Combination Chemotherapy for Influenza
Abstract
:1. Introduction
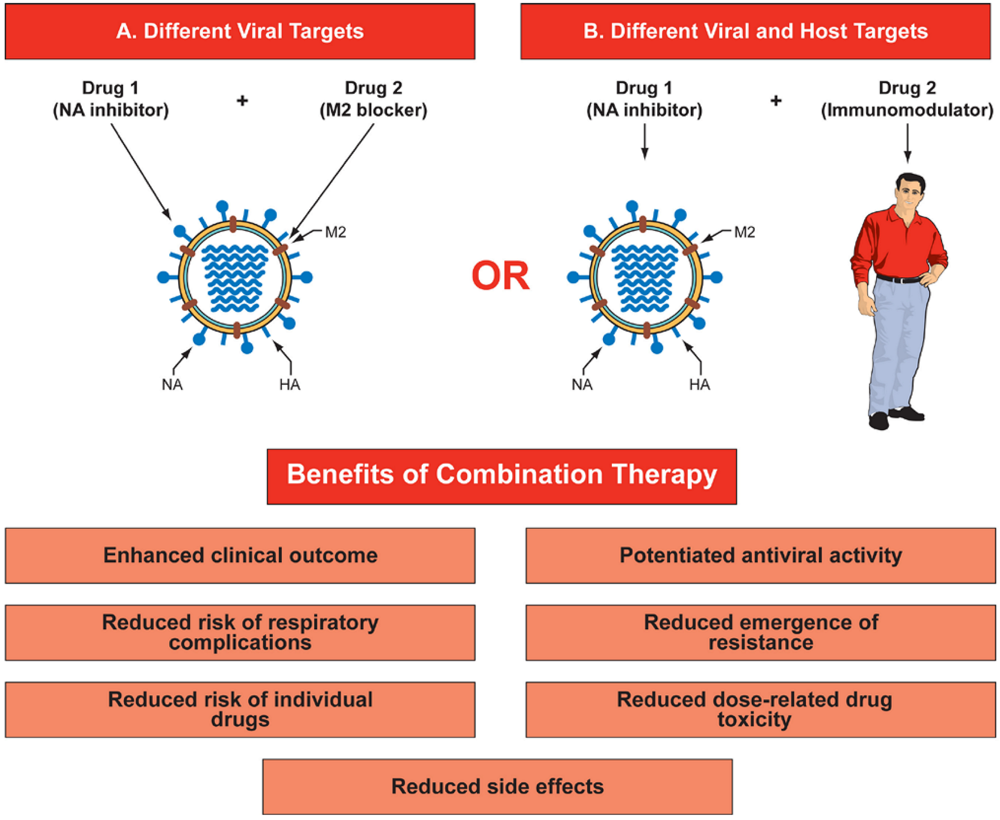
4. Clinical data
6. Conclusions
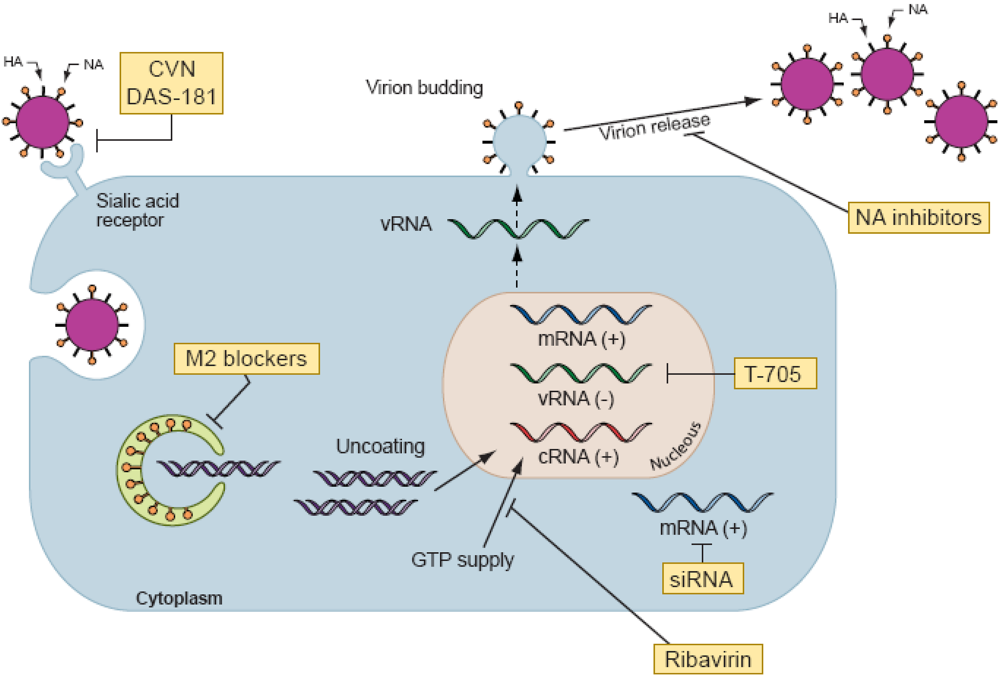
Conflict of Interest
Acknowledgments
References
- Stephenson, I.; Nicholson, K.G. Influenza: vaccination and treatment. Eur. Respir. J. 2001, 17, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Ong, A.K.; Hayden, F.G.; John, F. Enders lecture 2006: Antivirals for influenza . J. Infect. Dis. 2007, 196, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Moscona, A. Medical management of influenza infection. Annu. Rev. Med. 2008, 59, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Bright, R.A.; Medina, M.J.; Xu, X.; Perez-Oronoz, G.; Wallis, T.R.; Davis, X.M.; Povinelli, L.; Cox, N.J.; Klimov, A.I. Incidence of amantadine resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: a cause of concern. Lancet 2005, 366, 1175–1181. [Google Scholar] [CrossRef] [PubMed]
- Deyde, V.M.; Xu, X.; Bright, R.A.; Shaw, M.; Smith, C.B.; Zhang, Y.; Shu, Y.; Gubareva, L.V.; Cox, N.J.; Klimov, A.I. Surveillance of resistance to adamantanes among influenza A (H3N2) and A (H1N1) viruses isolated worldwide. J. Infect. Dis. 2007, 196, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Hurt, A.C.; Selleck, P.; Komadina, N.; Shaw, R.; Brown, L.; Barr, I.G. Susceptibility of highly pathogenic A (H5N1) avian influenza viruses to the neuraminidase inhibitors and adamantanes. Antiviral Res. 2007, 73, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Gubareva, L.; Okomo-Adhiambo, M.; Deyde, V.; Fry, A.M.; Sheu, T.G.; Garten, R.; Smith, D.C.; Barnes, J.; Myrick, A.; Hillman, M.; Shaw, M.; Bridges, C.; Klimov, A.; Cox, N. Centers for Disease Control and Prevention (CDC). Update: Drug susceptibility of swine origin influenza A (H1N1) viruses, April 2009. MMWR Morb. Mortal. Wkly Rep. 2009, 58, 433–435. [Google Scholar] [PubMed]
- Ferraris, O.; Lina, B. Mutations of neuraminidase implicated in neuraminidase inhibitors resistance. J. Clin. Virol. 2008, 41, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Herlocher, M.L.; Carr, J.; Ives, J.; Elias, S.; Truscon, R.; Roberts, N.; Monto, A.S. Influenza virus carrying an R292K mutation in the neuraminidase gene is not transmitted in ferrets. Antiviral Res. 2002, 54, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Yen, H-L; Herlocher, L.M.; Hoffmann, E.; Matrosovich, M.N.; Monto, A.S.; Webster, R.G.; Govorkova, E.A. Neuraminidase inhibitor-resistant influenza viruses may differ substantially in fitness and transmissibility . Antimicrob. Agents Chemother. 2005, 49, 4075–4084. [Google Scholar] [CrossRef] [PubMed]
