Drug–Drug Interactions with Oral Anticoagulants as Potentially Inappropriate Medications: Prevalence and Outcomes in Elderly Patients in Primary Care and Hospital Settings
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Study Population and Follow-Up
2.3. Exposure Assessment
2.3.1. Oral Anticoagulant Exposure
- -
- Apixaban: Patient ≥ 65 years old with renal impairment
- -
- Dabigatran: Patient ≥ 75 years old or patient ≥ 65 years old with renal impairment
- -
- Rivaroxaban: Patient ≥ 75 years old or patient ≥ 65 years old with renal impairment
- -
- Warfarin: Patient ≥ 65 years old with the following drug combinations:
- o
- Warfarin–Amiodarone,
- o
- Warfarin–Sulfamethoxazole/Trimethoprim,
- o
- Warfarin–Ciprofloxacin,
- o
- Warfarin–Macrolides (excepted Azithromycin)
- o
- Warfarin–Non-steroidal anti-inflammatory drugs (NSAIDs)
2.3.2. DDI and PIM–DDI Exposure
2.4. Detection of Patients with Bleeding ADE
2.5. Descriptive Analyses
2.6. Exploratory Analyses
3. Results
3.1. Descriptive Analyses
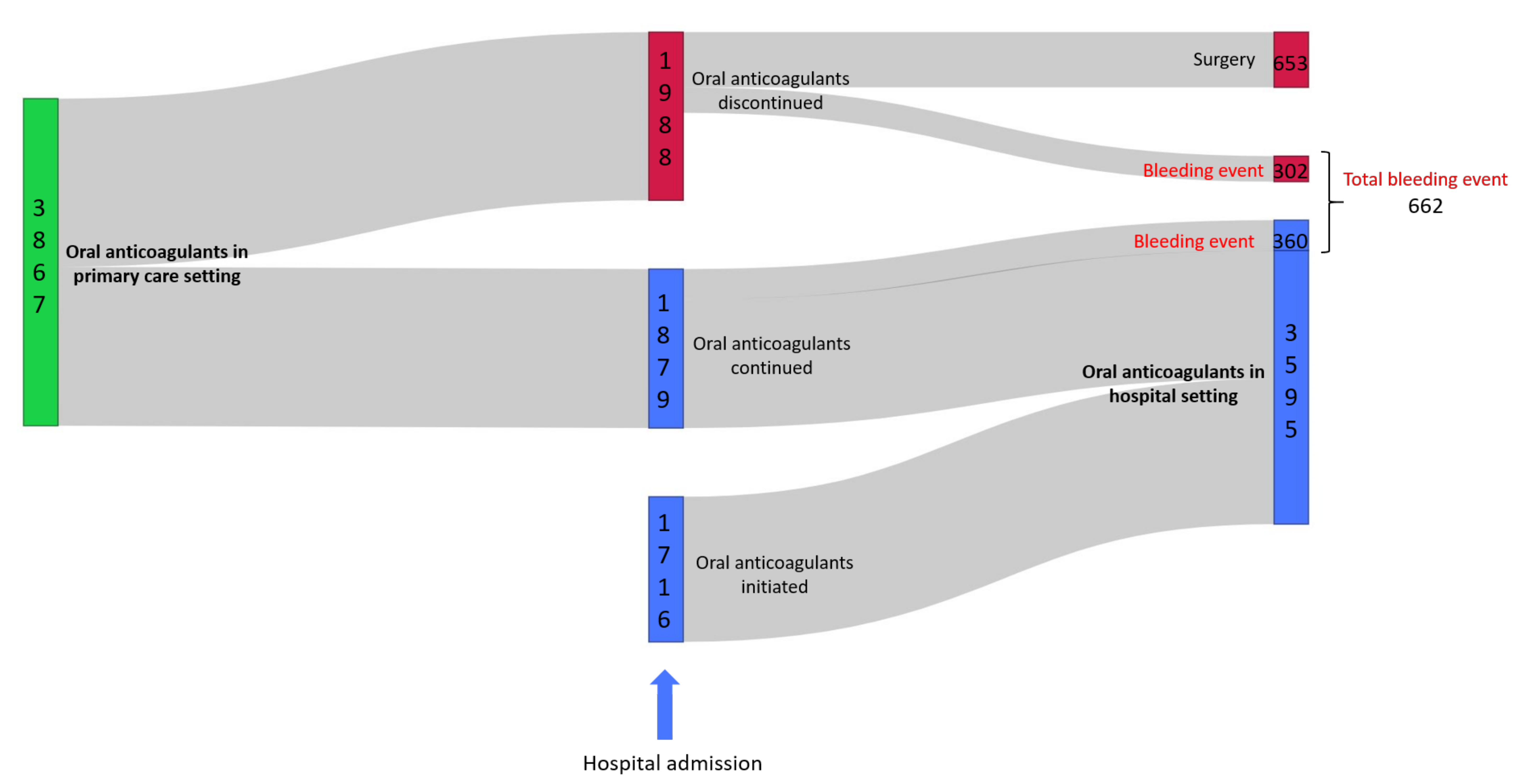
3.1.1. General Characteristics and Patient Drug Pathway
3.1.2. Prevalence of PIM, DDI and of PIM–DDI
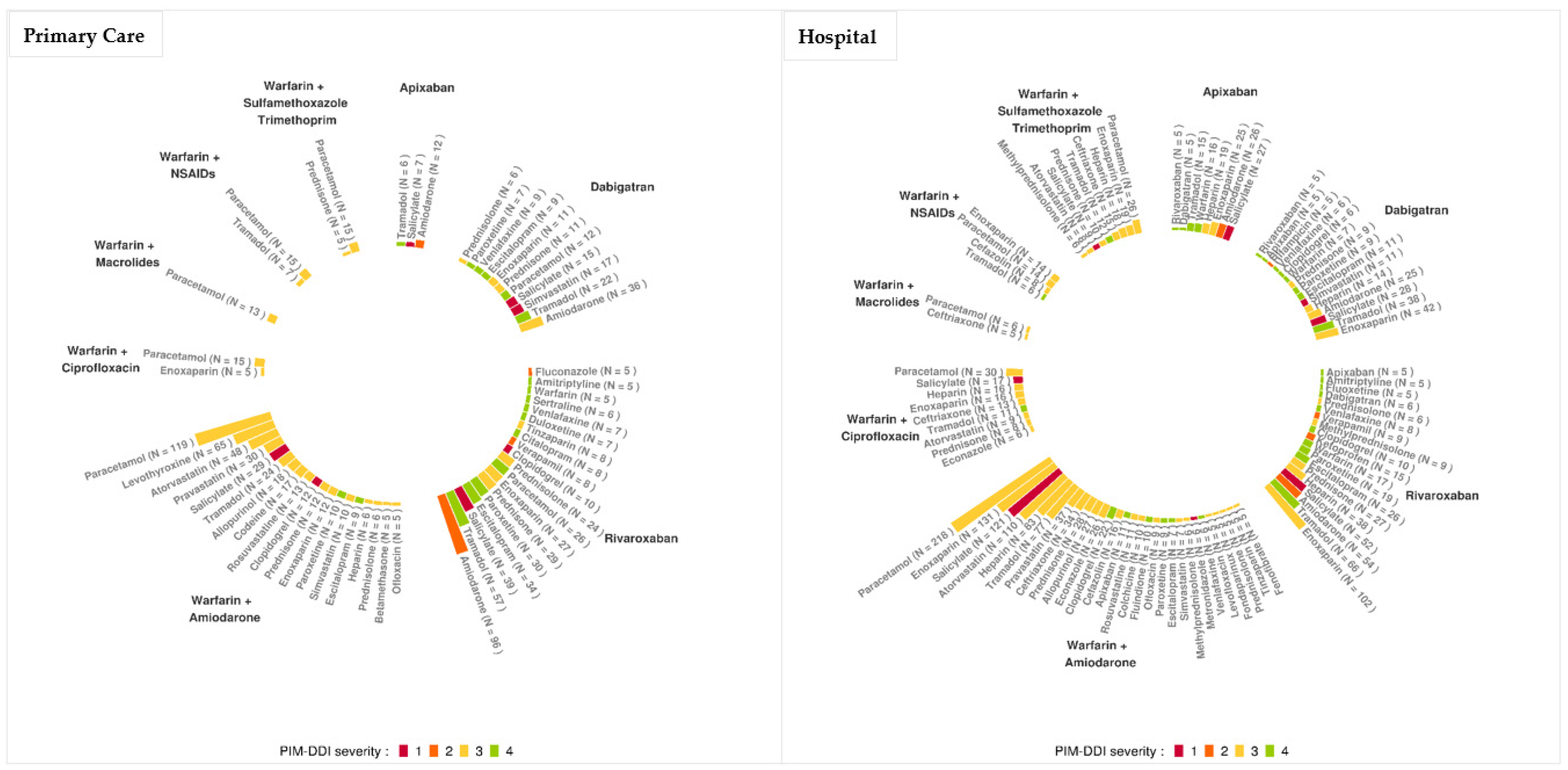
3.1.3. Mechanisms of DDI and of PIM–DDI
3.1.4. Bleeding ADE at Admission
3.2. Exploratory Analyses
3.2.1. Logistic Regression
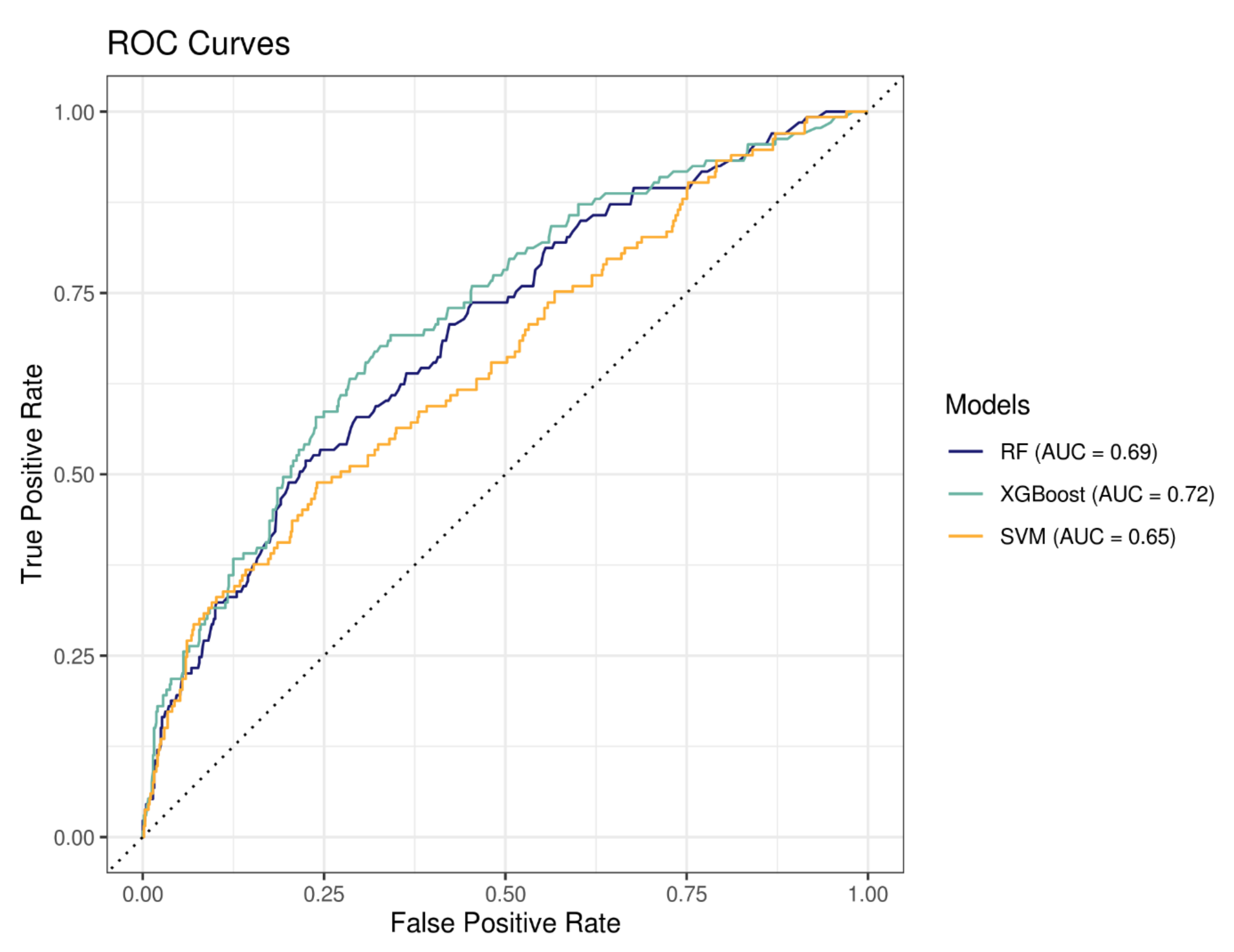
3.2.2. Machine Learning to Predict Hospitalization for Bleeding Event
4. Discussion
