The History and Applications of Phage Therapy in Pseudomonas aeruginosa
Abstract
:1. Introduction
2. Pseudomonas Aeruginosa
The Problem of Bacterial Resistance in P. aeruginosa
3. Bacteriophages
3.1. History of Phage Therapy
3.2. Phage Structure and Taxonomy
3.3. Phage Infection Process
3.3.1. Nucleic Acid Recognition, Adsorption, and Injection
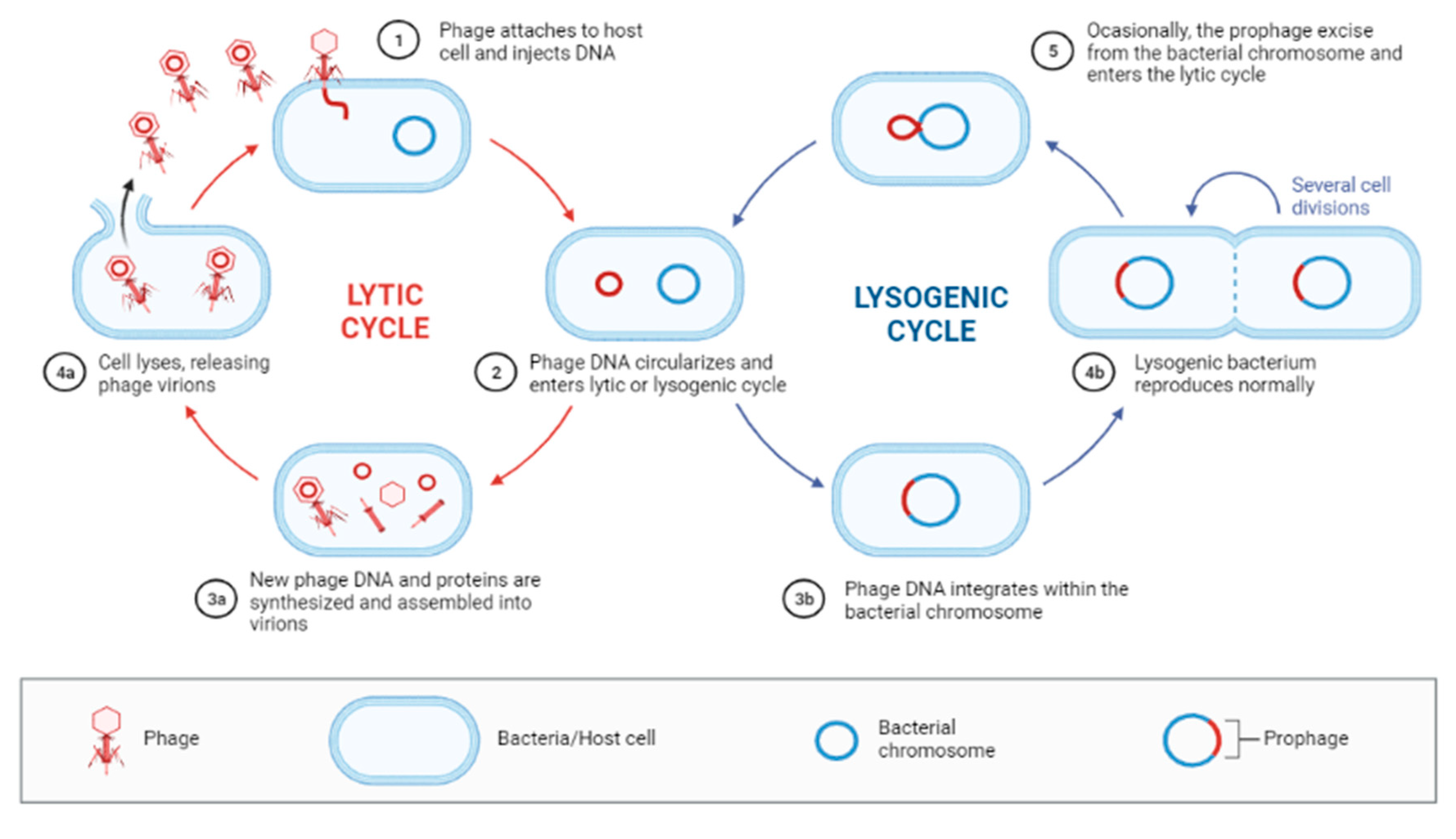
3.3.2. Lytic Cycle
3.3.3. Lysogenic Cycle
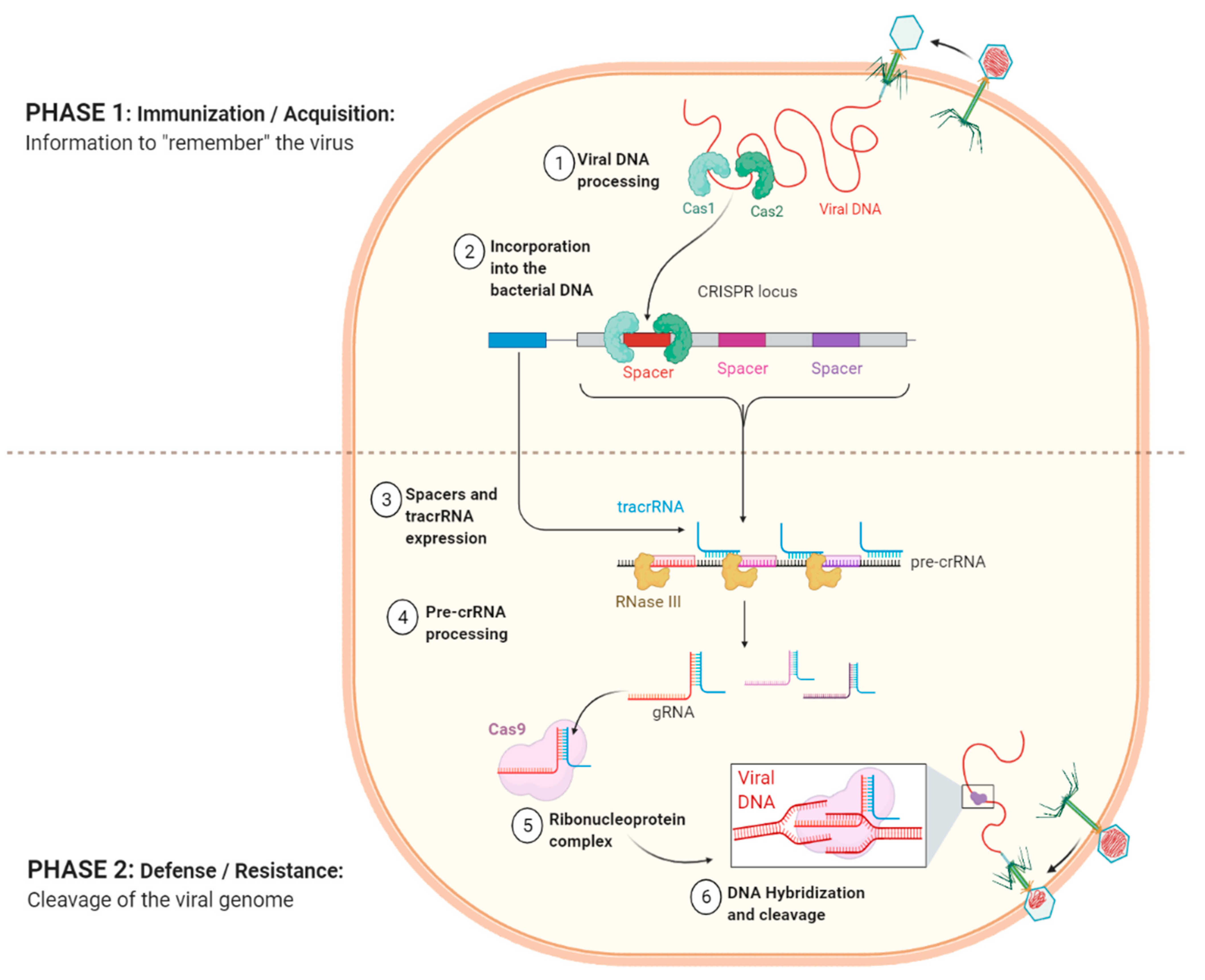
3.4. Phage-Host Interactions
3.5. Advantages and Disadvantages of Phage Therapy
3.6. Phage Therapy Applications
3.7. Phage Therapy and P. aeruginosa, Studies In Vivo and In Vitro
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Romero-Calle, D.; Guimarães Benevides, R.; Góes-Neto, A.; Billington, C. Bacteriophages as Alternatives to Antibiotics in Clinical Care. Antibiotics 2019, 8, 138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Divya Ganeshan, S.; Hosseinidoust, Z. Phage Therapy with a Focus on the Human Microbiota. Antibiotics 2019, 8, 131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, M.; Sun, M.; Huang, D.; Zhang, Z.; Zhang, H.; Zhang, S.; Hu, F.; Jiang, X.; Jiao, W. A review of bacteriophage therapy for pathogenic bacteria inactivation in the soil environment. Environ. Int. 2019, 129, 488–496. [Google Scholar] [CrossRef]
- Kakasis, A.; Panitsa, G. Bacteriophage therapy as an alternative treatment for human infections. A comprehensive review. Int. J. Antimicrob. Agents 2019, 53, 16–21. [Google Scholar] [CrossRef]
- Pires, D.P.; Vilas Boas, D.; Sillankorva, S.; Azeredo, J. Phage Therapy: A Step Forward in the Treatment of Pseudomonas aeruginosa Infections. J. Virol. 2015, 89, 7449–7456. [Google Scholar] [CrossRef] [Green Version]
- Pires, D.P.; Costa, A.R.; Pinto, G.; Meneses, L.; Azeredo, J. Current challenges and future opportunities of phage therapy. FEMS Microbiol. Rev. 2020, 44, 684–700. [Google Scholar] [CrossRef] [PubMed]
- Marques, D.R.A. Prevalência e Susceptibilidade de Isolados Clínicos de Pseudomonas aeruginosa numa unidade hospitalar de Portugal. Ph.D. Thesis, University of Minho, Braga, Portugal, 2015. [Google Scholar]
- Kerr, K.G.; Snelling, A.M. Pseudomonas aeruginosa: A formidable and ever-present adversary. J. Hosp. Infect. 2009, 73, 338–344. [Google Scholar] [CrossRef]
- Hernandez, E.; Segundo, N.V. Los bacteriófagos como una alternativa en el tratamiento de enfermedades infecciosas Bacterianas (Fagoterapia). Rev. Mex. Ciencias Farm. 2010, 41, 17–26. [Google Scholar]
- Pang, Z.; Raudonis, R.; Glick, B.R.; Lin, T.J.; Cheng, Z. Antibiotic resistance in Pseudomonas aeruginosa: Mechanisms and alternative therapeutic strategies. Biotechnol. Adv. 2019, 37, 177–192. [Google Scholar] [CrossRef]
- Pereira, S.G.; Rosa, A.; Ferreira, A.; Moreira, L.; Proença, D.; Morais, P.V.; Cardoso, O. Virulence factors and infection ability of Pseudomonas aeruginosa isolates from a hydropathic facility and respiratory infections. J. Appl. Microbiol. 2014, 116, 1359–1368. [Google Scholar] [CrossRef] [Green Version]
- Loureiro, R.J.; Roque, F.; Teixeira Rodrigues, A.; Herdeiro, M.T.; Ramalheira, E. Use of antibiotics and bacterial resistances: Brief notes on its evolution. Rev. Port. Saúde Publica 2016, 34, 77–84. [Google Scholar]
- Pachori, P.; Gothalwal, R.; Gandhi, P. Emergence of antibiotic resistance Pseudomonas aeruginosa in intensive care unit; a critical review. Genes Dis. 2019, 6, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Moradali, M.F.; Ghods, S.; Rehm, B.H.A. Pseudomonas aeruginosa Lifestyle: A Paradigm for Adaptation, Survival, and Persistence. Front. Cell Infect. Microbiol. 2017, 7, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, R.; Amador, P.; Prudencio, C. β-lactams: Chemical structure, mode of action and mechanisms of resistance. Rev. Med. Microbiol. 2013, 24, 7–17. [Google Scholar] [CrossRef] [Green Version]
- Tagliaferri, T.L.; Jansen, M.; Horz, H.-P. Fighting Pathogenic Bacteria on Two Fronts: Phages and Antibiotics as Combined Strategy. Front. Cell Infect. Microbiol. 2019, 9, 2. [Google Scholar] [CrossRef]
- Wittebole, X.; De Roock, S.; Opal, S.M. A historical overview of bacteriophage therapy as an alternative to antibiotics for the treatment of bacterial pathogens. Virulence 2014, 5, 226–235. [Google Scholar] [CrossRef]
- Sulakvelidze, A.; Alavidze, Z.; Morris, J.G., Jr. Bacteriophage therapy. Antimicrob Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef] [Green Version]
- Haq, I.U.; Chaudhry, W.N.; Akhtar, M.N.; Andleeb, S.; Qadri, I. Bacteriophages and their implications on future biotechnology: A review. Virol. J. 2012, 9, 9. [Google Scholar] [CrossRef] [Green Version]
