Progress in Polymeric Micelles for Drug Delivery Applications
Abstract
:1. Introduction
2. Types of Polymeric Micelles
3. Methods of Preparation
3.1. Direct Dissolution Method
3.2. Simple Mixing
3.3. Solvent Evaporation Method
3.4. Dialysis Method
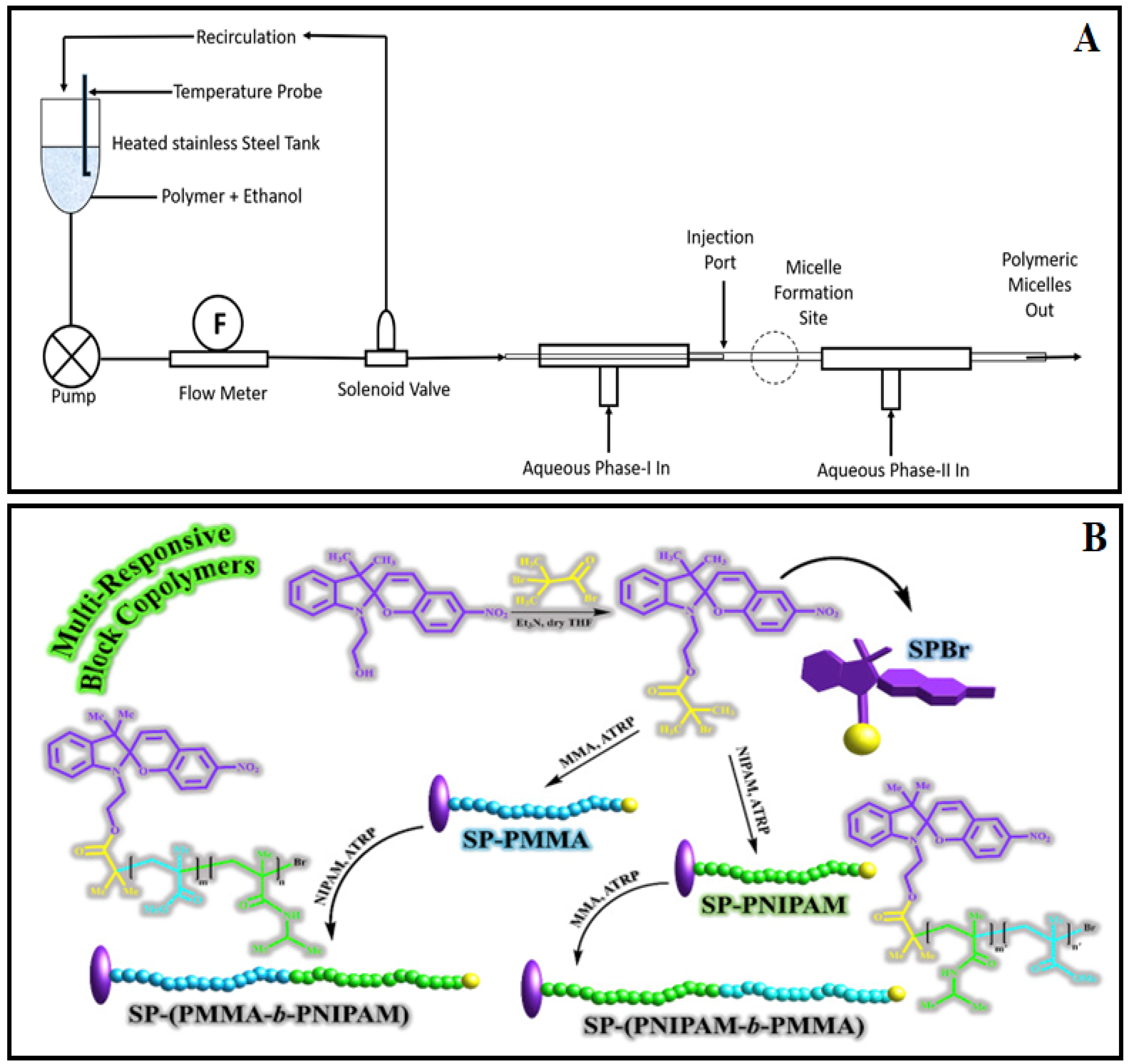
3.5. Continuous Processing
3.6. In Situ Charge-Neutralization-Controlled Particle Coagulation Mechanism
3.7. Spiropyran-Initiated Atom Transfer Polymerization
4. Methods of Characterization
4.1. Critical Micelle Concentration Determination (CMC)
4.2. Morphological Characterization
4.3. Physicochemical Characterization
4.4. Monitoring the Nano-Bio Interaction
5. Molecular Dynamics of Polymeric Micelles
6. Stability of Polymeric Micelles
7. Safety and Quality of Polymeric Micelles
8. Drug Delivery Applications
8.1. Oral
8.2. Parenteral
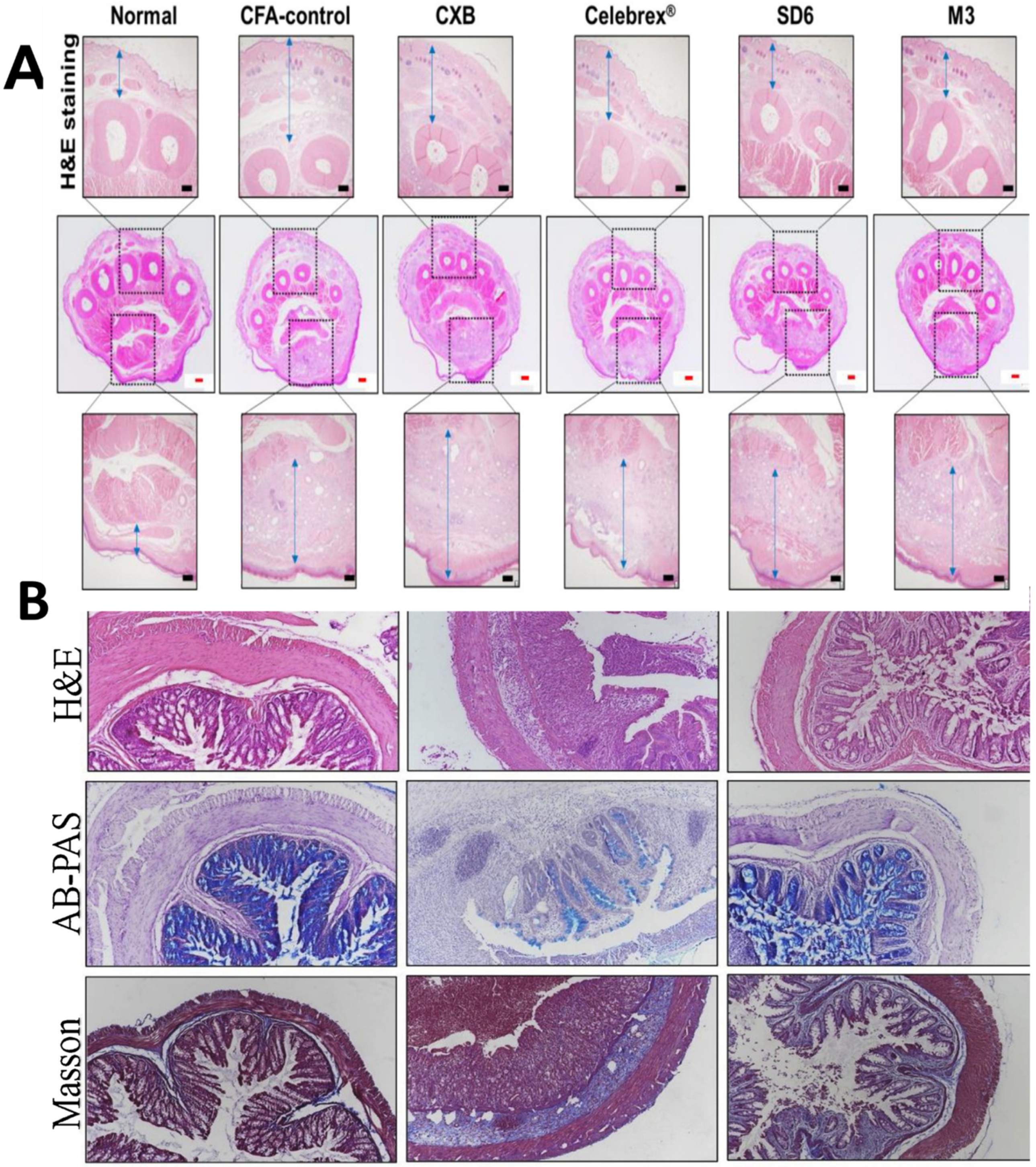
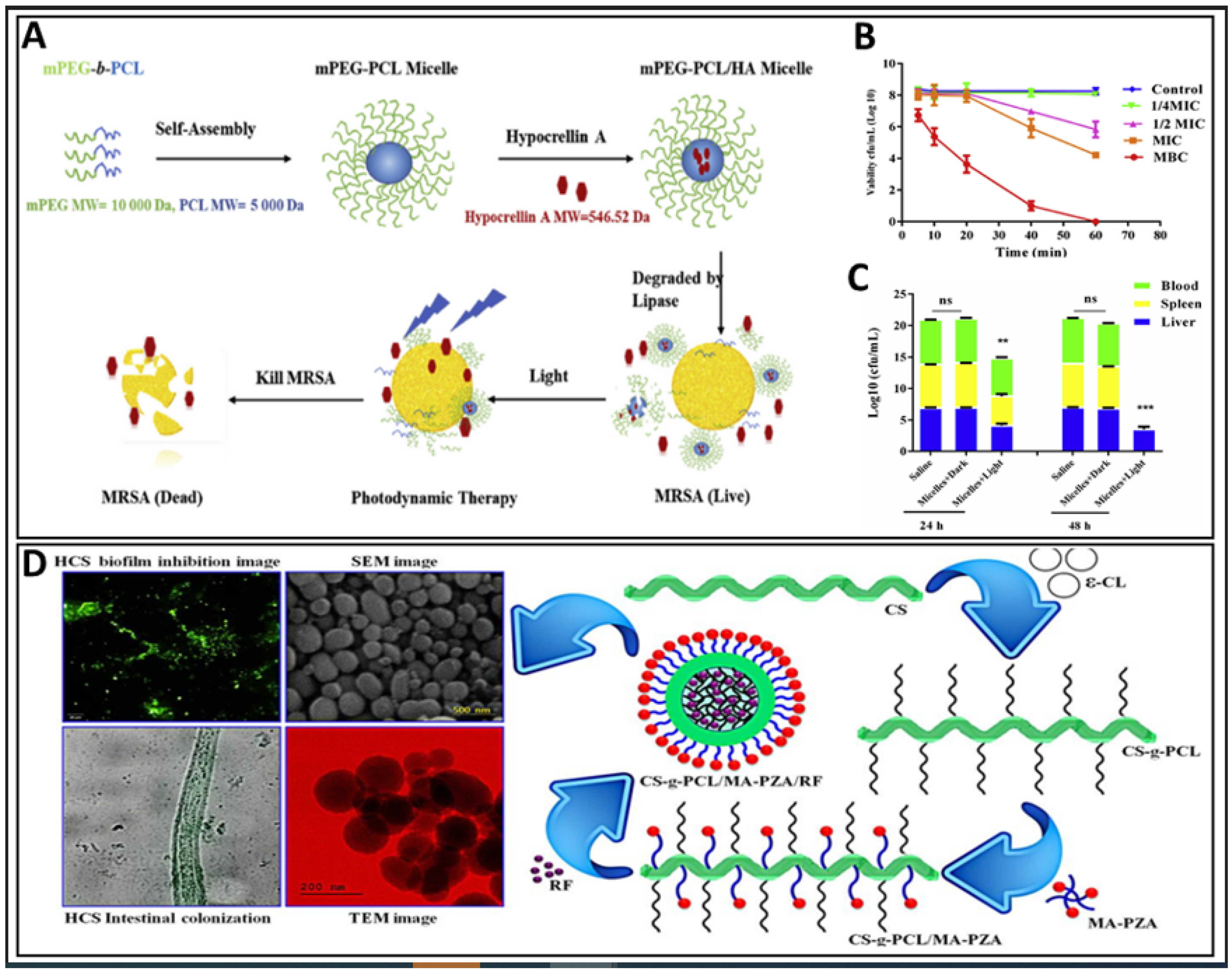
8.3. Antibacterial
8.4. Topical
8.5. Transdermal
8.6. Periodontal
8.7. Intranasal
8.8. Gene Delivery
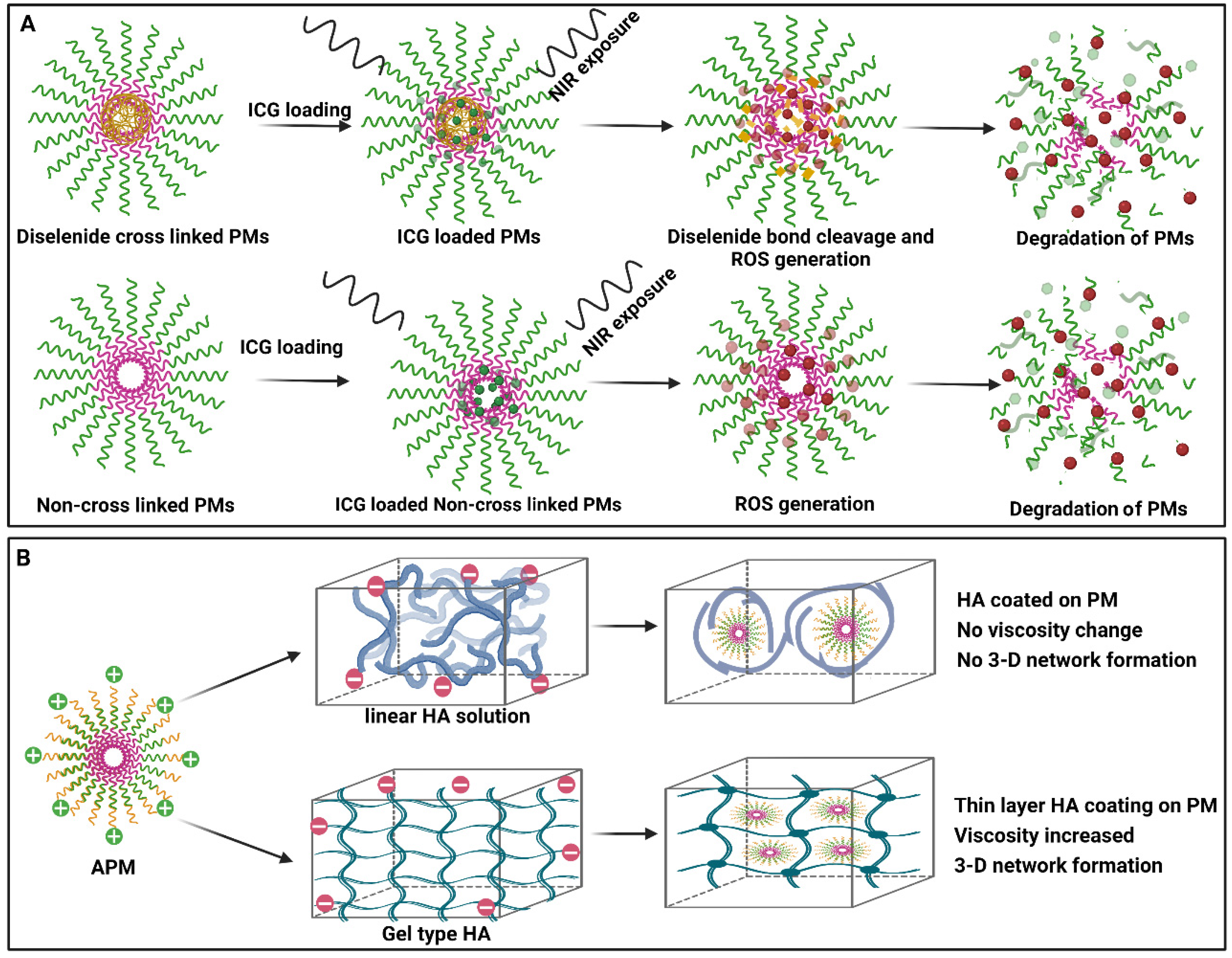
9. Theranostic Applications
10. Advanced Polymeric Micelle Systems
10.1. Brain-Targeted Systems
10.2. Mitochondria-Targeted Systems
10.3. Tumor-Targeted Systems
11. Mixed Micelle Systems
12. Other Special Types
13. In Vivo Stability of Polymeric Micelles
14. Patents on Polymeric Micelles
15. Clinical Applications of Polymeric Micelles
16. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Macha, I.J.; Ben-Nissan, B.; Vilchevskaya, E.N.; Morozova, A.S.; Abali, B.E.; Müller, W.H.; Rickert, W. Drug Delivery from Polymer-Based Nanopharmaceuticals—An Experimental Study Complemented by Simulations of Selected Diffusion Processes. Front. Bioeng. Biotechnol. 2019, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y.K.; Kim, S.W. Recent Advances in Polymeric Drug Delivery Systems. Biomater. Res. 2020, 24, 12. [Google Scholar] [CrossRef] [PubMed]
- Liechty, W.B.; Kryscio, D.R.; Slaughter, B.V.; Peppas, N.A. Polymers for Drug Delivery Systems. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 149–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Begines, B.; Ortiz, T.; Pérez-Aranda, M.; Martínez, G.; Merinero, M.; Argüelles-Arias, F.; Alcudia, A. Polymeric Nanoparticles for Drug Delivery: Recent Developments and Future Prospects. Nanomaterials 2020, 10, 1403. [Google Scholar] [CrossRef]
- Paul, D.R.; Robeson, L.M. Polymer Nanotechnology: Nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef] [Green Version]
- Bennet, D.; Kim, S. Polymer Nanoparticles for Smart Drug Delivery; Intechopen: London, UK, 2014; Volume 8. [Google Scholar]
- Tang, Z.; He, C.; Tian, H.; Ding, J.; Hsiao, B.S.; Chu, B.; Chen, X. Polymeric Nanostructured Materials for Biomedical Applications. Prog. Polym. Sci. 2016, 60, 86–128. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, B.; Biswas, S. Polymeric Micelles in Cancer Therapy: State of the Art. J. Control. Release 2021, 332, 127–147. [Google Scholar] [CrossRef]
- Wakaskar, R.R. Polymeric Micelles and Their Properties. J. Nanomed. Nanotechnol. 2017, 8, 433. [Google Scholar] [CrossRef] [Green Version]
- Jhaveri, A.M.; Torchilin, V.P. Multifunctional Polymeric Micelles for Delivery of Drugs and SiRNA. Front. Pharmacol. 2014, 5, 77. [Google Scholar] [CrossRef] [Green Version]
- Almgren, M. Mixed Micelles and Other Structures in the Solubilization of Bilayer Lipid Membranes by Surfactants. Biochim. Biophys. Acta Biomembr. 2000, 1508, 146–163. [Google Scholar] [CrossRef] [Green Version]
- Deshmukh, A.S.; Chauhan, P.N.; Noolvi, M.N.; Chaturvedi, K.; Ganguly, K.; Shukla, S.S.; Nadagouda, M.N.; Aminabhavi, T.M. Polymeric Micelles: Basic Research to Clinical Practice. Int. J. Pharm. 2017, 532, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Yadav, H.K.S.; Almokdad, A.A.; Shaluf, S.I.M.; Debe, M.S. Polymer-Based Nanomaterials for Drug-Delivery Carriers; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128140338. [Google Scholar]
- Gupta, A.; Costa, A.P.; Xu, X.; Lee, S.-L.; Cruz, C.N.; Bao, Q.; Burgess, D.J. Formulation and Characterization of Curcumin Loaded Polymeric Micelles Produced via Continuous Processing. Int. J. Pharm. 2020, 583, 119340. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; van Steenbergen, M.J.; Lou, B.; Liu, Y.; Hennink, W.E.; Kok, R.J. Folate Decorated Polymeric Micelles for Targeted Delivery of the Kinase Inhibitor Dactolisib to Cancer Cells. Int. J. Pharm. 2020, 582, 119305. [Google Scholar] [CrossRef]
