The Development of Toad Toxins as Potential Therapeutic Agents
Abstract
1. Introduction
2. Chemicals Components in Different Species of Toads
3. The Bioactivity Studies of Bufadienolides
3.1. Bufalin
3.2. Cinobufagin
3.3. Arenobufagin
3.4. Gamabufotalin
3.5. Other Key Bufadienolides
4. Indolealkylamines
4.1. Bufotenine
4.2. Bufotenidine
4.3. Dehydrobufotenine
4.3.1. Bufothionine
4.3.2. Other Indolealkylamines
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, Q.; Zhou, X.; Zhang, M.; Bi, L.; Miao, S.; Cao, W.; Xie, Y.; Sun, J.; Tang, H.; Li, Y. Angel of human health: Current research updates in toad medicine. Am. J. Transl. Res. 2015, 7, 1. [Google Scholar] [PubMed]
- Gao, H.; Zehl, M.; Leitner, A.; Wu, X.; Wang, Z.; Kopp, B. Comparison of toad venoms from different Bufo species by HPLC LC-DAD-MS/MS. J. Ethnopharmacol. 2010, 131, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Yau, L.-F.; Lu, J.-G.; Wu, Z.-Z.; Zhang, B.-X.; Wang, J.-R.; Jiang, Z.-H. Chemical profiling and cytotoxicity assay of bufadienolides in toad venom and toad skin. J. Ethnopharmacol. 2016, 187, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Tan, C.; Hashimi, S.M.; Zulfiker, A.H.M.; Good, D.; Wei, M.Q. Toad glandular secretions and skin extractions as anti-inflammatory and anticancer agents. Evid.-Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Yang, P.; Shen, Y.; Bei, W.; Zhang, Y.; Ge, Y.; Newman, R.A.; Cohen, L.; Liu, L.; Thornton, B. Pilot study of huachansu in patients with hepatocellular carcinoma, nonsmall-cell lung cancer, or pancreatic cancer. Cancer 2009, 115, 5309–5318. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Shen, Y.; Yang, P.; Robert, N.; Bei, W.; Zhang, Y.; Yongqian, G.; Lorenzo, C.; Razelle, K.; Liu, L. Phase I study of huachansu in hepatocellular carcinoma, non-small cell lung cancer, and pancreatic cancer: A preliminary report. Chin. Oncol. 2001, 5, 006. [Google Scholar]
- Wang, L.; Raju, U.; Milas, L.; Molkentine, D.; Zhang, Z.; Yang, P.; Cohen, L.; Meng, Z.; Liao, Z. Huachansu, containing cardiac glycosides, enhances radiosensitivity of human lung cancer cells. Anticancer Res. 2011, 31, 2141–2148. [Google Scholar] [PubMed]
- Efuet, E.T.; Ding, X.-P.; Cartwright, C.; Pan, Y.; Cohen, L.; Yang, P. Huachansu mediates cell death in non-Hodgkin’s lymphoma by induction of caspase-3 and inhibition of MAP kinase. Int. J. Oncol. 2015, 47, 592–600. [Google Scholar] [CrossRef] [PubMed]
- Laursen, M.; Gregersen, J.L.; Yatime, L.; Nissen, P.; Fedosova, N. Structures and characterization of digoxin-and bufalin-bound Na+, K+-ATPase compared with the ouabain-bound complex. Proc. Natl. Acad. Sci. USA 2015, 112, 1755–1760. [Google Scholar] [CrossRef] [PubMed]
- Prassas, I.; Diamandis, E.P. Novel therapeutic applications of cardiac glycosides. Nat. Rev. Drug Discov. 2008, 7, 926. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.A.; Smith, T.W. Recognition and management of digitalis toxicity. Am. J. Cardiol. 1992, 69, 108–119. [Google Scholar] [CrossRef]
- Kamboj, A.; Rathour, A.; Kaur, M. Bufadienolides and their medicinal utility: A review. Int. J. Pharm. Pharm. Sci. 2013, 5, 20–27. [Google Scholar]
- Baldo, E.C.F.; Anjolette, F.A.P.; Arantes, E.C.; Baldo, M.A. Toad Poison and Drug Discovery. Toxicon 2015, 1–22. [Google Scholar] [CrossRef]
- Davis, W.; Weil, A.T. Identity of a New World psychoactive toad. Anc. Mesoam. 1992, 3, 51–59. [Google Scholar] [CrossRef]
- Weil, A.T.; Davis, W. Bufo alvarius: A potent hallucinogen of animal origin. J. Ethnopharmacol. 1994, 41, 1–8. [Google Scholar] [CrossRef]
- Chamakura, R.P. Bufotenine-A Hallucinogen in Ancient Snuff Powders of South America and a Drug of Abuse on the Streets of New York City. Forensic Sci. Rev. 1994, 6, 1–18. [Google Scholar] [PubMed]
- Phillips, B.L.; Brown, G.P.; Greenlees, M.; Webb, J.K.; Shine, R. Rapid expansion of the cane toad (Bufo marinus) invasion front in tropical Australia. Aust. Ecol. 2007, 32, 169–176. [Google Scholar] [CrossRef]
- Leitch, I.; Lim, T.; Boura, A. Novel drugs from toad skins. RIDC Publ. Aust. 2000, 17, 1–65. [Google Scholar]
