Fungal Species and Mycotoxins Associated with Maize Ear Rots Collected from the Eastern Cape in South Africa
Abstract
1. Introduction
2. Results
2.1. Ear Rot Survey
2.2. Mycotoxins
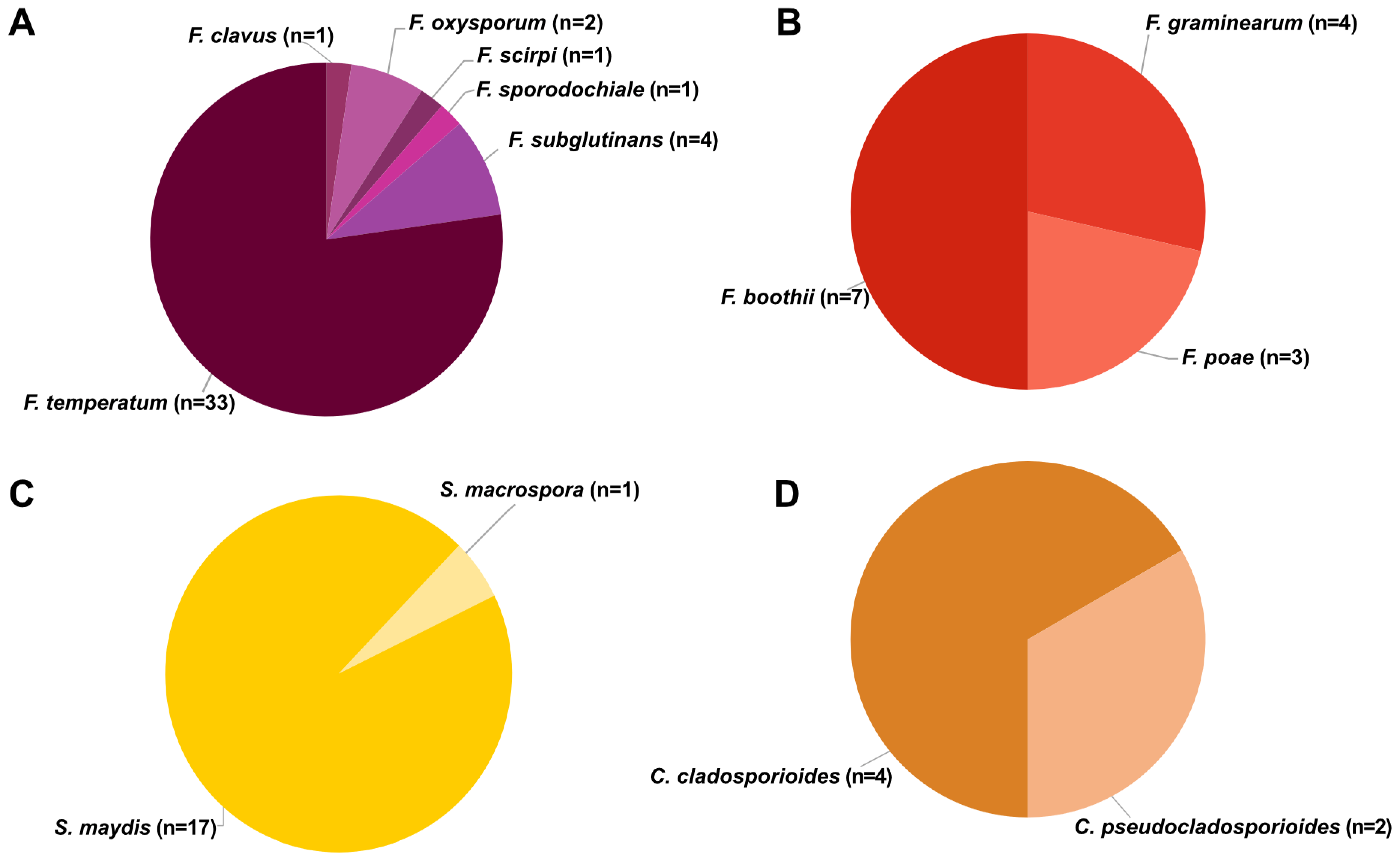
2.3. Fungal Identification
3. Discussion
4. Materials and Methods
4.1. Ear Rot Survey and Sample Collection
4.2. Mycotoxin Sample Preparation
4.3. Mycotoxin Analysis
4.4. Fungal Isolation and Identification
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chukwudi, U.P.; Kutu, F.R.; Mavengahama, S. Mycotoxins in maize and implications on food security: A Review. Agric. Rev. 2021, 42, 42–49. [Google Scholar] [CrossRef]
- Gravelet-Blondin, R. Prospectus on the South African Maize Industry. Available online: https://sacota.co.za/wp-content/uploads/Prospectus-on-the-South-African-Maize-Industry (accessed on 12 March 2020).