- Yang, X.; Lian, K.; Tan, Y.; Zhu, Y.; Liu, X.; Zeng, Y.; Yu, T.; Meng, T.; Yuan, H.; Hu, F. Selective Uptake of Chitosan Polymeric Micelles by Circulating Monocytes for Enhanced Tumor Targeting. Carbohydr. Polym. 2020, 229, 115435. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yang, Z.; Liu, M.; Tao, Y.; Li, Z.; Wu, Z.; Gui, S. Facile Nose-to-Brain Delivery of Rotigotine-Loaded Polymer Micelles Thermosensitive Hydrogels: In Vitro Characterization and in Vivo Behavior Study. Int. J. Pharm. 2020, 577, 119046. [Google Scholar] [CrossRef] [PubMed]
- Oda, C.M.R.; Malfatti-Gasperini, A.A.; Malachias, A.; Pound-Lana, G.; Mosqueira, V.C.F.; Fernandes, R.S.; Oliveira, M.C.D.; de Barros, A.L.B.; Leite, E.A. Physical and Biological Effects of Paclitaxel Encapsulation on Disteraroylphosphatidylethanolamine-Polyethyleneglycol Polymeric Micelles. Colloids Surf. B Biointerfaces 2020, 188, 110760. [Google Scholar] [CrossRef]
- Šmejkalová, D.; Muthný, T.; Nešporová, K.; Hermannová, M.; Achbergerová, E.; Huerta-Angeles, G.; Svoboda, M.; Čepa, M.; Machalová, V.; Luptáková, D.; et al. Hyaluronan Polymeric Micelles for Topical Drug Delivery. Carbohydr. Polym. 2017, 156, 86–96. [Google Scholar] [CrossRef]
- Starigazdová, J.; Nešporová, K.; Čepa, M.; Šínová, R.; Šmejkalová, D.; Huerta-Angeles, G.; Velebný, V. In Vitro Investigation of Hyaluronan-Based Polymeric Micelles for Drug Delivery into the Skin: The Internalization Pathway. Eur. J. Pharm. Sci. 2020, 143, 105168. [Google Scholar] [CrossRef]
- Zhang, L.; Pu, Y.; Li, J.; Yan, J.; Gu, Z.; Gao, W.; He, B. PH Responsive Coumarin and Imidazole Grafted Polymeric Micelles for Cancer Therapy. J. Drug Deliv. Sci. Technol. 2020, 58, 101789. [Google Scholar] [CrossRef]
- Guo, L.-Y.; Yan, S.-Z.; Tao, X.; Yang, Q.; Li, Q.; Wang, T.-S.; Yu, S.-Q.; Chen, S.-L. Evaluation of Hypocrellin A-Loaded Lipase Sensitive Polymer Micelles for Intervening Methicillin-Resistant Staphylococcus Aureus Antibiotic-Resistant Bacterial Infection. Mater. Sci. Eng. C 2020, 106, 110230. [Google Scholar] [CrossRef]
- Gener, P.; Montero, S.; Xandri-Monje, H.; Díaz-Riascos, Z.V.; Rafael, D.; Andrade, F.; Martínez-Trucharte, F.; González, P.; Seras-Franzoso, J.; Manzano, A.; et al. ZileutonTM Loaded in Polymer Micelles Effectively Reduce Breast Cancer Circulating Tumor Cells and Intratumoral Cancer Stem Cells. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102106. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Qiu, L.; Wang, X.; Ouyang, H.; Li, T.; Han, L.; Zhang, X.; Xu, W.; Chu, K. Evaluation of Intestinal Permeation Enhancement with Carboxymethyl Chitosan-Rhein Polymeric Micelles for Oral Delivery of Paclitaxel. Int. J. Pharm. 2020, 573, 118840. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Tu, L.; Wang, G.; Qi, N.; Wu, W.; Zhang, W.; Feng, J. Multi-Functional Chitosan Polymeric Micelles as Oral Paclitaxel Delivery Systems for Enhanced Bioavailability and Anti-Tumor Efficacy. Int. J. Pharm. 2020, 578, 119105. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xue, P.; Tong, M.; Chen, R.; Yang, W.; ZhuGe, D.; Yuan, J.; Yao, Q.; Zhao, Y.; Xu, H. Injected Laquinimod D-α-Tocopheryl Polyethylene Glycol-1000 Succinate Polymeric Micelles for the Treatment of Inflammatory Bowel Disease. Colloids Surf. B Biointerfaces 2020, 185, 110575. [Google Scholar] [CrossRef]
- Zhao, Y.; Alakhova, D.Y.; Zhao, X.; Band, V.; Batrakova, E.V.; Kabanov, A. V Eradication of Cancer Stem Cells in Triple Negative Breast Cancer Using Doxorubicin/Pluronic Polymeric Micelles. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102124. [Google Scholar] [CrossRef]
- Niu, J.; Wang, L.; Yuan, M.; Zhang, J.; Chen, H.; Zhang, Y. Dual-Targeting Nanocarrier Based on Glucose and Folic Acid Functionalized Pluronic P105 Polymeric Micelles for Enhanced Brain Distribution. J. Drug Deliv. Sci. Technol. 2020, 57, 101343. [Google Scholar] [CrossRef]
- Li, Y.; Gao, F.; Guo, J.; Ren, P.; Tian, Z.; Bai, J.; Hua, J. Polymeric Micelles with Aggregation-Induced Emission Based on Microbial ε-Polylysine for Doxorubicin Delivery. Eur. Polym. J. 2020, 122, 109355. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Wei, G.; Zhao, J.; Yang, G.; Zhou, S. Stepwise Dual Targeting and Dual Responsive Polymer Micelles for Mitochondrion Therapy. J. Control. Release 2020, 322, 157–169. [Google Scholar] [CrossRef]
- Supasena, W.; Muangnoi, C.; Thaweesest, W.; Songkram, C.; Ueda, K.; Higashi, K.; Moribe, K.; Tanasupawat, S.; Rojsitthisak, P. Enhanced Antipsoriatic Activity of Mycophenolic Acid against the TNF-α-Induced HaCaT Cell Proliferation by Conjugated Poloxamer Micelles. J. Pharm. Sci. 2020, 109, 1153–1160. [Google Scholar] [CrossRef]
