Bacterial Toxin-Antitoxin Systems’ Cross-Interactions—Implications for Practical Use in Medicine and Biotechnology
Abstract
1. Introduction
| TA System Type | Antitoxin/Toxin Chemical Nature | Antitoxin Mode of Action |
|---|---|---|
| I | sRNA/Protein | Silencing toxin expression [16] |
| II | Protein/Protein | Formation of neutralizing complex due to direct binding to toxin [17] |
| III | Small pseudoknot RNA/Protein | Formation of neutralizing complex due to direct binding to toxin [18] |
| IV | Protein/Protein | Direct interaction with the toxin’s target and its protection [19] |
| V | Protein/Protein | Degradation of toxin mRNA [20] |
| VI | Protein/Protein | Direct binding to toxin and targeting it to proteolysis by ATP-dependent proteases [21] |
| VII | Protein/Protein | Toxin inactivation due to its post-translational modification [22] |
| VIII | sRNA/sRNA | Silencing toxin expression [23] |
2. An Overview of Activation and Biological Function of the Bacterial Toxin-Antitoxin Systems
2.1. Toxin-Antitoxin System Classification and Distribution Issues
2.2. Mechanisms of TA Systems’ Activation
2.3. Contradictions Regarding Biological Function of TA Systems
3. Modes of the Toxin-Antitoxin System Cross-Talk
3.1. Specificity Determinants Allow Desired Interactions between the Toxin and Antitoxin
3.2. Different Levels of Cross-Interactions between TA Partners
3.2.1. Examples of Cross-Talk between Proteins of Chromosomal TA Cassettes
3.2.2. Cross-Interactions between TA Proteins Located on Different Replicons
3.2.3. Antitoxins as Regulators of Unrelated Gene Expression
3.2.4. Examples of Cross-Induction between Different TA Systems
4. Toxin-Antitoxin Systems—Practical Application in Medicine
4.1. The TA-Based Antibacterial Strategies
4.2. The TA-Based Antiviral Strategies
4.3. The TA-Based Anticancer Strategies
5. Practical Applications of Toxin-Antitoxin Systems in Biotechnology
5.1. Toxin-Antitoxin Modules as the Vector Stabilization Loci
5.2. Counterselection Systems Based on Toxins of TA Modules
5.3. Toxin of TA Cassette in a Single Protein Production Technology
5.4. Positive Selection of Inhibitors Based on Toxin-Antitoxin System
5.5. Toxin-Antitoxin Systems for Biological or Gene Containment Control
5.6. Toxin-Antitoxin Used to Control Bacterial Infections of Plants
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, G.; Yadav, M.; Ghosh, C.; Rathore, J.S. Bacterial toxin-antitoxin modules: Classification, functions, and association with persistence. Curr. Res. Microb. Sci. 2021, 2, 100047. [Google Scholar] [CrossRef]
- Jurėnas, D.; Fraikin, N.; Goormaghtigh, F.; Van Melderen, L. Biology and evolution of bacterial toxin–antitoxin systems. Nat. Rev. Microbiol. 2022, 20, 335–350. [Google Scholar] [CrossRef]
- Akarsu, H.; Bordes, P.; Mansour, M.; Bigot, D.-J.J.; Genevaux, P.; Falquet, L. TASmania: A bacterial toxin-antitoxin systems database. PLoS Comput. Biol. 2019, 15, e1006946. [Google Scholar] [CrossRef]
- Fiedoruk, K.; Daniluk, T.; Swiecicka, I.; Sciepuk, M.; Leszczynska, K. Type II toxin–antitoxin systems are unevenly distributed among Escherichia coli phylogroups. Microbiology 2015, 161, 158–167. [Google Scholar] [CrossRef]
- Dziewit, L.; Jazurek, M.; Drewniak, L.; Baj, J.; Bartosik, D. The SXT conjugative element and linear prophage N15 encode toxin-antitoxin-stabilizing systems homologous to the tad-ata module of the Paracoccus aminophilus plasmid pAMI2. J. Bacteriol. 2007, 189, 1983–1997. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, W.; Yao, J.; Wang, X.; Liu, D.; Wang, P. The HipAB Toxin–Antitoxin System Stabilizes a Composite Genomic Island in Shewanella putrefaciens CN-32. Front. Microbiol. 2022, 13, 858857. [Google Scholar] [CrossRef]
- Fraikin, N.; Goormaghtigh, F.; van Melderen, L. Type II Toxin-Antitoxin Systems: Evolution and Revolutions. J. Bacteriol. 2020, 202, e00763-19. [Google Scholar] [CrossRef]
- Tu, C.H.; Holt, M.; Ruan, S.; Bourne, C. Evaluating the Potential for Cross-Interactions of Antitoxins in Type II TA Systems. Toxins 2020, 12, 422. [Google Scholar] [CrossRef]
- De Bast, M.S.; Mine, N.; Van Melderen, L. Chromosomal toxin-antitoxin systems may act as antiaddiction modules. J. Bacteriol. 2008, 190, 4603–4609. [Google Scholar] [CrossRef]
- Mine, N.; Guglielmini, J.; Wilbaux, M.; Van Melderen, L. The decay of the chromosomally encoded ccdO157 toxin-antitoxin system in the Escherichia coli species. Genetics 2009, 181, 1557–1566. [Google Scholar] [CrossRef]
- Brown, B.L.; Grigoriu, S.; Kim, Y.; Arruda, J.M.; Davenport, A.; Wood, T.K.; Peti, W.; Page, R. Three Dimensional Structure of the MqsR:MqsA Complex: A Novel TA Pair Comprised of a Toxin Homologous to RelE and an Antitoxin with Unique Properties. PLoS Pathog. 2009, 5, e1000706. [Google Scholar] [CrossRef]
- Kim, Y.; Wang, X.; Zhang, X.-S.; Grigoriu, S.; Page, R.; Peti, W.; Wood, T.K. Escherichia coli toxin/antitoxin pair MqsR/MqsA regulate toxin CspD. Environ. Microbiol. 2010, 12, 1105–1121. [Google Scholar] [CrossRef]
- Fraikin, N.; Rousseau, C.J.; Goeders, N.; Van Melderen, L. Reassessing the role of the type II MqsRA toxin-antitoxin system in stress response and biofilm formation: MqsA is transcriptionally uncoupled from mqsR. MBio 2019, 10, e02678-19. [Google Scholar] [CrossRef] [PubMed]
- García-Contreras, R.; Zhang, X.-S.; Kim, Y.; Wood, T.K. Protein Translation and Cell Death: The Role of Rare tRNAs in Biofilm Formation and in Activating Dormant Phage Killer Genes. PLoS ONE 2008, 3, 2394. [Google Scholar] [CrossRef] [PubMed]
- Bukowski, M.; Lyzen, R.; Helbin, W.M.; Bonar, E.; Szalewska-Palasz, A.; Wegrzyn, G.; Dubin, G.; Dubin, A.; Wladyka, B. A regulatory role for Staphylococcus aureus toxin–antitoxin system PemIKSa. Nat. Commun. 2013, 4, 2012. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, K.; Helin, K.; Christensen, O.W.; Løbner-Olesen, A. Translational control and differential RNA decay are key elements regulating postsegregational expression of the killer protein encoded by the parB locus of plasmid R1. J. Mol. Biol. 1988, 203, 119–129. [Google Scholar] [CrossRef]
- Tam, J.E.; Kline, B.C. The F plasmid ccd autorepressor is a complex of CcdA and CcdB proteins. MGG Mol. Gen. Genet. 1989, 219, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Fineran, P.C.; Blower, T.R.; Foulds, I.J.; Humphreys, D.P.; Lilley, K.S.; Salmond, G.P.C. The phage abortive infection system, ToxIN, functions as a protein-RNA toxin-antitoxin pair. Proc. Natl. Acad. Sci. USA 2009, 106, 894–899. [Google Scholar] [CrossRef]
- Masuda, H.; Tan, Q.; Awano, N.; Wu, K.-P.; Inouye, M. YeeU enhances the bundling of cytoskeletal polymers of MreB and FtsZ, antagonizing the CbtA (YeeV) toxicity in Escherichia coli. Mol. Microbiol. 2012, 84, 979–989. [Google Scholar] [CrossRef]
- Wang, X.; Lord, D.M.; Cheng, H.Y.; Osbourne, D.O.; Hong, S.H.; Sanchez-Torres, V.; Quiroga, C.; Zheng, K.; Herrmann, T.; Peti, W.; et al. A new type V toxin-antitoxin system where mRNA for toxin GhoT is cleaved by antitoxin GhoS. Nat. Chem. Biol. 2012, 8, 855–861. [Google Scholar] [CrossRef]
