Rapid Enzyme-Linked Immunosorbent Assay for the Detection of Hantavirus-Specific Antibodies in Divergent Small Mammals
Abstract
:1. Introduction
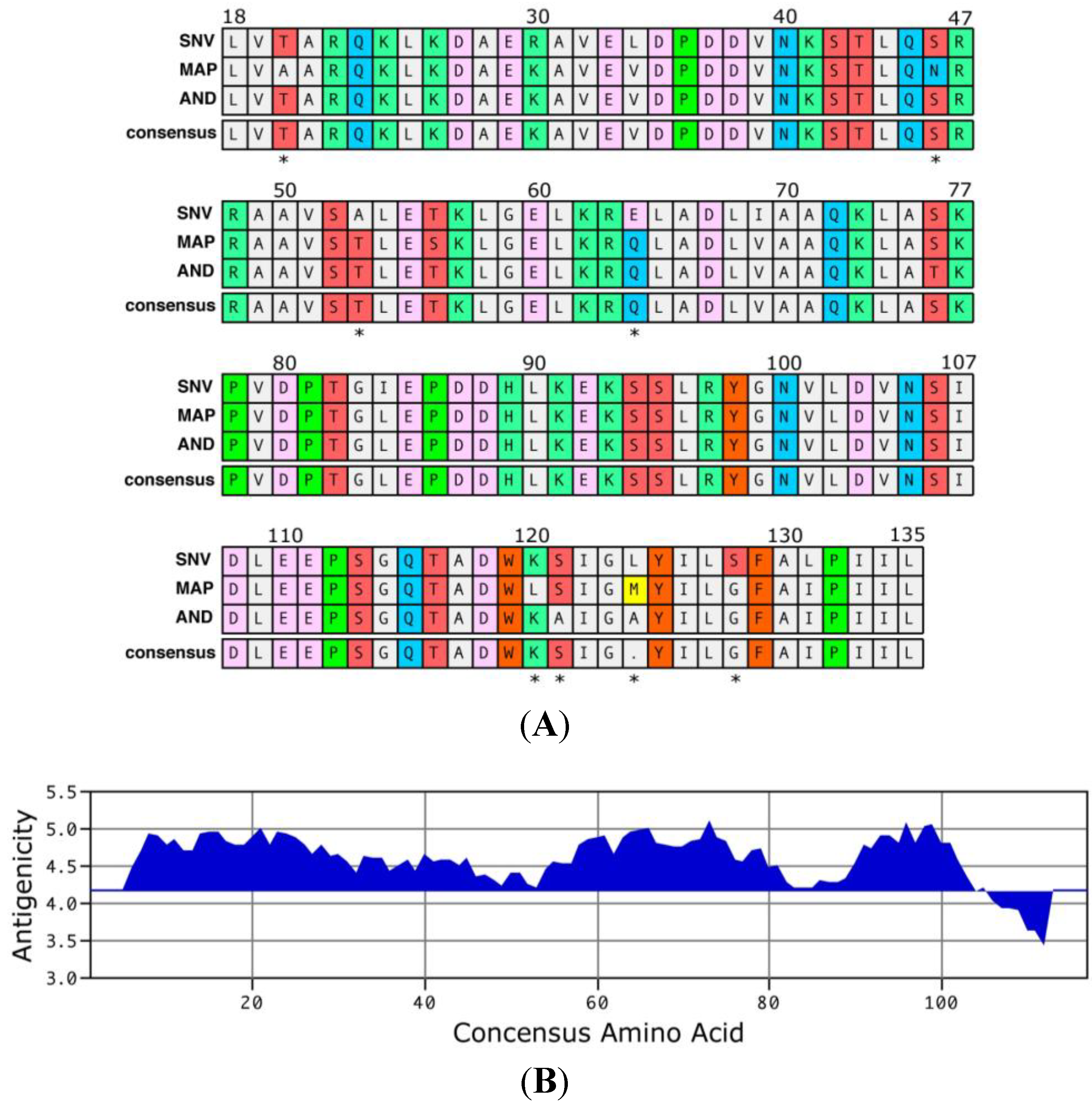
2. Results

| Locality | Species | Number of captures | Number of seropositives (ELISA) |
|---|---|---|---|
| Villarica, Region IX 39°25'S; 71°45'W | O. longicaudatus A. olivaceus A. longipilis | 6 2 4 0 | 3 0 0 |
| El Sauce, Chimbarongo, Region VI 70°59'W; 34°48'S | O. longicaudatus A. olivaceus | 2 1 | 1 0 |
| Rupanquito, Puerto Octay, Region X 40°46'S; 72°46'W | O. longicaudatus A. olivaceus A. longipilis R. rattus R. norvrgicus M. musculus | 28 13 3 2 1 1 | 2 0 0 0 0 0 |
| Boroa, Toltén, Region IX 39°17'S; 73°04'W | O. longicaudatus A. olivaceus R. rattus R. norvrgicus | 19 1 11 1 | 0 0 0 0 |
| Pichi-Juan, Pto. Varas, Region X 41°14'S; 72°43'W | O. longicaudatus A. olivaceus A. longipilis L. micropus | 32 11 2 1 | 0 0 0 0 |
| Miraflores, Lanco, Region XIV 39°32'S; 72°37'W | O. longicaudatus A. olivaceus A. longipilis | 59 7 5 | 7 0 1 |
| Llacolén, Contulmo, Region VII 37°54'S; 73°16'W | O. longicaudatus A. olivaceus A. longipilis D. gliroides | 22 7 4 1 | 0 0 0 0 |
| Accession | Species | Sex | Age | Titer |
|---|---|---|---|---|
| 18 | O. longicaudatus | Female | Adult | 1600 |
| 29 | O. longicaudatus | Male | Adult | 800 |
| 31 | O. longicaudatus | Male | Adult | 800 |
| 352 | O. longicaudatus | Female | Adult | 400 |
| 130 | O. longicaudatus | Male | Adult | 6400 |
| 131 | O. longicaudatus | Male | Adult | 6400 |
| 560 | O. longicaudatus | Female | Adult | 400 |
| 569 | A. longipilis | Male | Adult | 3200 |
| 577 | O. longicaudatus | Male | Adult | 400 |
| 584 | O. longicaudatus | Male | Adult | 400 |
| 589 | O. longicaudatus | Male | Adult | 800 |
| 600 | O. longicaudatus | Male | Adult | 400 |
| 601 | O. longicaudatus | Female | Adult | 800 |
| 604 | O. longicaudatus | Male | Adult | 200 |
3. Discussion
4. Experimental Section
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Hjelle, B.; Torres-Pérez, F. Hantaviruses in the Americas and their role as emerging pathogens. Viruses 2010, 2, 2559–2586. [Google Scholar] [CrossRef]