- Lu, Y.; Gao, X.; Cao, M.; Wu, B.; Su, L.; Chen, P.; Miao, J.; Wang, S.; Xia, R.; Qian, J. Interface Crosslinked MPEG-b-PAGE-b-PCL Triblock Copolymer Micelles with High Stability for Anticancer Drug Delivery. Colloids Surf. B Biointerfaces 2020, 189, 110830. [Google Scholar] [CrossRef]
- Liao, J.; Peng, H.; Wei, X.; Song, Y.; Liu, C.; Li, D.; Yin, Y.; Xiong, X.; Zheng, H.; Wang, Q. A Bio-Responsive 6-Mercaptopurine/Doxorubicin Based “Click Chemistry” Polymeric Prodrug for Cancer Therapy. Mater. Sci. Eng. C 2020, 108, 110461. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.S.; Jang, M.-S.; Fu, Y.; Lee, J.H.; Lee, D.S.; Li, Y.; Yang, H.Y. Intracellular Delivery of Cytochrome C Using Hypoxia-Responsive Polypeptide Micelles for Efficient Cancer Therapy. Mater. Sci. Eng. C 2020, 114, 111069. [Google Scholar] [CrossRef] [PubMed]
- Negi, P.; Sharma, G.; Verma, C.; Garg, P.; Rathore, C.; Kulshrestha, S.; Lal, U.R.; Gupta, B.; Pathania, D. Novel Thymoquinone Loaded Chitosan-Lecithin Micelles for Effective Wound Healing: Development, Characterization, and Preclinical Evaluation. Carbohydr. Polym. 2020, 230, 115659. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhao, L.; Shen, C.; He, Y.; Yang, F.; Zhang, G.; Jia, M.; Zeng, R.; Li, C.; Qiao, R. Reactive Oxygen Species-Responsive Amino Acid-Based Polymeric Nanovehicles for Tumor-Selective Anticancer Drug Delivery. Mater. Sci. Eng. C 2020, 106, 110159. [Google Scholar] [CrossRef] [PubMed]
- Mehnath, S.; Chitra, K.; Karthikeyan, K.; Jeyaraj, M. Localized Delivery of Active Targeting Micelles from Nanofibers Patch for Effective Breast Cancer Therapy. Int. J. Pharm. 2020, 584, 119412. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.-Q.; Zhang, C.-L.; Qiao, J.-B.; Cui, P.-F.; Xing, L.; Oh, Y.-K.; Jiang, H.-L. Extracellular Matrix-Penetrating Nanodrill Micelles for Liver Fibrosis Therapy. Biomaterials 2020, 230, 119616. [Google Scholar] [CrossRef]
- Wu, Z.; Li, S.; Cai, Y.; Chen, F.; Chen, Y.; Luo, X. Synergistic Action of Doxorubicin and 7-Ethyl-10-Hydroxycamptothecin Polyphosphorylcholine Polymer Prodrug. Colloids Surf. B Biointerfaces 2020, 189, 110741. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, W.; Wang, Y.; Chen, Y.; Xie, J.; Su, J.; Huang, C. One-Step Treatment of Periodontitis Based on a Core-Shell Micelle-in-Nanofiber Membrane with Time-Programmed Drug Release. J. Control. Release 2020, 320, 201–213. [Google Scholar] [CrossRef]
- Sotoudegan, F.; Amini, M.; Faizi, M.; Aboofazeli, R. Nimodipine-Loaded Pluronic® Block Copolymer Micelles: Preparation, Characterization, In-Vitro and In-Vivo Studies. Iran. J. Pharm. Res. IJPR 2016, 15, 641. [Google Scholar] [CrossRef]
- Cholkar, K.; Patel, A.; Vadlapudi, A.D.; MitraMitra, A.K. Novel Nanomicellar Formulation Approaches for Anterior and Posterior Segment Ocular Drug Delivery. Recent Pat. Nanotechnol. 2012, 2, 82–95. [Google Scholar] [CrossRef]
- Florinas, S.; Liu, M.; Fleming, R.; Van Vlerken-Ysla, L.; Ayriss, J.; Gilbreth, R.; Dimasi, N.; Gao, C.; Wu, H.; Xu, Z.-Q. A Nanoparticle Platform to Evaluate Bioconjugation and Receptor-Mediated Cell Uptake Using Cross-Linked Polyion Complex Micelles Bearing Antibody Fragments. Biomacromolecules 2016, 17, 1818–1833. [Google Scholar] [CrossRef] [PubMed]
- Mathew, C.; Sebastian, K.; Daisy, P.A. A Comprehensive Review on Polymeric Micelles. Int. J. Pharm. Sci. Rev. Res. 2020, 61, 36–39. [Google Scholar]
- Costa, A.P.; Xu, X.; Khan, M.A.; Burgess, D.J. Liposome Formation Using a Coaxial Turbulent Jet in Co-Flow. Pharm. Res. 2016, 33, 404–416. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, X.; Han, Y.; Liu, B.; Hu, Y.; Liu, X. Preparation of Monodisperse Polymer Particle via an in Situ Charge Neutralization-Controlled Particle Coagulation Mechanism in Macroemulsion Polymerization. Colloids Surf. A Physicochem. Eng. Asp. 2020, 587, 124345. [Google Scholar] [CrossRef]
- Razavi, B.; Abdollahi, A.; Roghani-Mamaqani, H.; Salami-Kalajahi, M. Light- and Temperature-Responsive Micellar Carriers Prepared by Spiropyran-Initiated Atom Transfer Polymerization: Investigation of Photochromism Kinetics, Responsivities, and Controlled Release of Doxorubicin. Polymer 2020, 187, 122046. [Google Scholar] [CrossRef]
- European Medicines Agency. 2 Committee for Medicinal Products for Human Use (CHMP) 3 Joint MHLW/EMA Reflection Paper on the Development of 4 Block Copolymer Micelle Medicinal Products; European Medicines Agency: Amsterdam, The Netherlands, 2013.
