Current Knowledge on Snake Dry Bites
Abstract
1. Introduction
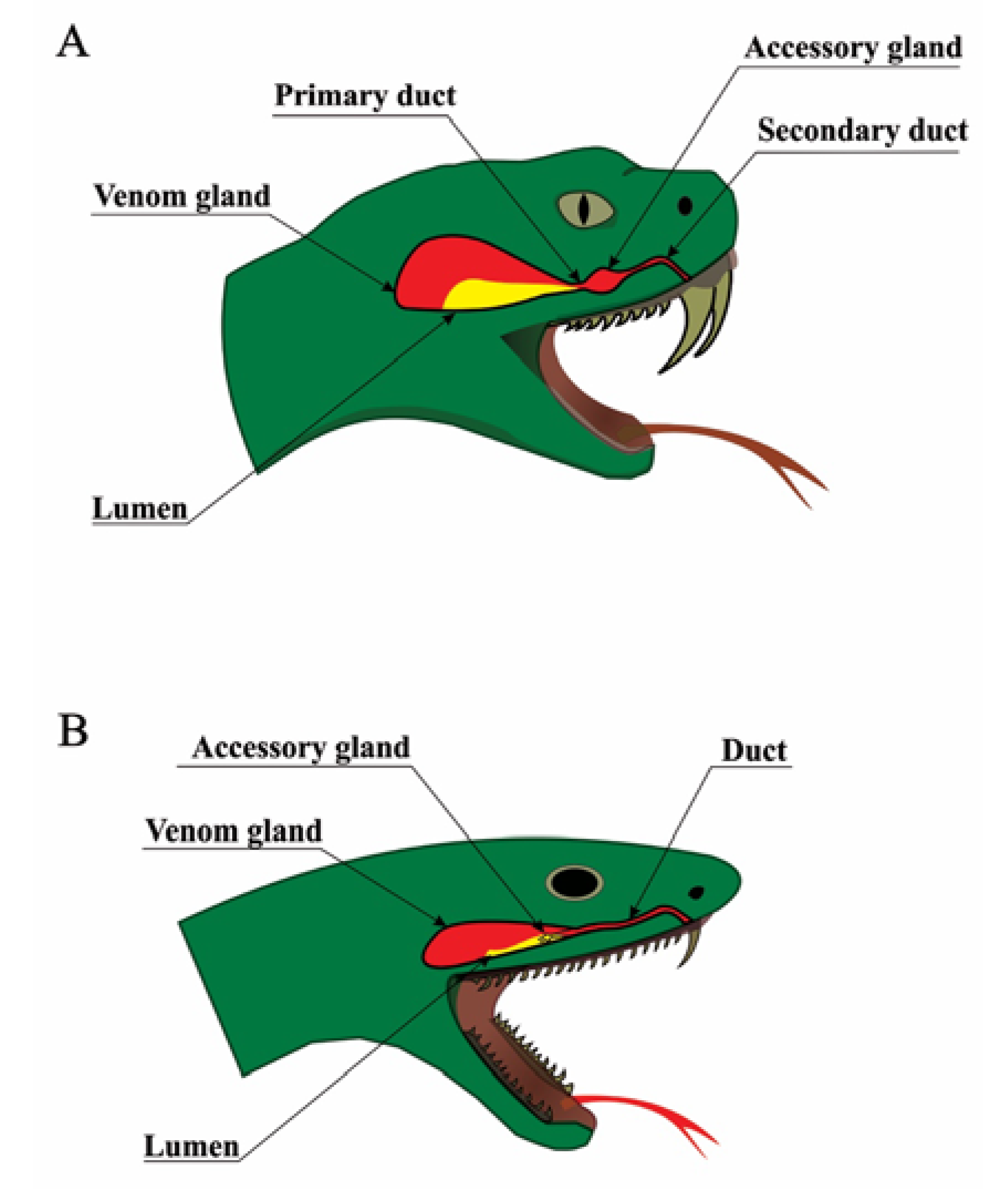
2. Snake Venom Apparatus and Venom Production
3. History of Snake Dry Bites
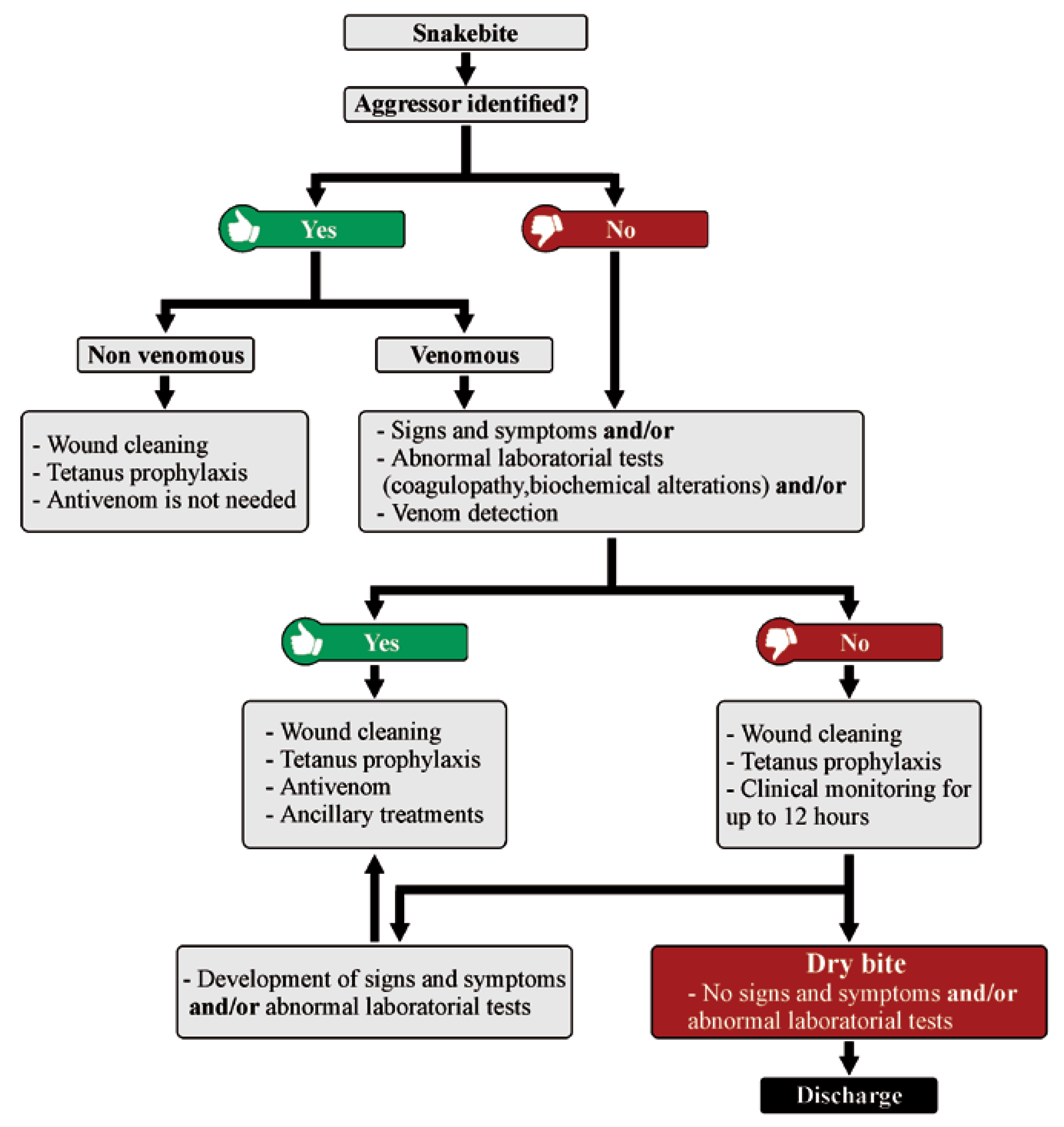
4. Diagnosis of Snake Dry Bites
5. The Snake Dry Bite Phenomenon
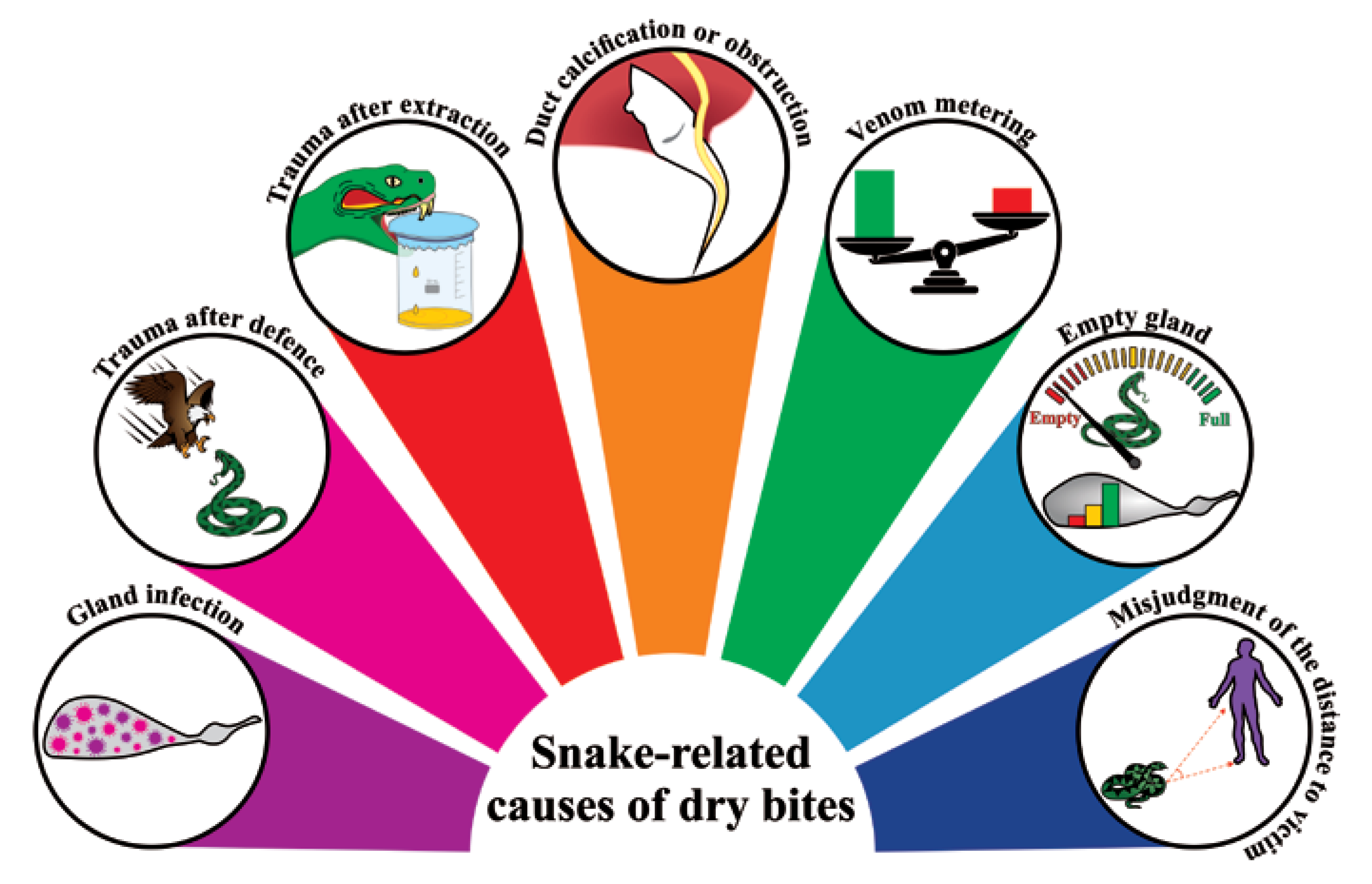
5.1. Snake-Related Factors Involved in Dry Bites
5.2. Human-Related Factors Involved in Dry Bites
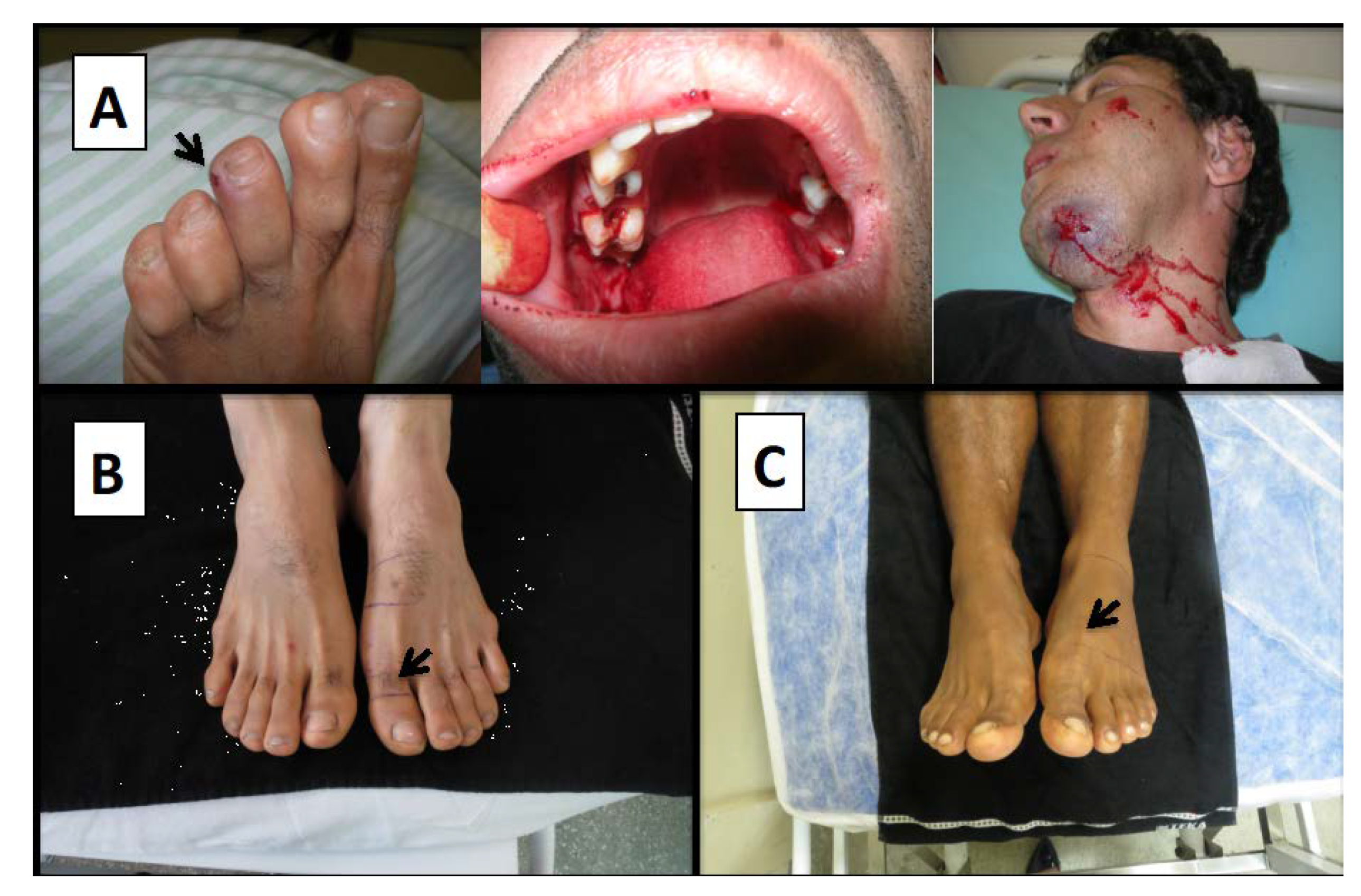
6. Clinical Features of Dry Bites: To Treat or Not to Treat?
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO Snakebite Envenoming—Key Facts 2019. Available online: https://www.who.int/news-room/fact-sheets/detail/snakebite-envenoming (accessed on 22 April 2020).
- Bolon, I.; Durso, A.M.; Mesa, S.B.; Ray, N.; Alcoba, G.; Chappuis, F.; de Castañeda, R.R. Identifying the snake: First scoping review on practices of communities and healthcare providers confronted with snakebite across the world. PLoS ONE 2020, 15, e0229989. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, B.; Warrell, D.A.; Suraweera, W.; Bhatia, P.; Dhingra, N.; Jotkar, R.M.; Rodriguez, P.S.; Mishra, K.; Whitaker, R.; Jha, P.; et al. Snakebite mortality in India: A nationally representative mortality survey. PLoS Negl. Trop. Dis. 2011, 5, e1018. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.-P. Snake-bites: Appraisal of the global situation. Bull. World Health Organ. 1998, 76, 515–524. [Google Scholar] [PubMed]
- Chippaux, J.-P. Estimate of the burden of snakebites in sub-Saharan Africa: A meta-analytic approach. Toxicon 2011, 57, 586–599. [Google Scholar] [CrossRef] [PubMed]
- Habib, A.G.; Kuznik, A.; Hamza, M.; Abdullahi, M.I.; Chedi, B.A.; Chippaux, J.-P.; Warrell, D.A. Snakebite is Under Appreciated: Appraisal of Burden from West Africa. PLoS Negl. Trop. Dis. 2015, 9. [Google Scholar] [CrossRef]
- Kasturiratne, A.; Wickremasinghe, A.R.; de Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite envenoming. Nat. Rev. Dis. Primers 2017, 3, 1–21. [Google Scholar] [CrossRef]
- Chippaux, J.-P. Epidemiology of snakebites in Europe: A systematic review of the literature. Toxicon 2012, 59, 86–99. [Google Scholar] [CrossRef]
- Chippaux, J.-P.; Saz-Parkinson, Z.; Amate Blanco, J.M. Epidemiology of snakebite in Europe: Comparison of data from the literature and case reporting. Toxicon 2013, 76, 206–213. [Google Scholar] [CrossRef]
- Harrison, R.A.; Hargreaves, A.; Wagstaff, S.C.; Faragher, B.; Lalloo, D.G. Snake envenoming: A disease of poverty. PLoS Negl. Trop. Dis. 2009, 3, e569. [Google Scholar] [CrossRef] [PubMed]
- Habib, A.G.; Abubakar, S.B.; Abubakar, I.S.; Larnyang, S.; Durfa, N.; Nasidi, A.; Yusuf, P.O.; Garnvwa, J.; Theakston, R.D.G.; Salako, L.; et al. Envenoming after carpet viper (Echis ocellatus) bite during pregnancy: Timely use of effective antivenom improves maternal and foetal outcomes. Trop. Med. Int. Health 2008, 13, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- WHO Snakebite: WHO Targets 50% Reduction in Deaths and Disabilities. Available online: https://www.who.int/news-room/detail/23-05-2019-who-launches-global-strategy-for-prevention-and-control-of-snanebite-envenoming (accessed on 23 April 2020).
- Morgenstern, D.; King, G.F. The venom optimization hypothesis revisited. Toxicon 2013, 63, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Kularatne, K.; Budagoda, S.; Maduwage, K.; Naser, K.; Kumarasiri, R.; Kularatne, S. Parallels between Russell’s viper (Daboia russelii) and hump–nosed viper (Hypnale species) bites in the central hills of Sri Lanka amidst the heavy burden of unidentified snake bites. Asian Pac. J. Trop. Med. 2011, 4, 564–567. [Google Scholar] [CrossRef]
- Kochva, E. The origin of snakes and evolution of the venom apparatus. Toxicon 1987, 25, 65–106. [Google Scholar] [CrossRef]
- Mackessy, S.P.; Baxter, L.M. Bioweapons synthesis and storage: The venom gland of front-fanged snakes. Zool. Anz. J. Comp. Zool. 2006, 245, 147–159. [Google Scholar] [CrossRef]
- Kochva, E.; Gans, C. Histology and Histochemistry of Venom Glands of Some Crotaline Snakes. Copeia 1966, 1966, 506–515. [Google Scholar] [CrossRef]
- Gans, C.; Kochva, E. The accessory gland in the venom apparatus of viperid snakes. Toxicon 1965, 3, 61–63. [Google Scholar] [CrossRef]
- Vonk, F.J.; Admiraal, J.F.; Jackson, K.; Reshef, R.; de Bakker, M.A.G.; Vanderschoot, K.; van den Berge, I.; van Atten, M.; Burgerhout, E.; Beck, A.; et al. Evolutionary origin and development of snake fangs. Nature 2008, 454, 630–633. [Google Scholar] [CrossRef]
