Preparation and Evaluation of Topically Applied Azithromycin Based on Sodium Hyaluronate in Treatment of Conjunctivitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Degradation Kinetics of AZI
2.2.2. Preparation of AZI-SH and AZI-HPMC Complex Powders
2.2.3. Differential Scanning Calorimetry (DSC)
2.2.4. X-Ray Diffraction Analysis
2.2.5. 1H-Nuclear Magnetic Resonance Spectroscopy (1H-NMR)
2.2.6. Rheological Study
2.2.7. Azithromycin Quantification by HPLC
2.2.8. Precorneal Pharmcokinetic Study
2.2.9. Safety Evaluation
Cytotoxicity Assay
In Vivo Eye Rrritation Assessment in Rabbits
Acute Toxicity Test
2.2.10. Statistical Analysis
3. Results
3.1. Degradation Kinetics of Azithromycin
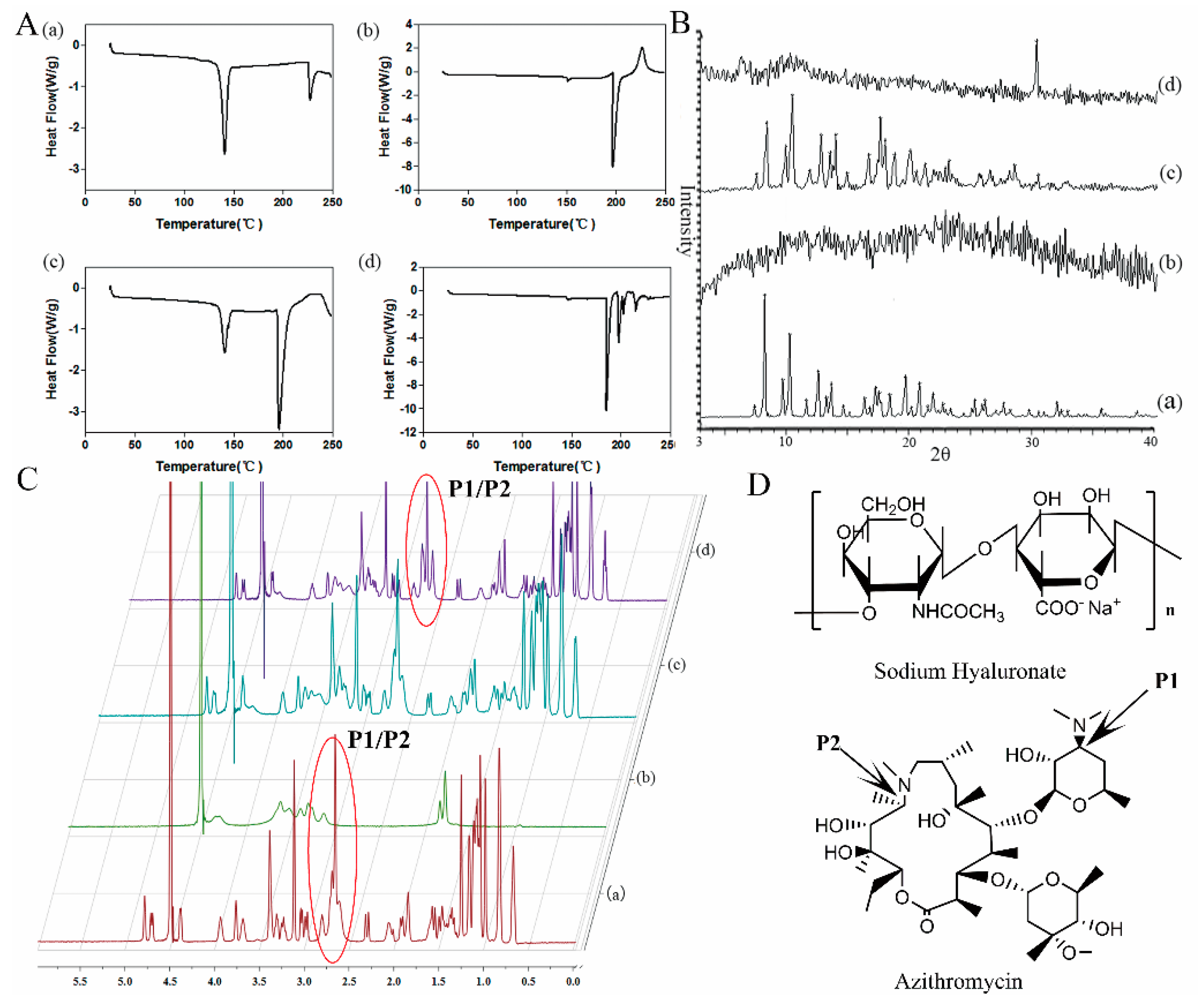
3.2. Differential Scanning Calorimetry (DSC)
3.3. XRD Analysis
3.4. 1H-NMR Analysis
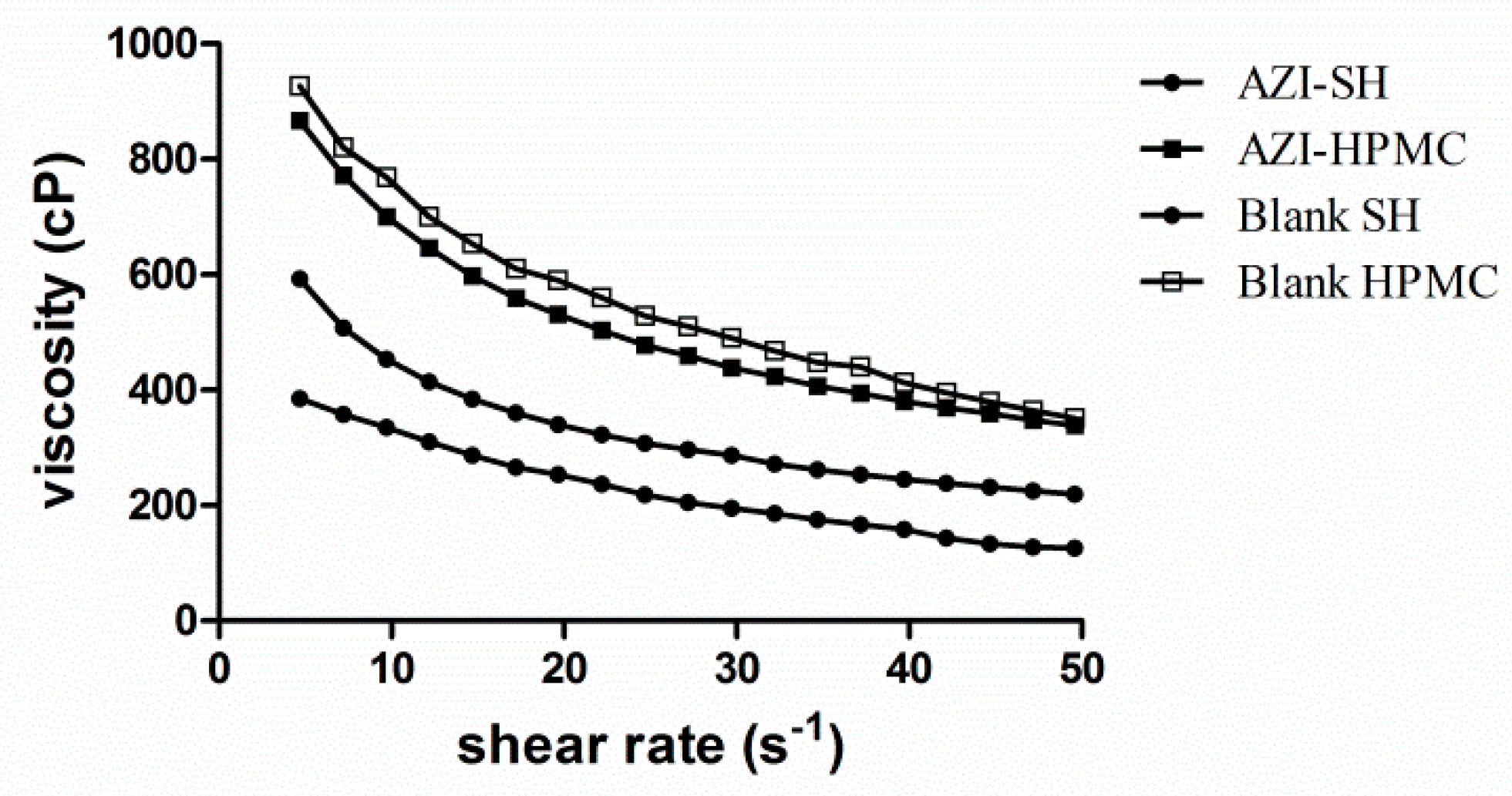
3.5. Rheological Study
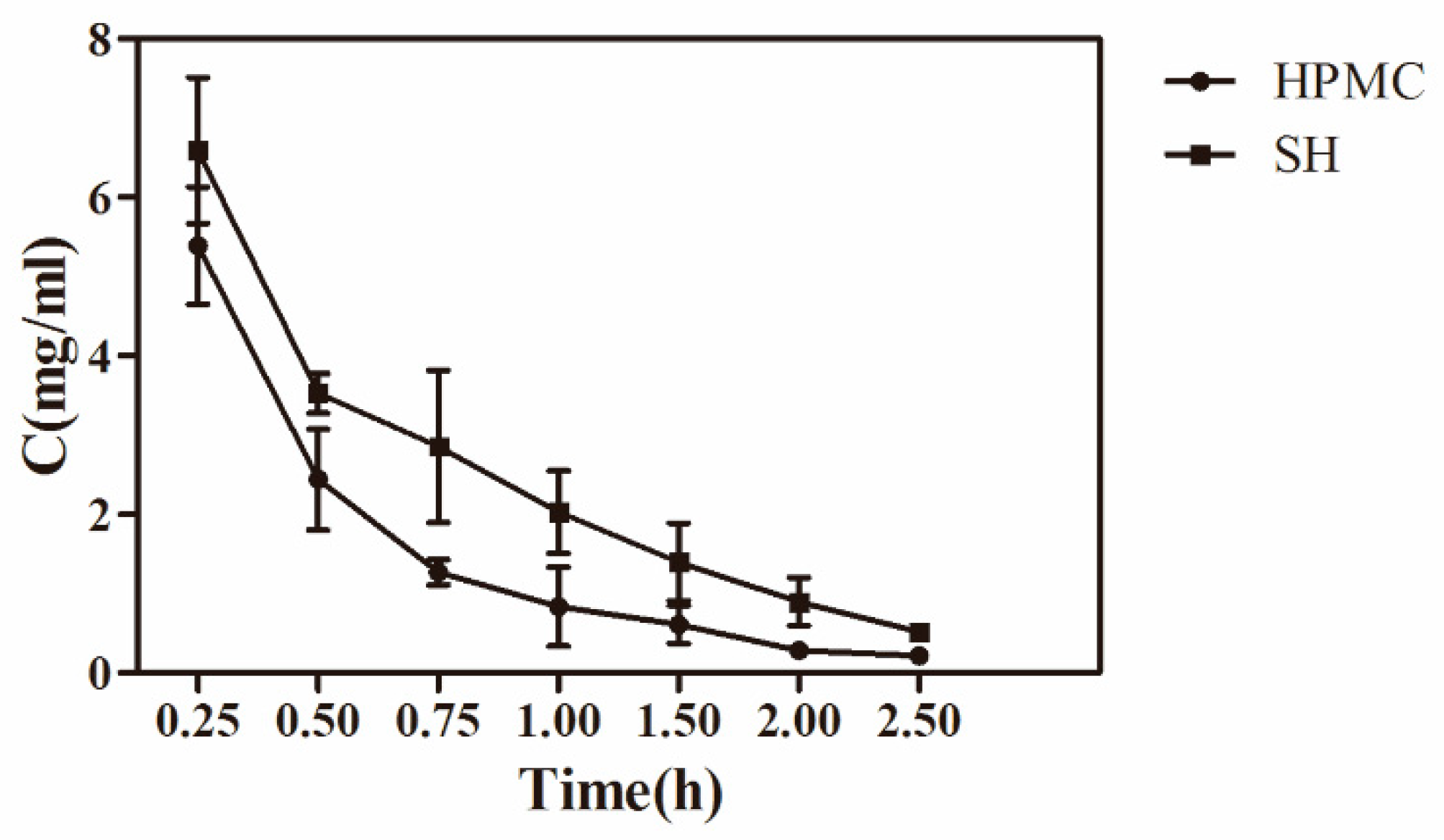
3.6. Precorneal Pharmacokinetic Study
3.7. Safety Evaluation
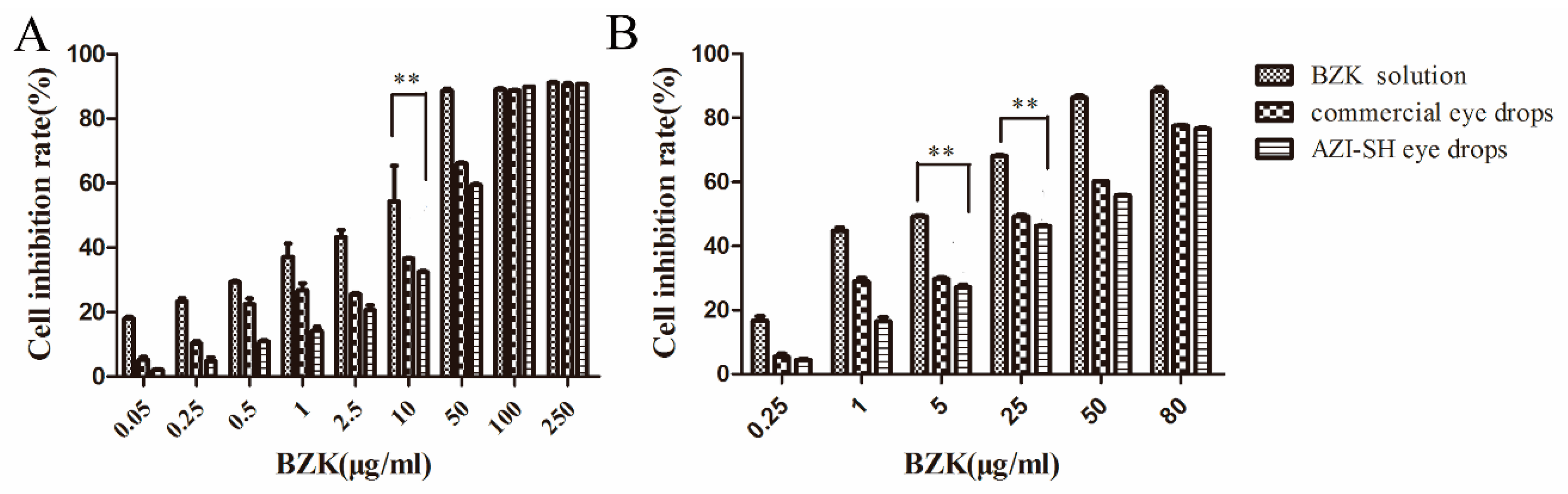
3.7.1. Cytotoxicity Assay
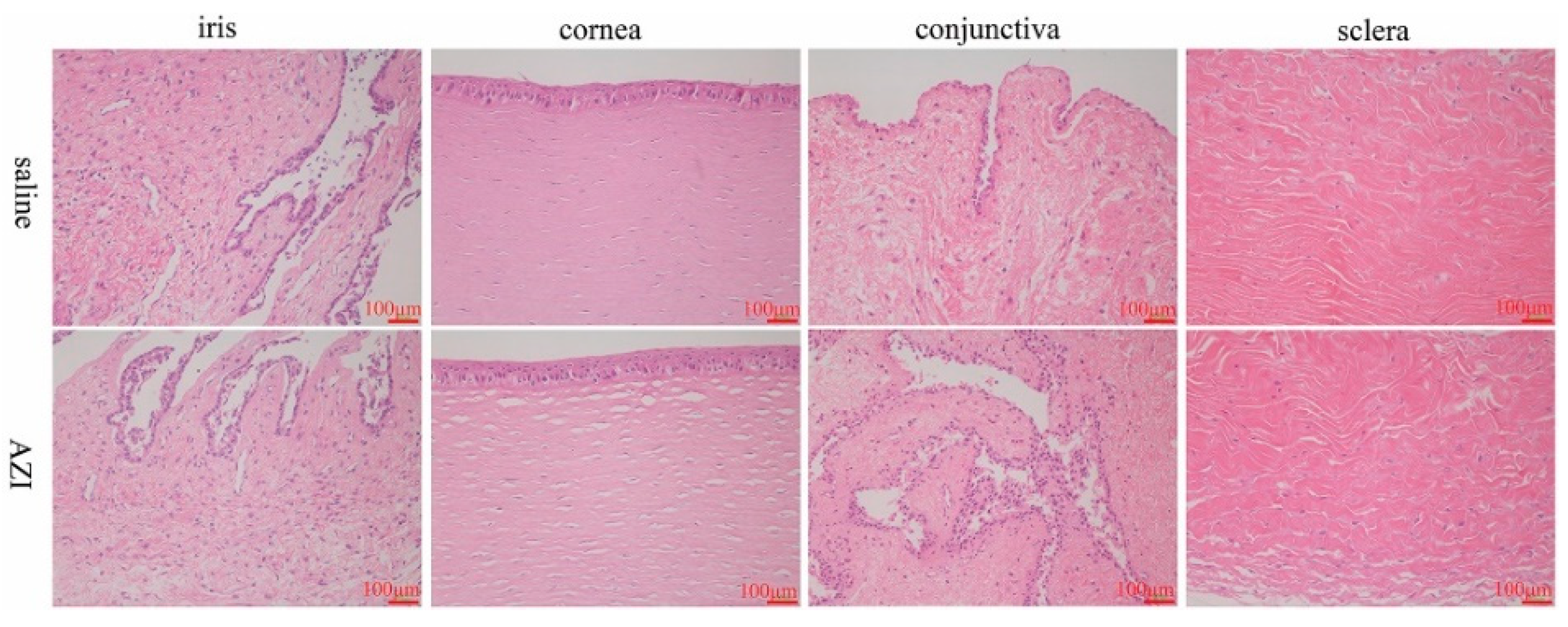
3.7.2. In Vivo Eye Irritation Assessment in Rabbits
3.7.3. Acute Toxicity Test in Rabbits
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Taylor, H.R. Trachoma—The future for a disease of the past. Br. J. Ophthalmol. 1993, 77, 66. [Google Scholar] [CrossRef][Green Version]
- Tung, N.T.; Tran, C.S.; Nguyen, T.L.; Hoang, T.; Trinh, T.D.; Nguyen, T.N. Formulation and biopharmaceutical evaluation of bitter taste masking microparticles containing azithromycin loaded in dispersible tablets. Eur. J. Pharm. Biopharm. 2017. [Google Scholar] [CrossRef]
- Liu, F.; Porco, T.C.; Mkocha, H.A.; Muñoz, B.; Ray, K.J.; Bailey, R.L.; Lietman, T.M.; West, S.K. The efficacy of oral azithromycin in clearing ocular chlamydia: Mathematical modeling from a community-randomized trachoma trial. Epidemics 2014, 6, 10–17. [Google Scholar] [CrossRef]
