Development and Evaluation of a Reconstitutable Dry Suspension Containing Isoniazid for Flexible Pediatric Dosing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Formulation of the Reconstitutable Dry Suspension Employing a Direct Dispersion Emulsification Method
2.3. Formulation Yield
2.4. Physicochemical Characterization of the Reconstitutable Dry Suspension
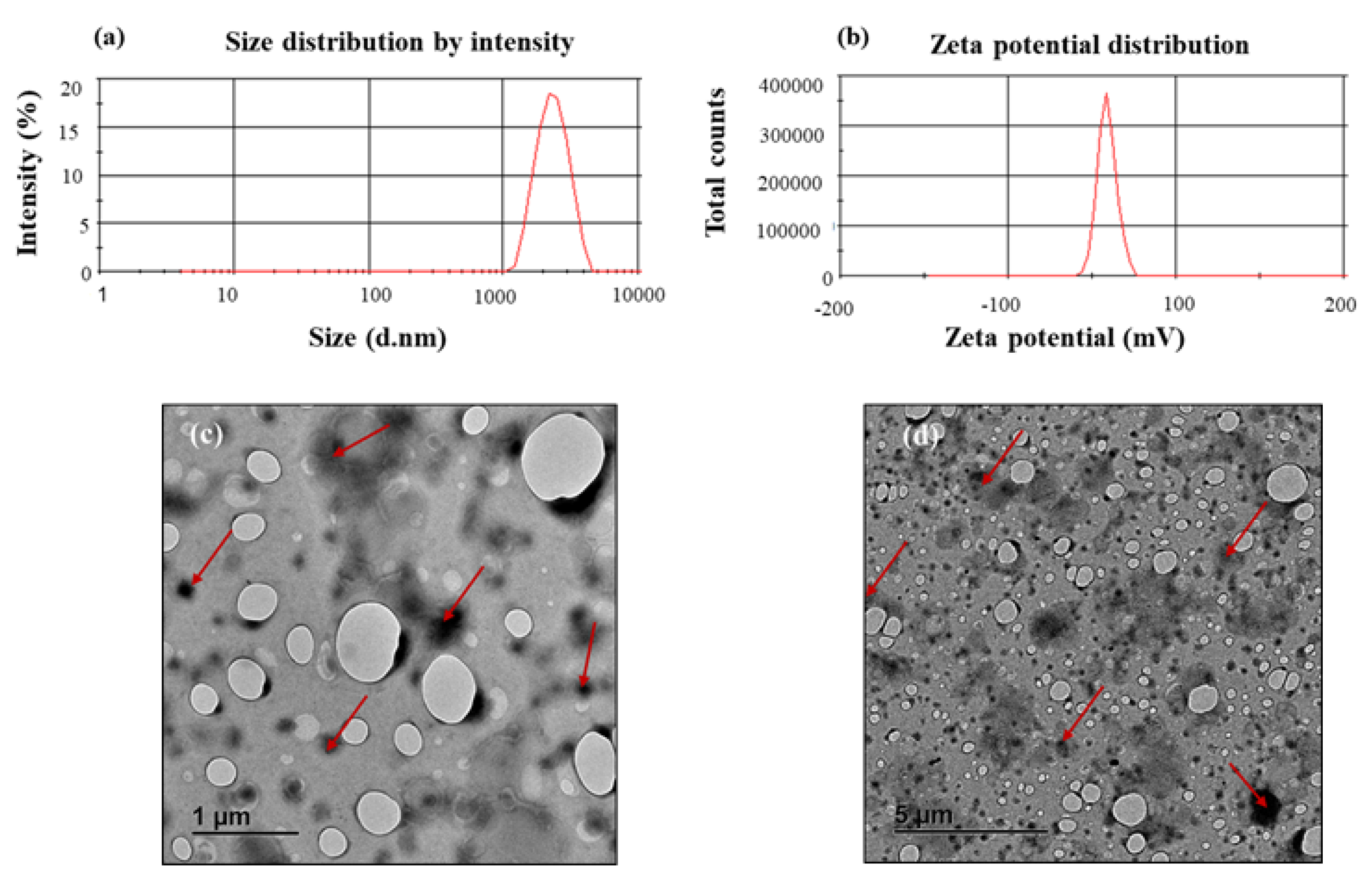
