Combination of [177Lu]Lu-DOTA-TATE Targeted Radionuclide Therapy and Photothermal Therapy as a Promising Approach for Cancer Treatment: In Vivo Studies in a Human Xenograft Mouse Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Gold Nanoshells
2.2. Flow Cytometry
2.3. Animal Model
2.4. Histology
2.5. [64Cu]Cu-DOTA-TATE PET/CT
2.6. Autoradiography
2.7. Radiolabeling of [177Lu]Lu-DOTA-TATE
2.8. SPECT/CT
2.9. Photothermal Treatment
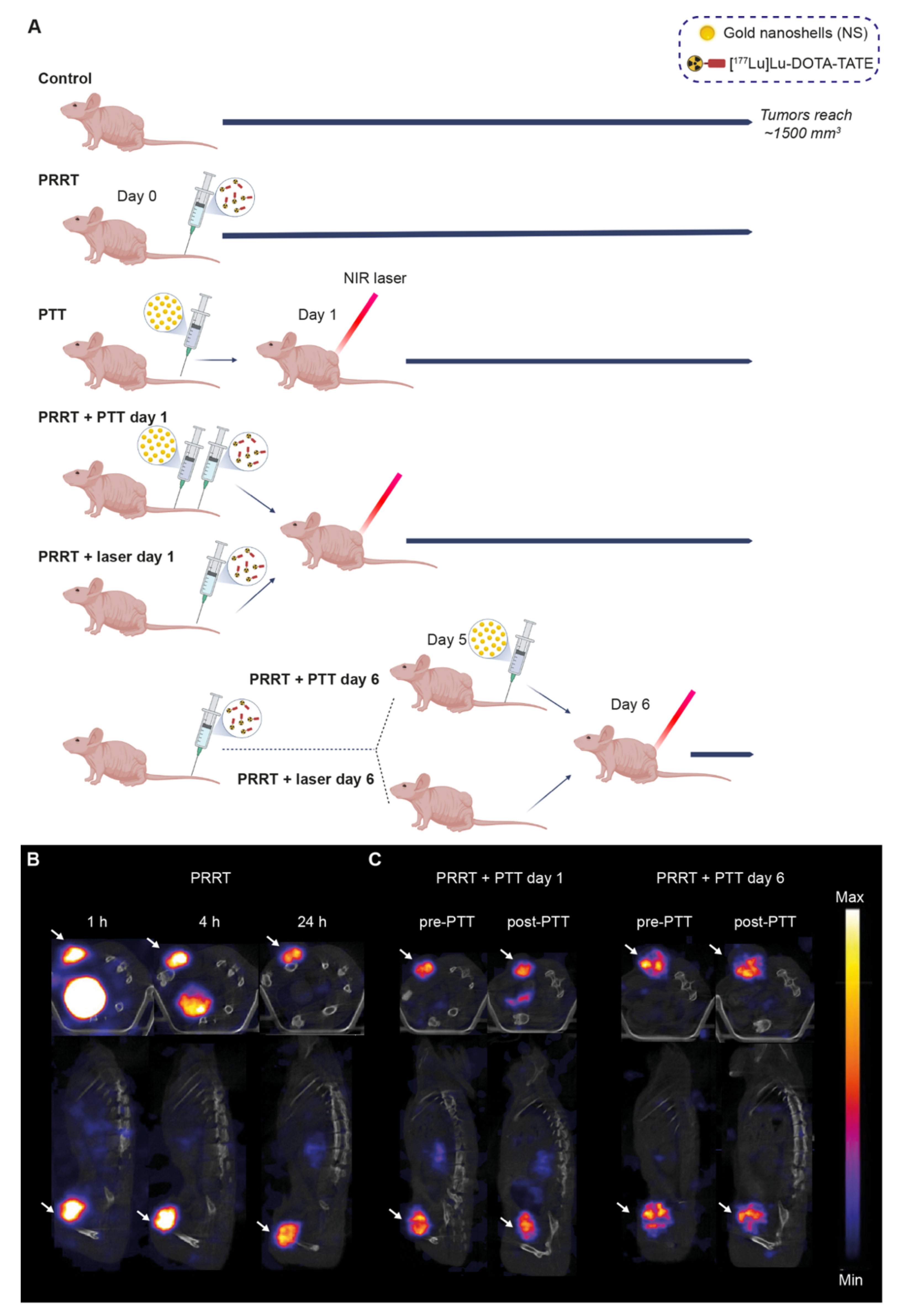
2.10. Combination Treatment
2.11. Blood Analysis
2.12. Statistical Analysis
3. Results
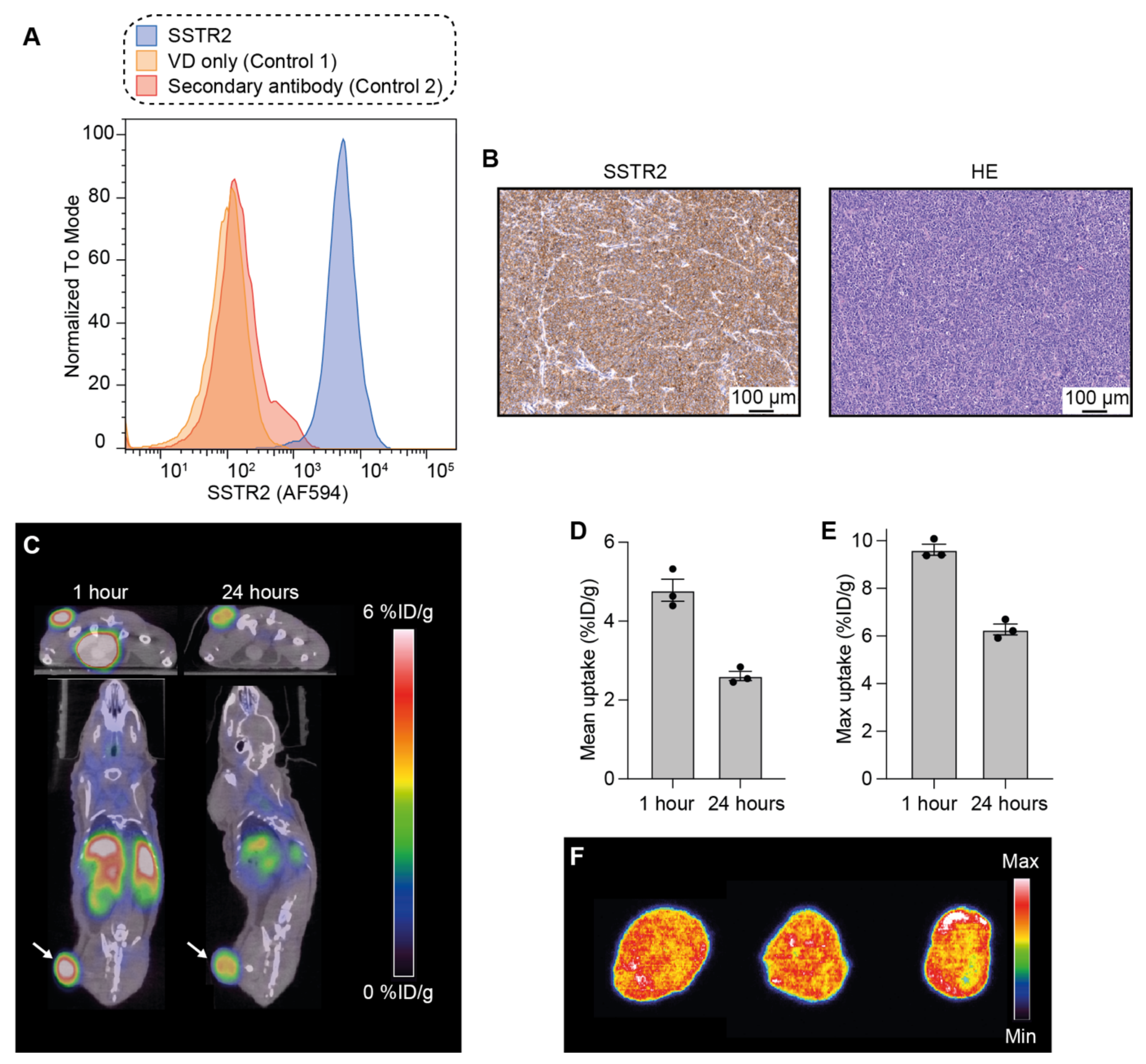
3.1. Analysis of SSTR2 Expression in NCI-H69 Tumor Cells
3.2. Uptake and Intratumoral Distribution of [64Cu]Cu-DOTA-TATE in Mice Bearing NCI-H69 Tumors
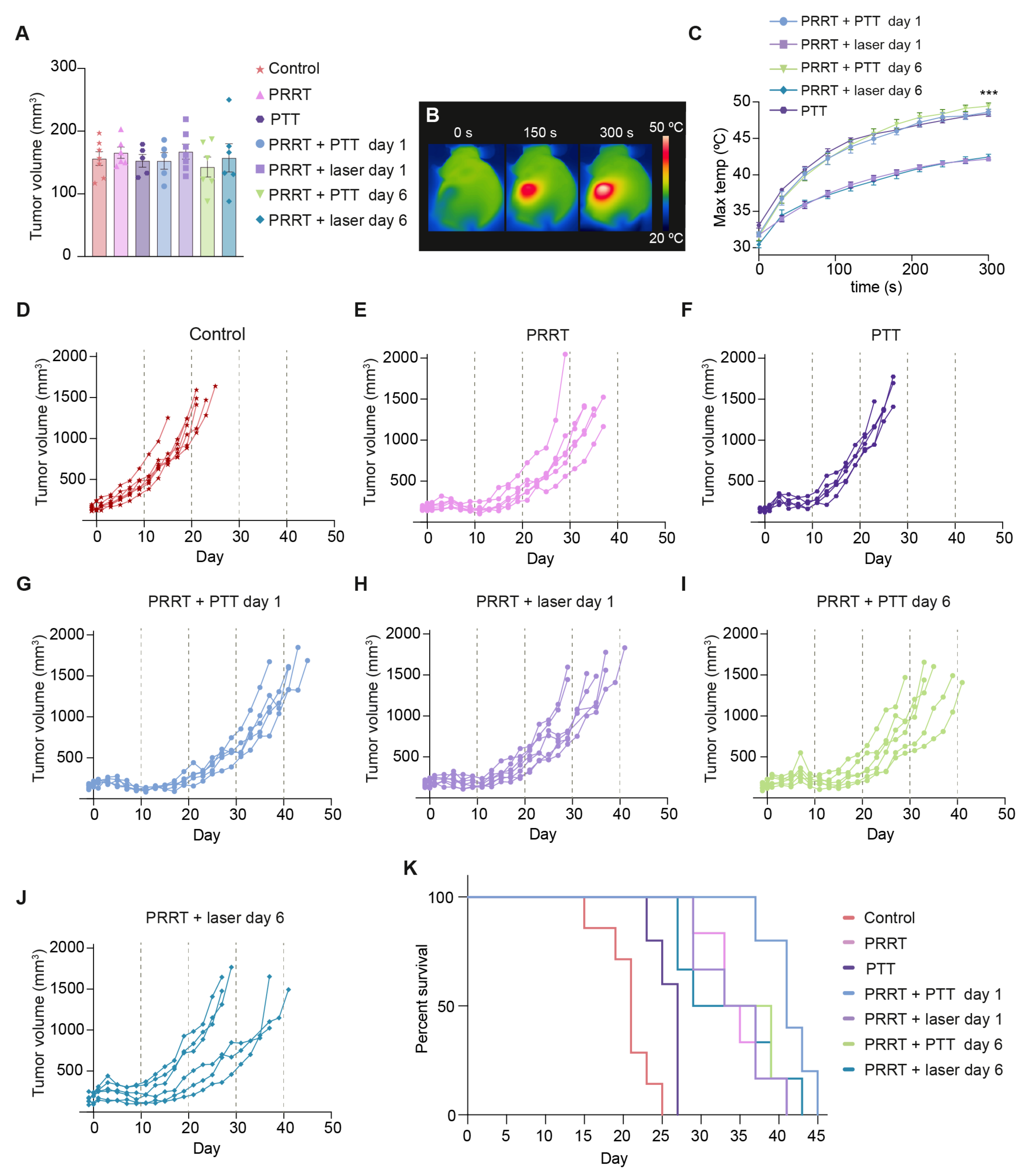
3.3. [177Lu]Lu-DOTA-TATE and Photothermal Therapy for the Treatment of SSTR2-Expressing Tumors In Vivo
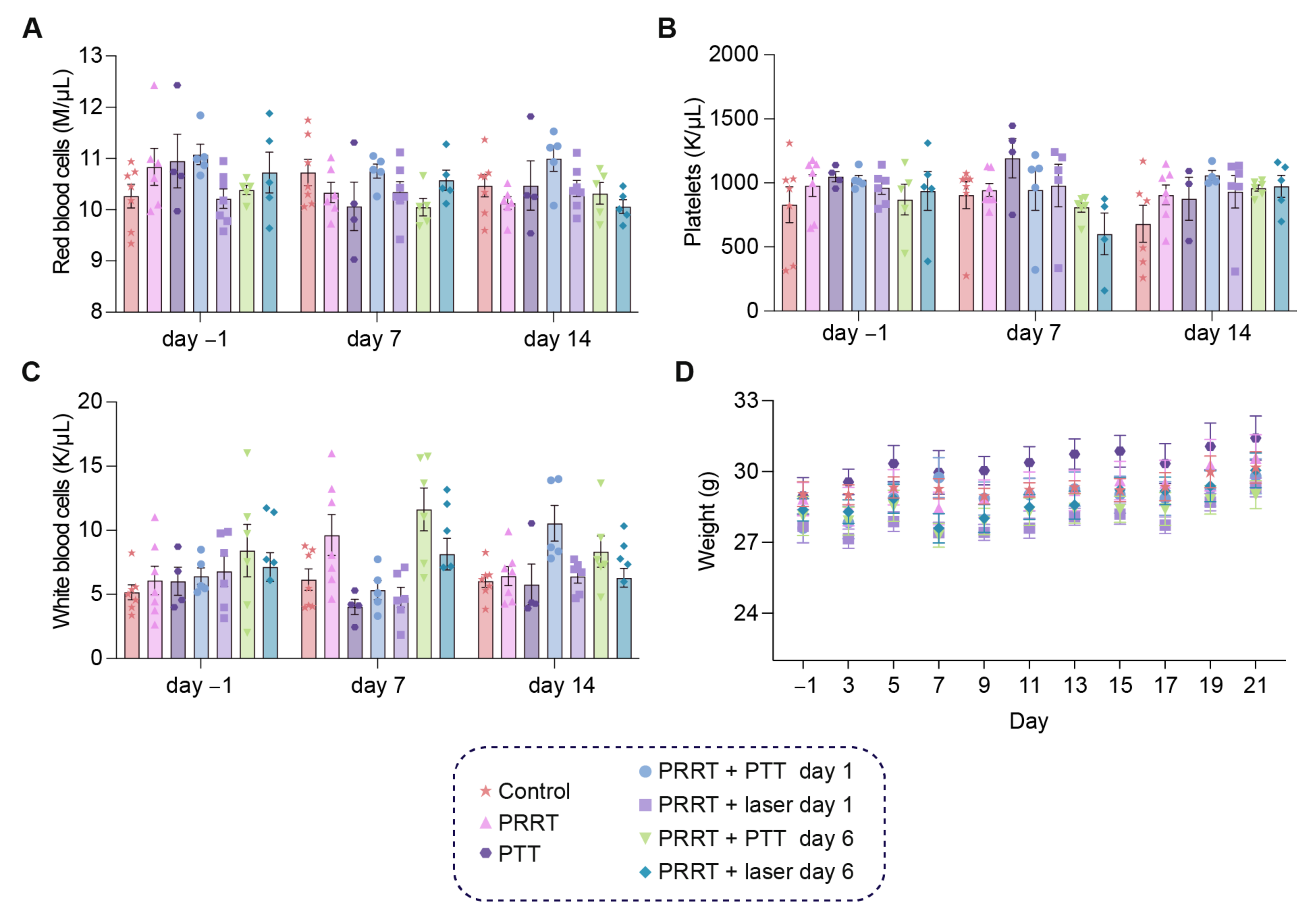
3.4. The Combination of [177Lu]Lu-DOTA-TATE and Photothermal Therapy Is Well-Tolerated in Tumor-Bearing Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ersahin, D.; Doddamane, I.; Cheng, D. Targeted Radionuclide Therapy. Cancers 2011, 3, 3838–3855. [Google Scholar] [CrossRef] [PubMed]
- La Salvia, A.; Espinosa-Olarte, P.; Riesco-Martinez, M.; Anton-Pascual, B.; Garcia-Carbonero, R. Targeted Cancer Therapy: What’s New in the Field of Neuroendocrine Neoplasms? Cancers 2021, 13, 1701. [Google Scholar] [CrossRef] [PubMed]
- Navalkissoor, S.; Flux, G.; Bomanji, J. Molecular radiotheranostics for neuroendocrine tumours. Clin. Med. 2017, 17, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Yordanova, A.; Ahmadzadehfar, H. Combination Therapies with PRRT. Pharmaceuticals 2021, 14, 1005. [Google Scholar] [CrossRef] [PubMed]
- Bison, S.M.; Konijnenberg, M.W.; Melis, M.; Pool, S.E.; Bernsen, M.; Teunissen, J.J.M.; Kwekkeboom, D.J.; De Jong, M. Peptide receptor radionuclide therapy using radiolabeled somatostatin analogs: Focus on future developments. Clin. Transl. Imaging 2014, 2, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Feijtel, D.; de Jong, M.; Nonnekens, J. Peptide Receptor Radionuclide Therapy: Looking Back, Looking Forward. Curr. Top. Med. Chem. 2020, 20, 2959–2969. [Google Scholar] [CrossRef]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Mittra, E.S. Neuroendocrine Tumor Therapy: 177Lu-DOTATATE. Am. J. Roentgenol. 