Current Trends on Protein Driven Bioinks for 3D Printing
Abstract
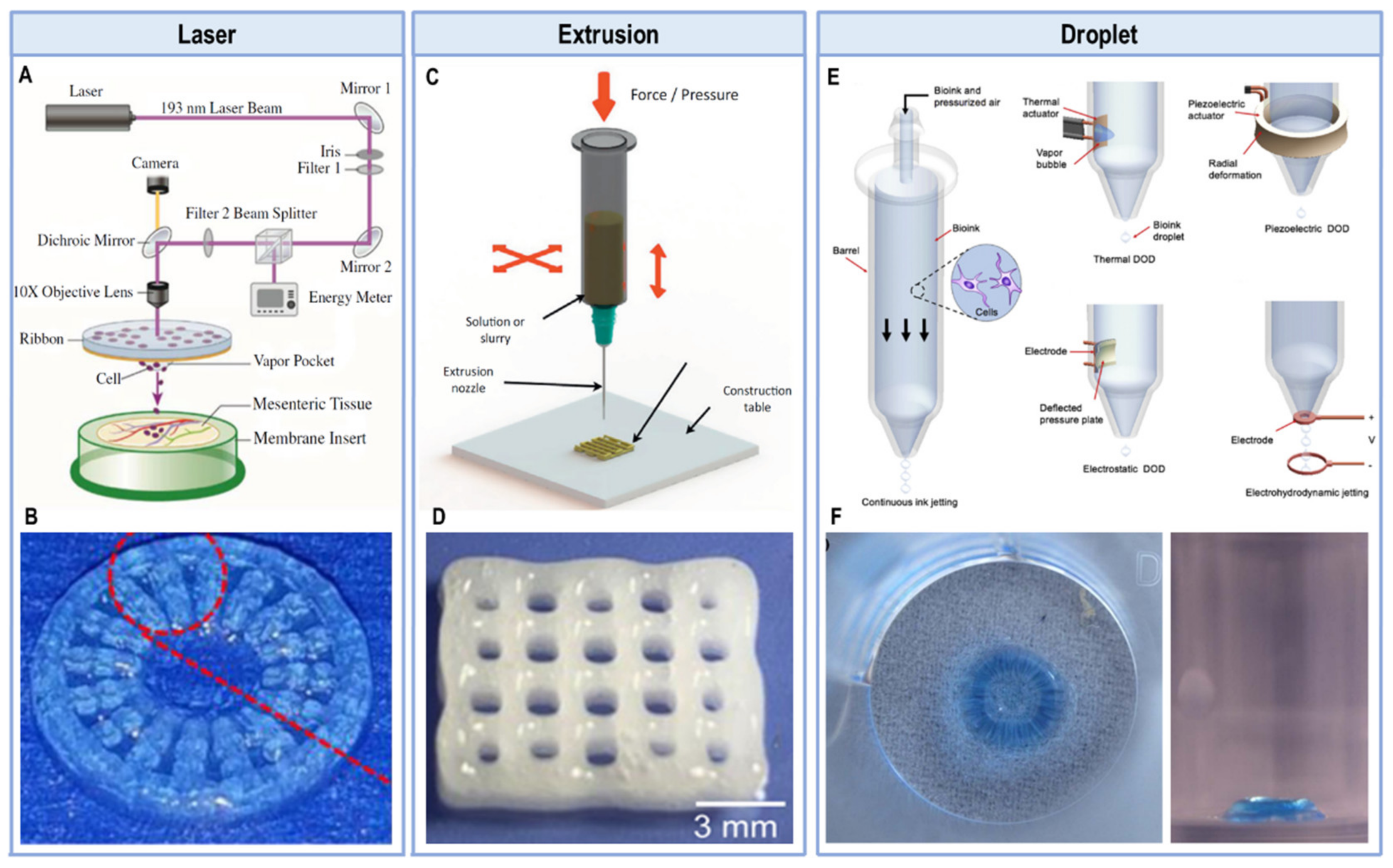
:1. Introduction
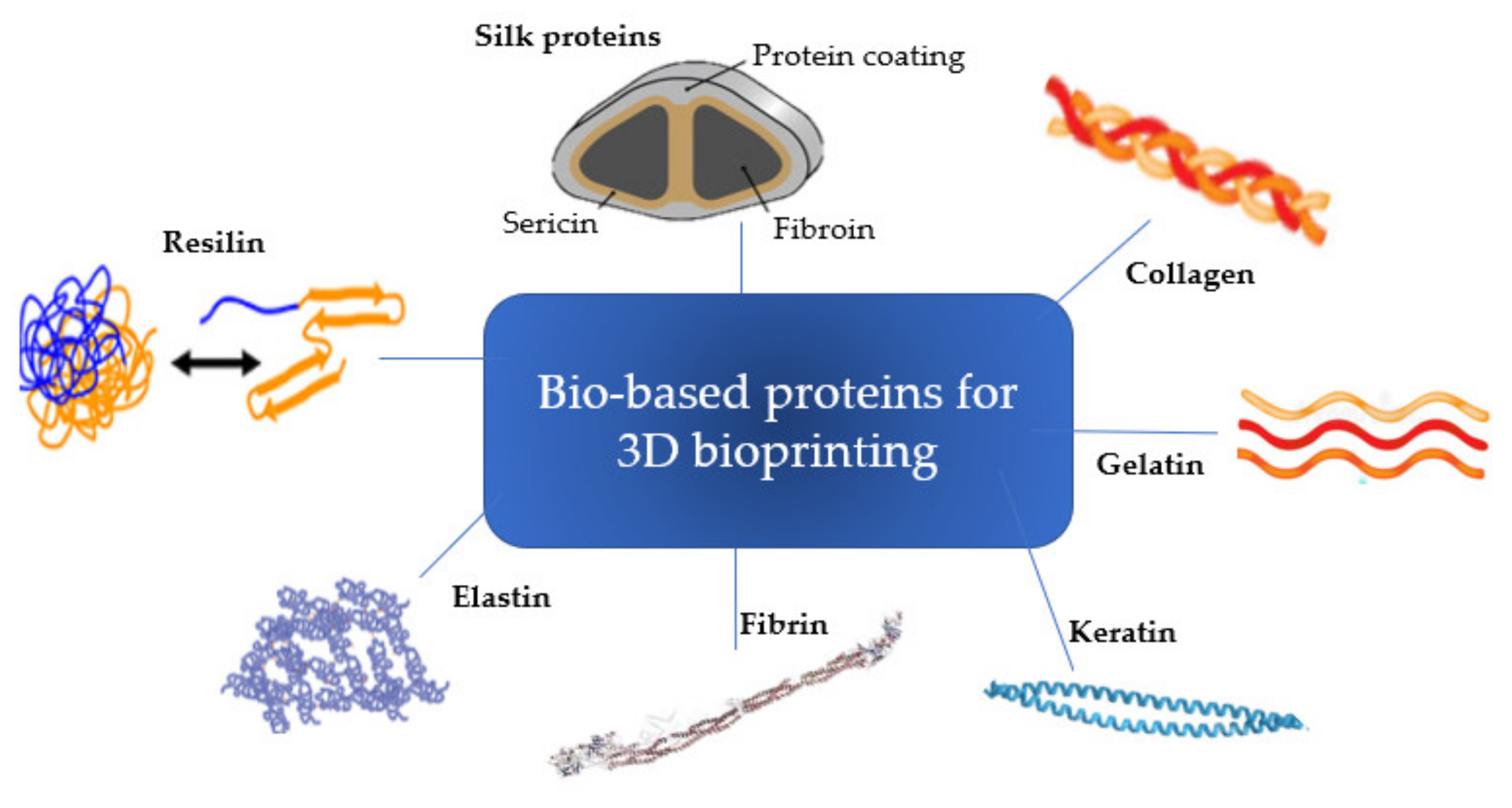
2. Protein-Based Bioinks
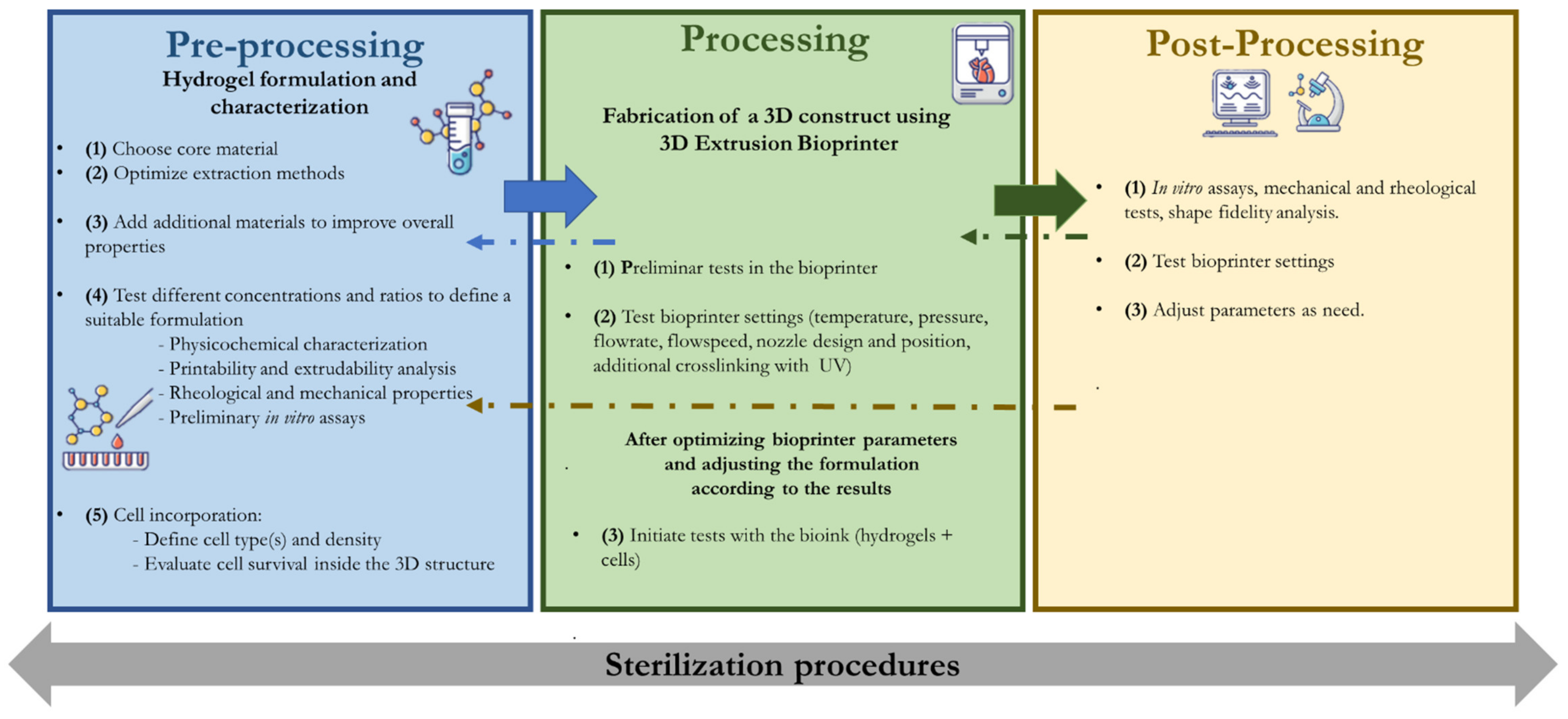
2.1. Pre-Processing Methods
2.1.1. Bioink Formulation
| Protein | Origin | Extraction | Structure | Amino Acid Sequence | Main Characteristics | Main Applications | Main Challenges for 3D Bioprinting | Ref. |
|---|---|---|---|---|---|---|---|---|
| Collagen | Animal tissue, including tendons, ligaments and skin | - Neutral saline solutions, acid solutions, and acid solutions with added enzymes. | - trimeric molecules (polypeptide α-chains) - triple helix-tertiary structure | - (GPX)n, where X is any amino acid other than glycine, proline or hydroxyproline | - high tensile strength and flexibility - cell adhesive cues - low mechanical properties Gelatin: - improved biodegradability and biocompatibility - Temperature responsive | - Skin tissue engineering (bone, cartilage, skin, liver, nervous system models, cornea) - Food additives - Cosmetics | - low mechanical properties | [28,29] |
| Gelatin | Obtained by partially hydrolyzing collagen under the action of an enzyme such as neutrase or under the action of an acid or an alkaline. | - random coiled domains | - poor rheological properties | [3,40,54] | ||||
| Keratin | - wool, horn, hair, hooves, shells, beaks, fingernails, toenails, feathers, and claws | - by oxidative and reductive chemistry | Twisted helices | - CCXPX and CCXS(T)S(T) | -high stability - low solubility | - Cosmetic (hair products) - Soft tissue regeneration | - low extensibility - insolubility | [33,34] |
| Fibrin | - blood | - isolation and concentration of blood fibrinogen by centrifugation combined with cryoprecipitation. | - (GPRP)n and (GHRP)n | - elastic and viscous properties - Short gelation time | fibrin-based sealants | - suitable crosslinking required - poor mechanical properties and rapid degradation | [30,31,32,61] | |
| Elastin | - lungs, blood vessels, aorta, and skin | Animal-derived tropoelastin, recombinant production | β-spirals | - (VAPGVG)n | - flexibility | - Skin tissue engineering (vascular grafts, heart valves and elastic cartilage) | - elastin purification is required. During this purification process, contaminations often take place. | [62,63,77] |
| Resilin | - insect cuticles | β-turns | -(AQTPSSQYGAP)n | - rubber-elasticity - high mechanical properties | - conductive, elastic and adhesive hydrogels suitable as biosensor | - difficult to identify the primary sequence and molecular structure of resilin due to the reduced stability during purification | [49,50,78] | |
| Silk Fibroin | Domestic silkworms: Bombyx mori; wild silkworms: Antheraea pernyi and Samia cynthia ricini; Spiders: Nephila clavipes and Araneus diadematus; recombinant silk proteins in different host systems | - Dissolving in a concentrated solution of lithium bromide or in a ternary solvent system of calcium chloride/ethanol/water | β-sheet structures connected by amorphous links | - Silk fiber: (GGXaa)n - Xaa = A, Y, L or Q - predominance of glycine and alanine in fibroin predominance of serine in sericin | - structural integrity, | - Skin tissue engineering - sutures | - low mechanical parameters, and high enzymatic degradation rate. | [26,55,66,67] |
| Silk Sericin | - reaction types (acidic, alkaline, enzymatic) and different conditions (time, temperature, pressure, pH) | amorphous random spiral/β-sheets | - antioxidant, antibacterial, UV-resistant, and ability to release moisturizing factors | - Cosmetic (creams) - Skin tissue engineering - Bone tissue engineering (regulate hydroxyapatite biomineralization process) | - poor structural integrity | [51,68] | ||
| Decellularized extracellular matrix (dECM) | allograft and xenograft tissue | Combination of decellularization methods (chemical, biological and physical) applied using different tecnhqiues (perfusion decellularization, pressuregradient, supercritical fluid, or immersion and agitation) | n/a | n/a | - Improves and regulate cellular functions of specific tissue duo to naturally present growth and differentiation factors - Decrease risk of immune-mediated rejection | - In vitro diseased tissue models - Tissue repair - Tissue replacement | - define the most suitable formulations | [79,80,81] |
2.1.2. Extraction Methods
2.1.3. Preliminary Evaluation Techniques
2.1.4. Crosslinkers
2.1.5. Conjugation with Other Molecules/Polymers
2.1.6. Rheological Properties
2.2. Processing and Pos-Processing
2.2.1. Printing Parameters
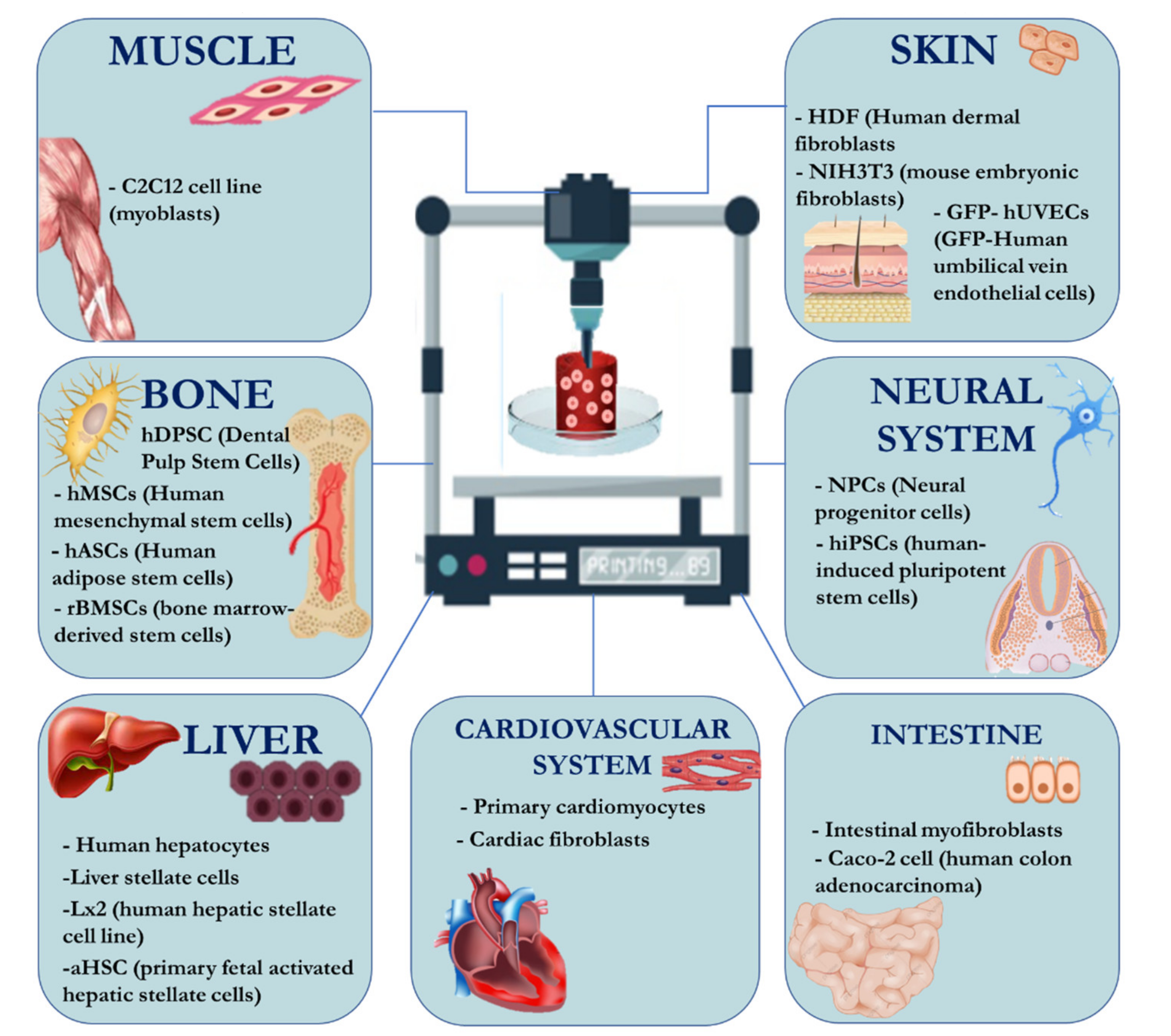
2.2.2. Adding Cells to the System
3. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Schröder, H.C.; Wang, X.; Tremel, W.; Ushijima, H.; Müller, W.E.G. Biofabrication of biosilica-glass by living organisms. Nat. Prod. Rep. 2008, 25, 455–474. [Google Scholar] [CrossRef]
- Groll, J.; Boland, T.; Blunk, T.; Burdick, J.A.; Cho, D.W.; Dalton, P.D.; Derby, B.; Forgacs, G.; Li, Q.; Mironov, V.A.; et al. Biofabrication: Reappraising the definition of an evolving field. Biofabrication 2016, 8, 013001. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, E.M.; Gómez-Blanco, J.C.; Nieto, E.L.; Casado, J.G.; Macías-García, A.; Díez, M.A.; Pagador, J.B. Hydrogels for bioprinting: A systematic review of hydrogels synthesis, bioprinting parameters, and bioprinted structures behavior. Front. Bioeng. Biotechnol. 2020, 8. [Google Scholar] [CrossRef]
- Silva, L.P. Current Trends and Challenges in Biofabrication Using Biomaterials and Nanomaterials: Future Perspectives for 3D/4D Bioprinting. In 3D and 4D Printing in Biomedical Applications: Process Engineering and Additive Manufacturing; Wiley: Hoboken, NJ, USA, 2018. [Google Scholar] [CrossRef]
- De Santis, M.M.; Alsafadi, H.N.; Tas, S.; Bölükbas, D.A.; Prithiviraj, S.; Da Silva, I.A.; Wagner, D.E. Extracellular-matrix-reinforced bioinks for 3D bioprinting human tissue. Adv. Mater. 2021, 33, 2005476. [Google Scholar] [CrossRef]
