Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals
Abstract
1. Introduction
2. Implementation of Lutetium-177 Therapy
2.1. Patient Eligibility
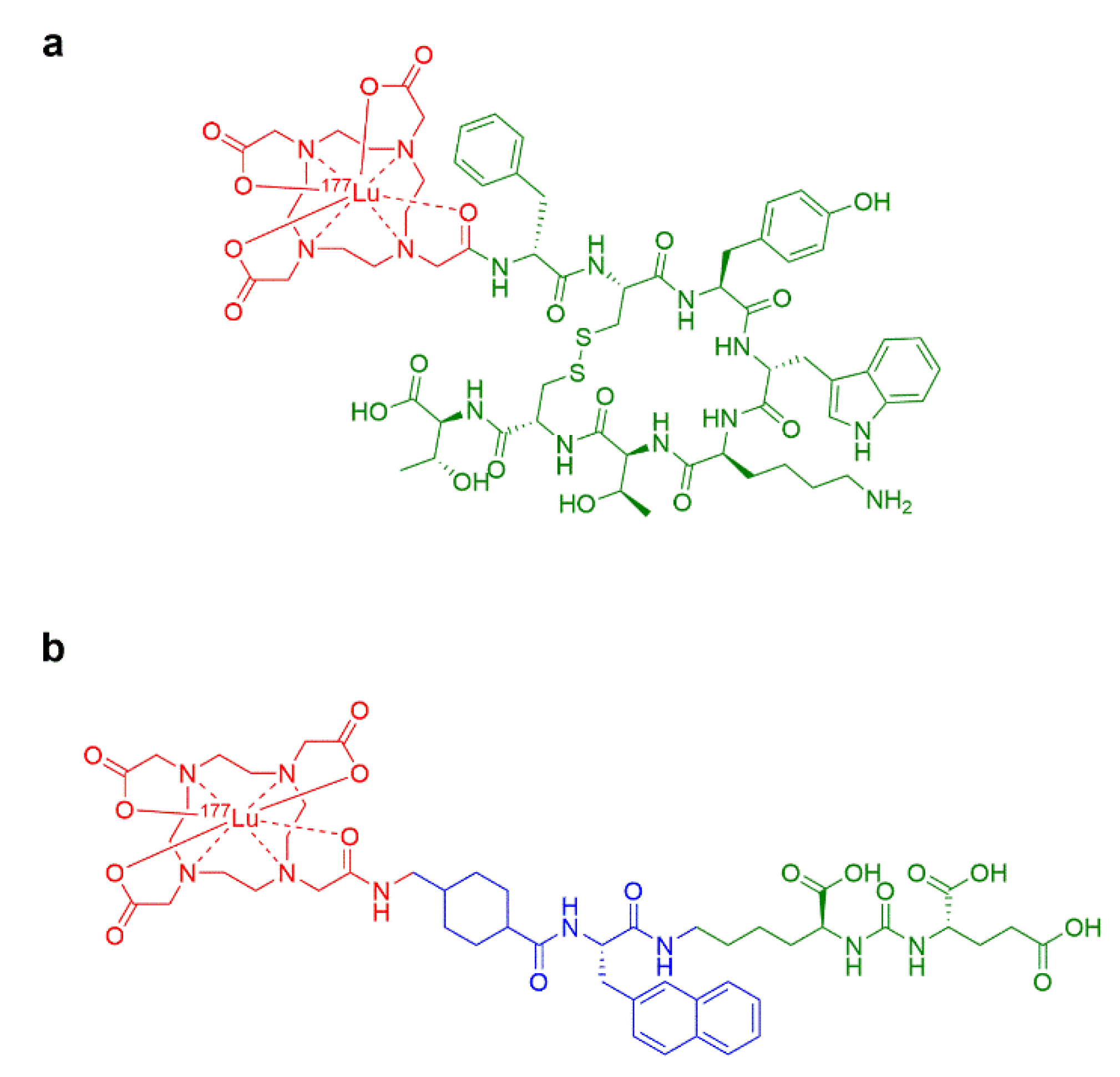
2.1.1. [177Lu]Lu-DOTATATE (Lutathera®)
2.1.2. [177Lu]Lu-vipivotide Tetraxetan (Pluvicto®)
2.2. Procedure of Treatment
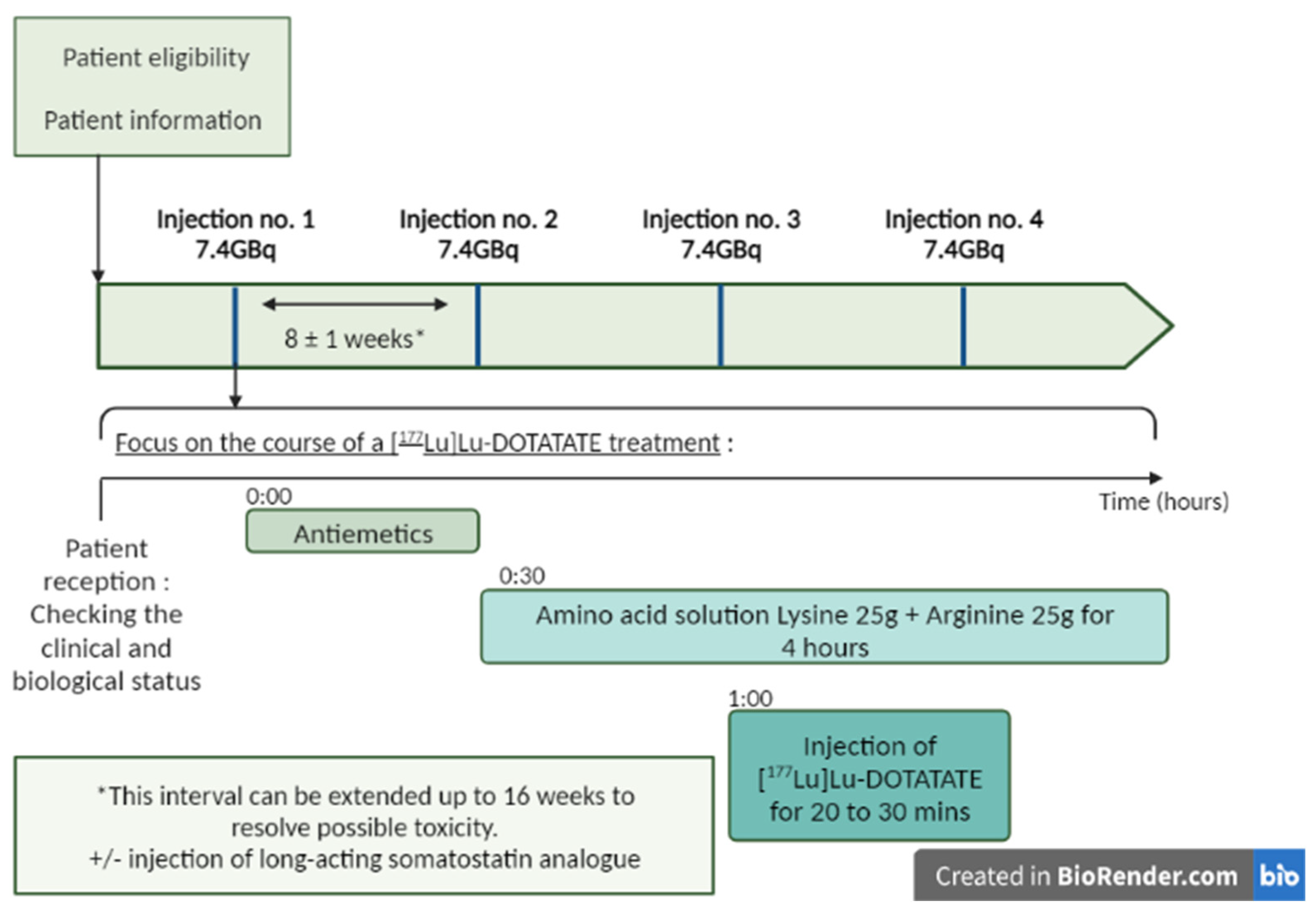
2.2.1. [¹⁷⁷Lu]Lu-DOTATATE (Lutathera®)
2.2.2. [177Lu]Lu-Vipivotide Tetraxetan (Pluvicto®)
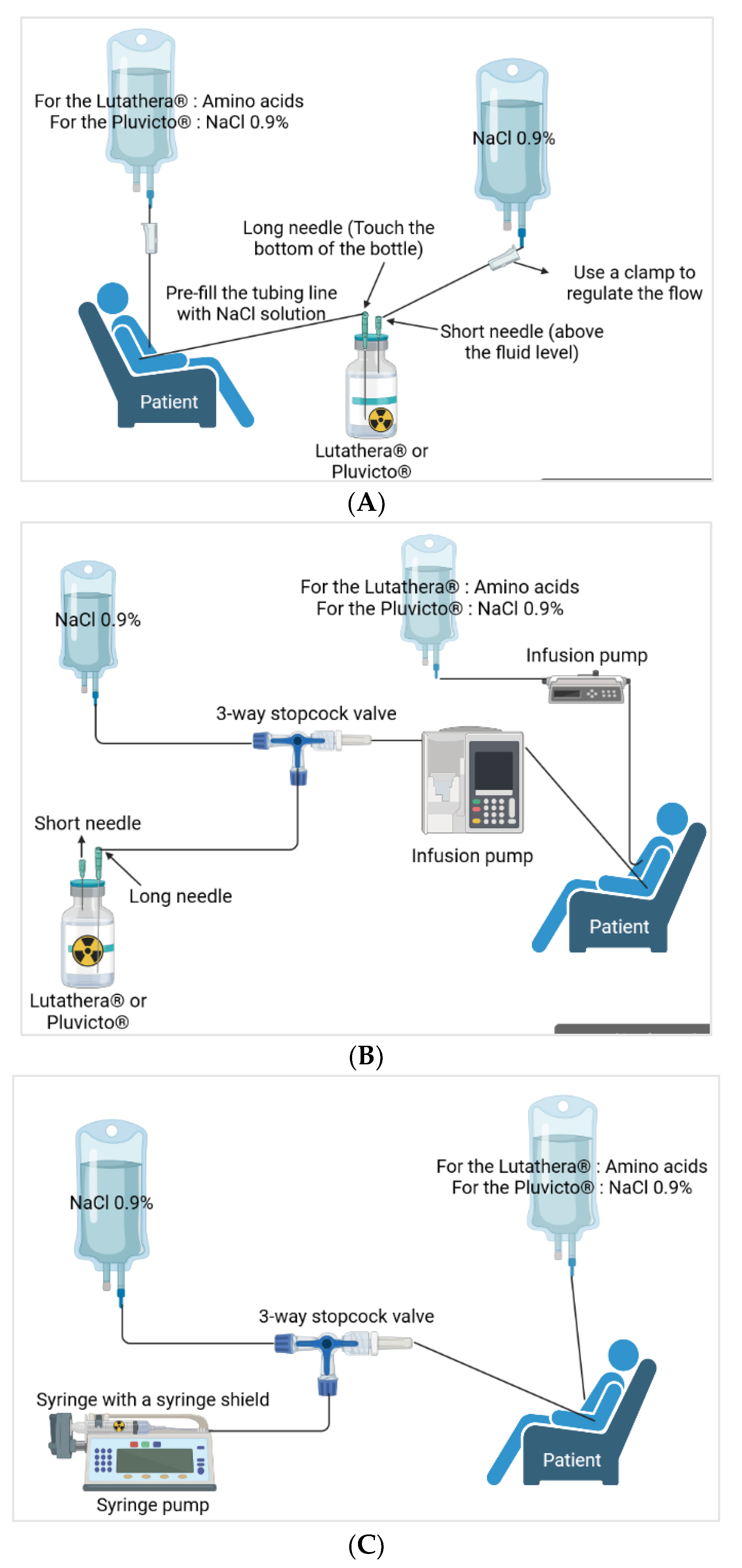
2.3. Premedication and Administration
2.3.1. [¹⁷⁷Lu]Lu-DOTATATE (Lutathera®)
2.3.2. [177Lu]Lu-Vipivotide Tetraxetan (Pluvicto®)
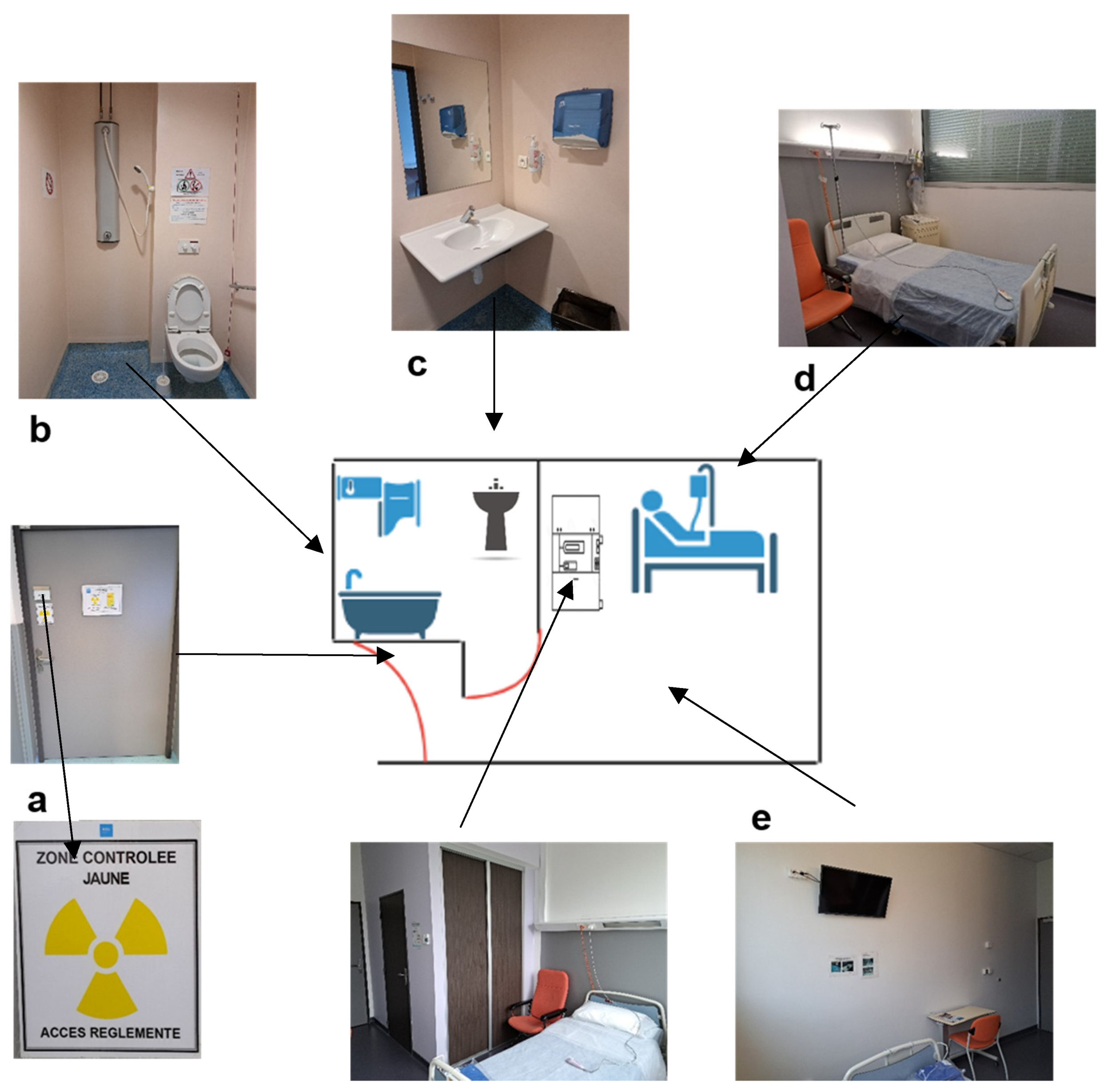
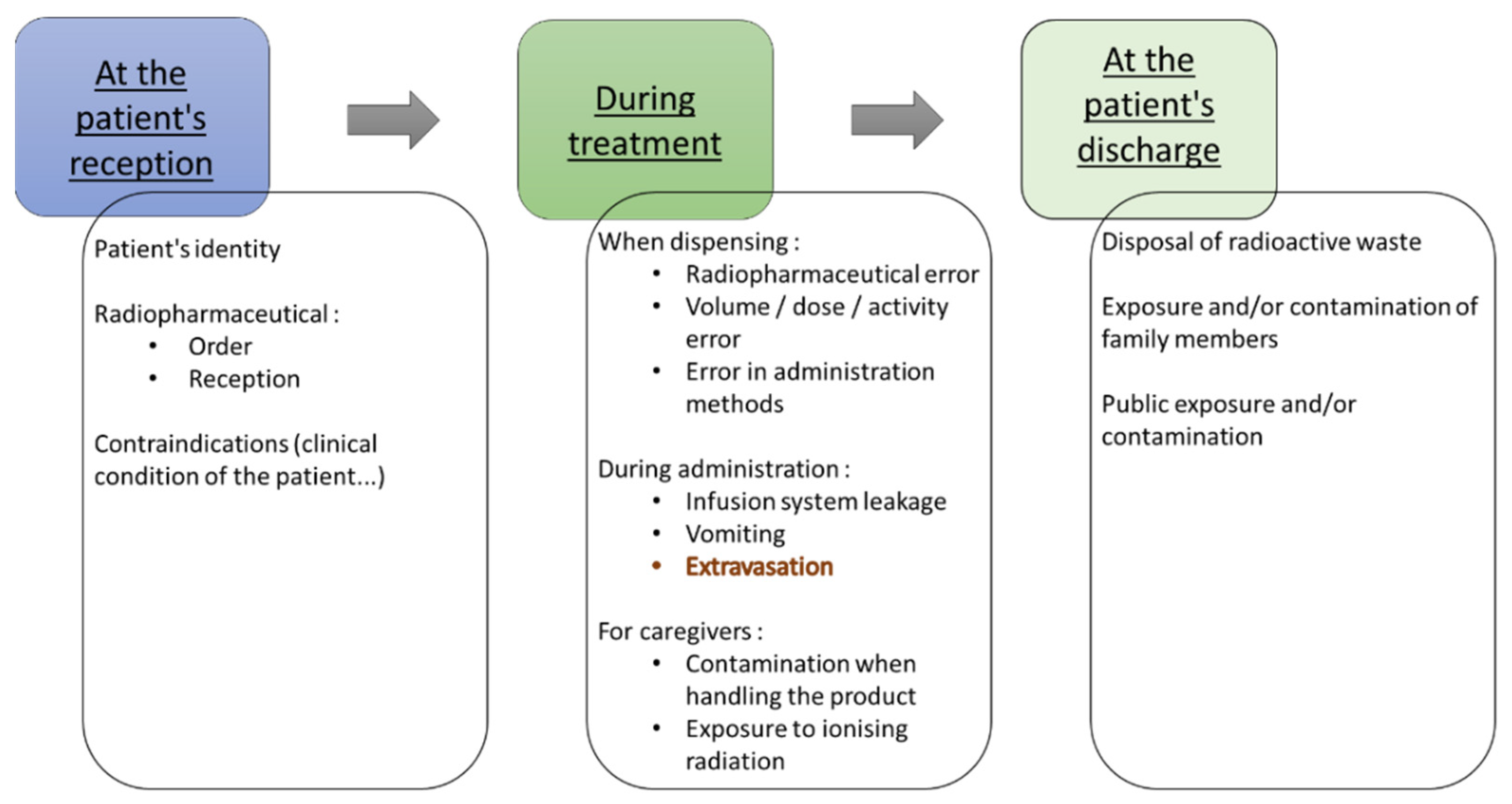
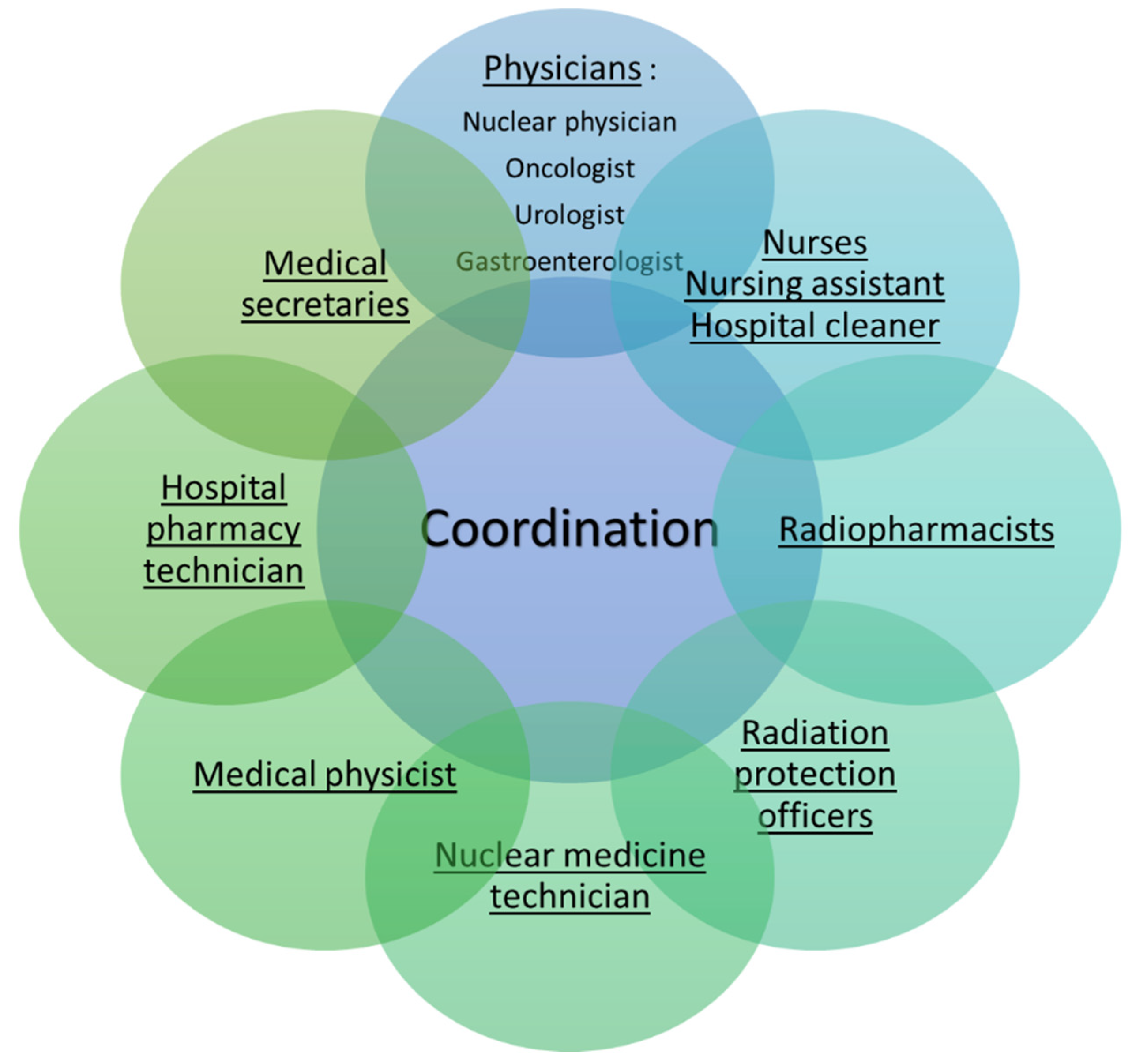
3. Radioprotection and Risk Management
4. Safety and Pharmacovigilance
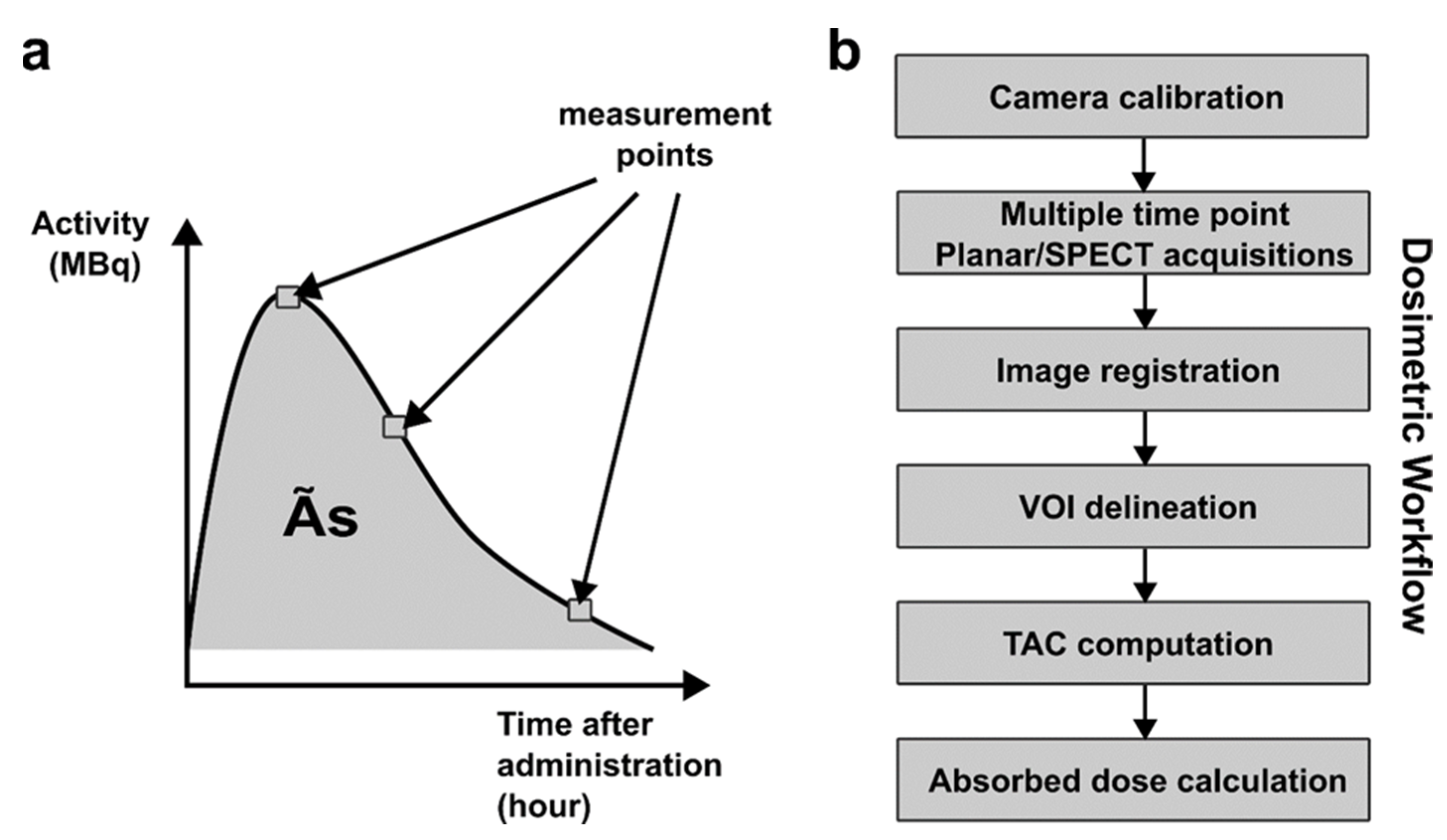
5. Internal radiotherapy with 177Lu: Toward Personalized Dosimetry
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Levine, R.; Krenning, E.P. Clinical History of the Theranostic Radionuclide Approach to Neuroendocrine Tumors and Other Types of Cancer: Historical Review Based on an Interview of Eric P. Krenning by Rachel Levine. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2017, 58, 3S–9S. [Google Scholar] [CrossRef] [PubMed]
- Seidlin, S.M.; Marinelli, L.D.; Oshry, E. Radioactive iodine therapy: Effect on functioning metastases of adenocarcinoma of the thyroid. CA. Cancer J. Clin. 1990, 40, 299–317. [Google Scholar] [CrossRef]
- Bodei, L.; Herrmann, K.; Schöder, H.; Scott, A.M.; Lewis, J.S. Radiotheranostics in oncology: Current challenges and emerging opportunities. Nat. Rev. Clin. Oncol. 2022, 19, 534–550. [Google Scholar] [CrossRef] [PubMed]
- Dolgin, E. Radioactive drugs emerge from the shadows to storm the market. Nat. Biotechnol. 2018, 36, 1125–1127. [Google Scholar] [CrossRef] [PubMed]
- Sherman, M.; Levine, R. Nuclear Medicine and Wall Street: An Evolving Relationship. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2019, 60, 20S–24S. [Google Scholar] [CrossRef] [PubMed]
- Boros, E.; Packard, A.B. Radioactive Transition Metals for Imaging and Therapy. Chem. Rev. 2019, 119, 870–901. [Google Scholar] [CrossRef]
