Physiological Stress Integrates Resistance to Rattlesnake Venom and the Onset of Risky Foraging in California Ground Squirrels
Abstract
1. Introduction
2. Results
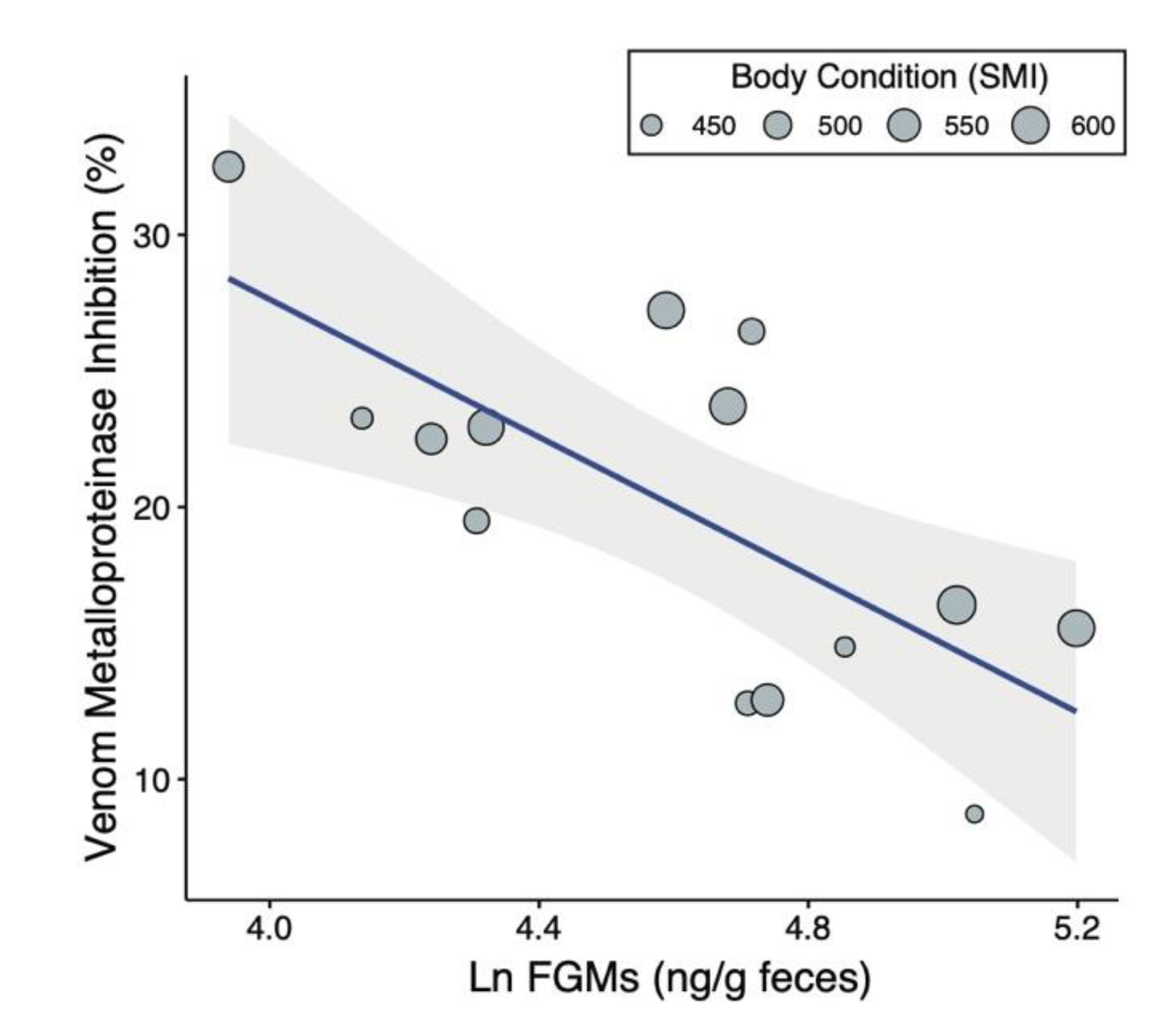
2.1. Predictors of Venom Resistance
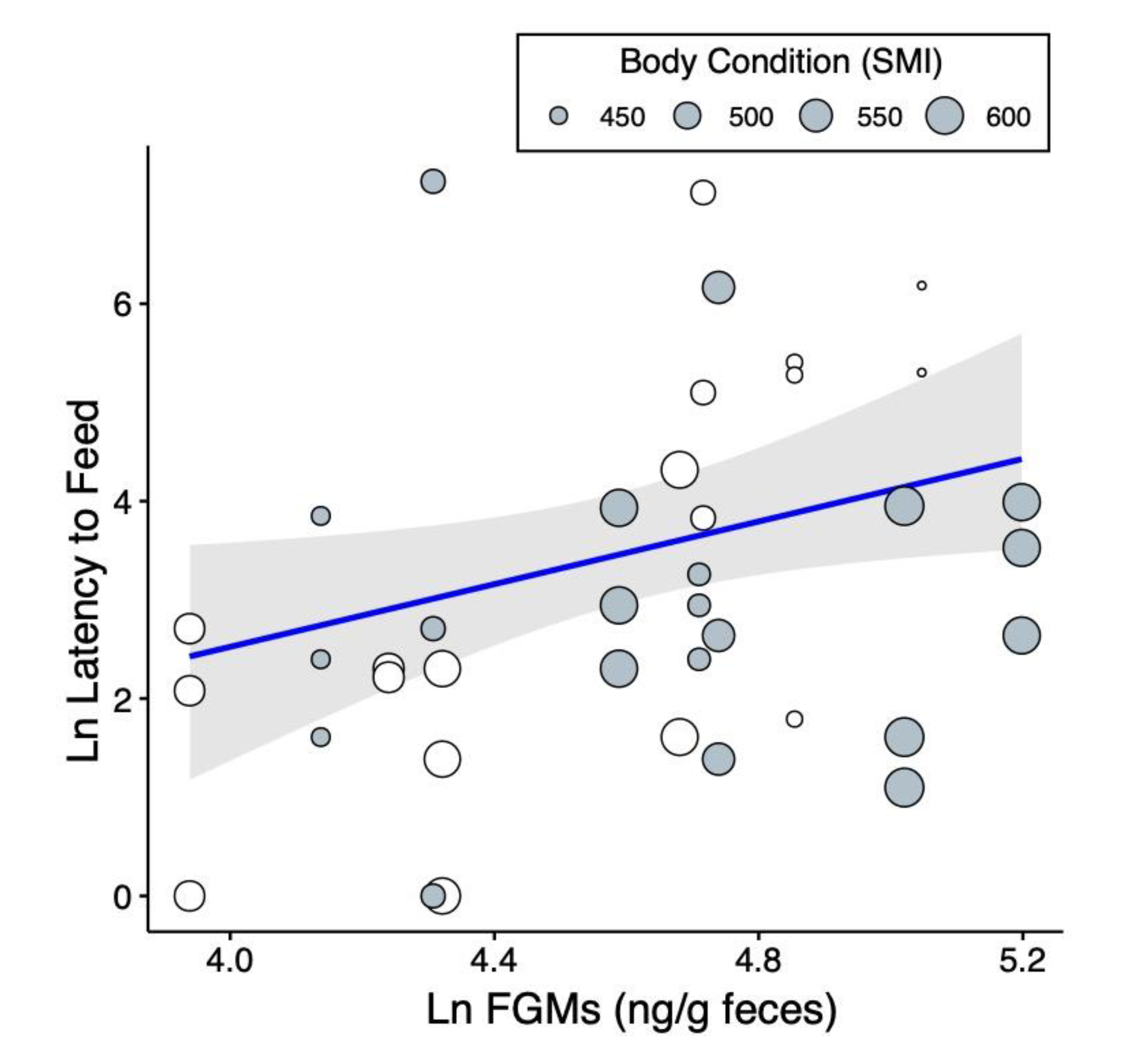
2.2. Predictors of Risk-Taking Behaviors
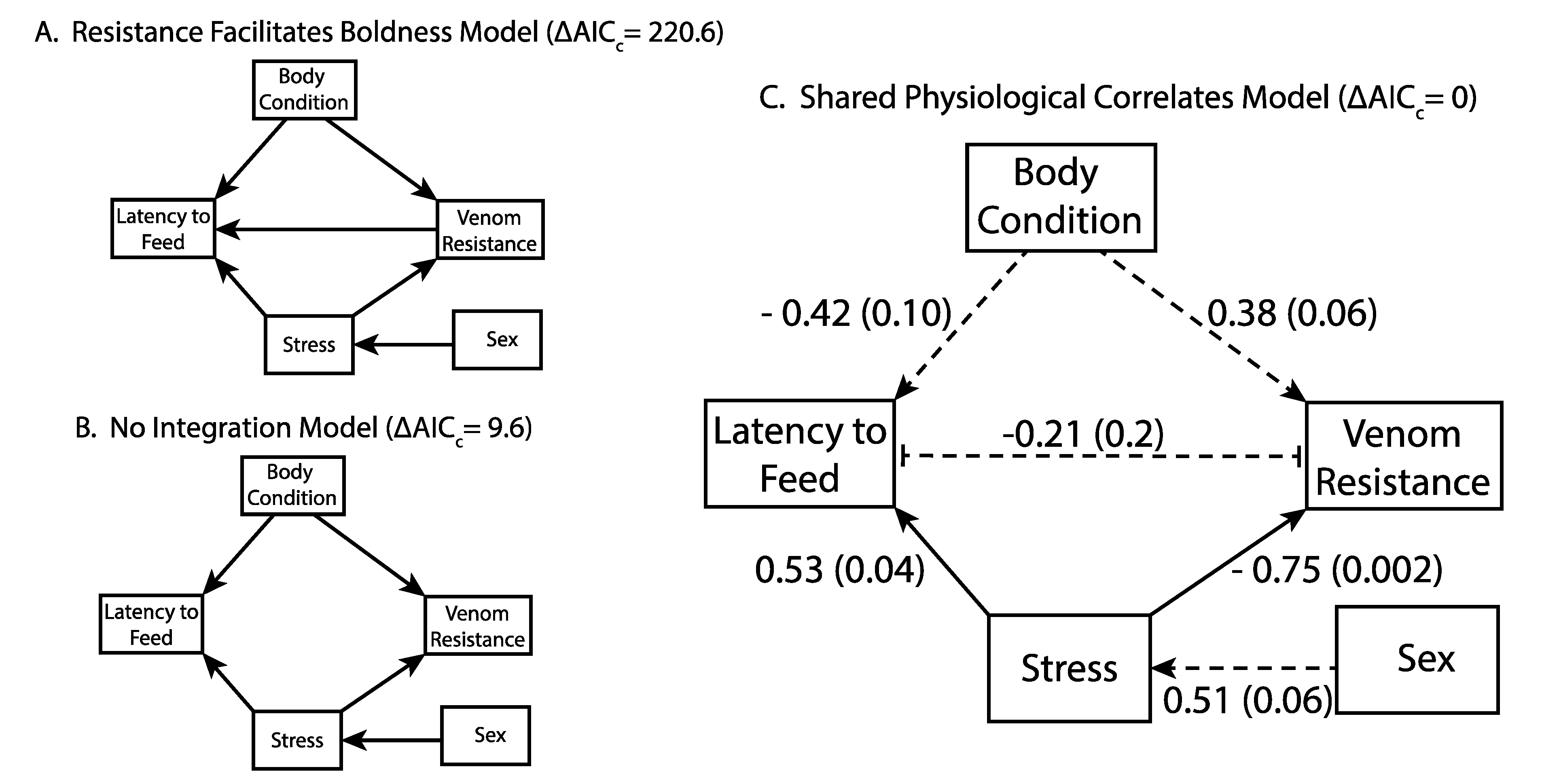
2.3. Integration of Resistance and Behavior
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Study Site and Squirrel Trapping
5.2. Squirrel Risk-Taking Behaviors
5.3. Fecal Glucocorticoid Metabolites
5.4. Serum Inhibition of Venom Acitivty
5.5. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barbour, M.A.; Clark, R.W. Ground squirrel tail-flag displays alter both predatory strike and ambush site selection behaviours of rattlesnakes. Proc. R. Soc. B Biol. Sci. 2012, 279, 3827–3833. [Google Scholar] [CrossRef] [PubMed]
- Arnold, S.J. Behavioral variation in natural populations. I. Phenotypic, genetic and environmental correlations between chemoreceptive responses to prey in the garter snake, Thamnophis elegans. Evolution 1981, 35, 489–509. [Google Scholar] [CrossRef] [PubMed]
- Putman, B.; Coss, R.; Clark, R. The ontogeny of antipredator behavior: Age differences in California ground squirrels (Otospermophilus beecheyi) at multiple stages of rattlesnake encounters. Behav. Ecol. Sociobiol. 2015, 1–11. [Google Scholar] [CrossRef]
- Toju, H.; Sota, T. Imbalance of predator and prey armament: Geographic clines in phenotypic interface and natural selection. Am. Nat. 2006, 167, 105–117. [Google Scholar] [CrossRef] [PubMed]
- West, K.; Cohen, A.; Baron, M. Morphology and behavior of crabs and gastropods from Lake Tanganyika, Africa: Implications for lacustrine predator-prey coevolution. Evolution 1991, 45, 589–607. [Google Scholar] [CrossRef]
- Feldman, C.R.; Brodie, E.D.; Brodie, E.D.; Pfrender, M.E. The evolutionary origins of beneficial alleles during the repeated adaptation of garter snakes to deadly prey. Proc. Natl. Acad. Sci. USA 2009, 106, 13415–13420. [Google Scholar] [CrossRef]
- Tarvin, R.D.; Borghese, C.M.; Sachs, W.; Santos, J.C.; Lu, Y.; O’Connell, L.A.; Cannatella, D.C.; Harris, R.A.; Zakon, H.H. Interacting amino acid replacements allow poison frogs to evolve epibatidine resistance. Science 2017, 357, 1261–1266. [Google Scholar] [CrossRef]
- Gilman, R.T.; Nuismer, S.L.; Jhwueng, D.-C. Coevolution in multidimensional trait space favours escape from parasites and pathogens. Nature 2012, 483, 328–330. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Pélabon, C.; Bolstad, G.H.; Hansen, T.F. Integrated phenotypes: Understanding trait covariation in plants and animals. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369. [Google Scholar] [CrossRef]
- Sih, A.; Bell, A.; Johnson, J.C.; Robert, Z. Behavioral syndromes: An integrative overview. Q. Rev. Biol. 2004, 79, 241–277. [Google Scholar] [CrossRef]
- Penna, A.; Melo, D.; Bernardi, S.; Oyarzabal, M.I.; Marroig, G. The evolution of phenotypic integration: How directional selection reshapes covariation in mice. Evolution 2017, 71, 2370–2380. [Google Scholar] [CrossRef] [PubMed]