- Green, S.; Ma, L.; Maresso, A. Phage therapy. Encycl. Microbiol. 2019, 1915, 485–495. [Google Scholar]
- Twort, F.W. An investigation on the nature of ultra-microscopic viruses. Lancet. 1915, 186, 1241–1243. [Google Scholar] [CrossRef] [Green Version]
- Lawrence, D.; Baldridge, M.T.; Handley, S.A. Phages and Human Health: More Than Idle Hitchhikers. Viruses 2019, 11, 587. [Google Scholar] [CrossRef] [Green Version]
- Fruciano, D.E.; Bourne, S. Phage as an antimicrobial agent: d’Herelle’s heretical theories and their role in the decline of phage prophylaxis in the West. Can. J. Infect. Dis. Med. Microbiol. 2007, 18, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Hyman, P.; Abedon, S.T. Bacteriophage: Overview. In Schmidt TMBT-E of M; Fourth, E., Ed.; Oxford Academic Press: Oxford, UK, 2019; pp. 441–457. [Google Scholar] [CrossRef]
- Nikolich, M.P.; Filippov, A.A. Bacteriophage Therapy: Developments and Directions. Antibiotics 2020, 9, 135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCallin, S.; Sacher, J.C.; Zheng, J.; Chan, B.K. Current State of Compassionate Phage Therapy. Viruses 2019, 11, 343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keen, E.C. A century of phage research: Bacteriophages and the shaping of modern biology. Bioessays 2015, 37, 6–9. [Google Scholar] [CrossRef] [PubMed]
- White, H.E. Bacteriophages: Their Structural Organisation and Function. In Bacteriophages-Perspectives and Future; Savva, E.-R., Ed.; IntechOpen: Rijeka, Croatia, 2020. [Google Scholar] [CrossRef] [Green Version]
- Adriaenssens, E.M.; Rodney Brister, J. How to name and classify your phage: An informal guide. Viruses 2017, 9, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chibani, C.M.; Farr, A.; Klama, S.; Dietrich, S.; Liesegang, H. Classifying the Unclassified: A Phage Classification Method. Viruses 2019, 11, 195. [Google Scholar] [CrossRef] [Green Version]
- Koskella, B.; Meaden, S. Understanding bacteriophage specificity in natural microbial communities. Viruses 2013, 5, 806–823. [Google Scholar] [CrossRef] [Green Version]
- Rakhuba, D.V.; Kolomiets, E.I.; Dey, E.; Novik, G.I. Bacteriophage Receptors, Mechanisms of Phage Adsorption and Penetration into Host Cell. Pol. J. Microbiol. 2010, 59, 145–155. [Google Scholar] [CrossRef]
- Chiaruttini, N.; de Frutos, M.; Augarde, E.; Boulanger, P.; Letellier, L.; Viasnoff, V. Is the In Vitro Ejection of Bacteriophage DNA Quasistatic? A Bulk to Single Virus Study. Biophys J. 2010, 99, 447–455. [Google Scholar] [CrossRef] [Green Version]
- Kropinski, A.M. Phage Therapy-Everything Old is New Again. Can. J. Infect. Dis. Med. Microbiol. 2006, 17, 297–306. [Google Scholar] [CrossRef] [PubMed]
- De Smet, J.; Hendrix, H.; Blasdel, B.G.; Danis-Wlodarczyk, K.; Lavigne, R. Pseudomonas predators: Understanding and exploiting phage-host interactions. Nat. Rev. Microbiol. 2017, 15, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Weigele, P.; Raleigh, E.A. Biosynthesis and Function of Modified Bases in Bacteria and Their Viruses. Chem. Rev. 2016, 116, 12655–12687. [Google Scholar] [CrossRef] [PubMed]
- Garneau, J.E.; Dupuis, M.-È.; Villion, M.; Romero, D.A.; Barrangou, R.; Boyaval, P.; Fremaux, C.; Horvath, P.; Magadan, A.H.; Moineau, S. The CRISPR/cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature 2010, 468, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Bondy-Denomy, J.; Pawluk, A.; Maxwell, K.L.; Davidson, A.R. Bacteriophage genes that inactivate the CRISPR/Cas bacterial immune system. Nature 2013, 493, 429–432. [Google Scholar] [CrossRef] [Green Version]
- Le Rhun, A.; Escalera-Maurer, A.; Bratovič, M.; Charpentier, E. CRISPR-Cas in Streptococcus pyogenes. RNA Biol. 2019, 16, 380–389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Principi, N.; Silvestri, E.; Esposito, S. Advantages and limitations of bacteriophages for the treatment of bacterial infections. Front. Pharmacol. 2019, 10, 513. [Google Scholar] [CrossRef] [Green Version]
- Wright, A.; Hawkins, C.H.; Änggård, E.E.; Harper, D.R. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa: A preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef]
- Chan, B.K.; Turner, P.E.; Kim, S.; Mojibian, H.R.; Elefteriades, J.A.; Narayan, D. Phage treatment of an aortic graft infected with Pseudomonas aeruginosa. Evol. Med. Public Health 2018, 2018, 60–66. [Google Scholar] [CrossRef] [Green Version]
- Kutter, E.; De Vos, D.; Gvasalia, G.; Alavidze, Z.; Gogokhia, L.; Kuhl, S.; Abedon, S.T. Phage Therapy in Clinical Practice: Treatment of Human Infections. Curr. Pharm. Biotechnol. 2010, 11, 69–86. [Google Scholar] [CrossRef]
