Optimization of a Cefuroxime Axetil-Loaded Liquid Self-Nanoemulsifying Drug Delivery System: Enhanced Solubility, Dissolution and Caco-2 Cell Uptake
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. HPLC Analysis
2.2.2. Preparation of Blank SNEDDS
- Selection of the SNEDDS Components
- Construction of the Phase Diagram
2.2.3. Preparation of CA-Loaded SNEDDS
2.2.4. Characterization of CA-Loaded SNEDDS
- Thermodynamic Stability and Self-Nanoemulsification Tests
- Grade A: Clear nanoemulsion that forms quickly and spontaneously.
- Grade B: Bluish, slightly less clear nanoemulsion that forms quickly and spontaneously.
- Grade C: Turbid emulsion that forms slowly.
- Grade D: Turbid emulsion that is dull and grayish.
- Grade E: Turbid emulsion with visible oil globules on the surface.
- Physicochemical Characterization of the CA- SNEDDS
2.2.5. In Vitro Release Studies
2.2.6. Cell Culture Studies
- Cytotoxicity Study
- Cellular Uptake Study
2.2.7. Statistical Analysis
3. Results and Discussion
3.1. Solubility Studies, Excipient Screening and Selection
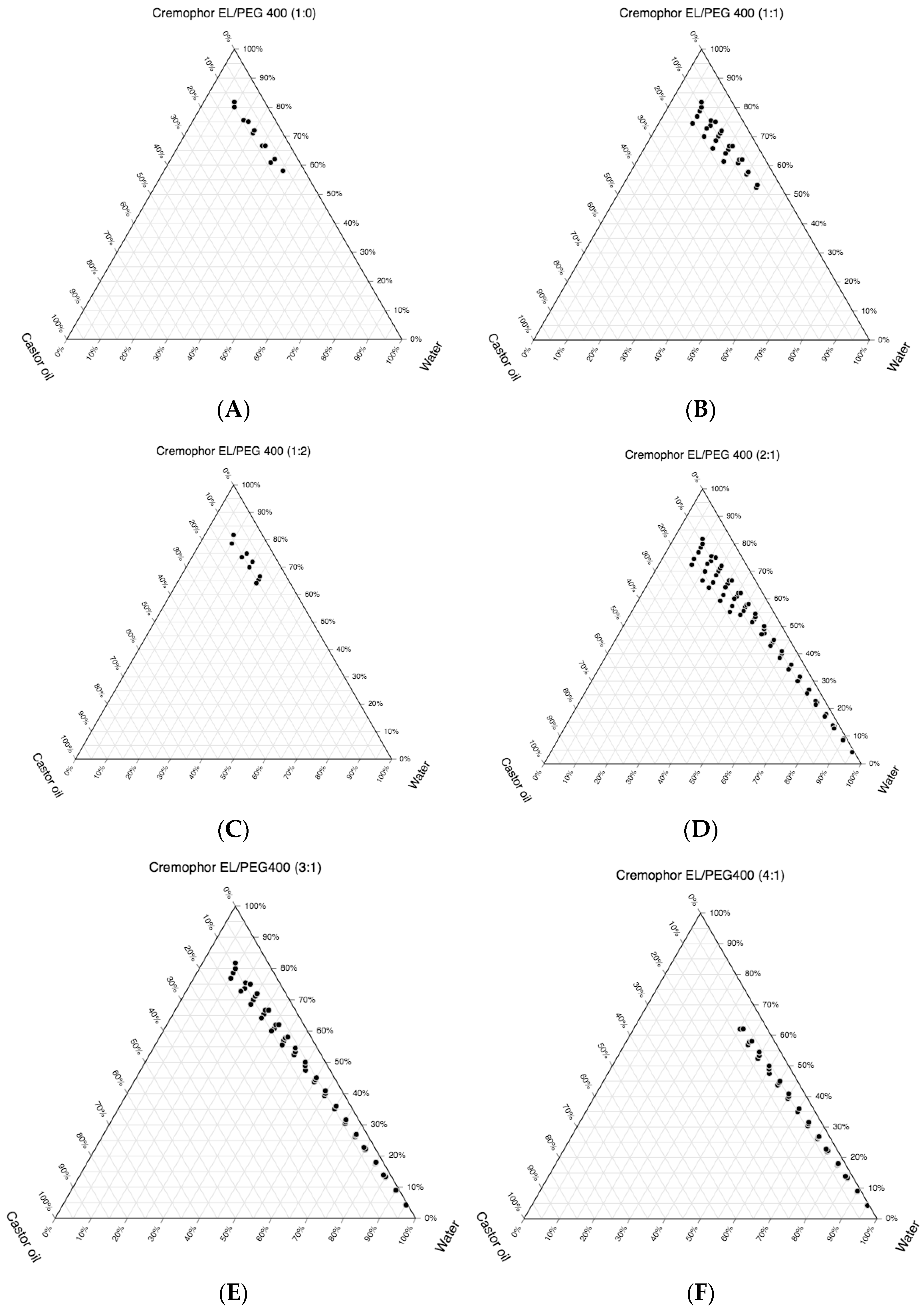
3.2. Construction of the Pseudo-Ternary Phase Diagrams and Drug Loading
3.3. Characterization of the CA-Loaded SNEDDS
- Thermodynamic Stability Study
- Physicochemical Characterization of the CA-SNEDDS
- Drug Loading Efficiency
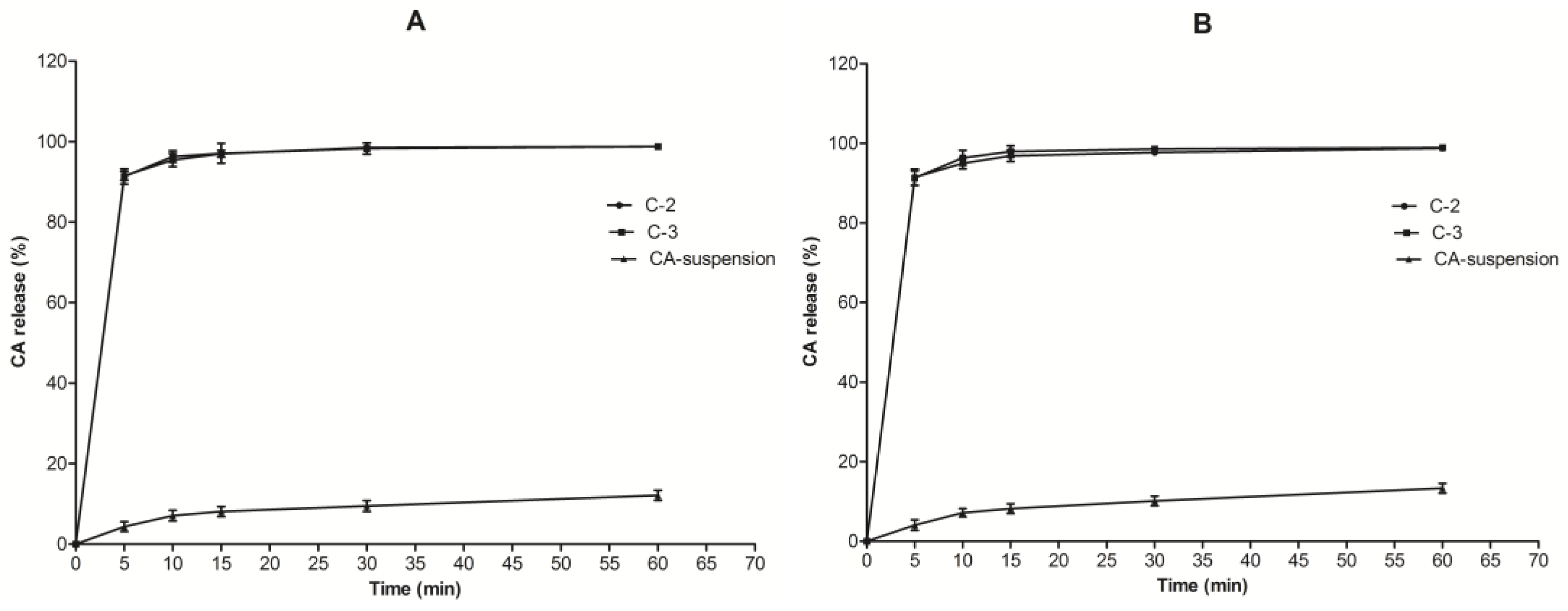
3.4. In Vitro Drug Release Studies
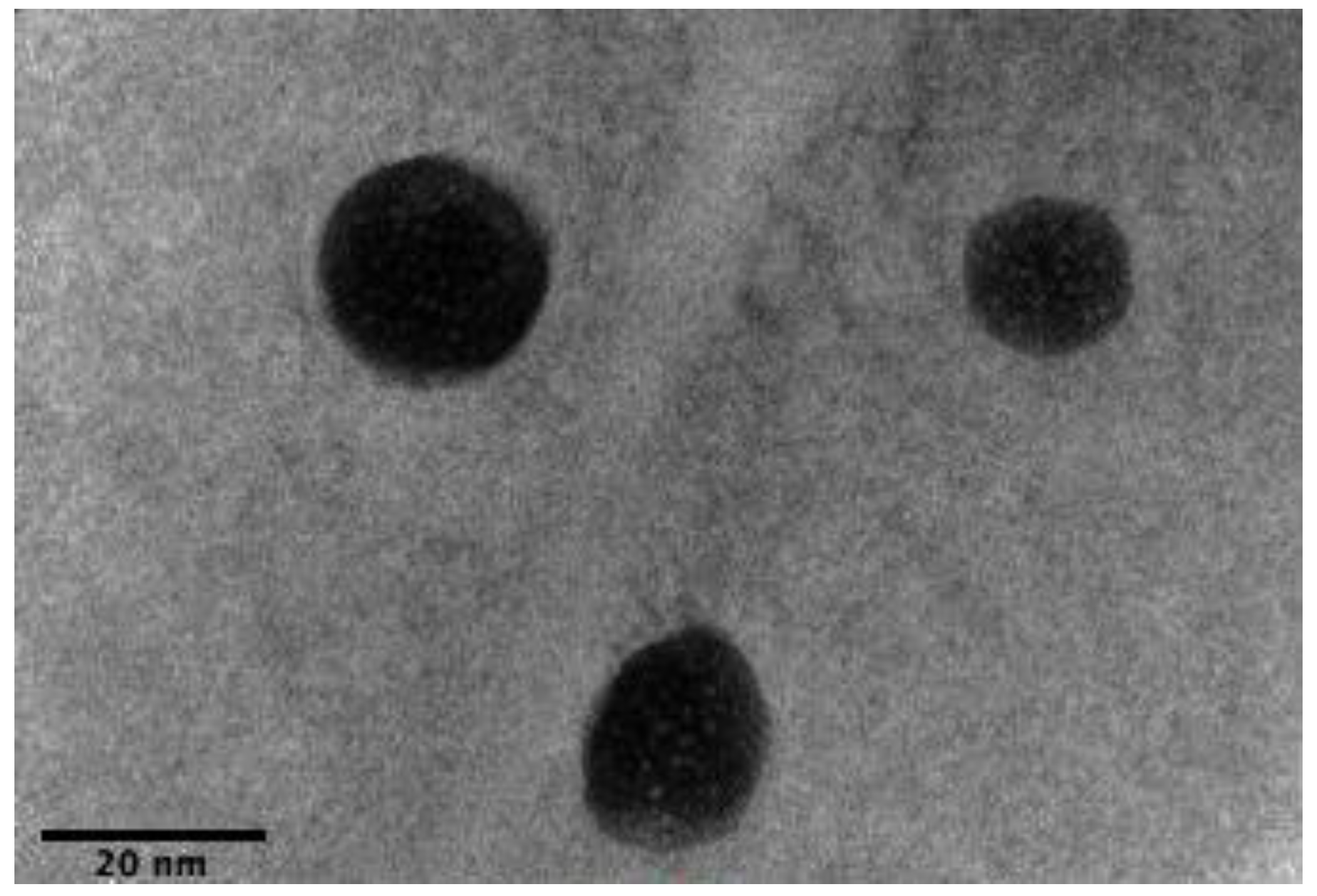
3.5. High-Resolution Transmission Electron Microscope (HRTEM)
3.6. Cell Culture
- Cytotoxicity Assay
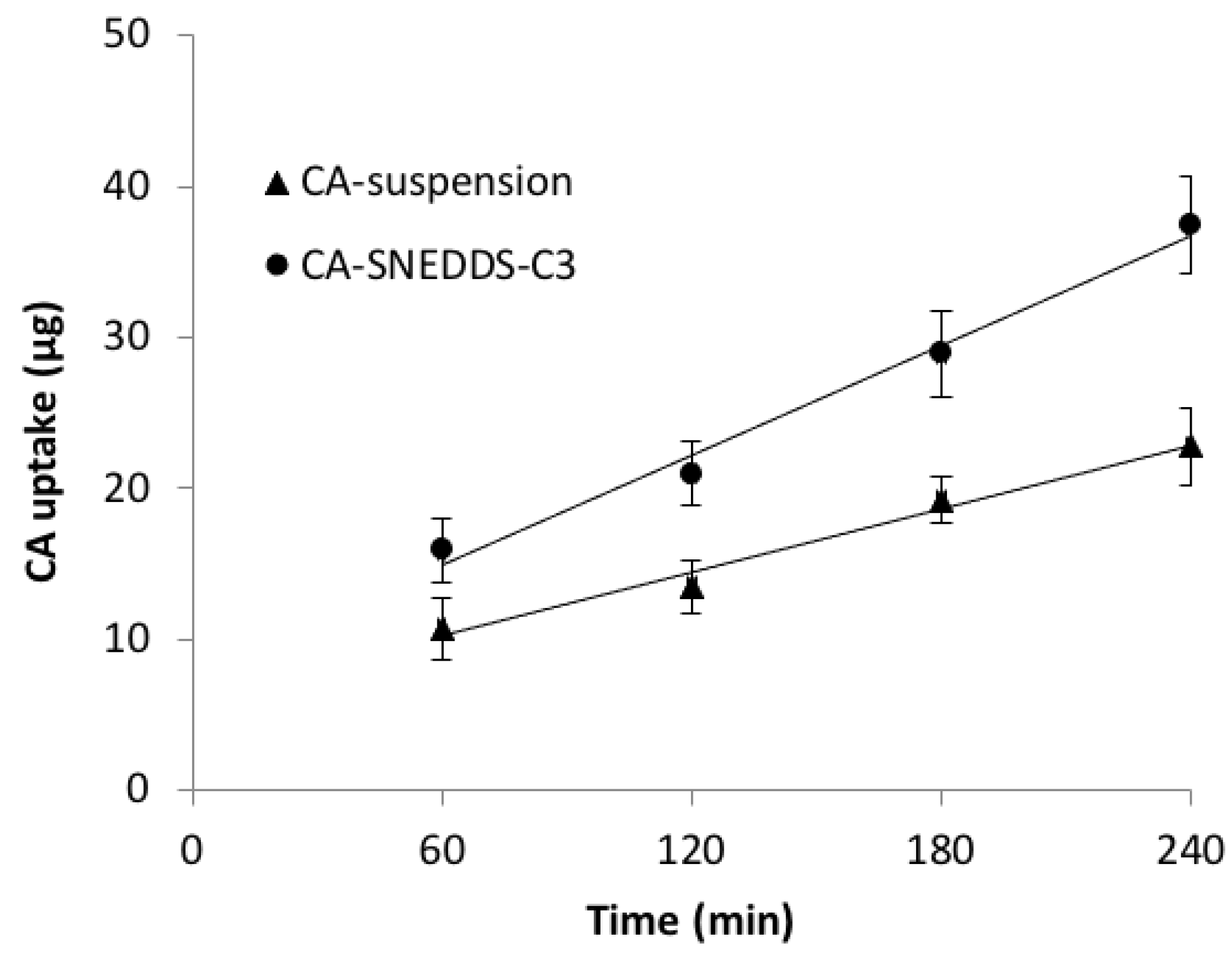
- Cellular Uptake Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barrett, M.A.; Lawrence, M.J.; Hutt, A.J.; Lansley, A.B. Stereoselective Absorption and Hydrolysis of Cefuroxime Axetil Diastereomers Using the Caco-2 Cell Monolayer Model. Eur. J. Drug Metab. Pharmacokinet. 1997, 22, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.M.; Brogden, R.N. Cefuroxime Axetil: A Review of Its Antibacterial Activity, Pharmacokinetic Properties and Therapeutic Efficacy. Drugs 1996, 52, 125–158. [Google Scholar] [CrossRef] [PubMed]