- Bouvier, N.M.; Lowen, A.C.; Palese, P. Oseltamivir-resistant influenza A viruses are transmitted efficiently among guinea pigs by direct contact but not by aerosol. J. Virol. 2008, 82, 10052–10058. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.; Small, I.; Smith, J.; Suter, P.; Dutkowski, R. Oseltamivir (TamifluR) and its potential for use in the event of an influenza pandemic. J. Antimicrob. Chemother. 2005, 55, 5–21. [Google Scholar] [CrossRef]
- Lackenby, A.; Hungnes, O.; Dudman, S.G.; Meijer, A.; Paget, W.J.; Hay, A. J.; Zambon, M.C. Emergence of resistance to oseltamivir among influenza A (H1N1) viruses in Europe. Eurosurveillance 2008, 13, 1–3. [Google Scholar]
- Dharan, N.J.; Gubareva, L.V.; Meyer, J.J.; Okomo-Adhiambo, M.; McClinton, R.C.; Marshall, S.A.; George, K. St.; Epperson, S.; Brammer, L.; Klimov, A.I.; Bresee, J.S.; Fry, A.M. For the Oseltamivir-Resistance Working Group. Infections with oseltamivir-resistant influenza A (H1N1) virus in the United States. JAMA 2009, 301, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
- Hurt, A.C.; Ernest, J.; Deng, Y-M.; Iannello, P.; Besselaar, T.G.; Birch, C.; Buchy, P.; Chittaganpitch, M.; Chiu, S-C.; Dwyer, D.; Guigoni, A.; Harrower, B.; Kei, I.P.; Kok, T.; Lin, C.; McPhie, K.; Mohd, A.; Olveda, R.; Panayotou, T.; Rawlinson, W.; Scott, L.; Smith, D.; D’Souza, H.; Komadina, N.; Shaw, R.; Kelso, A.; Barr, I.G. Emergence and spread of oseltamivir-resistant A (H1N1) influenza viruses in Oceania, South East Asia and South Africa . Antiviral Res. 2009, 83, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Bautista, E.; Chotpitayasunondh, T.; Gao, Z.; Harper, S.A.; Shaw, M.; Uyeki, T.M.; Zaki, S.R.; Hayden, F.G.; Hui, D.S.; Kettner, J.D.; Kumar, A.; Lim, M.; Shindo, N.; Penn, C.; Nicholson, K.G. Writing Committee of the WHO Consultation on the Clinical Aspects of Pandemic (H1N1) 2009 Influenza. Clinical aspects of pandemic 2009 influenza A (H1N1) virus infection. N. Engl. J. Med. 2010, 362, 1708–1719. [Google Scholar] [CrossRef] [PubMed]
- Li, I.W.; Hung, I.F.; To, K.K.; Chan, K.H.; Wong, S.S.; Chan, J.F.; Cheng, V.C.; Tsang, O.T.; Lai, S.T.; Lau, Y.L.; Yuen, K.Y. The natural viral load profile of patients with pandemic 2009 influenza A (H1N1) and the effect of oseltamivir treatment. Chest 2010, 137, 759–768. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.M.; Chow, A.L.; Lye, D.C.; Tan, A.S.; Krishnan, P.; Cui, L.; Win, N.N.; Chan, M.; Lim, P.L.; Lee, C.C.; Leo, Y.S. Effects of early oseltamivir therapy on viral shedding in 2009 pandemic influenza A (H1N1) virus infection. Clin. Infect. Dis. 2010, 50, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Gulick, R. Combination therapy for patients with HIV-1 infection: the use of dual nucleoside analogues with protease inhibitors and other agents . AIDS 1998, 12, S17–S22. [Google Scholar] [PubMed]
