Development of a Polyclonal Antibody for the Immunoanalysis of Ochratoxin A (OTA) by Employing a Specially Designed Synthetic OTA Derivative as the Immunizing Hapten
Abstract
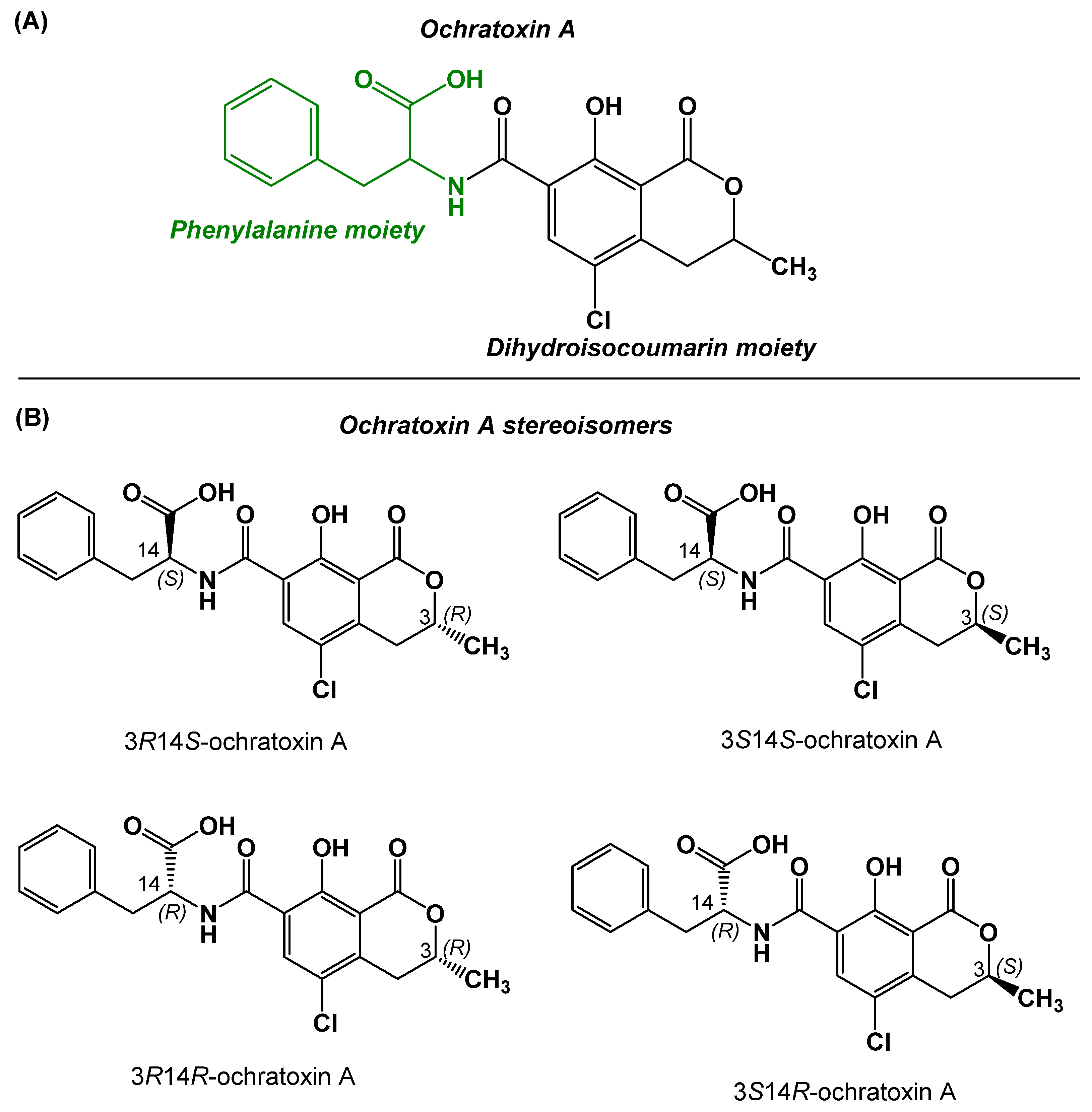
1. Introduction
2. Results
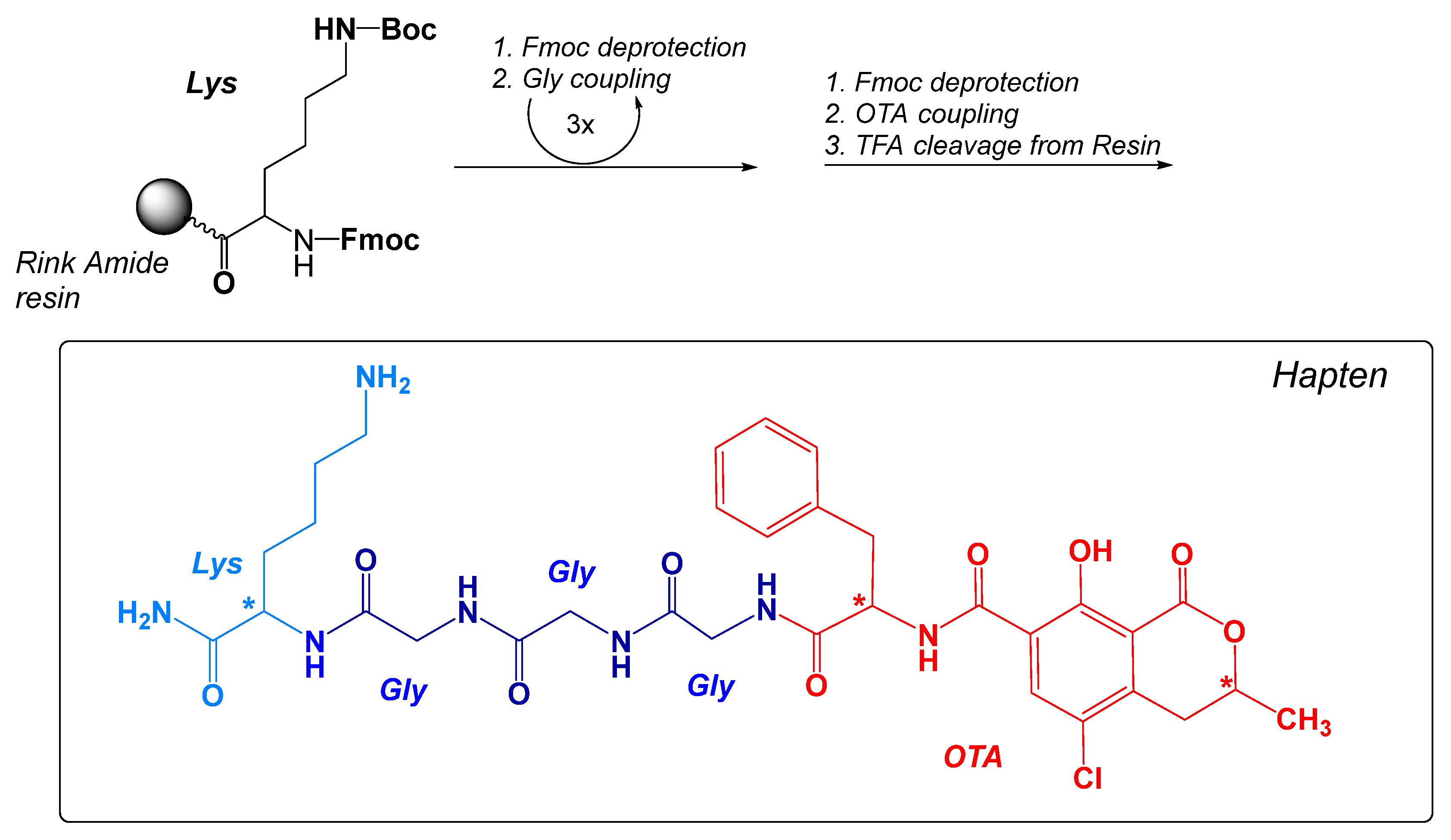
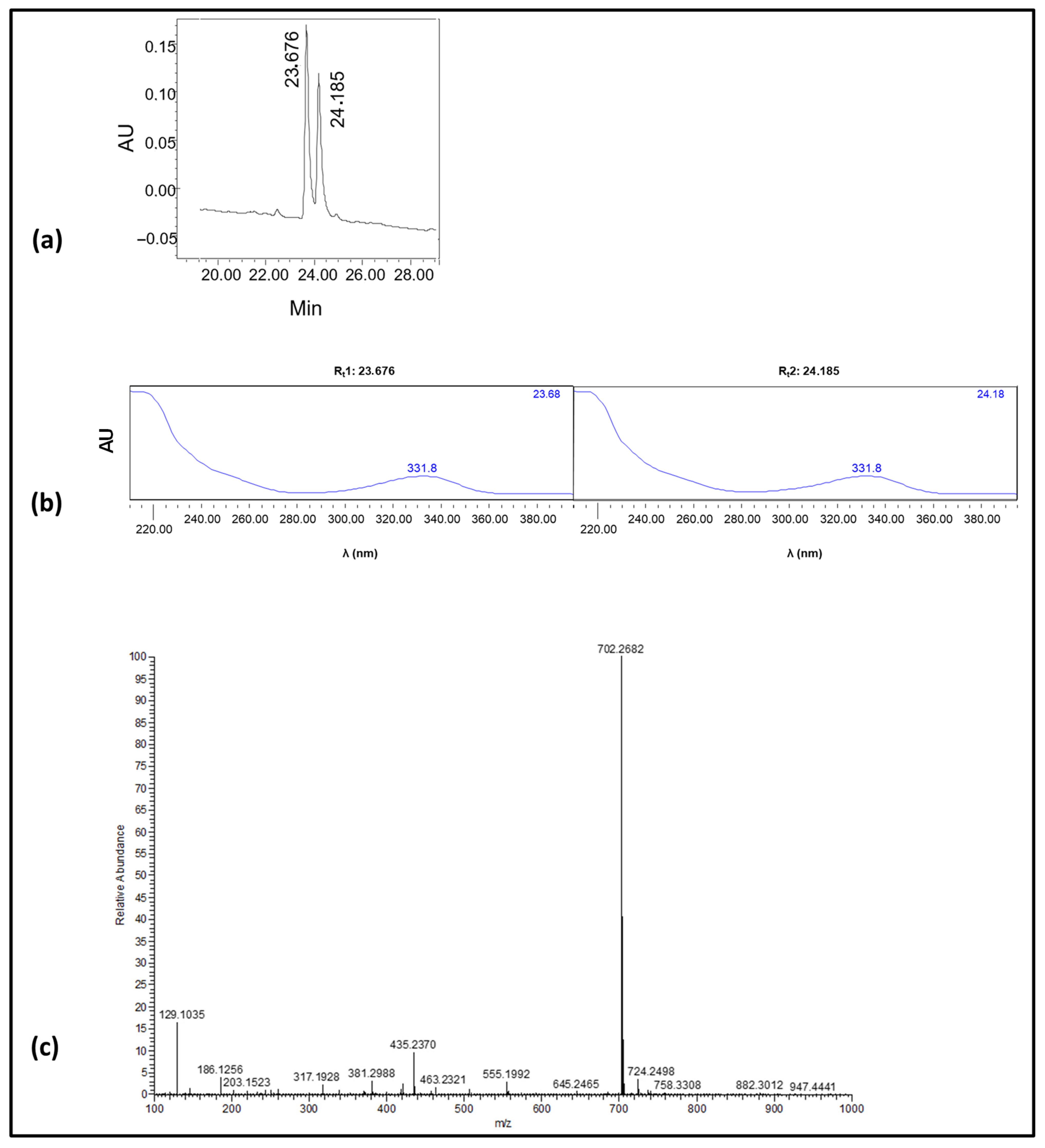
2.1. Design, Preparation, and Characterization of the Immunizing Hapten for OTA
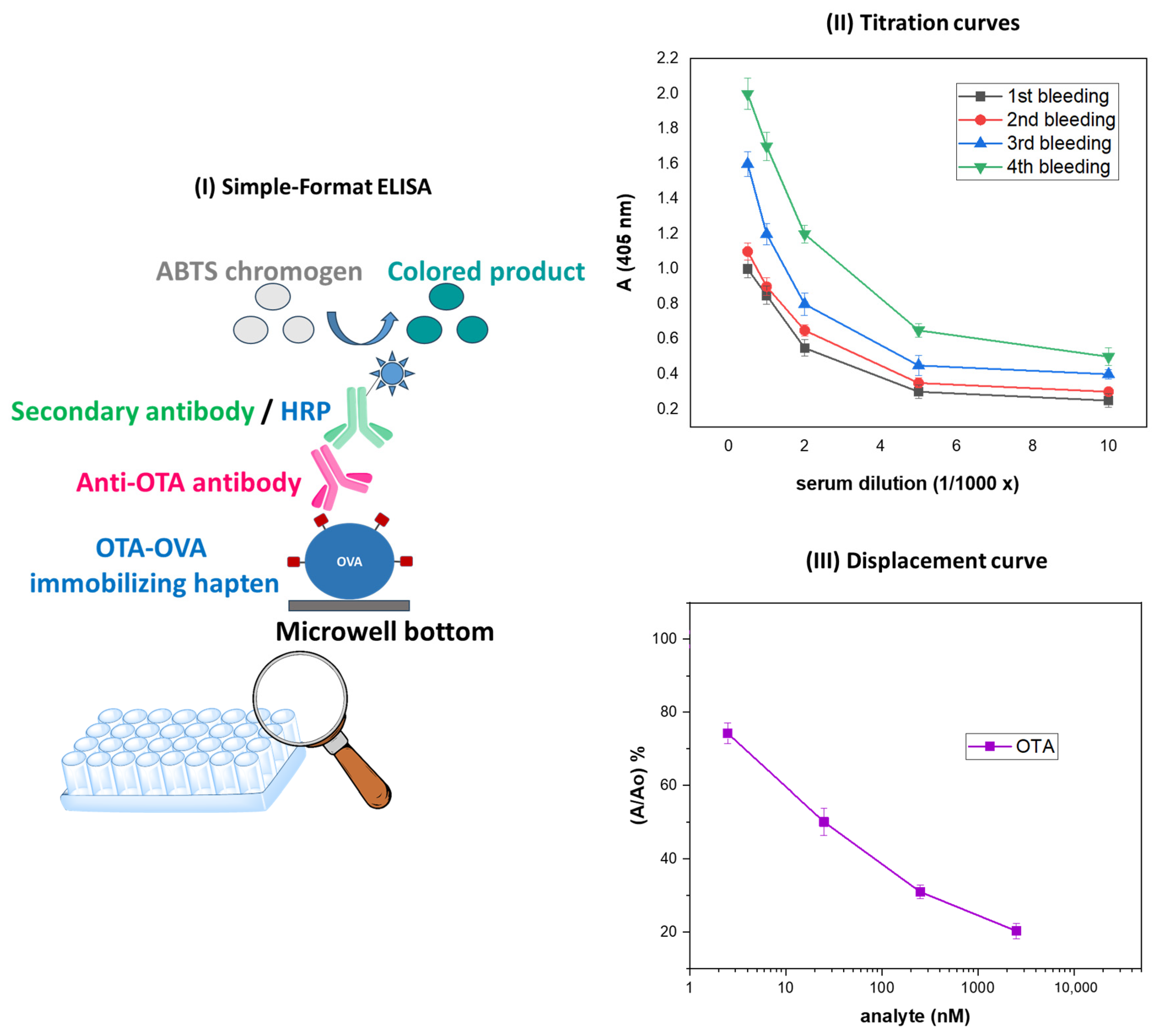
2.2. Development of the Anti-OTA Antibody
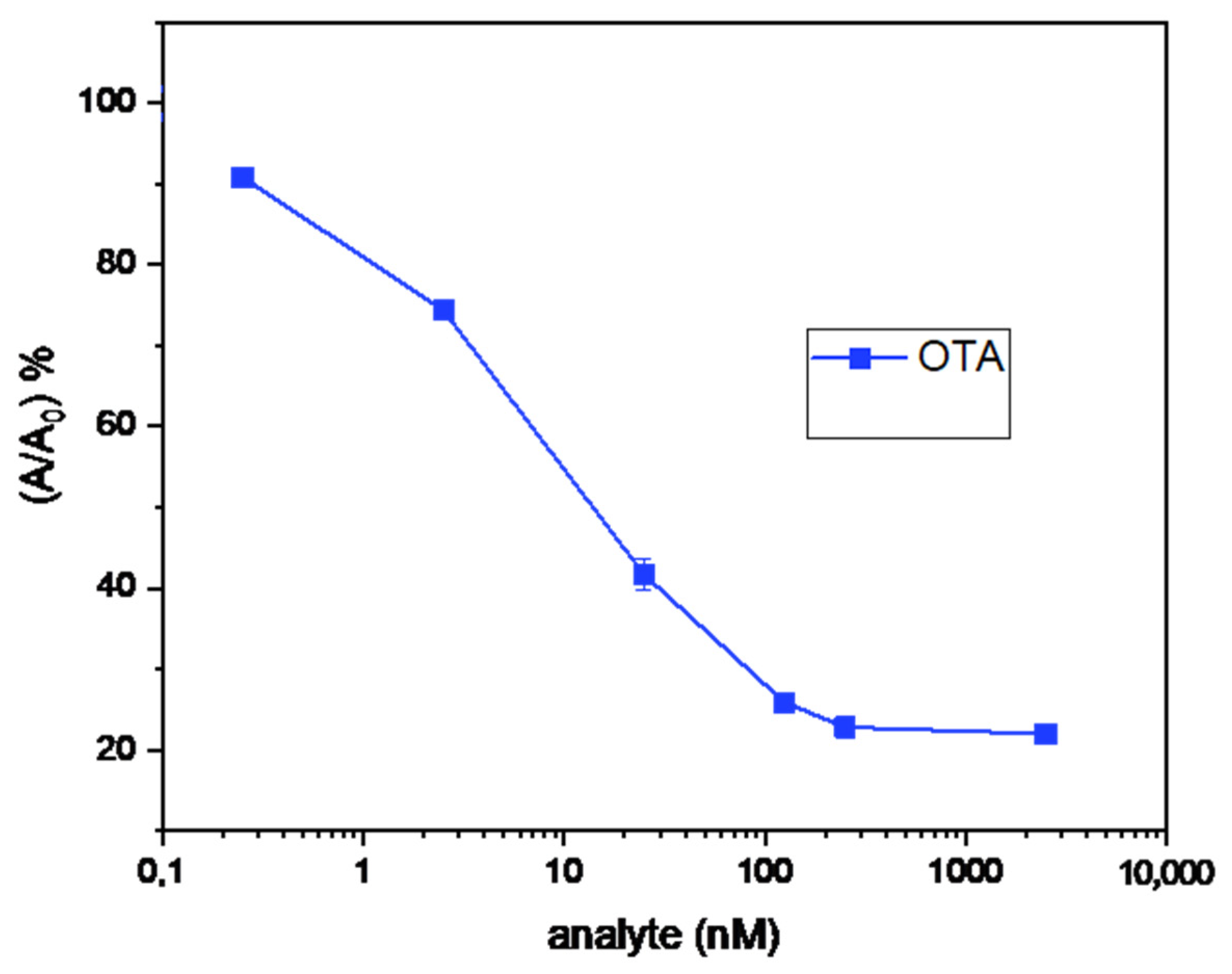
2.3. Evaluation of the Anti-OTA Antiserum with a Simple-Format ELISA
2.4. Various Applications of the Anti-OTA Antibody
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Immunizing Hapten for OTA (OTA-GGGK)
Chemistry
5.2. Development of the Anti-OTA Antibody
5.2.1. Conjugation of OTA-GGGK to bTGB
5.2.2. Immunization Protocol (Injections and Bleedings)
5.3. Evaluation of the Anti-OTA Antibody with a Simple-Format ELISA
5.3.1. Conjugation of OTA to Ovalbumin
5.3.2. ELISA Buffers
5.3.3. Titration ELISA Protocol
5.3.4. Displacement ELISA Protocol
5.4. Application of the Anti-OTA Antibody to a Multiple Mycotoxin Microarray Platform, an OTA-Optical Immunosensor, and a Biotin–Streptavidin ELISA
5.4.1. Multiple Mycotoxin Microarray Platform
5.4.2. Optical Immunosensor
5.4.3. Biotin–Streptavidin ELISA
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
Abbreviations
| A | Absorbance |
| A0 | Absorbance of the zero standard |
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| AFB1 | Aflatoxin B1 |
| AU | Absorbance units |
| Boc | tert-Butyloxycarbonyl |
| BSA | Bovine serum albumin |
| bTGB | Bovine thyroglobulin |
| Chlorpyrifos | O,O-diethyl O-3,5,6-trichloro-2-pyridyl phosphorothioate |
| DCM | Dichloromethane |
| DIC | N,N′-Diisopropylcarbodiimide |
| DMF | N,N-Dimethylformamide |
| DMSO | Dimethylsulfoxide |
| DON | Deoxynivalenol |
| EDC | 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide |
| ELISA | Enzyme-linked immunosorbent assay |
| ESI-MS | Electrospray ionization mass spectrometry |
| Fmoc | 9-Fluorenylmethyloxycarbonyl |
| Fmoc-SPPS | 9-Fluorenylmethyloxycarbonyl-based solid-phase peptide synthesis |
| FUM-B1 | Fumonisin B1 |
| GGGK | Glycyl-glycyl-glycyl-lysine |
| HPLC | High-performance liquid chromatography |
| HPLC-FLD | High-performance liquid chromatography with fluorescence detection |
| ID | Internal diameter |
| LC-MS | Liquid chromatography–mass spectrometry |
| LC-MS/MS | Liquid chromatography–tandem mass spectrometry |
| LoD | Limit of detection |
| MCPA | Methyl-4-chloro-phenoxyacetic acid |
| MRL | Maximum residue level |
| MW | Molecular weight |
| m/z | Mass-to-charge ratio |
| OPP | Ortho-phenyl-phenol |
| OTA | Ochratoxin A |
| OTA-GGGK | Ochratoxin A-glycyl-glycyl-glycyl-lysine |
| OTB | Ochratoxin B |
| OTC | Ochratoxin C |
| OVA | Ovalbumin |
| Oxyma | Ethyl 2-cyano-2-(hydroxyimino) acetate |
| PB | Phosphate buffer, 0.01 M, pH 7.4 |
| PBS | Phosphate-buffered saline (phosphate buffer, 0.01 M, pH 7.4, with 0.9% (w/v) NaCl) |
| PBS-T | PBS containing 0.05% (v/v) Tween 20 |
| PDA | Photodiode array |
| PMMA | Oxygen plasma micro-nanostructured poly (methyl methacrylate) |
| RP-HPLC | Reversed-phase high-performance liquid chromatography |
