1. Introduction
Therapeutic peptides are today widely used due to their high specificity giving more predictive responses and fewer side effects than conventional synthetic small molecule drugs. Because of the physico-chemical characteristics of peptide-based therapeutics, such as their proteolytic instability, hydrophilic nature, and a large molecular size, the mode of administration is mainly via injections. However, non-injectable delivery would in many cases be the preferred route when taking patient compliance into consideration.
To increase the transepithelial permeation of therapeutic peptide drugs, cell-penetrating peptides (CPPs) has shown promising potential as carriers [
1,
2]. A widely used methodology is to co-administer the therapeutic peptide with the CPP as a physical mixture. Co-administration eases sample preparation and enables optimization of the drug-to-CPP molar ratio when compared to covalent conjugation of the drug to the CPP. Also, the co-administration approach possibly lowers the risk of negatively affecting the biological activity of the drug and the cell-penetrating efficiency of the CPP when compared to covalent conjugation [
3]. The co-administration approach may, however, result in the formation of a pool of poorly defined complexes differing in size and morphology [
4,
5]. On the other hand, complex formation may have beneficial effects such as serving as protection against enzymatic degradation [
6] or facilitate high local concentrations of CPP and the cargo drug at the site of absorption in the intestine.
So far, studies investigating the effects of complexation between carrier peptides and therapeutic peptides in order to obtain sufficient transepithelial permeation of the latter are limited. Previous studies have concluded that electrostatic interactions between a CPP and a co-administered therapeutic peptide or protein are important for CPP-mediated transepithelial delivery of the cargo [
7,
8,
9]. In contrast, another study demonstrated that the CPP penetratin [
10] mediated transepithelial insulin permeation mainly at pH-values which did not favor complexation between insulin and penetratin [
11]. With the present study, we will provide further knowledge to explain the effects of intermolecular interactions and sample pH when exploiting CPPs as carriers for transepithelial permeation of co-administered therapeutic peptides.
In the present study, insulin and the pharmacologically active part of parathyroid hormone (PTH(1-34)) [
12] were included as relevant therapeutic peptides, differing in isoelectric points and molecular weights. The cationic 16-mer penetratin sequence was employed as carrier. Penetratin has earlier shown potential for improving the transepithelial delivery of co-administered cargo peptides [
13]. pH 5, 6.5, and 7.4 were included as relevant pH-values mimicking the neutral and slightly acidic parts of the intestine [
14]. Using simple bulk-mixing, the complexes formed between insulin or PTH(1-34) and penetratin were examined with respect to size-distributions employing dynamic light scattering (DLS), association efficiency using ultracentrifugation, and folding propensity using circular dichroism (CD) spectroscopy. The effect of complexation on the potential of penetratin to interact with/disrupt lipid membranes was studied using fluorescence confocal microscopy. The chemical stability of insulin, PTH(1-34), and penetratin was assessed following apical incubation with the human colon adenocarcinoma Caco-2 cell culture model [
15]. Finally, the penetratin-mediated transepithelial permeation of insulin and PTH(1-34) was evaluated in vitro using the Caco-2 cell culture model and in vivo following intra-intestinal injection in rats.
2. Materials and Methods
Human recombinant insulin (5807.6 g/mol, Zn-free) was provided by Sanofi (Frankfurt, Germany). Synthetic PTH(1-34) (4117.8 g/mol, purity > 95%) and penetratin (RQIKIWFQNRRMKWKK) (2246.7 g/mol, purity > 95%) were purchased from Bachem (Torrance, CA, USA) and Synpeptide (Shanghai, China), respectively. Thioflavin T (ThT) (T3516), Nile Red (N3013), and all other chemicals were obtained from Merck (Buchs, Switzerland) unless otherwise stated.
2.1. Preparation of Insulin-Penetratin and PTH(1-34)-Penetratin Samples
Using low-binding Eppendorf tubes, insulin, PTH(1-34), or penetratin was dissolved in ultrapure water (18.2 MΩ × cm) obtained from a BarnsteadTM water purification system (Thermo Scientific, Wilmington, NC, USA). For insulin, volumes of 0.1 M HCl were added until completely dissolved before neutralization by the addition of equal volumes 0.1 M NaOH. The insulin, PTH(1-34), and penetratin solutions were passed through a 0.22 µm Millex Millipore filter (MB Millipore, Billerica, MA, USA) before the concentrations were determined using a Nanodrop 2000c (Thermo Scientific, Wilmington, NC, USA). The insulin, PTH(1-34), and penetratin stock solutions were further diluted to relevant test concentrations in Hanks’ Balanced Salt Solution (HBSS) (Gibco, Invitrogen, Nærum, Denmark) added 10 mM 2-(N-morpholino)ethanesulfonic acid (MES) (mHBSS) (AppliChem, Darmstadt, Germany), and adjusted to pH 5 or 6.5, or 10 mM 4-(2-hydroxyethyl)-1- piperazineethanesulfonic acid (HEPES) (hHBSS) (AppliChem) and adjusted to pH 7.4.
2.2. Cell Culture Model
Caco-2 cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA) and grown in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 2 mM L-glutamine, 0.1 mM non-essential amino acids, 90 U/mL penicillin, 90 µg/mL streptomycin and 10% (v/v) fetal bovine serum (FBS) (Thermo Fischer Scientific, Slangerup, Denmark) at 37 °C and 5% CO2 in T175 cell culture flasks (Corning Costar, Costar, NY, USA). At 80% confluency the cells were sub-cultured by detaching them from the culture flasks by trypsin-EDTA treatment. For the permeation study 1 × 105 cells were grown on polycarbonate filters with a diameter of 1.12 cm2 and a pore size of 0.4 µm placed in a 12-well Transwell® plate (Corning Costar, Costar, NY, USA) for 20 days prior experimental use.
2.3. Size Distribution
Size distributions according to the distribution by volume were determined using a Zetasizer Nano ZSP (Malvern Instruments, Worcestershire, UK) equipped with a 633 nm laser at 25 °C with 10–20 runs of 10 s and N = 3. The size measurements were performed on 180 µM insulin or PTH(1-34) and 720 µM penetratin in mHBSS adjusted to pH 5 or 6.5 or in hHBSS adjusted to pH 7.4.
