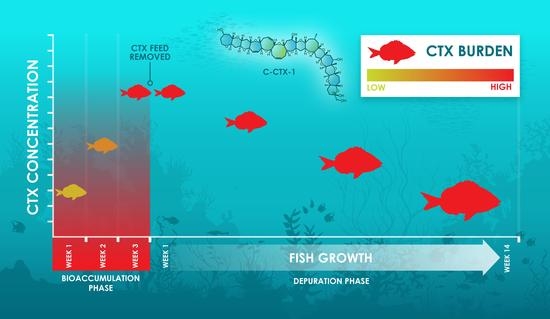
Depuration Kinetics and Growth Dilution of Caribbean Ciguatoxin in the Omnivore Lagodon rhomboides: Implications for Trophic Transfer and Ciguatera Risk
Abstract
:1. Introduction
2. Results
2.1. Experimental Design, Diet Formulation and Consistency
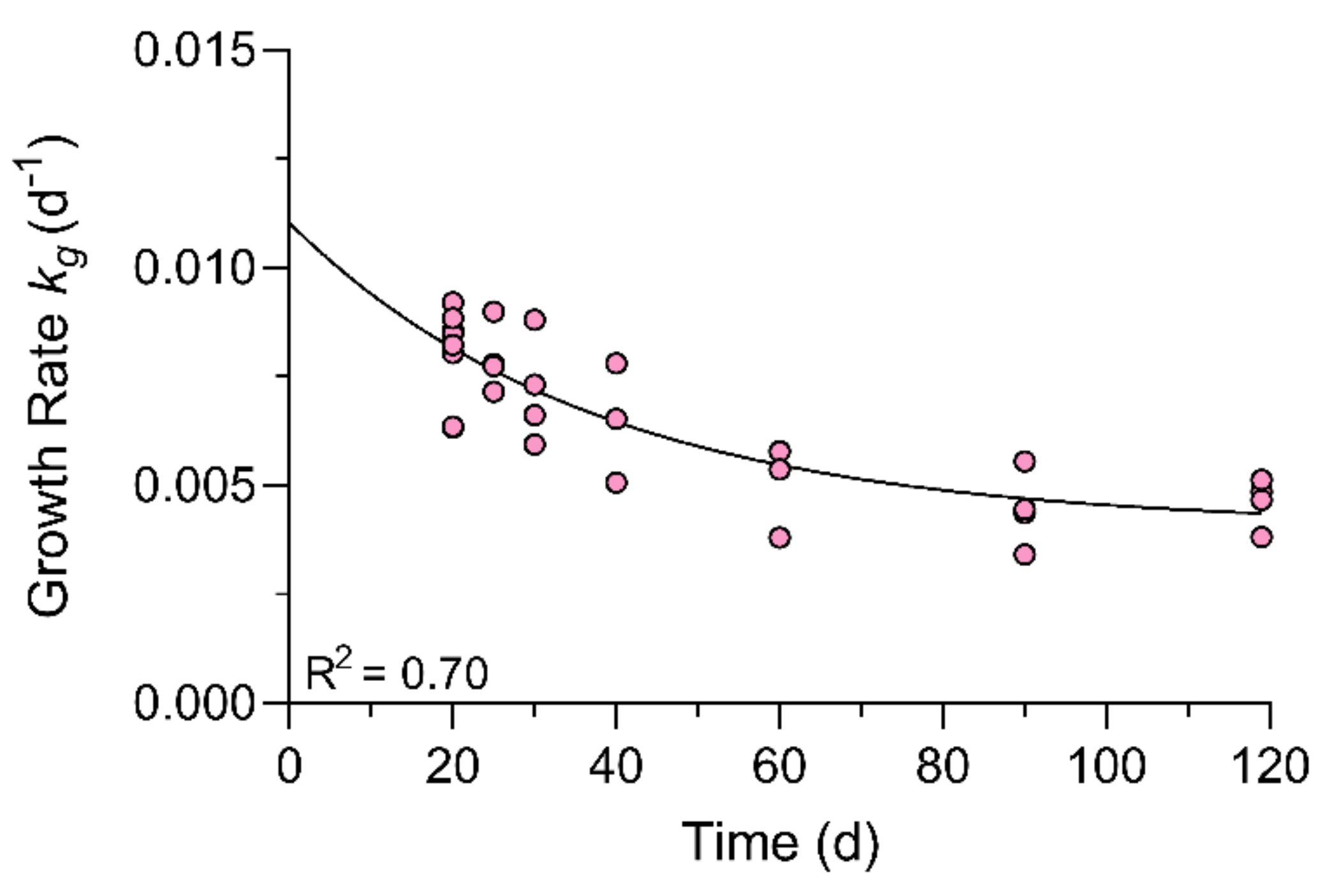
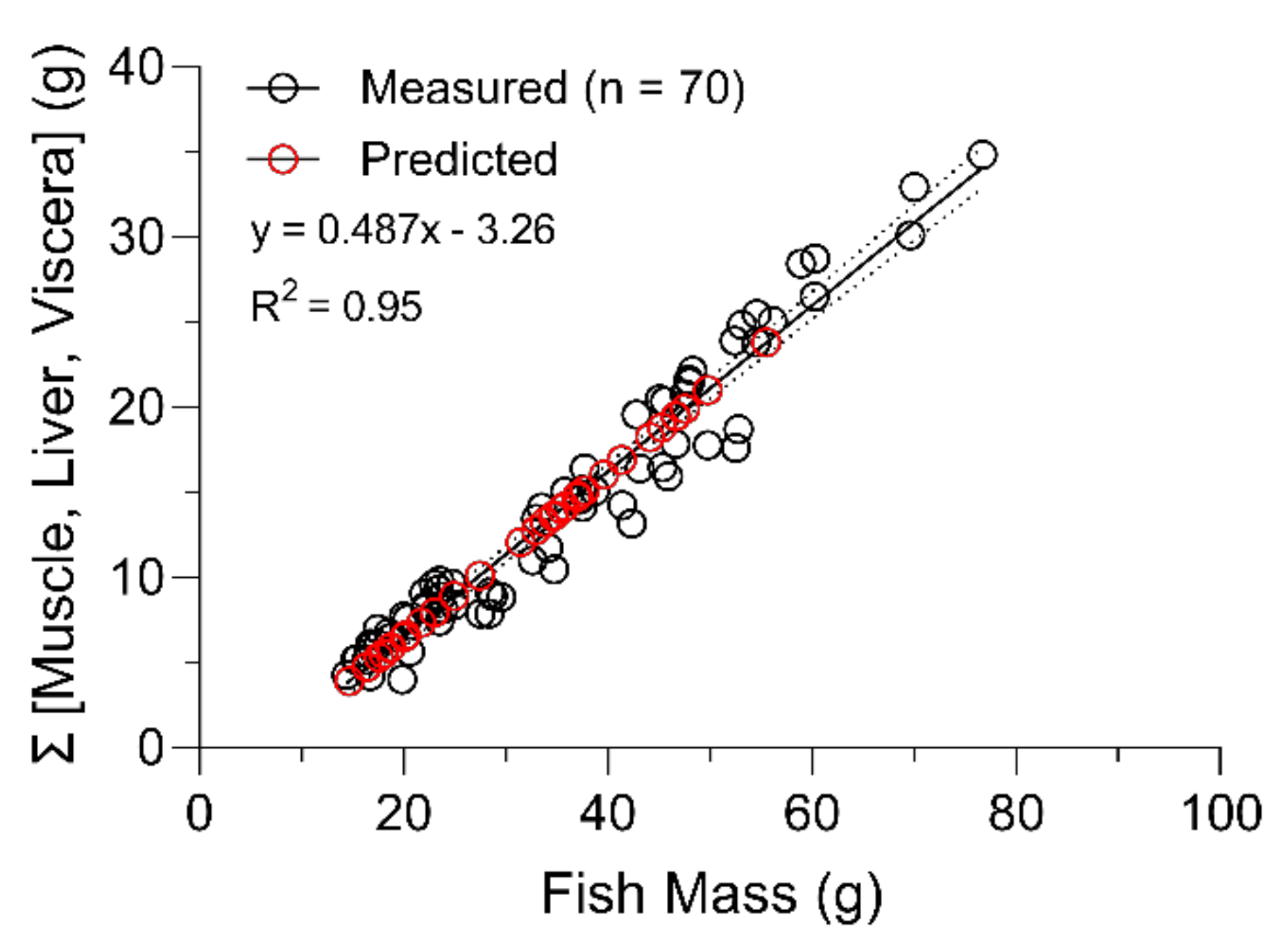
2.2. Fish Growth
2.3. Toxin Distribution in the Bioaccumulation Phase
2.4. Toxin Distribution in the Depuration Phase
2.5. Muscle, Liver, and Viscera CTX Kinetics
2.6. Muscle, Liver, and Viscera CTX Burdens
2.7. One-Compartment Model Kinetics and Growth Correction
3. Discussion
3.1. Experimental Considerations and Outcomes
3.2. Muscle, Liver, and Viscera CTX Kinetics: Bioaccumulation
3.3. Muscle, Liver, and Viscera CTX Kinetics: Depuration
3.4. CTX Tissue Distribution
3.5. One-Compartment Model Kinetics and Growth Correction
3.6. Conclusions and Implications for Trophic Transfer
4. Materials and Methods
4.1. Reagents and Chemicals
4.2. Controlled Exposure
4.2.1. Fish Collection and Acclimation
4.2.2. Feed Formulation
4.2.3. Experimental Design
4.2.4. Fish Sampling, Dissection, and Extraction
4.3. Toxin Analysis
4.3.1. Maintenance of Neuroblastoma Cells
4.3.2. Neuroblastoma MTT Assay (N2a-MTT)
4.3.3. N2a-MTT Data Analysis
4.4. Ciguatoxin Kinetics
4.4.1. Muscle, Liver, and Viscera Ciguatoxin Kinetics
4.4.2. One-Compartment Model Kinetics
4.4.3. Kinetic Modeling and Correction of Growth Dilution
4.5. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adachi, R.; Fukuyo, Y. The thecal structure of a marine toxic dinoflagellate Gambierdiscus toxicus gen. et sp. nov. collected in a ciguatera-endemic area. Bull. Jpn. Soc. Sci. Fish 1979, 45, 67–71. [Google Scholar] [CrossRef] [Green Version]
- Bagnis, R.; Chanteau, S.; Chungue, E.; Hurtel, J.; Yasumoto, T.; Inoue, A. Origins of ciguatera fish poisoning: A new dinoflagellate, Gambierdiscus toxicus Adachi and Fukuyo, definitively involved as a causal agent. Toxicon 1980, 18, 199–208. [Google Scholar] [CrossRef]
- Laza-Martínez, A.; David, H.; Riobó, P.; Miguel, I.; Orive, E. Characterization of a strain of Fukuyoa paulensis (Dinophyceae) from the Western Mediterranean Sea. J. Eukaryot. Microbiol. 2016, 63, 481–497. [Google Scholar] [CrossRef] [Green Version]