| Rt | Retention time |
| RT | Room temperature |
| sdAb | Single-domain antibody |
| SPPS | Solid phase peptide synthesis |
| TFA | Trifluoroacetic acid |
| TIS | Tri-isopropylsilane |
| TMB | 3,3′,5,5′-tetramethylbenzidine |
| Triclopyr | [(3,5,6-trichloropyridin-2-yl)oxy]acetic acid |
| UV-vis | Ultraviolet–visible |
References
- Banahene, J.C.M.; Ofosu, I.W.; Odai, B.T.; Lutterodt, H.E.; Agyemang, P.A.; Ellis, W.O. Ochratoxin A in food commodities: A review of occurrence, toxicity, and management strategies. Heliyon 2024, 10, e39313. [Google Scholar] [CrossRef] [PubMed]
- Ben Miri, Y.; Benabdallah, A.; Chentir, I.; Djenane, D.; Luvisi, A.; De Bellis, L. Comprehensive insights into ochratoxin A: Occurrence, analysis, and control strategies. Foods 2024, 13, 1184. [Google Scholar] [CrossRef]
- Cramer, B.; Harrer, H.; Nakamura, K.; Uemura, D.; Humpf, H.U. Total synthesis and cytotoxicity evaluation of all ochratoxin A stereoisomers. Bioorg. Med. Chem. 2010, 18, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Arce-López, B.; Coton, M.; Coton, E.; Hymery, N. Occurrence of the two major regulated mycotoxins, ochratoxin A and fumonisin B1, in cereal and cereal-based products in Europe and toxicological effects: A review. Environ. Toxicol. Pharmacol. 2024, 109, 104489. [Google Scholar] [CrossRef] [PubMed]
- Sharafi, H.; Alizadeh, N.; Moradi, M.; Sharafi, K.; Mousavi Khaneghah, A. The prevalence and concentration of ochratoxin A in meat and edible offal: A global systematic review and meta-analysis. Food Chem. Toxicol. 2023, 178, 113921. [Google Scholar] [CrossRef] [PubMed]
- Bonerba, E.; Manfredi, A.; Dimuccio, M.M.; Lorusso, P.; Pandiscia, A.; Terio, V.; Di Pinto, A.; Panseri, S.; Ceci, E.; Bozzo, G. Ochratoxin A in poultry supply chain: Overview of feed occurrence, carry-over, and pathognomonic lesions in target organs to promote food safety. Toxins 2024, 16, 487. [Google Scholar] [CrossRef]
- Aranda, C.; Rodriguez, R.; Fernández-Baldo, M.A.; Durán, P. Mycotoxins in cheese: Assessing risks, fungal contaminants, and control strategies for food safety. Foods 2025, 14, 351. [Google Scholar] [CrossRef]
- González-Curbelo, M.Á.; Kabak, B. Occurrence of mycotoxins in dried fruits worldwide, with a focus on aflatoxins and ochratoxin A: A review. Toxins 2023, 15, 576. [Google Scholar] [CrossRef]
- La Placa, L.; Tsitsigiannis, D.; Camardo Leggieri, M.; Battilani, P. From grapes to wine: Impact of the vinification process on ochratoxin A contamination. Foods 2023, 12, 260. [Google Scholar] [CrossRef]
- Zjalic, S.; Markov, K.; Loncar, J.; Jakopovic, Z.; Beccaccioli, M.; Reverberi, M. Biocontrol of occurrence ochratoxin A in wine: A review. Toxins 2024, 16, 277. [Google Scholar] [CrossRef]
- Fakhri, Y.; Ranaei, V.; Pilevar, Z.; Belaia, O.F.; Kolaeva, N.V.; Sarafraz, M.; Mousavi Khaneghah, A. Prevalence and concentration of Ochratoxin A in beer: A global systematic review, meta-analysis, and health risk assessment. Food Sci. Nutr. 2024, 12, 8503–8514. [Google Scholar] [CrossRef]
- Ahmad, M.S.; Alanazi, Y.A.; Alrohaimi, Y.; Shaik, R.A.; Alrashidi, S.; Al-Ghasham, Y.A.; Alkhalifah, Y.S.; Ahmad, R.K. Occurrence, evaluation, and human health risk assessment of ochratoxin a in infant formula and cereal-based baby food: A global literature systematic review. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2024, 41, 1171–1186. [Google Scholar] [CrossRef]