2.4. Association Efficiency
Samples containing 180 µM insulin or PTH(1-34) and 720 µM penetratin were subjected to ultracentrifugation (25,000× g, 30 min, 20 °C) using a 5417 R centrifuge equipped with a FA-45-24-11 rotor (Eppendorf, Hamburg, Germany). Insulin, PTH(1-34), and penetratin left in the supernatant was quantified by reverse phase high-pressure liquid chromatography (HPLC) using a Prominence HPLC system (Shimadzu, Kyoto, Japan) equipped with an Aeris PEPTIDE XB-C18 column (3.6 µm core shell particles; Torrance, CA, USA). A gradient elution from 20–70% mobile phase B (acetonitrile/ultrapure water 95:5 and 0.1% trifluoroacetic acid (TFA) (v/v)) in mobile phase A (ultrapure water/acetonitrile 95:5 and 0.1% TFA (v/v)) over 10 min was applied using a flow rate of 0.8 mL/min and an injection volume of 10 µL. UV-detection was performed at 218 nm.
2.5. Secondary Structure
CD spectra were recorded in the range of 180–260 nm with 0.5 nm steps on a Chirascan CD spectrometer (Applied Photophysics, Leatherhead, UK) using a 1 mm cuvette (Helma Analytics, Müllheim, Germany). Measurements were performed at 37 °C in 10 mM phosphate buffer (pH 5, 6.5, or 7.4) with 50 µM insulin or PTH(1-34) and 200 µM penetratin. All spectra represent an average of 3 scans which have been normalized to baseline and transformed into mean residue ellipticity (MRE):
where
MRW is the mean residue weigh,
Ө is the ellipticity,
l is the path length, and
c is the sample concentration.
2.6. Liposomes
For fluorescence confocal microscopy, anionic POPC:POPG (80:20 molar ratio) liposomes were produced by the thin film method as described by Foged et al. [
16]. Briefly, dry lipid films were formed in round bottom flasks by applying vacuum over night before hydration in buffer (10 mM HEPES, 150 mM KCl, pH 7.4) with agitation every 10 min for 1 h before annealing for 1 h.
2.7. Visualization of Supramolecular Structures
Samples containing 50 µM insulin or PTH(1-34) and 200 µM penetratin were added 100 µM ThT and 100 µM Nile Red. 50 µL were placed into microscope slides and imaged at 1024 × 1024 pixel resolution using a Leica TCS SP5 confocal laser scanning microscope with a 63× oil objective NA = 1.4 (Leica Microsystems, Wetzlar, Germany) and a scanning frequency of 200 Hz. ThT fluorescence was measured under 458 nm excitation in the range 470–550 nm (green channel) and Nile Red fluorescence was measured under 543 excitation in the range 600–700 nm (red channel). In the two color experiments, images were sequentially acquired in order to avoid bleed through artifacts.
2.8. Degree of ThT and Nile Red Binding to Supramolecular Complexes
The fluorescence in samples containing 50 µM insulin or PTH(1-34), 200 µM penetratin and 100 µM ThT (ex 450/em 485) or 100 µM Nile Red (ex 552/em 636) were recorded in black clear-bottom 96-well over 3 h using a CLARIOstar plate reader (BMG Labtech, Offenburg, Germany).
2.9. In Vitro Stability
The stability of insulin, PTH(1-34), and penetratin was assessed during apical incubation in Caco-2 cell monolayers as previously described by Trehín et al. [
17]. Briefly, The Caco-2 cell monolayers were washed twice apical and basolateral with 37 °C hHBSS and equilibrated to room temperature in the last washing volume. The transepithelial electrical resistance (TEER) was measured using an EVOM equipped with an Endohm-12 cup (World Precision Instruments, Sarasota, FL, USA) to validate the barrier integrity of the monolayer. Following equilibration to 37 °C the filter inserts were transferred to empty 6-well plates in order to block permeation across the monolayer. The buffer was removed from the apical compartment and the stability study was initiated by adding 800 µL of the prepared test samples. 50 µL samples were collected in HPLC vials prefilled with 5 µL 1% (
v/
v) formic acid in methanol at time-point 0, 30, 60, 90, 120, 150, 180, 210, and 240 min. The kinetics of the metabolic degradation was evaluated by analytical RP-HPLC as described under the ultracentrifugation procedure. Data for penetratin alone or in the presence of insulin or PTH(1-34) was analyzed according pseudo-first order kinetics and the half-lives (t
1/2) calculated:
where C is the peptide concentration and t is the incubation time.
2.10. In Vitro Transepithelial Permeation
Caco-2 cell monolayers were washed twice apical and basolateral with 37 °C hHBSS. The cells were equilibrated to room temperature in the last washing volume before TEER was measured using an EVOM equipped with an Endohm-12 cup (World Precision Instruments, Sarasota, FL, USA) and equilibration to 37 °C. The buffer was removed from the basolateral and apical compartments and 500 µL test samples containing 5 µM insulin or PTH(1-34) and 20 µM penetratin was added to the apical chamber. The permeation study was initiated by careful transfer of the filter inserts to fresh 12-well plates containing 1 mL 37 °C hHBSS. The experiment was performed on a 37 °C shaking table (50 rpm).
100 µL test samples were withdrawn from the basolateral compartment at time-points 0, 60, 120 and 180 min and stored at −20 °C. The sample volume was immediately replaced with 37 °C hHBSS. Insulin and PTH(1-34) were quantified using an insulin ELISA kit (Mercodia, Uppsala, Sweden) or a PTH(1-34) EIA kit (Bachem, Torrance, CA, USA) according to the manufacturer’s directions with absorbance measurement at 450 nm employing a FLUOstar OPTIMA plate reader (BMG Labtech, Offenburg, Germany).
The apparent permeability coefficient (P
app) was calculated:
where dQ/dt is the steady state flux, A (1.12 cm
2) is the area of the Caco-2 monolayer and C
0 is the initial donor concentration.
