A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells
Abstract
:1. Introduction
2. Experimental Section
2.1. Synthetic membranes
| Membrane | Polymera | MWCO (kDa) | Pore size (µm) | Thickness(µm) | ρ (%) | τ | Source | Batch no. |
|---|---|---|---|---|---|---|---|---|
| Cellulose-based | ||||||||
| Visking | RC | 12-14 | - | 20b | - | - | Medicell (London, UK) | DTV12000.05.000 |
| Cuprophan | RC | 10 | - | 10b | - | - | Medicell (London, UK) | N/A |
| Benzoylated tubing | RC | 1.2-2 | - | 35b | - | - | Sigma (Dorset,UK) | 074K7012 |
| Cellulose ester | CE | 0.5 | - | 80b | - | - | Spectrumlab (California) | 131060 |
| Cellulose nitrate | CN | 0.45 | 125 | 66-84 | - | Whatman (UK) | N/A | |
| Polymeric-based | ||||||||
| AN 69 | PAN | 40 | - | 25b | - | - | Hospal (Huntingdon, UK) | N/A |
| Biodyne | PA | na | 0.45 | 152 | 50- | - | Pall (Portsmouth, UK) | b- 50046, c-189051 |
| Supor | PES | na | 0.45 | 145 | 75 | ~1-1.5 | Pall (Portsmouth, UK) | 55083 |
| Tuffryn | PS | na | 0.45 | 145 | 80 | ~1-1.5 | Pall (Portsmouth, UK) | 60669 |
| Nuclepore | PC | na | 0.1 | 10 | 60 | ~1 | Whatman (New Jersey, USA) | 6018023 |
| Cyclopore | PC | na | 0.1 | 10 | 8 | ~1 | Whatman (New Jersey, USA) | 060.0131/6E8/L-3-L |
| Celgard 3500 | PP | na | 0.05 | 20 | 4 | - | Hoechst (New Jersey, USA) | 293485 |
| Silicone | PDMS | na | - | 400 | 35-48 | - | SAMCO (Nuneaton, UK) | 19T0.3-1000-60M1 |
2.2. Franz diffusion cells apparatus
2.3. Other materials
2.4. Preparation of ibuprofen saturated solution
2.5. Ibuprofen Saturated solubility
2.6. Preparation of ibuprofen crystals
2.7. Membrane treatment
2.8. Franz diffusion cell studies
2.8.1. Effect of synthetic membranes on ibuprofen drug diffusion
2.8.2. Comparison of ibuprofen drug diffusion through membrane of different pore sizes and surface groups
2.8.3. Regression analysis of ibuprofen flux with membrane parameters
2.9. Statistical analysis
3. Results and Discussion
3.1. Drug solubility and mass balance
| Preparation # | Amount of ibuprofen added (g) | Amount remaining on the filter paper (g) | Amout of ibuprofen dissolved in 50 mL (g) | Concentration% (w/v) |
|---|---|---|---|---|
| 1 | 1.657 | 0.616 | 1.041 | 2.082 |
| 2 | 1.621 | 0.591 | 1.030 | 2.060 |
