Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Methods
2.2.1. Box–Behnken Design (BBD)
2.2.2. Sample Preparation
2.2.3. Rheological Measurements
Stress Amplitude Sweep
Frequency Sweep
Creep Test
2.2.4. Peeling Test
2.2.5. Statistical Analysis
3. Results and Discussion
3.1. BBD Statistical Analysis
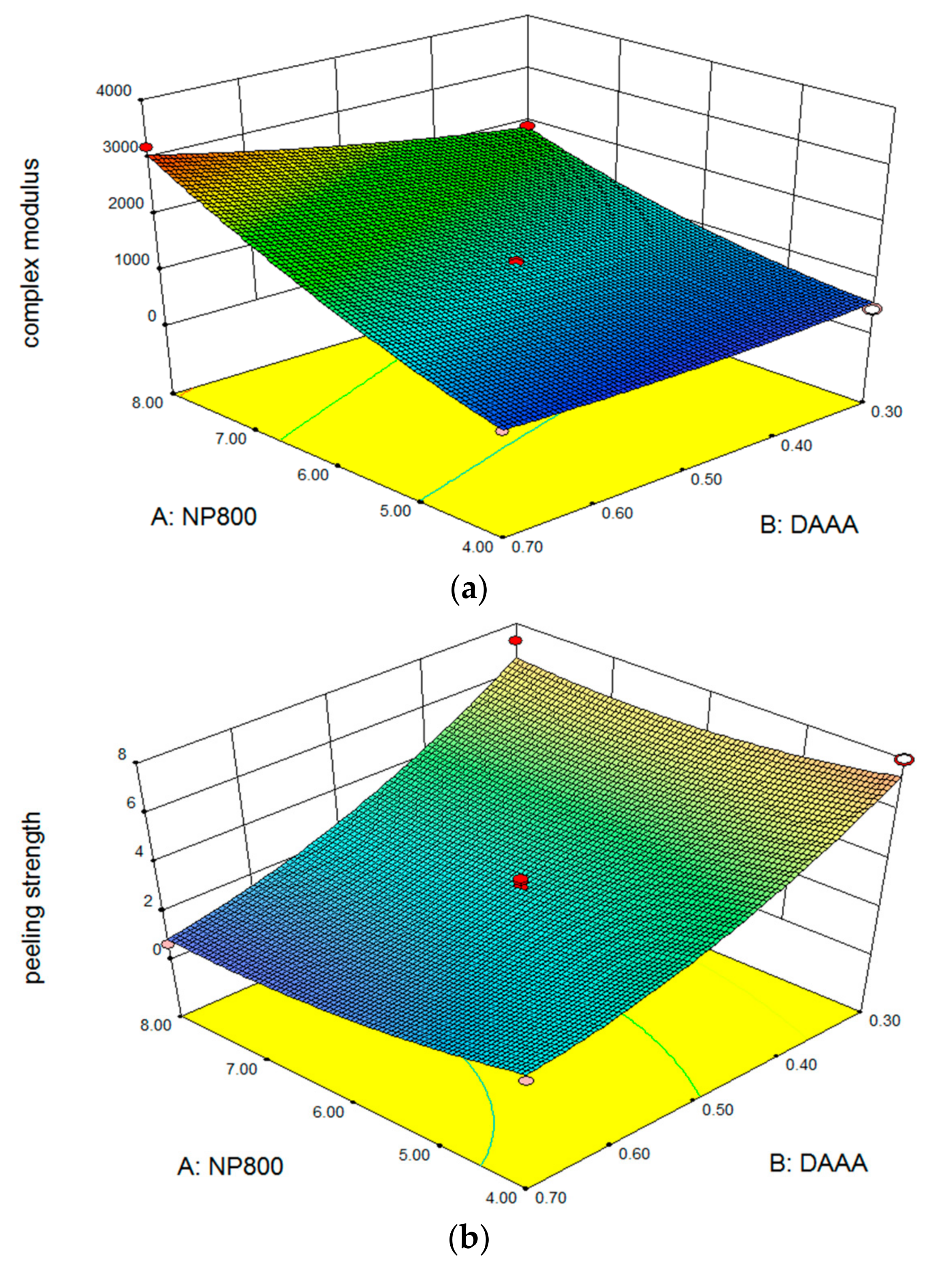
3.2. Multiple Response Optimization and Optimum Range
3.3. Rheological Testing
3.3.1. Stress Sweep
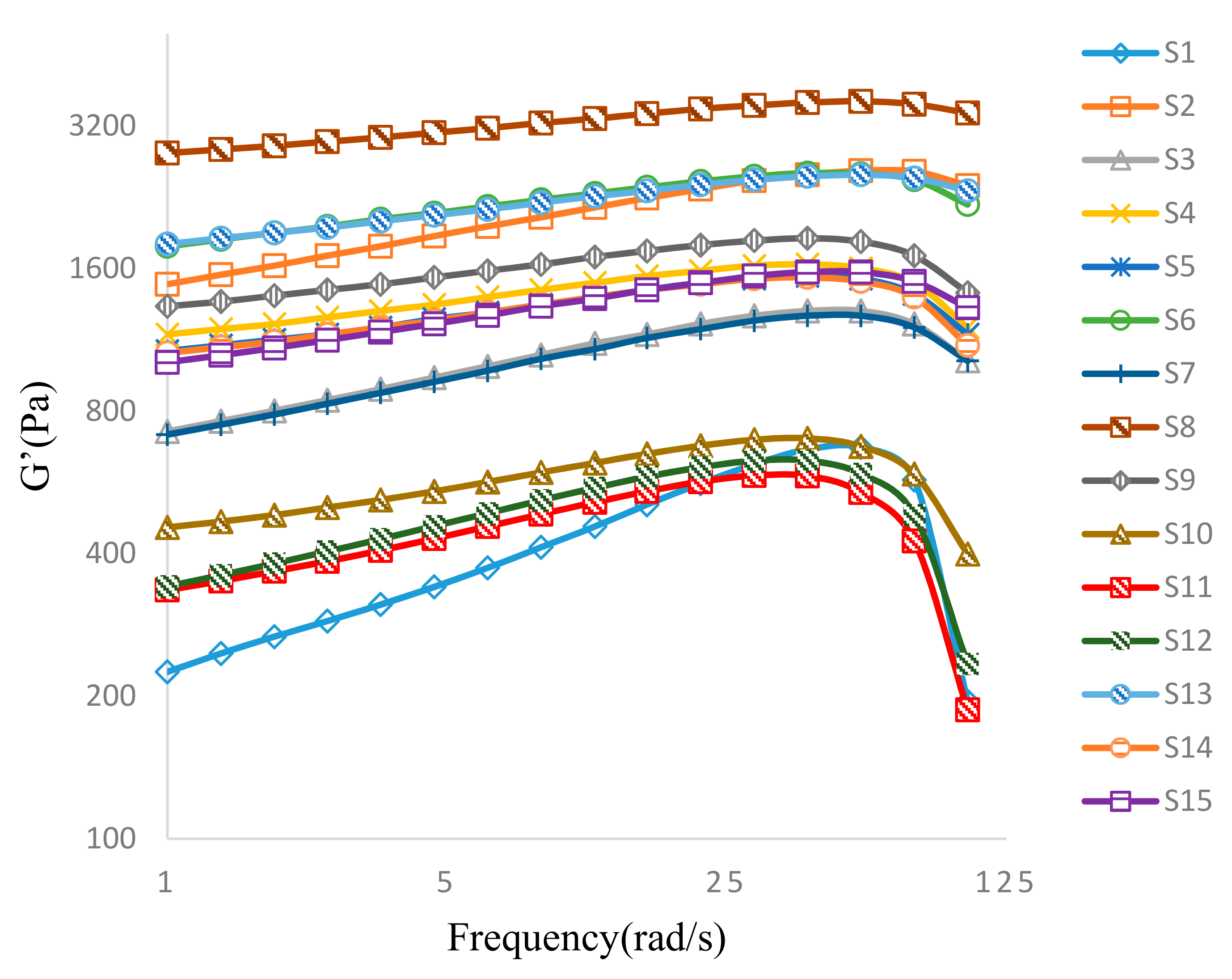
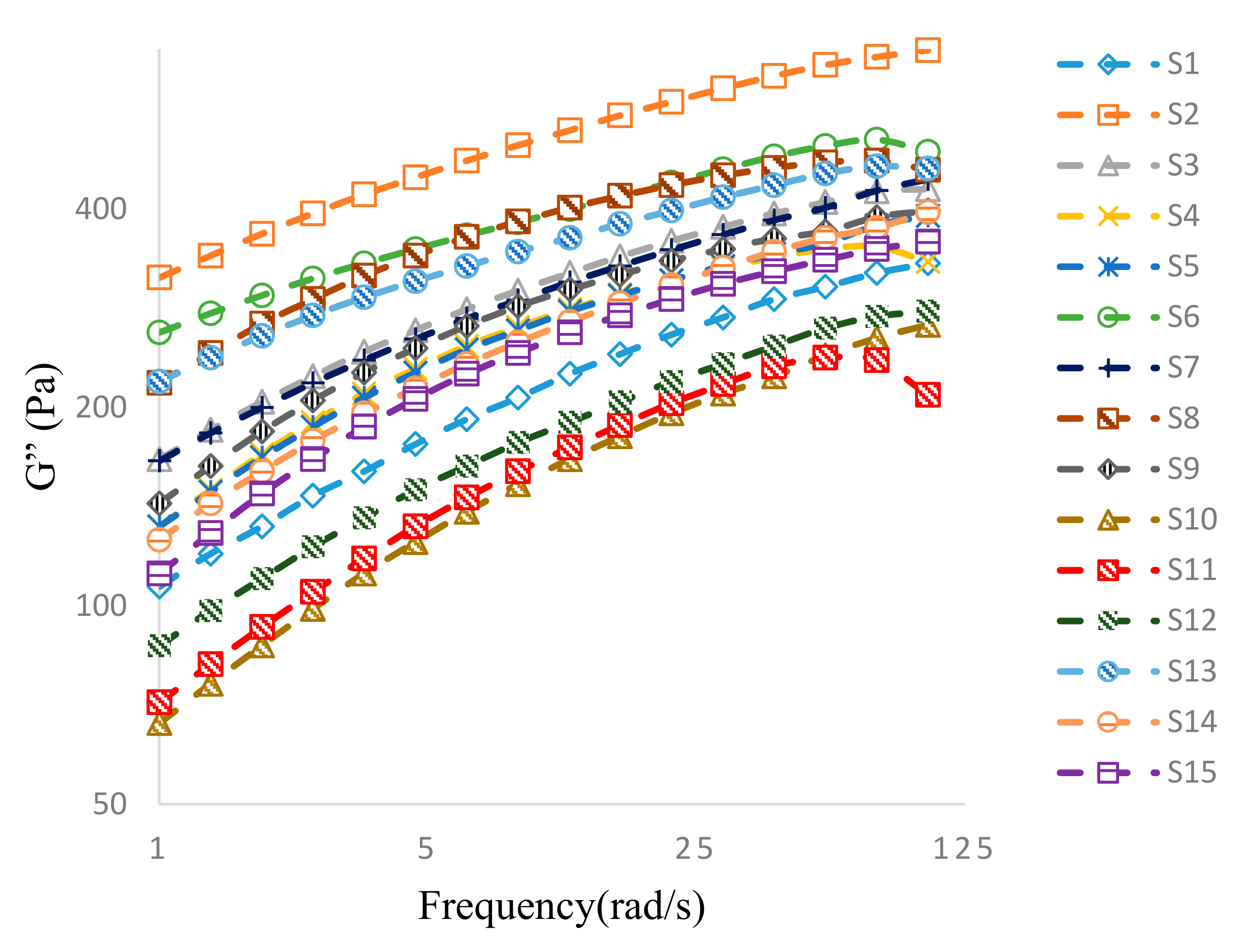
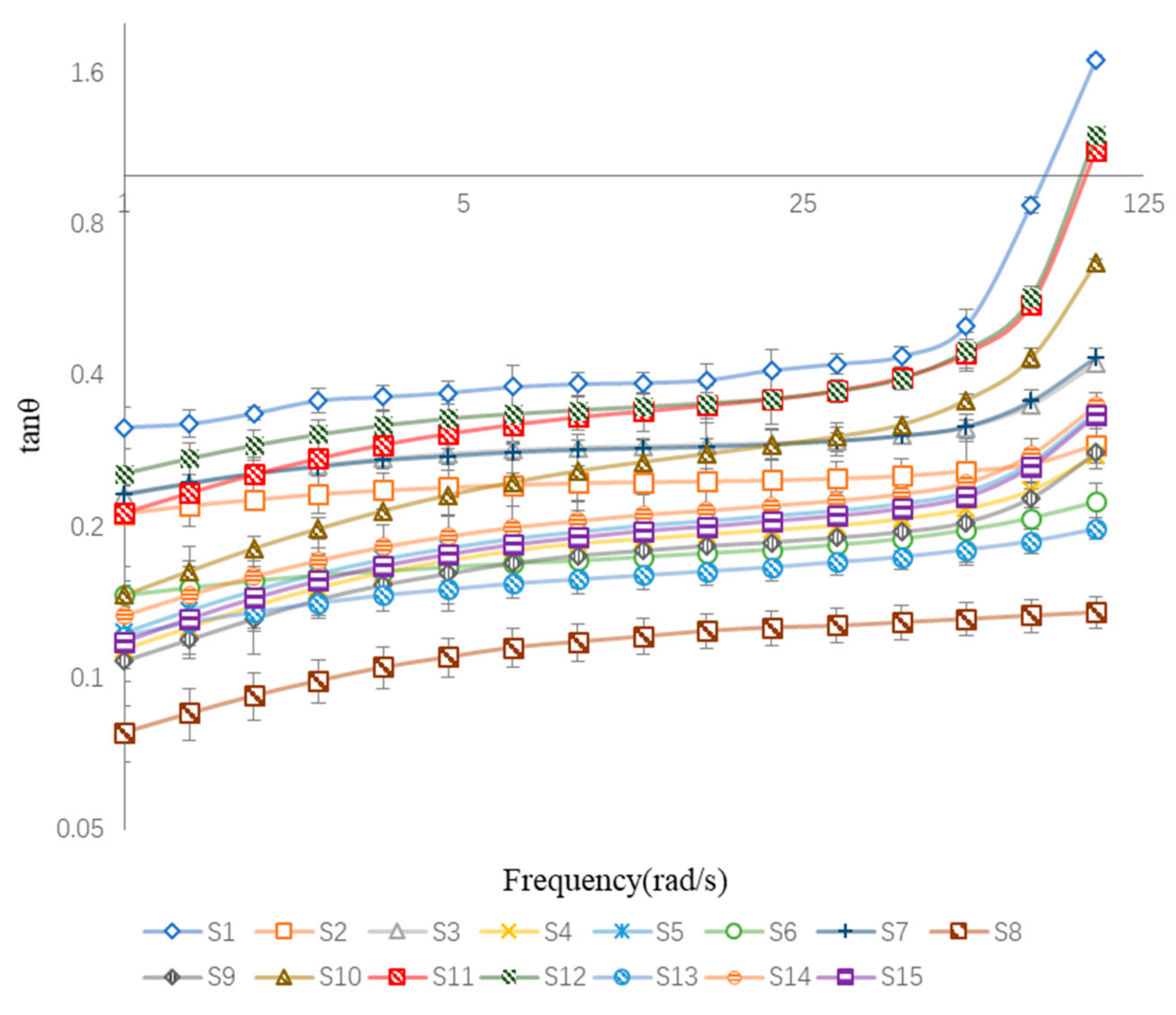
3.3.2. Oscillatory Frequency Sweep
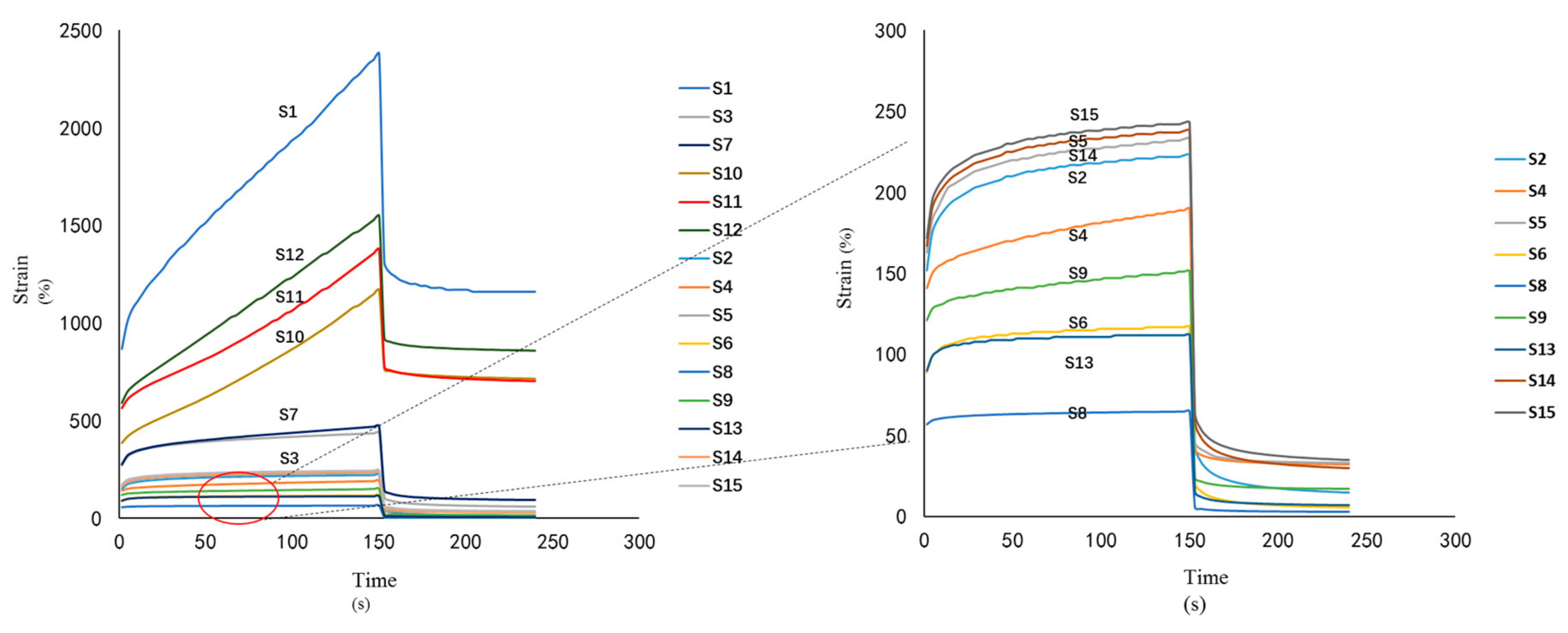
3.3.3. Creep-Recovery Study
3.4. Peeling Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, J.; Zhang, H.; An, D.; Yu, J.; Li, W.; Shen, T.; Wang, J. Rheological characterization of cataplasm bases composed of cross-linked partially neutralized polyacrylate hydrogel. AAPS PharmSciTech 2014, 15, 1149–1154. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.; Liu, S.-Z.; Zhang, Y.-S.; Du, M.-B.; Liang, A.-H.; Song, L.-H.; Ye, Z.-G. Compound antimalarial ethosomal cataplasm: Preparation, evaluation, and mechanism of penetration enhancement. Int. J. Nanomed. 