2.4.1. Particle Size, Polydispersity Index, and Zeta Potential
2.4.2. Visualization of Surface Morphology
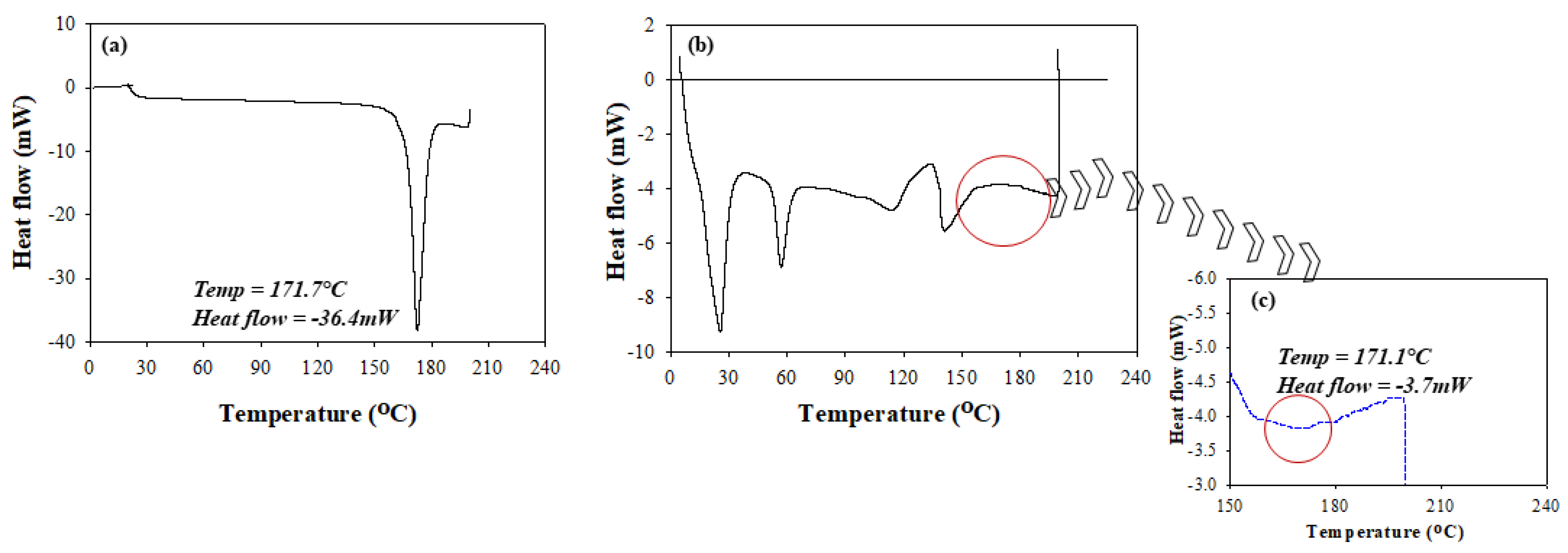
2.4.3. Thermal Analysis
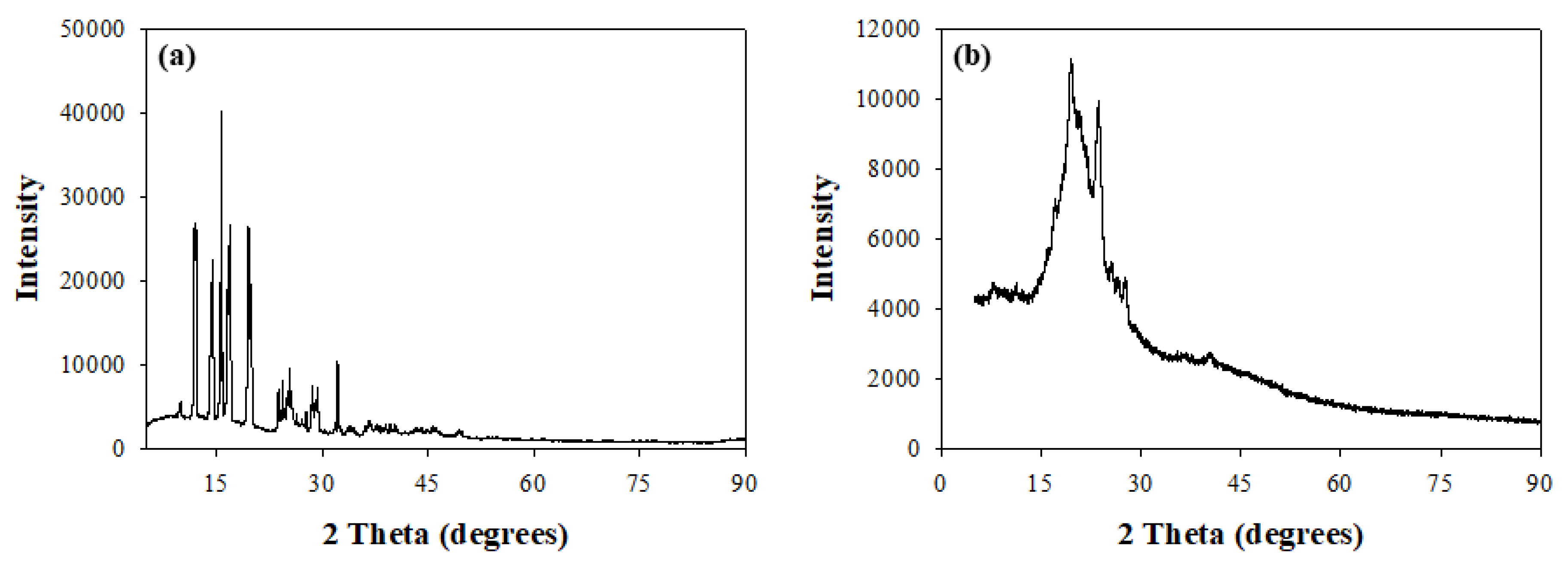
2.4.4. Determination of Crystallinity
2.4.5. Structural Elucidation
2.5. Drug Loading Efficiency
2.6. In Vitro Dissolution Studies
2.7. Stability Evaluations
2.7.1. Short-Term Stability Testing under Different Environmental Conditions
