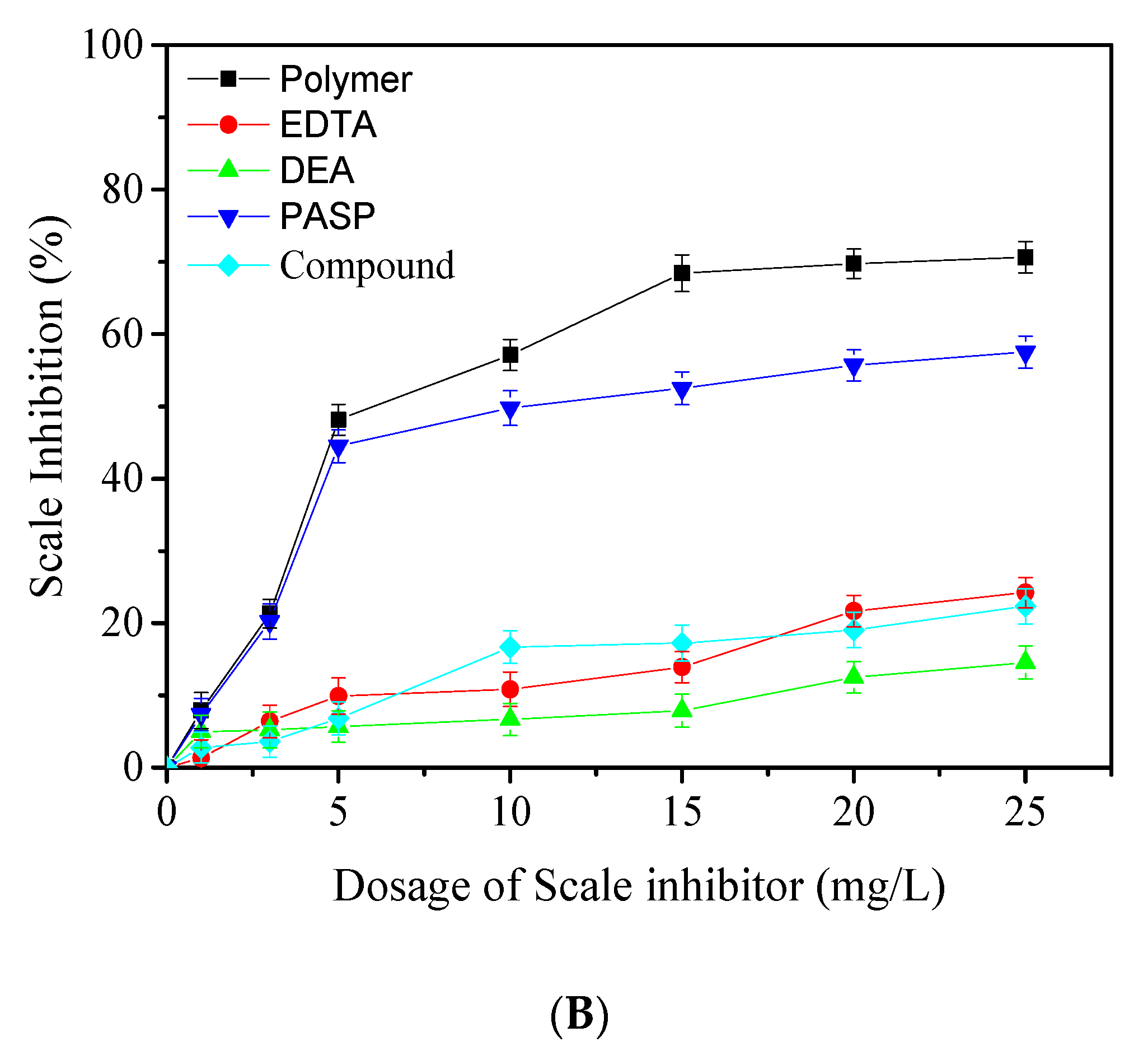
3.2. Precipitation Inhibition Performance of Prepared Polymers
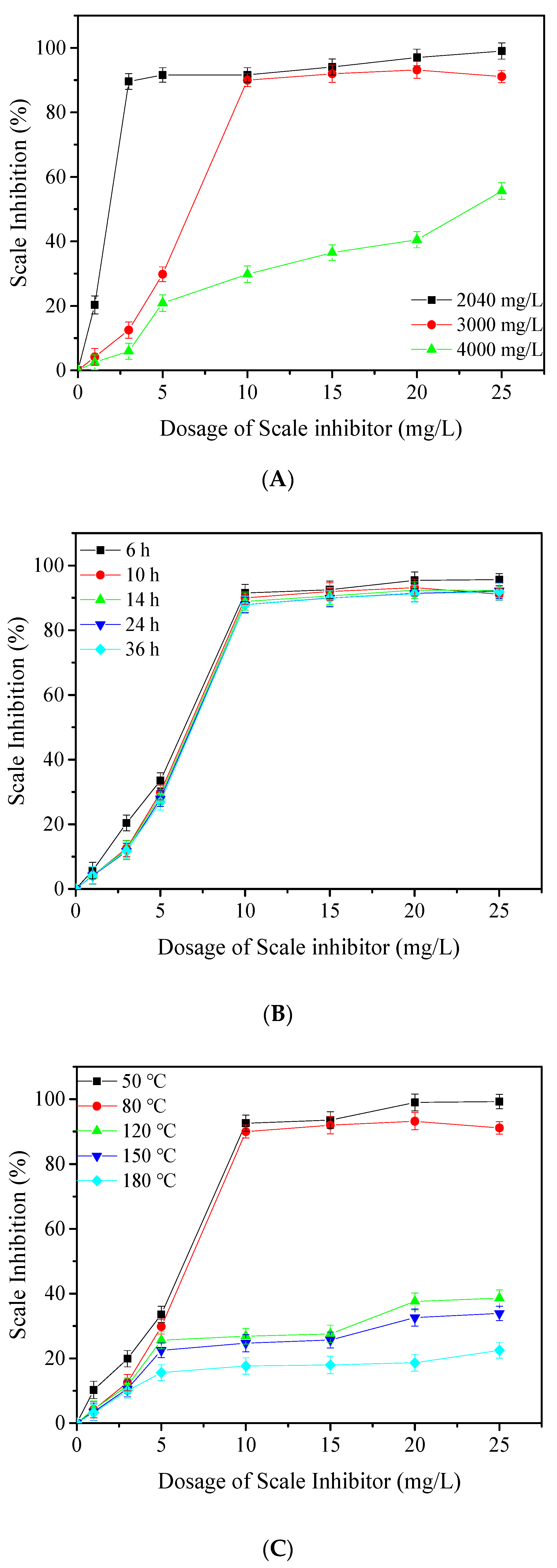
3.3. Different Conditions of Inhibition Test
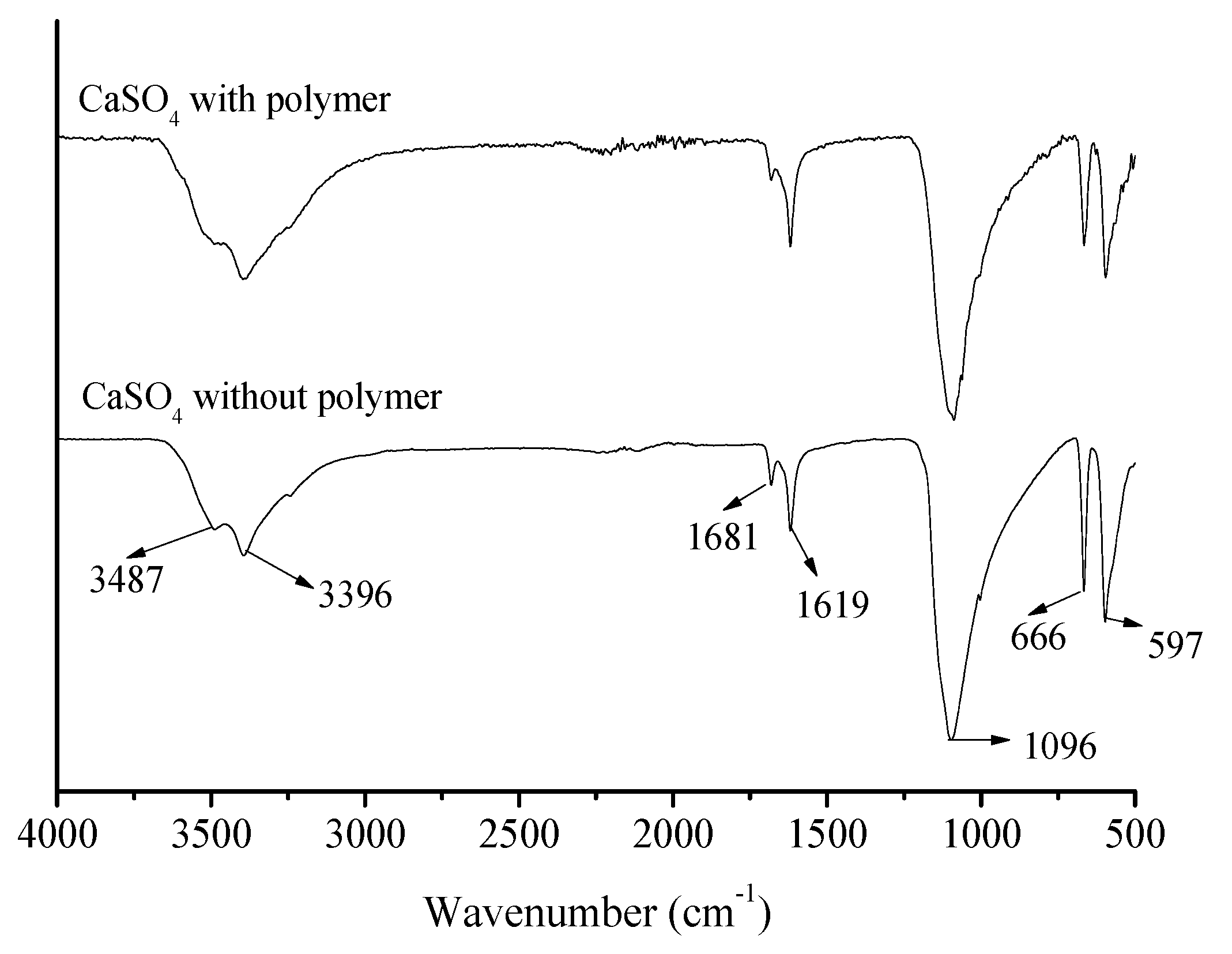
3.4. Characterization and Analysis of Calcium Sulfate Precipitate
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rahmani, K.; Jadidian, R.; Haghtalab, S. Evaluation of inhibitors and biocides on the corrosion, scaling and biofouling control of carbon steel and copper-nickel alloys in a power plant cooling water system. Desalination 2015, 393, 174–185. [Google Scholar] [CrossRef]
- Kavitha, A.L.; Vasudevan, T.; Prabu, H.G. Evaluation of synthesized antiscalants for cooling water system application. Desalination 2011, 268, 38–45. [Google Scholar] [CrossRef]
- Touir, R.; Cenoui, M.; Bakri, M.E.; Touhami, M.E. Sodium gluconate as corrosion and scale inhibitor of ordinary steel in simulated cooling water. Corros. Sci. 2008, 50, 1530–1537. [Google Scholar] [CrossRef]
- Chauhan, K.; Kumar, R.; Kumar, M.; Sharma, P.; Chauhan, G.S. Modified pectin-based polymers as green antiscalants for calcium sulfate scale inhibition. Desalination 2012, 305, 31–37. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, Z.M.; Wang, B.B.; He, J.J. Scale inhibition performance of sodium carboxymethyl cellulose on heat transfer surface at various temperatures: Experiments and molecular dynamics simulation. Int. J. Heat Mass Trans. 2019, 141, 457–463. [Google Scholar] [CrossRef]
- Liu, F.; Lu, X.; Yang, W.; Lu, J.J.; Zhong, H.Y.; Chang, X.; Zhao, C.C. Optimizations of inhibitors compounding and applied conditions in simulated circulating cooling water system. Desalination 2013, 313, 18–27. [Google Scholar] [CrossRef]
- Ou, H.H.; Tran, Q.T.P.; Lin, P.H. A synergistic effect between gluconate and molybdate on corrosion inhibition of recirculating cooling water systems. Corros. Sci. 2018, 133, 231–239. [Google Scholar] [CrossRef]
