Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience
Abstract
1. Introduction
2. Blood Brain Barrier Physiology and Pathophysiology in Glioma
3. Convection Enhanced Delivery—Rationale and Mechanisms
3.1. Biophysical Properties
3.2. CED Logistics
3.3. Pitfalls of CED
4. Convection Enhanced Delivery—The Columbia University Medical Center Experience
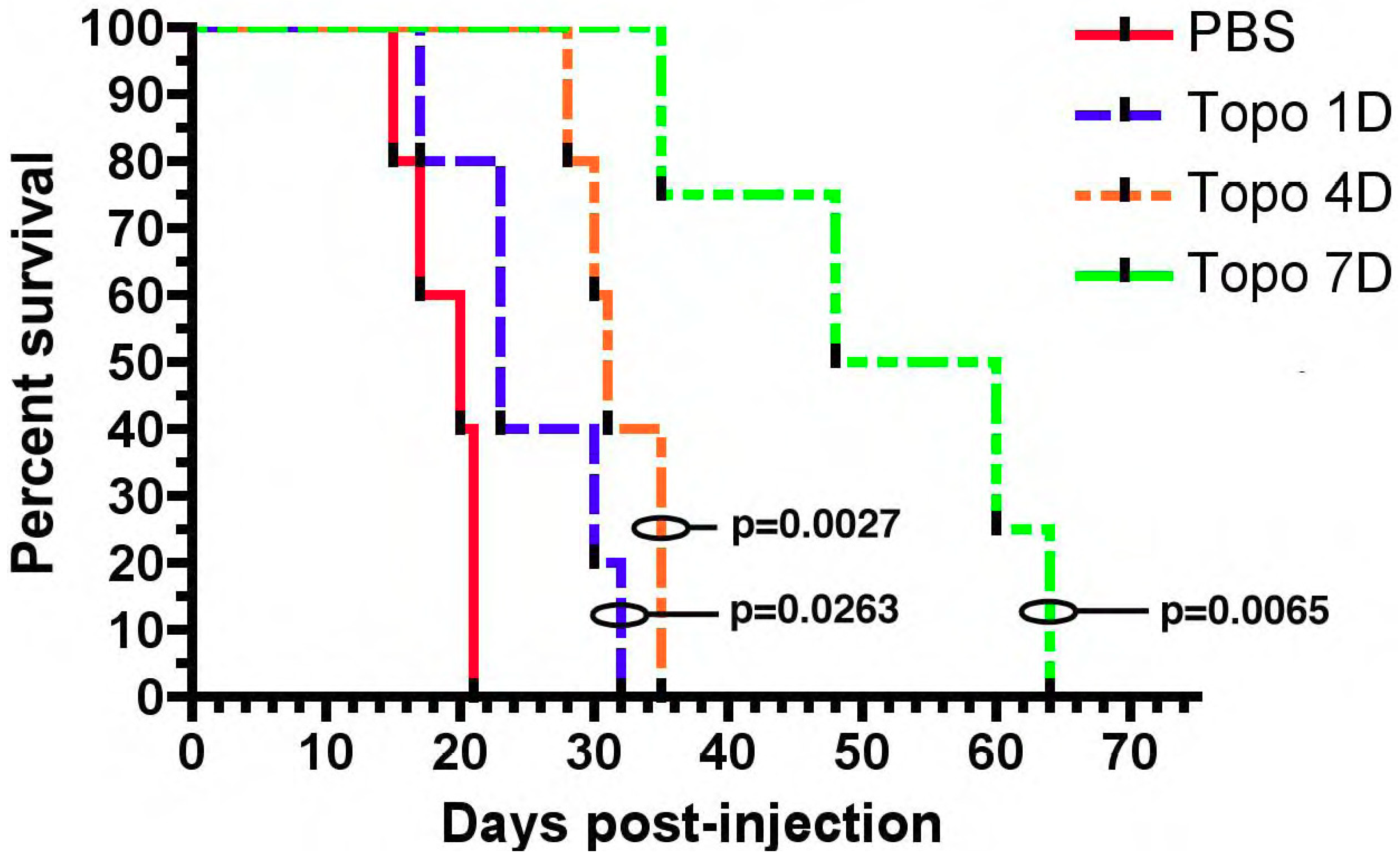
4.1. Preclinical Data—Small Animal Models
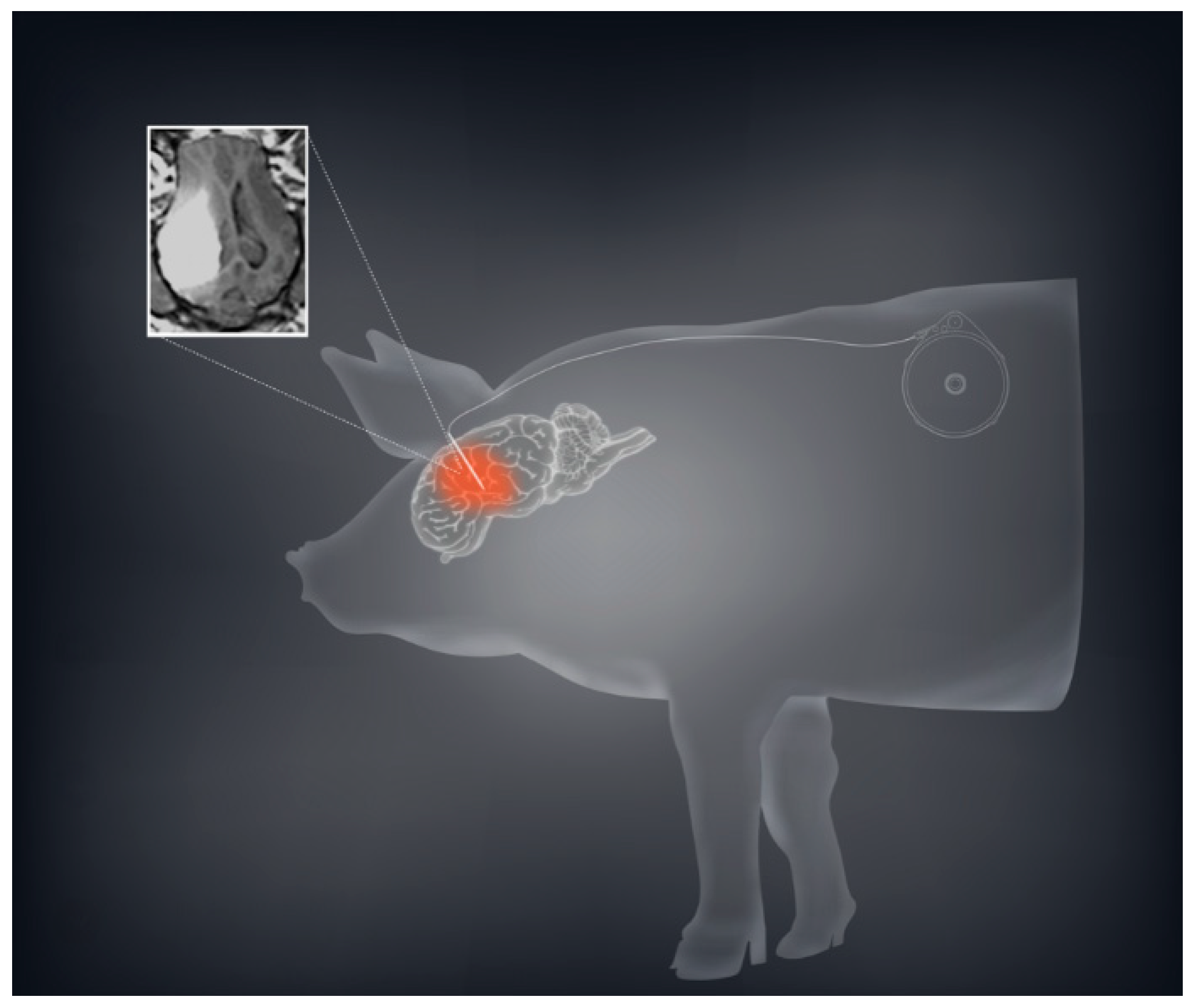
4.2. Pre-Clinical Data—Large Animal Models
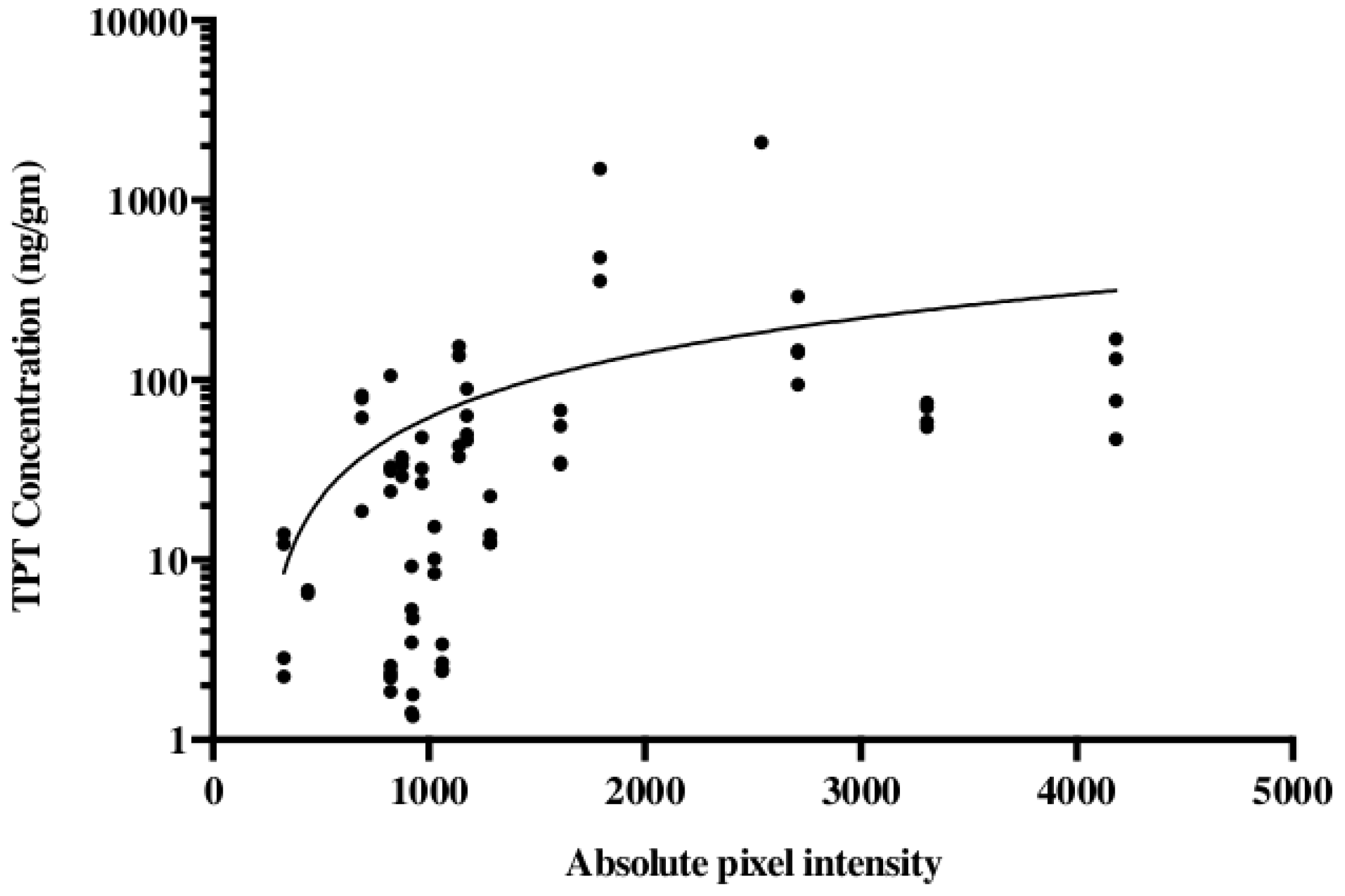
4.3. Gadolinium as a Surrogate for Convection Volumes
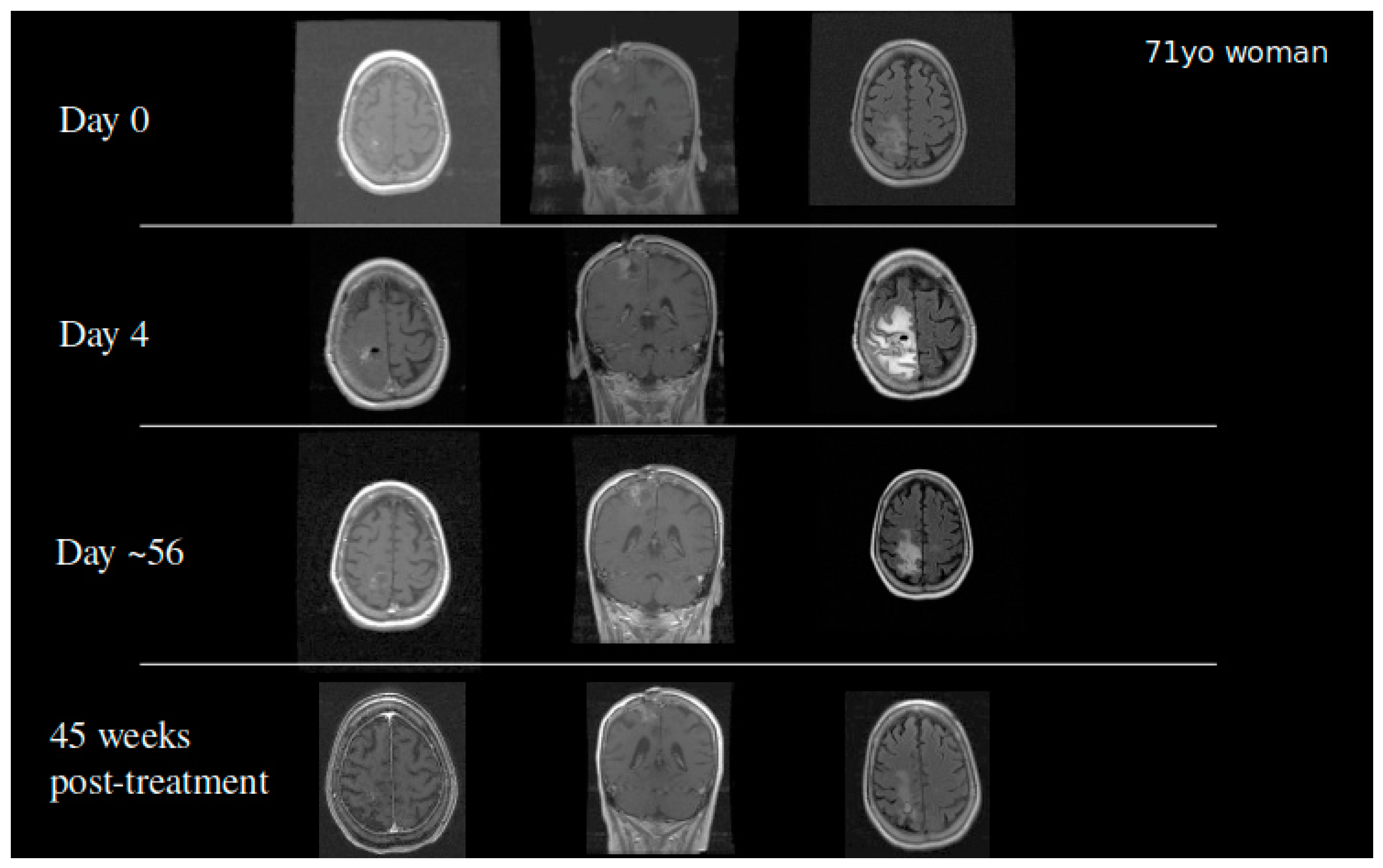
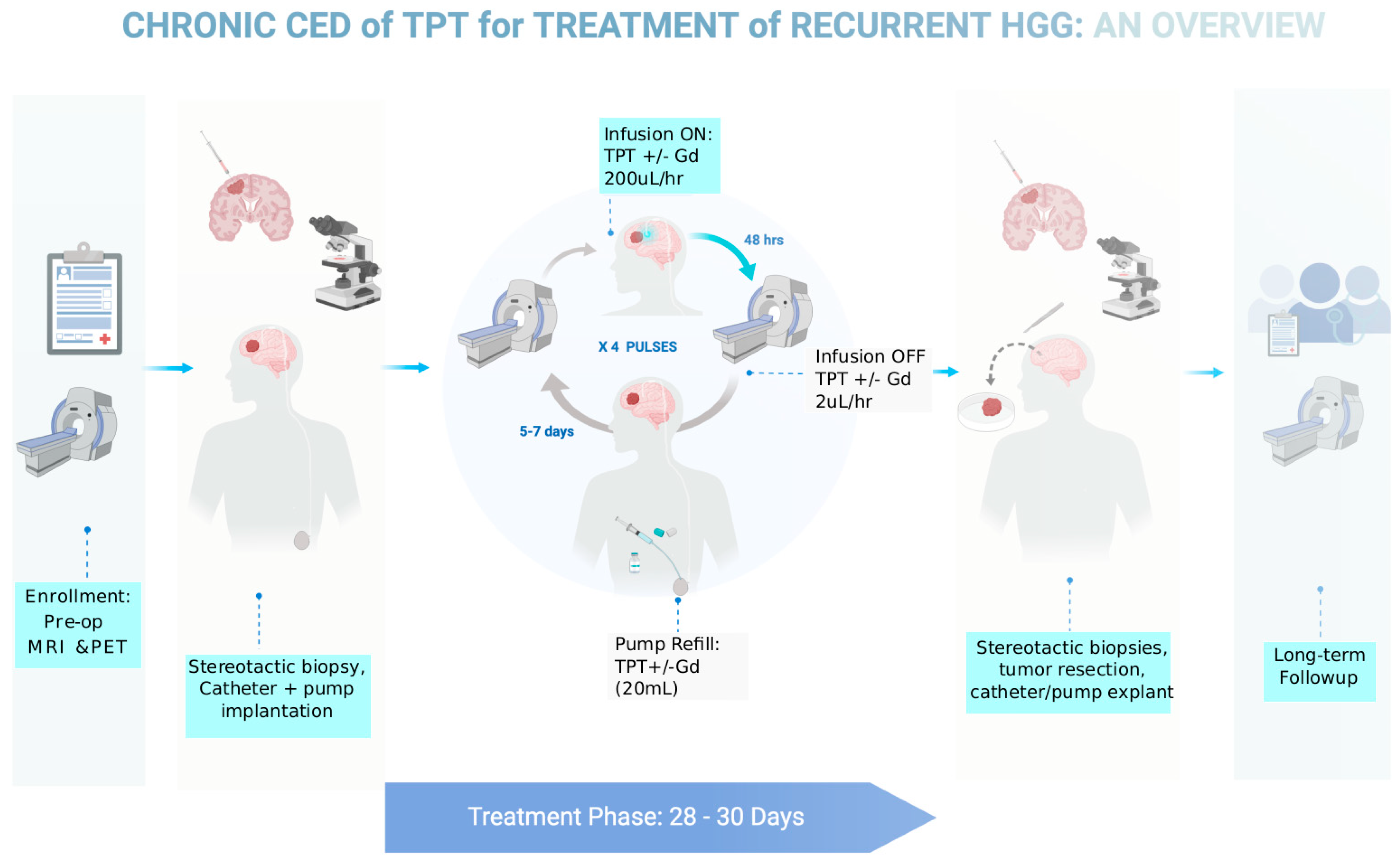
4.4. Clinical Experience with Topotecan
4.5. Current Chronic CED Clinical Studies
4.6. Best Practices
5. Current Landscape of CED Clinical Treatments
6. Investigational Drug Formulation
7. Potential Applications of CED beyond Glioma Therapy
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gill, B.J.; Pisapia, D.J.; Malone, H.R.; Goldstein, H.; Lei, L.; Sonabend, A.; Yun, J.; Samanamud, J.; Sims, J.S.; Banu, M.; et al. MRI-localized biopsies reveal subtype-specific differences in molecular and cellular composition at the margins of glioblastoma. Proc. Natl. Acad. Sci. USA 2014, 111, 12550–12555. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Levitin, H.M.; Frattini, V.; Bush, E.C.; Boyett, D.M.; Samanamud, J.; Ceccarelli, M.; Dovas, A.; Zanazzi, G.; Canoll, P.; et al. Single-cell transcriptome analysis of lineage diversity in high-grade glioma. Genome Med. 2018, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus Concomitant and Adjuvant Temozolomide for Glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Walker, M.D.; Strike, T.A.; Sheline, G.E. An analysis of dose-effect relationship in the radiotherapy of malignant gliomas. Int. J. Radiat. Oncol. 1979, 5, 1725–1731. [Google Scholar] [CrossRef]
- Lesimple, T.; Riffaud, L.; Frappaz, D.; Ben Hassel, M.; Gedouin, D.; Bay, J.-O.; Linassier, C.; Hamlat, A.; Piot, G.; Fabbro, M.; et al. Topotecan in combination with radiotherapy in unresectable glioblastoma: A phase 2 study. J. Neuro-Oncol. 2009, 93, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Falzone, L.; Salomone, S.; Libra, M. Evolution of Cancer Pharmacological Treatments at the Turn of the Third Millennium. Front. Pharmacol. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.V.; Khatri, D.; D’Amico, R.; Abrams, M.; Reichman, N.; Filippi, C.G.; Anderson, T.; Ratzon, F.; Wong, T.; Fralin, S.; et al. Vascularized Temporoparietal Fascial Flap: A Novel Surgical Technique to Bypass the Blood-Brain Barrier in Glioblastoma. World Neurosurg. 2020, 143, 38–45. [Google Scholar] [CrossRef]
- Guthkelch, A.N.; Carter, L.P.; Cassady, J.R.; Hynynen, K.H.; Iacono, R.P.; Johnson, P.C.; Obbens, E.A.M.T.; Roemer, R.B.; Seeger, J.F.; Shimm, D.S.; et al. Treatment of malignant brain tumors with focused ultrasound hyperthermia and radiation: Results of a phase I trial. J. Neuro-Oncol. 1991, 10, 271–284. [Google Scholar] [CrossRef]
- Ananda, S.; Nowak, A.K.; Cher, L.; Dowling, A.; Brown, C.; Simes, J.; Rosenthal, M.A. Phase 2 trial of temozolomide and pegylated liposomal doxorubicin in the treatment of patients with glioblastoma multiforme following concurrent radiotherapy and chemotherapy. J. Clin. Neurosci. 2011, 18, 1444–1448. [Google Scholar] [CrossRef]
- Bobo, R.H.; Laske, D.W.; Akbasak, A.; Morrison, P.F.; Dedrick, R.L.; Oldfield, E.H. Convection-enhanced delivery of macromolecules in the brain. Proc. Natl. Acad. Sci. USA 1994, 91, 2076–2080. [Google Scholar] [CrossRef]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef]
- Wolburg, H.; Noell, S.; Mack, A.; Wolburg-Buchholz, K.; Fallier-Becker, P. Brain endothelial cells and the glio-vascular complex. Cell Tissue Res. 2008, 335, 75–96. [Google Scholar] [CrossRef] [PubMed]
