Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) in Preclinical Studies of Antivascular Treatments
Abstract
:1. Introduction
1.1. Tumour Microvasculature and Antivascular Treatments
1.2. In vivo Imaging of Antivascular Treatments
2. DCE-MRI
2.1. Principle
2.2. Semiquantitative Analysis
2.3. Model Analysis


2.4. Contrast Agents
3. Antiangiogenic Treatments
3.1. Antibodies and Specialized Proteins
| Compound | Angiogenic target | Schedule/dose | References |
|---|---|---|---|
| Antibodies and specialized proteins | |||
| Bevacizumab * | VEGF-A (VEGF165) | 1–6 × 5–45 mg/kg | [78,79,80,81,82,83,84,85,86,87,88] |
| DC101 | VEGFR-2 | 7 × 30mg/kg | [89] |
| VEGF-trap | VEGF-A/B, PlGF | 4 × 25mg/kg | [90] |
| Tyrosine Kinase Inhibitors | |||
| Vatalanib | PDGFR-β, c-kit, VEGFRs | 7–14 × 50–100 mg/kg | [91,92,93,94] |
| Sunitinib | VEGFRs, PDGFRs | 1–7 × 40–45 mg/kg | [95,96] |
| Orantinib | VEGFR-2, c-kit, FGFR, PDGFR | 1–14 × 200 mg/kg | [97,98] |
| Vandetanib | VEGFR-2, EGFR | 2 × 12.5–100 mg/kg | [51,99] |
| Axitinib | PDGFR, c-kit, VEGFR-1/2/3 | 14 × 25 mg/kg | [100] |
| Imatinib | PDGFR-β, c-kit, abl, VEGFR-2 | 3 × 50 mg/kg | [101] |
| Cediranib | VEGFRs | 1–20 × 6 mg/kg | [102] |
| Sorafenib | VEGFRs, PDGFRs, Raf | 25 × 7 mg/kg | [103] |
| Others | |||
| TNP-470 | MetAP2 | 3–7 × 6.7–30 mg/kg | [104] |
| Everolimus | mTOR | 1–7 × 5–10 mg/kg | [105] |
| KR-31831 | Unknown | 21 × 50 mg/kg | [106] |
| Thalidomide | FGF-2 | 2–3 × 60–200 mg/kg | [107,108,109] |
3.2. Tyrosine Kinase Inhibitors
3.3. Other Mechanisms of Action
4. Vascular Disrupting Agents
4.1. Tubulin Binding Agents
| Compounds | Dose/schedule | DCE-MRI | References |
|---|---|---|---|
| Tubulin binding | |||
| CA4P | 1-2 × 10-250 mg/kg (mouse) | 1-24 h | [77,111,112,113,114,115,116,117,118,119,120,121] |
| 1 × 10-100 mg/kg (rat) | 1 h-9 days | [70,122,123,124,125] | |
| OXi4503 | 1-2 × 25-100 mg/kg (mouse) | 4-144 h | [114] |
| ZD6126 | 1 × 50-200 mg/kg (mouse) | 24 h | [126,127] |
| 1 × 2.5-50 mg/kg (rat) | 1-120 h | [125,128,129,130,131] | |
| NPI2358 | 1 × 2.5-15 mg/kg (mouse) | 1-24 h | [132] |
| Stilbene 5c and 6c | 1 × 50 mg/kg (mouse) | 4 h | [133] |
| TZT-1027 | 1 × 0.5 mg/kg (rat) | 1-3 h | [134] |
| ABT-751 | 1 × 30 mg/kg (rat) | 1-6 h | [135] |
| TNF-α inducing | |||
| DMXAA | 1-4 × 22-30 mg/kg (mouse) | 3-24 h | [113,136,137,138,139,141,142,143] |
| 1 × 100-350 mg/kg (rat) | 4-24 h | [144] | |
| TNF-α | 300 μg/kg or viral overexpression (mouse) | 2-96 h or 3 days | [145,146] |
| AP/1649, AP/1897 | 4 × 27 mg/kg (mouse) | 3-24 h | [143] |
| Other | |||
| Radiation | 8-18 Gy single dose or fractionated (rat) | 2-25 days | [147,148] |
| 20 Gy single dose or fractionated (mouse) | 3-120 h | [32] | |
| Photosensitizers | |||
| Bacteriochlorophyll-serine | 1 × 20 mg/kg (mouse) | 1-24 h | [136,149] |
| Targeted chemotherapy | |||
| MBT-0206 | 3 × 5 mg/kg (hamster) | 24 h | [150] |
| EndoTAG®-2 | 6 × 2.5 mg/kg (mouse) | 24 h | [151] |
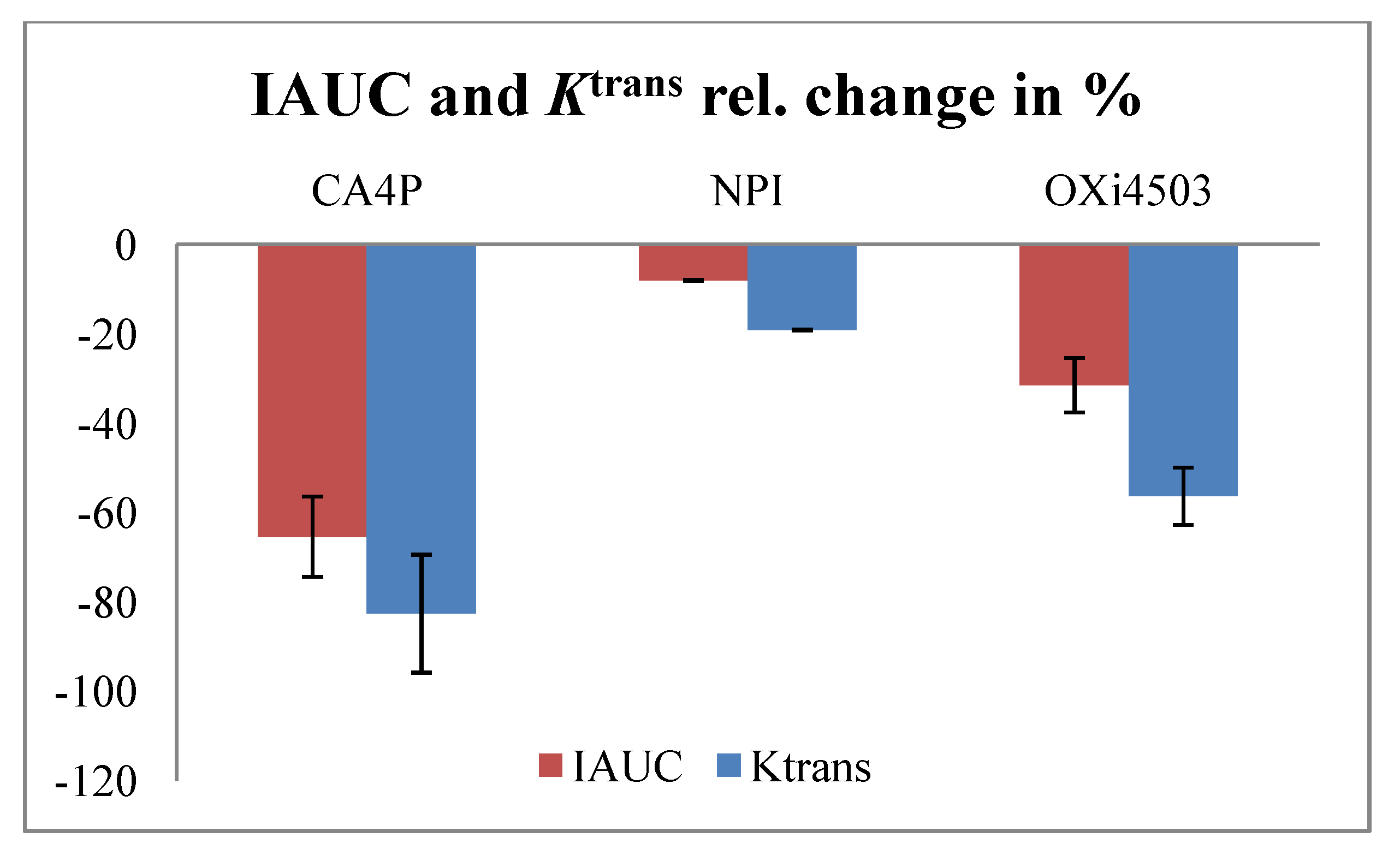
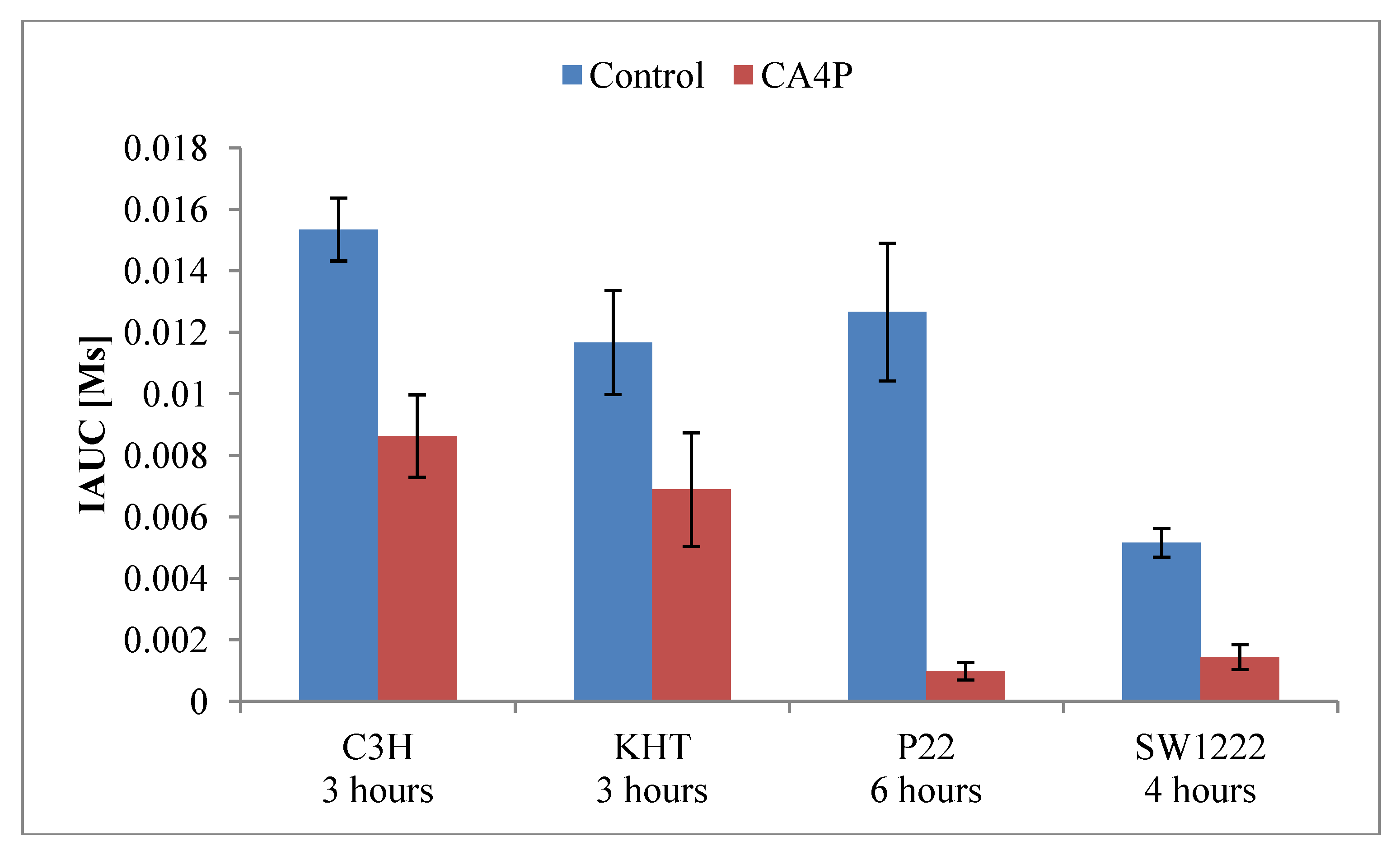
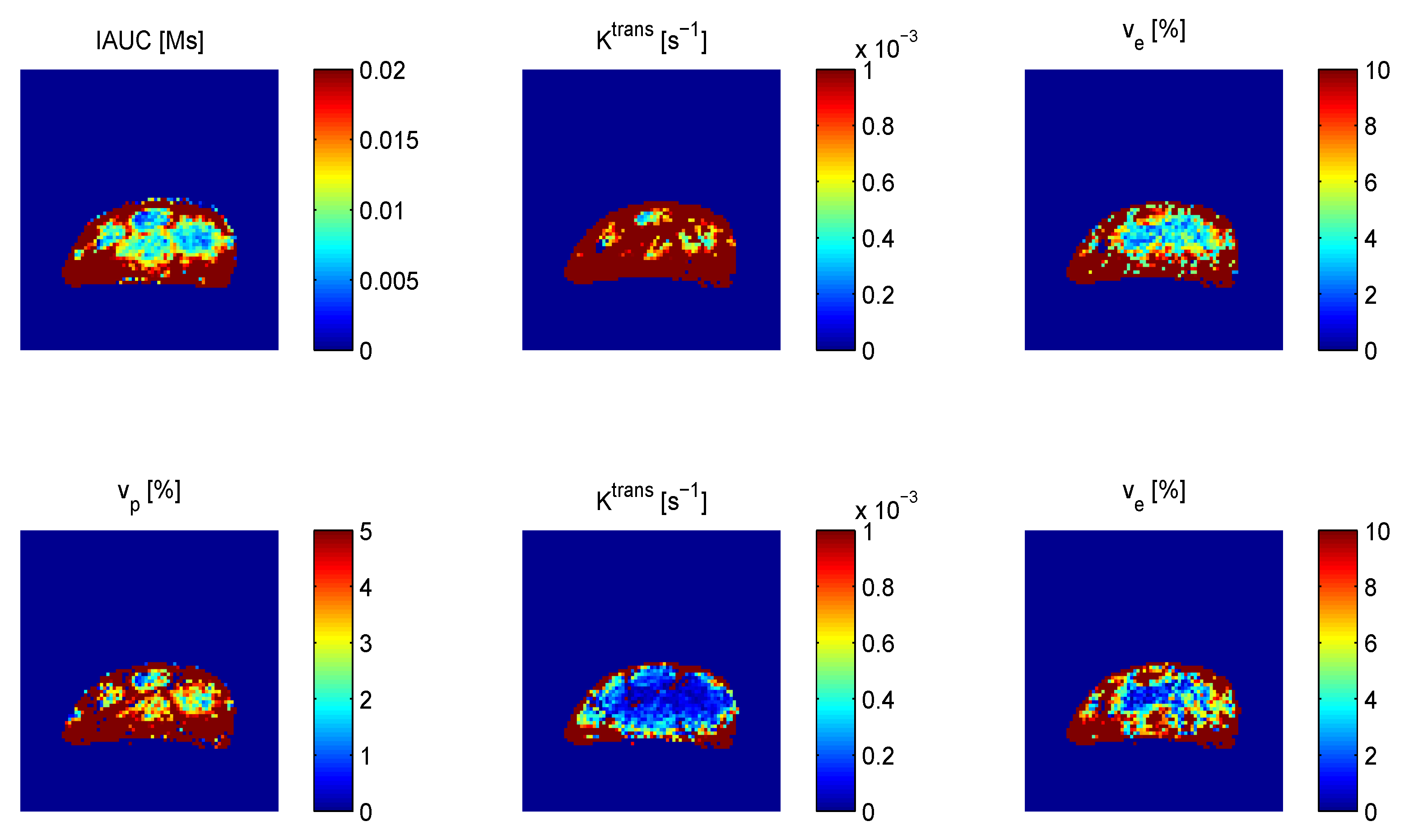
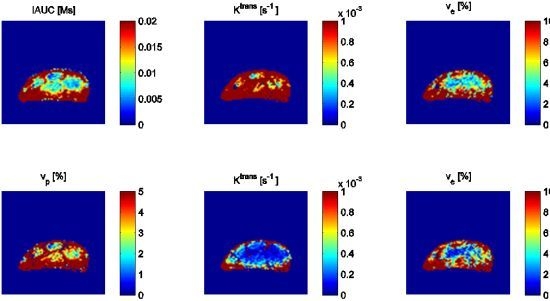
4.2. TNF-α Inducing Agents
4.3. Other Vascular Disrupting Treatments
5. Conclusions and Future Perspectives
Conflict of Interest
Acknowledgements
References
- Brem, S.; Brem, H.; Folkman, J.; Finkelstein, D.; Patz, A. Prolonged tumor dormancy by prevention of neovascularization in the vitreous. Cancer Res. 1976, 8, 2807–2812. [Google Scholar]
- Folkman, J. How is blood vessel growth regulated in normal and neoplastic tissue? G.H.A. Clowes memorial Award lecture. Cancer Res. 1986, 2, 467–473. [Google Scholar]
- Hahnfeldt, P.; Panigrahy, D.; Folkman, J.; Hlatky, L. Tumor development under angiogenic signaling: A dynamical theory of tumor growth, treatment response, and postvascular dormancy. Cancer Res. 1999, 19, 4770–4775. [Google Scholar]
- Bergers, G.; Benjamin, L.E. Tumorigenesis and the angiogenic switch. Nat. Rev. Cancer. 2003, 6, 401–410. [Google Scholar] [CrossRef]
- Vaupel, P.; Kallinowski, F.; Okunieff, P. Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: A review. Cancer Res. 1989, 23, 6449–6465. [Google Scholar]
- Stoeltzing, O.; Ellis, L.M. The role of microvasculature in metastasis formation. In Vascular-Targeted Therapies in Oncology; Siemann, D.W., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2006; pp. 31–62. [Google Scholar]
- Vaupel, P. Tumor microenvironmental physiology and its implications for radiation oncology. Semin.Radiat. Oncol. 2004, 3, 198–206. [Google Scholar] [CrossRef]
- Harris, A.L. Hypoxia—A key regulatory factor in tumour growth. Nat. Rev. Cancer. 2002, 1, 38–47. [Google Scholar]
- Siemann, D.W.; Bibby, M.C.; Dark, G.G.; Dicker, A.P.; Eskens, F.A.L.M.; Horsman, M.R.; Marmé, D.; Lorusso, P.M. Differentiation and definition of vascular-targeted therapies. Clin.Cancer Res. 2005, 2, 416–420. [Google Scholar]
- Horsman, M.R.; Siemann, D.W. Pathophysiologic effects of vascular-targeting agents and the implications for combination with conventional therapies. Cancer Res. 2006, 24, 11520–11539. [Google Scholar]
- Ferrara, N.; Gerber, H.P.; Le Couter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 6, 669–676. [Google Scholar]
- Pugh, C.W.; Ratcliffe, P.J. Regulation of angiogenesis by hypoxia: role of the HIF system. Nat. Med. 2003, 6, 677–684. [Google Scholar]