2018, 211, 278–285. [Google Scholar] [CrossRef]
- Hennrich, U.; Kopka, K. Lutathera®: The First FDA- and EMA-Approved Radiopharmaceutical for Peptide Receptor Radionuclide Therapy. Pharmaceuticals 2019, 12, 114. [Google Scholar] [CrossRef]
- Kendi, A.T.; Halfdanarson, T.R.; Packard, A.; Dundar, A.; Subramaniam, R.M. Therapy With 177Lu-DOTATATE: Clinical Implementation and Impact on Care of Patients with Neuroendocrine Tumors. Am. J. Roentgenol. 2019, 213, 309–317. [Google Scholar] [CrossRef]
- Zukotynski, K.; Jadvar, H.; Capala, J.; Fahey, F. Targeted Radionuclide Therapy: Practical Applications and Future Prospects: Supplementary Issue: Biomarkers and Their Essential Role in the Development of Personalised Therapies (A). Biomarkers Cancer 2016, 8 (Suppl. S2), 35–38. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.-X.; Zhou, P.-K. DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct. Target. Ther. 2020, 5, 60. [Google Scholar] [CrossRef]
- Pillai, M.; Chakraborty, S.; Das, T.; Venkatesh, M.; Ramamoorthy, N. Production logistics of 177Lu for radionuclide therapy. Appl. Radiat. Isot. 2003, 59, 109–118. [Google Scholar] [CrossRef]
- Kim, K.; Kim, S.-J. Lu-177-Based Peptide Receptor Radionuclide Therapy for Advanced Neuroendocrine Tumors. Nucl. Med. Mol. Imaging 2018, 52, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Del Olmo-García, M.I.; Prado-Wohlwend, S.; Bello, P.; Segura, A.; Merino-Torres, J.F. Peptide Receptor Radionuclide Therapy with [177Lu]Lu-DOTA-TATE in Patients with Advanced GEP NENS: Present and Future Directions. Cancers 2022, 14, 584. [Google Scholar] [CrossRef]
- Bison, S.M.; Haeck, J.C.; Bol, K.; Koelewijn, S.J.; Groen, H.C.; Melís, M.; Veenland, J.F.; Bernsen, M.; De Jong, M. Optimization of combined temozolomide and peptide receptor radionuclide therapy (PRRT) in mice after multimodality molecular imaging studies. EJNMMI Res. 2015, 5, 62. [Google Scholar] [CrossRef]