- Arismendi-Morillo, G.; Castellano, A. Tumoral micro-blood vessels and vascular microenvironment in human astrocytic tumors. A transmission electron microscopy study. J. Neuro-Oncol. 2005, 73, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Bruce, J.N.; Criscuolo, G.R.; Merrill, M.J.; Moquin, R.R.; Blacklock, J.B.; Oldfield, E.H. Vascular permeability induced by protein product of malignant brain tumors: Inhibition by dexamethasone. J. Neurosurg. 1987, 67, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Mendes, N.F.; Pansani, A.P.; Carmanhães, E.R.; Tange, P.; Meireles, J.V.; Ochikubo, M.; Chagas, J.R.; da Silva, A.V.; Monteiro de Castro, G.; Sueur-Maluf, L. The Blood-Brain Barrier Breakdown during Acute Phase of the Pilocarpine Model of Epilepsy Is Dynamic and Time-Dependent. Front. Neurol. 2019, 10, 382. [Google Scholar] [CrossRef]
- Mehta, A.M.; Sonabend, A.M.; Bruce, J.N. Convection-Enhanced Delivery. Neurotherapeutics 2017, 14, 358–371. [Google Scholar] [CrossRef]
- Souweidane, M.M. Editorial: Convection-enhanced delivery for diffuse intrinsic pontine glioma. J. Neurosurg. Pediatr. 2014, 13, 273–275. [Google Scholar] [CrossRef]
- Ung, T.H.; Malone, H.; Canoll, P.; Bruce, J.N. Convection-enhanced delivery for glioblastoma: Targeted delivery of antitumor therapeutics. CNS Oncol. 2015, 4, 225–234. [Google Scholar] [CrossRef]
- Mohammed, W.; Hong, X.N.; Haibin, S.; Jingzhi, M. Clinical applications of susceptibility-weighted imaging in detecting and grading intracranial gliomas: A review. Cancer Imaging 2013, 13, 186–195. [Google Scholar] [CrossRef]
- Barua, N.U.; Hopkins, K.; Woolley, M.; O’Sullivan, S.; Harrison, R.; Edwards, R.J.; Bienemann, A.S.; Wyatt, M.J.; Arshad, A.; Gill, S.S. A novel implantable catheter system with transcutaneous port for intermittent convection-enhanced delivery of carboplatin for recurrent glioblastoma. Drug Deliv. 2014, 23, 167–173. [Google Scholar] [CrossRef]
- Sonabend, A.M.; Stuart, R.M.; Yun, J.; Yanagihara, T.; Mohajed, H.; Dashnaw, S.; Bruce, S.S.; Brown, T.; Romanov, A.; Sebastian, M.; et al. Prolonged intracerebral convection-enhanced delivery of topotecan with a subcutaneously implantable infusion pump. Neuro-Oncology 2011, 13, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Singh, R.; Souweidane, M.M. Convection-Enhanced Delivery for Diffuse Intrinsic Pontine Glioma Treatment. Curr. Neuropharmacol. 2016, 15, 116–128. [Google Scholar] [CrossRef]
- Sillay, K.A.; McClatchy, S.G.; Shepherd, B.A.; Venable, G.T.; Fuehrer, T.S. Image-guided Convection-enhanced Delivery into Agarose Gel Models of the Brain. J. Vis. Exp. 2014, e51466. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.; Forsayeth, J.; Bankiewicz, K.S. Optimized cannula design and placement for convection-enhanced delivery in rat striatum. J. Neurosci. Methods 2010, 187, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Lewis, O.; Woolley, M.; Johnson, D.; Rosser, A.E.; Barua, N.U.; Bienemann, A.S.; Gill, S.S.; Evans, S.L. Chronic, intermittent convection-enhanced delivery devices. J. Neurosci. Methods 2016, 259, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Lidar, Z.; Mardor, Y.; Jonas, T.; Pfeffer, R.; Faibel, M.; Nass, D.; Hadani, M.; Ram, Z. Convection-enhanced delivery of paclitaxel for the treatment of recurrent malignant glioma: A Phase I/II clinical study. J. Neurosurg. 2004, 100, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Sanche, L. Convection-Enhanced Delivery in Malignant Gliomas: A Review of Toxicity and Efficacy. J. Oncol. 2019, 2019, 1–13. [Google Scholar] [CrossRef]
- Varenika, V.; Dickinson, P.; Bringas, J.; Lecouteur, R.; Higgins, R.; Park, J.; Fiandaca, M.; Berger, M.; Sampson, J.; Bankiewicz, K.S. Detection of infusate leakage in the brain using real-time imaging of convection-enhanced delivery. J. Neurosurg. 2008, 109, 874–880. [Google Scholar] [CrossRef]
- Van Putten, E.H.; Wembacher-Schröder, E.; Smits, M.; Dirven, C.M. Magnetic Resonance Imaging–Based Assessment of Gadolinium-Conjugated Diethylenetriamine Penta-Acetic Acid Test-Infusion in Detecting Dysfunction of Convection-Enhanced Delivery Catheters. World Neurosurg. 2016, 89, 272–279. [Google Scholar] [CrossRef]