- Raboud, J.M.; Harris, M.; Rae, S.; Montaner, J.S. Impact of adherence on duration of virological suppression among patients receiving combination antiretroviral therapy. HIV Med. 2002, 3, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Suhnel, J. Evaluation of synergism or antagonism for the combined action of antiviral agents. Antiviral Res. 1990, 13, 23–40. [Google Scholar] [CrossRef] [PubMed]
- Galegov, G.A.; Pushkarskaya, N.L.; Obrosova-Serova, N.P.; Zhdanov, V.M. Combined action of ribavirin and rimantadine in experimental myxovirus infection. Experientia 1977, 33, 905–906. [Google Scholar] [CrossRef] [PubMed]
- de Clercq, E. Antiviral agents active against influenza A viruses. Nature Rev. 2006, 5, 1015–1025. [Google Scholar] [CrossRef]
- Sidwell, R.W.; Bailey, K.W.; Wong, M.H.; Barnard, D.L.; Smee, D.F. In vitro and in vivo influenza virus-inhibitory effects of viramidine. Antiviral Res. 2005, 68, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Smee, D.F.; Wong, M.H.; Bailey, K.W.; Sidwell, R.W. Activities of oseltamivir and ribavirin used alone and in combination against infections in mice with recent isolates of influenza A (H1N1) and B viruses. Antiviral Chem. Chemother. 2006, 17, 185–192. [Google Scholar]
- Hayden, F.G.; Douglas Jr., R.G.; Simons, R. Enhancement of activity against influenza viruses by combinations of antiviral agents . Antimicrob. Agents Chemother. 1980, 18, 536–541. [Google Scholar] [PubMed]
- Hayden, F.G.; Slepushkin, A.N.; Pushkarskaya, N.L. Combined interferon-α2, rimantadine hydrochloride, and ribavirin inhibition of influenza virus replication in vitro. Antimicrob. Agents Chemother. 1984, 25, 53–57. [Google Scholar] [PubMed]
- Shigeta, S.; Mori, S.; Watanabe, J.; Soeda, S.; Takahashi, K.; Yamase, T. Synergistic anti-influenza virus A (H1N1) activities of PM-523 (polyoxometalate) and ribavirin in vitro and in vivo. Antimicrob. Agents Chemother. 1997, 41, 1423–1427. [Google Scholar] [PubMed]
- Serkedjieva, J. Combined antiinfluenza virus activity of Flos verbasci infusion and amantadine derivatives . Phytother. Res. 2000, 14, 571–574. [Google Scholar] [CrossRef] [PubMed]
- Gegova, G.; Manolova, N.; Serkedzhieva, Iu.; Maksimova, V.; Uzunov, S.; Dzeguze, D.; Indulen, M. Combined effect of selected antiviral substances of natural and synthetic origin. II. Anti-influenza activity of a combination of a polyphenolic complex isolated from Geranium sanguineum L. and rimantadine in vivo. Acta Microbiol Bulg. 1993, 30, 37–40. [Google Scholar] [PubMed]