- Zulfiker, A.H.M.; Hashimi, S.M.; Qi, J.; Grice, I.D.; Wei, M.Q. Aqueous and Ethanol Extracts of Australian Cane Toad Skins Suppress Pro-Inflammatory Cytokine Secretion in U937 Cells via NF-κB Signaling Pathway. J. Cell. Biochem. 2016, 117, 2769–2780. [Google Scholar] [CrossRef] [PubMed]
- Zulfiker, A.H.M.; Hashimi, S.M.; Good, D.A.; Grice, I.D.; Wei, M.Q. Cane Toad Skin Extract—Induced Upregulation and Increased Interaction of Serotonin 2A and D2 Receptors via Gq/11 Signaling Pathway in CLU213 Cells. J. Cell. Biochem. 2017, 118, 979–993. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.L.; Qi, F.H.; Tang, W.; Wang, F.S. Chemical constituents and bioactivities of the skin of Bufo bufo gargarizans Cantor. Chem. Biodivers. 2011, 8, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Zulfiker, A.H.M.; Sohrabi, M.; Qi, J.; Matthews, B.; Wei, M.Q.; Grice, I.D. Multi-constituent identification in Australian cane toad skin extracts using high-performance liquid chromatography high-resolution tandem mass spectrometry. J. Pharm. Biomed. Anal. 2016, 129, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Cunha Filho, G.A.; Schwartz, C.A.; Resck, I.S.; Murta, M.M.; Lemos, S.S.; Castro, M.S.; Kyaw, C.; Pires, O.R., Jr.; Leite, J.R.S.; Bloch, C., Jr. Antimicrobial activity of the bufadienolides marinobufagin and telocinobufagin isolated as major components from skin secretion of the toad Bufo rubescens. Toxicon 2005, 45, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Cunha-Filho, G.A.; Resck, I.S.; Cavalcanti, B.C.; Pessoa, C.Ó.; Moraes, M.O.; Ferreira, J.R.; Rodrigues, F.A.; dos Santos, M.L. Cytotoxic profile of natural and some modified bufadienolides from toad Rhinella schneideri parotoid gland secretion. Toxicon 2010, 56, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Zulfiker, M.; Hasanat, A.; Mariottini, G.L.; Qi, J.; Grice, I.D.; Wei, M.Q. Indolealkylamines from toad vertebrates and sea invertebrates-their identification and potential activities on the central nervous system. Cent. Nerv. Syst. Agents Med. Chem. 2016, 16, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Hippargi, R.V.; Gandhare, A.N. Toad skin-secretions: Potent source of pharmacologically and therapeutically significant compounds. Int. J. Pharmacol. 2008, 5, 17. [Google Scholar]
- Liu, M.; Feng, L.-X.; Hu, L.-H.; Liu, X.; Guo, D.-A. Advancement in research of anti-cancer effects of toad venom (ChanSu) and perspectives. World J. Tradit. Chin. Med. 2015, 1, 12–23. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.; Lin, Y.; Wang, X.; Fang, L.; Geng, Y.; Zhang, Q. Preparative separation and purification of bufadienolides from ChanSu by high-speed counter-current chromatography combined with preparative HPLC. Química Nova 2013, 36, 686–690. [Google Scholar] [CrossRef]
- Ye, M.; Guo, H.; Guo, H.; Han, J.; Guo, D. Simultaneous determination of cytotoxic bufadienolides in the Chinese medicine ChanSu by high-performance liquid chromatography coupled with photodiode array and mass spectrometry detections. J. Chromatogr. B 2006, 838, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Cui, Z.; Liu, Y.-S.; Sheng, Y. Isolation and identification of the indolealkylamines from the traditional Chinese medicine Toad Venom. J. Shenyang Pharm. Univ. 2006, 4, 005. [Google Scholar]
- Wu, X.; Zhao, H.; Wang, H.; Gao, B.; Yang, J.; Si, N.; Bian, B. Simultaneous determination of eight bufadienolides in cinobufacini injection by HPLC coupled with triple quadrupole mass spectrometry. J. Sep. Sci. 2012, 35, 1893–1898. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Cao, W.; Chen, Y.; Qu, D.; Zhou, J. Comparison of toad skins Bufo bufo gargarizans Cantor from different regions for their active constituents content and cytotoxic activity on lung carcinoma cell lines. Pharmacogn. Mag. 2014, 10, 207. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Garrett, C.; Shen, Y.; Liu, L.; Yang, P.; Huo, Y.; Zhao, Q.; Spelman, A.; Ng, C.; Chang, D. Prospective randomised evaluation of traditional Chinese medicine combined with chemotherapy: A randomised phase II study of wild toad extract plus gemcitabine in patients with advanced pancreatic adenocarcinomas. Br. J. Cancer 2012, 107, 411. [Google Scholar] [CrossRef] [PubMed]
