EV-A71 Mechanism of Entry: Receptors/Co-Receptors, Related Pathways and Inhibitors
Abstract
1. Introduction
2. Enterovirus A71 Receptors/Co-Receptors
2.1. hSCARB-2
2.2. PSGL-1
2.3. Sialylated Glycans
2.4. Anx 2
2.5. HS
2.6. HSP90
2.7. CypA
2.8. Vimentin
2.9. Nucleolin
2.10. Fibronectin
2.11. Prohibitin
2.12. hWARS
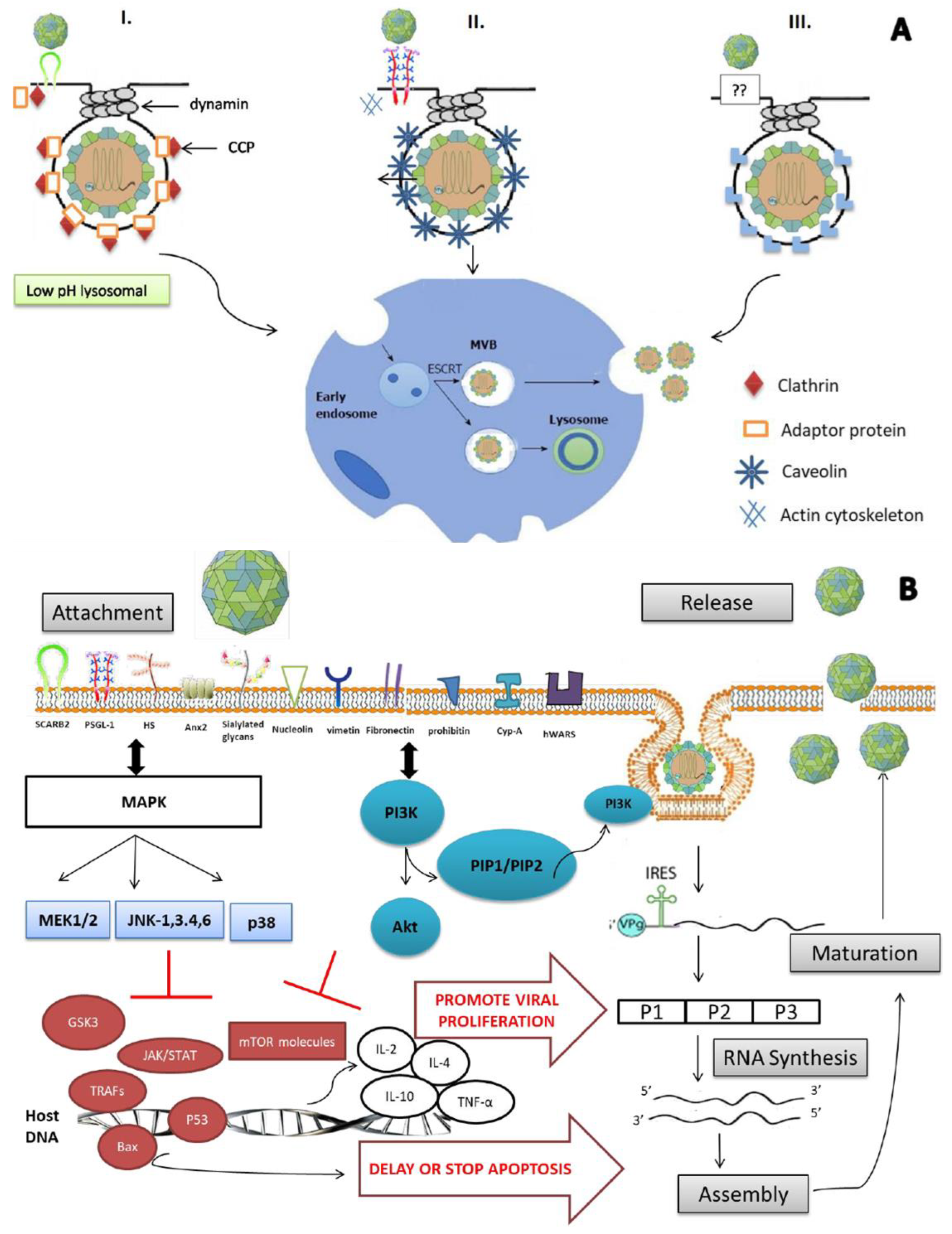
3. EV-A71 Entry into Cells
3.1. Adsorption to Cells
3.2. hSCARB-2 Uses Clathrin-Mediated Endocytosis (CME)
3.3. PSGL-1 Uses Caveolae-Mediated Endocytosis (CaME)
3.4. Endophilin-A2-Mediated Endocytosis
3.5. Relevant Signaling Pathways
3.5.1. MAPK Signaling Pathway
3.5.2. PI3K/Akt Signaling Pathway
3.5.3. IFN Signaling Pathway
3.5.4. Apoptosis Signaling Pathway
3.5.5. Autophagy Signaling Pathway
4. Drug Development Targeting EV-A71 Entry
4.1. Virus Receptor Inhibitors
4.2. Virus Capsid Protein Inhibitors
4.3. Related Signaling Pathway Inhibitors
4.4. Other Strategies Blocking Entry
| Drug Name | Target | EC50 | Cell Line/Animal Model | Ref. |
|---|---|---|---|---|
| Host receptor inhibitors | ||||
| Soluble form of receptors | hSCARB-2/PSGL-1 | — | RD/L-PSGL-1.1 | [24,34] |
| SP40 and L-SP40 | Nucleolin/Anx-2/hSCARB-2/hWARS/HS | 6–9.3 µM | RD/Hela/HT-29/Vero/neonatal mice | [114,115,116] |
| Lactoferrin bovine/human | HS | 10.5–24.5 µg/mL 103.3–185 µg/mL | RD | [119] |
| HS mimetics | HS | 205 µg/mL | Vero | [111] |
| Heparin/heparan | 290 µg/mL | |||
| sulfates/pentosan | 238 µg/mL | |||
| Capsid protein inhibitors | ||||
| Pyridyl Imidazolinone | ||||
| VP1 | 0.31–1.26 µM | Vero/MRC-5 | [123,124] |
| 0.51–1.37 µM | |||
| 0.04 µM | |||
| 0.339 µM | RD | [126] | |
| 0.04 µM | |||
| 0.011 µM | |||
| 0.04 µM | |||
| 0.13 µM | |||
| 0.0012 µM | |||
| 0.008 µM | |||
| 0.0088 µM | |||
| — | [125] | ||
| — | [128] | ||
| Pleconaril | VP1 | 0.13–0.54 µg/mL | RD/1-day-old ICR | [129] |
| Pirodavir | 0.36–0.727 µg/mL | RD | [130] | |
| Vapendavir | 0.498–0.957 µg/mL | [131] | ||
| NF449 | Capsid protein | 6.5 µM | [132] | |
| Kappa carrageenan | VP1/HS | — | Vero | [136] |
| Sertraline | EV-A71 entry/uncoating | 1.92/1.67 µM | RD/Hela | [138] |
| Ni(2+) chitosan beads | Capsid protein | 5.5 µM | Vero | [141] |
| CPZ and DNS | — | RD | [76] | |
| PTC-209HBr | hSCARB2/VP1 | 0.79–2.92 µM | RD/Hela/HEK293T/Vero | [142] |
| Related signaling pathway inhibitors | ||||
| Saururus Chinensis | MEK/ERK, AP-1, PI3K | — | Vero/SK--SH | [147] |
| U0126 | 3.45/3.98 µM | Mouse model | [148] | |
| Formononetin | PI3K/Akt | — | Vero | [149] |
| GS-9620 | MEK/ERK | 73.10 µM | SK-N-SH | [150] |
| Berberine | bax apoptosis | — | [151] | |
| Selenium | Akt | — | [106] | |
| Picochlorumsp.122 | P53, STAT1 | 13.7 µg/ml | RD | [152] |
| Durvillae A. | — | 2-day-old Balb/c | [105] | |
| Other strategies blocking entry | ||||
| Lipid rafts | Capsid protein | 31.5 µg/mL | Vero | [154] |
| Monoclonal antibodies | ||||
| EV-A71 endocytosis | — | RD/Vero/Jurkat T cells/2-day-old BALB/c mice | [155] |
| VP1 | — | Hela | [156] |
| VP1 | — | 2-week-old AG129 mice | [158] |
| VP3 | — | [159] | |
| siRNA | ||||
| VP1 GH Loop | — | Vero/Hela/HepG2/293T/L929 | [160] |
| hSCARB-2 | — | 293T/L929 | [161] |
| MEK/ERK | — | HEK293T | [88] |
5. Discussion and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALV | avian leucosis virus |
| Anx-2 | annexin 2 |
| AP-1 | activating protein-1 |
| ATM/ATR | ataxia-telangiectasia-mutated/ataxia telangiectasia |
| CCP | clathrin-coated pit |
| CCV | clathrin-coated vesicle |
| CVA 7/14/16 | coxsackievirus A 7/14/16 |
| CME | clathrin-mediated endocytosis |
| CaME | caveolae-mediated endocytosis |
| CPE | cytopathic effect |
| CPZ | chlorpromazine |
| CypA | cyclophilin A |
| DNS | dynasore |
| EMT | epithelial–mesenchymal transition |
| ESCRT-MVBs | endosomal sorting complex required for transport to multivesicular bodies |
| EV-A71 | enterovirus 71 |
| FDA | food and drug administration |
| GSK3 | glycogen synthase kinase-3 |
| HCV | hepatitis C virus |
| HIV | human immunodeficiency virus |
| HS | heparan sulfate |
| hSCARB-2 | human scavenger receptor class B, member 2 |
| HSP90 | heat shock protein 90 |
| HSV | herpex simplex virus |
| hWARS | human tryptophanyl-tRNA synthetase |
| IFN | interferon |
| IP | immunoprecipitation |
| IRES | internal ribosome sites |
| JAK | Janus kinase |
| mAbs | monoclonal antibodies |
| MAPKs | itogen-activated protein kinases |
| non-PB | non-PSGL-1 binding |
| ORF | open reading frame |
| PCR | polymerase chain reaction |
| PB | PSGL-1 binding |
| PI3K/Akt | phosphatidylinositol-3-kinase/protein kinase B |
| PKC ζ | protein kinase C zeta |
| PSGL-1 | P-selectin glycoprotein ligand 1 |
| RdRp | RNA dependent RNA-polymerase |
| RNA | ribonucleic acid |
| RSV | respiratory syncytial virus |
| SA | sialic acid |
| SG | sialic glycans |
| SI | selectivity index |
| SNPs | single nucleotide polymorphisms |
| siRNA | small interfering RNA |
| STAT | signal transducer and activator of transcription |
| TLR7 | toll-like receptor 7 |
| TRAF3 | TNF receptor-associated factor 3 |
References
- Zell, R. Picornaviridae-the ever-growing virus family. Arch. Virol. 2018, 163, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Plevka, P.; Perera, R.; Cardosa, J.; Kuhn, R.J.; Rossmann, M.G. Structure determination of enterovirus 71. Acta Crystallogr. D. Biol. Crystallogr. 2012, 68, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Liu, Y.; Ma, H.C.; Paul, A.V.; Wimmer, E. Picornavirus morphogenesis. Microbiol. Mol. Biol. Rev. 2014, 78, 418–437. [Google Scholar] [CrossRef] [PubMed]
