Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice
Abstract
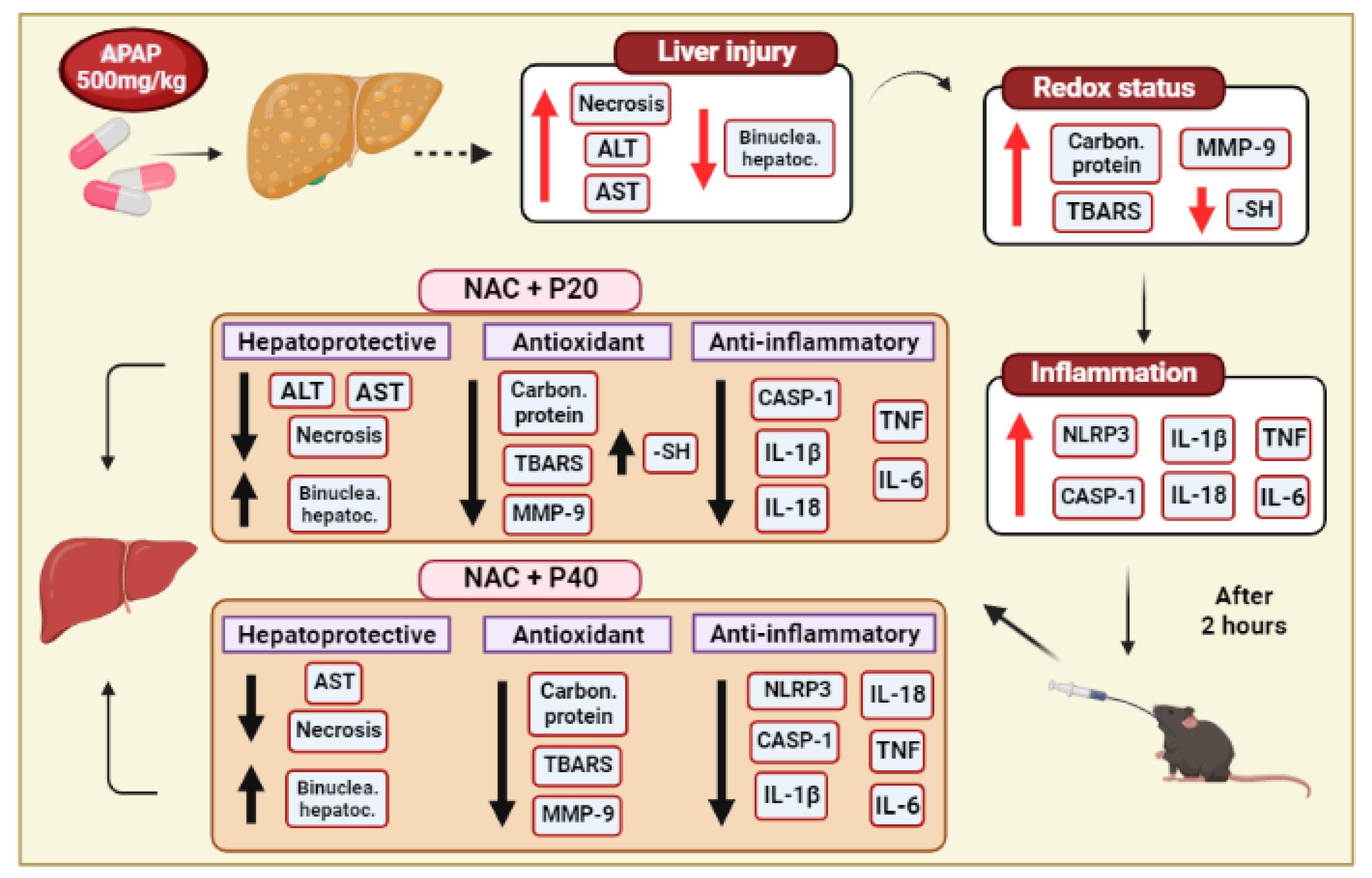
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Design
2.3. Liver Function
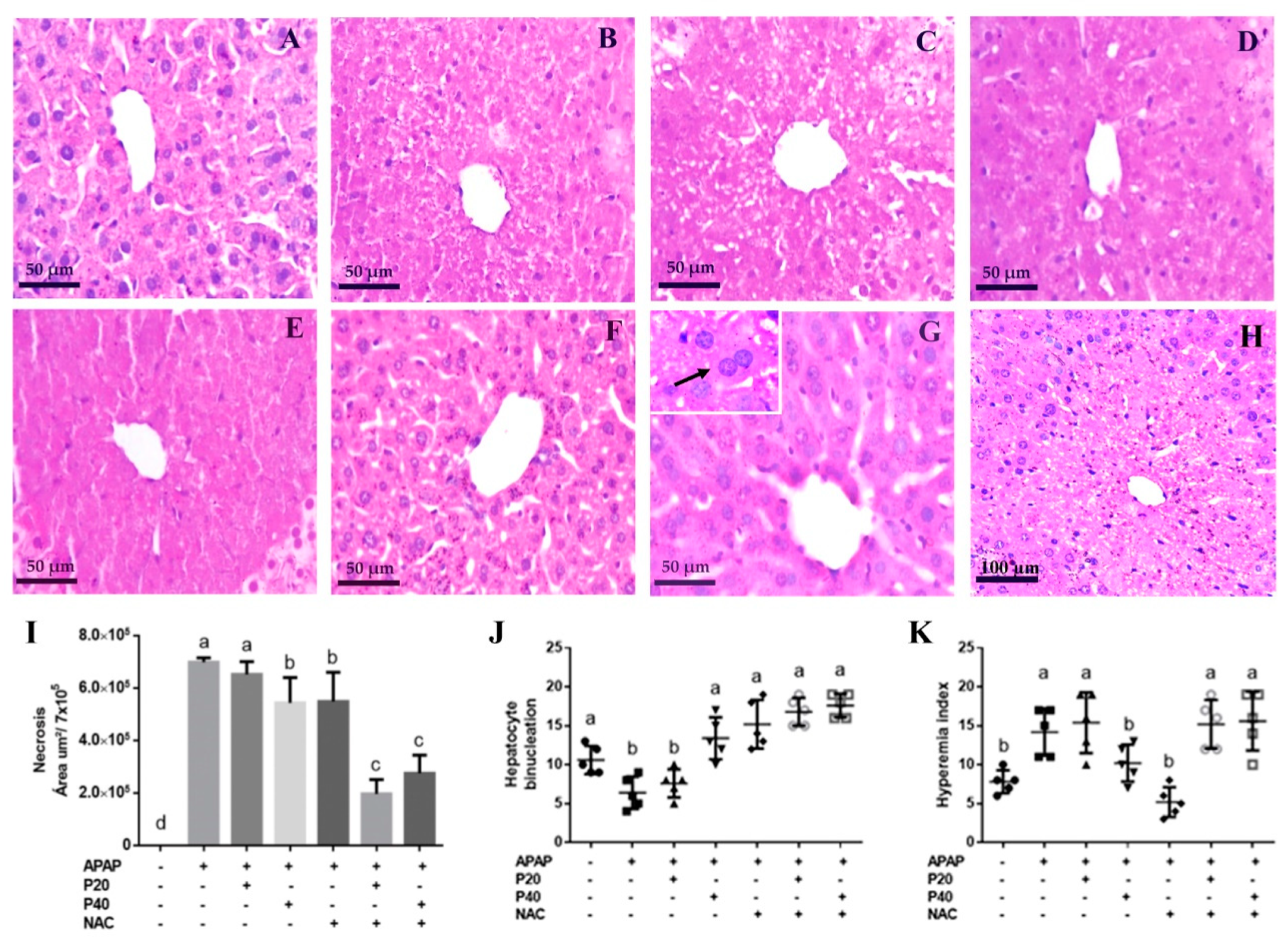
2.4. Liver Histological Analysis
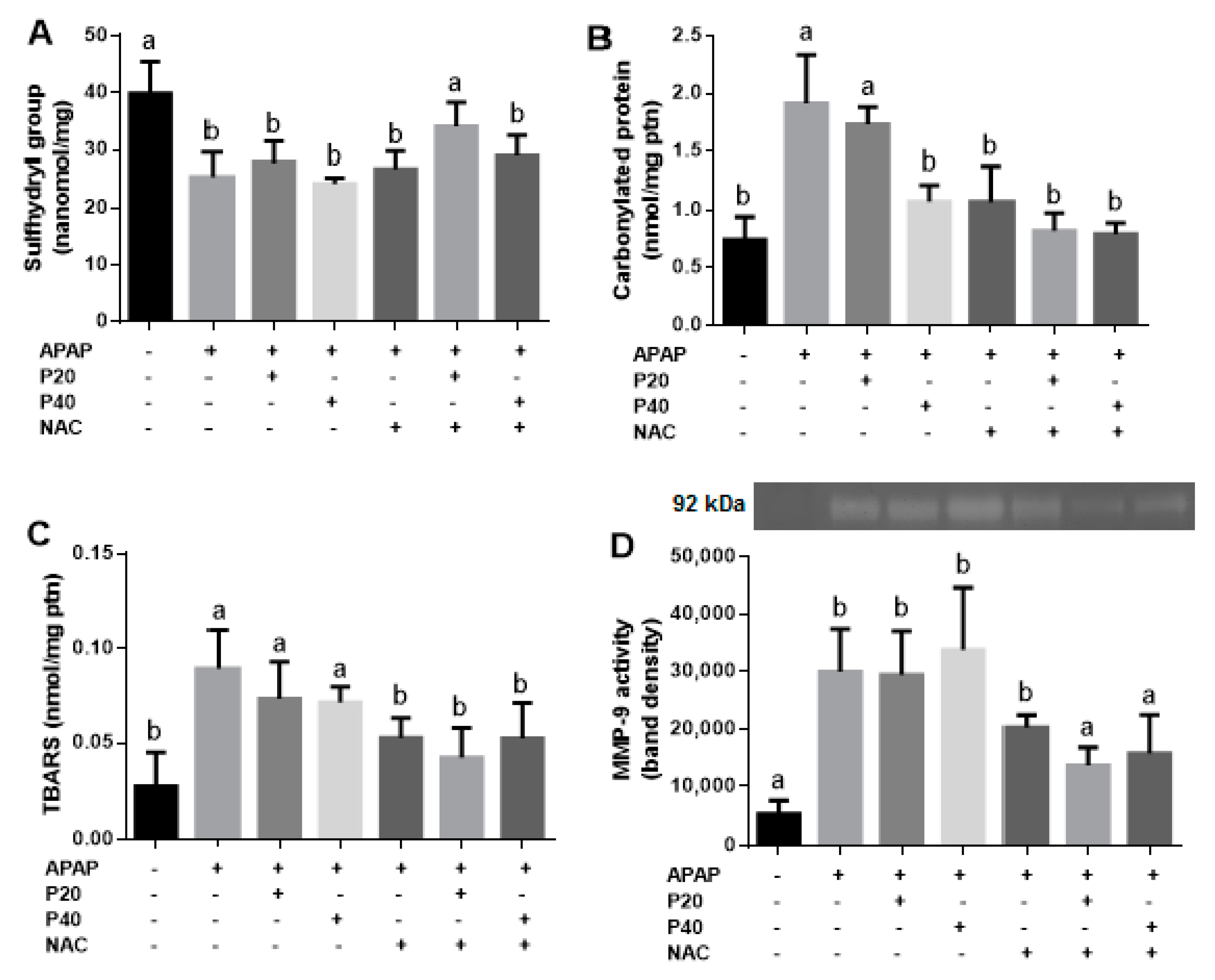
2.5. Biomarkers of Damage Caused by Lipid and Protein Oxidation
2.6. Analysis of Antioxidant Defenses
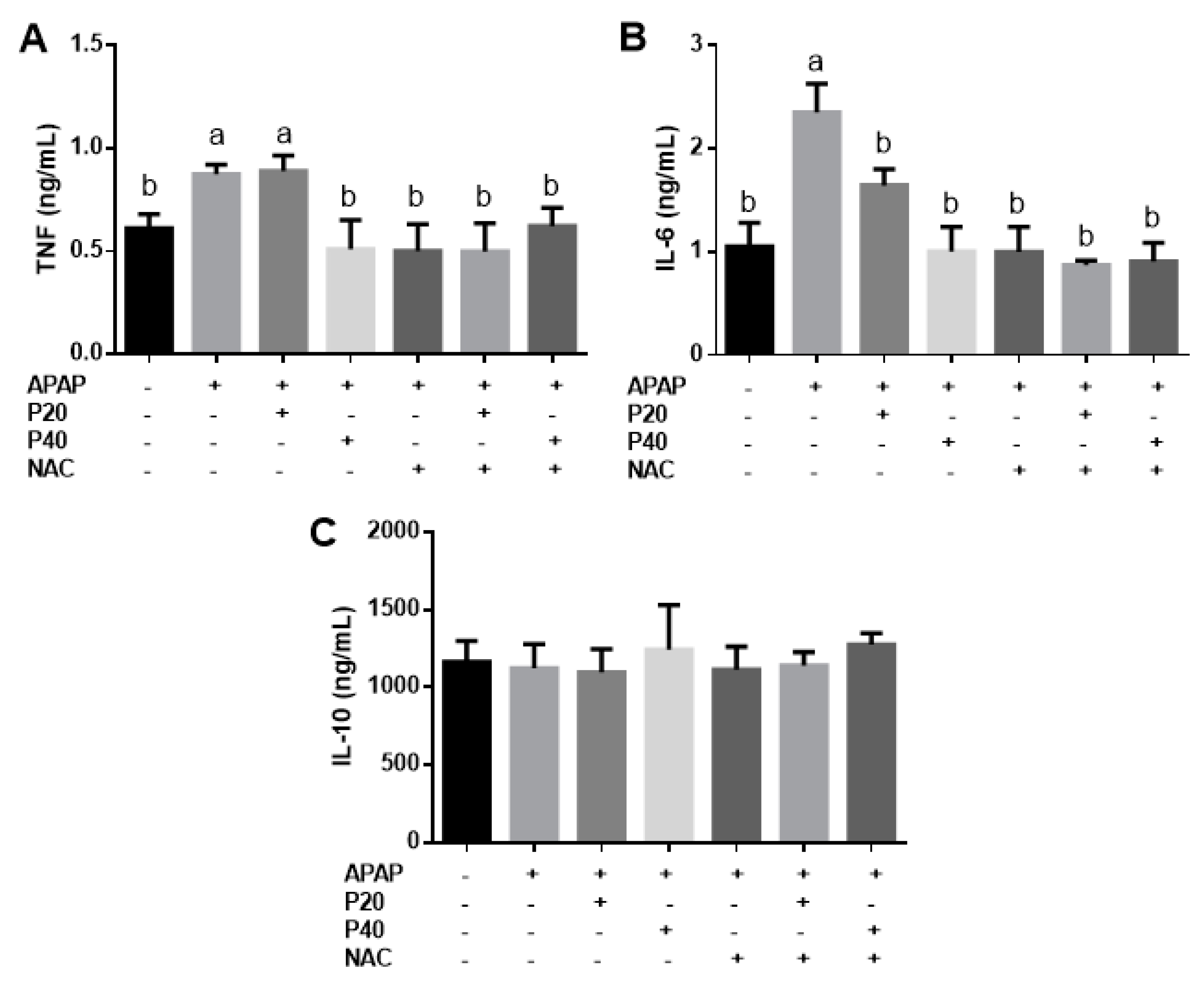
2.7. Cytokine Analysis
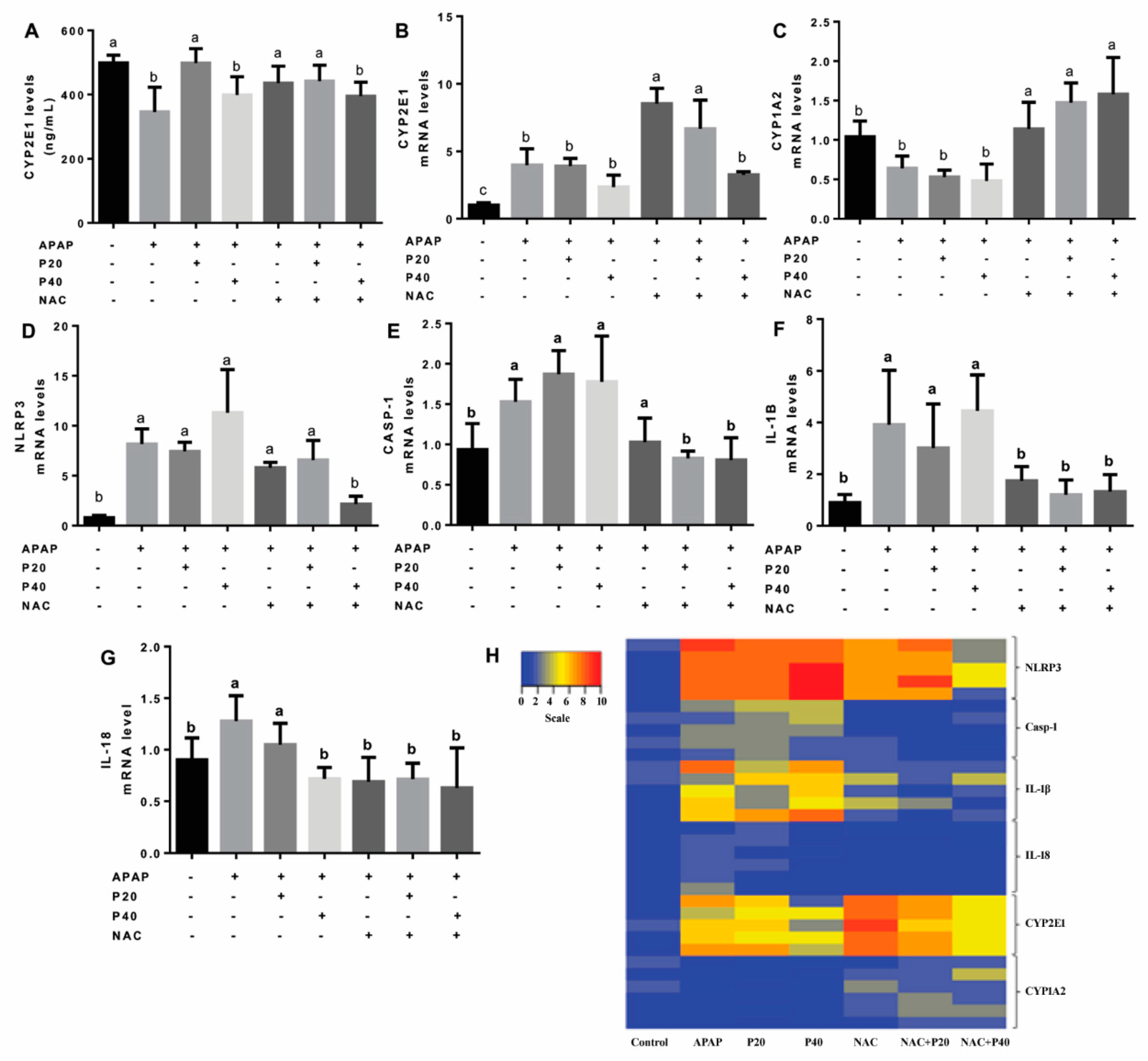
2.8. Cytochrome P450 2E1 (CYP2E1) Assay
2.9. Real-Time Reverse Transcription PCR (qRT-PCR)
2.10. Zymography
2.11. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thusius, N.J.; Romanowicz, M.; Bostwick, J.M. Intentional or Inadvertent Acetaminophen Overdose—How Lethal It Really Is? Psychosomatics 2019, 60, 574–581. [Google Scholar] [CrossRef]
- Klein, T.E.; Alvarellos, M.L.; McDonagh, E.M.; Patel, S.; McLeod, H.L.; Altman, R.B. Pathways of Acetaminophen Metabolism at the Therapeutic versus Toxic Doses. Pharmacogenet. Genom. 2015, 25, 622–630. [Google Scholar] [CrossRef]
- Xu, S.; Liu, J.; Shi, J.; Wang, Z.; Ji, L. 2,3,4′,5-Tetrahydroxystilbene-2-O-β-D-Glucoside Exacerbates Acetaminophen-Induced Hepatotoxicity by Inducing Hepatic Expression of CYP2E1, CYP3A4 and CYP1A2. Sci. Rep. 2017, 7, 16511. [Google Scholar] [CrossRef] [PubMed]
- Mazraati, P.; Minaiyan, M. Hepatoprotective Effect of Metadoxine on Acetaminophen-induced Liver Toxicity in Mice. Adv. Biomed. Res. 2018, 7, 67. [Google Scholar] [CrossRef]
- Salem, G.A.; Shaban, A.; Diab, H.A.; Elsaghayer, W.A.; Mjedib, M.D.; Hnesh, A.M.; Sahu, R.P. Phoenix Dactylifera Protects against Oxidative Stress and Hepatic Injury Induced by Paracetamol Intoxication in Rats. Biomed. Pharmacother. 2018, 104, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Woolbright, B.L.; Jaeschke, H. Xenobiotic and Endobiotic Mediated Interactions between the Cytochrome P450 System and the Inflammatory Response In the Liver. Adv. Pharmacol. 2015, 74, 131–161. [Google Scholar] [CrossRef]
