Frequency and Pathological Phenotype of Bovine Astrovirus CH13/NeuroS1 Infection in Neurologically-Diseased Cattle: Towards Assessment of Causality
Abstract
:1. Introduction
2. Materials and Methods
3. Results
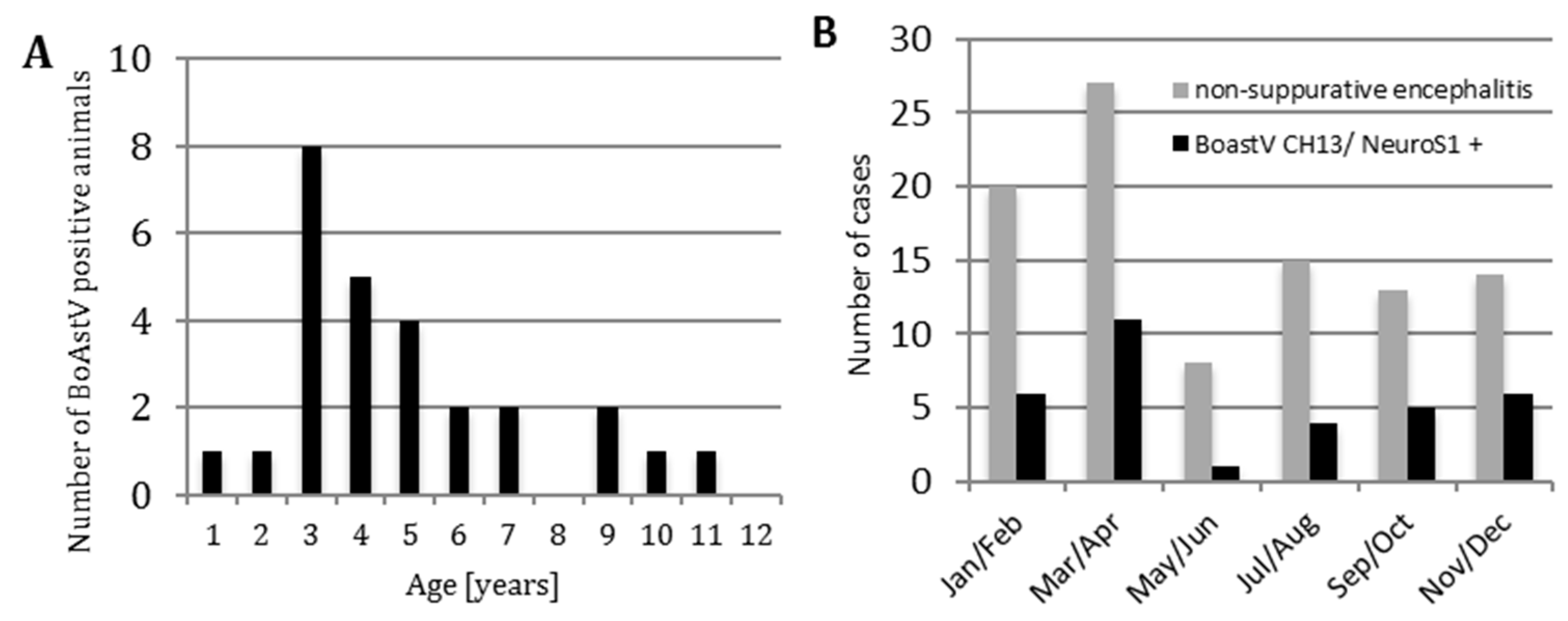
3.1. Frequency of Bovine Astrovirus (BoAstV) CH13/NeuroS1 Infection
3.2. Anamnestic Clinical Data
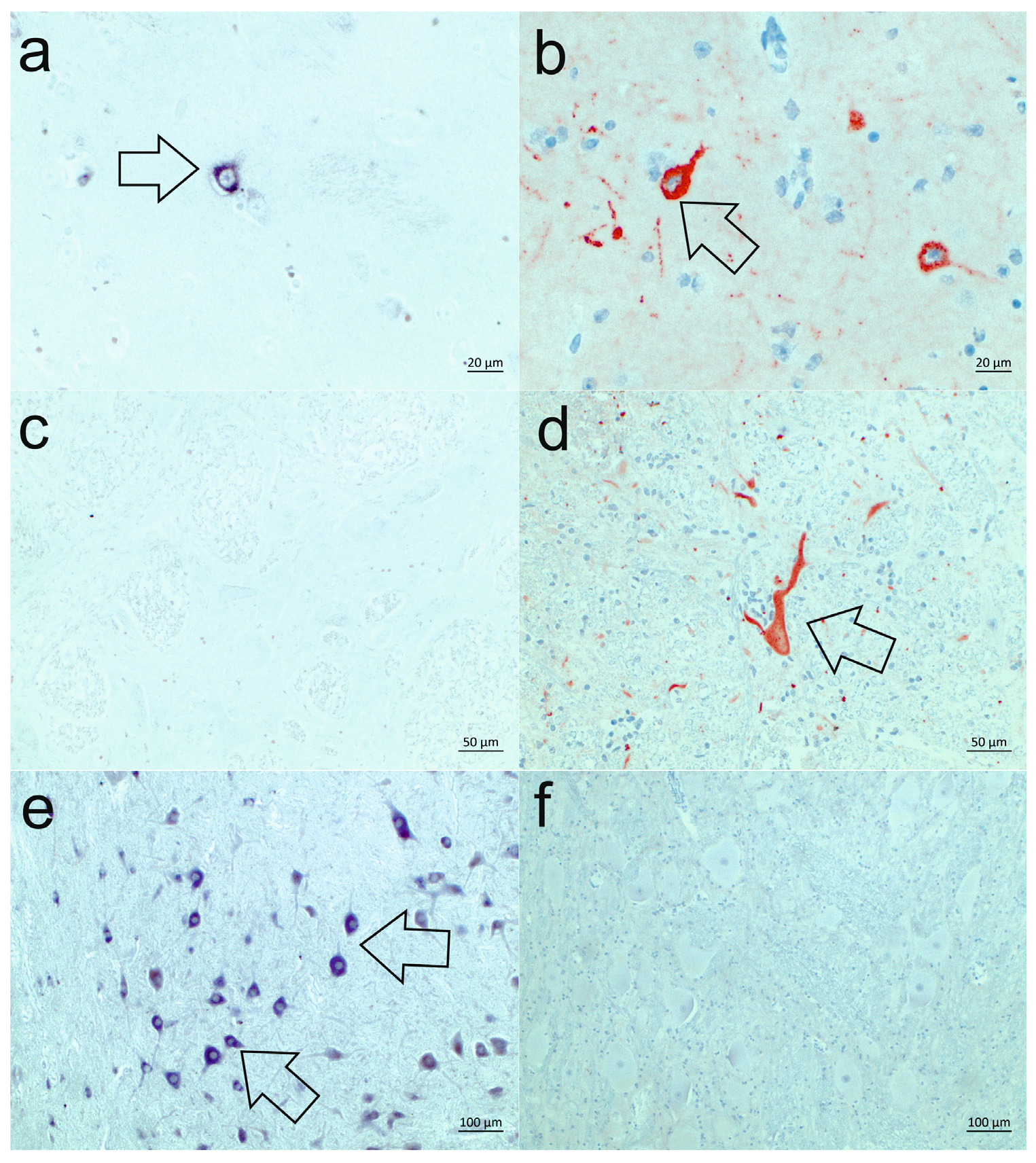
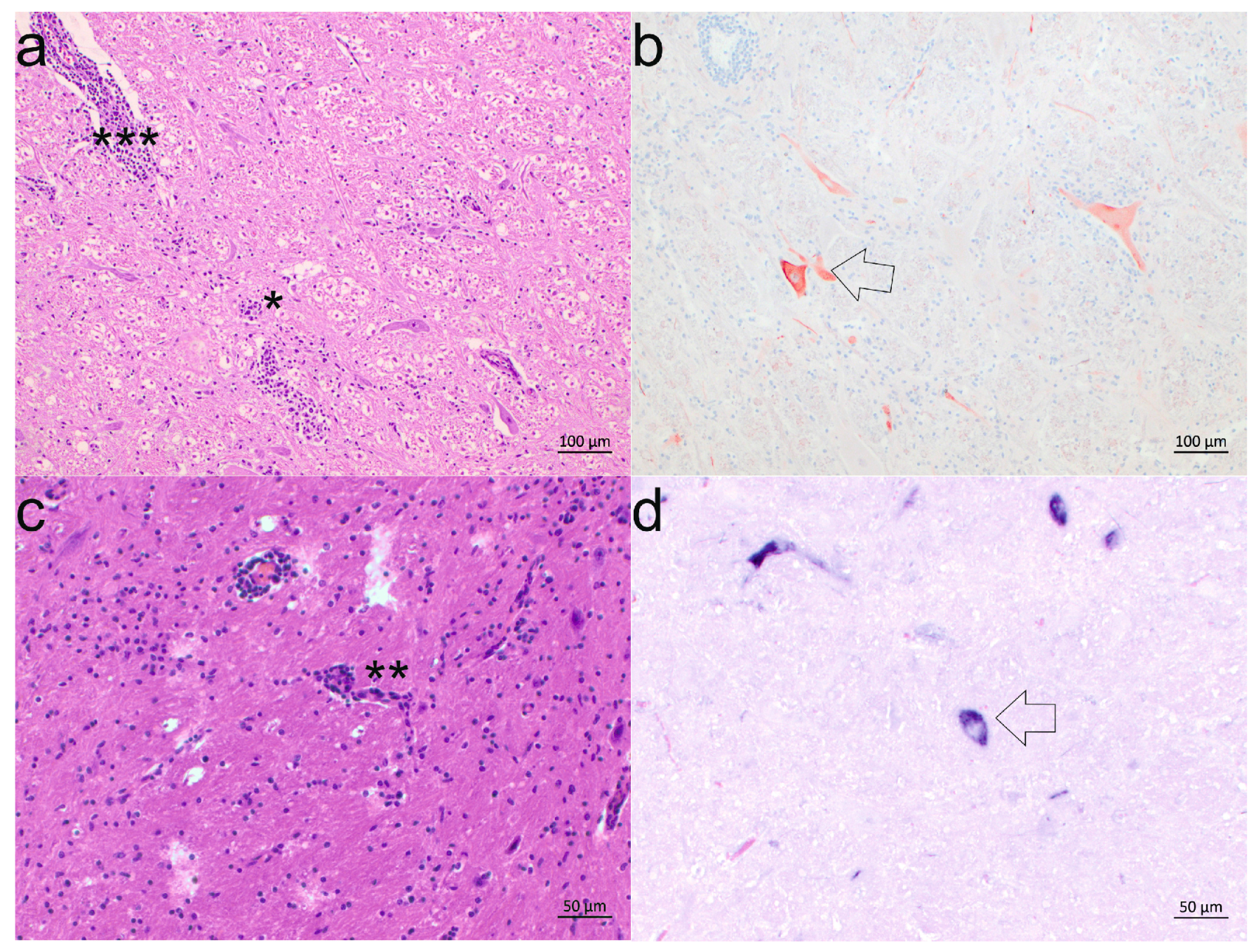
3.3. Neuroanatomical Distribution of BoAstV CH13/NeuroS1
3.4. Cell Tropism
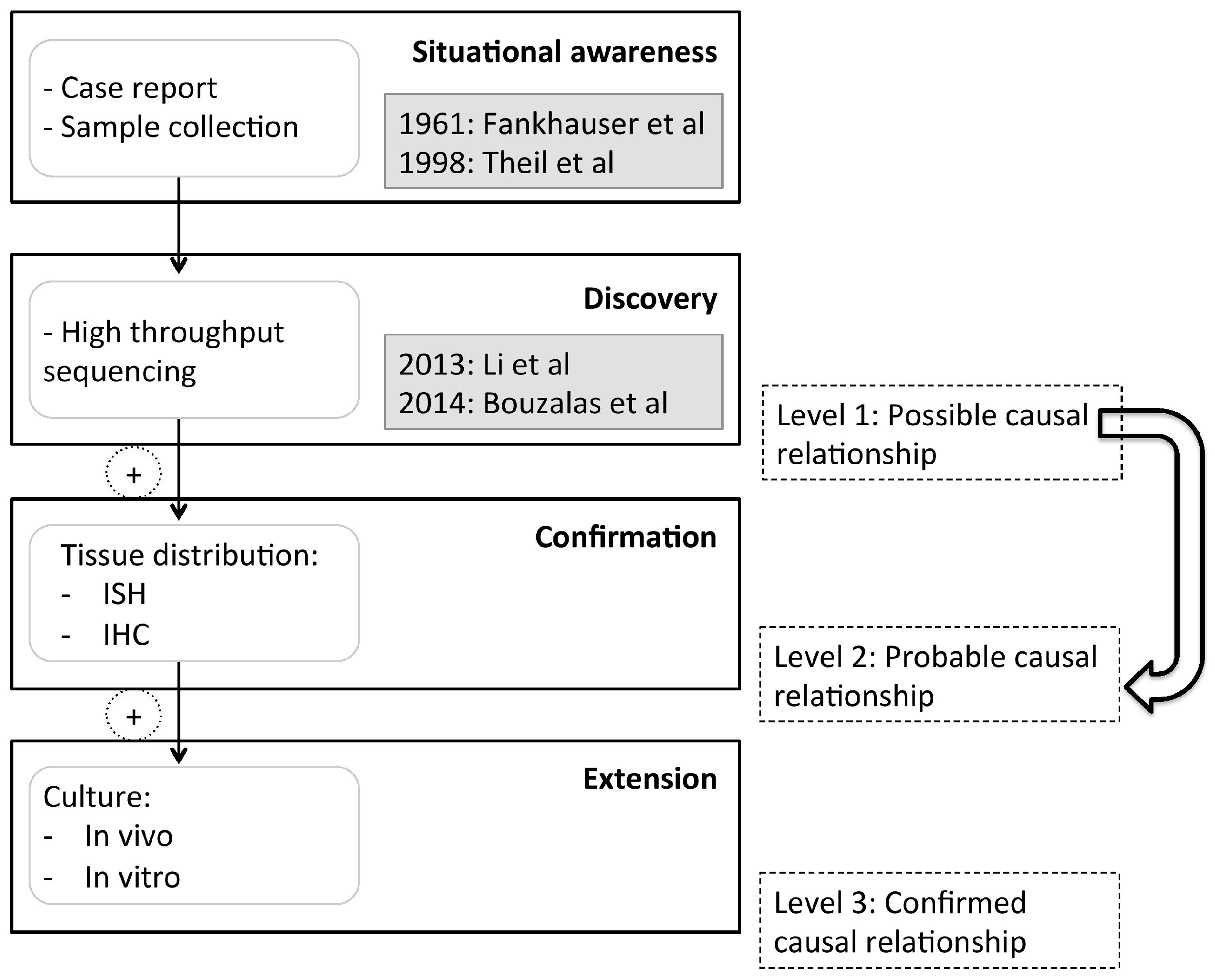
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Retief, F.; Cilliers, L. Measles in antiquity and the Middle Ages. S. Afr. Med. J. 2010, 100, 216–217. [Google Scholar] [CrossRef] [PubMed]
- Appleton, H.; Higgins, P.G. Letter: Viruses and gastroenteritis in infants. Lancet 1975, 1, 1297. [Google Scholar] [CrossRef]
- De Benedictis, P.; Schultz-Cherry, S.; Burnham, A.; Cattoli, G. Astrovirus infections in humans and animals — molecular biology, genetic diversity, and interspecies transmissions. Infect. Genet. Evol. 2011, 11, 1529–1544. [Google Scholar] [CrossRef] [PubMed]
- Blomstrom, A.L.; Widen, F.; Hammer, A.S.; Belak, S.; Berg, M. Detection of a novel astrovirus in brain tissue of mink suffering from shaking mink syndrome by use of viral metagenomics. J. Clin. Microbiol. 2010, 48, 4392–4396. [Google Scholar] [CrossRef] [PubMed]