- Bruce, J.N.; Falavigna, A.; Johnson, J.P.; Hall, J.S.; Birch, B.D.; Yoon, J.T.; Wu, E.X.; Fine, R.L.; Parsa, A.T. Intracerebral clysis in a rat glioma model. Neurosurgery 2000, 46, 683–691. [Google Scholar] [CrossRef]
- Yamashita, Y.; Krauze, M.T.; Kawaguchi, T.; Noble, C.O.; Drummond, D.C.; Park, J.W.; Bankiewicz, K.S. Convection-enhanced delivery of a topoisomerase I inhibitor (nanoliposomal topotecan) and a topoisomerase II inhibitor (pegylated liposomal doxorubicin) in intracranial brain tumor xenografts1. Neuro-Oncology 2007, 9, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.B.; Vasilyeva, A.; Gajjar, A.; Santana, V.M.; Stewart, C.F. Determining success rates of the current pharmacokinetically guided dosing approach of topotecan in pediatric oncology patients. Pediatr. Blood Cancer 2018, 66, e27578. [Google Scholar] [CrossRef] [PubMed]
- Perkins, J.B.; Goldstein, S.C.; Dawson, J.L.; Kim, J.; Field, T.L.; Partyka, J.S.; Fields, K.K.; Maddox, B.L.; Simonelli, C.E.; Neuger, A.M.; et al. Phase I Study of Topotecan, Ifosfamide, and Etoposide (TIME) with Autologous Stem Cell Transplant in Refractory Cancer: Pharmacokinetic and Pharmacodynamic Correlates. Clin. Cancer Res. 2011, 17, 7743–7753. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.G.; Parsa, A.T.; Fine, R.L.; Hall, J.S.; Chakrabarti, I.; Bruce, J.N. Tissue distribution and antitumor activity of topotecan delivered by intracerebral clysis in a rat glioma model. Neurosurgery 2000, 47, 1391–1398; discussion 1398–1399. [Google Scholar] [CrossRef]
- Lopez, K.A.; Tannenbaum, A.M.; Assanah, M.C.; Linskey, K.; Yun, J.; Kangarlu, A.; Gil, O.D.; Canoll, P.; Bruce, J.N. Convection-Enhanced Delivery of Topotecan into a PDGF-Driven Model of Glioblastoma Prolongs Survival and Ablates Both Tumor-Initiating Cells and Recruited Glial Progenitors. Cancer Res. 2011, 71, 3963–3971. [Google Scholar] [CrossRef]
- Mei, C.; Lei, L.; Tan, L.-M.; Xu, X.-J.; He, B.-M.; Luo, C.; Yin, J.-Y.; Li, X.; Zhang, W.; Zhou, H.-H.; et al. The role of single strand break repair pathways in cellular responses to camptothecin induced DNA damage. Biomed. Pharmacother. 2020, 125, 109875. [Google Scholar] [CrossRef]
- Sonabend, A.M.; Carminucci, A.S.; Amendolara, B.; Bansal, M.; Leung, R.; Lei, L.; Realubit, R.; Li, H.; Karan, C.; Yun, J.; et al. Convection-enhanced delivery of etoposide is effective against murine proneural glioblastoma. Neuro-Oncology 2014, 16, 1210–1219. [Google Scholar] [CrossRef]
- Sonabend, A.M.; Bansal, M.; Guarnieri, P.; Lei, L.; Amendolara, B.; Soderquist, C.; Leung, R.; Yun, J.; Kennedy, B.; Sisti, J.; et al. The transcriptional regulatory network of proneural glioma determines the genetic alterations selected during tumor progression. Cancer Res. 2014, 74, 1440–1451. [Google Scholar] [CrossRef]
- PubChem. Gadodiamide. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Gadodiamide (accessed on 18 December 2020).
- PubChem. Topotecan. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/topotecan (accessed on 8 December 2020).
- D’Amico, R.S.; Neira, J.A.; Yun, J.; Alexiades, N.G.; Banu, M.; Englander, Z.K.; Kennedy, B.C.; Ung, T.H.; Rothrock, R.J.; Romanov, A.; et al. Validation of an effective implantable pump-infusion system for chronic convection-enhanced delivery of intracerebral topotecan in a large animal model. J. Neurosurg. 2020, 133, 614–623. [Google Scholar] [CrossRef]
- Bruce, J.N.; Fine, R.L.; Canoll, P.; Yun, J.; Kennedy, B.C.; Rosenfeld, S.S.; Sands, S.A.; Surapaneni, K.; Lai, R.; Yanes, C.L.; et al. Regression of Recurrent Malignant Gliomas with Convection-Enhanced Delivery of Topotecan. Neurosurgery 2011, 69, 1272–1279; discussion 1279–1280. [Google Scholar] [CrossRef]
- Oberg, J.A.; Dave, A.N.; Bruce, J.N.; Sands, S.A. Neurocognitive functioning and quality of life in patients with recurrent malignant gliomas treated on a phase Ib trial evaluating topotecan by convection-enhanced delivery. Neuro-Oncol. Pract. 2014, 1, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.E.; Kennedy, B.; Yanes, C.L.; Garvin, J.; Needle, M.; Canoll, P.; Feldstein, N.A.; Bruce, J.N. Convection-enhanced delivery of topotecan into diffuse intrinsic brainstem tumors in children. J. Neurosurg. Pediatr. 2013, 11, 289–295. [Google Scholar] [CrossRef]