- Pigliucci, M. Phenotypic integration: Studying the ecology and evolution of complex phenotypes. Ecol. Lett. 2003, 6, 265–272. [Google Scholar] [CrossRef]
- Ahlgren, J.; Chapman, B.B.; Nilsson, P.A.; Brönmark, C. Individual boldness is linked to protective shell shape in aquatic snails. Biol. Lett. 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, L.T.; McAdam, A.G.; Hipsley, C.A.; Sinervo, B.R. Frequency-dependent and correlational selection pressures have conflicting consequences for assortative mating in a color-polymorphic lizard, Uta stansburiana. Am. Nat. 2014, 184, 188–197. [Google Scholar] [CrossRef]
- Brodie III, E.D. Correlational selection for color pattern and antipredator behavior in the garter snake Thamnophis ordinoides. Evolution 1992, 46, 1284. [Google Scholar] [CrossRef]
- Creel, S. The control of risk hypothesis: Reactive vs. proactive antipredator responses and stress-mediated vs. food-mediated costs of response. Ecol. Lett. 2018, 21, 947–956. [Google Scholar] [CrossRef]
- Lofeu, L.; Brandt, R.; Kohlsdorf, T. Phenotypic integration mediated by hormones: Associations among digit ratios, body size and testosterone during tadpole development. BMC Evol. Biol. 2017, 17, 1–12. [Google Scholar] [CrossRef]
- Lipshutz, S.E.; George, E.M.; Bentz, A.B.; Rosvall, K.A. Evaluating testosterone as a phenotypic integrator: From tissues to individuals to species. Mol. Cell. Endocrinol. 2019, 496, 110531. [Google Scholar] [CrossRef]
- Taff, C.C.; Vitousek, M.N. Endocrine flexibility: Optimizing phenotypes in a dynamic world? Trends Ecol. Evol. 2016, 31, 476–488. [Google Scholar] [CrossRef]
- Lema, S.C. Hormones and phenotypic plasticity in an ecological context: Linking physiological mechanisms to evolutionary processes. Integr. Comp. Biol. 2014, 54, 850–863. [Google Scholar] [CrossRef]
- Ketterson, E.D.; Atwell, J.W.; McGlothlin, J.W. Phenotypic integration and independence: Hormones, performance, and response to environmental change. Integr. Comp. Biol. 2009, 49, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Hau, M.; Goymann, W. Endocrine mechanisms, behavioral phenotypes and plasticity: Known relationships and open questions. Front. Zool. 2015, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Creel, S.; Dantzer, B.; Goymann, W.; Rubenstein, D.R. The ecology of stress: Effects of the social environment. Funct. Ecol. 2013, 27, 66–80. [Google Scholar] [CrossRef]
- Wingfield, J.C. Ecological processes and the ecology of stress: The impacts of abiotic environmental factors. Funct. Ecol. 2013, 27, 37–44. [Google Scholar] [CrossRef]
- McCormick, G.L.; Robbins, T.R.; Cavigelli, S.A.; Langkilde, T. Population history with invasive predators predicts innate immune function response to early-life glucocorticoid exposure in lizards. J. Exp. Biol. 2019, 222. [Google Scholar] [CrossRef]
- French, S.S.; Denardo, D.F.; Greives, T.J.; Strand, C.R.; Demas, G.E. Human disturbance alters endocrine and immune responses in the Galapagos marine iguana (Amblyrhynchus cristatus). Horm. Behav. 2010, 58, 792–799. [Google Scholar] [CrossRef]
- Sinclair, A.R.E.; Arcese, P. Population consequences of predation-sensitive foraging: The Serengeti wildebeest. Ecology 1995, 76, 882–891. [Google Scholar] [CrossRef]