- Mohammadpour, M.; Abrishami, M.; Masoumi, A.; Hashemi, H. Trachoma: Past, present and future. J. Curr. Ophthalmol. 2016, 28, 165–169. [Google Scholar] [CrossRef]
- Schachter, J.; West, S.K.; Mabey, D.; Dawson, C.R.; Bobo, L.; Bailey, R.; Vitale, S.; Quinn, T.C.; Sheta, A.; Sallam, S. Azithromycin in control of trachoma. Lancet 1999, 354, 630–635. [Google Scholar] [CrossRef]
- Solomon, A.W. Trachoma Control: A Guide for Programme Managers; World Health Organization: Geneva, Switzerland, 2006; Volume 21, p. 42. [Google Scholar]
- Urtti, A.; Pipkin, J.D.; Rork, G.; Sendo, T.; Finne, U.; Repta, A.J. Controlled drug delivery devices for experimental ocular studies with timolol 2. Ocular and systemic absorption in rabbits. Int. J. Pharm. 1990, 61, 241–249. [Google Scholar] [CrossRef]
- Leeming, D.J.P. Treatment of Ocular Infections with Topical Antibacterials. Clin. Pharmacokinet. 1999, 37, 351–360. [Google Scholar] [CrossRef]
- Bowman, L.M.; Si, E.; Pang, J.; Archibald, R.; Friedlaender, M. Development of a topical polymeric mucoadhesive ocular delivery system for azithromycin. J. Ocul. Pharmacol. Ther. 2009, 25, 133. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, X.; Tang, X. Lipid emulsions as a potential delivery system for ocular use of azithromycin. Drug Dev. Ind. Pharm. 2009, 35, 887. [Google Scholar] [CrossRef]
- Salzillo, R.; Schiraldi, C.; Corsuto, L.; D’Agostino, A.; Filosa, R.; De, R.M.; La, G.A. Optimization of hyaluronan-based eye drop formulations. Carbohydr. Polym. 2016, 153, 275–283. [Google Scholar] [CrossRef]