- Shine, R. The ecological impact of invasive cane toads (Bufo marinus) in Australia. Q. Rev. Biol. 2010, 85, 253–291. [Google Scholar] [CrossRef] [PubMed]
- Jing, J.; Ren, W.C.; Li, C.; Bose, U.; Parekh, H.S.; Wei, M.Q. Rapid identification of primary constituents in parotoid gland secretions of the Australian cane toad using HPLC/MS-Q-TOF. Biomed. Chromatogr. 2013, 27, 685–687. [Google Scholar] [CrossRef] [PubMed]
- Maciel, N.M.; Schwartz, C.A.; Junior, O.R.P.; Sebben, A.; Castro, M.S.; Sousa, M.V.; Fontes, W.; Schwartz, E.N.F. Composition of indolealkylamines of Bufo rubescens cutaneous secretions compared to six other Brazilian bufonids with phylogenetic implications. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2003, 134, 641–649. [Google Scholar] [CrossRef]
- Barry, T.L.; Petzinger, G.; Zito, S.W. GC/MS comparison of the West Indian aphrodisiac “Love Stone” to the Chinese medication “chan su”: Bufotenine and related bufadienolides. J. Forensic. Sci. 1996, 41, 1068–1073. [Google Scholar] [CrossRef] [PubMed]
- Moreno, Y.; Banuls, L.; Urban, E.; Gelbcke, M.; Dufrasne, F.o.; Kopp, B.; Kiss, R.; Zehl, M. Structure—Activity relationship analysis of bufadienolide-induced in vitro growth inhibitory effects on mouse and human cancer cells. J. Natl. Prod. 2013, 76, 1078–1084. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Yin, S.; Li, J.; Jiang, C.; Ye, M.; Hu, H. Bufadienolide compounds sensitize human breast cancer cells to TRAIL-induced apoptosis via inhibition of STAT3/Mcl-1 pathway. Apoptosis 2011, 16, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.H.; Kan, S.F.; Pu, H.F.; Chien, E.J.; Wang, P.S. Apoptotic signaling in bufalin-and cinobufagin-treated androgen-dependent and-independent human prostate cancer cells. Cancer Sci. 2008, 99, 2467–2476. [Google Scholar] [CrossRef] [PubMed]
- Yuan, B.; He, J.; Kisoh, K.; Hayashi, H.; Tanaka, S.; Si, N.; Zhao, H.-Y.; Hirano, T.; Bian, B.; Takagi, N. Effects of active bufadienolide compounds on human cancer cells and CD4+ CD25+ Foxp3+ regulatory T cells in mitogen-activated human peripheral blood mononuclear cells. Oncol. Rep. 2016, 36, 1377–1384. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.-H.; Liu, X.; Qiu, Y.-Y.; Cai, J.-F.; Qin, J.-M.; Zhu, H.-R.; Li, Q. Anti-tumor activity and apoptosis-regulation mechanisms of bufalin in various cancers: New hope for cancer patients. Asian Pac. J. Cancer Prev. 2012, 13, 5339–5343. [Google Scholar] [CrossRef] [PubMed]
- Karin, M. NF-κB as a critical link between inflammation and cancer. Cold Spring Harbor Perspect. Biol. 2009. [Google Scholar] [CrossRef]
- Zhakeer, Z.; Hadeer, M.; Tuerxun, Z.; Tuerxun, K. Bufalin inhibits the inflammatory effects in asthmatic mice through the suppression of nuclear factor-kappa B activity. Pharmacology 2017, 99, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Huang, Y.; Xie, X.; Huang, W.; Yin, J.; Lin, W.; Jia, Q.; Zeng, W. Anti-Inflammatory and Antinociceptive Activities of Bufalin in Rodents. Mediat. Inflamm. 2014, 2014, 171839. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Lu, H.F.; Hsu, S.C.; Kuo, C.L.; Chang, S.J.; Lin, J.J.; Wu, P.P.; Liu, J.Y.; Lee, C.H.; Chung, J.G. Bufalin inhibits migration and invasion in human hepatocellular carcinoma SK-Hep1 cells through the inhibitions of NF-kB and matrix metalloproteinase-2/-9-signaling pathways. Environ. Toxicol. 2015, 30, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-H.; Hsiao, Y.-T.; Kuo, C.-L.; Yu, F.-S.; Hsu, S.-C.; Wu, P.-P.; Chen, J.-C.; Hsia, T.-C.; Liu, H.-C.; Hsu, W.-H. Bufalin inhibits NCI-H460 human lung cancer cell metastasis in vitro by inhibiting MAPKs, MMPs, and NF-κB pathways. Am J. Chin. Med. 2015, 43, 1247–1264. [Google Scholar] [CrossRef] [PubMed]
