Bouganin, an Attractive Weapon for Immunotoxins
Abstract
1. Introduction
2. Purification and Antiviral Properties of Bouganin
3. Recombinant Bouganin and Structure/Function Studies
4. Antigenic Properties of Bouganin
5. Bouganin-Containing Immunotoxins, Preclinical Evaluations
6. Bouganin-Containing Immunotoxins, Clinical Evaluations
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bolognesi, A.; Bortolotti, M.; Maiello, S.; Battelli, M.G.; Polito, L. Ribosome-Inactivating Proteins from Plants: A Historical Overview. Molecules 2016, 21, 1627. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, L.; Valbonesi, P.; Bonora, E.; Gorini, P.; Bolognesi, A.; Stirpe, F. Polynucleotide: Adenosine glycosidase activity of ribosome-inactivating proteins: Effect on DNA, RNA and poly(A). Nucleic Acids Res. 1997, 25, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Battelli, M.G.; Barbieri, L.; Bolognesi, A.; Buonamici, L.; Valbonesi, P.; Polito, L.; Van Damme, E.J.; Peumans, W.J.; Stirpe, F. Ribosome-inactivating lectins with polynucleotide: Adenosine glycosidase activity. FEBS Lett. 1997, 408, 355–359. [Google Scholar] [CrossRef]
- Stirpe, F.; Bolognesi, A.; Bortolotti, M.; Farini, V.; Lubelli, C.; Pelosi, E.; Polito, L.; Dozza, B.; Strocchi, P.; Chambery, A.; et al. Characterization of highly toxic type 2 ribosome-inactivating proteins from Adenia lanceolata and Adenia stenodactyla (Passifloraceae). Toxicon 2007, 50, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Zheng, M.; Lu, D.; Wang, J.; Jiang, W.; Sha, O. Anti-tumor activities and apoptotic mechanism of ribosome-inactivating proteins. Chin. J. Cancer 2015, 34, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Polito, L.; Bortolotti, M.; Farini, V.; Battelli, M.G.; Barbieri, L.; Bolognesi, A. Saporin induces multiple death pathways in lymphoma cells with different intensity and timing as compared to ricin. Int. J. Biochem. Cell Biol. 2009, 41, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Huang, Q.; Huang, S.; Mai, W.; Zhong, X. Trichosanthin-induced autophagy in gastric cancer cell MKN-45 is dependent on reactive oxygen species (ROS) and NF-κB/p53 pathway. J. Pharmacol. Sci. 2016, 131, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Polito, L.; Bortolotti, M.; Pedrazzi, M.; Mercatelli, D.; Battelli, M.G.; Bolognesi, A. Apoptosis and necroptosis induced by stenodactylin in neuroblastoma cells can be completely prevented through caspase inhibition plus catalase or necrostatin-1. Phytomedicine 2016, 23, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Parikh, B.A.; Tumer, N.E. Antiviral activity of ribosome inactivating proteins in medicine. Mini Rev. Med. Chem. 2004, 4, 523–543. [Google Scholar] [CrossRef] [PubMed]
- Duggar, B.M.; Armstrong, J.K. The effect of treating the Virus of Tobacco Mosaic with the juices of various plants. Ann. Missouri Bot. Gard. 1925, 12, 359–366. [Google Scholar] [CrossRef]