- Górski, A.; Międzybrodzki, R.; Łobocka, M.; Głowacka-Rutkowska, A.; Bednarek, A.; Borysowski, J.; Jończyk-Matysiak, E.; Łusiak-Szelachowska, M.; Weber-Dąbrowska, B.; Bagińska, N.; et al. Phage therapy: What have we learned? Viruses 2018, 10, 288. [Google Scholar] [CrossRef] [Green Version]
- Hoggarth, A.; Weaver, A.; Pu, Q.; Huang, T.; Schettler, J.; Chen, F.; Yuan, X.; Wu, M. Mechanistic research holds promise for bacterial vaccines and phage therapies for Pseudomonas aeruginosa. Drug Des. Dev. Ther. 2019, 13, 909–924. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, W.; Forster, T.; Mayer, O.; Curtin, J.J.; Lehman, S.M.; Donlan, R.M. Bacteriophage cocktail for the prevention of biofilm formation by Pseudomonas aeruginosa on catheters in an in vitro model system. Antimicrob. Agents Chemother. 2010, 54, 397–404. [Google Scholar] [CrossRef] [Green Version]
- Torres-Barceló, C.; Arias-Sánchez, F.I.; Vasse, M.; Ramsayer, J.; Kaltz, O.; Hochberg, M.E. A window of opportunity to control the bacterial pathogen Pseudomonas aeruginosa combining antibiotics and phages. PLoS ONE 2014, 9, e106628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tümmler, B. Emerging therapies against infections with Pseudomonas aeruginosa [version 1; peer review: 2 approved]. F1000Research 2019, 8, 1–14. [Google Scholar] [CrossRef]
- Rose, T.; Verbeken, G.; De Vos, D.; Merabishvili, M.; Vaneechoutte, M.; Lavigne, R.; Jennes, S.; Zizi, M.; Pirnay, J.-P. Experimental phage therapy of burn wound infection: Difficult first steps. Int. J. Burns Trauma. 2014, 4, 66–73. [Google Scholar]
- Markwitz, P.; Olszak, T.; Gula, G.; Kowalska, M.; Arabski, M.; Drulis-Kawa, Z. Emerging Phage Resistance in Pseudomonas aeruginosa PAO1 Is Accompanied by an Enhanced Heterogeneity and Reduced Virulence. Viruses 2021, 13, 1332. [Google Scholar] [CrossRef]
- Tkhilaishvili, T.; Merabishvili, M.; Pirnay, J.P.; Starck, C.; Potapov, E.; Falk, V.; Schoenrath, F. Successful case of adjunctive intravenous bacteriophage therapy to treat left ventricular assist device infection. J. Infect. 2021, 83, e1–e3. [Google Scholar] [CrossRef]
- Antoine, C.; Laforêt, F.; Blasdel, B.; Glonti, T.; Kutter, E.; Pirnay, J.; Mainil, J.; Delcenserie, V.; Thiry, D. Efficacy assessment of PEV2 phage on Galleria mellonella larvae infected with a Pseudomonas aeruginosa dog otitis isolate. Res. Vet. Sci. 2021, 136, 598–601. [Google Scholar] [CrossRef]
- Aghaee, B.L.; Khan Mirzaei, M.; Alikhani, M.Y.; Mojtahedi, A.; Maurice, C.F. Improving the Inhibitory Effect of Phages against Pseudomonas aeruginosa Isolated from a Burn Patient Using a Combination of Phages and Antibiotics. Viruses 2021, 13, 334. [Google Scholar] [CrossRef] [PubMed]
- Krylov, V.; Bourkaltseva, M.; Pleteneva, E.; Shaburova, O.; Krylov, S.; Karaulov, A.; Zhavoronok, S.; Svitich, O.; Zverev, V. Phage phikz—The first of giants. Viruses 2021, 13, 149. [Google Scholar] [CrossRef] [PubMed]
- Alvi, I.A.; Asif, M.; ur Rehman, S. A single dose of a virulent bacteriophage vB PaeP-SaPL, rescues bacteremic mice infected with multi drug resistant Pseudomonas aeruginosa. Virus. Res. 2021, 292, 198250. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, D.; Jabbour, J.F.; Kanj, S.S. Current choices of antibiotic treatment for Pseudomonas aeruginosa infections. Curr. Opin. Infect. Dis. 2020, 33, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Chow, M.Y.; Chang, R.Y.K.; Li, M.; Wang, Y.; Lin, Y.; Morales, S.; McLachlan, A.J.; Kutter, E.; Li, J.; Chan, H.K. Pharmacokinetics and time-kill study of inhaled antipseudomonal bacteriophage therapy in mice. Antimicrob. Agents Chemother. 2021, 65, 1–12. [Google Scholar] [CrossRef]
- Chegini, Z.; Khoshbayan, A.; Taati Moghadam, M.; Farahani, I.; Jazireian, P.; Shariati, A. Bacteriophage therapy against Pseudomonas aeruginosa biofilms: A review. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 1–17. [Google Scholar] [CrossRef]
- McCallin, S.; Sarker, S.A.; Sultana, S.; Oechslin, F.; Brüssow, H. Metagenome analysis of Russian and Georgian Pyophage cocktails and a placebo-controlled safety trial of single phage versus phage cocktail in healthy Staphylococcus aureus carriers. Environ. Microbiol. 2018, 20, 3278–3293. [Google Scholar] [CrossRef]
- Fujiki, J.; Furusawa, T.; Munby, M.; Kawaguchi, C.; Matsuda, Y.; Shiokura, Y.; Nakamura, K.; Nakamura, T.; Sasaki, M.; Usui, M.; et al. Susceptibility of Pseudomonas aeruginosa veterinary isolates to Pbunavirus PB1-like phages. Microbiol. Immunol. 2020, 64, 778–782. [Google Scholar] [CrossRef]
- Shao, X.; Xie, Y.; Zhang, Y.; Liu, J.; Ding, Y.; Wu, M.; Wang, X.; Deng, X. Novel therapeutic strategies for treating Pseudomonas aeruginosa infection. Expert Opin. Drug Discov. 2020, 15, 1403–1423. [Google Scholar] [CrossRef]
