The GluN2B-Selective Antagonist Ro 25-6981 Is Effective against PTZ-Induced Seizures and Safe for Further Development in Infantile Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Drugs
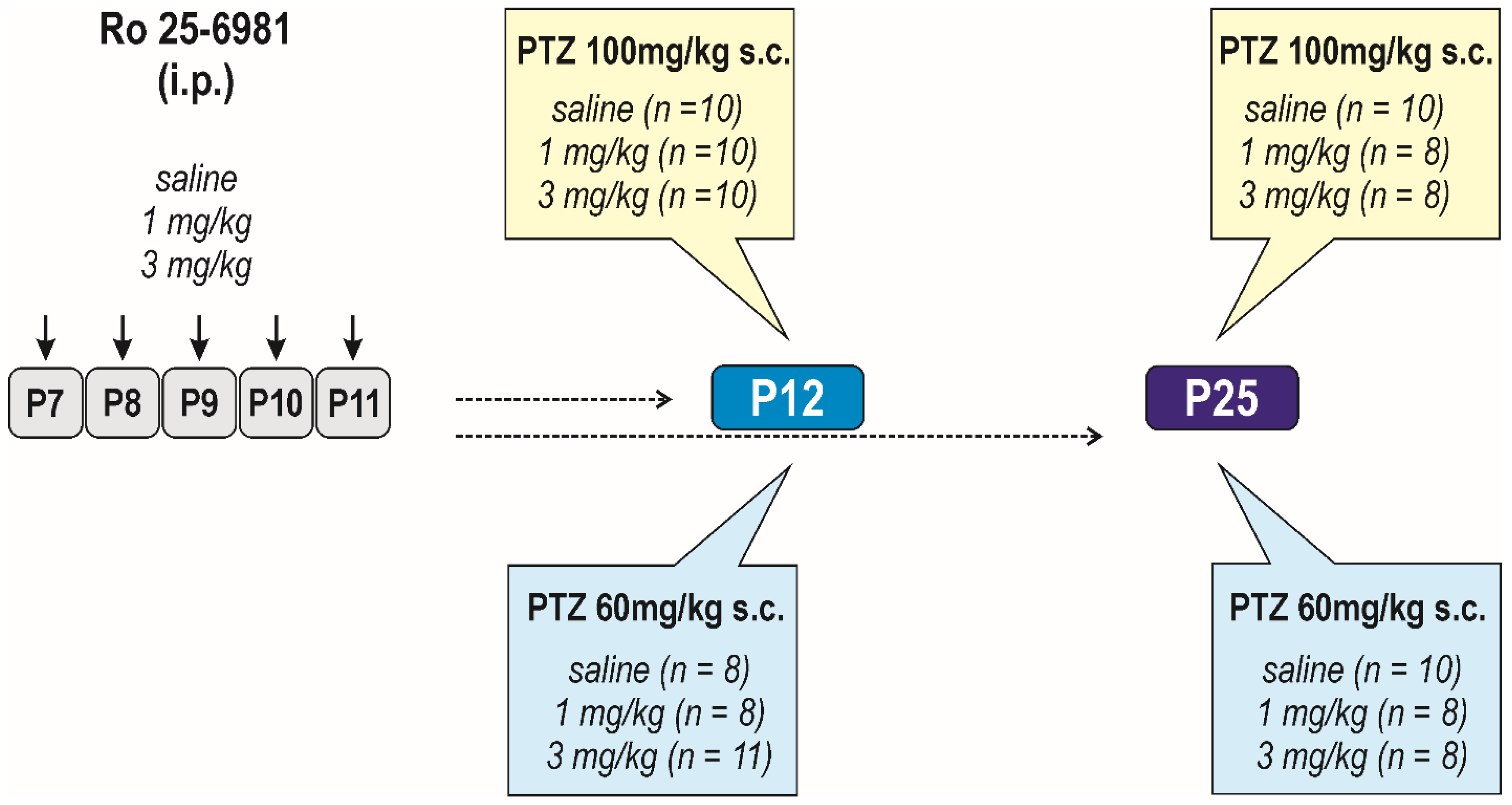
2.3. Experimental Design
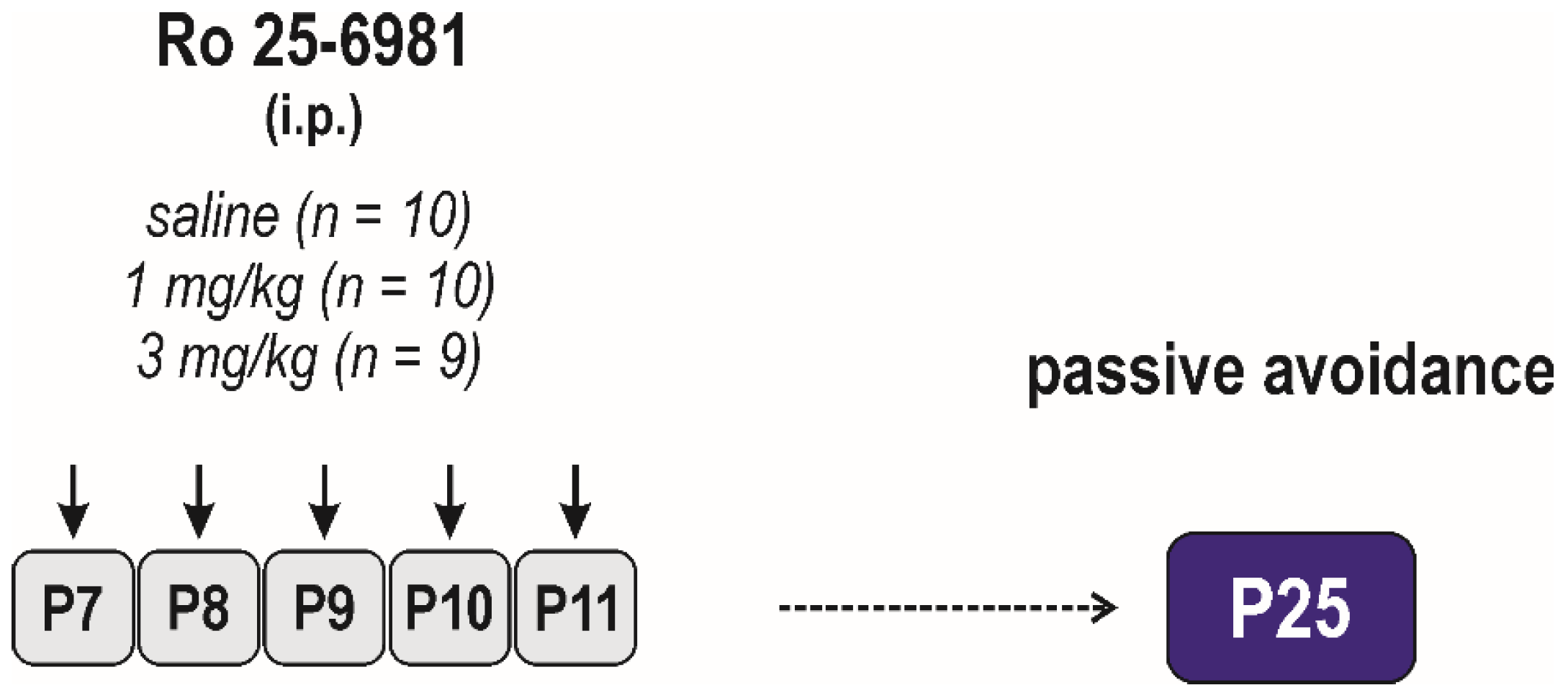
2.3.1. Acute Effects of Ro 25-6981
Anticonvulsant Effects
- 0—no changes;
- 0.5—abnormal behavior (e.g., automatisms, increased orienting reaction);
- 1—isolated myoclonic jerks;
- 2—typical minimal seizures or some parts of their symptomatology;
- 3—clonic seizures (mS) involving head and forelimb muscles with preserved righting reflexes (older term minimal metrazol seizures);
- 4—generalized seizures without the tonic phase (GCS);
- 5—complete generalized tonic-clonic seizures (GTCS).
Adverse Side Effects on Passive-Avoidance Responding
2.3.2. Effects of Early-Life Exposure to Ro 25-6981 on Brain Functions and Seizure Susceptibility
Long-Term Effects on Seizure Susceptibility
Long-Term Effects on Passive Avoidance Responding
Long-Term Effects on Sensorimotor Performance and the Open-Field Behavior
2.4. Statistics
3. Results
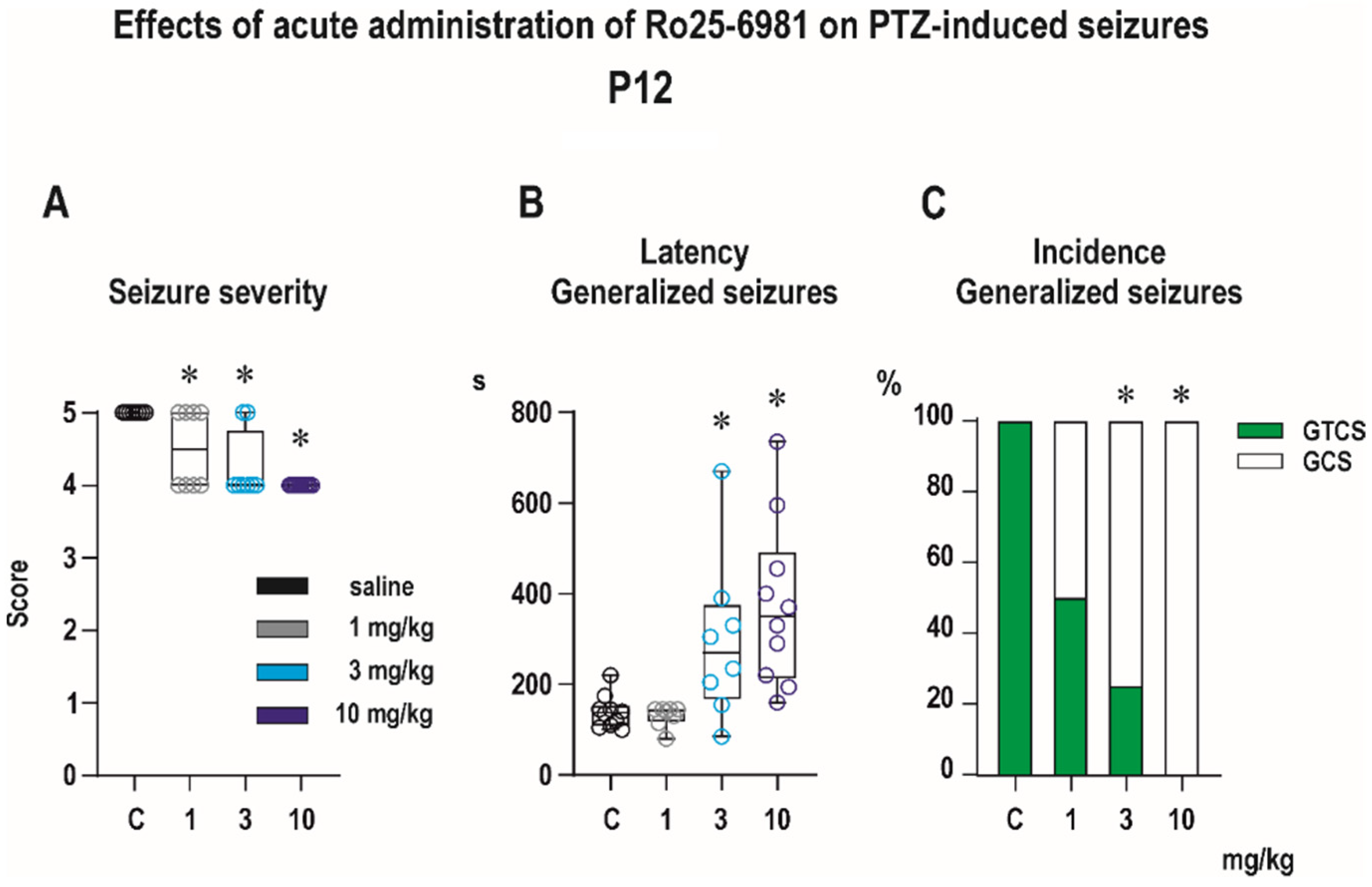
3.1. Acute Effects of Ro 25-6981
3.1.1. Anticonvulsant Effects
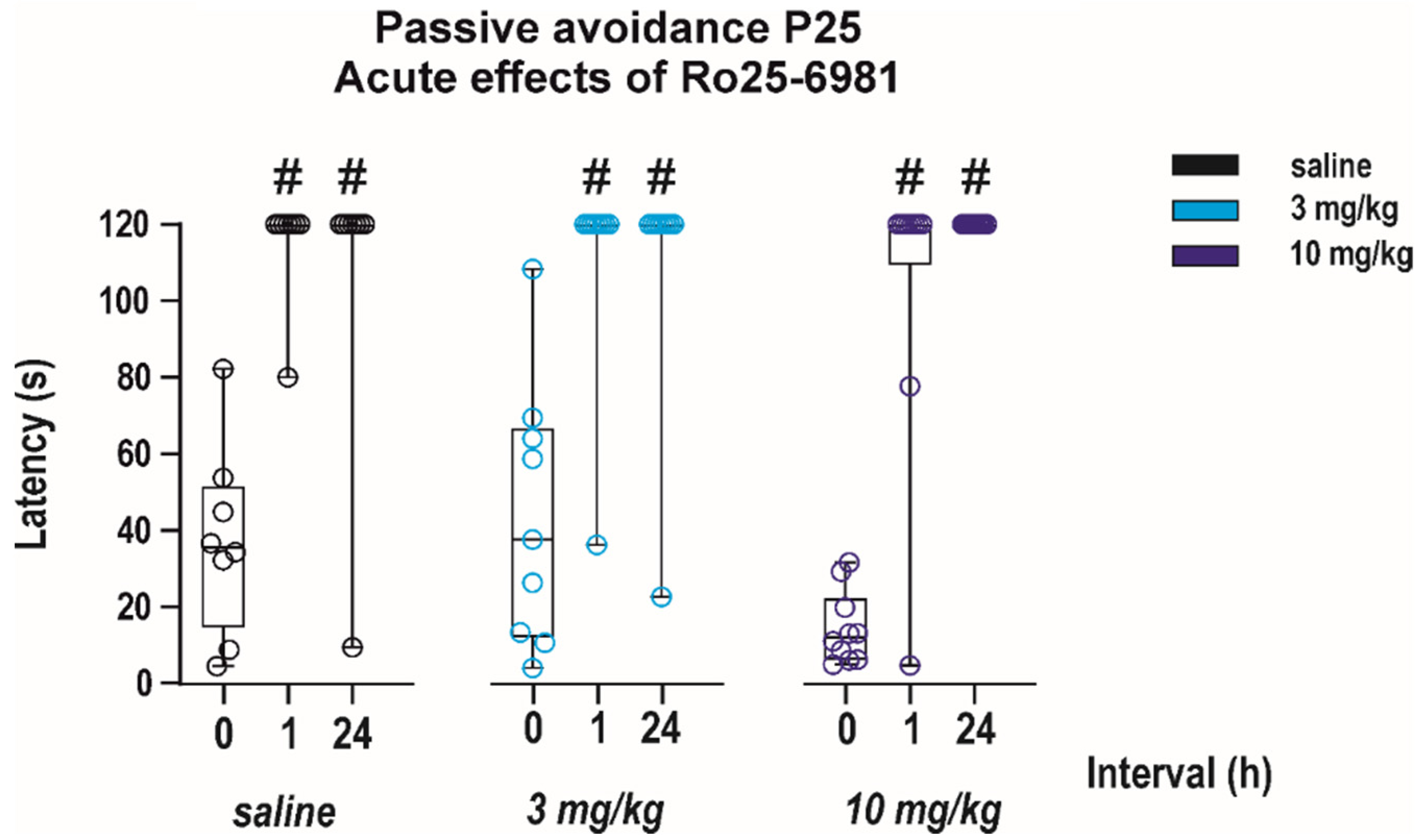
3.1.2. Adverse Side Effects on Passive Avoidance Responding
3.2. Effects of Early Life Exposure to Ro 25-6981 on Brain Functions and Excitability
3.2.1. Long-Term Effects on Seizure Susceptibility
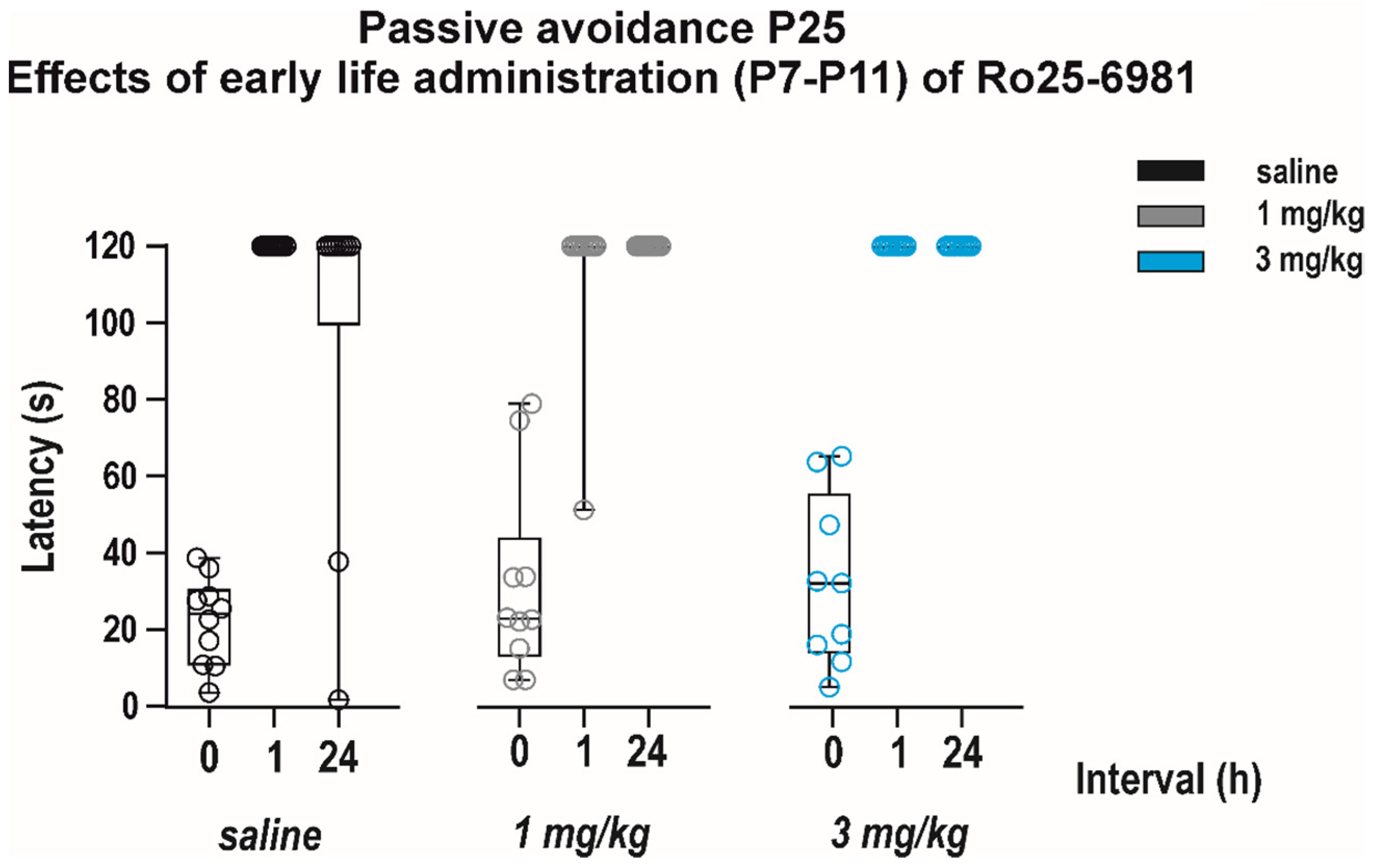
3.2.2. Long-Term Effects on Memory
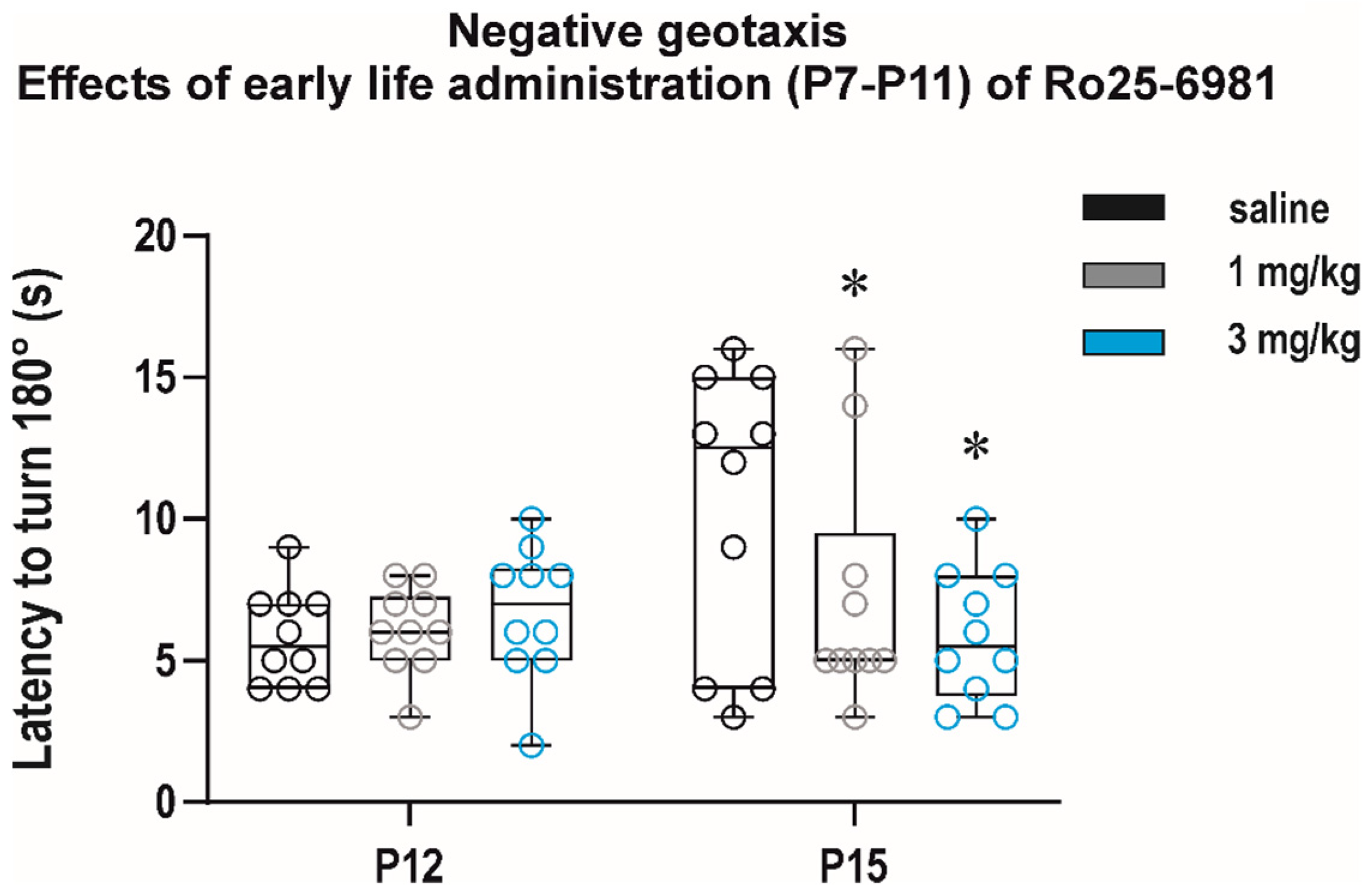
3.2.3. Long-Term Effects on Sensorimotor Development and Behavior in the Open Field
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hernan, A.E.; Holmes, G.L. Antiepileptic drug treatment strategies in neonatal epilepsy. Prog. Brain Res. 2016, 226, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Meador, K.J. Neurodevelopmental effects of antiepileptic drugs. Curr. Neurol. Neurosci. Rep. 2002, 2, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Kondratyev, A.; Tomita, Y.; Gale, K. Neurodevelopmental impact of antiepileptic drugs and seizures in the immature brain. Epilepsia 2007, 48 (Suppl. 5), 19–26. [Google Scholar] [CrossRef]
