Atypical Streptococcal Meningitis with Fatal Cerebrovascular Complications: A Case Report
Abstract
1. Introduction
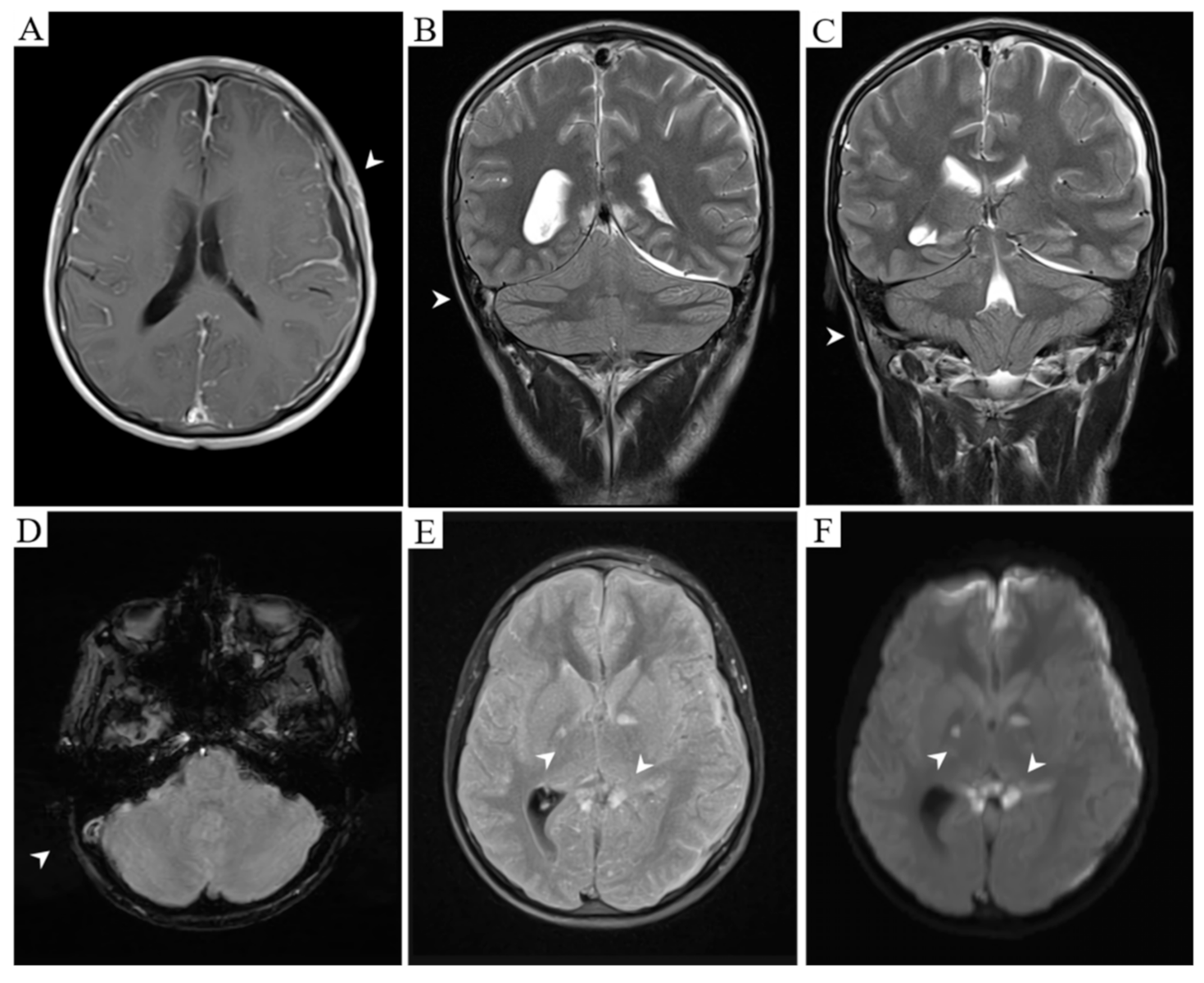
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Ethics Approval
Conflicts of Interest
Appendix A. Full References of Case Reports Included in Table 1
- Myint S. Cavernous sinus thrombosis due to Streptococcus milleri. J Infect. 1986;13:202–3.
- Pearson CR, Riden DK, Garth RJ, Thomas MR. Two cases of lateral sinus thrombosis presenting with extracranial head and neck abscesses. J Laryngol Otol. 1994;108:779–82.