- Aslam, S. Bacteriophage therapy as a treatment option for transplant infections. Curr. Opin. Infect. Dis. 2020, 33, 298–303. [Google Scholar] [CrossRef]
- Lin, Y.-W.; Chang, R.Y.K.; Rao, G.G.; Jermain, B.; Han, M.-L.; Zhao, J.; Chen, K.; Wang, J.; Barr, J.J.; Schooley, R.T.; et al. Pharmacokinetics/pharmacodynamics of antipseudomonal bacteriophage therapy in rats: A proof-of-concept study. Clin. Microbiol. Infect. 2020, 26, 1229–1235. [Google Scholar] [CrossRef]
- Peng, H.; Borg, R.E.; Dow, L.P.; Pruitt, B.L.; Chen, I.A. Controlled phage therapy by photothermal ablation of specific bacterial species using gold nanorods targeted by chimeric phages. Proc. Natl. Acad. Sci. USA 2020, 117, 1951–1961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olszak, T.; Danis-Wlodarczyk, K.; Arabski, M.; Gula, G.; Maciejewska, B.; Wasik, S.; Lood, C.; Higgins, G.; Harvey, B.J.; Lavigne, R.; et al. Pseudomonas aeruginosa PA5oct Jumbo Phage Impacts Planktonic and Biofilm Population and Reduces Its Host Virulence. Viruses 2019, 11, 1089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nilsson, A.S. Pharmacological limitations of phage therapy. Upsala J. Med. Sci. 2019, 124, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Tkhilaishvili, T.; Winkler, T.; Müller, M.; Perka, C.; Trampuz, A. Bacteriophages as Adjuvant to Antibiotics for the Treatment of Periprosthetic Joint Infection Caused by Multidrug-Resistant Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2020, 64, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Maddocks, S.; Fabijan, A.P.; Ho, J.; Lin, R.; Ben Zakour, N.L.; Dugan, C.; Kliman, I.; Branston, S.; Morales, S.; Iredell, J.R. Bacteriophage therapy of ventilator-associated pneumonia and empyema caused by Pseudomonas aeruginosa. Am. J. Respir. Crit. Care Med. 2019, 200, 1179–1181. [Google Scholar] [CrossRef]
- Law, N.; Logan, C.; Yung, G.; Furr, C.-L.L.; Lehman, S.M.; Morales, S.; Rosas, F.; Gaidamaka, A.; Bilinsky, I.; Grint, P.; et al. Successful adjunctive use of bacteriophage therapy for treatment of multidrug-resistant Pseudomonas aeruginosa infection in a cystic fibrosis patient. Infection 2019, 47, 665–668. [Google Scholar] [CrossRef]
- Gupta, P.; Singh, H.S.; Shukla, V.K.; Nath, G.; Bhartiya, S.K. Bacteriophage Therapy of Chronic Nonhealing Wound: Clinical Study. Int. J. Low. Extrem. Wounds 2019, 18, 171–175. [Google Scholar] [CrossRef]
- Yuan, Y.; Qu, K.; Tan, D.; Li, X.; Wang, L.; Cong, C.; Xiu, Z.; Xu, Y. Isolation and characterization of a bacteriophage and its potential to disrupt multi-drug resistant Pseudomonas aeruginosa biofilms. Microb. Pathog. 2019, 128, 329–336. [Google Scholar] [CrossRef]
- Abd El-Aziz, A.M.; Elgaml, A.; Ali, Y.M. Bacteriophage Therapy Increases Complement-Mediated Lysis of Bacteria and Enhances Bacterial Clearance after Acute Lung Infection with Multidrug-Resistant Pseudomonas aeruginosa. J. Infect. Dis. 2019, 219, 1439–1447. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.-P.; Que, Y.-A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2019, 19, 35–45. [Google Scholar] [CrossRef]
- Breederveld, R.S. Phage therapy 2.0: Where do we stand? Lancet Infect. Dis. 2019, 19, 2–3. [Google Scholar] [CrossRef]
- Forti, F.; Roach, D.R.; Cafora, M.; Pasini, M.E.; Horner, D.S.; Fiscarelli, E.V.; Rossitto, M.; Cariani, L.; Briani, F.; Debarbieux, L.; et al. Design of a broad-range bacteriophage cocktail that reduces Pseudomonas aeruginosa biofilms and treats acute infections in two animal models. Antimicrob. Agents Chemother. 2018, 62, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uchiyama, J.; Shigehisa, R.; Nasukawa, T.; Mizukami, K.; Takemura-Uchiyama, I.; Ujihara, T.; Murakami, H.; Imanishi, I.; Nishifuji, K.; Sakaguchi, M.; et al. Piperacillin and ceftazidime produce the strongest synergistic phage–antibiotic effect in Pseudomonas aeruginosa. Arch. Virol. 2018, 163, 1941–1948. [Google Scholar] [CrossRef]
- Międzybrodzki, R.; Borysowski, J.; Kłak, M.; Jończyk-Matysiak, E.; Obmińska-Mrukowicz, B.; Suszko-Pawłowska, A.; Bubak, B.; Weber-Dąbrowska, B.; Górski, A. In Vivo Studies on the Influence of Bacteriophage Preparations on the Autoimmune Inflammatory Process. BioMed Res. Int. 2017, 2017, 3612015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villarroel, J.; Larsen, M.V.; Kilstrup, M.; Nielsen, M. Metagenomic Analysis of Therapeutic PYO Phage Cocktails from 1997 to 2014. Viruses 2017, 9, 328. [Google Scholar] [CrossRef] [Green Version]
- Fong, S.A.; Drilling, A.; Morales, S.; Cornet, M.E.; Woodworth, B.A.; Fokkens, W.J.; Psaltis, A.J.; Vreugde, S.; Wormald, P.-J. Activity of Bacteriophages in Removing Biofilms of Pseudomonas aeruginosa Isolates from Chronic Rhinosinusitis Patients. Front. Cell Infect. Microbiol. 2017, 7, 418. [Google Scholar] [CrossRef] [Green Version]
