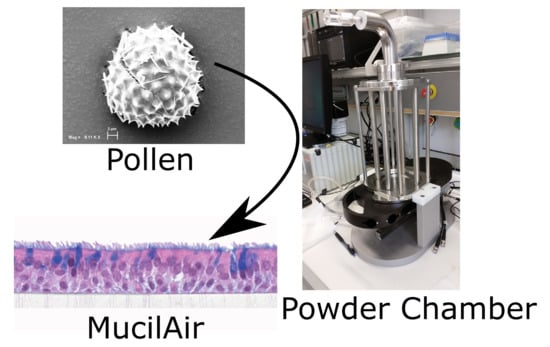
Combining MucilAir™ and Vitrocell® Powder Chamber for the In Vitro Evaluation of Nasal Ointments in the Context of Aerosolized Pollen
Abstract
:1. Introduction
2. Materials and Methods
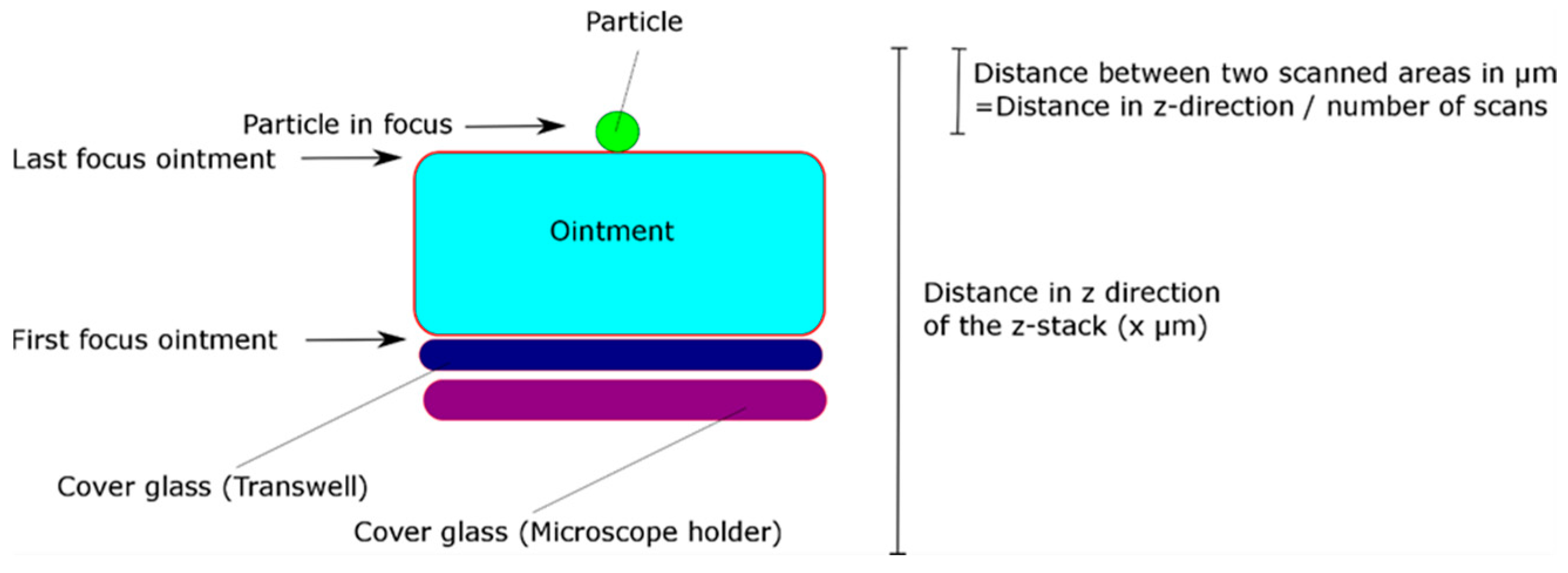
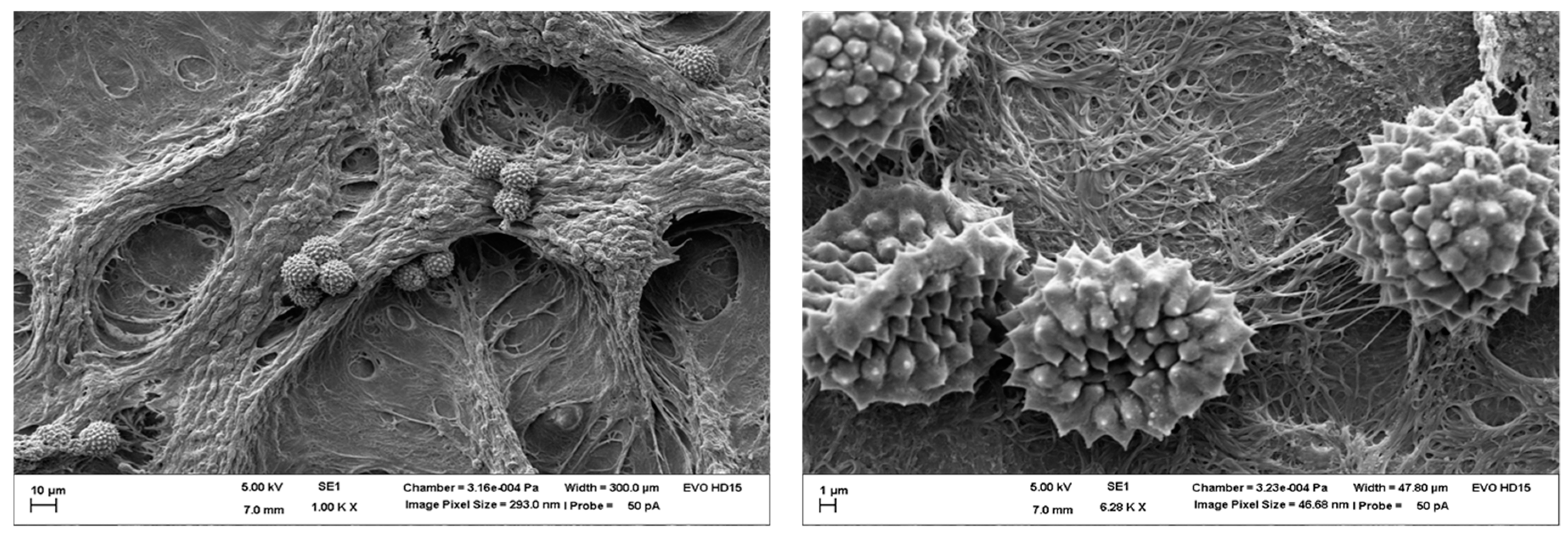
2.1. Microscopy
2.2. Parameters for Pollen Deposition with the Vitrocell® Powder Chamber
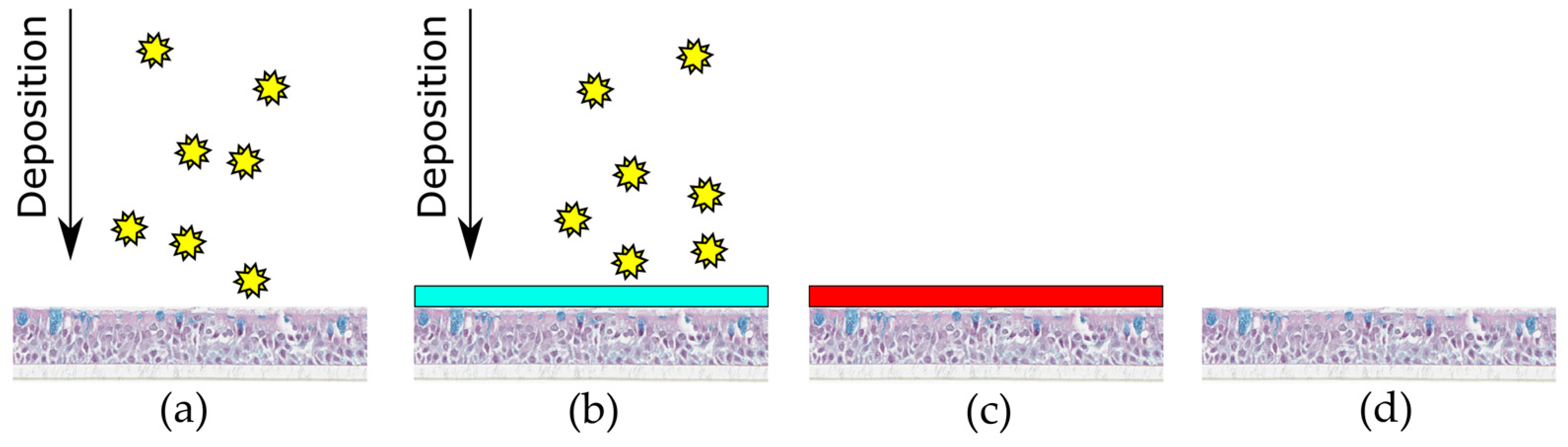
2.3. Penetration of Pollen into an Ointment Layer
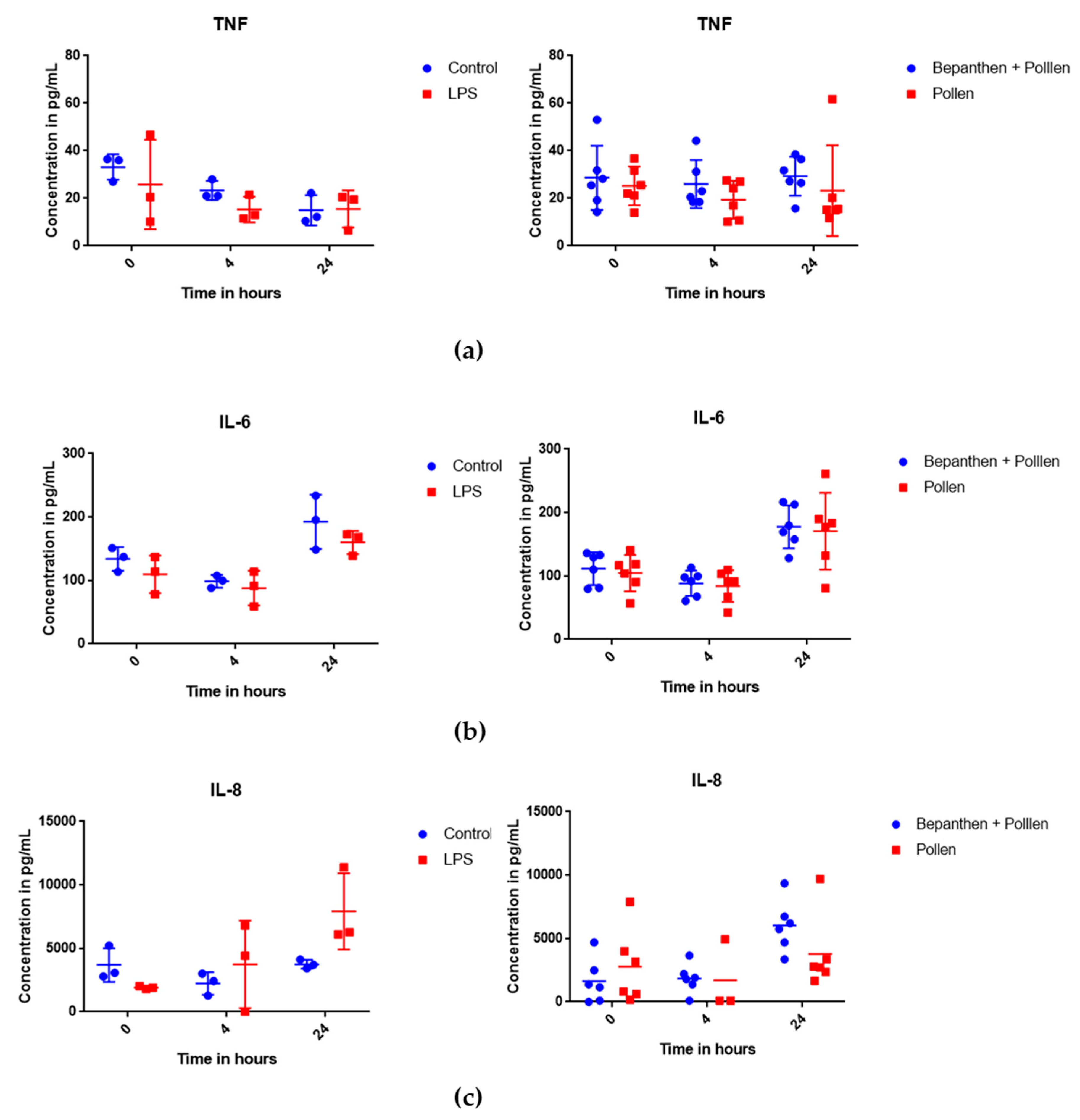
2.4. Cytokine Release after Deposition on MucilAir™
3. Results
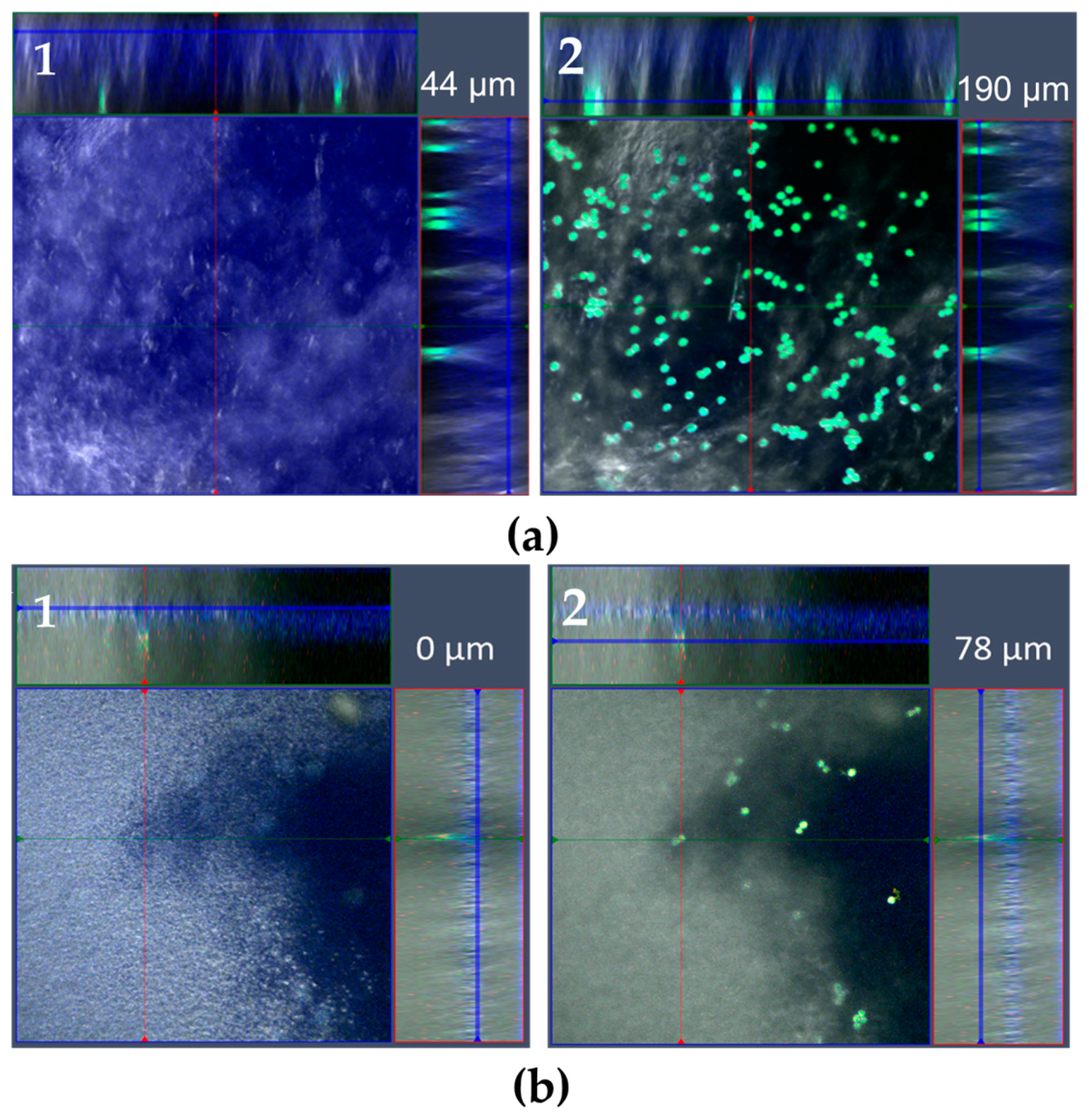
3.1. Pollen Selection
3.2. Penetration of Pollen into an Ointment Layer
3.3. Cytokine Release after Deposition on MucilAir™