- Marsh, E.D.; Brooks-Kayal, A.R.; Porter, B.E. Seizures and antiepileptic drugs: Does exposure alter normal brain development? Epilepsia 2006, 47, 1999–2010. [Google Scholar] [CrossRef]
- Holmes, G.L. Drug Treatment of Epilepsy Neuropsychiatric Comorbidities in Children. Paediatr. Drugs 2021, 23, 55–73. [Google Scholar] [CrossRef]
- Wasterlain, C.G.; Gloss, D.S.; Niquet, J.; Wasterlain, A.S. Epileptogenesis in the developing brain. Handb. Clin. Neurol. 2013, 111, 427–439. [Google Scholar] [CrossRef]
- Cull-Candy, S.; Brickley, S.; Farrant, M. NMDA receptor subunits: Diversity, development and disease. Curr. Opin. Neurobiol. 2001, 11, 327–335. [Google Scholar] [CrossRef]
- Monyer, H.; Burnashev, N.; Laurie, D.J.; Sakmann, B.; Seeburg, P.H. Developmental and regional expression in the rat brain and functional properties of four NMDA receptors. Neuron 1994, 12, 529–540. [Google Scholar] [CrossRef]
- Szczurowska, E.; Mareš, P. NMDA and AMPA receptors: Development and status epilepticus. Physiol. Res. 2013, 62 (Suppl. 1), S21–S38. [Google Scholar] [CrossRef]
- Gallagher, M.J.; Huang, H.; Pritchett, D.B.; Lynch, D.R. Interactions between ifenprodil and the NR2B subunit of the N-methyl-D-aspartate receptor. J. Biol. Chem. 1996, 271, 9603–9611. [Google Scholar] [CrossRef]
- Williams, K. Ifenprodil discriminates subtypes of the N-methyl-D-aspartate receptor: Selectivity and mechanisms at recombinant heteromeric receptors. Mol. Pharmacol. 1993, 44, 851–859. [Google Scholar]
- McAllister, K.H. N-methyl-D-aspartate receptor antagonists and channel blockers have different effects upon a spinal seizure model in mice. Eur. J. Pharmacol. 1992, 211, 105–108. [Google Scholar] [CrossRef]
- Wang, X.M.; Bausch, S.B. Effects of distinct classes of N-methyl-D-aspartate receptor antagonists on seizures, axonal sprouting and neuronal loss in vitro: Suppression by NR2B-selective antagonists. Neuropharmacology 2004, 47, 1008–1020. [Google Scholar] [CrossRef]