- Tomlinson, J.A.; Walker, V.M.; Flewett, T.H.; Barclay, G.R. The inhibition of infection by cucumber mosaic virus and influenza virus by extracts from Phytolacca americana. J. Gen. Virol. 1974, 22, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Ragetli, H.W.J.; Weintraub, M. Purification and characteristics of a virus inhibitor from Dianthus caryophyllus L.: I. Purification and activity. Virology 1962, 18, 232–240. [Google Scholar] [CrossRef]
- Obrig, T.G.; Irvin, J.D.; Hardesty, B. The effect of an antiviral peptide on the ribosomal reactions of the peptide elongation enzymes, EF-I and EF-II. Arch. Biochem. Biophys. 1973, 155, 278–289. [Google Scholar] [CrossRef]
- Foà-Tomasi, L.; Campadelli-Fiume, G.; Barbieri, L.; Stirpe, F. Effect of ribosome-inactivating proteins on virus-infected cells. Inhibition of virus multiplication and of protein synthesis. Arch. Virol. 1982, 71, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Stirpe, F.; Williams, D.G.; Onyon, L.J.; Legg, R.F.; Stevens, W.A. Dianthins, ribosome-damaging proteins with anti-viral properties from Dianthus caryophyllus L. (carnation). Biochem. J. 1981, 195, 399–405. [Google Scholar] [CrossRef] [PubMed]
- FitzGerald, D.J.; Wayne, A.S.; Kreitman, R.J.; Pastan, I. Treatment of hematologic malignancies with immunotoxins and antibody-drug conjugates. Cancer Res. 2011, 71, 6300–6309. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Nakajima, O.; Morishita, R.; Nagai, R. Targeted anticancer immunotoxins and cytotoxic agents with direct killing moieties. Sci. World J. 2006, 6, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Polito, L.; Bortolotti, M.; Maiello, S.; Battelli, M.G.; Bolognesi, A. Plants Producing Ribosome-Inactivating Proteins in Traditional Medicine. Molecules 2016, 21, 1560. [Google Scholar] [CrossRef] [PubMed]
- Guzmán Gutiérrez, S.L.; Reyes-Chilpa, R.; Jaime, H.B. Medicinal plants for the treatment of “nervios”, anxiety, and depression in Mexican Traditional Medicine. Rev. Bras. Farmacogn. 2014, 24, 591–608. [Google Scholar] [CrossRef]
- Argueta, A. Atlas de las Plantas de la Medicina Tradicional Mexicana; Instituto Nacional Indigenista: Ciudad de México, México, 1994; p. 1786.
- Ghogar, A.; Jiraungkoorskul, K.; Jiraungkoorskul, W. Paper Flower, Bougainvillea spectabilis: Update properties of traditional medicinal plant. J. Nat. Remed. 2016, 16, 82–87. [Google Scholar] [CrossRef]
- Murthy, N.S.; Nagarajan, K.; Sastry, A.B. Effect of prophylactic sprays of leaf extracts on the infection of tobacco by tobacco mosaic virus. Indian J. Agric. Sci. 1981, 51, 792–795. [Google Scholar]
- Verma, H.N.; Dwivedi, S.D. Properties of a virus inhibiting agent, isolated from plants which have been treated with leaf extracts from Bougainvillea spectabilis. Physiol. Plant Pathol. 1984, 25, 93–101. [Google Scholar] [CrossRef]