2015, 10, 4239–4253. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Song, W.; Song, T.; Fang, L. Design and evaluation of a novel felbinac transdermal patch: Combining ion-pair and chemical enhancer strategy. AAPS PharmSciTech 2016, 17, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Heng, P.W.S.; Chan, L.W.; Chow, K.T. Development of novel nonaqueous ethylcellulose gel matrices: Rheological and mechanical characterization. Pharm. Res. 2005, 22, 676–684. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.T.; Rodriguez-Hornedo, N.; Ciotti, S.; Ackermann, C. Rheological characterization of topical carbomer gels neutralized to different pH. Pharm. Res. 2004, 21, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Bonacucina, G.; Cespi, M.; Misici-Falzi, M.; Palmieri, G.F. Rheological, adhesive and release characterisation of semisolid carbopol/tetraglycol systems. Int. J. Pharm. 2006, 307, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.A.; El-Say, K.M.; Aljaeid, B.M.; Fahmy, U.A.; Abd-Allah, F.I. Transdermal glimepiride delivery system based on optimized ethosomal nano-vesicles: Preparation, characterization, in vitro, ex vivo and clinical evaluation. Int. J. Pharm. 2016, 500, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Lahooti, B.; Khorram, M.; Karimi1, G.; Mohammadi, A.; Emami, A. Modeling and optimization of antibacterial activity of the chitosan-based hydrogel films using central composite design. J. Biomed. Mater. Res. A 2016, 104, 2544–2553. [Google Scholar] [CrossRef] [PubMed]
- Ghafari, S.; Aziz, H.A.; Isa, M.H.; Zinatizadeh, A.A. Application of response surface methodology (RSM) to optimize coagulation-flocculation treatment of leachate using poly-aluminum chloride (PAC) and alum. J. Hazard. Mater. 2009, 163, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, S.; Maswal, M.; Chat, O.A.; Rather, G.M.; Dar, A.A. Rheological behavior and ibuprofen delivery applications of pH responsive composite alginate hydrogels. Coll. Surf. B Biointerface 2016, 139, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Quaroni, G.M.G.; Gennari, C.G.M.; Cilurzo, F.; Ducouret, G.; Creton, C.; Minghetti, P. Tuning the rheological properties of an ammonium methacrylate copolymer for the design of adhesives suitable for transdermal patches. Eur. J. Pharm. Sci. 2018, 111, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.M.; Ding, B.Y.; Cai, Z.; Zhang, W.; Gao, J.; Ding, X.Y.; Li, W.H.; Gao, S. Cross-linking mechanism of the matrix of hydrogel patch. Yao Xue Xue Bao 2012, 47, 785–791. [Google Scholar] [PubMed]
- Tamburic, S.; Craig, D.Q.M. An investigation into the rheological, dielectric and mucoadhesive properties of poly(acrylic acid) gel systems. J. Control. Release 1995, 37, 59–68. [Google Scholar] [CrossRef]
- Peppas, N.A.; Sahlin, J.J. Hydrogels as mucoadhesive and bioadhesive materials: A review. Biomaterials 1996, 17, 1553–1561. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, W.; Shahsavan, H.; Arunbabu, D. Poly(AAc-co-MBA) hydrogel films: Adhesive and mechanical properties in aqueous medium. J. Phys. Chem. B 2013, 117, 441–449. [Google Scholar]
- Tung, C.-Y.M.; Dynes, P.J. Relationship between viscoelastic properties and gelation in thermosetting systems. J. Appl. Polym. Sci. 1982, 27, 569–574. [Google Scholar] [CrossRef]
- Moczo, P.; Bard, P.-Y. Wave diffraction, amplification and differential motion near strong lateral discontinuities. Bull. Seismol. Soc. Am. 1993, 83, 85–106. [Google Scholar]