- Jelińska, A.; Dudzińska, I.; Zajac, M.; Oszczpowicz, I. The Stability of the Amorphous Form of Cefuroxime Axetil in Solid State. J. Pharm. Biomed. Anal. 2006, 41, 1075–1081. [Google Scholar] [CrossRef] [PubMed]
- Sullins, A.K.; Abdel-Rahman, S.M. Pharmacokinetics of Antibacterial Agents in the CSF of Children and Adolescents. Pediatr. Drugs 2013, 15, 93–117. [Google Scholar] [CrossRef]
- Al-Said, M.S.; Al-Khamis, K.I.; Niazy, E.M.; El-Sayed, Y.M.; Al-Rashood, K.A.; Al-Bella, S.; Al-Yamani, M.A.; Al-Najjar, T.A.; Alam, S.M.; Dham, R.; et al. Bioequivalence Evaluation of Two Brands of Cefuroxime 500 Mg Tablets (Cefuzime® and Zinnat®) in Healthy Human Volunteers. Biopharm. Drug Dispos. 2000, 21, 205–210. [Google Scholar] [CrossRef]
- Dellamonica, P. Cefuroxime Axetil. Int. J. Antimicrob. Agents 1994, 4, 23–36. [Google Scholar] [CrossRef]
- Jewesson, P.J. Pharmaceutical, Pharmacokinetic and Other Considerations for Intravenous to Oral Stepdown Therapy. Can. J. Infect. Dis. 1995, 6, 11A–16A. [Google Scholar] [CrossRef] [Green Version]
- Donn, K.H.; James, N.C.; Powell, J.R. Bioavailability of Cefuroxime Axetil Formulations. J. Pharm. Sci. 1994, 83, 842–844. [Google Scholar] [CrossRef]
- Harding, S.M.; Williams, P.E.O.; Ayrton, J. Pharmacology of Cefuroxime as the 1-Acetoxyethyl Ester in Volunteers. Antimicrob. Agents Chemother. 1984, 25, 78–82. [Google Scholar] [CrossRef] [Green Version]
- Finn, A.; Straughn, A.; Meyer, M.; Chubb, J. Effect of Dose and Food on the Bioavailability of Cefuroxime Axetil. Biopharm. Drug Dispos. 1987, 8, 519–526. [Google Scholar] [CrossRef]
- Williams, P.E.O.; Harding, S.M. The Absolute Bioavailability of Oral Cefuroxime Axetil in Male and Female Volunteers after Fasting and after Food. J. Antimicrob. Chemother. 1984, 13, 191–196. [Google Scholar] [CrossRef]