- Werner, E.E.; Gilliam, J.F.; Hall, D.J.; Mittelbach, G.G. An experimental test of the effects of predation risk on habitat use in fish. Ecology 1983, 64, 1540–1548. [Google Scholar] [CrossRef]
- Lima, S.L.; Dill, L.M. Behavioral decisions made under the risk of predation: A review and prospectus. Can. J. Zool. 1990, 68, 619–640. [Google Scholar] [CrossRef]
- Boonstra, R.; Hik, D.; Singleton, G.R.; Tinnikov, A. The impact of predator-induced stress on the snowshoe hare cycle. Ecol. Monogr. 1998, 68, 371. [Google Scholar] [CrossRef]
- Clinchy, M.; Zanette, L.; Boonstra, R.; Wingfield, J.C.; Smith, J.N.M. Balancing food and predator pressure induces chronic stress in songbirds. Proc. R. Soc. London. Ser. B Biol. Sci. 2004, 271, 2473–2479. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Horm. Behav. 2003, 43, 2–15. [Google Scholar] [CrossRef]
- Korte, S.M.; Koolhaas, J.M.; Wingfield, J.C.; McEwen, B.S. The Darwinian concept of stress: Benefits of allostasis and costs of allostatic load and the trade-offs in health and disease. Neurosci. Biobehav. Rev. 2005, 29, 3–38. [Google Scholar] [CrossRef]
- Bonier, F.; Martin, P.R.; Moore, I.T.; Wingfield, J.C. Do baseline glucocorticoids predict fitness? Trends Ecol. Evol. 2009, 24, 634–642. [Google Scholar] [CrossRef]
- Casewell, N.R.; Wüster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: The evolutionary novelty of venoms. Trends Ecol. Evol. 2012, 28, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.H.; Rowe, M.P. Risk assessment by grasshopper mice (Onychomys spp.) feeding on neurotoxic prey (Centruroides spp.). Anim. Behav. 2006, 71, 725–734. [Google Scholar] [CrossRef]
- Putman, B.J.; Clark, R.W. The fear of unseen predators: Ground squirrel tail flagging in the absence of snakes signals vigilance. Behav. Ecol. 2015, 26, 185–193. [Google Scholar] [CrossRef]
- Drabeck, D.H.; Dean, A.M.; Jansa, S.A. Why the honey badger don’t care: Convergent evolution of venom-targeted nicotinic acetylcholine receptors in mammals that survive venomous snake bites. Toxicon 2015, 99, 68–72. [Google Scholar] [CrossRef]
- Poran, N.S.; Coss, R.G.; Benjamini, E. Resistance of California ground squirrels (Spermophilus beecheyi) to the venom of the northern Pacific rattlesnake (Crotalus viridis oreganus): A study of adaptive variation. Toxicon 1987, 25, 767–777. [Google Scholar] [CrossRef]
- Rowe, A.H.; Xiao, Y.; Rowe, M.P.; Cummins, T.R.; Zakon, H.H. Voltage-gated sodium channel in grasshopper mice defends against bark scorpion toxin. Science 2013, 342, 441–446. [Google Scholar] [CrossRef]
- Pomento, A.M.; Perry, B.W.; Denton, R.D.; Gibbs, H.L.; Holding, M.L. No safety in the trees: Local and species-level adaptation of an arboreal squirrel to the venom of sympatric rattlesnakes. Toxicon 2016, 118. [Google Scholar] [CrossRef] [PubMed]
- Biardi, J.E. The ecological and evolutionary context of mammalian resistance to rattlesnake venoms. In Biology of the Rattlesnakes; Hayes, W.K., Beaman, K.R., Cardwell, M.D., Bush, S.P., Eds.; Loma Linda University Press: Loma Linda, CA, USA, 2008; pp. 557–568. [Google Scholar]