- Quan, P.L.; Wagner, T.A.; Briese, T.; Torgerson, T.R.; Hornig, M.; Tashmukhamedova, A.; Firth, C.; Palacios, G.; Baisre-De-Leon, A.; Paddock, C.D.; et al. Astrovirus encephalitis in boy with X-linked agammaglobulinemia. Emerg. Infect. Dis. 2010, 16, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Diab, S.; McGraw, S.; Barr, B.; Traslavina, R.; Higgins, R.; Talbot, T.; Blanchard, P.; Rimoldi, G.; Fahsbender, E.; et al. Divergent astrovirus associated with neurologic disease in cattle. Emerg. Infect. Dis. 2013, 19, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Bouzalas, I.G.; Wuthrich, D.; Walland, J.; Drogemuller, C.; Zurbriggen, A.; Vandevelde, M.; Oevermann, A.; Bruggmann, R.; Seuberlich, T. Neurotropic astrovirus in cattle with nonsuppurative encephalitis in Europe. J. Clin. Microbiol. 2014, 52, 3318–3324. [Google Scholar] [CrossRef] [PubMed]
- Cordey, S.; Vu, D.L.; Schibler, M.; L'Huillier, A.G.; Brito, F.; Docquier, M.; Posfay-Barbe, K.M.; Petty, T.J.; Turin, L.; Zdobnov, E.M.; et al. Astrovirus MLB2, a New Gastroenteric Virus Associated with Meningitis and Disseminated Infection. Emerg. Infect. Dis. 2016, 22, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Seuberlich, T.; Wuthrich, D.; Selimovic-Hamza, S.; Drogemuller, C.; Oevermann, A.; Bruggmann, R.; Bouzalas, I. Identification of a second encephalitis-associated astrovirus in cattle. Emerg. Microbes Infect. 2016, 5, e5. [Google Scholar] [CrossRef] [PubMed]