- Lauterburg, B.H.; Corcoran, G.B.; Mitchell, J.R. Mechanism of Action of N-Acetylcysteine in the Protection Against the Hepatotoxicity of Acetaminophen in Rats In Vivo. J. Clin. Investig. 1983, 71, 980–991. [Google Scholar] [CrossRef]
- More, S.S.; Nugent, J.; Vartak, A.P.; Nye, S.M.; Vince, R. Hepatoprotective Effect of ψ-Glutathione in a Murine Model of Acetaminophen-Induced Liver Toxicity. Chem. Res. Toxicol. 2017, 30, 777–784. [Google Scholar] [CrossRef]
- Rumack, B.H.; Bateman, D.N. Acetaminophen and Acetylcysteine Dose and Duration: Past, Present and Future. Clin. Toxicol. 2012, 50, 91–98. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, H.Y.; Back, S.Y.; Han, H. Piperine-Mediated Drug Interactions and Formulation Strategy for Piperine: Recent Advances and Future Perspectives. Expert Opin. Drug Metab. Toxicol. 2018, 14, 43–57. [Google Scholar] [CrossRef]
- Yang, W.; Chen, Y.H.; Liu, H.; Qu, H.D. Neuroprotective Effects of Piperine on the 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine-Induced Parkinson’s Disease Mouse Model. Int. J. Mol. Med. 2015, 36, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Praneetha, P.; Patil, A. Characterization of Stable and Reactive Metabolites of Piperine Formed on Incubation with Human Liver Microsomes. Mass Spectrom. 2019, 54, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P.; Sahebkar, A. Piperine and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016, 928, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Rinwa, P.; Kumar, A. Piperine Potentiates the Protective Effects of Curcumin against Chronic Unpredictable Stress-Induced Cognitive Impairment and Oxidative Damage in Mice. Brain Res. 2012, 1488, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Sethiya, N.K.; Shah, P.; Rajpara, A.; Nagar, P.A.; Mishra, S.H. Antioxidant and Hepatoprotective Effects of Mixed Micellar Lipid Formulation of Phyllanthin and Piperine in Carbon Tetrachloride-Induced Liver Injury in Rodents. Food Funct. 2015, 6, 3593–3603. [Google Scholar] [CrossRef]