- Brandt, M.; Cardinale, J.; Aulsebrook, M.L.; Gasser, G.; Mindt, T.L. An Overview of PET Radiochemistry, Part 2: Radiometals. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2018, 59, 1500–1506. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczak, R.; van der Meulen, N.P.; Lapi, S.E. Radiometals for imaging and theranostics, current production, and future perspectives. J. Label. Compd. Radiopharm. 2019, 62, 615–634. [Google Scholar] [CrossRef]
- Notni, J.; Wester, H.-J. Re-thinking the role of radiometal isotopes: Towards a future concept for theranostic radiopharmaceuticals. J. Label. Compd. Radiopharm. 2018, 61, 141–153. [Google Scholar] [CrossRef]
- Dash, A.; Pillai, M.R.A.; Knapp, F.F. Production of 177Lu for Targeted Radionuclide Therapy: Available Options. Nucl. Med. Mol. Imaging 2015, 49, 85–107. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Pillai, M.R.A.; Knapp, F.F. (Russ) Lutetium-177 Therapeutic Radiopharmaceuticals: Linking Chemistry, Radiochemistry, and Practical Applications. Chem. Rev. 2015, 115, 2934–2974. [Google Scholar] [CrossRef]
- Chakravarty, R.; Chakraborty, S. A review of advances in the last decade on targeted cancer therapy using 177Lu: Focusing on 177Lu produced by the direct neutron activation route. Am. J. Nucl. Med. Mol. Imaging 2021, 11, 443–475. [Google Scholar]
- Mathur, A.; Prashant, V.; Sakhare, N.; Chakraborty, S.; Vimalnath, K.V.; Mohan, R.K.; Arjun, C.; Karkhanis, B.; Seshan, R.; Basu, S.; et al. Bulk Scale Formulation of Therapeutic Doses of Clinical Grade Ready-to-Use 177Lu-DOTA-TATE: The Intricate Radiochemistry Aspects. Cancer Biother. Radiopharm. 2017, 32, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K.; Schwaiger, M.; Lewis, J.S.; Solomon, S.B.; McNeil, B.J.; Baumann, M.; Gambhir, S.S.; Hricak, H.; Weissleder, R. Radiotheranostics: A roadmap for future development. Lancet Oncol. 2020, 21, e146–e156. [Google Scholar] [CrossRef] [PubMed]
- Talip, Z.; Favaretto, C.; Geistlich, S.; van der Meulen, N.P. A Step-by-Step Guide for the Novel Radiometal Production for Medical Applications: Case Studies with 68Ga, 44Sc, 177Lu and 161Tb. Molecules 2020, 25, 966. [Google Scholar] [CrossRef]
