Single-Crystal Growth and Small Anisotropy of the Lower Critical Field in Oxypnictides: NdFeAsO1 − xFx
Abstract
1. Introduction
- The compounds decompose above ~1200 °C, well below the melting point;
- The presence of stable secondary phases (i.e., stable rare earth oxides compounds);
- The multicomponent phase diagrams are unknown.
- Growth by the flux method using metallic Tin (Sn), an equivalent mixture of sodium and potassium chlorides (NaCl/KCl), and iodides (NaI/KI), and sodium arsenide (NaAs) as the flux. The following points were noticed:
- Sodium arsenide (NaAs) was shown to yield large and well-shaped single crystals [11]. However, the quality of these crystals is under debate. Unfortunately, F-doping was not yet possible using this flux;
- Tin (Sn) is a suitable flux for the CeFe(As1 − xPx)O system, but not for the LaFeAsO compound [12];
- NaI/KI flux was shown to give crystals with several 100 μm lengths [13]. High temperatures need to be applied to increase the solubility of the elements in the salt flux.
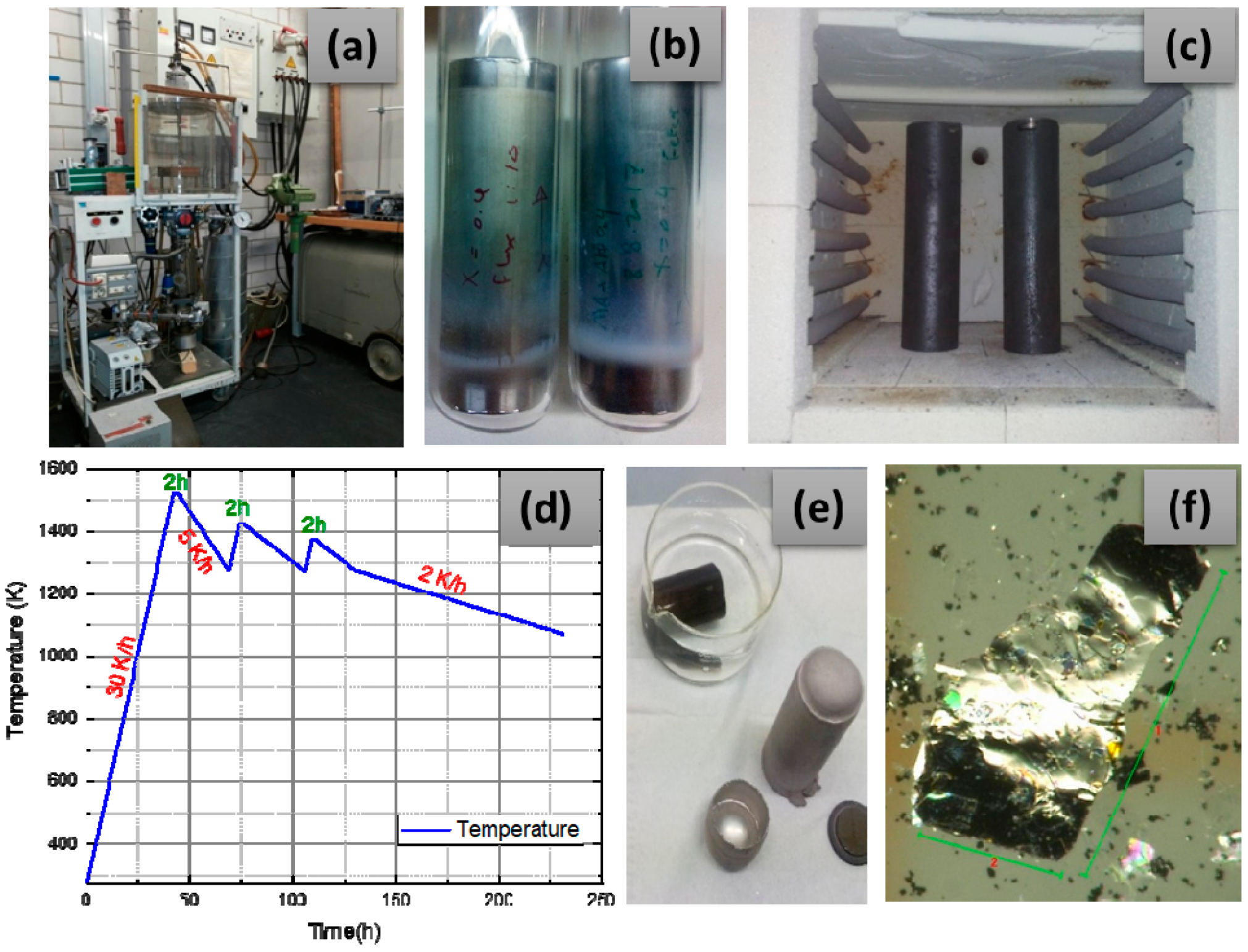
2. Materials and Methods
3. Results and Discussion
3.1. Crystal Quality
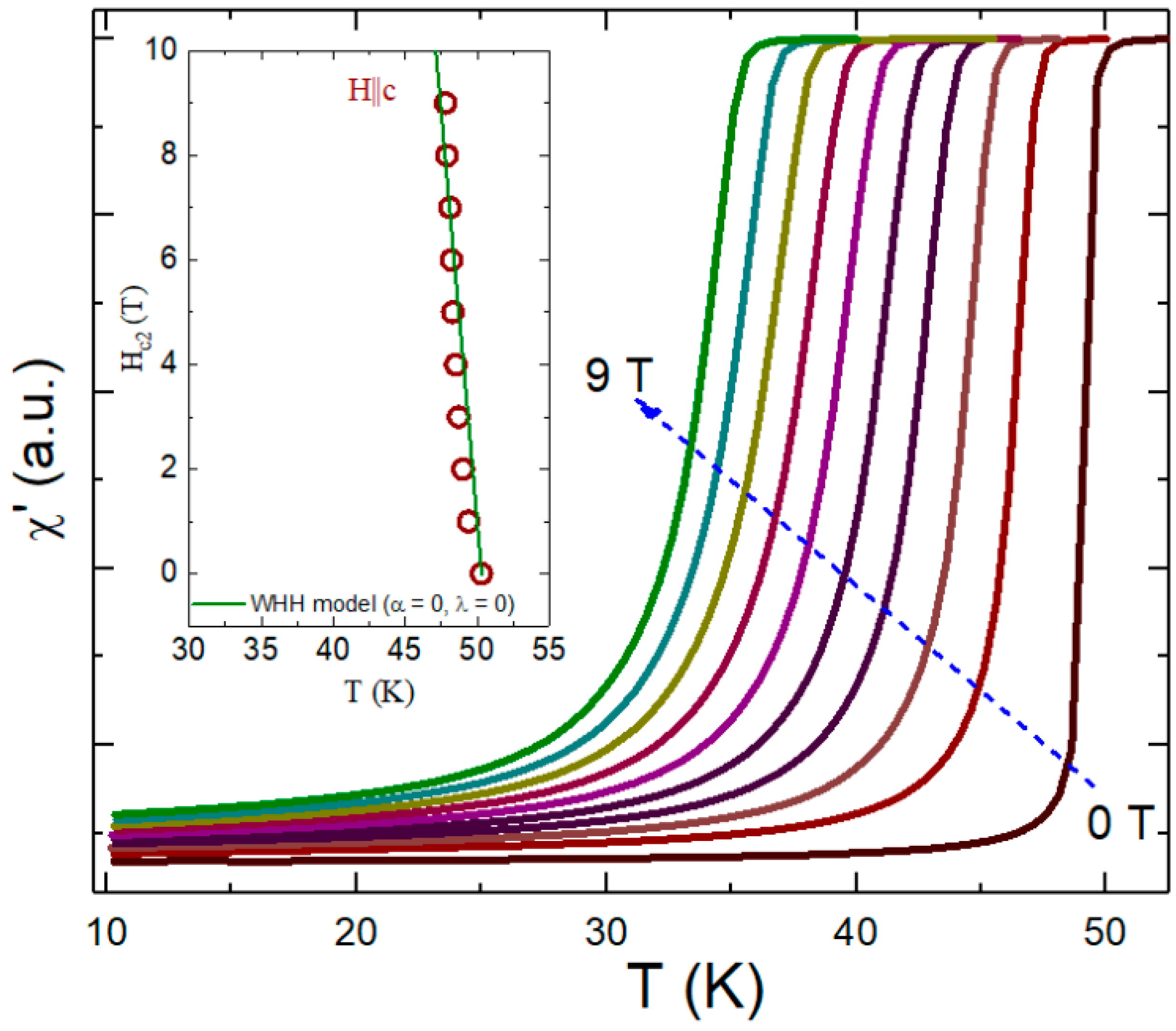
3.2. Transport Properties
3.3. AC Measurements
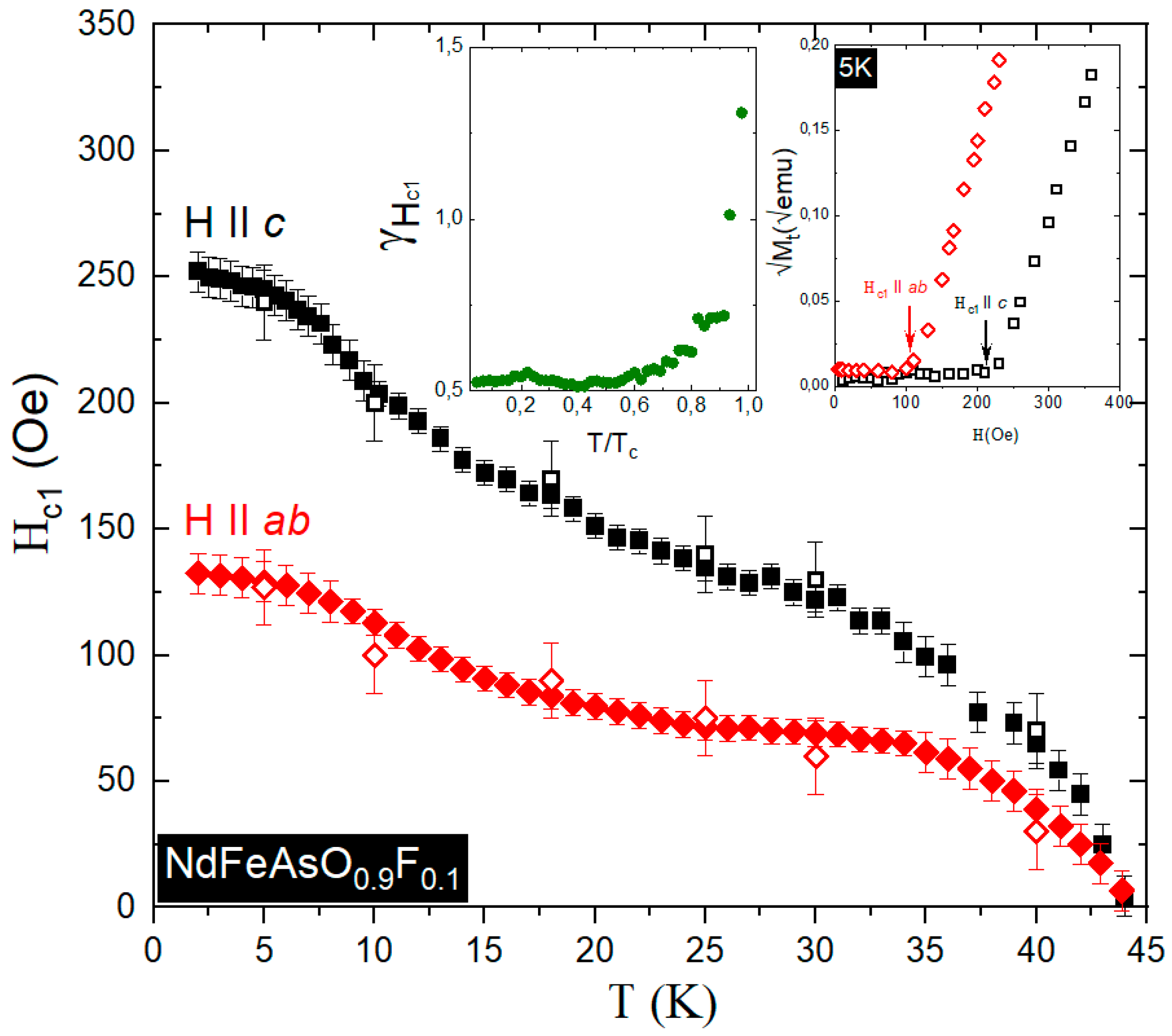
3.4. Experimental Determination of the Lower Critical Field
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Coleman, P.; Schofield, A.J. Quantum Criticality. Nature 2005, 433, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Löhneysen, H.v.; Rosch, A.; Vojta, M.; Wölfle, P. Fermi-liquid instabilities at magnetic quantum phase transitions. Rev. Mod. Phys. 2007, 79, 1015–1075. [Google Scholar] [CrossRef]