- Horsley, E.; Just, E.; Torres, C.; Huhtinen, E.; Forssman, B.; Slade, R. Enterovirus 71 outbreak in Northern Sydney, 2013: Case series and initial response. J. Paediatr. Child Health 2014, 50, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Duong, V.; Mey, C.; Eloit, M.; Zhu, H.; Danet, L.; Huang, Z.; Zou, G.; Tarantola, A.; Cheval, J.; Perot, P.; et al. Molecular epidemiology of human enterovirus 71 at the origin of an epidemic of fatal hand, foot and mouth disease cases in Cambodia. Emerg. Microbes Infect. 2016, 5, e104. [Google Scholar] [CrossRef] [PubMed]
- Xing, W.; Liao, Q.; Viboud, C.; Zhang, J.; Sun, J.; Wu, J.T.; Chang, Z.; Liu, F.; Fang, V.J.; Zheng, Y.; et al. Hand, foot, and mouth disease in China, 2008–2012: An epidemiological study. Lancet Infect. Dis. 2014, 14, 308–318. [Google Scholar] [CrossRef]
- Tagaya, I.; Takayama, R.; Hagiwara, A. A large-scale epidemic of hand, foot and mouth disease associated with enterovirus 71 infection in Japan in 1978. Jpn. J. Med. Sci. Biol. 1981, 34, 191–196. [Google Scholar] [CrossRef]
- Chua, K.B.; Kasri, A.R. Hand foot and mouth disease due to enterovirus 71 in Malaysia. Virol. Sin. 2011, 26, 221–228. [Google Scholar] [CrossRef]
- Lim, C.T.; Jiang, L.; Ma, S.; James, L.; Ang, L.W. Basic reproduction number of coxsackievirus type A6 and A16 and enterovirus 71: Estimates from outbreaks of hand, foot and mouth disease in Singapore, a tropical city-state. Epidemiol. Infect. 2016, 144, 1028–1034. [Google Scholar] [CrossRef]
- Baek, K.; Yeo, S.; Lee, B.; Park, K.; Song, J.; Yu, J.; Rheem, I.; Kim, J.; Hwang, S.; Choi, Y.; et al. Epidemics of enterovirus infection in Chungnam Korea, 2008 and 2009. Virol. J. 2011, 8, 297. [Google Scholar] [CrossRef]
- Chia, M.Y.; Chiang, P.S.; Chung, W.Y.; Luo, S.T.; Lee, M.S. Epidemiology of enterovirus 71 infections in Taiwan. Pediatr. Neonatol. 2014, 55, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Linsuwanon, P.; Puenpa, J.; Huang, S.W.; Wang, Y.F.; Mauleekoonphairoj, J.; Wang, J.R.; Poovorawan, Y. Epidemiology and seroepidemiology of human enterovirus 71 among Thai populations. J. Biomed. Sci. 2014, 21, 16. [Google Scholar] [CrossRef] [PubMed]
- Khanh, T.H.; Sabanathan, S.; Thanh, T.T.; Thoa, L.P.K.; Thuong, T.C.; Hang, V.T.; Farrar, J.F.; Hien, T.T.; Chau, N.V.; van Doorn, H.R. Enterovirus 71-associated hand, foot, and mouth disease, Southern Vietnam, 2011. Emerg. Infect. Dis. 2012, 18, 2002–2005. [Google Scholar] [CrossRef]
- Puenpa, J.; Wanlapakorn, N.; Vongpunsawad, S.; Poovorawan, Y. The history of enterovirus A71 outbreaks and molecular epidemiology in the asia-pacific region. J. Biomed. Sci. 2019, 26, 75. [Google Scholar] [CrossRef]
- Li, M.L.; Shih, S.R.; Tolbert, B.S.; Brewer, G. Enterovirus A71 vaccines. Vaccines 2021, 9, 199. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Zhao, Y.; Kotecha, A.; Fry, E.E.; Kelly, J.T.; Wang, X.; Rao, Z.; Rowlands, D.J.; Ren, J.; Stuart, D.I. Unexpected mode of engagement between enterovirus 71 and its receptor SCARB2. Nat. Microbiol. 2019, 4, 414–419. [Google Scholar] [CrossRef]
- Sangar, D.V. The replication of picornaviruses. J. Gen Virol. 1979, 45, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.Y.; Shih, S.R. Cell and tissue tropism of enterovirus 71 and other enteroviruses infections. J. Biomed. Sci. 2014, 21, 18. [Google Scholar] [CrossRef]
- Chang, C.S.; Liao, C.C.; Liou, A.T.; Chang, Y.S.; Chang, Y.T.; Tzeng, B.H.; Chen, C.C.; Shih, C. Enterovirus 71 targets the cardiopulmonary system in a robust oral infection mouse model. Sci. Rep. 2019, 9, 11108. [Google Scholar] [CrossRef]
- Lin, Y.W.; Wang, S.W.; Tung, Y.Y.; Chen, S.H. Enterovirus 71 infection of human dendritic cells. Exp. Biol. Med. 2009, 234, 1166–1173. [Google Scholar] [CrossRef]
- Huang, H.I.; Lin, J.Y.; Chen, S.H. EV71 infection induces IFNβ expression in neural cells. Viruses 2019, 11, 1121. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Koike, S. Cellular receptors for enterovirus A71. J. Biomed. Sci. 2020, 27, 23. [Google Scholar] [CrossRef]
- Calvo, D.; Dopazo, J.; Vega, M.A. The CD36, CLA-1 (CD36L1), and LIMPII (CD36L2) gene family: Cellular distribution, chromosomal location, and genetic evolution. Genomics 1995, 25, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Alquraini, A.; Khoury, J.E. Scavenger receptors. Curr. Biol. 2020, 30, R790–R795. [Google Scholar] [CrossRef] [PubMed]
- Yamayoshi, S.; Fujii, K.; Koike, S. Receptors for enterovirus 71. Emerg. Microbes Infect. 2014, 3, e53. [Google Scholar] [CrossRef]
- Zhu, J.; Chen, N.; Zhou, S.; Zheng, K.; Sun, L.; Zhang, Y.; Cao, L.; Zhang, X.; Xiang, Q.; Chen, Z.; et al. Severity of enterovirus A71 infection in a human SCARB2 knock-in mouse model is dependent on infectious strain and route. Emerg. Microbes Infect. 2018, 7, 205. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, P.; Wang, N.; Zhang, J.; Li, J.; Guo, H.; Yin, X.; Rao, Z.; Wang, X.; Zhang, L. The binding of a monoclonal antibody to the apical region of SCARB2 blocks EV71 infection. Protein Cell 2017, 8, 590–600. [Google Scholar] [CrossRef]
- Nishimura, Y.; Shimizu, H. Cellular receptors for human enterovirus species a. Front. Microbiol. 2012, 3, 105. [Google Scholar] [CrossRef]