- Aragona, P.; Papa, V.; Micali, A.; Santocono, M.; Milazzo, G. Long term treatment with sodium hyaluronate-containing artificial tears reduces ocular surface damage in patients with dry eye. Br. J. Ophthalmol. 2002, 86, 181–184. [Google Scholar] [CrossRef]
- Jiménez, M.M.; Fresno, M.J.; Ramírez, A. Rheological study of binary gels with Carbopol Ultrez 10 and hyaluronic acid. Chem. Pharm. Bull. 2007, 55, 1157. [Google Scholar] [CrossRef] [PubMed]
- Rozier, A.; Mazuel, C.; Grove, J.; Plazonnet, B. Functionality testing of gellan gum, a polymeric excipient material for ophthalmic dosage forms. Int. J. Pharm. 1997, 153, 191–198. [Google Scholar] [CrossRef]
- Tatavarti, A.S.; Hoag, S.W. Microenvironmental pH modulation based release enhancement of a weakly basic drug from hydrophilic matrices. J. Pharm. Sci. 2006, 95, 1459–1468. [Google Scholar] [CrossRef]
- Winterton, L.C.; Lally, J.M.; Sentell, K.B.; Chapoy, L.L. The elution of poly (vinyl alcohol) from a contact lens: The realization of a time release moisturizing agent/artificial tear. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 80, 424. [Google Scholar] [CrossRef]
- Abelson, M.; Protzko, E.; Shapiro, A.; Garces-Soldana, A.; Bowman, L. A randomized trial assessing the clinical efficacy and microbial eradication of 1% azithromycin ophthalmic solution vs tobramycin in adult and pediatric subjects with bacterial conjunctivitis. Clin. Ophthalmol. 2007, 1, 177–182. [Google Scholar]
- Protzko, E.; Bowman, L.; Abelson, M.; Shapiro, A. Phase 3 safety comparisons for 1.0% azithromycin in polymeric mucoadhesive eye drops versus 0.3% tobramycin eye drops for bacterial conjunctivitis. Investig. Ophthalmol. Vis. Sci. 2007, 48, 3425. [Google Scholar] [CrossRef]
- Anirudhan, T.S.; Nair, S.S.; Nair, A.S. Fabrication of a bioadhesive transdermal device from chitosan and hyaluronic acid for the controlled release of lidocaine. Carbohydr. Polym. 2016, 152, 687–698. [Google Scholar] [CrossRef]