| 3 | 1.637 | 0.579 | 1.058 | 2.116 |
| 4 | 1.620 | 0.559 | 1.061 | 2.122 |
| 5 | 1.767 | 0.746 | 1.021 | 2.042 |
3.2. Ibuprofen drug diffusion across synthetic membranes
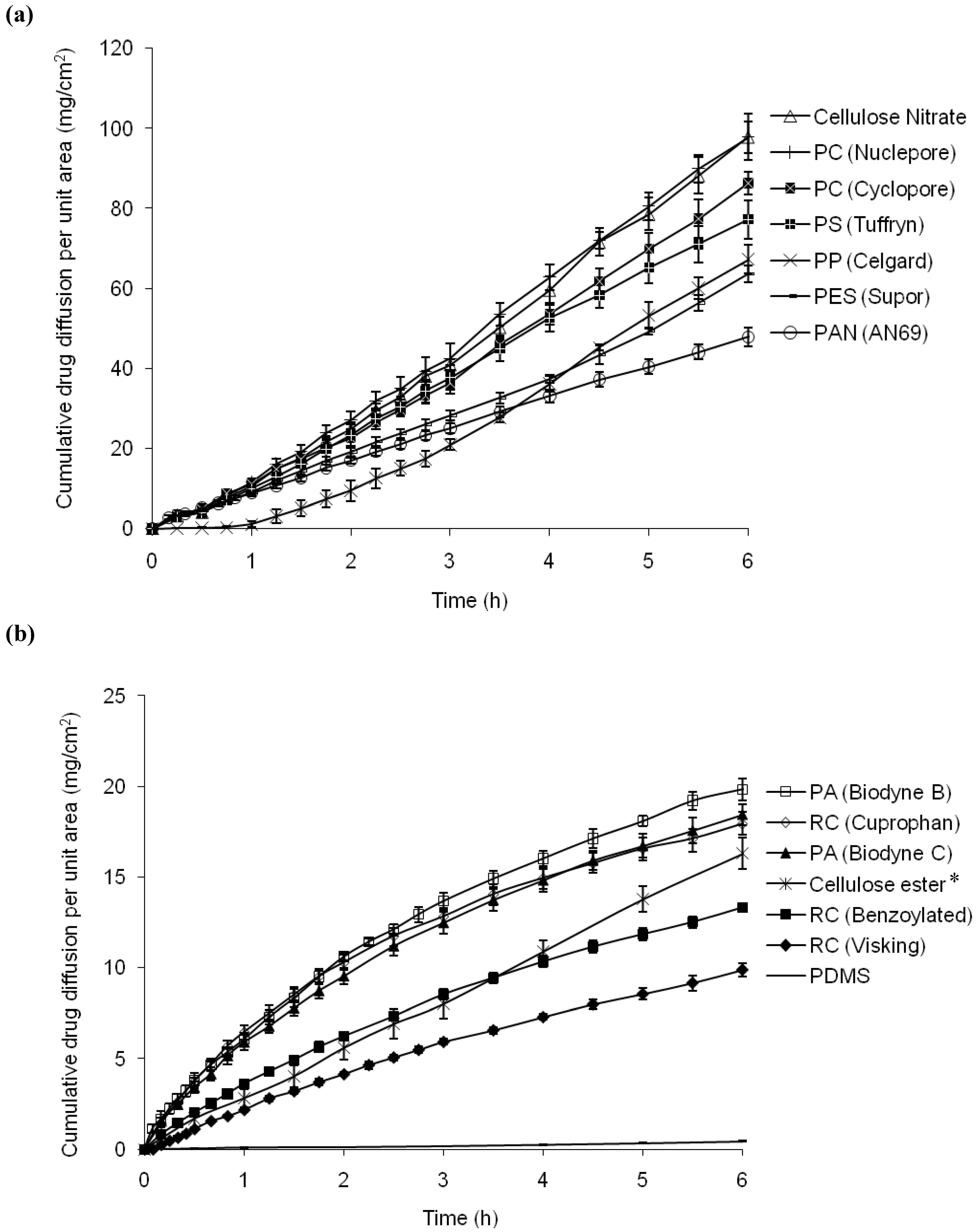
| Membrane | Flux (mg/cm2/h) | Total diffusion after 6 h (mg/cm2) | CV (%) |
|---|---|---|---|
| Cellulose nitrate | 17.65 ± 2.06 | 97.89 ± 5.79 | 11.7 |
| Nuclepore | 17.38 ± 0.79 | 97.75 ± 4.01 | 4.6 |
| Celgard | 15.45 ± 1.02 | 67.28 ± 3.66 | 6.6 |
| Cyclopore | 14.87 ± 0.50 | 86.32 ± 2.77 | 3.4 |
| Tuffryn | 13.54 ± 0.49 | 77.23 ± 4.80 | 3.6 |
| Supor | 10.48 ± 0.31 | 63.63 ± 2.08 | 2.9 |
| AN69 | 8.14 ± 0.38 | 47.84 ± 2.25 | 4.7 |
| *Cellulose ester | 2.66 ± 0.19 | 16.30 ± 0.84 | 7.3 |
| Biodyne B | 1.96 ± 0.07 | 19.32 ± 0.62 | 3.6 |
| Biodyne C | 1.77 ± 0.17 | 18.44 ± 0.59 | 4.4 |
| Cuprophan | 1.57 ± 0.15 | 17.94 ± 0.69 | 4.7 |
| Benzoylated cellulose | 1.51 ± 0.04 | 13.32 ± 0.24 | 2.9 |
| Visking | 1.39 ± 0.09 | 9.88 ± 0.37 | 6.2 |
| PDMS | 0.09 ± 0.01 | 0.45 ± 0.002 | 6.0 |
3.3. Correlation of flux with membrane parameters
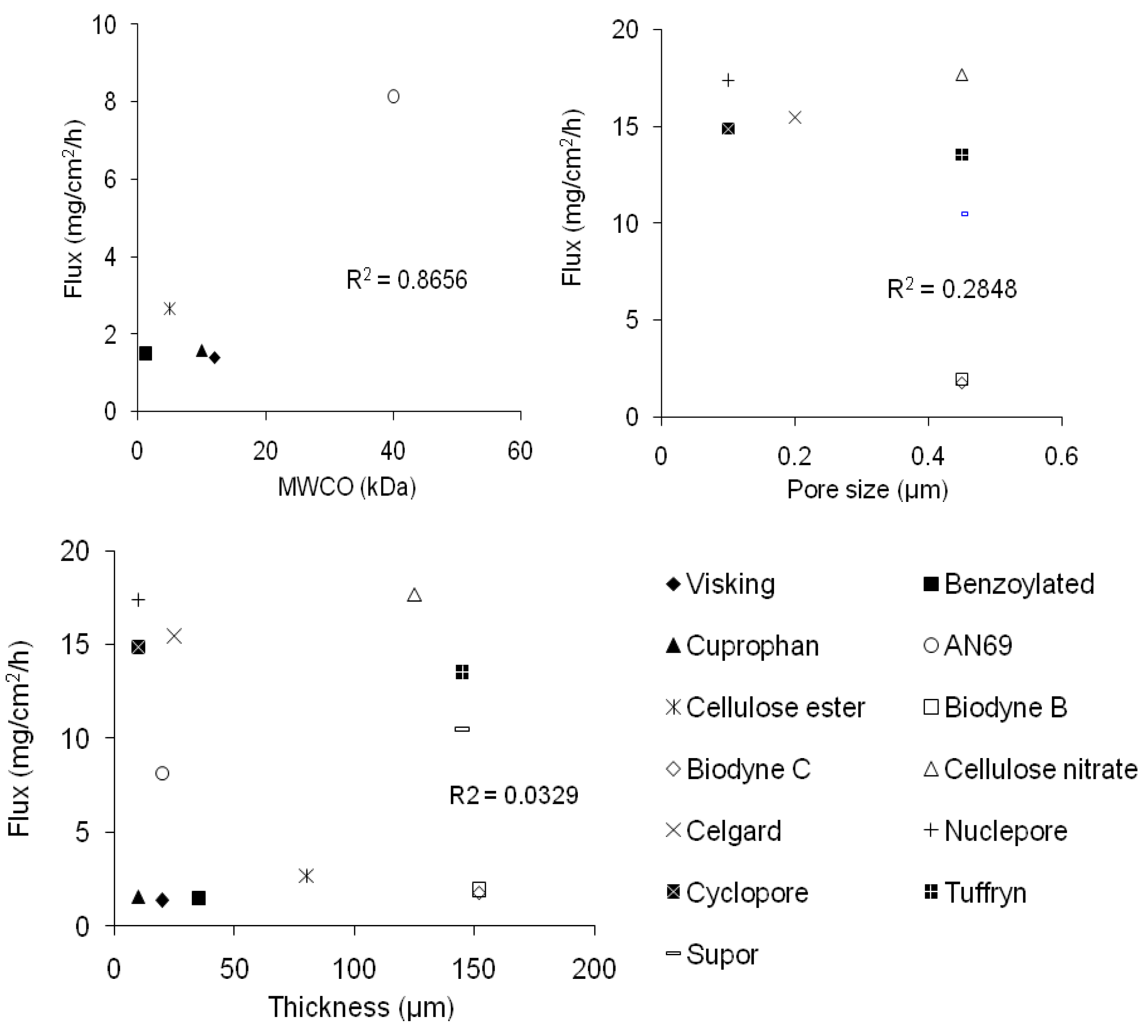

3.4. Discussion
4. Conclusions
Acknowledgements
References
- Franz, T.J. Percutaneous absorption. On the relevance of In vitro data. J. Invest. Dermatol. 1975, 64, 190–195. [Google Scholar]
- Franz, T.J. The finite dose technique as a valid in vitro model for the study of percutaneous absorption. Curr. Probl. Dermatol. 1978, 7, 58–68. [Google Scholar]
- Zorin, S.; Kuylenstierna, F.; Thulin, H. In vitro test of Nicotine's permeability through human skin. Risk evaluation and safety aspects. Am. Occup. Hyg. 1999, 43, 405–413. [Google Scholar]
- Siewert, M.; Dressman, J.; Brown, C.K.; Shah, V.P. FIP/AAPS guidelines to dissolution/In vitro release testing of novel/special dosage forms. AAPS PharmSci. Tech. 2003, 4, Article 7. [Google Scholar]
- Shah, V.; Elkins, J.; Williams, R. Evaluation of the test system used for in vitro release of drugs for topical dermatological drug products. Pharm. Dev. Technol. 1999, 4, 377–385. [Google Scholar] [CrossRef]
- Ueda, C.T.; Shah, V.P.; Derdzinski, K.; Ewing, G.; Flynn, G.; Maibach, H.; Marques, M.; Rytting, H.; Shaw, S.; Thakker, K.; Yacobi, A. Topical and Transdermal Drug Products. Pharmacopeial Forum. 2009, 35, 750–764. [Google Scholar]