- Yen, W.; Williamson, J.; Bertram, E.H.; Kapur, J. A comparison of three NMDA receptor antagonists in the treatment of prolonged status epilepticus. Epilepsy Res. 2004, 59, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Yourick, D.L.; Repasi, R.T.; Rittase, W.B.; Staten, L.D.; Meyerhoff, J.L. Ifenprodil and arcaine alter amygdala-kindling development. Eur. J. Pharmacol. 1999, 371, 147–152. [Google Scholar] [CrossRef]
- Mareš, P. Age and activation determines the anticonvulsant effect of ifenprodil in rats. Naunyn Schmiedebergs Arch. Pharmacol. 2014, 387, 753–761. [Google Scholar] [CrossRef] [PubMed]
- Mareš, P.; Mikulecká, A. Different effects of two N-methyl-D-aspartate receptor antagonists on seizures, spontaneous behavior, and motor performance in immature rats. Epilepsy Behav. 2009, 14, 32–39. [Google Scholar] [CrossRef]
- Mareš, P.; Tsenov, G.; Kubová, H. Anticonvulsant Action of GluN2A-Preferring Antagonist PEAQX in Developing Rats. Pharmaceutics 2021, 13, 415. [Google Scholar] [CrossRef]
- Fischer, G.; Mutel, V.; Trube, G.; Malherbe, P.; Kew, J.N.; Mohacsi, E.; Heitz, M.P.; Kemp, J.A. Ro 25-6981, a highly potent and selective blocker of N-methyl-D-aspartate receptors containing the NR2B subunit. Characterization in vitro. J. Pharmacol. Exp. Ther. 1997, 283, 1285–1292. [Google Scholar]
- Lynch, D.R.; Shim, S.S.; Seifert, K.M.; Kurapathi, S.; Mutel, V.; Gallagher, M.J.; Guttmann, R.P. Pharmacological characterization of interactions of RO 25-6981 with the NR2B (epsilon2) subunit. Eur. J. Pharmacol. 2001, 416, 185–195. [Google Scholar] [CrossRef]
- Dong, F.; Yao, R.; Yu, H.; Liu, Y. Neuroprotection of Ro 25-6981 Against Ischemia/Reperfusion-Induced Brain Injury via Inhibition of Autophagy. Cell. Mol. Neurobiol. 2017, 37, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Yellepeddi, V.K.; Zhudeva, M.Y.; Movahedi, F.; Vo, A.; Phan, J.; Kirsh, R.D.; Rawlins, D.B.; Talbot, J.N. Biopharmaceutical Characterization and Oral Efficacy of a New Rapid Acting Antidepressant Ro 25-6981. J. Pharm. Sci. 2018, 107, 2472–2478. [Google Scholar] [CrossRef]
