The Emerging Profile of Cross-Resistance among the Nonnucleoside HIV-1 Reverse Transcriptase Inhibitors
Abstract
:1. Reverse Transcription
2. Reverse Transcriptase Inhibitors
3. Mechanism of Action of NNRTIs
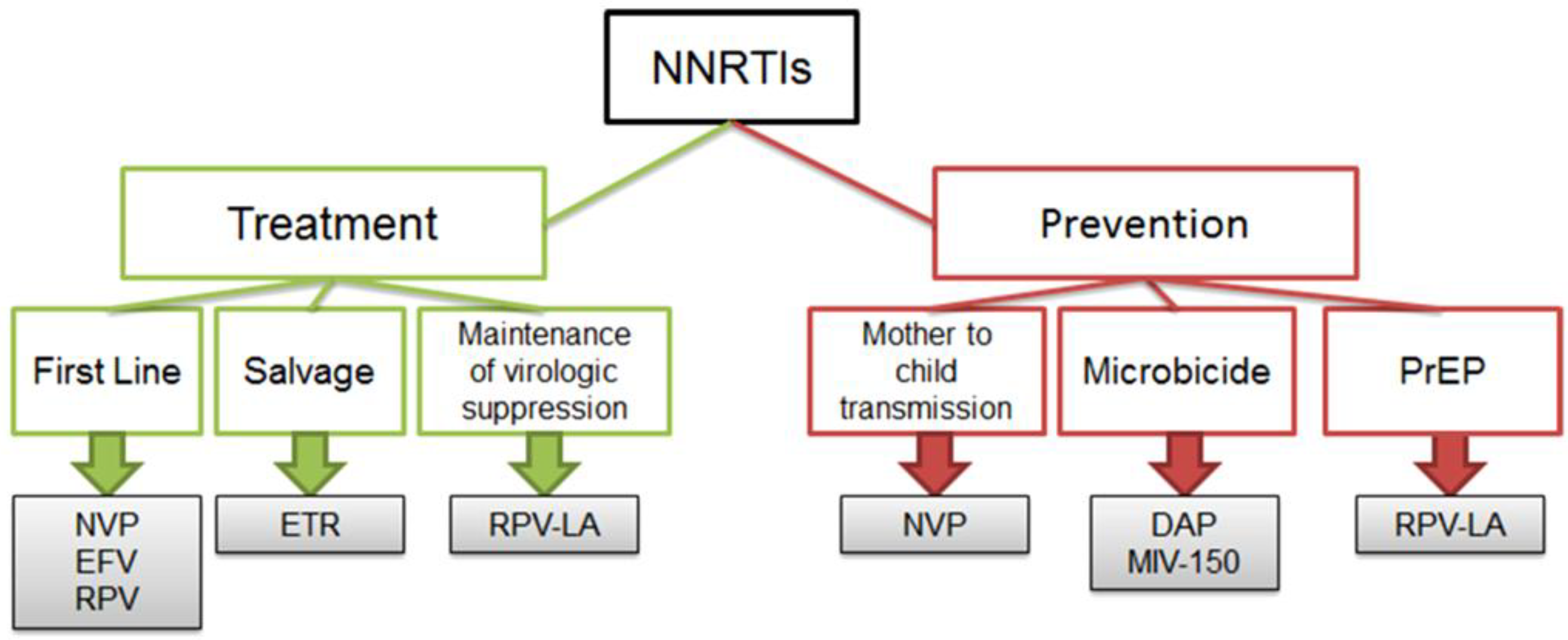
4. Clinical Use of NNRTIs
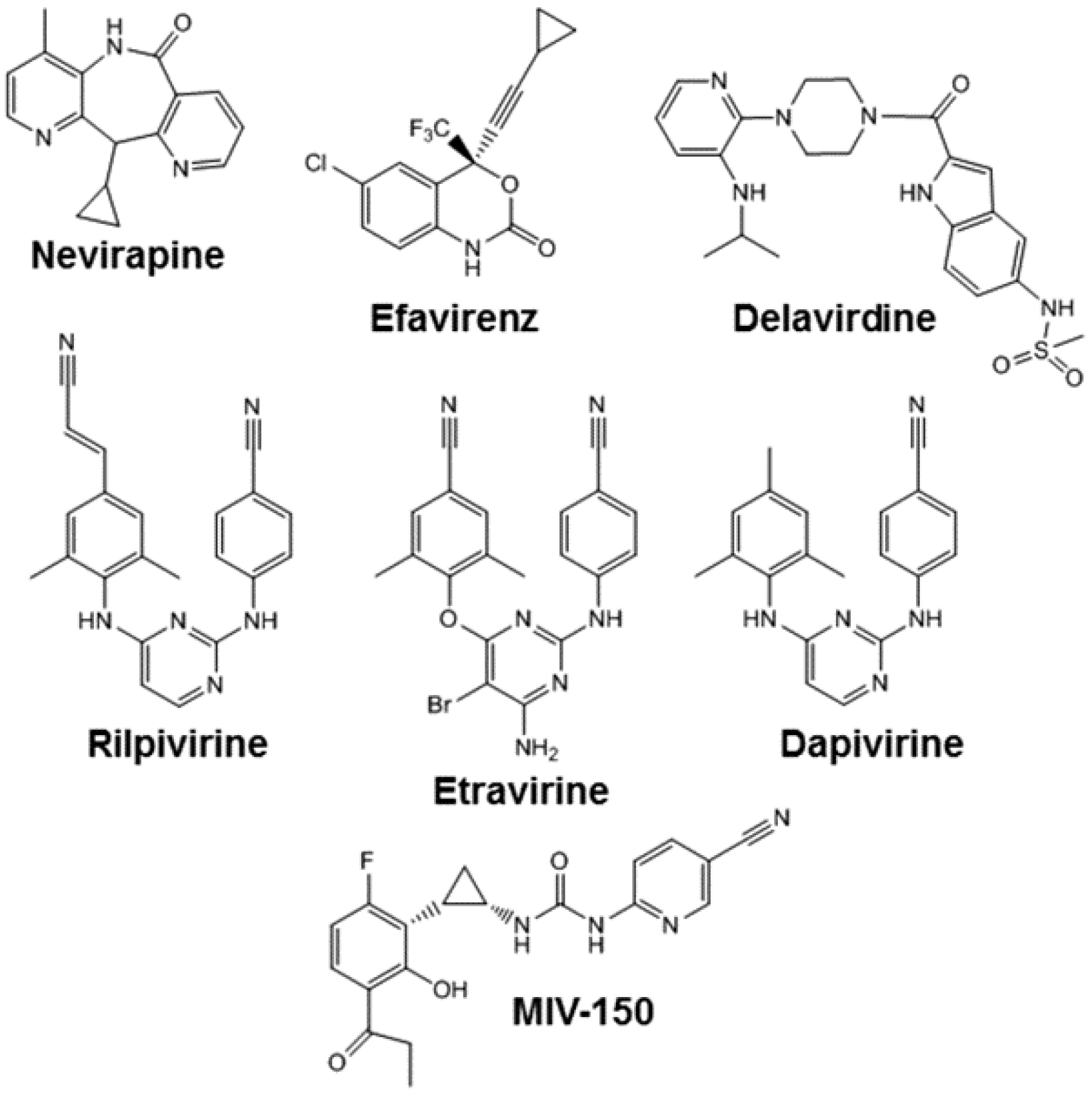
5. Nevirapine
6. Efavirenz
7. Etravirine
8. Rilpivirine
9. Long Acting Rilpivirine
10. Dapivirine
11. MIV-150
12. Potential for Cross-Resistance between NNRTIs Used for Prevention and Treatment of HIV-1 Infection
- (i)
- Nearly all of the NNRTI resistance mutations are within or adjacent to the NNRTI-binding pocket. Indeed there is no evidence that any one mutation only confers resistance to a single agent: most NNRTI-resistance mutations reduce susceptibility to two or more NNRTIs (Table 1).
