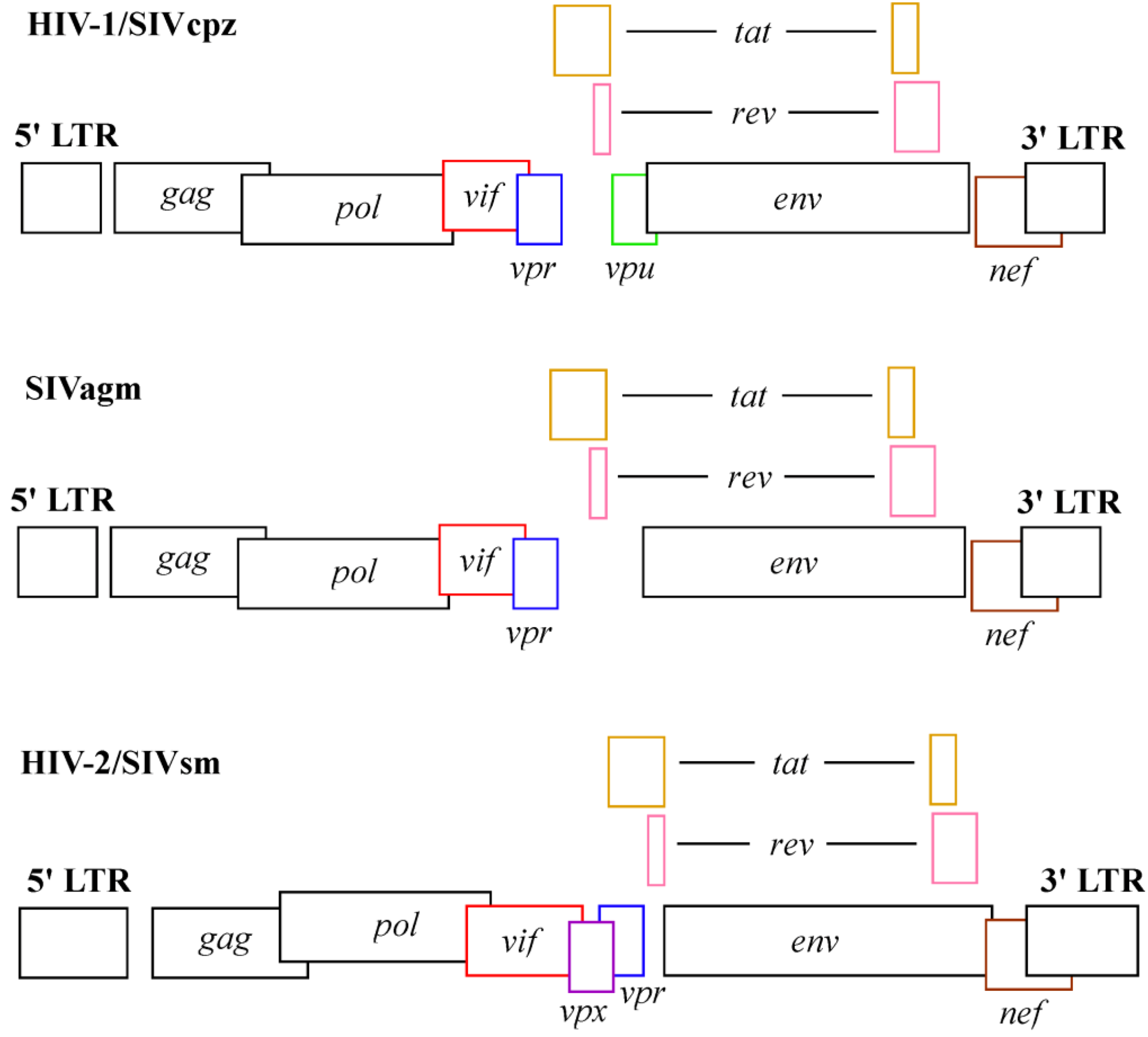
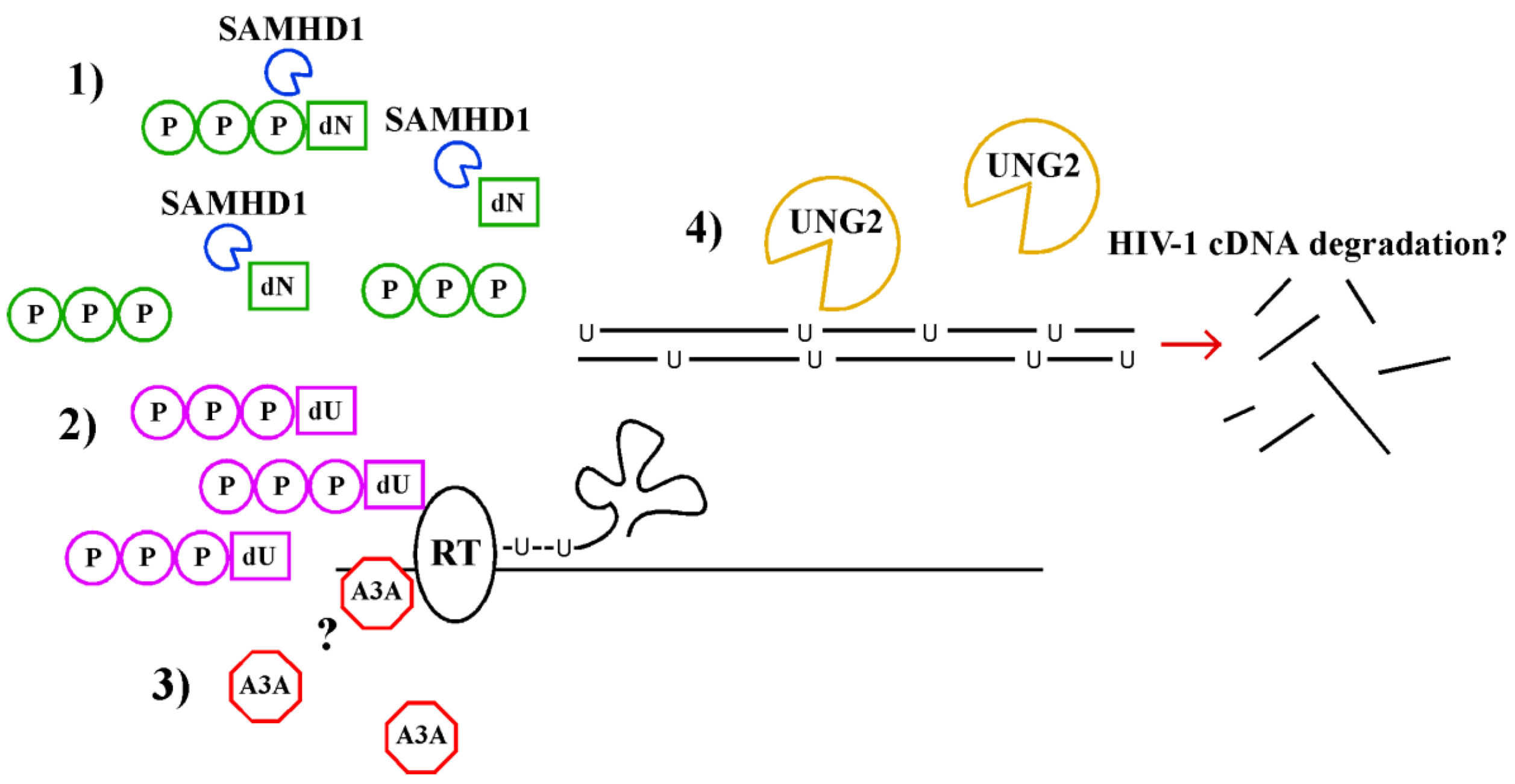
Molecular Mechanisms of HIV Immune Evasion of the Innate Immune Response in Myeloid Cells
Abstract
:1. Introduction
Intrinsic Anti-Viral Factors Limit Infection of Myeloid Cells by HIV and SIV
5. Conclusion
Acknowledgements
Conflict of Interest
References and Notes
- Cameron, P.U.; Forsum, U.; Teppler, H.; Granelli-Piperno, A.; Steinman, R.M. During hiv-1 infection most blood dendritic cells are not productively infected and can induce allogeneic cd4+ t cells clonal expansion. Clin. Exp. Immunol. 1992, 88, 226–236. [Google Scholar]
- Kaushik, R.; Zhu, X.; Stranska, R.; Wu, Y.; Stevenson, M. A cellular restriction dictates the permissivity of nondividing monocytes/macrophages to lentivirus and gammaretrovirus infection. Cell Host Microbe 2009, 6, 68–80. [Google Scholar] [CrossRef]
- Gupta, P.; Collins, K.B.; Ratner, D.; Watkins, S.; Naus, G.J.; Landers, D.V.; Patterson, B.K. Memory cd4(+) t cells are the earliest detectable human immunodeficiency virus type 1 (hiv-1)-infected cells in the female genital mucosal tissue during hiv-1 transmission in an organ culture system. J. Virol. 2002, 76, 9868–9876. [Google Scholar]
- Zhang, Z.; Schuler, T.; Zupancic, M.; Wietgrefe, S.; Staskus, K.A.; Reimann, K.A.; Reinhart, T.A.; Rogan, M.; Cavert, W.; Miller, C.J.; et al. Sexual transmission and propagation of siv and hiv in resting and activated cd4+ t cells. Science 1999, 286, 1353–1357. [Google Scholar] [CrossRef]
- Hladik, F.; Sakchalathorn, P.; Ballweber, L.; Lentz, G.; Fialkow, M.; Eschenbach, D.; McElrath, M.J. Initial events in establishing vaginal entry and infection by human immunodeficiency virus type-1. Immunity 2007, 26, 257–270. [Google Scholar] [CrossRef]
- Hu, J.; Gardner, M.B.; Miller, C.J. Simian immunodeficiency virus rapidly penetrates the cervicovaginal mucosa after intravaginal inoculation and infects intraepithelial dendritic cells. J. Virol. 2000, 74, 6087–6095. [Google Scholar] [CrossRef]
- Cohen, M.S.; Shaw, G.M.; McMichael, A.J.; Haynes, B.F. Acute hiv-1 infection. New Engl. J. Med. 2011, 364, 1943–1954. [Google Scholar] [CrossRef]
- Reinhart, T.A.; Rogan, M.J.; Huddleston, D.; Rausch, D.M.; Eiden, L.E.; Haase, A.T. Simian immunodeficiency virus burden in tissues and cellular compartments during clinical latency and aids. J. Infect. Dis. 1997, 176, 1198–1208. [Google Scholar]