- Shen, M.; Li, L.; Sun, Y.; Xu, J.; Guo, X.; Prud’homme, R.K. Rheology and adhesion of poly(acrylic acid)/laponite nanocomposite hydrogels as biocompatible adhesives. Langmuir 2014, 30, 1636–1642. [Google Scholar] [CrossRef] [PubMed]
- Dou, P.; Zhang, J.Y. Study on adhesion properties of a hot-melt pressure-sensitive adhesive based on epoxidized styreneisoprene-styrene triblock copolymers (ESIS) for transdermal drug delivery systems. J. Adhes. 2013, 89, 358–368. [Google Scholar] [CrossRef]

| Independent Variables | Level | |
|---|---|---|
| High | Low | |
| NP800 (%) | 8 | 4 |
| DAAA (%) | 0.7 | 0.3 |
| Felbinac (%) | 1 | 0.6 |
| Dependent responses | Aim | |
| Complex modulus (Pa) | 800–1000 | |
| Peeling strength (N/m) | maximize | |
| No. | Independent Variables | Dependent Responses * | |||
|---|---|---|---|---|---|
| X1 (%) | X2 (%) | X3 (%) | Y1 (Pa) | Y2 (N/m) | |
| S1 | 4 | 0.3 | 0.8 | 450 (±5) | 7.96 (±0.19) |
| S2 | 8 | 0.3 | 0.8 | 1910 (±8) | 7.33 (±0.007) |
| S3 | 6 | 0.3 | 1.0 | 994 (±6) | 5.81 (±0.55) |
| S4 | 6 | 0.7 | 1.0 | 1370 (±5) | 2.51 (±0.002) |
| S5 | 6 | 0.5 | 0.8 | 1270 (±3) | 2.86 (±0.19) |
| S6 | 8 | 0.5 | 0.6 | 2130 (±5) | 2.00 (±0.08) |
| S7 | 6 | 0.3 | 0.6 | 973 (±8) | 5.73 (±0.27) |
| S8 | 8 | 0.7 | 0.8 | 3200 (±7) | 1.06 (±0.04) |