- Ahmad, M.S.; Alanazi, Y.A.; Alrohaimi, Y.; Shaik, R.A.; Alrashidi, S.; Al-Ghasham, Y.A.; Alkhalifah, Y.S.; Ahmad, R.K. Infant nutrition at risk: A global systematic review of ochratoxin A in human breast milk-human health risk assessment. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2024, 41, 1611–1624. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, H.D.; Ryu, D. Practical strategies to reduce ochratoxin A in Foods. Toxins 2024, 16, 58. [Google Scholar] [CrossRef]
- Ding, L.; Han, M.; Wang, X.; Guo, Y. Ochratoxin A: Overview of prevention, removal, and detoxification methods. Toxins 2023, 15, 565. [Google Scholar] [CrossRef] [PubMed]
- Więckowska, M.; Szelenberger, R.; Niemcewicz, M.; Harmata, P.; Poplawski, T.; Bijak, M. Ochratoxin A—The current knowledge concerning hepatotoxicity, mode of action and possible prevention. Molecules 2023, 28, 6617. [Google Scholar] [CrossRef] [PubMed]
- Khoi, C.S.; Chen, J.H.; Lin, T.Y.; Chiang, C.K.; Hung, K.Y. Ochratoxin A-induced nephrotoxicity: Up-to-date evidence. Int. J. Mol. Sci. 2021, 22, 11237. [Google Scholar] [CrossRef] [PubMed]
- Obafemi, B.A.; Adedara, I.A.; Rocha, J.B.T. Neurotoxicity of ochratoxin A: Molecular mechanisms and neurotherapeutic strategies. Toxicology 2023, 497–498, 153630. [Google Scholar] [CrossRef]
- Serrano-Civantos, M.; Beraza, E.; Álvarez-Erviti, L.; de Cerain, A.L.; Vettorazzi, A. Potential role of ochratoxin A in Parkinson’s disease: A systematic review of current evidence. Arch. Toxicol. 2025, 99, 1769–1790. [Google Scholar] [CrossRef]
- Więckowska, M.; Cichon, N.; Szelenberger, R.; Gorniak, L.; Bijak, M. Ochratoxin A and its role in cancer development: A comprehensive review. Cancers 2024, 16, 3473. [Google Scholar] [CrossRef]
- Aydemir, M.C.; Yaman, İ.; Kilic, M.A. Membrane receptor-mediated disruption of cellular homeostasis: Changes in intracellular signaling pathways increase the toxicity of ochratoxin A. Mol. Nutr. Food Res. 2024, 68, e2300777. [Google Scholar] [CrossRef]
- Frangiamone, M.; Lázaro, Á.; Cimbalo, A.; Font, G.; Manyes, L. In vitro and in vivo assessment of AFB1 and OTA toxic effects and the beneficial role of bioactive compounds. A systematic review. Food Chem. 2024, 447, 138909. [Google Scholar] [CrossRef]
- Bui-Klimke, T.R.; Wu, F. Ochratoxin A and human health risk: A review of the evidence. Crit. Rev. Food. Sci. Nutr. 2015, 55, 1860–1869. [Google Scholar] [CrossRef] [PubMed]
- Coronel, M.B.; Marin, S.; Tarragó, M.; Cano-Sancho, G.; Ramos, A.J.; Sanchis, V. Ochratoxin A and its metabolite ochratoxin alpha in urine and assessment of the exposure of inhabitants of Lleida, Spain. Food Chem. Toxicol. 2011, 49, 1436–1442. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, K.; Blaszkewicz, M.; Degen, G.H. Simultaneous analysis of ochratoxin A and its major metabolite ochratoxin alpha in plasma and urine for an advanced biomonitoring of the mycotoxin. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2010, 878, 2623–2629. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) 2023/915 of 25 April 2023 on Maximum Levels for Certain Contaminants in Food and Repealing Regulation (EC) No 1881/2006; OJEU: Luxembourg, 2023; Volume L119, pp. 103–157. [Google Scholar]