- Kılıç, D.Ç.; Akat, E.; Arıkan, H. A histological study on venom apparatus of Montivipera xanthina (Gray, 1849) (Serpentes, Viperidae). Braz. Arch. Biol. Technol. 2016, 59. [Google Scholar] [CrossRef]
- Kerchove, C.M. Stimulation of the -adrenoceptor triggers the venom production cycle in the venom gland of Bothrops jararaca. J. Exp. Biol. 2004, 207, 411–416. [Google Scholar] [CrossRef]
- Yamanouye, N.; Britto, L.R.; Carneiro, S.M.; Markus, R.P. Control of venom production and secretion by sympathetic outflow in the snake Bothrops jararaca. J. Exp. Biol. 1997, 200, 2547–2556. [Google Scholar] [PubMed]
- Currier, R.B.; Calvete, J.J.; Sanz, L.; Harrison, R.A.; Rowley, P.D.; Wagstaff, S.C. Unusual Stability of Messenger RNA in Snake Venom Reveals Gene Expression Dynamics of Venom Replenishment. PLoS ONE 2012, 7, e41888. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, S.M.; Assakura, M.T.; Barrence, F.A.C.; Cardoso, S.R.T.; de Martins Camargo, A.C.; Sesso, A. Immunolocalization of venom metalloproteases in venom glands of adult and of newborn snakes of Bothrops jararaca. Tissue Cell 2002, 34, 381–389. [Google Scholar] [CrossRef]
- Young, B.A.; Blair, M.; Zahn, K.; Marvin, J. Mechanics of venom expulsion in Crotalus, with special reference to the role of the fang sheath. Anat. Rec. 2001, 264, 415–426. [Google Scholar] [CrossRef]
- Vonk, F.J.; Jackson, K.; Doley, R.; Madaras, F.; Mirtschin, P.J.; Vidal, N. Snake venom: From fieldwork to the clinic. BioEssays 2011, 33, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Young, B.A.; Kardong, K.V. Mechanisms controlling venom expulsion in the western diamondback rattlesnake, Crotalus atrox. J. Exp. Zool. Ecol. Genet. Physiol. 2007, 307, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Fayrer, J. Case of Rattlesnake Bite in London. Br. Med. J. 1892, 2, 728. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bannerman, W.B. Treatment of Snake-Bite with Potassium Permanganate. Ind. Med. Gaz. 1910, 45, 201. [Google Scholar]
- Fry, B. Snakebite: When the Human Touch Becomes a Bad Touch. Toxins 2018, 10, 170. [Google Scholar] [CrossRef]
- Parrish, H.M. Poisonous snakebites resulting in lack of venom poisoning. Va. Med. Mon. 1959, 86, 396–401. [Google Scholar]
- Parrish, H.M.; Goldner, J.C.; Silberg, S.L. Poisonous Snakebites Causing No Venenation. Postgrad. Med. 1966, 39, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.G.; Warrell, D.A.; Griffiths, E. Report of a WHO workshop on the standardization and control of antivenoms. Toxicon 2003, 41, 541–557. [Google Scholar] [CrossRef]
- Russell, F.E. Snake Venom Poisoning; Lippincott; Scholium International: Philadelphia, PA, USA, 1980. [Google Scholar]
- Kardong, K.V. Predatory strike behavior of the rattlesnake, Crotalus viridis oreganus. J. Comp. Psychol. 1986, 100, 304–314. [Google Scholar] [CrossRef]
- Silveira, P.V.; de A Nishioka, S. Venomous snake bite without clinical envenoming (‘dry-bite’). A neglected problem in Brazil. Trop. Geogr. Med. 1995, 47, 82–85. [Google Scholar] [PubMed]
- Walter, F.G.; Stolz, U.; Shirazi, F.; Mcnally, J. Temporal analyses of coral snakebite severity published in the American Association of Poison Control Centers’ Annual Reports from 1983 through 2007. Clin. Toxicol. 2010, 48, 72–78. [Google Scholar] [CrossRef]
- Zhang, X.; World Health Organization. WHO Traditional Medicine Strategy 2002–2005; WHO: Geneva, Switzerland, 2002. [Google Scholar]
- Russell, F.E. Snake venom poisoning in Southern California. Calif. Med. 1960, 93, 347–350. [Google Scholar]
- Campbell, C.H. The treatment of suspected venomous snake bite. Med. J. Aust. 1963, 17, 493–495. [Google Scholar] [CrossRef]
- Reid, H.A.; Thean, P.C.; Martin, W.J. Specific antivenene and prednisone in viper-bite poisoning: Controlled trial. Br. Med. J. 1963, 2, 1378–1380. [Google Scholar] [CrossRef]
- Parrish, H.M. Incidence of treated snakebites in the United States. Public Health Rep 1966, 81, 269–276. [Google Scholar] [CrossRef]
- Myint, L.; Warrell, D.A.; Phillips, R.E.; Tin, N.-S.; Tun, P.; Maung-Maung, L. Bites by Russell’s viper (Vipera russelli siamensis) in Burma: Haemostatic, vascular, and renal disturbances and response to treatment. Lancet 1985, 2, 1259–1264. [Google Scholar] [CrossRef]
- Kitchens, C.S.; Van Mierop, L.H.S. Envenomation by the Eastern Coral Snake (Micrurus fulvius fulvius): A Study of 39 Victims. JAMA 1987, 258, 1615. [Google Scholar] [CrossRef] [PubMed]
