Monitoring HPV Prevalence and Risk Cofactors for Abnormal Cytology in the Post-Vaccination Period among Croatian Women
Abstract
1. Introduction
2. Materials and Methods
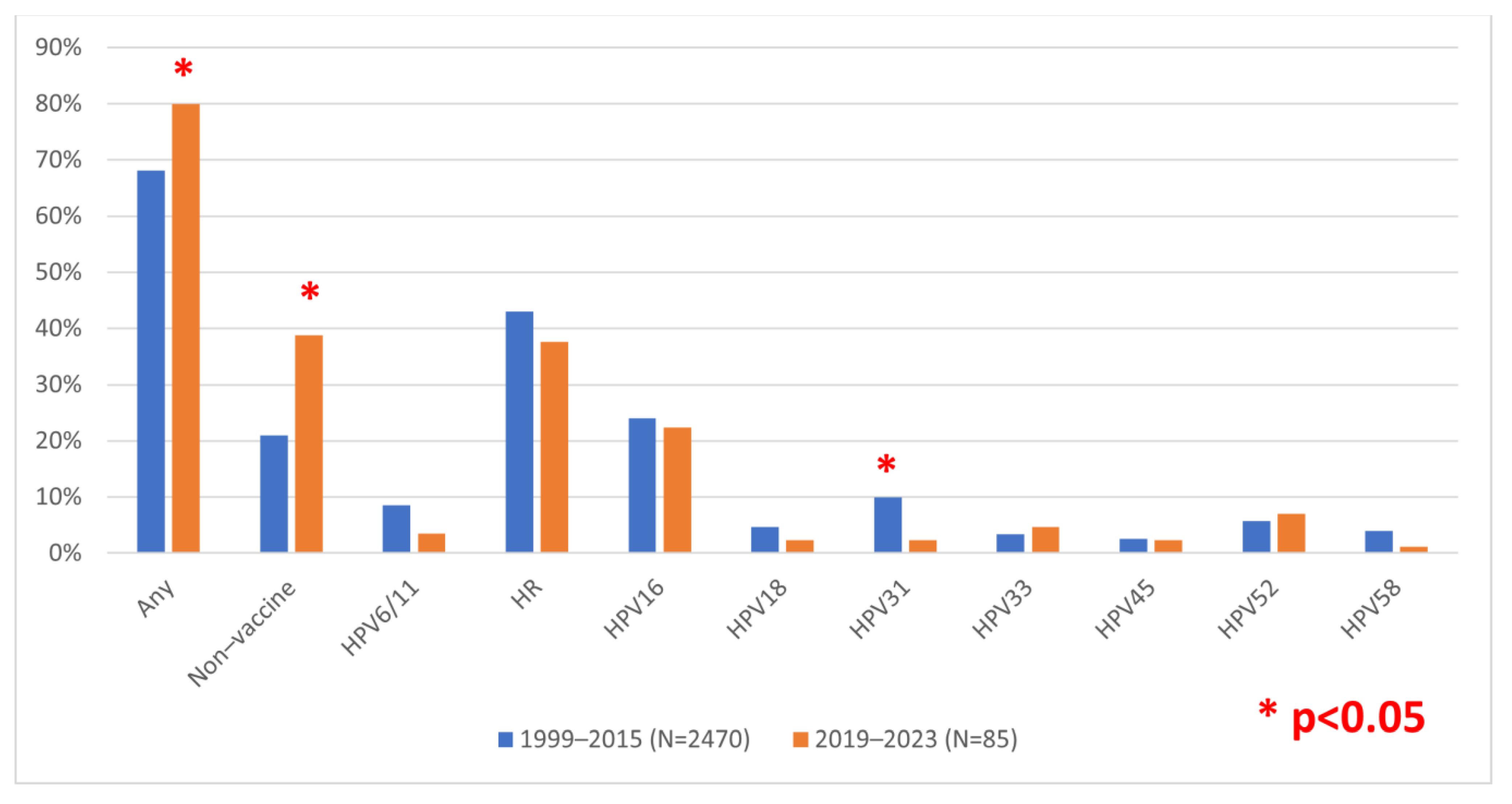
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohen, P.A.; Jhingran, A.; Oaknin, A.; Denny, L. Cervical Cancer. Lancet 2019, 393, 169–182. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Comprehensive Cervical Cancer Control: A Guide to Essential Practice, 2nd ed.; World Health Organization: Geneva, Switzerland, 2014; ISBN 978-92-4-154895-3.