- Bolognesi, A.; Polito, L.; Olivieri, F.; Valbonesi, P.; Barbieri, L.; Battelli, M.G.; Carusi, M.V.; Benvenuto, E.; Del Vecchio Blanco, F.; Di Maro, A.; et al. New ribosome-inactivating proteins with polynucleotide: Adenosine glycosidase and antiviral activities from Basella rubra L. and Bougainvillea spectabilis Willd. Planta 1997, 203, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, L.; Battelli, M.G.; Stirpe, F. Ribosome-inactivating proteins from plants. Biochim. Biophys. Acta 1993, 1154, 237–282. [Google Scholar] [CrossRef]
- Fermani, S.; Tosi, G.; Farini, V.; Polito, L.; Falini, G.; Ripamonti, A.; Barbieri, L.; Chambery, A.; Bolognesi, A. Structure/function studies on two type 1 ribosome inactivating proteins: Bouganin and lychnin. J. Struct. Biol. 2009, 168, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, A.; Polito, L.; Tazzari, P.L.; Lemoli, R.M.; Lubelli, C.; Fogli, M.; Boon, L.; de Boer, M.; Stirpe, F. In vitro anti-tumour activity of anti-CD80 and anti-CD86 immunotoxins containing type 1 ribosome-inactivating proteins. Br. J. Haematol. 2000, 110, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Jaffrézou, J.P.; Levade, T.; Kuhlein, E.; Thurneyssen, O.; Chiron, M.; Grandjean, H.; Carrière, D.; Laurent, G. Enhancement of ricin A chain immunotoxin activity by perhexiline on established and fresh leukemic cells. Cancer Res. 1990, 50, 5558–5566. [Google Scholar] [PubMed]
- Griffin, T.W.; Morgan, A.C.; Blythman, H.E. Immunotoxins therapy: Assessment by animal models. In Immunotoxins; Frankel, A.E., Ed.; Kluwer Academic Publishers: Boston, MA, USA, 1988; pp. 433–455. ISBN 0-89838-984-4. [Google Scholar]
- Den Hartog, M.T.; Lubelli, C.; Boon, L.; Heerkens, S.; Ortiz Buijsse, A.P.; de Boer, M.; Stirpe, F. Cloning and expression of cDNA coding for bouganin. Eur. J. Biochem. 2002, 269, 1772–1779. [Google Scholar] [CrossRef] [PubMed]
- Fabbrini, M.S.; Katayama, M.; Nakase, I.; Vago, R. Plant Ribosome-Inactivating Proteins: Progesses, Challenges and Biotechnological Applications (and a Few Digressions). Toxins 2017, 9, 314. [Google Scholar] [CrossRef] [PubMed]
- Di Maro, A.; Citores, L.; Russo, R.; Iglesias, R.; Ferreras, J.M. Sequence comparison and phylogenetic analysis by the Maximum Likelihood method of ribosome-inactivating proteins from angiosperms. Plant Mol. Biol. 2014, 85, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Kraulis, P.J. MOLSCRIPT: A program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 1991, 24, 946–950. [Google Scholar] [CrossRef]
- Merritt, E.A.; Bacon, D.J. Raster3D: Photorealistic molecular graphics. Methods Enzymol. 1997, 277, 505–524. [Google Scholar] [PubMed]
- Nicholls, A.; Sharp, K.A.; Honig, B. Protein folding and association: Insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins 1991, 11, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Pastan, I.; Hassan, R.; FitzGerald, D.J.; Kreitman, R.J. Immunotoxin treatment of cancer. Annu. Rev. Med. 2007, 58, 221–237. [Google Scholar] [CrossRef] [PubMed]