- Alborzi, Z.; Daadmehr, V. Synthesis and characterization of iron-based superconductor Nd-1111. Physica C: Supercond. 2018, 549, 116–118. [Google Scholar] [CrossRef]
- Tapia-Ignacio, C.; Ponce-Flores, H.; Corrales-Mendoza, I.; Conde-Gallardo, A. Superconductivity, and Paramagnetism in the Nd-Based 1111 Oxypnictide Compounds. IEEE Trans. Appl. Supercond. 2017, 27, 1–5. [Google Scholar] [CrossRef]
- Kito, H.; Eisaki, H.; Iyo, A. Superconductivity at 54 K in F-Free NdFeAsO1 − y. J. Phys. Soc. Jpn. 2008, 77, 063707. [Google Scholar] [CrossRef]
- McGuire, M.A.; Hermann, R.P.; Sefat, A.S.; Sales, B.C.; Jin, R.; Mandrus, D.; Grandjean, F.; Long, G.J. Influence of the rare-earth element on the effects of the structural and magnetic phase transitions in CeFeAsO, PrFeAsO and NdFeAsO. New J. Phys. 2009, 11, 025011. [Google Scholar] [CrossRef]
- Jia, Y.; Cheng, P.; Fang, L.; Luo, H.; Yang, H.; Ren, C.; Shan, L.; Gu, C.; Wen, H.-H. Critical fields and anisotropy of NdFeAsO0.82F0.18 single crystals. Appl. Phys. Lett. 2008, 93, 032503. [Google Scholar] [CrossRef]
- Ponce-Flores, H.; Conde-Gallardo, A. Influence of the Oxygen Excess in the Synthesis of NdFeAsO1 − x Fx Superconductors. J. Supercond. Nov. Magn. 2013, 27, 673–679. [Google Scholar] [CrossRef]
- Lamura, G.; Shiroka, T.; Bonfà, P.; Sanna, S.; De Renzi, R.; Putti, M.; Zhigadlo, N.D.; Katrych, S.; Khasanov, R.; Karpinski, J. Slow magnetic fluctuations and superconductivity in fluorine-doped NdFeAsO. Phys. Rev. B 2015, 91, 024513. [Google Scholar] [CrossRef]
- Tehrani, F.S.; Daadmehr, V. The Superconductivity Mechanism in Nd-1111 Iron-Based Superconductor Doped by Calcium. J. Low Temp. Phys. 2020, 1–15. [Google Scholar] [CrossRef]