- Schrenk, D.; Bodin, L.; Chipman, J.K.; Del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.R.; Leblanc, J.C.; Nebbia, C.S.; Nielsen, E.; et al. EFSA Panel on Contaminants in the Food Chain (CONTAM). Risk assessment of ochratoxin A in food. EFSA J. 2020, 18, e06113. [Google Scholar] [CrossRef]
- Singh, J.; Mehta, A. Rapid and sensitive detection of mycotoxins by advanced and emerging analytical methods: A review. Food Sci. Nutr. 2020, 8, 2183–2204. [Google Scholar] [CrossRef]
- Alhamoud, Y.; Yang, D.; Kenston, S.S.F.; Liu, G.; Liu, L.; Zhou, H.; Ahmed, F.; Zhao, J. Advances in biosensors for the detection of ochratoxin A: Bio-receptors, nanomaterials, and their applications. Biosens. Bioelectron. 2019, 141, 111418. [Google Scholar] [CrossRef]
- Li, X.; Ma, W.; Ma, Z.; Zhang, Q.; Li, H. Recent progress in determination of ochratoxin a in foods by chromatographic and mass spectrometry methods. Crit. Rev. Food Sci. Nutr. 2022, 62, 5444–5461. [Google Scholar] [CrossRef]
- Delfino, D.; Lucchetti, D.; Mauti, T.; Mancuso, M.; Di Giustino, P.; Triolone, D.; Vaccari, S.; Bonanni, R.C.; Neri, B.; Russo, K. Investigation of ochratoxin A in commercial cheeses and pork meat products by liquid chromatography-tandem mass spectrometry. J. Food Sci. 2022, 87, 4465–4475. [Google Scholar] [CrossRef]
- Ndoro, J.; Manduna, I.T.; Nyoni, M.; de Smidt, O. Multiple mycotoxin contamination in medicinal plants frequently sold in the free state province, South Africa detected using UPLC-ESI-MS/MS. Toxins 2022, 14, 690. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-S.; Zhao, J.; Ma, T.-T.; Li, Z.-Y.; Wang, L.-L.; Ji, S.-L.; Sun, M.-Y.; Liu, Y.-S.; Hu, Z.-H.; Liu, Q.-W.; et al. Magnetic covalent organic framework for effective solid-phase extraction and HPLC determination of ochratoxin A in food. LWT—Food Sci. Technol. 2023, 179, 114639. [Google Scholar] [CrossRef]
- Pakshir, K.; Dehghani, A.; Nouraei, H.; Zareshahrabadi, Z.; Zomorodian, K. Evaluation of fungal contamination and ochratoxin A detection in different types of coffee by HPLC-based method. J. Clin. Lab. Anal. 2021, 35, e24001. [Google Scholar] [CrossRef] [PubMed]
- Savastano, M.L.; Losito, I.; Pati, S. Rapid and automatable determination of ochratoxin A in wine based on microextraction by packed sorbent followed by HPLC-FLD. Food Control 2016, 68, 391–398. [Google Scholar] [CrossRef]
- Meira, D.I.; Barbosa, A.I.; Borges, J.; Reis, R.L.; Correlo, V.M.; Vaz, F. Label-free localized surface plasmon resonance (LSPR) biosensor, based on Au-Ag NPs embedded in TiO2 matrix, for detection of Ochratoxin-A (OTA) in wine. Talanta 2025, 284, 127238. [Google Scholar] [CrossRef]
- López-Puertollano, D.; Agulló, C.; Mercader, J.V.; Abad-Somovilla, A.; Abad-Fuentes, A. Immunoanalytical methods for ochratoxin A monitoring in wine and must based on innovative immunoreagents. Food Chem. 2021, 345, 128828. [Google Scholar] [CrossRef]