- Boyce, S.; Wyatt, A.; Webb, J.K.; O’Donnell, R.; Mason, G.; Rigby, M.; Sirinathsinghji, D.; Hill, R.G.; Rupniak, N.M. Selective NMDA NR2B antagonists induce antinociception without motor dysfunction: Correlation with restricted localisation of NR2B subunit in dorsal horn. Neuropharmacology 1999, 38, 611–623. [Google Scholar] [CrossRef]
- Velíšek, L.; Kubová, H.; Pohl, M.; Staňková, L.; Mareš, P.; Schickerová, R. Pentylenetetrazol-induced seizures in rats: An ontogenetic study. Naunyn Schmiedebergs Arch. Pharmacol. 1992, 346, 588–591. [Google Scholar] [CrossRef] [PubMed]
- de Casrilevitz, M.; Engelhardt, E.; Esberard, C.A. Maturation of convulsogenic activity induced by leptazol in the albino rat. Br. J. Pharmacol. 1971, 42, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Brožičková, C.; Mikulecká, A.; Otahál, J. Effect of 7-Nitroindazole, a Neuronal Nitric Oxide Synthase Inhibitor, on Behavioral and Physiological Parameters. Physiol. Res. 2014, 63, 637–648. [Google Scholar] [CrossRef]
- Mikulecká, A.; Mareš, P. NMDA receptor antagonists impair motor performance in immature rats. Psychopharmacology 2002, 162, 364–372. [Google Scholar] [CrossRef]
- Hrncić, D.; Rasić-Marković, A.; Susić, V.; Djurić, D.; Stanojlović, O. Influence of NR2B-selective NMDA antagonist on lindane-induced seizures in rats. Pharmacology 2009, 84, 234–239. [Google Scholar] [CrossRef]
- Burket, J.A.; Cannon, W.R.; Jacome, L.F.; Deutsch, S.I. MK-801, a noncompetitive NMDA receptor antagonist, elicits circling behavior in the genetically inbred Balb/c mouse strain. Brain Res. Bull. 2010, 83, 337–339. [Google Scholar] [CrossRef]
- Tsuda, M.; Suzuki, T.; Misawa, M. Age-related decrease in the antiseizure effect of ifenprodil against pentylenetetrazole in mice. Brain Res. Dev. Brain Res. 1997, 104, 201–204. [Google Scholar] [CrossRef]
- Szczurowska, E.; Mareš, P. Different action of a specific NR2B/NMDA antagonist Ro 25-6981 on cortical evoked potentials and epileptic afterdischarges in immature rats. Brain Res. Bull. 2015, 111, 1–8. [Google Scholar] [CrossRef]