- Schacker, T.; Little, S.; Connick, E.; Gebhard, K.; Zhang, Z.Q.; Krieger, J.; Pryor, J.; Havlir, D.; Wong, J.K.; Schooley, R.T.; et al. Productive infection of t cells in lymphoid tissues during primary and early human immunodeficiency virus infection. J. Infect. Dis. 2001, 183, 555–562. [Google Scholar] [CrossRef]
- Koenig, S.; Gendelman, H.E.; Orenstein, J.M.; Dal Canto, M.C.; Pezeshkpour, G.H.; Yungbluth, M.; Janotta, F.; Aksamit, A.; Martin, M.A.; Fauci, A.S. Detection of aids virus in macrophages in brain tissue from aids patients with encephalopathy. Science 1986, 233, 1089–1093. [Google Scholar]
- Desrosiers, R.C.; Hansen-Moosa, A.; Mori, K.; Bouvier, D.P.; King, N.W.; Daniel, M.D.; Ringler, D.J. Macrophage-tropic variants of siv are associated with specific aids-related lesions but are not essential for the development of aids. Am. J. Pathol. 1991, 139, 29–35. [Google Scholar]
- Berger, G.; Durand, S.; Fargier, G.; Nguyen, X.N.; Cordeil, S.; Bouaziz, S.; Muriaux, D.; Darlix, J.L.; Cimarelli, A. Apobec3a is a specific inhibitor of the early phases of hiv-1 infection in myeloid cells. PLoS Pathog. 2011, 7, e1002221. [Google Scholar] [CrossRef]
- Laguette, N.; Rahm, N.; Sobhian, B.; Chable-Bessia, C.; Munch, J.; Snoeck, J.; Sauter, D.; Switzer, W.M.; Heneine, W.; Kirchhoff, F.; et al. Evolutionary and functional analyses of the interaction between the myeloid restriction factor samhd1 and the lentiviral vpx protein. Cell Host Microbe 2012, 11, 205–217. [Google Scholar] [CrossRef]
- Geijtenbeek, T.B.; Kwon, D.S.; Torensma, R.; van Vliet, S.J.; van Duijnhoven, G.C.; Middel, J.; Cornelissen, I.L.; Nottet, H.S.; KewalRamani, V.N.; Littman, D.R.; Figdor, C.G.; van Kooyk, Y. Dc-sign, a dendritic cell-specific hiv-1-binding protein that enhances trans-infection of t cells. Cell 2000, 100, 587–597. [Google Scholar] [CrossRef]
- Saifuddin, M.; Hart, M.L.; Gewurz, H.; Zhang, Y.; Spear, G.T. Interaction of mannose-binding lectin with primary isolates of human immunodeficiency virus type 1. J. Gen. Virol. 2000, 81, 949–955. [Google Scholar]
- Bobardt, M.D.; Saphire, A.C.; Hung, H.C.; Yu, X.; Van der Schueren, B.; Zhang, Z.; David, G.; Gallay, P.A. Syndecan captures, protects, and transmits hiv to t lymphocytes. Immunity 2003, 18, 27–39. [Google Scholar] [CrossRef]
- Gummuluru, S.; Rogel, M.; Stamatatos, L.; Emerman, M. Binding of human immunodeficiency virus type 1 to immature dendritic cells can occur independently of dc-sign and mannose binding c-type lectin receptors via a cholesterol-dependent pathway. J. Virol. 2003, 77, 12865–12874. [Google Scholar] [CrossRef]