- Kouyoumdjian, J.A.; Polizelli, C. Acidentes ofidicos causados por Bothrops moojeni: Correlação do quadro clínico com o tamanho da serpente. Rev. Inst. Med. Trop. São Paulo 1989, 31, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Curry, S.C.; Horning, D.; Brady, P.; Requa, R.; Kunkel, D.B.; Vance, M.V. The legitimacy of rattlesnake bites in Central Arizona. Ann. Emerg. Med. 1989, 18, 658–663. [Google Scholar] [CrossRef]
- Tun, P.; Ba, -A.; Aye-Aye, -M.; Tin, -N.-S.; Warrell, D.A. Bites by Russell’s vipers (Daboia russelii siamensis) in Myanmar: Effect of the snake’s length and recent feeding on venom antigenaemia and severity of envenoming. Trans. R. Soc. Trop. Med. Hyg. 1991, 85, 804–808. [Google Scholar] [CrossRef]
- Tibbalis, J. Diagnosis and treatment of confirmed and suspected snake bite: Implications from an analysis of 46 paediatric cases. Med. J. Aust. 1992, 156, 270–274. [Google Scholar] [CrossRef]
- Mead, H.J.; Jelinek, G.A. Suspected snakebite in children: A study of 156 patients over 10 years. Med. J. Aust. 1996, 164, 467–470. [Google Scholar] [CrossRef]
- Milani Junior, R. Snake bites by the jararacucu (Bothrops jararacussu): Clinicopathological studies of 29 proven cases in Sao Paulo State, Brazil. QJM 1997, 90, 323–334. [Google Scholar] [CrossRef]
- Rezende, N.A.; Maia Torres, F.; Borges Dias, M.; Campolina, D.; Chavez-Olortegui, C.; Faria Santos Amaral, C. South American rattlesnake bite (Crotalus durissus sp) without envenoming: Insights on diagnosis and treatment. Toxicon 1998, 36, 2029–2032. [Google Scholar] [CrossRef]
- Tanen, D.A.; Ruha, A.-M.; Graeme, K.A.; Curry, S.C. Epidemiology and Hospital Course of Rattlesnake Envenomations Cared for at a Tertiary Referral Center in Central Arizona. Acad. Emerg. Med. 2001, 8, 177–182. [Google Scholar] [CrossRef]
- Kularatne, S.A.M. Common krait (Bungarus caeruleus) bite in Anuradhapura, Sri Lanka: A prospective clinical study, 1996–1998. Postgrad. Med. J. 2002, 78, 276–280. [Google Scholar] [CrossRef]
- Spiller, H.A.; Bosse, G.M. Prospective Study of Morbidity Associated with Snakebite Envenomation: Article. J. Toxicol. Clin. Toxicol. 2003, 41, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Bawaskar, H.S.; Bawaskar, P.H. Envenoming by the Common Krait (Bungarus caeruleus) and Asian Cobra (Naja naja): Clinical Manifestations and Their Management in a Rural Setting. Wilderness Environ. Med. 2004, 15, 257–266. [Google Scholar] [CrossRef]
- Bucaretchi, F.; Hyslop, S.; Vieira, R.J.; Toledo, A.S.; Madureira, P.R.; Capitani, E.M. de Bites by coral snakes (Micrurus spp.) in Campinas, State of São Paulo, Southeastern Brazil. Rev. Inst. Med. Trop. São Paulo 2006, 48, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Köse, R. The management of snake envenomation: Evaluation of twenty-one snake bite cases. Ulus. Travma Acil Cerrahi Derg. 2007, 13, 307–312. [Google Scholar]
- Ariaratnam, C.A.; Sheriff, M.H.R.; Theakston, R.D.G.; Warrell, D.A. Distinctive epidemiologic and clinical features of common krait (Bungarus caeruleus) bites in Sri Lanka. Am. J. Trop. Med. Hyg. 2008, 79, 458–462. [Google Scholar] [CrossRef]
- Kularatne, S.A.M.; Budagoda, B.D.S.S.; Gawarammana, I.B.; Kularatne, W.K.S. Epidemiology, clinical profile and management issues of cobra (Naja naja) bites in Sri Lanka: First authenticated case series. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 924–930. [Google Scholar] [CrossRef]
- Regional Office for South-East Asia, World Health Organization. In Guidelines for the Management of Snakebites; Warrell, D., Ed.; WHO: Geneva, Switzerland, 2016; ISBN 978-92-9022-530-0. [Google Scholar]
- Nicoleti, A.F.; de Medeiros, C.R.; Duarte, M.R.; França, F.O.D.S. Comparison of Bothropoides jararaca bites with and without envenoming treated at the Vital Brazil Hospital of the Butantan Institute, State of São Paulo, Brazil. Rev. Soc. Bras. Med. Trop. 2010, 43, 657–661. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kularatne, S.A.M.; Sivansuthan, S.; Medagedara, S.C.; Maduwage, K.; de Silva, A. Revisiting saw-scaled viper (Echis carinatus) bites in the Jaffna Peninsula of Sri Lanka: Distribution, epidemiology and clinical manifestations. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 591–597. [Google Scholar] [CrossRef]
- Spano, S.; Macias, F.; Snowden, B.; Vohra, R. Snakebite Survivors Club: Retrospective review of rattlesnake bites in Central California. Toxicon 2013, 69, 38–41. [Google Scholar] [CrossRef]