- Litaker, R.W.; Holland, W.C.; Hardison, D.R.; Pisapia, F.; Hess, P.; Kibler, S.R.; Tester, P.A. Ciguatoxicity of Gambierdiscus and Fukuyoa species from the Caribbean and Gulf of Mexico. PLoS ONE 2017, 12, e0185776. [Google Scholar] [CrossRef]
- Rhodes, L.; Harwood, T.; Smith, K.; Argyle, P.; Munday, R. Production of ciguatoxin and maitotoxin by strains of Gambierdiscus australes, G. pacificus and G. polynesiensis (Dinophyceae) isolated from Rarotonga, Cook Islands. Harmful Algae 2014, 39, 185–190. [Google Scholar] [CrossRef]
- Chinain, M.; Germain, M.; Deparis, X.; Pauillac, S.; Legrand, A.-M. Seasonal abundance and toxicity of the dinoflagellate Gambierdiscus spp. (Dinophyceae), the causative agent of ciguatera in Tahiti, French Polynesia. Mar. Biol. 1999, 135, 259–267. [Google Scholar] [CrossRef]
- Parsons, M.L.; Settlemier, C.J.; Bienfang, P.K. A simple model capable of simulating the population dynamics of Gambierdiscus, the benthic dinoflagellate responsible for ciguatera fish poisoning. Harmful Algae 2010, 10, 71–80. [Google Scholar] [CrossRef]
- Xu, Y.; Richlen, M.L.; Liefer, J.D.; Robertson, A.; Kulis, D.; Smith, T.B.; Parsons, M.L.; Anderson, D.M. Influence of environmental variables on Gambierdiscus spp.(Dinophyceae) growth and distribution. PLoS ONE 2016, 11, e0153197. [Google Scholar] [CrossRef] [PubMed]
- Liefer, J.D.; Richlen, M.L.; Smith, T.B.; DeBose, J.L.; Xu, Y.; Anderson, D.M.; Robertson, A. Asynchrony of Gambierdiscus spp. Abundance and Toxicity in the US Virgin Islands: Implications for Monitoring and Management of Ciguatera. Toxins 2021, 13, 413. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.J.; Lewis, R.J.; Poli, M.A.; Gillespie, N.C. Strain dependent production of ciguatoxin precursors (gambiertoxins) by Gambierdiscus toxicus (Dinophyceae) in culture. Toxicon 1991, 29, 761–775. [Google Scholar] [CrossRef]
- Lewis, R.; Sellin, M.; Street, R.; Holmes, M.; Gillespie, N. Excretion of ciguatoxin from moray eels (Muraenidae) of the central Pacific. In Proceedings of the Third International Conference on Ciguatera Fish Poisoning, La Parguera, Puerto Rico, 30 April–5 May 1990; Tosteson, T.R., Ed.; Polyscience Publications: Morin Heights, QC, Canada, 1992; pp. 131–143. [Google Scholar]
- Lewis, R.J.; Holmes, M.J. Origin and transfer of toxins involved in ciguatera. Comp. Biochem. Physiol. Part C Pharmacol. Toxicol. Endocrinol. 1993, 106, 615–628. [Google Scholar] [CrossRef]
