Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites
Abstract
1. Introduction
2. Experimental Procedures
2.1. Materials
2.1.1. Preparation and Characterization of AO Coating
2.1.2. Composites Coating
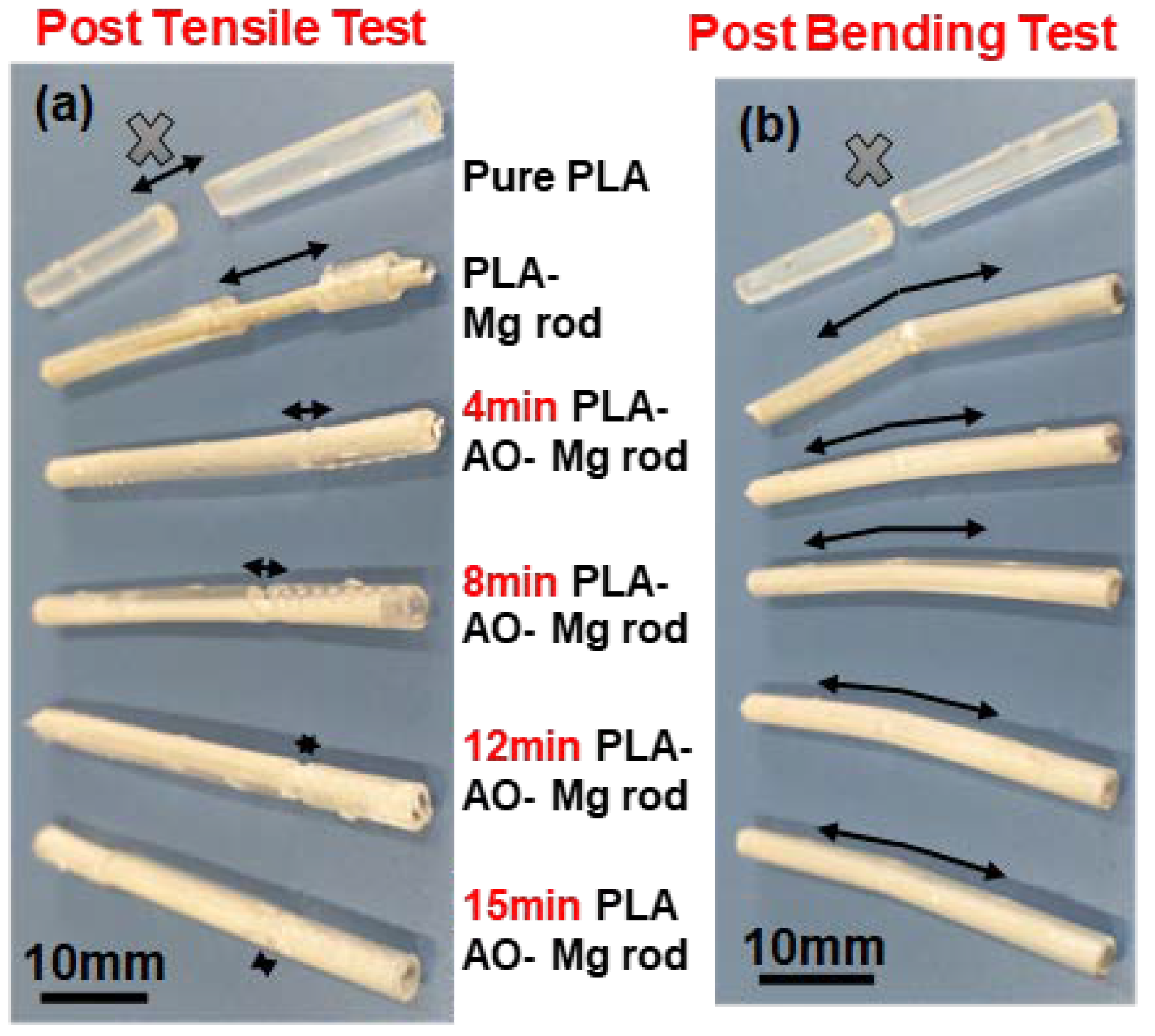
2.2. Mechanical Properties
2.3. In Vitro Degradation Test
3. Results and Discussions
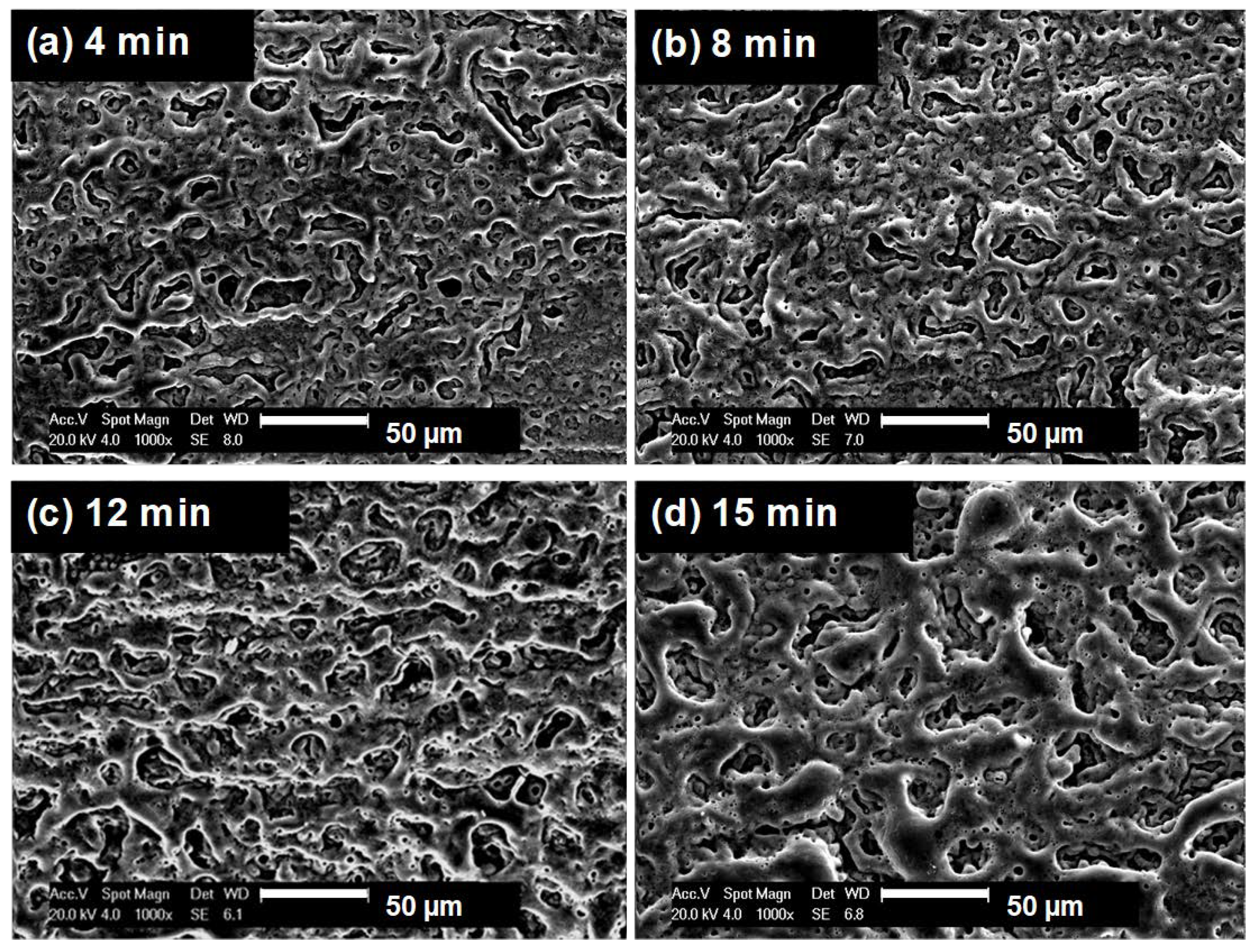
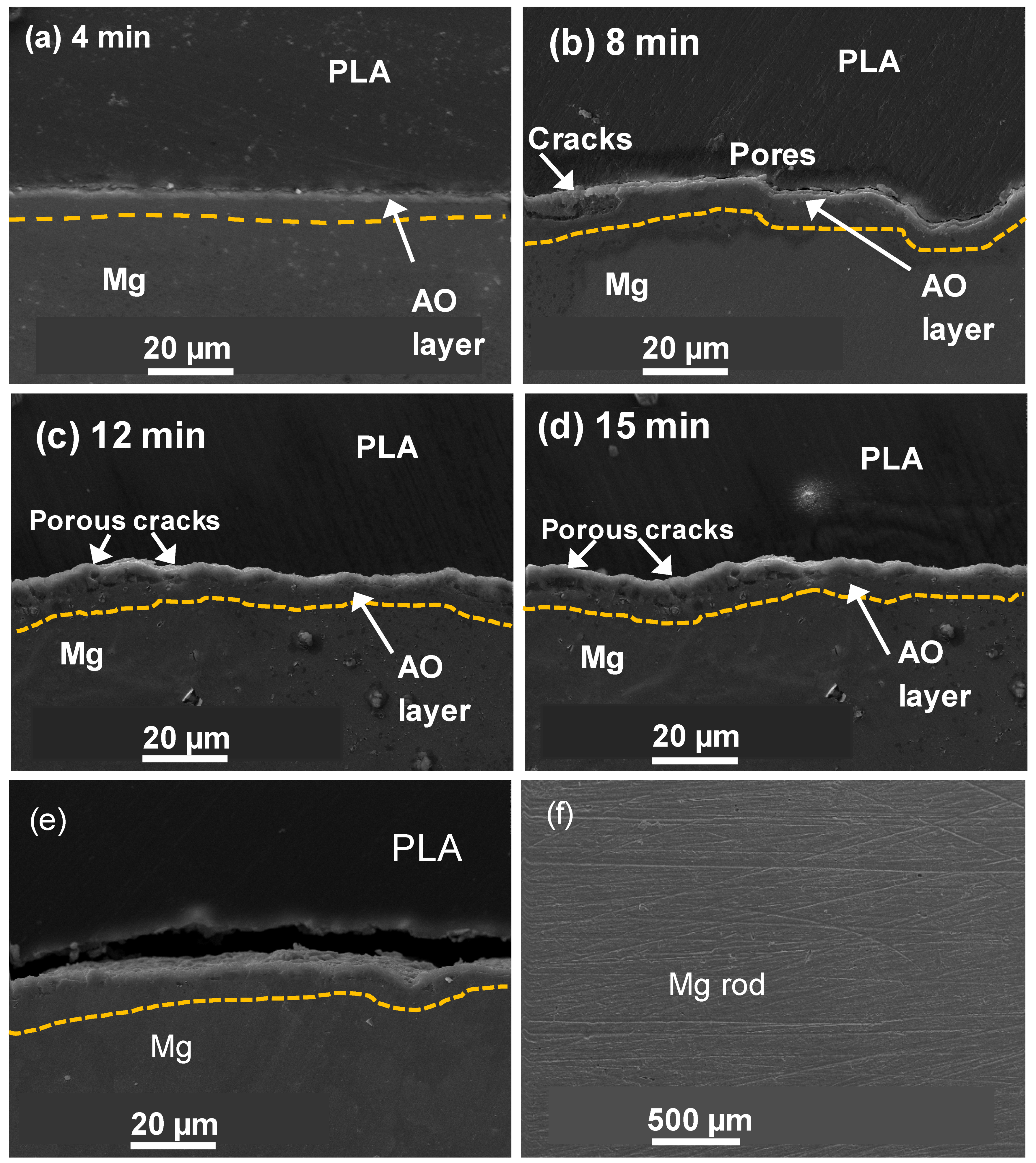
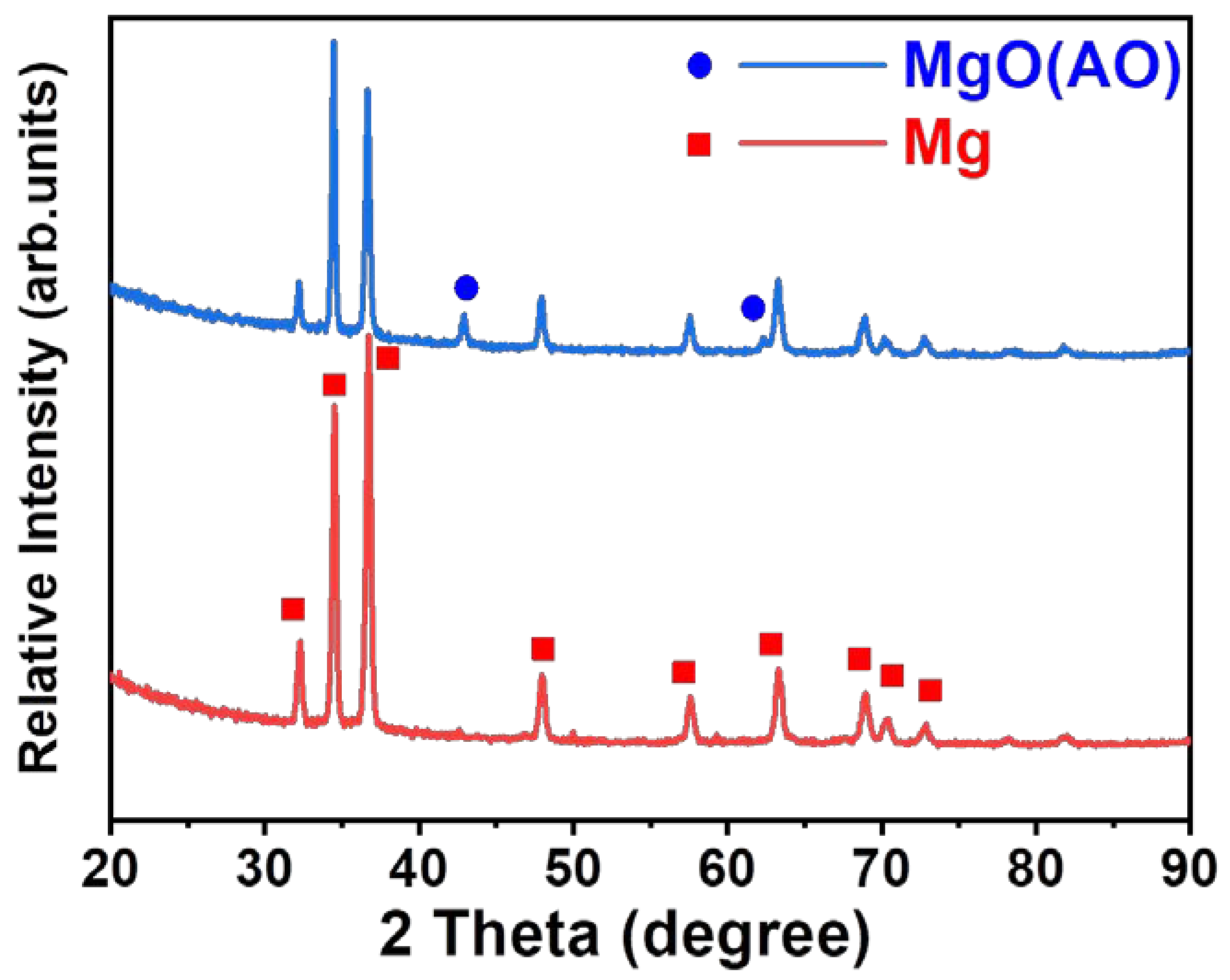
3.1. Microstructures
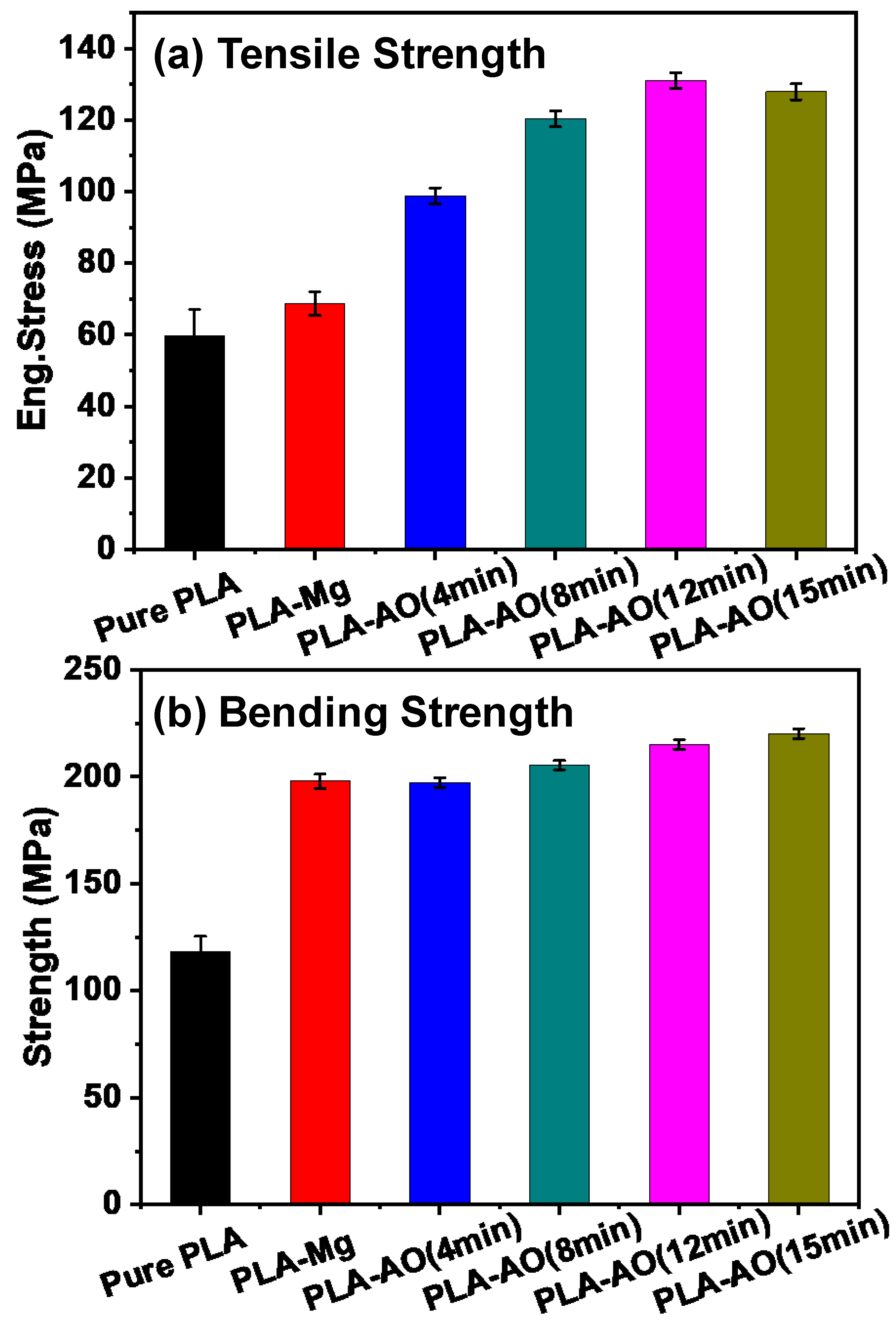
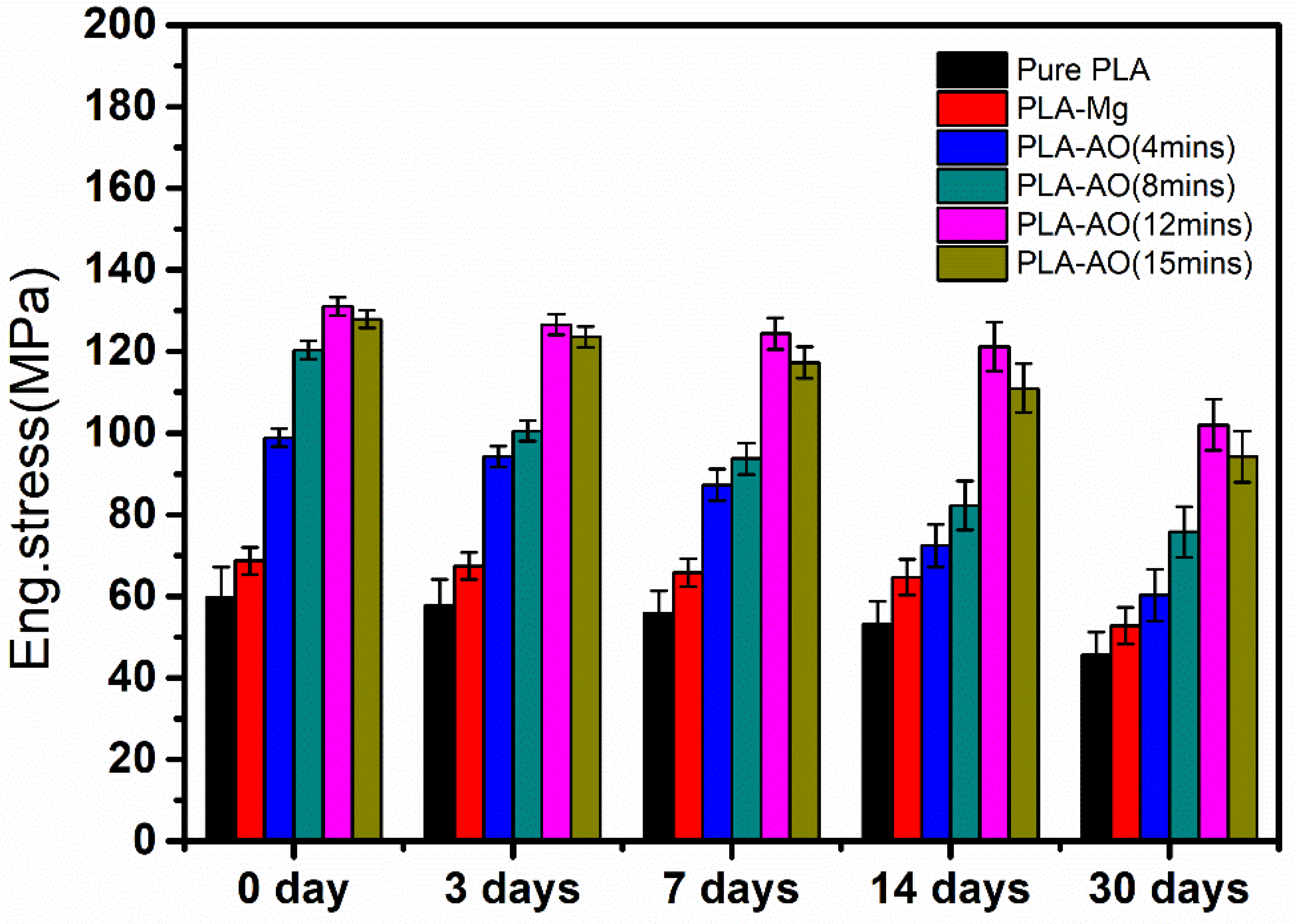
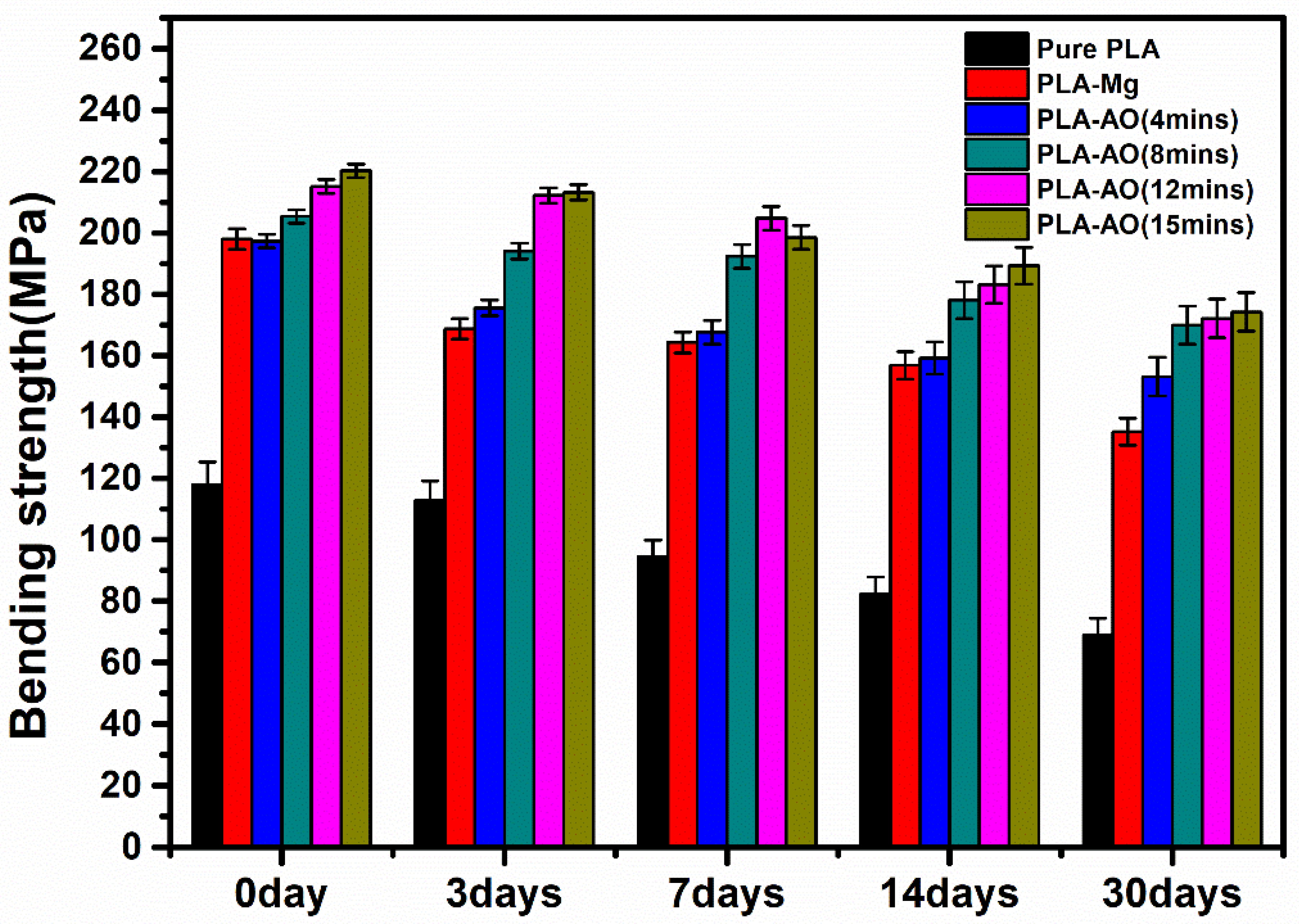
3.2. Mechanical Properties
3.3. Degradation Behavior
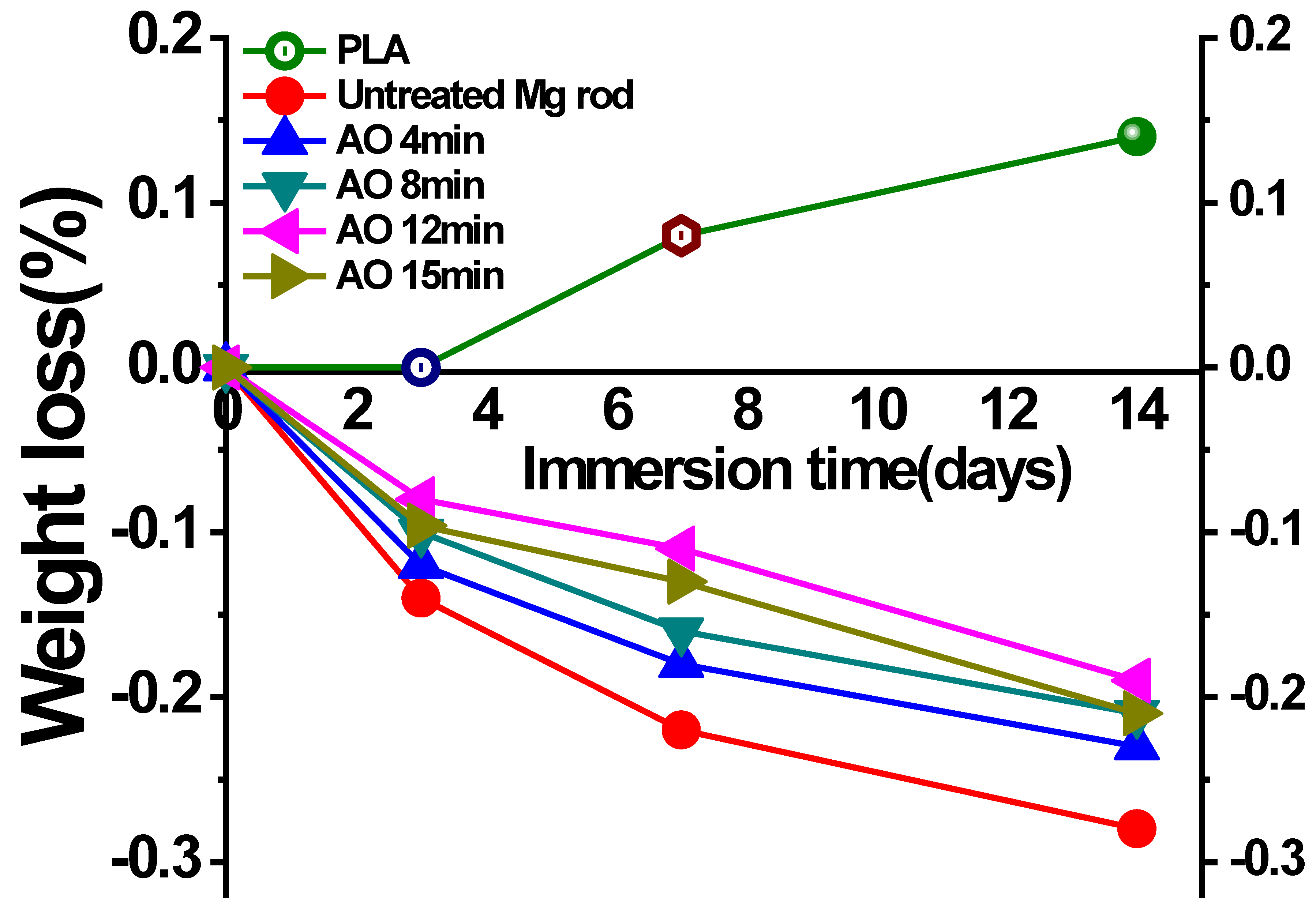
3.3.1. Weight Loss Test
3.3.2. Hydrogen Evolution
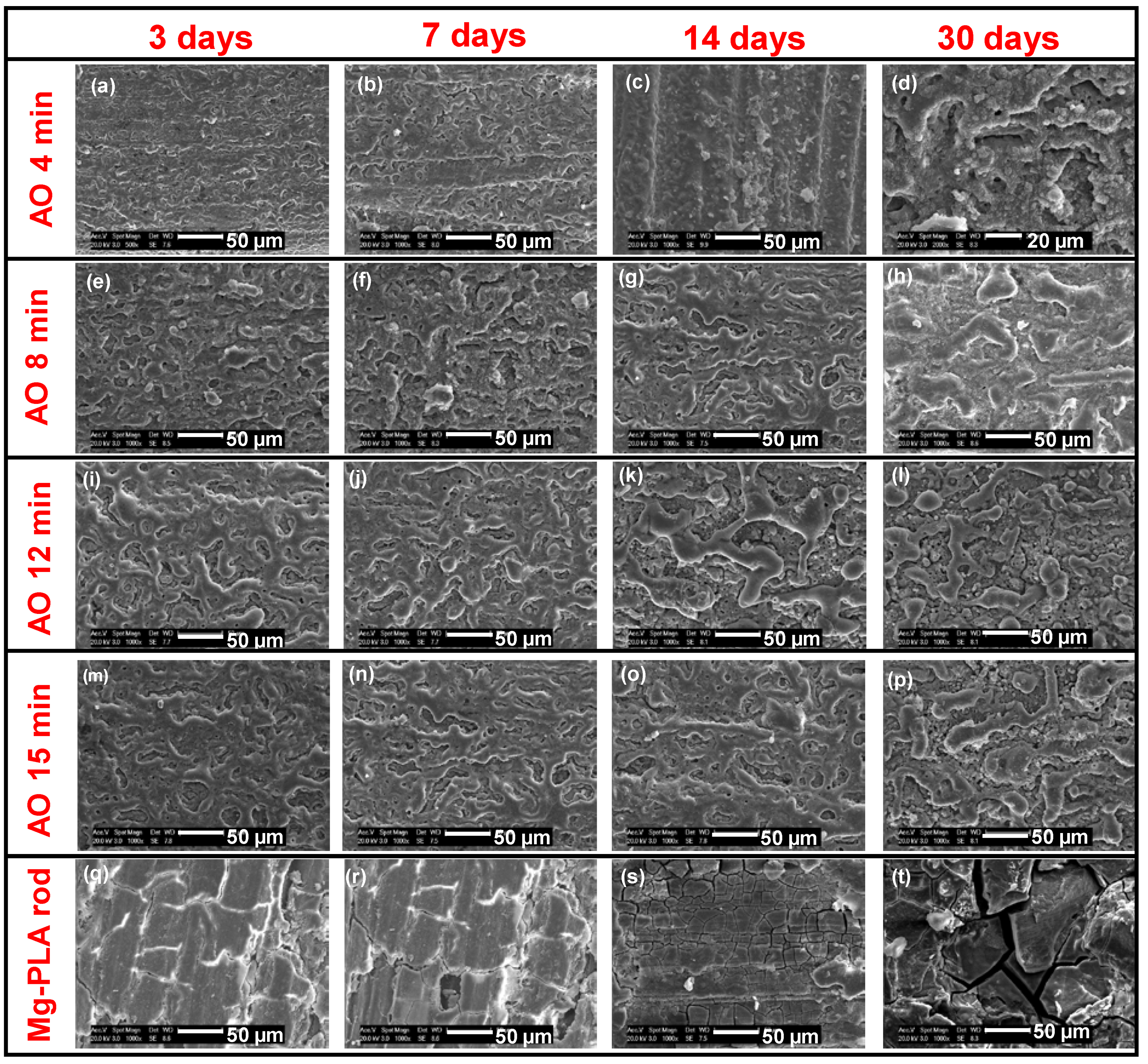
3.3.3. Morphologies
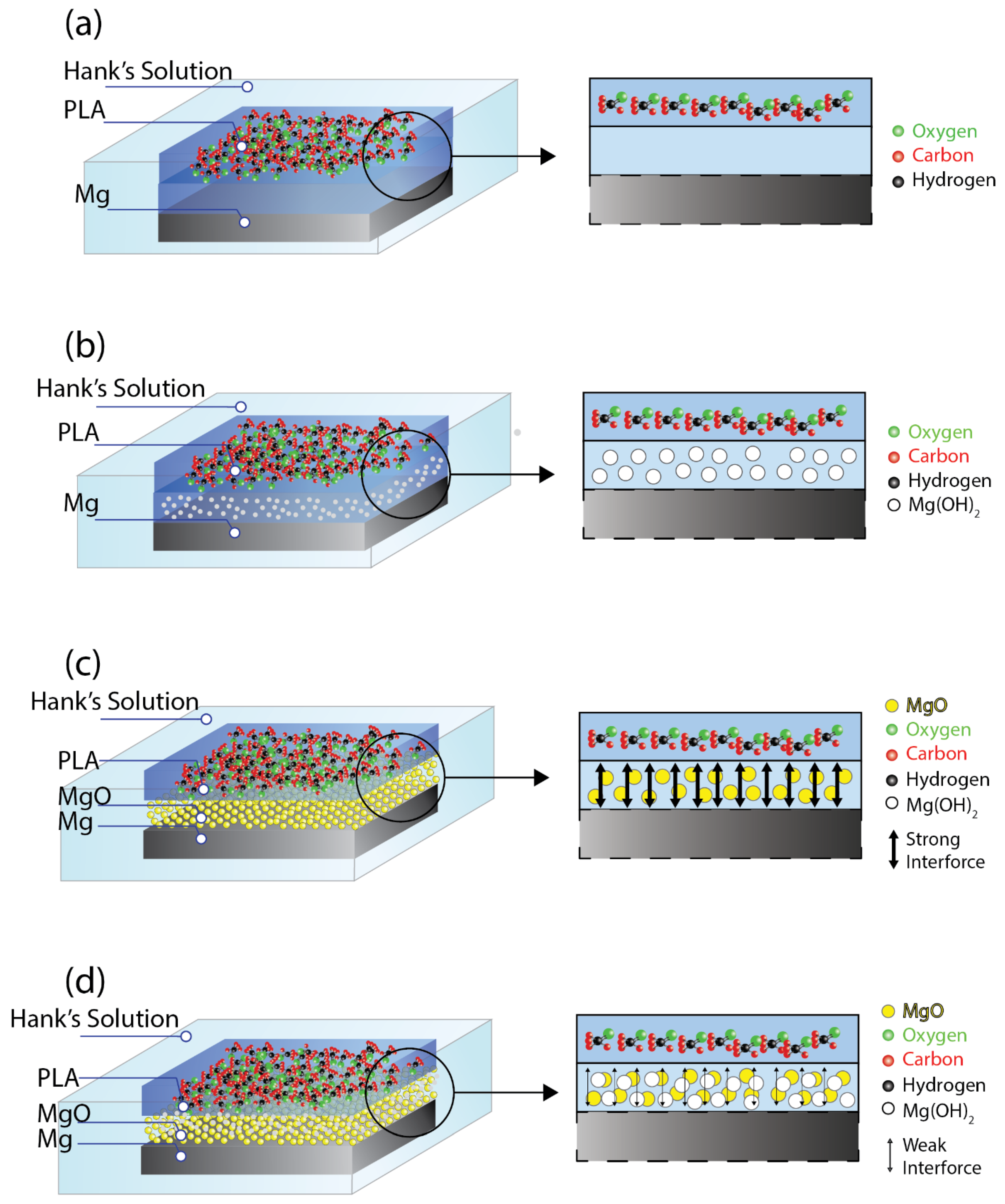
3.4. The Degradation Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moravej, M.; Mantovani, D. Biodegradable metals for cardiovascular stent application: Interests and new opportunities. Int. J. Mater. Sci. 2011, 12, 4250–4270. [Google Scholar] [CrossRef] [PubMed]
- Kunjukunju, S.; Roy, A.; Ramanathan, M.; Lee, B.; Candiello, J.E.; Kumta, P.N. A layer-by-layer approach to natural polymer-derived bioactive coatings on magnesium alloys. Acta Biomater. 2013, 9, 8690–8703. [Google Scholar] [CrossRef] [PubMed]