- Kim, C.U.; Lew, W.; Williams, M.A.; Liu, H.; Zhang, L.; Swaminathan, S.; Bischofberger, N.; Chen, M.S.; Mendel, D.B.; Tai, C.Y.; Laver, W.G.; Stevens, R.C. Influenza neuraminidase inhibitors possessing a novel hydrophobic interactions in the enzyme active site: design, synthesis, and structural analysis of carbocyclic sialic acid analogues with potent anti-influenza activity . J. Am. Chem. Soc. 1997, 119, 681–690. [Google Scholar] [CrossRef] [PubMed]
- von Itzstein, M.; Wu, W.-Y.; Kok, G.K.; Pegg, M.S.; Dyason, J.C.; Jin, B.; Van Phan, T.; Smithe, M.L.; White, H.F.; Oliver, S.W.; Colman, P.M.; Varghese, J.N.; Ryan, D.M.; Woods, J.M.; Bethell, R.C.; Hotham, V.J.; Cameron, J.M.; Penn, C.R. Rational design of potent sialidase-based inhibitors of influenza virus replication. Nature 1993, 363, 418–423. [Google Scholar] [CrossRef] [PubMed]
- Babu, Y.S.; Chand, P.; Bantia, S.; Kotian, P.L.; Dehghani, A.; El-Kattan, Y.; Lin, T.; Hutchison, T.L.; Elliot, A.J.; Parker, C.D.; Ananth, S.L.; Horn, L.L.; Laver, G.W.; Montgomery, J.A. BCX-1812 (RWJ-270201): discovery of a novel, highly potent, orally active and selective influenza neuraminidase inhibitor through structure-based drug design. J. Med. Chem. 2000, 43, 3482–3486. [Google Scholar] [CrossRef] [PubMed]
- Madren, L.K.; Shipman Jr., C.; Hayden, F.G. In vitro inhibitory effects of combinations of anti-influenza agents . Antivir. Chem. Chemother. 1995, 6, 109–113. [Google Scholar]
- Smee, D.F.; Bailey, K.W.; Morrison, A.C.; Sidwell, R.W. Combination treatment of influenza A virus infections in cell culture and in mice with the cyclopentane neuraminidase inhibitor RWJ-270201 and ribavirin. Chemotherapy 2002, 48, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Govorkova, E.A.; Fang, H.B.; Tan, M.; Webster, R.G. Neuraminidase inhibitor-rimantadine combinations exert additive and synergistic anti-influenza virus effects in MDCK cells. Antimicrob. Agents Chemother. 2004, 48, 4855–4863. [Google Scholar] [CrossRef] [PubMed]
- Ilyushina, N.A.; Bovin, N.V.; Webster, R.G.; Govorkova, E.A. Combination chemotherapy, a potential strategy for reducing the emergence of drug-resistant influenza A variants. Antiviral Res. 2006, 70, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Smee, D.F.; Hurst, B.L.; Wong, M.H.; Bailey, K.W.; Morrey, J.D. Effects of double combinations of amantadine, oseltamivir, and ribavirin on influenza A (H5N1) virus infections in cell culture and in mice. Antimicrob. Agents Chemother. 2009, 53, 2120–2128. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.T.; Hoopes, J.D.; Le, M.H.; Smee, D.F.; Patick, A.K.; Faix, D.J.; Blair, P.J.; de Jong, M.D.; Prichard, M.N.; Went, G.T. Triple combination of amantadine, ribavirin, and oseltamivir is highly active and synergistic against drug resistant influenza virus strains in vitro. PLoS One 2010, 5, e9332. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.T.; Hoopes, J.D.; Smee, D.F.; Prichard, M.N.; Driebe, E.M.; Engelthaler, D.M.; Le, M.H.; Keim, P.S.; Spence, R.P.; Went, G.T. Triple combination of oseltamivir, amantadine, and ribavirin displays synergistic activity against multiple influenza virus strains in vitro. Antimicrob. Agents Chemother. 2009, 53, 4115–4126. [Google Scholar] [CrossRef] [PubMed]
- Prichard, M.N.; Shipman Jr., C. A three-dimensional model to analyze drug-drug interactions . Antiviral Res. 1990, 14, 181–206. [Google Scholar] [CrossRef] [PubMed]
- Galabov, A.S.; Simeonova, L.; Gegova, G. Rimantadine and oseltamivir demonstrate synergistic combination effect in an experimental infection with type A (H3N2) influenza virus in mice . Antivir. Chem. Chemother. 2006, 17, 251–258. [Google Scholar] [PubMed]
- Ilyushina, N.A.; Hoffmann, E.; Salomon, R.; Webster, R.G.; Govorkova, E.A. Amantadine-oseltamivir combination therapy for H5N1 influenza virus infection in mice. Antiviral Ther. 2007, 12, 363–370. [Google Scholar]
- Ilyushina, N.A.; Hay, A.; Yilmaz, N.; Boon, A.C.M.; Webster, R.G.; Govorkova, E.A. Oseltamivir-ribavirin combination therapy for highly pathogenic H5N1 influenza virus infection in mice. Antimicrob. Agents Chemother. 2008, 52, 3889–3897. [Google Scholar] [CrossRef] [PubMed]
- Masihi, K.N.; Schweiger, B.; Finsterbusch, T.; Hengel, H. Low dose oral combination chemoprophylaxis with oseltamivir and amantadine for influenza A virus infection in mice. J. Chemother. 2007, 19, 295–303. [Google Scholar] [PubMed]
- Wilson, S.Z.; Knight, V.; Wyde, P.R.; Drake, S.; Couch, R.B. Amantadine and ribavirin aerosol treatment of influenza A and B infection in mice. Antimicrob. Agents Chemother. 1980, 17, 642–648. [Google Scholar] [PubMed]
- Hayden, F.G. Combinations of antiviral agents for treatment of influenza virus infections. J. Antimicrob. Chemother. 1986, 18, 77–83. [Google Scholar] [PubMed]
- Zhirnov, O.P. High protection of animals lethally infected with influenza virus by aprotinin-rimantadine combinations. J. Med. Virol. 1987, 21, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Peiris, J.S.M.; de Jong, M.D.; Guan, Y. Avian influenza virus (H5N1): a threat to human health. Clin. Mircob. Rev. 2007, 20, 243–267. [Google Scholar] [CrossRef]
- Leneva, I.A.; Roberts, N.; Govorkova, E.A.; Goloubeva, O.G.; Webster, R.G. The neuraminidase inhibitor GS 4101 (oseltamivir phosphate) is efficacious against A/Hong Kong/156/97 (H5N1) and A/Hong Kong/1074/99 (H9N2) influenza viruses. Antiviral Res. 2000, 48, 101–115. [Google Scholar] [CrossRef] [PubMed]