- (ii)
- The genetic barrier to NNRTI resistance is low. Typically, EFV, NVP and RPV require only a single mutation to reduce clinical efficacy. ETR requires two mutations, but in certain circumstances (i.e., Y181I/V) a single mutation may be sufficient.
| NNRTI | V 90 | L 100 | K 101 | K 103 | V 106 | V 108 | E 138 | V 179 | Y 181 | Y 188 | G 190 | H 221 | P 225 | F 227 | M 230 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NVP | I | I | EP | N | A/I/M | I | D/E/L | C/I/V | L/C/H | A/S/E | H | L/C | L | ||
| EFV | I | I | EP | N | A/I/M | I | D/E/L | C/I/V | L/C/H | A/S/E | H | L/C | L | ||
| ETR | I | I | EP | I | A/G/K/Q | D/E/F/I/T/L | C/I/V | L | A/S/E | C | L | ||||
| RPV | I | I | EP | I | A/G/K/Q | D/E/F/I/T/L | C/I/V | L | A/S/E | Y | C | L | |||
| DAP | I | I | E | I | K | C | L | ||||||||
| MIV-150 | E | N | I | I | A/K | A/I/L | C/I | H |
Acknowledgments
Conflicts of Interest
References and Notes
- Telesnitsky, A.; Goff, S. Reverse Transcriptase and the Generation of Retroviral DNA in Retroviruses; Coffin, J., Hughes, S., Varmus, H., Eds.; Cold Spring Harbor Laboratory Press: Plainview, NY, USA., 1997; pp. 121–160. [Google Scholar]
- Das, K.; Arnold, E. HIV-1 reverse transcriptase and antiviral drug resistance. Part 1. Curr. Opin. Virol. 2013, 3, 111–118. [Google Scholar] [CrossRef]
- Das, K.; Arnold, E. HIV-1 reverse transcriptase and antiviral drug resistance. Part 2. Curr. Opin. Virol. 2013, 3, 119–128. [Google Scholar] [CrossRef]
- Kohlstaedt, L.A.; Wang, J.; Friedman, J.M.; Rice, P.A.; Steitz, T.A. Crystal structure at 3.5 A resolution of HIV-1 reverse transcriptase complexed with an inhibitor. Science 1992, 26, 1783–1790. [Google Scholar]
- Esnouf, R.; Ren, J.; Ross, C.; Jones, Y.; Stammers, D.; Stuart, D. Mechanism of inhibition of HIV-1 reverse transcriptase by non-nucleoside inhibitors. Nat. Struct. Biol. 1995, 2, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Hsiou, Y.; Ding, J.; Das, K.; Clark, A.D., Jr.; Hughes, S.H.; Arnold, E. Structure of unliganded HIV-1 reverse transcriptase at 2.7 A resolution: Implications of conformational changes for polymerization and inhibition mechanisms. In Structure; Volume 15, 1996; pp. 853–860. [Google Scholar]
- Merluzzi, V.J.; Hargrave, K.D.; Labadia, M.; Grozinger, K.; Skoog, M.; Wu, J.C.; Shih, C.K.; Eckner, K.; Hattox, S.; Adams, J. Inhibition of HIV-1 replication by a nonnucleoside reverse transcriptase inhibitor. Science 1990, 250, 1411–1413. [Google Scholar] [CrossRef] [PubMed]
- Havlir, D.; McLaughlin, M.M.; Richman, D.D. A pilot study to evaluate the development of resistance to nevirapine in asymptomatic human immunodeficiency virus-infected patients with CD4 cell counts of >500/mm3: AIDS Clinical Trials Group Protocol 208. J. Infect. Dis. 1995, 172, 1379–1383. [Google Scholar] [CrossRef] [PubMed]