2.7.2. In Vitro Aqueous Stability Assessment
2.8. Preliminary Formulation Toxicity Assessment
3. Results and Discussions
3.1. Formulation Synthesis and Yield
3.2. Size, Polydispersity Index, and Zeta Potential Determination and Morphology
3.3. Thermal Behavior
3.4. X-ray Diffractometry
3.5. Structural Analysis
3.6. Drug Content
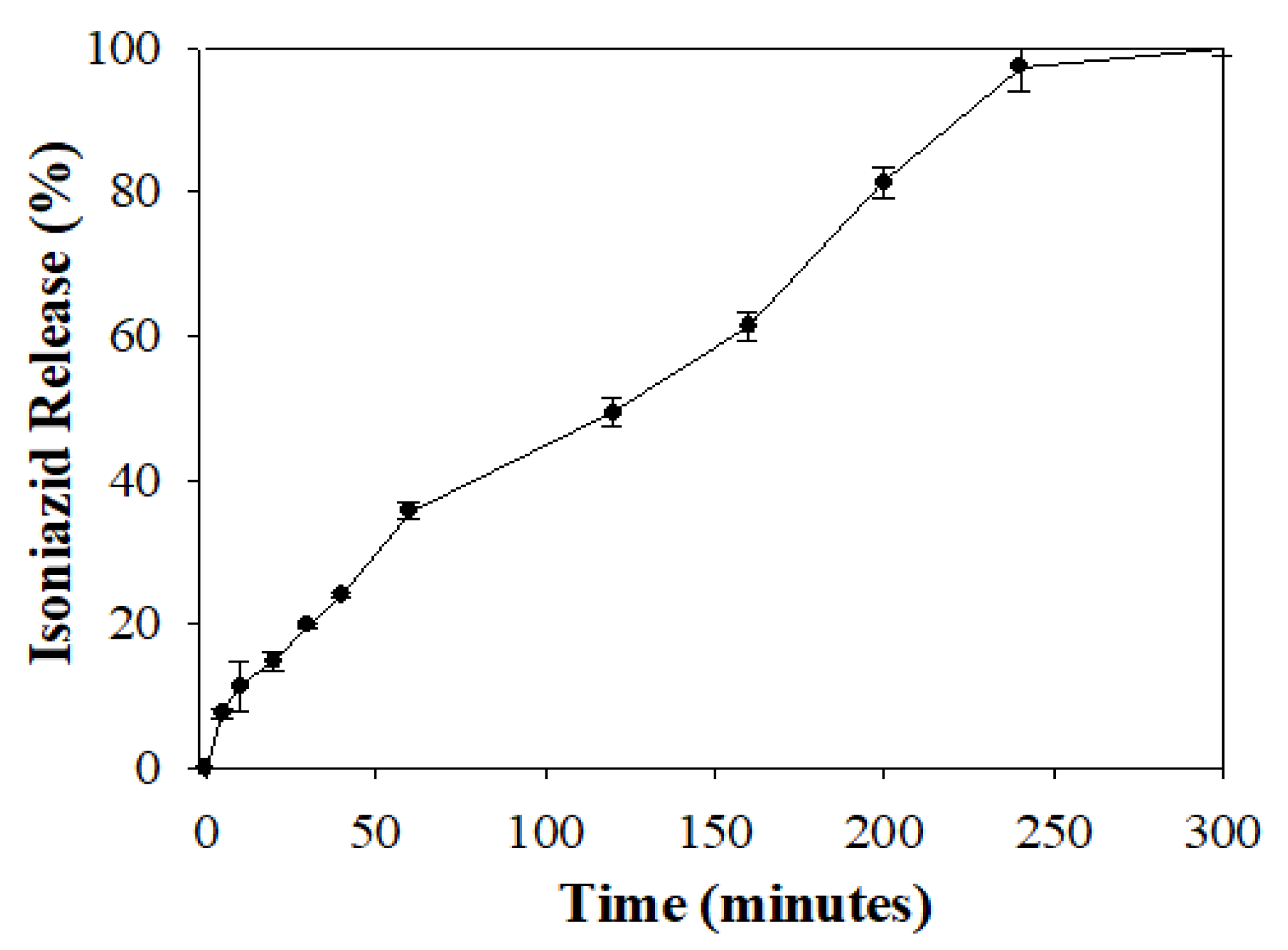
3.7. Drug Release Behavior