- Gao, M.; Yang, Y.; Bergfel, A.; Huang, L.; Zheng, L.; Bowden, T.M. Self-Assembly of Cholesterol End-Capped Polymer Micelles for Controlled Drug Delivery. J. Nanobiotechnol. 2020, 18, 13. [Google Scholar] [CrossRef] [Green Version]
- Scholz, N.; Behnke, T.; Resch-Genger, U. Determination of the Critical Micelle Concentration of Neutral and Ionic Surfactants with Fluorometry, Conductometry, and Surface Tension—A Method Comparison. J. Fluoresc. 2018, 28, 465–476. [Google Scholar] [CrossRef]
- Esteves, R.; Dikici, B.; Lehman, M.; Mazumder, Q.; Onukwuba, N. Determination of Aqueous Surfactant Solution Surface Tensions with a Surface Tensiometer. Undergrad. Res. J. 2016, 1, 4. [Google Scholar]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric Micelles in Drug Delivery: An Insight of the Techniques for Their Characterization and Assessment in Biorelevant Conditions. J. Control. Release 2021, 332, 312–336. [Google Scholar] [CrossRef]
- Bagheri, M.; Bresseleers, J.; Varela-Moreira, A.; Sandre, O.; Meeuwissen, S.A.; Schiffelers, R.M.; Metselaar, J.M.; Van Nostrum, C.F.; Van Hest, J.C.M.; Hennink, W.E. Effect of Formulation and Processing Parameters on the Size of MPEG- b-p(HPMA-Bz) Polymeric Micelles. Langmuir 2018, 34, 15495–15506. [Google Scholar] [CrossRef] [Green Version]
- Su, H.; Wang, F.; Ran, W.; Zhang, W.; Dai, W.; Wang, H.; Anderson, C.F.; Wang, Z.; Zheng, C.; Zhang, P.; et al. The Role of Critical Micellization Concentration in Efficacy and Toxicity of Supramolecular Polymers. Proc. Natl. Acad. Sci. USA 2020, 117, 4518–4526. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.; Oh, Y.T.; Seok Youn, Y.; Nam, M.; Park, B.; Yun, J.; Kim, J.H.; Song, H.-T.; Oh, K.T. Binary Mixing of Micelles Using Pluronics for a Nano-Sized Drug Delivery System. Colloids Surf. B Biointerfaces 2010, 82, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Pitt, W.G. A Polymeric Micelle System with a Hydrolysable Segment for Drug Delivery. J. Biomater. Sci. Polym. Ed. 2006, 17, 591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hait, S.K.; Moulik, S.P. Determination of Critical Micelle Concentration (CMC) of Nonionic Surfactants by Donor-Acceptor Interaction with Lodine and Correlation of CMC with Hydrophile-Lipophile Balance and Other Parameters of the Surfactants. J. Surfactants Deterg. 2001, 4, 303–309. [Google Scholar] [CrossRef]
- Topel, Ö.; Çakir, B.A.; Budama, L.; Hoda, N. Determination of Critical Micelle Concentration of Polybutadiene-Block- Poly(Ethyleneoxide) Diblock Copolymer by Fluorescence Spectroscopy and Dynamic Light Scattering. J. Mol. Liq. 2013, 177, 40–43. [Google Scholar] [CrossRef]
- McKenzie, B.E.; Holder, S.J.; Sommerdijk, N.A.J.M. Assessing Internal Structure of Polymer Assemblies from 2D to 3D CryoTEM: Bicontinuous Micelles. Curr. Opin. Colloid Interface Sci. 2012, 17, 343–349. [Google Scholar] [CrossRef] [Green Version]
- Gu, P.F.; Xu, H.; Sui, B.W.; Gou, J.X.; Meng, L.K.; Sun, F.; Wang, X.J.; Qi, N.; Zhang, Y.; He, H.B.; et al. Polymeric Micelles Based on Poly(Ethylene Glycol) Block Poly(Racemic Amino Acids) Hybrid Polypeptides: Conformation-Facilitated Drug-Loading Behavior and Potential Application as Effective Anticancer Drug Carriers. Int. J. Nanomed. 2012, 7, 109. [Google Scholar] [CrossRef] [Green Version]
- Touve, M.A.; Figg, C.A.; Wright, D.B.; Park, C.; Cantlon, J.; Sumerlin, B.S.; Gianneschi, N.C. Polymerization-Induced Self-Assembly of Micelles Observed by Liquid Cell Transmission Electron Microscopy. ACS Cent. Sci. 2018, 4, 543–547. [Google Scholar] [CrossRef]
- Puig-Rigall, J.; Fernández-Rubio, C.; González-Benito, J.; Houston, J.E.; Radulescu, A.; Nguewa, P.; González-Gaitano, G. Structural Characterization by Scattering and Spectroscopic Methods and Biological Evaluation of Polymeric Micelles of Poloxamines and TPGS as Nanocarriers for Miltefosine Delivery. Int. J. Pharm. 2020, 578, 119057. [Google Scholar] [CrossRef]
- Sakou, M.; Takechi, A.; Murakami, S.I.; Sakurai, K.; Akiba, I. Study of the Internal Structure of Polymer Micelles by Anomalous Small-Angle X-ray Scattering at Two Edges. J. Appl. Crystallogr. 2013, 46, 1407–1413. [Google Scholar] [CrossRef]
- Nakanishi, R.; Machida, G.; Kinoshita, M.; Sakurai, K.; Akiba, I. Anomalous Small-Angle X-ray Scattering Study on the Spatial Distribution of Hydrophobic Molecules in Polymer Micelles. Polym. J. 2016, 48, 801–806. [Google Scholar] [CrossRef]
- Salimi, A.; Makhmal Zadeh, B.S.; Kazemi, M. Preparation and Optimization of Polymeric Micelles as an Oral Drug Delivery System for Deferoxamine Mesylate: In Vitro and Ex Vivo Studies. Res. Pharm. Sci. 2019, 14, 293. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, T.; Tsuchiya, K.; Numata, K. Endosome-Escaping Micelle Complexes Dually Equipped with Cell-Penetrating and Endosome-Disrupting Peptides for Efficient DNA Delivery into Intact Plants. Nanoscale 2021, 13, 5679–5692. [Google Scholar] [CrossRef] [PubMed]
- Marques, P.E.; Antunes, M.M.; David, B.A.; Pereira, R.V.; Teixeira, M.M.; Menezes, G.B. Imaging Liver Biology in Vivo Using Conventional Confocal Microscopy. Nat. Protoc. 2015, 10, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Nichols, J.W.; Toh, K.; Nomoto, T.; Cabral, H.; Miura, Y.; Christie, R.J.; Yamada, N.; Ogura, T.; Kano, M.R.; et al. Vascular Bursts Enhance Permeability of Tumour Blood Vessels and Improve Nanoparticle Delivery. Nat. Nanotechnol. 2016, 11, 533–538. [Google Scholar] [CrossRef] [PubMed]
- Sindhwani, S.; Syed, A.M.; Ngai, J.; Kingston, B.R.; Maiorino, L.; Rothschild, J.; MacMillan, P.; Zhang, Y.; Rajesh, N.U.; Hoang, T.; et al. The Entry of Nanoparticles into Solid Tumours. Nat. Mater. 2020, 19, 566–575. [Google Scholar] [CrossRef]
- Dirisala, A.; Uchida, S.; Toh, K.; Li, J.; Osawa, S.; Tockary, T.A.; Liu, X.; Abbasi, S.; Hayashi, K.; Mochida, Y.; et al. Transient Stealth Coating of Liver Sinusoidal Wall by Anchoring Two-Armed PEG for Retargeting Nanomedicines. Sci. Adv. 2020, 6, eabb8133. [Google Scholar] [CrossRef]