- Valenta, J.; Stach, Z.; Stříteský, M.; Michálek, P. Common viper bites in the Czech Republic—epidemiological and clinical aspects during 15 year period (1999–2013). Prague Med. Rep. 2014, 115, 120–127. [Google Scholar] [CrossRef]
- Roth, B.; Sharma, K.; Onisko, N.; Chen, T. Prospective evaluation of pain, swelling, and disability from copperhead envenomation. Clin. Toxicol. 2016, 54, 271–276. [Google Scholar] [CrossRef]
- Silva, A.; Maduwage, K.; Sedgwick, M.; Pilapitiya, S.; Weerawansa, P.; Dahanayaka, N.J.; Buckley, N.A.; Johnston, C.; Siribaddana, S.; Isbister, G.K. Neuromuscular Effects of Common Krait (Bungarus caeruleus) Envenoming in Sri Lanka. PLoS Negl. Trop. Dis. 2016, 10, e0004368. [Google Scholar] [CrossRef] [PubMed]
- Bawaskar, H.; Bawaskar, P. Diagnosis of envenomation by Russell’s and Echis carinatus viper: A clinical study at rural Maharashtra state of India. J. Fam. Med. Prim. Care 2019, 8, 1386. [Google Scholar] [CrossRef] [PubMed]
- Naik, B.S. “Dry bite” in venomous snakes: A review. Toxicon 2017, 133, 63–67. [Google Scholar] [CrossRef] [PubMed]
- White, J.; Meier, J. Handbook of Clinical Toxicology of Animal Venoms and Poisons, 1st ed.; White, J., Ed.; CRC Press: Boca Raton, FL, USA, 2017; ISBN 978-0-203-71944-2. [Google Scholar]
- Punguyire, D.; Iserson, K.V.; Stolz, U.; Apanga, S. Bedside Whole-Blood Clotting Times: Validity after Snakebites. J. Emerg. Med. 2013, 44, 663–667. [Google Scholar] [CrossRef] [PubMed]
- de Brito Sousa, J.D.; Sachett, J.A.G.; de Oliveira, S.S.; Mendonça-da-Silva, I.; Marques, H.O.; de Lacerda, M.V.G.; Fan, H.W.; Monteiro, W.M. Accuracy of the Lee–White Clotting Time Performed in the Hospital Routine to Detect Coagulopathy in Bothrops atrox Envenomation. Am. J. Trop. Med. Hyg. 2018, 98, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Bucaretchi, F.; De Capitani, E.M.; Branco, M.M.; Fernandes, L.C.R.; Hyslop, S. Coagulopathy as the main systemic manifestation after envenoming by a juvenile South American rattlesnake (Crotalus durissus terrificus): Case report. Clin. Toxicol. 2013, 51, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Thongtonyong, N.; Chinthammitr, Y. Sensitivity and specificity of 20-minute whole blood clotting test, prothrombin time, activated partial thromboplastin time tests in diagnosis of defibrination following Malayan pit viper envenoming. Toxicon 2020, 185, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, E.S. Ácidos Nucleicos Circulantes Como Biomarcadores de Gravidade em Acidentes Bothrópicos. Master’s Thesis, Universidade do estado do Amazonas, Manaus, Brazil, 2020. [Google Scholar]
- Ibiapina, H.N.S.; Costa, A.G.; Sachett, J.A.G.; Silva, I.M.; Tarragô, A.M.; Neves, J.C.F.; Kerr, M.W.A.; Santana, M.F.; Martins-Filho, O.A.; Lacerda, M.V.G.; et al. An Immunological Stairway to Severe Tissue Complication Assembly in Bothrops atrox Snakebites. Front. Immunol. 2019, 10, 1882. [Google Scholar] [CrossRef]
- Castellanos, I.A.M.W.; Ibiapina, H.N.S.; Sachett, J.A.G.; Sartim, M.A.; Silva, I.M.; Oliveira, S.S.; Tarrago, A.M.; Moura-da-Silva, A.M.; Lacerda, M.V.G.; de Lima Ferreira, L.C.; et al. Correlating fibrinogen consumption and profiles of inflammatory molecules in human envenomation’s by Bothrops atrox in the Brazilian Amazon. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Brunda, G.; Sashidhar, R.B. Epidemiological profile of snake-bite cases from Andhra Pradesh using immunoanalytical approach. Indian J. Med. Res. 2007, 125, 661–668. [Google Scholar] [PubMed]
- Brunda, G.; Sashidhar, R.B.; Sarin, R.K. Use of egg yolk antibody (IgY) as an immunoanalytical tool in the detection of Indian cobra (Naja naja naja) venom in biological samples of forensic origin. Toxicon 2006, 48, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.P.; Yu, Y.J.; Hung, D.Z. Sandwich enzyme-linked immunosorbent assay for Taiwan cobra venom. Vet. Hum. Toxicol. 2002, 44, 200–204. [Google Scholar] [PubMed]
- Liu, C.-C.; Yu, J.-S.; Wang, P.-J.; Hsiao, Y.-C.; Liu, C.-H.; Chen, Y.-C.; Lai, P.-F.; Hsu, C.-P.; Fann, W.-C.; Lin, C.-C. Development of sandwich ELISA and lateral flow strip assays for diagnosing clinically significant snakebite in Taiwan. PLoS Negl. Trop. Dis. 2018, 12, e0007014. [Google Scholar] [CrossRef]
- Núñez Rangel, V.; Fernández Culma, M.; Rey-Suárez, P.; Pereañez, J. Development of a sensitive enzyme immunoassay (ELISA) for specific identification of Lachesis acrochorda venom. J. Venom. Anim. Toxins Incline. Trop. Dis 2012, 18, 173–179. [Google Scholar] [CrossRef]