- Aakre, C.D.; Phung, T.N.; Huang, D.; Laub, M.T. A Bacterial Toxin Inhibits DNA Replication Elongation Through a Direct Interaction with the β Sliding Clamp. Mol. Cell 2013, 52, 617. [Google Scholar] [CrossRef] [PubMed]
- Marimon, O.; Teixeira, J.M.C.; Cordeiro, T.N.; Soo, V.W.C.; Wood, T.L.; Mayzel, M.; Amata, I.; García, J.; Morera, A.; Gay, M.; et al. An oxygen-sensitive toxin-antitoxin system. Nat. Commun. 2016, 7, 13634. [Google Scholar] [CrossRef] [PubMed]
- Soo Choi, J.; Kim, W.; Suk, S.; Park, H.; Bak, G.; Yoon, J.; Lee, Y. RNA Biology The small RNA, SdsR, acts as a novel type of toxin in Escherichia coli The small RNA, SdsR, acts as a novel type of toxin in Escherichia coli. RNA Biol. 2018, 15, 1319–1335. [Google Scholar] [CrossRef] [PubMed]
- Lawarée, E.; Jankevicius, G.; Cooper, C.; Ahel, I.; Uphoff, S.; Tang, C.M. DNA ADP-Ribosylation Stalls Replication and Is Reversed by RecF-Mediated Homologous Recombination and Nucleotide Excision Repair. Cell Rep. 2020, 30, 1373–1384.e4. [Google Scholar] [CrossRef]
- Jankevicius, G.; Ariza, A.; Ahel, M.; Ahel, I. The Toxin-Antitoxin System DarTG Catalyzes Reversible ADP-Ribosylation of DNA. Mol. Cell 2016, 64, 1109–1116. [Google Scholar] [CrossRef]
- Blower, T.R.; Salmond, G.P.C.; Luisi, B.F. Balancing at survival’s edge: The structure and adaptive benefits of prokaryotic toxin-antitoxin partners. Curr. Opin. Struct. Biol. 2011, 21, 109–118. [Google Scholar] [CrossRef]
- Kawano, M.; Aravind, L.; Storz, G. An antisense RNA controls synthesis of an SOS-induced toxin evolved from an antitoxin. Mol. Microbiol. 2007, 64, 738–754. [Google Scholar] [CrossRef]
- Soo, V.W.C.; Cheng, H.Y.; Kwan, B.W.; Wood, T.K. De novo Synthesis of a Bacterial Toxin/Antitoxin System. Sci. Rep. 2014, 4, 4807. [Google Scholar] [CrossRef]
- Leplae, R.; Geeraerts, D.; Hallez, R.; Guglielmini, J.; Drze, P.; Van Melderen, L. Diversity of bacterial type II toxin-antitoxin systems: A comprehensive search and functional analysis of novel families. Nucleic Acids Res. 2011, 39, 5513–5525. [Google Scholar] [CrossRef]
- Guglielmini, J.; Van Melderen, L. Bacterial toxin-antitoxin systems: Translation inhibitors everywhere. Mob. Genet. Elem. 2011, 1, 283–306. [Google Scholar] [CrossRef]
- Triplett, L.R.; Shidore, T.; Long, J.; Miao, J.; Wu, S.; Han, Q.; Zhou, C.; Ishihara, H.; Li, J.; Zhao, B.; et al. AvrRxo1 Is a Bifunctional Type III Secreted Effector and Toxin-Antitoxin System Component with Homologs in Diverse Environmental Contexts. PLoS ONE 2016, 11, e0158856. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, T.; de Beer, T.A.P.; Tamegger, S.; Harms, A.; Dietz, N.; Dranow, D.M.; Edwards, T.E.; Myler, P.J.; Phan, I.; Dehio, C. Evolutionary Diversification of Host-Targeted Bartonella Effectors Proteins Derived from a Conserved FicTA Toxin-Antitoxin Module. Microorganisms 2021, 9, 1645. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Wei, Y.; Shen, Y.; Li, X.; Zhou, H.; Tai, C.; Deng, Z.; Ou, H.-Y.Y. TADB 2.0: An updated database of bacterial type II toxin-antitoxin loci. Nucleic Acids Res. 2018, 46, D749–D753. [Google Scholar] [CrossRef]
- Makarova, K.S.; Wolf, Y.I.; Koonin, E.V. Comprehensive comparative-genomic analysis of Type 2 toxin-antitoxin systems and related mobile stress response systems in prokaryotes. Biol. Direct 2009, 4, 19. [Google Scholar] [CrossRef]
- Ramisetty, B.C.M.; Santhosh, R.S. Horizontal gene transfer of chromosomal type ii toxin-antitoxin systems of Escherichia coli. FEMS Microbiol. Lett. 2015, 363, fnv238. [Google Scholar] [CrossRef] [PubMed]
- Christensen, S.K.; Mikkelsen, M.; Pedersen, K.; Gerdes, K. RelE, a global inhibitor of translation, is activated during nutritional stress. Proc. Natl. Acad. Sci. USA 2001, 98, 14328–14333. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, K.; Christensen, S.K.; Løbner-Olesen, A. Prokaryotic toxin–antitoxin stress response loci. Nat. Rev. Microbiol. 2005, 3, 371–382. [Google Scholar] [CrossRef]
- Gupta, A.; Venkataraman, B.; Vasudevan, M.; Gopinath Bankar, K. Co-expression network analysis of toxin-antitoxin loci in Mycobacterium tuberculosis reveals key modulators of cellular stress. Sci. Rep. 2017, 7, 5868. [Google Scholar] [CrossRef]
- Ariyachaokun, K.; Grabowska, A.D.; Gutierrez, C.; Neyrolles, O. Multi-Stress Induction of the Mycobacterium tuberculosis MbcTA Bactericidal Toxin-Antitoxin System. Toxins 2020, 12, 329. [Google Scholar] [CrossRef]
- Bordes, P.; Genevaux, P. Control of Toxin-Antitoxin Systems by Proteases in Mycobacterium tuberculosis. Front. Mol. Biosci. 2021, 8, 691399. [Google Scholar] [CrossRef]
- LeRoux, M.; Culviner, P.H.; Liu, Y.J.; Littlehale, M.L.; Laub, M.T. Stress Can Induce Transcription of Toxin-Antitoxin Systems without Activating Toxin. Mol. Cell 2020, 79, 280–292.e8. [Google Scholar] [CrossRef] [PubMed]
- Short, F.L.; Akusobi, C.; Broadhurst, W.R.; Salmond, G.P.C. The bacterial Type III toxin-antitoxin system, ToxIN, is a dynamic protein-RNA complex with stability-dependent antiviral abortive infection activity. Sci. Rep. 2018, 8, 1013. [Google Scholar] [CrossRef] [PubMed]
- Guegler, C.K.; Laub, M.T. Shutoff of host transcription triggers a toxin-antitoxin system to cleave phage RNA and abort infection. Mol. Cell 2021, 81, 2361–2373.e9. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Tang, K.; Sit, B.; Gu, J.; Chen, R.; Lin, J.; Lin, S.; Liu, X.; Wang, W.; Gao, X.; et al. Dual control of lysogeny and phage defense by a phosphorylation-based toxin/antitoxin system. bioRxiv 2022. bioRxiv:2022.09.05.506569. [Google Scholar] [CrossRef]
- Takada, K.; Hama, K.; Sasaki, T.; Otsuka, Y. The hokW-sokW Locus Encodes a Type I Toxin-Antitoxin System That Facilitates the Release of Lysogenic Sp5 Phage in Enterohemorrhagic Escherichia coli O157. Toxins 2021, 13, 796. [Google Scholar] [CrossRef]
- Dubiel, A.; Wegrzyn, K.; Kupinski, A.P.; Konieczny, I. ClpAP protease is a universal factor that activates the parDE toxin-antitoxin system from a broad host range RK2 plasmid. Sci. Rep. 2018, 8, 15287. [Google Scholar] [CrossRef]
- Vos, M.R.; Piraino, B.; LaBreck, C.J.; Rahmani, N.; Trebino, C.E.; Schoenle, M.; Peti, W.; Camberg, J.L.; Page, R. Degradation of the E. coli antitoxin MqsA by the proteolytic complex ClpXP is regulated by zinc occupancy and oxidation. J. Biol. Chem. 2022, 298, 101557. [Google Scholar] [CrossRef]
- Zhang, T.; Tamman, H.; Coppieters, K.; Kurata, T.; LeRoux, M.; Srikant, S.; Brodiazhenko, T.; Cepauskas, A.; Talavera, A.; Martens, C.; et al. Direct activation of a bacterial innate immune system by a viral capsid protein. Nature 2022, 612, 132–140. [Google Scholar] [CrossRef]
- Smith, L.M.; Fineran, P.C. Phage capsid recognition triggers activation of a bacterial toxin-antitoxin defense system. Mol. Cell 2023, 83, 165–166. [Google Scholar] [CrossRef]