2.11. Epithelial Cell Viability
The 3-(4,5-dimethyl-thiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS)/phenazine methosulfate (PMS) assay (as previously described by Cory et al. [
18]) was employed to determine the cellular viability of the Caco-2 cells after each permeation study. Briefly, 320 µL MTS/PMS solution containing 240 µg/mL MTS and 2.4 µg/mL PMS (Promega, Madison, WI, USA) was added the cell monolayers and incubated for 1.5 h with orbital shaking (50 rpm) at 37 °C. Two 100 µL samples were withdrawn from each well and the absorbance was measured at 492 nm on a FLUOstar OPTIMA plate reader (BMG Labtech, Offenburg, Germany).
The relative viability was calculated by using the equation:
where A
sample is the absorbance of the test sample, A
SDS is the absorbance of the positive control (0.2% (
w/v) sodium dodecyl sulphate (SDS)) corresponding to 0% cell viability, and A
buffer is the absorbance of the negative control corresponding to 100% cell viability.
2.12. In Situ Rat Intestinal Injection
Male Sprague Dawley rats (Taconic, Ejby, Denmark) weighing ~250 g were allowed to acclimatize for at least 8 days at 20–22 °C and 45–65% humidity with free access to water and food. The rats were fasted for 18 h prior to surgery. Hypnorm (fentanyl 0.315 mg/mL and fluanisone 10 mg/mL) (Glostrup apotek, Glostrup, Denmark) and Dormicum (midazolam 5 mg/mL) (Nomeco, Copenhagen, Denmark) were mixed 1:1 (
v/
v) with ultrapure water and used for anesthesia. The initial dose was 2 mL/kg and subsequent doses of 1 mL/kg were given every 20 min within the first hour and thereafter every 40 min to keep the animals anaesthetized. All doses of anesthetics were administered subcutaneously in the neck region. The abdominal wall was cut open and the small intestine localized. A 10 cm PE-50 catheter (Smiths Medical, Keene, NH, USA) was inserted into the middle of the intestine and secured with a 3/0 suture (Braun, Tuttlingen, Germany). Care was taken not to interrupt the mesenteric blood flow. The intestine was hydrated with 1–2 mL isotonic sodium chloride solution before the rat abdominal wall was closed with 9 mm clips using the AutoClip Wound Closure System (Alzet, Cupertino, CA, USA). The animal was left on a 37 °C heating plate (Scandidact, Kvistgaard, Denmark) for 30 min prior dosing of the test samples to allow normalization of the potentially elevated blood glucose level resulting from stress induced by handling and surgery. 500 µL test samples containing 180 µM insulin (50 IU/kg) or PTH(1-34) and 720 µM penetratin were injected into the catheter. A volume of 200 µL air was injected immediately after dosing to fully empty the catheter into the intestinal lumen. 200 µL blood samples were collected from the tail vein at time points 0, 5, 15, 30, 45, 60, 90, and 120 min into EDTA-coated microcuvette vials (Sarstedt, Nümbrecht, Germany). The blood samples were placed on ice prior isolation of the plasma by centrifugation at 2000×
g at 4 °C for 10 min (Eppendorf centrifuge 5424, Eppendorf, Hamburg, Germany). Blood glucose was measured at time points of 15, 30, 45, 60, 90, and 120 min after dosing using an On-Call blood glucose meter (Acon, San Diego, CA, USA). After the final blood sampling, the animals were euthanized while still under anesthesia using 90% CO
2 in O
2 [
19]. The abdominal wall of the rats was reopened after the experiment to ensure the catheter was still in place. Likewise, control of potential damage to the intestine was determined macroscopically. The experiments were conducted in strict compliance with the national animal research license 2009/561-1622.
2.13. Data and Statistical Analysis
Data processing was carried out using Microsoft Office Excel 2010 and GraphPad Prism version 6 (GraphPad Software, San Diego, CA, USA). Statistical analysis was done in GraphPad Prism using one-way analysis of variance (ANOVA) with Tukey’s multiple comparisons test. Data are presented as mean ± standard deviation (SD) or standard error of mean (SEM) with N representing the number of experimental replicates and n representing number of biological replicates.
4. Discussion
In the present study the effect of sample pH was applied in the study of complexes formed between the therapeutic peptides insulin or PTH(1-34) and penetratin applied as a carrier peptide to enhance intestinal delivery. Focus was on studying the properties of the self-associated complexes, structure, membrane interaction, and stability to gain knowledge on mode of action and parameters important for successful penetratin-mediated transepithelial delivery of co-administered insulin and PTH(1-34).