- Richman, D.D.; Havlir, D.; Corbeil, J.; Looney, D.; Ignacio, C.; Spector, S.A.; Sullivan, J.; Cheeseman, S.; Barringer, K.; Pauletti, D. Nevirapine resistance mutations of human immunodeficiency virus type 1 selected during therapy. J. Virol. 1994, 68, 1660–1666. [Google Scholar] [PubMed]
- Larder, B.A. 3'-Azido-3'-deoxythymidine resistance suppressed by a mutation conferring human immunodeficiency virus type 1 resistance to nonnucleoside reverse transcriptase inhibitors. Antimicrob. Agents Chemother. 1992, 36, 2664–2669. [Google Scholar] [CrossRef] [PubMed]
- Selmi, B.; Deval, J.; Alvarez, K.; Boretto, J.; Sarfati, S.; Guerreiro, C.; Canard, B. The Y181C substitution in 3'-azido-3'-deoxythymidine-resistant human immunodeficiency virus, type 1, reverse transcriptase suppresses the ATP-mediated repair of the 3'-azido-3'-deoxythymidine 5'-monophosphate-terminated primer. J. Biol. Chem. 2003, 278, 40464–40472. [Google Scholar] [CrossRef] [PubMed]
- Brenner, B.; Turner, D.; Oliveira, M.; Moisi, D.; Detorio, M.; Carobene, M.; Marlink, R.G.; Schapiro, J.; Roger, M.; Wainberg, M.A. A V106M mutation in HIV-1 clade C viruses exposed to efavirenz confers cross-resistance to non-nucleoside reverse transcriptase inhibitors. AIDS 2003, 17, F1–F5. [Google Scholar] [CrossRef] [PubMed]
- Yap, S.H.; Sheen, C.W.; Fahey, J.; Zanin, M.; Tyssen, D.; Lima, V.D.; Wynhoven, B.; Kuiper, M.; Sluis-Cremer, N.; Harrigan, P.R.; et al. N348I in the connection domain of HIV-1 reverse transcriptase confers zidovudine and nevirapine resistance. PLoS Med. 2007, 4, e335. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.H.; Koontz, D.L.; Wallis, C.L.; Shutt, K.A.; Sanne, I.; Wood, R.; McIntyre, J.A.; Stevens, W.S.; Sluis-Cremer, N.; Mellors, J.W. CIPRA-SA Project 1 Study Team. Frequent emergence of N348I in HIV-1 subtype C reverse transcriptase with failure of initial therapy reduces susceptibility to reverse-transcriptase inhibitors. Clin. Infect. Dis. 2012, 55, 737–745. [Google Scholar]
- Ton, Q.; Frenkel, L. HIV drug resistance in mothers and infants following use of antiretrovirals to prevent mother-to-child transmission. Curr. HIV Res. 2013, 11, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Young, S.D.; Britcher, S.F.; Tran, L.O.; Payne, L.S.; Lumma, W.C.; Lyle, T.A.; Huff, J.R.; Anderson, P.S.; Olsen, D.B.; Carroll, S.S. L-743,726 (DMP-266): A novel, highly potent nonnucleoside inhibitor of the human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 1995, 39, 2602–2605. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.; Staszewski, S.; Morales-Ramirez, J.O.; Barros, C.; Ferrer, E.; Milazzo, F. Sccessful virologic suppression with efavirenz in HIV-infected patients with low baseline CD4 cell counts: Post hoc results from study 006. In Proceedings of the Abstracts of the Tenth European Congress of Clinical Microbiology and Infectious Diseases, Stockholm, Sweden, 28–31 May 2000. Abstract 3–349.