- Gibbs, H.L.; Sanz, L.; Pérez, A.; Ochoa, A.; Hassinger, A.T.B.; Holding, M.L.; Calvete, J.J. The molecular basis of venom resistance in a rattlesnake-squirrel predator-prey system. Mol. Ecol. 2020, mec.15529. [Google Scholar] [CrossRef]
- de Wit, C.A.; Weström, B.R. Venom resistance in the Hedgehog, Erinaceus europaeus: Purification and identification of macroglobulin inhibitors as plasma antihemorrhagic factors. Toxicon 1987, 25, 315–323. [Google Scholar] [CrossRef]
- Biardi, J.E.; Ho, C.Y.L.; Marcinczyk, J.; Nambiar, K.P. Isolation and identification of a snake venom metalloproteinase inhibitor from California ground squirrel (Spermophilus beecheyi) blood sera. Toxicon 2011, 58, 486–493. [Google Scholar] [CrossRef]
- Holding, M.L.; Drabeck, D.H.; Jansa, S.A.; Gibbs, H.L. Venom resistance as a model for understanding the molecular basis of complex coevolutionary adaptations. Integr. Comp. Biol. 2016, 56. [Google Scholar] [CrossRef] [PubMed]
- McGlothlin, J.W.; Jawor, J.M.; Ketterson, E.D. Natural variation in a testosterone-mediated trade-off between mating effort and parental effort. Am. Nat. 2007, 170, 864–875. [Google Scholar] [CrossRef]
- Swanson, E.M.; Dantzer, B. Insulin-like growth factor-1 is associated with life-history variation across Mammalia. Proc. R. Soc. B Biol. Sci. 2014, 281, 20132458. [Google Scholar] [CrossRef]
- Herr, M.W.; Graham, S.P.; Langkilde, T. Stressed snakes strike first: Hormone levels and defensive behavior in free ranging cottonmouths (Agkistrodon piscivorus). Gen. Comp. Endocrinol. 2017, 243, 89–95. [Google Scholar] [CrossRef]
- Taff, C.C.; Schoenle, L.A.; Vitousek, M.N. The repeatability of glucocorticoids: A review and meta-analysis. Gen. Comp. Endocrinol. 2018, 260, 136–145. [Google Scholar] [CrossRef]
- Smith, J.E.; Long, D.J.; Russell, I.D.; Newcomb, K.L.; Muñoz, V.D. Otospermophilus beecheyi (Rodentia: Sciuridae). Mamm. Species 2016, 48, 91–108. [Google Scholar] [CrossRef]
- Fitch, H.S. Study of snake populations in Central California. Am. Midl. Nat. 1949, 41, 513–579. [Google Scholar] [CrossRef]
- Owings, D.H.; Coss, R.G. Snake mobbing by California Ground Squirrels: Adaptive variation and ontogeny. Behaviour 1977, 62. [Google Scholar]
- Coss, R.G. Context and animal behavior III: The relationship between early development and evolutionary persistence of ground qquirrel antisnake behavior. Ecol. Psychol. 1991, 3, 277–315. [Google Scholar] [CrossRef]
- Coss, R.G.; Gusé, K.L.; Poran, N.S.; Smith, D.G. Development of antisnake defenses in California ground squirrels (Spermophilus beecheyi): II. Microevolutionary effects of relaxed selection from rattlesnakes. Behaviour 1993, 124, 137–164. [Google Scholar] [CrossRef]
- Poran, N.S.; Coss, R.G. Development of antisnake defenses in California ground squirrels (Spermophilus beecheyi): I. Behavioral and immunological relationships. Behaviour 1990, 112, 222–245. [Google Scholar] [CrossRef]
- Holding, M.L.; Biardi, J.E.; Gibbs, H.L. Coevolution of venom function and venom resistance in a rattlesnake predator and its squirrel prey. Proc. R. Soc. B Biol. Sci. 2016, 283. [Google Scholar] [CrossRef]