- Dash, A.; Chakraborty, S.; Pillai, M.R.A.; Knapp, F.F.R. Peptide receptor radionuclide therapy: An overview. Cancer Biother. Radiopharm. 2015, 30, 47–71. [Google Scholar] [CrossRef]
- Valkema, R.; de Jong, M.; Bakker, W.H.; Breeman, W.A.P.; Kooij, P.P.M.; Lugtenburg, P.J.; de Jong, F.H.; Christiansen, A.; Kam, B.L.R.; de Herder, W.W.; et al. Phase I Study of Peptide Receptor Radionuclide Therapy With [lllIn-DTPA~ The Rotterdam Experience. Semin. Nucl. Med. 2002, 32, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Pinto, J.T.; Suffoletto, B.P.; Berzin, T.M.; Qiao, C.H.; Lin, S.; Tong, W.P.; May, F.; Mukherjee, B.; Heston, W.D. Prostate-specific membrane antigen: A novel folate hydrolase in human prostatic carcinoma cells. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 1996, 2, 1445–1451. [Google Scholar]
- Halsted, C.H.; Ling, E.H.; Luthi-Carter, R.; Villanueva, J.A.; Gardner, J.M.; Coyle, J.T. Folylpoly-gamma-glutamate carboxypeptidase from pig jejunum. Molecular characterization and relation to glutamate carboxypeptidase II. J. Biol. Chem. 1998, 273, 20417–20424. [Google Scholar] [CrossRef]
- Hofman, M.S.; Violet, J.; Hicks, R.J.; Ferdinandus, J.; Thang, S.P.; Akhurst, T.; Iravani, A.; Kong, G.; Ravi Kumar, A.; Murphy, D.G.; et al. [177Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): A single-centre, single-arm, phase 2 study. Lancet Oncol. 2018, 19, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Sartor, O.; de Bono, J.; Chi, K.N.; Fizazi, K.; Herrmann, K.; Rahbar, K.; Tagawa, S.T.; Nordquist, L.T.; Vaishampayan, N.; El-Haddad, G.; et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. N. Engl. J. Med. 2021, 385, 1091–1103. [Google Scholar] [CrossRef]
- Satapathy, S.; Mittal, B.R.; Sood, A.; Das, C.K.; Mavuduru, R.S.; Goyal, S.; Shukla, J.; Singh, S.K. 177Lu-PSMA-617 versus docetaxel in chemotherapy-naïve metastatic castration-resistant prostate cancer: A randomized, controlled, phase 2 non-inferiority trial. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1754–1764. [Google Scholar] [CrossRef]
