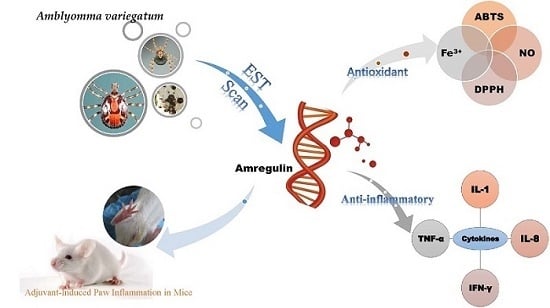
An Immunosuppressant Peptide from the Hard Tick Amblyomma variegatum
Abstract
:1. Introduction
2. Results
2.1. Sequence Analysis and the Effects of Amregulin on Cytokine Secretions Induced by LPS
2.2. Antioxidant Activities of Amregulin
2.2.1. Free Radical Scavenging Activity
2.2.2. Fe3+ Reducing Power
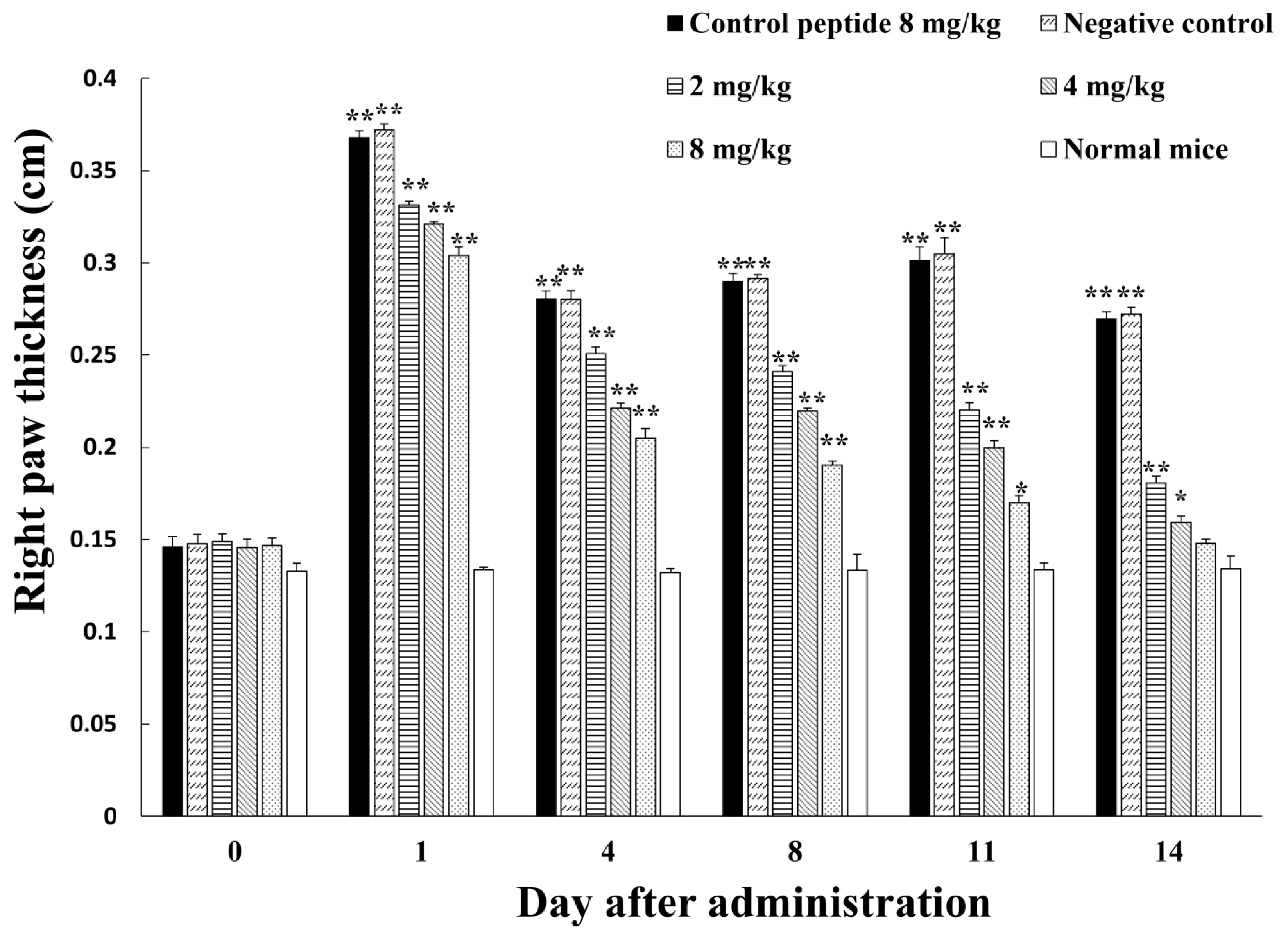
2.3. Inhibition of Adjuvant-Induced Paw Inflammation by Amregulin
3. Discussion
3.1. Effects of Amregulin on Cytokine Secretion Induced by LPS
3.2. Antioxidant Activities of Amregulin
3.3. Inhibition of Adjuvant-Induced Paw Inflammation by Amregulin
4. Conclusions
5. Experimental Section
5.1. cDNA, Peptide and Cytokine Assays
5.2. Free Radical Scavenging Activity
5.2.1. DPPH Scavenging
5.2.2. ABTS+ Scavenging
5.2.3. NO Scavenging
5.2.4. Fe3+ Reducing Power
5.3. Adjuvant-Induced Paw Inflammation in Mice
5.4. Statistical Analysis
5.5. Ethics Statement
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Burke, M.S.; Fordham, L.A.; Hamrick, H.J. Ticks and tick paralysis: Imaging findings on cranial MR. Pediatr. Radiol. 2005, 35, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Cabezas, C.A.; Valdes, J.J. Are ticks venomous animals? Front. Zool. 2014, 11. [Google Scholar] [CrossRef]
- Lysyk, T.J.; Veira, D.M.; Majak, W. Cattle can develop immunity to paralysis caused by Dermacentor andersoni. J. Med. Entomol. 2009, 46, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Lysyk, T.J.; Dergousoff, S.J. Can subclinical infestation by Paralyzing Dermacentor andersoni (Acari: Ixodidae) induce immunity to tick paralysis in sheep? J. Med. Entomol. 2016, 48. [Google Scholar] [CrossRef]
- Dworkin, M.S.; Shoemaker, P.C.; Anderson, D.E. Tick paralysis: 33 human cases in Washington State, 1946–1996. Clin. Infect. Dis. 1999, 29, 1435–1439. [Google Scholar] [CrossRef] [PubMed]
- Diaz, J.H. A comparative meta-analysis of tick paralysis in the United States and Australia. Clin. Toxicol. 2015, 53, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Edlow, J.A. Tick paralysis. Curr. Treat. Options Neurol. 2010, 12, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A. Lyme disease: Uphill struggle. Nature 2006, 439, 524–525. [Google Scholar] [CrossRef] [PubMed]
- Bernard, Q.; Jaulhac, B.; Boulanger, N. Skin and arthropods: An effective interaction used by pathogens in vector-borne diseases. Eur. J. Dermatol. 2015, 25 (Suppl. 1), 18–22. [Google Scholar] [PubMed]