- Hull, R.; Mbele, M.; Makhafola, T.; Hicks, C.; Wang, S.; Reis, R.; Mehrotra, R.; Mkhize-Kwitshana, Z.; Kibiki, G.; Bates, D.; et al. Cervical Cancer in Low and Middle-income Countries (Review). Oncol. Lett. 2020, 20, 2058–2074. [Google Scholar] [CrossRef] [PubMed]
- Šekerija, M.; Bubanović, L.; Lončar, J.; Čukelj, P.; Veltruski, J.; Mikolaj, L.; Stupnik, D.; HZJZ. Cancer Incidence in Croatia 2020, Bulletin 45/Incidencija Raka u Hrvatskoj 2020, Bilten 45; Cancer Incidence in Croatia; Croatian Institute of Public Health, National Cancer Registry: Zagreb, Croatia, 2022; p. 37. [Google Scholar]
- Markowitz, L.E.; Schiller, J.T. Human Papillomavirus Vaccines. J. Infect. Dis. 2021, 224, S367–S378. [Google Scholar] [CrossRef] [PubMed]
- Cervical Cancer Elimination Initiative. Global Strategy to Accelerate the Elimination of Cervical Cancer as a Public Health Problem; World Health Organization, Ed.; World Health Organization: Geneva, Switzerland, 2020; ISBN 978-92-4-001410-7. [Google Scholar]
- World Health Organization. Market Information for Access to Vaccines; Global Market Study: HPV; WHO: Geneva, Switzerland, 2022; p. 7.
- Pavić Šimetin, I.; Belavić, A.; Žehaček Živković, M. Promotion of vaccination against HPV infection at the national level. Paediatr. Croat. 2018, 62, 9–12. [Google Scholar]
- Croatian Institute of Public Health. Vaccination Against Human Papillomavirus (HPV)—Current Data. Available online: https://www.hzjz.hr/aktualnosti/cijepljenje-protiv-humanog-papiloma-virusa-hpv/ (accessed on 14 February 2024). (In Croatian).
- Croatian Bureau of Statistics Croatia. Population Mid-Year Estimate. 2023. Available online: https://podaci.dzs.hr/media/vb1ae2vm/procjene-stanovnistva.xlsx (accessed on 14 February 2024).