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swart, F.; Bianchi, G.; Lenzi, J.; Iommi, M.; Maestri, L.; Raschi, E.; Zoli, M.; De Ponti, F.; Poluzzi, E. Risk of hospitalization from drug–drug interactions in the Elderly: Real-world evidence in a large administrative database. Aging 2020, 12, 19711–19739. [Google Scholar] [CrossRef] [PubMed]
- Dechanont, S.; Maphanta, S.; Butthum, B.; Kongkaew, C. Hospital admissions/visits associated with drug–drug interactions: A systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2014, 23, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.; Kallewaard, M.; Caspers, P.W.J.; Visser, L.E.; Leufkens, H.G.M.; Stricker, B.H. Hospitalisations and emergency department visits due to drug–drug interactions: A literature review. Pharmacoepidemiol. Drug Saf. 2006, 16, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Bénard-Laribière, A.; Miremont-Salamé, G.; Pérault-Pochat, M.-C.; Noize, P.; Haramburu, F.; The EMIR Study Group on behalf of the French network of pharmacovigilance centres. Incidence of hospital admissions due to adverse drug reactions in France: The EMIR study. Fundam. Clin. Pharmacol. 2014, 29, 106–111. [Google Scholar] [CrossRef]
- Zerah, L.; Henrard, S.; Wilting, I.; O’Mahony, D.; Rodondi, N.; Dalleur, O.; Dalton, K.; Knol, W.; Haschke, M.; Spinewine, A. Prevalence of drug–drug interactions in older people before and after hospital admission: Analysis from the OPERAM trial. BMC Geriatr. 2021, 21, 1–11. [Google Scholar] [CrossRef]