- Yamayoshi, S.; Iizuka, S.; Yamashita, T.; Minagawa, H.; Mizuta, K.; Okamoto, M.; Nishimura, H.; Sanjoh, K.; Katsushima, N.; Itagaki, T.; et al. Human SCARB2-dependent infection by coxsackievirus A7, A14, and A16 and enterovirus 71. J. Virol. 2012, 86, 5686–5696. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.W.; Lin, H.Y.; Tsou, Y.L.; Chitra, E.; Hsiao, K.N.; Shao, H.Y.; Liu, C.C.; Sia, C.; Chong, P.; Chow, Y.H. Human SCARB2-mediated entry and endocytosis of EV71. PLoS ONE 2012, 7, e30507. [Google Scholar] [CrossRef]
- Yen, T.Y.; Shih, W.L.; Huang, Y.C.; Lee, J.T.; Huang, L.M.; Chang, L.Y. Polymorphisms in enterovirus 71 receptors associated with susceptibility and clinical severity. PLoS ONE 2018, 13, e0206769. [Google Scholar] [CrossRef] [PubMed]
- Laszik, Z.; Jansen, P.J.; Cummings, R.D.; Tedder, T.F.; McEver, R.P.; Moore, K.L. P-selectin glycoprotein ligand-1 is broadly expressed in cells of myeloid, lymphoid, and dendritic lineage and in some nonhematopoietic cells. Blood 1996, 88, 3010–3021. [Google Scholar] [CrossRef] [PubMed]
- Somers, W.S.; Tang, J.; Shaw, G.D.; Camphausen, R.T. Insights into the molecular basis of leukocyte tethering and rolling revealed by structures of P- and E-selectin bound to SLe(X) and PSGL-1. Cell 2000, 103, 467–479. [Google Scholar] [CrossRef]
- Nishimura, Y.; Shimojima, M.; Tano, Y.; Miyamura, T.; Wakita, T.; Shimizu, H. Human P-selectin glycoprotein ligand-1 is a functional receptor for enterovirus 71. Nat. Med. 2009, 15, 794–797. [Google Scholar] [CrossRef]
- Nishimura, Y.; Wakita, T.; Shimizu, H. Tyrosine sulfation of the amino terminus of PSGL-1 is critical for enterovirus 71 infection. PLoS Pathog. 2010, 6, e1001174. [Google Scholar] [CrossRef]
- Nishimura, Y.; Lee, H.; Hafenstein, S.; Kataoka, C.; Wakita, T.; Bergelson, J.M.; Shimizu, H. Enterovirus 71 binding to PSGL-1 on leukocytes: VP1-145 acts as a molecular switch to control receptor interaction. PLoS Pathog. 2013, 9, e1003511. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Tijsma, A.; Mirabelli, C.; Baggen, J.; Wahedi, M.; Franco, D.; De Palma, A.; Leyssen, P.; Verbeken, E.; van Kuppeveld, F.J.M.; et al. Intra-host emergence of an enterovirus A71 variant with enhanced PSGL1 usage and neurovirulence. Emerg. Microbes Infect. 2019, 8, 1076–1085. [Google Scholar] [CrossRef] [PubMed]
- Zarbock, A.; Müller, H.; Kuwano, Y.; Ley, K. PSGL-1-dependent myeloid leukocyte activation. J. Leukoc. Biol. 2009, 86, 1119–1124. [Google Scholar] [CrossRef]
- Gu, Y.Y.; Shi, K.; Yao, S.; Yang, X.; Liu, Y.H.; Tang, L.; Dang, Y.W.; Chen, G.; Feng, Z.B.; Pan, H.B. Morphological characteristics of fatal pediatric hand, foot and mouth disease: A clinicopathological study with related receptors of EV71. Pathol. Res. Pract. 2017, 213, 1144–1151. [Google Scholar] [CrossRef]
- Maginnis, M.S. Virus-Receptor Interactions: The Key to Cellular Invasion. J. Mol. Biol. 2018, 430, 2590–2611. [Google Scholar] [CrossRef]
- Varki, N.M.; Varki, A. Diversity in cell surface sialic acid presentations: Implications for biology and disease. Lab. Investig. 2007, 87, 851–857. [Google Scholar] [CrossRef]
- Yang, B.; Chuang, H.; Yang, K.D. Sialylated glycans as receptor and inhibitor of enterovirus 71 infection to DLD-1 intestinal cells. Virol. J. 2009, 6, 141. [Google Scholar] [CrossRef] [PubMed]
- Rintala-Dempsey, A.C.; Rezvanpour, A.; Shaw, G.S. S100-annexin complexes--structural insights. FEBS J. 2008, 275, 4956–4966. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.R.; Skeate, J.G.; Kast, W.M. Annexin A2 in Virus Infection. Front. Microbiol. 2018, 9, 2954. [Google Scholar] [CrossRef]
- Gerke, V.; Moss, S.E. Annexins: From structure to function. Physiol. Rev. 2002, 82, 331–371. [Google Scholar] [CrossRef]
- Yang, S.L.; Chou, Y.T.; Wu, C.N.; Ho, M.S. Annexin II binds to capsid protein VP1 of enterovirus 71 and enhances viral infectivity. J. Virol. 2011, 85, 11809–11820. [Google Scholar] [CrossRef] [PubMed]
- Kjellén, L.; Lindahl, U. Proteoglycans: Structures and interactions. Annu. Rev. Biochem. 1991, 60, 443–475. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, S.; Lamanna, W.C.; Esko, J.D. Heparan sulfate proteoglycans. Cold Spring Harb. Perspect. Biol. 2011, 3, a004952. [Google Scholar] [CrossRef]
- Cagno, V.; Tseligka, E.D.; Jones, S.T.; Tapparel, C. Heparan Sulfate Proteoglycans and Viral Attachment: True Receptors or Adaptation Bias? Viruses 2019, 11, 596. [Google Scholar] [CrossRef]
- Tan, C.W.; Poh, C.L.; Sam, I.C.; Chan, Y.F. Enterovirus 71 uses cell surface heparan sulfate glycosaminoglycan as an attachment receptor. J. Virol. 2013, 87, 611–620. [Google Scholar] [CrossRef]
- Kobayashi, K.; Sudaka, Y.; Takashino, A.; Imura, A.; Fujii, K.; Koike, S. Amino acid variation at VP1-145 of enterovirus 71 determines attachment receptor usage and neurovirulence in human scavenger receptor B2 transgenic mice. J. Virol. 2018, 92, e00681-18. [Google Scholar] [CrossRef] [PubMed]
- Neckers, L.; Ivy, S.P. Heat shock protein 90. Curr. Opin. Oncol. 2003, 15, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Tsou, Y.L.; Lin, Y.W.; Chang, H.W.; Lin, H.Y.; Shao, H.Y.; Yu, S.L.; Liu, C.C.; Chitra, E.; Sia, C.; Chow, Y.H. Heat shock protein 90: Role in enterovirus 71 entry and assembly and potential target for therapy. PLoS ONE 2013, 8, e77133. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Heitman, J. The cyclophilins. Genome Biol. 2005, 6, 226. [Google Scholar] [CrossRef]