3.8. Short-Term Formulation Stability Assessment
3.8.1. Stability Evaluation under Varying Storage Conditions
3.8.2. Formulation Stability in an Aqueous Environment
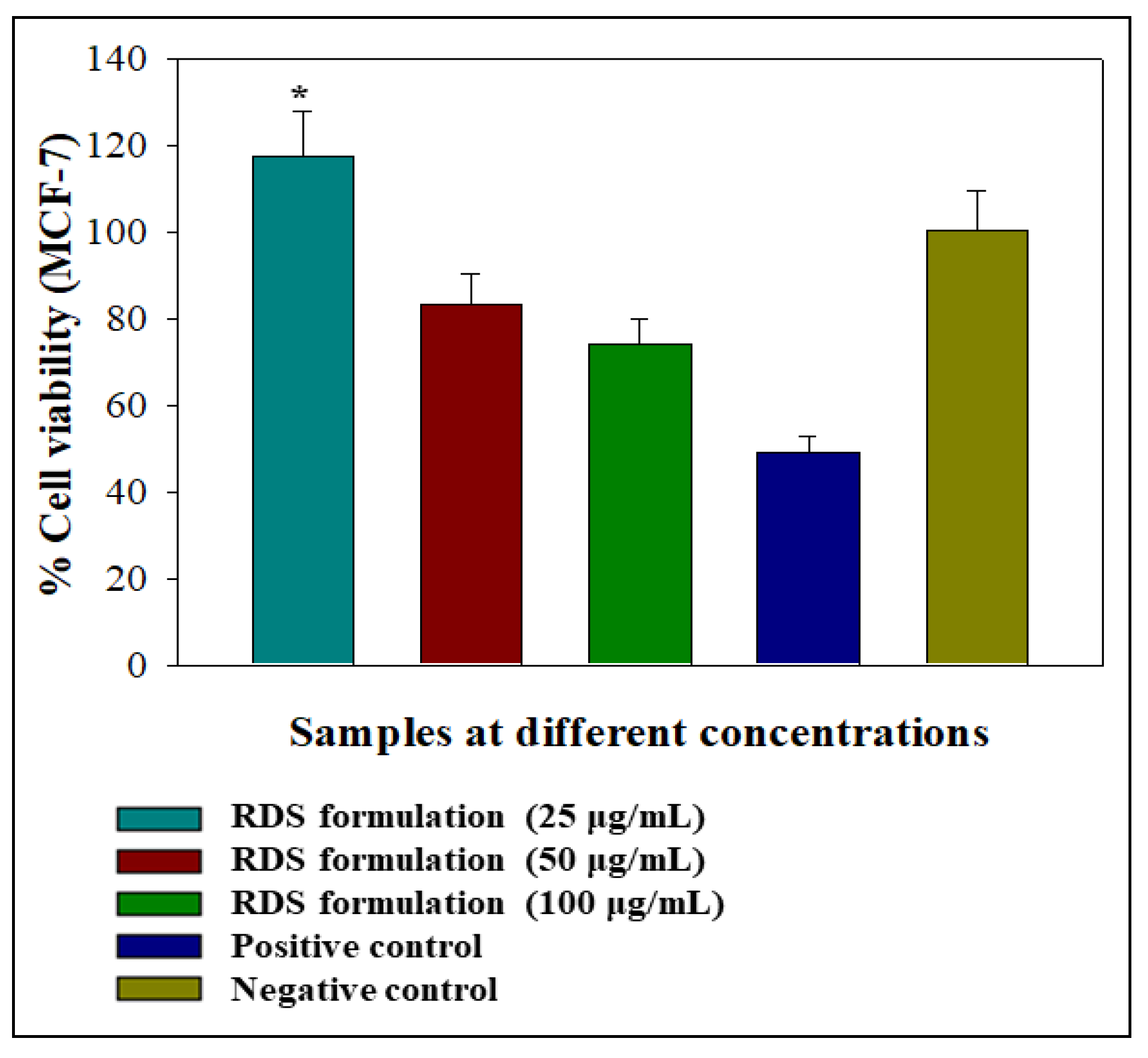
3.9. Cell Viability Assessment
4. Conclusions and Future Work
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. WHO Global TB Report. 2018. Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 10 December 2019).