- Mosher, G.L.; McBee, J.; Shaw, D.B. Esterase Activity toward the Diastereomers of Cefuroxime Axetil in the Rat and Dog. Pharm. Res. 1992, 9, 687–689. [Google Scholar] [CrossRef]
- Imada, C.; Takahashi, T.; Kuramoto, M.; Masuda, K.; Ogawara, K.I.; Sato, A.; Wataya, Y.; Kim, H.S.; Higaki, K. Improvement of Oral Bioavailability of N-251, a Novel Antimalarial Drug, by Increasing Lymphatic Transport with Long-Chain Fatty Acid-Based Self-Nanoemulsifying Drug Delivery System. Pharm. Res. 2015, 32, 2595–2608. [Google Scholar] [CrossRef]
- Baloch, J.; Sohail, M.F.; Sarwar, H.S.; Kiani, M.H.; Khan, G.M.; Jahan, S.; Rafay, M.; Chaudhry, M.T.; Yasinzai, M.; Shahnaz, G. Self-Nanoemulsifying Drug Delivery System (SNEDDS) for Improved Oral Bioavailability of Chlorpromazine: In Vitro and In Vivo Evaluation. Medicina 2019, 55, 210. [Google Scholar] [CrossRef] [Green Version]
- Date, A.A.; Desai, N.; Dixit, R.; Nagarsenker, M. Self-Nanoemulsifying Drug Delivery Systems: Formulation Insights, Applications and Advances. Nanomedicine 2010, 5, 1595–1616. [Google Scholar] [CrossRef]
- Narang, A.S.; Delmarre, D.; Gao, D. Stable Drug Encapsulation in Micelles and Microemulsions. Int. J. Pharm. 2007, 345, 9–25. [Google Scholar] [CrossRef]
- Kim, D.H.; Kim, J.Y.; Kim, R.M.; Maharjan, P.; Ji, Y.G.; Jang, D.J.; Min, K.A.; Koo, T.S.; Cho, K.H. Orlistat-Loaded Solid SNEDDS for the Enhanced Solubility, Dissolution, and in Vivo Performance. Int. J. Nanomed. 2018, 13, 7095–7106. [Google Scholar] [CrossRef] [Green Version]
- Gupta, S.; Chavhan, S.; Sawant, K.K. Self-Nanoemulsifying Drug Delivery System for Adefovir Dipivoxil: Design, Characterization, in Vitro and Ex Vivo Evaluation. Colloids Surf. A Physicochem. Eng. Asp. 2011, 392, 145–155. [Google Scholar] [CrossRef]
- Sanna, V.; Gavini, E.; Cossu, M.; Rassu, G.; Giunchedi, P. Solid Lipid Nanoparticles (SLN) as Carriers for the Topical Delivery of Econazole Nitrate: In-Vitro Characterization, Ex-Vivo and in-Vivo Studies. J. Pharm. Pharmacol. 2010, 59, 1057–1064. [Google Scholar] [CrossRef]