- Liu, Q.; Qian, Y.; Li, P.; Zhang, S.; Wang, Z.; Liu, J.; Sun, X.; Fulham, M.; Feng, D.; Chen, Z.; et al. The combined therapeutic effects of 131iodine-labeled multifunctional copper sulfide-loaded microspheres in treating breast cancer. Acta Pharm. Sin. B 2018, 8, 371–380. [Google Scholar] [CrossRef]
- Cullinane, C.; Waldeck, K.; Kirby, L.; Rogers, B.E.; Eu, P.; Tothill, R.W.; Hicks, R.J. Enhancing the anti-tumour activity of 177Lu-DOTA-octreotate radionuclide therapy in somatostatin receptor-2 expressing tumour models by targeting PARP. Sci. Rep. 2020, 10, 10196. [Google Scholar] [CrossRef]
- Adnan, A.; Basu, S. Combined 177Lu-DOTATATE Peptide Receptor Radionuclide Therapy and Platinum-Based Chemotherapy in Recurrent, Metastatic Sinonasal Neuroendocrine Carcinoma: A Promising Therapeutic Option. J. Nucl. Med. Technol. 2020, 48, 292–294. [Google Scholar] [CrossRef]
- Jaque, D.; Martínez Maestro, L.; del Rosal, B.; Haro-Gonzalez, P.; Benayas, A.; Plaza, J.L.; Rodríguez, E.M.; Solé, J.G. Nanoparticles for photothermal therapies. Nanoscale 2014, 6, 9494–9530. [Google Scholar] [CrossRef]
- De Melo-Diogo, D.; Pais-Silva, C.; Dias, D.R.; Moreira, A.F.; Correia, I.J. Strategies to Improve Cancer Photothermal Therapy Mediated by Nanomaterials. Adv. Healthc. Mater. 2017, 6, 1700073. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Choi, K. Advances in Nanomaterial-Mediated Photothermal Cancer Therapies: Toward Clinical Applications. Biomedicines 2021, 9, 305. [Google Scholar] [CrossRef] [PubMed]
- Day, E.S.; Thompson, P.A.; Zhang, L.; Lewinski, N.A.; Ahmed, N.; Drezek, R.A.; Blaney, S.M.; West, J.L. Nanoshell-mediated photothermal therapy improves survival in a murine glioma model. J. Neuro Oncol. 2011, 104, 55–63. [Google Scholar] [CrossRef]
- Rastinehad, A.R.; Anastos, H.; Wajswol, E.; Winoker, J.S.; Sfakianos, J.P.; Doppalapudi, S.K.; Carrick, M.R.; Knauer, C.J.; Taouli, B.; Lewis, S.C.; et al. Gold nanoshell-localized photothermal ablation of prostate tumors in a clinical pilot device study. Proc. Natl. Acad. Sci. USA 2019, 116, 18590–18596. [Google Scholar] [CrossRef] [PubMed]