- Rinke, A.; Müller, H.-H.; Schade-Brittinger, C.; Klose, K.-J.; Barth, P.; Wied, M.; Mayer, C.; Aminossadati, B.; Pape, U.-F.; Bläker, M.; et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: A report from the PROMID Study Group. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 4656–4663. [Google Scholar] [CrossRef] [PubMed]
- Kvols, L.K.; Moertel, C.G.; O’Connell, M.J.; Schutt, A.J.; Rubin, J.; Hahn, R.G. Treatment of the Malignant Carcinoid Syndrome. N. Engl. J. Med. 1986, 315, 663–666. [Google Scholar] [CrossRef]
- Caplin, M.E.; Pavel, M.; Ćwikła, J.B.; Phan, A.T.; Raderer, M.; Sedláčková, E.; Cadiot, G.; Wolin, E.M.; Capdevila, J.; Wall, L.; et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N. Engl. J. Med. 2014, 371, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.C.; Fazio, N.; Singh, S.; Buzzoni, R.; Carnaghi, C.; Wolin, E.; Tomasek, J.; Raderer, M.; Lahner, H.; Voi, M.; et al. Everolimus for the treatment of advanced, nonfunctional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): A randomised, placebo-controlled, phase 3 study. Lancet Lond. Engl. 2016, 387, 968–977. [Google Scholar] [CrossRef]
- Kulke, M.H.; Siu, L.L.; Tepper, J.E.; Fisher, G.; Jaffe, D.; Haller, D.G.; Ellis, L.M.; Benedetti, J.K.; Bergsland, E.K.; Hobday, T.J.; et al. Future Directions in the Treatment of Neuroendocrine Tumors: Consensus Report of the National Cancer Institute Neuroendocrine Tumor Clinical Trials Planning Meeting. J. Clin. Oncol. 2011, 29, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.R.; Caplin, M.E.; Kunz, P.L.; Ruszniewski, P.B.; Bodei, L.; Hendifar, A.; Mittra, E.; Wolin, E.M.; Yao, J.C.; Pavel, M.E.; et al. 177Lu-Dotatate plus long-acting octreotide versus high-dose long-acting octreotide in patients with midgut neuroendocrine tumours (NETTER-1): Final overall survival and long-term safety results from an open-label, randomised, controlled, phase 3 trial. Lancet Oncol. 2021, 22, 1752–1763. [Google Scholar] [CrossRef] [PubMed]
- EMA Annexe I—Résumé Des Caractéristiques Du Produit—Lutathera 370MBq/mL Solution Pour Perfusion [Internet]. Available online: https://www.ema.europa.eu/en/documents/product-information/lutathera-epar-product-information_fr.pdf (accessed on 11 January 2023).