- Gray-Munro, J.E.; Seguin, C.; Strong, M. Influence of surface modification on the in vitro corrosion rate of magnesium alloy AZ31. J. Biomed. Mater. Res. A 2009, 91, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yamamoto, A. In vitro degradation of biodegradable polymer-coated magnesium under cell culture condition. Appl. Surf. Sci. 2012, 258, 6353–6358. [Google Scholar] [CrossRef]
- Ostrowski, N.; Lee, B.; Roy, A.; Ramanathan, M.; Kumta, P. Biodegradable poly(lactide-co-glycolide) coatings on magnesium alloys for orthopedic applications. J. Mater. Sci. Mater. Med. 2013, 24, 85–96. [Google Scholar] [CrossRef]
- Hornberger, H.; Virtanen, S.; Boccaccini, A.R. Biomedical coatings on magnesium alloys—A review. Acta Biomater. 2012, 8, 2442–2455. [Google Scholar] [CrossRef]
- Bakhsheshi-Rad, H.R.; Hamzah, E.; Ismail, A.F.; Sharer, Z.; Abdul-Kadir, M.R.; Daroonparvar, M.; Saud, S.N.; Medraj, M. Synthesis and corrosion behavior of a hybrid bioceramic-biopolymer coating on biodegradable Mg alloy for orthopaedic implants. J. Alloys Compd. 2015, 648, 1067–1071. [Google Scholar] [CrossRef]
- Li, J.N.; Cao, P.; Zhang, X.N.; Zhang, S.X.; He, Y.H. In vitro degradation and cell attachment of a PLGA coated biodegradable Mg–6Zn based alloy. J. Mater. Sci. 2010, 45, 6038–6045. [Google Scholar] [CrossRef]