- Simón, M.; Nørregaard, K.; Jørgensen, J.T.; Oddershede, L.B.; Kjaer, A. Fractionated photothermal therapy in a murine tumor model: Comparison with single dose. Int. J. Nanomed. 2019, 14, 5369–5379. [Google Scholar] [CrossRef]
- Simón, M.; Jørgensen, J.T.; Norregaard, K.; Kjaer, A. 18F-FDG positron emission tomography and diffusion-weighted magnetic resonance imaging for response evaluation of nanoparticle-mediated photothermal therapy. Sci. Rep. 2020, 10, 7595. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, X.; Wang, X.; Guan, X.; Zhang, W.; Ma, J. Recent advances in selective photothermal therapy of tumor. J. Nanobiotechnology 2021, 19, 335. [Google Scholar] [CrossRef]
- Xing, S.; Zhang, X.; Luo, L.; Cao, W.; Li, L.; He, Y.; An, J.; Gao, D. Doxorubicin/gold nanoparticles coated with liposomes for chemo-photothermal synergetic antitumor therapy. Nanotechnology 2018, 29, 405101. [Google Scholar] [CrossRef]
- Nam, J.; Son, S.; Ochyl, L.J.; Kuai, R.; Schwendeman, A.; Moon, J.J. Chemo-photothermal therapy combination elicits anti-tumor immunity against advanced metastatic cancer. Nat. Commun. 2018, 9, 1074. [Google Scholar] [CrossRef]
- Wang, S.; Ma, X.; Hong, X.; Cheng, Y.; Tian, Y.; Zhao, S.; Liu, W.; Tang, Y.; Zhao, R.; Song, L.; et al. Adjuvant Photothermal Therapy Inhibits Local Recurrences after Breast-Conserving Surgery with Little Skin Damage. ACS Nano 2018, 12, 662–670. [Google Scholar] [CrossRef]
- Li, Z.; Chen, Y.; Yang, Y.; Yu, Y.; Zhang, Y.; Zhu, D.; Yu, X.; Ouyang, X.; Xie, Z.; Zhao, Y.; et al. Recent Advances in Nanomaterials-Based Chemo-Photothermal Combination Therapy for Improving Cancer Treatment. Front. Bioeng. Biotechnol. 2019, 7, 293. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, J.; Yu, Y.; Chen, S.; Huang, F.; Yang, C.; Chang, J.; Yang, L.; Fan, S.; Liu, J. Enhanced radiotherapy using photothermal therapy based on dual-sensitizer of gold nanoparticles with acid-induced aggregation. Nanomed. Nanotechnol. Biol. Med. 2020, 29, 102241. [Google Scholar] [CrossRef] [PubMed]