| S9 | 6 | 0.7 | 0.6 | 1550 (±5) | 1.25 (±0.20) |
| S10 | 4 | 0.7 | 0.8 | 563 (±7) | 2.82 (±0.005) |
| S11 | 4 | 0.5 | 0.6 | 465 (±3) | 5.80 (±0.16) |
| S12 | 4 | 0.5 | 1.0 | 496 (±3) | 5.72 (±0.34) |
| S13 | 8 | 0.5 | 1.0 | 2340 (±9) | 1.61 (±0.001) |
| S14 | 6 | 0.5 | 0.8 | 1230 (±8) | 2.36 (±0.14) |
| S15 | 6 | 0.5 | 0.8 | 1170 (±7) | 2.55 (±0.3) |
| Source | Y1 | Source | Y2 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimated Effect | F Value | p Value | R2 | Estimated Effect | F Value | p Value | R2 | ||
| Model | 1223.33 | 50.65 | 0.0020 * | 0.9892 | Model | 2.66 | 18.78 | 0.0002 * | 0.9125 |
| X1 | 950.75 | 386.30 | <0.0001 * | X1 | −1.29 | 18.34 | 0.0020 * | ||
| X2 | 294.50 | 37.07 | 0.0017 * | X2 | −2.40 | 63.67 | <0.0001 * | ||
| X3 | 10.25 | 0.045 | 0.8406 | X1X2 | −0.28 | 0.44 | 0.5230 | ||
| X1X2 | 294.25 | 18.50 | 0.0077 * | X12 | 1.07 | 5.9 | 0.0381 * | ||
| X1X3 | 44.75 | 0.43 | 0.5419 | X22 | 1.11 | 6.37 | 0.0325 * | ||
| X2X3 | −50.25 | 0.54 | 0.4956 | ||||||
| X12 | 221.71 | 9.70 | 0.0264 * | ||||||
| X22 | 85.71 | 1.45 | 0.2826 | ||||||
| X32 | −87.29 | 1.50 | 0.2748 | ||||||
| Source | Lack of Fit | R2 | Adj R2 | Adeq Precisior | |||||