- Zhang, S.P.; Qu, H.J.; Yang, Z.; Fu, C.E.; Tian, Z.Q.; Yang, W.B. Scale inhibition performance and mechanism of sulfamic/amino acids modified polyaspartic acid against calcium sulfate. Desalination 2017, 419, 152–159. [Google Scholar] [CrossRef]
- Rahardianto, A.; Shih, W.Y.; Lee, R.W.; Cohen, Y. Diagnostic characterization of gypsum scale formation and control in RO membrane desalination of brackish water. J. Membr. Sci. 2006, 279, 655–668. [Google Scholar] [CrossRef]
- Fu, L.P.; Lv, J.; Zhou, L.; Li, Z.D.; Tang, M.J.; Li, J.B. Study on corrosion and scale inhibition mechanism of polyaspartic acid grafted β-cyclodextrin. Mater. Lett. 2020, 264, 127–276. [Google Scholar]
- Shakkthivel, P.; Vasudevan, T. Acrylic acid-diphenylamine sulphonic acid copolymer threshold inhibitor for sulphate and carbonate scales in cooling water systems. Desalination 2006, 197, 179–189. [Google Scholar] [CrossRef]
- Shi, W.Y.; Ding, C.; Yan, J.L.; Han, X.Y.; Lv, Z.M.; Lei, W.; Xia, M.Z.; Wang, F.Y. Molecular dynamics simulation for interaction of PESA and acrylic copolymers with calcite crystal surfaces. Desalination 2012, 291, 8–14. [Google Scholar] [CrossRef]
- Senthilmurugan, B.; Ghosh, B.; Kundu, S.S.; Haroun, M.; Kameshwari, B. Maleic acid-based scale inhibitors for calcium sulfate scale inhibition in high temperature application. J. Petrol. Sci. Eng. 2010, 75, 189–195. [Google Scholar] [CrossRef]
- Amjad, Z.; Koutsoukos, P.G. Evaluation of maleic acid-based polymers as scale inhibitors and dispersants for industrial water applications. Desalination 2014, 335, 55–63. [Google Scholar] [CrossRef]
- Wang, T.; Zhao, C.H.; Xu, J.; Sun, D.J. Enhanced Ca2+ binding with sulfonic acid type polymers at increased temperatures. Colloids Surf. A Physicochem. Eng. Asp. 2013, 417, 256–263. [Google Scholar] [CrossRef]