- Anderson, J.M.; Shive, S.M. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 2012, 64, 72–82. [Google Scholar] [CrossRef]
- Li, L.; Ding, S.; Zhou, C. Preparation and degradation of PLA/chitosan composite materials. J. Appl. Polym. Sci. 2004, 91, 274–277. [Google Scholar] [CrossRef]
- Li, H.; Chang, J. pH-compensation effect of bioactive inorganic fillers on the degradation of PLGA. Compos. Sci. Technol. 2005, 65, 2226–2232. [Google Scholar] [CrossRef]
- Wu, Y.H.; Li, N.; Cheng, Y.; Zheng, Y.F.; Han, Y. In vitro study on biodegradable AZ31 magnesium alloy fibers reinforced PLGA composite. J. Mater. Sci. Technol. 2013, 29, 545–550. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Saris, N.E.L.; Mervaala, E.; Karppanen, H.; Khawaja, J.A.; Lewenstam, A. Magnesium: An update on physiological, clinical and analytical aspects. Clin. Chim. Acta 2000, 294, 1–26. [Google Scholar] [CrossRef]
- Yamasaki, Y.; Yoshida, Y.; Okazaki, M.; Shimazu, A.; Kubo, T.; Akagawa, Y.; Uchida, T. Action of FGMgCO3 Ap-collagen composite in promoting bone formation. Biomaterials 2003, 24, 4913–4920. [Google Scholar] [CrossRef]