- Pöpperl, G.; Goldbrunner, R.; Gildehaus, F.J.; Kreth, F.W.; Tanner, P.; Holtmannspötter, M.; Tonn, J.C.; Tatsch, K. O-(2-[18F]fluoroethyl)-l-tyrosine PET for monitoring the effects of convection-enhanced delivery of paclitaxel in patients with recurrent glioblastoma. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 1018–1025. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Zhou, Z.; Tisnado, J.; Haque, S.; Peck, K.K.; Young, R.J.; Tsiouris, A.J.; Thakur, S.B.; Souweidane, M.M. A novel magnetic resonance imaging segmentation technique for determining diffuse intrinsic pontine glioma tumor volume. J. Neurosurg. Pediatr. 2016, 18, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Guisado, D.I.; Singh, R.; Minkowitz, S.; Zhou, Z.; Haque, S.; Peck, K.K.; Young, R.J.; Tsiouris, A.J.; Souweidane, M.M.; Thakur, S.B. A Novel Methodology for Applying Multivoxel MR Spectroscopy to Evaluate Convection-Enhanced Drug Delivery in Diffuse Intrinsic Pontine Gliomas. Am. J. Neuroradiol. 2016, 37, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Laske, D.W.; Youle, R.J.; Oldfield, E.H. Tumor regression with regional distribution of the targeted toxin TF-CRM107 in patients with malignant brain tumors. Nat. Med. 1997, 3, 1362–1368. [Google Scholar] [CrossRef]
- White, E.; Bienemann, A.; Taylor, H.; Hopkins, K.; Cameron, A.L.; Gill, S.S. A phase I trial of carboplatin administered by convection-enhanced delivery to patients with recurrent/progressive glioblastoma multiforme. Contemp. Clin. Trials 2012, 33, 320–331. [Google Scholar] [CrossRef]
- Souweidane, M.M.; Kramer, K.; Pandit-Taskar, N.; Zhou, Z.; Haque, S.; Zanzonico, P.; Carrasquillo, J.A.; Lyashchenko, S.K.; Thakur, S.B.; Donzelli, M.; et al. Convection-enhanced delivery for diffuse intrinsic pontine glioma: A single-centre, dose-escalation, phase 1 trial. Lancet Oncol. 2018, 19, 1040–1050. [Google Scholar] [CrossRef]
- Heiss, J.D.; Jamshidi, A.; Shah, S.; Martin, S.; Wolters, P.L.; Argersinger, D.P.; Warren, K.E.; Lonser, R.R. Phase I trial of convection-enhanced delivery of IL13-Pseudomonas toxin in children with diffuse intrinsic pontine glioma. J. Neurosurg. Pediatr. 2019, 23, 333–342. [Google Scholar] [CrossRef]
- Saito, R.; Kanamori, M.; Sonoda, Y.; Yamashita, Y.; Nagamatsu, K.; Murata, T.; Mugikura, S.; Kumabe, T.; Wembacher-Schroeder, E.; Thomson, R.; et al. Phase I trial of convection-enhanced delivery of nimustine hydrochloride (ACNU) for brainstem recurrent glioma. Neuro-Oncol. Adv. 2020, 2. [Google Scholar] [CrossRef]
- Rand, R.W.; Kreitman, R.J.; Patronas, N.; Varricchio, F.; Pastan, I.; Puri, R.K. Intratumoral administration of recombinant circularly permuted interleukin-4-Pseudomonas exotoxin in patients with high-grade glioma. Clin. Cancer Res. 2000, 6, 2157–2165. [Google Scholar] [PubMed]
- Weber, F.W.; Floeth, F.; Asher, A.; Bucholz, R.; Berge, M.; Pradoss, M.; Chang, S.; Bruces, J.; Hall, W.; Raino, N.G.; et al. Local convection enhanced delivery of IL4-Pseudomonas exotoxin (NBI-3001) for treatment of patients with recurrent malignant glioma. Oper. Neuromodul. 2003, 88, 93–103. [Google Scholar] [CrossRef]
- Kunwar, S. Convection enhanced delivery of IL13-PE38QQR for treatment of recurrent malignant glioma: Presentation of interim findings from ongoing phase 1 studies. Oper. Neuromodul. 2003, 88, 105–111. [Google Scholar] [CrossRef]
- Sampson, J.H.; Akabani, G.; Archer, G.E.; Bigner, D.D.; Berger, M.S.; Friedman, A.H.; Friedman, H.S.; Ii, J.E.H.; Kunwar, S.; Marcus, S.; et al. Progress Report of a Phase I Study of the Intracerebral Microinfusion of a Recombinant Chimeric Protein Composed of Transforming Growth Factor (TGF)-α and a Mutated form of the Pseudomonas Exotoxin Termed PE-38 (TP-38) for the Treatment of Malignant Brain Tumors. J. Neuro-Oncol. 2003, 65, 27–35. [Google Scholar] [CrossRef]