- Kerbel, R.; Folkman, J. Clinical translation of angiogenesis inhibitors. Nat Rev Cancer 2002, 10, 727–739. [Google Scholar] [CrossRef]
- Marme, D. The impact of anti-angiogenic agents on cancer therapy. J. Cancer Res. Clin. Oncol. 2003, 11, 607–620. [Google Scholar]
- Eskens, F.A. Angiogenesis inhibitors in clinical development; Where are we now and where are we going? Br. J. Cancer 2004, 1, 1–7. [Google Scholar] [CrossRef]
- Ferrara, N.; Kerbel, R.S. Angiogenesis as a therapeutic target. Nature 2005, 7070, 967–974. [Google Scholar] [CrossRef]
- Heath, V.L.; Bicknell, R. Anticancer strategies involving the vasculature. Nat. Rev. Clin. Oncol. 2009, 7, 395–404. [Google Scholar]
- National Cancer Institute. Available online: www.cancer.gov/clinicaltrials (accessed on 1 May 2012).
- Horsman, M.R.; Overgaard, J. Thermal radiosensitization in animal tumors: the potential for therapeutic gain. In Hyperthermia and Oncology; Urano, M., Douple, E., Eds.; VSP: Utrecht, The Netherland, 1985; Volume 2, pp. 113–145. [Google Scholar]
- Chaplin, D.J.; Dougherty, G.J. Tumour vasculature as a target for cancer therapy. Br. J. Cancer 1999, 80, 57–64. [Google Scholar]
- Thorpe, P.E. Vascular targeting agents as cancer therapeutics. Clin.Cancer Res. 2004, 2, 415–427. [Google Scholar]
- Siemann, D.W.; Chaplin, D.J.; Horsman, M.R. Vascular-targeting therapies for treatment of malignant disease. Cancer 2004, 12, 2491–2499. [Google Scholar]
- Baguley, B.C. Antivascular therapy of cancer: DMXAA. Lancet Oncol. 2003, 3, 141–148. [Google Scholar]
- Tozer, G.M.; Kanthou, C.; Baguley, B.C. Disrupting tumour blood vessels. Nat. Rev. Cancer 2005, 6, 423–435. [Google Scholar] [CrossRef]
- Weidner, N.; Semple, J.P.; Welch, W.R.; Folkman, J. Tumor angiogenesis and metastasis—Correlation in invasive breast carcinoma. N. Engl. J. Med. 1991, 1, 1–8. [Google Scholar]
- Borre, M.; Offersen, B.V.; Nerstrøm, B.; Overgaard, J. Microvessel density predicts survival in prostate cancer patients subjected to watchful waiting. Br. J. Cancer 1998, 7, 940–944. [Google Scholar]
- Hansen, S.; Grabau, D.A.; Sørensen, F.B.; Bak, M.; Rose, W.V.A.C. The prognostic value of angiogenesis by Chalkley counting in a confirmatory study design on 836 breast cancer patients. Clin.Cancer Res. 2000, 1, 139–146. [Google Scholar]
- Borre, M.; Nerstrøm, B.; Overgaard, J. Association between immunohistochemical expression of vascular endothelial growth factor (VEGF), VEGF-expressing neuroendocrine-differentiated tumor cells, and outcome in prostate cancer patients subjected to watchful waiting. Clin.Cancer Res. 2000, 5, 1882–1890. [Google Scholar]
- Gasparini, G. Clinical significance of determination of surrogate markers of angiogenesis in breast cancer. Crit. Rev. Oncol. 2001, 2, 97–114. [Google Scholar] [CrossRef]
- Robinson, S.P.; Rijken, P.F.J.W.; Howe, F.A.; McSheehy, P.M.J.; van der Sanden, B.P.J.; Heerschap, A.; Stubbs, M.; van der Kogel, A.J.; Griffiths, J.R. Tumor vascular architecture and function evaluated by non-invasive susceptibility MRI methods and immunohistochemistry. J. Magn. Reson. Imaging 2003, 4, 445–454. [Google Scholar]
- Garcia-Barros, M.; Paris, F.; Cordon-Cardo, C.; Lyden, D.; Rafii, S.; Haimovitz-Friedman, A.; Fuks, Z.; Kolesnick, R. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003, 5622, 1155–1159. [Google Scholar]
- Horsman, M.R.; Nielsen, T.; Østergaard, L.; Overgaard, J. Radiation administered as a large single dose or in a fractionated schedule: Role of the tumour vasculature as a target for influencing response. Acta Oncol. 2006, 7, 876–880. [Google Scholar]
- Miller, J.C.; Pien, H.H.; Sahani, D.; Sorensen, A.G.; Thrall, J.H. Imaging angiogenesis: Applications and potential for drug development. J. Natl. Cancer Inst. 2005, 3, 172–187. [Google Scholar]
- Lassau, N.; Chebil, M.; Chami, L.; Bidault, S.; Girard, E.; Roche, A. Dynamic contrast-enhanced ultrasonography (DCE-US): A new tool for the early evaluation of antiangiogenic treatment. Target Oncol. 2010, 1, 53–58. [Google Scholar]
- Mason, R.P.; Zhao, D.; Liu, L.; Trawick, M.L.; Pinney, K.G. A perspective on vascular disrupting agents that interact with tubulin: preclinical tumor imaging and biological assessment. Integr. Biol. (Camb) 2011, 4, 375–387. [Google Scholar]
- Tai, J.H.; Tessier, J.; Ryan, A.J.; Hoffman, L.; Chen, X.; Lee, T.Y. Assessment of acute antivascular effects of vandetanib with high-resolution dynamic contrast-enhanced computed tomographic imaging in a human colon tumor xenograft model in the nude rat. Neoplasia 2010, 9, 697–707. [Google Scholar]
- Anderson, H.L.; Yap, J.T.; Miller, M.P.; Robbins, A.; Jones, T.; Price, P.M. Assessment of pharmacodynamic vascular response in a phase I trial of combretastatin A4 phosphate. J. Clin. Oncol. 2003, 15, 2823–2830. [Google Scholar]
- Leach, M.O.; Brindle, K.M.; Evelhoch, J.L.; Griffiths, J.R.; Horsman, M.R.; Jackson, A.; Jayson, G.C.; Judson, I.R.; Knopp, M.V.; Maxwell, R.J.; et al. Pharmacodynamic/Pharmacokinetic Technologies Advisory Committee, Drug Development Office, Cancer Research UK The assessment of antiangiogenic and antivascular therapies in early-stage clinical trials using magnetic resonance imaging: issues and recommendations. Br. J. Cancer 2005, 9, 1599–1610. [Google Scholar]
- Petersen, E.T.; Zimine, I.; Ho, Y.C.; Golay, X. Non-invasive measurement of perfusion: A critical review of arterial spin labelling techniques. Br. J. Radiol. 2006, 944, 688–701. [Google Scholar]