- Mueller, W.D.; Nascimento, M.L.; Mele, D.; Lee, M.F. Critical discussion of the results from different corrosion studies of Mg and Mg alloys for biomaterial applications. Acta Biomater. 2010, 6, 1749–1755. [Google Scholar] [CrossRef]
- Xin, Y.; Hu, T.; Chu, P.K. Influence of test solutions on in vitro studies of biomedical magnesium alloys. J. Electrochem. Soc. 2010, 157, 238–243. [Google Scholar] [CrossRef]
- Mathew, A.P.; Oksman, K.; Sain, M. Mechanical properties of biodegradable composites from poly lactic acid (PLA) and microcrystalline cellulose (MCC). J. Appl. Polym. Sci. 2005, 97, 2014–2025. [Google Scholar] [CrossRef]
- Cifuentes, S.C.; Gavilán, R.; Lieblich, M.; Benavente, R.; González-Carrasco, J.L. In vitro degradation of biodegradable polylactic acid/magnesium composites: Relevance of Mg particle shape. Acta Biomater. 2016, 32, 348–357. [Google Scholar] [CrossRef]
- Song, Y.W.; Shan, D.Y.; Han, E.H. Electrodeposition of hydroxyapatite coating on AZ91D magnesium alloy for biomaterial application. Mater. Lett. 2008, 62, 3276–3279. [Google Scholar] [CrossRef]
- Li, L.; Gao, J.; Wang, Y. Evaluation of cyto-toxicity and corrosion behavior of alkali-heat-treated magnesium in simulated body fluid. Surf. Coat. Technol. 2004, 185, 92–98. [Google Scholar] [CrossRef]
- Chiu, K.Y.; Wong, M.H.; Cheng, F.T.; Man, H.C. Characterization and corrosion studies of fluoride conversion coating on degradable Mg implants. Surf. Coat. Technol. 2007, 202, 590–598. [Google Scholar] [CrossRef]