- Yang, L.; Yang, W.Z.; Xu, B.; Yin, X.S.; Chen, Y.; Liu, Y.; Ji, Y.; Huan, Y. Synthesis and scale inhibition performance of a novel environmentally friendly and hydrophilic terpolymer inhibitor. Desalination 2017, 416, 166–174. [Google Scholar] [CrossRef]
- Jensen, M.K.; Kelland, M.A. A new class of hyper branched polymeric scale inhibitors. J. Petrol. Sci. Eng. 2012, 94, 66–72. [Google Scholar] [CrossRef]
- Ou, H.H.; Hsieh, L.H.C. A synergistic effect of sodium gluconate and 2-phosphonobutane-1, 2, 4-tricarboxylic acid on the inhibition of CaCO3 scaling formation. Powder Technol. 2016, 302, 160–167. [Google Scholar] [CrossRef]
- Khormali, A.; Sharifov, A.R.; Torba, D.I. Increasing efficiency of calcium sulfate scale prevention using a new mixture of phosphonate scale inhibitors during water flooding. J. Petrol. Sci. Eng. 2018, 164, 245–258. [Google Scholar] [CrossRef]
- Klepetsanis, P.G.; Koutsoukos, P.G. Kinetics of calcium sulfate formation in aqueous media: Effect of organ phosphorus compounds. J. Cryst. Growth 1998, 193, 156–163. [Google Scholar] [CrossRef]
- Migahed, M.A.; Rashwan, S.M.; Kamel, M.M.; Habib, R.E. Synthesis, characterization of polyaspartic acid-glycine adduct and evaluation of their performance as scale and corrosion inhibitor in desalination water plants. J. Mol. Liq. 2016, 224, 849–858. [Google Scholar] [CrossRef]
- Gao, Y.H.; Fan, L.H.; Ward, L.; Liu, Z.F. Synthesis of polyaspartic acid derivative and evaluation of its corrosion and scale inhibition performance in seawater utilization. Desalination 2015, 365, 220–226. [Google Scholar] [CrossRef]
- Zhang, Y.; Yin, H.Q.; Zhang, Q.S.; Li, Y.Z.; Yao, P.J. Synthesis and characterization of novel polyaspartic acid/urea graft copolymer with acylamino group and its scale inhibition performance. Desalination 2016, 395, 92–98. [Google Scholar] [CrossRef]
- Huang, H.H.; Yao, Q.; Jiao, Q.; Liu, B.L.; Chen, H.L. Polyepoxysuccinic acid with hyper-branched structure as an environmentally friendly scale inhibitor and its scale inhibition mechanism. J. Saudi Chem. Soc. 2019, 23, 61–74. [Google Scholar] [CrossRef]