- Bobonis, J.; Mitosch, K.; Mateus, A.; Karcher, N.; Kritikos, G.; Selkrig, J.; Zietek, M.; Monzon, V.; Pfalz, B.; Garcia-Santamarina, S.; et al. Bacterial retrons encode phage-defending tripartite toxin–antitoxin systems. Nature 2022, 609, 144–150. [Google Scholar] [CrossRef]
- Cui, Y.; Su, X.; Wang, C.; Xu, H.; Hu, D.; Wang, J.; Pei, K.; Sun, M.; Zou, T. Bacterial MazF/MazE toxin-antitoxin suppresses lytic propagation of arbitrium-containing phages. Cell Rep. 2022, 41, 111752. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.W. CRISPR-Cas Toxin–Antitoxin Systems: Selfishness as a Constructive Evolutionary Force. Trends Microbiol. 2021, 29, 869–870. [Google Scholar] [CrossRef] [PubMed]
- Ramisetty, B.C.M.; Santhosh, R.S. Endoribonuclease type II toxin-antitoxin systems: Functional or selfish? Microbiology 2017, 163, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Rosendahl, S.; Tamman, H.; Brauer, A.; Remm, M.; Hõrak, R. Chromosomal toxin-antitoxin systems in Pseudomonas putida are rather selfish than beneficial. Sci. Rep. 2020, 10, 9230. [Google Scholar] [CrossRef]
- Song, S.; Wood, T.K. A Primary Physiological Role of Toxin/Antitoxin Systems Is Phage Inhibition. Front. Microbiol. 2020, 11, 1895. [Google Scholar] [CrossRef]
- Song, S.; Wood, T.K. Toxin/Antitoxin System Paradigms: Toxins Bound to Antitoxins Are Not Likely Activated by Preferential Antitoxin Degradation. Adv. Biosyst. 2020, 4, 1900290. [Google Scholar] [CrossRef]
- Song, S.; Wood, T.K. Post-segregational Killing and Phage Inhibition Are Not Mediated by Cell Death Through Toxin/Antitoxin Systems. Front. Microbiol. 2018, 9, 814. [Google Scholar] [CrossRef]
- Kim, J.-S.; Wood, T.K. Persistent Persister Misperceptions. Front. Microbiol. 2016, 7, 2134. [Google Scholar] [CrossRef]
- Van Melderen, L.; Wood, T.K. Commentary: What Is the Link between Stringent Response, Endoribonuclease Encoding Type II Toxin-Antitoxin Systems and Persistence? Front. Microbiol. 2017, 8, 191. [Google Scholar] [CrossRef]
- Kamruzzaman, M.; Wu, A.Y.; Iredell, J.R. Biological functions of type ii toxin-antitoxin systems in bacteria. Microorganisms 2021, 9, 1276. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Inouye, M. Regulation of growth and death in Escherichia coli by toxin-antitoxin systems. Nat. Rev. Microbiol. 2011, 9, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Helaine, S.; Cheverton, A.M.; Watson, K.G.; Faure, L.M.; Matthews, S.A.; Holden, D.W. Internalization of salmonella by macrophages induces formation of nonreplicating persisters. Science 2014, 343, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Harms, A.; Maisonneuve, E.; Gerdes, K. Mechanisms of bacterial persistence during stress and antibiotic exposure. Science 2016, 354, aaf4268. [Google Scholar] [CrossRef] [PubMed]
- Harms, A.; Fino, C.; Sørensen, M.A.; Semsey, S.; Gerdes, K. Prophages and growth dynamics confound experimental results with antibiotic-tolerant persister cells. MBio 2017, 8, e01964-17. [Google Scholar] [CrossRef] [PubMed]
- Pontes, M.H.; Groisman, E.A. Slow growth dictates non-heritable antibiotic resistance in Salmonella enterica. Sci. Signal. 2019, 12, eaax3938. [Google Scholar] [CrossRef]
- Claudi, B.; Spröte, P.; Chirkova, A.; Personnic, N.; Zankl, J.; Schürmann, N.; Schmidt, A.; Bumann, D. Phenotypic Variation of Salmonella in Host Tissues Delays Eradication by Antimicrobial Chemotherapy. Cell 2014, 158, 722–733. [Google Scholar] [CrossRef]
- Tsilibaris, V.; Maenhaut-Michel, G.; Mine, N.; Van Melderen, L. What is the benefit to Escherichia coli of having multiple toxin-antitoxin systems in its genome? J. Bacteriol. 2007, 189, 6101–6108. [Google Scholar] [CrossRef]
- Norton, J.P.; Mulvey, M.A. Toxin-Antitoxin Systems Are Important for Niche-Specific Colonization and Stress Resistance of Uropathogenic Escherichia coli. PLoS Pathog. 2012, 8, e1002954. [Google Scholar] [CrossRef]
- Agarwal, S.; Sharma, A.; Bouzeyen, R.; Deep, A.; Sharma, H.; Mangalaparthi, K.K.; Datta, K.K.; Kidwai, S.; Gowda, H.; Gowda, H.; et al. VapBC22 toxin-antitoxin system from Mycobacterium tuberculosis is required for pathogenesis and modulation of host immune response. Sci. Adv. 2020, 6, aba6944. [Google Scholar] [CrossRef]
- Du, C.; Zhang, W.; Gu, H.; Dong, X.; Hu, Y. Type II toxin–antitoxin system, RatAB, contributes to oxidative resistance, biofilm formation and virulence of Edwardsiella piscicida. Aquac. Res. 2022, 53, 2575–2585. [Google Scholar] [CrossRef]
- Nordstrom, K.; Austin, S.J. Mechanisms That Contribute to the Stable Segregation of Plasmids. Annu. Rev. Genet. 2003, 23, 37–69. [Google Scholar] [CrossRef] [PubMed]
- Ogura, T.; Hiraga, S. Mini-F plasmid genes that couple host cell division to plasmid proliferation. Proc. Natl. Acad. Sci. USA 1983, 80, 4784–4788. [Google Scholar] [CrossRef] [PubMed]
- Hiraga, S.; Jaffe, A.; Ogura, T.; Mori, H.; Takahashi, H. F plasmid ccd mechanism in Escherichia coli. J. Bacteriol. 1986, 166, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Kawano, M.; Oshima, T.; Kasai, H.; Mori, H. Molecular characterization of long direct repeat (LDR) sequences expressing a stable mRNA encoding for a 35-amino-acid cell-killing peptide and a cis-encoded small antisense RNA in Escherichia coli. Mol. Microbiol. 2002, 45, 333–349. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, P.; Tello, M.; Orellana, O. Toxin-Antitoxin Systems in the Mobile Genome of Acidithiobacillus ferrooxidans. PLoS ONE 2014, 9, e112226. [Google Scholar] [CrossRef]
- Yao, J.; Guo, Y.; Wang, P.; Zeng, Z.; Li, B.; Tang, K.; Liu, X.; Wang, X. Type II toxin/antitoxin system ParESO/CopASO stabilizes prophage CP4So in Shewanella oneidensis. Environ. Microbiol. 2018, 20, 1224–1239. [Google Scholar] [CrossRef]
- Yao, X.; Chen, T.; Shen, X.; Zhao, Y.; Wang, M.; Rao, X.; Yin, S.; Wang, J.; Gong, Y.; Lu, S.; et al. The chromosomal SezAT toxin-antitoxin system promotes the maintenance of the SsPI-1 pathogenicity island in epidemic Streptococcus suis. Mol. Microbiol. 2015, 98, 243–257. [Google Scholar] [CrossRef]
- Peltier, J.; Hamiot, A.; Garneau, J.R.; Boudry, P.; Maikova, A.; Hajnsdorf, E.; Fortier, L.C.; Dupuy, B.; Soutourina, O. Type I toxin-antitoxin systems contribute to the maintenance of mobile genetic elements in Clostridioides difficile. Commun. Biol. 2020, 3, 718. [Google Scholar] [CrossRef]
- Cooper, T.F.; Heinemann, J.A. Postsegregational killing does not increase plasmid stability but acts to mediate the exclusion of competing plasmids. Proc. Natl. Acad. Sci. USA 2000, 97, 12643–12648. [Google Scholar] [CrossRef]
- Jurėnas, D.; Garcia-Pino, A.; Van Melderen, L. Novel toxins from type II toxin-antitoxin systems with acetyltransferase activity. Plasmid 2017, 93, 30–35. [Google Scholar] [CrossRef]
- Martins, P.M.M.; Machado, M.A.; Silva, N.V.; Takita, M.A.; De Souza, A.A. Type II toxin-antitoxin distribution and adaptive aspects on Xanthomonas genomes: Focus on Xanthomonas citri. Front. Microbiol. 2016, 7, 652. [Google Scholar] [CrossRef] [PubMed]