- Da Silveira, J.A.; Rabelo, E.M.; Ribeiro, M.F. Detection of Theileria and Babesia in brown brocket deer (Mazama gouazoubira) and marsh deer (Blastocerus dichotomus) in the State of Minas Gerais, Brazil. Vet. Parasitol. 2011, 177, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Galay, R.L.; Maeda, H.; Aung, K.M.; Umemiya-Shirafuji, R.; Xuan, X.; Igarashi, I.; Tsuji, N.; Tanaka, T.; Fujisaki, K. Anti-babesial activity of a potent peptide fragment derived from longicin of Haemaphysalis longicornis. Trop. Anim. Health Prod. 2012, 44, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.R.; Oteo, J.A. Human granulocytic ehrlichiosis in Europe. Clin. Microbiol. Infect. 2002, 8, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Ogden, N.H.; Woldehiwet, Z.; Hart, C.A. Granulocytic ehrlichiosis: An emerging or rediscovered tick-borne disease? J. Med. Microbiol. 1998, 47, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Andoh, M.; Sakata, A.; Takano, A.; Kawabata, H.; Fujita, H.; Une, Y.; Goka, K.; Kishimoto, T.; Ando, S. Detection of Rickettsia and Ehrlichia spp. in ticks associated with exotic reptiles and amphibians imported into Japan. PLoS ONE 2015, 10, e0133700. [Google Scholar] [CrossRef] [PubMed]
- Ytrehus, B.; Vainio, K.; Dudman, S.G.; Gilray, J.; Willoughby, K. Tick-borne encephalitis virus and louping-ill virus may co-circulate in Southern Norway. Vector Borne Zoonotic Dis. 2013, 13, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.; Bloch, K.C.; McBride, J.W. Human ehrlichiosis and anaplasmosis. Clin. Lab. Med. 2010, 30, 261–292. [Google Scholar] [CrossRef] [PubMed]
- Nakao, R.; Qiu, Y.; Salim, B.; Hassan, S.M.; Sugimoto, C. Molecular detection of Rickettsia africae in Amblyomma variegatum collected from Sudan. Vector Borne Zoonotic Dis. 2015, 15, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Natala, A.J.; Balogun, E.O.; Balogun, J.A.; Inuwa, H.M.; Nok, A.J.; Shiba, T.; Harada, S.; Kita, K.; Agbede, R.I.; Esievo, K.A. Identification and characterization of sialidase-like activity in the developmental stages of Amblyomma variegatum. J. Med. Entomol. 2013, 50, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Hovius, J.W.; Levi, M.; Fikrig, E. Salivating for knowledge: Potential pharmacological agents in tick saliva. PLoS Med. 2008, 5, e43. [Google Scholar] [CrossRef] [PubMed]