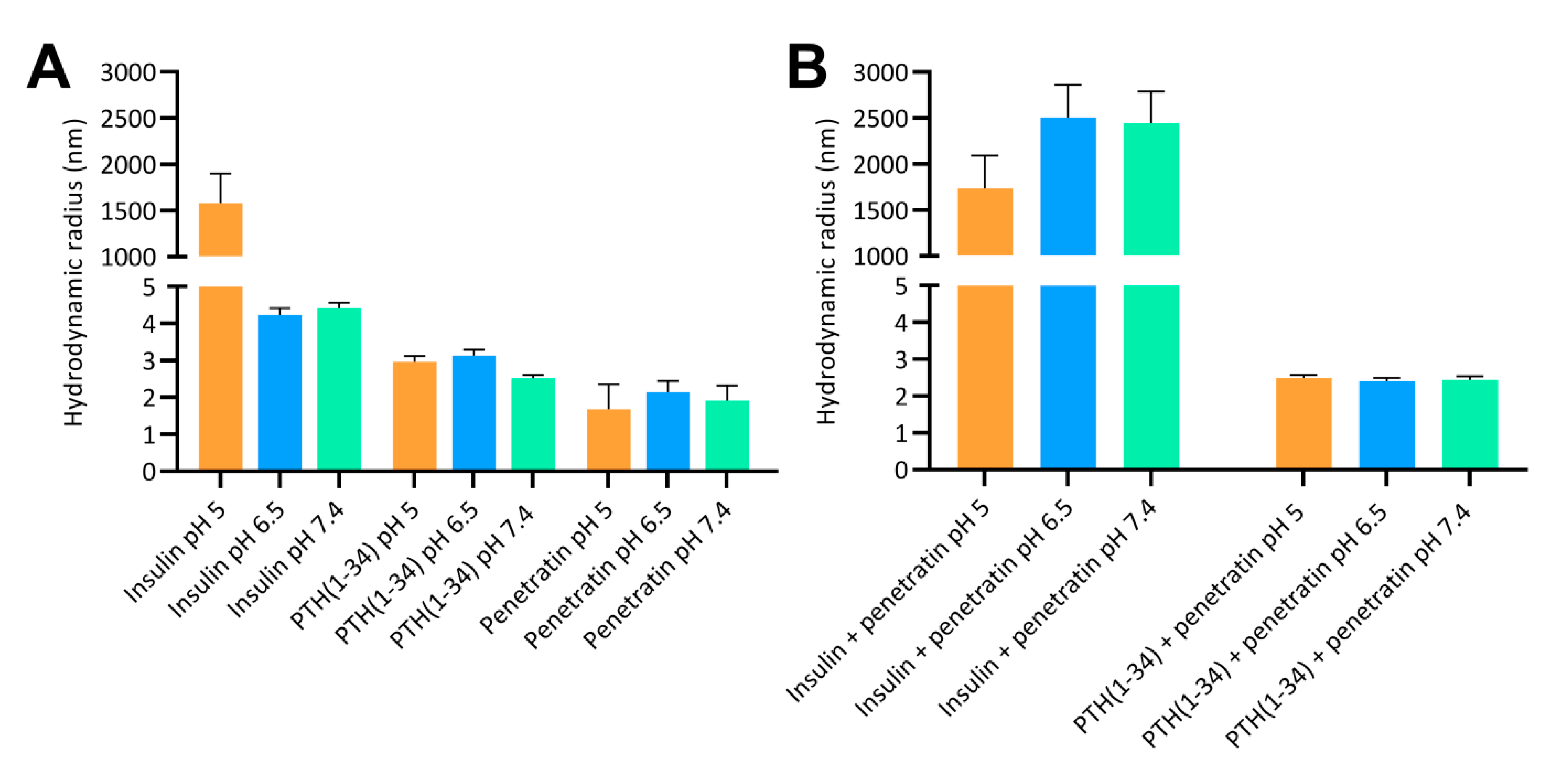
Insulin self-association was observed at pH 5 using DLS and confocal- microscopy; likely due to the fact that pH 5 is close to the isoelectric point (5.3) of monomeric insulin [
27]. Neither insulin at pH 6.5 or 7.4 nor PTH(1-34) appeared to self-associate to the same extent. Some penetratin self-association was observed at all pH-values; similar to previously reported for penetratin [
28], Tat, nona-arginine [
29] and other membrane translocating peptides [
30]. Supramolecular µm-sized insulin-penetratin complexes dominated the samples at pH 6.5 and 7.4, which is in agreement with earlier studies, documenting supramolecular complexes as a result of mixing insulin with penetratin or analogues thereof [
11] or with octaa-arginine [
31]. The size distribution for the pH 5 insulin-penetratin sample did not differ from the pH 5 insulin sample, thereby indicating that the insulin-penetratin complexation is more pronounced at pH 6.5 and 7.4 as compared to pH 5. The pI of insulin and PTH(1-34) is 5.3 and 8.3, respectively, and the pI of penetratin is 12.3 leaving it positively charged at pH 5, 6.5, and 7.4. Thus, electrostatic interactions appear to be main determinants for the observed complex formation within the insulin-penetratin samples. The association efficiency between insulin or PTH(1-34) and penetratin was indirectly quantitated by subjecting the samples to ultracentrifugation. Insulin sedimentation was observed in samples of pH 6.5 and 7.4; thereby supporting the earlier observation of larger complexes resulting from mixing insulin with penetratin. In line with this result, an earlier study investigated the binding strength between insulin and penetratin at pH 5, 6, and 7, demonstrating limited interaction between insulin and penetratin at pH 5, whereas the strongest interactions were observed at pH 6 and 7 [
32]. Almost no sedimentation of penetratin was observed in the ultracentrifugated pH 6.5 and 7.4 insulin-penetratin samples; thereby pointing to that insulin may be the dominant molecule present in the insulin-penetratin complexes. Though penetratin appears to self-associate, no sedimentation was observed. Similarly, neither PTH(1-34) nor penetratin were lost to sedimentation following ultracentrifugation alone or in mixture; despite some complexation was observed using DLS and confocal microscopy. We ascribe this discrepancy to that the observed complexes in those samples may have been only loosely associated complexes that de-assembled during the centrifugation step or were of such small size and density that they could not be efficiently spun down.