- Liu, D.; Dong, W.B.; Li, F.T.; Hui, F.; Lédion, J. Comparative performance of polyepoxysuccinic acid and polyaspartic acid on scaling inhibition by static and rapid controlled precipitation methods. Desalination 2012, 304, 1–10. [Google Scholar] [CrossRef]
- Zhao, Y.Z.; Jia, L.L.; Liu, K.Y.; Gao, P.; Ge, H.H.; Fu, L.J. Inhibition of calcium sulfate scale by poly (citric acid). Desalination 2016, 392, 1–7. [Google Scholar] [CrossRef]
- Evstatiev, R.; Cervenka, A.; Lang, M.; Frick, A.P.; Gmainer, C.; Krnjic, A.; Khare, V.; Gasche, C. EDTA compounds, as used in food additives, aggravate intestinal inflammation and drive tumorigenesis in a mouse model of colitis-associated cancer. Gastroenterology 2017, 152, S735. [Google Scholar] [CrossRef]
- Evstatiev, R.; Deim, G.; Khare, V.; Gmainer, C.; Krnjic, A.; Cervenka, A.; Lang, M.; Baumgartner, M.; Frick, A.P.; Gasche, C. The food additive EDTA increases intestinal inflammation and colorectal carcinogenesis by disrupting the intestinal epithelial barrier. Z. Gastroenterol. 2019, 57, V05. [Google Scholar]
- Ying, C.; Chen, X.S.; Liang, Y.N. Synthesis of polyaspartic acid/graphene oxide grafted copolymer and evaluation of scale inhibition and dispersion performance. Diam. Relat. Mater. 2020, 108, 107949. [Google Scholar]
- Dayarathne, H.N.P.; Jeong, S.; Jang, A. Chemical-free scale inhibition method for seawater reverse osmosis membrane process: Air micro-nano bubbles. Desalination 2019, 461, 1–9. [Google Scholar] [CrossRef]
- Benecke, J.; Haas, M.; Baur, F.; Ernst, M. Investigating the development and reproducibility of heterogeneous gypsum scaling on reverse osmosis membranes using real-time membrane surface imaging. Desalination 2018, 428, 161–171. [Google Scholar] [CrossRef]