- Olejnik, P.; Nuc, K. Cyclophilins—proteins with many functions. (Article in Polish). Postepy. Biochem. 2018, 64, 46–54. [Google Scholar] [CrossRef]
- Zhou, D.; Mei, Q.; Li, J.; He, H. Cyclophilin A and viral infections. Biochem. Biophys. Res. Commun. 2012, 424, 647–650. [Google Scholar] [CrossRef]
- Liu, J.; Farmer, J.D., Jr.; Lane, W.S.; Friedman, J.; Weissman, I.; Schreiber, S.L. Calcineurin is a common target of cyclophilin-cyclosporin A and FKBP-FK506 complexes. Cell 1991, 66, 807–815. [Google Scholar] [CrossRef]
- Qing, J.; Wang, Y.; Sun, Y.; Huang, J.; Yan, W.; Wang, J.; Su, D.; Ni, C.; Li, J.; Rao, Z.; et al. Cyclophilin A associates with enterovirus-71 virus capsid and plays an essential role in viral infection as an uncoating regulator. PLoS Pathog. 2014, 10, e1004422. [Google Scholar] [CrossRef]
- Goldman, R.D.; Khuon, S.; Chou, Y.H.; Opal, P.; Steinert, P.M. The function of intermediate filaments in cell shape and cytoskeletal integrity. J. Cell Biol. 1996, 134, 971–983. [Google Scholar] [CrossRef]
- Sukhotnik, I.; Shahar, Y.B.; Pollak, Y.; Dorfman, T.; Shefer, H.K.; Assi, Z.E.; Mor-Vaknin, N.; Coran, A.G. The role of intermediate filaments in maintaining integrity and function of intestinal epithelial cells after massive bowel resection in a rat. Pediatr. Surg. Int. 2018, 3, 217–225. [Google Scholar] [CrossRef]
- Du, N.; Cong, H.; Tian, H.; Zhang, H.; Zhang, W.; Song, L.; Tien, P. Cell surface vimentin is an attachment receptor for enterovirus 71. J. Virol. 2014, 88, 5816–5833. [Google Scholar] [CrossRef] [PubMed]
- Ginisty, H.; Sicard, H.; Roger, B.; Bouvet, P. Structure and functions of nucleolin. J. Cell Sci. 1999, 112, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Mastrangelo, P.; Griffin, J.K.; Moraes, T.J.; Hegele, R.G. Respiratory syncytial virus receptor expression in the mouse and viral tropism. Histol. Histopathol. 2015, 30, 401–411. [Google Scholar] [PubMed]
- Mastrangelo, P.; Chin, A.A.; Tan, S.; Jeon, A.H.; Ackerley, C.A.; Siu, K.K.; Lee, J.E.; Hegele, R.G. Identification of RSV Fusion Protein Interaction Domains on the Virus Receptor, Nucleolin. Viruses 2021, 13, 261. [Google Scholar] [CrossRef]
- Hovanessian, A.G. Midkine, a cytokine that inhibits HIV infection by binding to the cell surface expressed nucleolin. Cell Res. 2006, 16, 174–181. [Google Scholar] [CrossRef]
- Su, P.Y.; Wang, Y.F.; Huang, S.W.; Lo, Y.C.; Wang, Y.H.; Wu, S.R.; Shieh, D.B.; Chen, S.H.; Wang, J.R.; Lai, M.D.; et al. Cell surface nucleolin facilitates enterovirus 71 binding and infection. J. Virol. 2015, 89, 4527–4538. [Google Scholar] [CrossRef]
- Pankov, R.; Yamada, K.M. Fibronectin at a glance. J. Cell Sci. 2002, 115, 3861–3863. [Google Scholar] [CrossRef]
- Adams, J.C.; Chiquet-Ehrismann, R.; Tucker, R.P. The evolution of tenascins and fibronectin. Cell Adh. Migr. 2015, 9, 22–33. [Google Scholar] [CrossRef]
- He, Q.Q.; Ren, S.; Xia, Z.C.; Cheng, Z.K.; Peng, N.F.; Zhu, Y. Fibronectin Facilitates Enterovirus 71 Infection by Mediating Viral Entry. J. Virol. 2018, 92, e02251-17. [Google Scholar] [CrossRef]
- Wei, Y.; Chiang, W.C.; Sumpter, R., Jr.; Mishra, P.; Levine, B. Prohibitin 2 Is an Inner Mitochondrial Membrane Mitophagy Receptor. Cell 2017, 168, 224–238. [Google Scholar] [CrossRef]
- Chowdhury, I.; Thompson, W.E.; Thomas, K. Prohibitins role in cellular survival through Ras-Raf-MEK-ERK pathway. J. Cell Physiol. 2014, 229, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Too, I.H.K.; Bonne, I.; Tan, E.L.; Chu, J.J.H.; Alonso, S. Prohibitin plays a critical role in Enterovirus 71 neuropathogenesis. PLoS Pathog. 2018, 14, e1006778. [Google Scholar] [CrossRef] [PubMed]
- Too, I.H.; Yeo, H.; Sessions, O.M.; Yan, B.; Libau, E.A.; Howe, J.L.; Lim, Z.Q.; Suku-Maran, S.; Ong, W.Y.; Chua, K.B.; et al. Enterovirus 71 infection of motor neuron-like NSC-34 cells undergoes a non-lytic exit pathway. Sci. Rep. 2016, 6, 36983. [Google Scholar] [CrossRef] [PubMed]
- Kisselev, L.L. Mammalian tryptophanyl-tRNA synthetases. Biochimie 1993, 75, 1027–1039. [Google Scholar] [CrossRef]
- Yeung, M.L.; Jia, L.; Yip, C.C.Y.; Chan, J.F.W.; Teng, J.L.L.; Chan, K.H.; Cai, J.P.; Zhang, C.; Zhang, A.J.; Wong, W.M.; et al. Human tryptophanyl-tRNA synthetase is an IFN-γ-inducible entry factor for Enterovirus. J. Clin. Investig. 2018, 128, 5163–5177. [Google Scholar] [CrossRef]
- Yuan, M.; Yan, J.; Xun, J.; Chen, C.; Zhang, Y.; Wang, M.; Chu, W.; Song, Z.; Hu, Y.; Zhang, S.; et al. Enhanced human enterovirus 71 infection by endocytosis inhibitors reveals multiple entry pathways by enterovirus causing hand-foot-and-mouth diseases. Virol. J. 2018, 15, 1. [Google Scholar] [CrossRef]
- Dang, M.; Wang, X.; Wang, Q.; Wang, Y.; Lin, J.; Sun, Y.; Li, X.; Zhang, L.; Lou, Z.; Wang, J.; et al. Molecular mechanism of SCARB2-mediated attachment and uncoating of EV71. Protein Cell 2014, 5, 692–703. [Google Scholar] [CrossRef]
- Hussain, K.M.; Leong, K.L.; Ng, M.M.; Chu, J.J. The essential role of clathrin-mediated endocytosis in the infectious entry of human enterovirus 71. J. Biol. Chem. 2011, 286, 309–321. [Google Scholar] [CrossRef]
- Mudhakir, D.; Harashima, H. Learning from the viral journey: How to enter cells and how to overcome intracellular barriers to reach the nucleus. AAPS J. 2009, 11, 65–77. [Google Scholar] [CrossRef]