- Ison, M.G.; Gnann Jr., J.W.; Nagy-Agren, S.; Treannor, J.; Paya, C.; Steigbigel, R.; Elliott, M.; Weiss, H.L.; Hayden, F.G. Safety and efficacy of nebulized zanamivir in hospitalized patients with serious influenza . Antiviral Ther. 2003, 8, 183–190. [Google Scholar]
- Morrison, D.; Roy, S.; Rayner, C.; Amer, A.; Howard, D.; Smith, J.R.; Evans, T.G. A randomized, crossover study to evaluate the pharmacokinetics of amantadine and oseltamivir administered alone and in combination . PLoS One 2007, 2, e1305. [Google Scholar] [CrossRef] [PubMed]
- Hill, G.; Cihlar, T.; Oo, C.; Ho, E.S.; Prior, K.; Wiltshire, H.; Barrett, J.; Liu, B.; Ward, P. The anti-influenza drug oseltamivir exhibits low potential to induce pharmacokinetic drug interactions via renal secretion-correlation of in vivo and in vitro studies. Drug Metab. Dispos. 2002, 30, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Holodniy, M.; Penzak, S.R.; Straight, T.M.; Davey, R.T.; Lee, K.K.; Goetz, M.B.; Raisch, D.W.; Cunningham, F.; Lin, E.T.; Olivo, N.; Deyton, L.R. Pharmacokinetics and tolerability of oseltamivir combined with probenecid. Antimicrob. Agents Chemother. 2008, 52, 3013–3021. [Google Scholar] [CrossRef] [PubMed]
- De Bony, F.; Tod, M.; Bidault, R.; On, N.T.; Posner, J.; Rolan, P. Multiple interactions of cimetidine and probenecid with valaciclovir and its metabolite acyclovir. Antimicrob. Agents Chemother. 2002, 46, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, J.S.; Devane, C.L.; Liston, H.L.; Boulton, D.W.; Risch, S.C. The effects of probenecid on the disposition of risperidone and olanzapine in healthy volunteers. Clin. Pharmacol. Ther. 2002, 71, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Takahashi, K.; Fukuda, Y.; Kuno, M.; Kamiyama, T.; Kozaki, K.; Nomura, N.; Egawa, H.; Minami, S.; Watanabe, Y.; Narita, H.; Shiraki, K. In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob. Agents Chemother. 2002, 46, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Takahashi, K.; Kuno-Maekawa, M.; Sangawa, H.; Uehara, S.; Kozaki, K.; Nomura, N.; Egawa, H.; Shiraki, K. Mechanism of action of T-705 against influenza virus. Antimicrob. Agents Chemother. 2005, 49, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Streeter, D.G.; Witkowski, J.T.; Khare, G.P.; Sidwell, R.W.; Bauer, R.J.; Robins, R.K.; Simon, L.N. Mechanism of action of 1- -D-ribofuranosyl-1,2,4-triazole-3-carboxamide (Virazole), a new broad-spectrum antiviral agent. Proc. Natl. Acad. Sci. U S A 1973, 70, 1174–1178. [Google Scholar] [CrossRef] [PubMed]