- Schmaljohn, C.; Hjelle, B. Hantaviruses: A global disease problem. Emerg. Infect. Dis. 1997, 3, 95–104. [Google Scholar] [CrossRef]
- Botten, J.; Mirowsky, K.; Ye, C.Y.; Gottlieb, K.; Saavedra, M.; Ponce, L.; Hjelle, B. Shedding and intracage transmission of Sin Nombre hantavirus in the deer mouse (Peromyscus maniculatus) model. J. Virol. 2002, 76, 7587–7594. [Google Scholar] [CrossRef]
- Padula, P.; Figueroa, R.; Navarrete, M.; Pizarro, E.; Cadiz, R.; Bellomo, C.; Jofre, C.; Zaror, L.; Rodriguez, E.; Murua, R. Transmission study of Andes hantavirus infection in wild sigmodontine rodents. J. Virol. 2004, 78, 11972–11979. [Google Scholar] [CrossRef]
- Kang, H.J.; Bennett, S.N.; Hope, A.G.; Cook, J.A.; Yanagihara, R. Shared ancestry between a newfound mole-borne hantavirus and hantaviruses harbored by cricetid rodents. J. Virol. 2011, 85, 7496–7503. [Google Scholar] [CrossRef]
- Padula, P.J.; Colavecchia, S.B.; Martinez, V.P.; Della Valle, M.O.G.; Edelstein, A.; Miguel, S.D.L.; Russi, J.; Riquelme, J.M.; Colucci, N.; Almiron, M.; et al. Genetic diversity, distribution, and serological features of hantavirus infection in five countries in South America. J. Clin. Microbiol. 2000, 38, 3029–3035. [Google Scholar]
- Maes, P.; Klempa, B.; Clement, J.; Matthijnssens, J.; Gajdusek, D.C.; Kruger, D.H.; van Ranst, M. A proposal for new criteria for the classification of hantaviruses, based on S and M segment protein sequences. Infect. Genet. Evol. 2009, 9, 813–820. [Google Scholar] [CrossRef]
- Ferres, M.; Vial, P.; Marco, C.; Yanez, L.; Godoy, P.; Castillo, C.; Hjelle, B.; Delgado, I.; Lee, S.J.; Mertz, G.J. Prospective evaluation of household contacts of persons with hantavirus cardiopulmonary syndrome in Chile. J. Infect. Dis. 2007, 195, 1563–1571. [Google Scholar] [CrossRef]
- Martinez, V.P.; Bellomo, C.; San Juan, J.; Pinna, D.; Forlenza, R.; Elder, M.; Padula, P.J. Person-to-person transmission of Andes virus. Emerg. Infect. Dis. 2005, 11, 1848–1853. [Google Scholar] [CrossRef]
- Ministerio de Salud, Gobierno de Chile. Available online: http://epi.minsal.cl/ (accessed on 1 October 2013).