- Juncadella, I.J.; Garg, R.; Ananthnarayanan, S.K.; Yengo, C.M.; Anguita, J. T-cell signaling pathways inhibited by the tick saliva immunosuppressor, Salp15. FEMS. Immunol. Med. Microbiol. 2007, 49, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, S.; Sukumaran, B.; Bozdogan, U.; Thomas, V.; Liang, X.; DePonte, K.; Marcantonio, N.; Koski, R.A.; Anderson, J.F.; Kantor, F.; et al. A tick antioxidant facilitates the Lyme disease agent’s successful migration from the mammalian host to the arthropod vector. Cell Host Microbe 2007, 2, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Preston, S.G.; Majtan, J.; Kouremenou, C.; Rysnik, O.; Burger, L.F.; Cabezas Cruz, A.; Chiong Guzman, M.; Nunn, M.A.; Paesen, G.C.; Nuttall, P.A.; et al. Novel immunomodulators from hard ticks selectively reprogramme human dendritic cell responses. PLoS Pathog. 2013, 9, e1003450. [Google Scholar] [CrossRef] [PubMed]
- Lieskovska, J.; Palenikova, J.; Langhansova, H.; Chagas, A.C.; Calvo, E.; Kotsyfakis, M.; Kopecky, J. Tick sialostatins L and L2 differentially influence dendritic cell responses to Borrelia spirochetes. Parasit. Vectors 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, S.; Perez, O.; Mootien, S.; DePonte, K.; Koski, R.A.; Fikrig, E.; Ledizet, M. Characterization of Ixophilin, a thrombin inhibitor from the gut of Ixodes scapularis. PLoS ONE 2013, 8, e68012. [Google Scholar] [CrossRef] [PubMed]
- Pekarikova, D.; Rajska, P.; Kazimirova, M.; Pechanova, O.; Takac, P.; Nuttall, P.A. Vasoconstriction induced by salivary gland extracts from ixodid ticks. Int. J. Parasitol. 2015, 45, 879–883. [Google Scholar] [CrossRef] [PubMed]
- Kotsyfakis, M.; Sa-Nunes, A.; Francischetti, I.M.; Mather, T.N.; Andersen, J.F.; Ribeiro, J.M. Antiinflammatory and immunosuppressive activity of sialostatin L, a salivary cystatin from the tick Ixodes scapularis. J. Biol. Chem. 2006, 281, 26298–26307. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.; Lomas, L.O.; Jonczy, J.; Turner, P.C.; Rees, H.H. Two novel non-cationic defensin-like antimicrobial peptides from haemolymph of the female tick, Amblyomma hebraeum. Biochem. J. 2004, 379, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.; Takeuchi, H.; Lomas, L.O.; Jonczy, J.; Rigden, D.J.; Rees, H.H.; Turner, P.C. A new type of antimicrobial protein with multiple histidines from the hard tick, Amblyomma hebraeum. FASEB J. 2004, 18, 1447–1449. [Google Scholar] [CrossRef] [PubMed]
- Petnicki-Ocwieja, T.; Brissette, C.A. Lyme disease: Recent advances and perspectives. Front. Cell. Infect. Microbiol. 2015, 5. [Google Scholar] [CrossRef]