- Timmers, E.M.; Magana, J.R.; Schoenmakers, S.M.C.; Michel Fransen, P.; Janssen, H.M.; Voets, I.K. Sequence of Polyurethane Ionomers Determinative for Core Structure of Surfactant–Copolymer Complexes. Int. J. Mol. Sci. 2021, 22, 337. [Google Scholar] [CrossRef]
- Sasaki, S.; Machida, G.; Nakanishi, R.; Kinoshita, M.; Akiba, I. Elucidation of Spatial Distribution of Hydrophobic Aromatic Compounds Encapsulated in Polymer Micelles by Anomalous Small-Angle X-ray Scattering. Polymers 2018, 10, 180. [Google Scholar] [CrossRef] [Green Version]
- El Aferni, A.; Guettari, M.; Kamli, M.; Tajouri, T.; Ponton, A. A Structural Study of a Polymer-Surfactant System in Dilute and Entangled Regime: Effect of High Concentrations of Surfactant and Polymer Molecular Weight. J. Mol. Struct. 2020, 1199, 127052. [Google Scholar] [CrossRef]
- Cheng, M.H.; Yao, I.C.; Chiu, T.Y.; Chen, J.T. Block Copolymer Micelles Confined in Cylindrical Nanopores: Effects of Annealing Solvents and Hybridization. React. Funct. Polym. 2020, 150, 104534. [Google Scholar] [CrossRef]
- Siboro, S.A.P.; Anugrah, D.S.B.; Jeong, Y.T.; Yoo, S.I.; Lim, K.T. Systematic Investigation to the Effects of Near-Infrared Light Exposure on Polymeric Micelles of Poly(Ethylene Glycol)-Block-Poly(Styrene-Alt-Maleic Anhydride) Loaded with Indocyanine Green. Polym. Degrad. Stab. 2019, 167, 241–249. [Google Scholar] [CrossRef]
- Sze, L.P.; Li, H.Y.; Lai, K.L.A.; Chow, S.F.; Li, Q.; KennethTo, K.W.; Lam, T.N.T.; Lee, W.Y.T. Oral Delivery of Paclitaxel by Polymeric Micelles: A Comparison of Different Block Length on Uptake, Permeability and Oral Bioavailability. Colloids Surf. B Biointerfaces 2019, 184, 110554. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Park, D.; Yang, T.; Lee, J.Y.; Kang, J.Y.; Kim, D.; Kim, J.W.; Jin, L.; Kim, J.W. Controlled Rheological Behaviors of Hyaluronic Acid Solutions through Attractive Polymeric Micelle-Mediated Interchain Association. J. Ind. Eng. Chem. 2019, 75, 61–68. [Google Scholar] [CrossRef]
- Kacar, G. Thermodynamic Stability of Ibuprofen Loaded Poloxamer Micelles. Chem. Phys. 2020, 533, 110713. [Google Scholar] [CrossRef]
- Chen, Q.; Luo, L.; Xue, Y.; Han, J.; Liu, Y.; Zhang, Y.; Yin, T.; Wang, L.; Cun, D.; Gou, J.; et al. Cisplatin-Loaded Polymeric Complex Micelles with a Modulated Drug/Copolymer Ratio for Improved in Vivo Performance. Acta Biomater. 2019, 92, 205–218. [Google Scholar] [CrossRef]
- Feitosa, V.A.; Almeida, V.C.D.; Malheiros, B.; Castro, R.D.D.; Barbosa, L.R.S.; Cerize, N.N.P.; Rangel-Yagui, C.D.O. Polymeric Micelles of Pluronic F127 Reduce Hemolytic Potential of Amphiphilic Drugs. Colloids Surf. B Biointerfaces 2019, 180, 177–185. [Google Scholar] [CrossRef]
- Choi, J.-S.; Lee, D.-H.; Ahn, J.B.; Sim, S.; Heo, K.-S.; Myung, C.-S.; Park, J.-S. Therapeutic Effects of Celecoxib Polymeric Systems in Rat Models of Inflammation and Adjuvant-Induced Rheumatoid Arthritis. Mater. Sci. Eng. C 2020, 114, 111042. [Google Scholar] [CrossRef]
- Almeida, A.; Araújo, M.; Novoa-Carballal, R.; Andrade, F.; Gonçalves, H.; Reis, R.L.; Lúcio, M.; Schwartz, S.; Sarmento, B. Novel Amphiphilic Chitosan Micelles as Carriers for Hydrophobic Anticancer Drugs. Mater. Sci. Eng. C 2020, 112, 110920. [Google Scholar] [CrossRef]
- Wei, X.; Liu, L.; Li, X.; Wang, Y.; Guo, X.; Zhao, J.; Zhou, S. Selectively Targeting Tumor-Associated Macrophages and Tumor Cells with Polymeric Micelles for Enhanced Cancer Chemo-Immunotherapy. J. Control. Release 2019, 313, 42–53. [Google Scholar] [CrossRef]
- Shi, X.; Amarnath Praphakar, R.; Suganya, K.; Murugan, M.; Sasidharan, P.; Rajan, M. In Vivo Approach of Simply Constructed Pyrazinamide Conjugated Chitosan-g-Polycaprolactone Micelles for Methicillin Resistance Staphylococcus Aureus. Int. J. Biol. Macromol. 2020, 158, 636–647. [Google Scholar] [CrossRef] [PubMed]
- Gerola, A.P.; Costa, P.F.A.; de Morais, F.A.P.; Tsubone, T.M.; Caleare, A.O.; Nakamura, C.V.; Brunaldi, K.; Caetano, W.; Kimura, E.; Hioka, N. Liposome and Polymeric Micelle-Based Delivery Systems for Chlorophylls: Photodamage Effects on Staphylococcus Aureus. Colloids Surf. B Biointerfaces 2019, 177, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Witting, M.; Boreham, A.; Brodwolf, R.; Vávrová, K.; Alexiev, U.; Friess, W.; Hedtrich, S. Interactions of Hyaluronic Acid with the Skin and Implications for the Dermal Delivery of Biomacromolecules. Mol. Pharm. 2015, 12, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, Y.; Chen, X.; Su, J.; Huang, C. Enhanced Efficacy of Baicalin-Loaded TPGS Polymeric Micelles against Periodontitis. Mater. Sci. Eng. C 2019, 101, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Garg, C.; Priyam, A.; Kumar, P.; Sharma, A.K.; Gupta, A. In Vitro Assessment of Core-Shell Micellar Nanostructures of Amphiphilic Cationic Polymer-Peptide Conjugates as Efficient Gene and Drug Carriers. J. Pharm. Sci. 2020, 109, 2847–2853. [Google Scholar] [CrossRef]
- Shao, C.; Xiao, F.; Guo, H.; Yu, J.; Jin, D.; Wu, C.; Xi, L.; Tian, L. Utilizing Polymer Micelle to Control Dye J-Aggregation and Enhance Its Theranostic Capability. iScience 2019, 22, 229–239. [Google Scholar] [CrossRef] [Green Version]
- Trubitsyn, G.; Nguyen, V.N.; Di Tommaso, C.; Borchard, G.; Gurny, R.; Möller, M. Impact of Covalently Nile Red and Covalently Rhodamine Labeled Fluorescent Polymer Micelles for the Improved Imaging of the Respective Drug Delivery System. Eur. J. Pharm. Biopharm. 2019, 142, 480–487. [Google Scholar] [CrossRef]
- Lu, L.; Zhao, X.; Fu, T.; Li, K.; He, Y.; Luo, Z.; Dai, L.; Zeng, R.; Cai, K. An IRGD-Conjugated Prodrug Micelle with Blood-Brain-Barrier Penetrability for Anti-Glioma Therapy. Biomaterials 2020, 230, 119666. [Google Scholar] [CrossRef]
- Karami, Z.; Sadighian, S.; Rostamizadeh, K.; Hosseini, S.H.; Rezaee, S.; Hamidi, M. Magnetic Brain Targeting of Naproxen-Loaded Polymeric Micelles: Pharmacokinetics and Biodistribution Study. Mater. Sci. Eng. C 2019, 100, 771–780. [Google Scholar] [CrossRef]