- Smee, D.F.; Hurst, B.L.; Wong, M.H.; Bailey, K.W.; Tarbet, E.B.; Morrey, J.D.; Furuta, Y. Effects of the combination of favipiravir (T-705) and oseltamivir on influenza a virus infections in mice. Antimicrob. Agents Chemother. 2010, 54, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.C.; Cheung, C.Y.; Chui, W.H.; Tsao, S.W.; Nicholls, J.M.; Chan, Y.O.; Chan, R.W.; Long, H.T.; Poon, L.L.; Guan, Y.; Peiris, J.S. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir. Res. 2005, 11, 1–135. [Google Scholar]
- de Jong, M.D.; Simmons, C.P.; Thanh, T.T.; Hien, V.M.; Smith, G.J.; Chau, T.N.; Hoang, D.M.; Chau, N.V.; Khanh, T.H.; Dong, V.C.; Qui, P.T.; Cam, B.V.; Ha, do Q.; Guan, Y.; Peiris, J.S.; Chinh, N.T.; Hien, T.T.; Farrar, J. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia . Nat. Med. 2006, 12, 1203–1207. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.J.; Chan, K.W.; Lin, Y.P.; Zhao, G.Y.; Chan, C.; Zhang, H.J.; Chen, H.L.; Wong, S.S.; Lau, S.K.; Woo, P.C.; Chan, K.H.; Jin, D.Y.; Yuen, K.Y. Delayed antiviral plus immunomodulator treatment still reduces mortality in mice infected by high inoculum of influenza A/H5N1 virus. Proc. Natl. Acad Sci. U S A 2008, 105, 8091–8096. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, P.; Ungheri, D. Synergistic combination of N-acetylcysteine and ribavirin to protect from lethal influenza viral infection in a mouse model. Int. J. Immunopathol. Pharmacol. 2004, 17, 99–102. [Google Scholar] [PubMed]
- Garozzo, A.; Tempera, G.; Ungheri, D.; Timpanaro, R.; Castro, A. N-acetylcysteine synergizes with oseltamivir in protecting mice from lethal influenza infection. Int. J. Immunopathol. Pharmacol. 2007, 20, 349–354. [Google Scholar] [PubMed]
- Serkedjieva, J.; Ivanova, E. Combined protective effect of an immunostimulatory bacterial preparation and rimantadine in experimental influenza A virus infection. Acta Virol. 1997, 41, 65–70. [Google Scholar] [PubMed]
- Kohno, S.; Yen, M.Y.; Cheong, H.J.; Hirotsu, N.; Ishida, T.; Kadota, J.; Mizuguchi, M.; Kida, H.; Shimada, J. Single-intravenous peramivir vs. oral oseltamivir to treat acute, uncomplicated influenza in the outpatient setting: a phase III randomized, double-blind trial . In Proceedings of the 49th Interscience Conference on Antimicrobial Agents and Chemotherapy; September 2009. [Google Scholar]
- GlaxoSmithKline. An open-label, multi-center, single arm study to evaluate the safety and tolerability of intravenous zanamivir in the treatment of hospitalized adult, adolescent and pediatric subjects with confirmed influenza infection . Available online: http://www.gsk-clinicalstudyregister.com/protocol_detail.jsp?protocolId=113678&studyId=29102&compound=Zanamivir (accessed on 20 April 2010).