- Aveling, T.A.S.; de Ridder, K.; Olivier, N.A.; Berger, D.K. Seasonal variation in mycoflora associated with asymptomatic maize grain from small-holder farms in two provinces of South Africa. J. Agric. Rural Dev. Trop. Subtrop. 2020, 121, 265–275. [Google Scholar] [CrossRef]
- Logrieco, A.; Battilani, P.; Leggieri, M.C.; Jiang, Y.; Haesaert, G.; Lanubile, A.; Mahuku, G.; Mesterházy, A.; Ortega-Beltran, A.; Pasti, M.; et al. Perspectives on global mycotoxin issues and management from the MycoKey maize working group. Plant Dis. 2021, 105, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Boutigny, A.L.; Beukes, I.; Small, I.; Zühlke, S.; Spiteller, M.; Van Rensburg, B.J.; Flett, B.; Viljoen, A. Quantitative detection of Fusarium pathogens and their mycotoxins in South African maize. Plant Pathol. 2012, 61, 522–531. [Google Scholar] [CrossRef]
- White, D.G.; Carson, M.L.; Munkvold, G.P. Compendium of Corn Diseases, 4th ed.; Munkvold, G.P., White, D.G., Eds.; American Phytopathological Society Press: St. Paul, MN, USA, 2016; Volume 165, p. 165. [Google Scholar]
- Mabuza, L. Monitoring Fusarium, Gibberella and Diplodia Ear Rots and Associated Mycotoxins in Maize Grown under Different Cropping Systems. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, 2017. [Google Scholar]
- Flett, B. A Look at the Most Important Ear Rots in Maize Production. Available online: https://www.grainsa.co.za/a-look-at-the-most-important-ear-rots-in-maize-production#:~:text=The%20major%20maize%20ear%20rot,and%20potential%20human%20toxicity%20problems (accessed on 15 April 2022).
- Reid, L.M.; Nicol, R.W.; Ouellet, T.; Savard, M.; Miller, J.D.; Young, J.C.; Stewart, D.W.; Schaafsma, A.W. Interaction of Fusarium graminearum and F. Moniliforme in maize ears: Disease progress, fungal biomass, and mycotoxin accumulation. Phytopathology 1999, 89, 1028–1037. [Google Scholar] [CrossRef] [PubMed]
- Fandohan, P.; Hell, K.; Marasas, W.F.O.; Wingfield, M.J. Infection of maize by Fusarium species and contamination with fumonisin in Africa. Afr. J. Biotechnol. 2003, 2, 570–579. [Google Scholar] [CrossRef]
- Munkvold, G.P.; Proctor, R.H.; Moretti, A. Mycotoxin production in Fusarium according to contemporary species concepts. Annu. Rev. Phytopathol. 2021, 59, 373–402. [Google Scholar] [CrossRef]
- Munkvold, G.P. Fusarium species and their associated mycotoxins. Methods Mol. Biol. 2017, 1542, 51–106. [Google Scholar]
- Logrieco, A.; Mule, G.; Moretti, A.; Bottalico, A. Toxigenic Fusarium species and mycotoxins associated with maize ear rot in Europe. In Mycotoxins in Plant Disease; Logrieco, A., Bailey, J.A., Corazza, L., Cooke, B.M., Eds.; Springer: Dordrecht, The Netherlands, 2002; pp. 597–609. [Google Scholar]
- Bottalico, A. Fusarium diseases of cereals: Species complex and related mycotoxin profiles, in Europe. J. Plant Pathol. 1998, 80, 85–103. [Google Scholar]
- Nelson, P.E. Taxonomy and biology of Fusarium moniliforme. Mycopathologia 1992, 117, 29–36. [Google Scholar] [CrossRef]