- Shiley, J.R.; Comfort, K.K.; Robinson, J.B. Immunogenicity and antimicrobial effectiveness of Pseudomonas aeruginosa specific bacteriophage in a human lung in vitro model. Appl. Microbiol. Biotechnol. 2017, 101, 7977–7985. [Google Scholar] [CrossRef] [PubMed]
- Trend, S.; Fonceca, A.M.; Ditcham, W.G.; Kicic, A.C.F.; Cf, A. The potential of phage therapy in cystic fibrosis: Essential human-bacterial-phage interactions and delivery considerations for use in Pseudomonas aeruginosa-infected airways. J. Cyst. Fibros. 2017, 16, 663–670. [Google Scholar] [CrossRef] [Green Version]
- Roach, D.R.; Leung, C.Y.; Henry, M.; Morello, E.; Singh, D.; Di Santo, J.P.; Weitz, J.S.; Debarbieux, L. Synergy between the host immune system and bacteriophage is essential for successful phage therapy against an acute respiratory pathogen. Cell Host Microbe 2017, 22, 38–47.e4. [Google Scholar] [CrossRef]
- Waters, E.M.; Neill, D.; Kaman, B.; Sahota, J.S.; Clokie, M.R.J.; Winstanley, C.; Kadioglu, A. Phage therapy is highly effective against chronic lung infections with Pseudomonas aeruginosa. Thorax 2017, 72, 666–667. [Google Scholar] [CrossRef] [Green Version]
- Al-Wrafy, F.; Brzozowska, E.; Górska, S.; Gamian, A. Pathogenic factors of Pseudomonas aeruginosa-the role of biofilm in pathogenicity and as a target for phage therapy. Adv. Hyg. Exp. Med. Hig. Med. Dosw. 2017, 71. [Google Scholar] [CrossRef] [PubMed]
- Stratton, C.W. Phages, fitness, virulence, and synergy: A novel approach for the therapy of infections caused by Pseudomonas aeruginosa. J. Infect. Dis. 2017, 215, 668–670. [Google Scholar] [CrossRef] [PubMed]
- Oechslin, F.; Piccardi, P.; Mancini, S.; Gabard, J.; Moreillon, P.; Entenza, J.M.; Resch, G.; Que, Y.-A. Synergistic interaction between phage therapy and antibiotics clears Pseudomonas aeruginosa infection in endocarditis and reduces virulence. J. Infect. Dis. 2017, 215, 703–712. [Google Scholar] [CrossRef] [Green Version]
- Høyland-Kroghsbo, N.M.; Paczkowski, J.; Mukherjee, S.; Broniewski, J.; Westra, E.; Bondy-Denomy, J.; Bassler, B.L. Quorum sensing controls the Pseudomonas aeruginosa CRISPR-Cas adaptive immune system. Proc. Natl. Acad. Sci. USA 2017, 114, 131–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furusawa, T.; Iwano, H.; Hiyashimizu, Y.; Matsubara, K.; Higuchi, H.; Nagahata, H.; Niwa, H.; Katayama, Y.; Kinoshita, Y.; Hagiwara, K.; et al. Phage therapy is effective in a mouse model of bacterial equine keratitis. Appl. Environ. Microbiol. 2016, 82, 5332–5339. [Google Scholar] [CrossRef] [Green Version]
- Danis-Wlodarczyk, K.; Vandenheuvel, D.; Jang, H.B.; Briers, Y.; Olszak, T.; Arabski, M.; Wąsik, S.; Drabik, M.; Higgins, G.; Tyrrell, J.; et al. A proposed integrated approach for the preclinical evaluation of phage therapy in Pseudomonas infections. Sci Rep. 2016, 6, 1–13. [Google Scholar]
- Latz, S.; Wahida, A.; Arif, A.; Häfner, H.; Hoß, M.; Ritter, K.; Horz, H.P. Preliminary survey of local bacteriophages with lytic activity against multi-drug resistant bacteria. J. Basic Microbiol. 2016, 56, 1117–1123. [Google Scholar] [CrossRef]
- Khawaja, K.A.; Rauf, M.; Abbas, Z.; Rehman, S. A virulent phage JHP against Pseudomonas aeruginosa showed infectivity against multiple genera. J. Basic Microbiol. 2016, 56, 1090–1097. [Google Scholar] [CrossRef] [PubMed]
- Sagona, A.P.; Grigonyte, A.M.; MacDonald, P.R.; Jaramillo, A. Genetically modified bacteriophages. Integr. Biol. 2016, 8, 465–474. [Google Scholar] [CrossRef] [Green Version]
- Cafora, M.; Deflorian, G.; Forti, F.; Ferrari, L.; Binelli, G.; Briani, F.; Ghisotti, D.; Pistocchi, A. Phage therapy against Pseudomonas aeruginosa infections in a cystic fibrosis zebrafish model. Sci. Rep. 2019, 9, 1527. [Google Scholar] [CrossRef] [Green Version]
- Jang, H.-J.; Bae, H.-W.; Cho, Y.-H. Exploitation of Drosophila Infection Models to Evaluate Antibacterial Efficacy of Phages BT-Bacteriophages: Methods and Protocols; Clokie, M.R.J., Kropinski, A., Lavigne, R., Eds.; Springer: New York, NY, USA, 2019; pp. 183–190. [Google Scholar] [CrossRef]
- Chang, R.Y.K.; Wallin, M.; Kutter, E.; Morales, S.; Britton, W.; Li, J.; Chan, H.-K. Storage stability of inhalable phage powders containing lactose at ambient conditions. Int. J. Pharm. 2019, 560, 11–18. [Google Scholar] [CrossRef] [PubMed]

| Form | Nucleic Acid | Order | Family | Details | Examples |
|---|---|---|---|---|---|
| With tail | dsDNA (L) | Caudovi-rales | Myoviridae | Contractile tail | T4, Um, P1, P2 |