- TB Alliance. The Pandemic. 2019. Available online: https://www.tballiance.org/why-new-tb-drugs/global-pandemic (accessed on 2 December 2019).
- Suárez-González, J.; Santoveña-Estévez, A.; Soriano, M.; Fariña, J.B. Design and optimization of a child-friendly dispersible tablet containing Isoniazid, Pyrazinamide and Rifampicin for treating Tuberculosis in pediatrics. Drug Dev. Ind. Pharm. 2020, 46, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Venturini, E.; Turkova, A.; Chiappini, E.; Gali, L.; de Martino, M.; Thorne, C. Tuberculosis and HIV co-infection in children. BMC Infect. Dis. 2014, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.; Shenoi, S.V.; Friedland, G.H. Opportunities for community health workers to contribute to global efforts to end tuberculosis. Glob. Public Health 2020, 15, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.G.Y.; Chan, H.K.; Prestidge, C.A.; Denman, J.A.; Young, P.M.; Traini, D. A novel dry powder inhalable formulation incorporating three first-line anti-tubercular antibiotics. Eur. J. Pharm. Biopharm. 2013, 83, 285–292. [Google Scholar] [CrossRef]
- Hussain, A.; Shakeel, F.; Singh, S.K.; Alsarra, I.A.; Faruk, A.; Alanazi, F.K.; Christoper, G.P. Solidified SNEDDS for the oral delivery of rifampicin: Evaluation, proof of concept, in vivo kinetics, and in silico GastroPlusTM simulation. Int. J. Pharm. 2019, 566, 203–217. [Google Scholar] [CrossRef]
- Hamzaoui, A.; Yalaoui, A.; Cherif, F.A.; Slim, L.; Barraies, A. Childhood tuberculosis: A concern for the modern world. Eur. Respir. Update Tuberc. 2014, 23, 278–291. [Google Scholar] [CrossRef]
- Jenkins, H.E. Global burden of childhood tuberculosis. Pneumonia 2016, 8, 24. [Google Scholar] [CrossRef]
- Peña, M.J.M.; García, B.S.; Baquero-Artigao, F.; Pérez, D.M.; Pérez, R.P.; Echevarría, A.M.; Amador, J.T.R.; Durán, D.G.P.; Julian, A.N.; Tuberculosis, W.G.; et al. Tuberculosis treatment for children: An update. An. Pediatr. 2018, 88, 52.e1–52.e12. [Google Scholar]
- Pérez, R.P.; García, B.S.; Fernández-Llamazares, C.M.; Artigao, F.B.; Julia, A.N.; Péna, M.J.M. The challenge of administering anti-tuberculosis treatment in infants and pre-school children. An. Pediatr. 2016, 8, 4–12. [Google Scholar]
- Samad, A.; Sultana, Y.; Khar, R.K.; Aqil, M.; Chuttani, K.; Mishra, A.K. Reconstituted powder for formulation of antitubercular drugs formulated as microspheres for paediatric use. Drug Discov. Ther. 2008, 2, 108–114. [Google Scholar]
- Shanbhag, P.P.; Bhalerao, S.S. Development and evaluation of oral reconstitutable systems of cephalexin. Int. J. Pharm. Tech. Res. 2010, 2, 502–506. [Google Scholar]
- Pouplin, T.; Phuong, P.N.; Van Toi, P.; Pouplin, J.N.; Farrar, J. Isoniazid, pyrazinamide and rifampicin content variation in split fixed-dose combination tablets. PLoS ONE 2014, 9, e102047. [Google Scholar] [CrossRef] [PubMed]