- Chateau-Degat, M.-L.; Chinain, M.; Cerf, N.; Gingras, S.; Hubert, B.; Dewailly, E. Seawater temperature, Gambierdiscus spp. variability and incidence of ciguatera poisoning in French Polynesia. Harmful Algae 2005, 4, 1053–1062. [Google Scholar] [CrossRef]
- Llewellyn, L.E. Revisiting the association between sea surface temperature and the epidemiology of fish poisoning in the South Pacific: Reassessing the link between ciguatera and climate change. Toxicon 2010, 56, 691–697. [Google Scholar] [CrossRef]
- Darius, H.; Ponton, D.; Revel, T.; Cruchet, P.; Ung, A.; Fouc, M.T.; Chinain, M. Ciguatera risk assessment in two toxic sites of French Polynesia using the receptor-binding assay. Toxicon 2007, 50, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Chinain, M.; Darius, H.T.; Ung, A.; Fouc, M.T.; Revel, T.; Cruchet, P.; Pauillac, S.; Laurent, D. Ciguatera risk management in French Polynesia: The case study of Raivavae Island (Australes Archipelago). Toxicon 2010, 56, 674–690. [Google Scholar] [CrossRef] [PubMed]
- Banner, A.H.; Helfrich, P.; Piyakarnchana, T. Retention of ciguatera toxin by the red snapper, Lutjanus bohar. Copeia 1966, 2, 297–301. [Google Scholar] [CrossRef]
- Davin, W.T.; Kohler, C.C.; Tindall, D.R. Ciguatera toxins adversely affect piscivorous fishes. Trans. Am. Fish. Soc. 1988, 117, 374–384. [Google Scholar] [CrossRef]
- Ledreux, A.; Brand, H.; Chinain, M.; Bottein, M.-Y.D.; Ramsdell, J.S. Dynamics of ciguatoxins from Gambierdiscus polynesiensis in the benthic herbivore Mugil cephalus: Trophic transfer implications. Harmful Algae 2014, 39, 165–174. [Google Scholar] [CrossRef]
- Li, J.; Mak, Y.L.; Chang, Y.-H.; Xiao, C.; Chen, Y.-M.; Shen, J.; Wang, Q.; Ruan, Y.; Lam, P.K. Uptake and Depuration Kinetics of Pacific Ciguatoxins (P-CTXs) in Orange-spotted Grouper (Epinephelus coioides). Environ. Sci. Technol. 2020, 54, 4475–4483. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Henao, A.; García-Álvarez, N.; Padilla, D.; Ramos-Sosa, M.; Silva Sergent, F.; Fernández, A.; Estévez, P.; Gago-Martínez, A.; Diogène, J.; Real, F. Accumulation of C-CTX1 in Muscle Tissue of Goldfish (Carassius auratus) by Dietary Experience. Animals 2021, 11, 242. [Google Scholar] [CrossRef]