- Mackessy, S.P. Evolutionary trends in venom composition in the western rattlesnakes (Crotalus viridis sensu lato): Toxicity vs. tenderizers. Toxicon 2010, 55, 1463–1474. [Google Scholar] [CrossRef]
- Holding, M.L.; Margres, M.J.; Rokyta, D.R.; Gibbs, H.L. Local prey community composition and genetic distance predict venom divergence among populations of the northern Pacific rattlesnake ( Crotalus oreganus ). J. Evol. Biol. 2018. [Google Scholar] [CrossRef]
- Ortiz, C.A.; Pendleton, E.L.; Newcomb, K.L.; Smith, J.E. Conspecific presence and microhabitat features influence foraging decisions across ontogeny in a facultatively social mammal. Behav. Ecol. Sociobiol. 2019, 73, 1–14. [Google Scholar] [CrossRef]
- Owings, D.H.; Coss, R.G.; McKernon, D.; Rowe, M.P.; Arrowood, P.C. Snake-directed antipredator behavior of Rock Squirrels (Spermophilus variegatus): Population differences and snake-species discrimination. Behaviour 2001, 138, 575–595. [Google Scholar]
- Coss, R.G.; Biardi, J.E. Individual Variation in the Antisnake Behavior of California Ground Squirrels (Spermophilus beecheyi). J. Mammal. 1997, 78, 294–310. [Google Scholar] [CrossRef]
- Hammond, T.T.; Vo, M.; Burton, C.T.; Surber, L.L.; Lacey, E.A.; Smith, J.E. Physiological and behavioral responses to anthropogenic stressors in a human-tolerant mammal. J. Mammal. 2019, 100, 1928–1940. [Google Scholar] [CrossRef]
- Smith, J.E.; Gamboa, D.A.; Spencer, J.M.; Travenick, S.J.; Ortiz, C.A.; Hunter, R.D.; Sih, A. Split between two worlds: Automated sensing reveals links between above- and belowground social networks in a free-living mammal. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2018, 373, 20170249. [Google Scholar] [CrossRef]
- Swaisgood, R.R.; Rowe, M.P.; Owings, D.H. Assessment of rattlesnake dangerousness by California Ground Squirrels: Exploration of cues from rattling sounds. Anim. Behav. 1999, 57, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Wolf, E.J.; Harrington, K.M.; Clark, S.L.; Miller, M.W. Sample size requirements for structural equation models. Educ. Psychol. Meas. 2013, 73, 913–934. [Google Scholar] [CrossRef]
- Boellstorff, D.E.; Owings, D.H. Home range, population structure, and spatial organization of California ground squirrels. J. Mammal. 1995, 76, 551–561. [Google Scholar] [CrossRef]
- Leger, D.W.; Owings, D.H.; Coss, R.G. Behavioral ecology of time allocation in California ground squirrels (Spermophilus beecheyi): Microhabitat effects. J. Comp. Psychol. 1983, 97, 283–291. [Google Scholar] [CrossRef]
- Smith, J.E. Hamilton’s legacy: Kinship, cooperation and social tolerance inmammalian groups. Anim. Behav. 2014, 92, 291–304. [Google Scholar] [CrossRef]
- Brown, J.S.; Kotler, B.P. Hazardous duty pay and the foraging cost of predation. Ecol. Lett. 2004, 7, 999–1014. [Google Scholar] [CrossRef]
- Dender, M.G.E.; Capelle, P.M.; Love, O.P.; Heath, D.D.; Heath, J.W.; Semeniuk, C.A.D. Phenotypic integration of behavioural and physiological traits is related to variation in growth among stocks of Chinook salmon. Can. J. Fish. Aquat. Sci. 2018, 75, 2271–2279. [Google Scholar] [CrossRef]