- Fogh, J.; Fogh, J.M.; Orfeo, T. One Hundred and Twenty Seven Cultured Human Tumor Cell Lines Producing Tumors in Nude Mice. J. Natl. Cancer Inst. 1977, 59, 221–226. [Google Scholar] [CrossRef]
- Hidalgo, I.J.; Raub, T.J.; Borchardt, R.T. Characterization of the Human Colon Carcinoma Cell Line (Caco-2) as a Model System for Intestinal Epithelial Permeability. Gastroenterology 1989, 96, 736–749. [Google Scholar] [CrossRef]
- Artursson, P. Cell Cultures as Models for Drug Absorption across the Intestinal Mucosa. Crit. Rev. Ther. Drug Carr. Syst. 1991, 8, 305–330. [Google Scholar]
- Gochoco, C.H.; Ryan, F.M.; Miller, J.; Smith, P.L.; Hidalgo, I.J. Uptake and Transepithelial Transport of the Orally Absorbed Cephalosporin Cephalexin, in the Human Intestinal Cell Line, Caco-2. Int. J. Pharm. 1994, 104, 187–202. [Google Scholar] [CrossRef]
- Dantzig, A.H.; Duckworth, D.C.; Tabas, L.B. Transport Mechanisms Responsible for the Absorption of Loracarbef, Cefixime, and Cefuroxime Axetil into Human Intestinal Caco-2 Cells. BBA Biomembr. 1994, 1191, 7–13. [Google Scholar] [CrossRef]
- Inui, K.I.; Yamamoto, M.; Saito, H. Transepithelial Transport of Oral Cephalosporins by Monolayers of Intestinal Epithelial Cell Line Caco-2: Specific Transport Systems in Apical and Basolateral Membranes. J. Pharmacol. Exp. Ther. 1992, 261, 195–201. [Google Scholar]
- Hovgaard, L.; Brøndsted, H.; Buur, A.; Bundgaard, H. Drug Delivery Studies in Caco-2 Monolayers. Synthesis, Hydrolysis, and Transport of O-Cyclopropane Carboxylic Acid Ester Prodrugs of Various β-Blocking Agents. Pharm. Res. 1995, 12, 387–392. [Google Scholar] [CrossRef]
- Khan, A.A.; Kumar, A.; Ali, J.; Sahni, J.K.; Baboota, S. Formulation, Optimization and Characterization of Self Nanoemulsifying Drug Delivery System (SNEDDS) of Paclitaxel for Solubility Enhancement. Nanosci. Nanotechnol. Lett. 2013, 5, 861–867. [Google Scholar] [CrossRef]