- Rinwa, P.; Kumar, A. Quercetin along with Piperine Prevents Cognitive Dysfunction, Oxidative Stress and Neuro-Inflammation Associated with Mouse Model of Chronic Unpredictable Stress. Arch. Pharm. Res. 2013, 40, 1166–1175. [Google Scholar] [CrossRef]
- Wightman, E.L.; Reay, J.L.; Haskell, C.F.; Williamson, G.; Dew, T.P.; Kennedy, D.O. Effects of Resveratrol Alone or in Combination with Piperine on Cerebral Blood Flow Parameters and Cognitive Performance in Human Subjects: A Randomised, Double-Blind, Placebo-Controlled, Cross-over Investigation. Br. J. Nutr. 2014, 112, 203–213. [Google Scholar] [CrossRef]
- Shamsi, S.; Tran, H.; Tan, R.S.J.; Tan, Z.J.; Lim, L.Y. Curcumin, Piperine, and Capsaicin: A Comparative Study of Spice-Mediated Inhibition of Human Cytochrome P450 Isozyme Activities. Drug Metab. Dispos. 2017, 45, 49–55. [Google Scholar] [CrossRef]
- Kangt, M.H.; Wont, S.M.; Parks, S.S.; Kim, S.G.; Novak, R.F.; Kim, N.D. Piperine Effects on the Expression of P4502E1, P4502B and P4501A in Rat. Xenobiotica 1994, 24, 1195–1204. [Google Scholar] [CrossRef]
- Bang, J.S.; Oh, D.H.; Choi, H.M.; Sur, B.; Lim, S.; Kim, J.Y.; Yang, H.; Yoo, M.C.; Hahm, D.; Kim, K.S. Anti-Inflammatory and Antiarthritic Effects of Piperine in Human Interleukin 1 β -Stimulated Fibroblast-like Synoviocytes and in Rat Arthritis Models. Arthritis Res. Ther. 2009, 11, R49. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Singhal, V.; Roshan, R.; Sharma, A.; Rembhotkar, G.W.; Ghosh, B. Piperine Inhibits TNF-α Induced Adhesion of Neutrophils to Endothelial Monolayer through Suppression of NF-κB and IκB Kinase Activation. Eur. J. Pharmacol. 2007, 575, 177–186. [Google Scholar] [CrossRef]