- Jarnum, H.; Steffensen, E.G.; Knutsson, L.; Frund, E.T.; Simonsen, C.W.; Lundbye-Christensen, S.; Shankaranarayanan, A.; Alsop, D.C.; Jensen, F.T.; Larsson, E.M. Perfusion MRI of brain tumours: A comparative study of pseudo-continuous arterial spin labelling and dynamic susceptibility contrast imaging. Neuroradiology 2010, 4, 307–317. [Google Scholar]
- Van Westen, D.; Petersen, E.T.; Wirestam, R.; Siemund, R.; Bloch, K.M.; Stahlberg, F.; Bjorkman-Burtscher, I.M.; Knutsson, L. Correlation between arterial blood volume obtained by arterial spin labelling and cerebral blood volume in intracranial tumours. MagmaMagn.Reson. Mater. Phy. Biol. Med. 2011, 4, 211–223. [Google Scholar]
- Østergaard, L.; Weisskoff, R.M.; Chesler, D.A.; Gyldensted, C.; Rosen, B.R. High resolution measurement of cerebral blood flow using intravascular tracer bolus passages. Part I: Mathematical approach and statistical analysis. Magn. Reson. Med. 1996, 5, 715–725. [Google Scholar]
- Østergaard, L.; Sørensen, A.G.; Kwong, K.K.; Gyldensted, R.M.W.a.C.; Rosen, B.R. High resolution measurement of cerebral blood flow using intravascular tracer bolus passages. Part II: Experimental comparison and preliminary results. Magn. Reson. Med. 1996, 5, 726–736. [Google Scholar]
- Kiselev, V.G.; Strecker, R.; Ziyeh, S.; Speck, O.; Hennig, J. Vessel size imaging in humans. Magn.Reson. Med. 2005, 3, 553–563. [Google Scholar]
- Dennie, J.; Mandeville, J.B.; Boxerman, J.L.; Packard, S.D.; Rosen, B.R.; Weisskoff, R.M. NMR imaging of changes in vascular morphology due to tumor angiogenesis. Magn.Reson. Med. 1998, 6, 793–799. [Google Scholar]
- Troprès, I.; Grimault, S.; Vaeth, A.; Grillon, E.; Julien, C.; Payen, J.F.; Lamalle, L.; Décorps, M. Vessel size imaging. Magn.Reson. Med. 2001, 3, 397–408. [Google Scholar]
- Zahra, M.A.; Hollingsworth, K.G.; Sala, E.; Lomas, D.J.; Tan, L.T. Dynamic contrast-enhanced MRI as a predictor of tumour response to radiotherapy. Lancet Oncol 2007, 1, 63–74. [Google Scholar]
- Gadian, D.G.; Payne, J.A.; Bryant, D.J.; Young, I.R.; Carr, D.H.; Bydder, G.M. Gadolinium-DTPA as a contrast agent in MR imaging—Theoretical projections and practical observations. J. Comput. Assist. Tomogr. 1985, 2, 242–251. [Google Scholar]
- Grossman, R.I.; Gonzalez-Scarano, F.; Atlas, S.W.; Galetta, S.; Silberberg, D.H. Multiple sclerosis: Gadolinium enhancement in MR imaging. Radiology 1986, 3, 721–725. [Google Scholar]
- Miller, D.H.; Rudge, P.; Johnson, G.; Kendall, B.E.; Macmanus, D.G.; Moseley, I.F.; Barnes, D.; McDonald, W.I. Serial gadolinium enhanced magnetic resonance imaging in multiple sclerosis. Brain 1988, Pt 4, 927–939. [Google Scholar]
- Pradel, C.; Siauve, N.; Bruneteau, G.; Clement, O.; de Bazelaire, C.; Frouin, F.; Wedge, S.R.; Tessier, J.L.; Robert, P.H.; Frija, G.; et al. Reduced capillary perfusion and permeability in human tumour xenografts treated with the VEGF signalling inhibitor ZD4190: An in vivo assessment using dynamic MR imaging and macromolecular contrast media. Magn. Reson. Imaging 2003, 8, 845–851. [Google Scholar]
- De Bazelaire, C.; Siauve, N.; Fournier, L.; Frouin, F.; Robert, P.; Clement, O.; de Kerviler, E.; Cuenod, C.A. Comprehensive model for simultaneous MRI determination of perfusion and permeability using a blood-pool agent in rats rhabdomyosarcoma. Eur. Radiol. 2005, 12, 2497–2505. [Google Scholar]
- Sourbron, S.P.; Buckley, D.L. Tracer kinetic modelling in MRI: Estimating perfusion and capillary permeability. Phys. Med. Biol. 2012, 2, R1–R33. [Google Scholar]
- Evelhoch, J.L. Key factors in the acquisition of contrast kinetic data for oncology. J. Magn. Reson. Imaging 1999, 3, 254–259. [Google Scholar] [CrossRef]
- Kety, S.S. The theory and applications of the exchange of inert gas at the lungs and tissues. Pharmacol.Rev. 1951, 3, 1–41. [Google Scholar]
- Renkin, E.M. Transport of potassium-42 from blood to tissue in isolated mammalian skeletal muscles. Am. J. Physiol. 1959, 197, 1205–1210. [Google Scholar]
- Crone, C. The permeability of capillaries in various organs as determined by use of the “indicator diffusion” method. Acta Physiol. Scand. 1963, 58, 292–305. [Google Scholar] [CrossRef]
- Patlak, C.S.; Fenstermacher, J.D. Measurements of dog blood-brain transfer constants by ventriculocisternal perfusion. Am. J. Physiol. 1975, 4, 877–884. [Google Scholar]
- Tofts, P.S. Modeling tracer kinetics in dynamic Gd-DTPA MR imaging. J. Magn. Reson. Imaging 1997, 1, 91–101. [Google Scholar]
- Tofts, P.S.; Kermode, A.G. Measurement of the blood-brain barrier permeability and leakage space using dynamic MR imaging. 1. Fundamental concepts. Magn. Reson. Med. 1991, 2, 357–367. [Google Scholar]
- Larsson, H.B.; Stubgaard, M.; Frederiksen, J.L.; Jensen, M.; Henriksen, O.; Paulson, O.B. Quantitation of blood-brain barrier defect by magnetic resonance imaging and gadolinium-DTPA in patients with multiple sclerosis and brain tumors. Magn.Reson. Med. 1990, 1, 117–131. [Google Scholar]
- Brix, G.; Semmler, W.; Port, R.; Schad, L.R.; Layer, G.; Lorenz, W.J. Pharmacokinetic parameters in CNS Gd-DTPA enhanced MR imaging. J. Comput. Assist. Tomogr. 1991, 4, 621–628. [Google Scholar]