- Fan, X.; Tan, C.; Mei, X.; Ma, J.; Wu, K.; Deng, A.; Feng, X.; Li, J. Highly efficient electrochemiluminescent properties of porphyrin-based metal-organic framework Zn-TCPP and its immunoassay application to the detection of ochratoxin A. Anal. Chim. Acta 2024, 1330, 343267. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, X.; Yang, H.; Zhou, Y. Triple-readout immunoassay based on copper ion trigger for the detection of ochratoxin A. Anal. Chim. Acta 2025, 1345, 343750. [Google Scholar] [CrossRef]
- Kong, Y.; Qian, X.; Mei, X.; Ma, J.; Wu, K.; Deng, A.; Li, J. Electrochemiluminescence immunoassay system based on PCN-224-Mn and gold-platinum bimetallic nanoflowers for sensitive detection of ochratoxin A. Talanta 2025, 281, 126937. [Google Scholar] [CrossRef]
- Long, X.; Zhang, T.; Yang, L.; Guo, C.; Zhao, Q.; Cui, Y.; Wang, C.; Zhang, Y.; He, Y. CRISPR/Cas12a-based indirect competitive enzyme-linked immunosorbent assay for sensitive detection of ochratoxin A. J. Agric. Food Chem. 2024, 72, 21912–21921. [Google Scholar] [CrossRef]
- Tobias, C.; López-Puertollano, D.; Abad-Somovilla, A.; Mercader, J.V.; Abad-Fuentes, A.; Rurack, K. Development of simple and rapid bead-based cytometric immunoassays using superparamagnetic hybrid core-shell microparticles. ACS Meas. Sci. Au 2024, 4, 678–688. [Google Scholar] [CrossRef]
- de Andrade Silva, T.; Arcadio, F.; Zeni, L.; Martins, R.; de Oliveira, J.P.; Marques, C.; Cennamo, N. Plasmonic immunosensors based on spoon-shaped waveguides for fast and on-site ultra-low detection of ochratoxin A in coffee samples. Talanta 2024, 271, 125648. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Dong, Y.; Li, S.; Ma, X.; Pang, C.; Wang, S.; Zhang, J.; Chen, Y. A particle counting immunosensor for the sensitive detection of ochratoxin A via click chemistry-mediated signal amplification. J. Hazard. Mater. 2025, 488, 137381. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhao, X.; Song, X.; He, J. Diazo-functionalised immunoelectrochemical sensor for the detection of ochratoxin a in foods. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2024, 41, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jia, X.X.; Wang, Y.; Li, S.; Ren, S.; Wang, Y.; Han, D.; Qin, K.; Chang, X.; Zhou, H.; et al. A facile dual-mode immunosensor based on speckle Ag-doped nanohybrids for ultrasensitive detection of Ochratoxin A. Food Chem. 2024, 439, 138102. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, M.; Peng, D.; Zheng, H.; Qin, H.; Xiao, J.; Wu, Y.; Yang, N. A self-supported electrochemical immunosensor based on Cu2O/CuO@AuNPs heterostructures for sensitive and selective detection of ochratoxin A in food. Talanta 2025, 287, 127657. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, W.; Yang, J.; Li, Z.; Li, Q.; Xiao, L.; Tan, T.; Li, J. Photonic microbead array digital time-resolved fluorescence ultrasensitive platform for simultaneous detection of multiple mycotoxins. Anal. Chem. 2024, 96, 16842–16853. [Google Scholar] [CrossRef]
- Sheng, J.; Gao, H.; Zhang, M.; Xu, D. Simultaneous detection of five mycotoxins in traditional Chinese medicines (TCMs) by visual protein microarray. J. Pharm. Biomed. Anal. 2024, 249, 116333. [Google Scholar] [CrossRef]