- Singh, A.; Rao, A.R. Evaluation of the Modulatory Influence of Black Pepper (Piper nigrum, L.) on the Hepatic Detoxication System. Cancer Lett. 1993, 72, 5–9. [Google Scholar] [CrossRef]
- Jaeschke, H.; McGill, M.R.; David Williams, A.; Ramachandran, A. Current Issues with Acetaminophen Hepatotoxicity—A Clinically Relevant Model to Test the Efficacy of Natural Products. Life Sci. 2011, 88, 737–745. [Google Scholar] [CrossRef]
- Singh, A.; Bhat, T.K.; Sharma, O.P. Clinical Biochemistry of Hepatotoxicity. J. Clin. Toxicol. 2011, 4, 2161-0495. [Google Scholar] [CrossRef]
- Bandeira, A.C.B.; Silva, R.C.; da Júnior, J.V.R.; Figueiredo, V.P.; Talvani, A.; Cangussú, S.D.; Bezerra, F.S.; Costa, D.C. Lycopene Pretreatment Improves Hepatotoxicity Induced by Acetaminophen in C57BL/6 Mice. Bioorg. Med. Chem. 2017, 25, 1057–1065. [Google Scholar] [CrossRef]
- Bandeira, A.C.B.; da Silva, T.P.; de Araujo, G.R.; Araujo, C.M.; da Silva, R.C.; Lima, W.G.; Bezerra, F.S.; Costa, D.C. Lycopene Inhibits Reactive Oxygen Species Production in SK-Hep-1 Cells and Attenuates Acetaminophen-Induced Liver Injury in C57BL/6 Mice. Chem. Biol. Interact. 2017, 263, 7–17. [Google Scholar] [CrossRef]
- Coelho, A.M.; Queiroz, I.F.; Lima, W.G.; Talvani, A.; Perucci, L.O.; Souza, M.O. De Temporal Analysis of Paracetamol-Induced Hepatotoxicity. Drug Chem. Toxicol. 2022, 45, 1–10. [Google Scholar] [CrossRef]
- Draper, H.; Squires, E.; Mahmoodi, H.; Wu, J.; Agarwal, S.; Hadley, M. A Comparative Evaluation of Thiobarbituric Acid Methods for the Determination of Malondialdehyde in Biological Materials. Free Radic. Biol. Med. 1993, 15, 353–363. [Google Scholar] [CrossRef]
- Levine, R.L.; Williams, J.A.; Earl, R.S.; Emily, S. Carbonyl Assays for Determination of Oxidatively Modified Proteins. Methods Enzymol. 1991, 186, 346–357. [Google Scholar]