- Ewing, J.R.; Knight, R.A.; Nagaraja, T.N.; Yee, J.S.; Nagesh, V.; Whitton, P.A.; Li, L.; Fenstermacher, J.D. Patlak plots of Gd-DTPA MRI data yield blood-brain transfer constants concordant with those of 14C-sucrose in areas of blood-brain opening. Magn.Reson. Med. 2003, 2, 283–292. [Google Scholar]
- Tofts, P.S.; Brix, G.; Buckley, D.L.; Evelhoch, J.L.; Henderson, E.; Knopp, M.V.; Larsson, H.B.; Lee, T.Y.; Mayr, N.A.; Parker, G.J.; et al. Estimating kinetic parameters from dynamic contrast-enhanced T(1)-weighted MRI of a diffusable tracer: standardized quantities and symbols. J. Magn. Reson. Imaging 1999, 3, 223–232. [Google Scholar]
- Patlak, C.S.; Blasberg, R.G. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Generalizations. J. Cereb. Blood Flow Metab. 1985, 4, 584–590. [Google Scholar]
- Ewing, J.R.; Brown, S.L.; Lu, M.; Panda, S.; Ding, G.; Knight, R.A.; Cao, Y.; Jiang, Q.; Nagaraja, T.N.; Churchman, J.L.; et al. Model selection in magnetic resonance imaging measurements of vascular permeability: Gadomer in a 9L model of rat cerebral tumor. J. Cereb. Blood Flow Metab. 2006, 3, 310–320. [Google Scholar]
- Roberts, T. Technical and practical considerations for permeability modeling of dynamic contrast enhanced MRI. Acad. Radiol. 2005, S34–S37. [Google Scholar] [CrossRef]
- Weinmann, H.J.; Laniado, M.; Mützel, W. Pharmacokinetics of GdDTPA/dimeglumine after intravenous injection into healthy volunteers. Physiol. Chem. Phys. Med. NMR 1984, 2, 167–172. [Google Scholar]
- Wedeking, P.; Eaton, S.; Covell, D.G.; Nair, S.; Tweedle, M.F.; Eckelman, W.C. Pharmacokinetic analysis of blood distribution of intravenously administered 153Gd-labeled Gd(DTPA)2- and 99mTc(DTPA) in rats. Magn.Reson. Imaging 1990, 5, 567–575. [Google Scholar]
- Maxwell, R.J.; Wilson, J.; Prise, V.E.; Vojnovic, B.; Rustin, G.J.; Lodge, M.A.; Tozer, G.M. Evaluation of the anti-vascular effects of combretastatin in rodent tumours by dynamic contrast enhanced MRI. NMR Biomed. 2002, 2, 89–98. [Google Scholar]
- Furman-Haran, E.; Grobgeld, D.; Degani, H. Dynamic contrast-enhanced imaging and analysis at high spatial resolution of MCF7 human breast tumors. J. Magn. Reson. 1997, 2, 161–171. [Google Scholar]
- Yang, C.; Karczmar, G.S.; Medved, M.; Stadler, W.M. Estimating the arterial input function using two reference tissues in dynamic contrast-enhanced MRI studies: Fundamental concepts and simulations. Magn.Reson. Med. 2004, 5, 1110–1117. [Google Scholar]
- Yankeelov, T.E.; Luci, J.J.; Lepage, M.; Li, R.; Debusk, L.; Lin, P.C.; Price, R.R.; Gore, J.C. Quantitative pharmacokinetic analysis of DCE-MRI data without an arterial input function: A reference region model. Magn.Reson. Imaging 2005, 4, 519–529. [Google Scholar]
- Turetschek, K.; Preda, A.; Novikov, V.; Brasch, R.C.; Weinmann, H.J.; Wunderbaldinger, P.; Roberts, T.P. Tumor microvascular changes in antiangiogenic treatment: Assessment by magnetic resonance contrast media of different molecular weights. J. Magn. Reson. Imaging 2004, 1, 138–144. [Google Scholar]
- Brasch, R.; Turetschek, K. MRI characterization of tumors and grading angiogenesis using macromolecular contrast media: status report. Eur. J. Radiol. 2000, 3, 148–155. [Google Scholar]
- Daldrup-Link, H.E.; Kaiser, A.; Helbich, T.; Werner, M.; Bjørnerud, A.; Link, T.M.; Rummeny, E.J. Macromolecular contrast medium (feruglose) versus small molecular contrast medium (gadopentetate) enhanced magnetic resonance imaging: differentiation of benign and malignant breast lesions. Acad. Radiol. 2003, 11, 1237–1246. [Google Scholar]
- Bentzen, L.; Vestergaard-Poulsen, P.; Nielsen, T.; Overgaard, J.; Bjørnerud, A.; Briley-Saebø, K.; Horsman, M.R.; Ostergaard, L. Intravascular contrast agent-enhanced MRI measuring contrast clearance and tumor blood volume and the effects of vascular modifiers in an experimental tumor. Int. J. Radiat. Oncol. Biol. Phys. 2005, 4, 1208–1215. [Google Scholar]
- Brasch, R.; Pham, C.; Shames, D.; Roberts, T.; van Dijke, K.; van Bruggen, N.; Mann, J.; Ostrowitzki, S.; Melnyk, O. Assessing tumor angiogenesis using macromolecular MR imaging contrast media. J. Magn. Reson. Imaging 1997, 1, 68–74. [Google Scholar]
- Pham, C.D.; Roberts, T.P.; Bruggen, N. v.; Melnyk, O.; Mann, J.; Ferrara, N.; Cohen, R.L.; Brasch, R.C. Magnetic resonance imaging detects suppression of tumor vascular permeability after administration of antibody to vascular endothelial growth factor. Cancer Invest. 1998, 4, 225–230. [Google Scholar]
- Gossmann, A.; Helbich, T.H.; Mesiano, S.; Shames, D.M.; Wendland, M.F.; Roberts, T.P.; Ferrara, N.; Jaffe, R.B.; Brasch, R.C. Magnetic resonance imaging in an experimental model of human ovarian cancer demonstrating altered microvascular permeability after inhibition of vascular endothelial growth factor. Am. J. Obstet. Gynecol. 2000, 4, 956–963. [Google Scholar]
- Gossmann, A.; Helbich, T.H.; Kuriyama, N.; Ostrowitzki, S.; Roberts, T.P.; Shames, D.M.; van Bruggen, N.; Wendland, M.F.; Israel, M.A.; Brasch, R.C. Dynamic contrast-enhanced magnetic resonance imaging as a surrogate marker of tumor response to anti-angiogenic therapy in a xenograft model of glioblastoma multiforme. J. Magn. Reson. Imaging 2002, 3, 233–240. [Google Scholar]
- Daldrup-Link, H.E.; Okuhata, Y.; Wolfe, A.; Srivastav, S.; Øie, S.; Ferrara, N.; Cohen, R.L.; Shames, D.M.; Brasch, R.C. Decrease in tumor apparent permeability-surface area product to a MRI macromolecular contrast medium following angiogenesis inhibition with correlations to cytotoxic drug accumulation. Microcirculation 2004, 5, 387–396. [Google Scholar]