- Kalam, M.A. Development of chitosan nanoparticles coated with hyaluronic acid for topical ocular delivery of dexamethasone. Int. J. Biol. Macromol. 2016, 89, 127–136. [Google Scholar] [CrossRef]
- Vasi, A.M.; Popa, M.I.; Butnaru, M.; Dodi, G.; Verestiuc, L. Chemical functionalization of hyaluronic acid for drug delivery applications. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 38, 177–185. [Google Scholar] [CrossRef]
- Härfstrand, A.; Molander, N.; Stenevi, U.; Apple, D.; Schenholm, M.; Madsen, K. Evidence of hyaluronic acid and hyaluronic acid binding sites on human corneal endothelium. J. Cataract. Refract. Surg. 1992, 18, 265–269. [Google Scholar] [CrossRef]
- Liu, X.; Yu, F.F.; Zhong, Y.M.; Guo, X.X.; Mao, Z. Therapeutic Effects of Sodium Hyaluronate on Ocular Surface Damage Induced by Benzalkonium Chloride Preserved Anti-glaucoma Medications. Chin. Med. J. 2015, 128, 2444–2449. [Google Scholar] [CrossRef] [PubMed]
- Pauloin, T.; Dutot, M.; Warnet, J.M.; Rat, P. In vitro modulation of preservative toxicity: High molecular weight hyaluronan decreases apoptosis and oxidative stress induced by benzalkonium chloride. Eur. J. Pharm. Sci. 2008, 34, 263–273. [Google Scholar] [CrossRef]
- Langford, M.P.; Foreman, B.D.; Srur, L.; Ganley, J.P.; Redens, T.B. Bilateral acute pyogenic conjunctivitis with iritis induced by unilateral topical application of bacterial peptidoglycan muramyl dipeptide in adult rabbits. Exp. Eye Res. 2013, 116, 324. [Google Scholar] [CrossRef][Green Version]
- Schoenwald, R.D.; Vidvauns, S.; Wurster, D.E.; Barfknecht, C.F. Tear film stability of protein extracts from dry eye patients administered a sigma agonist. J. Ocul. Pharmacol. Ther. 1997, 13, 151–161. [Google Scholar] [CrossRef]