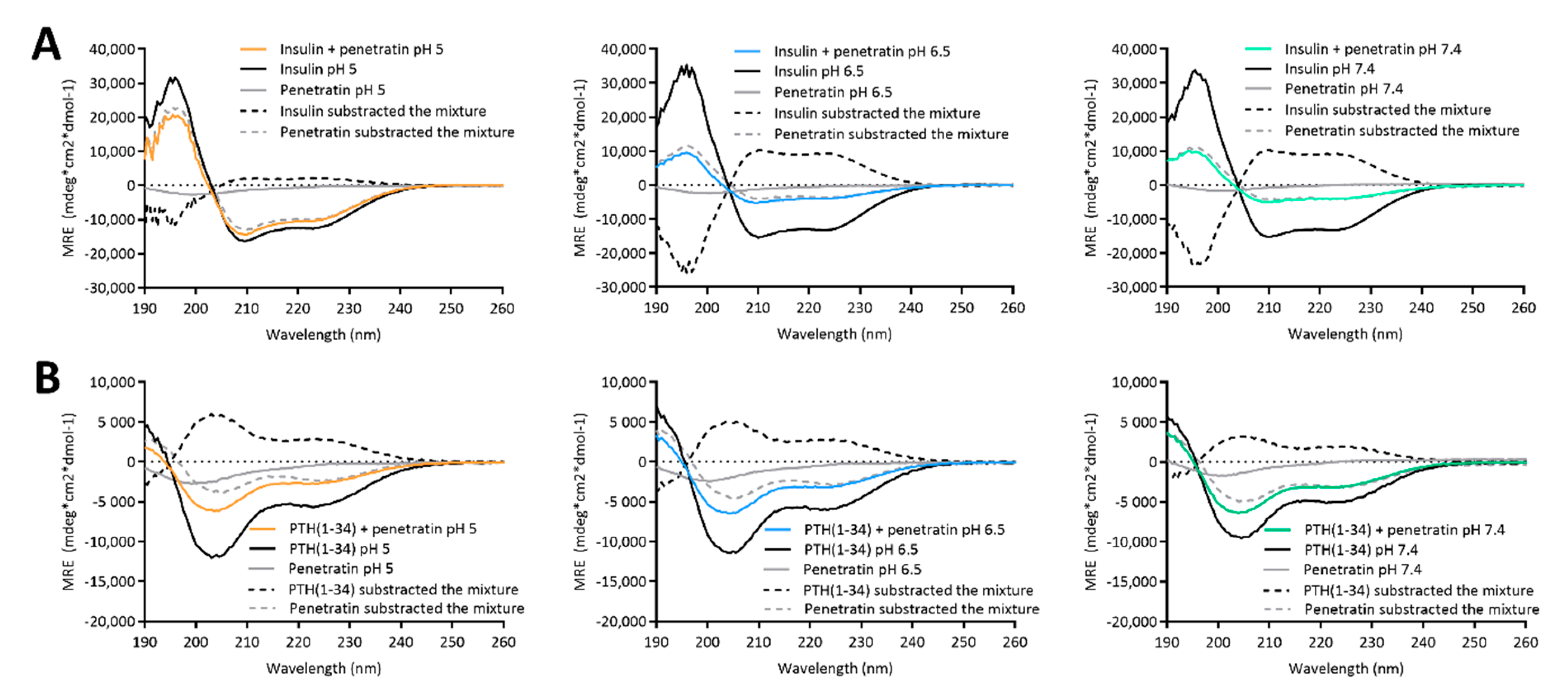
Effects on the secondary structure content was studied in the physical mixtures containing insulin or PTH(1-34) and penetratin using CD spectroscopy. The secondary structure in the insulin-penetratin sample was highly pH-dependent as evident by the minor amount of α-helical content in the pH 6.5 and 7.4 samples as compared to the pH 5 sample. Subtracting the penetratin spectra from the insulin-penetratin samples did not affect the resulting profile, thus implying little impact from penetratin on the overall secondary structure content in the mixtures. The more pronounced interaction between insulin and penetratin in the pH 6.5 and 7.4 samples may therefore affect the ability of insulin to adapt an α-helical conformation.
No pH-dependence was observed in the resulting secondary structure within the PTH(1-34)-penetratin samples, which is well in line with the lack of efficient association between PTH(1-34) and penetratin at the pH-values included in the present study. Thus, the ability to form large complexes with strong intermolecular interactions appears to be associated with some structural changes at the level of secondary structure as illustrated for the pH 6.5 and 7.4 insulin-penetratin samples.
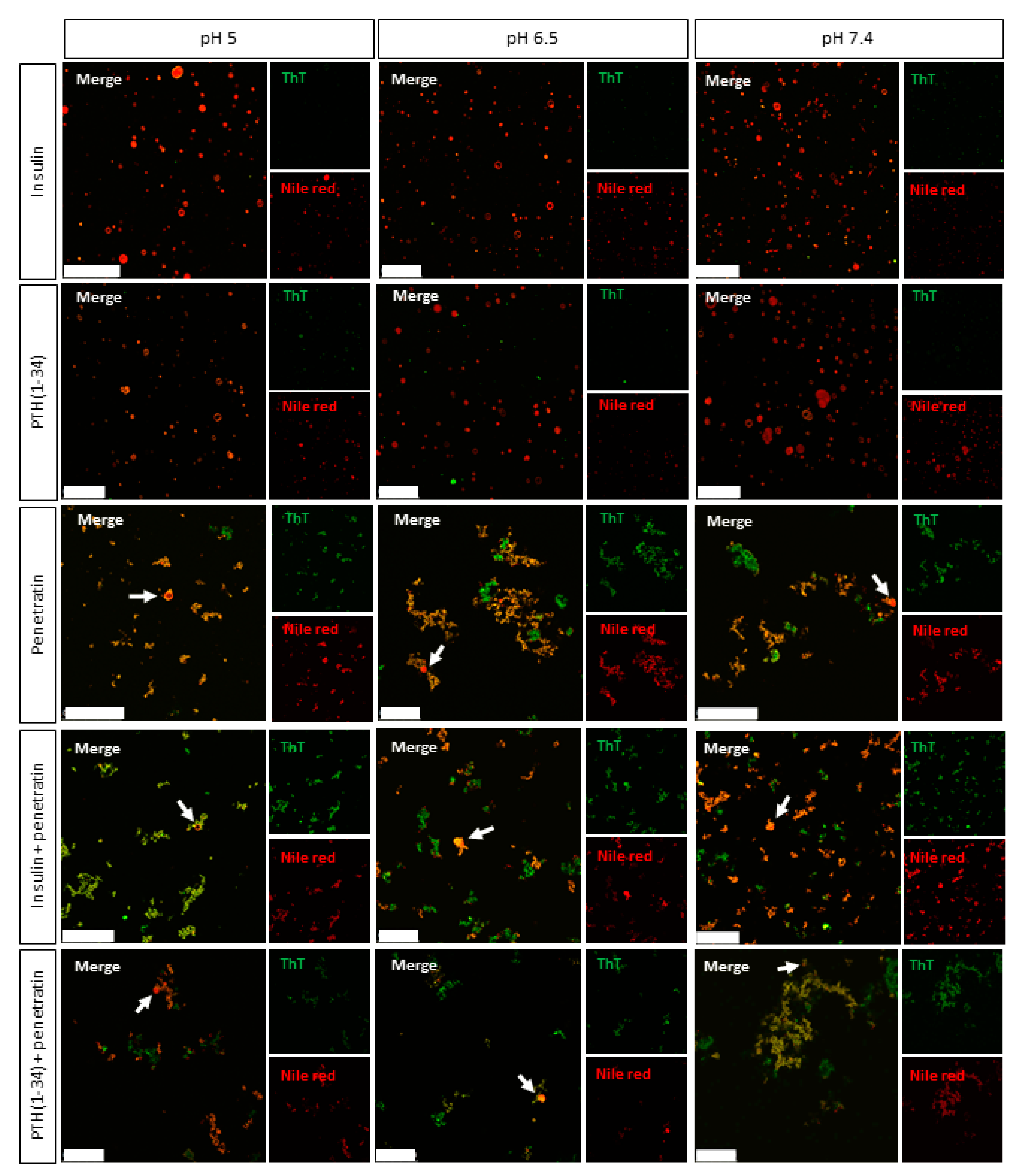
The effect of mixing penetratin with insulin or PTH(1-34) with respect to interaction with lipid membranes was studied using fluorescence confocal microscopy. ThT and Nile Red were included as hydrophobic domain dyes to allow detection of the complexes and the liposomal structures. Neither Nile Red- nor ThT-staining was observed in the pH 6.5 or 7.4 insulin samples or in any of the PTH(1-34) samples, which in line with the DLS measurements, indicates absence of larger complexes. It should be noted, though, that confocal microscopy and DLS should be considered complementary acknowledging that they are applied to investigate different assembly size scales. All penetratin samples as well as samples containing mixtures of penetratin and insulin or PTH(1-34) stained positive for both dyes. The morphology of the observed aggregates and complexes appeared similar to the earlier observed so-called crinkled aggregates of concanavalin A staining positive for ThT [