- Hazard, L.C.; Nagy, K.A.; Miles, D.B.; Svensson, E.I.; Costa, D.; Sinervo, B. Integration of genotype, physiological performance, and survival in a lizard (Uta stansburiana) with alternative mating strategies. Physiol. Biochem. Zool. 2019, 92, 303–315. [Google Scholar] [CrossRef] [PubMed]
- Levis, N.A.; Martin, R.A.; O’Donnell, K.A.; Pfennig, D.W. Intraspecific adaptive radiation: Competition, ecological opportunity, and phenotypic diversification within species. Evolution (N. Y). 2017, 71, 2496–2509. [Google Scholar] [CrossRef]
- Santini, F.; Climent, J.M.; Voltas, J. Phenotypic integration and life history strategies among populations of Pinus halepensis: An insight through structural equation modelling. Ann. Bot. 2019, 124, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.N. The Geographic Mosaic of Coevolution; University of Chicago Press: Chicago, IL, USA, 2005; ISBN 9780226797625. [Google Scholar]
- Swaisgood, R.R.; Owings, D.H.; Rowe, M.P. Conflict and assessment in a predator-prey system: Ground squirrels versus rattlesnakes. Anim. Behav. 1999, 57, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Swaisgood, R.R.; Rowe, M.P.; Owings, D.H. Antipredator responses of California ground squirrels to rattlesnakes and rattling sounds: The roles of sex, reproductive parity, and offspring age in assessment and decision-making rules. Behav. Ecol. Sociobiol. 2003, 55, 22–31. [Google Scholar] [CrossRef]
- Gao, S.; Deviche, P.J. The causative effects of corticosterone on innate immunity during the stress response in the House Sparrow, Passer domesticus. Gen. Comp. Endocrinol. 2019, 275, 30–37. [Google Scholar] [CrossRef]
- DeVries, A.C.; Gerber, J.M.; Richardson, H.N.; Moffatt, C.A.; Demas, G.E.; Taymans, S.E.; Nelson, R.J. Stress affects corticosteroid and immunoglobulin concentrations in male house mice (Mus musculus) and prairie voles (Microtus ochrogaster). Comp. Biochem. Physiol. A Physiol. 1997, 118, 655–663. [Google Scholar] [CrossRef]
- Hews, D.K.; Abell Baniki, A.J. The breeding season duration hypothesis: Acute handling stress and total plasma concentrations of corticosterone and androgens in male and female striped plateau lizards (Sceloporus virgatus). J. Comp. Physiol. B Biochem. Syst. Environ. Physiol. 2013, 183, 933–946. [Google Scholar] [CrossRef]
- Jessop, T.S.; Lane, M.L.; Teasdale, L.; Stuart-Fox, D.; Wilson, R.S.; Careau, V.; Moore, I.T. Multiscale evaluation of thermal dependence in the glucocorticoid response of vertebrates. Am. Nat. 2016, 188, 342–356. [Google Scholar] [CrossRef]
- Réale, D.; Reader, S.M.; Sol, D.; McDougall, P.T.; Dingemanse, N.J. Integrating animal temperament within ecology and evolution. Biol. Rev. 2007, 82, 291–318. [Google Scholar] [CrossRef]
- Putman, B.J.; Azure, K.R.; Swierk, L. Dewlap size in male water anoles associates with consistent inter-individual variation in boldness. Curr. Zool. 2018, 65, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Dosmann, A.; Brooks, K.C.; Mateo, J.M. Evidence for a mechanism of phenotypic integration of behaviour and innate immunity in a wild rodent: Implications for animal personality and ecological immunology. Anim. Behav. 2015, 101, 179–189. [Google Scholar] [CrossRef]