- Hines, L.E.; Murphy, J.E. Potentially Harmful Drug–Drug Interactions in the Elderly: A Review. Am. J. Geriatr. Pharmacother. 2011, 9, 364–377. [Google Scholar] [CrossRef]
- Magro, L.; Moretti, U.; Leone, R. Epidemiology and characteristics of adverse drug reactions caused by drug–drug interactions. Expert Opin. Drug Saf. 2011, 11, 83–94. [Google Scholar] [CrossRef]
- Oscanoa, T.; Lizaraso, F.; Carvajal, A. Hospital admissions due to adverse drug reactions in the elderly. A meta-analysis. Eur. J. Clin. Pharmacol. 2017, 73, 759–770. [Google Scholar] [CrossRef]
- Alhawassi, T.M.; Krass, I.; Bajorek, B.V.; Pont, L.G. A systematic review of the prevalence and risk factors for adverse drug reactions in the elderly in the acute care setting. Clin. Interv. Aging 2014, 9, 2079–2086. [Google Scholar] [CrossRef] [Green Version]
- By the 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 Updated AGS Beers Criteria® for Potentially Inappropriate Medication Use in Older Adults. J. Am. Geriatr. Soc. 2019, 67, 674–694. [Google Scholar] [CrossRef]
- Bories, M.; Bouzillé, G.; Cuggia, M.; Le Corre, P. Drug–drug Interactions in Elderly Patients with Potentially Inappropriate Medications in Primary Care, Nursing Home and Hospital Settings: A Systematic Review and a Preliminary Study. Pharmaceutics 2021, 13, 266. [Google Scholar] [CrossRef]