- Twist, J.; Zatz, J. Membrane - solvent - solute interaction in a model permeation system. J. Pharm. Sci. 1988, 77, 538–540. [Google Scholar]
- Twist, J.N.; Zatz, J.L. Influence of solvents on paraben permeation through idealized skin model membranes. J. Soc. Cosmet. Chem. 1986, 37, 429–444. [Google Scholar]
- Corbo, M.; Schultz, T.W.; Wong, G.K.; van Buskirk, G.A. Development and validation of in vitro release testing methods for semisolid formulations. Pharm. Tech. 1993, 9, 112–128. [Google Scholar]
- Pellett, M.A.; Castellano, S.; Hadgraft, J.; Davis, A.F. The penetration of supersaturated solutions of piroxicam across silicone membranes and human skin in vitro. J. Control. Release 1997, 46, 205–214. [Google Scholar] [CrossRef]
- Addicks, W.J.; Flynn, G.L.; Weiner, N. Validation of a flow-through diffusion cell for use in transdermal research. Pharm. Res. 1987, 4, 337–341. [Google Scholar] [CrossRef]
- Bonferoni, M.C.; Rossi, S.; Ferrari, F.; Caramella, C. A modified Franz diffusion cell for simultaneous assessment of drug release and washability of mucoadhesive gels. Pharm. Dev. Tech. 1999, 4, 45–53. [Google Scholar]
- Chattaraj, S.; Kanfer, I. The insertion cell': a novel approach to monitor drug release from semi-solid dosage forms. Int. J. Pharm. 1996, 133, 59–63. [Google Scholar] [CrossRef]
- Chattaraj, S.C.; Kanfer, I. Release of acyclovir from semisolid dosage forms: a semi-automated procedure using a simple plexiglass flow-through cell. Int. J. Pharm. 1995, 125, 215–222. [Google Scholar] [CrossRef]
- Clement, P.; Laugel, C.; Marty, J.-P. Influence of three synthetic membranes on the release of caffeine from concentrated w/o emulsions. J. Controlled Release 2000, 66, 243–254. [Google Scholar] [CrossRef]
- Dias, M.; Raghavan, S.L.; Hadgraft, J. ATR-FTIR spectroscopic investigation on the effect of solvents on the permeation of benzoic acid and salicylic acid through silicone membranes. Int. J. Pharm. 2001, 216, 51–59. [Google Scholar] [CrossRef]
- Friend, D.; Catz, P.; Heller, J.; Okagaki, M. Transdermal delivery of Levonorgestrel IV: Evaluation of membranes. J. Pharm. Sci. 1989, 78, 477–481. [Google Scholar] [CrossRef]