- Logrieco, A.; Moretti, A.; Perrone, G.; Mulè, G. Biodiversity of complexes of mycotoxigenic fungal species associated with Fusarium ear rot of maize and Aspergillus rot of grape. Int. J. Food Microbiol. 2007, 119, 11–16. [Google Scholar] [CrossRef]
- Pitt, J.I. Toxigenic fungi and mycotoxins. Br. Med. Bull. 2000, 56, 184–192. [Google Scholar] [CrossRef]
- Lillehoj, E.B.; Fennell, D.I.; Kwolek, W.F. Aspergillus flavus and aflatoxin in Iowa corn before harvest. Science 1976, 193, 495–496. [Google Scholar] [CrossRef]
- Shotwell, O.L. Mycotoxins in hot spots in grains. I. Aflatoxin and zearalenone occurrence in stored corn. Cereal Chem. 1975, 52, 687–697. [Google Scholar]
- Sweeney, M.J.; Dobson, A.D.W. Mycotoxin production by Aspergillus, Fusarium and Penicillium species. Int. J. Food Microbiol. 1998, 43, 141–158. [Google Scholar] [CrossRef] [PubMed]
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajšlová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nut. 2020, 60, 2773–2789. [Google Scholar] [CrossRef]
- Bezuidenhout, S.C.; Gelderblom, W.C.A.; Gorst-Allman, C.P.; Horak, R.M.; Marasas, W.F.O.; Spiteller, G.; Vleggaar, R. Structure elucidation of the fumonisins, mycotoxins from Fusarium moniliforme. J. Chem. Soc., Chem. Commun. 1988, 11, 743–745. [Google Scholar] [CrossRef]
- Rheeder, J.P.; Van der Westhuizen, L.; Imrie, G.; Shephard, G.S. Fusarium species and fumonisins in subsistence maize in the former Transkei region, South Africa: A multi-year study in rural villages. Food Addit. Contam. Part B 2016, 9, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, W.C.; Jaskiewicz, K.; Marasas, W.F.; Thiel, P.G.; Horak, R.M.; Vleggaar, R.; Kriek, N.P. Fumonisins—Novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1988, 54, 1806–1811. [Google Scholar] [CrossRef] [PubMed]
- Flett, B.C.; McLaren, N.W.; Wehner, F.C. Incidence of ear rot pathogens under alternating corn tillage practices. Plant Dis. 1998, 82, 781–784. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Flett, B.C.; Wehner, F.C. Incidence of Stenocarpella and Fusarium cob rots in monoculture maize under different tillage systems. J. Phytopathol. 1991, 133, 327–333. [Google Scholar] [CrossRef]
- Ncube, E.; Flett, B.C.; Van den Berg, J.; Erasmus, A.; Viljoen, A. Fusarium ear rot and fumonisins in maize kernels when comparing a Bt hybrid with its non-Bt isohybrid and under conventional insecticide control of Busseola fusca infestations. J. Crop Prot. 2018, 110, 183–190. [Google Scholar] [CrossRef]
- Phokane, S. Mycotoxin Contamination of Maize and Groundnut Produced by Subsistence Farmers in Northern KwaZulu-Natal. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, 2018. [Google Scholar]
- Rheeder, J.P.; Marasas, W.F.O.; Van Wyk, P.S.; Du Toit, W.; Pretorius, A.J.; Van Schalkwyk, D.J. Incidence of Fusarium and Diplodia species and other fungi in naturally infected grain of South African maize cultivars. Phytophylactica 1990, 22, 97–102. [Google Scholar]
- Flett, B.C.; McLaren, N.W.; Wehner, F.C. Incidence of Stenocarpella maydis ear rot of corn under crop rotation systems. Plant Dis. 2001, 85, 92–94. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Multi-mycotoxin screening of feed and feed raw materials from Africa. World Mycotoxin J. 2018, 11, 369–383. [Google Scholar] [CrossRef]