- Kreitman, R.J. Immunotoxins for targeted cancer therapy. AAPS J. 2006, 8, 532–551. [Google Scholar] [CrossRef] [PubMed]
- Cizeau, J.; Grenkow, D.M.; Brown, J.G.; Entwistle, J.; MacDonald, G.C. Engineering and biological characterization of VB6-845, an anti-EpCAM immunotoxin containing a T-cell epitope-depleted variant of the plant toxin bouganin. J. Immunother. 2009, 32, 574–584. [Google Scholar] [CrossRef] [PubMed]
- Giansanti, F.; Flavell, D.J.; Angelucci, F.; Fabbrini, M.S.; Ippoliti, R. Strategies to Improve the Clinical Utility of Saporin-Based Targeted Toxins. Toxins 2018, 10, 82. [Google Scholar] [CrossRef] [PubMed]
- Vooijs, W.C.; Otten, H.G.; van Vliet, M.; van Dijk, A.J.G.; de Weger, R.A.; de Boer, M.; Bohlen, H.; Bolognesi, A.; Polito, L.; de Gast, G.C. B7-1 (CD80) as target for immunotoxin therapy for Hodgkin’s disease. Br. J. Cancer 1997, 76, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Chaboureau, A.; Ragon, I.; Stibbard, S.; Cizeau, J.; Glover, N.; MacDonald, G.C. Intracellular trafficking of VB6-845, an immunocytotoxin containing a de-immunized variant of bouganin. In Proceedings of the American Association of Cancer Research Annual Meeting, San Diego, CA, USA, 12–16 April 2008. [Google Scholar]
- Entwistle, J.; Brown, J.G.; Chooniedass, S.; Cizeau, J.; MacDonald, G.C. Preclinical evaluation of VB6-845: An anti-EpCAM immunotoxin with reduced immunogenic potential. Cancer Biother. Radiopharm. 2012, 27, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Entwistle, J.; Kowalski, M.; Brown, J.; Cizeau, J.; MacDonald, G.C. The Preclinical and Clinical Evaluation of VB6-845: An Immunotoxin with a De-Immunized Payload for the Systemic Treatment of Solid Tumors. In Antibody-Drug Conjugates and Immunotoxins. Cancer Drug Discovery and Development; Phillips, G., Ed.; Springer: New York, NY, USA, 2013; pp. 349–367. ISBN 978-1-4614-5455-7. [Google Scholar]
- Di Massimo, A.M.; Di Loreto, M.; Pacilli, A.; Raucci, G.; D’Alatri, L.; Mele, A.; Bolognesi, A.; Polito, L.; Stirpe, F.; De Santis, R. Immunoconjugates made of an anti-EGF receptor monoclonal antibody and type 1 ribosome-inactivating proteins from Saponaria ocymoides or Vaccaria pyramidata. Br. J. Cancer 1997, 75, 822–828. [Google Scholar] [CrossRef] [PubMed]
- Ricci, C.; Polito, L.; Nanni, P.; Landuzzi, L.; Astolfi, A.; Nicoletti, G.; Rossi, I.; De Giovanni, C.; Bolognesi, A.; Lollini, P.L. HER/erbB receptors as therapeutic targets of immunotoxins in human rhabdomyosarcoma cells. J. Immunother. 2002, 25, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Qiu, J.; Wang, Z.; Huang, N.; Li, X.; Li, Q.; Zhang, Y.; Zhao, C.; Luo, C.; Zhang, N.; et al. In vitro and in vivo anti-tumor activities of anti-EGFR single-chain variable fragment fused with recombinant gelonin toxin. J. Cancer Res. Clin. Oncol. 