- Haute Autorité de Santé Avis de la Commission de la Transparence Du 11 Juillet 2018 Concernant le 177Lutécium-Oxodotréotide. Available online: https://www.has-sante.fr/upload/docs/evamed/CT-16606_LUTATHERA_PIC_INS_Avis3_CT16606.pdf (accessed on 27 January 2023).
- Société Savante Des Maladies Et Cancers de L’appareil Digestif Néoplasies Neuroendocrines (NNe) Digestives—Thésaurus National de Cancérologie Digestive. Available online: https://www.snfge.org/sites/default/files/SNFGE/TNCD/tncd_chap-11_nne_2020_03_17.pdf (accessed on 1 February 2023).
- Falconi, M.; Eriksson, B.; Kaltsas, G.; Bartsch, D.K.; Capdevila, J.; Caplin, M.; Kos-Kudla, B.; Kwekkeboom, D.; Rindi, G.; Klöppel, G.; et al. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology 2016, 103, 153–171. [Google Scholar] [CrossRef]
- Pavel, M.; O’’Toole, D.; Costa, F.; Capdevila, J.; Gross, D.; Kianmanesh, R.; Krenning, E.; Knigge, U.; Salazar, R.; Pape, U.-F.; et al. ENETS Consensus Guidelines Update for the Management of Distant Metastatic Disease of Intestinal, Pancreatic, Bronchial Neuroendocrine Neoplasms (NEN) and NEN of Unknown Primary Site. Neuroendocrinology 2016, 103, 172–185. [Google Scholar] [CrossRef]
- Hicks, R.J.; Kwekkeboom, D.J.; Krenning, E.; Bodei, L.; Grozinsky-Glasberg, S.; Arnold, R.; Borbath, I.; Cwikla, J.; Toumpanakis, C.; Kaltsas, G.; et al. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Neoplasms: Peptide Receptor Radionuclide Therapy with Radiolabelled Somatostatin Analogues. Neuroendocrinology 2017, 105, 295–309. [Google Scholar] [CrossRef]
- Kwekkeboom, D.J.; Krenning, E.P.; Lebtahi, R.; Komminoth, P.; Kos-Kudła, B.; de Herder, W.W.; Plöckinger, U. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: Peptide Receptor Radionuclide Therapy with Radiolabeled Somatostatin Analogs. Neuroendocrinology 2009, 90, 220–226. [Google Scholar] [CrossRef]
- Hope, T.A.; Bodei, L.; Chan, J.A.; El-Haddad, G.; Fidelman, N.; Kunz, P.L.; Mailman, J.; Menda, Y.; Metz, D.C.; Mittra, E.S.; et al. NANETS/SNMMI Consensus Statement on Patient Selection and Appropriate Use of 177Lu-DOTATATE Peptide Receptor Radionuclide Therapy. J. Nucl. Med. 2020, 61, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Hendifar, A.E.; Mehr, S.H.; McHaffie, D.R. Best Practices for the Coordinated Care of Patients with Neuroendocrine Tumors Undergoing Peptide Receptor Radionuclide Therapy. Pancreas 2022, 51, 213–218. [Google Scholar] [CrossRef]
- Apostolidis, L.; Dal Buono, A.; Merola, E.; Jann, H.; Jäger, D.; Wiedenmann, B.; Winkler, E.C.; Pavel, M. Multicenter Analysis of Treatment Outcomes for Systemic Therapy in Well Differentiated Grade 3 Neuroendocrine Tumors (NET G3). Cancers 2021, 13, 1936. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, V.; Zanoni, L.; Filice, A.; Lamberti, G.; Argalia, G.; Fortunati, E.; Campana, D.; Versari, A.; Fanti, S. Radiolabeled Somatostatin Analogues for Diagnosis and Treatment of Neuroendocrine Tumors. Cancers 2022, 14, 1055. [Google Scholar] [CrossRef]
- Ezziddin, S.; Attassi, M.; Yong-Hing, C.J.; Ahmadzadehfar, H.; Willinek, W.; Grünwald, F.; Guhlke, S.; Biersack, H.-J.; Sabet, A. Predictors of Long-Term Outcome in Patients with Well-Differentiated Gastroenteropancreatic Neuroendocrine Tumors After Peptide Receptor Radionuclide Therapy with 177Lu-Octreotate. J. Nucl. Med. 2014, 55, 183–190. [Google Scholar] [CrossRef]
- Sansovini, M.; Severi, S.; Ianniello, A.; Nicolini, S.; Fantini, L.; Mezzenga, E.; Ferroni, F.; Scarpi, E.; Monti, M.; Bongiovanni, A.; et al. Long-term follow-up and role of FDG PET in advanced pancreatic neuroendocrine patients treated with 177Lu-D OTATATE. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 490–499. [Google Scholar] [CrossRef]
- Aalbersberg, E.A.; Huizing, D.M.V.; Walraven, I.; de Wit-van der Veen, B.J.; Kulkarni, H.R.; Singh, A.; Stokkel, M.P.M.; Baum, R.P. Parameters to Predict Progression-Free and Overall Survival After Peptide Receptor Radionuclide Therapy: A Multivariate Analysis in 782 Patients. J. Nucl. Med. 2019, 60, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Mittra, E.S. Neuroendocrine Tumor Therapy: 177Lu-DOTATATE. Am. J. Roentgenol. 2018, 211, 278–285. [Google Scholar] [CrossRef]
- Zaknun, J.J.; Bodei, L.; Mueller-Brand, J.; Pavel, M.E.; Baum, R.P.; Hörsch, D.; O’Dorisio, M.S.; O’Dorisiol, T.M.; Howe, J.R.; Cremonesi, M.; et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 800–816. [Google Scholar] [CrossRef] [PubMed]
- Médicaments Radiopharmaceutiques: Du Diagnostic Au Thranostique Des Tumeurs Neuroendocrines. Available online: https://reader.elsevier.com/reader/sd/pii/S092812582100019X?token=5AD3413788D8F96FBA939BD43322BB1B2E50F75604042F460F0D77C4202FEAABA18EDBAA394289780F60C4F92ACD6BC8&originRegion=eu-west-1&originCreation=20230111143928 (accessed on 11 January 2023).
- Endocan—Renaten. GTE—Groupes d’étude des Tumeurs Endocrines. Available online: https://www.reseau-gte.org/renaten/ (accessed on 27 January 2023).