- Lin, H.Y.; Yang, Y.T.; Yu, S.L.; Hsiao, K.N.; Liu, C.C.; Sia, C.; Chow, Y.H. Caveolar endocytosis is required for human PSGL-1-mediated enterovirus 71 infection. J. Virol. 2013, 87, 9064–9076. [Google Scholar] [CrossRef]
- Parton, R.G.; Howes, M.T. Revisiting caveolin trafficking: The end of the caveosome. J. Cell Biol. 2010, 191, 439–441. [Google Scholar] [CrossRef] [PubMed]
- Hayer, A.; Stoeber, M.; Ritz, D.; Engel, S.; Meyer, H.H.; Helenius, A. Caveolin-1 is ubiquitinated and targeted to intralumenal vesicles in endolysosomes for degradation. J. Cell Biol. 2010, 191, 615–629. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.L.; Liu, Y.G.; Zhou, Y.T.; Zhao, P.; Ren, H.; Xiao, M.; Zhu, Y.Z.; Qi, Z.T. Endophilin-A2-mediated endocytic pathway is critical for enterovirus 71 entry into caco-2 cells. Emerg. Microbes Infect. 2019, 8, 773–786. [Google Scholar] [CrossRef] [PubMed]
- Tung, W.H.; Lee, I.T.; Hsieh, H.L.; Yang, C.M. EV71 induces COX-2 expression via c-Src/PDGFR/PI3K/Akt/p42/p44 MAPK/AP-1 and NF-kappaB in rat brain astrocytes. J. Cell Physiol. 2010, 224, 376–386. [Google Scholar] [CrossRef]
- Bian, L.; Wang, Y.; Liu, Q.; Xia, J.; Long, J.E. Prediction of signaling pathways involved in enterovirus 71 infection by algorithm analysis based on miRNA profiles and their target genes. Arch. Virol. 2015, 160, 173–182. [Google Scholar] [CrossRef]
- Fu, F.; Zhao, J.; Xi, X. Identification of genes involved in enterovirus 71 infected SK-N-SH cells. Int. J. Clin. Exp. Pathol. 2017, 1, 11588–11595. [Google Scholar]
- Pleschka, S. RNA viruses and the mitogenic Raf/MEK/ERK signal transduction cascade. Biol. Chem. 2008, 389, 1273–1282. [Google Scholar] [CrossRef]
- Shi, W.; Hou, X.; Peng, H.; Zhang, L.; Li, Y.; Gu, Z.; Jiang, Q.; Shi, M.; Ji, Y.; Jiang, J. MEK/ERK signaling pathway is required for enterovirus 71 replication in immature dendritic cells. Virol. J. 2014, 11, 227. [Google Scholar] [CrossRef]
- Wang, C.; Wang, P.; Chen, X.; Wang, W.; Jin, Y. Saururus chinensis (Lour.) Baill blocks enterovirus 71 infection by hijacking MEK1-ERK signaling pathway. Antivir. Res. 2015, 119, 47–56. [Google Scholar] [CrossRef]
- Plotnikov, A.; Zehorai, E.; Procaccia, S.; Seger, R. The MAPK cascades: Signaling components, nuclear roles and mechanisms of nuclear translocation. Biochim. Biophys. Acta 2011, 1813, 1619–1633. [Google Scholar] [CrossRef]
- Shi, W.; Hou, X.; Li, X.; Peng, H.; Shi, M.; Jiang, Q.; Liu, X.; Ji, Y.; Yao, Y.; He, C.; et al. Differential gene expressions of the MAPK signaling pathway in enterovirus 71-infected rhabdomyosarcoma cells. Braz. J. Infect. Dis. 2013, 17, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.R.; Chen, Y.Y.; Yang, S.M.; Chen, Y.L.; Horng, J.T. Phosphorylation of PI3K/Akt and MAPK/ERK in an early entry step of enterovirus 71. Life Sci. 2005, 78, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Diehl, N.; Schaal, H. Make yourself at home: Viral hijacking of the PI3K/Akt signaling pathway. Viruses 2013, 5, 3192–3212. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.T.; Liu, H.J. PI3K-Akt signaling and viral infection. Recent Pat. Biotechnol. 2008, 2, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Eaton, H.E.; Saffran, H.A.; Wu, F.W.; Quach, K.; Smiley, J.R. Herpes simplex virus protein kinases US3 and UL13 modulate VP11/12 phosphorylation, virion packaging, and phosphatidylinositol 3-kinase/Akt signaling activity. J. Virol. 2014, 88, 7379–7388. [Google Scholar] [CrossRef]
- Izmailyan, R.; Hsao, J.C.; Chung, C.S.; Chen, C.H.; Su, P.W.; Liao, C.L.; Chang, W. Integrin β1 mediates vaccinia virus entry through activation of PI3K/Akt signaling. J. Virol. 2012, 86, 6677–6687. [Google Scholar] [CrossRef]
- Zhang, Q.; Xie, T.; Mo, G.; Zhang, Z.; Lin, L.; Zhang, X. ACSL1 Inhibits ALV-J Replication by IFN-I Signaling and PI3K/Akt Pathway. Front. Immunol. 2021, 12, 774323. [Google Scholar] [CrossRef]
- Gongora, C.; Mechti, N. Interferon signaling pathways. (Article in French). Bull. Cancer 1999, 86, 911–919. [Google Scholar]
- Wen, W.; Qi, Z.; Wang, J. The function and mechanism of enterovirus 71 (EV71) 3C protease. Curr. Microbiol. 2020, 77, 968–1975. [Google Scholar] [CrossRef]
- Visser, L.J.; Langereis, M.A.; Rabouw, H.H.; Wahedi, M.; Muntjewerff, E.M.; de Groot, R.J.; van Kuppeveld, F.J.M. Essential role of enterovirus 2A protease in counteracting stress granule formation and the induction of type I interferon. J. Virol. 2019, 93, e00222-19. [Google Scholar] [CrossRef]
- Wang, C.; Sun, M.; Yuan, X.; Ji, L.; Jin, Y.; Cardona, C.J.; Xing, Z. Enterovirus 71 suppresses interferon responses by blocking Janus kinase (JAK)/signal transducer and activator of transcription (STAT) signaling through inducing karyopherin-α1 degradation. J. Biol Chem. 2017, 292, 10262–10274. [Google Scholar] [CrossRef]
- Li, D.; Chen, S.; Zhang, W.; Zhang, C.; Sun, T.; Du, Y.; Ding, R.; Gao, Y.; Jin, Y.; Duan, G. MicroRNA-628-5p facilitates enterovirus 71 infection by suppressing TRAF3 signaling. Cell Mol. Immunol. 2021, 18, 1320–1322. [Google Scholar] [CrossRef]
- Good, C.; Wells, A.I.; Coyne, C.B. Type III interferon signaling restricts enterovirus 71 infection of goblet cells. Sci. Adv. 2019, 5, eaau4255. [Google Scholar] [CrossRef] [PubMed]