- Amjad, Z. Kinetic and morphological investigation of calcium sulfate dihydrate (gypsum) scale formation on heat exchanger surfaces in the presence of inhibitors. Int. J. Corros. Scale Inhib. 2017, 6, 276–290. [Google Scholar]
- Roomi, Y.A.; Hussein, K.F.; Riazi, M.R. Inhibition efficiencies of synthesized anhydride based polymers as scale control additives in petroleum production. J. Petrol. Sci. Eng. 2012, 81, 151–160. [Google Scholar] [CrossRef]
- Chaúque, E.F.C.; Ngila, J.C.; Ray, S.C.; Ndlwana, L. Degradation of methyl orange on Fe/Ag nanoparticles immobilized on polyacrylonitrile nanofibers using EDTA chelating agents. J. Environ. Manag. 2019, 236, 481–489. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Y.; Yao, Q.; Huang, J.; Liu, G.; Wang, H.; Cao, K.; Chen, Y.; Bu, Y.; Wu, W.; et al. Double-hydrophilic polyether antiscalant used as a crystal growth modifier of calcium scales in cooling-water systems. J. Appl. Polym. Sci. 2014, 131, 39792. [Google Scholar] [CrossRef]
- Li, H.Y.; Ma, W.; Wang, L.; Liu, R.; Wei, L.S.; Wang, Q. Inhibition of calcium and magnesium-containing scale by a new antiscalant polymer in laboratory tests and a field trial. Desalination 2006, 196, 237–247. [Google Scholar] [CrossRef]
- Oshchepkov, M.S.; Kamagurov, S.; Tkachenko, S.; Popov, K.; Ryabova, A. An Insight into the Mechanisms of the Scale Inhibition. A Case Study of a Novel Task-specific Fluorescent-tagged Scale Inhibitor Location on Gypsum Crystals. ChemNanoMat 2019, 5, 586–592. [Google Scholar] [CrossRef]
- Popov, K.; Rudakova, G.; Larchenko, V.; Tusheva, M.; Kamagurov, S.; Dikareva, J.; Kovaleva, N. A Comparative Performance Evaluation of Some Novel “Green” and Traditional Antiscalants in Calcium Sulfate Scaling. Adv. Mat. Sci. Eng. 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, D.P.; Lv, X.G.; Xu, Y.; Cui, Y.C. Synthesis of polyaspartic acid/3-amino-1H-1,2,4-triazole-5-carboxylic acid hydrate graft copolymer and evaluation of its corrosion inhibition and scale inhibition performance. Desalination 2013, 327, 32–38. [Google Scholar] [CrossRef]