- EMA Annexe I—Résumé Des Caractéristiques Du Produit—Pluvicto 1000Mbq/mL Solution Injectable/Pour Perfusion. Available online: https://www.ema.europa.eu/en/documents/product-information/pluvicto-epar-product-information_fr.pdf (accessed on 11 January 2023).
- Fendler, W.P.; Eiber, M.; Beheshti, M.; Bomanji, J.; Ceci, F.; Cho, S.; Giesel, F.; Haberkorn, U.; Hope, T.A.; Kopka, K.; et al. 68Ga-PSMA PET/CT: Joint EANM and SNMMI procedure guideline for prostate cancer imaging: Version 1.0. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1014–1024. [Google Scholar] [CrossRef] [PubMed]
- Oken, M.M.; Creech, R.H.; Tormey, D.C.; Horton, J.; Davis, T.E.; McFadden, E.T.; Carbone, P.P. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am. J. Clin. Oncol. 1982, 5, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Kratochwil, C.; Fendler, W.P.; Eiber, M.; Baum, R.; Bozkurt, M.F.; Czernin, J.; Delgado Bolton, R.C.; Ezziddin, S.; Forrer, F.; Hicks, R.J.; et al. EANM procedure guidelines for radionuclide therapy with 177Lu-labelled PSMA-ligands (177Lu-PSMA-RLT). Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 2536–2544. [Google Scholar] [CrossRef]
- AlSadi, R.; Bouhali, O.; Dewji, S.; Djekidel, M. 177Lu-PSMA Therapy for Metastatic Castration-Resistant Prostate Cancer: A Mini-Review of State-of-the-Art. The Oncologist 2022, 27, e957–e966. [Google Scholar] [CrossRef] [PubMed]
- Hennrich, U.; Kopka, K. Lutathera®: The First FDA- and EMA-Approved Radiopharmaceutical for Peptide Receptor Radionuclide Therapy. Pharmaceuticals 2019, 12, 114. [Google Scholar] [CrossRef]
- Fallah, J.; Agrawal, S.; Gittleman, H.; Fiero, M.H.; Subramaniam, S.; John, C.; Chen, W.; Ricks, T.K.; Niu, G.; Fotenos, A.; et al. FDA Approval Summary: Lutetium Lu 177 vipivotide tetraxetan for patients with metastatic castration-resistant prostate cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2022, CCR-22-2875. [Google Scholar] [CrossRef]
- Sanli, Y.; Simsek, D.H.; Sanli, O.; Subramaniam, R.M.; Kendi, A.T. 177Lu-PSMA Therapy in Metastatic Castration-Resistant Prostate Cancer. Biomedicines 2021, 9, 430. [Google Scholar] [CrossRef] [PubMed]
- Radecki, J.-J.; Aubert, B.; Cordier, G.; Fracas, P. La mise en œuvre de l’arrêté zonage. Radioprotection 2007, 42, 463–476. [Google Scholar] [CrossRef]
- Bournaud, C.; Lombard-Bohas, C.; Habouzit, V.; Carlier, T.; Hindié, E.; Ansquer, C. La radiothérapie interne vectorisée par les analogues de la somatostatine, en pratique, en 2019. Médecine Nucl. 2019, 43, 251–266. [Google Scholar] [CrossRef]
- Hennrich, U.; Eder, M. [177Lu]Lu-PSMA-617 (PluvictoTM): The First FDA-Approved Radiotherapeutical for Treatment of Prostate Cancer. Pharmaceuticals 2022, 15, 1292. [Google Scholar] [CrossRef]
- Advanced Accelerator Applications Dosing and Administration Guide—Pluvicto®. Available online: https://www.hcp.novartis.com/siteassets/vilupsa/dosing/184981-pluvicto-branded-hcp-dosing-guide-digital_3.29.22.pdf (accessed on 10 January 2023).
- Das, S.; Al-Toubah, T.; El-Haddad, G.; Strosberg, J. 177Lu-DOTATATE for the treatment of gastroenteropancreatic neuroendocrine tumors. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 1023–1031. [Google Scholar] [CrossRef]
- Hope, T.A.; Abbott, A.; Colucci, K.; Bushnell, D.L.; Gardner, L.; Graham, W.S.; Lindsay, S.; Metz, D.C.; Pryma, D.A.; Stabin, M.G.; et al. NANETS/SNMMI Procedure Standard for Somatostatin Receptor–Based Peptide Receptor Radionuclide Therapy with 177 Lu-DOTATATE. J. Nucl. Med. 2019, 60, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Courault, P.; Deville, A.; Habouzit, V.; Gervais, F.; Bolot, C.; Bournaud, C.; Levigoureux, E. Amino Acid Solutions for 177Lu-Oxodotreotide Premedication: A Tolerance Study. Cancers 2022, 14, 5212. [Google Scholar] [CrossRef] [PubMed]
- Love, C.; Desai, N.B.; Abraham, T.; Banks, K.P.; Bodei, L.; Boike, T.; Brown, R.K.J.; Bushnell, D.L.; DeBlanche, L.E.; Dominello, M.M.; et al. ACR-ACNM-ASTRO-SNMMI Practice Parameter for Lutetium-177 (Lu-177) DOTATATE Therapy. Clin. Nucl. Med. 2022, 47, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Advanced Accelerator Applications Monographie de Produit—Lutathera®. Available online: https://canm-acmn.ca/resources/Documents/Lutathera%20Product%20Monograph_FR.pdf (accessed on 31 January 2023).
- Advanced Accelerator Applications. Dosing and Administration—LUTATHERA®. Available online: https://www.hcp.novartis.com/products/lutathera/gep-nets/dosing-and-administration/ (accessed on 1 February 2023).
- Autorité de Sureté Nucléaire ASN. Lettre Circulaire Du 12 Juin 2020—Evolution Des Conditions D’autorisation Des Services de Médecine Nucléaire par L’ASN Pour la Détention Et L’utilisation Du Lutétium-177; Autorité de Sureté Nucléaire ASN: Montrouge, France, 2020. [Google Scholar]
- Herrmann, K.; Giovanella, L.; Santos, A.; Gear, J.; Kiratli, P.O.; Kurth, J.; Denis-Bacelar, A.M.; Hustinx, R.; Patt, M.; Wahl, R.L.; et al. Joint EANM, SNMMI and IAEA enabling guide: How to set up a theranostics centre. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 2300–2309. [Google Scholar] [CrossRef] [PubMed]
- Hosono, M.; Ikebuchi, H.; Nakamura, Y.; Nakamura, N.; Yamada, T.; Yanagida, S.; Kitaoka, A.; Kojima, K.; Sugano, H.; Kinuya, S.; et al. Manual on the proper use of lutetium-177-labeled somatostatin analogue (Lu-177-DOTA-TATE) injectable in radionuclide therapy (2nd ed.). Ann. Nucl. Med. 2018, 32, 217–235. [Google Scholar] [CrossRef] [PubMed]
- Kurth, J. External radiation exposure, excretion, and effective half-life in 177Lu-PSMA-targeted therapies. EJNMMI Res. 2018, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Institut de Radioprotection Et de Sûreté Nucléaire IRSN—Rapport D’expertise n°2021-00484—Nouveaux Radionucléides en Médecine Nucléaire. Available online: https://www.irsn.fr/FR/expertise/rapports_expertise/Documents/radioprotection/IRSN_Rapport-2021-00484-nouveaux-radionucleides-medecine.pdf (accessed on 11 January 2023).
- Valentin, J.; International Commission on Radiological Protection. Radiological Protection in Medicine; ICRP Publication; Elsevier: Oxford, UK, 2008; 63p, ISBN 978-0-7020-3102-1. [Google Scholar]
- Autorité de Sûreté Nucléaire Décision n°2021-DC-0708 de L’ASN Du 6 Avril 2021- Obligations D’assurance de la Qualité Pour Les Actes utilisant Des Rayonnements Ionisants Réalisés à Des Fins de Prise en Charge Thérapeutique; Autorité de Sureté Nucléaire ASN: Montrouge, France, 2021.