- Sealy, M.P.; Guo, Y.B. Surface integrity and process mechanics of laser shock peening of novel biodegradable magnesium–calcium (Mg–Ca) alloy. J. Mech. Behav. Biomed. Mater. 2010, 3, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Blawert, C.; Dietzel, W.; Ghali, E.; Song, G. Anodizing treatments for magnesium alloys and their effect on corrosion resistance in various environments. Adv. Eng. Mater. 2006, 8, 511–533. [Google Scholar] [CrossRef]
- Ren, M.; Cai, S.; Liu, T.; Huang, K.; Wang, X.; Zhao, H.; Niu, S.; Zhang, R.; Wu, X. Calcium phosphate glass/MgF2 double layered composite coating for improving the corrosion resistance of magnesium alloy. J. Alloys Compd. 2014, 591, 34–40. [Google Scholar] [CrossRef]
- Li, N.; Zheng, Y. Novel magnesium alloys developed for biomedical application: A review. J. Mater. Sci. Technol. 2013, 29, 489–502. [Google Scholar] [CrossRef]
- Li, X.; Chu, C.L.; Liu, L.; Liu, X.K.; Bai, J.; Guo, C.; Xue, F.; Lin, P.H.; Chu, P.K. Biodegradable poly-lactic acid based composite reinforced unidirectionally with high-strength magnesium alloy wires. Biomaterials 2015, 49, 135–144. [Google Scholar] [CrossRef]