33]. At the applied peptide to lipid ratios, the addition of liposomes revealed penetratin-mediated rupture of the majority of the liposomal structures both without and with the presence of insulin or PTH(1-34), and without any obvious pH-dependence. Thus, penetratin appears to exert its effect on lipid membranes even as self-associated or in strong association with its therapeutic cargo, as observed for the pH 6.5 and 7.4 insulin-penetratin samples. In addition to the disappearance of the liposomal structures, some fusion and/or aggregation of the liposomal structures was detected (
Figure S8); similar to recently observed as a result of the application of nona-arginine to giant unilamellar vesicles (GUVs) [
34] or various cationic CPPs to POPC:POPG liposomes [
28]. Liposomes do, however, not reflect the complexity of the intestinal epithelium, which likely explain the lack of observed penetratin-mediated transepithelial transport at pH 6.5 and 7.4 in vitro and limited effect in vivo.
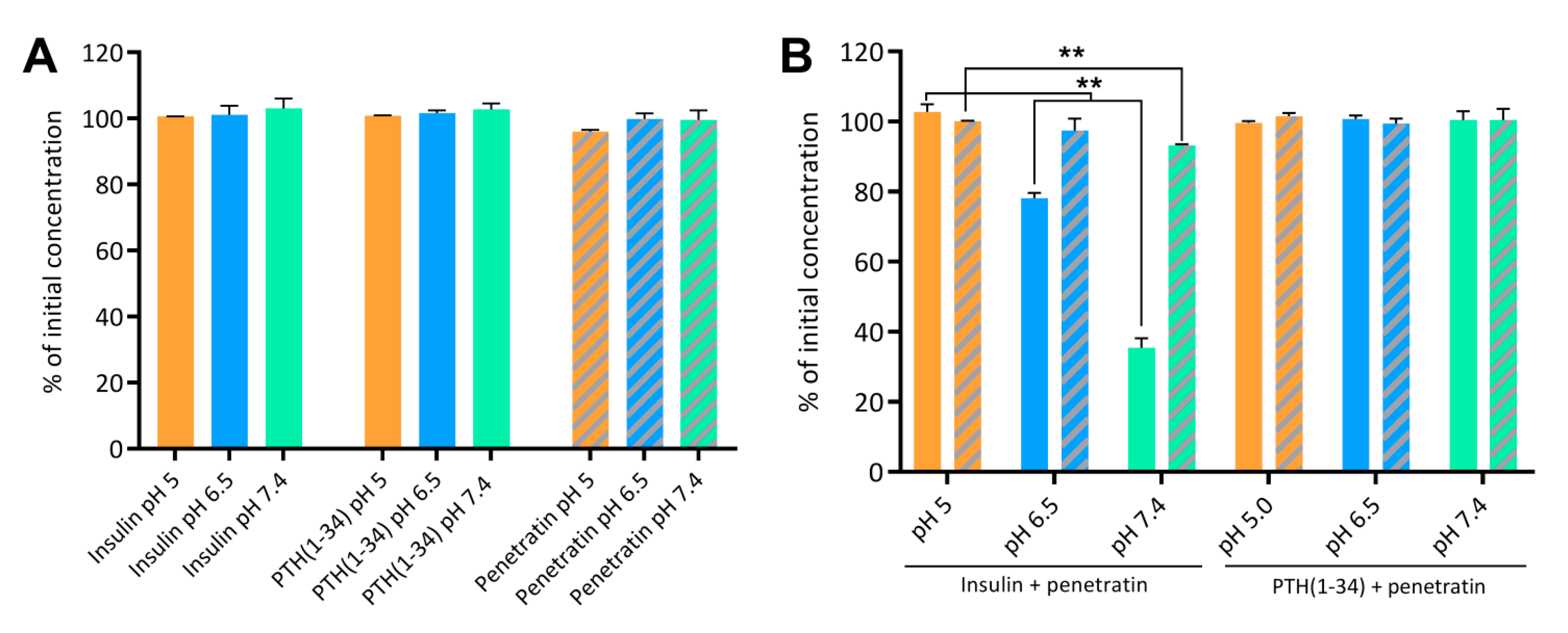
Peptides and proteins are inherently prone to degradation when presented to the metabolic barrier of an epithelium [
35,
36]. Therefore, the enzymatic stability of penetratin is of great importance for its ability to mediate transepithelial permeation of co-administered cargo drugs such as insulin and PTH(1-34). The co-administration approach may positively influence the stability of penetratin when conditions favor complex formation, thereby retaining the cell-penetrating propensity of penetratin for a longer time-period. On the other hand, degradation of penetratin may have beneficial effects in terms of releasing the therapeutic cargo as well as the physiological clearance of penetratin. In the present study, the penetratin, insulin, and PTH(1-34) resistance towards enzymatic degradation was assessed during apical incubation on polarized Caco-2 cell monolayers. Penetratin was less stable when compared to insulin and PTH(1-34); and that in a pH-dependent manner, with longer half-lives obtained at pH 5 than at pH 6.5 and 7.4. In addition, the complexation with insulin at pH 7.4 appears to positively influence the penetratin stability; which was, however, not the case at pH 6.5. Thus, the better association efficiency observed in the pH 7.4 insulin-penetratin sample, likely adds to the longer half-life observed at pH 7.4 but not at pH 6.5, when compared to applying penetratin alone or together with PTH(1-34) at pH 6.5 or 7.4.