- Janse van Rensburg, B.; McLaren, N.W.; Flett, B.C.; Schoeman, A. Fumonisin producing Fusarium spp. and fumonisin contamination in commercial South African maize. Eur. J. Plant Pathol. 2015, 141, 491–504. [Google Scholar] [CrossRef]
- Meyer, H.; Skhosana, Z.D.; Motlanthe, M.; Louw, W.; Rohwer, E. Long term monitoring (2014–2018) of multi-mycotoxins in South African commercial maize and wheat with a locally developed and validated LC-MS/MS method. Toxins 2019, 11, 271. [Google Scholar] [CrossRef]
- Ekwomadu, T.I.; Dada, T.A.; Nleya, N.; Gopane, R.; Sulyok, M.; Mwanza, M. Variation of Fusarium free, masked, and emerging mycotoxin metabolites in maize from agriculture regions of South Africa. Toxins 2020, 12, 149. [Google Scholar] [CrossRef]
- Ekwomadu, T.I.; Dada, T.A.; Akinola, S.A.; Nleya, N.; Mwanza, M. Analysis of selected mycotoxins in maize from North-West South Africa using high performance liquid chromatography (HPLC) and other analytical techniques. Separations 2021, 8, 143–155. [Google Scholar] [CrossRef]
- Rheeder, J.; Marasas, W.; van Schalkwyk, D. Incidence of Fusarium and Diplodia species in naturally infected grain of South African maize cultivars: A follow-up study. Phytophylactica 1993, 25, 43–48. [Google Scholar]
- Czembor, E.; Stępień, Ł.; Waśkiewicz, A. Fusarium temperatum as a new species causing ear rot on maize in Poland. Plant Dis. 2014, 98, 1001–1019. [Google Scholar] [CrossRef]
- Fumero, M.V.; Reynoso, M.M.; Chulze, S. Fusarium temperatum and Fusarium subglutinans isolated from maize in Argentina. Int. J. Food Microbiol. 2015, 199, 86–92. [Google Scholar] [CrossRef]
- Njeru, N.K.; Midega, C.A.O.; Muthomi, J.W.; Wagacha, J.M.; Khan, Z.R. Impact of push–pull cropping system on pest management and occurrence of ear rots and mycotoxin contamination of maize in western Kenya. Plant Pathol. 2020, 69, 1644–1654. [Google Scholar] [CrossRef]
- Pfordt, A.; Ramos, R.L.; Schiwek, S.; Karlovsky, P.; von Tiedemann, A. Impact of environmental conditions and agronomic practices on the prevalence of Fusarium species associated with ear- and stalk rot in maize. Pathogens 2020, 9, 236. [Google Scholar] [CrossRef]
- Robertson, A.E.; Munkvold, G.P.; Hurburgh, C.R.; Ensley, S. Effects of natural hail damage on ear rots, mycotoxins, and grain quality characteristics of corn. J. Agron. 2011, 103, 193–199. [Google Scholar] [CrossRef]
- Stępień, Ł.; Gromadzka, K.; Chełkowski, J.; Basińska-Barczak, A.; Lalak-Kańczugowska, J. Diversity and mycotoxin production by Fusarium temperatum and Fusarium subglutinans as causal agents of pre-harvest Fusarium maize ear rot in Poland. J. Appl. Genet. 2019, 60, 113–121. [Google Scholar] [CrossRef]
- Zhang, H.; Brankovics, B.; Luo, W.; Xu, J.; Xu, J.S.; Guo, C.; Guo, J.G.; Jin, S.L.; Chen, W.Q.; Feng, J. Crops are a main driver for species diversity and the toxigenic potential of Fusarium isolates in maize ears in China. World Mycotoxin J. 2016, 9, 701–715. [Google Scholar] [CrossRef]