- Schlottau, K.; Schulze, C.; Bilk, S.; Hanke, D.; Hoper, D.; Beer, M.; Hoffmann, B. Detection of a Novel Bovine Astrovirus in a Cow with Encephalitis. Transbound Emerg. Dis. 2016, 63, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Kuroda, M.; Kasai, M.; Matsui, H.; Fukuyama, T.; Katano, H. Acute encephalopathy in an immunocompromised boy with astrovirus-MLB1 infection detected by next generation sequencing. J. Clin. Virol. 2016, 78, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Theil, D.; Fatzer, R.; Schiller, I.; Caplazi, P.; Zurbriggen, A.; Vandevelde, M. Neuropathological and aetiological studies of sporadic non-suppurative meningoencephalomyelitis of cattle. Vet. Rec. 1998, 143, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Fankhauser, R. Cerebellar encephalitis in cattle. Schweiz Arch. Tierheilkd 1961, 103, 11. [Google Scholar]
- Fankhauser, R. Sporadic meningo-encephalomyelitis in cattle. Schweiz Arch. Tierheilkd 1961, 103, 225–235. [Google Scholar]
- Frauchiger, E.; Hofmann, W. Nervous Diseases of Cattle; Verlag Hans Huber: Bern, Germany, 1961. [Google Scholar]