- Blaser B, Greusing B, Häusler R. Bezold’s abscess with wide extension to the lateral skull base. Schweiz Med Wochenschr. 2000;Suppl 125:23S–26S.
- Benhayoun M, Llor J, Van-Den-Abbeele T, Elmaleh M, Mariani P, Beaufils F, et al. Bilateral jugular thrombosis in Lemierre syndrome. Arch Pediatr. 2003;10:1071–4.
- Chang W-N, Chen S-D, Lui C-C, Huang C-R, Lu C-H. Septic cavernous sinus thrombosis due to Streptococcus constellatus infection. J Formos Med Assoc. 2003;102:733–6.
- Watkins LM, Pasternack MS, Banks M, Kousoubris P, Rubin PAD. Bilateral cavernous sinus thromboses and intraorbital abscesses secondary to Streptococcus milleri. Ophthalmology. 2003;110:569–74.
- Ching H-Y, Ramsden JD, Bottrill I. A unique presentation: Bezold’s abscess and glomerulonephritis. Eur J Pediatr. 2006;165:569–70.
- Hoshino C, Satoh N, Sugawara S, Kuriyama C, Kikuchi A, Ohta M. Septic cavernous sinus thrombosis complicated by narrowing of the internal carotid artery, subarachnoid abscess and multiple pulmonary septic emboli. Intern Med. 2007;46:317–23.
- Pavlovich P, Looi A, Rootman J. Septic thrombosis of the cavernous sinus: two different mechanisms. Orbit. 2006;25:39–43.
- Goawalla A, Mansell N, Pearson A. Septic cavernous sinus thrombosis with bilateral secondary orbital infection. Orbit. 2007;26:113–6.
- Udaondo P, Garcia-Delpech S, Díaz-Llopis M, Salom D, Garcia-Pous M, Strottmann JM. Bilateral intraorbital abscesses and cavernous sinus thromboses secondary to Streptococcus milleri with a favorable outcome. Ophthal Plast Reconstr Surg. 2008;24:408–10.
- Jones RG, Arnold B. Sudden onset proptosis secondary to cavernous sinus thrombosis from underlying mandibular dental infection. BMJ Case Rep. 2009;2009:bcr0320091671.
- Gonzalez L, Holznecth C, Apoorv B. Not a typical sinus infection. In: ACP Clinical Vignette Presentations. Medical College of Wisconsin; 2012. p. 1–22.
- Imholz B, Becker M, Lombardi T, Scolozzi P. Septic thrombosis of the cavernous sinus secondary to a Streptococcus milleri oral infection. Dentomaxillofac Radiol. 2012;41:525–8.
- Noel N, Pandit R, Kale T, Jacobs N. A 13-year-old male with ptosis. Pediatr Ann. 2013;42:61–3.
- Sakaida H, Kobayashi M, Ito A, Takeuchi K. Cavernous sinus thrombosis: linking a swollen red eye and headache. Lancet. 2014;384:928.
- Suzuki Y, Ogawa K, Oishi M, Kamei S. A case of Streptococcus intermedius-induced subdural abscess and left transverse sinus thrombosis occurring subsequent to treatment for gingivitis. Neurology Asia. 2014;19:405–7.
- Karssemakers LHE, Forouzanfar T, Schulten EAJM, Karagozoglu KH. Bilateral conjunctival swelling after dental extraction. Lancet Infect Dis. 2015;15:746.
- Selvitop O, Poretti A, Huisman TA, Wagner MW. Cerebral sinovenous thrombosis in a child with Crohn’s disease, otitis media, and meningitis. Neuroradiol J. 2015;28:274–7.
- Shams PN, Policeni B, Carter KD, Shriver E, Thurtell MJ. Bilateral septic cavernous sinus thrombosis, congestive orbitopathy, and ischemic optic neuropathy. Can J Ophthalmol. 2016;51:e75–7.
- Allegrini D, Reposi S, Nocerino E, Pece A. Odontogenic orbital cellulitis associated with cavernous sinus thrombosis and pulmonary embolism: a case report. J Med Case Rep. 2017;11:164.
- López F, Santamarta E, Martínez P, Sáiz-Ayala A, Llorente JL. Cavernous sinus thrombosis during pregnancy. Auris Nasus Larynx. 2017;44:232–6.
- Martel A. Septic thrombosis of cavernous sinus extended to the ipsilateral internal jugular vein and transversal sinus with favorable outcome: Clinical and radiological features of a Lemierre syndrome. Orbit. 2018;37:94–6.