- Kolarová, A.; Ringer, R.; Tauber, M.G.; Leib, S.L. Blockade of NMDA receptor subtype NR2B prevents seizures but not apoptosis of dentate gyrus neurons in bacterial meningitis in infant rats. BMC Neurosci. 2003, 4, 21. [Google Scholar] [CrossRef]
- Loftis, J.M.; Janowsky, A. The N-methyl-D-aspartate receptor subunit NR2B: Localization, functional properties, regulation, and clinical implications. Pharmacol. Ther. 2003, 97, 55–85. [Google Scholar] [CrossRef]
- Klinger, W. Developmental pharmacology and toxicology: Biotransformation of drugs and other xenobiotics during postnatal development. Eur. J. Drug Metab. Pharm. 2005, 30, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Kato, R.; Takanaka, A. Effect of phenobarbital on electron transport system, oxidation and reduction of drugs in liver microsomes of rats of different age. J. Biochem. 1968, 63, 406–408. [Google Scholar] [PubMed]
- Schmitt, G.; Parrott, N.; Prinssen, E.; Barrow, P. The great barrier belief: The blood-brain barrier and considerations for juvenile toxicity studies. Reprod. Toxicol. 2017, 72, 129–135. [Google Scholar] [CrossRef]
- Soares, R.V.; Do, T.M.; Mabondzo, A.; Pons, G.; Chhun, S. Ontogeny of ABC and SLC transporters in the microvessels of developing rat brain. Fundam. Clin. Pharmacol. 2016, 30, 107–116. [Google Scholar] [CrossRef]
- Burket, J.A.; Mastropaolo, J.; Rosse, R.B.; Katz, E.U.; Deutsch, S.I. NMDA NR2B subtype-selective receptor antagonists fail to antagonize electrically-precipitated seizures and elicit popping in mice. Eur. Neuropsychopharmacol. 2010, 20, 207–210. [Google Scholar] [CrossRef]
- Browning, R.A.; Nelson, D.K. Modification of electroshock and pentylenetetrazol seizure patterns in rats after precollicular transections. Exp. Neurol. 1986, 93, 546–556. [Google Scholar] [CrossRef]
- Mareš, P.; Kubová, H. What is the role of neurotransmitter systems in cortical seizures? Physiol. Res. 2008, 57 (Suppl. 3), S111–S120. [Google Scholar] [CrossRef]
- Löscher, W. Animal Models of Seizures and Epilepsy: Past, Present, and Future Role for the Discovery of Antiseizure Drugs. Neurochem. Res. 2017, 42, 1873–1888. [Google Scholar] [CrossRef] [PubMed]