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- WHO. Global Surveillance, Prevention and Control of Chronic Respiratory Diseases; World Health Organization: Geneva, Switzerland, 2007; pp. 1–146. [Google Scholar]
- Asher, M.I.; Montefort, S.; Björkstén, B.; Lai, C.K.; Strachan, D.P.; Weiland, S.K. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 2006, 368, 733–743. [Google Scholar] [CrossRef]
- Burr, M.L.; Emberlin, J.C.; Treu, R.; Cheng, S.; Pearce, N.E. Pollen counts in relation to the prevalence of allergic rhinoconjunctivitis, asthma and atopic eczema in the International Study of Asthma and Allergies in Childhood (ISAAC). Clin. Exp. Allergy 2003, 33, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- Bachert, C.; Borchard, U.; Wedi, B.; Klimek, L.; Rasp, G.; Riechelmann, H. Allergische rhinokonjunktivitis: Leitlinie der deutschen gesellschaft für allergologie und klinische Immunologie (DGAI). Allergo J. 2003, 12, 182–194. [Google Scholar] [CrossRef]
- Day, J. Pros and cons of the use of antihistamines in managing allergic rhinitis. J. Allergy Clin. Immunol. 1999, 103, 395–399, a96504. [Google Scholar] [CrossRef]
- Estelle, R.F.; Simons, R.; Simons, K.J. The pharmacology and use of H1-Receptor-antagonist drugs. Drug Ther. 1994, 330, 1663–1670. [Google Scholar] [CrossRef]
- Golightly, L.; Greos, L. Second-Generation Antihistamines Allergic Disorders. Drugs 2005, 65, 341–384. [Google Scholar] [CrossRef] [PubMed]
- Frass, M.; Strassl, R.P.; Friehs, H.; Mullner, M.; Kundi, M.; Kaye, A. Use and Acceptance of Complementary and Alternative Medicine Among the General Population and Medical Personnel: A Systematic Review. Ochsner J. 2012, 12, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Engel, L.W.; Straus, S.E. Development of therapeutics: Opportunities within complementary and alternative medicine. Nat. Rev. Drug Discov. 2002, 1, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Ziment, I.; Tashkin, D.P. Alternative medicine for allergy and asthma. J. Allergy Clin. Immunol. 2000, 106, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Sharma, S.; Garg, S. Permeability issues in nasal drug delivery. Drug Discov. Today 2002, 7, 967–975. [Google Scholar] [CrossRef]
- Kloecker, N.; Rudolph, P.; Verse, T. Evaluation of protective and therapeutic effects of dexpanthenol on nasal decongestants and preservatives: Results of cytotoxic studies In vitro. Am. J. Rhinopl. 2004, 18, 315–320. [Google Scholar]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Hittinger, M.; Barthold, S.; Siebenbürger, L.; Zäh, K.; Gress, A.; Guenther, S.; Wiegand, B.; Boerger, C.; Berger, M.; Krebs, T.; et al. Proof of concept of the Vitrocell® dry powder chamber: A new in vitro test system for the controlled deposition of aerosol formulation. In Proceedings of the Europe Respiratory Drug Delivery 2017 Scientific Conference, Antibes, France, 25–28 April 2017; pp. 283–288. [Google Scholar]
- Huang, S.; Wiszniewski, L.; Constant, S.; Roggen, E. Potential of in vitro reconstituted 3D human airway epithelia (MucilAir) to assess respiratory sensitizers. Toxicol. Vitr. 2013, 27, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Epithelix. MucilAir by Epithelix. Available online: http://www.epithelix.com/products/mucilair (accessed on 13 January 2018).