- Takai, N.; Kira, N.; Ishii, T.; Yoshida, T.; Nishida, M.; Nishida, Y.; Nasu, K.; Narahara, H. Bufalin, a traditional oriental medicine, induces apoptosis in human cancer cells. Asian Pac. J. Cancer Prev. 2012, 13, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Han, K.Q.; Huang, G.; Gu, W.; Su, Y.H.; Huang, X.Q.; Ling, C.Q. Anti-tumor activities and apoptosis-regulated mechanisms of bufalin on the orthotopic transplantation tumor model of human hepatocellular carcinoma in nude mice. World J. Gastroenterol. 2007, 13, 3374–3379. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Yang, Y.K.; Wu, W.Z. Bufalin attenuates the stage and metastatic potential of hepatocellular carcinoma in nude mice. J. Transl. Med. 2014, 12, 57. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, C.; Wang, S.; Zhang, Y.; Yin, P.; Gao, Z.; Xu, J.; Feng, D.; Zuo, Q.; Zhao, R.; et al. Bufalin Inhibits HCT116 Colon Cancer Cells and Its Orthotopic Xenograft Tumor in Mice Model through Genes Related to Apoptotic and PTEN/AKT Pathways. Gastroenterol. Res. Pract. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.H.; Bau, D.T.; Hsiao, Y.T.; Lu, K.W.; Hsia, T.C.; Lien, J.C.; Ko, Y.C.; Hsu, W.H.; Yang, S.T.; Huang, Y.P. Bufalin induces apoptosis in vitro and has Antitumor activity against human lung cancer xenografts in vivo. Environ. Toxicol. 2017, 32, 1305–1317. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Hashimi, S.M.; Cao, S.; Mellick, A.S.; Duan, W.; Good, D.; Wei, M.Q. The mechanisms of chansu in inducing efficient apoptosis in colon cancer cells. Evid.-Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Hashimi, S.M.; Cao, S.; Qi, J.; Good, D.; Duan, W.; Wei, M.Q. Chansu inhibits the expression of cortactin in colon cancer cell lines in vitro and in vivo. BMC Complement. Altern. Med. 2015, 15, 207. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Yu, L.; Dai, G.; Zhang, S.; Zhang, Z.; Gao, T.; Guo, W. Cinobufagin induces apoptosis of osteosarcoma cells through inactivation of Notch signaling. Eur. J. Pharmacol. 2017, 794, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Lin, S.; Peng, P.; Cai, C.; Deng, J.; Wang, M.; Li, X.; Lin, R.; Lin, Y.; Fang, A.; et al. Arenobufagin activates p53 to trigger esophageal squamous cell carcinoma cell apoptosis in vitro and in vivo. OncoTargets Ther. 2017, 10, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.-M.; Liu, J.-S.; Deng, L.-J.; Chen, M.-F.; Yiu, A.; Cao, H.-H.; Tian, H.-Y.; Fung, K.-P.; Kurihara, H.; Pan, J.-X. Arenobufagin, a natural bufadienolide from toad venom, induces apoptosis and autophagy in human hepatocellular carcinoma cells through inhibition of PI3K/Akt/mTOR pathway. Carcinogenesis 2013, 34, 1331–1342. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Mai, W.; Chen, M.; Hu, J.; Zhuo, Z.; Lei, X.; Deng, L.; Liu, J.; Yao, N.; Huang, M. Arenobufagin inhibits prostate cancer epithelial-mesenchymal transition and metastasis by down-regulating β-catenin. Pharmacol. Res. 2017, 123, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wu, S.; Liu, Z.; Zhang, W.; Xu, J.; Wang, Y.; Liu, J.; Zhang, D.; Tian, H.; Li, Y. Arenobufagin, a bufadienolide compound from toad venom, inhibits VEGF-mediated angiogenesis through suppression of VEGFR-2 signaling pathway. Biochem. Pharmacol. 2012, 83, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Tang, N.; Shi, L.; Yu, Z.; Dong, P.; Wang, C.; Huo, X.; Zhang, B.; Huang, S.; Deng, S.; Liu, K. Gamabufotalin, a major derivative of bufadienolide, inhibits VEGF-induced angiogenesis by suppressing VEGFR-2 signaling pathway. Oncotarget 2016, 7, 3533. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Li, T.; Wang, C.; Deng, S.; Zhang, B.; Huo, X.; Zhang, B.; Wang, X.; Zhong, Y.; Ma, X. Gamabufotalin triggers c-Myc degradation via induction of WWP2 in multiple myeloma cells. Oncotarget 2016, 7, 15725. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Guo, W.; Ma, X.; Zhang, B.; Dong, P.; Huang, L.; Wang, X.; Wang, C.; Huo, X.; Yu, W. Gamabufotalin, a bufadienolide compound from toad venom, suppresses COX-2 expression through targeting IKKβ/NF-κB signaling pathway in lung cancer cells. Mol. Cancer 2014, 13, 203. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Song, Y.; An, N.; Zeng, S.; Wang, D.; Yu, L.; Zhu, T.; Zhang, T.; Cui, J.; Zhou, C. The effects of telocinobufagin isolated from Chan Su on the activation and cytokine secretion of immunocytes in vitro. Fundam. Clin. Pharmacol. 2009, 23, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Sementsov, A. Poisons of toads. Farm. Zh. (Kharkov) 1939, 12, 19–22. [Google Scholar]