- Desjardins, A.; Gromeier, M.; Ii, J.E.H.; Beaubier, N.; Bolognesi, D.P.; Friedman, A.H.; Friedman, H.S.; McSherry, F.; Muscat, A.; Nair, S.; et al. Recurrent Glioblastoma Treated with Recombinant Poliovirus. N. Engl. J. Med. 2018, 379, 150–161. [Google Scholar] [CrossRef]
- Shimamura, T.; Husain, S.R.; Puri, R.K. The IL-4 and IL-13 pseudomonas exotoxins: New hope for brain tumor therapy. Neurosurg. Focus 2006, 20, E11. [Google Scholar] [CrossRef]
- Carpentier, A.; Laigle-Donadey, F.; Zohar, S.; Capelle, L.; Behin, A.; Tibi, A.; Martin-Duverneuil, N.; Sanson, M.; Lacomblez, L.; Taillibert, S.; et al. Phase 1 trial of a CpG oligodeoxynucleotide for patients with recurrent glioblastoma. Neuro-Oncology 2006, 8, 60–66. [Google Scholar] [CrossRef]
- Carpentier, A.F.; Metellus, P.; Ursu, R.; Zohar, S.; Lafitte, F.; Barrié, M.; Meng, Y.; Richard, M.; Parizot, C.; Laigle-Donadey, F.; et al. Intracerebral administration of CpG oligonucleotide for patients with recurrent glioblastoma: A phase II study. Neuro-Oncology 2010, 12, 401–408. [Google Scholar] [CrossRef]
- Hadjipanayis, C.G.; Machaidze, R.; Kaluzova, M.; Wang, L.; Schuette, A.J.; Chen, H.; Wu, X.; Mao, H. EGFRvIII Antibody-Conjugated Iron Oxide Nanoparticles for Magnetic Resonance Imaging-Guided Convection-Enhanced Delivery and Targeted Therapy of Glioblastoma. Cancer Res. 2010, 70, 6303–6312. [Google Scholar] [CrossRef]
- Platt, S.R.; Nduom, E.K.; Kent, M.; Freeman, C.; Machaidze, R.; Kaluzova, M.; Wang, L.; Mao, H.; Hadjipanayis, C.G. Canine Model of Convection-Enhanced Delivery of Cetuximab-Conjugated Iron-Oxide Nanoparticles Monitored with Magnetic Resonance Imaging. Clin. Neurosurg. 2012, 59, 107–113. [Google Scholar] [CrossRef]
- Chandramohan, V.; Bao, X.; Keir, S.T.; Pegram, C.N.; Szafranski, S.E.; Piao, H.; Wikstrand, C.J.; McLendon, R.E.; Kuan, C.-T.; Pastan, I.H.; et al. Construction of an Immunotoxin, D2C7-(scdsFv)-PE38KDEL, Targeting EGFRwt and EGFRvIII for Brain Tumor Therapy. Clin. Cancer Res. 2013, 19, 4717–4727. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sivakumar, W.; Torres, S.; Jhaveri, N.; Vaikari, V.P.; Gong, A.; Howard, A.; Golden, E.B.; Louie, S.G.; Schönthal, A.H.; et al. Effects of convection-enhanced delivery of bevacizumab on survival of glioma-bearing animals. Neurosurg. Focus 2015, 38, E8. [Google Scholar] [CrossRef] [PubMed]
- Bogdahn, U.; Hau, P.; Stockhammer, G.; Venkataramana, N.K.; Mahapatra, A.K.; Suri, A.A.; Balasubramaniam, A.; Nair, S.; Oliushine, V.; Parfenov, V.; et al. Targeted therapy for high-grade glioma with the TGF-β2 inhibitor trabedersen: Results of a randomized and controlled phase IIb study. Neuro-Oncology 2011, 13, 132–142. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, S.; Chang, S.M.; Westphal, M.; Vogelbaum, M.; Sampson, J.; Barnett, G.; Shaffrey, M.; Ram, Z.; Piepmeier, J.; Prados, M.; et al. Phase III randomized trial of CED of IL13-PE38QQR vs Gliadel wafers for recurrent glioblastoma. Neuro-Oncology 2010, 12, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Sampson, J.H.; Akabani, G.; Archer, G.E.; Berger, M.S.; Coleman, R.E.; Friedman, A.H.; Friedman, H.S.; Greer, K.; Herndon, J.E.; Kunwar, S.; et al. Intracerebral infusion of an EGFR-targeted toxin in recurrent malignant brain tumors. Neuro-Oncology 2008, 10, 320–329. [Google Scholar] [CrossRef]
- Vogelbaum, M.A.; Sampson, J.H.; Kunwar, S.; Chang, S.M.; Shaffrey, M.; Asher, A.L.; Lang, F.F.; Croteau, D.; Parker, K.; Grahn, A.Y.; et al. Convection-enhanced delivery of cintredekin besudotox (interleukin-13-PE38QQR) followed by radiation therapy with and without temozolomide in newly diagnosed malignant gliomas: Phase 1 study of final safety results. Neurosurgery 2007, 61, 1031–1037; discussion 1037–1038. [Google Scholar] [CrossRef]
- Kunwar, S.; Prados, M.D.; Chang, S.M.; Berger, M.S.; Lang, F.F.; Piepmeier, J.M.; Sampson, J.H.; Ram, Z.; Gutin, P.H.; Gibbons, R.D.; et al. Direct Intracerebral Delivery of Cintredekin Besudotox (IL13-PE38QQR) in Recurrent Malignant Glioma: A Report by the Cintredekin Besudotox Intraparenchymal Study Group. J. Clin. Oncol. 2007, 25, 837–844. [Google Scholar] [CrossRef]