- Aakre, C.D.; Herrou, J.; Phung, T.N.; Perchuk, B.S.; Crosson, S.; Laub, M.T. Evolving new protein-protein interaction specificity through promiscuous intermediates. Cell 2015, 163, 594. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Green, A.G.; Wang, B.; Lite, T.L.V.; Weinstein, E.N.; Marks, D.S.; Laub, M.T. Co-evolution of interacting proteins through non-contacting and non-specific mutations. Nat. Ecol. Evol. 2022, 6, 590–603. [Google Scholar] [CrossRef] [PubMed]
- Leroux, M.; Laub, M.T. Toxin-Antitoxin Systems as Phage Defense Elements. Annu. Rev. Microbiol. 2022, 76, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Pecota, D.C.; Wood, T.K. Exclusion of T4 phage by the hok/sok killer locus from plasmid R1. J. Bacteriol. 1996, 178, 2044–2050. [Google Scholar] [CrossRef] [PubMed]
- Blower, T.R.; Chai, R.; Przybilski, R.; Chindhy, S.; Fang, X.; Kidman, S.E.; Tan, H.; Luisi, B.F.; Fineran, P.C.; Salmond, G.P.C. Evolution of Pectobacterium bacteriophage ΦM1 to escape two bifunctional type III toxin-antitoxin and abortive infection systems through mutations in a single viral gene. Appl. Environ. Microbiol. 2017, 83, e03229-16. [Google Scholar] [CrossRef]
- Koga, M.; Otsuka, Y.; Lemire, S.; Yonesaki, T. Escherichia coli rnlA and rnlB Compose a Novel Toxin-Antitoxin System. Genetics 2011, 187, 123–130. [Google Scholar] [CrossRef]
- Otsuka, Y.; Yonesaki, T. Dmd of bacteriophage T4 functions as an antitoxin against Escherichia coli LsoA and RnlA toxinsm. Mol. Microbiol. 2012, 83, 669–681. [Google Scholar] [CrossRef]
- Ni, M.; Lin, J.; Gu, J.; Lin, S.; He, M.; Guo, Y. Antitoxin CrlA of CrlTA Toxin–Antitoxin System in a Clinical Isolate Pseudomonas aeruginosa Inhibits Lytic Phage Infection. Front. Microbiol. 2022, 1, 892021. [Google Scholar] [CrossRef]
- Blower, T.R.; Evans, T.J.; Przybilski, R.; Fineran, P.C.; Salmond, G.P.C. Viral Evasion of a Bacterial Suicide System by RNA-Based Molecular Mimicry Enables Infectious Altruism. PLoS Genet. 2012, 8, 1003023. [Google Scholar] [CrossRef]
- Leroux, M.; Srikant, S.; Littlehale, M.H.; Teodoro, G.; Doron, S.; Badiee, M.; Leung, A.K.L.; Sorek, R.; Laub, M.T. The DarTG toxin-antitoxin system provides phage defense by ADP-ribosylating viral DNA. bioRxiv 2021. [Google Scholar] [CrossRef]
- Hilliard, J.J.; Maurizi, M.R.; Simon, L.D. Isolation and characterization of the phage T4 PinA protein, an inhibitor of the ATP-dependent Lon protease of Escherichia coli. J. Biol. Chem. 1998, 273, 518–523. [Google Scholar] [CrossRef]
- Sberro, H.; Leavitt, A.; Kiro, R.; Koh, E.; Peleg, Y.; Qimron, U.; Sorek, R. Discovery of Functional Toxin/Antitoxin Systems in Bacteria by Shotgun Cloning. Mol. Cell 2013, 50, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Van Melderen, L.; De Bast, M.S. Bacterial toxin-Antitoxin systems: More than selfish entities? PLoS Genet. 2009, 5, e1000437. [Google Scholar] [CrossRef] [PubMed]
- Lite, T.L.V.; Grant, R.A.; Nocedal, I.; Littlehale, M.L.; Guo, M.S.; Laub, M.T. Uncovering the basis of protein-protein interaction specificity with a combinatorially complete library. eLife 2020, 9, e60924. [Google Scholar] [CrossRef] [PubMed]
- Fiebig, A.; Castro Rojas, C.M.; Siegal-Gaskins, D.; Crosson, S. Interaction specificity, toxicity and regulation of a paralogous set of ParE/RelE-family toxin-antitoxin systems. Mol. Microbiol. 2010, 77, 236–251. [Google Scholar] [CrossRef]
- Połom, D.; Boss, L.; Wȩgrzyn, G.; Hayes, F.; Kȩdzierska, B. Amino acid residues crucial for specificity of toxin-antitoxin interactions in the homologous Axe-Txe and YefM-YoeB complexes. FEBS J. 2013, 280, 5906–5918. [Google Scholar] [CrossRef]
- Kurata, T.; Saha, C.K.; Buttress, J.A.; Mets, T.; Brodiazhenko, T.; Turnbull, K.J.; Awoyomi, O.F.; Oliveira, S.R.A.; Jimmy, S.; Ernits, K.; et al. A hyperpromiscuous antitoxin protein domain for the neutralization of diverse toxin domains. Proc. Natl. Acad. Sci. USA 2022, 119, e2102212119. [Google Scholar] [CrossRef]
- Grabe, G.J.; Giorgio, R.T.; Hall, A.M.J.; Morgan, R.M.L.; Dubois, L.; Sisley, T.A.; Rycroft, J.A.; Hare, S.A.; Helaine, S. Auxiliary interfaces support the evolution of specific toxin–antitoxin pairing. Nat. Chem. Biol. 2021, 17, 1296–1304. [Google Scholar] [CrossRef]
- Zhu, L.; Sharp, J.D.; Kobayashi, H.; Woychik, N.A.; Inouye, M. Noncognate Mycobacterium tuberculosis toxin-antitoxins can physically and functionally interact. J. Biol. Chem. 2010, 285, 39732–39738. [Google Scholar] [CrossRef]
- Yang, M.; Gao, C.; Wang, Y.; Zhang, H.; He, Z.G. Characterization of the interaction and cross-regulation of three Mycobacterium tuberculosis RelBE modules. PLoS ONE 2010, 5, e10672. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Tu, J.; Tan, Y.; Cai, X.; Yang, C.; Deng, X.; Su, B.; Ma, S.; Liu, X.; Ma, P.; et al. Structural and Biochemical Characterization of the Cognate and Heterologous Interactions of the MazEF-mt9 TA System. ACS Infect. Dis. 2019, 5, 1306–1316. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Li, Y.; Yang, F.; Wu, Q.; Liu, D.; Li, X.; Hua, H.; Liu, X.; Wang, Y.; Zheng, K.; et al. Physical and Functional Interplay between MazF1Bif and Its Noncognate Antitoxins from Bifidobacterium longum. Appl. Environ. Microbiol. 2017, 83, e03232-16. [Google Scholar] [CrossRef]
- Goulard, C.; Langrand, S.; Carniel, E.; Chauvaux, S. The Yersinia pestis chromosome encodes active addiction toxins. J. Bacteriol. 2010, 192, 3669–3677. [Google Scholar] [CrossRef] [PubMed]
- Kamphuis, M.; Chiara Monti, M.H.; van den Heuvel, R.H.; Lopez-Villarejo, J.; Diaz-Orejas, R.; Boelens, R. Structure and Function of Bacterial Kid-Kis and Related Toxin-Antitoxin Systems. Protein Pept. Lett. 2007, 14, 113–124. [Google Scholar] [CrossRef]
- Smith, A.B.; López-Villarejo, J.; Diago-Navarro, E.; Mitchenall, L.A.; Barendregt, A.; Heck, A.J.; Lemonnier, M.; Maxwell, A.; Díaz-Orejas, R. A Common Origin for the Bacterial Toxin-Antitoxin Systems parD and ccd, Suggested by Analyses of Toxin/Target and Toxin/Antitoxin Interactions. PLoS ONE 2012, 7, e46499. [Google Scholar] [CrossRef] [PubMed]
- Guérout, A.M.; Iqbal, N.; Mine, N.; Ducos-Galand, M.; Van Melderen, L.; Mazel, D. Characterization of the phd-doc and ccd toxin-antitoxin cassettes from Vibrio superintegrons. J. Bacteriol. 2013, 195, 2270–2283. [Google Scholar] [CrossRef]
- Wei, Y.; Gao, Z.; Zhang, H.; Dong, Y. Structural characterizations of phage antitoxin Dmd and its interactions with bacterial toxin RnlA. Biochem. Biophys. Res. Commun. 2016, 472, 592–597. [Google Scholar] [CrossRef]
- Guo, Y.; Yao, J.; Sun, C.; Wen, Z.; Wang, X. Characterization of the deep-sea streptomyces sp. SCSIO 02999 derived VapC/VapB toxin-antitoxin system in Escherichia coli. Toxins 2016, 8, 195. [Google Scholar] [CrossRef]
- Agarwal, S.; Tiwari, P.; Deep, A.; Kidwai, S.; Gupta, S.; Thakur, K.G.; Singh, R. System-wide analysis unravels the differential regulation and in vivo essentiality of virulence-associated proteins B and C toxin-antitoxin systems of Mycobacterium tuberculosis. J. Infect. Dis. 2018, 217, 1809–1820. [Google Scholar] [CrossRef]