- Balogh Sivars, K.; Sivars, U.; Hornberg, E.; Zhang, H.; Brändén, L.; Bonfante, R. A 3D Human airway model enables prediction of respiratory toxicity of inhaled drugs in vitro. Toxicol. Sci. 2018, 162, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Vitrocell-Powder-Chamber. Available online: https://www.vitrocell.com/inhalation-toxicology/exposure-systems/vitrocell-powder-chamber (accessed on 29 March 2018).
- Kooter, I.M.; Gröllers-Mulderij, M.; Duistermaat, E.; Kuper, F.; Schoen, E.D. Factors of concern in a human 3D cellular airway model exposed to aerosols of nanoparticles. Toxicol. In Vitro 2017, 44, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Balloy, V.; Deveaux, A.; Lebeaux, D.; Tabary, O.; Le Rouzic, P.; Ghigo, J.M. Azithromycin analogue CSY0073 attenuates lung inflammation induced by LPS challenge. Br. J. Pharmacol. 2014, 171, 1783–1794. [Google Scholar] [CrossRef] [PubMed]
- Follak, S. Notizen zum Vorkommen von Iva xanthiifolia in der Südwestslowakei. Stapfia 2014, 101, 71–77. [Google Scholar]
- Cariñanos, P.; Casares, P.M. Urban green zones and related pollen allergy: A review. Some guidelines for designing spaces with low allergy impact. Landsc. Urban Plan. 2011, 101, 205–214. [Google Scholar] [CrossRef]
- Heinzerling, L.; Frew, A.J.; Bindslev, J.C.; Bonini, S.; Bousquet, J.; Bresciani, M. Standard skin prick testing and sensitization to inhalant allergens across Europe-A survey from the GA2LEN network. Allergy Eur. J. Allergy Clin. Immunol. 2005, 60, 1287–1300. [Google Scholar] [CrossRef] [PubMed]
- Christiane, S.M.; Peter, H.; Lang, S.R.; Ditzinger, G.; Merkle, H.P. In vitro cell models to study nasal mucosal permeability and metabolism. Adv. Drug Deliv. Rev. 1998, 29, 51–79. [Google Scholar] [CrossRef]
- Huang, S.; Boda, B.; Vernaz, J.; Ferreira, E.; Wiszniewski, L.; Constant, S. Establishment and characterization of an in vitro human small airway model (SmallAirTM). Eur. J. Pharm. Biopharm. 2017, 118, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, P.; Mickenhagen, A.; Siefer, O.; Walger, M. A new approach to pharmacological effects on ciliary beat frequency in cell cultures—Exemplary measurements under Pelargonium sidoides extract (EPs 7630). Phytomedicine 2005, 12, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, G.L.; Marousi, S.; Ellul, J.; Mougiou, A.; Theodori, E.; Mouzaki, A. T helper 1 (Th1)/Th2 cytokine expression shift of peripheral blood CD4 + and CD8+ T cells in patients at the post-acute phase of stroke. Clin. Exp. Immunol. 2008, 152, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Wiszniewski, L.; Constant, S. Chapter 8: The Use of In Vitro 3D Cell Models in Drug Development for Respiratory Diseases. In The Use of In Vitro 3D Cell Models in Drug Development for Respiratory Diseases, Drug Discovery and Development—Present and Future; InTech: Rijeka, Croatia, 2011; pp. 169–191. ISBN 978-953-307-615-7. [Google Scholar]