- Liaw, J.; Lin, Y.-C. Evaluation of poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) (PEO-PPO-PEO) gel as a release vehicle for percutaneous fentanyl. J. Control. Release 2000, 68, 273–282. [Google Scholar] [CrossRef]
- Morell, J.L.P.; Claramonte, M.D.C.; Vialard, A.P. Validation of a release diffusion cell for topical dosage forms. Int. J. Pharm. 1996, 137, 49–55. [Google Scholar] [CrossRef]
- Proniuk, S.; Dixon, S.E.; Blanchard, J. Investigation of the utility of an in vitro release test for optimizing semisolid dosage forms. Pharm. Dev. Technol. 2001, 6, 469–476. [Google Scholar] [CrossRef]
- Santoyo, S.; Arrelano, A.; Ygartua, P.; Martin, C. In vitro percutaneous absorption of piroxicam through synthetic membranes and abdominal rat skin. Pharm. Acta. Helv. 1996, 71, 141–146. [Google Scholar] [CrossRef]
- Schwarb, F.P.; Imanidis, G.; Smith, E.W.; Haigh, J.M.; Surber, C. Effect of concentration and degree of saturation of topical fluocinonide formulations on in vitro membrane transport and in vivo availability on human skin. Pharm. Res. 1999, 16, 909–915. [Google Scholar] [CrossRef]
- Shah, V.; Elkins, J.; Hanus, J.; Noorizadeh, C.; Skelly, J. In vitro release of Hydrocortisone from topical preparations and automated procedure. Pharm. Res. 1991, 8, 55–59. [Google Scholar] [CrossRef]
- Shah, V.; Elkins, J.; Williams, R. In vitro drug release measurement for topical glucocorticoid creams. Pharmacopeial Forum 1993, 19, 5048–5060. [Google Scholar]
- Smith, E.W.; Haigh, J.M. In vitro diffusion cell design and validation II. Temperature, agitation and membrane effects on betamethasone 17-valerate permeation. Acta. Pharm. Nord. 1992, 4, 171–178. [Google Scholar]
- Thakker, K.D.; Chern, H. Development and validation of in vitro release tests for semisolid dosage forms - case study. Disso. Tech. 2003, 10, 10–15. [Google Scholar]
- Welin-Berger, K.; Neelissen, J.A.M.; Bergenstahl, B. The effect of rheological behaviour of a topical anaesthetic formulations on the release and permeation rates of the active compound. Eur. J. Pharm. Sci. 2001, 13, 309–318. [Google Scholar]
- FDA-SUPAC-SS. Guidance for Industry. SUPAC-SS Non-sterile Semisolid Dosage Forms. Scale-up and Postapproval Changes: Chemistry, Manufacturing and Controls. In In vitro Release Testing and In Vivo Bioequivalence Documentation ; 1997; pp. 19–24.