- Luo, Y.; Pan, H.; Jiang, J.; Zhao, C.; Zhang, J.; Chen, P.; Lin, X.; Fan, S. Desktop-Stereolithography 3D Printing of a Polyporous Extracellular Matrix Bioink for Bone Defect Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 589094. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Lee, H.; Lee, J.; Atala, A.; Yoo, J.J.; Lee, S.J.; Kim, G.H. Efficient myotube formation in 3D bioprinted tissue construct by biochemical and topographical cues. Biomaterials 2019, 230, 119632. [Google Scholar] [CrossRef] [PubMed]
- Carlberg, B.; Wang, T.; Liu, J. Direct Photolithographic Patterning of Electrospun Films for Defined Nanofibrillar Microarchitectures. Langmuir 2010, 26, 2235–2239. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, G.; Chen, L.; Li, H.; Yin, T.; Wang, B.; Lee, J.C.-M.; Yu, Q. Electrospun nanofiber meshes with tailored architectures and patterns as potential tissue-engineering scaffolds. Biofabrication 2009, 1, 015001. [Google Scholar] [CrossRef]
- Landers, R.; Hübner, U.; Schmelzeisen, R.; Mülhaupt, R. Rapid prototyping of scaffolds derived from thermoreversible hydrogels and tailored for applications in tissue engineering. Biomaterials 2002, 23, 4437–4447. [Google Scholar] [CrossRef]
- Moroni, L.; Boland, T.; Burdick, J.A.; De Maria, C.; Derby, B.; Forgacs, G.; Groll, J.; Li, Q.; Malda, J.; Mironov, V.A.; et al. Biofabrication: A Guide to Technology and Terminology. Trends Biotechnol. 2017, 36, 384–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sidorov, V.Y.; Samson, P.C.; Sidorova, T.N.; Davidson, J.; Lim, C.C.; Wikswo, J.P. I-Wire Heart-on-a-Chip I: Three-dimensional cardiac tissue constructs for physiology and pharmacology. Acta Biomater. 2016, 48, 68–78. [Google Scholar] [CrossRef] [Green Version]
- Pati, F.; Ha, D.-H.; Jang, J.; Han, H.H.; Rhie, J.-W.; Cho, D.-W. Biomimetic 3D tissue printing for soft tissue regeneration. Biomaterials 2015, 62, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-J.; Kim, T.G.; Jeong, J.; Yi, H.-G.; Park, J.W.; Hwang, W.; Cho, D.-W. 3D Cell Printing of Functional Skeletal Muscle Constructs Using Skeletal Muscle-Derived Bioink. Adv. Healthc. Mater. 2016, 5, 2636–2645. [Google Scholar] [CrossRef]
- Mao, Q.; Wang, Y.; Li, Y.; Juengpanich, S.; Li, W.; Chen, M.; Yin, J.; Fu, J.; Cai, X. Fabrication of liver microtissue with liver decellularized extracellular matrix (dECM) bioink by digital light processing (DLP) bioprinting. Mater. Sci. Eng. C 2020, 109, 110625. [Google Scholar] [CrossRef]
- Daly, A.; Davidson, M.D.; Burdick, J.A. 3D bioprinting of high cell-density heterogeneous tissue models through spheroid fusion within self-healing hydrogels. Nat. Commun. 2021, 12, 1–13. [Google Scholar] [CrossRef]
- Yan, W.-C.; Davoodi, P.; Vijayavenkataraman, S.; Tian, Y.; Ng, W.C.; Fuh, J.Y.; Robinson, K.S.; Wang, C.-H. 3D bioprinting of skin tissue: From pre-processing to final product evaluation. Adv. Drug Deliv. Rev. 2018, 132, 270–295. [Google Scholar] [CrossRef]
- Włodarczyk-Biegun, M.K.; del Campo, A. 3D bioprinting of structural proteins. Biomaterials 2017, 134, 180–201. [Google Scholar] [CrossRef] [PubMed]
- Groll, J.; Burdick, J.A.; Cho, D.W.; Derby, B.; Gelinsky, M.; Heilshorn, S.C.; Jüngst, T.; Malda, J.; Mironov, V.A.; Nakayama, K.; et al. A definition of bioinks and their distinction from biomaterial inks. Biofabrication 2018, 11, 013001. [Google Scholar] [CrossRef] [PubMed]
- Crowder, S.W.; Leonardo, V.; Whittaker, T.E.; Papathanasiou, P.; Stevens, M.M. Material Cues as Potent Regulators of Epigenetics and Stem Cell Function. Cell Stem Cell 2016, 18, 39–52. [Google Scholar] [CrossRef] [Green Version]
- Gungor-Ozkerim, P.S.; Inci, I.; Zhang, Y.S.; Khademhosseini, A.; Dokmeci, M.R. Bioinks for 3D bioprinting: An overview. Biomater. Sci. 2018, 6, 915–946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loo, Y.; Lakshmanan, A.; Ni, M.; Toh, L.L.; Wang, S.; Hauser, C.A.E. Peptide Bioink: Self-Assembling Nanofibrous Scaffolds for Three-Dimensional Organotypic Cultures. Nano Lett. 2015, 15, 6919–6925. [Google Scholar] [CrossRef]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2018, 7, 843–855. [Google Scholar] [CrossRef]
- Adamiak, K.; Sionkowska, A. Current methods of collagen cross-linking: Review. Int. J. Biol. Macromol. 2020, 161, 550–560. [Google Scholar] [CrossRef]
- Khoeini, R.; Nosrati, H.; Akbarzadeh, A.; Eftekhari, A.; Kavetskyy, T.; Khalilov, R.; Ahmadian, E.; Nasibova, A.; Datta, P.; Roshangar, L.; et al. Natural and Synthetic Bioinks for 3D Bioprinting. Adv. NanoBiomed Res. 2021, 1, 2000097. [Google Scholar] [CrossRef]
- Veiga, A.; Castro, F.; Rocha, F.; Oliveira, A. Protein-Based Hydroxyapatite Materials: Tuning Composition toward Biomedical Applications. ACS Appl. Bio Mater. 2020, 3, 3441–3455. [Google Scholar] [CrossRef]
- Abascal, N.C.; Regan, L. The past, present and future of protein-based materials. Open Biol. 2018, 8, 180113. [Google Scholar] [CrossRef] [Green Version]
- Khan, R.; Khan, M.H. Use of collagen as a biomaterial: An update. J. Indian Soc. Periodontol. 2013, 17, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Osidak, E.O.; Kozhukhov, V.I.; Osidak, M.S.; Domogatskiy, S.P. Collagen as Bioink for Bioprinting: A Comprehensive Review. Int. J. Bioprinting 2020, 6, 270. [Google Scholar] [CrossRef]
- Sharma, R.; Smits, I.P.M.; De La Vega, L.; Lee, C.; Willerth, S.M. 3D Bioprinting Pluripotent Stem Cell Derived Neural Tissues Using a Novel Fibrin Bioink Containing Drug Releasing Microspheres. Front. Bioeng. Biotechnol. 2020, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Kim, D.S.; Jang, I.H.; Kim, H.-R.; Kang, H.-W. Bioprinting of three-dimensional dentin–pulp complex with local differentiation of human dental pulp stem cells. J. Tissue Eng. 2019, 10, 2041731419845849. [Google Scholar] [CrossRef]