| Siphoviri-dae | Long tail, not contractile | Λ, T5, HK97, N15 | |||
| Podoviri-dae | Short tail | T7, T3, φ29, P22 | |||
| Polyhedral | ssDNA (C) | Petitvira-les | Microviri-dae | Capsomers conspicuous | PM2 |
| dsDNA (C,S) | Linavira-les | Corticovi-ridae | Complex capsid, lipids | PRD1 | |
| dsDNA (L) | Kalamavi-rales | Tectiviri-dae | Double capsid, lipids, pseudo-tail | PRD1 | |
| dsDNA (L) | SH1 * | Double capsid, lipids | SH1 | ||
| dsDNA (C) | STIV * | Tower-shaped protrusions | STIV | ||
| ssRNA (L) | Levivira-les | Leviviridae | Like poliovirus | MS2 | |
| dsRNA (L, M) | Mindivi-rales | Cystoviri-dae | Envelope, lipids | Φ6 | |
| Filamentous | ssDNA (C) | Tubulavi-rales | Inoviridae | Long filamentous, short stem | M13 |
| dsDNA (L) | Ligamen-virales | Lipothixvi-ridae | Envelope, lipids | TTV1 | |
| dsDNA (L) | Rudiviro-dae | Rigid rods type, TMV | SIRV-1 | ||
| Pleomorphic | dsDNA (C,S) | Plasmavi-ridae | Envelope, without lipid capsid | L2 | |
| dsDNA (C, S) | Fusellovi-ridae | Lemon shape, envelope | SSV1 | ||
| dsDNA (L,S) | ** | Lemon shape, envelope | His1 | ||
| dsDNA (C, S) | Guttaviri-dae | Drop shape | SNDV | ||
| dsDNA (L) | Ampulla-viridae * | Bottle shape, NC helical | ABV |
| Advantages | Description |
|---|---|
| Specificity | Very specific to the host. infections with more than one host, it is necessary to use a phage cocktail |
| Bactericidal versus bacteriostatic | Lytic phages infect target host bacteria and cause cell death, compared to certain bacteriostatic antibiotics |
| Active on-site propagation | Phages increase the concentration in the host as they replicate, theoretically requiring only one therapeutic dose |
| Low inherent toxicity | Phages exist in large amounts in the biosphere, it is possible to isolate and purify the phage required to achieve certain bacteria. |
| Formulation and application versatility | Various phages can be converted in a cocktail to target several bacteria simultaneously. The type of administration can also vary, liquid, powder, ointment, tablets. |
| Narrow potential for antibiotic cross-resistance | Since the phage and bacterial resistance mechanisms are different, bacteria resistant to certain antibiotics can be treated with the use of phage therapy. |
| Biofilm clearance | Phages can penetrate through biofilms. Part of this capacity is due to the presence of depolymerases and lysins |
| Relatively low discovery and production cost | The costs associated with discovering phage isolation and purification are relatively low |
| Low environmental impact | Phages are natural components of the environment |
| Study | Subject | Pathology | Bact Strain | Phage | Duration | Outcome |
|---|---|---|---|---|---|---|
| Chang, R.Y.K. [93] | Lung alveolar macrophage and epithelial cells | Cystic fibrosis | PAV237 | PEV1, PEV20 and PEV61 | 24 h | Significantly reduces the multidrug-resistant (MDR) P. aeruginosa burden in mouse lungs. |
| Fong, S.A. [73] | 44 isolates Pseudomonas aeruginosa from humans | Chronic rhinosinusi-tis | PAO1 | Pa 193, Pa 204, Pa 222, Pa 223 | 48 h | A single dose of phages is able to significantly reduce biofilms formed in vitro. |
| Shiley, J.R. [74] | Lung cell A549 and U937 monocyte | Cancer | PAO1-WT, pilin lacking PAO1-NP | DMS3, PEV2 | 24 h | DMS3 and PEV2 were able to inhibit bacterial growth in a PAO1-WT and PAO1-NP infection model, respectively. |
| Aghaee, B.L. [88] | Human | Burn wound infection | P. aeruginosa | vB_PaeM_GUMS6, vB_PaeM_GUMS32, vB_PaeM_GUMS45 | 4, 8, 12, 24 and 48 h | The combination of two phage-antibiotics had the highest inhibition efficiency against the P. aeruginosa strain. The phages tested showed low stability at high temperatures, acid pH values, and in both lotions. |
| Study | Subject | Participants | Age | Pathology | Bacterial Strain | Phage | Duration | Outcome |
|---|---|---|---|---|---|---|---|---|
| Antoine, C. [82] | Galleria mellonella | 450 Larvae | N.D. | Canine otitis | PAV237 | PEV2 | 24, 48 and 72 h | Despite the ineffectiveness as life-saving antimicrobials in this model, PEV2 is active against P. aeruginosa in G. mellonella, even if this did not result in the complete elimination of bacteria at the tested MOIs. |
| Olszak, T. [60] | Galleria mellonella | Larvae | N.D. | N.D. | PAO1 | PA5oct Jumbo phage | 8, 18, 24, 48 and 72 h | The use of a phage mixture targeting different extracellular structures as receptors causes a huge impact on bacterial fitness, virulence, and pathogenicity. |
| Cafora, M. [94] | Danio rerio | Embryos | N.D. | Cystic fibrosis (CF) | PAO1 | vB_PaeP_PYO2, vB_PaeP_DEV, vB_PaeM_E215, vB_PaeM_E217 | 20, 24, 26, 48 hpf (to block the embryo pigmentation from 24 h post fertilization) | The lethality and bacterial burden were both decreased following phage therapy application. |