- Shah, V.P.; Elkins, J.S.; Lam, S.Y.; Skelly, J.P. Determination of in vitro drug release from hydrocortisone creams. Int. J. Pharm. 1989, 53, 53–39. [Google Scholar] [CrossRef]
- Wu, S.T.; Shiu, G.K.; Simmons, J.E.; Bronaugh, R.L.; Skelly, J.P. In vitro release of nitroglycerin from topical products by use of artificial membranes. J. Pharm. Sci. 1992, 81, 1153–1156. [Google Scholar] [CrossRef]
- Gallagher, S.J.; Trottet, L.; Carter, T.P.; Heard, C.M. Effects of membrane type and liquid/liquid phase boundary on in vitro release of Ketoprofen from gel formulations. J. Drug target. 2003, 11, 373–379. [Google Scholar] [CrossRef]
- Hatanaka, T.; Inuma, M.; Morimoto, Y. Prediction of skin permeability of drugs. 1. Comparison with artificial membrane. Chem. Pharm. Bull. 1990, 38, 3452–3459. [Google Scholar] [CrossRef]
- Chenoweth, D.E.; Cheung, A.K.; Henderson, L.W. Anaphylatoxin formation during hemodialysis: Effects of different dialyzer membranes. Kidney Int. 1983, 24, 764–769. [Google Scholar] [CrossRef]
- Bellomo, R.; Ronco, C. An Introduction to continuous renal replacement therapy. In Atlas of Hemodialysis; Bellomo, R., Baldwin, I., Ronco, C., Thomas, G., Eds.; Bailliere Tindall: London, UK, 2001; Volume 1, pp. 1–10. [Google Scholar]
- Cronin, M.T.D.; Dearden, J.C.; Moss, G.P.; Murray-Dickson, G. Investigation of the mechanism of flux across human skin in vitro by quantitative structure-permeability relationship. Eur. J. Pharm. Sci. 1998, 7, 325–330. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ng, S.-F.; Rouse, J.; Sanderson, D.; Eccleston, G. A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells. Pharmaceutics 2010, 2, 209-223. https://doi.org/10.3390/pharmaceutics2020209
Ng S-F, Rouse J, Sanderson D, Eccleston G. A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells. Pharmaceutics. 2010; 2(2):209-223. https://doi.org/10.3390/pharmaceutics2020209
Chicago/Turabian StyleNg, Shiow-Fern, Jennifer Rouse, Dominic Sanderson, and Gillian Eccleston. 2010. "A Comparative Study of Transmembrane Diffusion and Permeation of Ibuprofen across Synthetic Membranes Using Franz Diffusion Cells" Pharmaceutics 2, no. 2: 209-223. https://doi.org/10.3390/pharmaceutics2020209