- Wang, Z.; Lee, S.J.; Cheng, H.-J.; Yoo, J.J.; Atala, A. 3D bioprinted functional and contractile cardiac tissue constructs. Acta Biomater. 2018, 70, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Placone, J.K.; Navarro, J.; Laslo, G.W.; Lerman, M.J.; Gabard, A.R.; Herendeen, G.J.; Fisher, J.P. Development and characterization of a 3D printed, keratin-based hydrogel. Ann. Biomed. Eng. 2017, 45, 237–248. [Google Scholar] [CrossRef]
- Kwak, H.; Shin, S.; Lee, H.; Hyun, J. Formation of a keratin layer with silk fibroin-polyethylene glycol composite hydrogel fabricated by digital light processing 3D printin. J. Ind. Eng. Chem. 2018, 72, 232–240. [Google Scholar] [CrossRef]
- Ahn, G.; Min, K.-H.; Kim, C.; Lee, J.-S.; Kang, D.; Won, J.-Y.; Cho, D.-W.; Kim, J.-Y.; Jin-Hyung, S.; Kyung-Hyun, M.; et al. Precise stacking of decellularized extracellular matrix based 3D cell-laden constructs by a 3D cell printing system equipped with heating modules. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Toprakhisar, B.; Nadernezhad, A.; Bakirci, E.; Khani, N.; Skvortsov, G.A.; Koc, B. Development of Bioink from Decellularized Tendon Extracellular Matrix for 3D Bioprinting. Macromol. Biosci. 2018, 18, e1800024. [Google Scholar] [CrossRef] [PubMed]
- Benwood, C.; Chrenek, J.; Kirsch, R.; Masri, N.; Richards, H.; Teetzen, K.; Willerth, S. Natural Biomaterials and Their Use as Bioinks for Printing Tissues. Bioengineering 2021, 8, 27. [Google Scholar] [CrossRef]
- Sakai, S.; Yoshii, A.; Sakurai, S.; Horii, K.; Nagasuna, O. Silk fibroin nanofibers: A promising ink additive for extrusion three-dimensional bioprinting. Mater. Today Bio 2020, 8, 100078. [Google Scholar] [CrossRef]
- Diamantides, N.; Wang, L.; Pruiksma, T.; Siemiatkoski, J.; Dugopolski, C.; Shortkroff, S.; Kennedy, S.; Bonassar, L.J. Correlating rheological properties and printability of collagen bioinks: The effects of riboflavin photocrosslinking and pH. Biofabrication 2017, 9, 034102. [Google Scholar] [CrossRef]
- Mirzaei, M.; Okoro, O.; Nie, L.; Petri, D.; Shavandi, A. Protein-Based 3D Biofabrication of Biomaterials. Bioengineering 2021, 8, 48. [Google Scholar] [CrossRef]
- Gillispie, G.; Prim, P.; Copus, J.; Fisher, J.; Mikos, A.G.; Yoo, J.J.; Atala, A.; Lee, S.J. Assessment methodologies for extrusion-based bioink printability. Biofabrication 2020, 12, 022003. [Google Scholar] [CrossRef] [PubMed]
- Mobaraki, M.; Ghaffari, M.; Yazdanpanah, A.; Luo, Y.; Mills, D. Bioinks and bioprinting: A focused review. Bioprinting 2020, 18, e00080. [Google Scholar] [CrossRef]
- Burks, H.E.; Phamduy, T.B.; Azimi, M.S.; Saksena, J.; Burow, M.E.; Collins-Burow, B.M.; Chrisey, D.B.; Murfee, W.L. Laser Direct-Write Onto Live Tissues: A Novel Model for Studying Cancer Cell Migration. J. Cell. Physiol. 2016, 231, 2333–2338. [Google Scholar] [CrossRef] [Green Version]
- Visscher, D.O.; Lee, H.; van Zuijlen, P.P.; Helder, M.N.; Atala, A.; Yoo, J.J.; Lee, S.J. A photo-crosslinkable cartilage-derived extracellular matrix bioink for auricular cartilage tissue engineering. Acta Biomater. 2020, 121, 193–203. [Google Scholar] [CrossRef]
- Gudapati, H.; Dey, M.; Ozbolat, I. A comprehensive review on droplet-based bioprinting: Past, present and future. Biomaterials 2016, 102, 20–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faulkner-Jones, A.; Fyfe, C.; Cornelissen, D.-J.; Gardner, J.; King, J.; Courtney, A.; Shu, W. Bioprinting of human pluripotent stem cells and their directed differentiation into hepatocyte-like cells for the generation of mini-livers in 3D. Biofabrication 2015, 7, 044102. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Kim, H.J.; Wong, C.; Vepari, C.; Matsumoto, A.; Kaplan, D.L. Fibrous proteins and tissue engineering. Mater. Today 2006, 9, 44–53. [Google Scholar] [CrossRef]
- Veiga, A.; Castro, F.; Reis, C.C.; Sousa, A.; Oliveira, A.L.; Rocha, F. Hydroxyapatite/sericin composites: A simple synthesis route under near-physiological conditions of temperature and pH and preliminary study of the effect of sericin on the biomineralization process. Mater. Sci. Eng. C 2019, 108, 110400. [Google Scholar] [CrossRef]
- McGann, C.L.; Levenson, E.; Kiick, K.L. Resilin-Based Hybrid Hydrogels for Cardiovascular Tissue Engineering. Macromol. Chem. Phys. 2012, 214, 203–213. [Google Scholar] [CrossRef] [Green Version]
- Li, L.; Tong, Z.; Jia, X.; Kiick, K.L. Resilin-like polypeptide hydrogels engineered for versatile biological function. Soft Matter 2012, 9, 665–673. [Google Scholar] [CrossRef] [Green Version]
- Punyamoonwongsa, P.; Klayya, S.; Sajomsang, W.; Kunyanee, C.; Aueviriyavit, S. Silk Sericin Semi-interpenetrating Network Hydrogels Based on PEG-Diacrylate for Wound Healing Treatment. Int. J. Polym. Sci. 2019, 2019, 4740765. [Google Scholar] [CrossRef]
- Shoulders, M.D.; Raines, R.T. Collagen structure and stability. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef] [Green Version]
- Bello, A.B.; Kim, D.; Kim, D.; Park, H.; Lee, S.-H. Engineering and Functionalization of Gelatin Biomaterials: From Cell Culture to Medical Applications. Tissue Eng. Part B Rev. 2020, 26, 164–180. [Google Scholar] [CrossRef] [Green Version]
- Bohidar, H.B.; Jena, S.S. Kinetics of sol–gel transition in thermoreversible gelation of gelatin. J. Chem. Phys. 1993, 98, 8970–8977. [Google Scholar] [CrossRef]
- Wang, F.; Yang, C.; Hu, X. Advanced protein composite materials. In Lightweight Materials from Biopolymers and Biofibers; Yang, Y., Xu, H., Yu, X., Eds.; ACS Symposium Series; ACS Publications: Washington, DC, USA, 2014; pp. 177–208. [Google Scholar]
- Zhang, Z.; Ortiz, O.; Goyal, R.; Kohn, J. Biodegradable polymers. In Handbook of Polymer Applications in Medicine and Medical Devices; Elsevier: Amsterdam, The Netherlands, 2014; pp. 303–335. [Google Scholar]
- Balaji, S.; Kumar, R.; Sripriya, R.; Rao, U.; Mandal, A.; Kakkar, P.; Reddy, P.N.; Sehgal, P.K. Characterization of keratin-collagen 3D scaffold for biomedical applications. Polym. Adv. Technol. 2011, 23, 500–507. [Google Scholar] [CrossRef]