- Cong, Y.; Geng, J.; Wang, H.; Su, J.; Arif, M.; Dong, Q.; Chi, Z.; Liu, C. Ureido-Modified Carboxymethyl Chitosan-Graft-Stearic Acid Polymeric Nano-Micelles as a Targeted Delivering Carrier of Clarithromycin for Helicobacter Pylori: Preparation and in Vitro Evaluation. Int. J. Biol. Macromol. 2019, 129, 686–692. [Google Scholar] [CrossRef]
- Li, S.; Zhao, W.; Liang, N.; Xu, Y.; Kawashima, Y.; Sun, S. Multifunctional Micelles Self-Assembled from Hyaluronic Acid Conjugate for Enhancing Anti-Tumor Effect of Paclitaxel. React. Funct. Polym. 2020, 152, 104608. [Google Scholar] [CrossRef]
- Gao, Q.-Q.; Zhang, C.-M.; Zhang, E.-X.; Chen, H.-Y.; Zhen, Y.-H.; Zhang, S.-B.; Zhang, S.-F. Zwitterionic PH-Responsive Hyaluronic Acid Polymer Micelles for Delivery of Doxorubicin. Colloids Surf. B Biointerfaces 2019, 178, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Muddineti, O.S.; Kiran Rompicharla, S.V.; Kumari, P.; Bhatt, H.; Ghosh, B.; Biswas, S. Lipid and Poly (Ethylene Glycol)-Conjugated Bi-Functionalized Chlorine E6 Micelles for NIR-Light Induced Photodynamic Therapy. Photodiagnosis Photodyn. Ther. 2020, 29, 101633. [Google Scholar] [CrossRef]
- Su, M.; Xiao, S.; Shu, M.; Lu, Y.; Zeng, Q.; Xie, J.; Jiang, Z.; Liu, J. Enzymatic Multifunctional Biodegradable Polymers for PH- and ROS-Responsive Anticancer Drug Delivery. Colloids Surf. B Biointerfaces 2020, 193, 111067. [Google Scholar] [CrossRef]
- Huang, Y.; Tang, Z.; Peng, S.; Zhang, J.; Wang, W.; Wang, Q.; Lin, W.; Lin, X.; Zu, X.; Luo, H.; et al. PH/Redox/UV Irradiation Multi-Stimuli Responsive Nanogels from Star Copolymer Micelles and Fe3+ Complexation for “on-Demand” Anticancer Drug Delivery. React. Funct. Polym. 2020, 149, 104532. [Google Scholar] [CrossRef]
- Yang, X.-L.; Li, J.; Wu, W.-X.; Liu, Y.-H.; Wang, N.; Yu, X.-Q. Preparation of Fluorophore-Tagged Polymeric Drug Delivery Vehicles with Multiple Biological Stimuli-Triggered Drug Release. Mater. Sci. Eng. C 2020, 108, 110358. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, J.; Huang, H.; Zhao, Z.; Gunatillake, P.A.; Hao, X. Brush-Shaped RAFT Polymer Micelles as Nanocarriers for a Ruthenium (II) Complex Photodynamic Anticancer Drug. Eur. Polym. J. 2019, 113, 267–275. [Google Scholar] [CrossRef]
- Sang, X.; Yang, Q.; Wen, Q.; Zhang, L.; Ni, C. Preparation and Controlled Drug Release Ability of the Poly[N-Isopropylacryamide-Co-Allyl Poly(Ethylene Glycol)]-b-Poly(γ-Benzyl-l-Glutamate) Polymeric Micelles. Mater. Sci. Eng. C 2019, 98, 910–917. [Google Scholar] [CrossRef]
- Gao, M.; Deng, J.; Liu, F.; Fan, A.; Wang, Y.; Wu, H.; Ding, D.; Kong, D.; Wang, Z.; Peer, D.; et al. Triggered Ferroptotic Polymer Micelles for Reversing Multidrug Resistance to Chemotherapy. Biomaterials 2019, 223, 119486. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Zhang, X.; Xia, X.; Zhang, C. Biodegradable Reduction-Responsive Polymeric Micelles for Enhanced Delivery of Melphalan to Retinoblastoma Cells. Int. J. Biol. Macromol. 2019, 141, 997–1003. [Google Scholar] [CrossRef]
- Yao, Q.; Liu, Y.; Kou, L.; Tu, Y.; Tang, X.; Zhu, L. Tumor-Targeted Drug Delivery and Sensitization by MMP2-Responsive Polymeric Micelles. Nanomed. Nanotechnol. Biol. Med. 2019, 19, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Lamch, Ł.; Kulbacka, J.; Dubińska-Magiera, M.; Saczko, J.; Wilk, K.A. Folate-Directed Zinc (II) Phthalocyanine Loaded Polymeric Micelles Engineered to Generate Reactive Oxygen Species for Efficacious Photodynamic Therapy of Cancer. Photodiagnosis Photodyn. Ther. 2019, 25, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Lan, Y.; Cao, M.; Ma, X.; Cao, A.; Sun, Y.; Yang, J.; Li, L.; Liu, Y. Glycyrrhetinic Acid-Conjugated Polymeric Prodrug Micelles Co-Delivered with Doxorubicin as Combination Therapy Treatment for Liver Cancer. Colloids Surf. B Biointerfaces 2019, 175, 106–115. [Google Scholar] [CrossRef]
- Wan, X.; Beaudoin, J.J.; Vinod, N.; Min, Y.; Makita, N.; Bludau, H.; Jordan, R.; Wang, A.; Sokolsky, M.; Kabanov, A.V. Co-Delivery of Paclitaxel and Cisplatin in Poly(2-Oxazoline) Polymeric Micelles: Implications for Drug Loading, Release, Pharmacokinetics and Outcome of Ovarian and Breast Cancer Treatments. Biomaterials 2019, 192, 1–14. [Google Scholar] [CrossRef]
- Sun, C.; Li, W.; Ma, P.; Li, Y.; Zhu, Y.; Zhang, H.; Adu-Frimpong, M.; Deng, W.; Yu, J.; Xu, X. Development of TPGS/F127/F68 Mixed Polymeric Micelles: Enhanced Oral Bioavailability and Hepatoprotection of Syringic Acid against Carbon Tetrachloride-Induced Hepatotoxicity. Food Chem. Toxicol. 2020, 137, 111126. [Google Scholar] [CrossRef]
- Takashima, H.; Koga, Y.; Tsumura, R.; Yasunaga, M.; Tsuchiya, M.; Inoue, T.; Negishi, E.; Harada, M.; Yoshida, S.; Matsumura, Y. Reinforcement of Antitumor Effect of Micelles Containing Anticancer Drugs by Binding of an Anti-Tissue Factor Antibody without Direct Cytocidal Effects. J. Control. Release 2020, 323, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Bastakoti, B.P.; Bentley, J.; McLaurin, D.; Yusa, S.; Shaji, S.; Mucha, N.R.; Kumar, D.; Ahamad, T. Synthesis of Magnetite Loaded Fluorescence Micelles of Triblock Copolymer. J. Mol. Liq. 2020, 305, 112785. [Google Scholar] [CrossRef]
- Wang, J.; Qian, Y.; Qian, C.; Yao, J.; Bi, X. Paclitaxel-Loaded Cyclodextrin-Cored Unimolecular Micelles and Their in Vivo Behavior. React. Funct. Polym. 2020, 150, 104542. [Google Scholar] [CrossRef]
- Kang, C.; Jung, E.; Hyeon, H.; Seon, S.; Lee, D. Acid-Activatable Polymeric Curcumin Nanoparticles as Therapeutic Agents for Osteoarthritis. Nanomed. Nanotechnol. Biol. Med. 2020, 23, 102104. [Google Scholar] [CrossRef]
- Yang, X.; Chen, X.; Wang, Y.; Xu, G.; Yu, L.; Ding, J. Sustained Release of Lipophilic Gemcitabine from an Injectable Polymeric Hydrogel for Synergistically Enhancing Tumor Chemoradiotherapy. Chem. Eng. J. 2020, 396, 125320. [Google Scholar] [CrossRef]