- Shen, H.W.; Jiang, X.L.; Winter, J.C.; Yu, A.M. Psychedelic 5-methoxy-N,N-dimethyltryptamine: Metabolism, pharmacokinetics, drug interactions, and pharmacological actions. Curr. Drug Metab. 2010, 11, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Roth, B.; Choudhary, M.; Khan, N.; Uluer, A. High-affinity agonist binding is not sufficient for agonist efficacy at 5-hydroxytryptamine2A receptors: Evidence in favor of a modified ternary complex model. J. Pharmacol. Exp. Ther. 1997, 280, 576–583. [Google Scholar] [PubMed]
- Raisanen, M.; Karkkainen, J. Mass fragmentographic quantification of urinary N,N-dimethyltryptamine and bufotenine. J. Chromatogr. 1979, 162, 579–584. [Google Scholar] [CrossRef]
- Takeda, N.; Ikeda, R.; Ohba, K.; Kondo, M. Bufotenine reconsidered as a diagnostic indicator of psychiatric disorders. Neuroreport 1995, 6, 2378–2380. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, E.; Colombo, R.; Martinelli, V.; Brondino, N.; Marini, M.; Boso, M.; Barale, F.; Politi, P. Elevated urine levels of bufotenine in patients with autistic spectrum disorders and schizophrenia. Neuro Endocrinol. Lett. 2010, 31, 117–121. [Google Scholar] [PubMed]
- Kryukova, E.V.; Lebedev, D.S.; Ivanov, I.A.; Ivanov, D.A.; Starkov, V.G.; Tsetlin, V.I.; Utkin, Y.N. N-methyl serotonin analogues from the Bufo bufo toad venom interact efficiently with the alpha7 nicotinic acetylcholine receptors. Dokl. Biochem. Biophys. 2017, 472, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Erspamer, G.F.; Cei, J.M. Biogenic amines and active polypeptides in the skin of Leptodactylus vilarsi melin. Biochem. Pharmacol. 1970, 19, 321–325. [Google Scholar] [CrossRef]
- Gyermek, L.; Bindler, E. Action of indole alkylamines and amidines on the inferior mesenteric ganglion of the cat. J. Pharmacol. Exp. Ther. 1962, 138, 159–164. [Google Scholar] [PubMed]
- Dai, L.P.; Gao, H.M.; Wang, Z.M.; Wang, W.H. Isolation and structure identification of chemical constituents from the skin of Bufo bufo gargarizans. Yao Xue Xue Bao 2007, 42, 858–861. [Google Scholar] [PubMed]
- Radisky, D.C.; Radisky, E.S.; Barrows, L.R.; Copp, B.R.; Kramer, R.A.; Ireland, C.M. Novel cytotoxic topoisomerase II inhibiting pyrroloiminoquinones from Fijian sponges of the genus Zyzzya. J. Am. Chem. Soc. 1993, 115, 1632–1638. [Google Scholar] [CrossRef]
- Barrows, L.R.; Radisky, D.C.; Copp, B.R.; Swaffar, D.S.; Kramer, R.A.; Warters, R.L.; Ireland, C.M. Makaluvamines, marine natural-products, are active anticancer agents and DNA topo-ii inhibitors. Anti-Cancer Drug Des. 1993, 8, 333–347. [Google Scholar]
- Zhang, Z.; Chen, Y.; Jia, X.; He, J.; Wang, J. Dehydrobufotenine dry powder inhaler (DPI), its preparation method and application in preparation of antitumor drug for treating lung tumor. Am. Chem. Soc. 2012, 8, 55–66. [Google Scholar]
- Xie, R.-F.; Li, Z.-C.; Gao, B.; Shi, Z.-N.; Zhou, X. Bufothionine, a possible effective component in cinobufocini injection for hepatocellular carcinoma. J. Ethnopharmacol. 2012, 141, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Luo, C. Method for Extracting Bufothionine from Skin of Bufo Bufo Gargarizans Cantor; Anhui Jinchan Biochemical Co., Ltd.: Huaibei, China, 2012; 10p. [Google Scholar]
- Dai, Y.H.; Shen, B.; Xia, M.Y.; Wang, A.D.; Chen, Y.L.; Liu, D.C.; Wang, D. A New Indole Alkaloid from the Toad Venom of Bufo bufo gargarizans. Molecules 2016, 21, 349. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liu, J. New Formulations of Alkaloid from Toad Skin for Cancer Therapy, China, 2017; 14p.