- Sando, L.; Kim, M.; Colgrave, M.; Ramshaw, J.A.M.; Werkmeister, J.A.; Elvin, C.M. Photochemical crosslinking of soluble wool keratins produces a mechanically stable biomaterial that supports cell adhesion and proliferation. J. Biomed. Mater. Res. Part A 2010, 95, 901–911. [Google Scholar] [CrossRef]
- Wang, S.; Taraballi, F.; Tan, L.P.; Ng, K.W. Human keratin hydrogels support fibroblast attachment and proliferation in vitro. Cell Tissue Res. 2012, 347, 795–802. [Google Scholar] [CrossRef] [PubMed]
- Sproul, E.; Nandi, S.; Brown, A. Fibrin biomaterials for tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Elsevier: Amsterdam, The Netherlands, 2018; pp. 151–173. [Google Scholar]
- de Melo, B.A.; Jodat, Y.A.; Cruz, E.M.; Benincasa, J.C.; Shin, S.R.; Porcionatto, M.A. Strategies to use fibrinogen as bioink for 3D bioprinting fibrin-based soft and hard tissues. Acta Biomater. 2020, 117, 60–76. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Sani, E.S.; Spencer, A.R.; Guan, Y.; Weiss, A.S.; Annabi, N. Human-recombinant-elastin-based bioinks for 3D bioprinting of vascularized soft tissues. Adv. Mater. 2020, 32, 1–10. [Google Scholar] [CrossRef]
- Wu, Y.; Fortunato, G.M.; Okesola, B.O.; Di Brocchetti, F.L.P.; Suntornnond, R.; Connelly, J.; De Maria, C.; Rodriguez-Cabello, J.C.; Vozzi, G.; Wang, W.; et al. An interfacial self-assembling bioink for the manufacturing of capillary-like structures with tuneable and anisotropic permeability. Biofabrication 2021, 13, 035027. [Google Scholar] [CrossRef]
- Michels, J.; Appel, E.; Gorb, S.N. Resilin—The pliant protein. In Extracellular Composite Matrices in Arthropods; Springer: Cham, Switzerland, 2016; pp. 89–136. [Google Scholar]
- Mu, X.; Sahoo, J.K.; Cebe, P.; Kaplan, D.L. Photo-Crosslinked Silk Fibroin for 3D Printing. Polymers 2020, 12, 2936. [Google Scholar] [CrossRef]
- Veiga, A.; Castro, F.; Rocha, F.; Oliveira, A. Recent Advances in Silk Sericin/Calcium Phosphate Biomaterials. Front. Mater. 2020, 7, 24. [Google Scholar] [CrossRef] [Green Version]
- Mu, X.; Agostinacchio, F.; Xiang, N.; Pei, Y.; Khan, Y.; Guo, C.; Cebe, P.; Motta, A.; Kaplan, D.L. Recent advances in 3D printing with protein-based inks. Prog. Polym. Sci. 2021, 115, 101375. [Google Scholar] [CrossRef] [PubMed]
- Baptista-Silva, S.; Borges, S.; Costa-Pinto, A.R.; Costa, R.; Amorim, M.; Dias, J.R.; Ramos, O.; Alves, P.; Granja, P.L.; Soares, R.; et al. In situ forming silk sericin-based hydrogel: A novel wound healing biomaterial. ACS Biomater. Sci. Eng. 2021, 7, 1573–1586. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, D.; Tun, H.W.; Wang, T.; Naing, M.W. Organ-Derived Decellularized Extracellular Matrix: A Game Changer for Bioink Manufacturing? Trends Biotechnol. 2018, 36, 787–805. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Lee, H.; Luo, L.; Kyriakides, T.R. Extracellular matrix-derived biomaterials in engineering cell function. Biotechnol. Adv. 2019, 42, 107421. [Google Scholar] [CrossRef] [PubMed]
- Duarte, M.M.; Ribeiro, N.; Silva, I.V.; Dias, J.R.; Alves, N.M.; Oliveira, A.L. Fast decellularization process using supercritical carbon dioxide for trabecular bone. J. Supercrit. Fluids 2021, 172, 105194. [Google Scholar] [CrossRef]
- Beachley, V.Z.; Wolf, M.T.; Sadtler, K.; Manda, S.S.; Jacobs, H.; Blatchley, M.R.; Bader, J.S.; Pandey, A.; Pardoll, D.M.; Elisseeff, J.H. Tissue matrix arrays for high-throughput screening and systems analysis of cell function. Nat. Methods 2015, 12, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Han, W.; Singh, N.K.; Kim, J.J.; Kim, H.; Kim, B.S.; Park, J.Y.; Jang, J.; Cho, D.-W. Directed differential behaviors of multipotent adult stem cells from decellularized tissue/organ extracellular matrix bioinks. Biomaterials 2019, 224, 119496. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, Y.; Luo, C.; Zhai, C.; Li, Z.; Zhang, Y.; Yuan, T.; Dong, S.; Zhang, J.; Fan, W. Crosslinker-free silk/decellularized extracellular matrix porous bioink for 3D bioprinting-based cartilage tissue engineering. Mater. Sci. Eng. C 2020, 118, 111388. [Google Scholar] [CrossRef]
- Jorgensen, A.M.; Chou, Z.; Gillispie, G.; Lee, S.J.; Yoo, J.J.; Soker, S.; Atala, A. Decellularized Skin Extracellular Matrix (dsECM) Improves the Physical and Biological Properties of Fibrinogen Hydrogel for Skin Bioprinting Applications. Nanomaterials 2020, 10, 1484. [Google Scholar] [CrossRef]
- Kim, W.; Kim, G.H. An intestinal model with a finger-like villus structure fabricated using a bioprinting process and collagen/SIS-based cell-laden bioink. Theranostics 2020, 10, 2495–2508. [Google Scholar] [CrossRef]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of natural hydrogels for regenerative medicine applications. J. Mater. Sci. Mater. Med. 2019, 30, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costa, F.; Silva, R.; Boccaccini, A.R. Fibrous protein-based biomaterials (silk, keratin, elastin, and resilin proteins) for tissue regeneration and repair. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Barbosa, M., Martins, C., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 175–204. [Google Scholar]
- Da Silva, K.; Kumar, P.; Choonara, Y.E.; Du Toit, L.C.; Pillay, V. Three-dimensional printing of extracellular matrix (ECM)-mimicking scaffolds: A critical review of the current ECM materials. J. Biomed. Mater. Res. Part A 2020, 108, 2324–2350. [Google Scholar] [CrossRef]
- Garreta, E.; Oria, R.; Tarantino, C.; Pla-Roca, M.; Prado, P.; Fernández-Avilés, F.; Campistol, J.M.; Samitier, J.; Montserrat, N. Tissue engineering by decellularization and 3D bioprinting. Mater. Today 2017, 20, 166–178. [Google Scholar] [CrossRef] [Green Version]
- Ashammakhi, N.; Ahadian, S.; Xu, C.; Montazerian, H.; Ko, H.; Nasiri, R.; Barros, N.; Khademhosseini, A. Bioinks and bioprinting technologies to make heterogeneous and biomimetic tissue constructs. Mater. Today Bio 2019, 1, 100008. [Google Scholar] [CrossRef]