- Nemeth Blažić, T.; Božičević, I.; Kosanović Ličina, M.L.; Štulhofer, A.; Nola, I.A. Self-Reported HPV Vaccination Status and HPV Vaccine Hesitancy in a Nationally Representative Sample of Emerging Adults in Croatia. Front. Public Health 2023, 11, 1182582. [Google Scholar] [CrossRef] [PubMed]
- Sabol, I.; Milutin Gašperov, N.; Matovina, M.; Božinović, K.; Grubišić, G.; Fistonić, I.; Belci, D.; Alemany, L.; Džebro, S.; Dominis, M.; et al. Cervical HPV Type-Specific Pre-Vaccination Prevalence and Age Distribution in Croatia. PLoS ONE 2017, 12, e0180480. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, N.; Yousefi, Z.; Khosravi, G.; Malayeri, F.E.; Golabi, M.; Askarzadeh, M.; Shams, M.H.; Ghezelbash, B.; Eskandari, N. Human Papillomavirus Vaccination in Low-and Middle-Income Countries: Progression, Barriers, and Future Prospective. Front. Immunol. 2023, 14, 1150238. [Google Scholar] [CrossRef] [PubMed]
- IARC Working Group on the Evaluation of Cancer-Preventive Strategies. Cervix Cancer Screening; IARC Press: Lyon, France, 2005; ISBN 978-92-832-3010-6. [Google Scholar]
- The International Collaboration of Epidemiological Studies of Cervical Cancer. Comparison of Risk Factors for Invasive Squamous Cell Carcinoma and Adenocarcinoma of the Cervix: Collaborative Reanalysis of Individual Data on 8097 Women with Squamous Cell Carcinoma and 1374 Women with Adenocarcinoma from 12 Epidemiological Studies. Int. J. Cancer 2007, 120, 885–891. [Google Scholar] [CrossRef]
- Wudtisan, J.; Tantipalakorn, C.; Charoenkwan, K.; Sreshthaputra, R.-A.; Srisomboon, J. Factors Associated with Development of High-Grade Squamous Intraepithelial Lesions of the Uterine Cervix in Women Younger than 30 Years. Asian Pac. J. Cancer Prev. 2019, 20, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Šimić, I.; Božinović, K.; Milutin Gašperov, N.; Kordić, M.; Pešut, E.; Manojlović, L.; Grce, M.; Dediol, E.; Sabol, I. Head and Neck Cancer Patients’ Survival According to HPV Status, miRNA Profiling, and Tumour Features—A Cohort Study. IJMS 2023, 24, 3344. [Google Scholar] [CrossRef] [PubMed]
- Scott-Wittenborn, N.; Fakhry, C. Epidemiology of HPV Related Malignancies. Semin. Radiat. Oncol. 2021, 31, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Grce, M.; Husnjak, K.; Bozikov, J.; Magdic, L.; Zlacki, M.; Lukac, J.; Fistonic, I.; Sikanic-Dugic, N.; Pavelic, K. Evaluation of Genital Human Papillomavirus Infections by Polymerase Chain Reaction among Croatian Women. Anticancer Res. 2001, 21, 579–584. [Google Scholar] [PubMed]
- Milutin-Gasperov, N.; Sabol, I.; Halec, G.; Matovina, M.; Grce, M. Retrospective Study of the Prevalence of High-Risk Human Papillomaviruses among Croatian Women. Coll. Antropol. 2007, 31 (Suppl. S2), 89–96. [Google Scholar] [PubMed]