- Branson SV, McClintic E, Yeatts RP. Septic cavernous sinus thrombosis associated with orbital cellulitis: A report of 6 cases and review of literature. Ophthal Plast Reconstr Surg. 2018.
- Deliran S, Sondag L, Leijten Q, Tuladhar A, Meijer F. Hoofdpijn: denk aan sinus-cavernosus tromboflebitis. Ned Tijdschr Geneeskd. 2018;162:D2907.
- Mo S, Wei L, Chen H, Li R, Li S, Luo G. A Chinese case of Prevotella intermedia and Streptococcus constellatus intracranial mixed infection. Metab Brain Dis. 2018;33:161–6.
References
- Institute for Health Metrics and Evaluation. Global Burden of Disease (GBD) Results Tool. Seattle: University of Washington, 2017. Available online: http://ghdx.healthdata.org/gbd-results-tool (accessed on 20 December 2019).
- Instituto Nacional de Salud. Boletín Epidemiológico Semanal: Semana epidemiológica 15. In Bogotá D.C.: Ministerio de Salud de Colombia; 2019. Available online: http://www.ins.gov.co/buscador-eventos/BoletinEpidemiologico/2019_Boletín_epidemiológico_semana_15.pdf (accessed on 20 December 2019).
- Zunt, J.R.; Kassebaum, N.J.; Blake, N.; Glennie, L.; Wright, C.; Nichols, E.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; Adamu, A.A.; et al. Global, regional, and national burden of meningitis, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 1061–1082. [Google Scholar] [CrossRef]
- Hughes, D.C.; Raghavan, A.; Mordekar, S.R.; Griffiths, P.D.; Connolly, D.J.A. Role of imaging in the diagnosis of acute bacterial meningitis and its complications. Postgrad. Med J. 2010, 86, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Julayanont, P.; Ruthirago, D.; Detoledo, J.C. Bacterial meningitis and neurological complications in adults. Southwest Respir. Crit. Care Chron. 2016, 4, 5–16. [Google Scholar]
- Gideon Online. Cerebrospinal meningitis. Worldwide diseases distribution. Los Angeles: GIDEON Informatics. 2019. Available online: http://web.gideonoline.com/web/epidemiology/index.php?disease=11480&country=G100# (accessed on 20 December 2019).
- Yau, B.; Hunt, N.H.; Mitchell, A.J.; Too, L.K. Blood‒Brain Barrier Pathology and CNS Outcomes in Streptococcus pneumoniae Meningitis. Int. J. Mol. Sci. 2018, 19, 3555. [Google Scholar] [CrossRef] [PubMed]
- Vergouwen, M.D.I.; Schut, E.S.; Troost, D.; Van De Beek, D. Diffuse Cerebral Intravascular Coagulation and Cerebral Infarction in Pneumococcal Meningitis. Neurocritical Care 2010, 13, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.J.; Brouwer, M.C.; Van De Beek, D. Neurological sequelae of bacterial meningitis. J. Infect. 2016, 73, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.J.; Brouwer, M.C.; Van De Beek, D. Delayed cerebral thrombosis in bacterial meningitis: A prospective cohort study. Intensiv. Care Med. 2012, 39, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Gallegos, C.; Tobolowsky, F.; Nigo, M.; Hasbun, R. Delayed Cerebral Injury in Adults with Bacterial Meningitis. Crit. Care Med. 2018, 46, e811–e814. [Google Scholar] [CrossRef] [PubMed]
- Kastenbauer, S.; Pfister, H. Pneumococcal meningitis in adults: Spectrum of complications and prognostic factors in a series of 87 cases. Brain 2003, 126, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Bodur, H.; Colpan, A.; Gozukucuk, R.; Akinci, E.; Cevik, M.A.; Balaban, N. Venous Sinus Thrombosis After Proteus Vulgaris Meningitis and Concomitant Clostridium abscess Formation. Scand. J. Infect. Dis. 2002, 34, 694–696. [Google Scholar] [CrossRef] [PubMed]