- Inoue, M.; Hirasawa, I. The relationship between crystal morphology and XRD peak intensity on CaSO4·2H2O. J. Cryst. Growth 2013, 380, 169–175. [Google Scholar] [CrossRef]
- Silva, A.M.; Junot, D.O.; Caldas, L.V.; Souza, D.N. Structural, optical and dosimetric characterization of CaSO4: Tb, CaSO4: Tb, Ag and CaSO4: Tb, Ag (NP). J. Lumin. 2020, 224, 117286. [Google Scholar] [CrossRef]
- Böke, H.; Akkurt, S.; Özdemir, S.; Göktürk, E.H.; Saltik, E.N.C. Quantification of CaCO3–CaSO3 0.5 H2O–CaSO4·2H2O mixtures by FTIR analysis and its ANN model. Mater. Lett. 2004, 58, 723–726. [Google Scholar] [CrossRef]
- Wang, Y.B. Study on the surface properties and hydration meehanism of calcium sulfate whiskers. Ind. Miner. Process. 2008, 6, 1–4. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.-Q.; Liu, T.-R.; Sun, M.-M.; Zhao, Y.-Z.; Ge, H.-H. Inhibition of Poly(ethylenediaminetetraacetic acid-diethanolamine) on Deposition of Calcium Sulfate Crystal in Simulated Industrial Water. Crystals 2020, 10, 544. https://doi.org/10.3390/cryst10060544
Chen J-Q, Liu T-R, Sun M-M, Zhao Y-Z, Ge H-H. Inhibition of Poly(ethylenediaminetetraacetic acid-diethanolamine) on Deposition of Calcium Sulfate Crystal in Simulated Industrial Water. Crystals. 2020; 10(6):544. https://doi.org/10.3390/cryst10060544
Chicago/Turabian StyleChen, Jian-Qiu, Ting-Ran Liu, Miao-Miao Sun, Yu-Zeng Zhao, and Hong-Hua Ge. 2020. "Inhibition of Poly(ethylenediaminetetraacetic acid-diethanolamine) on Deposition of Calcium Sulfate Crystal in Simulated Industrial Water" Crystals 10, no. 6: 544. https://doi.org/10.3390/cryst10060544
APA StyleChen, J.-Q., Liu, T.-R., Sun, M.-M., Zhao, Y.-Z., & Ge, H.-H. (2020). Inhibition of Poly(ethylenediaminetetraacetic acid-diethanolamine) on Deposition of Calcium Sulfate Crystal in Simulated Industrial Water. Crystals, 10(6), 544. https://doi.org/10.3390/cryst10060544