- Chen, Y.; Zhang, Z.; Jia, X.; Zhou, L.; Wu, Q. Bufothionine Powder for Inhalation and its Preparation and Application for Treating Pulmonary Neoplasm, China, 2011; 8p.
- Ujváry, I. Psychoactive natural products: Overview of recent developments. Annali dell’Istituto Superiore Sanita 2014, 50, 12–27. [Google Scholar]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Anti-inflammatory and antipyretic activities of indo-methacin, 1-(p-chlorobenzoyl)-5-methoxy-2-methyl-indole-3-acetic acid. J. Pharmacol. Exp. Ther. 1963, 141, 369–376. [Google Scholar] [PubMed]
- Khamna, S.; Yokota, A.; Lumyong, S. Actinomycetes isolated from medicinal plant rhizosphere soils: Diversity and screening of antifungal compounds, indole-3-acetic acid and siderophore production. World J. Microbiol. Biotechnol. 2009, 25, 649–655. [Google Scholar] [CrossRef]
- Seltzer, H.S. Quantitative effects of glucose, sulfonylureas, salicylate, and indole-3-acetic acid on the secretion of insulin activity into pancreatic venous blood. J. Clin. Investig. 1962, 41, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Folkes, L.K.; Wardman, P. Oxidative activation of indole-3-acetic acids to cytotoxic species—A potential new role for plant auxins in cancer therapy. Biochem. Pharmacol. 2001, 61, 129–136. [Google Scholar] [CrossRef]
- Gowda, R.M.; Cohen, R.A.; Khan, I.A. Toad venom poisoning: Resemblance to digoxin toxicity and therapeutic implications. Heart 2003, 89, e14. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Popescu, R.; Kopp, B.; Wang, Z. Bufadienolides and their antitumor activity. Nat. Prod. Rep. 2011, 28, 953–969. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Zhu, L.; Liang, H.; Hu, F.; Feng, J. Improved antitumor efficacy and reduced toxicity of liposomes containing bufadienolides. Arch. Pharm. Res. 2011, 34, 1487. [Google Scholar] [CrossRef] [PubMed]