- Shakeel, F.; Shazly, G.A.; Raish, M.; Ahmad, A.; Kalam, M.A.; Ali, N.; Ansari, M.A.; Elosaily, G.M. Biological Investigation of a Supersaturated Self-Nanoemulsifying Drug Delivery System of Piper Cubeba Essential Oil. RSC Adv. 2015, 5, 105206–105217. [Google Scholar] [CrossRef]
- Shafiq, S.; Shakeel, F.; Talegaonkar, S.; Ahmad, F.J.; Khar, R.K.; Ali, M. Development and Bioavailability Assessment of Ramipril Nanoemulsion Formulation. Eur. J. Pharm. Biopharm. 2007, 66, 227–243. [Google Scholar] [CrossRef]
- Khan, A.W.; Kotta, S.; Ansari, S.H.; Sharma, R.K.; Ali, J. Self-Nanoemulsifying Drug Delivery System (SNEDDS) of the Poorly Water-Soluble Grapefruit Flavonoid Naringenin: Design, Characterization, in Vitro and in Vivo Evaluation. Drug Deliv. 2015, 22, 552–561. [Google Scholar] [CrossRef]
- Na, Y.G.; Byeon, J.J.; Wang, M.; Huh, H.W.; Son, G.H.; Jeon, S.H.; Bang, K.H.; Kim, S.J.; Lee, H.J.; Lee, H.K.; et al. Strategic Approach to Developing a Self-Microemulsifying Drug Delivery System to Enhance Antiplatelet Activity and Bioavailability of Ticagrelor. Int. J. Nanomed. 2019, 14, 1193–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, A.A.; Mudassir, J.; Akhtar, S.; Murugaiyah, V.; Darwis, Y. Freeze-Dried Lopinavir-Loaded Nanostructured Lipid Carriers for Enhanced Cellular Uptake and Bioavailability: Statistical Optimization, in Vitro and in Vivo Evaluations. Pharmaceutics 2019, 11, 97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nazzal, S.; Smalyukh, I.I.; Lavrentovich, O.D.; Khan, M.A. Preparation and in Vitro Characterization of a Eutectic Based Semisolid Self-Nanoemulsified Drug Delivery System (SNEDDS) of Ubiquinone: Mechanism and Progress of Emulsion Formation. Int. J. Pharm. 2002, 235, 247–265. [Google Scholar] [CrossRef]