- Bakardzhiev, P.; Toncheva-Moncheva, N.; Mladenova, K.; Petrova, S.; Videv, P.; Moskova-Doumanova, V.; Topouzova-Hristova, T.; Doumanov, J.; Rangelov, S. Assembly of Amphiphilic Nucleic Acid-Polymer Conjugates into Complex Superaggregates: Preparation, Properties, and in Vitro Performance. Eur. Polym. J. 2020, 131, 109692. [Google Scholar] [CrossRef]
- Chu, L.; Zhang, Y.; Feng, Z.; Yang, J.; Tian, Q.; Yao, X.; Zhao, X.; Tan, H.; Chen, Y. Synthesis and Application of a Series of Amphipathic Chitosan Derivatives and the Corresponding Magnetic Nanoparticle-Embedded Polymeric Micelles. Carbohydr. Polym. 2019, 223, 114966. [Google Scholar] [CrossRef] [PubMed]
- Yan, G.; Huang, Y.; Li, D.; Xu, Y.; Wang, J.; Wang, X.; Tang, R. Sequentially Dynamic Polymeric Micelles with Detachable PEGylation for Enhanced Chemotherapeutic Efficacy. Eur. J. Pharm. Biopharm. 2019, 145, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Mejías, F.J.R.; Gutiérrez, M.T.; Durán, A.G.; Molinillo, J.M.G.; Valdivia, M.M.; Macías, F.A. Provitamin Supramolecular Polymer Micelle with PH Responsiveness to Control Release, Bioavailability Enhancement and Potentiation of Cytotoxic Efficacy. Colloids Surf. B Biointerfaces 2019, 173, 85–93. [Google Scholar] [CrossRef]
- Talelli, M.; Barz, M.; Rijcken, C.J.F.; Kiessling, F.; Hennink, W.E.; Lammers, T. Core-Crosslinked Polymeric Micelles: Principles, Preparation, Biomedical Applications and Clinical Translation. Nano Today 2015, 10, 93–117. [Google Scholar] [CrossRef] [Green Version]
- Kulthe, S.S.; Choudhari, Y.M.; Inamdar, N.N.; Mourya, V. Polymeric Micelles: Authoritative Aspects for Drug Delivery. Des. Monomers Polym. 2012, 15, 465–521. [Google Scholar] [CrossRef]
- Richter, A.; Olbrich, C.; Krause, M.; Hoffmann, J.; Kissel, T. Polymeric Micelles for Parenteral Delivery of Sagopilone: Physicochemical Characterization, Novel Formulation Approaches and Their Toxicity Assessment in Vitro as Well as in Vivo. Eur. J. Pharm. Biopharm. 2010, 75, 80–89. [Google Scholar] [CrossRef]
- Chen, H.; Kim, S.; He, W.; Wang, H.; Low, P.S.; Park, K.; Cheng, J.-X. Fast Release of Lipophilic Agents from Circulating PEG-PDLLA Micelles Revealed by in Vivo Förster Resonance Energy Transfer Imaging. Langmuir 2008, 24, 5213–5217. [Google Scholar] [CrossRef]
- Liu, J.; Zeng, F.; Allen, C. In Vivo Fate of Unimers and Micelles of a Poly(Ethylene Glycol)-Block-Poly(Caprolactone) Copolymer in Mice Following Intravenous Administration. Eur. J. Pharm. Biopharm. 2007, 65, 309–319. [Google Scholar] [CrossRef]
- Zhang, H.; Li, H.; Cao, Z.; Du, J.; Yan, L.; Wang, J. Investigation of the in Vivo Integrity of Polymeric Micelle via Large Stokes Shift Fluorophore-Based FRET. J. Control. Release 2020, 324, 47–54. [Google Scholar] [CrossRef]
- Langridge, T.D.; Gemeinhart, R.A. Toward Understanding Polymer Micelle Stability: Density Ultracentrifugation Offers Insight into Polymer Micelle Stability in Human Fluids. J. Control. Release 2020, 319, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Šimek, M.; Hermannová, M.; Šmejkalová, D.; Foglová, T.; Souček, K.; Binó, L.; Velebný, V. LC–MS/MS Study of in Vivo Fate of Hyaluronan Polymeric Micelles Carrying Doxorubicin. Carbohydr. Polym. 2019, 209, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. Progress in Nanomedicine: Approved and Investigational Nanodrugs. Pharm. Ther. 2017, 42, 742. [Google Scholar]
- Varela-Moreira, A.; Shi, Y.; Fens, M.H.A.M.; Lammers, T.; Hennink, W.E.; Schiffelers, R.M. Clinical Application of Polymeric Micelles for the Treatment of Cancer. Mater. Chem. Front. 2017, 1, 1485–1501. [Google Scholar] [CrossRef]


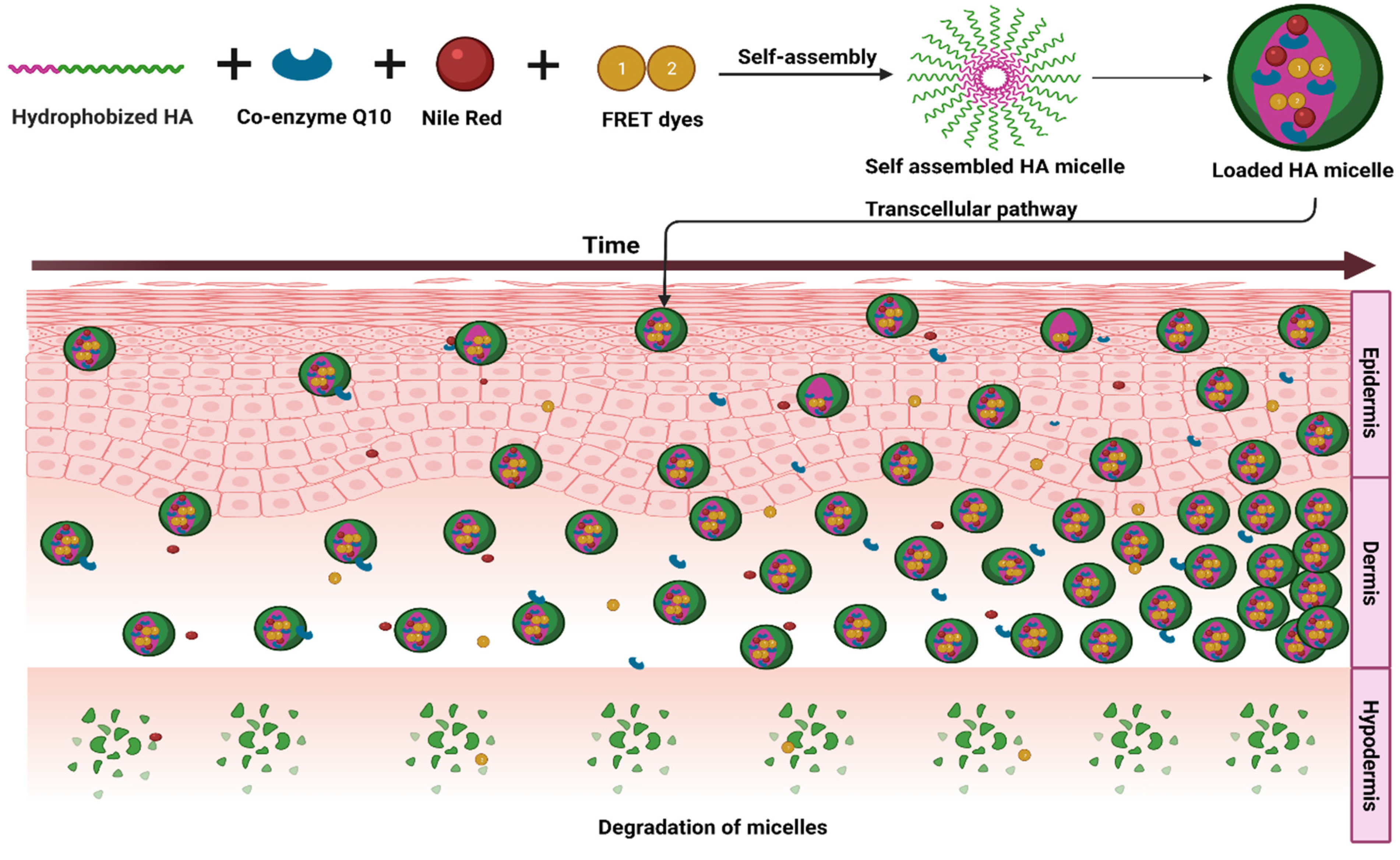
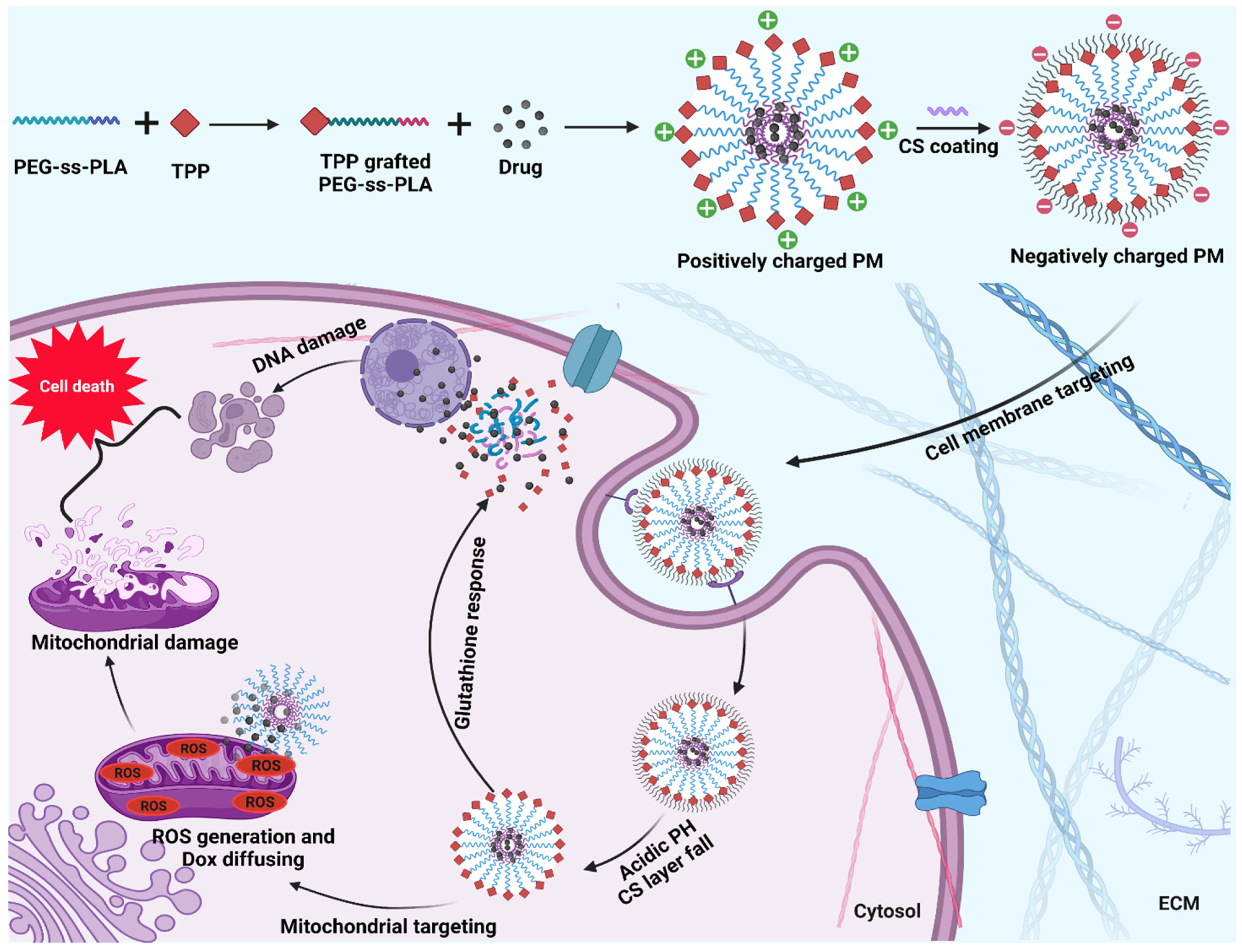
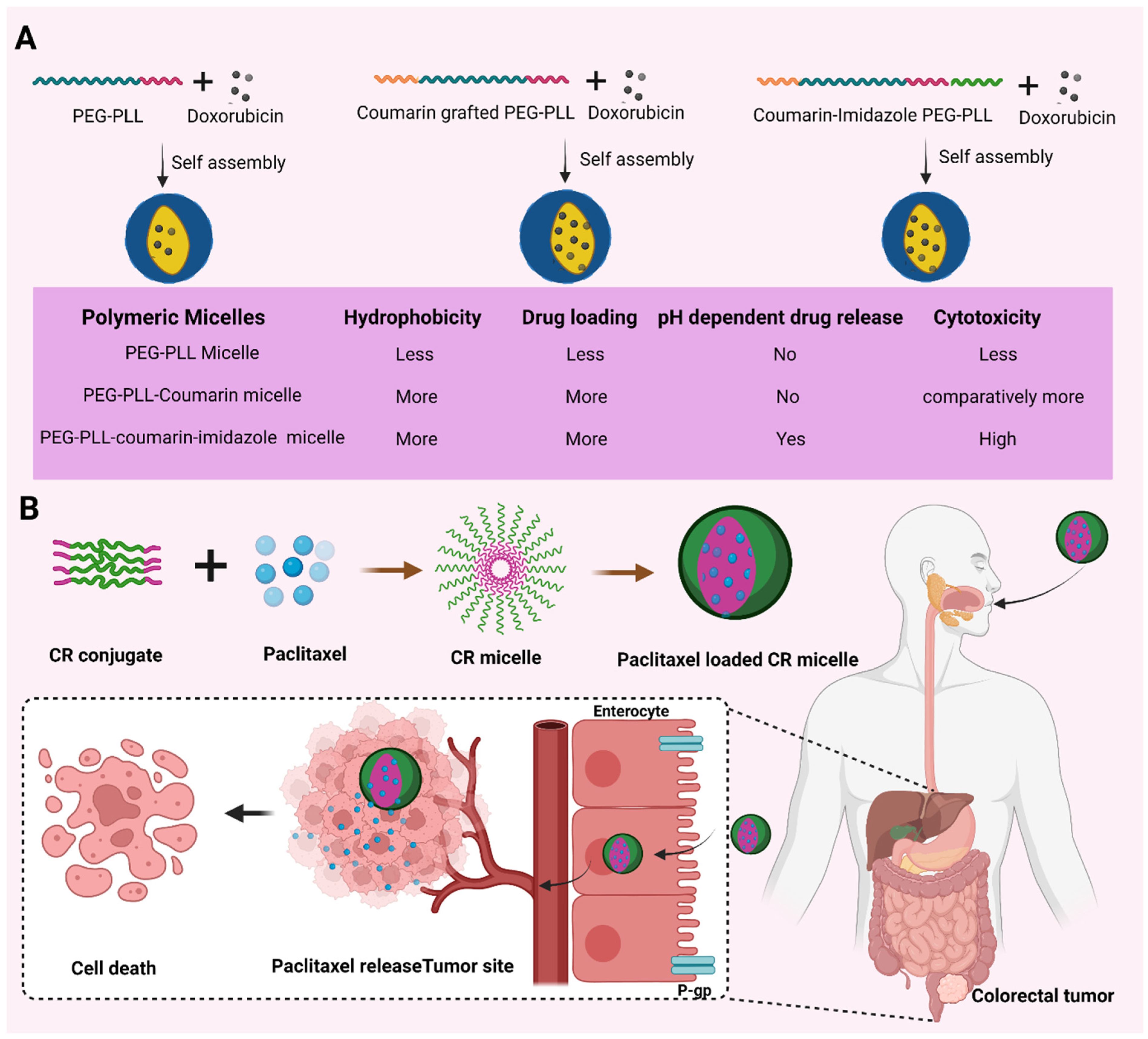

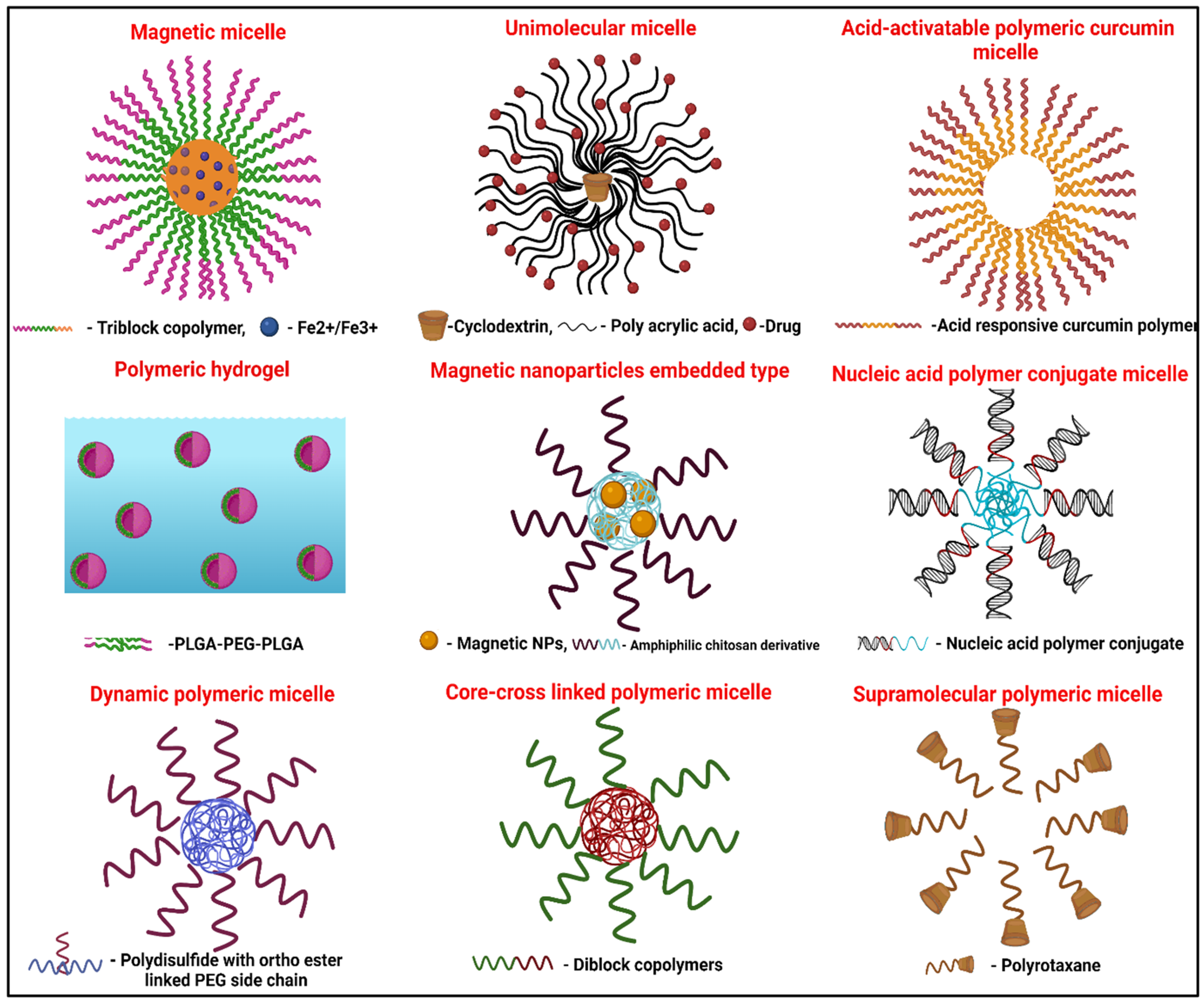
| Sl. No. | Polymer | Method of Preparation | Drug | Reference |
|---|---|---|---|---|
| 1 | Block copolymer of mPEG (5 kDa)-PCL (2 kDa) | Continuous processing | Curcumin | [14] |
| 2 | Poly(ethylene glycol)-b-poly(acrylic acid) | Self-assembly | Dactolisib | [15] |
| 3 | Chitosan-stearic acid | Self-assembly | Doxorubicin | [16] |
| 4 | mPEG-PLGA | Solvent evaporation method | Rotigotine | [17] |
| 5 | disteraroylphosphatidylethanolamine-(PEG) | Solvent evaporation method | Paclitaxel | [18] |
| 6 | Hyaluronan | Solvent evaporation method | Coenzyme Q10 | [19] |
| 7 | Hexyl-hyaluronan and oleyl-hyaluronan | Solvent evaporation method | Curcumin | [20] |
| 8 | Coumarin and imidazole grafted poly(ethylene glycol)-b-poly(l-lysine) | Mixing and dialysis Nanoprecipitation | Doxorubicin | [21] |
| 9 | mPEG poly(ethylene glycol)-block-poly(ε-caprolactone) (PEG-b-PCL) | Thin-film hydration method | Hypocrellin A (photosensitizer) | [22] |
| 10 | Pluronic® F127 | Thin-film hydration technique | ZileutonTM | [23] |
| 11 | Carboxymethyl chitosan-rhein conjugate | Self-assembly | Paclitaxel | [24] |
| 12 | Gallic acid-Chitosan-D-α-tocopherol PEG 1000 succinate | Ultrasonic emulsification | Paclitaxel | [25] |
| 13 | D-α-tocopherol PEG 1000 succinate (TPGS) | Thin-film dispersion | Laquinimod | [26] |
| 14 | Poloxamer | Not mentioned | Doxorubicin | [27] |
| 15 | Functionalized poloxamer | Not mentioned | Doxorubicin | [28] |
| 16 | Methoxypoly(ethylene glycol)-graft-ε-polylysine-tetraphenylethylene | Self-assembly | Doxorubicin | [29] |
| 17 | triphenylphosphonium (TPP) grafted poly(ethylene glycol)(PEG)-poly(D,L-lactide)(PLA) | Solvent evaporation method | Doxorubicin | [30] |
| 18 | Poloxamer | Self-assembly | Mycophenolic acid | [31] |
| 19 | Methoxy poly(ethylene glycol)-b-poly(allyl glycidyl ether)-b-poly(ε-caprolactone) | Solvent evaporation method | Doxorubicin | [32] |
| 20 | PDEA-b-P(ABMA-co-OEGMA) | Sonication | 6-mercaptopurine and doxorubicin | [33] |
| 21 | mPEG-b-P(Deta-NBCF)LG | Self-assembly | Cytochrome C | [34] |
| 22 | Chitosan-lecithin | Sonication | Thymoquinone | [35] |