- Sedlak, J.; Lindsay, R.H. Estimation of Total, Protein-Bound, and Nonprotein Sulfhydryl Groups in Tissue with Ellman’s Reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Sung, M.M.; Schulz, C.G.; Wang, W.; Sawicki, G.; Bautista-lópez, N.L.; Schulz, R. Matrix Metalloproteinase-2 Degrades the Cytoskeletal Protein α-Actinin in Peroxynitrite Mediated Myocardial Injury. J. Mol. Cell. Cardiol. 2007, 43, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Rabelo, A.C.S.; de Pádua Lúcio, K.; Araújo, C.M.; de Araújo, G.R.; de Amorim Miranda, P.H.; Carneiro, A.C.A.; de Castro Ribeiro, É.M.; de Melo Silva, B.; de Lima, W.G.; Costa, D.C. Baccharis Trimera Protects against Ethanol Induced Hepatotoxicity in Vitro and in Vivo. J. Ethnopharmacol. 2018, 215, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Martínez, M. Paracetamol: Overdose-Induced Oxidative Stress Toxicity, Metabolism, and Protective Effects of Various Compounds in Vivo and in Vitro. Drug Metab. Rev. 2017, 49, 395–437. [Google Scholar] [CrossRef]
- Alvarenga, D.; Mattos, M.; Lopes, M.; Marchesi, S.; Araújo, A.; Nakagaki, B.; Santos, M.; David, B.; De Souza, V.; Carvalho, É.; et al. Paradoxical Role of Matrix Metalloproteinases in Liver Injury and Regeneration after Sterile Acute Hepatic Failure. Cells 2018, 7, 247. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Ramachandran, A.; Jaeschke, H. Oxidative Stress during Acetaminophen Hepatotoxicity: Sources, Pathophysiological Role and Therapeutic Potential. Redox Biol. 2016, 10, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Huo, Y.; Yin, S.; Hu, H. Mechanisms of Acetaminophen-Induced Liver Injury and Its Implications for Therapeutic Interventions. Redox Biol. 2018, 17, 274–283. [Google Scholar] [CrossRef]
- Zhai, W.J.; Zhang, Z.B.; Xu, N.N.; Guo, Y.F.; Qiu, C.; Li, C.Y.; Deng, G.Z.; Guo, M.Y. Piperine Plays an Anti-Inflammatory Role in Staphylococcus Aureus Endometritis by Inhibiting Activation of NF-κB and MAPK Pathways in Mice. Evid.-Based Complement. Altern. Med. 2016, 2016, 8597208. [Google Scholar] [CrossRef]