- Lee, J.Y.; Oh, I.-Y.; Lee, J.-H.; Kim, S.-Y.; Kwon, S.S.; Yang, H.-J.; Kim, Y.-K.; Bang, S.-M. The increased risk of bleeding due to drug–drug interactions in patients administered direct oral anticoagulants. Thromb. Res. 2020, 195, 243–249. [Google Scholar] [CrossRef]
- Hollowell, J.; Meier, C.R.; Haefeli, W.E.; Gasse, C. Drug interactions and risk of acute bleeding leading to hospitalisation or death in patients with chronic atrial fibrillation treated with warfarin. Thromb. Haemost. 2005, 94, 537–543. [Google Scholar] [CrossRef] [Green Version]
- Nair, N.P.; Chalmers, L.; Peterson, G.M.; Bereznicki, B.J.; Castelino, R.L.; Bereznicki, L.R. Hospitalization in older patients due to adverse drug reactions—The need for a prediction tool. Clin. Interv. Aging. 2016, 11, 497–505. [Google Scholar] [CrossRef] [Green Version]
- Bannay, A.; Bories, M.; Le Corre, P.; Riou, C.; Lemordant, P.; Van Hille, P.; Chazard, E.; Dode, X.; Cuggia, M.; Bouzillé, G. Leveraging National Claims and Hospital Big Data: Cohort Study on a Statin-Drug Interaction Use Case. JMIR Med. Inform. 2021, 9, e29286. [Google Scholar] [CrossRef]
- Bezin, J.; Duong, M.; Lassalle, R.; Droz, C.; Pariente, A.; Blin, P.; Moore, N. The national healthcare system claims databases in France, SNIIRAM and EGB: Powerful tools for pharmacoepidemiology. Pharmacoepidemiol. Drug Saf. 2017, 26, 954–962. [Google Scholar] [CrossRef]
- Madec, J.; Bouzillé, G.; Riou, C.; Van Hille, P.; Merour, C.; Artigny, M.-L.; Delamarre, D.; Raimbert, V.; Lemordant, P.; Cuggia, M. eHOP Clinical Data Warehouse: From a Prototype to the Creation of an Inter-Regional Clinical Data Centers Network. Stud. Health Technol. Inform. 2019, 264, 1536–1537. [Google Scholar]