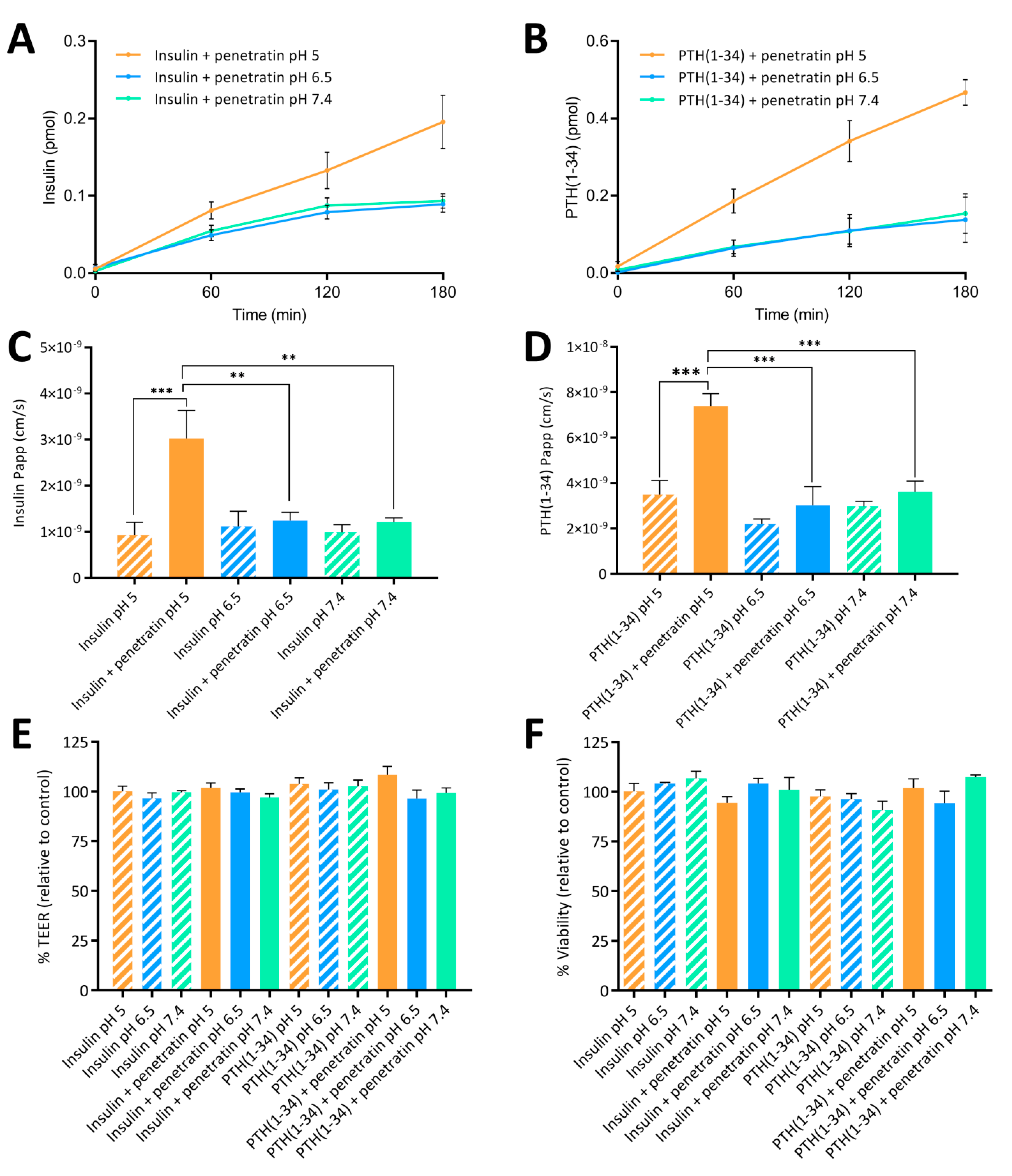
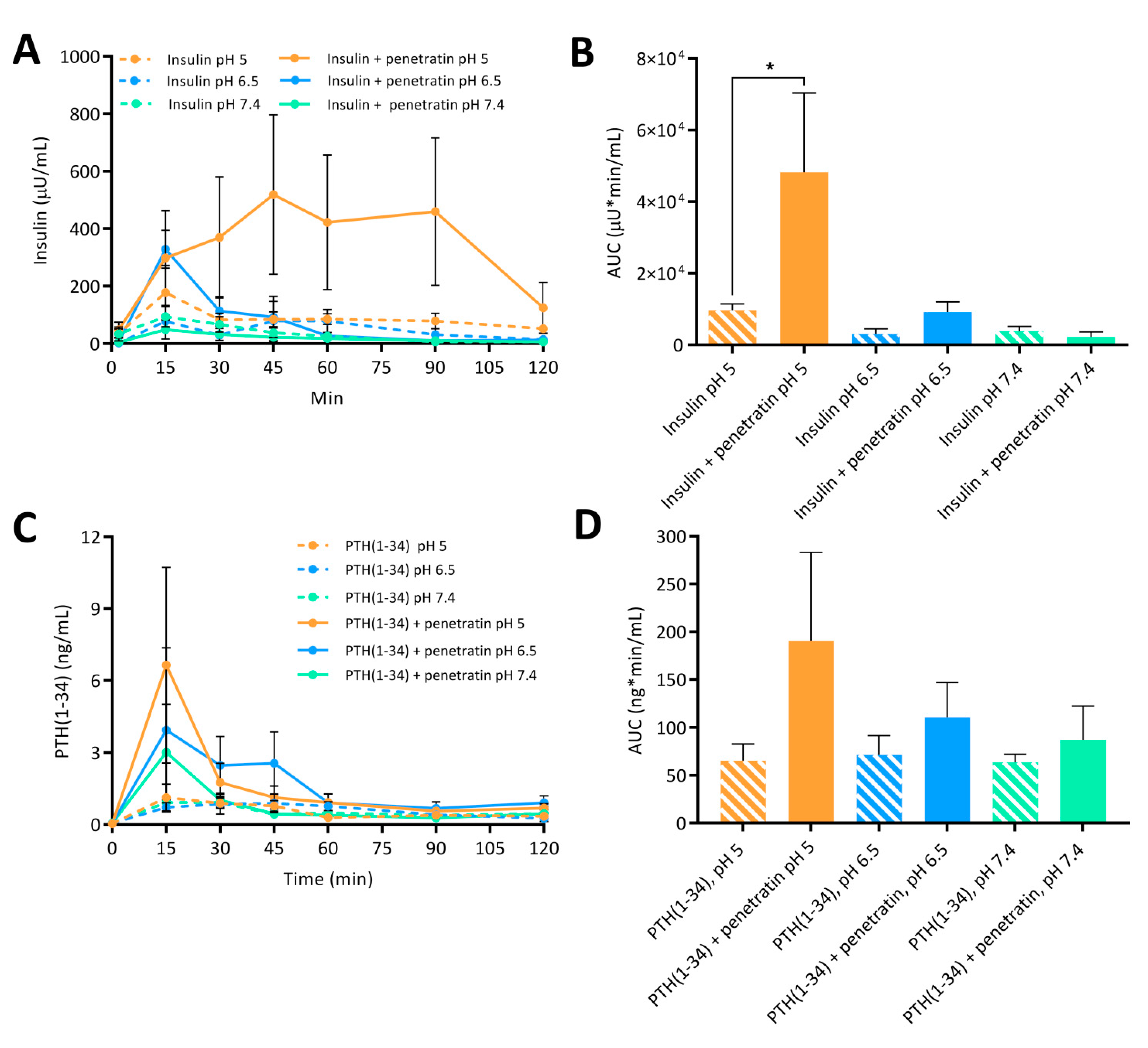
Finally, the ability of penetratin to facilitate transepithelial insulin and PTH(1-34) permeation was assessed in vitro using polarized Caco-2 cell monolayers and in vivo following intestinal injection in rats. A pH-dependent penetratin-mediated transepithelial permeation of insulin and PTH(1-34) was observed both in vitro and in vivo. In addition, the increase in the penetratin-mediated insulin delivery observed at pH 5 when compared to pH 6.5 and 7.4 was associated with a decrease in the blood glucose level. Thus, the formation of complexes observed in the insulin-penetratin samples at pH 6.5 and 7.4, which displayed stronger intermolecular interactions than at pH 5, did not have a positive impact on the transepithelial insulin permeation. In contrast, mixing insulin with penetratin at pH 6.5 and 7.4 resulted in visible precipitates in the concentrations applied in vivo (
Figure S9). Visible precipitation was not evident for PTH(1-34) when mixed with penetratin, which is in line with the results from DLS and the ultracentrifugation experiments. Despite that, some effect of co-administering insulin with penetratin was observed at pH 6.5 with respect to plasma insulin and an associated decrease in blood glucose level. This observation corresponds well with an earlier study, demonstrating dissociation of insulin-penetratin aggregates following addition to isolated rat intestinal fluid [
13]. Poor association between insulin and penetratin was observed at pH 5, thereby indicating that the ability of penetratin as a carrier for transmucosal insulin delivery is impaired by high association efficiencies resulting from physical mixing of molecules with distinctly opposing net charges. However, completely avoiding the formation of complexes appears not to be a general solution to increase the penetratin-mediated transepithelial permeation of a co-administered cargo. No