- Sampson, J.H.; Brady, M.L.; Petry, N.A.; Croteau, D.; Friedman, A.H.; Friedman, H.S.; Wong, T.; Bigner, D.D.D.; Pastan, I.; Puri, R.K.K.; et al. Intracerebral Infusate Distribution by Convection-enhanced Delivery in Humans with Malignant Gliomas: Descriptive Effects of Target Anatomy and Catheter Positioning. Oper. Neurosurg. 2007, 60, ONS-89–ONS-98; discussion ONS98–ONS99. [Google Scholar] [CrossRef]
- Seo, Y.-E.; Bu, T.; Saltzman, W.M. Nanomaterials for convection-enhanced delivery of agents to treat brain tumors. Curr. Opin. Biomed. Eng. 2017, 4, 1–12. [Google Scholar] [CrossRef]
- Nance, E.; Zhang, C.; Shih, T.-Y.; Xu, Q.; Schuster, B.S.; Hanes, J. Brain-Penetrating Nanoparticles Improve Paclitaxel Efficacy in Malignant Glioma Following Local Administration. ACS Nano 2014, 8, 10655–10664. [Google Scholar] [CrossRef]
- Zhang, C.; Nance, E.A.; Mastorakos, P.; Chisholm, J.; Berry, S.; Eberhart, C.; Tyler, B.; Brem, H.; Suk, J.S.; Hanes, J. Convection enhanced delivery of cisplatin-loaded brain penetrating nanoparticles cures malignant glioma in rats. J. Control. Release 2017, 263, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, P.J.; LeCouteur, R.A.; Higgins, R.J.; Bringas, J.R.; Roberts, B.; Larson, R.F.; Yamashita, Y.; Krauze, M.; Noble, C.O.; Drummond, D.; et al. Canine model of convection-enhanced delivery of liposomes containing CPT-11 monitored with real-time magnetic resonance imaging: Laboratory investigation. J. Neurosurg. 2008, 108, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Krauze, M.T.; Vandenberg, S.R.; Yamashita, Y.; Saito, R.; Forsayeth, J.; Noble, C.; Park, J.; Bankiewicz, K.S. Safety of real-time convection-enhanced delivery of liposomes to primate brain: A long-term retrospective. Exp. Neurol. 2008, 210, 638–644. [Google Scholar] [CrossRef] [PubMed]
- MTX110 by Convection-Enhanced Delivery in Treating Participants with Newly-Diagnosed Diffuse Intrinsic Pontine Glioma—Full Text View—ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT03566199?term=convection+enhanced+delivery+nanoparticle&cond=glioma&draw=2&rank=1 (accessed on 4 December 2020).
- Case Medical Research CED of MTX110 Newly Diagnosed Diffuse Midline Gliomas. Case Med. Res. 2020. [CrossRef]
- Ryan, D.A.; Mastrangelo, M.A.; Narrow, W.C.; Sullivan, M.A.; Federoff, H.J.; Bowers, W.J. Abeta-directed single-chain antibody delivery via a serotype-1 AAV vector improves learning behavior and pathology in Alzheimer’s disease mice. Mol. Ther. 2010, 18, 1471–1481. [Google Scholar] [CrossRef]
- Carty, N.; Lee, D.C.; Dickey, C.A.; Ceballos-Diaz, C.; Jansen-West, K.; Golde, T.E.; Gordon, M.N.; Morgan, D.; Nash, K.R. Convection-enhanced delivery and systemic mannitol increase gene product distribution of AAV vectors 5, 8, and 9 and increase gene product in the adult mouse brain. J. Neurosci. Methods 2010, 194, 144–153. [Google Scholar] [CrossRef]
- Barua, N.; Miners, J.S.; Bienemann, A.S.; Wyatt, M.J.; Welser, K.; Tabor, A.B.; Hailes, H.C.; Love, S.; Gill, S.S. Convection-Enhanced Delivery of Neprilysin: A Novel Amyloid-β-Degrading Therapeutic Strategy. J. Alzheimer’s Dis. 2012, 32, 43–56. [Google Scholar] [CrossRef]
- Ksendzovsky, A.; Walbridge, S.; Saunders, R.C.; Asthagiri, A.R.; Heiss, J.D.; Lonser, R.R. Convection-enhanced delivery of M13 bacteriophage to the brain. J. Neurosurg. 2012, 117, 197–203. [Google Scholar] [CrossRef]
- Ates, G.; Goldberg, J.; Currais, A.; Maher, P. CMS121, a fatty acid synthase inhibitor, protects against excess lipid peroxidation and inflammation and alleviates cognitive loss in a transgenic mouse model of Alzheimer’s disease. Redox Biol. 2020, 36, 101648. [Google Scholar] [CrossRef]
- Currais, A.; Huang, L.; Goldberg, J.; Petrascheck, M.; Ates, G.; Pinto-Duarte, A.; Shokhirev, M.N.; Schubert, D.; Maher, P. Elevating acetyl-CoA levels reduces aspects of brain aging. Elife 2019, 8, e47866. [Google Scholar] [CrossRef]
- Nagahara, A.H.; Wilson, B.R.; Ivasyk, I.; Kovacs, I.; Rawalji, S.; Bringas, J.R.; Pivirotto, P.J.; Sebastian, W.S.; Samaranch, L.; Bankiewicz, K.S.; et al. MR-guided delivery of AAV2-BDNF into the entorhinal cortex of non-human primates. Gene Ther. 2018, 25, 104–114. [Google Scholar] [CrossRef]