- Squires, K.; Lazzarin, A.; Gatell, J.M.; Powderly, W.G.; Pokrovskiy, V.; Delfraissy, J.F.; Jemsek, J.; Rivero, A.; Rozenbaum, W.; Schrader, S.; et al. Comparison of once-daily atazanavir with efavirenz, each in combination with fixed-dose zidovudine and lamivudine, as initial therapy for patients infected with HIV. J. Acquir. Immune Defic. Syndr. 2004, 36, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Yeni, P.; Cooper, D.A.; Aboulker, J.P.; Babiker, A.G.; Carey, D.; Darbyshire, J.H.; Floridia, M.; Girard, P.M.; Goodall, R.L.; Hooker, M.H.; et al. Virological and immunological outcomes at 3 years after starting antiretroviral therapy with regimens containing non-nucleoside reverse transcriptase inhibitor, protease inhibitor, or both in INITIO: Open-label randomised trial. Lancet 2006, 368, 287–298. [Google Scholar]
- Van Leth, F.; Phanuphak, P.; Ruxrungtham, K.; Baraldi, E.; Miller, S.; Gazzard, B.; Cahn, P.; Lalloo, U.G.; van der Westhuizen, I.P.; Malan, D.R.; et al. Comparison of first-line antiretroviral therapy with regimens including nevirapine, efavirenz, or both drugs, plus stavudine and lamivudine: A randomised open-label trial, the 2NN Study. Lancet 2004, 363, 1253–1263. [Google Scholar]
- Gulick, R.M.; Ribaudo, H.J.; Shikuma, C.M.; Lustgarten, S.; Squires, K.E.; Meyer, W.A., 3rd; Acosta, E.P.; Schackman, B.R.; Pilcher, C.D.; Murphy, R.L.; et al. Triple-nucleoside regimens versus efavirenz-containing regimens for the initial treatment of HIV-1 infection. N. Engl. J. Med. 2004, 350, 1850–1861. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.A.; Heera, J.; Goodrich, J.; Tawadrous, M.; Saag, M.; Dejesus, E.; Clumeck, N.; Walmsley, S.; Ting, N.; Coakley, E.; et al. Maraviroc versus efavirenz, both in combination with zidovudine-lamivudine, for the treatment of antiretroviral-naive subjects with CCR5-tropic HIV-1 infection. J. Infect. Dis. 2010, 201, 803–813. [Google Scholar]
- Markowitz, M.; Nguyen, B.Y.; Gotuzzo, E.; Mendo, F.; Ratanasuwan, W.; Kovacs, C.; Prada, G.; Morales-Ramirez, J.O.; Crumpacker, C.S.; Isaacs, R.D.; et al. Rapid and durable antiretroviral effect of the HIV-1 integrase inhibitor raltegravir as part of combination therapy in treatment-naive patients with HIV-1 infection: Results of a 48-week controlled study. J. Acquir. Immune Defic. Syndr. 2007, 46, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Lennox, J.L.; DeJesus, E.; Lazzarin, A.; Pollard, R.B.; Madruga, J.V.; Berger, D.S.; Zhao, J.; Xu, X.; Williams-Diaz, A.; Rodgers, A.J.; et al. STARTMRK investigators. Safety and efficacy of raltegravir-based versus efavirenz-based combination therapy in treatment-naive patients with HIV-1 infection: A multicentre, double-blind randomised controlled trial. Lancet 2009, 374, 796–806. [Google Scholar]
- Lennox, J.L.; Dejesus, E.; Berger, D.S.; Lazzarin, A.; Pollard, R.B.; Ramalho Madruga, J.V.; Zhao, J.; Wan, H.; Gilbert, C.L.; Teppler, H.; et al. STARTMRK Investigators. Raltegravir versus Efavirenz regimens in treatment-naive HIV-1-infected patients: 96-week efficacy, durability, subgroup, safety, and metabolic analyses. J. Acquir. Immune Defic. Syndr. 2010, 55, 39–48. [Google Scholar]
- Rockstroh, J.K.; Lennox, J.L.; Dejesus, E.; Saag, M.S.; Lazzarin, A.; Wan, H.; Walker, M.L.; Xu, X.; Zhao, J.; Teppler, H.; et al. STARTMRK Investigators. Long-term treatment with raltegravir or efavirenz combined with tenofovir/emtricitabine for treatment-naive human immunodeficiency virus-1-infected patients: 156-week results from STARTMRK. Clin. Infect. Dis. 2011, 53, 807–816. [Google Scholar]