- Medina, R.A.; Torres-Pérez, F.; Galeno, H.; Navarrete, M.; Vial, P.A.; Palma, R.E.; Ferres, M.; Cook, J.A.; Hjelle, B. Ecology, genetic diversity, and phylogeographic structure of Andes virus in humans and rodents in Chile. J. Virol. 2009, 83, 2446–2459. [Google Scholar] [CrossRef]
- Palma, R.E.; Boric-Bargetto, D.; Torres-Pérez, F.; Hernández, C.E.; Yates, T.L. Glaciation Effects on the Phylogeographic Structure of Oligoryzomys longicaudatus (Rodentia: Sigmodontinae) in the Southern Andes. PLoS One 2012, 7, e32206. [Google Scholar]
- Belmar-Lucero, S.; Godoy, P.; Ferres, M.; Vial, P.; Palma, R.E. Range expansion of Oligoryzomys longicaudatus (Rodentia, Sigmodontinae) in Patagonian Chile, and first record of Hantavirus in the region. Rev. Chil. Hist. Nat. 2009, 82, 265–275. [Google Scholar]
- Carbajo, A.E.; Pardinas, U.F.J. Spatial distribution model of a Hantavirus reservoir, the long-tailed colilargo. J. Mammal. 2007, 88, 1555–1568. [Google Scholar] [CrossRef]
- Torres-Pérez, F.; Navarrete-Droguett, J.; Aldunate, R.; Yates, T.L.; Mertz, G.J.; Vial, P.A.; Ferres, M.; Marquet, P.A.; Palma, R.E. Peridomestic small mammals associated with confirmed cases of human hantavirus disease on southcentral Chile. Am. J. Trop. Med. Hyg. 2004, 70, 305–309. [Google Scholar]
- Toro, J.; Vega, J.D.; Khan, A.S.; Mills, J.N.; Padula, P.; Terry, W.; Yadon, Z.; Valderrama, R.; Ellis, B.A.; Pavletic, C.; et al. An outbreak of hantavirus pulmonary syndrome, Chile, 1997. Emerg. Infect. Dis. 1998, 4, 687–694. [Google Scholar]
- Yee, J.; Wortman, I.A.; Nofchissey, R.A.; Goade, D.; Bennett, S.G.; Webb, J.P.; Irwin, W.; Hjelle, B. Rapid and simple method for screening wild rodents for antibodies to Sin Nombre hantavirus. J. Wildl. Dis. 2003, 39, 271–277. [Google Scholar]
- Padula, P.J.; Rossi, C.M.; della Valle, M.O.; Martinez, P.V.; Colavecchia, S.B.; Edelstein, A.; Miguel, S.D.L.; Rabinovich, R.D.; Segura, E.L. Development and evaluation of a solid-phase enzyme immunoassay based on Andes hantavirus recombinant nucleoprotein. J. Med. Microbiol. 2000, 49, 149–155. [Google Scholar]