- Husson, M.-C. Theriaque: Independent-drug database for good use of drugs by health practitioners. Ann. Pharm. Fr. 2008, 66, 268–277. [Google Scholar] [CrossRef]
- Micromedex. Available online: https://www.micromedexsolutions.com/home/dispatch/ (accessed on 4 April 2022).
- Kuhn, M.; Letunic, I.; Jensen, L.J.; Bork, P. The SIDER database of drugs and side effects. Nucleic Acids Res. 2015, 44, D1075–D1079. [Google Scholar] [CrossRef]
- Franco, L.; Becattini, C.; Beyer-Westendorf, J.; Vanni, S.; Nitti, C.; Re, R.; Manina, G.; Pomero, F.; Cappelli, R.; Conti, A.; et al. Definition of major bleeding: Prognostic classification. J. Thromb. Haemost. 2020, 18, 2852–2860. [Google Scholar] [CrossRef]
- Zhu, W.; He, W.; Guo, L.; Wang, X.; Hong, K. The HAS-BLED Score for Predicting Major Bleeding Risk in Anticoagulated Patients With Atrial Fibrillation: A Systematic Review and Meta-analysis. Clin. Cardiol. 2015, 38, 555–561. [Google Scholar] [CrossRef]
- Fang, M.C.; Go, A.S.; Chang, Y.; Borowsky, L.H.; Pomernacki, N.K.; Udaltsova, N.; Singer, D.E. A New Risk Scheme to Predict Warfarin-Associated Hemorrhage: The ATRIA (Anticoagulation and Risk Factors in Atrial Fibrillation) Study. J. Am. Coll. Cardiol. 2011, 58, 395–401. [Google Scholar] [CrossRef] [Green Version]
- Tidymodels. Available online: https://www.tidymodels.org (accessed on 5 March 2022).