- Butt, M.S.; Bai, J.; Wan, X.; Chu, C.; Xue, F.; Ding, H.; Zhou, G. Mechanical and degradation properties of biodegradable Mg strengthened poly-lactic acid composite through plastic injection molding. Mater. Sci. Eng. C 2017, 70, 141–147. [Google Scholar] [CrossRef]
- Cai, H.; Zhang, Y.; Meng, J.; Li, X.; Xue, F.; Chu, C.; Bai, J. Enhanced fully- biodegradable Mg/PLA composite rod: Effect of surface modification of Mg-2Zn wire on the interfacial bonding. Surf. Coat. Technol. 2018, 350, 722–731. [Google Scholar] [CrossRef]
- Butt, M.S.; Bai, J.; Wan, X.; Chu, C.; Xue, F.; Ding, H.; Zhou, G. Mg alloy rod reinforced biodegradable poly-lactic acid composite for load bearing bone replacement. Surf. Coat. Technol. 2017, 309, 471–479. [Google Scholar] [CrossRef]
- Hanks, J.H.; Wallace, R.E. Relation of oxygen and temperature in the preservation of tissues by refrigeration. Proc. Soc. Exp. Biol. Med. 1949, 71, 196–200. [Google Scholar] [CrossRef]
- Tkacz, J.; Slouková, K.; Minda, J.; Drábiková, J.; Fintová, S.; Doležal, P.; Wasserbauer, J. Influence of the composition of the Hank’s balanced salt solution on the corrosion behavior of AZ31 and AZ61 magnesium alloys. Metals 2017, 7, 465. [Google Scholar] [CrossRef]
- Zhang, C.Y.; Zeng, R.C.; Liu, C.L. Comparison of calcium phosphate coatings on Mg–Al and Mg–Ca alloys and their corrosion behavior in Hank’s solution. Surf. Coat. Technol. 2010, 204, 3636–3640. [Google Scholar]