- Simón, M.; Jørgensen, J.T.; Melander, F.; Andresen, T.L.; Christensen, A.; Kjaer, A. Photothermal Therapy as Adjuvant to Surgery in an Orthotopic Mouse Model of Human Fibrosarcoma. Cancers 2021, 13, 5820. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, A.; Knigge, U.; Mortensen, J.; Oturai, P.; Berthelsen, A.K.; Loft, A.; Binderup, T.; Rasmussen, P.; Elema, D.; Klausen, T.L.; et al. Clinical PET of Neuroendocrine Tumors Using 64Cu-DOTATATE: First-in-Humans Study. J. Nucl. Med. 2012, 53, 1207–1215. [Google Scholar] [CrossRef]
- Pfeifer, A.; Knigge, U.; Binderup, T.; Mortensen, J.; Oturai, P.; Loft, A.; Berthelsen, A.K.; Langer, S.W.; Rasmussen, P.; Elema, D.; et al. 64Cu-DOTATATE PET for Neuroendocrine Tumors: A Prospective Head-to-Head Comparison with 111In-DTPA-Octreotide in 112 Patients. J. Nucl. Med. 2015, 56, 847–854. [Google Scholar] [CrossRef]
- Norregaard, K.; Jørgensen, J.T.; Simon, M.; Melander, F.; Kristensen, L.K.; Bendix, P.M.; Andresen, T.L.; Oddershede, L.B.; Kjaer, A. 18F-FDG PET/CT-based early treatment response evaluation of nanoparticle-assisted photothermal cancer therapy. PLoS ONE 2017, 12, e0177997. [Google Scholar] [CrossRef]
- Jørgensen, J.T.; Norregaard, K.; Martín, M.S.; Oddershede, L.B.; Kjaer, A. Non-invasive Early Response Monitoring of Nanoparticle-assisted Photothermal Cancer Therapy Using 18F-FDG, 18F-FLT, and 18F-FET PET/CT Imaging. Nanotheranostics 2018, 2, 201–210. [Google Scholar] [CrossRef][Green Version]
- Loke, K.; Padhy, A.; Ng, D.; Goh, A.; Divgi, C. Dosimetric Considerations in Radioimmunotherapy and Systemic Radionuclide Therapies: A Review. World J. Nucl. Med. 2011, 10, 122–138. [Google Scholar] [CrossRef]
- Liberini, V.; Huellner, M.; Grimaldi, S.; Finessi, M.; Thuillier, P.; Muni, A.; Pellerito, R.; Papotti, M.; Piovesan, A.; Arvat, E.; et al. The Challenge of Evaluating Response to Peptide Receptor Radionuclide Therapy in Gastroenteropancreatic Neuroendocrine Tumors: The Present and the Future. Diagnostics 2020, 10, 1083. [Google Scholar] [CrossRef]