- Marasas, W.F.O.; Jaskeiwicz, K.; Venter, F.S.; van Schalkwyk, D.J. Fusarium moniliforme contamination of maize in oesophageal cancer areas in Transkei. S. Afr. Med. J. 1988, 74, 110–114. [Google Scholar] [PubMed]
- Marasas, W.F.O.; Naude, T.W.; Pienaar, J.G.; Kellerman, T.S. Leukoencephalomacia: A mycotoxicosis of equidae caused by Fusarium moniliforme Sheldon. Onderstpoort J. Vet. Res. 1976, 43, 113–122. [Google Scholar]
- Marasas, W.F.O.; Wehner, F.C.; Van Rensburg, S.J.; Van Schalkwyk, D.J. Mycoflora of corn produced in human esophageal cancer areas in Transkei, southern Africa. Phytopathology 1981, 71, 792–796. [Google Scholar] [CrossRef]
- Steenkamp, E.T.; Wingfield, B.D.; Desjardins, A.E.; Marasas, W.F.O.; Wingfield, M.J. Cryptic speciation in Fusarium subglutinans. Mycologia 2002, 94, 1032–1043. [Google Scholar] [CrossRef]
- O’Donnell, K.; Cigelnik, E.; Nirenberg, H.I. Molecular systematics and phylogeography of the Gibberella fujikuroi species complex. Mycologia 1998, 90, 465–493. [Google Scholar] [CrossRef]
- Scauflaire, J.; Gourgue, M.; Munaut, F. Fusarium temperatum sp. nov. from maize, an emergent species closely related to Fusarium subglutinans. Mycologia 2011, 103, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Munkvold, G.P.; Arias, S.; Taschl, I.; Gruber-Dorninger, C. Mycotoxins in corn: Occurrence, impacts, and management. In Corn, 3rd ed.; Serna-Saldivar, S.O., Ed.; AACC International Press: Oxford, UK, 2019; pp. 235–287. [Google Scholar]
- Vigier, B.; Reid, L.M.; Seifert, K.A.; Stewart, D.S.W.; Hamilton, R.I. Distribution and prediction of Fusarium species associated with maize ear rot in Ontario. Can. J. Plant Pathol. 1997, 19, 60–65. [Google Scholar] [CrossRef]
- Nelson, P.E.; Plattner, R.D.; Shackelford, D.D.; Desjardins, A.E. Fumonisin B1 production by Fusarium species other than F. moniliforme in section Liseola and by some related species. Appl. Environ. Microbiol. 1992, 58, 984–989. [Google Scholar] [CrossRef] [PubMed]
- Fumero, V.M.; Villani, A.; Susca, A.; Haidukowski, M.; Cimmarusti, M.T.; Toomajian, C.; Leslie, J.F.; Chulze, S.N.; Moretti, A. Fumonisin and beauvericin chemotypes and genotypes of the sister species Fusarium subglutinans and Fusarium temperatum. Appl. Environ. Microbiol. 2020, 86, e00133-00120. [Google Scholar] [CrossRef] [PubMed]
- Jestoi, M. Emerging Fusarium -mycotoxins fusaproliferin, beauvericin, enniatins, and moniliformin—A review. Crit. Rev. Food Sci. Nutr. 2008, 48, 21–49. [Google Scholar] [CrossRef] [PubMed]
- Kouri, K.; Lemmens, M.; Lemmens-Gruber, R. Beauvericin-induced channels in ventricular myocytes and liposomes. Biochim Biophys. Acta (BBA)—Biomembr. 2003, 1609, 203–210. [Google Scholar] [CrossRef]
- Boutigny, A.; Ward, T.J.; Van Coller, G.J.; Flett, B.; Lamprecht, S.C.; O’Donnell, K.; Viljoen, A. Analysis of the Fusarium graminearum species complex from wheat, barley and maize in South Africa provides evidence of species-specific differences in host preference. Fungal Genet. Biol. 2011, 48, 914–920. [Google Scholar] [CrossRef]