- Lopez, F.L.; Ernest, T.B.; Tuleu, C.; Gul, M.O. Formulation approaches to paediatric oral drug delivery: Benefits and limitations of current platforms. Expert Opin. Drug Deliv. 2015, 12, 1727–1740. [Google Scholar] [CrossRef] [PubMed]
- Long, S.S.; Prober, C.G.; Fischer, M. Principles and Practice of Pediatric Infectious Diseases E-Book, 5th ed.; Elsevier Health Sciences: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Grange, J.M.; Schaaf, H.S.; Zumla, A. Tuberculosis: A Comprehensive Clinical Reference; Saunders/Elsevier: Philadelphia, PA, USA, 2009. [Google Scholar]
- Moretton, M.A.; Cagel, M.; Bernabeu, E.; Gonzalez, L.; Chiappetta, D.A. Nanopolymersomes as potential carriers for rifampicin pulmonary delivery. Colloids Surf. B Biointerfaces 2015, 136, 1017–1025. [Google Scholar] [CrossRef]
- Adeleke, O.A.; Tsai, P.C.; Karry, K.M.; Monama, N.O.; Michniak-Kohn, B.B. Isoniazid-loaded orodispersible strips: Methodical design, optimization and in vitro-in silico characterization. Int. J. Pharm. 2018, 547, 347–359. [Google Scholar] [CrossRef]
- Brennan, P.J.; Young, D.B. Handbook of Anti-Tuberculosis Agents. Tuberculosis 2008, 88, 112–116. [Google Scholar]
- Fülöp, V.; Jakab, G.; Bozó, T.; Tóth, B.; Endrésik, D.; Balogh, E.; Kellermayer, M.; Antal, I. Study on the dissolution improvement of albendazole using reconstitutable dry nanosuspension formulation. Eur. J. Pharm. Sci. 2018, 123, 70–78. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. An overview of clinical and commercial impact of drug delivery systems. J. Control. Release 2014, 190, 15–28. [Google Scholar] [CrossRef]
- Yin Win, K.; Feng, S. Effects of particle size and surface coating on cellular uptake of polymeric nanoparticles for oral delivery of anticancer drugs. Biomaterials 2005, 26, 2713–2722. [Google Scholar] [CrossRef]
- Öztürk, A.A.; Yenilmez, E.; Özarda, M.G. Clarithromycin-Loaded Poly (Lactic-co-glycolic acid) (PLGA) Nanoparticles for Oral Administration: Effect of Polymer Molecular Weight and Surface Modification with Chitosan on Formulation, Nanoparticle Characterization and Antibacterial Effects. Polymers 2019, 11, 1632. [Google Scholar] [CrossRef]
- Kodama, T.; Tomita, N.; Horie, S.; Sax, N.; Iwasaki, H.; Suzuki, R.; Maruyama, K.; Mori, S.; Manabu, F. Morphological study of acoustic liposomes using transmission electron microscopy. J. Electron Microsc. 2009, 59, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Jermy, B.R.; Alomari, M.; Ravinayagam, V.; Almofty, S.A.; Akhtar, S.; Borgio, J.F.; AbdulAzeez, S. SPIONs/3D SiSBA-16 based Multifunctional Nanoformulation for target specific cisplatin release in colon and cervical cancer cell lines. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Trinh, H.M.; Agrahari, V.; Sheng, Y.; Pal, D.; Mitra, A.K. Nanoparticle-based topical ophthalmic gel formulation for sustained release of hydrocortisone butyrate. AAPS Pharm. Sci. Tech. 2016, 17, 294–306. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Stability Testing of Pharmaceutical Products Containing Well-Established Drug Substances in Conventional Dosage Forms. WHO Technical Report Series 863, Annex 5. 1996. Available online: https://apps.who.int/medicinedocs/pdf/s5516e/s5516e.pdf (accessed on 24 February 2020).