- Bouzalas, I.G.; Wuthrich, D.; Selimovic-Hamza, S.; Drogemuller, C.; Bruggmann, R.; Seuberlich, T. Full-genome based molecular characterization of encephalitis-associated bovine astroviruses. Infect. Genet. Evol. 2016, 44, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Rivers, T.M. Viruses and Koch's Postulates. J. Bacteriol. 1937, 33, 1–12. [Google Scholar] [PubMed]
- Lipkin, W.I.; Anthony, S.J. Virus hunting. Virology 2015, 479–480, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Boujon, C.; Selimovic-Hamza, S.; Bouzalas, I.G.; Seuberlich, T. Development and validation of an immunohistochemistry procedure for the detection of a neurotropic bovine astrovirus. J. Virol. Methods 2016, 239, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Selimovic-Hamza, S.; Bouzalas, I.G.; Vandevelde, M.; Oevermann, A.; Seuberlich, T. Detection of Astrovirus in Historical Cases of European Sporadic Bovine Encephalitis, Switzerland 1958–1976. Front. Vet. Sci. 2016, 3, 91. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.D.; Basetse, H.R.; Blacklow, N.R.; Herrmann, J.E. Astrovirus infection in South Africa: A pilot study. Ann. Trop. Paediatr. 1998, 18, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Wüthrich, D.; Boujon, C.L.; Truchet, L.; Selimovic-Hamza, S.; Oevermann, A.; Bouzalas, I.G.; Bruggmann, R.; Seuberlich, T. Exploring the virome of