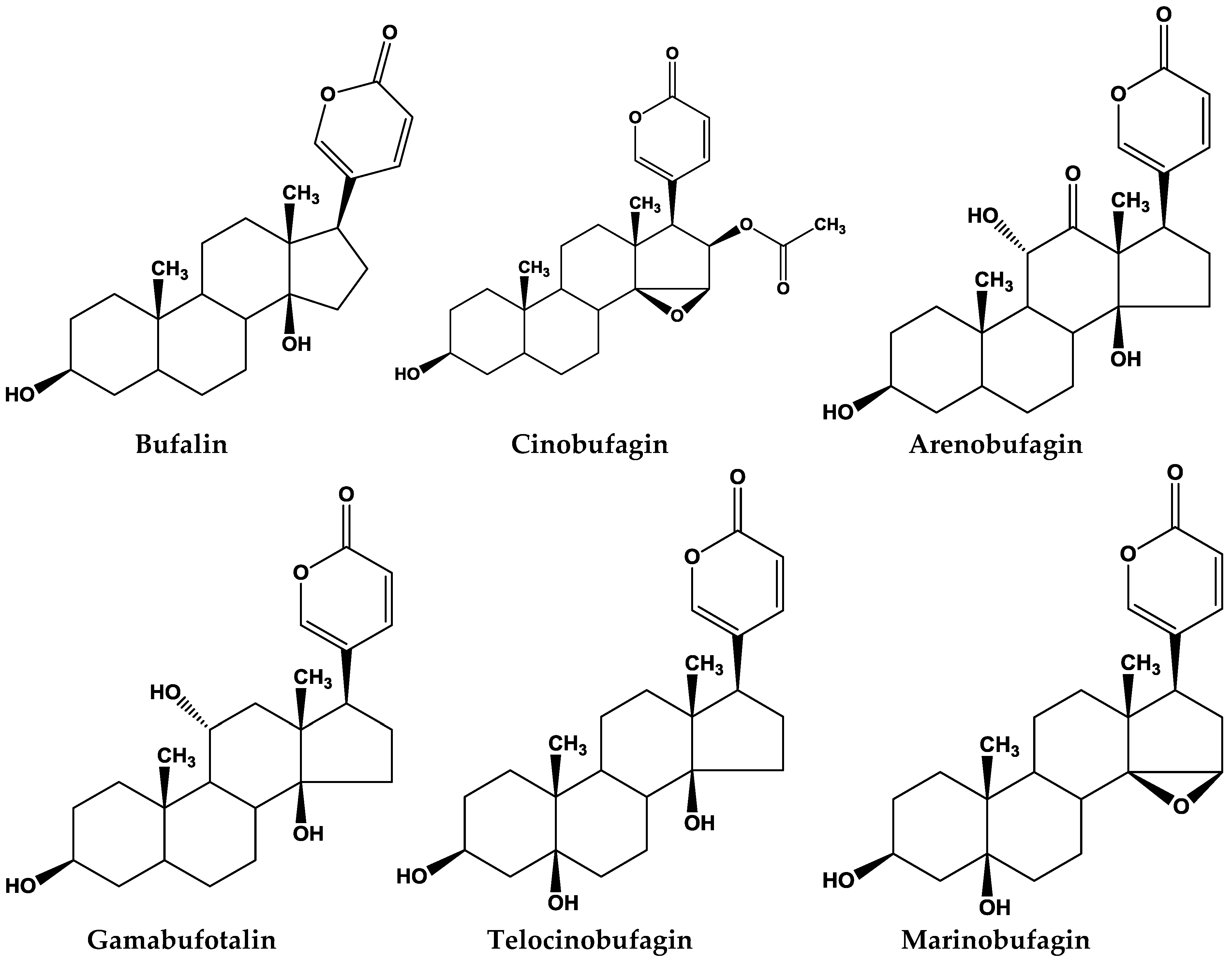
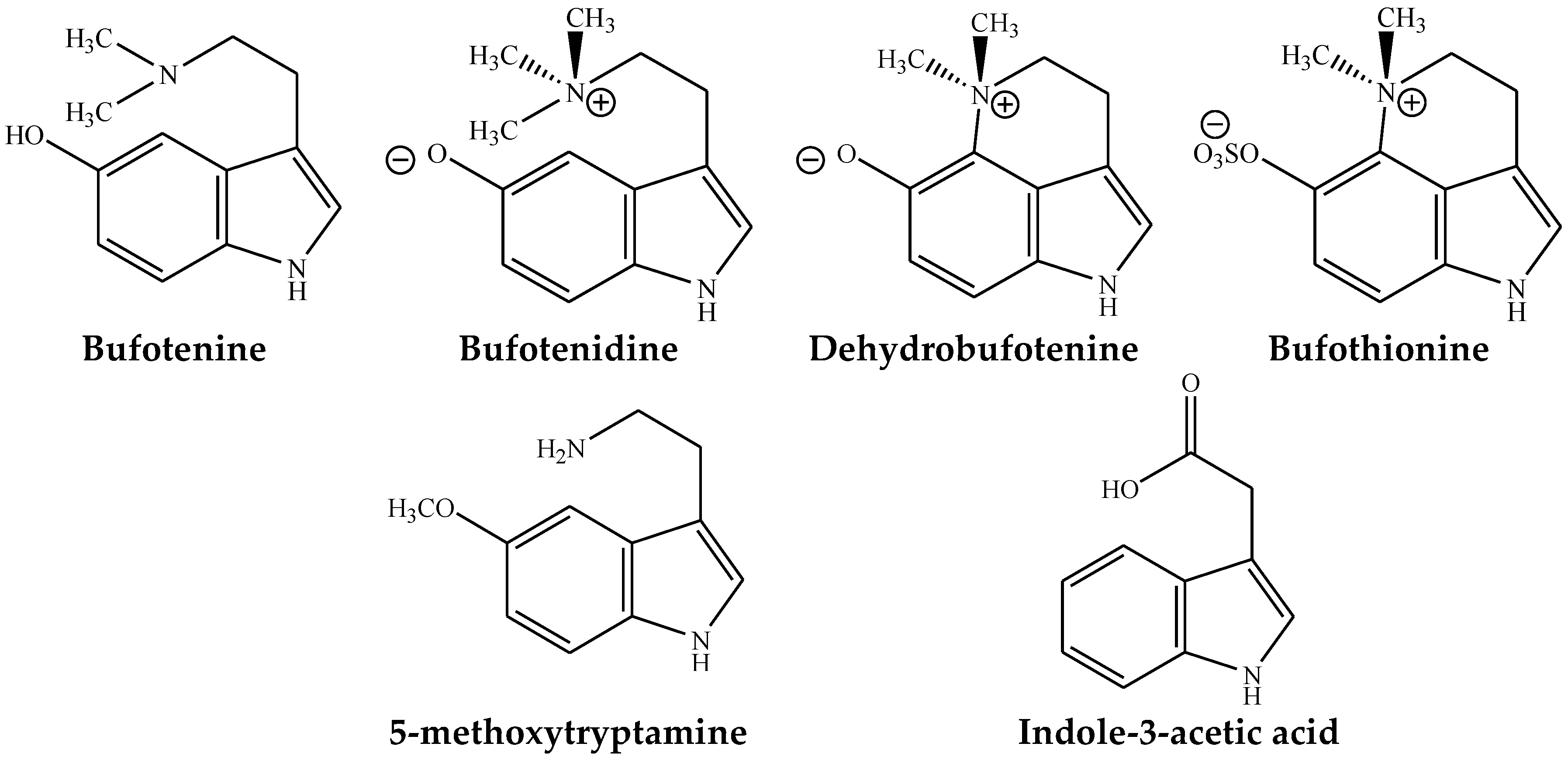
| Name | Classification | Formula | Species of Toad | |||
|---|---|---|---|---|---|---|
| B. b. gargarizans | B. marinus | B. alvarius | B. melanosticus | |||
| Bufalin | Bufadienolides | C24H34O4 | + | + | + | + |
| Cinobufagin | Bufadienolides | C26H34O6 | + | − | − | − |
| Arenobufagin | Bufadienolides | C24H32O6 | + | + | + | + |
| Gamabufotalin | Bufadienolides | C24H34O5 | + | − | + | + |
| Telocinobufagin | Bufadienolides | C24H34O5 | + | + | + | + |
| Marinobufagin | Bufadienolides | C24H32O5 | + | + | + | + |
| Bufotenine | Indolealkylamine | C12H16N2O | + | + | + | + |
| Bufotenidine | Indolealkylamine | C13H18N2O | + | − | − | + |
| Dehydrobufotenine | Indolealkylamine | C12H14N2O | + | + | − | + |
| Bufothionine | Indolealkylamine | C12H15N2O3S | + | + | + | − |
| 5-methoxytryptamine | Indolealkylamine | C11H14N2O | − | + | + | − |
| Indole-3-acetic acid | Indolealkylamine | C10H9NO2 | − | − | + | − |
| Compound | Experimental Models | Molecular Targets | References |
|---|---|---|---|
| Bufalin | In vitro/In vivo | Macrophages, eosinophils, lymphocytes, and neutrophils and cytokines including IL-4, IL-5, and IL-13, NF-κB | [44] |
| In vivo | iNOS, COX-2, IL-1β, IL-6, TNF-α, NF-κB | [45] | |
| In vitro | MMP-2, MMP-9, PI3K, AKT, NF-κB | [46,47] | |
| In vivo | Bax | [49] | |
| In vivo | AKT/GSK3β/β-catenin/E-cadherin | [50] | |
| In vivo | PTEN/phosphate-PTEN, AKT/phosphate-AKT, Bad, Bcl-xl, Bax, or Caspase-3 | [51] | |
| Cinobufagin | In vitro/In vivo | Caspase-3, hypoxia-inducing factor-1 alpha | [53] |
| In vivo | Cortactin | [54] | |
| In vitro | Notch pathway | [55] | |
| Arenobufagin | In vitro/In vivo | p53 pathway | [56] |