- Van der Pol, J.; Vöö, S.; Bucerius, J.; Mottaghy, F.M. Consequences of radiopharmaceutical extravasation and therapeutic interventions: A systematic review. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1234–1243. [Google Scholar] [CrossRef] [PubMed]
- Mazzara, C.; Salvadori, J.; Ritzenthaler, F.; Martin, S.; Porot, C.; Imperiale, A. 177Lu-DOTA-0-Tyr3-octreotate infusion modeling for real-time detection and characterization of extravasation during PRRT. EJNMMI Phys. 2022, 9, 33. [Google Scholar] [CrossRef]
- Tylski, P.; Pina-Jomir, G.; Bournaud-Salinas, C.; Jalade, P. Tissue dose estimation after extravasation of 177Lu-DOTATATE. EJNMMI Phys. 2021, 8, 33. [Google Scholar] [CrossRef] [PubMed]
- Arveschoug, A.K.; Bekker, A.C.; Iversen, P.; Bluhme, H.; Villadsen, G.E.; Staanum, P.F. Extravasation of [177Lu]Lu-DOTATOC: Case report and discussion. EJNMMI Res. 2020, 10, 68. [Google Scholar] [CrossRef]
- Sakulpisuti, C.; Chamroonrat, W.; Tepmongkol, S. Cutaneous Management after Extravasation of High-Concentrated Amino Acid Solution Administered for Renal Protection in PRRT. Tomography 2022, 8, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Divband, G.; Alavi, S.H.; Adinehpour, Z.; Kalantari, F.; Zarehparvar Moghadam, S. Amino acid extravasation: A rare red flag to keep in mind during peptide receptor radioligand therapy (PRRT) with [177Lu]Lu-DOTATATE. Nucl. Med. Rev. Cent. East. Eur. 2022, 25, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Rolleman, E.; Valkema, R.; de Jong, M.; Kooij, P.; Krenning, E. Safe and effective inhibition of renal uptake of radiolabelled octreotide by a combination of lysine and arginine. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 9–15. [Google Scholar] [CrossRef]
- De Keizer, B.; van Aken, M.O.; Feelders, R.A.; de Herder, W.W.; Kam, B.L.R.; van Essen, M.; Krenning, E.P.; Kwekkeboom, D.J. Hormonal crises following receptor radionuclide therapy with the radiolabeled somatostatin analogue [177Lu-DOTA0,Tyr3]octreotate. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 749–755. [Google Scholar] [CrossRef] [PubMed]
- Van der Zwan, W.A.; Brabander, T.; Kam, B.L.R.; Teunissen, J.J.M.; Feelders, R.A.; Hofland, J.; Krenning, E.P.; de Herder, W.W. Salvage peptide receptor radionuclide therapy with [177Lu-DOTA,Tyr3]octreotate in patients with bronchial and gastroenteropancreatic neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 704–717. [Google Scholar] [CrossRef] [PubMed]
- Valkema, R.; Pauwels, S.A.; Kvols, L.K.; Kwekkeboom, D.J.; Jamar, F.; de Jong, M.; Barone, R.; Walrand, S.; Kooij, P.P.M.; Bakker, W.H.; et al. Long-term follow-up of renal function after peptide receptor radiation therapy with (90)Y-DOTA(0),Tyr(3)-octreotide and (177)Lu-DOTA(0), Tyr(3)-octreotate. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2005, 46 (Suppl. 1), 83S–91S. [Google Scholar]
- Goncalves, I.; Burbury, K.; Michael, M.; Iravani, A.; Ravi Kumar, A.S.; Akhurst, T.; Tiong, I.S.; Blombery, P.; Hofman, M.S.; Westerman, D.; et al. Characteristics and outcomes of therapy-related myeloid neoplasms after peptide receptor radionuclide/chemoradionuclide therapy (PRRT/PRCRT) for metastatic neuroendocrine neoplasia: A single-institution series. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Vigne, J.; Chrétien, B.; Bignon, A.-L.; Bouhier-Leporrier, K.; Dolladille, C. [177Lu]Lu-DOTATATE peptide receptor radionuclide therapy–associated myeloid neoplasms: Insights from the WHO pharmacovigilance database. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3332–3333. [Google Scholar] [CrossRef]
- Hofman, M.S.; Emmett, L.; Sandhu, S.; Iravani, A.; Joshua, A.M.; Goh, J.C.; Pattison, D.A.; Tan, T.H.; Kirkwood, I.D.; Ng, S.; et al. [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): A randomised, open-label, phase 2 trial. Lancet Lond. Engl. 2021, 397, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Violet, J.; Jackson, P.; Ferdinandus, J.; Sandhu, S.; Akhurst, T.; Iravani, A.; Kong, G.; Kumar, A.R.; Thang, S.P.; Eu, P.; et al. Dosimetry of 177Lu-PSMA-617 in Metastatic Castration-Resistant Prostate Cancer: Correlations Between Pretherapeutic Imaging and Whole-Body Tumor Dosimetry with Treatment Outcomes. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2019, 60, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Ling, S.W.; de Blois, E.; Hooijman, E.; van der Veldt, A.; Brabander, T. Advances in 177Lu-PSMA and 225Ac-PSMA Radionuclide Therapy for Metastatic Castration-Resistant Prostate Cancer. Pharmaceutics 2022, 14, 2166. [Google Scholar] [CrossRef] [PubMed]
- Klein Nulent, T.J.W.; Valstar, M.H.; de Keizer, B.; Willems, S.M.; Smit, L.A.; Al-Mamgani, A.; Smeele, L.E.; van Es, R.J.J.; de Bree, R.; Vogel, W.V. Physiologic distribution of PSMA-ligand in salivary glands and seromucous glands of the head and neck on PET/CT. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2018, 125, 478–486. [Google Scholar] [CrossRef]
- Taïeb, D.; Foletti, J.-M.; Bardiès, M.; Rocchi, P.; Hicks, R.J.; Haberkorn, U. PSMA-Targeted Radionuclide Therapy and Salivary Gland Toxicity: Why Does It Matter? J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2018, 59, 747–748. [Google Scholar] [CrossRef]
- Lucaroni, L.; Georgiev, T.; Prodi, E.; Puglioli, S.; Pellegrino, C.; Favalli, N.; Prati, L.; Manz, M.G.; Cazzamalli, S.; Neri, D.; et al. Cross-reactivity to glutamate carboxypeptidase III causes undesired salivary gland and kidney uptake of PSMA-targeted small-molecule radionuclide therapeutics. Eur. J. Nucl. Med. Mol. Imaging 2022, 50, 957–961. [Google Scholar] [CrossRef]
- Silver, D.A.; Pellicer, I.; Fair, W.R.; Heston, W.D.; Cordon-Cardo, C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 1997, 3, 81–85. [Google Scholar]
- Rosar, F.; Kochems, N.; Bartholomä, M.; Maus, S.; Stemler, T.; Linxweiler, J.; Khreish, F.; Ezziddin, S. Renal Safety of [177Lu]Lu-PSMA-617 Radioligand Therapy in Patients with Compromised Baseline Kidney Function. Cancers 2021, 13, 3095. [Google Scholar] [CrossRef]
- Zhang, J.; Kulkarni, H.R.; Singh, A.; Schuchardt, C.; Niepsch, K.; Langbein, T.; Baum, R.P. Lu-PSMA-617 Radioligand Therapy in Metastatic Castration-Resistant Prostate Cancer Patients with a Single Functioning Kidney. J. Nucl. Med. 2019, 60, 1579–1586. [Google Scholar] [CrossRef]
- Groener, D.; Nguyen, C.T.; Baumgarten, J.; Bockisch, B.; Davis, K.; Happel, C.; Mader, N.; Nguyen Ngoc, C.; Wichert, J.; Banek, S.; et al. Hematologic safety of 177Lu-PSMA-617 radioligand therapy in patients with metastatic castration-resistant prostate cancer. EJNMMI Res. 2021, 11, 61. [Google Scholar] [CrossRef] [PubMed]
- Sjögreen Gleisner, K.; Chouin, N.; Gabina, P.M.; Cicone, F.; Gnesin, S.; Stokke, C.; Konijnenberg, M.; Cremonesi, M.; Verburg, F.A.; Bernhardt, P.; et al. EANM dosimetry committee recommendations for dosimetry of 177Lu-labelled somatostatin-receptor- and PSMA-targeting ligands. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1778–1809. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.A.; Beauregard, J.-M.; Hofman, M.S.; Kron, T.; Hogg, A.; Hicks, R.J. An automated voxelized dosimetry tool for radionuclide therapy based on serial quantitative SPECT/CT imaging. Med. Phys. 2013, 40, 112503. [Google Scholar] [CrossRef] [PubMed]
- Huizing, D.M.V.; de Wit-van der Veen, B.J.; Verheij, M.; Stokkel, M.P.M. Dosimetry methods and clinical applications in peptide receptor radionuclide therapy for neuroendocrine tumours: A literature review. EJNMMI Res. 2018, 8, 89. [Google Scholar] [CrossRef] [PubMed]