- Winther, K.S.; Gerdes, K. Ectopic production of VapCs from Enterobacteria inhibits translation and trans-activates YoeB mRNA interferase. Mol. Microbiol. 2009, 72, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Kasari, V.; Mets, T.; Tenson, T.; Kaldalu, N. Transcriptional cross-activation between toxin-antitoxin systems of Escherichia coli. BMC Microbiol. 2013, 13, 45. [Google Scholar] [CrossRef]
- Kędzierska, B.; Potrykus, K.; Szalewska-Pałasz, A.; Wodzikowska, B. Insights into transcriptional repression of the homologous toxin-antitoxin cassettes YeFm-YoeB and Axe-Txe. Int. J. Mol. Sci. 2020, 21, 62. [Google Scholar] [CrossRef]
- Guo, Y.; Sun, C.; Li, Y.; Tang, K.; Ni, S.; Wang, X. Antitoxin HigA inhibits virulence gene mvfR expression in Pseudomonas aeruginosa. Environ. Microbiol. 2019, 21, 2707–2723. [Google Scholar] [CrossRef] [PubMed]
- Ni, S.; Li, B.; Tang, K.; Yao, J.; Wood, T.K.; Wang, P.; Wang, X. Conjugative plasmid-encoded toxin–antitoxin system PrpT/PrpA directly controls plasmid copy number. Proc. Natl. Acad. Sci. USA 2021, 118, e2011577118. [Google Scholar] [CrossRef] [PubMed]
- Soo, V.W.C.; Wood, T.K. Antitoxin MqsA represses curli formation through the master biofilm regulator CsgD. Sci. Rep. 2013, 3, 3186. [Google Scholar] [CrossRef]
- Hu, Y.; Benedik, M.J.; Wood, T.K. Antitoxin DinJ influences the general stress response through transcript stabilizer CspE. Environ. Microbiol. 2012, 14, 669–679. [Google Scholar] [CrossRef]
- Lin, C.Y.; Awano, N.; Masuda, H.; Park, J.H.; Inouye, M. Transcriptional Repressor HipB Regulates the Multiple Promoters in Escherichia coli. J. Mol. Microbiol. Biotechnol. 2013, 23, 440–447. [Google Scholar] [CrossRef]
- Sala, A.; Bordes, P.; Genevaux, P. Multiple toxin-antitoxin systems in Mycobacterium tuberculosis. Toxins 2014, 6, 1002–1020. [Google Scholar] [CrossRef]
- Wilbaux, M.; Mine, N.; Guérout, A.-M.; Mazel, D.; Melderen, L. Van Functional Interactions between Coexisting Toxin-Antitoxin Systems of the ccd Family in Escherichia coli O157:H7. J. Bacteriol. 2007, 189, 2712–2719. [Google Scholar] [CrossRef]
- Goh, Y.-X.; Li, P.; Wang, M.; Djordjevic, M.; Tai, C.; Wang, H.; Deng, Z.; Chen, Z.; Ou, H.-Y. Comparative Analysis of Diverse Acetyltransferase-Type Toxin-Antitoxin Loci in Klebsiella pneumoniae. Microbiol. Spectr. 2022, 10, e00320-22. [Google Scholar] [CrossRef] [PubMed]
- Wessner, F.; Lacoux, C.; Goeders, N.; d’Héerouel, A.F.; Matos, R.; Serror, P.; van Melderen, L.; Repoila, F. Regulatory crosstalk between type I and type II toxin-antitoxin systems in the human pathogen Enterococcus faecalis. RNA Biol. 2015, 12, 1099–1108. [Google Scholar] [CrossRef] [PubMed]
- Kedzierska, B.; Hayes, F. Emerging roles of toxin-antitoxin modules in bacterial pathogenesis. Molecules 2016, 21, 790. [Google Scholar] [CrossRef]
- Wang, X.; Lord, D.M.; Hong, S.H.; Peti, W.; Benedik, M.J.; Page, R.; Wood, T.K. Type II toxin/antitoxin MqsR/MqsA controls type V toxin/antitoxin GhoT/GhoS. Environ. Microbiol. 2013, 15, 1734–1744. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Pati, S.; Kaushik, H.; Singh, S.; Garg, L.C. Toxin-antitoxin systems and their medical applications: Current status and future perspective. Appl. Microbiol. Biotechnol. 2021, 105, 1803–1821. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Guo, Y.; Yao, J.; Tang, K.; Wang, X. Applications of toxin-antitoxin systems in synthetic biology. Eng. Microbiol. 2023, 3, 100069. [Google Scholar] [CrossRef]
- Riffaud, C.; Pinel-Marie, M.L.; Felden, B. Cross-Regulations between Bacterial Toxin–Antitoxin Systems: Evidence of an Interconnected Regulatory Network? Trends Microbiol. 2020, 28, 851–866. [Google Scholar] [CrossRef]
- Kang, S.-M.; Kim, D.-H.; Lee, K.-Y.; Park, S.J.; Yoon, H.-J.; Lee, S.J.; Im, H.; Lee, B.-J. Functional details of the Mycobacterium tuberculosis VapBC26 toxin-antitoxin system based on a structural study: Insights into unique binding and antibiotic peptides. Nucleic Acids Res. 2017, 45, 8564–8580. [Google Scholar] [CrossRef]
- Kang, S.-M.; Moon, H.; Han, S.-W.; Wook Kim, B.; Kim, D.-H.; Moon Kim, B.; Lee, B.-J.; Toxin-Activating, B. Toxin-Activating Stapled Peptides Discovered by Structural Analysis Were Identified as New Therapeutic Candidates That Trigger Antibacterial Activity against Mycobacterium tuberculosis in the Mycobacterium smegmatis Model. Microorganisms 2021, 9, 568. [Google Scholar] [CrossRef]
- Kang, S.M.; Moon, H.; Han, S.W.; Kim, D.H.; Kim, B.M.; Lee, B.J. Structure-Based De Novo Design of Mycobacterium tuberculosis VapC-Activating Stapled Peptides. ACS Chem. Biol. 2020, 15, 2493–2498. [Google Scholar] [CrossRef]
- Chauhan, U.; Barth, V.C.; Woychik, N.A. tRNAfMet Inactivating Mycobacterium tuberculosis VapBC Toxin-Antitoxin Systems as Therapeutic Targets. Antimicrob. Agents Chemother. 2022, 66, e01896-21. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.M.; Jin, C.; Kim, D.H.; Lee, Y.; Lee, B.J. Structural and Functional Study of the Klebsiella pneumoniae VapBC Toxin-Antitoxin System, including the Development of an Inhibitor That Activates VapC. J. Med. Chem. 2020, 63, 13669–13679. [Google Scholar] [CrossRef] [PubMed]
- Cristina, B.; Sanches, P.; Rocha, C.A.; Gregorio, J.; Bedoya, M.; Luiz Da Silva, V.; Bento Da Silva, P.; Fusco-Almeida, A.M.; Chorilli, M.; Contiero, J.; et al. Rhamnolipid-Based Liposomes as Promising Nano-Carriers for Enhancing the Antibacterial Activity of Peptides Derived from Bacterial Toxin-Antitoxin Systems. Int. J. Nanomed. 2021, 10, 925–939. [Google Scholar] [CrossRef]
- You, Y.H.; Liu, H.Y.; Zhu, Y.Z.; Zheng, H. Rational design of stapled antimicrobial peptides. Amin. Acids 2023, 55, 421–442. [Google Scholar] [CrossRef]
- May, K.L.; Yan, Q.; Tumer, N.E. Targeting ricin to the ribosome. Toxicon 2013, 69, 143–151. [Google Scholar] [CrossRef]
- Grela, P.; Szajwaj, M.; Horbowicz-Dro, P.; Tchórzewski, M. How Ricin Damages the Ribosome. Toxins 2019, 11, 241. [Google Scholar] [CrossRef]
- Olombrada, M.; Peña, C.; Rodríguez-Galán, O.; Klingauf-Nerurkar, P.; Portugal-Calisto, D.; Oborská-Oplová, M.; Altvater, M.; Gavilanes, J.G.; Martínez-Del-Pozo, Á.; De La Cruz, J.; et al. The ribotoxin α-sarcin can cleave the sarcin/ricin loop on late 60S pre-ribosomes. Nucleic Acids Res. 2020, 48, 6210–6222. [Google Scholar] [CrossRef]
- Yeo, C.C.; Bakar, F.A.; Chan, W.T.; Espinosa, M.; Harikrishna, J.A. Heterologous expression of toxins from bacterial toxin-antitoxin systems in eukaryotic cells: Strategies and applications. Toxins 2016, 8, 49. [Google Scholar] [CrossRef]
- Audoly, G.; Vincentelli, R.; Edouard, S.; Georgiades, K.; Mediannikov, O.; Gimenez, G.; Socolovschi, C.; Mège, J.L.; Cambillau, C.; Raoult, D. Effect of Rickettsial Toxin VapC on Its Eukaryotic Host. PLoS ONE 2011, 6, 26528. [Google Scholar] [CrossRef]