- Kubová, H.; Mareš, P. Partial agonist of benzodiazepine receptors Ro 19-8022 elicits withdrawal symptoms after short-term administration in immature rats. Physiol. Res. 2012, 61, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Chazot, P.L. The NMDA receptor NR2B subunit: A valid therapeutic target for multiple CNS pathologies. Curr. Med. Chem. 2004, 11, 389–396. [Google Scholar] [CrossRef]
- Kumar, A. NMDA Receptor Function During Senescence: Implication on Cognitive Performance. Front. Neurosci. Switz. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Dalton, G.L.; Wu, D.C.; Wang, Y.T.; Floresco, S.B.; Phillips, A.G. NMDA GluN2A and GluN2B receptors play separate roles in the induction of LTP and LTD in the amygdala and in the acquisition and extinction of conditioned fear. Neuropharmacology 2012, 62, 797–806. [Google Scholar] [CrossRef]
- Radiske, A.; Gonzalez, M.C.; Noga, D.A.; Rossato, J.I.; Bevilaqua, L.R.M.; Cammarota, M. GluN2B and GluN2A-containing NMDAR are differentially involved in extinction memory destabilization and restabilization during reconsolidation. Sci. Rep. 2021, 11, 186. [Google Scholar] [CrossRef]
- Kutsuwada, T.; Sakimura, K.; Manabe, T.; Takayama, C.; Katakura, N.; Kushiya, E.; Natsume, R.; Watanabe, M.; Inoue, Y.; Yagi, T.; et al. Impairment of suckling response, trigeminal neuronal pattern formation, and hippocampal LTD in NMDA receptor epsilon 2 subunit mutant mice. Neuron 1996, 16, 333–344. [Google Scholar] [CrossRef]
- Bhardwaj, S.K.; Forcelli, P.A.; Palchik, G.; Gale, K.; Srivastava, L.K.; Kondratyev, A. Neonatal exposure to phenobarbital potentiates schizophrenia-like behavioral outcomes in the rat. Neuropharmacology 2012, 62, 2337–2345. [Google Scholar] [CrossRef]
- Mikulecká, A.; Šubrt, M.; Pařízková, M.; Mareš, P.; Kubová, H. Consequences of early postnatal benzodiazepines exposure in rats. II. Social behavior. Front. Behav. Neurosci. 2014, 8, 169. [Google Scholar] [CrossRef]