- Kaliterna, V.; Kaliterna, M.; Pejković, L.; Drmić Hofman, I.; Anđelinović, Š. Prevalence and Genotyping of the Human Papillomavirus in the Cervical Specimens among Women of Southern Croatia (Dalmatia County). Cent. Eur. J. Public Health 2013, 21, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Kaliterna, V.; Kaliterna, P.; Pejkovic, L.; Vulic, R.; Zanchi, L.; Cerskov, K. Prevalence of Human Papillomavirus (HPV) among Females in the General Population of the Split and Dalmatia County and Its Association with Genital Microbiota and Infections: A Prospective Study. Viruses 2023, 15, 443. [Google Scholar] [CrossRef] [PubMed]
- Freire-Salinas, J.; Benito, R.; Azueta, A.; Gil, J.; Mendoza, C.; Nicolás, M.; García-Berbel, P.; Algarate, S.; Gómez-Román, J. Genotype Distribution Change After Human Papillomavirus Vaccination in Two Autonomous Communities in Spain. Front. Cell. Infect. Microbiol. 2021, 11, 633162. [Google Scholar] [CrossRef] [PubMed]
- García, F.R.; Norenhag, J.; Edfeldt, G.; Cheng, L.; Hugerth, L.W.; Pennhag, A.A.L.; Schuppe-Koistinen, I.; Engstrand, L.; Olovsson, M.; Du, J. Prevalence of the Human Papillomavirus (HPV) Types among Cervical Dysplasia Women Attending a Gynaecological Clinic in Sweden. BJC Rep. 2023, 1, 11. [Google Scholar] [CrossRef]
- Ährlund-Richter, A.; Cheng, L.; Hu, Y.O.O.; Svensson, M.; Pennhag, A.A.L.; Ursu, R.G.; Haeggblom, L.; Grün, N.; Ramqvist, T.; Engstrand, L.; et al. Changes in Cervical Human Papillomavirus (HPV) Prevalence at a Youth Clinic in Stockholm, Sweden, a Decade After the Introduction of the HPV Vaccine. Front. Cell. Infect. Microbiol. 2019, 9, 59. [Google Scholar] [CrossRef] [PubMed]
- Kahn, J.A.; Brown, D.R.; Ding, L.; Widdice, L.E.; Shew, M.L.; Glynn, S.; Bernstein, D.I. Vaccine-Type Human Papillomavirus and Evidence of Herd Protection After Vaccine Introduction. Pediatrics 2012, 130, e249–e256. [Google Scholar] [CrossRef] [PubMed]
- Pimenoff, V.N.; Gray, P.; Louvanto, K.; Eriksson, T.; Lagheden, C.; Söderlund-Strand, A.; Dillner, J.; Lehtinen, M. Ecological Diversity Profiles of Non-Vaccine-Targeted HPVs after Gender-Based Community Vaccination Efforts. Cell Host Microbe 2023, 31, 1921–1929.e3. [Google Scholar] [CrossRef] [PubMed]
- Sabeena, S.; Ravishankar, N. Postvaccination Prevalence of Vaccine-Human Papillomavirus (vHPV) Genotypes among the Target Population: A Systematic Review and Meta-Analysis. J. Med. Virol. 2021, 93, 4659–4667. [Google Scholar] [CrossRef] [PubMed]
- Feiring, B.; Laake, I.; Christiansen, I.K.; Hansen, M.; Stålcrantz, J.; Ambur, O.H.; Magnus, P.; Jonassen, C.M.; Trogstad, L. Substantial Decline in Prevalence of Vaccine-Type and Nonvaccine-Type Human Papillomavirus (HPV) in Vaccinated and Unvaccinated Girls 5 Years After Implementing HPV Vaccine in Norway. J. Infect. Dis. 2018, 218, 1900–1910. [Google Scholar] [CrossRef] [PubMed]
- Carozzi, F.; Puliti, D.; Ocello, C.; Anastasio, P.S.; Moliterni, E.A.; Perinetti, E.; Serradell, L.; Burroni, E.; Confortini, M.; Mantellini, P.; et al. Monitoring Vaccine and Non-Vaccine HPV Type Prevalence in the Post-Vaccination Era in Women Living in the Basilicata Region, Italy. BMC Infect. Dis. 2018, 18, 38. [Google Scholar] [CrossRef] [PubMed]