- Rockstroh, J.K.; DeJesus, E.; Lennox, J.L.; Yazdanpanah, Y.; Saag, M.S.; Wan, H.; Rodgers, A.J.; Walker, M.L.; Miller, M.; DiNubile, M.J.; et al. STARTMRK Investigators. Durable efficacy and safety of raltegravir versus efavirenz when combined with tenofovir/emtricitabine in treatment-naive HIV-1-infected patients: Final 5-year results from STARTMRK. J. Acquir. Immune Defic. Syndr. 2013, 63, 77–85. [Google Scholar]
- Walmsley, S.L.; Antela, A.; Clumeck, N.; Duiculescu, D.; Eberhard, A.; Gutiérrez, F.; Hocqueloux, L.; Maggiolo, F.; Sandkovsky, U.; Granier, C.; et al. SINGLE Investigators. Dolutegravir plus abacavir-lamivudine for the treatment of HIV-1 infection. N. Engl. J. Med. 2013, 369, 1807–1818. [Google Scholar]
- Vingerhoets, J.; Azijn, H.; Fransen, E.; De Baere, I.; Smeulders, L.; Jochmans, D.; Andries, K.; Pauwels, R.; de Béthune, M.P. TMC125 displays a high genetic barrier to the development of resistance: Evidence from in vitro selection experiments. J. Virol. 2005, 79, 12773–12782. [Google Scholar] [CrossRef] [PubMed]
- Madruga, J.V.; Cahn, P.; Grinsztejn, B.; Haubrich, R.; Lalezari, J.; Mills, A.; Pialoux, G.; Wilkin, T.; Peeters, M.; Vingerhoets, J.; et al. Efficacy and safety of TMC125 (etravirine) in treatment-experienced HIV-1-infected patients in DUET-1: 24-week results from a randomised,double-blind, placebo-controlled trial. Lancet 2007, 370, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Lazzarin, A.; Campbell, T.; Clotet, B.; Johnson, M.; Katlama, C.; Moll, A.; Towner, W.; Trottier, B.; Peeters, M.; Vingerhoets, J.; et al. Efficacy and safety of TMC125 (etravirine) in treatment-experienced HIV-1-infected patients in DUET-2: 24-week results from a randomised, double-blind, placebo-controlled trial. Lancet 2007, 370, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Tambuyzer, L.; Vingerhoets, J.; Azijn, H.; Daems, B.; Nijs, S.; de Béthune, M.P.; Picchio, G. Characterization of genotypic and phenotypic changes in HIV-1-infected patients with virologic failure on an etravirine-containing regimen in the DUET-1 and DUET-2 clinical studies. AIDS Res. Hum. Retroviruses 2010, 26, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Vingerhoets, J.; Tambuyzer, L.; Azijn, H.; Hoogstoel, A.; Nijs, S.; Peeters, M.; de Béthune, M.P.; De Smedt, G.; Woodfall, B.; Picchio, G. Resistance profile of etravirine: Combined analysis of baseline genotypic and phenotypic data from the randomized, controlled Phase III clinical studies. AIDS 2010, 24, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Ruxrungtham, K.; Pedro, R.J.; Latiff, G.H.; Conradie, F.; Domingo, P.; Lupo, S.; Pumpradit, W.; Vingerhoets, J.H.; Peeters, M.; Peeters, I.; et al. TMC125-C227 study group. Impact of reverse transcriptase resistance on the efficacy of TMC125 (etravirine) with two nucleoside reverse transcriptase inhibitors in protease inhibitor-naïve, nonnucleoside reverse transcriptase inhibitor-experienced patients: Study TMC125-C227. HIV Med. 2008, 9, 883–896. [Google Scholar]
- Azijn, H.; Tirry, I.; Vingerhoets, J.; de Béthune, M.P.; Kraus, G.; Boven, K.; Jochmans, D.; Van Craenenbroeck, E.; Picchio, G.; Rimsky, L.T. TMC278,a next-generation nonnucleoside reverse transcriptase inhibitor (NNRTI),active against wild-type and NNRTI-resistant HIV-1. Antimicrob. Agents Chemother. 2010, 54, 718–727. [Google Scholar] [CrossRef] [PubMed]