- Leite, I.d.P.; Sdiri, K.; Taylor, A.; Viallon, J.; Gharbia, H.B.; Mafra Júnior, L.L.; Swarzenski, P.; Oberhaensli, F.; Darius, H.T.; Chinain, M. Experimental Evidence of Ciguatoxin Accumulation and Depuration in Carnivorous Lionfish. Toxins 2021, 13, 564. [Google Scholar] [CrossRef]
- Holmes, M.J.; Venables, B.; Lewis, R.J. Critical Review and Conceptual and Quantitative Models for the Transfer and Depuration of Ciguatoxins in Fishes. Toxins 2021, 13, 515. [Google Scholar] [CrossRef] [PubMed]
- Hansen, D.J. Food, growth, migration, reproduction, and abundance of pinfish, Lagodon rhomboides, and Atlantic croaker, Micropogon undulatus, near Pensacola, Florida, 1963–1965. Fish. Bull. 1969, 68, 135–146. [Google Scholar]
- Stoner, A.W. Feeding ecology of Lagodon rhomboides (Pisces: Sparidae): Variation and functional responses. Fish. Bull. 1980, 78, 337–352. [Google Scholar]
- Luczkovich, J.J.; Norton, S.R.; Gilmore, R.G. The influence of oral anatomy on prey selection during the ontogeny of two percoid fishes, Lagodon rhomboides and Centropomus undecimalis. Environ. Biol. Fishes 1995, 44, 79–95. [Google Scholar] [CrossRef]
- Luczkovich, J.J. The Patterns and Mechanisms of Selective Feeding on Seagrass-Meadow Epifauna by Juvenile Pinfish, Lagodon Rhomboides (Linnaeus); The Florida State University: Tallahassee, FL, USA, 1987. [Google Scholar]
- Hoese, H.D.; Moore, R.H. Fishes of the Gulf of Mexico, Texas, Louisiana, and Adjacent Waters, 2nd ed.; Texas A&M University: College Station, TX, USA, 1977. [Google Scholar]
- Nelson, J.; Wilson, R.; Coleman, F.; Koenig, C.; DeVries, D.; Gardner, C.; Chanton, J. Flux by fin: Fish-mediated carbon and nutrient flux in the northeastern Gulf of Mexico. Mar. Biol. 2012, 159, 365–372. [Google Scholar] [CrossRef]
- Nelson, J.A.; Stallings, C.D.; Landing, W.M.; Chanton, J. Biomass transfer subsidizes nitrogen to offshore food webs. Ecosystems 2013, 16, 1130–1138. [Google Scholar] [CrossRef] [Green Version]
- Naughton, S.P.; Saloman, C.H. Food of Gag (Mycteroperca microlepis) from North Carolina and Three Areas of Florida; US Department of Commerce, National Oceanic and Atmospheric Administration: Panama City, FL, USA, 1985; Volume 160. [Google Scholar]
- Naar, J.P.; Flewelling, L.J.; Lenzi, A.; Abbott, J.P.; Granholm, A.; Jacocks, H.M.; Gannon, D.; Henry, M.; Pierce, R.; Baden, D.G. Brevetoxins, like ciguatoxins, are potent ichthyotoxic neurotoxins that accumulate in fish. Toxicon 2007, 50, 707–723. [Google Scholar] [CrossRef] [Green Version]