- Kvansakul, M. Viral infection and apoptosis. Viruses 2017, 9, 356. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Li, Y.; Wu, H.L.; Chen, H.; Wu, H.; Guo, M.; Zhao, M.; Wang, C.; Lin, T.; Lin, Z.; et al. The inhibition of enterovirus 71 induced apoptosis by Durvillaea antarctica through P53 and STAT1 signaling pathway. J. Med. Virol. 2021, 93, 3532–3538. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Li, Y.; Xu, T.; Guo, M.; Wang, C.; Zhao, M.; Chen, H.; Kuang, J.; Li, W.; Zhang, Y.; et al. Inhibition of enterovirus 71 by selenium nanoparticles loaded with siRNA through bax signaling pathways. ACS. Omega. 2020, 13, 12495–12500. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Lin, E.; He, L.; Yu, J.; Tan, P.; Zhou, Y. Autophagy and viral infection. Adv. Exp. Med. Biol. 2019, 1209, 55–78. [Google Scholar]
- Liu, Z.W.; Zhuang, Z.C.; Chen, R.; Wang, X.R.; Zhang, H.L.; Li, S.H.; Wang, Z.Y.; Wen, H.L. Enterovirus 71 VP1 protein regulates viral replication in SH-SY5Y cells via the mTOR autophagy signaling pathway. Viruses 2019, 12, 11. [Google Scholar] [CrossRef]
- Lin, Y.W.; Yu, S.L.; Shao, H.Y.; Lin, H.Y.; Liu, C.C.; Hsiao, K.N.; Chitra, E.; Tsou, Y.L.; Chang, H.W.; Sia, C.; et al. Human SCARB2 transgenic mice as an infectious animal model for enterovirus 71. PLoS ONE 2013, 8, e57591. [Google Scholar] [CrossRef]
- Fujii, K.; Nagata, N.; Sato, Y.; Ong, K.C.; Wong, K.T.; Yamayoshi, S.; Shimanuki, M.; Shitara, H.; Taya, C.; Koike, S. Transgenic mouse model for the study of enterovirus 71 neuropathogenesis. Proc. Natl. Acad. Sci. USA 2013, 110, 14753–14758. [Google Scholar] [CrossRef]
- Pourianfar, H.R.; Poh, C.L.; Fecondo, J.; Grollo, L. In vitro evaluation of the antiviral activity of heparan sulfate mimetic compounds against Enterovirus 71. Virus Res. 2012, 169, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Mizuta, K.; Koike, S. Heparan sulfate attachment receptor is a major selection factor for attenuated enterovirus 71 mutants during cell culture adaptation. PLoS Pathog. 2020, 16, e1008428. [Google Scholar] [CrossRef] [PubMed]
- Su, P.Y.; Liu, Y.T.; Chang, H.Y.; Huang, S.W.; Wang, Y.F.; Yu, C.K.; Wang, J.R.; Chang, C.F. Cell surface sialylation affects binding of enterovirus 71 to rhabdomyosarcoma and neuroblastoma cells. BMC Microbiol. 2012, 12, 162. [Google Scholar] [CrossRef]
- Masomian, M.; Lalani, S.; Poh, C.L. Molecular Docking of SP40 Peptide towards Cellular Receptors for Enterovirus 71 (EV-A71). Molecules 2021, 26, 6576. [Google Scholar] [CrossRef] [PubMed]
- Lalani, S.; Tan, S.H.; Tan, K.O.; Lim, H.X.; Ong, K.C.; Wong, K.T.; Poh, C.L. Molecular mechanism of L-SP40 peptide and in vivo efficacy against EV-A71 in neonatal mice. Life Sci. 2021, 287, 120097. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.W.; Chan, Y.F.; Sim, K.M.; Tan, E.L.; Poh, C.L. Inhibition of enterovirus 71 (EV-71) infections by a novel antiviral peptide derived from EV-71 capsid protein VP1. PLoS ONE 2012, 7, e34589. [Google Scholar] [CrossRef]
- Berlutti, F.; Pantanella, F.; Natalizi, T.; Frioni, A.; Paesano, R.; Polimeni, A.; Valenti, P. Antiviral properties of lactoferrin--a natural immunity molecule. Molecules 2011, 16, 6992–7018. [Google Scholar] [CrossRef] [PubMed]
- Antoshin, A.A.; Shpichka, A.I.; Huang, G.; Chen, K.; Lu, P.; Svistunov, A.A.; Lychagin, A.V.; Lipina, M.M.; Sinelnikov, M.Y.; Reshetov, I.V.; et al. Lactoferrin as a regenerative agent: The old-new panacea. Pharmacol. Res. 2021, 167, 105564. [Google Scholar] [CrossRef]
- Lin, T.Y.; Chu, C.; Chiu, C.H. Lactoferrin inhibits enterovirus 71 infection of human embryonal rhabdomyosarcoma cells in vitro. J. Infect. Dis. 2002, 186, 1161–1164. [Google Scholar] [CrossRef]
- Weng, T.Y.; Chen, L.C.; Shyu, H.W.; Chen, S.H.; Wang, J.R.; Yu, C.K.; Lei, H.Y.; Yeh, T.M. Lactoferrin inhibits enterovirus 71 infection by binding to VP1 protein and host cells. Antivir. Res. 2005, 67, 31–34. [Google Scholar] [CrossRef]
- Swain, S.P.; Mohanty, S. Imidazolidinones and Imidazolidine-2,4-diones as Antiviral Agents. ChemMedChem 2019, 14, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Dove, A.W.; Racaniello, V.R. An antiviral compound that blocks structural transitions of poliovirus prevents receptor binding at low temperatures. J. Virol. 2000, 74, 3929–3931. [Google Scholar] [CrossRef]
- Shia, K.-S.; Li, W.-T.; Chang, C.-M.; Hsu, M.-C.; Chern, J.-H.; Leong, M.K.; Tseng, S.-N.; Lee, C.-C.; Lee, Y.-C.; Chen, S.-J.; et al. Design, synthesis, and structure-activity relationship of pyridyl imidazolidinones: A novel class of potent and selective human enterovirus 71 inhibitors. J. Med. Chem. 2002, 45, 1644–1655. [Google Scholar] [CrossRef] [PubMed]
- Chern, J.-H.; Shia, K.-S.; Hsu, T.-A.; Tai, C.-L.; Lee, C.-C.; Lee, Y.-C.; Chang, C.-S.; Tseng, S.-N.; Shih, S.-R. Design, synthesis, and structure-activity relationships of pyrazolo[3,4-d] pyrimidines: A novel class of potent enterovirus inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 2519–2525. [Google Scholar] [CrossRef] [PubMed]
- Plevka, P.; Perera, R.; Yap, M.L.; Cardosa, J.; Kuhn, R.J.; Rossmann, M.G. Structure of human enterovirus 71 in complex with a capsid-binding inhibitor. Proc. Natl. Acad. Sci. USA 2013, 110, 5463–5467. [Google Scholar] [CrossRef]
- Shih, S.-R.; Tsai, M.-C.; Tseng, S.-N.; Won, K.-F.; Shia, K.-S.; Li, W.-T.; Chern, J.-H.; Chen, G.-W.; Lee, C.-C.; Lee, Y.-C.; et al. Mutation in enterovirus 71 capsid protein VP1 confers resistance to the inhibitory effects of pyridyl imidazolidinone. Antimicrob. Agents Chemother. 2004, 48, 3523–3529. [Google Scholar] [CrossRef]