- Izquierdo-Useros, N.; Lorizate, M.; Contreras, F.X.; Rodriguez-Plata, M.T.; Glass, B.; Erkizia, I.; Prado, J.G.; Casas, J.; Fabrias, G.; Krausslich, H.G.; et al. Sialyllactose in viral membrane gangliosides is a novel molecular recognition pattern for mature dendritic cell capture of hiv-1. PLoS Biol. 2012, 10, e1001315. [Google Scholar] [CrossRef]
- Puryear, W.B.; Yu, X.; Ramirez, N.P.; Reinhard, B.M.; Gummuluru, S. Hiv-1 incorporation of host-cell-derived glycosphingolipid gm3 allows for capture by mature dendritic cells. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 7475–7480. [Google Scholar]
- Magerus-Chatinet, A.; Yu, H.; Garcia, S.; Ducloux, E.; Terris, B.; Bomsel, M. Galactosyl ceramide expressed on dendritic cells can mediate hiv-1 transfer from monocyte derived dendritic cells to autologous t cells. Virology 2007, 362, 67–74. [Google Scholar] [CrossRef]
- de Silva, S.; Planelles, V.; Wu, L. Differential effects of vpr on single-cycle and spreading hiv-1 infections in cd4+ t-cells and dendritic cells. PLoS One 2012, 7, e35385. [Google Scholar]
- Wu, L.; KewalRamani, V.N. Dendritic-cell interactions with hiv: Infection and viral dissemination. Nat. Rev. Immunol. 2006, 6, 859–868. [Google Scholar] [CrossRef]
- Cameron, P.U.; Freudenthal, P.S.; Barker, J.M.; Gezelter, S.; Inaba, K.; Steinman, R.M. Dendritic cells exposed to human immunodeficiency virus type-1 transmit a vigorous cytopathic infection to cd4+ t cells. Science 1992, 257, 383–387. [Google Scholar]
- Altfeld, M.; Fadda, L.; Frleta, D.; Bhardwaj, N. Dcs and nk cells: Critical effectors in the immune response to hiv-1. Nat. Rev. Immunol. 2011, 11, 176–186. [Google Scholar] [CrossRef]
- Coleman, C.M.; Wu, L. Hiv interactions with monocytes and dendritic cells: Viral latency and reservoirs. Retrovirology 2009, 6, 51. [Google Scholar] [CrossRef]
- Sharova, N.; Swingler, C.; Sharkey, M.; Stevenson, M. Macrophages archive hiv-1 virions for dissemination in trans. EMBO J. 2005, 24, 2481–2489. [Google Scholar] [CrossRef]
- Lim, E.S.; Fregoso, O.I.; McCoy, C.O.; Matsen, F.A.; Malik, H.S.; Emerman, M. The ability of primate lentiviruses to degrade the monocyte restriction factor samhd1 preceded the birth of the viral accessory protein vpx. Cell Host Microbe 2012, 11, 194–204. [Google Scholar] [CrossRef]