- Clausing, R.J.; Losen, B.; Oberhaensli, F.R.; Darius, H.T.; Sibat, M.; Hess, P.; Swarzenski, P.W.; Chinain, M.; Bottein, M.-Y.D. Experimental evidence of dietary ciguatoxin accumulation in an herbivorous coral reef fish. Aquat. Toxicol. 2018, 200, 257–265. [Google Scholar] [CrossRef] [Green Version]
- Friedman, M.A.; Fleming, L.E.; Fernandez, M.; Bienfang, P.; Schrank, K.; Dickey, R.; Bottein, M.-Y.; Backer, L.; Ayyar, R.; Weisman, R.; et al. Ciguatera Fish Poisoning: Treatment, Prevention and Management. Mar. Drugs 2008, 6, 456–479. [Google Scholar] [CrossRef]
- Ledreux, A.; Ramsdell, J.S. Bioavailability and intravenous toxicokinetic parameters for Pacific ciguatoxin P-CTX-1 in rats. Toxicon 2013, 64, 81–86. [Google Scholar] [CrossRef]
- Roué, M.; Darius, H.T.; Picot, S.; Ung, A.; Viallon, J.; Gaertner-Mazouni, N.; Sibat, M.; Amzil, Z.; Chinain, M. Evidence of the bioaccumulation of ciguatoxins in giant clams (Tridacna maxima) exposed to Gambierdiscus spp. cells. Harmful Algae 2016, 57, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Vernoux, J.P.; Lahlou, N.; Abbad El Andaloussi, S.; Riyeche, N.; Magras, L. A study of the distribution of ciguatoxin in individual Caribbean fish. Acta Tropica 1985, 42, 225–233. [Google Scholar] [CrossRef]
- Welling, P.G. Influence of food and diet on gastrointestinal drug absorption: A review. J. Pharmacokinet. Biopharm. 1977, 5, 291–334. [Google Scholar] [CrossRef]
- Takagaki, R.; Ishida, Y.; Sadakiyo, T.; Taniguchi, Y.; Sakurai, T.; Mitsuzumi, H.; Watanabe, H.; Fukuda, S.; Ushio, S. Effects of isomaltodextrin in postprandial lipid kinetics: Rat study and human randomized crossover study. PLoS ONE 2018, 13, e0196802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Have, G.A.M.T.; Engelen, M.P.K.J.; Luiking, Y.C.; Deutz, N.E.P. Absorption Kinetics of Amino Acids, Peptides, and Intact Proteins. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, S23–S36. [Google Scholar] [CrossRef]