cattle with non-suppurative encephalitis of unknown etiology by metagenomics. Virology 2016, 493, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Murillo, A.; Vera-Estrella, R.; Barkla, B.J.; Mendez, E.; Arias, C.F. Identification of Host Cell Factors Associated with Astrovirus Replication in Caco-2 Cells. J. Virol. 2015, 89, 10359–10370. [Google Scholar] [CrossRef] [PubMed]
| BoAstV CH13/NeuroS1 ISH | Total | ||
|---|---|---|---|
| Non-suppurative encephalitis | + | - | |
| + | 33 | 64 | 97 |
| - | 0 | 58 | 58 |
| Total | 33 | 122 | 155 |
| Case No. | Brainstem | Cerebellum | Cerebrum | Hippocampus | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HE | ISH | IHC | HE | ISH | IHC | HE | ISH | IHC | HE | ISH | IHC | |
| 24231 | +++ | - | - | +++ | +++ | +++ | - | + | + | +++ | +++ | +++ |
| 24594 | ++ | ++ | ++ | +++ | ++ | + | ++ | +++ | ++ | ++ | ++ | + |
| 24595 | +++ | + | + | +++ | + | + | +++ | ++ | ++ | +++ | +++ | ++ |
| 25018 | + | +++ * | - * | +++ | ++ * | - * | - | - | - | - | - | - |
| 26730 | ++ | + | ++ | +++ | ++ | + | - | + | n.a. | ++ | +++ | ++ |
| 26875 | ++ | ++ | +++ | +++ | + | + | + | ++ | + | + | + | + |
| 42799 | +++ | ++ | ++ | +++ | + | + | ++ | + | + | +++ | - | - |
| 43660 | ++ | + | + | - | - | - | - | - | - | ++ | - | - |
| 45664 | +++ | + | + | +++ | + | + | +++ | ++ | ++ | +++ | ++ | ++ |