| Y1 | 0.0801 | 0.9892 | 0.9696 | 24.149 | |||||
| Y2 | 0.0669 | 0.9125 | 0.8640 | 13.710 | |||||
| Formulation | Predicted Value | Measured Value | Relative Error | Predicted Value | Measured Value | Relative Error |
|---|---|---|---|---|---|---|
| Y1 (Pa) | Y1 (Pa) | (%) | Y2 (N/m) | Y2 (N/m) | (%) | |
| A | 895.06 | 925.43 | 3.28 | 6.51 | 6.43 | 1.24 |
| B | 883.08 | 873.35 | 1.10 | 2.87 | 3.04 | 5.59 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Zhu, Y.; Diao, Y.; Yin, C. Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design. Pharmaceutics 2018, 10, 88. https://doi.org/10.3390/pharmaceutics10030088
Yang J, Zhu Y, Diao Y, Yin C. Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design. Pharmaceutics. 2018; 10(3):88. https://doi.org/10.3390/pharmaceutics10030088
Chicago/Turabian StyleYang, Jie, Yishen Zhu, Yongqin Diao, and Caiyun Yin. 2018. "Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design" Pharmaceutics 10, no. 3: 88. https://doi.org/10.3390/pharmaceutics10030088
APA StyleYang, J., Zhu, Y., Diao, Y., & Yin, C. (2018). Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design. Pharmaceutics, 10(3), 88. https://doi.org/10.3390/pharmaceutics10030088