- Leeman, D.; Allan, A.B.; Cameron, H.; Donelly, C.; Tramaseur, A.; Stratton, J.; MacDonald, S.J. Validation of the 11+ Myco MS-PREP® method for determination of aflatoxins, fumonisins, deoxynivalenol, ochratoxin A, zearalenone, HT-2, and T-2 toxins in cereals, baby food, spices, and animal feed by immunoaffinity column with LC-MS/MS: AOAC performance tested methodSM 112401. J. AOAC Int. 2025, 108, 207–252. [Google Scholar] [CrossRef]
- Andronaco, P.; Di Sanzo, R.; Ioppolo, F.; Ligato, F.; Alberto, S.; Galluccio, M.A.; Carabetta, S.; Russo, M. An Innovative analytical approach for multi-mycotoxin detection in craft beer using freeze-dried samples, IAC column and HPLC/ESI-MS/MS. Foods 2025, 14, 956. [Google Scholar] [CrossRef]
- Chu, F.S.; Chang, F.C.; Hinsdill, R.D. Production of antibody against ochratoxin A. Appl. Environ. Microbiol. 1976, 31, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Gyöngyösi-Horváth, A.; Barna-Vetró, I.; Solti, L. A new monoclonal antibody detecting ochratoxin A at the picogram level. Lett. Appl. Microbiol. 1996, 22, 103–105. [Google Scholar] [CrossRef]
- Mao, F.; He, Z.; Sun, Z.; Zhang, S.; Cao, H.; Liu, X. Plasmonic enzyme immunoassay via nanobody-driven controllable aggregation of gold nanoparticles for detection of ochratoxin A in pepper. Food Chem. 2024, 453, 139623. [Google Scholar] [CrossRef]
- Zhang, Y.; He, Z.; Sun, Z.; Zhang, S.; Liu, X. Genetic engineering-powered dual-mode lateral flow immunosensor for colorimetric and fluorescent detection of ochratoxin A in pepper. J. Hazard. Mater. 2025, 489, 137636. [Google Scholar] [CrossRef]
- Chen, X.; Gao, D.; Sun, F.; Li, Z.; Wang, Y.; Qiu, C.; He, K.; Wang, J. Nanomaterial-based aptamer biosensors for ochratoxin A detection: A review. Anal. Bioanal. Chem. 2022, 414, 2953–2969. [Google Scholar] [CrossRef]
- Huang, H.; Ouyang, W.; Feng, K.; Camarada, M.B.; Liao, T.; Tang, X.; Liu, R.; Hou, D.; Liao, X. Rational design of molecularly imprinted electrochemical sensor based on Nb2C-MWCNTs heterostructures for highly sensitive and selective detection of Ochratoxin a. Food Chem. 2024, 456, 140007. [Google Scholar] [CrossRef]
- Duncan, H.; Agulló, C.; Mercader, J.V.; Abad-Somovilla, A.; Abad-Fuentes, A. Harnessing the intrinsic chemical reactivity of the mycotoxin patulin for immunosensing. Anal. Chem. 2024, 96, 12370–12377. [Google Scholar] [CrossRef]
- Duncan, H.; Mercader, J.V.; Agulló, C.; Gil-Sepulcre, M.; Abad-Somovilla, A.; Abad-Fuentes, A. Chemical strategies for triggering the immune response to the mycotoxin patulin. Sci. Rep. 2021, 11, 23438. [Google Scholar] [CrossRef]
- Ceballos-Alcantarilla, E.; Abad-Somovilla, A.; Agulló, C.; Abad-Fuentes, A.; Mercader, J.V. Protein-free hapten-carbon nanotube constructs induce the secondary immune response. Bioconjug. Chem. 2017, 28, 1630–1638. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, R.; White, P.; Offer, J. Advances in Fmoc solid-phase peptide synthesis. J. Pept. Sci. 2016, 22, 4–27. [Google Scholar] [CrossRef] [PubMed]
- Fields, G.B.; Noble, R.L. Solid phase peptide synthesis utilizing 9-fluorenylmethoxycarbonyl amino acids. Int. J. Pept. Protein Res. 1990, 35, 161–214. [Google Scholar] [CrossRef]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: Behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 2004, 37, 790–802. [Google Scholar] [CrossRef]