- Park, H.W.; Park, C.G.; Park, M.; Lee, S.H.; Park, H.R.; Lim, J.; Paek, S.H.; Choy, Y.B. Intrastriatal administration of coenzyme Q10 enhances neuroprotection in a Parkinson’s disease rat model. Sci. Rep. 2020, 10, 9572. [Google Scholar] [CrossRef] [PubMed]
- Heiss, J.D.; Walbridge, S.; Argersinger, D.P.; Hong, C.S.; Ray-Chaudhury, A.; Lonser, R.R.; Elias, W.J.; Zaghloul, K.A. Convection-Enhanced Delivery of Muscimol Into the Bilateral Subthalamic Nuclei of Nonhuman Primates. Neurosurgery 2018, 84, E420–E429. [Google Scholar] [CrossRef] [PubMed]
- Heiss, J.D.; Lungu, C.; Hammoud, D.A.; Herscovitch, P.; Ehrlich, D.J.; Bs, D.P.A.; Sinharay, S.; Scott, G.; Wu, T.; Federoff, H.J.; et al. Trial of magnetic resonance–guided putaminal gene therapy for advanced Parkinson’s disease. Mov. Disord. 2019, 34, 1073–1078. [Google Scholar] [CrossRef]
- Valles, F.; Fiandaca, M.S.; Eberling, J.L.; Starr, P.A.; Larson, P.S.; Christine, C.W.; Forsayeth, J.; Richardson, R.M.; Su, X.; Aminoff, M.J.; et al. Qualitative Imaging of Adeno-Associated Virus Serotype 2–Human Aromatic L-Amino Acid Decarboxylase Gene Therapy in a Phase I Study for the Treatment of Parkinson Disease. Neurosurgery 2010, 67, 1377–1385. [Google Scholar] [CrossRef] [PubMed]
- Slevin, J.T.; Gash, D.M.; Smith, C.D.; Gerhardt, G.A.; Kryscio, R.; Chebrolu, H.; Walton, A.; Wagner, R.; Young, A.B. Unilateral intraputamenal glial cell line–derived neurotrophic factor in patients with Parkinson disease: Response to 1 year of treatment and 1 year of withdrawal. J. Neurosurg. 2007, 106, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Whone, A.; Luz, M.; Boca, M.; Woolley, M.; Mooney, L.; Dharia, S.; Broadfoot, J.; Cronin, D.; Schroers, C.; Barua, N.U.; et al. Randomized trial of intermittent intraputamenal glial cell line-derived neurotrophic factor in Parkinson’s disease. Brain 2019, 142, 512–525. [Google Scholar] [CrossRef]
| Author | Description | Investigational Drug | Device(s) Used | Flow Rate/Time | Results |
|---|---|---|---|---|---|
| Heiss et al. 2018 [51] | Phase I study of five pediatric DIPG patients. NCT00088061 | IL13-Pseudomonas toxin | Navigus catheter guide, Medfusion 3500 pump | 5–10 uL/min, maximum 13 h | Infusions led to transient CN deficits. All patients had disease progression by 12 weeks post-infusion. |
| Desjardins et al. 2018 [57] | Phase I dose escalation in 61 patients with recurrent GBM. NCT01491893 | PVSRIPO | Vygon PIC030 Sophysa catheter, Medfusion 3500 pump | 500 ul/h, 6.5 h | 21% of patients survived at 24 and 36 months. |
| Souweidane et al. 2018 [50] | Phase I dose escalation study in 28 pediatric DIPG patients. NCT01502917 | 124I-8H9 | SmartFlow cannula | Max 7.5 uL/min | No dose limiting toxicities observed. Across the seven doses, a significant dose dependent increase in overall survival was observed. |
| Anderson et al. 2012 [44] | Phase IB in two pediatric patients with DIPG. NCT00308165 | Topotecan | Silastic infusion catheter, Medfusion 2010 pump | <0.04 mL/h, Vi 5–6 mL over 100 h | Brainstem CED is feasible. Flow rates over 12 mL/h were associated with new neurological deficits. |
| White et al. 2012 [49] | Phase I dose escalation study in 18 patients with recurrent or progressive GBM | Carboplatin | In-house catheter | 8 or 16 h | N/A |
| Bogdahn et al. 2011 [65] | Phase IIB for 145 patients with recurrent GBM/AA | Trabedersen (TGF-B2 inhibitor) | Subcutaneous port access system with external pump | 1 uL/min up, up to 11 treatment cycles | Trend towards a survival benefit for CED of 10 uM Trabedersen (39.1 months), 80 uM Trabedersen (35.2 months), chemotherapy alone (21.7 months). |
| Bruce et al. 2011 [40] | Phase IB dose escalation study of 10 GBM patients and six AA patients. NCT00308165 | Topotecan | Silastic infusion catheter, 2.5 mm diameter | 200 uL/h for 100 h | Six month PFS for GBM patients was 44%, AA patients 75%. Sixty-nine percent demonstrated radiographic response. |