- Laraba, I.; McCormick, S.P.; Vaughan, M.M.; Geiser, D.M.; O’Donnell, K. Phylogenetic diversity, trichothecene potential, and pathogenicity within Fusarium sambucinum species complex. PLoS ONE 2021, 16, e0245037. [Google Scholar] [CrossRef]
- Boutigny, A.L.; Beukes, I.; Viljoen, A. Head blight of barley in South Africa is caused by Fusarium graminearum with a 15-ADON chemotype. J. Plant Pathol. 2011, 93, 321–329. [Google Scholar]
- Beukes, I.; Rose, L.J.; Shephard, G.S.; Flett, B.C.; Viljoen, A. Mycotoxigenic Fusarium species associated with grain crops in South Africa-A review. S. Afr. J. Sci. 2017, 113, 1–12. [Google Scholar] [CrossRef]
- Phoku, J.Z.; Dutton, M.F.; Njobeh, P.B.; Mwanza, M.; Egbuta, M.A.; Chilaka, C.A. Fusarium infection of maize and maize-based products and exposure of a rural population to fumonisin B1 in Limpopo Province, South Africa. Food Addit. Contam. Part A 2012, 29, 1743–1751. [Google Scholar] [CrossRef]
- Hocking, A.D.; Pitt, J.I.; Samson, R.A.; Thrane, U. Advances in Food Mycology; Springer: Berlin/Heidelberg, Germany, 2006; Volume 384. [Google Scholar]
- Miller, J.D. Significance of grain mycotoxins for health and nutrition. In Proceedings of the Fungi and Mycotoxins in Stored Products, Bangkok, Thailand, 23–26 April 1991; pp. 126–135. [Google Scholar]
- Stob, M.; Baldwin, R.S.; Tuite, J.; Andrews, F.N.; Gillette, K.G. Isolation of an anabolic, uterotrophic compound from corn infected with Gibberella zeae. Nature 1962, 196, 1318. [Google Scholar] [CrossRef] [PubMed]
- DSM. DSM World Mycotoxin Survey 2021. Available online: https://www.dsm.com/anh/news/downloads/whitepapers-and-reports/dsm-mycotoxin-survey-2021-focus-ruminants.html (accessed on 24 January 2024).
- Biomin. Biomin World Mycotoxin Survey 2019. Available online: https://www.biomin.net/science-hub/world-mycotoxin-survey-impact-2020/ (accessed on 24 January 2020).
- Biomin. Biomin World Mycotoxin Survey 2020. Available online: https://www.biomin.net/science-hub/world-mycotoxin-survey-impact-2021/ (accessed on 24 January 2024).
- van der Lee, T.; Zhang, H.; van Diepeningen, A.; Waalwijk, C. Biogeography of Fusarium graminearum species complex and chemotypes: A Review. Food Addit. Contam. Part A 2015, 32, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Rheeder, J.P.; Marasas, W.F.; Thiel, P.G.; Sydenham, E.W.; Shephard, G.S.; Van Schalkwyk, D.J. Fusarium moniliforme and fumonisins in corn in relation to human esophageal cancer in Transkei. Post Pathol. Mycotoxins 1992, 82, 353–357. [Google Scholar]
- Ncube, E.; Flett, B.C.; Waalwijk, C.; Viljoen, A. Fusarium spp. and levels of fumonisins in maize produced by subsistence farmers in South Africa: Research article. S. Afr. J. Sci. 2011, 107, 1–7. [Google Scholar] [CrossRef]
- Miller, J.D. Fungi and mycotoxins in grain: Implications for stored product research. J. Stored Prod. Res. 1995, 31, 1–16. [Google Scholar] [CrossRef]