or little penetratin-mediated PTH(1-34) permeation was observed at pH 6.5 and 7.4 in vitro and in vivo, respectively; and that despite poor association efficiency between PTH(1-34) and penetratin. Importantly, the increased penetratin stability obtained when lowering the sample pH from 7.4 or 6.5 to pH 5, may be the determining factor for the significant increase in insulin and PTH(1-34) oral delivery in vitro and in vivo.
5. Conclusions
The present work paves the way for further understanding the effect of formulation pH and complex formation when simply bulk-mixing insulin or PTH(1-34) with penetratin in order to obtain better transepithelial permeation of the cargo drug. The intermolecular association efficiency within complexes resulting from mixing insulin or PTH(1-34) with penetratin was pronounced at pH-values favoring electrostatic interactions, i.e., within the insulin-penetratin samples at pH 6.5 and 7.4. The formation of complexes in the insulin-penetratin samples observed at pH 6.5 and 7.4 resulted in less α-helical structures when compared to the pH 5 sample but did not affect the potential of penetratin to disrupt liposomal structures. The ability of penetratin to facilitate transepithelial permeation of insulin and PTH(1-34) in vitro and in vivo was, however, clearly favored at pH 5 when compared to pH 6.5 and 7.4. The penetratin stability was moreover positively affected by lowering the sample pH from 7.4 or 6.5 to pH 5. Thus, the better penetratin stability observed at pH 5 when compared to pH 6.5 and 7.4, rather than strong intermolecular interactions, may be the critical parameter in order to obtain successful transepithelial penetratin-mediated insulin and PTH(1-34) permeation.
The outcome of the present study implies that co-administration with penetratin may be a viable strategy to obtain intestinal delivery of therapeutic peptides, but control of the sample pH appears to be of utmost importance for future successful use.