- Ortiz, J.C.; Venegas, W.; Sandoval, J.A.; Chandia, P.; Torres-Pérez, F. Hantavirus in rodents of the VIII Region of Chile. Rev. Chil. Hist. Nat. 2004, 77, 251–256. [Google Scholar]
- Piudo, L.; Monteverde, M.J.; Walker, R.S.; Douglass, R.J. Características de Oligoryzomys longicaudatus asociadas a la presencia del virus Andes (Hantavirus). Rev. Chil. Infectol. 2012, 29, 200–206. [Google Scholar] [CrossRef]
- Schountz, T.; Calisher, C.H.; Richens, T.R.; Rich, A.A.; Doty, J.B.; Hughes, M.T.; Beaty, B.J. Rapid field immunoassay for detecting antibody to Sin Nombre virus in deer mice. Emerg. Infect. Dis. 2007, 13, 1604–1607. [Google Scholar] [CrossRef]
- Ramsden, C.; Holmes, E.C.; Charleston, M.A. Hantavirus Evolution in Relation to its Rodent and Insectivore Hosts: No Evidence for Co-divergence. Mol. Biol. Evol. 2009, 26, 143–153. [Google Scholar]
- Londoño, A.F.; Díaz, F.J.; Agudelo-Flórez, P.; Levis, S.; Rodas, J.D. Genetic evidence of hantavirus infections in wild rodents from Northwestern Colombia. Vector Borne Zoonotic. Dis. 2011, 11, 701–708. [Google Scholar] [CrossRef]
- Schountz, T.; Colorado State University, Fort Collins, CO, USA. Unpublished work. 2013.
- Machado, A.M.; de Souza, W.M.; de Padua, M.; da Silva Rodrigues Machado, A.R.; Figueiredo, L.T. Development of a one-step SYBR Green I real-time RT-PCR assay for the detection and quantitation of Araraquara and Rio Mamore hantavirus. Viruses 2013, 5, 2272–2281. [Google Scholar] [CrossRef]
- Machado, A.M.; de Figueiredo, G.G.; Sabino dos Santos Jr, G.; Figueiredo, L.T.M. Laboratory diagnosis of human hantavirus infection: novel insights and future potential. Fut. Virol. 2009, 4, 383–389. [Google Scholar] [CrossRef]
- Navarrete, M.; Barrera, C.; Zaror, L.; Otth, C. Rapid immunochromatographic test for hantavirus Andes contrasted with capture-IgM ELISA for detection of andes-specific IgM antibodies. J. Med. Virol. 2007, 79, 41–44. [Google Scholar] [CrossRef]
- Pierce (Technical Resource) Binding characteristics of Protein A, Protein G, Protein A/G and Protein L. Available online: http://fscimage.fishersci.com/cmsassets/downloads/segment/Scientific/pdf/pierce_binding.pdf (accessed on 30 December 2013).