- Mesher, D.; Soldan, K.; Lehtinen, M.; Beddows, S.; Brisson, M.; Brotherton, J.M.L.; Chow, E.P.F.; Cummings, T.; Drolet, M.; Fairley, C.K.; et al. Population-Level Effects of Human Papillomavirus Vaccination Programs on Infections with Nonvaccine Genotypes. Emerg. Infect. Dis. 2016, 22, 1732–1740. [Google Scholar] [CrossRef] [PubMed]
- Cornall, A.M.; Phillips, S.; Cummins, E.; Garland, S.M.; Tabrizi, S.N. In Vitro Assessment of the Effect of Vaccine-Targeted Human Papillomavirus (HPV) Depletion on Detection of Non-Vaccine HPV Types: Implications for Post-Vaccine Surveillance Studies. J. Virol. Methods 2015, 214, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Patel, C.; Brotherton, J.M.; Pillsbury, A.; Jayasinghe, S.; Donovan, B.; Macartney, K.; Marshall, H. The Impact of 10 Years of Human Papillomavirus (HPV) Vaccination in Australia: What Additional Disease Burden Will a Nonavalent Vaccine Prevent? Eurosurveillance 2018, 23, 1700737. [Google Scholar] [CrossRef] [PubMed]
- Palmer, T.J.; Kavanagh, K.; Cuschieri, K.; Cameron, R.; Graham, C.; Wilson, A.; Roy, K. Invasive Cervical Cancer Incidence Following Bivalent Human Papillomavirus Vaccination: A Population-Based Observational Study of Age at Immunization, Dose, and Deprivation. JNCI J. Natl. Cancer Inst. 2024, djad263. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Huu, N.-H.; Thilly, N.; Derrough, T.; Sdona, E.; Claudot, F.; Pulcini, C.; Agrinier, N. HPV Policy working group Human Papillomavirus Vaccination Coverage, Policies, and Practical Implementation across Europe. Vaccine 2020, 38, 1315–1331. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Diaz, M.; Barrionuevo-Rosas, L.; Herrero, R.; Bray, F.; Bosch, F.X.; de Sanjosé, S.; Castellsagué, X. Global Estimates of Human Papillomavirus Vaccination Coverage by Region and Income Level: A Pooled Analysis. Lancet Glob. Health 2016, 4, e453–e463. [Google Scholar] [CrossRef] [PubMed]
- Karafillakis, E.; Simas, C.; Jarrett, C.; Verger, P.; Peretti-Watel, P.; Dib, F.; De Angelis, S.; Takacs, J.; Ali, K.A.; Pastore Celentano, L.; et al. HPV Vaccination in a Context of Public Mistrust and Uncertainty: A Systematic Literature Review of Determinants of HPV Vaccine Hesitancy in Europe. Hum. Vaccines Immunother. 2019, 15, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- La Vecchia, C.; Boccia, S. Oral Contraceptives, Human Papillomavirus and Cervical Cancer. Eur. J. Cancer Prev. 2014, 23, 110–112. [Google Scholar] [CrossRef] [PubMed]
- Berrington De González, A.; Sweetland, S.; Green, J. Comparison of Risk Factors for Squamous Cell and Adenocarcinomas of the Cervix: A Meta-Analysis. Br. J. Cancer 2004, 90, 1787–1791. [Google Scholar] [CrossRef] [PubMed]
- Daly, S.F.; Doyle, M.; English, J.; Turner, M.; Clinch, J.; Prendiville, W. Can the Number of Cigarettes Smoked Predict High-Grade Cervical Intraepithelial Neoplasia among Women with Mildly Abnormal Cervical Smears? Am. J. Obstet. Gynecol. 1998, 179, 399–402. [Google Scholar] [CrossRef] [PubMed]
| Variable | Normal/ASCUS (N = 236) N (%) | LSIL+ (N = 85) N (%) | p-Value | |
|---|---|---|---|---|
| Age groups | 18–30 | 67 (28.4) | 29 (34.1) | 0.2580 |