- Haq, I.U.; Imran, M.; Nadeem, M.; Tufail, T.; Gondal, T.A.; Mubarak, M.S. Piperine: A Review of Its Biological Effects. Phyther. Res. 2021, 35, 680–700. [Google Scholar] [CrossRef]
- Lim, A.Y.L.; Segarra, I.; Chakravarthi, S.; Akram, S.; Judson, J.P. Histopathology and Biochemistry Analysis of the Interaction between Sunitinib and Paracetamol in Mice. BMC Pharmacol. 2010, 10, 14. [Google Scholar] [CrossRef]
- Booranasubkajorn, S.; Huabprasert, S.; Wattanarangsan, J.; Chotitham, P.; Jutasompakorn, P.; Laohapand, T.; Akarasereenont, P.; Tripatara, P. Vasculoprotective and Vasodilatation Effects of Herbal Formula (Sahatsatara) and Piperine in Spontaneously Hypertensive Rats. Phytomedicine 2016, 24, 148–156. [Google Scholar] [CrossRef]
- Intasar, S.; Taqvi, H.; Shah, A.J.; Gilani, A.H. Blood Pressure Lowering and Vasomodulator Effects of Piperine. J. Cardiovasc. Pharmacol. 2008, 52, 452–458. [Google Scholar] [CrossRef] [Green Version]
- Dhivya, V.; Bharathi, L.; Chirayil, H.T.; Sathiskumar, S.; Huang, C.; Vijaya, V. Piperine Modulates Isoproterenol Induced Myocardial Ischemia through Antioxidant and Anti-Dyslipidemic Effect in Male Wistar Rats. Biomed. Pharmacother. 2017, 87, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Dutta, M.; Ghosh, K.; Mishra, P.; Jain, G.; Rangari, V. Function Ascorbate Induced Toxic Injury to Goat Cardiac Mitochondria in Vitro. Food Funct. 2014, 5, 2252–2267. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, K. Black Pepper and Its Pungent Principle-Piperine: A Review of Diverse Physiological Effects. Taylor Fr. 2007, 47, 37–41. [Google Scholar] [CrossRef]
- Geervliet, E.; Bansal, R. Matrix Metalloproteinases as Potential Biomarkers. Cells 2020, 9, 1212. [Google Scholar] [CrossRef]