- Haider, M.; Das, S.; Al-Toubah, T.; Pelle, E.; El-Haddad, G.; Strosberg, J. Somatostatin receptor radionuclide therapy in neuroendocrine tumors. Endocr. Relat. Cancer 2021, 28, R81–R93. [Google Scholar] [CrossRef]
- Sandström, M.; Garske-Román, U.; Granberg, D.; Johansson, S.; Widström, C.; Eriksson, B.; Sundin, A.; Lundqvist, H.; Lubberink, M. Individualized Dosimetry of Kidney and Bone Marrow in Patients Undergoing 177Lu-DOTA-Octreotate Treatment. J. Nucl. Med. 2013, 54, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Seidl, C.; Zöckler, C.; Beck, R.; Quintanilla-Martinez, L.; Bruchertseifer, F.; Senekowitsch-Schmidtke, R. 177Lu-immunotherapy of experimental peritoneal carcinomatosis shows comparable effectiveness to 213Bi-immunotherapy, but causes toxicity not observed with 213Bi. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Exner, S.; Arrey, G.; Prasad, V.; Grötzinger, C. mTOR Inhibitors as Radiosensitizers in Neuroendocrine Neoplasms. Front. Oncol. 2021, 10, 578380. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, T.N.; van Velthuysen, M.-L.; Van Eijck, C.H.J.; Teunissen, J.J.; Hofland, J.; De Herder, W.W. Successful neoadjuvant peptide receptor radionuclide therapy for an inoperable pancreatic neuroendocrine tumour. Endocrinol. Diabetes Metab. Case Rep. 2018, 2018, 18-0015. [Google Scholar] [CrossRef] [PubMed]
- Bucharskaya, A.; Maslyakova, G.; Terentyuk, G.; Yakunin, A.; Avetisyan, Y.; Bibikova, O.; Tuchina, E.; Khlebtsov, B.; Khlebtsov, N.; Tuchin, V. Towards Effective Photothermal/Photodynamic Treatment Using Plasmonic Gold Nanoparticles. Int. J. Mol. Sci. 2016, 17, 1295. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Shao, Z.; Zhao, Y. Solutions to the Drawbacks of Photothermal and Photodynamic Cancer Therapy. Adv. Sci. 2021, 8, 2002504. [Google Scholar] [CrossRef]
- Yagawa, Y.; Tanigawa, K.; Kobayashi, Y.; Yamamoto, M. Cancer immunity and therapy using hyperthermia with immunotherapy, radiotherapy, chemotherapy, and surgery. J. Cancer Metastasis Treat. 2017, 3, 218. [Google Scholar] [CrossRef]