- Yan, J.-Q.; Jensen, B.; Dennis, K.W.; McCallum, R.W.; Lograsso, T.A. Flux requirements for the growth of RFeAsO (R=rare earth) superconductors. Appl. Phys. Lett. 2011, 98, 072504. [Google Scholar] [CrossRef]
- Krellner, C.; Geibel, C. Single-crystal growth and anisotropy of CeRuPO. J. Cryst. Growth 2008, 310, 1875–1880. [Google Scholar] [CrossRef]
- Nitsche, F.; Jesche, A.; Hieckmann, E.; Doert, T.; Ruck, M. Structural trends from a consistent set of single-crystal data of RFeAsO (R=La, Ce, Pr, Nd, Sm, Gd, and Tb). Phys. Rev. B 2010, 82, 134514. [Google Scholar] [CrossRef]
- Zhigadlo, N.D.; Katrych, S.; Bukowski, Z.; Weyeneth, S.; Puzniak, R.; Karpinski, J. Single crystals of superconducting SmFeAsO1 − xFygrown at high pressure. J. Phys. Condens. Matter 2008, 20, 342202. [Google Scholar] [CrossRef]
- Karpinski, J.; Zhigadlo, N.D.; Katrych, S.; Bukowski, Z.; Moll, P.; Weyeneth, S.; Keller, H.; Puzniak, R.; Tortello, M.; Daghero, D.; et al. Single crystals of LnFeAsO1 − xFx (Ln=La, Pr, Nd, Sm, Gd) and Ba1 − xRbxFe2As2: Growth, structure and superconducting properties. Physica C: Supercond. 2009, 469, 370–380. [Google Scholar] [CrossRef]
- Adamski, A.; Krellner, C.; Abdel-Hafiez, M. Signature of multigap nodeless superconductivity in fluorine-doped NdFeAsO. Phys. Rev. B 2017, 96, 100503. [Google Scholar] [CrossRef]
- Adamski, A.; Nassief, A.R.; Krellner, C.; Abdel-Hafiez, M. Vortex dynamics and the critical current density in NdFeAsO0.9F0.1 single crystal. J. Magn. Magn. Mater. 2018, 459, 301–304. [Google Scholar] [CrossRef]