- Marier, J.-F.; Vachon, P.; Gritsas, A.; Zhang, J.; Moreau, J.-P.; Ducharme, M.P. Metabolism and disposition of resveratrol in rats: Extent of absorption, glucuronidation, and enterohepatic recirculation evidenced by a linked-rat model. J. Pharmacol. Exp. Ther. 2002, 302, 369–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herman, R.J.; Van Pham, J.D.; Szakacs, C.B. Disposition of lorazepam in human beings: Enterohepatic recirculation and first-pass effect. Clin. Pharmacol. Ther. 1989, 46, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Saker, M.L.; Metcalf, J.S.; Codd, G.A.; Vasconcelos, V.M. Accumulation and depuration of the cyanobacterial toxin cylindrospermopsin in the freshwater mussel Anodonta cygnea. Toxicon 2004, 43, 185–194. [Google Scholar] [CrossRef]
- Gwinn, J.K.; Uhlig, S.; Ivanova, L.; Fæste, C.K.; Kryuchkov, F.; Robertson, A. In Vitro Glucuronidation of Caribbean Ciguatoxins in Fish: First Report of Conjugative Ciguatoxin Metabolites. Chem. Res. Toxicol. 2021, 34, 1910–1925. [Google Scholar] [CrossRef]
- Brooke, D.; Crookes, M. Depuration Rate Constant: Growth Correction and Use as an Indicator of Bioaccumulation Potential; Environment Agency: Bristol, UK, 2012; Volume 7371. [Google Scholar]
- Brooke, D.; Crookes, M.; Gray, D.; Robertson, S. Environmental Risk Assessment Report: Octamethylcyclotetrasiloxane; Environment Agency of England and Wales: Bristol, UK, 2009. [Google Scholar]
- Dechraoui, M.-Y.B.; Wacksman, J.J.; Ramsdell, J.S. Species selective resistance of cardiac muscle voltage gated sodium channels: Characterization of brevetoxin and ciguatoxin binding sites in rats and fish. Toxicon 2006, 48, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Randall, J.E. A review of ciguatera, tropical fish poisoning, with a tentative explanation of its cause. Bull. Mar. Sci. 1958, 8, 236–267. [Google Scholar]
- Robertson, A.; Richlen, M.L.; Erdner, D.; Smith, T.B.; Anderson, D.M.; Liefer, J.D.; Xu, Y.; McCarron, P.; Miles, C.O.; Parsons, M.L. Toxicity, Chemistry, and Implications of Gamberdiscus silvae: A Ciguatoxin Superbug in the Greater Caribbean Region. In Proceedings of the 18th International Conference for Harmful Algae, Nantes, France, 21–26 October 2018; p. 115. [Google Scholar]
- Kryuchkov, F.; Robertson, A.; Miles, C.O.; Mudge, E.M.; Uhlig, S. LC–HRMS and Chemical Derivatization Strategies for the Structure Elucidation of Caribbean Ciguatoxins: Identification of C-CTX-3 and-4. Mar. Drugs 2020, 18, 182. [Google Scholar] [CrossRef] [Green Version]
- Ward, D.M.; Nislow, K.H.; Chen, C.Y.; Folt, C.L. Rapid, efficient growth reduces mercury concentrations in stream-dwelling Atlantic salmon. Trans. Am. Fish. Soc. 2010, 139, 1–10. [Google Scholar] [CrossRef] [Green Version]