- Wilhelmus, K.R. The Draize eye test. Surv. Ophthalmol. 2001, 45, 493–515. [Google Scholar] [CrossRef]
- Kishore, A.S.; Surekha, P.; Murthy, P.B. Assessment of the dermal and ocular irritation potential of multi-walled carbon nanotubes by using in vitro and in vivo methods. Toxicol. Lett. 2009, 191, 268–274. [Google Scholar] [CrossRef]
- Li, Q.; Li, Z.; Zeng, W.; Ge, S.; Lu, H.; Wu, C.; Ge, L.; Liang, D.; Xu, Y. Proniosome-derived niosomes for tacrolimus topical ocular delivery: In vitro cornea permeation, ocular irritation, and in vivo anti-allograft rejection. Eur J. Pharm. Sci. 2014, 62, 115. [Google Scholar] [CrossRef] [PubMed]
- Diebold, Y.; Jarrín, M.; Sáez, V.; Carvalho, E.L.; Orea, M.; Calonge, M.; Seijo, B.; Alonso, M.J. Ocular drug delivery by liposome-chitosan nanoparticle complexes (LCS-NP). Biomaterials 2007, 28, 1553–1564. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Lin, X.; Liu, X.; Tian, B.; Tang, X. High azithromycin loading powders for inhalation and their in vivo evaluation in rats. Int. J. Pharm. 2010, 395, 205. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Li, P.; Mi, G.; Tu, J.; Sun, L.; Webster, T.J.; Shen, Y. Ion-paired pirenzepine-loaded micelles as an ophthalmic delivery system for the treatment of myopia. Nanomedicine 2017, 13, 2079–2089. [Google Scholar] [CrossRef] [PubMed]
- Kauss, T.; Gaubert, A.; Boyer, C.; Ba, B.B.; Manse, M.; Massip, S.; Léger, J.M.; Fawaz, F.; Lembege, M.; Boiron, J.M. Pharmaceutical development and optimization of azithromycin suppository for paediatric use. Int. J. Pharm. 2013, 441, 218–226. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khachatryan, G.; Khachatryan, K.; Grzyb, J.; Fiedorowicz, M. Formation and properties of hyaluronan/nano Ag and hyaluronan-lecithin/nano Ag films. Carbohydr. Polym. 2016, 151, 452–457. [Google Scholar] [CrossRef]