| 50898 | +++ | - * | ++ * | +++ | ++ | ++ | + | + | ++ | + | ++ | ++ |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selimovic-Hamza, S.; Boujon, C.L.; Hilbe, M.; Oevermann, A.; Seuberlich, T. Frequency and Pathological Phenotype of Bovine Astrovirus CH13/NeuroS1 Infection in Neurologically-Diseased Cattle: Towards Assessment of Causality. Viruses 2017, 9, 12. https://doi.org/10.3390/v9010012
Selimovic-Hamza S, Boujon CL, Hilbe M, Oevermann A, Seuberlich T. Frequency and Pathological Phenotype of Bovine Astrovirus CH13/NeuroS1 Infection in Neurologically-Diseased Cattle: Towards Assessment of Causality. Viruses. 2017; 9(1):12. https://doi.org/10.3390/v9010012
Chicago/Turabian StyleSelimovic-Hamza, Senija, Céline L. Boujon, Monika Hilbe, Anna Oevermann, and Torsten Seuberlich. 2017. "Frequency and Pathological Phenotype of Bovine Astrovirus CH13/NeuroS1 Infection in Neurologically-Diseased Cattle: Towards Assessment of Causality" Viruses 9, no. 1: 12. https://doi.org/10.3390/v9010012
APA StyleSelimovic-Hamza, S., Boujon, C. L., Hilbe, M., Oevermann, A., & Seuberlich, T. (2017). Frequency and Pathological Phenotype of Bovine Astrovirus CH13/NeuroS1 Infection in Neurologically-Diseased Cattle: Towards Assessment of Causality. Viruses, 9(1), 12. https://doi.org/10.3390/v9010012