- Chen, T.-C.; Liu, S.-C.; Huang, P.-N.; Chang, H.-Y.; Chern, J.-H.; Shih, S.-R.J. Antiviral activity of pyridyl imidazolidinones against enterovirus 71 variants. Biomed. Sci. 2008, 15, 291–300. [Google Scholar] [CrossRef]
- Tan, Y.W.; Yam, W.K.; Kooi, R.J.W.; Westman, J.; Arbrandt, G.; Chu, J.J.H. Novel capsid binder and PI4KIIIbeta inhibitors for EV-A71 replication inhibition. Sci. Rep. 2021, 11, 9719. [Google Scholar] [CrossRef]
- Zhang, G.; Zhou, F.; Gu, B.; Ding, C.; Feng, D.; Xie, F.; Wang, J.; Zhang, C.; Cao, Q.; Deng, Y.; et al. In vitro and in vivo evaluation of ribavirin and pleconaril antiviral activity against enterovirus 71 infection. Arch. Virol. 2012, 157, 669–679. [Google Scholar] [CrossRef]
- Barnard, D.L.; Hubbard, V.D.; Smee, D.F.; Sidwell, R.W.; Watson, K.G.; Tucker, S.P.; Reece, P.A. In vitro activity of expanded-spectrum pyridazinyl oxime ethers related to pirodavir: Novel capsid-binding inhibitors with potent antipicornavirus activity. Antimicrob. Agents Chemother. 2004, 48, 1766–1772. [Google Scholar] [CrossRef]
- Tijsma, A.; Franco, D.; Tucker, S.; Hilgenfeld, R.; Froeyen, M.; Leyssen, P.; Neyts, J. The capsid binder Vapendavir and the novel protease inhibitor SG85 inhibit enterovirus 71 replication. Antimicrob. Agents Chemother. 2014, 58, 6990–6992. [Google Scholar] [CrossRef] [PubMed]
- Arita, M.; Wakita, T.; Shimizu, H. Characterization of pharmacologically active compounds that inhibit poliovirus and enterovirus 71 infectivity. J. Gen. Virol. 2008, 89, 2518–2530. [Google Scholar] [CrossRef] [PubMed]
- Leiro, J.M.; Castro, R.; Arranz, J.A.; Lamas, J. Immunomodulating activities of acidic sulphated polysaccharides obtained from the seaweed Ulva rigida C. Agardh. Int. Immunopharmacol. 2007, 7, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Zhang, Q.; Zhao, T.; Chen, R.; Zhang, H.; Niu, X.; Li, Z. Antioxidant activity of different sulfate content derivatives of polysaccharide extracted from Ulva pertusa (Chlorophyta) in vitro. Int. J. Biol. Macromol. 2005, 37, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Berteau, O.; Mulloy, B. Sulfated fucans, fresh perspectives: Structures, functions, and biological properties of sulfated fucans and an overview of enzymes active toward this class of polysaccharide. Glycobiology 2003, 13, 29R–40R. [Google Scholar] [CrossRef]
- Chiu, H.; Chan, Y.L.; Tsai, L.W.; Li, T.L.; Wu, C.J. Prevention of human enterovirus 71 infection by kappa carrageenan. Antivir. Res. 2012, 95, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Groult, H.; Cousin, R.; Chot-Plassot, C.; Maura, M.; Bridiau, N.; Piot, J.M.; Maugard, T.; Fruitier-Arnaudin, I. λ-Carrageenan Oligosaccharides of Distinct Anti-Heparanase and Anticoagulant Activities Inhibit MDA-MB-231 Breast Cancer Cell Migration. Mar. Drugs 2019, 17, 140. [Google Scholar] [CrossRef]
- Tseng, K.C.; Hsu, B.Y.; Ling, P.; Lu, W.W.; Lin, C.W.; Kung, S.H. Antidepressant Sertraline Is a Broad-Spectrum Inhibitor of Enteroviruses Targeting Viral Entry through Neutralization of Endolysosomal Acidification. Viruses 2022, 14, 109. [Google Scholar] [CrossRef]
- Wang, W.; Meng, Q.; Li, Q.; Liu, J.; Zhou, M.; Jin, Z.; Zhao, K. Chitosan Derivatives and Their Application in Biomedicine. Int. J. Mol. Sci. 2020, 21, 487. [Google Scholar] [CrossRef]
- Thangavelu, A.; Stelin, K.S.; Vannala, V.; Mahabob, N.; Hayyan, F.M.B.; Sundaram, R. An Overview of Chitosan and Its Role in Periodontics. J. Pharm. Bioallied Sci. 2021, 13, S15–S18. [Google Scholar] [CrossRef]
- Lin, Y.C.; Lin, S.T.; Chen, C.Y.; Wu, S.C. Enterovirus 71 adsorption on metal ion-composite chitosan beads. Biotechnol. Prog. 2012, 28, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Xu, Z.C.; Zhang, F.; Cai, Y.; Chen, Y.N.; Lu, B.J.; Zhou, H.B.; Lan, K.; Wu, S.W. Identification of a novel binding inhibitor that blocks the interaction between hSCARB2 and VP1 of enterovirus 71. Cell Insight 2022, 1, 100016. [Google Scholar] [CrossRef]
- Yoo, H.J.; Kang, H.J.; Jung, H.J.; Kim, K.; Lim, C.J.; Park, E.H. Anti-inflammatory, anti-angiogenic and anti-nociceptive activities of Saururus chinensis extract. J. Ethnopharmacol. 2008, 120, 282–286. [Google Scholar] [CrossRef]
- Cho, H.Y.; Cho, C.W.; Song, Y.S. Antioxidative and anti-inflammatory effects of Saururus chinensis methanol extract in RAW 264.7 macrophages. J. Med. Food 2005, 8, 190–197. [Google Scholar] [PubMed]
- Gong, Q.Z.; Xiao, D.; Feng, F.; Wen, X.D.; Wei, Q. Ent-Sauchinone as Potential Anticancer Agent Inhibiting Migration and Invasion of Human Liver Cancer Cells via Suppressing the STAT3 Signaling Pathway. Chem. Biodivers. 2018, 15, e1800024. [Google Scholar] [CrossRef]
- Park, J.S.; Bang, O.S.; Kim, J. Screening of Stat3 inhibitory effects of Korean herbal medicines in the A549 human lung cancer cell line. Integr. Med. Res. 2014, 3, 67–73. [Google Scholar] [CrossRef]
- Tian, L.; Li, X.Y.; Xu, Y.X.; Li, E. Effect and mechanism of Saururus chinensis against herpes dimplex virus. Zhongguo Zhong Yao Za Zhi China J. Chin. Mater. Med. 2012, 37, 1642–1645. (In Chinese) [Google Scholar]
- Wang, B.; Zhang, H.; Zhu, M.; Luo, Z.; Peng, Y. MEK1-ERKs signal cascade is required for the replication of Enterovirus 71 (EV71). Antivir. Res. 2012, 93, 110–117. [Google Scholar] [CrossRef]