- Edsman, K.; Carlfors, J.; Harju, K. Rheological evaluation and ocular contact time of some carbomer gels for ophthalmic use. Int. J. Pharm. 1996, 137, 233–241. [Google Scholar] [CrossRef]
- Üstündağ-Okur, N.; Gökçe, E.H.; Bozbıyık, D.İ.; Eğrilmez, S.; Ozer, O.; Ertan, G. Preparation and in vitro-in vivo evaluation of ofloxacin loaded ophthalmic nano structured lipid carriers modified with chitosan oligosaccharide lactate for the treatment of bacterial keratitis. Eur. J. Pharm. Sci. 2014, 63, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Ooteghem, M.V. The evaluation of viscous ophthalmic vehicles by slit lamp fluorophotometry in humans. Int. J. Pharm. 1989, 54, 95–102. [Google Scholar] [CrossRef]
- Parrott, E.L. Pharmaceutical Technology: Fundamental Pharmaceutics; Burgess Pub. Co.: Minneapolis, MN, USA, 1970. [Google Scholar]
- Ozaki, T.; Oda, H. Non-Linear Time Series Model Identification by Akaike’s Information Criterion. IFAC Proc. Vol. 1977, 10, 83–91. [Google Scholar] [CrossRef]
- Bother, H.; Waaler, T. Rheological Characterization of Tear Substitutes. Drug Dev. Ind. Pharm. 2008, 16, 755–768. [Google Scholar] [CrossRef]
- Durrani, A.M.; Farr, S.J.; Kellaway, I.W. Influence of molecular weight and formulation pH on the precorneal clearance rate of hyaluronic acid in the rabbit eye. Int. J. Pharm. 1995, 118, 243–250. [Google Scholar] [CrossRef]
- Robinson, J.R.; Mlynek, G.M. Bioadhesive and phase-change polymers for ocular drug delivery. Adv. Drug Deliv. Rev. 1995, 16, 45–50. [Google Scholar] [CrossRef]
- Denizot, F.; Lang, R. Rapid colorimetric assay for cell growth and survival. Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Methods 1986, 89, 271–277. [Google Scholar] [CrossRef]