- Kato, H.; Kuriyama, N.; Duarte, S.; Clavien, P.-A.; Busuttil, R.W.; Coito, A.J. MMP-9 Deficiency Shelters Endothelial Pecam-1 Expression and Enhances Regeneration of Steatotic Livers after Ischemia and Reperfusion Injury. J. Hepatol. 2015, 60, 1032–1039. [Google Scholar] [CrossRef]
- Ito, Y.; Abril, E.R.; Bethea, N.W.; Mccuskey, R.S. Inhibition of Matrix Metalloproteinases Minimizes Hepatic Microvascular Injury in Response to Acetaminophen in Mice. Toxicol. Sci. 2005, 83, 190–196. [Google Scholar] [CrossRef]
- McGill, M.R.; Jaeschke, H. Metabolism and Disposition of Acetaminophen: Recent Advances in Relation to Hepatotoxicity and Diagnosis. Pharm. Res. 2013, 30, 2174–2187. [Google Scholar] [CrossRef]
- Cheung, C.; Yu, A.; Ward, J.M.; Krausz, K.W.; Akiyama, T.E.; Feigenbaum, L.; Gonzalez, F.J. The CYP2E1-Humanized Transgenic Mouse: Role of CYP2E1 in Acetaminophen Hepatotoxicity. Drug Metab. Dispos. 2005, 33, 449–457. [Google Scholar] [CrossRef]
- Papackova, Z.; Heczkova, M.; Dankova, H.; Sticova, E.; Lodererova, A.; Bartonova, L.; Poruba, M.; Cahova, M. Silymarin Prevents Acetaminophen-Induced Hepatotoxicity in Mice. PLoS ONE 2018, 13, e0191353. [Google Scholar] [CrossRef]
- Xie, W.; Wang, M.; Chen, C.; Zhang, X.; Melzig, M.F. Hepatoprotective Effect of Isoquercitrin against Acetaminophen-Induced Liver Injury. Life Sci. 2016, 152, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Murray, M. Toxicological Actions of Plant-Derived and Anthropogenic Methylenedioxyphenyl-Substituted Chemicals in Mammals and Insects. J. Toxicol. Environ. Health Part B Crit. Rev. 2012, 15, 365–395. [Google Scholar] [CrossRef] [PubMed]
- Imaeda, A.B.; Watanabe, A.; Sohail, M.A.; Mahmood, S.; Mohamadnejad, M.; Sutterwala, F.S.; Flavell, R.A.; Mehal, W.Z. Acetaminophen-Induced Hepatotoxicity in Mice Is Dependent on Tlr9 and the Nalp3 Inflammasome. J. Clin. Investig. 2009, 119, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Woolbright, B.L.; Jaeschke, H. Role of the Inflammasome in Acetaminophen-Induced Liver Injury and Acute Liver Failure. J. Hepatol. 2018, 66, 836–848. [Google Scholar] [CrossRef]