- Tristem, M.; Marshall, C.; Karpas, A.; Hill, F. Evolution of the primate lentiviruses: Evidence from vpx and vpr. EMBO J. 1992, 11, 3405–3412. [Google Scholar]
- Ayinde, D.; Maudet, C.; Transy, C.; Margottin-Goguet, F. Limelight on two hiv/siv accessory proteins in macrophage infection: Is vpx overshadowing vpr? Retrovirology 2010, 7, 35. [Google Scholar]
- Le Rouzic, E.; Benichou, S. The vpr protein from hiv-1: Distinct roles along the viral life cycle. Retrovirology 2005, 2, 11. [Google Scholar] [CrossRef]
- Malim, M.H.; Emerman, M. Hiv-1 accessory proteins—Ensuring viral survival in a hostile environment. Cell Host Microbe 2008, 3, 388–398. [Google Scholar] [CrossRef]
- Peeters, M.; Courgnaud, V. Overview of primate lentiviruses and their evolution in non-human primates in Africa. In HIV Sequence Compendium; HIV sequence compendium; Theoretical Biology and Biophysics Group, Los Alamos National Laboratory: Los Alamos, NM, USA, 2002; pp. 2–23. [Google Scholar]
- Belshan, M.; Kimata, J.T.; Brown, C.; Cheng, X.; McCulley, A.; Larsen, A.; Thippeshappa, R.; Hodara, V.; Giavedoni, L.; Hirsch, V.; et al. Vpx is critical for sivmne infection of pigtail macaques. Retrovirology 2012, 9, 32. [Google Scholar] [CrossRef]
- Hirsch, V.M.; Sharkey, M.E.; Brown, C.R.; Brichacek, B.; Goldstein, S.; Wakefield, J.; Byrum, R.; Elkins, W.R.; Hahn, B.H.; Lifson, J.D.; et al. Vpx is required for dissemination and pathogenesis of siv(sm) pbj: Evidence of macrophage-dependent viral amplification. Nat. Med. 1998, 4, 1401–1408. [Google Scholar]
- Gibbs, J.S.; Lackner, A.A.; Lang, S.M.; Simon, M.A.; Sehgal, P.K.; Daniel, M.D.; Desrosiers, R.C. Progression to aids in the absence of a gene for vpr or vpx. J. Virol. 1995, 69, 2378–2383. [Google Scholar]
- Mandal, D.; Prasad, V.R. Analysis of 2-ltr circle junctions of viral DNA in infected cells. Meth. Mol. Biol. 2009, 485, 73–85. [Google Scholar] [CrossRef]
- Sharova, N.; Wu, Y.; Zhu, X.; Stranska, R.; Kaushik, R.; Sharkey, M.; Stevenson, M. Primate lentiviral vpx commandeers ddb1 to counteract a macrophage restriction. PLoS Pathog. 2008, 4, e1000057. [Google Scholar] [CrossRef]
- Hrecka, K.; Hao, C.; Gierszewska, M.; Swanson, S.K.; Kesik-Brodacka, M.; Srivastava, S.; Florens, L.; Washburn, M.P.; Skowronski, J. Vpx relieves inhibition of hiv-1 infection of macrophages mediated by the samhd1 protein. Nature 2011, 474, 658–661. [Google Scholar] [CrossRef]