- Borenfreund, E.; Puerner, J.A. Toxicity determined in vitro by morphological alterations and neutral red absorption. Toxicol. Lett. 1985, 24, 119–124. [Google Scholar] [CrossRef]
- Ubels, J.L.; Clousing, D.P. In Vitro Alternatives to the Use of Animals in Ocular Toxicology Testing. Ocul. Surf. 2005, 3, 126–142. [Google Scholar] [CrossRef]
- Ayaki, M.; Iwasawa, A. Cell viability of four corneoconjunctival cell lines exposed to five preservatives and a surfactant used for infection control in eyedrops. Biocontrol Sci. 2011, 16, 117–121. [Google Scholar] [CrossRef][Green Version]
- Iwasawa, A.; Ayaki, M.; Niwano, Y. Cell viability score (CVS) as a good indicator of critical concentration of benzalkonium chloride for toxicity in cultured ocular surface cell lines. Regul. Toxicol. Pharmacol. 2013, 66, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Zhang, H.; Wang, C.; Wu, Y.; Xie, J.; Jin, X.; Yang, J.; Ye, J. Genoprotective effect of hyaluronic acid against benzalkonium chloride-induced DNA damage in human corneal epithelial cells. Mol. Vis. 2011, 17, 3364–3370. [Google Scholar] [CrossRef] [PubMed]
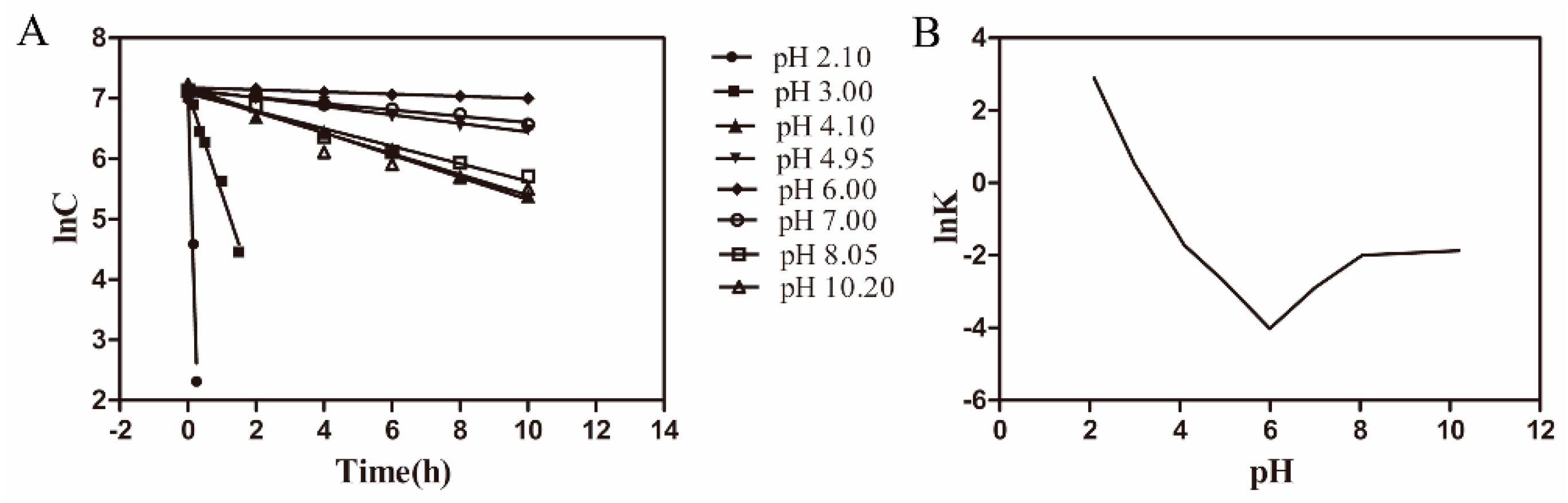
| pH | K (h−1) | lnK | R |
|---|---|---|---|
| 2.1 | 18.1000 | 2.90 | 0.9849 |
| 3.0 | 1.6747 | 0.52 | 0.9955 |
| 4.1 | 0.1805 | −1.71 | 0.9965 |
| 4.95 | 0.0689 | −2.68 | 0.9947 |
| 6.0 | 0.0179 | −4.02 | 0.9953 |
| 7.0 | 0.0557 | −2.89 | 0.9918 |
| 8.05 | 0.1354 | −2.00 | 0.9849 |
| 10.2 | 0.1534 | −1.87 | 0.9852 |
| Pharmacokinetics Parameters | HPMC Group | SH Group |
|---|---|---|
| A (μg/mL) | 12.39 | 31.35 |
| α (1/h) | 4.42 | 10.83 |
| B (μg/mL) | 1.58 | 5.34 |
| β (1/hr) | 0.81 | 0.90 |
| V(c) (mg)/(mg/mL) | 71.60 | 13.63 |
| T1/2α (h) | 0.16 | 0.06 |
| T1/2β (h) | 0.86 | 0.77 |