| 31–45 | 117 (49.6) | 44 (51.8) | ||
| 46+ | 52 (22) | 12 (14.1) | ||
| Smoking history | Non-smoker | 175 (74.2) | 52 (61.2) | 0.0357 |
| Smoker | 60 (25.4) | 31 (36.5) | ||
| Not specified | 1 (0.4) | 2 (2.3) | ||
| Number of cigarettes per day | Not reported | 3 (1.3) | 9 (10.6) | 0.0004 |
| Non-smoker | 175 (74.1) | 52 (61.2) | ||
| 1–5/day | 18 (7.6) | 5 (5.9) | ||
| 6–10/day | 16 (6.8) | 12 (14.1) | ||
| 11–20/day | 24 (10.2) | 7 (8.2) | ||
| Parity | Nulliparous | 93 (39.4) | 34 (40) | 0.4861 |
| 1–2 | 119 (50.4) | 46 (54.1) | ||
| 3+ | 24 (10.2) | 5 (5.9) | ||
| Abortions | Yes | 203 (86.0) | 68 (80.0) | 0.1903 |
| No | 33 (14.0) | 17 (20.0) | ||
| Cancer history | Nothing reported | 140 (59.3) | 57 (67.1) | 0.2098 |
| Family or personal history | 96 (40.7) | 28 (32.9) | ||
| HPV positivity 1 | Negative | 157 (66.5) | 17 (20.0) | <0.0001 |
| Positive | 79 (33.5) | 68 (80.0) | ||
| LR-HPV | 2 (0.8) | 3 (3.5) | ||
| Non-vaccine types | 38 (16.1) | 33 (38.8) | ||
| HR-HPV | 39 (16.5) | 32 (37.6) |
| HPV Positivity 1 | Normal (N = 236) N (%) | LSIL+ (N = 85) N (%) | Total (N = 321) N (%) |
|---|---|---|---|
| Negative | 157 (66.5) | 17 (20.0) | 174 (54.2) |
| Any HPV | 79 (33.5) | 68 (80.0) | 147 (45.8) |
| Non-vaccine types | 38 (16.1) | 33 (38.8) | 71 (22.1) |
| LR-HPV (HPV 6/11) | 3 (1.3) | 3 (3.5) | 6 (1.9) |
| HR-HPV | 39 (16.5) | 32 (37.6) | 71 (22.1) |
| HPV 16 | 21 (8.9) | 19 (22.4) | 40 (12.5) |
| HPV 18 | 2 (0.8) | 2 (2.4) | 4 (1.2) |
| HPV 31 | 5 (2.1) | 2 (2.4) | 7 (2.2) |
| HPV 33 | 2 (0.8) | 4 (4.7) | 6 (1.9) |
| HPV 45 | 5 (2.1) | 2 (2.4) | 7 (2.2) |
| HPV 52 | 8 (3.4) | 6 (7.1) | 14 (4.4) |
| HPV 58 | 2 (0.8) | 1 (1.2) | 3 (0.9) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pešut, E.; Šimić, I.; Fureš, R.; Milutin Gašperov, N.; Lež, C.; Feratović, F.; Kukina Žvigač, T.; Grce, M.; Erceg Ivkošić, I.; Sabol, I. Monitoring HPV Prevalence and Risk Cofactors for Abnormal Cytology in the Post-Vaccination Period among Croatian Women. Viruses 2024, 16, 642. https://doi.org/10.3390/v16040642
Pešut E, Šimić I, Fureš R, Milutin Gašperov N, Lež C, Feratović F, Kukina Žvigač T, Grce M, Erceg Ivkošić I, Sabol I. Monitoring HPV Prevalence and Risk Cofactors for Abnormal Cytology in the Post-Vaccination Period among Croatian Women. Viruses. 2024; 16(4):642. https://doi.org/10.3390/v16040642
Chicago/Turabian StylePešut, Ena, Ivana Šimić, Rajko Fureš, Nina Milutin Gašperov, Cvjetko Lež, Fabijan Feratović, Tomica Kukina Žvigač, Magdalena Grce, Ivana Erceg Ivkošić, and Ivan Sabol. 2024. "Monitoring HPV Prevalence and Risk Cofactors for Abnormal Cytology in the Post-Vaccination Period among Croatian Women" Viruses 16, no. 4: 642. https://doi.org/10.3390/v16040642
APA StylePešut, E., Šimić, I., Fureš, R., Milutin Gašperov, N., Lež, C., Feratović, F., Kukina Žvigač, T., Grce, M., Erceg Ivkošić, I., & Sabol, I. (2024). Monitoring HPV Prevalence and Risk Cofactors for Abnormal Cytology in the Post-Vaccination Period among Croatian Women. Viruses, 16(4), 642. https://doi.org/10.3390/v16040642