- Lamprecht, S.C.; Crous, P.W.; Groenewald, J.Z.; Tewoldemedhin, Y.T.; Marasas, W.F.O. Diaporthaceae associated with root and crown rot of maize. IMA Fungus 2011, 2, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Wicklow, D.T.; Rogers, K.D.; Dowd, P.F.; Gloer, J.B. Bioactive metabolites from Stenocarpella maydis, a stalk and ear rot pathogen of maize. Fungal Biol. 2011, 115, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Shurtleff, M.C. Compendium of Corn Diseases, 2nd ed.; American Phytopathological Society Press: St. Paul, MN, USA, 1980; p. 105. [Google Scholar]
- Kellerman, T.S.; Schultz, R.A.; Rabie, C.J.; Van Ark, H.; Maartens, B.P.; Lubben, A.; Prozesky, L. Perinatal mortality in lambs of ewes exposed to cultures of Diplodia maydis (=Stenocarpella maydis) during gestation. Onderstpoort J. Vet. Res. 1991, 58, 297–308. [Google Scholar]
- Steyn, P.S.; Wessels, P.L.; Holzapfel, C.W.; Potgieter, D.J.J.; Louw, W.K.A. The isolation and structure of a toxic metabolite from Diplodia maydis (Berk.) Sacc. Tetrahedron 1972, 28, 4775–4785. [Google Scholar] [CrossRef]
- Odriozola, E.; Odeón, A.; Canton, G.; Clemente, G.; Escande, A. Diplodia maydis: A cause of death of cattle in Argentina. N. Z. Vet. J. 2005, 53, 160–161. [Google Scholar] [CrossRef]
- Riet-Correa, F.; Rivero, R.; Odriozola, E.; Adrien, M.d.L.; Medeiros, R.M.T.; Schild, A.L. Mycotoxicoses of ruminants and horses. J. Vet. Diagn. Investig. 2013, 25, 692–708. [Google Scholar] [CrossRef]
- Zaccaron, A.Z.; Woloshuk, C.P.; Bluhm, B.H. Comparative genomics of maize ear rot pathogens reveals expansion of carbohydrate-active enzymes and secondary metabolism backbone genes in Stenocarpella maydis. Fungal Biol. 2017, 121, 966–983. [Google Scholar] [CrossRef] [PubMed]
- Botts, E.A. Case Study: Local Government and Civil Society: Climate Change Response in Alfred Nzo District Municipality; SANBI: Rhodes Drive, Newlands, 2015. [Google Scholar]
- Nkamisa, M.; Ndhleve, S.; Nakin, M.D.V.; Mngeni, A.; Kabiti, H.M. Analysis of trends, recurrences, severity and frequency of droughts using standardised precipitation index: Case of OR Tambo District Municipality, Eastern Cape, South Africa. Jamba 2022, 14, 1147. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.F.O.; Kellerman, T.S.; Gelderblom, W.C.A.; Thiel, P.G.; Van der Lugt, J.J.; Coetzer, J.A.W. Leukoencephalomalacia in a horse induced by fumonisin B₁ isolated from Fusarium moniliforme. Onderstpoort J. Vet. Res. 1988, 55, 197–203. [Google Scholar]
- Marasas, W.F.O.; Van Rensburg, S.J.; Mirocha, C.J. Incidence of Fusarium species and the mycotoxins, deoxynivalenol and zearalenone, in corn produced in esophageal cancer areas in Transkei. J. Agric. Food Chem. 1979, 27, 1108–1112. [Google Scholar] [CrossRef] [PubMed]
- Bayer. Gibberella Ear Rot and Stalk Rot in Corn. Available online: https://www.cropscience.bayer.ca/en/articles/2021/gibberella-ear-rot-and-stalk-rot-in-corn (accessed on 4 April 2021).
- Pierce, P. Corn Ear Rots: Identification, Quantification and Testing for Mycotoxins. Available online: https://agcrops.osu.edu/newsletter/corn-newsletter/2016-32/corn-ear-rots-identification-quantification-and-testing (accessed on 4 April 2021).