- Swenson, E.S.; Milisen, W.B.; Curatolo, W. Intestinal Permeability Enhancement: Efficacy, Acute Local Toxicity, and Reversibility. Pharm. Res. 1994, 11, 1132–1142. [Google Scholar] [CrossRef]
- Shakeel, F.; Haq, N.; El-Badry, M.; Alanazi, F.K.; Alsarra, I.A. Ultra Fine Super Self-Nanoemulsifying Drug Delivery System (SNEDDS) Enhanced Solubility and Dissolution of Indomethacin. J. Mol. Liq. 2013, 180, 89–94. [Google Scholar] [CrossRef]
- Kathe, N.; Henriksen, B.; Chauhan, H. Physicochemical Characterization Techniques for Solid Lipid Nanoparticles: Principles and Limitations. Drug Dev. Ind. Pharm. 2014, 40, 1565–1575. [Google Scholar] [CrossRef]
- Villar, A.M.S.; Naveros, B.C.; Campmany, A.C.C.; Trenchs, M.A.; Rocabert, C.B.; Bellowa, L.H. Design and Optimization of Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Enhanced Dissolution of Gemfibrozil. Int. J. Pharm. 2012, 431, 161–175. [Google Scholar] [CrossRef] [Green Version]
- Nielsen, F.S.; Petersen, K.B.; Müllertz, A. Bioavailability of Probucol from Lipid and Surfactant Based Formulations in Minipigs: Influence of Droplet Size and Dietary State. Eur. J. Pharm. Biopharm. 2008, 69, 553–562. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Liu, Y.; Peterson, D.A.; Kimura, H.; Schubert, D. Mechanism of Cellular 3-(4,5-Dimethylthiazol-2-Yl)-2,5- Diphenyltetrazolium Bromide (MTT) Reduction. J. Neurochem. 1997, 69, 581–593. [Google Scholar] [CrossRef]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium Dyes as Tools in Cell Biology: New Insights into Their Cellular Reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [CrossRef]
- Campbell, C.J.; Chantrell, L.J.; Eastmond, R. Purification and Partial Characterization of Rat Intestinal Cefuroxime Axetil Esterase. Biochem. Pharmacol. 1987, 36, 2317–2324. [Google Scholar] [CrossRef]
- Cordon-Cardo, C.; O’Brien, J.P.; Boccia, J.; Casals, D.; Bertino, J.R.; Melamed, M.R. Expression of the Multidrug Resistance Gene Product (P-Glycoprotein) in Human Normal and Tumor Tissues. J. Histochem. Cytochem. 1990, 38, 1277–1287. [Google Scholar] [CrossRef]