- Vasylieva, N.; Barnych, B.; Rand, A.; Inceoglu, B.; Gee, S.J.; Hammock, B.D. Sensitive immunoassay for detection and quantification of the neurotoxin, tetramethylenedisulfotetramine (TETS). Anal. Chem. 2017, 89, 5612–5619. [Google Scholar] [CrossRef] [PubMed]
- Karachaliou, C.E.; Koukouvinos, G.; Zisis, G.; Kizis, D.; Krystalli, E.; Siragakis, G.; Goustouridis, D.; Kakabakos, S.; Petrou, P.; Livaniou, E.; et al. Fast and accurate determination of minute ochratoxin A levels in cereal flours and wine with the label-free white light reflectance spectroscopy biosensing platform. Biosensors 2022, 12, 877. [Google Scholar] [CrossRef] [PubMed]
- Koukouvinos, G.; Karachaliou, C.-E.; Kanioura, A.; Tsougeni, K.; Livaniou, E.; Kakabakos, S.E.; Petrou, P.S. Fluorescence enhancement on silver-plated plasma micro-nanostructured 3D polymeric microarray substrates for multiplex mycotoxin detection. Processes 2021, 9, 392. [Google Scholar] [CrossRef]
- López-Puertollano, D.; Agulló, C.; Mercader, J.V.; Abad-Somovilla, A.; Abad-Fuentes, A. Click chemistry-assisted bioconjugates for hapten immunodiagnostics. Bioconjug. Chem. 2020, 31, 956–964. [Google Scholar] [CrossRef]
- López-Puertollano, D.; Mercader, J.V.; Agulló, C.; Abad-Somovilla, A.; Abad-Fuentes, A. Novel haptens and monoclonal antibodies with subnanomolar affinity for a classical analytical target, ochratoxin A. Sci. Rep. 2018, 8, 9761. [Google Scholar] [CrossRef]
- Papasarantos, I.; Klimentzou, P.; Koutrafouri, V.; Anagnostouli, M.; Zikos, C.; Paravatou-Petsotas, M.; Livaniou, E. Solid-phase synthesis of a biotin derivative and its application to the development of anti-biotin antibodies. Appl. Biochem. Biotechnol. 2010, 162, 221–232. [Google Scholar] [CrossRef]
- Sauceda-Friebe, J.C.; Karsunke, X.Y.; Vazac, S.; Biselli, S.; Niessner, R.; Knopp, D. Regenerable immuno-biochip for screening ochratoxin A in green coffee extract using an automated microarray chip reader with chemiluminescence detection. Anal. Chim. Acta 2011, 689, 234–242. [Google Scholar] [CrossRef]
- Cagnasso, I.; Tonachini, G.; Berto, S.; Giacomino, A.; Mandrile, L.; Maranzana, A.; Durbiano, F. Comprehensive study on the degradation of ochratoxin A in water by spectroscopic techniques and DFT calculations. RSC Adv. 2019, 9, 19844–19854. [Google Scholar] [CrossRef]
- Sueck, F.; Hemp, V.; Specht, J.; Torres, O.; Cramer, B.; Humpf, H.U. Occurrence of the ochratoxin A degradation product 2′R-ochratoxin A in coffee and other food: An update. Toxins 2019, 11, 329. [Google Scholar] [CrossRef] [PubMed]
- Zapaśnik, A.; Bryła, M.; Waśkiewicz, A.; Ksieniewicz-Woźniak, E.; Podolska, G. Ochratoxin A and 2′R-ochratoxin A in selected foodstuffs and dietary risk assessment. Molecules 2021, 27, 188. [Google Scholar] [CrossRef] [PubMed]
- Bittner, A.; Cramer, B.; Harrer, H.; Humpf, H.U. Structure elucidation and in vitro cytotoxicity of ochratoxin α amide, a new degradation product of ochratoxin A. Mycotoxin Res. 2015, 31, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Duengo, S.; Muhajir, M.I.; Hidayat, A.T.; Musa, W.J.A.; Maharani, R. Epimerisation in peptide synthesis. Molecules 2023, 28, 8017. [Google Scholar] [CrossRef]
- Brenna, E.; Dalla Santa, F.; Gatti, F.G.; Gatti, G.; Tessaro, D. Exploiting the vicinal disubstituent effect on the diastereoselective synthesis of γ and δ lactones. Org. Biomol. Chem. 2019, 17, 813–821. [Google Scholar] [CrossRef]