- Wang, H.Q.; Zhang, D.J.; Ge, M.; Li, Z.R.; Jiang, J.D.; Li, Y.H. Formononetin inhibits enterovirus 71 replication by regulating COX-2/PGE₂ expression. Virol. J. 2015, 12, 35. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, B.; Chen, X.; Song, N.; Wu, J.; Li, G.; Yu, P.; Han, Y.; Liu, J.; Qin, C. GS-9620 inhibits enterovirus 71 replication mainly through the NF-κB and PI3K-AKT signaling pathways. Antivir. Res. 2018, 153, 39–48. [Google Scholar] [CrossRef]
- Wang, H.; Li, K.; Ma, L.; Wu, S.; Hu, J.; Yan, H.; Jiang, J.; Li, Y. Berberine inhibits enterovirus 71 replication by downregulating the MEK/ERK signaling pathway and autophagy. Virol. J. 2017, 14, 2. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Zheng, R.; Wu, H.L.; Chen, D.; Su, J.; Xu, T.; Wu, H.; Xiang, W.; Li, Y.; Zhu, B. Inhibition of enterovirus 71 infection by polysaccharides extracted from Picochlorum sp. 122 via the AKT and ATM/ATR signaling pathways. Arch. Virol. 2021, 166, 3269–3274. [Google Scholar] [CrossRef] [PubMed]
- Ripa, I.; Andreu, S.; López-Guerrero, J.A.; Bello-Morales, R. Membrane R: Portals for viral entry. Front. Microbiol. 2021, 12, e631274. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.Z.; Wu, D.G.; Ren, H.; Xu, Q.Q.; Zheng, K.C.; Chen, W.; Chen, S.L.; Qian, X.J.; Tao, Q.Y.; Wang, Y.; et al. The role of lipid rafts in the early stage of Enterovirus 71 infection. Cell Physiol. Biochem. 2015, 35, 1347–1359. [Google Scholar] [CrossRef] [PubMed]
- Chang, G.H.; Luo, Y.J.; Wu, X.Y.; Si, B.Y.; Lin, L.; Zhu, Q.Y. Monoclonal antibody induced with inactived EV71-Hn2 virus protects mice against lethal EV71-Hn2 virus infection. Virol. J. 2010, 7, 106. [Google Scholar] [CrossRef]
- Ku, Z.; Ye, X.; Shi, J.; Wang, X.; Liu, Q.; Huang, Z. Single neutralizing monoclonal antibodies targeting the VP1 GH loop of enterovirus 71 inhibit both virus attachment and internalization during viral entry. J. Virol. 2015, 89, 12084–12095. [Google Scholar] [CrossRef]
- Lee, H.; Cifuente, J.O.; Ashley, R.E.; Conway, J.F.; Makhov, A.M.; Tano, Y.; Shimizu, H.; Nishimura, Y.; Hafenstein, S. A strain-specific epitope of enterovirus 71 identified by cryo-electron microscopy of the complex with fab from neutralizing antibody. J. Virol. 2013, 87, 11363–11370. [Google Scholar] [CrossRef]
- Lim, X.F.; Jia, Q.; Khong, W.X.; Yan, B.; Premanand, B.; Alonso, S.; Chow, V.T.; Kwang, J. Characterization of an isotype-dependent monoclonal antibody against linear neutralizing epitope effective for prophylaxis of enterovirus 71 infection. PLoS ONE 2012, 7, e29751. [Google Scholar] [CrossRef]
- Kiener, T.K.; Jia, Q.; Meng, T.; Chow, V.T.; Kwang, J. A novel universal neutralizing monoclonal antibody against enterovirus 71 that targets the highly conserved “knob” region of VP3 protein. PLoS Negl. Trop. Dis. 2014, 8, e2895. [Google Scholar] [CrossRef]
- Li, X.; Huang, Y.; Sun, M.; Ji, H.; Dou, H.; Hu, J.; Yan, Y.; Wang, X.; Chen, L. Honeysuckle-encoded microRNA2911 inhibits Enterovirus 71 replication via targeting VP1 gene. Antivir. Res. 2018, 152, 117–123. [Google Scholar] [CrossRef]
- Feng, C.; Fu, Y.; Chen, D.; Wang, H.; Su, A.; Zhang, L.; Chang, L.; Zheng, N.; Wu, Z. miR-127-5p negatively regulates enterovirus 71 replication by directly targeting SCARB2. FEBS Open Bio. 2017, 7, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Song, Z.; Qi, Y.; Feng, X.; Xu, N.; Sun, Y.; Wu, X.; Yao, X.; Mao, Q.; Li, X.; et al. Molecular determinants of enterovirus 71 viral entry: Cleft around GLN-172 on VP1 protein interacts with variable region on scavenge receptor B 2. J. Biol. Chem. 2012, 287, 6406–6420. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Yu, X.; Wang, D.; Li, Z.; Zhou, Y.; Xu, G.; Yuan, B.; Qin, Y.; Chen, M. SLC35B2 acts in a dual role in the host sulfation required for EV71 infection. J. Virol. 2022, 96, e0204221. [Google Scholar] [CrossRef] [PubMed]
- Falah, N.; Montserret, R.; Lelogeais, V.; Schuffenecker, I.; Lina, B.; Cortay, J.C.; Violot, S. Blocking human enterovirus 71 replication by targeting viral 2A protease. J. Antimicrob. Chemother. 2012, 67, 2865–2869. [Google Scholar] [CrossRef] [PubMed]
- Diarimalala, R.O.; Hu, M.; Wei, Y.; Hu, K. Recent advances of enterovirus 71 3Cpro targeting Inhibitors. Virol. J. 2020, 17, 173. [Google Scholar] [CrossRef]
- Yao, C.; Xi, C.; Hu, K.; Gao, W.; Cai, X.; Qin, J.; Lv, S.; Du, C.; Wei, Y. Inhibition of enterovirus 71 replication and viral 3C protease by quercetin. Virol. J. 2018, 15, 116. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, K.; Onintsoa Diarimalala, R.; Yao, C.; Li, H.; Wei, Y. EV-A71 Mechanism of Entry: Receptors/Co-Receptors, Related Pathways and Inhibitors. Viruses 2023, 15, 785. https://doi.org/10.3390/v15030785
Hu K, Onintsoa Diarimalala R, Yao C, Li H, Wei Y. EV-A71 Mechanism of Entry: Receptors/Co-Receptors, Related Pathways and Inhibitors. Viruses. 2023; 15(3):785. https://doi.org/10.3390/v15030785
Chicago/Turabian StyleHu, Kanghong, Rominah Onintsoa Diarimalala, Chenguang Yao, Hanluo Li, and Yanhong Wei. 2023. "EV-A71 Mechanism of Entry: Receptors/Co-Receptors, Related Pathways and Inhibitors" Viruses 15, no. 3: 785. https://doi.org/10.3390/v15030785
APA StyleHu, K., Onintsoa Diarimalala, R., Yao, C., Li, H., & Wei, Y. (2023). EV-A71 Mechanism of Entry: Receptors/Co-Receptors, Related Pathways and Inhibitors. Viruses, 15(3), 785. https://doi.org/10.3390/v15030785