- Oei, A.; Kok, H.; Oei, S.; Horsman, M.; Stalpers, L.; Franken, N.; Crezee, J. Molecular and biological rationale of hyperthermia as radio- and chemosensitizer. Adv. Drug Deliv. Rev. 2020, 163–164, 84–97. [Google Scholar] [CrossRef]
- Refaat, T.; Sachdev, S.; Sathiaseelan, V.; Helenowski, I.; Abdelmoneim, S.; Pierce, M.C.; Woloschak, G.; Small, W., Jr.; Mittal, B.; Kiel, K.D. Hyperthermia and radiation therapy for locally advanced or recurrent breast cancer. Breast 2015, 24, 418–425. [Google Scholar] [CrossRef]
- Horsman, M.R.; Overgaard, J. Hyperthermia: A Potent Enhancer of Radiotherapy. Clin. Oncol. 2007, 19, 418–426. [Google Scholar] [CrossRef]
- Kaur, P.; Hurwitz, M.D.; Krishnan, S.; Asea, A. Combined Hyperthermia and Radiotherapy for the Treatment of Cancer. Cancers 2011, 3, 3799–3823. [Google Scholar] [CrossRef] [PubMed]
- Peeken, J.C.; Vaupel, P.; Combs, S.E. Integrating Hyperthermia into Modern Radiation Oncology: What Evidence Is Necessary? Front. Oncol. 2017, 7, 132. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhong, X.; Yi, X.; Huang, M.; Ning, P.; Liu, T.; Ge, C.; Chai, Z.; Liu, Z.; Yang, K. Radionuclide 131I labeled reduced graphene oxide for nuclear imaging guided combined radio- and photothermal therapy of cancer. Biomaterials 2015, 66, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Yook, S.; Cai, Z.; Lu, Y.; Winnik, M.A.; Pignol, J.-P.; Reilly, R.M. Intratumorally Injected 177Lu-Labeled Gold Nanoparticles: Gold Nanoseed Brachytherapy with Application for Neoadjuvant Treatment of Locally Advanced Breast Cancer. J. Nucl. Med. 2016, 57, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Kwekkeboom, D.J.; De Herder, W.W.; Kam, B.L.; Van Eijck, C.H.; Van Essen, M.; Kooij, P.P.; Feelders, R.A.; Van Aken, M.O.; Krenning, E.P. Treatment With the Radiolabeled Somatostatin Analog [177Lu-DOTA0,Tyr3]Octreotate: Toxicity, Efficacy, and Survival. J. Clin. Oncol. 2008, 26, 2124–2130. [Google Scholar] [CrossRef]
- Singh, S.; Han, L.; Murali, R.; Solis, L.; Roth, J.; Ji, L.; Wistuba, I.; Kundra, V. SSTR2-Based Reporters for Assessing Gene Transfer into Non–Small Cell Lung Cancer: Evaluation Using an Intrathoracic Mouse Model. Hum. Gene Ther. 2011, 22, 55–64. [Google Scholar] [CrossRef]