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- De Hoog, G.S.; van den Ende, A.H.G. Molecular diagnostics of clinical strains of filamentous Basidiomycetes: Molekulare Diagnostik klinischer Stämme filamentöser Basidiomyzeten. Mycoses 1998, 41, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Masclaux, F.; Guého, E.; de Hoog, G.S.; Christen, R. Phylogenetic relationships of human-pathogenic Cladosporium (Xylohypha) species inferred from partial LSU rRNA sequences. Med. Mycol. J. 1995, 33, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Vilgalys, R.; Hester, H. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [PubMed]
- Rehner, S.A.; Samuels, G.J. Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol. Res. 1994, 98, 625–634. [Google Scholar] [CrossRef]
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous Ascomycetes. Mycologia 1999, 91, 553–556. [Google Scholar] [CrossRef]
- Geiser, D.M.; del Mar Jiménez-Gasco, M.; Kang, S.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’Donnell, K. FUSARIUM-ID v. 1.0: A DNA Sequence Database for Identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479. [Google Scholar] [CrossRef]

| Farm Number | Ear Rot | Species Detected | District e | General Disease | |
|---|---|---|---|---|---|
| Incidence (%) | Severity (Average) | ||||
| Farm 1 | CER a | C. pseudocladosporioides * | AN | 14 | 1 |
| DER b | S. maydis ** | ||||
| FER c | F. temperatum *** | ||||
| Farm 2 | DER | S. maydis | AN | 6 | 1 |
| FER | F. temperatum | ||||
| Farm 3 | FER | F. temperatum | AN | 2 | 2 |
| Farm 4 | CER | C. cladosporioides, C. pseudocladosporioides | CH | 3 | 2 |
| FER | F. temperatum, F. scirpi | ||||
| Farm 5 | DER | S. maydis | CH | 35 | 7 |
| FER | F. oxysporum, F. temperatum | ||||
| GER d | F. boothii, F. poae | ||||
| Farm 6 | DER | S. maydis | CH | 5 | 2 |
| FER | F. clavus, F. oxysporum, F. subglutinans, F. temperatum | ||||
| Farm 7 | FER | F. oxysporum, F. temperatum | JG | 3 | 2 |
| GER | F. boothii, F. graminearum, F. poae | ||||
| Farm 8 | CER | C. cladosporioides | JG | 6 | 1 |
| Farm 9 | CER | C. cladosporioides | JG | 11 | 1 |
| Farm 10 | DER | S. macrospora, S. maydis | ORT | 2 | 2 |
| FER | F. temperatum | ||||
| Farm 11 | FER | F. subglutinans, F. sporodochiale | ORT | 1 | 2 |
| Farm 12 | CER | C. cladosporioides | ORT | 0.8 | 1 |
| DER | S. maydis | ||||
| FER | F. temperatum | ||||
| GER | F. boothii | ||||
| Farm Number | Mycotoxins Detected (Average µg/kg) | |||
|---|---|---|---|---|
| DON a | 15-ADON b | DIP c | ZEN d | |
| LOQ e: 100 µg/kg | LOQ: 100 µg/kg | LOQ: 50 µg/kg | LOQ: 20 µg/kg | |
| LOD f: 50 µg/kg | LOD: 50 µg/kg | LOD: 25 µg/kg | LOD: 10 µg/kg | |
| Farm 1 | ND | ND | 726 | ND |
| Farm 2 | 879 | 174 | 122 | ND |
| Farm 3 | ND | ND | ND | ND |
| Farm 4 | 3143 | 301 | ND | ND |
| Farm 5 | 5678 | 257 | 33,808 | 23 |
| Farm 6 | 254 | ND | 2648 | ND |
| Farm 7 | 18,306 | 1263 | ND | 639 |
| Farm 8 | <LOQ | ND | ND | ND |
| Farm 9 | ND | ND | <LOQ | ND |
| Farm 10 | ND | ND | 1268 | ND |
| Farm 11 | ND | ND | ND | ND |
| Farm 12 | 7273 | 736 | 23,004 | 67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Price, J.-L.; Visagie, C.M.; Meyer, H.; Yilmaz, N. Fungal Species and Mycotoxins Associated with Maize Ear Rots Collected from the Eastern Cape in South Africa. Toxins 2024, 16, 95. https://doi.org/10.3390/toxins16020095
Price J-L, Visagie CM, Meyer H, Yilmaz N. Fungal Species and Mycotoxins Associated with Maize Ear Rots Collected from the Eastern Cape in South Africa. Toxins. 2024; 16(2):95. https://doi.org/10.3390/toxins16020095
Chicago/Turabian StylePrice, Jenna-Lee, Cobus Meyer Visagie, Hannalien Meyer, and Neriman Yilmaz. 2024. "Fungal Species and Mycotoxins Associated with Maize Ear Rots Collected from the Eastern Cape in South Africa" Toxins 16, no. 2: 95. https://doi.org/10.3390/toxins16020095
APA StylePrice, J.-L., Visagie, C. M., Meyer, H., & Yilmaz, N. (2024). Fungal Species and Mycotoxins Associated with Maize Ear Rots Collected from the Eastern Cape in South Africa. Toxins, 16(2), 95. https://doi.org/10.3390/toxins16020095