| K21 (1/h) | 1.22 | 2.3438 |
| K10 (1/h) | 2.94 | 4.15 |
| K12 (1/h) | 1.08 | 5.24 |
| AUC (mg/mL) × h | 4.76 | 8.85 |
| CL(s) μg/h/(mg/mL) | 210.18 | 56.52 |
| Pharmacokinetics Parameters | HPMC Group | SH Group |
|---|---|---|
| Area under the curve (AUC)(mg × h/mL) | 4.60 ± 0.51 | 7.30 ± 0.42 |
| Area under the moment curve (AUMC)(mg × h2/mL) | 3.16 ± 0.52 | 7.77 ± 0.58 |
| Mean residence time (MRT)(h) *** | 0.69 ± 0.03 | 1.07 ± 0.05 |
| Variance of the residence time (VRT)(h × h) | 1.07 ± 0.02 | 1.61 ± 0.02 |
| Groups | IC50 (μg/mL) | ED50 (μg/mL) |
|---|---|---|
| BZK solution | 5.09 ± 0.96 | 9.69 ± 5.66 |
| AZI-SH eye drops | 29.07 ± 1.87 | 40.57 ± 1.36 * |
| commercial AZI eye drops | 24.80 ± 2.06 | 32.79 ± 0.48 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Q.; Yin, C.; Ma, J.; Tu, J.; Shen, Y. Preparation and Evaluation of Topically Applied Azithromycin Based on Sodium Hyaluronate in Treatment of Conjunctivitis. Pharmaceutics 2019, 11, 183. https://doi.org/10.3390/pharmaceutics11040183
Chen Q, Yin C, Ma J, Tu J, Shen Y. Preparation and Evaluation of Topically Applied Azithromycin Based on Sodium Hyaluronate in Treatment of Conjunctivitis. Pharmaceutics. 2019; 11(4):183. https://doi.org/10.3390/pharmaceutics11040183
Chicago/Turabian StyleChen, Qian, Chun Yin, Jiang Ma, Jiasheng Tu, and Yan Shen. 2019. "Preparation and Evaluation of Topically Applied Azithromycin Based on Sodium Hyaluronate in Treatment of Conjunctivitis" Pharmaceutics 11, no. 4: 183. https://doi.org/10.3390/pharmaceutics11040183
APA StyleChen, Q., Yin, C., Ma, J., Tu, J., & Shen, Y. (2019). Preparation and Evaluation of Topically Applied Azithromycin Based on Sodium Hyaluronate in Treatment of Conjunctivitis. Pharmaceutics, 11(4), 183. https://doi.org/10.3390/pharmaceutics11040183