- Preda, A.; Novikov, V.; Moglich, M.; Turetschek, K.; Shames, D.M.; Brasch, R.C.; Cavagna, F.M.; Roberts, T.P. MRI monitoring of Avastin antiangiogenesis therapy using B22956/1, a new blood pool contrast agent, in an experimental model of human cancer. J. Magn. Reson. Imaging 2004, 5, 865–873. [Google Scholar]
- Jahnke, K.; Muldoon, L.L.; Varallyay, C.G.; Lewin, S.J.; Kraemer, D.F.; Neuwelt, E.A. Bevacizumab and carboplatin increase survival and asymptomatic tumor volume in a glioma model. Neuro Oncol. 2009, 2, 142–150. [Google Scholar]
- Bauerle, T.; Bartling, S.; Berger, M.; Schmitt-Graff, A.; Hilbig, H.; Kauczor, H.U.; Delorme, S.; Kiessling, F. Imaging anti-angiogenic treatment response with DCE-VCT, DCE-MRI and DWI in an animal model of breast cancer bone metastasis. Eur. J. Radiol. 2010, 2, 280–287. [Google Scholar]
- Muldoon, L.L.; Gahramanov, S.; Li, X.; Marshall, D.J.; Kraemer, D.F.; Neuwelt, E.A. Dynamic magnetic resonance imaging assessment of vascular targeting agent effects in rat intracerebral tumor models. Neuro Oncol. 2011, 1, 51–60. [Google Scholar]
- Keunen, O.; Johansson, M.; Oudin, A.; Sanzey, M.; Rahim, S.A.; Fack, F.; Thorsen, F.; Taxt, T.; Bartos, M.; Jirik, R.; et al. Anti-VEGF treatment reduces blood supply and increases tumor cell invasion in glioblastoma. Proc. Natl. Acad. Sci. USA 2011, 9, 3749–3754. [Google Scholar]
- Fruth, K.; Weber, S.; Okcu, Y.; Noppens, R.; Klein, K.U.; Joest, E.; Hedrich, J.; Thilemann, S.; Pogorzelski, B.; Koutsimpelas, D.; et al. Increased basic fibroblast growth factor release and proliferation in xenotransplanted squamous cell carcinoma after combined irradiation/anti-vascular endothelial growth factor treatment. Oncol. Rep. 2012, 5, 1573–1579. [Google Scholar]
- Kiessling, F.; Farhan, N.; Lichy, M.P.; Vosseler, S.; Heilmann, M.; Krix, M.; Bohlen, P.; Miller, D.W.; Mueller, M.M.; Semmler, W.; et al. Dynamic contrast-enhanced magnetic resonance imaging rapidly indicates vessel regression in human squamous cell carcinomas grown in nude mice caused by VEGF receptor 2 blockade with DC101. Neoplasia 2004, 3, 213–223. [Google Scholar]
- Hoff, B.A.; Bhojani, M.S.; Rudge, J.; Chenevert, T.L.; Meyer, C.R.; Galban, S.; Johnson, T.D.; Leopold, J.S.; Rehemtulla, A.; Ross, B.D.; et al. DCE and DW-MRI monitoring of vascular disruption following VEGF-Trap treatment of a rat glioma model. NMR Biomed. 2012, 25, 935–942. [Google Scholar] [CrossRef]
- Turetschek, K.; Preda, A.; Floyd, E.; Shames, D.M.; Novikov, V.; Roberts, T.P.; Wood, J.M.; Fu, Y.; Carter, W.O.; Brasch, R.C. MRI monitoring of tumor response following angiogenesis inhibition in an experimental human breast cancer model. Eur. J. Nucl. Med. Mol. Imaging 2003, 3, 448–455. [Google Scholar]
- Rudin, M.; McSheehy, P.M.; Allegrini, P.R.; Rausch, M.; Baumann, D.; Becquet, M.; Brecht, K.; Brueggen, J.; Ferretti, S.; Schaeffer, F.; et al. PTK787/ZK222584, a tyrosine kinase inhibitor of vascular endothelial growth factor receptor, reduces uptake of the contrast agent GdDOTA by murine orthotopic B16/BL6 melanoma tumours and inhibits their growth in vivo. NMR Biomed. 2005, 5, 308–321. [Google Scholar]
- Ali, M.M.; Janic, B.; Babajani-Feremi, A.; Varma, N.R.; Iskander, A.S.; Anagli, J.; Arbab, A.S. Changes in vascular permeability and expression of different angiogenic factors following anti-angiogenic treatment in rat glioma. PLoS One 2010, 1, e8727. [Google Scholar]
- Drevs, J.; Muller-Driver, R.; Wittig, C.; Fuxius, S.; Esser, N.; Hugenschmidt, H.; Konerding, M.A.; Allegrini, P.R.; Wood, J.; Hennig, J.; et al. PTK787/ZK 222584, a specific vascular endothelial growth factor-receptor tyrosine kinase inhibitor, affects the anatomy of the tumor vascular bed and the functional vascular properties as detected by dynamic enhanced magnetic resonance imaging. Cancer Res. 2002, 14, 4015–4022. [Google Scholar]
- Marzola, P.; Degrassi, A.; Calderan, L.; Farace, P.; Nicolato, E.; Crescimanno, C.; Sandri, M.; Giusti, A.; Pesenti, E.; Terron, A.; et al. Early antiangiogenic activity of SU11248 evaluated in vivo by dynamic contrast-enhanced magnetic resonance imaging in an experimental model of colon carcinoma. Clin.Cancer Res. 2005, 16, 5827–5832. [Google Scholar]
- Casneuf, V.F.; Delrue, L.; van Damme, N.; Demetter, P.; Robert, P.; Corot, C.; Duyck, P.; Ceelen, W.; Boterberg, T.; Peeters, M. Noninvasive monitoring of therapy-induced microvascular changes in a pancreatic cancer model using dynamic contrast-enhanced magnetic resonance imaging with P846, a new low-diffusible gadolinium-based contrast agent. Radiat.Res. 2011, 1, 10–20. [Google Scholar]
- Marzola, P.; Degrassi, A.; Calderan, L.; Farace, P.; Crescimanno, C.; Nicolato, E.; Giusti, A.; Pesenti, E.; Terron, A.; Sbarbati, A.; et al. In vivo assessment of antiangiogenic activity of SU6668 in an experimental colon carcinoma model. Clin. Cancer Res. 2004, 2, 739–750. [Google Scholar]
- Farace, P.; Galie, M.; Merigo, F.; Daducci, A.; Calderan, L.; Nicolato, E.; Degrassi, A.; Pesenti, E.; Sbarbati, A.; Marzola, P. Inhibition of tyrosine kinase receptors by SU6668 promotes abnormal stromal development at the periphery of carcinomas. Br. J. Cancer 2009, 10, 1575–1580. [Google Scholar]