- Dodiya, S.; Chavhan, S.; Korde, A.; Sawant, K.K. Solid lipid nanoparticles and nanosuspension of adefovir dipivoxil for bioavailability improvement: Formulation, characterization, pharmacokinetic and biodistribution studies. Drug Dev. Ind. Pharm. 2013, 39, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Manaia, E.B.; Abuçafy, M.P.; Chiari-Andréo, B.G.; Silva, B.L.; Junior, J.A.O.; Chiavacci, L.A. Physicochemical characterization of drug nanocarriers. Int. J. Nanomed. 2017, 12, 4991. [Google Scholar] [CrossRef]
- Maciel, V.B.; Yoshida, C.M.; Pereira, S.M.; Goycoolea, F.M.; Franco, T.T. Electrostatic self-assembled chitosan-pectin nano-and microparticles for insulin delivery. Molecules 2017, 22, E1707. [Google Scholar] [CrossRef]
- Afinjuomo, F.; Barclay, T.G.; Parikh, A.; Chung, R.; Song, Y.; Nagalingam, G.; Triccas, J.; Wang, L.; Liu, L.; Hayball, J.D.; et al. Synthesis and Characterization of pH-Sensitive Inulin Conjugate of Isoniazid for Monocyte-Targeted Delivery. Pharmaceutics 2019, 11, 555. [Google Scholar] [CrossRef]
- Siepmann, J.; Siepmann, F. Mathematical modeling of drug delivery. Int. J. Pharm. 2008, 364, 328–343. [Google Scholar] [CrossRef]
- Paolino, D.; Tudose, A.; Celia, C.; Di Marzio, L.; Cilurzo, F.; Mircioiu, C. Mathematical Models as Tools to Predict the Release Kinetic of Fluorescein from Lyotropic Colloidal Liquid Crystals. Materials 2019, 12, 693. [Google Scholar] [CrossRef]
- Bácskay, I.; Nemes, D.; Fenyvesi, F.; Váradi, J.; Vasvári, G.; Fehér, P.; Vecsernyés, M.; Ujhelyi, Z. Role of Cytotoxicity Experiments in Pharmaceutical Development. In Cytotoxicity; Çelik, T.A., Ed.; InTech: London, UK, 2018; ISBN 978-1-78923-430-5. [Google Scholar]
- Senthilraja, P.; Kathiresan, K. In vitro cytotoxicity MTT assay in Vero, HepG2 and MCF-7 cell lines study of Marine Yeast. J. Appl. Pharm. Sci. 2015, 5, 80–84. [Google Scholar] [CrossRef]
- Garcia-Fernandez, J.; Turiel, D.; Bettmer, J.; Jakubowski, N.; Panne, U.; Rivas García, L.; Llopis, J.; Sánchez González, C.; Montes-Bayón, M. In vitro and in situ experiments to evaluate the biodistribution and cellular toxicity of ultrasmall iron oxide nanoparticles potentially used as oral iron supplements. Nanotoxicology 2020, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Hormesis: Path and progression to significance. Int. J. Mol. Sci. 2018, 19, 2871. [Google Scholar] [CrossRef] [PubMed]
- Kloner, R.A. Remote Ischemic Conditioning as a Form of Hormesis. In The Science of Hormesis in Health and Longevity; Academic Press: Amsterdam, The Netherlands, 2019. [Google Scholar]
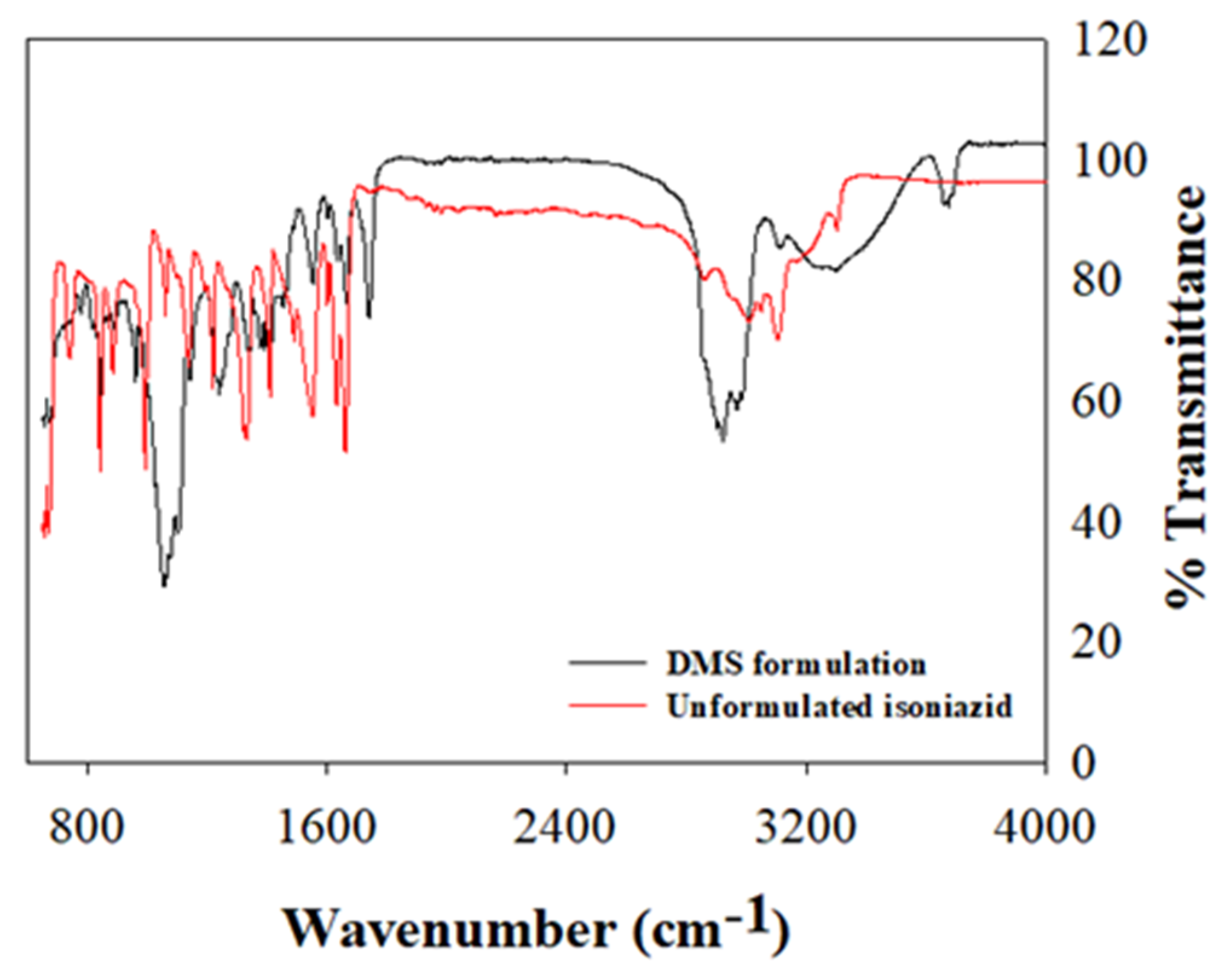
| Functional Groups and Remarks | Vibrational Frequencies (cm−1) | |
|---|---|---|