- Baig, M.A.; Banthorpe, D.V.; Carr, G.; Whittaker, D. Reactions of some cyclic ethers in superacids. J. Chem. Soc. Perkin Trans. 2 1989, 12, 1981–1986. [Google Scholar] [CrossRef]
- Bouisseau, A.; Roland, A.; Reillon, F.; Schneider, R.; Cavelier, F. First synthesis of a stable isotope of Ochratoxin A metabolite for a reliable detoxification monitoring. Org. Lett. 2013, 15, 3888–3890. [Google Scholar] [CrossRef]
- Gillman, I.G.; Clark, T.N.; Manderville, R.A. Oxidation of ochratoxin A by an Fe-porphyrin system: Model for enzymatic activation and DNA cleavage. Chem. Res. Toxicol. 1999, 12, 1066–1076. [Google Scholar] [CrossRef]
- Sanders, M.; Guo, Y.; Iyer, A.; García, Y.R.; Galvita, A.; Heyerick, A.; Deforce, D.; Risseeuw, M.D.; Van Calenbergh, S.; Bracke, M.; et al. An immunogen synthesis strategy for the development of specific anti-deoxynivalenol monoclonal antibodies. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 1751–1759. [Google Scholar] [CrossRef]
- Subirós-Funosas, R.; Prohens, R.; Barbas, R.; El-Faham, A.; Albericio, F. Oxyma: An efficient additive for peptide synthesis to replace the benzotriazole-based HOBt and HOAt with a lower risk of explosion. Chemistry 2009, 15, 9394–9403. [Google Scholar] [CrossRef]
- Sarin, V.K.; Kent, S.B.; Tam, J.P.; Merrifield, R.B. Quantitative monitoring of solid-phase peptide synthesis by the ninhydrin reaction. Anal. Biochem. 1981, 117, 147–157. [Google Scholar] [CrossRef]
- Vaitukaitis, J.L. Production of antisera with small doses of immunogen: Multiple intradermal injections. Methods Enzymol. 1981, 73 Pt B, 46–52. [Google Scholar] [CrossRef]



| Assay Parameter | Value |
|---|---|
| Assay time 1 | 4 h |
| LoD | 0.1 ng/mL in assay buffer 1 ng/mL in white and red wine |
| Accuracy | 86.3–115.1% in white and red wine |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karachaliou, C.-E.; Zikos, C.; Liolios, C.; Pelecanou, M.; Livaniou, E. Development of a Polyclonal Antibody for the Immunoanalysis of Ochratoxin A (OTA) by Employing a Specially Designed Synthetic OTA Derivative as the Immunizing Hapten. Toxins 2025, 17, 415. https://doi.org/10.3390/toxins17080415
Karachaliou C-E, Zikos C, Liolios C, Pelecanou M, Livaniou E. Development of a Polyclonal Antibody for the Immunoanalysis of Ochratoxin A (OTA) by Employing a Specially Designed Synthetic OTA Derivative as the Immunizing Hapten. Toxins. 2025; 17(8):415. https://doi.org/10.3390/toxins17080415
Chicago/Turabian StyleKarachaliou, Chrysoula-Evangelia, Christos Zikos, Christos Liolios, Maria Pelecanou, and Evangelia Livaniou. 2025. "Development of a Polyclonal Antibody for the Immunoanalysis of Ochratoxin A (OTA) by Employing a Specially Designed Synthetic OTA Derivative as the Immunizing Hapten" Toxins 17, no. 8: 415. https://doi.org/10.3390/toxins17080415
APA StyleKarachaliou, C.-E., Zikos, C., Liolios, C., Pelecanou, M., & Livaniou, E. (2025). Development of a Polyclonal Antibody for the Immunoanalysis of Ochratoxin A (OTA) by Employing a Specially Designed Synthetic OTA Derivative as the Immunizing Hapten. Toxins, 17(8), 415. https://doi.org/10.3390/toxins17080415