- Sih, A.; Mathot, K.J.; Moirón, M.; Montiglio, P.O.; Wolf, M.; Dingemanse, N.J. Animal personality and state-behaviour feedbacks: A review and guide for empiricists. Trends Ecol. Evol. 2015, 30, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Clark, C.W. Antipredator behavior and the asset-protection principle. Behav. Ecol. 1994, 5, 159–170. [Google Scholar] [CrossRef]
- Luttbeg, B.; Sih, A. Risk, resources and state-dependent adaptive behavioural syndromes. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3977–3990. [Google Scholar] [CrossRef]
- Alzaga, V.; Vicente, J.; Villanua, D.; Acevedo, P.; Casas, F.; Gortazar, C. Body condition and parasite intensity correlates with escape capacity in Iberian hares (Lepus granatensis). Behav. Ecol. Sociobiol. 2008, 62, 769–775. [Google Scholar] [CrossRef]
- Hoefler, C.D.; Persons, M.H.; Rypstra, A.L. Evolutionarily costly courtship displays in a wolf spider: A test of viability indicator theory. Behav. Ecol. 2008, 19, 974–979. [Google Scholar] [CrossRef]
- Brooks, K.C.; Mateo, J.M. Chronically raised glucocorticoids reduce innate immune function in Belding’s ground squirrels (Urocitellus beldingi) after an immune challenge. Gen. Comp. Endocrinol. 2013, 193, 149–157. [Google Scholar] [CrossRef]
- Ebensperger, L.A.; Ramírez-Estrada, J.; León, C.; Castro, R.A.; Tolhuysen, L.O.; Sobrero, R.; Quirici, V.; Burger, J.R.; Soto-Gamboa, M.; Hayes, L.D. Sociality, glucocorticoids and direct fitness in the communally rearing rodent, Octodon degus. Horm. Behav. 2011, 60, 346–352. [Google Scholar] [CrossRef]
- Hanson, M.T.; Coss, R.G. Age Differences in the Response of California Ground Squirrels (Spermophilus beecheyi) to Avian and Mammalian Predators. J. Comp. Psychol. 1997, 111, 174–184. [Google Scholar] [CrossRef]
- Peig, J.; Green, A.J. New perspectives for estimating body condition from mass/length data: The scaled mass index as an alternative method. Oikos 2009, 118, 1883–1891. [Google Scholar] [CrossRef]
- Putman, B.J.; Barbour, M.A.; Clark, R.W. The foraging behavior of free-ranging rattlesnakes (Crotalus oreganus) in California ground qquirrel (Otospermophilus beecheyi) colonies. Herpetologica 2016, 72, 55–63. [Google Scholar] [CrossRef]
- Wilson, A.D.M.; Stevens, E.D. Consistency in context-specific measures of shyness and boldness in rainbow trout, Oncorhynchus mykiss. Ethology 2005, 111, 849–862. [Google Scholar] [CrossRef]
- Dammhahn, M.; Almeling, L. Is risk taking during foraging a personality trait? A field test for cross-context consistency in boldness. Anim. Behav. 2012, 84, 1131–1139. [Google Scholar] [CrossRef]
- Biardi, J.E.; Nguyen, K.T.; Lander, S.; Whitley, M.; Nambiar, K.P. A rapid and sensitive fluorometric method for the quantitative analysis of snake venom metalloproteases and their inhibitors. Toxicon 2011, 57, 342–347. [Google Scholar] [CrossRef]
- Stoffel, M.A.; Nakagawa, S.; Schielzeth, H. rptR: Repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol. Evol. 2017, 8, 1639–1644. [Google Scholar] [CrossRef]
- Zhang, D. rsq: R-Squared and Related Measures. R Package. 2020. Available online: https://CRAN.R-project.org/package=rsq (accessed on 27 September 2020).