- Kubo, S.; Tomozawa, T.; Kakuta, M.; Tokumitsu, A.; Yamashita, M. Laninamivir prodrug CS-8958, a long-acting neuraminidase inhibitor, shows superior anti-influenza virus activity after a single administration. Antimicrob. Agents Chemother. 2010, 54, 1256–1264. [Google Scholar] [CrossRef] [PubMed]
- Kiso, M.; Kubo, S.; Ozawa, M.; Le, Q.M.; Nidom, C.A.; Yamashita, M.; Kawaoka, Y. Efficacy of the new neuraminidase inhibitor CS-8958 against H5N1 influenza viruses . PLoS Pathog. 2010, 6, e1000786. [Google Scholar] [CrossRef] [PubMed]
- Biota Holdings. LANI phase II completed-phase III scheduled (press release 2008). Available online: http://www.biota.com.au/uploaded/154/1021393_18laniphaseiicompleted3Dp.pdf (accessed on 20 April 2010) .
- Sugaya, N.; Ohashi, Y. Long-acting neuraminidase inhibitor laninamivir octanoate (CS-8958) versus oseltamivir as treatment for children with influenza virus infection. Antimicrob. Agents Chemother. 2010, 54, 2575–2582. [Google Scholar] [CrossRef] [PubMed]
- Malakhov, M.P.; Aschenbrenner, L.M.; Smee, D.F.; Wandersee, M.K.; Sidwell, R.W.; Gubareva, L.V.; Mishin, V.P.; Hayden, F.G.; Kim, D.H.; Ing, A.; Campbell, E.R.; Yu, M.; Fang, F. Sialidase fusion protein as a novel broad-spectrum inhibitor of influenza virus infection. Antimicrob. Agents Chemother. 2006, 50, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Belser, J.A.; Lu, X.; Szretter, K.J.; Jin, X.; Aschenbrenner, L.M.; Lee, A.; Hawley, S.; Kim, do H.; Malakhov, M.P.; Yu, M.; Fang, F.; Katz, J.M. DAS181, a novel sialidase fusion protein, protects mice from lethal avian influenza H5N1 virus infection . J. Infect. Dis. 2007, 196, 1493–1499. [Google Scholar] [CrossRef] [PubMed]
- NexBio. NexBio Initiates Phase II Trial of DAS181 (Fludase®*) for Treatment of Influenza, Including Pandemic Influenza A (H1N1), 2010. Available online: http://www.nexbio.com/docs/DAS181_Phase_2_Press_Release_010710.pdf (accessed on 20 April 2010).
- O'Keefe, B.R.; Smee, D.F.; Turpin, J.A.; Saucedo, C.J.; Gustafson, K.R.; Mori, T.; Blakeslee, D.; Buckheit, R.; Boyd, M.R. Potent anti-influenza activity of cyanovirin-N and interactions with viral hemagglutinin. Antimicrob. Agents Chemother. 2003, 47, 2518–2525. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Govorkova, E.A.; Webster, R.G. Combination Chemotherapy for Influenza. Viruses 2010, 2, 1510-1529. https://doi.org/10.3390/v2081510
Govorkova EA, Webster RG. Combination Chemotherapy for Influenza. Viruses. 2010; 2(8):1510-1529. https://doi.org/10.3390/v2081510
Chicago/Turabian StyleGovorkova, Elena A., and Robert G. Webster. 2010. "Combination Chemotherapy for Influenza" Viruses 2, no. 8: 1510-1529. https://doi.org/10.3390/v2081510
APA StyleGovorkova, E. A., & Webster, R. G. (2010). Combination Chemotherapy for Influenza. Viruses, 2(8), 1510-1529. https://doi.org/10.3390/v2081510