- Laguette, N.; Sobhian, B.; Casartelli, N.; Ringeard, M.; Chable-Bessia, C.; Segeral, E.; Yatim, A.; Emiliani, S.; Schwartz, O.; Benkirane, M. Samhd1 is the dendritic- and myeloid-cell-specific hiv-1 restriction factor counteracted by vpx. Nature 2011, 474, 654–657. [Google Scholar] [CrossRef]
- Lim, E.S.; Emerman, M. Hiv: Going for the watchman. Nature 2011, 474, 587–588. [Google Scholar] [CrossRef]
- Planelles, V. Samhd1 joins the red queen’s court. Cell Host Microbe 2012, 11, 103–105. [Google Scholar] [CrossRef]
- Baldauf, H.M.; Pan, X.; Erikson, E.; Schmidt, S.; Daddacha, W.; Burggraf, M.; Schenkova, K.; Ambiel, I.; Wabnitz, G.; Gramberg, T.; et al. Samhd1 restricts hiv-1 infection in resting cd4(+) t cells. Nat. Med. 2012, 18, 1682–1689. [Google Scholar] [CrossRef]
- Descours, B.; Cribier, A.; Chable-Bessia, C.; Ayinde, D.; Rice, G.; Crow, Y.; Yatim, A.; Schwartz, O.; Laguette, N.; Benkirane, M. Samhd1 restricts hiv-1 reverse transcription in quiescent cd4+ t-cells. Retrovirology 2012, 9, 87. [Google Scholar] [CrossRef]
- Aravind, L.; Koonin, E.V. The hd domain defines a new superfamily of metal-dependent phosphohydrolases. Trends Biochem. Sci. 1998, 23, 469–472. [Google Scholar] [CrossRef]
- Goldstone, D.C.; Ennis-Adeniran, V.; Hedden, J.J.; Groom, H.C.; Rice, G.I.; Christodoulou, E.; Walker, P.A.; Kelly, G.; Haire, L.F.; Yap, M.W.; et al. Hiv-1 restriction factor samhd1 is a deoxynucleoside triphosphate triphosphohydrolase. Nature 2011, 480, 379–382. [Google Scholar]
- Lahouassa, H.; Daddacha, W.; Hofmann, H.; Ayinde, D.; Logue, E.C.; Dragin, L.; Bloch, N.; Maudet, C.; Bertrand, M.; Gramberg, T.; et al. Samhd1 restricts the replication of human immunodeficiency virus type 1 by depleting the intracellular pool of deoxynucleoside triphosphates. Nat. Immunol. 2012, 13, 223–228. [Google Scholar]
- Jermy, A. Viral infection: Samhd1 cuts the power to hiv-1. Nat. Rev. Microbiol. 2012, 10, 237. [Google Scholar] [CrossRef]
- Schaller, T.; Goujon, C.; Malim, M.H. Aids/hiv. Hiv interplay with samhd1. Science 2012, 335, 1313–1314. [Google Scholar] [CrossRef]
- Manel, N.; Hogstad, B.; Wang, Y.; Levy, D.E.; Unutmaz, D.; Littman, D.R. A cryptic sensor for hiv-1 activates antiviral innate immunity in dendritic cells. Nature 2010, 467, 214–217. [Google Scholar] [CrossRef]
- Xu, W.; Santini, P.A.; Sullivan, J.S.; He, B.; Shan, M.; Ball, S.C.; Dyer, W.B.; Ketas, T.J.; Chadburn, A.; Cohen-Gould, L.; et al. Hiv-1 evades virus-specific igg2 and iga responses by targeting systemic and intestinal b cells via long-range intercellular conduits. Nat. Immunol. 2009, 10, 1008–1017. [Google Scholar] [CrossRef]