- Maqbool, A.; Hussain, M.A.; Khalid, F.A.; Bakhsh, N.; Hussain, A.; Kim, M.H. Mechanical characterization of copper coated carbon nanotubes reinforced aluminum matrix composites. Mater. Char. 2013, 86, 39–48. [Google Scholar] [CrossRef]
- Saleem, M.; Hwan, L.D.; Kim, I.; Kim, M.S.; Maqbool, A.; Nisar, U.; Pervez, S.A. Revealing of core shell effect on frequency-dependent properties of Bi-based relaxor/ferroelectric ceramic composites. Sci. Rep. 2018, 8, 14146. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Butt, M.S.; Maqbool, A.; Umer, M.A.; Shahid, M.; Javaid, F.; Malik, R.A.; Jabbar, H.; Khalil, H.M.W.; Hwan, L.D.; et al. Percolation phenomena of dielectric permittivity of a microwave-sintered BaTiO3–Ag nanocomposite for high energy capacitor. J. Alloys Compd. 2020, 822, 153525. [Google Scholar] [CrossRef]
- Kotsikos, G.; Sutcliffe, J.M.; Holroyd, N.J.H. Hydrogen effects in the corrosion fatigue behaviour of the white zone of 7xxx series aluminium alloy welds. Corr. Sci. 2000, 42, 17–33. [Google Scholar] [CrossRef]
- Alabbasi, A.; Liyanaarachchi, S.; Kannan, M.B. Polylactic acid coating on a biodegradable magnesium alloy: An in vitro degradation study by electrochemical impedance spectroscopy. Thin Solid Films 2012, 520, 6841–6844. [Google Scholar] [CrossRef]