| In vitro/In vivo | Bax/Bcl-2, PI3K/Akt/ mTOR pathway. ADP-ribose polymerase, light chain 3-II | [57] | |
| In vivo | β-catenin | [58] | |
| In vitro/In vivo | VEGFR-2 pathway | [59] | |
| Gamabufotalin | In vivo | VEGFR-2 pathway | [60] |
| In vitro/In vivo | c-Myc | [61] | |
| In vitro/In vivo | IKKβ, NF-κB, COX-2, p65 | [62] | |
| Telocinobufagin | In vitro | CD4, CD8, IL-2, IL-12, IFN-γ, TNF-α, IL-4 | [63] |
| Recipe Name | Treatment Purpose | Main Ingredients |
|---|---|---|
| Liu Shen Wan | Inflammatory and infectious diseases, etc. | Chansu, Pearl Powder, Bezoar Bovis, Musk, Realgar, and Bornel |
| She Xiang Bao Xin Wan | Congestive heart failure | Chansu, Ginseng, Bezoar Bovis, Musk, Cinnamon, Liquidambar, and Borneol |
| Mei Hua Dian She Wan | Relieves swelling and pain | Chansu, Borneol, Cinnabar, Myrrh, Bezoar Bovis, Borax, Frankincense, Musk, Draco Seed, Realgar, Bear Gall, Blood Dracon, Pearl Powder, and Cinnabar |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, J.; Zulfiker, A.H.M.; Li, C.; Good, D.; Wei, M.Q. The Development of Toad Toxins as Potential Therapeutic Agents. Toxins 2018, 10, 336. https://doi.org/10.3390/toxins10080336
Qi J, Zulfiker AHM, Li C, Good D, Wei MQ. The Development of Toad Toxins as Potential Therapeutic Agents. Toxins. 2018; 10(8):336. https://doi.org/10.3390/toxins10080336
Chicago/Turabian StyleQi, Ji, Abu Hasanat Md Zulfiker, Chun Li, David Good, and Ming Q. Wei. 2018. "The Development of Toad Toxins as Potential Therapeutic Agents" Toxins 10, no. 8: 336. https://doi.org/10.3390/toxins10080336
APA StyleQi, J., Zulfiker, A. H. M., Li, C., Good, D., & Wei, M. Q. (2018). The Development of Toad Toxins as Potential Therapeutic Agents. Toxins, 10(8), 336. https://doi.org/10.3390/toxins10080336