- Sadiku-Agboola, O.; Sadiku, E.R.; Adegbola, A.T.; Biotidara, O.F. Rheological Properties of Polymers: Structure and Morphology of Molten Polymer Blends. Mater. Sci. Appl. 2011, 2, 30–41. [Google Scholar] [CrossRef] [Green Version]
- Das, A.; Gilmer, E.L.; Biria, S.; Bortner, M.J. Importance of Polymer Rheology on Material Extrusion Additive Manufacturing: Correlating Process Physics to Print Properties. ACS Appl. Polym. Mater. 2021, 3, 1218–1249. [Google Scholar] [CrossRef]
- Kim, W.; Kim, G. Collagen/bioceramic-based composite bioink to fabricate a porous 3D hASCs-laden structure for bone tissue regeneration. Biofabrication 2019, 12, 015007. [Google Scholar] [CrossRef]
- Yeo, M.G.; Kim, G.H. A cell-printing approach for obtaining hASC-laden scaffolds by using a collagen/polyphenol bioink. Biofabrication 2017, 9, 025004. [Google Scholar] [CrossRef]
- León-López, A.; Morales-Peñaloza, A.; Martínez-Juárez, V.M.; Vargas-Torres, A.; Zeugolis, D.I.; Aguirre-Álvarez, G. Hydrolyzed collagen—Sources and applications. Molecules 2019, 24, 4031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campos, D.F.D.; Lindsay, C.D.; Roth, J.; LeSavage, B.L.; Seymour, A.J.; Krajina, B.A.; Ribeiro, R.; Costa, P.F.; Blaeser, A.; Heilshorn, S.C. Bioprinting Cell- and Spheroid-Laden Protein-Engineered Hydrogels as Tissue-on-Chip Platforms. Front. Bioeng. Biotechnol. 2020, 8, 374. [Google Scholar] [CrossRef]
- Hull, S.M.; Lindsay, C.D.; Brunel, L.G.; Shiwarski, D.J.; Tashman, J.W.; Roth, J.G.; Myung, D.; Feinberg, A.W.; Heilshorn, S.C. 3D Bioprinting using UNIversal Orthogonal Network (UNION) Bioinks. Adv. Funct. Mater. 2020, 31, 2007983. [Google Scholar] [CrossRef]
- Technical Proteins Nanobiotechnology. Product Information—Recombinant Elastin-Like Protein Polymer for Tissue Engineering; No. 14998; Technical Proteins Nanobiotechnology: Valladolid, Spain, 1920; pp. 1919–1920. [Google Scholar]
- Kim, H.; Song, D.; Kim, M.; Ryu, S.; Um, I.; Ki, C.; Park, Y. Effect of silk fibroin molecular weight on physical property of silk hydrogel. Polymer 2016, 90, 26–33. [Google Scholar] [CrossRef]
- Webb, B.; Doyle, B. Parameter optimization for 3D bioprinting of hydrogels. Bioprinting 2017, 8, 8–12. [Google Scholar] [CrossRef]
- GhavamiNejad, A.; Ashammakhi, N.; Wu, X.Y.; Khademhosseini, A. Crosslinking Strategies for 3D Bioprinting of Polymeric Hydrogels. Small 2020, 16, 2002931. [Google Scholar] [CrossRef]
- Piras, C.C.; Smith, D.K. Multicomponent polysaccharide alginate-based bioinks. J. Mater. Chem. B 2020, 8, 8171–8188. [Google Scholar] [CrossRef]
- Gu, Y.; Schwarz, B.; Forget, A.; Barbero, A.; Martin, I.; Shastri, V. Advanced Bioink for 3D Bioprinting of Complex Free-Standing Structures with High Stiffness. Bioengineering 2020, 7, 141. [Google Scholar] [CrossRef]
- Berg, J.; Hiller, T.; Kissner, M.S.; Qazi, T.H.; Duda, G.N.; Hocke, A.C.; Hippenstiel, S.; Elomaa, L.; Weinhart, M.; Fahrenson, C.; et al. Optimization of cell-laden bioinks for 3D bioprinting and efficient infection with influenza A virus. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Kim, E.; Seok, J.M.; Bin Bae, S.; Park, S.; Park, W.H. Silk fibroin enhances cytocompatibilty and dimensional stability of alginate hydrogels for light-based three-dimensional bioprinting. Biomacromolecules 2021, 22, 1921–1931. [Google Scholar] [CrossRef] [PubMed]
- Trucco, D.; Sharma, A.; Manferdini, C.; Gabusi, E.; Petretta, M.; Desando, G.; Ricotti, L.; Chakraborty, J.; Ghosh, S.; Lisignoli, G. Modeling and Fabrication of Silk Fibroin–Gelatin-Based Constructs Using Extrusion-Based Three-Dimensional Bioprinting. ACS Biomater. Sci. Eng. 2021, 7, 3306–3320. [Google Scholar] [CrossRef]
- Yang, X.; Lu, Z.; Wu, H.; Li, W.; Zheng, L.; Zhao, J. Collagen-alginate as bioink for three-dimensional (3D) cell printing based cartilage tissue engineering. Mater. Sci. Eng. C 2018, 83, 195–201. [Google Scholar] [CrossRef]
- Khalil, S.; Sun, W. Bioprinting Endothelial Cells With Alginate for 3D Tissue Constructs. J. Biomech. Eng. 2009, 131, 111002. [Google Scholar] [CrossRef]
- Demirtaş, T.T.; Irmak, G.; Gümüşderelioğlu, M. A bioprintable form of chitosan hydrogel for bone tissue engineering. Biofabrication 2017, 9, 035003. [Google Scholar] [CrossRef]
- Saarai, A.; Kasparkova, V.; Sedlacek, T.; Saha, P. A comparative study of crosslinked sodium alginate/gelatin hydrogels for wound dressing. In Proceedings of the 4th WSEAS International Conference on Engineering Mechanics, Structures, Engineering Geology, Corfu Island, Greece, 14–16 July 2011; pp. 384–389. [Google Scholar]
- Lee, J.; Yeo, M.; Kim, W.; Koo, Y.; Kim, G.H. Development of a tannic acid cross-linking process for obtaining 3D porous cell-laden collagen structure. Int. J. Biol. Macromol. 2018, 110, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Kim, G. Intestinal Villi Model with Blood Capillaries Fabricated Using Collagen-Based Bioink and Dual-Cell-Printing Process. ACS Appl. Mater. Interfaces 2018, 10, 41185–41196. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhu, K.; Yan, S.; Li, J.; Pan, T.; Abudupataer, M.; Alam, F.; Sun, X.; Wang, L.; Wang, C. Using Multilayered Hydrogel Bioink in Three-Dimensional Bioprinting for Homogeneous Cell Distribution. J. Vis. Exp. 2020, e60920. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Häder, D.-P. UV-induced DNA damage and repair: A review. Photochem. Photobiol. Sci. 2002, 1, 225–236. [Google Scholar] [CrossRef]
- Heichel, D.L.; Tumbic, J.A.; Boch, M.E.; Ma, A.W.K.; Burke, K.A. Silk fibroin reactive inks for 3D printing crypt-like structures. Biomed. Mater. 2020, 15, 055037. [Google Scholar] [CrossRef]