| Material | Injection Speed | Injection Temperature | Cooling Time | Pressure Time | Backing Pressure | Cylinder Temperature |
|---|---|---|---|---|---|---|
| Mg rod | 20 mm/s | 190 °C | 10 s | 3 s | 750 bar | 200-195-190-185-35 °C |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butt, M.S.; Maqbool, A.; Umer, M.A.; Saleem, M.; Malik, R.A.; Alarifi, I.M.; Alrobei, H. Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites. Crystals 2020, 10, 381. https://doi.org/10.3390/cryst10050381
Butt MS, Maqbool A, Umer MA, Saleem M, Malik RA, Alarifi IM, Alrobei H. Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites. Crystals. 2020; 10(5):381. https://doi.org/10.3390/cryst10050381
Chicago/Turabian StyleButt, Muhammad Shoaib, Adnan Maqbool, Malik Adeel Umer, Mohsin Saleem, Rizwan Ahmed Malik, Ibrahim M. Alarifi, and Hussein Alrobei. 2020. "Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites" Crystals 10, no. 5: 381. https://doi.org/10.3390/cryst10050381
APA StyleButt, M. S., Maqbool, A., Umer, M. A., Saleem, M., Malik, R. A., Alarifi, I. M., & Alrobei, H. (2020). Enhanced Mechanical Properties of Surface Treated AZ31 Reinforced Polymer Composites. Crystals, 10(5), 381. https://doi.org/10.3390/cryst10050381