- Ramamoorthi, N.; Narasimhan, S.; Pal, U.; Bao, F.; Yang, X.F.; Fish, D.; Anguita, J.; Norgard, M.V.; Kantor, F.S.; Anderson, J.F. The Lyme disease agent exploits a tick protein to infect the mammalian host. Nature 2005, 436, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Hovius, J.W.; de Jong, M.A.; den Dunnen, J.; Litjens, M.; Fikrig, E.; van der Poll, T.; Gringhuis, S.I.; Geijtenbeek, T.B. Salp15 binding to DC-SIGN inhibits cytokine expression by impairing both nucleosome remodeling and mRNA stabilization. PLoS Pathog. 2008, 4, e31. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Liang, J.; Yu, H.; Wu, H.; Xu, C.; Liu, J.; Lai, R. A tick B-cell inhibitory protein from salivary glands of the hard tick, Hyalomma asiaticum asiaticum. Biochem. Biophys. Res. Commun. 2006, 343, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Hong, J.; Yang, H.; Wu, J.; Ma, D.; Li, D.; Lin, D.; Lai, R. Frog skins keep redox homeostasis by antioxidant peptides with rapid radical scavenging ability. Free Radic. Biol. Med. 2010, 48, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.F. The natural history of ticks. Med. Clin. N. Am. 2002, 86, 205–218. [Google Scholar] [CrossRef]
- Juncadella, I.J.; Bates, T.C.; Suleiman, R.; Monteagudo-Mera, A.; Olson, C.M.; Navasa, N.; Olivera, E.R.; Osborne, B.A.; Anguita, J. The tick saliva immunosuppressor, Salp15, contributes to Th17-induced pathology during Experimental Autoimmune Encephalomyelitis. Biochem. Biophys. Res. Commun. 2010, 402, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Juncadella, I.J.; Garg, R.; Bates, T.C.; Olivera, E.R.; Anguita, J. The Ixodes scapularis salivary protein, salp15, prevents the association of HIV-1 gp120 and CD4. Biochem. Biophys. Res. Commun. 2008, 367, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, Y.; Liu, H.; Yang, H.; Ma, D.; Li, J.; Li, D.; Lai, R.; Yu, H. Two immunoregulatory peptides with antioxidant activity from tick salivary glands. J. Biol. Chem. 2010, 285, 16606–16613. [Google Scholar] [CrossRef] [PubMed]
- Diaz, P.V.; Valdivia, G.; Gaggero, A.A.; Bono, M.R.; Zepeda, G.; Rivas, M.; Uasapud, P.; Pinto, R.A.; Boza, M.L.; Guerrero, J. Pro-Inflammatory cytokines in Nasopharyngeal Aspirate from hospitalized children with respiratory syncytial virus infection with or without Rhinovirus Bronchiolitis, and use of the cytokines as predictors of illness severity. Medicine (Baltimore) 2015, 94, e1512. [Google Scholar] [CrossRef] [PubMed]