- Schwab, A.; Helary, C.; Richards, R.G.; Alini, M.; Eglin, D.; D’Este, M. Tissue mimetic hyaluronan bioink containing collagen fibers with controlled orientation modulating cell migration and alignment. Mater. Today Bio 2020, 7, 100058. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.W.; Choi, J.H.; Lee, Y.; Park, K.D. Horseradish peroxidase-catalysed in situ-forming hydrogels for tissue-engineering applications. J. Tissue Eng. Regen. Med. 2014, 12, 181–204. [Google Scholar]
- Freeman, S.; Ramos, R.; Chando, P.A.; Zhou, L.; Reeser, K.; Jin, S.; Soman, P.; Ye, K. A bioink blend for rotary 3D bioprinting tissue engineered small-diameter vascular constructs. Acta Biomater. 2019, 95, 152–164. [Google Scholar] [CrossRef]
- Jorgensen, A.; Varkey, M.; Gorkun, A.; Clouse, C.; Xu, L.; Chou, Z.; Murphy, S.V.; Molnar, J.; Lee, S.J.; Yoo, J.J.; et al. Bioprinted Skin Recapitulates Normal Collagen Remodeling in Full-Thickness Wounds. Tissue Eng. Part A 2020, 26, 512–526. [Google Scholar] [CrossRef]
- Abelseth, E.; Abelseth, L.; De La Vega, L.; Beyer, S.T.; Wadsworth, S.J.; Willerth, S.M. 3D Printing of Neural Tissues Derived from Human Induced Pluripotent Stem Cells Using a Fibrin-Based Bioink. ACS Biomater. Sci. Eng. 2018, 5, 234–243. [Google Scholar] [CrossRef]
- Forth, J.; Liu, X.; Hasnain, J.; Toor, A.; Miszta, K.; Shi, S.; Geissler, P.L.; Emrick, T.; Helms, B.A.; Russell, T.P. Reconfigurable Printed Liquids. Adv. Mater. 2018, 30, e1707603. [Google Scholar] [CrossRef] [PubMed]
- Alruwaili, M.; Lopez, J.A.; McCarthy, K.; Reynaud, E.G.; Rodriguez, B.J. Liquid-phase 3D bioprinting of gelatin alginate hydrogels: Influence of printing parameters on hydrogel line width and layer height. Bio-Des. Manuf. 2019, 2, 172–180. [Google Scholar] [CrossRef]
- Nishida, H.; Tokiwa, Y. Biomedical applications of biodegradable polymers. J. Environ. Polym. Degrad. 1993, 1, 65–80. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, S.C.N.; Tobias, G.; Roy, A.K.; Vacanti, C.A.; Bonassar, L.J. Tissue Engineering of Autologous Cartilage for Craniofacial Reconstruction by Injection Molding. Plast. Reconstr. Surg. 2003, 112, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Ratanavaraporn, J.; Yodmuang, S. Alginate-silk fibroin Bioink: Printable hydrogel for tissue engineering. In Proceedings of the IEEE 12th Biomedical Engineering International Conference, Ubon Ratchathani, Thailand, 19–22 November 2019. [Google Scholar]
- Singh, Y.P.; Bandyopadhyay, A.; Mandal, B.B. 3D Bioprinting Using Cross-Linker-Free Silk–Gelatin Bioink for Cartilage Tissue Engineering. ACS Appl. Mater. Interfaces 2019, 11, 33684–33696. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Barua, R.; Das, J. Importance of Alginate Bioink for 3D Bioprinting in Tissue Engineering and Regenerative Medicine. In Alginates—Recent Uses of This Natural Polymer; IntechOpen: London, UK, 2020. [Google Scholar]
- Salati, M.A.; Khazai, J.; Tahmuri, A.M.; Samadi, A.; Taghizadeh, A.; Taghizadeh, M.; Zarrintaj, P.; Ramsey, J.D.; Habibzadeh, S.; Seidi, F.; et al. Agarose-Based Biomaterials: Opportunities and Challenges in Cartilage Tissue Engineering. Polymers 2020, 12, 1150. [Google Scholar] [CrossRef]
- Campos, D.F.D.; Marquez, A.B.; O’Seanain, C.; Fischer, H.; Blaeser, A.; Vogt, M.; Corallo, D.; Aveic, S. Exploring Cancer Cell Behavior In Vitro in Three-Dimensional Multicellular Bioprintable Collagen-Based Hydrogels. Cancers 2019, 11, 180. [Google Scholar] [CrossRef] [Green Version]
- Luo, W.; Song, Z.; Wang, Z.; Wang, Z.; Li, Z.; Wang, C.; Liu, H.; Liu, Q.; Wang, J. Printability Optimization of Gelatin-Alginate Bioinks by Cellulose Nanofiber Modification for Potential Meniscus Bioprinting. J. Nanomater. 2020, 2020, 3863428. [Google Scholar] [CrossRef]
- Köpf, M.; Campos, D.F.D.; Blaeser, A.; Sen, K.S.; Fischer, H. A tailored three-dimensionally printable agarose-collagen blend allows encapsulation, spreading, and attachment of human umbilical artery smooth muscle cells. Biofabrication 2016, 8, 1–15. [Google Scholar] [CrossRef]
- Heidenreich, A.C.; Pérez-Recalde, M.; Wusener, A.G.; Hermida, É.B. Collagen and chitosan blends for 3D bioprinting: A rheological and printability approach. Polym. Test. 2020, 82, 106297. [Google Scholar] [CrossRef]
- Snetkov, P.; Zakharova, K.; Morozkina, S.; Olekhnovich, R.; Uspenskaya, M. Hyaluronic acid: The influence of molecular weight on structural, physical, physico-chemical, and degradable properties of biopolymer. Polymers 2020, 12, 1800. [Google Scholar] [CrossRef]
- Pescosolido, L.; Schuurman, W.; Malda, J.; Matricardi, P.; Alhaique, F.; Coviello, T.; Van Weeren, P.R.; Dhert, W.; Hennink, W.E.; Vermonden, T. Hyaluronic Acid and Dextran-Based Semi-IPN Hydrogels as Biomaterials for Bioprinting. Biomacromolecules 2011, 12, 1831–1838. [Google Scholar] [CrossRef] [PubMed]
- Petta, D.; Grijpma, D.W.; Alini, M.; Eglin, D.; D’Este, M. Three-Dimensional Printing of a Tyramine Hyaluronan Derivative with Double Gelation Mechanism for Independent Tuning of Shear Thinning and Postprinting Curing. ACS Biomater. Sci. Eng. 2018, 4, 3088–3098. [Google Scholar] [CrossRef] [PubMed]
- Mazzocchi, A.; Devarasetty, M.; Huntwork, R.; Soker, S.; Skardal, A. Optimization of collagen type I-hyaluronan hybrid bioink for 3D bioprinted liver microenvironments. Biofabrication 2018, 11, 015003. [Google Scholar] [CrossRef]
- Clark, C.C.; Aleman, J.; Mutkus, L.; Skardal, A. A mechanically robust thixotropic collagen and hyaluronic acid bioink supplemented with gelatin nanoparticles. Bioprinting 2019, 16, e00058. [Google Scholar] [CrossRef]
- Shin, J.H.; Kang, H.-W. The Development of Gelatin-Based Bio-Ink for Use in 3D Hybrid Bioprinting. Int. J. Precis. Eng. Manuf. 2018, 19, 767–771. [Google Scholar] [CrossRef]
- Kim, W.; Jang, C.H.; Kim, G.H. A Myoblast-Laden Collagen Bioink with Fully Aligned Au Nanowires for Muscle-Tissue Regeneration. Nano Lett. 2019, 19, 8612–8620. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Yan, M.; Wang, Y.; Fu, J.; Suo, H. 3D Bioprinting of Low-Concentration Cell-Laden Gelatin Methacrylate (GelMA) Bioinks with a Two-Step Cross-linking Strategy. ACS Appl. Mater. Interfaces 2018, 10, 6849–6857. [Google Scholar] [CrossRef]