- Abdel-Hafiez, M.; Mito, M.; Shibayama, K.; Takagi, S.; Ishizuka, M.; Vasiliev, A.N.; Krellner, C.; Mao, H.K. High-pressure phase diagram of NdFeAsO0.9F0.1: Disappearance of superconductivity on the verge of ferromagnetism from Nd moments. Phys. Rev. B 2018, 98, 094504. [Google Scholar] [CrossRef]
- Abdel-Hafiez, M.; Ge, J.; Vasiliev, A.N.; Chareev, D.A.; Van de Vondel, J.; Moshchalkov, V.V.; Silhanek, A.V. Temperature dependence of lower critical field Hc1(T) shows nodeless superconductivity in FeSe. Phys. Rev. B 2013, 88, 174512. [Google Scholar] [CrossRef]
- Abdel-Hafiez, M.; Zhang, Y.-Y.; Cao, Z.-Y.; Duan, C.-G.; Karapetrov, G.; Pudalov, V.M.; Vlasenko, V.A.; Sadakov, A.V.; Knyazev, D.A.; Romanova, T.A.; et al. Superconducting properties of sulfur-doped iron selenide. Phys. Rev. B 2015, 91, 165109. [Google Scholar] [CrossRef]
- Abdel-Hafiez, M.; Pereira, P.J.; Kuzmichev, S.A.; Kuzmicheva, T.E.; Pudalov, V.M.; Harnagea, L.; Kordyuk, A.A.; Silhanek, A.V.; Moshchalkov, V.V.; Shen, B.; et al. Lower critical field and SNS-Andreev spectroscopy of 122-arsenides: Evidence of nodeless superconducting gap. Phys. Rev. B 2014, 90, 054524. [Google Scholar] [CrossRef]
- Abdel-Hafiez, M.; Zhao, Y.; Huang, Z.; Cho, C.W.; Wong, C.H.; Hassen, A.; Ohkuma, M.; Fang, Y.W.; Pan, B.J.; Ren, Z.A.; et al. High-pressure effects on isotropic superconductivity in the iron-free layered pnictide superconductor BaPd2As2. Phys. Rev. B 2018, 97, 134508. [Google Scholar] [CrossRef]
- Song, Y.J.; Ghim, J.S.; Yoon, J.H.; Lee, K.J.; Jung, M.H.; Ji, H.-S.; Shim, J.H.; Bang, Y.; Kwon, Y.S. Small anisotropy of the lower critical field and the s ± -wave two-gap feature in single-crystal LiFeAs. EPL (Europhysics Letters) 2011, 94, 57008. [Google Scholar] [CrossRef][Green Version]