| Unformulated Isoniazid | Isoniazid Loaded RDS Formulation | |
| C–C–C–C and C–N–C–C torsion | 653 | 665 |
| NH2 rock | 677 | 677 |
| C–C–C out of plane bending | 746 | 757 |
| C–C–H out of plane bending | 850 and 1022 | 850 and 1023 |
| C–N–C and C–C–C in plane bending | 1063 | 1062 |
| Aromatic C–N stretching | 1344 | 1330 |
| C–C–H in plane bending | 1210 | 1212 |
| O=C-N in plane bending | 1330 | 1333 |
| C–N–H in plane bending | 1411 | 1408 |
| C–C stretching | 1477 | 1471 |
| C=O stretching | 1558 | 1552 |
| NH2 scissoring | 1632 | 1635 |
| Aromatic C–H stretching | 3052 | 3054 |
| N–H stretching | 3307 | 3308 |
| O–H stretching | - | 3676 |
| Mathematical Models | R2 | AIC |
|---|---|---|
| Zero order | 0.976 | 74.080 |
| First order | 0.159 | 176.881 |
| Second order | 0.091 | 129.633 |
| Korsmeyer-Peppas | 0.997 | 110.291 |
| Weibull | 0.990 | 92.616 |
| Michaelis-Menten | 1.000 | 120.737 |
| Higuchi | -0.341 | 127.138 |
| RDS Formulation Test Conditions | Time (Months) | Stability Indicators | ||||
|---|---|---|---|---|---|---|
| Stability tester | PDIa | ZPb (mV) | PSc (µm) | DCd (%) | Color | |
| 0 | 0.37 | −41.10 | 1.63 | 94.12 | No change | |
| 1 | 0.40 | −42.59 | 1.58 | 94.07 | No change | |
| 4 | 0.39 | −43.60 | 1.82 | 93.76 | Slight change | |
| Room | 0 | 0.37 | −41.10 | 1.63 | 94.12 | No change |
| 1 | 0.44 | −42.71 | 1.69 | 93.95 | No change | |
| 4 | 0.46 | −40.51 | 1.97 | 91.05 | Slight change | |
| Refrigerator | 0 | 0.37 | −41.10 | 1.63 | 94.12 | No change |
| 1 | 0.39 | −41.65 | 1.61 | 93.95 | No change | |
| 4 | 0.38 | −40.91 | 1.70 | 94.02 | No change | |
| Test Conditions | Time-Points (Days) | Drug Content (%) | Discoloration |
|---|---|---|---|
| Room/Ambient | 0 | 94.12 | None |
| 1 | 93.18 | None | |
| 5 | 93.61 | None | |
| 11 | 92.89 | None | |
| Refrigerator | 0 | 94.12 | None |
| 1 | 93.33 | None | |
| 5 | 93.79 | None | |
| 11 | 92.56 | None |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adeleke, O.A.; Hayeshi, R.K.; Davids, H. Development and Evaluation of a Reconstitutable Dry Suspension Containing Isoniazid for Flexible Pediatric Dosing. Pharmaceutics 2020, 12, 286. https://doi.org/10.3390/pharmaceutics12030286
Adeleke OA, Hayeshi RK, Davids H. Development and Evaluation of a Reconstitutable Dry Suspension Containing Isoniazid for Flexible Pediatric Dosing. Pharmaceutics. 2020; 12(3):286. https://doi.org/10.3390/pharmaceutics12030286
Chicago/Turabian StyleAdeleke, Oluwatoyin A., Rose K. Hayeshi, and Hajierah Davids. 2020. "Development and Evaluation of a Reconstitutable Dry Suspension Containing Isoniazid for Flexible Pediatric Dosing" Pharmaceutics 12, no. 3: 286. https://doi.org/10.3390/pharmaceutics12030286
APA StyleAdeleke, O. A., Hayeshi, R. K., & Davids, H. (2020). Development and Evaluation of a Reconstitutable Dry Suspension Containing Isoniazid for Flexible Pediatric Dosing. Pharmaceutics, 12(3), 286. https://doi.org/10.3390/pharmaceutics12030286