- Theus, A.S.; Ning, L.; Hwang, B.; Gil, C.; Chen, S.; Wombwell, A.; Mehta, R.; Serpooshan, V. Bioprintability: Physiomechanical and Biological Requirements of Materials for 3D Bioprinting Processes. Polymers 2020, 12, 2262. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.G.; Gopi, S. Induction assisted friction stir welding: A review. Aust. J. Mech. Eng. 2018, 18, 119–123. [Google Scholar] [CrossRef]
- Lepowsky, E.; Muradoglu, M.; Tasoglu, S. Towards preserving post-printing cell viability and improving the resolution: Past, present, and future of 3D bioprinting theory. Bioprinting 2018, 11, 1–17. [Google Scholar] [CrossRef]
- Osidak, E.O.; Karalkin, P.A.; Osidak, M.S.; Parfenov, V.A.; Sivogrivov, D.E.; Pereira, F.D.A.S.; Gryadunova, A.A.; Koudan, E.V.; Khesuani, Y.D.; Kasyanov, V.A.; et al. Viscoll collagen solution as a novel bioink for direct 3D bioprinting. J. Mater. Sci. Mater. Med. 2019, 30, 1–12. [Google Scholar] [CrossRef]
- Genova, T.; Roato, I.; Carossa, M.; Motta, C.; Cavagnetto, D.; Mussano, F. Advances on Bone Substitutes through 3D Bioprinting. Int. J. Mol. Sci. 2020, 21, 7012. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, S.; Zhang, Y.; Cormier, D.R.; Rivero, I.V.; Harrysson, O.L.; Rao, P.K.; Tamayol, A. Extrusion bioprinting: Recent progress, challenges, and future opportunities. Bioprinting 2021, 21, e00116. [Google Scholar] [CrossRef]
- Gillispie, G.; Han, A.; Uzun-Per, M.; Fisher, J.; Mikos, A.G.; Niazi, M.K.K.; Yoo, J.J.; Lee, S.J.; Atala, A. The Influence of Printing Parameters and Cell Density on Bioink Printing Outcomes. Tissue Eng. Part A 2020, 26, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Öztürk, E.; Arlov, Ø.; Gatenholm, P.; Zenobi-Wong, M. Alginate sulfate-nanocellulose bioinks for cartilage bioprinting applications. Ann. Biomed. Eng. 2017, 45, 210–223. [Google Scholar] [CrossRef]
- Piard, C.; Baker, H.; Kamalitdinov, T.; Fisher, J. Bioprinted osteon-like scaffolds enhance in vivo neovascularization. Biofabrication 2019, 11, 025013. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Nam, J.; Sun, W. Effects of dispensing pressure and nozzle diameter on cell survival from solid freeform fabrication-based direct cell writing. Tissue Eng. Part A 2008, 14, 41–48. [Google Scholar] [CrossRef]
- O’Connell, C.; Ren, J.; Pope, L.; Li, Y.; Mohandas, A.; Blanchard, R.; Duchi, S.; Onofrillo, C. Characterizing Bioinks for Extrusion Bioprinting: Printability and Rheology. Methods Mol. Biol. 2020, 2140, 111–133. [Google Scholar] [CrossRef] [PubMed]
- Nerger, B.A.; Brun, P.-T.; Nelson, C.M. Microextrusion printing cell-laden networks of type I collagen with patterned fiber alignment and geometry. Soft Matter 2019, 15, 5728–5738. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, J.; Ghosh, S. Cellular Proliferation, Self-Assembly, and Modulation of Signaling Pathways in Silk Fibroin Gelatin-Based 3D Bioprinted Constructs. ACS Appl. Bio Mater. 2020, 3, 8309–8320. [Google Scholar] [CrossRef]
- Moncal, K.K.; Ozbolat, V.; Datta, P.; Heo, D.N.; Ozbolat, I.T. Thermally-controlled extrusion-based bioprinting of collagen. J. Mater. Sci. Mater. Med. 2019, 30, 1–14. [Google Scholar] [CrossRef]
- Schwab, A.; Levato, R.; D’Este, M.; Piluso, S.; Eglin, D.; Malda, J. Printability and Shape Fidelity of Bioinks in 3D Bioprinting. Chem. Rev. 2020, 120, 11028–11055. [Google Scholar] [CrossRef]
- Dias, J.R.; Ribeiro, N.; da Silva, S.B.; Costa-Pinto, A.R.; Alves, N.; Oliveira, A. In situ Enabling Approaches for Tissue Regeneration: Current Challenges and New Developments. Front. Bioeng. Biotechnol. 2020, 8, 85. [Google Scholar] [CrossRef] [Green Version]
- Hakimi, N.; Cheng, R.; Leng, L.; Sotoudehfar, M.; Ba, P.Q.; Bakhtyar, N.; Amini-Nik, S.; Jeschke, M.G.; Guenther, A. Handheld skin printer: In situ formation of planar biomaterials and tissues. Lab. Chip 2018, 18, 1440–1451. [Google Scholar] [CrossRef] [PubMed]
- Ozbolat, I.T. The Bioink ∗ ∗ with contributions by Monika Hospodiuk and Madhuri Dey, The Pennsylvania State University. In 3D Bioprinting; Elsevier: Amsterdam, The Netherlands, 2017; pp. 41–92. [Google Scholar]
- Galante, R.; Pinto, T.J.A.; Colaço, R.; Serro, A.P. Sterilization of hydrogels for biomedical applications: A review. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 2472–2492. [Google Scholar] [CrossRef]
- Rafael, D.; Andrade, F.; Martinez-Trucharte, F.; Basas, J.; Seras-Franzoso, J.; Palau, M.; Gomis, X.; Pérez-Burgos, M.; Blanco, A.; López-Fernández, A.; et al. Sterilization Procedure for Temperature-Sensitive Hydrogels Loaded with Silver Nanoparticles for Clinical Applications. Nanomaterials 2019, 9, 380. [Google Scholar] [CrossRef] [Green Version]
- Das, S.; Pati, F.; Choi, Y.-J.; Rijal, G.; Shim, J.-H.; Kim, S.W.; Ray, A.R.; Cho, D.-W.; Ghosh, S. Bioprintable, cell-laden silk fibroin–gelatin hydrogel supporting multilineage differentiation of stem cells for fabrication of three-dimensional tissue constructs. Acta Biomater. 2014, 11, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Soares, G.; Learmonth, D.A.; Vallejo, M.; Davila, S.P.; González, P.; Sousa, R.A.; Oliveira, A. Supercritical CO2 technology: The next standard sterilization technique? Mater. Sci. Eng. C 2019, 99, 520–540. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, N.; Soares, G.C.; Santos-Rosales, V.; Concheiro, A.; Alvarez-Lorenzo, C.; García-González, C.A.; Oliveira, A. A new era for sterilization based on supercritical CO 2 technology. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 108, 399–428. [Google Scholar] [CrossRef] [PubMed]
- USA Food and Drug. FDA Innovation Challenge 1: Identify New Sterilization Methods and Technologies. 2019. Available online: https://www.fda.gov/medical-devices/general-hospital-devices-and-supplies/fda-innovation-challenge-1-identify-new-sterilization-methods-and-technologies (accessed on 19 July 2021).