- Molina, J.M.; Cahn, P.; Grinsztejn, B.; Lazzarin, A.; Mills, A.; Saag, M.; Supparatpinyo, K.; Walmsley, S.; Crauwels, H.; Rimsky, L.T.; et al. ECHO study group. Rilpivirine versus efavirenz with tenofovir and emtricitabine in treatment-naïve adults infected with HIV-1 (ECHO): A phase 3 randomised double-blind active-controlled trial. Lancet 2011, 378, 238–246. [Google Scholar]
- Cohen, C.J.; Andrade-Villanueva, J.; Clotet, B.; Fourie, J.; Johnson, M.A.; Ruxrungtham, K.; Wu, H.; Zorrilla, C.; Crauwels, H.; Rimsky, L.T.; et al. THRIVE study group. Rilpivirine versus efavirenz with two background nucleoside or nucleotide reverse transcriptase inhibitors in treatment-naïve adults infected with HIV-1 (THRIVE): A Phase 3, randomized, non-inferiority trial. Lancet 2011, 378, 229–237. [Google Scholar]
- Xu, H.T.; Asahchop, E.L.; Oliveira, M.; Quashie, P.K.; Quan, Y.; Brenner, B.G.; Wainberg, M.A. Compensation by the E138K mutation in HIV-1 reverse transcriptase for deficits in viral replication capacity and enzyme processivity associated with the M184I/V mutations. J. Virol. 2011, 85, 11300–11308. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Kuritzkes, D.R. Interaction of reverse transcriptase (RT) mutations conferring resistance to lamivudine and etravirine: Effects on fitness and RT activity of human immunodeficiency virus type 1. J. Virol. 2011, 85, 11309–11314. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.; Babaoglu, K.; Lansdon, E.B.; Rimsky, L.; van Eygen, V.; Picchio, G.; Svarovskaia, E.; Miller, M.D.; White, K.L. The HIV-1 reverse transcriptase M184I mutation enhances the E138K-associated resistance to rilpivirine and decreases viral fitness. J. Acquir. Immune Defic. Syndr. 2012, 59, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Sluis-Cremer, N.; Jordan, M.R.; Huber, K.; Wallis, C.L.; Bertagnolio, S.; Mellors, J.W.; Parkin, N.T.; Harrigan, P.R. E138A in HIV-1 reverse transcriptase is more common in subtype C than B: Implications for rilpivirine use in resource-limited settings. Antivir. Res. 2014, 107, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Siegel, M.O.; Swierzbinski, M.; Kan, V.L.; Parenti, D.M. Baseline E138 reverse transcriptase resistance-associated mutations in antiretroviral-naive HIV-infected patients. AIDS 2012, 26, 1181–1182. [Google Scholar] [CrossRef] [PubMed]
- Baert, L.; van’t Klooster, G.; Dries, W.; François, M.; Wouters, A.; Basstanie, E.; Iterbeke, K.; Stappers, F.; Stevens, P.; Schueller, L.; et al. Development of a long-acting injectable formulation with nanoparticles of rilpivirine (TMC278) for HIV treatment. Eur. J. Pharm. Biopharm. 2009, 72, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Van’t Klooster, G.; Hoeben, E.; Borghys, H.; Looszova, A.; Bouche, M.P.; van Velsen, F.; Baert, L. Pharmacokinetics and disposition of rilpivirine (TMC278) nanosuspension as a long-acting injectable antiretroviral formulation. Antimicrob. Agents Chemother. 2010, 54, 2042–2050. [Google Scholar] [CrossRef] [PubMed]
- Verloes, R.; van’t Klooster, G.; Baert, L.; van Velsen, F.; Bouche, M.P.; Spittaels, K.; Leempoels, J.; Williams, P.; Kraus, G.; Wigerinck, P. TMC278 Long Acting—A Parenteral Nanosuspension Formulation that Provides Sustained Clinically Relevant Plasma Concentrations in HIV-Negative Volunteers. In Proceedings of the 17th International AIDS Conference, Mexico City, Mexico, 23 November 2008.