- Heikkonen, J.; Mäenpää, H.; Hippeläinen, E.; Reijonen, V.; Tenhunen, M. Effect of calculation method on kidney dosimetry in 177Lu-octreotate treatment. Acta Oncol. Stockh. Swed. 2016, 55, 1069–1076. [Google Scholar] [CrossRef]
- Sundlöv, A.; Sjögreen-Gleisner, K.; Svensson, J.; Ljungberg, M.; Olsson, T.; Bernhardt, P.; Tennvall, J. Individualised 177Lu-DOTATATE treatment of neuroendocrine tumours based on kidney dosimetry. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1480–1489. [Google Scholar] [CrossRef]
- Sandström, M.; Garske, U.; Granberg, D.; Sundin, A.; Lundqvist, H. Individualized dosimetry in patients undergoing therapy with (177)Lu-DOTA-D-Phe (1)-Tyr (3)-octreotate. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 212–225. [Google Scholar] [CrossRef]
- Sandström, M.; Garske-Román, U.; Granberg, D.; Johansson, S.; Widström, C.; Eriksson, B.; Sundin, A.; Lundqvist, H.; Lubberink, M. Individualized dosimetry of kidney and bone marrow in patients undergoing 177Lu-DOTA-octreotate treatment. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2013, 54, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Thieme, A.; Allmann, J.; D’Alessandria, C.; Maurer, T.; Retz, M.; Tauber, R.; Heck, M.M.; Wester, H.-J.; Tamaki, N.; et al. Radiation Dosimetry for 177Lu-PSMA I&T in Metastatic Castration-Resistant Prostate Cancer: Absorbed Dose in Normal Organs and Tumor Lesions. J. Nucl. Med. 2017, 58, 445–450. [Google Scholar] [CrossRef]
- Goetz, T.I.; Lang, E.W.; Prante, O.; Maier, A.; Cordes, M.; Kuwert, T.; Ritt, P.; Schmidkonz, C. Three-dimensional Monte Carlo-based voxel-wise tumor dosimetry in patients with neuroendocrine tumors who underwent 177Lu-DOTATOC therapy. Ann. Nucl. Med. 2020, 34, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Bolch, W.E.; Eckerman, K.F.; Sgouros, G.; Thomas, S.R. MIRD pamphlet No. 21: A generalized schema for radiopharmaceutical dosimetry--standardization of nomenclature. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2009, 50, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Dewaraja, Y.K.; Frey, E.C.; Sgouros, G.; Brill, A.B.; Roberson, P.; Zanzonico, P.B.; Ljungberg, M. MIRD pamphlet No. 23: Quantitative SPECT for patient-specific 3-dimensional dosimetry in internal radionuclide therapy. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2012, 53, 1310–1325. [Google Scholar] [CrossRef] [PubMed]
- Ljungberg, M.; Celler, A.; Konijnenberg, M.W.; Eckerman, K.F.; Dewaraja, Y.K.; Sjögreen-Gleisner, K.; SNMMI MIRD Committee; Bolch, W.E.; Brill, A.B.; Fahey, F.; et al. MIRD Pamphlet No. 26: Joint EANM/MIRD Guidelines for Quantitative 177Lu SPECT Applied for Dosimetry of Radiopharmaceutical Therapy. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2016, 57, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Uribe, C.; Peterson, A.; Van, B.; Fedrigo, R.; Carlson, J.; Sunderland, J.; Frey, E.; Dewaraja, Y.K. An International Study of Factors Affecting Variability of Dosimetry Calculations, Part 1: Design and Early Results of the SNMMI Dosimetry Challenge. J. Nucl. Med. Off. Publ. Soc. Nucl. Med. 2021, 62, 36S–47S. [Google Scholar] [CrossRef]
- Mora-Ramirez, E.; Santoro, L.; Cassol, E.; Ocampo-Ramos, J.C.; Clayton, N.; Kayal, G.; Chouaf, S.; Trauchessec, D.; Pouget, J.-P.; Kotzki, P.-O.; et al. Comparison of commercial dosimetric software platforms in patients treated with 177 Lu-DOTATATE for peptide receptor radionuclide therapy. Med. Phys. 2020, 47, 4602–4615. [Google Scholar] [CrossRef]
- Rosar, F.; Schön, N.; Bohnenberger, H.; Bartholomä, M.; Stemler, T.; Maus, S.; Khreish, F.; Ezziddin, S.; Schaefer-Schuler, A. Comparison of different methods for post-therapeutic dosimetry in [177Lu]Lu-PSMA-617 radioligand therapy. EJNMMI Phys. 2021, 8, 40. [Google Scholar] [CrossRef]
| Toxicity | Action | Result | Next Step |
|---|---|---|---|
| Grade 2 toxicity platelet count 40% increase in serum creatinine with a 40% decrease in clearance | Postpone treatment up to 16 weeks interval and monitor every 2 weeks | If resolution: re-treat with decrease of 50% radioactivity dose | No toxicity: full dose |
| If persisting toxicity: stop treatment | Toxicity: stop treatment | ||
| Any Grade 3 (except pre-existing serum liver enzyme abnormalities) | Postpone treatment up to 16 weeks interval and monitor every 2 weeks | If resolution: re-treat with decrease of 50% radioactivity dose | No toxicity: full dose |
| If persisting toxicity: stop treatment | Toxicity: stop treatment | ||
| Any Grade 4 | Postpone treatment up to 16 weeks interval and monitor frequently. Act as necessary | If resolution: re-treat with decrease of 50% radioactivity dose | No toxicity: full dose |
| If persisting toxicity: stop treatment | Toxicity: stop treatment |
| Adverse events | Severity | Action |
|---|---|---|
| Myelosuppression | Grade 2 | Withhold Pluvicto® until improvement to Grade 1 or the baseline. |
| Grade ≥3 | Withhold Pluvicto® until improvement to Grade 1 or the baseline and reduce the Pluvicto® dose by 20%to 5.9 GBq (160 mCi). | |
| Recurrent Grade ≥3 after one dose reduction | Permanently discontinue Pluvicto®. | |
| Renal toxicity | Confirmed serum creatinine increase (Grade ≥2) confirmed CLcr <30 mL/min | Withhold Pluvicto® until improvement. |
| Confirmed ≥40% increase from baseline serum creatinine confirmed >40% decrease from baseline CLcr | Withhold Pluvicto® until improvement or a return to baseline. Reduce Pluvicto® dose by 20% to 5.9 GBq (160 mCi). | |
| Grade ≥3 renal toxicity and recurrent renal toxicity after one dose reduction | Permanently discontinue Pluvicto®. | |
| Gastrointestinal toxicity | Grade ≥3 | Withhold Pluvicto® until improvement to Grade 2 or the baseline. Reduce the Pluvicto® dose by 20% to 5.9 GBq (160 mCi). |
| Recurrent Grade ≥3 gastrointestinal toxicity after one dose reduction | Permanently discontinue Pluvicto®. | |
| Dry mouth | Grade 2 | Withhold Pluvicto® until improvement or a return to the baseline. Consider reducing the Pluvicto® dose by 20% to 5.9 GBq (160 mCi). |
| Grade 3 | Withhold Pluvicto® until improvement or a return to the baseline. Reduce the Pluvicto® dose by 20% to 5.9 GBq (160 mCi). | |
| Recurrent Grade 3 dry mouth after one dose reduction | Permanently discontinue Pluvicto®. | |
| Fatigue | Grade ≥3 | Withhold Pluvicto® until improvement to Grade 2 or the baseline. |
| [177Lu]Lu-DOTATATE (Lutathera®) | [177Lu]Lu-vipivotide tetraxetan (Pluvicto®) |
|---|---|
| |
| |
| |
| |
|
|
| |
|
|
| |
| |
| |
| |
| |
|
|
| |
| |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladrière, T.; Faudemer, J.; Levigoureux, E.; Peyronnet, D.; Desmonts, C.; Vigne, J. Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals. Pharmaceutics 2023, 15, 1240. https://doi.org/10.3390/pharmaceutics15041240
Ladrière T, Faudemer J, Levigoureux E, Peyronnet D, Desmonts C, Vigne J. Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals. Pharmaceutics. 2023; 15(4):1240. https://doi.org/10.3390/pharmaceutics15041240
Chicago/Turabian StyleLadrière, Typhanie, Julie Faudemer, Elise Levigoureux, Damien Peyronnet, Cédric Desmonts, and Jonathan Vigne. 2023. "Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals" Pharmaceutics 15, no. 4: 1240. https://doi.org/10.3390/pharmaceutics15041240
APA StyleLadrière, T., Faudemer, J., Levigoureux, E., Peyronnet, D., Desmonts, C., & Vigne, J. (2023). Safety and Therapeutic Optimization of Lutetium-177 Based Radiopharmaceuticals. Pharmaceutics, 15(4), 1240. https://doi.org/10.3390/pharmaceutics15041240