- Ogawa, K.; Shibata, R.; Kiyomasu, T.; Higuchi, I.; Kishida, Y.; Ishimoto, A.; Adachi, A. Mutational analysis of the human immunodeficiency virus vpr open reading frame. J. Virol. 1989, 63, 4110–4114. [Google Scholar]
- Balliet, J.W.; Kolson, D.L.; Eiger, G.; Kim, F.M.; McGann, K.A.; Srinivasan, A.; Collman, R. Distinct effects in primary macrophages and lymphocytes of the human immunodeficiency virus type 1 accessory genes vpr, vpu, and nef: Mutational analysis of a primary hiv-1 isolate. Virology 1994, 200, 623–631. [Google Scholar] [CrossRef]
- Rey, F.; BouHamdan, M.; Navarro, J.M.; Agostini, I.; Willetts, K.; Bouyac, M.; Tamalet, C.; Spire, B.; Vigne, R.; Sire, J. A role for human immunodeficiency virus type 1 vpr during infection of peripheral blood mononuclear cells. J. Gen. Virol. 1998, 79, 1083–1087. [Google Scholar]
- Heinzinger, N.K.; Bukinsky, M.I.; Haggerty, S.A.; Ragland, A.M.; Kewalramani, V.; Lee, M.A.; Gendelman, H.E.; Ratner, L.; Stevenson, M.; Emerman, M. The vpr protein of human immunodeficiency virus type 1 influences nuclear localization of viral nucleic acids in nondividing host cells. Proc. Natl. Acad. Sci. U. S. A. 1994, 91, 7311–7315. [Google Scholar]
- Lang, S.M.; Weeger, M.; Stahl-Hennig, C.; Coulibaly, C.; Hunsmann, G.; Muller, J.; Muller-Hermelink, H.; Fuchs, D.; Wachter, H.; Daniel, M.M.; et al. Importance of vpr for infection of rhesus monkeys with simian immunodeficiency virus. J. Virol. 1993, 67, 902–912. [Google Scholar]
- Hoch, J.; Lang, S.M.; Weeger, M.; Stahl-Hennig, C.; Coulibaly, C.; Dittmer, U.; Hunsmann, G.; Fuchs, D.; Muller, J.; Sopper, S.; et al. Vpr deletion mutant of simian immunodeficiency virus induces aids in rhesus monkeys. J. Virol. 1995, 69, 4807–4813. [Google Scholar]
- Somasundaran, M.; Sharkey, M.; Brichacek, B.; Luzuriaga, K.; Emerman, M.; Sullivan, J.L.; Stevenson, M. Evidence for a cytopathogenicity determinant in hiv-1 vpr. Proc. Natl. Acad. Sci. U. S. A. 2002, 99, 9503–9508. [Google Scholar]
- Schrofelbauer, B.; Hakata, Y.; Landau, N.R. Hiv-1 vpr function is mediated by interaction with the damage-specific DNA-binding protein ddb1. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 4130–4135. [Google Scholar]