- Jackson, A.; Else, L.; Tjia, J.; Seymour, N.; Stafford, M.; Back, D.; Gazzard, B.; Boffito, M. Rilpivirine-LA Formulation: Pharmacokinetics in Plasma, Genital Tract in HIV-Females and Rectum in Males [Abstract no35]. In Proceedings of the 19th Conference on Retroviruses and Opportunistic Infections, Seattle, WA, USA,, 5–8 March 2012.
- Fletcher, P.; Harman, S.; Azijn, H.; Armanasco, N.; Manlow, P.; Perumal, D.; de Bethune, M.P.; Nuttall, J.; Romano, J.; Shattock, R. Inhibition of human immunodeficiency virus type 1 infection by the candidate microbicide dapivirine, a nonnucleoside reverse transcriptase inhibitor. Antimicrob. Agents Chemother. 2009, 53, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Schader, S.M.; Oliveira, M.; Ibanescu, R.I.; Moisi, D.; Colby-Germinario, S.P.; Wainberg, M.A. In vitro resistance profile of the candidate HIV-1 microbicide drug dapivirine. Antimicrob. Agents Chemother. 2012, 56, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Kenney, J.; Aravantinou, M.; Singer, R.; Hsu, M.; Rodriguez, A.; Kizima, L.; Abraham, C.J.; Menon, R.; Seidor, S.; Chudolij, A.; et al. An Antiretroviral/Zinc combination gel provides 24 h of complete protection against vaginal SHIV infection in macaques. PLoS One 2011, 6, e15835. [Google Scholar] [CrossRef] [PubMed]
- Singer, R.; Mawson, P.; Derby, N.; Rodriguez, A.; Kizima, L.; Menon, R.; Goldman, D.; Kenney, J.; Aravantinou, M.; Seidor, S.; et al. An Intravaginal Ring That Releases the NNRTI MIV-150 Reduces SHIV Transmission in Macaques. Sci. Transl. Med. 2012, 4, 150ra123. [Google Scholar] [CrossRef] [PubMed]
- Hsu, M.; Keele, B.F.; Aravantinou, M.; Krawczyk, N.; Seidor, S.; Abraham, C.J.; Zhang, S.; Rodriguez, A.; Kizima, L.; Derby, N.; et al. Exposure to MIV-150 from a high-dose intravaginal ring results in limited emergence of drug resistance mutations in SHIV-RT infected rhesus macaques. PLoS One 2014, 9, e89300. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sluis-Cremer, N. The Emerging Profile of Cross-Resistance among the Nonnucleoside HIV-1 Reverse Transcriptase Inhibitors. Viruses 2014, 6, 2960-2973. https://doi.org/10.3390/v6082960
Sluis-Cremer N. The Emerging Profile of Cross-Resistance among the Nonnucleoside HIV-1 Reverse Transcriptase Inhibitors. Viruses. 2014; 6(8):2960-2973. https://doi.org/10.3390/v6082960
Chicago/Turabian StyleSluis-Cremer, Nicolas. 2014. "The Emerging Profile of Cross-Resistance among the Nonnucleoside HIV-1 Reverse Transcriptase Inhibitors" Viruses 6, no. 8: 2960-2973. https://doi.org/10.3390/v6082960
APA StyleSluis-Cremer, N. (2014). The Emerging Profile of Cross-Resistance among the Nonnucleoside HIV-1 Reverse Transcriptase Inhibitors. Viruses, 6(8), 2960-2973. https://doi.org/10.3390/v6082960