| 23 | Poly(aspartic acid) derivatives with phenylborate serine side groups | Dialysis | Doxorubicin | [36] |
| 24 | Cholic acid conjugated poly (bis (carboxyphenoxy) phosphazene) | Ultrasonication | Paclitaxel | [37] |
| 25 | Poly-(lactic-co-glycolic)-b-poly (ethylene glycol)-maleimide | Dialysis method | Nilotinib | [38] |
| 26 | Poly (α-azide caprolactone-co-caprolactone)-b-poly (2-methacryloyloxyethyl phosphorylcholine) | Dialysis method | Doxorubicin and 7-Ethyl-10-hydroxycamptothecin | [39] |
| 27 | Poly (ethylene glycol)-block-poly (ε-caprolactone) | Thin-film hydration | SP600125 and Bone Morphogenetic Protein-2 | [40] |
| Sl. No. | Publication No. (Year) | Title | Summary of Invention |
|---|---|---|---|
| 1. | WO/2020/228265 (2020) | “Pharmaceutical compositions containing mixed PMs” | Describes drug-loaded 1–1000 nm-sized mixed PMs. |
| 2. | 111686261 (2020) | “Adriamycin-polymeric micelles-nucleophosmin-binding protein (ADR-PMs-NMBP) with anti-acute lymphoblastic leukemia (ALL) activity and preparation method of ADR-PMs-NMBP” | Describes the preparation method of the ADR-PMs-NMBP. |
| 3. | 111686076 (2020) | “Doxorubicin-loaded polymeric micelle as well as preparation method and application thereof” | Describes the preparation and applications of DOX-loaded PMs. Two amphipathic PMs were described for the purpose. |
| 4. | 201811043353 (2020) | “Improved wound healing topical composition of thymoquinone” | Thymoquinone-loaded PMs are prepared using chitosan and soy lecithin for wound healing. |
| 5. | CN111658783A (2020) | “Switch type glucose-responsive double-layer cross-linked polymer micelle drug delivery system and preparation method and application thereof” | Glucose-responsive double-layered cross-linked PM. |
| 6. | CN111978520A (2020) | “PEG monomethyl ether-polylactic acid segmented copolymer, polymer micelle medicine and preparation method” | Preparation of PMs using PEG monomethyl ether-polylactic acid segmented copolymer for medical application. |
| 7. | CN111978553A (2020) | “Triple-stimulus responsive interfacial crosslinked polymer micelle and preparation method and application thereof” | Triple stimulus-responsive hydrophobic polymer-based PMs. Enhanced drug loading and rapid drug release to stimulus. |
| 8. | CN111330014A (2020) | “Acid-responsive cross-linked polymer prodrug and preparation method and application thereof” | Includes a vinyl alkyl ether acrylate monomer. Provides high drug loading for hydroxyl-group-containing drugs. |
| 9. | CN111888357A (2020) | “Chemotherapy drug-co-loaded crizotinib prodrug polymer micelle and preparation method thereof” | PM is prepared by self-assembling an amphiphilic diblock copolymer and hydrophobic crizotinib. |
| 10. | CN111214438A (2020) | “Method for preparing doxorubicin-loaded polymer micelles with different sizes” | Size controlled doxorubicin-loaded PMs using a micro-reaction system. |
| 11. | United States Patent 10,799,455 (2020) | “Micelles containing alpha-lipoic acid as a transdermal drug delivery system” | PMs with alpha-lipoic acid and vinpocetine for transdermal delivery. |
| 12. | 109966508 (2019) | “pH-sensitive targeting polymeric micelles PPi-Far-PMs, and preparation method and application thereof” | PMs for the release of farnesol to the decayed tooth by targeting the pyrophosphate and acid environment. |
| 13. | WO/2019/129657 (2019) | “Actively targeted polymeric micelles for drug and gene delivery” | PMs prepared of an amphiphilic block copolymer of polyoxyethylene and polyoxypropylene blocks. |
| 14. | CN108524933 (2018) | “Carrier capable of inhibiting tumor multidrug resistance and preparation method thereof” | PMs are prepared by targeting molecule-PEG-pyrene amphiphilic polymer system. |
| Sl. No. | ClinicalTrials.gov Identifier | Details | Phase (Status) |
|---|---|---|---|
| 1. | NCT00912639 | Cremophor EL-free PMs of paclitaxel (Genexol-PM®) for taxane-pretreated recurrent breast cancer | Phase 4 (Not available) |
| 2. | NCT01023347 | A comparative trial between two strategies; Paclitaxel (Genexol®) and cisplatin vs. Genexol-PM® and cisplatin, in non-small cell lung cancer | Phase 2 (Completed) |
| 3. | NCT00886717 | Paclitaxel-loaded PMs in advanced ovarian cancer | Phase 2 (Not available) |
| 4. | NCT03585673 | Docetaxel-loaded PMs in esophagus squamous cell carcinoma | Phase 2 (Recruiting) |
| 5. | NCT01426126 | Genexol-PM® in advanced urothelial cancer earlier treated with gemcitabine and platinum | Phase 2 (Completed) |
| 6. | NCT02639858 | Docetaxel-loaded PMs in recurrent or metastatic head and neck squamous cell carcinoma | Phase 2 (Not available) |
| 7. | NCT00111904 | Paclitaxel-loaded PMs in unresectable locally advanced or metastatic pancreatic cancer | Phase 2 (Completed) |
| 8. | NCT01770795 | Genexol-PM® in untreated metastatic non-small cell lung cancer patients | Phase 2 (Completed) |
| 9. | NCT02739529 | Genexol-PM® in gynecologic cancer | Phase 1 (Not available) |
| 10. | NCT00882973 | Genexol-PM® in advanced pancreatic cancer | Phase 1 (Completed) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotta, S.; Aldawsari, H.M.; Badr-Eldin, S.M.; Nair, A.B.; YT, K. Progress in Polymeric Micelles for Drug Delivery Applications. Pharmaceutics 2022, 14, 1636. https://doi.org/10.3390/pharmaceutics14081636
Kotta S, Aldawsari HM, Badr-Eldin SM, Nair AB, YT K. Progress in Polymeric Micelles for Drug Delivery Applications. Pharmaceutics. 2022; 14(8):1636. https://doi.org/10.3390/pharmaceutics14081636
Chicago/Turabian StyleKotta, Sabna, Hibah Mubarak Aldawsari, Shaimaa M. Badr-Eldin, Anroop B. Nair, and Kamal YT. 2022. "Progress in Polymeric Micelles for Drug Delivery Applications" Pharmaceutics 14, no. 8: 1636. https://doi.org/10.3390/pharmaceutics14081636
APA StyleKotta, S., Aldawsari, H. M., Badr-Eldin, S. M., Nair, A. B., & YT, K. (2022). Progress in Polymeric Micelles for Drug Delivery Applications. Pharmaceutics, 14(8), 1636. https://doi.org/10.3390/pharmaceutics14081636