- Ramage, H.R.; Connolly, L.E.; Cox, J.S. Comprehensive Functional Analysis of Mycobacterium tuberculosis Toxin-Antitoxin Systems: Implications for Pathogenesis, Stress Responses, and Evolution. PLoS Genet. 2009, 5, e1000767. [Google Scholar] [CrossRef]
- Równicki, M.; Pieńko, T.; Czarnecki, J.; Kolanowska, M.; Bartosik, D.; Trylska, J. Artificial Activation of Escherichia coli mazEF and hipBA Toxin-Antitoxin Systems by Antisense Peptide Nucleic Acids as an Antibacterial Strategy. Front. Microbiol. 2018, 9, 2870. [Google Scholar] [CrossRef] [PubMed]
- López-Igual, R.; Bernal-Bayard, J.; Rodríguez-Patón, A.; Ghigo, J.M.; Mazel, D. Engineered toxin-intein antimicrobials can selectively target and kill antibiotic-resistant bacteria in mixed populations. Nat. Biotechnol. 2019, 37, 755–760. [Google Scholar] [CrossRef] [PubMed]
- DiRita, V.J. Co-ordinate expression of virulence genes by ToxR in Vibrio cholerae. Mol. Microbiol. 1992, 6, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Nelapati, S.; Krishnaiah, N. Detection of total and pathogenic Vibrio parahaemolyticus by Polymerase chain reaction using toxR, tdh and trh genes. Vet. World 2010, 3, 268–271. [Google Scholar]
- Beaber, J.W.; Waldor, M.K. Identification of operators and promoters that control SXT conjugative transfer. J. Bacteriol. 2004, 186, 5945–5949. [Google Scholar] [CrossRef] [PubMed]
- Paul, P.; Verma, S.; Panda, K.; Jaiswal, S.; Sahu, B.R.; Suar, M.; Kumar Panda, P. Artificial Cells, Nanomedicine, and Biotechnology Molecular insight to influential role of Hha-TomB toxin-antitoxin system for antibacterial activity of biogenic silver nanoparticles Molecular insight to influential role of Hha-TomB toxin-antitoxin system. Artif. Cells Nanomed. Biotechnol. 2018, 46 (Suppl. S3), 572–584. [Google Scholar] [CrossRef]
- Jaiswal, S.; Paul, P.; Padhi, C.; Ray, S.; Ryan, D.; Dash, S.; Suar, M. The Hha-TomB Toxin-Antitoxin System Shows Conditional Toxicity and Promotes Persister Cell Formation by Inhibiting Apoptosis-Like Death in S. Typhimurium OPEN. Nat. Publ. Gr. 2016, 6, 38204. [Google Scholar] [CrossRef]
- Balsalobre, C.; Johansson, J.; Uhlin, B.E.; Juárez, A.; Muñoa, F.J. Alterations in protein expression caused by the hha mutation in Escherichia coli: Influence of growth medium osmolarity. J. Bacteriol. 1999, 181, 3018–3024. [Google Scholar] [CrossRef]
- Nieto, J.M.; Madrid, C.; Prenafeta, A.; Miquelay, E.; Balsalobre, C.; Carrascal, M.; Juárez, A. Expression of the hemolysin operon in Escherichia coli is modulated by a nucleoid-protein complex that includes the proteins Hha and H-NS. Mol. Gen. Genet. 2000, 263, 349–358. [Google Scholar] [CrossRef]
- Nieto, J.M.; Madrid, C.; Miquelay, E.; Parra, J.L.; Rodríguez, S.; Juárez, A. Evidence for direct protein-protein interaction between members of the enterobacterial Hha/YmoA and H-NS families of proteins. J. Bacteriol. 2002, 184, 629–635. [Google Scholar] [CrossRef]
- Shabazi, S.; Shivaee, A.; Nasiri, M.; Mirshekar, M.; Sabzi, S.; Saria, O.K. Zinc oxide nanoparticles impact the expression of the genes involved in toxin–antitoxin systems in multidrug-resistant Acinetobacter baumannii. J. Basic Microbiol. 2022. Early View. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhao, C.; Xue, B.; Li, C.; Zhang, X.; Yang, X.; Li, Y.; Yang, Y.; Shen, Z.; Wang, J.; et al. Nanoalumina triggers the antibiotic persistence of Escherichia coli through quorum sensing regulators lrsF and qseB. J. Hazard. Mater. 2022, 436, 129198. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, V. Hepatitis C virus RNA replication. Curr. Top. Microbiol. Immunol. 2013, 369, 167–198. [Google Scholar] [CrossRef] [PubMed]
- Shapira, A.; Shapira, S.; Gal-Tanamy, M.; Zemel, R.; Tur-Kaspa, R. Removal of Hepatitis C Virus-Infected Cells by a Zymogenized Bacterial Toxin. PLoS ONE 2012, 7, 32320. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, J.H.; Choi, W.; Lim, B.K. Specific elimination of coxsackievirus B3 infected cells with a protein engineered toxin-antitoxin system. Mol. Cell. Toxicol. 2019, 15, 425–430. [Google Scholar] [CrossRef]
- Chono, H.; Matsumoto, K.; Tsuda, H.; Saito, N.; Lee, K.; Kim, S.; Shibata, H.; Ageyama, N.; Terao, K.; Yasutomi, Y.; et al. Acquisition of HIV-1 Resistance in T Lymphocytes Using an ACA-Specific E. coli mRNA Interferase. Hum. Gene Ther. 2010, 22, 35–43. [Google Scholar] [CrossRef]
- Okamoto, M.; Chono, H.; Kawano, Y.; Saito, N.; Tsuda, H.; Inoue, K.; Kato, I.; Mineno, J.; Baba, M. Sustained Inhibition of HIV-1 Replication by Conditional Expression of the E. coli-Derived Endoribonuclease MazF in CD4+ T cells. Hum. Gene Ther. 2013, 24, 94–103. [Google Scholar] [CrossRef]
- Okamoto, M.; Chono, H.; Hidaka, A.; Toyama, M.; Mineno, J.; Baba, M. Induction of E. coli-derived endonuclease MazF suppresses HIV-1 production and causes apoptosis in latently infected cells. Biochem. Biophys. Res. Commun. 2020, 530, 597–602. [Google Scholar] [CrossRef]
- Martyn, J.; Gomez-Valero, L.; Buchrieser, C. The evolution and role of eukaryotic-like domains in environmental intracellular bacteria: The battle with a eukaryotic cell. FEMS Microbiol. Rev. 2022, 46, fuac012. [Google Scholar] [CrossRef]
- Al Marjani, M.F.; Authman, S.H.; Ali, F.S. Toxin-antitoxin systems and biofilm formation in bacteria. Rev. Res. Med. Microbiol. 2020, 31, 61–69. [Google Scholar] [CrossRef]
- Chieng Yeo, C. MicroCommentary GNAT toxins of bacterial toxin-antitoxin systems: Acetylation of charged tRNAs to inhibit translation A multitude of type II toxin-antitoxin (TA) systems in prokaryotic genomes. Mol. Microbiol. 2018, 108, 331–335. [Google Scholar] [CrossRef]
- Matelska, D.; Steczkiewicz, K.; Ginalski, K. SURVEY AND SUMMARY Comprehensive classification of the PIN domain-like superfamily. Nucleic Acids Res. 2017, 45, 6995–7020. [Google Scholar] [CrossRef] [PubMed]
- Senissar, M.; Manav, M.C.; Brodersen, D.E. Structural conservation of the PIN domain active site across all domains of life. Protein Sci. 2017, 26, 1474–1492. [Google Scholar] [CrossRef] [PubMed]
- DiAntonio, A.; Milbrandt, J.; Figley, M.D. The SARM1 TIR NADase: Mechanistic Similarities to Bacterial Phage Defense and Toxin-Antitoxin Systems. Front. Immunol. 2021, 12, 3735. [Google Scholar] [CrossRef]
- Shimazu, T.; Degenhardt, K.; Nur-E-Kamal, A.; Zhang, J.; Yoshida, T.; Zhang, Y.; Mathew, R.; White, E.; Inouye, M. NBK/BIK antagonizes MCL-1 and BCL-X L and activates BAK-mediated apoptosis in response to protein synthesis inhibition. Genes Dev. 2007, 21, 929–941. [Google Scholar] [CrossRef]
- De la Cueva-Méndez, G.; Mills, A.D.; Clay-Farrace, L.; Diaz-Orejas, R.; Laskey, R.A. Regulatable killing of eukaryotic cells by the prokaryotic proteins Kid and Kis. EMBO J. 2003, 22, 246–251. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, J.; Hoeflich, K.P.; Ikura, M.; Qing, G.; Inouye, M. MazF cleaves cellular mRNAs specifically at ACA to block protein synthesis in Escherichia coli. Mol. Cell 2003, 12, 913–923. [Google Scholar] [CrossRef]