- Kennedy, E.M.; Daddacha, W.; Slater, R.; Gavegnano, C.; Fromentin, E.; Schinazi, R.F.; Kim, B. Abundant non-canonical dutp found in primary human macrophages drives its frequent incorporation by hiv-1 reverse transcriptase. J. Biol. Chem. 2011, 286, 25047–25055. [Google Scholar]
- Sire, J.; Querat, G.; Esnault, C.; Priet, S. Uracil within DNA: An actor of antiviral immunity. Retrovirology 2008, 5, 45. [Google Scholar] [CrossRef]
- Norman, J.M.; Mashiba, M.; McNamara, L.A.; Onafuwa-Nuga, A.; Chiari-Fort, E.; Shen, W.; Collins, K.L. The antiviral factor apobec3g enhances the recognition of hiv-infected primary t cells by natural killer cells. Nat. Immunol. 2011, 12, 975–983. [Google Scholar] [CrossRef]
- Croxford, J.L.; Gasser, S. Damage control: How hiv survives the editor apobec3g. Nat. Immunol. 2011, 12, 925–927. [Google Scholar] [CrossRef]
- Harris, R.S.; Bishop, K.N.; Sheehy, A.M.; Craig, H.M.; Petersen-Mahrt, S.K.; Watt, I.N.; Neuberger, M.S.; Malim, M.H. DNA deamination mediates innate immunity to retroviral infection. Cell 2003, 113, 803–809. [Google Scholar] [CrossRef]
- Chen, R.; Le Rouzic, E.; Kearney, J.A.; Mansky, L.M.; Benichou, S. Vpr-mediated incorporation of ung2 into hiv-1 particles is required to modulate the virus mutation rate and for replication in macrophages. J. Biol. Chem. 2004, 279, 28419–28425. [Google Scholar]
- Zimmerman, E.S.; Sherman, M.P.; Blackett, J.L.; Neidleman, J.A.; Kreis, C.; Mundt, P.; Williams, S.A.; Warmerdam, M.; Kahn, J.; Hecht, F.M.; Grant, R.M.; de Noronha, C.M.; Weyrich, A.S.; Greene, W.C.; Planelles, V. Human immunodeficiency virus type 1 vpr induces DNA replication stress in vitro and in vivo. J. Virol. 2006, 80, 10407–10418. [Google Scholar]
- Lavin, M.F.; Kozlov, S. Atm activation and DNA damage response. Cell Cycle 2007, 6, 931–942. [Google Scholar] [CrossRef]
- Cimprich, K.A.; Cortez, D. Atr: An essential regulator of genome integrity. Nat. Rev. Mol. Cell Biol. 2008, 9, 616–627. [Google Scholar] [CrossRef]
- Priet, S.; Gros, N.; Navarro, J.M.; Boretto, J.; Canard, B.; Querat, G.; Sire, J. Hiv-1-associated uracil DNA glycosylase activity controls dutp misincorporation in viral DNA and is essential to the hiv-1 life cycle. Mol. Cell 2005, 17, 479–490. [Google Scholar] [CrossRef]
- Gu, Y.; Sundquist, W.I. Good to cu. Nature 2003, 424, 21–22. [Google Scholar] [CrossRef]
- Yang, B.; Chen, K.; Zhang, C.; Huang, S.; Zhang, H. Virion-associated uracil DNA glycosylase-2 and apurinic/apyrimidinic endonuclease are involved in the degradation of apobec3g-edited nascent hiv-1 DNA. J. Biol. Chem. 2007, 282, 11667–11675. [Google Scholar]