- Tibussek, D.; Sinclair, A.; Yau, I.; Teatero, S.; Fittipaldi, N.; Richardson, S.E.; Mayatepek, E.; Jahn, P.; Askalan, R. Late-Onset Group B Streptococcal Meningitis Has Cerebrovascular Complications. J. Pediatr. 2015, 166, 1187–1192. [Google Scholar] [CrossRef] [PubMed]
- Siegman-Igra, Y.; Azmon, Y.; Schwartz, D. Milleri group Streptococcus—A stepchild in the viridans family. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2453–2459. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.V.; Moreno, E.S.; Rodrigo, J.L.; Lletí, M.S.; Burdalo, G.S.; Pérez-Bellés, C. Meningitis and brain abscess caused by streptococcus intermedius in a patient infected with HIV-1. An. Med. Interna 2005, 22, 279–282. [Google Scholar]
- Verma, S.; Landisch, R.; Quirk, B.; Schmainda, K.; Prah, M.; Whelan, H.T.; Willoughby, R.E. Presumed Hydrogen Sulfide-mediated Neurotoxicity After Streptococcus anginosus Group Meningitis. Pediatr. Infect. Dis. J. 2013, 32, 189–191. [Google Scholar] [CrossRef] [PubMed]
- Tabata, A.; Sato, Y.; Maya, K.; Nakano, K.; Kikuchi, K.; Whiley, R.A.; Ohkura, K.; Tomoyasu, T.; Nagamune, H. A streptolysin S homologue is essential for β-haemolytic Streptococcus constellatus subsp. constellatus cytotoxicity. Microbiology 2014, 160, 980–991. [Google Scholar] [PubMed]
- Møller, K. Meningitis Caused by Streptococci Other than Streptococcus pneumoniae: A Retrospective Clinical Study. Scand. J. Infect. Dis. 1999, 31, 375–381. [Google Scholar] [PubMed]
- Bringas-Bollada, M.; Ortuño-Andériz, F.; Cabo, C.M.-D.; García-Sánchez, E. Bacteriemia, pneumonía and meningitis due to Streptococcus constellatum. Med. Clin. 2006, 126, 719. [Google Scholar] [CrossRef] [PubMed]
- Stratton, C.W. Infections due to the Streptococcus anginosus (Streptococcus milleri) Group; UpToDate: Waltham, MA, USA, 2019; Available online: https://www.uptodate.com (accessed on 20 December 2019).
- Kojan, S.; Al Jumah, M. Infection related cerebral venous thrombosis. J. Pak. Med. Assoc. 2006, 56, 494–497. [Google Scholar] [PubMed]
| Author (Year) | Country | Age, Sex | Predisposing Factor | Agent | Abscess | Thrombosis | Antimicrobial | Anticoagulant | Sequelae |
|---|---|---|---|---|---|---|---|---|---|
| Myint (1986) [I] | UK | 13, M | Orbital cellulitis | S. milleri | No | CS | Penicillin, chloramphenicol, metronidazole | Unknown | |
| Pearson (1994) [II] | UK | 37, M | Otitis media | S. milleri | Yes | TS | Ciprofloxacin, cefuroxime | None | |
| UK | 58, M | Scalp abscess | S. intermedius | Yes | TS | Erythromycin, cefuroxime, cefotaxime, etc. | Yes | ||
| Blaser (2000) [III] | Switzerland | 72, F | Mastoiditis, diabetes | S. milleri | Yes | SGS | Ceftriaxone | Unknown | |
| Benhayoun (2003) [IV] | France | 13, M | Rhinosinusitis | S. constellatus | No | IJV | Metronidazole, cefotaxime, netilmicin | NFH | None |
| Chang (2003) [V] | Taiwan | 39, M | Sinusitis, alcoholism | S. constellatus | No | CS | Penicillin | None | |
| Watkins (2003) [VI] | United States | 17, F | Intraorbital abscess | S. milleri | Yes | CS | Vancomycin, ceftriaxone, metronidazole | WARF | Yes |
| Ching (2006) [VII] | UK | 14, M | Otitis media | S. milleri | Yes | SGS | Ceftriaxone and metronidazole | Yes | |
| Hoshino (2006) [VIII] | Japan | 56, F | Orbital cellulitis | S. constellatus | Yes | CS | Sulbactam/ampicillin | Yes | |
| Pavlovich (2006) [IX] | Canada | 55, M | Orbital cellulitis, periodontitis | S. anginosus | No | CS | Vancomycin, ceftriaxone, metronidazole, etc. | LMWH | No |