- EFSA Panel on Contaminants in the Food Chain. Scientific Opinion on marine biotoxins in shellfish–Emerging toxins: Ciguatoxin group. EFSA J. 2010, 8, 1627. [Google Scholar]
- Viallon, J.; Chinain, M.; Darius, H.T. Revisiting the neuroblastoma cell-based assay (CBA-N2a) for the improved detection of marine toxins active on voltage gated sodium channels (VGSCs). Toxins 2020, 12, 281. [Google Scholar] [CrossRef]
- Gobas, F.A. A model for predicting the bioaccumulation of hydrophobic organic chemicals in aquatic food-webs: Application to Lake Ontario. Ecol. Model. 1993, 69, 1–17. [Google Scholar] [CrossRef]
- Munschy, C.; Héas-Moisan, K.; Tixier, C.; Olivier, N.; Gastineau, O.; Le Bayon, N.; Buchet, V. Dietary exposure of juvenile common sole (Solea solea L.) to polybrominated diphenyl ethers (PBDEs): Part 1. Bioaccumulation and elimination kinetics of individual congeners and their debrominated metabolites. Environ. Pollut. 2011, 159, 229–237. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Trial 1 (N) | Trial 2 (N) | ||||
|---|---|---|---|---|---|
| Phase of Experiment | Experiment Day | Control | CTX | Control | CTX |
| Bioaccumulation | 0 | 4 | - | 3 | - |
| 6 | 4 | 4 | - | - | |
| 10 | 4 | 4 | - | - | |
| 20 | 4 | 4 | 2 | 4 | |
| Depuration | 25 (5) | 4 | 4 | - | - |
| 30 (10) | 4 | 4 | - | - | |
| 40 (20) | 4 | 4 | - | - | |
| 60 (40) | - | - | 2 | 4 | |
| 90 (70) | - | - | - | 4 | |
| 119 (99) | - | - | 2 | 4 | |
| Total | 28 | 24 | 9 | 16 | |
| Quality Control Parameter | Trial 1 | Trial 2 | ||
|---|---|---|---|---|
| Treatment | ||||
| Control | CTX | Control | CTX | |
| Pellet Water Content (%) a | 35.8 ± 0.5 | 27.0 ± 4.0 | 37.3 ± 1.5 | 33.7 ± 2.9 |
| Pellet Weight (g) b | 0.10 ± 0.00 | 0.09 ± 0.01 | 0.09 ± 0.01 | 0.09 ± 0.01 |
| Food CTX Concentration (ng g−1) c | 0 | 1.06 ± 0.06 | 0 | 0.96 ± 0.04 |
| Pellet CTX Concentration (ng pellet−1) c | 0 | 0.096 ± 0.020 | 0 | 0.091 ± 0.019 |
| % of Initial Weight Fed Daily (whole) d | 1.9 ± 0.2 | 1.8 ± 0.2 | 1.7 ± 0.1 | 1.8 ± 0.1 |
| % of Initial Weight Fed Daily (dry) e | 1.2 ± 0.1 | 1.3 ± 0.1 | 1.1 ± 0.1 | 1.2 ± 0.0 |
| Consumption Rate (ng g−1 fish day−1) f | 0 | 0.019 ± 0.002 | 0 | 0.017 ± 0.001 |
| Tissue | CTX3C Eq. | Statistics | ||
|---|---|---|---|---|