- Noah, T.L.; Ivins, S.S.; Murphy, P.; Kazachkova, I.; Moats-Staats, B.; Henderson, F.W. Chemokines and inflammation in the nasal passages of infants with respiratory syncytial virus bronchiolitis. Clin. Immunol. 2002, 104, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Schroder, K.; Hertzog, P.J.; Ravasi, T.; Hume, D.A. Interferon-gamma: An overview of signals, mechanisms and functions. J. Leukoc. Biol. 2004, 75, 163–189. [Google Scholar] [CrossRef] [PubMed]
- Gadow, A.V.; Joubert, E.; Hansmann, C.F. Effect of extraction time and additional heating on the antioxidant activity of rooibos tea (Aspalathus linearis) extracts. J. Agric. Food Chem. 1997, 45, 1370–1374. [Google Scholar] [CrossRef]
- Gurpreet, K.M.; Sarwar, A.; Zoobi, J.; Kaleem, J.; Mohammad, A. Evaluation of antioxidant activity of Cassia siamea flowers. J. Ethnopharmacol. 2006, 108, 340–348. [Google Scholar]
- SignalP 4.0 Server. Available online: http://www.cbs.dtu.dk/services/SignalP/ (accessed on 12 January 2015).


| Samples | Free Radical Scavenging Capacity (%) | Fe3+ Reducing Power (Absorbance at 700 nm) | ||
|---|---|---|---|---|
| ABTS+ | DPPH | NO | ||
| H2O | 0 ± 0.0 | 0 ± 5.4 | NA | NA |
| BHT | 73.6 ± 7.9 | 92.1 ± 10.4 | 41.7 ± 6.5 | 0.27 ± 0.06 |
| Amregulin (2.5 µg/mL) | 53.1 ± 12.3 | 39.5 ± 6.9 | 17.6 ± 4.2 | NA |
| Amregulin (5 µg/mL) | 79.9 ± 11.5 | 63.1± 9.2 | 30.3 ± 4.9 | 0.08 ± 0.01 |
| Amregulin (10 µg/mL) | 88.3 ± 17.2 | 70.8 ± 10.4 | 42.4 ± 6.7 | 0.10 ± 0.03 |
| Amregulin (20 µg/mL) | 93.5 ± 13.6 | 79.2 ± 13.4 | 48.7 ± 7.3 | 0.13 ± 0.02 |
| Control peptide (20 µg/mL) | 12.7 ± 3.1 | 7.6 ± 1.5 | 3.3 ± 0.8 | NA |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, Y.; Chen, W.; Mo, G.; Chen, R.; Fang, M.; Yedid, G.; Yan, X. An Immunosuppressant Peptide from the Hard Tick Amblyomma variegatum. Toxins 2016, 8, 133. https://doi.org/10.3390/toxins8050133
Tian Y, Chen W, Mo G, Chen R, Fang M, Yedid G, Yan X. An Immunosuppressant Peptide from the Hard Tick Amblyomma variegatum. Toxins. 2016; 8(5):133. https://doi.org/10.3390/toxins8050133
Chicago/Turabian StyleTian, Yufeng, Wenlin Chen, Guoxiang Mo, Ran Chen, Mingqian Fang, Gabriel Yedid, and Xiuwen Yan. 2016. "An Immunosuppressant Peptide from the Hard Tick Amblyomma variegatum" Toxins 8, no. 5: 133. https://doi.org/10.3390/toxins8050133
APA StyleTian, Y., Chen, W., Mo, G., Chen, R., Fang, M., Yedid, G., & Yan, X. (2016). An Immunosuppressant Peptide from the Hard Tick Amblyomma variegatum. Toxins, 8(5), 133. https://doi.org/10.3390/toxins8050133