- Checkley, D.; Tessier, J.J.; Kendrew, J.; Waterton, J.C.; Wedge, S.R. Use of dynamic contrast-enhanced MRI to evaluate acute treatment with ZD6474, a VEGF signalling inhibitor, in PC-3 prostate tumours. Br. J. Cancer 2003, 10, 1889–1895. [Google Scholar]
- Wilmes, L.J.; Pallavicini, M.G.; Fleming, L.M.; Gibbs, J.; Wang, D.; Li, K.L.; Partridge, S.C.; Henry, R.G.; Shalinsky, D.R.; Hu-Lowe, D.; et al. AG-013736, a novel inhibitor of VEGF receptor tyrosine kinases, inhibits breast cancer growth and decreases vascular permeability as detected by dynamic contrast-enhanced magnetic resonance imaging. Magn.Reson. Imaging 2007, 3, 319–327. [Google Scholar]
- Dafni, H.; Kim, S.J.; Bankson, J.A.; Sankaranarayanapillai, M.; Ronen, S.M. Macromolecular dynamic contrast-enhanced (DCE)-MRI detects reduced vascular permeability in a prostate cancer bone metastasis model following anti-platelet-derived growth factor receptor (PDGFR) therapy, indicating a drop in vascular endothelial growth factor receptor (VEGFR) activation. Magn.Reson. Med. 2008, 4, 822–833. [Google Scholar]
- Kamoun, W.S.; Ley, C.D.; Farrar, C.T.; Duyverman, A.M.; Lahdenranta, J.; Lacorre, D.A.; Batchelor, T.T.; di Tomaso, E.; Duda, D.G.; Munn, L.L.; et al. Edema control by cediranib, a vascular endothelial growth factor receptor-targeted kinase inhibitor, prolongs survival despite persistent brain tumor growth in mice. J. Clin. Oncol. 2009, 15, 2542–2552. [Google Scholar]
- Merz, M.; Komljenovic, D.; Zwick, S.; Semmler, W.; Bauerle, T. Sorafenib tosylate and paclitaxel induce anti-angiogenic, anti-tumour and anti-resorptive effects in experimental breast cancer bone metastases. Eur. J. Cancer 2011, 2, 277–286. [Google Scholar]
- Bhujwalla, Z.M.; Artemov, D.; Natarajan, K.; Solaiyappan, M.; Kollars, P.; Kristjansen, P.E. Reduction of vascular and permeable regions in solid tumors detected by macromolecular contrast magnetic resonance imaging after treatment with antiangiogenic agent TNP-470. Clin.Cancer Res. 2003, 1, 355–362. [Google Scholar]
- Lane, H.A.; Wood, J.M.; McSheehy, P.M.; Allegrini, P.R.; Boulay, A.; Brueggen, J.; Littlewood-Evans, A.; Maira, S.M.; Martiny-Baron, G.; Schnell, C.R.; et al. mTOR inhibitor RAD001 (everolimus) has antiangiogenic/vascular properties distinct from a VEGFR tyrosine kinase inhibitor. Clin. Cancer Res. 2009, 5, 1612–1622. [Google Scholar]
- Yang, J.; Kim, J.H.; Im, G.H.; Heo, H.; Yoon, S.; Lee, J.; Lee, J.H.; Jeon, P. Evaluation of antiangiogenic effects of a new synthetic candidate drug KR-31831 on xenografted ovarian carcinoma using dynamic contrast enhanced MRI. Korean J. Radiol. 2011, 5, 602–610. [Google Scholar]
- Ansiaux, R.; Baudelet, C.; Jordan, B.F.; Beghein, N.; Sonveaux, P.; de Wever, J.; Martinive, P.; Gregoire, V.; Feron, O.; Gallez, B. Thalidomide radiosensitizes tumors through early changes in the tumor microenvironment. Clin.Cancer Res. 2005, 2, 743–750. [Google Scholar]
- Cyran, C.C.; Sennino, B.; Chaopathomkul, B.; Fu, Y.; Rogut, V.S.; Shames, D.M.; Wendland, M.F.; McDonald, D.M.; Brasch, R.C. Magnetic resonance imaging for monitoring the effects of thalidomide on experimental human breast cancers. Eur. Radiol. 2009, 1, 121–131. [Google Scholar]
- Zhao, D.; Jiang, L.; Hahn, E.W.; Mason, R.P. Continuous low-dose (metronomic) chemotherapy on rat prostate tumors evaluated using MRI in vivo and comparison with histology. Neoplasia 2005, 7, 678–687. [Google Scholar] [CrossRef]
- Lee, D.F.; Hung, M.C. All roads lead to mTOR: Integrating inflammation and tumor angiogenesis. Cell.Cycle 2007, 24, 3011–3014. [Google Scholar]
- Beauregard, D.A.; Thelwall, P.E.; Chaplin, D.J.; Hill, S.A.; Adams, G.E.; Brindle, K.M. Magnetic resonance imaging and spectroscopy of combretastatin A4 prodrug-induced disruption of tumour perfusion and energetic status. Br. J. Cancer 1998, 11, 1761–1767. [Google Scholar]
- Beauregard, D.A.; Hill, S.A.; Chaplin, D.J.; Brindle, K.M. The susceptibility of tumors to the antivascular drug combretastatin A4 phosphate correlates with vascular permeability. Cancer Res. 2001, 18, 6811–6815. [Google Scholar]
- Beauregard, D.A.; Pedley, R.B.; Hill, S.A.; Brindle, K.M. Differential sensitivity of two adenocarcinoma xenografts to the anti-vascular drugs combretastatin A4 phosphate and 5,6-dimethylxanthenone-4-acetic acid, assessed using MRI and MRS. NMR Biomed. 2002, 2, 99–105. [Google Scholar]
- Salmon, H.W.; Siemann, D.W. Effect of the second-generation vascular disrupting agent OXi4503 on tumor vascularity. Clin.Cancer Res. 2006, 13, 4090–4094. [Google Scholar]
- Lankester, K.J.; Maxwell, R.J.; Pedley, R.B.; Dearling, J.L.; Qureshi, U.A.; El-Emir, E.; Hill, S.A.; Tozer, G.M. Combretastatin A-4-phosphate effectively increases tumor retention of the therapeutic antibody, 131I-A5B7, even at doses that are sub-optimal for vascular shut-down. Int. J. Oncol. 2007, 2, 453–460. [Google Scholar]
- Salmon, B.A.; Salmon, H.W.; Siemann, D.W. Monitoring the treatment efficacy of the vascular disrupting agent CA4P. Eur. J. Cancer 2007, 10, 1622–1629. [Google Scholar]
- Nielsen, T.; Murata, R.; Maxwell, R.J.; Stødkilde-Jørgensen, H.; Ostergaard, L.; Horsman, M.R. Preclinical studies to predict efficacy of vascular changes induced by combretastatin a-4 disodium phosphate in patients. Int. J. Radiat. Oncol. Biol. Phys. 2008, 3, 859–866. [Google Scholar]