- Selvanayagam, Z.E.; Gnanavendhan, S.G.; Ganesh, K.A.; Rajagopal, D.; Subba Rao, P.V. ELISA for the detection of venoms from four medically important snakes of India. Toxicon 1999, 37, 757–770. [Google Scholar] [CrossRef]
- Shaikh, I.K.; Dixit, P.P.; Pawade, B.S.; Waykar, I.G. Development of dot-ELISA for the detection of venoms of major Indian venomous snakes. Toxicon 2017, 139, 66–73. [Google Scholar] [CrossRef]
- Theakston, R.D.; Lloyd-Jones, M.J.; Reid, H.A. Micro-ELISA for detecting and assaying snake venom and venom-antibody. Lancet 1977, 2, 639–641. [Google Scholar] [CrossRef]
- Hung, D.Z.; Lin, J.H.; Mo, J.F.; Huang, C.F.; Liau, M.Y. Rapid diagnosis of Naja atra snakebites. Clin. Toxicol. 2014, 52, 187–191. [Google Scholar] [CrossRef]
- Pawade, B.S.; Salvi, N.C.; Shaikh, I.K.; Waghmare, A.B.; Jadhav, N.D.; Wagh, V.B.; Pawade, A.S.; Waykar, I.G.; Potnis-Lele, M. Rapid and selective detection of experimental snake envenomation—Use of gold nanoparticle based lateral flow assay. Toxicon 2016, 119, 299–306. [Google Scholar] [CrossRef]
- de Faria, R.A.D.; Lins, V. de F.C.; Nappi, G.U.; Matencio, T.; Heneine, L.G.D. Development of an Impedimetric Immunosensor for Specific Detection of Snake Venom. BioNanoScinece 2018, 8, 988–996. [Google Scholar] [CrossRef]
- Chandler, H.M.; Hurrell, J.G. A new enzyme immunoassay system suitable for field use and its application in a snake venom detection kit. Clin. Chim. Acta 1982, 121, 225–230. [Google Scholar] [CrossRef]
- Li, Y.L. Natural latex agglutination inhibition test-a method for rapid identification of the specific venom in snakebite. Zhonghua Yi Xue Za Zhi 1982, 62, 709–711. [Google Scholar] [PubMed]
- Lión, V.; Rojas, A.; Rodríguez-Acosta, A. Design of an agglutination latex test to detect venoms from Viperidae snakes in patients’ serum. Rev. Cubana Med. Trop. 2004, 56, 13–20. [Google Scholar]
- Sutherland, S.K.; Couter, A.R.; Broad, A.J. Human snake bite victims: The successful detection of circulating snake venom by radiommunoassay. Med. J. Aust. 1975, 1, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Kuch, U.; Höde, P.; Bruhse, L.; Pandey, D.P.; Ghimire, A.; Chappuis, F.; Alirol, E. Use of molecular diagnostic tools for the identification of species responsible for snakebite in Nepal: A pilot study. PLoS Negl. Trop. Dis. 2016, 10, e0004620. [Google Scholar] [CrossRef] [PubMed]
- Suntrarachun, S.; Pakmanee, N.; Tirawatnapong, T.; Chanhome, L.; Sitprija, V. Development of a polymerase chain reaction to distinguish monocellate cobra (Naja khouthia) bites from other common Thai snake species, using both venom extracts and bite-site swabs. Toxicon 2001, 39, 1087–1090. [Google Scholar] [CrossRef]
- Supikamolseni, A.; Ngaoburanawit, N.; Sumontha, M.; Chanhome, L.; Suntrarachun, S.; Peyachoknagul, S.; Srikulnath, K. Molecular barcoding of venomous snakes and species-specific multiplex PCR assay to identify snake groups for which antivenom is available in Thailand. Genet. Mol. Res. 2015, 14, 13981–13997. [Google Scholar] [CrossRef]
- Maduwage, K.; O’Leary, M.A.; Isbister, G.K. Diagnosis of snake envenomation using a simple phospholipase A2 assay. Sci. Rep. 2015, 4, 4827. [Google Scholar] [CrossRef]
- Steuten, J.; Winkel, K.; Carroll, T.; Williamson, N.A.; Ignjatovic, V.; Fung, K.; Purcell, A.W.; Fry, B.G. The molecular basis of cross-reactivity in the Australian Snake Venom Detection Kit (SVDK). Toxicon 2007, 50, 1041–1052. [Google Scholar] [CrossRef]
- Nimorakiotakis, V.B.; Winkel, K.D. Prospective assessment of the false positive rate of the Australian snake venom detection kit in healthy human samples. Toxicon 2016, 111, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.I.; Ryan, N.M.; Page, C.B.; Buckley, N.A.; Brown, S.G.; O’Leary, M.A.; Isbister, G.K. The Australian Snakebite Project, 2005–2015 (ASP-20). Med. J. Aust. 2017, 207, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A. Observation of snakebite victims: Is twelve hours still necessary? Emerg. Med. Australas. 2003, 15, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Churchman, A.; O’Leary, M.A.; Buckley, N.A.; Page, C.B.; Tankel, A.; Gavaghan, C.; Holdgate, A.; Brown, S.G.A.; Isbister, G.K. Clinical effects of red-bellied black snake (Pseudechis porphyriacus ) envenoming and correlation with venom concentrations: Australian Snakebite Project (ASP-11). Med. J. Aust. 2010, 193, 696–700. [Google Scholar] [CrossRef] [PubMed]