- Torres-Pérez, F.; Palma, R.E.; Hjelle, B.; Ferres, M.; Cook, J.A. Andes virus infections in the rodent reservoir and in humans vary across contrasting landscapes in Chile. Infect. Genet. Evol. 2010, 10, 820–825. [Google Scholar]
- Godoy, P.; Marsac, D.; Stefas, E.; Ferrer, P.; Tischler, N.D.; Pino, K.; Ramdohr, P.; Vial, P.; Valenzuela, P.D.; Ferres, M.; et al. Andes Virus Antigens are shed in urine of patients with acute Hantavirus Cardiopulmonary Syndrome. J. Virol. 2009, 83, 5046–5055. [Google Scholar] [CrossRef]
- Escadafal, C.; Avsic-Zupanc, T.; Vapalahti, O.; Niklasson, B.; Teichmann, A.; Niedrig, M.; Donoso-Mantke, O. Second External Quality Assurance Study for the Serological Diagnosis of Hantaviruses in Europe. PLoS Negl. Trop. Dis. 2012, 6, e1607, doi:1610.1371/journal.pntd.0001607. [Google Scholar]
- Kuenzi, A.J.; Douglass, R.J.; Bond, C.W.; Calisher, C.H.; Mills, J.N. Long-term dynamics of Sin Nombre viral RNA and antibody in deer mice in Montana. J. Wildl. Dis. 2005, 41, 473–481. [Google Scholar] [CrossRef]
- Tischler, N.D.; Rosemblatt, M.; Valenzuela, P.D. Characterization of cross-reactive and serotype-specific epitopes on the nucleocapsid proteins of hantaviruses. Virus Res. 2008, 135, 1–9. [Google Scholar] [CrossRef]
- Mills, J.N.; Yates, T.L.; Childs, J.E.; Parmenter, R.R.; Ksiazek, T.G.; Rollin, P.E.; Peters, C.J. Guidelines for working with rodents potentially infected with hantavirus. J. Mammal. 1995, 76, 716–722. [Google Scholar] [CrossRef]
- Gannon, W.L.; Sikes, R.S. Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J. Mammal. 2007, 88, 809–823. [Google Scholar] [CrossRef]
- Galeno, H.; Mora, J.; Villagra, E.; Fernandez, J.; Hernandez, J.; Mertz, G.J.; Ramirez, E. First human isolate of Hantavirus (Andes virus) in the Americas. Emerg. Infect. Dis. 2002, 8, 657–661. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cautivo, K.; Schountz, T.; Acuña-Retamar, M.; Ferrés, M.; Torres-Pérez, F. Rapid Enzyme-Linked Immunosorbent Assay for the Detection of Hantavirus-Specific Antibodies in Divergent Small Mammals. Viruses 2014, 6, 2028-2037. https://doi.org/10.3390/v6052028
Cautivo K, Schountz T, Acuña-Retamar M, Ferrés M, Torres-Pérez F. Rapid Enzyme-Linked Immunosorbent Assay for the Detection of Hantavirus-Specific Antibodies in Divergent Small Mammals. Viruses. 2014; 6(5):2028-2037. https://doi.org/10.3390/v6052028
Chicago/Turabian StyleCautivo, Karla, Tony Schountz, Mariana Acuña-Retamar, Marcela Ferrés, and Fernando Torres-Pérez. 2014. "Rapid Enzyme-Linked Immunosorbent Assay for the Detection of Hantavirus-Specific Antibodies in Divergent Small Mammals" Viruses 6, no. 5: 2028-2037. https://doi.org/10.3390/v6052028
APA StyleCautivo, K., Schountz, T., Acuña-Retamar, M., Ferrés, M., & Torres-Pérez, F. (2014). Rapid Enzyme-Linked Immunosorbent Assay for the Detection of Hantavirus-Specific Antibodies in Divergent Small Mammals. Viruses, 6(5), 2028-2037. https://doi.org/10.3390/v6052028