- Lyard, L.; Szabó, P.; Klein, T.; Marcus, J.; Marcenat, C.; Kim, K.H.; Kang, B.W.; Lee, H.S.; Lee, S.I. Anisotropies of the Lower and Upper Critical Fields in MgB2 Single Crystals. Phys. Rev. Lett. 2004, 92, 057001. [Google Scholar] [CrossRef]
- Hashimoto, K.; Shibauchi, T.; Kato, T.; Ikada, K.; Okazaki, R.; Shishido, H.; Ishikado, M.; Kito, H.; Iyo, A.; Eisaki, H.; et al. Microwave Penetration Depth and Quasiparticle Conductivity of PrFeAsO1 − y Single Crystals: Evidence for a Full-Gap Superconductor. Phys. Rev. Lett. 2009, 102, 017002. [Google Scholar] [CrossRef]
- Singh, D.J. Electronic structure and doping in BaFe2As2 and LiFeAs: Density functional calculations. Phys. Rev. B 2008, 78, 094511. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanna, A.R.N.; Abdel-Hafiez, M. Single-Crystal Growth and Small Anisotropy of the Lower Critical Field in Oxypnictides: NdFeAsO1 − xFx. Crystals 2020, 10, 362. https://doi.org/10.3390/cryst10050362
Hanna ARN, Abdel-Hafiez M. Single-Crystal Growth and Small Anisotropy of the Lower Critical Field in Oxypnictides: NdFeAsO1 − xFx. Crystals. 2020; 10(5):362. https://doi.org/10.3390/cryst10050362
Chicago/Turabian StyleHanna, Abanoub R. N., and Mahmoud Abdel-Hafiez. 2020. "Single-Crystal Growth and Small Anisotropy of the Lower Critical Field in Oxypnictides: NdFeAsO1 − xFx" Crystals 10, no. 5: 362. https://doi.org/10.3390/cryst10050362
APA StyleHanna, A. R. N., & Abdel-Hafiez, M. (2020). Single-Crystal Growth and Small Anisotropy of the Lower Critical Field in Oxypnictides: NdFeAsO1 − xFx. Crystals, 10(5), 362. https://doi.org/10.3390/cryst10050362