- Zhao, D.; Richer, E.; Antich, P.P.; Mason, R.P. Antivascular effects of combretastatin A4 phosphate in breast cancer xenograft assessed using dynamic bioluminescence imaging and confirmed by MRI. FASEB J. 2008, 7, 2445–2451. [Google Scholar]
- Nielsen, T.; Mouridsen, K.; Maxwell, R.J.; Stødkilde-Jørgensen, H.; Østergaard, L.; Horsman, M.R. Segmentation of dynamic contrast enhanced magnetic resonance imaging data. Acta Oncol. 2008, 7, 1265–1270. [Google Scholar]
- Nielsen, T.; Murata, R.; Maxwell, R.J.; Stodkilde-Jorgensen, H.; Ostergaard, L.; Ley, C.D.; Kristjansen, P.E.; Horsman, M.R. Non-invasive imaging of combretastatin activity in two tumor models: Association with invasive estimates. Acta Oncol. 2010, 7, 906–913. [Google Scholar]
- Bohndiek, S.E.; Kettunen, M.I.; Hu, D.E.; Witney, T.H.; Kennedy, B.W.; Gallagher, F.A.; Brindle, K.M. Detection of tumor response to a vascular disrupting agent by hyperpolarized 13C magnetic resonance spectroscopy. Mol. Cancer. Ther. 2010, 12, 3278–3288. [Google Scholar]
- Galbraith, S.M.; Maxwell, R.J.; Lodge, M.A.; Tozer, G.M.; Wilson, J.; Taylor, N.J.; Stirling, J.J.; Sena, L.; Padhani, A.R.; Rustin, G.J. S. Combretastatin A4 phosphate has tumor antivascular activity in rat and man as demonstrated by dynamic magnetic resonance imaging. J. Clin. Oncol. 2003, 15, 2831–2842. [Google Scholar]
- Zhao, D.; Jiang, L.; Hahn, E.W.; Mason, R.P. Tumor physiologic response to combretastatin A4 phosphate assessed by MRI. Int. J. Radiat. Oncol. Biol. Phys. 2005, 3, 872–880. [Google Scholar]
- Thoeny, H.C.; Keyzer, F.D.; Vandecaveye, V.; Chen, F.; Sun, X.; Bosmans, H.; Hermans, R.; Verbeken, E.K.; Boesch, C.; Marchal, G.; et al. Effect of vascular targeting agent in rat tumor model: Dynamic contrast-enhanced versus diffusion-weighted MR imaging. Radiology 2005, 2, 492–499. [Google Scholar]
- Wang, H.; Cona, M.M.; Chen, F.; Yu, J.; Feng, Y.; Li, J.; Keyzer, F.D.; Marchal, G.; Ni, Y. Comparison of two vascular-disrupting agents at a clinically relevant dose in rodent liver tumors with multiparametric magnetic resonance imaging biomarkers. Anticancer Drugs 2012, 1, 12–21. [Google Scholar]
- Evelhoch, J.L.; LoRusso, P.M.; He, Z.; DelProposto, Z.; Polin, L.; Corbett, T.H.; Langmuir, P.; Wheeler, C.; Stone, A.; Leadbetter, J.; et al. Magnetic resonance imaging measurements of the response of murine and human tumors to the vascular-targeting agent ZD6126. Clin. Cancer Res. 2004, 11, 3650–3657. [Google Scholar]
- Vogel-Claussen, J.; Gimi, B.; Artemov, D.; Bhujwalla, Z.M. Diffusion-weighted and macromolecular contrast enhanced MRI of tumor response to antivascular therapy with ZD6126. Cancer.Biol. Ther. 2007, 9, 1469–1475. [Google Scholar]
- Robinson, S.P.; McIntyre, D.J.O.; Checkley, D.; Tessier, J.J.; Howe, F.A.; Griffiths, J.R.; Ashton, S.E.; Ryan, A.J.; Blakey, D.C.; Waterton, J.C. Tumour dose response to the antivascular agent ZD6126 assessed by magnetic resonance imaging. Br. J. Cancer 2003, 10, 1592–1597. [Google Scholar]
- McIntyre, D.J.; Robinson, S.P.; Howe, F.A.; Griffiths, J.R.; Ryan, A.J.; Blakey, D.C.; Peers, I.S.; Waterton, J.C. Single dose of the antivascular agent, ZD6126 (N-acetylcolchinol-O-phosphate), reduces perfusion for at least 96 hours in the GH3 prolactinoma rat tumor model. Neoplasia 2004, 2, 150–157. [Google Scholar]
- Bradley, D.P.; Tessier, J.J.; Ashton, S.E.; Waterton, J.C.; Wilson, Z.; Worthington, P.L.; Ryan, A.J. Correlation of MRI biomarkers with tumor necrosis in Hras5 tumor xenograft in athymic rats. Neoplasia 2007, 5, 382–391. [Google Scholar]
- Wang, H.; Li, J.; Chen, F.; De Keyzer, F.; Yu, J.; Feng, Y.; Nuyts, J.; Marchal, G.; Ni, Y. Morphological, functional and metabolic imaging biomarkers: Assessment of vascular-disrupting effect on rodent liver tumours. Eur. Radiol. 2010, 8, 2013–2026. [Google Scholar]
- Bertelsen, L.B.; Shen, Y.Y.; Nielsen, T.; Stodkilde-Jorgensen, H.; Lloyd, G.K.; Siemann, D.W.; Horsman, M.R. Vascular effects of plinabulin (NPI-2358) and the influence on tumour response when given alone or combined with radiation. Int. J. Radiat. Biol. 2011, 11, 1126–1134. [Google Scholar]
- Durrant, D.; Corwin, F.; Simoni, D.; Zhao, M.; Rudek, M.A.; Salloum, F.N.; Kukreja, R.C.; Fatouros, P.P.; Lee, R.M. cis-3,4',5-Trimethoxy-3'-aminostilbene disrupts tumor vascular perfusion without damaging normal organ perfusion. Cancer Chemother. Pharmacol. 2009, 2, 191–200. [Google Scholar]
- Natsume, T.; Watanabe, J.; Ogawa, K.; Yasumura, K.; Kobayashi, M. Tumor-specific antivascular effect of TZT-1027 (Soblidotin) elucidated by magnetic resonance imaging and confocal laser scanning microscopy. Cancer.Sci. 2007, 4, 598–604. [Google Scholar]
- Luo, Y.; Hradil, V.P.; Frost, D.J.; Rosenberg, S.H.; Gordon, G.B.; Morgan, S.J.; Gagne, G.D.; Cox, B.F.; Tahir, S.K.; Fox, G.B. ABT-751, a novel tubulin-binding agent, decreases tumor perfusion and disrupts tumor vasculature. Anticancer Drugs 2009, 6, 483–492. [Google Scholar]
- Seshadri, M.; Spernyak, J.A.; Mazurchuk, R.; Camacho, S.H.; Oseroff, A.R.; Cheney, R.T.; Bellnier, D.A. Tumor vascular response to photodynamic therapy and the antivascular agent 5,6-dimethylxanthenone-4-acetic acid: implications for combination therapy. Clin.Cancer Res. 2005, 11, 4241–4250. [Google Scholar]
- Seshadri, M.; Mazurchuk, R.; Spernyak, J.A.; Bhattacharya, A.; Rustum, Y.M.; Bellnier, D.A. Activity of the vascular-disrupting agent 5,6-dimethylxanthenone-4-acetic acid against human head and neck carcinoma xenografts. Neoplasia 2006, 7, 534–542. [Google Scholar]