- Nascimento da Costa, T.; Mota-da-Silva, A.; Colombini, M.; Moura-da-Silva, A.M.; Medeiros de Souza, R.; Monteiro, W.M.; Bernarde, P.S. Relationship between snake size and clinical, epidemiological and laboratory aspects of Bothrops atrox snakebites in the Western Brazilian Amazon. Toxicon 2020, 186, 160–167. [Google Scholar] [CrossRef]
- Bernal, J.C.C.; Bisneto, P.F.; Pereira, J.P.T.; Ibiapina, H.N.D.S.; Sarraff, L.K.S.; Monteiro-Júnior, C.; da Silva Pereira, H.; Santos, B.; de Moura, V.M.; de Oliveira, S.S.; et al. “Bad things come in small packages”: Predicting venom-induced coagulopathy in Bothrops atrox bites using snake ontogenetic parameters. Clin. Toxicol. 2020, 58, 388–396. [Google Scholar] [CrossRef]
- Giannotti, K.C.; Sesso, A.; Grego, K.F.; Fernandes, W.; Cardoso, R.P.; Camargo, G.G.; Carneiro, S.M. Viperid venom glands with defective venom production. Morphological study. Toxicon 2013, 70, 32–43. [Google Scholar] [CrossRef]
- Kardong, K.V. The predatory strike of the rattlesnake: When things go amiss. Copeia 1986, 1986, 816. [Google Scholar] [CrossRef]
- Hayes, W.; Herbert, S.; Rehling, G.; Gennaro, J. Factors that influence venom expenditure in viperids and other snake species during predatory and defensive contexts. In Biology of the Vipers; Eagle Mountain Publishing: Eagle Mountain, UT, USA, 2002; pp. 207–233. [Google Scholar]
- Young, B.A.; Lee, C.E.; Daley, K.M. Do Snakes Meter Venom? BioScience 2002, 52, 1121. [Google Scholar] [CrossRef]
- Schaeffer, R.C.; Bernick, S.; Rosenquist, T.H.; Russell, F.E. The histochemistry of the venom glands of the rattlesnake Crotalus viridis helleri—II: Monoamine oxidase, acid and alkaline phosphatase. Toxicon 1972, 10, 295–297. [Google Scholar] [CrossRef]
- Chiszar, D.; Radcliffe, C.W.; O’Connell, B.; Smith, H.M. Strike-induced chemosensory searching in rattlesnakes (Crotalus enyo) as a function of disturbance prior to presentation of prey. Trans. Kans. Acad. Sci. 1980, 83, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Klauber, L.M. Rattlesnakes: Their Habits, Life Histories, and Influence on Mankind, Abridged Edition; University of California Press: Berkeley, CA, USA, 1982; ISBN 978-0-520-04039-7. [Google Scholar]
- Herbert, S.S.; Hayes, W.K. Denim clothing reduces venom expenditure by rattlesnakes striking defensively at model human limbs. Ann. Emerg. Med. 2009, 54, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Daniel, Z.-M.; Fox, E.G.P.; Monteiro, A.P.; Gama, D.; Poublan, L.E.; de Araujo, A.F.; Araujo, M.F.C.; Atella, G.C.; Machado, E.A.; Diaz, B.L. The allergic response mediated by fire ant venom proteins. Sci. Rep. 2018, 8, 1–10. [Google Scholar]
- Leon, G.; Sanchez, L.; Hernandez, A.; Villalta, M.; Herrera, M.; Segura, A.; Estrada, R.; Maria Gutierrez, J. Immune response towards snake venoms. IADT 2011, 10, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.G.; Reid, H.A.; Iddon, D.; Larrick, J.W. Protective effect of snake venom antibodies in sera of previous snake bite victims. Ann. Trop. Med. Parasitol. 1983, 77, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.; Reid, H.A.; Larrick, J.W.; Kaplan, J.; Yost, J.A. Snake venom antibodies in Ecuadorian Indians. J. Trop. Med. Hyg. 1981, 84, 199–202. [Google Scholar] [PubMed]
- Chippaux, J.P.; Theakston, R.D. Epidemiological studies of snake bite in French Guiana. Ann. Trop. Med. Parasitol. 1987, 81, 301–304. [Google Scholar] [CrossRef] [PubMed]
- New, R.R.; Theakston, R.D.; Zumbuhl, O.; Iddon, D.; Friend, J. Immunization against snake venoms. N. Engl. J. Med. 1984, 311, 56–57. [Google Scholar] [CrossRef]
- Pucca, M.B.; Peigneur, S.; Cologna, C.T.; Cerni, F.A.; Zoccal, K.F.; de CF Bordon, K.; Faccioli, L.H.; Tytgat, J.; Arantes, E.C. Electrophysiological characterization of the first Tityus serrulatus alpha-like toxin, Ts5: Evidence of a pro-inflammatory toxin on macrophages. Biochimie 2015, 115, 8–16. [Google Scholar] [CrossRef]
- Isbister, G.K.; Halkidis, L.; O’Leary, M.A.; Whitaker, R.; Cullen, P.; Mulcahy, R.; Bonnin, R.; Brown, S.G.A. Human anti-snake venom IgG antibodies in a previously bitten snake-handler, but no protection against local envenoming. Toxicon 2010, 55, 646–649. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Gutiérrez, J.M.; Lohse, B.; Rasmussen, A.R.; Fernández, J.; Milbo, C.; Lomonte, B. Snake venomics of monocled cobra (Naja kaouthia) and investigation of human IgG response against venom toxins. Toxicon 2015, 99, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Engmark, M.; Clouser, C.; Timberlake, S.; Vigneault, F.; Gutiérrez, J.M.; Lomonte, B. Exploration of immunoglobulin transcriptomes from mice immunized with three-finger toxins and phospholipases A 2 from the Central American coral snake, Micrurus nigrocinctus. PeerJ 2017, 5, e2924. [Google Scholar] [CrossRef]