| Group | Treatment | n |
|---|---|---|
| Control | No treatment | 7 |
| PRRT | [177Lu]Lu-DOTA-TATE injection (day 0) | 6 |
| PTT | NS-based PTT (day 1) | 5 |
| PRRT + PTT day 1 | [177Lu]Lu-DOTA-TATE injection (day 0) and NS-based PTT (day 1) | 5 |
| PRRT + laser day 1 | [177Lu]Lu-DOTA-TATE injection (day 0) and laser treatment (without NS, day 1) | 7 |
| PRRT + PTT day 6 | [177Lu]Lu-DOTA-TATE injection (day 0) and NS-based PTT (day 6) | 6 |
| PRRT + laser day 6 | [177Lu]Lu-DOTA-TATE injection (day 0) and laser treatment (without NS, day 6) | 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simón, M.; Jørgensen, J.T.; Khare, H.A.; Christensen, C.; Nielsen, C.H.; Kjaer, A. Combination of [177Lu]Lu-DOTA-TATE Targeted Radionuclide Therapy and Photothermal Therapy as a Promising Approach for Cancer Treatment: In Vivo Studies in a Human Xenograft Mouse Model. Pharmaceutics 2022, 14, 1284. https://doi.org/10.3390/pharmaceutics14061284
Simón M, Jørgensen JT, Khare HA, Christensen C, Nielsen CH, Kjaer A. Combination of [177Lu]Lu-DOTA-TATE Targeted Radionuclide Therapy and Photothermal Therapy as a Promising Approach for Cancer Treatment: In Vivo Studies in a Human Xenograft Mouse Model. Pharmaceutics. 2022; 14(6):1284. https://doi.org/10.3390/pharmaceutics14061284
Chicago/Turabian StyleSimón, Marina, Jesper Tranekjær Jørgensen, Harshvardhan A. Khare, Camilla Christensen, Carsten Haagen Nielsen, and Andreas Kjaer. 2022. "Combination of [177Lu]Lu-DOTA-TATE Targeted Radionuclide Therapy and Photothermal Therapy as a Promising Approach for Cancer Treatment: In Vivo Studies in a Human Xenograft Mouse Model" Pharmaceutics 14, no. 6: 1284. https://doi.org/10.3390/pharmaceutics14061284
APA StyleSimón, M., Jørgensen, J. T., Khare, H. A., Christensen, C., Nielsen, C. H., & Kjaer, A. (2022). Combination of [177Lu]Lu-DOTA-TATE Targeted Radionuclide Therapy and Photothermal Therapy as a Promising Approach for Cancer Treatment: In Vivo Studies in a Human Xenograft Mouse Model. Pharmaceutics, 14(6), 1284. https://doi.org/10.3390/pharmaceutics14061284