- Wu, C.-Y.; Benet, L.Z. Predicting Drug Disposition via Application of BCS: Transport/Absorption/Elimination Interplay and Development of a Biopharmaceutics Drug Disposition Classification System. Pharm. Res. 2005, 22, 11–23. [Google Scholar] [CrossRef]
- Gronich, N.; Stein, N.; Muszkat, M. Association Between Use of Pharmacokinetic-Interacting Drugs and Effec-tiveness and Safety of Direct Acting Oral Anticoagulants: Nested Case-Control Study. Clin. Pharmacol. Ther. 2021, 110, 1526–1536. [Google Scholar] [CrossRef]
- Li, A.; Li, M.K.; Crowther, M.; Vazquez, S.R. Drug–drug interactions with direct oral anticoagulants associated with adverse events in the real world: A systematic review. Thromb. Res. 2020, 194, 240–245. [Google Scholar] [CrossRef]
- Lam, J.; Gomes, T.; Juurlink, D.N.; Mamdani, M.M.; Pullenayegum, E.M.; Kearon, C.; Spencer, F.A.; Paterson, M.; Zheng, H.; Holbrook, A.M. Hospitalization for hemor-rhage among warfarin recipients prescribed amiodarone. Am. J. Cardiol. 2013, 112, 420–423. [Google Scholar] [CrossRef] [Green Version]
- Miano, T.A.; Yang, W.; Shashaty, M.G.S.; Zuppa, A.; Brown, J.R.; Hennessy, S. The Magnitude of the Warfa-rin-Amiodarone Drug–drug Interaction Varies With Renal Function: A Propensity-Matched Cohort Study. Clin. Pharmacol. Ther. 2020, 107, 1446–1456. [Google Scholar] [CrossRef]
- Fohtung, R.B.; Novak, E.; Rich, M.W. Effect of New Oral Anticoagulants on Prescribing Practices for Atrial Fibrillation in Older Adults. J. Am. Geriatr. Soc. 2017, 65, 2405–2412. [Google Scholar] [CrossRef]
- Kim, I.-S.; Kim, H.-J.; Kim, T.-H.; Uhm, J.-S.; Joung, B.; Lee, M.-H.; Pak, H.-N. Non-vitamin K antagonist oral anticoagulants have better efficacy and equivalent safety compared to warfarin in elderly patients with atrial fibrillation: A systematic review and meta-analysis. J. Cardiol. 2018, 72, 105–112. [Google Scholar] [CrossRef] [Green Version]
- Gage, B.F.; Yan, Y.; Milligan, P.E.; Waterman, A.D.; Culverhouse, R.; Rich, M.W.; Radford, M.J. Clinical classification schemes for predicting hemorrhage: Results from the National Registry of Atrial Fibrillation (NRAF). Am. Heart J. 2006, 151, 713–719. [Google Scholar] [CrossRef]
- Claxton, J.S.; MacLehose, R.F.; Lutsey, P.L.; Norby, F.L.; Chen, L.; O’Neal, W.T.; Chamberlain, A.M.; Bengtson, L.G.S.; Alonso, A. A new model to predict major bleeding in patients with atrial fibrillation using warfarin or direct oral anticoagulants. PLoS ONE 2018, 13, e0203599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.-C.; Cheng, Y.-C.; Jhou, M.-J.; Chen, M.; Lu, C.-J. Important Risk Factors in Patients with Nonvalvular Atrial Fibrillation Taking Dabigatran Using Integrated Machine Learning Scheme—A Post Hoc Analysis. J. Pers. Med. 2022, 12, 756. [Google Scholar] [CrossRef] [PubMed]
- Le Corre, P.A. Prescriptome analytics: An opportunity for clinical pharmacy. Int. J. Clin. Pharm. 2019, 41, 1394–1397. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, M.; O’Mahony, D.; Cherubini, A. Inappropriate prescribing: Hazards and solutions. Age Ageing 2022, 51, afab269. [Google Scholar] [CrossRef]