- Shimazu, T.; Mirochnitchenko, O.; Phadtare, S.; Inouye, M. Regression of Solid Tumors by Induction of MazF, a Bacterial mRNA Endoribonuclease. Microb. Physiol. 2014, 24, 228–233. [Google Scholar] [CrossRef]
- Shapira, S.; Shapira, A.; Kazanov, D.; Hevroni, G.; Kraus, S.; Arber, N. Selective eradication of cancer cells by delivery of adenovirus-based toxins. Oncotarget 2017, 8, 38581. [Google Scholar] [CrossRef]
- Shapira, S.; Boustanai, I.; Kazanov, D.; Ben Shimon, M.; Fokra, A.; Arber, N. Innovative dual system approach for selective eradication of cancer cells using viral-based delivery of natural bacterial toxin–antitoxin system. Oncogene 2021, 40, 4967–4979. [Google Scholar] [CrossRef]
- Bulcha, J.T.; Wang, Y.; Ma, H.; Tai, P.W.L.; Gao, G. Viral vector platforms within the gene therapy landscape. Sig. Transduct. Target. Ther. 2021, 6, 53. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, M.S.A. Bacteria-Mediated Delivery of mazF mRNA into Cancer Cells for Bacteria-Mediated Delivery of mazF mRNA into Cancer Cells for Induction of Apoptosis Induction of Apoptosis. Ph.D. Thesis, Clemson University, Clemson, SC, USA, 2018. [Google Scholar]
- Saffarian, M.; Zadeh, A.; Macpherson, R.A.; Huang, G.; Ding, H.; Powell, R.R.; Bruce, T.; Tzeng, T.-R.J.; Tzeng, J. Bacterial Ribonuclease MazF-Mediated Apoptosis as Potential Cancer Therapy. Res. Artic. 2021. preprint. [Google Scholar] [CrossRef]
- Yahya, E.B.; Alqadhi, A.M. Recent trends in cancer therapy: A review on the current state of gene delivery. Life Sci. 2021, 269, 119087. [Google Scholar] [CrossRef] [PubMed]
- Oladejo, M.; Paterson, Y.; Wood, L.M. Clinical Experience and Recent Advances in the Development of Listeria-Based Tumor Immunotherapies. Front. Immunol. 2021, 12, 642316. [Google Scholar] [CrossRef]
- Belete, T.M. The Current Status of Gene Therapy for the Treatment of Cancer. Biol. Targets Ther. 2021, 15, 67–77. [Google Scholar] [CrossRef]
- Pimentel, B.; Nair, R.; Bermejo-Rodríguez, C.; Preston, M.A.; Agu, C.A.; Wang, X.; Bernal, J.A.; Sherratt, D.J.; De La Cueva-Méndez, G. Toxin Kid uncouples DNA replication and cell division to enforce retention of plasmid R1 in Escherichia coli cells. Proc. Natl. Acad. Sci. USA 2014, 111, 2734–2739. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, Y.; Li, Y.; Bahar Ali, Z.Z. Functional characterization of Kid-Kis and MazF-MazE in Sf9 cells and Mythimna separata embryos. Pestic. Biochem. Physiol. 2021, 174, 104814. [Google Scholar] [CrossRef]
- Muñoz-Gómez, A.J.; Lemonnier, M.; Santos-Sierra, S.; Berzal-Herranz, A.; Díaz-Orejas, R. RNase/Anti-RNase Activities of the Bacterial parD Toxin-Antitoxin System. J. Bacteriol. 2005, 187, 3151. [Google Scholar] [CrossRef]
- Preston, M.A.; Pimentel, B.; Bermejo-Rodríguez, C.; Dionne, I.; Turnbull, A.; De La Cueva-Méndez, G. Repurposing a Prokaryotic Toxin-Antitoxin System for the Selective Killing of Oncogenically Stressed Human Cells. ACS Synth. Biol. 2016, 5, 540–546. [Google Scholar] [CrossRef]
- Turnbull, A.; Bermejo-Rodríguez, C.; Preston, M.A.; Garrido-Barros, M.; Pimentel, B.; De La Cueva-Méndez, G. Targeted cancer cell killing by highly selective miRNA-triggered activation of a prokaryotic toxin-antitoxin system. bioRxiv 2019. [Google Scholar] [CrossRef]
- Houri, H.; Ghalavand, Z.; Faghihloo, E.; Fallah, F.; Mohammadi-Yeganeh, S. Exploiting yoeB-yefM toxin-antitoxin system of Streptococcus pneumoniae on the selective killing of miR-21 overexpressing breast cancer cell line (MCF-7). J. Cell. Physiol. 2020, 235, 2925–2936. [Google Scholar] [CrossRef]
- Unterholzner, S.J.; Poppenberger, B.; Rozhon, W. Toxin—Antitoxin systems Biology, identification, and application. Mob. Genet. Elem. 2016, 3, e26219. [Google Scholar] [CrossRef] [PubMed]
- Schuster, C.F.; Bertram, R. Toxin-antitoxin systems are ubiquitous and versatile modulators of prokaryotic cell fate. FEMS Microbiol. Lett. 2013, 340, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Bukowski, M.; Rojowska, A.; Wladyka, B. Prokaryotic toxin-antitoxin systems—The role in bacterial physiology and application in molecular biology. Acta Biochim. Pol. 2011, 58, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pecota, D.C.; Kim, C.S.; Wu, K.; Gerdes, K.; Wood, T.K. Combining the hok/sok, parDE, and pnd postsegregational killer loci to enhance plasmid stability. Appl. Environ. Microbiol. 1997, 63, 1917–1924. [Google Scholar] [CrossRef] [PubMed]
- Szpirer, C.Y.; Milinkovitch, M.C. Separate-component-stabilization system for protein and DNA production without the use of antibiotics. Biotechniques 2005, 38, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Danino, T.; Prindle, A.; Kwong, G.A.; Skalak, M.; Li, H.; Allen, K.; Hasty, J.; Bhatia, S.N. Programmable probiotics for detection of cancer in urine. Sci. Transl. Med. 2015, 7, aaa3519. [Google Scholar] [CrossRef]
- Abedi, M.H.; Yao, M.S.; Mittelstein, D.R.; Bar-Zion, A.; Swift, M.B.; Lee-Gosselin, A.; Barturen-Larrea, P.; Buss, M.T.; Shapiro, M.G. Ultrasound-controllable engineered bacteria for cancer immunotherapy. Nat. Commun. 2022, 13, 1585. [Google Scholar] [CrossRef]
- Fedorec, A.J.H.; Ozdemir, T.; Doshi, A.; Ho, Y.K.; Rosa, L.; Rutter, J.; Velazquez, O.; Pinheiro, V.B.; Danino, T.; Barnes, C.P. Two New Plasmid Post-segregational Killing Mechanisms for the Implementation of Synthetic Gene Networks in Escherichia coli. iScience 2019, 14, 323–334. [Google Scholar] [CrossRef]
- Wu, A.Y.; Kamruzzaman, M.; Iredell, J.R. Specialised functions of two common plasmid mediated toxin-Antitoxin systems, ccdAB and pemIK, in Enterobacteriaceae. PLoS ONE 2020, 15, e0230652. [Google Scholar] [CrossRef]
- Faridani, O.R.; Nikravesh, A.; Pandey, D.P.; Gerdes, K.; Good, L. Competitive inhibition of natural antisense Sok-RNA interactions activates Hok-mediated cell killing in Escherichia coli. Nucleic Acids Res. 2006, 34, 5915–5922. [Google Scholar] [CrossRef] [PubMed]
- JEON, H.; CHOI, E.; HWANG, J. Identification and characterization of VapBC toxin-antitoxin system in Bosea sp. PAMC 26642 isolated from Arctic lichens. RNA 2021, 27, 1374–1389. [Google Scholar] [CrossRef] [PubMed]
- Ames, J.R.; McGillick, J.; Murphy, T.; Reddem, E.; Bourne, C.R. Identifying a Molecular Mechanism That Imparts Species-Specific Toxicity to YoeB Toxins. Front. Microbiol. 2020, 11, 959. [Google Scholar] [CrossRef]
- Stieber, D.; Gabant, P.; Szpirer, C.Y. The art of selective killing: Plasmid toxin/antitoxin systems and their technological applications. Biotechniques 2008, 45, 344–346. [Google Scholar] [CrossRef]
- Bernard, P. New ccdB positive-selection cloning vectors with kanamycin or chloramphenicol selectable markers. Gene 1995, 162, 159–160. [Google Scholar] [CrossRef] [PubMed]
- Young-Jun, C.; Tsung-Tsan, W.; Lee, B.H. Positive selection vectors. Crit. Rev. Biotechnol. 2002, 22, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Katzen, F. Gateway® recombinational cloning: A biological operating system. Expert Opin. Drug Discov. 2007, 2, 571–589. [Google Scholar] [CrossRef]