| Goawalla (2007) [X] | UK | 38, M | Orbital cellulitis | S. constellatus | Yes | CS | Cefuroxime, metronidazole, amoxicillin, etc. | Unknown | Yes |
| Udaondo (2008) [XI] | Spain | 51, F | Orbital cellulitis | S. constellatus | Yes | CS | Ceftriaxone, vancomycin, metronidazole | NFH/LWMH | Yes |
| Jones (2009) [XII] | New Zealand | 54, F | Dental abscess, RA | S. constellatus | Yes | CS | Meropenem, clindamycin | LMWH/WARF | Yes |
| Gonzalez (2012) [XIII] | United States | 26, M | Sinusitis, substance abuse | S. intermedius | No | CS | Broad-spectrum antibiotics | LMWH | Unknown |
| Imholz (2012) [XIV] | Switzerland | 45, M | Orbital cellulitis, periodontitis | S. constellatus | No | CS | Ceftriaxone, vancomycin | LMWH | None |
| Noel (2013) [XV] | United States | 12, M | Sinusitis | S. constellatus | No | CS | Broad-spectrum antibiotics, ceftriaxone | Unknown | |
| Sakaida (2012) [XVI] | Japan | 75, F | Sinusitis | S. constellatus | No | CS | Unknown | NFH | None |
| Suzuki (2014) [XVII] | Japan | 50, F | Otitis media, gingivitis | S. intermedius | Yes | TS | Meropenem, vancomycin | Heparin | Yes |
| Karssemakers (2015) [XVIII] | Netherlands | 54, M | Dental caries, conjunctivitis | S. anginosus | Yes | CS | Unknown | None | |
| Selvitop (2015) [XIX] | United States | 10, F | Mastoiditis, Crohn’s disease | S. anginosus | No | CS/SGS/TS | Ceftriaxone | LMWH | None |
| Shams (2016) [XX] | United States | 73, F | Sinusitis | S. anginosus | No | CS/TS | Vancomycin, aztreonam, clindamycin, etc. | LMWH/WARF | Yes |
| Allegrini (2017) [XXI] | Italy | 46, F | Dental caries, orbital cellulitis | S. constellatus | Yes | CS | Piperacillin/tazobactam | Heparin | None |
| López (2016) [XXII] | Spain | 24, F | Pregnancy, orbital abscess | S. milleri | Yes | CS | Meropenem | LMWH/WARF | Yes |
| Martel (2018) [XXIII] | France | 86, M | Sinusitis | S. intermedius | No | CS/IJV | Amoxicillin/clavulanic acid | Unknown | None |
| Branson (2018) [XXIV] | United States | 12, M | Sinusitis, orbital cellulitis | S. anginosus | Yes | CS/IJV | Meropenem, vancomycin | NFH/LWMH | None |
| Deliran (2018) [XXV] | Netherlands | 38, F | Periodontitis, dental caries | S. intermedius | Yes | CS | Ceftriaxone, metronidazole, penicillin, etc. | Yes | |
| Mo (2018) [XXVI] | China | 48, M | Alcoholism | S. constellatus | No | SSS/TS | Ceftriaxone | Unknown | |
| Pinilla-Torres (2020) [XXVII] | Colombia | 23, M | Sinusitis | S. constellatus | Yes | SGS/TS | Ceftriaxone, vancomycin, metronidazole | Death |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinilla-Monsalve, G.D.; Torres-Cutiva, D.F.; Fernández-Cubillos, J.P. Atypical Streptococcal Meningitis with Fatal Cerebrovascular Complications: A Case Report. Infect. Dis. Rep. 2020, 12, 87-96. https://doi.org/10.3390/idr12030018
Pinilla-Monsalve GD, Torres-Cutiva DF, Fernández-Cubillos JP. Atypical Streptococcal Meningitis with Fatal Cerebrovascular Complications: A Case Report. Infectious Disease Reports. 2020; 12(3):87-96. https://doi.org/10.3390/idr12030018
Chicago/Turabian StylePinilla-Monsalve, Gabriel D., Daniel F. Torres-Cutiva, and Juan P. Fernández-Cubillos. 2020. "Atypical Streptococcal Meningitis with Fatal Cerebrovascular Complications: A Case Report" Infectious Disease Reports 12, no. 3: 87-96. https://doi.org/10.3390/idr12030018
APA StylePinilla-Monsalve, G. D., Torres-Cutiva, D. F., & Fernández-Cubillos, J. P. (2020). Atypical Streptococcal Meningitis with Fatal Cerebrovascular Complications: A Case Report. Infectious Disease Reports, 12(3), 87-96. https://doi.org/10.3390/idr12030018