| Kunwar et al. 2010 [66] | Phase III trial of 296 recurrent GBM patients. NCT00076986 | CB (IL13-PE) vs. gliadel wafers | 2–4 catheters used | 0.75 mL/h over 96 h | Median survival was 45.3 weeks for CED patients and 39.8 weeks for gliadel patients (p = 0.310). |
| Carpentier et al. 2010 [61] | Phase II trial in 34 recurrent GBM patients. NCT00190424 | CpG: Immunostimulating motifs | Seldiflex and Plastimed catheters | 3.3 uM/h over 6 h | 6 month PFS: 19%. Median OS 28 weeks. |
| Sampson et al. 2008 [67] | Phase I dose escalation study of 20 recurrent glioma patients | TGF-alpha PE (TP38) | PS Medical CSF ventricular catheter, outer diameter 2.1 mm | 0.4 mL/h over 50 h | Co-infusion with 123I-albumin showed Vd of > 4 cm away from catheter. A total of 2/15 patients with radiographic response. |
| Vogelbaum et al. 2007 [68] | Phase I trial in 21 patients with newly diagnosed GBM. NCT00089427 | CB (IL13-PE) | Vygon Neuro: 1 mm inner diameter barium impregnated catheter | 0.75 mL/h over 96 h | No dose limiting toxicities at a dose of 0.25 ug/mL with EBRT and TMZ. |
| Kunwar et al. 2007 [69] | Phase I trial in 51 GBM and AA patients. | CB (IL13-PE) | N/A | 0.75 mL/h over 96 h | Patients with optimally placed catheters survived longer (55.6 vs. 37.4 weeks p = 0.03). |
| Sampson et al. 2007 [70] | Phase I study of seven patients with recurrent malignant glioma | CB + 123I-labeled albumin | Vygon Neuro - 1 mm inner diameter barium impregnated catheter | 0.540–0.750 mL/h over 96 h | SPECT demonstrated that intratumoral infusion led to poor volume of distribution. |
| Carpentier et al. 2006 [59] | Phase I study with 24 recurrent GBM patients | CpG immuostimulatory oligonucleotides | N/A | Median survival 7.2 months with only one dose limiting toxicity observed. | |
| Popperl et al. 2005 [45] | Phase I study with eight recurrent GBM patients | Paclitaxel | Silicone catheter, Medfusion 2010 pump | 0.3 mL/h over 120 h | FET-PET uptake correlates with tumor burden and decreased following CED. |
| Lidar et al. 2004 [26] | Phase I/II trial of 15 recurrent GBM/AA patients | Paclitaxel | VPS Medtronic silicone catheter, Medfusion 2010 pump | 0.3 mL/h over 120 h | DW-MRI showed five complete responses and six partial responses out of 15 cases. Responses were confirmed via biopsies and en-bloc resection. |
| Sampson et al. 2003 [56] | Phase I dose escalation study of 20 recurrent malignant glioma patients | TGF-alpha PE (TP38) | N/A | 0. 4 mL/h over 50 h | A total of 3/15 patients with radiographic response and 4/20 have no evidence of tumor recurrence. |
| Weber et al. 2003 [54] | Phase I dose escalation of 25 GBM and six AA patients NCT00003842 | IL-4 PE | N/A | 96 h infusion | Median survival of GBM patients was 5.8 months following treatment. Thirty-nine percent of patients had drug related toxicity. |
| Laske et al. 1997 [48] | Phase I dose escalation of 10 GBM, five AA, one AO, and one LC patients | Tf-CRM107 | Silastic infusion catheters (2.5 mm outer diameter), Medfusion 2001 syringe pump | 4–10 µL/min over 4 h | A total of 9/15 patients had 50% reduction in tumor volume with two complete responses. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Upadhyayula, P.S.; Spinazzi, E.F.; Argenziano, M.G.; Canoll, P.; Bruce, J.N. Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience. Pharmaceutics 2021, 13, 39. https://doi.org/10.3390/pharmaceutics13010039
Upadhyayula PS, Spinazzi EF, Argenziano MG, Canoll P, Bruce JN. Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience. Pharmaceutics. 2021; 13(1):39. https://doi.org/10.3390/pharmaceutics13010039
Chicago/Turabian StyleUpadhyayula, Pavan S., Eleonora F. Spinazzi, Michael G. Argenziano, Peter Canoll, and Jeffrey N. Bruce. 2021. "Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience" Pharmaceutics 13, no. 1: 39. https://doi.org/10.3390/pharmaceutics13010039
APA StyleUpadhyayula, P. S., Spinazzi, E. F., Argenziano, M. G., Canoll, P., & Bruce, J. N. (2021). Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience. Pharmaceutics, 13(1), 39. https://doi.org/10.3390/pharmaceutics13010039