| Formulation Code | Castor Oil (% v/v) | Smix (Cremophor EL:PEG 400; 2:1) (% v/v) | Water (% v/v) |
|---|---|---|---|
| C1 | 5 | 35 | 60 |
| C2 | 5 | 40 | 55 |
| C3 | 5 | 45 | 50 |
| C4 | 5 | 30 | 65 |
| C5 | 7.84 | 47.06 | 45.1 |
| C6 | 8.13 | 56.91 | 34.96 |
| C7 | 11 | 54.14 | 34.86 |
| C8 | 11.95 | 57.36 | 30.7 |
| C9 | 13.79 | 55.17 | 31.03 |
| C10 | 14.81 | 59.26 | 25.93 |
| Figures | Smix Ratio | Self-Nanoemulsifying Area | Maximum Oil Phase Emulsified (% v/v) | Maximum Amount of Smix Used to Emulsify Respective Maximum Oil Phase (% v/v) |
|---|---|---|---|---|
| Figure 2A | 1:0 | Lowest | 10 | 80 |
| Figure 2B | 1:1 | Higher than 2A | 15.44 | 74.51 |
| Figure 2C | 1:2 | Lower than 2A | 11.24 | 78.65 |
| Figure 2D | 2:1 | Highest | 17.21 | 72.36 |
| Figure 2E | 3:1 | Lower than 2D | 12.82 | 76.92 |
| Figure 2F | 4:1 | Lower than 2D and 2E | 8.13 | 56.91 |
| Formulation Code | Emulsification Time (s) | Grade of Self-Nanoemulsification Test | Thermodynamic Stability Tests | ||
|---|---|---|---|---|---|
| Centrifugation | Heating and Cooling Cycles | Freeze-Thaw Cycles | |||
| C1 | 16.67 ± 0.59 | A | ✓ | ✓ | ✓ |
| C2 | 17.93 ± 0.61 | A | ✓ | ✓ | ✓ |
| C3 | 15.79 ± 0.40 | A | ✓ | ✓ | ✓ |
| C4 | 20.67 ± 0.62 | A | ✓ | ✓ | ✓ |
| C5 | 16.75 ± 0.84 | A | ✓ | ✓ | ✓ |
| C6 | 20.82 ± 0.54 | A | ✓ | ✓ | ✓ |
| C7 | 22.06 ± 0.64 | A | ✓ | ✓ | ✓ |
| C8 | 20.07 ± 0.63 | A | ✓ | ✓ | ✓ |
| C9 | 20.75 ± 0.51 | A | ✓ | ✓ | ✓ |
| C10 | 21.42 ± 0.68 | A | ✓ | ✓ | ✓ |
| Formulation Code | Mean Droplet Size (nm) | Poly Dispersity Index (PDI) | Zeta Potential (mV) | Transmittance (%) | Drug Loading Efficiency (%) |
|---|---|---|---|---|---|
| C1 | 23.33± 1.07 | 0.116 ± 0.008 | −19.71 ± 1.05 | 97.41 ± 0.34 | 96.44 ± 1.33 |
| C2 | 21.23± 1.90 | 0.084 ± 0.009 | −21.53 ± 1.64 | 98.23 ± 0.15 | 97.07 ± 1.47 |
| C3 | 18.50± 1.83 | 0.064 ± 0.008 | −22.12 ± 1.20 | 98.32 ± 0.27 | 97.62 ± 1.06 |