- Yan, N.; O'Day, E.; Wheeler, L.A.; Engelman, A.; Lieberman, J. Hiv DNA is heavily uracilated, which protects it from autointegration. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 9244–9249. [Google Scholar]
- Sheehy, A.M.; Gaddis, N.C.; Choi, J.D.; Malim, M.H. Isolation of a human gene that inhibits hiv-1 infection and is suppressed by the viral vif protein. Nature 2002, 418, 646–650. [Google Scholar] [CrossRef]
- Bishop, K.N.; Verma, M.; Kim, E.Y.; Wolinsky, S.M.; Malim, M.H. Apobec3g inhibits elongation of hiv-1 reverse transcripts. PLoS Pathog. 2008, 4, e1000231. [Google Scholar] [CrossRef]
- Koning, F.A.; Newman, E.N.; Kim, E.Y.; Kunstman, K.J.; Wolinsky, S.M.; Malim, M.H. Defining apobec3 expression patterns in human tissues and hematopoietic cell subsets. J. Virol. 2009, 83, 9474–9485. [Google Scholar]
- Malim, M.H. Apobec proteins and intrinsic resistance to hiv-1 infection. Phil. Trans. Roy. Soc. Lond. B Biol. Sci. 2009, 364, 675–687. [Google Scholar] [CrossRef]
- Peng, G.; Greenwell-Wild, T.; Nares, S.; Jin, W.; Lei, K.J.; Rangel, Z.G.; Munson, P.J.; Wahl, S.M. Myeloid differentiation and susceptibility to hiv-1 are linked to apobec3 expression. Blood 2007, 110, 393–400. [Google Scholar] [CrossRef]
- Koning, F.A.; Goujon, C.; Bauby, H.; Malim, M.H. Target cell-mediated editing of hiv-1 cdna by apobec3 proteins in human macrophages. J. Virol. 2011, 85, 13448–13452. [Google Scholar] [CrossRef]
- Chiu, Y.L.; Greene, W.C. The apobec3 cytidine deaminases: An innate defensive network opposing exogenous retroviruses and endogenous retroelements. Ann. Rev. Immunol. 2008, 26, 317–353. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mashiba, M.; Collins, K.L. Molecular Mechanisms of HIV Immune Evasion of the Innate Immune Response in Myeloid Cells. Viruses 2013, 5, 1-14. https://doi.org/10.3390/v5010001
Mashiba M, Collins KL. Molecular Mechanisms of HIV Immune Evasion of the Innate Immune Response in Myeloid Cells. Viruses. 2013; 5(1):1-14. https://doi.org/10.3390/v5010001
Chicago/Turabian StyleMashiba, Mike, and Kathleen L. Collins. 2013. "Molecular Mechanisms of HIV Immune Evasion of the Innate Immune Response in Myeloid Cells" Viruses 5, no. 1: 1-14. https://doi.org/10.3390/v5010001
APA StyleMashiba, M., & Collins, K. L. (2013). Molecular Mechanisms of HIV Immune Evasion of the Innate Immune Response in Myeloid Cells. Viruses, 5(1), 1-14. https://doi.org/10.3390/v5010001