| Trial 1 | Trial 2 | t | p-Value | |
| Muscle | 0.04 ± 0.02 | 0.06 ± 0.01 | 1.39 | 0.214 |
| Liver | 1.01 ± 0.99 | 0.91 ± 0.48 | 0.165 | 0.875 |
| Viscera | 4.40 ± 1.17 | 3.80 ± 0.37 | 0.971 | 0.369 |
| Tissue | Statistical Parameters | Bioaccumulation | Depuration |
|---|---|---|---|
| Muscle | Linear equation | y = 0.0034x − 0.0200 | y = 0.0005x + 0.0201 |
| R2 | 0.70 | 0.43 | |
| p-value | 0.0001 | 0.0005 | |
| Liver | Linear equation | y = 0.0630x − 0.3024 | y = 0.0063x + 0.5277 |
| R2 | 0.24 | 0.09 | |
| p-value | 0.1291 | 0.1518 | |
| Viscera | Linear equation | y = 0.1239x + 1.704 | y = −0.0331x + 6.093 |
| R2 | 0.47 | 0.43 | |
| p-value | 0.0034 | 0.0005 |
| Experiment Day | Trial | Average kg × 10−3 (day−1) | k2 growth-corrected × 10−3 (day−1) a |
|---|---|---|---|
| 20 | 1 | 7.615 ± 1.492 | −0.454 |
| 20 | 2 | 8.402 ± 0.345 | −1.241 |
| 25 | 1 | 7.747 ± 0.676 | −0.586 |
| 30 | 1 | 7.037 ± 1.132 | 0.124 |
| 40 | 1 | 6.489 ± 1.270 | 0.672 |
| 60 | 2 | 5.187 ± 0.943 | 1.975 |
| 90 | 2 | 4.447 ± 0.878 | 2.714 |
| 119 | 2 | 4.619 ± 0.567 | 2.542 |
| Experiment Day | Trial | Average kg × 10−3 (day−1) | k2 growth-corrected × 10−3 (day−1) a |
|---|---|---|---|
| 20–25 | 1, 2 | 7.921 | −0.760 |
| 30–40 | 1 | 6.763 | 0.398 |
| 60–119 | 2 | 4.785 | 2.410 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bennett, C.T.; Robertson, A. Depuration Kinetics and Growth Dilution of Caribbean Ciguatoxin in the Omnivore Lagodon rhomboides: Implications for Trophic Transfer and Ciguatera Risk. Toxins 2021, 13, 774. https://doi.org/10.3390/toxins13110774
Bennett CT, Robertson A. Depuration Kinetics and Growth Dilution of Caribbean Ciguatoxin in the Omnivore Lagodon rhomboides: Implications for Trophic Transfer and Ciguatera Risk. Toxins. 2021; 13(11):774. https://doi.org/10.3390/toxins13110774
Chicago/Turabian StyleBennett, Clayton T., and Alison Robertson. 2021. "Depuration Kinetics and Growth Dilution of Caribbean Ciguatoxin in the Omnivore Lagodon rhomboides: Implications for Trophic Transfer and Ciguatera Risk" Toxins 13, no. 11: 774. https://doi.org/10.3390/toxins13110774
APA StyleBennett, C. T., & Robertson, A. (2021). Depuration Kinetics and Growth Dilution of Caribbean Ciguatoxin in the Omnivore Lagodon rhomboides: Implications for Trophic Transfer and Ciguatera Risk. Toxins, 13(11), 774. https://doi.org/10.3390/toxins13110774