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. R Core Team nlme: Linear and Nonlinear Mixed Effects Models 2017. Available online: https://CRAN.R-project.org/package=nlme (accessed on 27 September 2020).
- Nakagawa, S.; Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Lefcheck, J.S. piecewiseSEM: Piecewise structural equation modeling in R for ecology, evolution, and systematics. Methods Ecol. Evol. 2016, 7, 573–579. [Google Scholar] [CrossRef]
- Zepeda, J.A.; Rödel, H.G.; Monclús, R.; Hudson, R.; Bautista, A. Sibling differences in litter huddle position contribute to overall variation in weaning mass in a small mammal. Behav. Ecol. Sociobiol. 2019, 73, 165. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
| Model Terms | Coefficient | Std. Error | t-Value | p-Value | Partial R2 |
|---|---|---|---|---|---|
| Intercept | 51.41 | 20.46 | 2.51 | 0.031 | N/A |
| Ln(Corticosterone) | −11.76 | 3.82 | −3.08 | 0.012 | 0.43 |
| Body Condition | 0.04 | 0.02 | 2.24 | 0.049 | 0.17 |
| Sex (Ref. = Male) | −2.90 | 3.18 | −0.91 | 0.382 | 0.03 |
| Behavior | Model Terms | Coefficient | Std. Error | t-Value | p-Value | Partial R2 |
|---|---|---|---|---|---|---|
| i. Tail-flagging Rate | Intercept | 0.73 | 5.20 | 0.14 | 0.890 | N/A |
| R2 = 16% | Snake Species a | 0.12 | 0.31 | 0.39 | 0.703 | 0.003 |
| Snake Size | 0.00 | 0.01 | 0.14 | 0.887 | 0 | |
| Squirrel Sex b | −0.07 | 0.78 | −0.10 | 0.926 | 0.001 | |
| Body Condition | −0.01 | 0.00 | −1.19 | 0.262 | 0.105 | |
| Ln(Corticosterone) | 0.76 | 0.93 | 0.82 | 0.430 | 0.059 | |
| Squirrel ID | Variance = 0.60 | SD = 0.77 | 10.1 | 0.002 | N/A | |
| ii. Proximity to Snake | Intercept | −0.96 | 3.47 | −0.28 | 0.784 | N/A |
| R2 = 28% | Snake Species a | −0.79 | 0.25 | −3.13 | 0.005 | 0.173 |
| Snake Size | 0.04 | 0.01 | 3.08 | 0.005 | 0.176 | |
| Squirrel Sex b | −0.43 | 0.51 | −0.85 | 0.415 | 0.044 | |
| Body Condition | 0.00 | 0.00 | −0.21 | 0.838 | 0.003 | |
| Ln(Corticosterone) | 0.21 | 0.60 | 0.34 | 0.738 | 0.008 | |
| Squirrel ID | Variance = 0.38 | SD = 0.62 | 4.2 | 0.040 | N/A | |
| iii. Latency to Feed | Intercept | −3.89 | 5.39 | −0.72 | 0.477 | N/A |
| R2 = 33% | Snake Species a | −0.48 | 0.63 | −0.76 | 0.453 | 0.018 |
| Snake Size | 0.02 | 0.03 | 0.53 | 0.601 | 0.009 | |
| Squirrel Sex b | −0.60 | 0.73 | −0.83 | 0.428 | 0.021 | |
| Body Condition | −0.01 | 0.00 | −2.24 | 0.049 | 0.136 | |
| Ln(Corticosterone) | 2.50 | 0.84 | 2.98 | 0.014 | 0.218 | |
| Squirrel ID | Variance = 2.7 | SD = 1.6 | −12.1 | 1 | N/A |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Holding, M.L.; Putman, B.J.; Kong, L.M.; Smith, J.E.; Clark, R.W. Physiological Stress Integrates Resistance to Rattlesnake Venom and the Onset of Risky Foraging in California Ground Squirrels. Toxins 2020, 12, 617. https://doi.org/10.3390/toxins12100617
Holding ML, Putman BJ, Kong LM, Smith JE, Clark RW. Physiological Stress Integrates Resistance to Rattlesnake Venom and the Onset of Risky Foraging in California Ground Squirrels. Toxins. 2020; 12(10):617. https://doi.org/10.3390/toxins12100617
Chicago/Turabian StyleHolding, Matthew L., Breanna J. Putman, Lauren M. Kong, Jennifer E. Smith, and Rulon W. Clark. 2020. "Physiological Stress Integrates Resistance to Rattlesnake Venom and the Onset of Risky Foraging in California Ground Squirrels" Toxins 12, no. 10: 617. https://doi.org/10.3390/toxins12100617
APA StyleHolding, M. L., Putman, B. J., Kong, L. M., Smith, J. E., & Clark, R. W. (2020). Physiological Stress Integrates Resistance to Rattlesnake Venom and the Onset of Risky Foraging in California Ground Squirrels. Toxins, 12(10), 617. https://doi.org/10.3390/toxins12100617