- Mikulecká, A.; Šubrt, M.; Stuchlík, A.; Kubová, H. Consequences of early postnatal benzodiazepines exposure in rats. I. Cognitive-like behavior. Front. Behav. Neurosci. 2014, 8, 101. [Google Scholar] [CrossRef]
- Furuie, H.; Yamada, K.; Ichitani, Y. Differential effects of N-methyl-D-aspartate receptor blockade during the second and third postnatal weeks on spatial working and reference memory in adult rats. Brain Res. 2019, 1721, 146339. [Google Scholar] [CrossRef] [PubMed]
- McLamb, R.L.; Williams, L.R.; Nanry, K.P.; Wilson, W.A.; Tilson, H.A. MK-801 impedes the acquisition of a spatial memory task in rats. Pharmacol. Biochem. Behav. 1990, 37, 41–45. [Google Scholar] [CrossRef]
- Clancy, B.; Darlington, R.B.; Finlay, B.L. Translating developmental time across mammalian species. Neuroscience 2001, 105, 7–17. [Google Scholar] [CrossRef]
- Workman, A.D.; Charvet, C.J.; Clancy, B.; Darlington, R.B.; Finlay, B.L. Modeling transformations of neurodevelopmental sequences across mammalian species. J. Neurosci. 2013, 33, 7368–7383. [Google Scholar] [CrossRef]
- Ikonomidou, C.; Bosch, F.; Miksa, M.; Bittigau, P.; Vöckler, J.; Dikranian, K.; Tenkova, T.I.; Stefovska, V.; Turski, L.; Olney, J.W. Blockade of NMDA receptors and apoptotic neurodegeneration in the developing brain. Science 1999, 283, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Kawabe, K. Effects of chronic forced-swim stress on behavioral properties in rats with neonatal repeated MK-801 treatment. Pharmacol. Biochem. Behav. 2017, 159, 48–54. [Google Scholar] [CrossRef]
- Uehara, T.; Sumiyoshi, T.; Seo, T.; Itoh, H.; Matsuoka, T.; Suzuki, M.; Kurachi, M. Long-term effects of neonatal MK-801 treatment on prepulse inhibition in young adult rats. Psychopharmacology 2009, 206, 623–630. [Google Scholar] [CrossRef]
- Lima-Ojeda, J.M.; Vogt, M.A.; Pfeiffer, N.; Dormann, C.; Köhr, G.; Sprengel, R.; Gass, P.; Inta, D. Pharmacological blockade of GluN2B-containing NMDA receptors induces antidepressant-like effects lacking psychotomimetic action and neurotoxicity in the perinatal and adult rodent brain. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 45, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Mysliveček, J.; Hassmannová, J. Step-down passive avoidance in the rat ontogeny. Acta Neurobiol. Exp. 1991, 51, 89–96. [Google Scholar]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