- da Silva, A.M.; da Graça Mendes, V.K.; Monteiro, W.M.; Bernarde, P.S. Non-venomous snakebites in the Western Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 2019, 52, e20190120. [Google Scholar] [CrossRef]
- Laustsen, A.H.; Gutiérrez, J.M.; Knudsen, C.; Johansen, K.H.; Bermúdez-Méndez, E.; Cerni, F.A.; Jürgensen, J.A.; Ledsgaard, L.; Martos-Esteban, A.; Øhlenschlæger, M.; et al. Pros and cons of different therapeutic antibody formats for recombinant antivenom development. Toxicon 2018, 146, 151–175. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, J.M.; Oliveira, I.S.; Ferreira, I.G.; Alexandre-Silva, G.M.; Cerni, F.A.; Zottich, U.; Pucca, M.B. Fatal Rattlesnake Envenomation in Northernmost Brazilian Amazon: A Case Report and Literature Overview. Rep. Med. Cases Images Videos 2020, 3, 9. [Google Scholar] [CrossRef]
- Peterson, M.E. Snake Bite: Coral Snakes. Clin. Tech. Small Anim. Pract. 2006, 21, 183–186. [Google Scholar] [CrossRef]
- Bucaretchi, F.; De Capitani, E.M.; Vieira, R.J.; Rodrigues, C.K.; Zannin, M.; Da Silva, N.J.; Casais-e-Silva, L.L.; Hyslop, S. Coral snake bites (Micrurus spp.) in Brazil: A review of literature reports. Clin. Toxicol. 2016, 54, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H. Recombinant Antivenoms. Ph.D. Thesis, University of Copenhagen, Copenhagen, Denmark, 2016. [Google Scholar]
- Kini, R.M.; Sidhu, S.S.; Laustsen, A.H. Biosynthetic oligoclonal antivenom (BOA) for snakebite and next-generation treatments for snakebite victims. Toxins 2018, 10, 534. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; León, G.; Lomonte, B. Pharmacokinetic-Pharmacodynamic Relationships of Immunoglobulin Therapy for Envenomation. Clin. Pharmacokinet. 2003, 42, 721–741. [Google Scholar] [CrossRef]
| Reference | Dry Bite Incidence (%) | Location of Snakebite | Data Collection Period # | Snake Species Involved | Criteria for Dry Bite Diagnosis |
|---|---|---|---|---|---|
| Silveira and Nishioka, 1995 [32] | 13/40 (32.5%) | Brazil | 1992–1994 | Lance-headed viper and rattlesnakes | No clinical or laboratory evidence of local or systemic envenoming. |
| Russell, 1960 [40] | 5/22 (22%) | USA | Not reported | Pacific rattlesnakes | No local or systemic signs and symptoms, No lab abnormalities, Presence of fang marks |
| Campbell, 1963 [41] | 29/152 (19%) | Papua New Guinea | 1960–1962 | Oxyuranus sp. Acanthophis sp. Pseudechis papuanus | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Reid et al., 1963 [42] | 107/212 (50%) | Malaya | 1960-1961 | Calloselasma rhodostoma | Minimal or no local signs and symptoms, Snake identified |
| Parrish et al., 1966 [33] | 335/1,315 (25%) | USA | 1958–1959 | Crotalus sp. Agkistrodon piscivorus Agkistrodon contortrix Coral snakes | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Parrish, 1966 [43] | 667/2,433 (27%) | USA | 1958–1959 | Crotalus sp. Agkistrodon sp. Sistrurus sp. Micrurus sp. | No local or systemic signs and symptoms, Presence of fang marks |
| Myint-Lwin & Warrell, 1985 [44] | 34/123 (27%) | Myanmar | 1983–1985 | Daboia siamensis Trimeresurus erythrurus Naja kaouthia | No local or systemic signs and symptoms, Snake identified |
| Kitchens & Mierop, 1987 [45] | 4/20 (20%) | USA | 1975–1986 | Micrurus fulvius | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Kouyoumdjian and Polizelli, 1989 [46] | 1/22 (4%) | Brazil | 1986–1987 | Bothrops moojeni | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy), Snake identified |
| Curry et al., 1989 [47] | 15/146 (10%) | USA | 1984–1986 | Rattlesnake | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy), Snake identified |
| Tun-Pe et al., 1991 [48] | 91/234 (38%) | Myanmar | 1984–1988 | Daboia siamensis | No local or systemic signs and symptoms, Snake identified |
| Tibballs, 1992 [49] | 10/46 (22%) | Australia | 1979–1990 | Pseudonaja textilis Notechis scutatus Austrelaps superbus Pseudechis porphyriacus | No local or systemic signs and symptoms, Presence of fang marks, No lab abnormalities (coagulopathy), Venom not detected (blood, urine, or washings from the suspected bite site) |