- Suntornnond, R.; Tan, E.Y.S.; Zander, N.; Chua, C.K. A Mathematical Model on the Resolution of Extrusion Bioprinting for the Development of New Bioinks. Materials 2016, 9, 756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, S.; Zhao, H.; Lewinski, N. Key parameters and applications of extrusion-based bioprinting. Bioprinting 2021, 23, e00156. [Google Scholar] [CrossRef]
- Momeni, F.; Hassani, N.; Liu, X.; Hasani, S. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- Gao, B.; Yang, Q.; Zhao, X.; Jin, G.; Ma, Y.; Xu, F. 4D Bioprinting for Biomedical Applications. Trends Biotechnol. 2016, 34, 746–756. [Google Scholar] [CrossRef]
- An, J.; Chua, C.K.; Mironov, V. A Perspective on 4D Bioprinting. Int. J. Bioprinting 2016, 2. [Google Scholar] [CrossRef]

| Protein characteristics |
MW determination Protein sequencing Standardization of extraction protocols |
Chromatography Mass spectrometry |
| Preliminary in vitro assays | Viability tests | Calcein AM Green Live Dead assay Propidum Iodide Alamar blue Hoechst 33342 |
| Metabolic tests | Cell Counting Kit-8 assay (CCK8) 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide assay (MTT) Positron emisson tomography activity (PET) | |
| Mechanical | Compressive tests Young or elastic modulus Compression modulus Yield stress Ultimate tensile strength (UTS) | |
| Quantitative measures (Extrudability) | Viscosity Rheology Extrusion uniformity Structural integrity. | |
| Extrusion Bioprinter | Parameters | Units | Most Used |
|---|---|---|---|
 | (1) Cartridge/Printhead temperature | °C | 37 °C Room temperature |
| (2) Bed/working plane temperature. | 20 °C–40 °C | ||
| (3) Extrusion pressure (pneumatic, piston, screw) | Pa (bar, psi, N/mm2) | 4–150 KPa | |
| (4) Nozzle | µm | 0.26 mm–250 mm | |
| Printing speed (Feed rate) | mm/s | 0.2–150 mm/s (most common 1–30 mm/s) | |
| Flow rate | mm3/s | Not described | |
| (5) Distance between the nozzle and substrate | cm | ½ nozzle diameter | |
| (6) 3D structure | - Conventional shapes: squares, rectangles and spheres - Fiber resolution in the microscale (µm) |
| Formulation | Cell Type | Cell Density (Cells/mL) | Cell Viability & Functionality | Ref. |
|---|---|---|---|---|
| Gelatin (5% w/v) and silk fibroin solution (5% w/v) | hMSCs | 1 × 107 | After 28 days: 95% cell viability and increase of SOX-9, the specific chondrogenic transcriptional factor, as well as of collagen type 2. | [97] |
| MeTro (5 % w/v), GelMA (7.5% w/v) and gelatin (20% w/v) | CMs, CFs, HUVECs | 2 × 107 | After 7 days of culture: 85% cell viability. Endothelium barrier function and spontaneous beating of cardiac muscle cells. | [62] |
| Elastin-like protein (3, 4, or 5 % w/v) | NPCs | 1 × 107 | After 7 days of culture: 88.3% cell viability. - after 5 days of presence of hiPSC-NPCs (Sox-2-positive, a marker of neural progenitor cell pluripotency) | [87] |
| Elastin recombinamer (2 % w/v) + graphene oxide (GO) (0.1 % w/v) | GFP- hUVECs | 5 × 103, 1 × 104, 5 × 104, and 1 × 105 | After 48 h of culture, membranes exhibited different cell viabilities ∼30%, 50%, 80%, and >95% (according to the different cell densities used) | [63] |
| Gelatin (7.5 % w/v) and 10 mg/mL fibrinogen | HDF-n | 3 × 106 | 80% cell viability after printing | [109] |
| Fibrinogen (20 mg/mL), gelatin (30 mg/mL), aprotinin (20 μg/mL), glycerol (10% 1/v), and hyaluronic acid (3 mg/mL) | Primary cardiomyocyte | 10 × 106 | Progressive cardiac tissue development was confirmed by immunostaining for α-actinin and connexin 43 | [32] |
| Fibrinogen (20 mg/mL), 0.5 (% w/v) of alginate and genipin (0.3 mg/mL) | NPCs | 1 × 106 | After 7 days of printing: 95%. - The bioprinted tissues expressed the early neuronal marker, TUJ1 and the early midbrain marker, Forkhead Box A2 (FOXA2) after 15 days of culture. Other glial markers such as glial fibrillary acidic protein and oligodendrocyte progenitor marker were present after 30 days. | [30] |
| Fibrin (20 mg/ mL), alginate (5 mg/mL), genipin (0.3 mg/mL). | hiPSCs | 1 × 104 | After 10 days: 94.72% cell viability. After 15 days: 64.12%. - hiPSCs in the presence of puro and RA differentiated into neurons as indicated by early neuronal expression marker TUJ1 and the long neurite extensions into the scaffold. | [111] |
| Fibrinogen (10% w/v) and (5% w/v) | hMSCs and HUVECs | 2 × 106 | After 7 days of culture: 94.8% cell viability. - Increase in gene expression of BMP2, ALP, VEGFA and PECAM1. | [141] |
| Fibrinogen (5–20 mg/mL), gelatin (37.5 mg/mL), hyaluronic acid (3 mg/mL), and glycerol (4% v/v) | hDPSC | - | After 7 days of culture: >90% cell viability | [31] |
| Collagen (5 w/v%) and mixed Au nanowires (GNWs) | myoblasts (C2C12 cell line) | 1 × 107 | After 1 days of culture: >90%. | [131] |
| Collagen (15 mg/mL), agarose (1:4) mixed with sodium alginate (0.1 g/mL) | chondrocytes | 1 × 107 | - High cell viability after culture for 14 days (>80%). - Expression of cartilage specific genes such as Acan, Col2al and Sox9. | [98] |
| Collagen branded Viscoll (2, 3 and 4 w/v) | NIH 3T3 fibroblasts | 0.5 × 106 | After 7 days of culture: 97.2%, 95.2% and 87.2% for bioinks at 2, 3 and 4% collagen concentrations | [136] |
| Collagen (5 % w/v) mixed with various weight fractions of β-TCP (0, 10, 20, and 45% w/v) | MC3T3-E1 | 1 × 107 | After 7 days of culture: 92% cell viability. For greater than 20 wt% of β-TCP, the cell-viability was significantly lowered. | [84] |
| Collagen (4 % w/v) | hASCs | 2 × 106 | After 5 days of culture: 93% cell viability. | [85] |
| Collagen (5 mg/mL) and tyramine derivative of hyaluronan (25 mg/mL) | hMSC | 3 × 106 | - The cells were viable after printing and remained viable over culture time. After 6 days, the number of dead cells were less than that observed on day 1. - hMSCs embedded in the isotropic bioink displayed chondrogenic differentiation comparable | [107] |
| Collagen (3 mg/mL) | rBMSCs | 10 × 106 | Cell viability recorded were >88% after printing. | [146] |
| Fibrinogen (30 mg/mL), gelatin (35 mg/mL), glycerol (100 μL/mL), and hyaluronic acid (3 mg/mL) | Human keratinocytes, melanocytes, fibroblasts, dermal microvascular endothelial cells, follicle dermal papilla cells, and adipocytes | 20 × 106 | All bioprinted skin treated wounds closed by day 21, compared with open control wounds. Wound | [110] |
| Collagen solution (4, 8, or 12 mg/mL) and riboflavin | chondrocytes | 1 × 106 | After 7 days of culture: >95% cell viability at all collagen concentrations tested. However, the addition of blue light activated riboflavin crosslinking decreased viability to 76–77%. | [39] |
| Collagen-based (4 % w/v) and tannic acid (0, 1, and 2 % w/v) | Caco-2 | 5 × 106 | After 7 days of culture: 93% cell viability. | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Veiga, A.; Silva, I.V.; Duarte, M.M.; Oliveira, A.L. Current Trends on Protein Driven Bioinks for 3D Printing. Pharmaceutics 2021, 13, 1444. https://doi.org/10.3390/pharmaceutics13091444
Veiga A, Silva IV, Duarte MM, Oliveira AL. Current Trends on Protein Driven Bioinks for 3D Printing. Pharmaceutics. 2021; 13(9):1444. https://doi.org/10.3390/pharmaceutics13091444
Chicago/Turabian StyleVeiga, Anabela, Inês V. Silva, Marta M. Duarte, and Ana L. Oliveira. 2021. "Current Trends on Protein Driven Bioinks for 3D Printing" Pharmaceutics 13, no. 9: 1444. https://doi.org/10.3390/pharmaceutics13091444
APA StyleVeiga, A., Silva, I. V., Duarte, M. M., & Oliveira, A. L. (2021). Current Trends on Protein Driven Bioinks for 3D Printing. Pharmaceutics, 13(9), 1444. https://doi.org/10.3390/pharmaceutics13091444