- Hartley, J.L. Use of the Gateway System for Protein Expression in Multiple Hosts. Curr. Protoc. Protein Sci. 2002, 30, 5.17.1–5.17.10. [Google Scholar] [CrossRef]
- Perehinec, T.M.; Qazi, S.N.A.; Gaddipati, S.R.; Salisbury, V.; Rees, C.E.D.; Hill, P.J. Construction and evaluation of multisite recombinatorial (Gateway) cloning vectors for Gram-positive bacteria. BMC Mol. Biol. 2007, 8, 80. [Google Scholar] [CrossRef]
- Suzuki, M.; Zhang, J.; Liu, M.; Woychik, N.A.; Inouye, M. Single protein production in living cells facilitated by an mRNA interferase. Mol. Cell 2005, 18, 253–261. [Google Scholar] [CrossRef]
- Schneider, W.M.; Inouye, M.; Montelione, G.T.; Roth, M.J. Independently inducible system of gene expression for condensed single protein production (cSPP) suitable for high efficiency isotope enrichment. J. Struct. Funct. Genom. 2009, 10, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Gómez, A.J.; Santos-Sierra, S.; Berzal-Herranz, A.; Lemonnier, M.; Díaz-Orejas, R. Insights into the specificity of RNA cleavage by the Escherichia coli MazF toxin. FEBS Lett. 2004, 567, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, L.; Zhang, J.; Inouye, M. Characterization of ChpBK, an mRNA interferase from Escherichia coli. J. Biol. Chem. 2005, 280, 26080–26088. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yamaguchi, Y.; Inouye, M. Bacillus subtilis MazF-bs (EndoA) is a UACAU-specific mRNA interferase. FEBS Lett. 2011, 585, 2526–2532. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Zhu, L.; Suzuki, M.; Inouye, M. Interference of mRNA function by sequence-specific endoribonuclease PemK. J. Biol. Chem. 2004, 279, 20678–20684. [Google Scholar] [CrossRef]
- Alalam, H.; Sigurdardóttir, S.; Bourgard, C.; Tiukova, I.; King, R.D.; Grøtli, M.; Sunnerhagen, P. A Genetic Trap in Yeast for Inhibitors of SARS-CoV-2 Main Protease. MSystems 2021, 6, e01087-21. [Google Scholar] [CrossRef]
- García, J.L.; Díaz, E. Plasmids as Tools for Containment. In Plasmids: Biology and Impact in Biotechnology and Discovery; John Wiley & Sons: Hoboken, NJ, USA, 2015; pp. 615–631. [Google Scholar] [CrossRef]
- Molin, S.; Klemm, P.; Poulsen, L.K.; Biehl, H.; Gerdes, K.; Andersson, P. Conditional suicide system for containment of bacteria and plasmids. Nat. Biotechnol. 1987, 5, 1315–1318. [Google Scholar] [CrossRef]
- Schweder, T.; Schmidt, I.; Herrmann, H.; Neubauer, P.; Hecker, M.; Hofmann, K. An expression vector system providing plasmid stability and conditional suicide of plasmid-containing cells. Appl. Microbiol. Biotechnol. 1992, 38, 91–93. [Google Scholar] [CrossRef]
- Stirling, F.; Bitzan, L.; O’Keefe, S.; Redfield, E.; Oliver, J.W.K.; Way, J.; Silver, P.A. Rational Design of Evolutionarily Stable Microbial Kill Switches. Mol. Cell 2017, 68, 686–697.e3. [Google Scholar] [CrossRef]
- Zhou, Y.; Sun, T.; Chen, Z.; Song, X.; Chen, L.; Zhang, W. Development of a New Biocontainment Strategy in Model Cyanobacterium Synechococcus Strains. ACS Synth. Biol. 2019, 8, 2576–2584. [Google Scholar] [CrossRef]
- Halvorsen, T.M.; Ricci, D.P.; Park, D.M.; Jiao, Y.; Yung, M.C. Comparison of Kill Switch Toxins in Plant-Beneficial Pseudomonas fluorescens Reveals Drivers of Lethality, Stability, and Escape. ACS Synth. Biol. 2022, 11, 3785–3796. [Google Scholar] [CrossRef] [PubMed]
- Kristoffersen, P.; Jensen, G.B.; Gerdes, K.; Piškur, J. Bacterial toxin-antitoxin gene system as containment control in yeast cells. Appl. Environ. Microbiol. 2000, 66, 5524–5526. [Google Scholar] [CrossRef] [PubMed]
- Bakar, F.A.; Yeo, C.C.; Harikrishna, J.A. Expression of the Streptococcus pneumoniae yoeB Chromosomal toxin gene causes Cell Death in the model plant Arabidopsis thaliana. BMC Biotechnol. 2015, 15, 26. [Google Scholar] [CrossRef]
- Baldacci-Cresp, F.; Houbaert, A.; Metuor Dabire, A.; Mol, A.; Monteyne, D.; El Jaziri, M.; Van Melderen, L.; Baucher, M. Escherichia coli mazEF Toxin-Antitoxin System as a Tool to Target Cell Ablation in Plants. J. Mol. Microbiol. Biotechnol. 2016, 26, 277–283. [Google Scholar] [CrossRef] [PubMed]
- de Souza-Neto, R.R.; Carvalho, I.G.B.; Martins, P.M.M.; Picchi, S.C.; Tomaz, J.P.; Caserta, R.; Takita, M.A.; de Souza, A.A. MqsR toxin as a biotechnological tool for plant pathogen bacterial control. Sci. Rep. 2022, 12, 2794. [Google Scholar] [CrossRef] [PubMed]
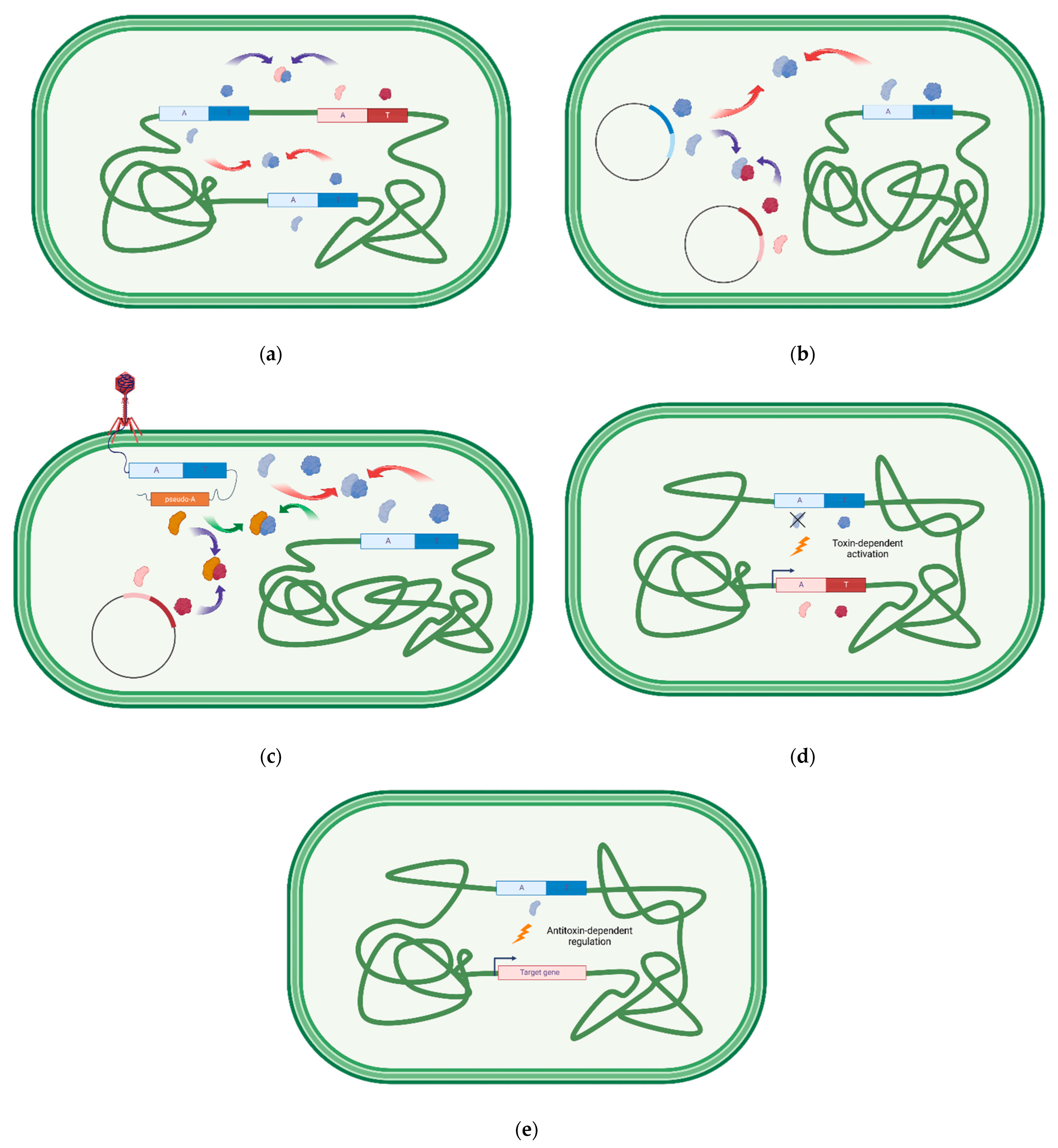
| Original TA Location | Type of Cross-Talk | Examples of Cross-Talk |
|---|---|---|
| chromosome/chromosome | homologous TAs cross-interactions | M. tuberculosis MazEF and RelBE [100,101,102] B. longum MazEF [103] Y. pestis RelBE [104] |
| chromosome/chromosome | non-homologous TAs cross-interactions | B. longum MazEF-RelBE [103], M. tuberculosis MazEF-VapBC [100] |
| chromosome/plasmid | homologous TAs cross-interactions | E. chrysanthemi—P1 CcdAB, E. coli O157:H7—pO157 CcdAB E. coli O157: H7—pB171-like AtaRT [80] E. coli MazEF—R1 Kis/Kid [105] |
| plasmid/plasmid | non- homologous TAs cross-interactions | E. coli R1 Kis/Kid—F1 CcdAB [106] |
| chromosome/phage | homologous TAs cross-interactions | V. cholerae Phd/Doc—P1 Phd/Doc [107] |
| chromosome/phage | non- homologous TAs cross-interactions | E. coli RnlAB—T4 Dmd [108] |
| plasmid/phage | non- homologous TAs cross-interactions | E. coli O157:H7 pOSAK1 IsoAB—T4 Dmd [88] |
| chromosome or plasmid or phage | TAs-indirect cross-activation | Streptomyces and M. tuberculosis VapBC-Lon-several TAS [109,110] Salmonella and Shigella VapBC—Lon-YefM/YoeB [111] E. coli RelBEF—Lon independent-several TAS [112] |
| chromosome or plasmid or phage | TAs-direct cross-regulation | S. oneidensis ParE-CopA-promoter of MR-1 PemIK [76] E. faecium pRUM Axe/Txe-promoter of E. coli YefM/YoeB and vice versa [113] |
| chromosome or plasmid or phage | TA-direct regulation of unrelated protein | P. aeruginosa HigA-mvfR promoter [114] P. rubra PrpA-ori iterons [115] E. coli MqsA-csgD promoter [116] E. coli DinJ-cspE promoter [117] E. coli HipB-relA, eutH and fadH promoters [118] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boss, L.; Kędzierska, B. Bacterial Toxin-Antitoxin Systems’ Cross-Interactions—Implications for Practical Use in Medicine and Biotechnology. Toxins 2023, 15, 380. https://doi.org/10.3390/toxins15060380
Boss L, Kędzierska B. Bacterial Toxin-Antitoxin Systems’ Cross-Interactions—Implications for Practical Use in Medicine and Biotechnology. Toxins. 2023; 15(6):380. https://doi.org/10.3390/toxins15060380
Chicago/Turabian StyleBoss, Lidia, and Barbara Kędzierska. 2023. "Bacterial Toxin-Antitoxin Systems’ Cross-Interactions—Implications for Practical Use in Medicine and Biotechnology" Toxins 15, no. 6: 380. https://doi.org/10.3390/toxins15060380
APA StyleBoss, L., & Kędzierska, B. (2023). Bacterial Toxin-Antitoxin Systems’ Cross-Interactions—Implications for Practical Use in Medicine and Biotechnology. Toxins, 15(6), 380. https://doi.org/10.3390/toxins15060380