| Mead and Jelinek, 1996 [50] | 32/156 (20%) | Australia | 1984–1993 | Pseudonaja sp. Notechis sp. Pseudechis sp. | No local or systemic signs and symptoms, Presence of fang marks, No lab abnormalities (coagulopathy), Snake identified |
| Milani et al., 1997 [51] | 1/29 (3%) | Brazil | 1975–1995 | Bothrops jararacussu | No local or systemic signs and symptoms, Snake identified |
| de Rezende et al., 1998 [52] | 5/41 (12%) | Brazil | 1994–1996 | C. durissus | No local or systemic signs and symptoms, Presence of fang marks, No lab abnormalities, Venom not detected (plasma) Snake identified |
| Tanen et al., 2001 [53] | 7/236 (3%) | USA | 1994–2000 | Crotalus sp. | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy + hematological), Snake identified |
| Kularatne, 2002 [54] | 22/210 (10%) | Sri Lanka | 1996–1998 | Bungarus caeruleus | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Spiller & Bosse, 2003 [55] | 31/128 (24%) | USA | 2001 | Agkistrodon contortrix Crotalus horridus Agkistrodon piscivorus | No local or systemic signs and symptoms, Presence of fang marks, No lab abnormalities (coagulopathy) |
| Bawaskar and Bawaskar, 2004 [56] | 1/29 (3%) | India | 2001–2003 | Bungarus caeruleus Naja naja | No local or systemic signs and symptoms, Snake identified |
| Bucaretchi et al., 2006 [57] | 1/11 (9%) | Brazil | 1984–2004 | Micrurus lemniscatus | No local or systemic signs and symptoms, Snake identified |
| Köse, 2007 [58] | 4/21 (19%) | Turkey | 2004–2005 | Macrovipera lebetinus | No local or systemic signs and symptoms, Presence of fang marks |
| Ariaratnam et al., 2008 [59] | 4/88 (4%) | Sri Lanka | 1993–1997 | Bungarus ceylonicus | No local or systemic signs and symptoms, Snake identified |
| Kularatne et al., 2009 [60] | 5/20 (20%) | Sri Lanka | 1995–1998; 2002–2007 | Naja naja | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Walter et al., 2010 [38] | 117/838 (13%) | USA | 1983–2007 | Micrurus sp. | No local or systemic signs and symptoms, Snake identified |
| Warrell, 2010 [61] | 5–50% | South East-Asia Countries | Not informed | Calloselasma sp. Daboia russelii Echis sp. | No local or systemic signs and symptoms |
| Nicoleti et al., 2010 [62] | 19/792 (2%) | Brazil | 1990–2004 | Bothrops jararaca | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy), Snake identified |
| Kularatne et al., 2011 [63] | 2/26 (8%) | Sri Lanka | 2009–2010 | Echis carinatus | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy), Snake identified |
| Kularatne et al., 2011 [15] | 1/19 (5%) 2/36 (5%) | Sri Lanka (Central hills) | 2006–2008 | Daboia russelii Hypnale species | No local or systemic signs and symptoms, Snake identified |
| Spano et al., 2013 [64] | 5/46 (10%) | USA | 2000–2010 | Rattlesnake | No local or systemic signs and symptoms, No lab abnormalities (coagulopathy) |
| Valenta et al., 2014 [65] | 51/191 (26%) | Czech Republic | 1999–2013 | Vipera berus | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Roth et al., 2016 [66] | 5/104 (4%) | USA | 2009–2011 | Agkistrodon sp. | No local or systemic signs and symptoms, Presence of fang marks, Snake identified |
| Silva et al., 2016 [67] | 8/33 (24%) | Sri Lanka | 2014–not informed | Bungarus caeruleus | No local or systemic signs and symptoms, |
| Bawaskar and Bawaskar, 2019 [68] | 1/77 (1.75%) | India | Not reported | Echis carinatus Daboia russelii | No local or systemic signs and symptoms, Presence of fang marks, No lab abnormalities (coagulopathy), Snake identified |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pucca, M.B.; Knudsen, C.; S. Oliveira, I.; Rimbault, C.; A. Cerni, F.; Wen, F.H.; Sachett, J.; Sartim, M.A.; Laustsen, A.H.; Monteiro, W.M. Current Knowledge on Snake Dry Bites. Toxins 2020, 12, 668. https://doi.org/10.3390/toxins12110668
Pucca MB, Knudsen C, S. Oliveira I, Rimbault C, A. Cerni F, Wen FH, Sachett J, Sartim MA, Laustsen AH, Monteiro WM. Current Knowledge on Snake Dry Bites. Toxins. 2020; 12(11):668. https://doi.org/10.3390/toxins12110668
Chicago/Turabian StylePucca, Manuela B., Cecilie Knudsen, Isadora S. Oliveira, Charlotte Rimbault, Felipe A. Cerni, Fan Hui Wen, Jacqueline Sachett, Marco A. Sartim, Andreas H. Laustsen, and Wuelton M. Monteiro. 2020. "Current Knowledge on Snake Dry Bites" Toxins 12, no. 11: 668. https://doi.org/10.3390/toxins12110668
APA StylePucca, M. B., Knudsen, C., S. Oliveira, I., Rimbault, C., A. Cerni, F., Wen, F. H., Sachett, J., Sartim, M. A., Laustsen, A. H., & Monteiro, W. M. (2020). Current Knowledge on Snake Dry Bites. Toxins, 12(11), 668. https://doi.org/10.3390/toxins12110668