- De, S.B.; Ranzini, F.; Piqué, N. Protective barrier properties of Rhinosectan® spray (containing xyloglucan) on an organotypic 3D airway tissue model (MucilAir): Results of an in vitro study. Allergy Asthma Clin. Immunol. BioMed Central 2017, 13, 37. [Google Scholar] [CrossRef]
- Murgi, X.; Lorentz, B.; Hartwig, O.; Hittinger, M.; Lehr, C.M. The role of mucus on drug transport and its potential to affect therapeutic outcomes. Adv. Drug Deliv. Rev. 2017, 124, 82–97. [Google Scholar] [CrossRef] [PubMed]
- Cohn, L.; Homer, R.J.; Niu, N.; Bottomly, K. T helper 1 cells and interferon gamma regulate allergic airway inflammation and mucus production. J. Exp. Med. 1999, 190, 1309–1318. [Google Scholar] [CrossRef] [PubMed]
- Scavuzzo, M.C.; Rocchi, V.; Fattori, B.; Ambrogi, F.; Carpi, A.; Ruffoli, R. Cytokine secretion in nasal mucus of normal subjects and patients with allergic rhinitis. Biomed. Pharmacother. 2003, 57, 366–371. [Google Scholar] [CrossRef]

| Pollen Species | Characteristics | Scanning Electron Microscopy | Confocal Light Scanning Microscopy |
|---|---|---|---|
| Iva xanthiifolia | Size: ~28 µm Shape: serrated, rough surface Allergic potential: allergenic Neophyt [21] |  |  |
| Populus nigra italic | Size: ~26 µm Shape: uneven, particulate surface Allergic potential: ♂ dioecious [22] |  |  |
| Populus tremuloides | Size: ~30 µm Shape: ovate, particulate surface Allergic potential: not known |  |  |
| Populus deltoides | Size: ~28 µm Shape: longish, porous surface Allergic potential: yes [23] |  |  |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Metz, J.; Knoth, K.; Groß, H.; Lehr, C.-M.; Stäbler, C.; Bock, U.; Hittinger, M. Combining MucilAir™ and Vitrocell® Powder Chamber for the In Vitro Evaluation of Nasal Ointments in the Context of Aerosolized Pollen. Pharmaceutics 2018, 10, 56. https://doi.org/10.3390/pharmaceutics10020056
Metz J, Knoth K, Groß H, Lehr C-M, Stäbler C, Bock U, Hittinger M. Combining MucilAir™ and Vitrocell® Powder Chamber for the In Vitro Evaluation of Nasal Ointments in the Context of Aerosolized Pollen. Pharmaceutics. 2018; 10(2):56. https://doi.org/10.3390/pharmaceutics10020056
Chicago/Turabian StyleMetz, Julia, Katharina Knoth, Henrik Groß, Claus-Michael Lehr, Carolin Stäbler, Udo Bock, and Marius Hittinger. 2018. "Combining MucilAir™ and Vitrocell® Powder Chamber for the In Vitro Evaluation of Nasal Ointments in the Context of Aerosolized Pollen" Pharmaceutics 10, no. 2: 56. https://doi.org/10.3390/pharmaceutics10020056
APA StyleMetz, J., Knoth, K., Groß, H., Lehr, C.-M., Stäbler, C., Bock, U., & Hittinger, M. (2018). Combining MucilAir™ and Vitrocell® Powder Chamber for the In Vitro Evaluation of Nasal Ointments in the Context of Aerosolized Pollen. Pharmaceutics, 10(2), 56. https://doi.org/10.3390/pharmaceutics10020056