- El-shishtawy, M.M.; Eissa, L.A. Cepharanthine and Piperine Ameliorate Diabetic Nephropathy in Rats: Role of NF-κB and NLRP3 Inflammasome; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 2010058925. [Google Scholar]
- Laskin, D.L.; Laskin, J.D. Role of Macrophages and Inflammatory Mediators in Chemically Induced Toxicity. Elsevier 2001, 160, 111–118. [Google Scholar] [CrossRef]
- Hashizume, M.; Higuchi, Y.; Uchiyama, Y.; Mihara, M. Cytokine IL-6 Plays an Essential Role in Neutrophilia under Inflammation. Cytokine 2011, 54, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in Inflammation, Immunity, and Disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coelho, A.M.; Queiroz, I.F.; Perucci, L.O.; Souza, M.O.d.; Lima, W.G.; Talvani, A.; Costa, D.C. Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice. Pharmaceutics 2022, 14, 1800. https://doi.org/10.3390/pharmaceutics14091800
Coelho AM, Queiroz IF, Perucci LO, Souza MOd, Lima WG, Talvani A, Costa DC. Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice. Pharmaceutics. 2022; 14(9):1800. https://doi.org/10.3390/pharmaceutics14091800
Chicago/Turabian StyleCoelho, Aline Meireles, Isabela Ferreira Queiroz, Luiza Oliveira Perucci, Melina Oliveira de Souza, Wanderson Geraldo Lima, André Talvani, and Daniela Caldeira Costa. 2022. "Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice" Pharmaceutics 14, no. 9: 1800. https://doi.org/10.3390/pharmaceutics14091800
APA StyleCoelho, A. M., Queiroz, I. F., Perucci, L. O., Souza, M. O. d., Lima, W. G., Talvani, A., & Costa, D. C. (2022). Piperine as Therapeutic Agent in Paracetamol-Induced Hepatotoxicity in Mice. Pharmaceutics, 14(9), 1800. https://doi.org/10.3390/pharmaceutics14091800