| C4 | 26.17± 2.05 | 0.132 ± 0.011 | −18.88 ± 1.78 | 96.42 ± 0.25 | 96.25 ± 1.22 |
| C5 | 23.80± 1.45 | 0.106 ± 0.005 | −19.91 ± 1.37 | 97.23 ± 0.06 | 98.76 ± 0.68 |
| C6 | 25.98± 1.48 | 0.129 ± 0.009 | −17.78 ± 1.52 | 96.43 ± 0.31 | 98.17 ± 0.83 |
| C7 | 32.07± 1.11 | 0.168 ± 0.018 | −12.35 ± 2.06 | 96.23 ± 0.15 | 99.41 ± 0.22 |
| C8 | 33.43± 0.83 | 0.170 ± 0.020 | −12.77 ± 1.79 | 96.47 ± 0.31 | 99.16 ± 0.48 |
| C9 | 27.33± 1.70 | 0.140 ± 0.014 | −14.36 ± 1.89 | 96.32 ± 0.26 | 99.04 ± 0.23 |
| C10 | 27.97± 2.32 | 0.156 ± 0.010 | −15.47 ± 1.75 | 96.31 ± 0.20 | 98.93 ± 0.55 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, A.A.; Atiya, A.; Akhtar, S.; Yadav, Y.; Qureshi, K.A.; Jaremko, M.; Mahmood, S. Optimization of a Cefuroxime Axetil-Loaded Liquid Self-Nanoemulsifying Drug Delivery System: Enhanced Solubility, Dissolution and Caco-2 Cell Uptake. Pharmaceutics 2022, 14, 772. https://doi.org/10.3390/pharmaceutics14040772
Khan AA, Atiya A, Akhtar S, Yadav Y, Qureshi KA, Jaremko M, Mahmood S. Optimization of a Cefuroxime Axetil-Loaded Liquid Self-Nanoemulsifying Drug Delivery System: Enhanced Solubility, Dissolution and Caco-2 Cell Uptake. Pharmaceutics. 2022; 14(4):772. https://doi.org/10.3390/pharmaceutics14040772
Chicago/Turabian StyleKhan, Arshad Ali, Akhtar Atiya, Safia Akhtar, Yogesh Yadav, Kamal A. Qureshi, Mariusz Jaremko, and Syed Mahmood. 2022. "Optimization of a Cefuroxime Axetil-Loaded Liquid Self-Nanoemulsifying Drug Delivery System: Enhanced Solubility, Dissolution and Caco-2 Cell Uptake" Pharmaceutics 14, no. 4: 772. https://doi.org/10.3390/pharmaceutics14040772
APA StyleKhan, A. A., Atiya, A., Akhtar, S., Yadav, Y., Qureshi, K. A., Jaremko, M., & Mahmood, S. (2022). Optimization of a Cefuroxime Axetil-Loaded Liquid Self-Nanoemulsifying Drug Delivery System: Enhanced Solubility, Dissolution and Caco-2 Cell Uptake. Pharmaceutics, 14(4), 772. https://doi.org/10.3390/pharmaceutics14040772