| Jang, H.J. [95] | Drosophila melanogaster | 15–30 M/F | 5–7 days | PA infection | PAO1 | MPK1,MPK6,D3112, and PP7 | 48 h | N.D. |
| Alvi, I.A. [90] | Mice | 81 F | 6–8 weeks | CF | PAO1 | vB PaeP- SaPL | 1, 6, 12, 24, 48 and 96 h | The majority of tested MDR P. aeruginosa isolates are highly susceptible to SaPL, which inhibits their growth for longer time. SaPL is a potential candidate that can be used in phage therapy against MDR P. aeruginosa infections. |
| Lin, Y. [92] | Mice | F | 8–10 weeks | CF, bronchiectasis and chronic obstructive pulmonary disease (COPD) | FADD1-PA001 | PEV20 | N.D. | Co-spray dried phage PEV20 and ciprofloxacin combination powder showed synergistic bacterial killing effect compared with single PEV20 or ciprofloxacin powders in an acute mouse lung infection model caused by P. aeruginosa. |
| Chow, M.Y.T. [93] | Mice | F | 6–8 weeks | Acute respiratory infection | FADDI-PA001 | PEV31 | 26 h | Pulmonary delivery of phage PEV31 in mice can reduce the MDR bacterial burden. |
| Lin, Y.W. [55] | Rats | 17 (14F; 3M) | 8 weeks | CF | Pandrug-resistant (PDR) isolate P. aeruginosa 112 and P. aeruginosa ADDG. | øPEV20 | 48–72 h | This studied showed positive outcomes in the utilization of PEV20 phage, in Rats, with simullation results predicting an infection erradication 12 h after the treatment, but some inconsistencie in in vivo models. |
| Abd El-Aziz, A.M. [65] | Mice | Three groups of F mice (15 each) | 10-week-old | Lung infection | P. aeruginosa clinical strains | MMI-Ps1 | 72 h post infection | In vitro and animal model studies showed that MMI-Ps1 effectively killed different serotypes of P. aeruginosa, including mucoid and nonmucoid strains. In addition, phage therapy significantly protected mice from lung and bloodstream infection caused by mucoid strains of P. aeruginosa. |
| Międzybrodzki, R. [70] | Mice | N.D. | 8–16 weeks at the begin-ning | Murine collagen-induced arthritis | P. aeruginosa 119x | 119x phage | 56 days | Preliminary results do not suggest that phages may aggravate the symptoms of rheumatoid arthritis |
| Chang, R.Y.K. [95] | Mice | 4 | 8 to 10 weeks | Lung infection | FADDI-PA001 | Phage PEV20 | 24 h | The study showed that intratracheally delivered aerosolized phage powder can significantly reduce MDR P. aeruginosa load in mouse lungs. |
| Roach, D.R. [76] | Mice | 6 | N.D. | CF, acute pneumonia | P. aeruginosa PAKlumi | PAK_P1 | 14 days | The study shows that successful phage curative and pro-phylactic therapies of acute respiratory infections depend on an essential synergy between phage lysis and the host’s own immune defenses. |
| Waters, E.M. [77] | Mice | 60 | 6–8 weeks | CF, COPD | P. aeruginosa LESB65 and NP22_2 | Phage PELP20 | 48–72 h and 7 days depending on the treatment. | The study shows that phage administered intranasally up to 6 days after establishment of chronic lung infection were efficient in reducing bacterial numbers in the lungs of mice infected with P. aeruginosa, demonstrating the potential for phage therapy in the treatment of established and recalcitrant chronic respiratory tract infections. |
| Stratton, C.W. [79] | Mice | Rodents | N.D. | Catheter-induced rat aortic valve endocarditis | P. aeruginosa strain CHA | Cocktail of 12 phages agaisnt P. aeruginosa | 6 h | This study provide a strong proof of concept for the use of phage cocktails combined with antimicrobial agents for therapy of deep-seated and systemic infections. |
| Oechslin, F. [51] | Mice | Rodents | N.D. | Induced rat aortic valve endocarditis | 33 strains of P. aeruginosa reference strains and clinical isolates | 12 phages contained in the PP1131 cocktail | 18 h | Single-dose phage therapy was active against P. aeruginosa EE and highly synergistic with ciprofloxacin. Phage-resistant mutants had impaired infectivity. |
| Furusawa, T. [83] | Mice, horse | Rodents | N.D. | Bacterial keratitis | Pseudomonas sp. (Pa12, Pa18, Pa26, and Pa50) and P. aeruginosa strain NE-126 | ΦR18 and ΦS12-1 | 3 h | The phages used in this study rapidly adsorbed to P. aeruginosa and widely killed these isolates from horse lesions. One of the phages was shown to completely prevent keratitis in a keratitis mouse model. |
| Danis-Wlodarczyk, K. [84] | Wax moth | Larvae | N.D. | Infection | Pseudomonas aeruginosa (PAO1) | KTN4 phage | 36 h | Considering all these characteristics, KTN4 phage is a suitable and promising for applications in treatment and prophylaxis in lung infections. |