- Vazquez, S.R.; Barnes, G.D. Anticoagulant drug–drug interactions: Highlighting the need for antithrombotic stewardship and shared decision making. Res. Pract. Thromb. Haemost. 2022, 6, e12662. [Google Scholar] [CrossRef]

| Oral Anticoagulant in Primary Care Setting | Oral Anticoagulant in Hospital Setting | |
|---|---|---|
| Total | n = 3867 | n = 3595 |
| Sex | ||
| Men | 57.8% (2236) | 52.6% (1890) |
| Women | 42.2% (1631) | 47.4% (1705) |
| Age (median-IQR) | 79 (73–85) | 80 (73–86) |
| 65–75 years old | 29.5% (1141) | 30.4% (1092) |
| ≥75 years old | 70.5% (2726) | 69.6% (2503) |
| Hospital stay | ||
| Medicine | 63.6% (2458) | 62.0% (2227) |
| Surgery | 36.4% (1409) | 38.0% (1368) |
| Time frame exposure to oral anticoagulants prior to hospitalization | ||
| ≤1 month | 6.5% (253) | - |
| 1 to ≤3 months | 9.3% (361) | - |
| 3 to ≤6 months | 8.2% (318) | - |
| >6 months | 75.9% (2935) | - |
| Time frame exposure to oral anticoagulants during hospitalization | ||
| ≤1 day | - | 31.8% (1144) |
| 1 to ≤8 days | - | 49.0% (1763) |
| 8 to ≤15 days | - | 12.5% (451) |
| >15 days | - | 6.6% (237) |
| Oral Anticoagulant in Primary Care Setting | Oral Anticoagulant in Hospital Setting | |
|---|---|---|
| Total | n= 3867 | n= 3595 |
| Anticoagulants | ||
| Apixaban | 18.3% (706) | 24.8% (890) |
| Dabigatran | 11.6% (450) | 7.3% (264) |
| Rivaroxaban | 23.9% (923) | 19.2% (690) |
| Warfarine | 46.6% (1801) | 50.8% (1826) |
| PIM | 22.9% (886) | 20.9% (750) |
| Apixaban | 0.8% (33) | 0.9% (31) |
| Dabigatran | 4.5% (175) | 3.0% (108) |
| Rivaroxaban | 9.7% (374) | 5.5% (199) |
| Warfarin | 7.9% (304) | 11.6% (417) |
| DDI | 47.2% (1825) | 58.9% (2117) |
| Apixaban | 8.5% (329) | 16.5% (593) |
| Dabigatran | 2.4% (94) | 1.5% (55) |
| Rivaroxaban | 3.6% (140) | 6.6% (239) |
| Warfarin | 32.7% (1266) | 34.9% (1254) |
| Average number of DDI per patient | 1.5 +/− 1.8 | 1.6 +/− 1.8 |
| Contraindicated DDI (level 1) | 8.6% (243) | 13.8% (607) |
| Not recommended DDI (level 2) | 15.9% (450) | 19.9%(871) |
| PIM–DDI | 19.5% (753) | 23.5% (847) |
| Apixaban | 0.9% (34) | 1.9% (71) |
| Dabigatran | 3.6% (138) | 3.6% (130) |
| Rivaroxaban | 8.0% (310) | 7.6% (274) |
| Warfarine | 7.2% (280) | 11.8% (423) |
| Average number of PIM–DDI per patient | 0.8 +/− 1.9 | 0.8 +/− 2.0 |
| Contraindicated PIM–DDI (level 1) | 9.8% (116) | 14.9% (266) |
| Not recommended PIM–DDI (level 2) | 20.0% (236) | 19.2% (344) |
| A | Primary Care Prevalence of DDI (n) | Hospital Prevalence of DDI (n) |
|---|---|---|
| Pharmacodynamics | 38.7% (507) | 59.8% (1489) |
| Pharmacokinetics | 61.3% (803) | 40.2% (1001) |
| ● Inhibition of CYP2C9 and CYP2C19 | 32.0% (257) | 25.5% (255) |
| ● Inhibition of CYP3A4 | 0.9% (7) | 0.6% (6) |
| ● Inhibition of P-glycoprotein | 5.4% (43) | 1.9% (19) |
| ● Combined inhibition CYP3A4 and P-glycoprotein | 24.5% (197) | 22.3% (223) |
| B | Primary care Prevalence of PIM–DDI (n) | Hospital Prevalence of PIM–DDI (n) |
| Pharmacodynamics | 28.9% (220) | 54.9% (588) |
| Pharmacokinetics | 71.1% (541) | 45.1% (483) |