- Seshadri, M.; Spernyak, J.A.; Maiery, P.G.; Cheney, R.T.; Mazurchuk, R.; Bellnier, D.A. Visualizing the acute effects of vascular-targeted therapy in vivo using intravital microscopy and magnetic resonance imaging: Correlation with endothelial apoptosis, cytokine induction, and treatment outcome. Neoplasia 2007, 2, 128–135. [Google Scholar]
- Seshadri, M.; Bellnier, D.A.; Cheney, R.T. Assessment of the early effects of 5,6-dimethylxanthenone-4-acetic acid using macromolecular contrast media-enhanced magnetic resonance imaging: Ectopic versus orthotopic tumors. Int. J. Radiat. Oncol. Biol. Phys. 2008, 4, 1198–1207. [Google Scholar]
- Seshadri, M.; Ciesielski, M.J. MRI-based characterization of vascular disruption by 5,6-dimethylxanthenone-acetic acid in gliomas. J. Cereb. Blood Flow Metab. 2009, 8, 1373–1382. [Google Scholar]
- Seshadri, M.; Toth, K. Acute vascular disruption by 5,6-dimethylxanthenone-4-acetic Acid in an orthotopic model of human head and neck cancer. Transl. Oncol. 2009, 3, 121–127. [Google Scholar]
- Seshadri, M.; Sacadura, N.T.; Coulthard, T. Monitoring antivascular therapy in head and neck cancer xenografts using contrast-enhanced MR and US imaging. Angiogenesis 2011, 4, 491–501. [Google Scholar]
- Barbera, M.; Kettunen, M.I.; Caputo, A.; Hu, D.E.; Gobbi, S.; Brindle, K.M.; Carrara, M. Immune-modulating and anti-vascular activities of two xanthenone acetic acid analogues: A comparative study to DMXAA. Int. J. Oncol. 2009, 1, 273–279. [Google Scholar]
- McPhail, L.D.; McIntyre, D.J.; Ludwig, C.; Kestell, P.; Griffiths, J.R.; Kelland, L.R.; Robinson, S.P. Rat tumor response to the vascular-disrupting agent 5,6-dimethylxanthenone-4-acetic acid as measured by dynamic contrast-enhanced magnetic resonance imaging, plasma 5-hydroxyindoleacetic acid levels, and tumor necrosis. Neoplasia 2006, 3, 199–206. [Google Scholar]
- Tang, J.S.; Choy, G.; Bernardo, M.; Thomasson, D.; Libutti, S.K.; Choyke, P.L. Dynamic contrast-enhanced magnetic resonance imaging in the assessment of early response to tumor necrosis factor alpha in a colon carcinoma model. Invest. Radiol. 2006, 9, 691–696. [Google Scholar]
- Haney, C.R.; Parasca, A.D.; Fan, X.; Bell, R.M.; Zamora, M.A.; Karczmar, G.S.; Mauceri, H.J.; Halpern, H.J.; Weichselbaum, R.R.; Pelizzari, C.A. Characterization of response to radiation mediated gene therapy by means of multimodality imaging. Magn.Reson. Med. 2009, 2, 348–356. [Google Scholar]
- Kiessling, F.; Huber, P.E.; Grobholz, R.; Heilmann, M.; Meding, J.; Lichy, M.P.; Fink, C.; Krix, M.; Peschke, P.; Schlemmer, H. Dynamic magnetic resonance tomography and proton magnetic resonance spectroscopy of prostate cancers in rats treated by radiotherapy. Invest. Radiol. 2004, 1, 34–44. [Google Scholar]
- De Keyzer, F.; Vandecaveye, V.; Thoeny, H.; Chen, F.; Ni, Y.; Marchal, G.; Hermans, R.; Nuyts, S.; Landuyt, W.; Bosmans, H. Dynamic contrast-enhanced and diffusion-weighted MRI for early detection of tumoral changes in single-dose and fractionated radiotherapy: Evaluation in a rat rhabdomyosarcoma model. Eur. Radiol. 2009, 11, 2663–2671. [Google Scholar]
- Zilberstein, J.; Schreiber, S.; Bloemers, M.C.; Bendel, P.; Neeman, M.; Schechtman, E.; Kohen, F.; Scherz, A.; Salomon, Y. Antivascular treatment of solid melanoma tumors with bacteriochlorophyll-serine-based photodynamic therapy. Photochem. Photobiol. 2001, 3, 257–266. [Google Scholar]
- Eichhorn, M.E.; Becker, S.; Strieth, S.; Werner, A.; Sauer, B.; Teifel, M.; Ruhstorfer, H.; Michaelis, U.; Griebel, J.; Brix, G.; et al. Paclitaxel encapsulated in cationic lipid complexes (MBT-0206) impairs functional tumor vascular properties as detected by dynamic contrast enhanced magnetic resonance imaging. Cancer. Biol. Ther. 2006, 1, 89–96. [Google Scholar]
- Eichhorn, M.E.; Luedemann, S.; Strieth, S.; Papyan, A.; Ruhstorfer, H.; Haas, H.; Michaelis, U.; Sauer, B.; Teifel, M.; Enders, G.; et al. Cationic lipid complexed camptothecin (EndoTAG-2) improves antitumoral efficacy by tumor vascular targeting. Cancer. Biol. Ther. 2007, 6, 920–929. [Google Scholar] [CrossRef]
- O’Connor, J.P.; Jackson, A.; Parker, G.J.; Roberts, C.; Jayson, G.C. Dynamic contrast-enhanced MRI in clinical trials of antivascular therapies. Nat. Rev. Clin. Oncol. 2012, 3, 167–177. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nielsen, T.; Wittenborn, T.; Horsman, M.R. Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) in Preclinical Studies of Antivascular Treatments. Pharmaceutics 2012, 4, 563-589. https://doi.org/10.3390/pharmaceutics4040563
Nielsen T, Wittenborn T, Horsman MR. Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) in Preclinical Studies of Antivascular Treatments. Pharmaceutics. 2012; 4(4):563-589. https://doi.org/10.3390/pharmaceutics4040563
Chicago/Turabian StyleNielsen, Thomas, Thomas Wittenborn, and Michael R. Horsman. 2012. "Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) in Preclinical Studies of Antivascular Treatments" Pharmaceutics 4, no. 4: 563-589. https://doi.org/10.3390/pharmaceutics4040563
APA StyleNielsen, T., Wittenborn, T., & Horsman, M. R. (2012). Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI) in Preclinical Studies of Antivascular Treatments. Pharmaceutics, 4(4), 563-589. https://doi.org/10.3390/pharmaceutics4040563