2012, 138, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Yip, W.L.; Weyergang, A.; Berg, K.; Tønnesen, H.H.; Selbo, P.K. Targeted delivery and enhanced cytotoxicity of cetuximab-saporin by photochemical internalization in EGFR-positive cancer cells. Mol. Pharm. 2007, 4, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.L.; Chooniedass, S.; Premsukh, A.; Adams, G.P.; Entwistle, J.; MacDonald, G.C.; Cizeau, J. Trastuzumab-deBouganin Conjugate Overcomes Multiple Mechanisms of T-DM1 Drug Resistance. J. Immunother. 2016, 39, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Chooniedass, S.; Dillon, R.L.; Premsukh, A.; Hudson, P.J.; Adams, G.P.; MacDonald, G.C.; Cizeau, J. DeBouganin Diabody Fusion Protein Overcomes Drug Resistance to ADCs Comprised of Anti-Microtubule Agents. Molecules 2016, 21, 1741. [Google Scholar] [CrossRef] [PubMed]
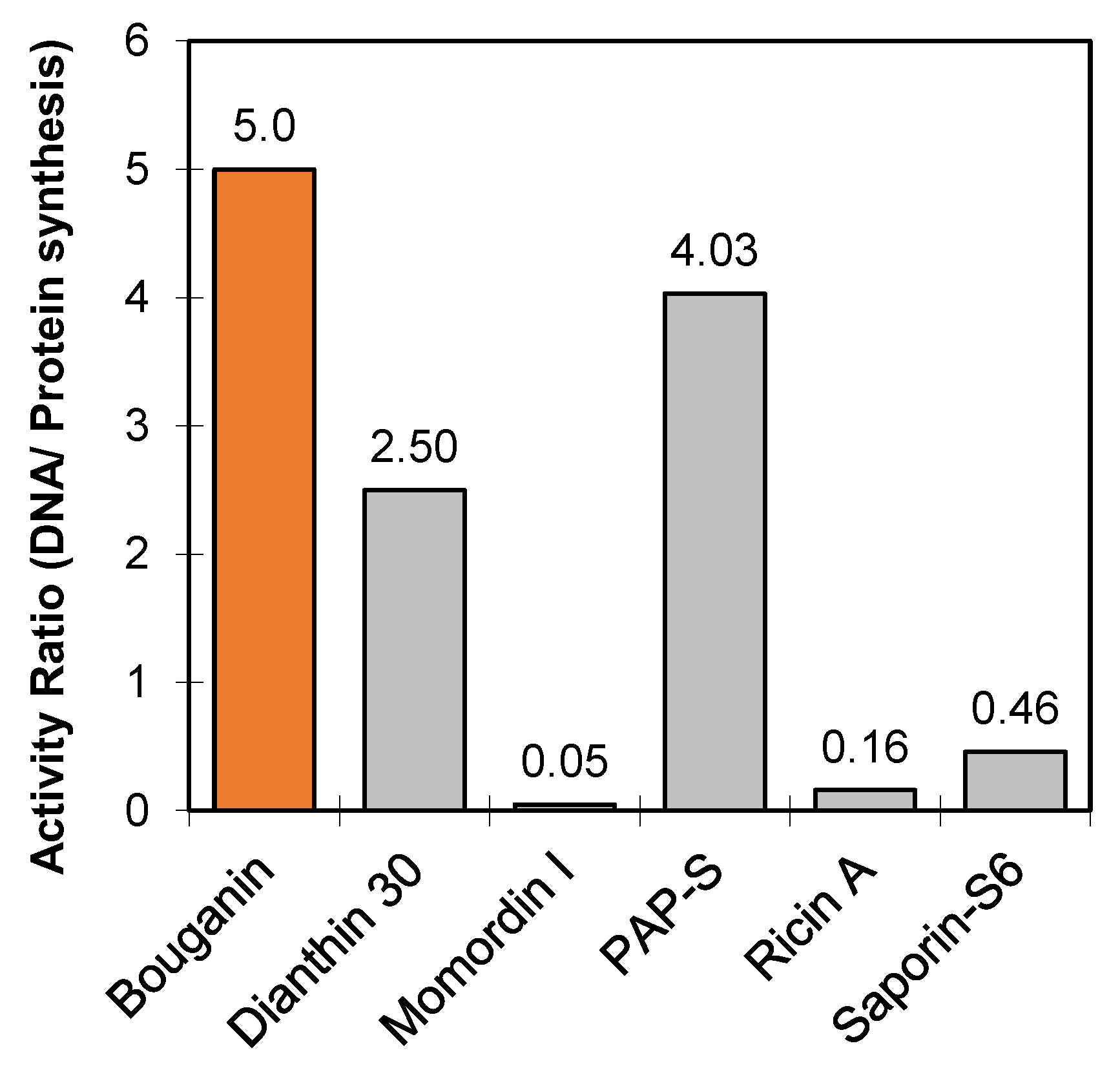

| RIP | Adenine Released | Protein Synthesis | Toxicity | ||
|---|---|---|---|---|---|
| hsDNA 1 pmol | Rat Ribosomes 1 pmol | Cell Free 1 103 U/mg * | Raji Cells 2 IC50 (nM) | Mouse 3 LD50 (mg/kg) | |
| Bouganin | 377.7 | 4.8 | 75 | 839 | >32 4,# |
| Dianthin 30 | 239.9 | 5.7 | 96 | 541 | 14 |
| Momordin I | 27.1 | 3.9 | 526 | n.a. § | 7.4 |
| PAP-S | 503.2 | 5.1 | 125 | n.a. § | 2.6 |
| Ricin A chain | 48.5 | 6.2 | 300 | 200 5 | 16 6 |
| Saporin-S6 | 376.1 | 19.1 | 813 | 23.6 | 4 |
| Bouganin | Carrier | Target | In Vitro Studies | In Vivo Studies | Ref. | ||
|---|---|---|---|---|---|---|---|
| Cell Line | IC50 (pM) * | Animals | Survival Rate | ||||
| native | M24 | CD80 | Raji (Burkitt’s lymphomaL) | 4.61 | n.a. # | n.a. # | [27] |
| native | 1G10 | CD86 | Raji (Burkitt’s lymphomaL) | 129 | n.a. # | n.a. # | [27] |
| de-bouganin | 4D5MOCB | EpCAM | NIH:OVCAR-3 (Ovarian) | 700 | SCID mice/NIH:OVCAR-3 | 100% | [38] |
| de-bouganin | 4D5MOCB | EpCAM | MCF-7 (Breast), NIH:OVCAR-3 (Ovarian) | 400 | HRLN nu/nu/MCF-7 | 70% | [42] |
| de-bouganin | Trastuzumab | HER2 | HCC1954 (Breast) | 45 | CB.17 SCID mice/BT-474 | 83% | [48] |
| de-bouganin | C6.5 diabody | HER2 | HCC202 (Breast) | 22 | n.a. # | n.a. # | [49] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bortolotti, M.; Bolognesi, A.; Polito, L. Bouganin, an Attractive Weapon for Immunotoxins. Toxins 2018, 10, 323. https://doi.org/10.3390/toxins10080323
Bortolotti M, Bolognesi A, Polito L. Bouganin, an Attractive Weapon for Immunotoxins. Toxins. 2018; 10(8):323. https://doi.org/10.3390/toxins10080323
Chicago/Turabian StyleBortolotti, Massimo, Andrea Bolognesi, and Letizia Polito. 2018. "Bouganin, an Attractive Weapon for Immunotoxins" Toxins 10, no. 8: 323. https://doi.org/10.3390/toxins10080323
APA StyleBortolotti, M., Bolognesi, A., & Polito, L. (2018). Bouganin, an Attractive Weapon for Immunotoxins. Toxins, 10(8), 323. https://doi.org/10.3390/toxins10080323