| ● Inhibition of CYP2C9 and CYP2C19 | 26.1% (141) | 23.0% (111) |
| ● Inhibition of CYP3A4 | 1.5% (8) | 1.4% (7) |
| ● Inhibition of P-glycoprotein | 10.0% (54) | 7.9% (38) |
| ● Combined inhibition CYP3A4 and P-glycoprotein | 21.4% (116) | 18.2% (88) |
| Association | % of Patient with the Drug Combination and Bleeding ADE (n) | Level of Severity | Type of Association |
|---|---|---|---|
| Rivaroxaban–Salicylate | 28.2% (11) | 1 | PIM–DDI |
| Warfarin–Salicylate | 25.8% (33) | 1 | DDI |
| Rivaroxaban–Amiodarone | 18.8% (18) | 2 | PIM–DDI |
| Warfarin–Amiodarone–Paracetamol | 23.5% (28) | 3 | PIM–DDI |
| Warfarin–Paracetamol | 22.6% (176) | 3 | DDI |
| Warfarin–Amiodarone–Atorvastatin | 20.8% (10) | 3 | PIM–DDI |
| Warfarin–Atorvastatin | 19.5% (42) | 3 | DDI |
| Warfarin–Levothyroxin | 17.0% (26) | 3 | DDI |
| Warfarin–Tramadol | 28.1% (34) | 4 | DDI |
| Rivaroxaban–Tramadol | 22.8% (13) | 4 | PIM–DDI |
| Characteristic | OR | 95% CI | p-Value |
|---|---|---|---|
| Sex | |||
| F | - | - | - |
| M | 1.19 | 1.00–1.43 | 0.053 |
| Age | 1.03 | 1.02–1.04 | <0.001 |
| History of stroke | 1.17 | 0.64–2.03 | 0.60 |
| Previous bleeding event | 4.23 | 3.00–5.94 | <0.001 |
| Diabetes | 1.29 | 0.99–1.66 | 0.058 |
| Renal disease | 1.98 | 1.48–2.64 | <0.001 |
| Liver disease | 1.72 | 0.90–3.14 | 0.085 |
| Cancer | 2.68 | 1.56–4.50 | <0.001 |
| Hypertension | 1.72 | 1.43–2.06 | <0.001 |
| PIM | |||
| 0 | - | - | - |
| 1 | 1.08 | 0.85–1.38 | 0.500 |
| DDI | |||
| 0 | - | - | - |
| 1 | 1.15 | 0.92–1.45 | 0.200 |
| PIM–DDI | |||
| 0 | - | - | - |
| 1 | 1.23 | 1.00–1.57 | 0.060 |
| RF | XGBoost | SVM | |
|---|---|---|---|
| Accuracy | 0.64 | 0.68 | 0.64 |
| Sensitivity | 0.65 | 0.70 | 0.56 |
| Specificity | 0.64 | 0.68 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bories, M.; Bouzillé, G.; Cuggia, M.; Le Corre, P. Drug–Drug Interactions with Oral Anticoagulants as Potentially Inappropriate Medications: Prevalence and Outcomes in Elderly Patients in Primary Care and Hospital Settings. Pharmaceutics 2022, 14, 1410. https://doi.org/10.3390/pharmaceutics14071410
Bories M, Bouzillé G, Cuggia M, Le Corre P. Drug–Drug Interactions with Oral Anticoagulants as Potentially Inappropriate Medications: Prevalence and Outcomes in Elderly Patients in Primary Care and Hospital Settings. Pharmaceutics. 2022; 14(7):1410. https://doi.org/10.3390/pharmaceutics14071410
Chicago/Turabian StyleBories, Mathilde, Guillaume Bouzillé, Marc Cuggia, and Pascal Le Corre. 2022. "Drug–Drug Interactions with Oral Anticoagulants as Potentially Inappropriate Medications: Prevalence and Outcomes in Elderly Patients in Primary Care and Hospital Settings" Pharmaceutics 14, no. 7: 1410. https://doi.org/10.3390/pharmaceutics14071410
APA StyleBories, M., Bouzillé, G., Cuggia, M., & Le Corre, P. (2022). Drug–Drug Interactions with Oral Anticoagulants as Potentially Inappropriate Medications: Prevalence and Outcomes in Elderly Patients in Primary Care and Hospital Settings. Pharmaceutics, 14(7), 1410. https://doi.org/10.3390/pharmaceutics14071410