| Forti, F. [94] | Mice and wax moth | F | 8–10 weeks | Acute respiratory infection (mice) and bacteremia (wax moth) | PAO1; PA14; PAK-lumi; LESB58; AG5; AG3; AA10; GJY9; CL1; CL2; VR8; AG6; DV4; CH7; AA2; AA43; AA44; TR1;TR66; TR67; E1 to E9 and PaPh1 a PaPh33. | Cocktail composed of four novel phages (PYO2, DEV, E215 and E217) and two previously characterized phages (PAK_P1 and PAK_P4) | After two hours post-infection (P.I.) (mice); 1 h P.I. (wax moth) | Our findings indicate that in mice, lethal acute respiratory infection can be cured by treatment with the cocktail. Compared to our previous data obtained with a single phage, the cocktail showed the advantage of having more rapid efficacy in reducing the bacterial load. |
| Study | Subject | Participants | Age | Pathology | P. aeruginosa Strain | Phage | Duration | Outcome |
|---|---|---|---|---|---|---|---|---|
| Tkhila-ishvili [71] | Human | 1F | 83 years old | Periprosthetic joint infection | Clinical isolate of P. aeruginosa | N.D. | 5 days | The wound healed and the patient was discharged after 14 days with oral antibiotics for six weeks. The patient died four months after bacteriophage treatment due to left ventricular assist device pump thrombosis. |
| Madd-ocks, S. [61] | Human | 1F | 77 years old | Adenocarcinoma | Clinical isolate of P. aeruginosa | Lytic anti-P.aeruginosa phages | 7 days | Intravenous antibiotics, bacteriophage therapy was well tolerated, with no adverse events detected either during therapy or subsequently. In combination with antimicrobials, bacteriophage therapy was associated with resolution of infection and with apparent eradication of Pseudomonas colonization. The successful decolonization would have occurred in the absence of phage treatment is unlikely, given the extent of disease and the sequential development of antimicrobial resistance. |
| Aslam, S. [62] | Human | 3 (2F;1M) | 51 years old | Lung transplant | Clinical isolate of P. aeruginosa among other strains | PA193; Pa204;Pa222;Pa223;Pa176; Paϕ1; PaSKWϕ17,PaSKWϕ22; PaATFϕ1; PaATFϕ3; BdPF16phi428. | 60 days | Bacteriophage therapy was well tolerated and associated with clinical improvement when used as an adjunct to antibiotics in lung transplant recipients with against multidrug-resistant respiratory infections otherwise not responsive to antibiotics alone. |
| Law, N. [69] | Human | 1M | 26 years old | Cystic fibrosis | Clinical isolate of P. aeruginosa | Cocktail AB-PAo1 | 9 months | Given the concern for multidrug-resistant P. aeruginosa infections in cystic fibrosis patients, bacteriophage therapy may offer a viable anti-infective adjunct to traditional antibiotic therapy. |
| Gupta, P. [63] | Human | 20 | 36 years old | Nonhealing wound | Clinical isolate of P. aeruginosa among other strains | Cocktail from water sources | 21 days | A significant improvement was observed in the wound healing, and there were no signs of infection clinically and microbiologically after 3 to 5 doses of topical bacteriophage therapy. Seven patients achieved complete healing on day 21 during follow up while in others healthy margins and healthy granulation tissue were observed. |
| Jault, P. [66] | Human | 27 | 18 years or older | Burn wound infection | Clinical isolate of P. aeruginosa | PP1131 | 8 days | Clinically relevant reduction in bacterial burden was observed in the phage group, with numerically fewer serious adverse events seen in those treated with the phage cocktail than the standard of care, indicating a favourable potential of phage therapy. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, C.; Sá, S.; Guedes, C.; Oliveira, C.; Lima, C.; Oliveira, M.; Mendes, J.; Novais, G.; Baylina, P.; Fernandes, R. The History and Applications of Phage Therapy in Pseudomonas aeruginosa. Microbiol. Res. 2022, 13, 14-37. https://doi.org/10.3390/microbiolres13010002
Silva C, Sá S, Guedes C, Oliveira C, Lima C, Oliveira M, Mendes J, Novais G, Baylina P, Fernandes R. The History and Applications of Phage Therapy in Pseudomonas aeruginosa. Microbiology Research. 2022; 13(1):14-37. https://doi.org/10.3390/microbiolres13010002
Chicago/Turabian StyleSilva, Carina, Sara Sá, Carla Guedes, Carla Oliveira, Cláudio Lima, Marco Oliveira, João Mendes, Gonçalo Novais, Pilar Baylina, and Ruben Fernandes. 2022. "The History and Applications of Phage Therapy in Pseudomonas aeruginosa" Microbiology Research 13, no. 1: 14-37. https://doi.org/10.3390/microbiolres13010002
APA StyleSilva, C., Sá, S., Guedes, C., Oliveira, C., Lima, C., Oliveira, M., Mendes, J., Novais, G., Baylina, P., & Fernandes, R. (2022). The History and Applications of Phage Therapy in Pseudomonas aeruginosa. Microbiology Research, 13(1), 14-37. https://doi.org/10.3390/microbiolres13010002