- Crawley, J.N. Behavioral phenotyping strategies for mutant mice. Neuron 2008, 57, 809–818. [Google Scholar] [CrossRef]
- Altman, J.; Sudarshan, K. Postnatal development of locomotion in the laboratory rat. Anim. Behav. 1975, 23, 896–920. [Google Scholar] [CrossRef]
- Geisler, H.C.; Westerga, J.; Gramsbergen, A. Development of posture in the rat. Acta Neurobiol. Exp. 1993, 53, 517–523. [Google Scholar]
- Gramsbergen, A. Posture and locomotion in the rat: Independent or interdependent development? Neurosci. Biobehav. Rev. 1998, 22, 547–553. [Google Scholar] [CrossRef]
- Jänicke, B.; Schulze, G.; Coper, H. Motor performance achievements in rats of different ages. Exp. Gerontol. 1983, 18, 393–407. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mareš, P.; Kozlová, L.; Mikulecká, A.; Kubová, H. The GluN2B-Selective Antagonist Ro 25-6981 Is Effective against PTZ-Induced Seizures and Safe for Further Development in Infantile Rats. Pharmaceutics 2021, 13, 1482. https://doi.org/10.3390/pharmaceutics13091482
Mareš P, Kozlová L, Mikulecká A, Kubová H. The GluN2B-Selective Antagonist Ro 25-6981 Is Effective against PTZ-Induced Seizures and Safe for Further Development in Infantile Rats. Pharmaceutics. 2021; 13(9):1482. https://doi.org/10.3390/pharmaceutics13091482
Chicago/Turabian StyleMareš, Pavel, Lucie Kozlová, Anna Mikulecká, and Hana Kubová. 2021. "The GluN2B-Selective Antagonist Ro 25-6981 Is Effective against PTZ-Induced Seizures and Safe for Further Development in Infantile Rats" Pharmaceutics 13, no. 9: 1482. https://doi.org/10.3390/pharmaceutics13091482
APA StyleMareš, P., Kozlová, L., Mikulecká, A., & Kubová, H. (2021). The GluN2B-Selective Antagonist Ro 25-6981 Is Effective against PTZ-Induced Seizures and Safe for Further Development in Infantile Rats. Pharmaceutics, 13(9), 1482. https://doi.org/10.3390/pharmaceutics13091482






